Abstract
The accumulation of microplastics (MPs) in agricultural soils and atrazine in agricultural soils creates compound pollution that severely threatens soil health. The present study aimed to evaluate the effect of polyethylene (PE), polyvinyl chloride (PVC), and polybutylene succinate (PBS) on the adsorption and degradation of atrazine in yellow-brown and black soil. Batch adsorption kinetic and isotherm experiments were conducted in two distinct soils amended with MPs. A 90-day degradation experiment was performed to monitor atrazine persistence and the activities of key soil enzymes. The adsorption process was best described by the pseudo-second-order model and the Freundlich isotherm model, suggesting dominant chemisorption and multilayer adsorption on heterogeneous surfaces of the soil–MP composites. All MPs significantly enhanced the adsorption capacity for atrazine (6.80–39.93 mg kg−1), with the order PBS > PE > PVC. Furthermore, the degradation of atrazine was impeded by all MPs, with PVC exhibiting the strongest inhibitory effect. The half-life of atrazine ranges from 22.97 to 81.76 days in two soils. The presence of MPs also influenced soil enzyme activities and the effects varied by MP type and soil property. These results demonstrate that MPs can modify the adsorption and persistence of atrazine in soil, thereby increasing its environmental risk. This study provides valuable insights for the long-term ecological risk assessment of co-existing MPs and pesticide pollution in terrestrial environments.
1. Introduction
The accumulation of microplastics (MPs) in terrestrial ecosystems has emerged as a critical global environmental challenge. According to previous studies [1,2,3], terrestrial soil has been recognized as a repository and dispersal site for MPs. Agricultural soil is subjected to the adverse effects of terrestrial MPs, making it a prominent hotspot for pollution [4,5,6], largely attributed to the use of crop mulch films and plastic greenhouses [7,8,9,10]. While plastic mulching is essential for enhancing agricultural productivity and food security, the steady increase in its use over time exacerbates the accumulation of residual plastics and associated ecological risks [11,12]. Concurrently, atrazine (ATZ)—a herbicide that serves a critical function in plant growth regulation—is heavily utilized worldwide [13,14]. With an estimated annual consumption of 10,000 tons in China alone, ATZ is a ubiquitous contaminant often detected in soils at mean concentrations of 5.7 ng g−1, and occasionally up to 113 ng g−1 in certain areas [15,16]. ATZ exhibits poor biodegradability, and studies report that approximately 19.5 g of ATZ per hectare remained extractable from soil two decades after the last application, and its metabolites are found even in soils without a recorded ATZ application [17]. Toxicological investigations demonstrate that doses as low as 5 μg L−1 of ATZ may cause gill and kidney inflammation or necrosis in some fish species [18]. It also has the potential to accumulate in the human body through the food chain, resulting in hormonal imbalance and carcinogenic effects [19,20]. Given the inherent persistence and toxicity of both MPs and ATZ [21,22], their co-occurrence in agricultural soils creates a complex “compound pollution” scenario that severely threatens soil health and food safety.
Previous studies have achieved substantial advancements in understanding the individual behaviors of these contaminants. Research on ATZ has thoroughly shown its environmental fate, with an emphasis on the adsorption and degradation processes [23,24], revealing that the maximal equilibrium adsorption capacity varies considerably according to soil type (alluvial soil > laterite soil > paddy soil) [25]. The co-occurrence of MPs and pesticides in ecosystems may result in interactions that exacerbate ecological hazards [26,27]. Studies on MP–pesticide interactions show that adsorption onto polymers like polyethylene (PE) is often governed by hydrophobic forces, whereas surface adsorption mostly regulates the adsorption of some pesticides, including imidacloprid, buprofezin, and difenoconazole [28]. Additionally, the capacity of MPs to adsorb organochlorine pesticides in soil is inversely related to their particle size [29,30]. However, previous studies have focused on a limited range of plastics, predominantly conventional PE, neglecting the comparative effects of different polymer types—specifically the contrast between conventional plastics and biodegradable alternatives like polybutylene succinate (PBS). While the physical adsorption of pesticides onto MPs is documented, the subsequent impact of this interaction on the degradation rate of the pesticide remains poorly understood. Moreover, the coexistence of MPs and pesticides in soil can impair soil enzyme activity and intensify ecological hazards [26]. Soil enzymes serve as a vital bioindicator of soil health. For example, dehydrogenase, urease, and sucrase are critical mediators in nutrient cycling, and the degradation of organic pollutants [31]. Current evidence indicates that MPs can reduce pesticide bioavailability through adsorption, thereby hindering essential enzyme-catalyzed degradation processes [32,33]. However, there is a lack of systematic data elucidating how specific polymer types differentially influence soil enzyme activity. Consequently, the specific mechanisms by which different MP types regulate the bioavailability, persistence, and biological toxicity of ATZ in a soil system have not been fully elucidated.
To bridge this gap, this study provides a systematic investigation into the environmental fate of ATZ in the presence of three distinct MP types: PE, polyvinyl chloride (PVC), and PBS. The novelty of this research lies in its comparative approach, simultaneously evaluating how these varying polymer structures influence both the physicochemical and biological behaviors of ATZ. Specifically, we aim to: (1) quantify the adsorption capacity of varying MPs for ATZ; (2) determine how MPs alter the degradation kinetics of ATZ in soil; and (3) assess the response of critical soil enzymes to the combined pollution. This research is significant as it provides essential data for risk assessment, offering new insights into the long-term ecological consequences of transitioning from conventional to biodegradable mulch films in ATZ-treated agricultural lands.
2. Materials and Methods
2.1. Materials
Soil S1 was acquired from an agricultural field located in Nanjing City (32.033657295 N, 118.8755106 E), while soil S2 was gathered from a farmland in Hongguang Farm in Hailun City (47.662785 N, 127.324170 E). All soil samples were acquired in the spring of 2022 from the 0–20 cm depth layer. Subsequently, the soils were air-dried, sieved through a 2 mm mesh, and pre-incubated in an artificial climate chamber (25 °C) in the dark for two weeks to reestablish soil microorganisms. ATZ (Yuanye, CAS: [1912-24-9], 99% purity) was obtained from the Jiangsu Academy of Agricultural Sciences. PE, PVC, and PBS were purchased from Ningxia Zhongcheng Plastic Corporation (80–100 µm in diameter). All these reagents and solvents were purchased from Nanjing Xianghai Biotechnology Co., Ltd. (Nanjing, China). The physicochemical properties of the two soils and MPs were summarized in Table 1 and Table 2.

Table 1.
Physico-chemical properties of the soils.

Table 2.
Physico-chemical properties of the microplastics.
2.2. Sorption Kinetics and Isotherm Experiments
A stock solution of ATZ (1000 mg L−1) was diluted with a background solution (0.01 M CaCl2 and 100 mg L−1 HgCl2) to produce an experimental solution at 10 mg L−1. The sorbent was composed of a 10% (w/w) mixture of soil and MPs. In each treatment, 5 mL experimental solution was transferred to 20 mL glass tubes with the addition of soil–MP composites.
Adsorption kinetics experiments: Experiments were conducted with eight treatments in triplicate: S1 (1.1 g of soil S1), S1-PE (1 g of soil S1 and 0.1 g of PE), S1-PVC (1 g of soil S1 and 0.1 g of PVC), S1-PBS (1 g of soil S1 and 0.1 g of PBS), S2 (1.1 g of soil S2), S2-PE (1 g of soil S2 and 0.1 g of PE), S2-PVC (1 g of soil S2 and 0.1 g of PVC), and S2-PBS (1 g of soil S2 and 0.1 g of PBS). The mixtures were shaken at 160 rpm and 25 °C for 0.5, 1, 2, 4, 8, 12, 24, and 48 h, respectively. The collected samples subsequently went through a 20 min centrifuge at 5000 rpm, a 0.22 µm filter, and a 2 mL injection vial before being analyzed. Preliminary kinetic data indicated that adsorption equilibrium was reached within 12 h; therefore, this duration was chosen for the subsequent adsorption isotherm investigations.
Adsorption isotherm experiment: Comparable to the kinetic experiments, isotherm experiments were performed by adding ATZ solutions to the sorbent mixtures at initial concentrations ranging from 1.0 to 20.0 mg L−1. The samples were then agitated for 12 h on a platform shaker before being centrifuged and filtered for analysis.
2.3. Degradation Experiment
Prior to the experiment, soil moisture content was adjusted to 60% of the water-holding capacity. The samples were equilibrated over a 14-day preincubation period in darkness at 25 °C. Degradation studies employed the same eight treatments as the adsorption experiments (S1, S1-PE, S1-PVC, S1-PBS, S2, S2-PE, S2-PVC, and S2-PBS). For each treatment, 200 g of soil was spiked with ATZ at 3.0 mg kg−1 and thoroughly homogenized with 10% (w/w) of the respective MP. The mixtures were transferred to the bottles, covered with porous foil, and stored in a climate chamber at 25 °C for 90 days. Deionized water was added periodically to compensate for moisture loss. Samples were taken for ATZ analysis on days 0, 7, 14, 28, 56, and 90. Soil enzyme activity was assessed after 56 days of incubation. Soil urease activity was determined by sodium phenate–sodium hypochlorite colorimetry, sucrase activity by 3,5-dinitrosalicylic acid (DNS) colorimetry, and dehydrogenase activity by potassium permanganate titration [34].
2.4. Chemical Analysis and Quality Control
Soil samples were prepared with the QuEChERS pretreatment method, and the ATZ quantification was performed by an ACCELA UHPLC instrument (Thermo, Waltham, MA, USA) equipped with a ZORBAX C18 analytical column (4.6 mm × 250 mm, 5 μm). Chromatographic separation was achieved under isocratic conditions using a mobile phase of acetonitrile and water (30:70, v/v) at a flow rate of 0.8 mL min−1. The column temperature remained at 30 °C, and UV detection was calibrated at 228 nm [35].
The identification of ATZ metabolites in the samples was conducted using an HRLC-Q-TOF-MS/MS instrument (Thermo, Waltham, MA, USA) in conjunction with an ultraviolet detector set at 220 nm. The analysis was conducted using a 20 mL injection volume on a reversed-phase C-18 column (4.6 mm × 250 mm, 5 μm). The analytes were separated under isocratic conditions using a water-methanol mobile phase (35:65, v/v) at a flow rate of 0.6 mL min−1, with the procedure performed at room temperature.
Five quality assurance/quality control (QA/QC) samples spiked with ATZ (1 mg kg−1) were added during the sample analysis procedure. The recovery rate of ATZ in QA/QC samples ranged from 91.23% to 113.22%, with RSDs less than 10%. These results indicate that satisfactory mean recovery values were acquired in QA/QC samples. Origin Pro (Version 8.5) software was used for data analysis.
2.5. Mathematical Models for Adsorption Mechanism, Adsorption Capacity, and Degradation
To acquire a comprehensive understanding of the adsorption mechanism, adsorption capacity, and degradation of ATZ onto soil, several mathematical models were used to describe the adsorption equilibrium.
Pseudo-first/second-order, Elovich, and Intraparticle diffusion models:
The Boyd model:
where qt and qe are the sorption amount at time t and sorption equilibrium, respectively, (mg kg−1); t is the sorption time (min); k1 and k2 are pseudo-first-order and pseudo-second-order rate constants, respectively. The constants a (mg kg−1) and b are associated with the activation energy for chemisorption and the extent of surface coverage, respectively.
ki (mg (kg t1/2)−1) is the rate constant of intra-particle diffusion, and Ci (mg kg−1) is the thickness of the boundary layer. F = qt/qe is the fractional attainment of equilibrium at time t (h).
The kinetic model was statistically validated by calculating the normalized standard deviation (NSD), using the following equation:
qt,exp and qt,cal indicate the experimental and calculated values, respectively, whereas i represents the number of data points.
The Freundlich and Langmuir isotherms models:
where qmax is the maximum sorption capacity (mg kg−1); KF is the Freundlich constant ((mg kg−1) (mg L−1)−n); and n is the sorption affinity constant
Degradation kinetics: The first-order kinetic equation was used to estimate the degradation kinetics of ATZ in soil, where C0 is the concentration of ATZ and C indicates the concentration at time t, respectively. k is the degradation rate.
3. Results and Discussion
3.1. Effects of MPs on the Adsorption Kinetics of ATZ in Soil
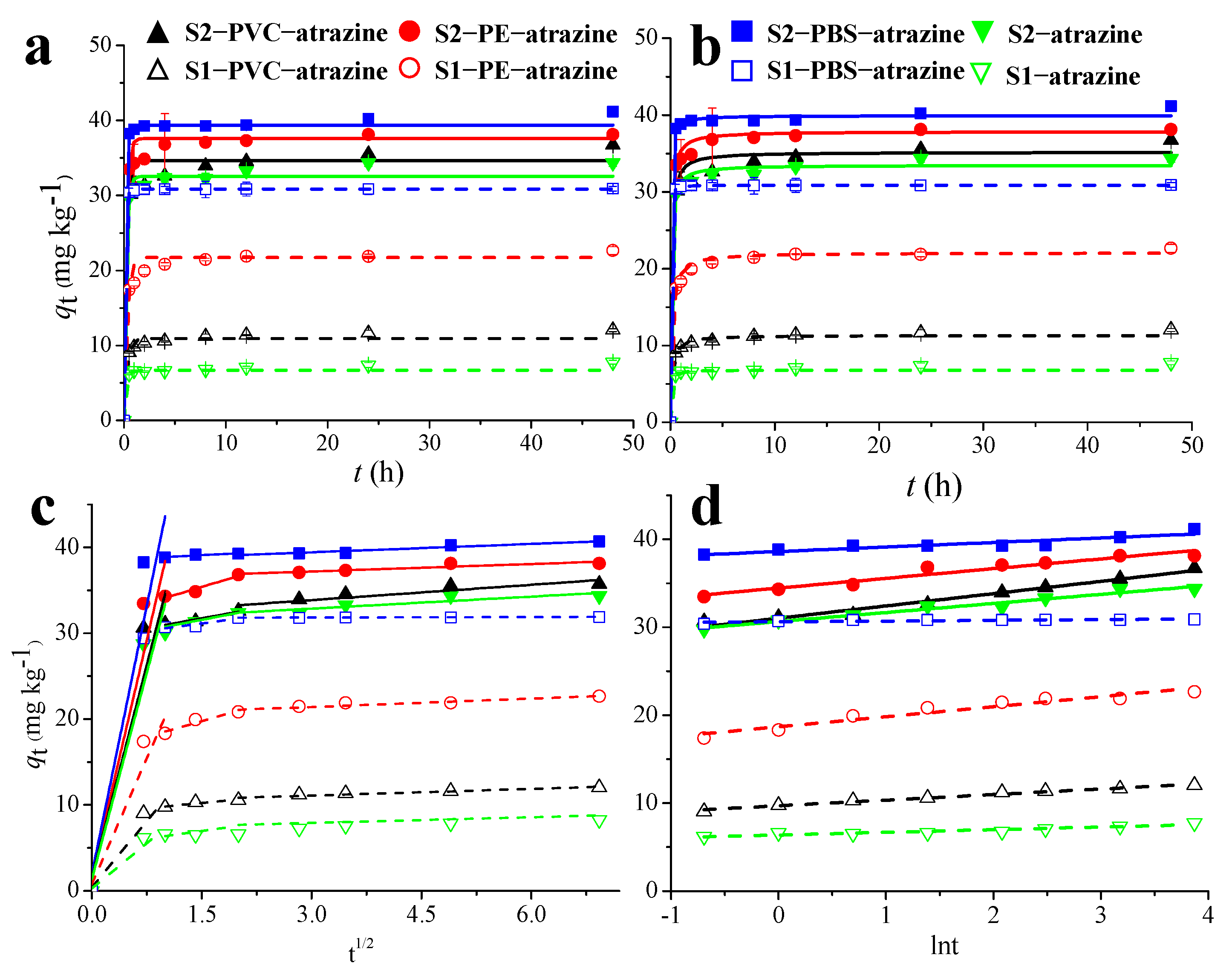
Kinetic experiments were carried out to explore the influence of MPs and soil types in the ATZ adsorption process. The adsorption of ATZ onto soils and MP-amended soils increased rapidly initially, then slowed before reaching equilibrium after approximately 12 h (Figure 1). Consequently, a 12 h contact time was selected for subsequent isotherm experiments. The adsorption kinetics data were fitted using pseudo-first-order, pseudo-second-order, and Elovich models. (Figure 1a–c and Table S1).
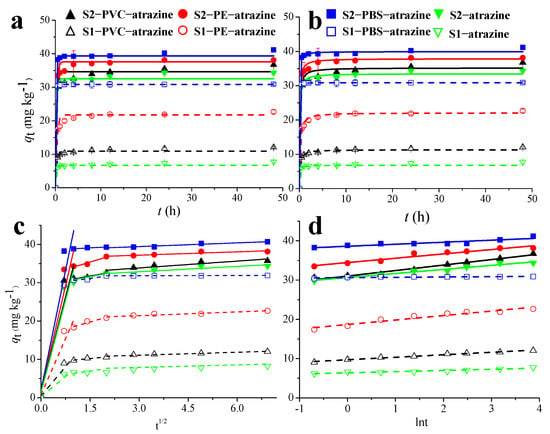
Figure 1.
Plots of the pseudo-first-order (a), pseudo-second-order (b), Elovich (c), and intra-particle diffusion (d) models for sorption kinetics of atrazine onto black soil samples (solid curves) and yellow-brown soil samples (dashed curves). S1, yellow-brown soil; S2, black soil.
The pseudo-second-order model yielded a superior fit than the pseudo-first-order and Elovich models, exhibiting higher R2 values (>0.990), lower NSD values (<11.64), and calculated equilibrium adsorption capacities (qe, cal) highly consistent with the experimental values (qe, exp). This indicates that the adsorption process is best described by the pseudo-second-order model, which often suggests that chemisorption may play a significant role in the adsorption process [35]. While chemical processes involving π-π interactions and hydrogen bonding are potential mechanisms based on this model, spectroscopic validation is required to confirm these specific molecular interactions. At equilibrium, the equilibrium adsorption capacity ranged from 6.80 to 30.89 mg kg−1 in yellow-brown soil and from 33.49 to 39.93 mg kg−1 in black soil. This corresponds to an experimental adsorption percentage ranging from 14.7% to 67.9% in yellow-brown soil, 71.6% to 86.7% in black soil. Furthermore, it was observed that the adsorption capacity and rates of ATZ followed the order: Soil + PBS > Soil + PE > Soil + PVC > Soil. This demonstrates that all MPs significantly increased the ATZ capacity and accelerated the ATZ adsorption process. Notably, the Elovich model can also accurately predict the ATZ adsorption kinetics data on soils and MP-amended soils (R2 > 0.904), indicating a diffusion-controlled process on heterogeneous surfaces [36].
Moreover, the investigation of mass transfer phases in the adsorption process was also conducted using the intra-particle diffusion model (Figure 1d). The adsorption process for the majority of the treatments was conducted in three distinct stages, including the first rapid adsorption phase characterized by liquid film diffusion, subsequent transit of ATZ through the particles with comparatively slow diffusion, and finally, the gradual establishment of equilibrium between adsorption and desorption. These findings are consistent with previous reports on pesticide adsorption by MPs, indicating that the adsorption mechanism of herbicides and insecticides onto MPs aligns well with the intra-particle diffusion model [37].
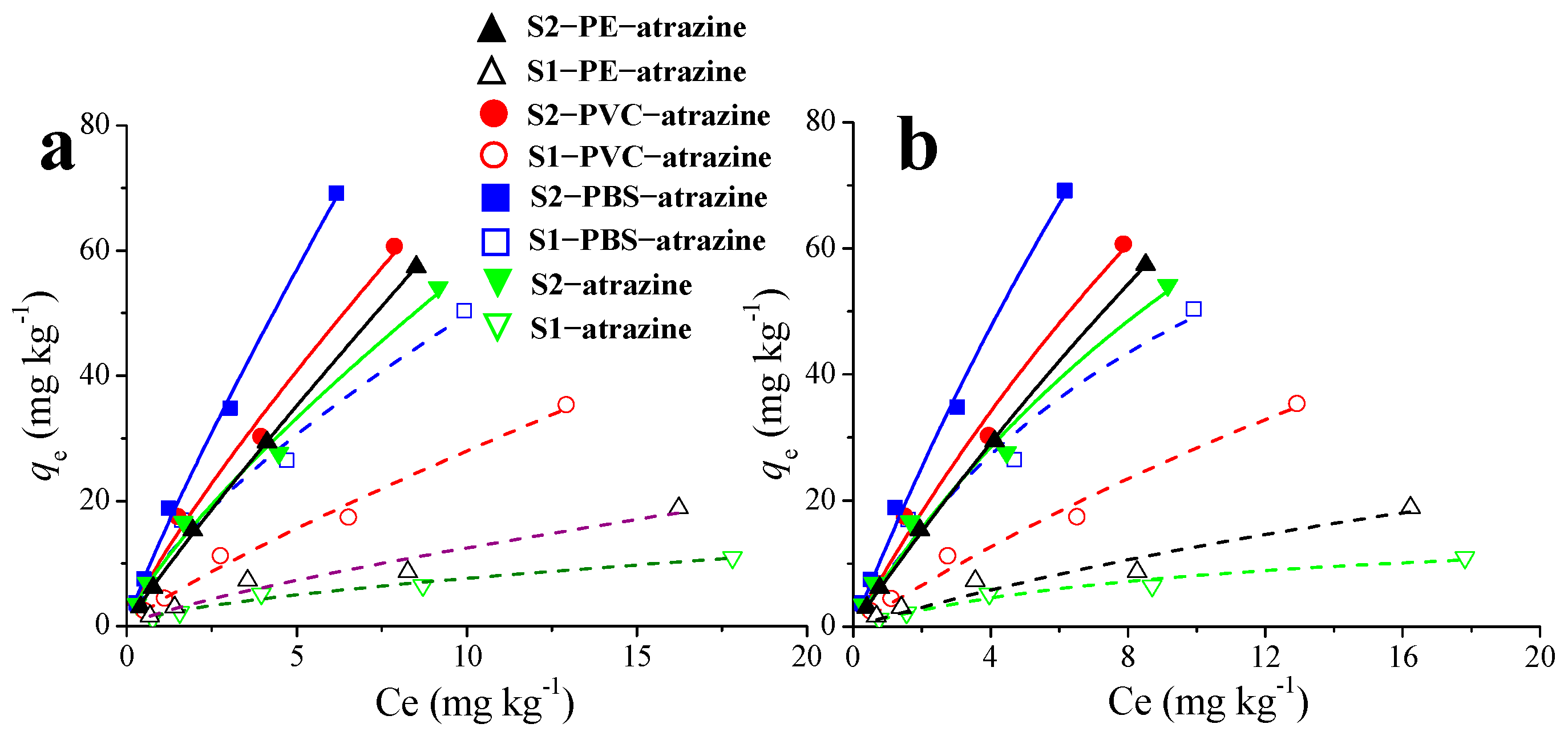
3.2. Effects of MPs on the Adsorption Isotherms of ATZ in Soil
The adsorption isotherms of ATZ in soil were significantly altered by MPs, showing a clear enhancement in adsorption capacity. Equilibrium data were fitted with nonlinear Freundlich and Langmuir models (Figure 2). Comparison of the determination coefficients (R2) and (Chi-sqr) values revealed that the Freundlich model provided a better description of the adsorption process across all treatments than the Langmuir model (Table S2). The suitability of the Freundlich isotherm indicates that ATZ adsorption onto the soil–MP composites involves multilayer adsorption on heterogeneous surfaces, through both physical and chemical mechanisms [36]. These findings align with previous studies reporting that the Freundlich isotherm model showed greater suitability for explaining the adsorption of pesticides onto MPs [28,38].
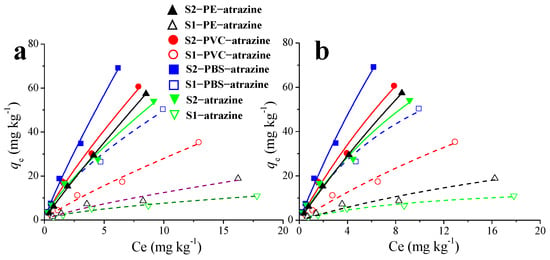
Figure 2.
Effects of MPs on the adsorption isotherms of atrazine (Freundlich, (a); Langmuir, (b)) in soil.
The Freundlich parameters kF and n provide key insights into adsorption capacity and intensity, respectively. The kF values, which represent adsorption capacity [39], increased substantially with MP addition in both soils (PBS > PVC > PE). The PBS-amended soil showed the highest kF, highlighting the importance of polar functional groups such as ester carbonyls in promoting specific interactions (e.g., hydrogen bonding). The exponent n reflects adsorption favorability and surface heterogeneity. All calculated 1/n values were below 1, confirming that adsorption was favorable under the experimental conditions. Furthermore, the n values also varied with MP type and generally increased with MP addition, indicating enhanced adsorption intensity.
Consequently, the isotherm analysis confirms that MPs act as effective adsorbents, substantially increasing the soil’s retention of ATZ. The extent of this enhancement depends strongly on the specific MP polymer; PBS exerted the greatest effect, thereby altering the partitioning and potential mobility of ATZ in soil.
3.3. The Effect of Microplastic Material on Sorption
The material composition of MPs dictates their interaction with ATZ in soil, significantly altering adsorption capacity and kinetics. The results reveal a distinct, polymer-specific order for enhancing ATZ sorption relative to unamended soil: PBS > PE > PVC. This order highlights the fundamental importance of MP surface chemistry and physicochemical properties in governing the adsorption process.
The enhanced adsorption capability in PBS-amended soils results from its chemical composition. PBS comprises polar ester carbonyl (C=O) groups. It could be hypothesized that these functional groups may facilitate specific interactions with ATZ, such as hydrogen bonding and dipole–dipole interactions, which could explain the higher adsorption affinity. However, future spectroscopic studies are needed to definitively verify this mechanism. The combination of polar interactions with non-specific hydrophobic partitioning yields a higher adsorption affinity compared to non-polar polymers. Conversely, PE interacts with ATZ primarily through hydrophobic partitioning and van der Waals interactions. Although PE enhances adsorption relative to unamended soil, its adsorption capacity remains lower than that of PBS, which might be due to the absence of polar functional groups necessary for specific interactions with ATZ. Meanwhile, PVC exhibits more complex behavior, providing a less pronounced adsorption enhancement than PE and PBS. This difference is likely attributable to its surface characteristics. Relevant investigations have supported the conclusion that the presence of polar functional groups on the surface of PBS might potentially influence the adsorption behavior of organic molecules [40,41]. Overall, the adsorption process is likely driven by a combination of hydrophobic partitioning, surface polarity, and specific chemical compatibility between the pesticides and the polymer matrix [42,43].
Furthermore, intrinsic soil characteristics also influence these MP-specific effects. Under comparable MP conditions, the adsorption rate constants (k) and the qe value in black soil were greater than those in yellow brown soil [44]. Consequently, the addition of MPs to black soil induced competition for primary sorption sites between MP surfaces and native soil organic matter. Nevertheless, even in organic-rich soil, the introduction of MPs, particularly PBS, still provided additional active sites, further enhancing the overall sequestration of ATZ. This suggests that the properties of MPs are the primary determinants governing their adsorption process. These characteristics interact with the soil’s components, but are not entirely limited by them. Ultimately, they collaboratively affect the fate and dynamics of pesticides inside the soil. Similar findings included the adsorption of three imidazolinone herbicides onto polypropylene MPs [37], the adsorption of three commonly used fungicides on PE in vegetable fields [45], and the adsorption of insecticide fipronil on four non-degradable and two degradable MPs (PE, polystyrene (PS), PVC, polylactic acid (PLA), and PBS [26].
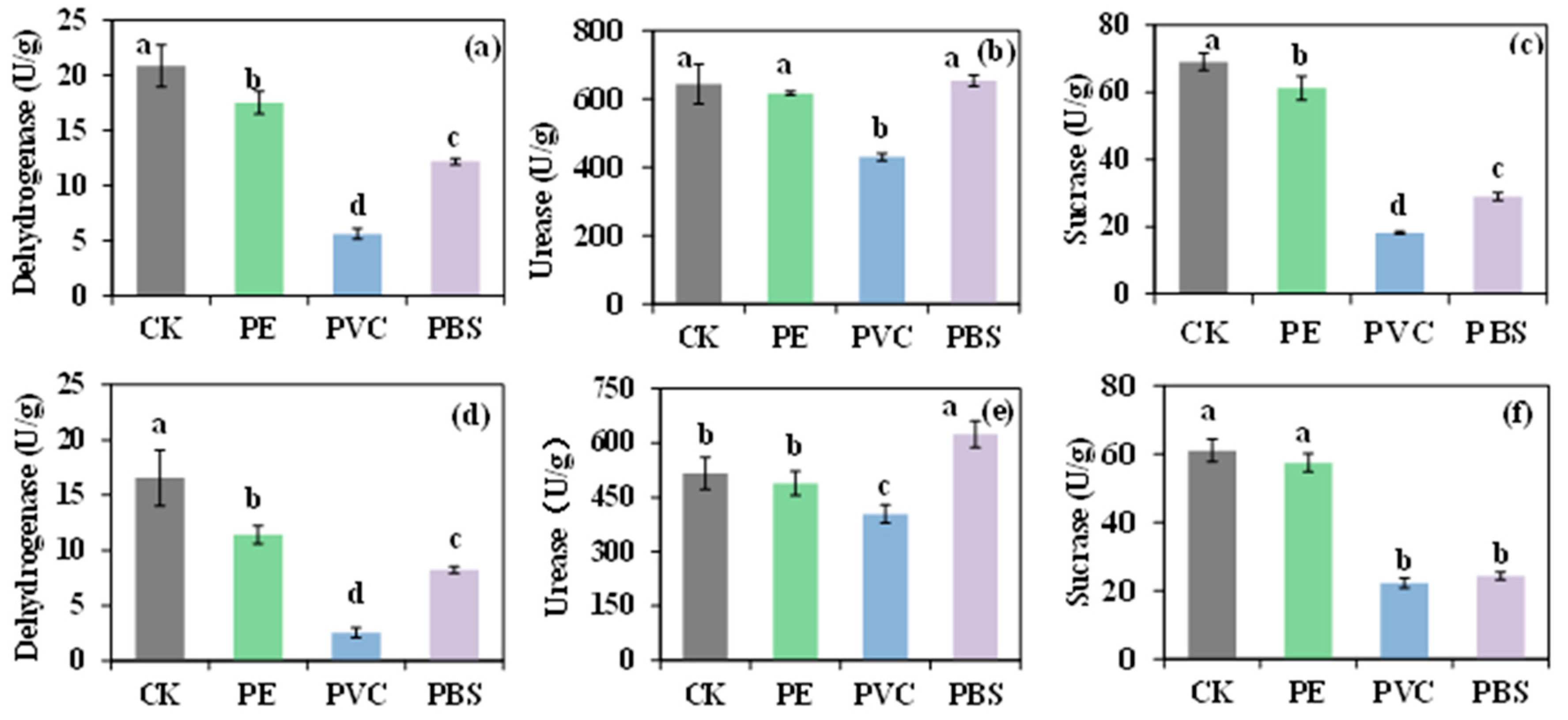
3.4. Soil Enzyme Activity
MPs exert a considerable type-specific influence on soil enzyme activity [46]. Notably, the activity of the three enzymes studied is consistently inhibited by PVC (Figure 3). This inhibition is primarily attributed to the leaching of toxic additives, such as plasticizers, which directly impair microbial cells and enzyme molecules [47]. Additionally, the stable structure of PVC facilitates the physical adsorption and immobilization of enzymes, which can potentially obstruct active sites and hinder substrate interactions. In contrast, dehydrogenase activity is suppressed by PE, likely through alterations to the soil microenvironment induced by its hydrophobic surface [48]. The biodegradable PBS can serve as a potential carbon source, stimulating specific microbial communities that consequently enhance urease secretion, thereby accelerating the mineralization of organic nitrogen [49]. However, concurrent inhibition of dehydrogenase and sucrase by PBS suggests a reallocation of microbial resources toward its degradation pathways, which may limit the synthesis of other enzyme systems. A significant variation in the impact of identical MPs on enzyme activity is observed between soil types, which is associated with their inherent properties [46]. Black soil is typically characterized by higher levels of organic matter and clay particles. Organic matter has the capacity to adsorb MPs or leached toxic compounds, thereby reducing their bioavailability and mitigating toxic effects [50]. This phenomenon may explain the differential impact of PE on sucrase activity observed between black and yellow-brown soils. Moreover, the higher inherent microbial biomass and diversity in fertile black soil contribute to greater ecosystem resilience and generally higher baseline enzyme activity compared to yellow-brown soil. Distinct soil types possess distinct microbial community structures. The introduction of MPs acts as a selective pressure, reshaping the microbial community [51]. For instance, PE can alter the relative abundance of Actinobacteria and Acidobacteria, influencing the activity of the enzymes they secrete [52]. These results indicate that the presence of MPs can significantly disrupt essential soil enzymatic functions, potentially altering degradation capacity and ecological balance. During the degradation of ATZ, MPs can alter the soil microenvironment, thereby influencing the degradation rate and pathways of the herbicide [53]. Currently, there are discrepancies in the research findings related to the influence of microbial communities on the activity of soil enzymes. This is likely due to the variable effects of different MP types under diverse soil conditions. Ultimately, interactions between pesticides and soil enzymes are inherently complex, and the introduction of MPs may further complicate these influencing factors.

Figure 3.
The activity of dehydrogenase, urease, and sucrase in black soil (a–c) and yellow brown soil (d–f) treated with atrazine. PE: polyethylene; PVC: polyvinyl chloride; PBS: polybutylene succinate. Different lowercase letters indicate significant difference at the p < 0.05 level.
3.5. Effects of MPs on ATZ Degradation in Soil
An indoor simulation experiment was conducted to assess the impact of various MPs on the ATZ degradation in soil. First-order degradation kinetics showed that the half-life (T1/2) of ATZ was significantly prolonged in MP-amended soils compared to the controls. The inhibitory effect was observed in both soils, but varied depending on the MP type and soil properties. The ATZ half-life ranged from 28.77 to 81.76 days in yellow-brown soil, and from 22.97 to 36.47 days in black soil (Table 3). PVC exhibited the strongest inhibitory effect, extending the half-life by factors of 2.84 in yellow-brown soil and 1.59 in black soil. The prolonged degradation is primarily attributed to enhanced ATZ adsorption onto MPs [54,55,56], as confirmed by kinetic and isotherm studies. The strong affinity, particularly for PBS and PE, reduces the bioavailability of ATZ to soil microorganisms, limiting its accessibility for enzyme-catalyzed breakdown. A similar occurrence was described, where MPs could hinder the degradation of imidazolinone herbicides in sediment and water [37]. Furthermore, PVC likely exerts a dual inhibitory effect: adsorption combined with intrinsic toxicity of its chlorine content and leachable additives (e.g., plasticizers) to microbial communities responsible for herbicide mineralization [57,58,59,60].

Table 3.
Degradation kinetic parameters of atrazine at 3.0 mg kg−1 in yellow-brown soil (S1) and black soil (S2) under different conditions.
Furthermore, MP-induced alterations in soil enzyme activities could affect the biodegradation ability for pesticides. PVC consistently suppressed the activity of dehydrogenase, urease, and sucrase, disrupting the microbial metabolic pathways involved in ATZ mineralization. While biodegradable PBS could stimulate specific microbial functions (e.g., urease activity), it concurrently inhibited other enzymes, indicating a complex reallocation of microbial resources that may not facilitate ATZ catabolism.
The differential resilience observed between the two soils highlights the role of soil properties. Black soil, characterized by higher organic matter and clay content, mitigated the adverse effects of MPs on ATZ degradation more effectively than yellow-brown soil. This indicates that soil type might influence the ecotoxicological risk associated with MP–pesticide interactions. This finding aligns with previous reports that pesticide degradation in soil is closely linked to soil physicochemical properties, such as pH, organic matter composition, and silt content [24,45].
3.6. Analysis of ATZ Degradation Pathway
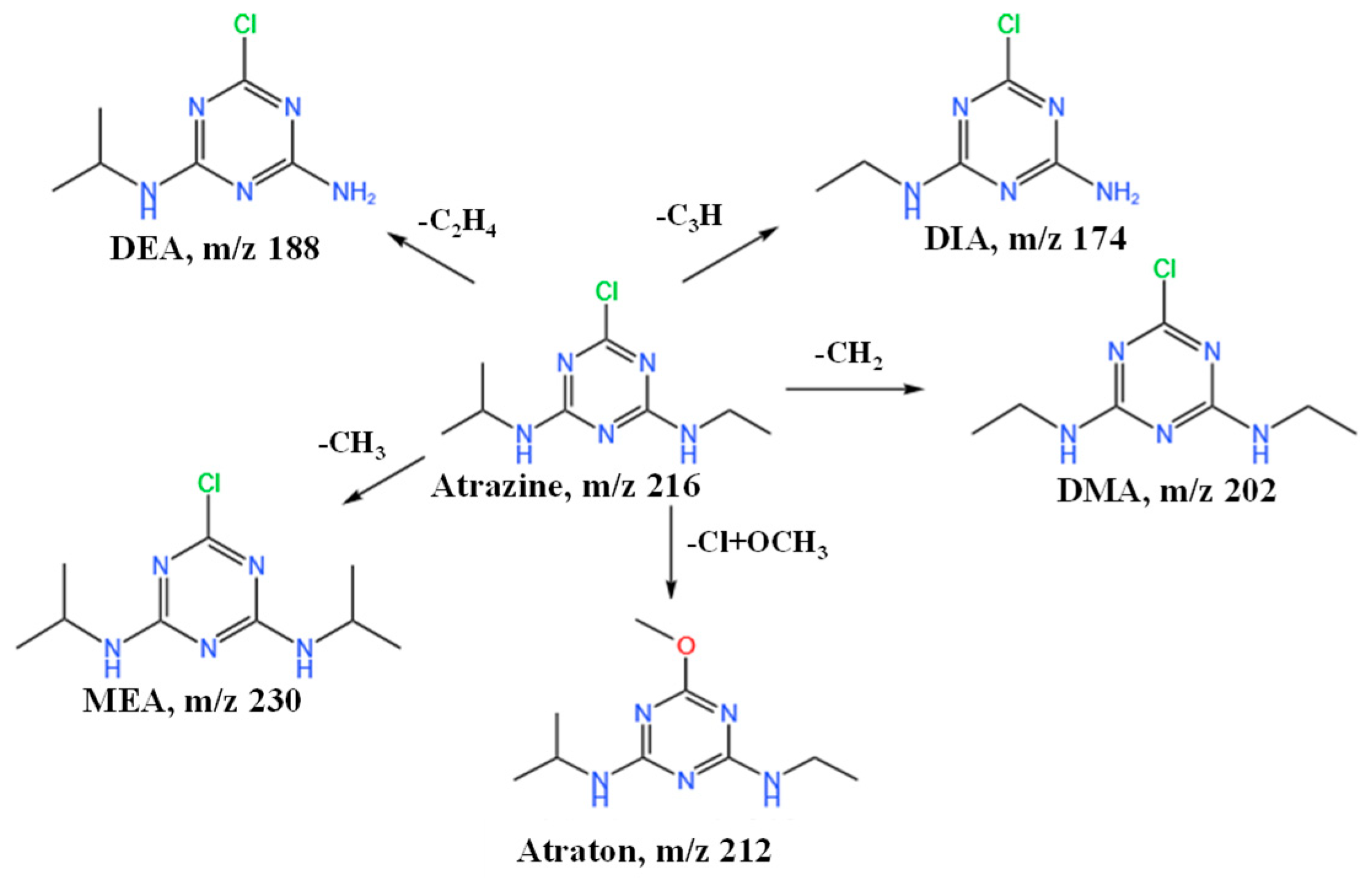
Given that ATZ degraded relatively more rapidly in soil S1, soil S1 was chosen for further examination of ATZ’s degradation products and degradation pathway. Using HRLC-Q-TOF-MS/MS, the metabolites of ATZ in the S1 soil matrix were identified. The validity and summary of the molecular and structural formulas of metabolites, including five degradation products, were determined by comparing observed and theoretical m/z values with fragmentation data (Figure 4) [24]. Atraton (N2-ethyl-N4 -isopropyl-6- methoxy-1,3,5-triazine-2,4-diamine) was detected at 6.83 min, synthesized by substituting the chlorine in ATZ with a methoxy group, displaying fragment ions with m/z 68 and 128 in the MS2 spectrum. DeethylATZ (DEA), a byproduct of ATZ resulting from N-dealkylation, peaked at 6.41 min with fragment ions (m/z 104 and 146) observed in the MS2 spectrum. The methylation of ATZ (MEA, 6-chloro-N2, N4-diisopropyl-1,3,5-triazine-2,4-diamine) peaked at 12.6 min, displaying fragment ions with m/z 146 and 188 in the MS2 spectrum. At m/z 68, 104, and 174, the product of isopropyl demethylation of ATZ, demethylated ATZ (DMA, 6-chloro-N2, N4-diethyl-1,3,5-triazine-2,4-diamine) was identified. At m/z 68, 104, and 146, demethylated product deisopropyltriATZ (DIA, 6-chloro-N2-ethyl-1,3,5-triazine-2,4 diamine) peaked at 10.79 min. The results acquired from HRLC-Q-TOF-MS/MS analysis suggest a potential mechanism for the breakdown of ATZ in soil (Figure 4). Therefore, Atraton, MEA, DEA, DMA, and DIA could be produced by physicochemical reactions, including dechlorination, hydroxylation, hydrodealkylation, methylation, dealkylation, elimination, and hydrolysis.
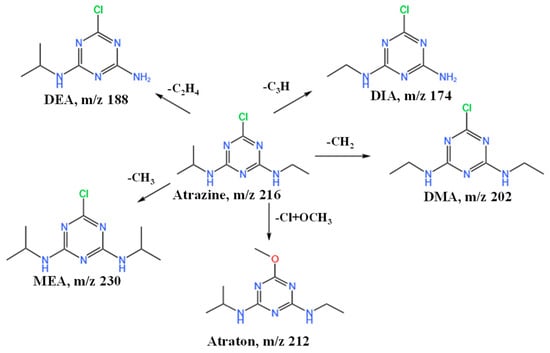
Figure 4.
Degradation pathway of atrazine in soil.
3.7. Environmental Implications
The coexistence of MPs and pesticides in agricultural soils constitutes a combined pollution issue. MPs serve as effective carriers for ATZ, enhancing its adsorption and retention in the soil matrix—with PBS exhibiting the most pronounced effect. Although this carrier effect likely reduces the mobility and leaching potential of ATZ, it concurrently reduces the herbicide’s bioavailability for microbial degradation. The resulting prolonged environmental persistence, as evidenced by the extended half-lives, increases the long-term exposure risk for soil organisms and raises the potential for accumulation in the food chain. Furthermore, the polymer-specific impacts of MPs are critical for comprehensive risk assessment. PVC poses an enhanced ecological hazard due to its dual inhibition of ATZ degradation and essential soil enzymatic activities (dehydrogenase, urease, and sucrase). The release of leachable additives combined with the physical toxicity of PVC toward soil microbiota can disrupt nutrient cycling, creating a synergistic negative effect with existing pesticide residues. Conversely, while PBS enhances adsorption, its biodegradable nature stimulates specific microbial functions (e.g., urease activity), indicating that the ecological implications of biodegradable plastics are multifaceted. Additionally, the differential responses between yellow-brown soil and black soil highlight the regulatory role of soil properties in MP–pesticide interactions. Specifically, the higher organic matter and clay contents of black soil conferred greater resilience, alleviating some of the negative impacts on enzyme activities. These findings highlight the necessity of soil ecological risk assessments.
Notably, the MP application rate of 10% (w/w) utilized in our experiments significantly exceeds typical environmental background levels, which generally remain below 1%. Consequently, there is a critical need for future studies to employ environmentally realistic MP concentrations (<1%) in order to accurately scale and evaluate true ecological risks within natural agricultural settings.
Overall, these interactions complicate the remediation of co-contaminated sites. Effective management strategies must adopt a multi-pollutant perspective, advocating for stricter regulation of plastic mulch usage, promotion of truly environmentally benign alternatives, and monitoring of enzymatic biomarkers as early indicators of the soil ecosystem.
4. Conclusions
This study systematically investigated the effects of PE, PVC, and PBS on the environmental fate of ATZ in two distinct soil types. All tested MPs enhanced the adsorption capacity and rate of ATZ. The adsorption process was best described by the pseudo-second-order kinetic model and the Freundlich isotherm model, indicating dominant chemisorption on heterogeneous surfaces of the soil–MP composites. Furthermore, the presence of MPs significantly inhibited ATZ degradation and prolonged its half-life. PVC exhibited the strongest inhibitory effect, primarily attributed to reduced ATZ bioavailability and intrinsic microbial toxicity of PVC. MPs altered soil enzyme activities in a polymer-specific manner. Notably, black soil, characterized by higher organic matter and clay content, demonstrated greater resilience against MP-induced perturbations compared to yellow-brown soil.
These findings have confirmed the necessity of incorporating the interaction between MPs and pesticides into ecological risk assessments and soil management policies. Furthermore, future research should prioritize spectroscopic validation (e.g., FTIR, XPS) to confirm the specific molecular mechanisms driving MP–pesticide adsorption. Additionally, while this study successfully identified the qualitative degradation pathways of ATZ, future investigations should employ the quantitative monitoring of metabolite dynamics over time to fully elucidate how different MP types influence specific degradation rates and the accumulation of metabolic byproducts.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agriculture16060705/s1, Table S1. Kinetic parameters for atrazine adsorption on yellow brown soil (S1) and black soil (S2); Table S2. Isotherm parameters for atrazine adsorption on yellow brown soil (S1) and black soil (S2).
Author Contributions
H.S. and X.Y. conceived the idea of the experiment; Z.C., W.Y. and X.Z. performed the test; J.C., H.Z., K.L. and L.M. analyzed the data; J.G., Y.C. and F.S. wrote and revised the paper; H.S., L.M. and J.G. funded this article. All authors have read and agreed to the published version of the manuscript.
Funding
This study was financially supported by the State Key Laboratory of Soil and Sustainable Agriculture (SKLSSA25K07), the Natural Science Foundation of Jiangsu Province (BK20191107, BK20220742), and the Agricultural Science and Technology Innovation Fund of Jiangsu Province, China (CX(22)2036).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Li, M.; Wang, Z.; Zhu, L.; Zhu, Y.; Yi, J.; Fu, X. Research advances on microplastics contamination in terrestrial geoenvironment: A review. Sci. Total Environ. 2024, 935, 173259. [Google Scholar] [CrossRef]
- Büks, F.; Loes van Schaik, N.; Kaupenjohann, M. What do we know about how the terrestrial multicellular soil fauna reacts to microplastic? Soil 2020, 6, 245–267. [Google Scholar] [CrossRef]
- de Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an emerging threat to terrestrial ecosystems. Glob. Chang. Biol. 2018, 24, 1405–1416. [Google Scholar] [CrossRef]
- Jin, T.; Tang, J.; Lyu, H.; Wang, L.; Gillmore, A.B.; Schaeffer, S.M. Activities of microplastics (MPs) in agricultural soil: A review of MPs pollution from the perspective of agricultural ecosystems. J. Agric. Food Chem. 2022, 70, 4182–4201. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, Y.; Tan, W.; Zhang, Z. Microplastics as an emerging environmental pollutant in agricultural soils: Effects on ecosystems and human health. Front. Environ. Sci. 2022, 10, 855292. [Google Scholar] [CrossRef]
- Ullah, R.; Tsui, M.T.-K.; Chow, A.; Chen, H.; Williams, C.; Ligaba-Osena, A. Micro(nano)plastic pollution in terrestrial ecosystem: Emphasis on impacts of polystyrene on soil biota, plants, animals, and humans. Environ. Monit. Assess. 2022, 195, 252. [Google Scholar] [CrossRef]
- Qi, R.; Tang, Y.; Jones, D.L.; He, W.; Yan, C. Occurrence and characteristics of microplastics in soils from greenhouse and open-field cultivation using plastic mulch film. Sci. Total Environ. 2023, 905, 166935. [Google Scholar] [CrossRef]
- Xu, D.; Jin, T.; Xi, B.; Gao, H.; Li, X.; Huang, L.; Song, J. Distribution characteristics, influencing factors, and future prospects of MPs derived from agricultural mulching film in farmland soil: A review. Curr. Opin. Environ. Sci. Health 2025, 47, 100652. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, Q.; Jia, W.; Yan, C.; Wang, J. Agricultural plastic mulching as a source of microplastics in the terrestrial environment. Environ. Pollut. 2020, 260, 114096. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Yan, C.; Liu, Q.; Ding, W.; Chen, B.; Li, Z. Effects of plastic mulching and plastic residue on agricultural production: A meta-analysis. Sci. Total Environ. 2019, 651, 484–492. [Google Scholar] [CrossRef]
- Mansoor, Z.; Tchuenbou-Magaia, F.; Kowalczuk, M.; Adamus, G.; Manning, G.; Parati, M.; Radecka, I.; Khan, H. Polymers use as mulch films in agriculture—A review of history, problems and current trends. Polymers 2022, 14, 5062. [Google Scholar] [CrossRef]
- Changrong, Y.; Wenqing, H.; Neil, C. Plastic-film mulch in Chinese agriculture: Importance and problems. World Agric 2014, 4, 32–36. [Google Scholar]
- Sharma, A.; Kumar, V.; Shahzad, B.; Tanveer, M.; Sidhu, G.P.S.; Handa, N.; Kohli, S.K.; Yadav, P.; Bali, A.S.; Parihar, R.D. Worldwide pesticide usage and its impacts on ecosystem. SN Appl. Sci. 2019, 1, 1446. [Google Scholar] [CrossRef]
- Carvalho, F.P. Pesticides, environment, and food safety. Food Energy Secur. 2017, 6, 48–60. [Google Scholar] [CrossRef]
- Zhang, W.; Jiang, F.; Ou, J. Global pesticide consumption and pollution: With China as a focus. Proc. Int. Acad. Ecol. Environ. Sci. 2011, 1, 125. [Google Scholar]
- Sun, J.T.; Pan, L.L.; Zhan, Y.; Tsang, D.C.W.; Zhu, L.Z.; Li, X.D. Atrazine contamination in agricultural soils from the Yangtze River Delta of China and associated health risks. Environ. Geochem. Health 2017, 39, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Jablonowski, N.D.; Hamacher, G.; Martinazzo, R.; Langen, U.; Köppchen, S.; Hofmann, D.; Burauel, P. Metabolism and persistence of atrazine in several field soils with different atrazine application histories. J. Agric. Food Chem. 2010, 58, 12869–12877. [Google Scholar] [CrossRef] [PubMed]
- Hayes, T.B.; Collins, A.; Lee, M.; Mendoza, M.; Noriega, N.; Stuart, A.A.; Vonk, A. Hermaphroditic, demasculinized frogs after exposure to the herbicide atrazine at low ecologically relevant doses. Proc. Natl. Acad. Sci. USA 2002, 99, 5476–5480. [Google Scholar] [CrossRef]
- Arabi, S.; Heidari-Beni, M.; Poursafa, P.; Roshanaei, M.; Kelishadi, R. A review of the potential adverse health impacts of atrazine in humans. Rev. Environ. Health 2025, 40, 347–359. [Google Scholar] [CrossRef]
- Galbiati, V.; Buoso, E.; d’Emmanuele di Villa Bianca, R.; Paola, R.D.; Morroni, F.; Nocentini, G.; Racchi, M.; Viviani, B.; Corsini, E. Immune and nervous systems interaction in endocrine disruptors toxicity: The case of atrazine. Front. Toxicol. 2021, 3, 649024. [Google Scholar] [CrossRef]
- Rohr, J.R. The atrazine saga and its importance to the future of toxicology, science, and environmental and human health. Environ. Toxicol. Chem. 2021, 40, 1544–1558. [Google Scholar] [CrossRef]
- Pathak, R.K.; Dikshit, A.K. Atrazine and human health. Int. J. Ecosyst 2011, 1, 14–23. [Google Scholar] [CrossRef]
- Mudhoo, A.; Garg, V. Sorption, transport and transformation of atrazine in soils, minerals and composts: A review. Pedosphere 2011, 21, 11–25. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, J.H.; Guo, Q.N.; Ma, L.Y.; Yang, H. Physiochemical assessment of environmental behaviors of herbicide atrazine in soils associated with its degradation and bioavailability to weeds. Chemosphere 2021, 262, 127830. [Google Scholar] [CrossRef]
- Yue, L.; Ge, C.; Feng, D.; Yu, H.; Deng, H.; Fu, B. Adsorption–desorption behavior of atrazine on agricultural soils in China. J. Environ. Sci. 2017, 57, 180–189. [Google Scholar] [CrossRef]
- Peña, A.; Rodríguez-Liébana, J.A.; Delgado-Moreno, L. Interactions of microplastics with pesticides in soils and their ecotoxicological implications. Agronomy 2023, 13, 701. [Google Scholar] [CrossRef]
- Zhang, T.; Jiang, B.; Xing, Y.; Ya, H.; Lv, M.; Wang, X. Current status of microplastics pollution in the aquatic environment, interaction with other pollutants, and effects on aquatic organisms. Environ. Sci. Pollut. Res. 2022, 29, 16830–16859. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Yu, C.; Chu, Q.; Wang, F.; Lan, T.; Wang, J. Adsorption behavior and mechanism of five pesticides on microplastics from agricultural polyethylene films. Chemosphere 2020, 244, 125491. [Google Scholar] [CrossRef]
- Zhang, C.; Lei, Y.; Qian, J.; Qiao, Y.; Liu, J.; Li, S.; Dai, L.; Sun, K.; Guo, H.; Sui, G.; et al. Sorption of organochlorine pesticides on polyethylene microplastics in soil suspension. Ecotoxicol. Environ. Saf. 2021, 223, 112591. [Google Scholar] [CrossRef]
- Mo, Q.; Yang, X.; Wang, J.; Xu, H.; Li, W.; Fan, Q.; Gao, S.; Yang, W.; Gao, C.; Liao, D. Adsorption mechanism of two pesticides on polyethylene and polypropylene microplastics: DFT calculations and particle size effects. Environ. Pollut. 2021, 291, 118120. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Asha, V.K.M.; Singh, D. Soil enzyme activities as bioindicators of soil health: Implications for sustainable agriculture. J. Nat. Resour. Conserv. Manag. 2024, 5, 124–136. [Google Scholar] [CrossRef]
- Tang, K.H.D. Effects of microplastics on bioavailability, persistence and toxicity of plant pesticides: An agricultural perspective. Agriculture 2025, 15, 356. [Google Scholar] [CrossRef]
- Chia, X.K.; Hadibarata, T.; Kristanti, R.A.; Jusoh, M.N.H.; Tan, I.S.; Foo, H.C.Y. The function of microbial enzymes in breaking down soil contaminated with pesticides: A review. Bioprocess Biosyst. Eng. 2024, 47, 597–620. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, Y.; An, N.; Jiang, D.; Cao, B.; Jiang, Z.; Yan, Y.; Ming, C.; Meng, Q.; Han, W. Short-term response of soil enzyme activities and bacterial communities in black soil to a herbicide mixture: Atrazine and Acetochlor. Appl. Soil Ecol. 2023, 181, 104652. [Google Scholar] [CrossRef]
- Hu, M.; Huang, L.; Wang, Y.; Tan, H.; Yu, X. Insight into the effect of microplastics on the adsorption and degradation behavior of thiamethoxam in agricultural soils. Chemosphere 2023, 337, 139262. [Google Scholar] [CrossRef]
- Sheng, H.; Wang, F.; Gu, C.; Stedtfeld, R.; Bian, Y.; Liu, G.; Wu, W.; Jiang, X. Sorption characteristics of N-acyl homserine lactones as signal molecules in natural soils based on the analysis of kinetics and isotherms. RSC Adv. 2018, 8, 9364–9374. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Liu, L.; Hou, N.; Li, X.; Zeng, D.; Tan, H. Insight into the adsorption mechanisms of ionizable imidazolinone herbicides in sediments: Kinetics, adsorption model, and influencing factors. Chemosphere 2021, 274, 129655. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, F.; Li, J.; Deng, S.; Zhang, S. Adsorption of three pesticides on polyethylene microplastics in aqueous solutions: Kinetics, isotherms, thermodynamics, and molecular dynamics simulation. Chemosphere 2021, 264, 128556. [Google Scholar] [CrossRef] [PubMed]
- Sheng, H.; Yin, Y.; Xiang, L.; Wang, Z.; Harindintwali, J.D.; Cheng, J.; Ge, J.; Zhang, L.; Jiang, X.; Yu, X. Sorption of N-acyl homoserine lactones on maize straw derived biochars: Characterization, kinetics and isotherm analysis. Chemosphere 2022, 299, 134446. [Google Scholar] [CrossRef]
- Zhao, L.; Rong, L.; Xu, J.; Lian, J.; Wang, L.; Sun, H. Sorption of five organic compounds by polar and nonpolar microplastics. Chemosphere 2020, 257, 127206. [Google Scholar] [CrossRef]
- Sun, M.; Yang, Y.; Huang, M.; Fu, S.; Hao, Y.; Hu, S.; Lai, D.; Zhao, L. Adsorption behaviors and mechanisms of antibiotic norfloxacin on degradable and nondegradable microplastics. Sci. Total Environ. 2022, 807, 151042. [Google Scholar] [CrossRef]
- Guo, Y.; Shao, W.; Zhao, W.; Zhu, H. The occurrence, distribution, environmental effects, and interactions of microplastics and antibiotics in the aquatic environment of China. Water 2024, 16, 1435. [Google Scholar] [CrossRef]
- Wu, C.; Pan, S.; Shan, Y.; Ma, Y.; Wang, D.; Song, X.; Hu, H.; Ren, X.; Ma, X.; Cui, J. Microplastics mulch film affects the environmental behavior of adsorption and degradation of pesticide residues in soil. Environ. Res. 2022, 214, 114133. [Google Scholar] [CrossRef]
- Đurović, R.; Gajić-Umiljendić, J.; Đorđević, T. Effects of organic matter and clay content in soil on pesticide adsorption processes. Pestic. Fitomed. 2009, 24, 51–57. [Google Scholar] [CrossRef]
- Qiu, S.; Shen, H.; Song, J.; Fang, H.; Yu, Y.; Zhang, L. Different effects of polyethylene microplastics on bioaccumulation of three fungicides in maize (Zea mays L.). Crop Health 2024, 2, 7. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, X.; Zhang, J.; Guo, X.; Li, Y.; Andom, O.; Li, Z. Microplastics alter microbial structure and assembly processes in different soil types: Driving effects of environmental factors. Environ. Res. 2025, 278, 121672. [Google Scholar] [CrossRef]
- Iftikhar, A.; Qaiser, Z.; Sarfraz, W.; Ejaz, U.; Aqeel, M.; Rizvi, Z.F.; Khalid, N. Understanding the leaching of plastic additives and subsequent risks to ecosystems. Emerg. Contam. Environ. Health 2024, 3, 5. [Google Scholar] [CrossRef]
- Liu, S.; Huang, K.; Yuan, G.; Yang, C. Effects of polyethylene microplastics and phenanthrene on soil properties, enzyme activities and bacterial communities. Processes 2022, 10, 2128. [Google Scholar] [CrossRef]
- Song, D.; Jin, G.; Su, Z.; Ge, C.; Fan, H.; Yao, H. Influence of biodegradable microplastics on soil carbon cycling: Insights from soil respiration, enzyme activity, carbon use efficiency and microbial community. Environ. Res. 2025, 266, 120558. [Google Scholar] [CrossRef]
- Prajapati, A.; Narayan Vaidya, A.; Kumar, A.R. Microplastic properties and their interaction with hydrophobic organic contaminants: A review. Environ. Sci. Pollut. Res. 2022, 29, 49490–49512. [Google Scholar] [CrossRef]
- Han, L.; Chen, L.; Feng, Y.; Kuzyakov, Y.; Chen, Q.a.; Zhang, S.; Chao, L.; Cai, Y.; Ma, C.; Sun, K.; et al. Microplastics alter soil structure and microbial community composition. Environ. Int. 2024, 185, 108508. [Google Scholar] [CrossRef]
- Yi, M.; Zhou, S.; Zhang, L.; Ding, S. The effects of three different microplastics on enzyme activities and microbial communities in soil. Water Environ. Res. 2021, 93, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Du, Y.; Yang, L.; Luo, Y.; Zhong, G.; Zhao, H.-M.; Liu, J. Effects of microplastics on the environmental behaviors of the herbicide atrazine in soil: Dissipation, adsorption, and bioconcentration. J. Hazard. Mater. 2024, 465, 133085. [Google Scholar] [CrossRef]
- Liu, Z.; Han, L.; Zhang, X.; Chen, S.; Wang, X.; Fang, H. Core bacteria carrying the genes associated with the degradation of atrazine in different soils. Environ. Int. 2023, 181, 108303. [Google Scholar] [CrossRef]
- Fan, X.; Song, F. Bioremediation of atrazine: Recent advances and promises. J. Soils Sediments 2014, 14, 1727–1737. [Google Scholar] [CrossRef]
- Fang, H.; Lian, J.; Wang, H.; Cai, L.; Yu, Y. Exploring bacterial community structure and function associated with atrazine biodegradation in repeatedly treated soils. J. Hazard. Mater. 2015, 286, 457–465. [Google Scholar] [CrossRef]
- Li, Z.; Li, J.; Zhang, J.; Wang, C. The response of soil ecosystem multifunctionality to two different microplastics of polyethylene and polyvinyl chloride. J. Soils Sediments 2025, 25, 2435–2449. [Google Scholar] [CrossRef]
- Kudzin, M.H.; Piwowarska, D.; Festinger, N.; Chruściel, J.J. Risks associated with the presence of polyvinyl chloride in the environment and methods for its disposal and utilization. Materials 2023, 17, 173. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Chen, Z.; Zhu, F.; Zhu, C.; Wang, C.; Gu, C. Effect of polyvinyl chloride microplastics on bacterial community and nutrient status in two agricultural soils. Bull. Environ. Contam. Toxicol. 2021, 107, 602–609. [Google Scholar] [CrossRef]
- Barili, S.; Bernetti, A.; Sannino, C.; Montegiove, N.; Calzoni, E.; Cesaretti, A.; Pinchuk, I.; Pezzolla, D.; Turchetti, B.; Buzzini, P. Impact of PVC microplastics on soil chemical and microbiological parameters. Environ. Res. 2023, 229, 115891. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.