Precise Monitoring of Lettuce Functional Responses to Minimal Nutrient Supplementation Identifies Aquaponic System’s Nutrient Limitations and Their Time-Course
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Laboratory Conditions
- (a)
- Control, incorporating only the recirculating aquaponic water with no further nutrient input,
- (b)
- Fe treatment, where iron was supplemented,
- (c)
- Fe+K treatment, where iron and potassium were added.
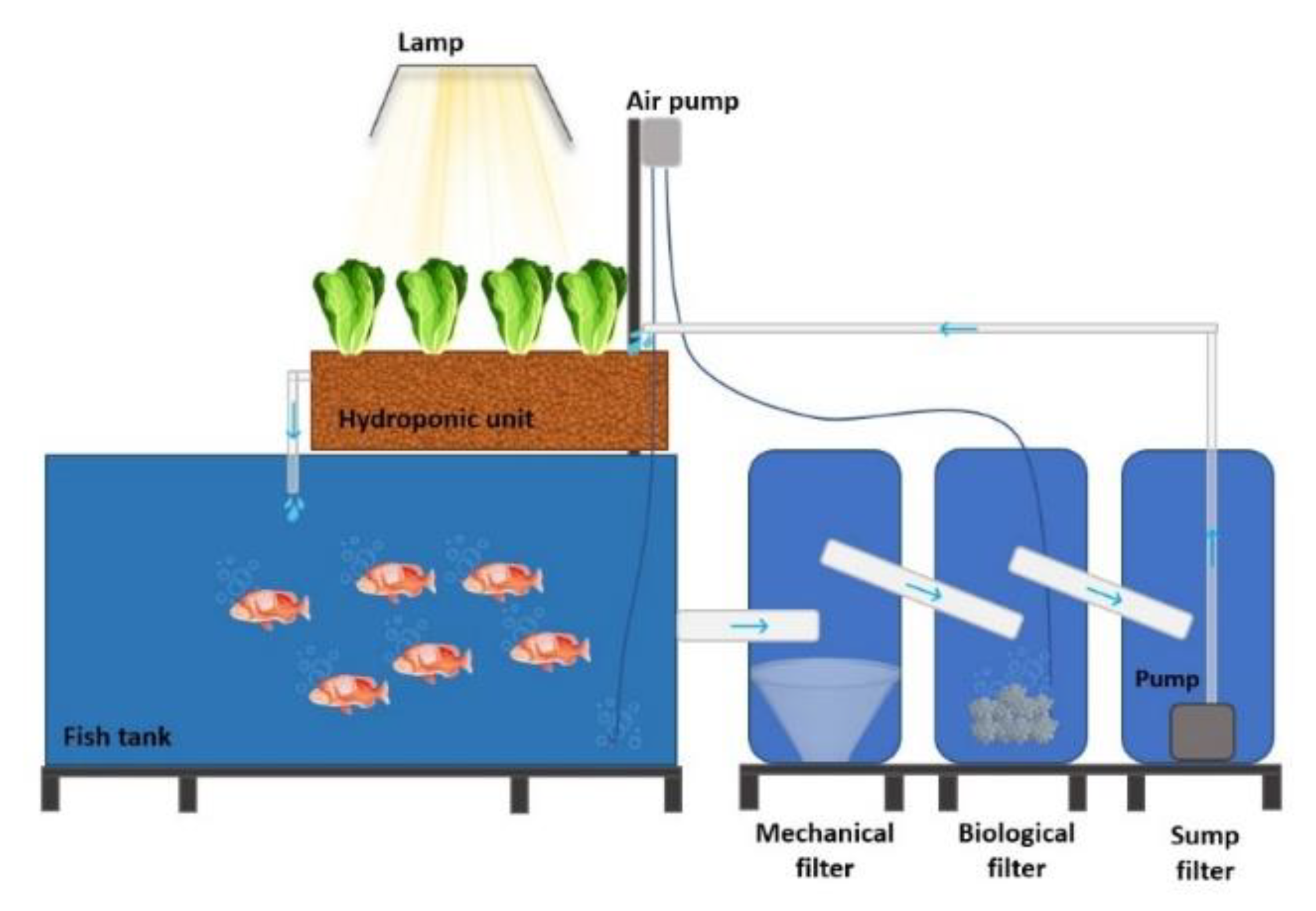
2.2. Aquaponic Systems
2.3. Monitoring of Water Physicochemical Parameters and Nutrient Concentrations
2.4. Tilapia Rearing Conditions and Measurements
2.5. Lettuce Growth Conditions
2.6. Measurement of Plant Physiological and Biochemical Characteristics during the Experimental Period
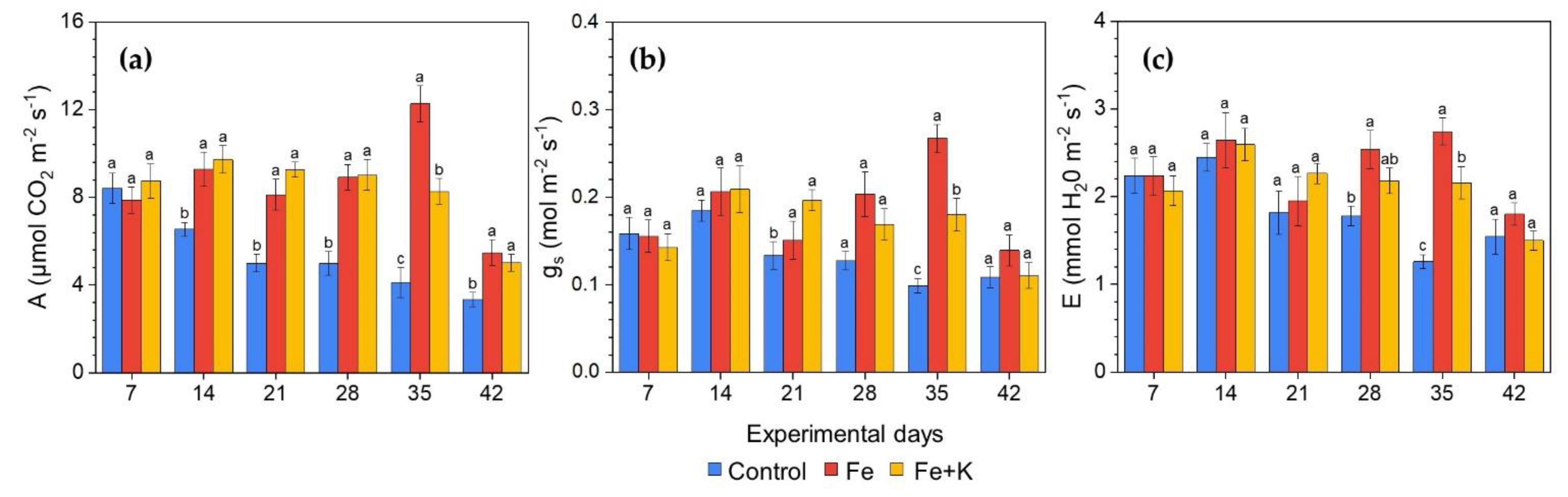
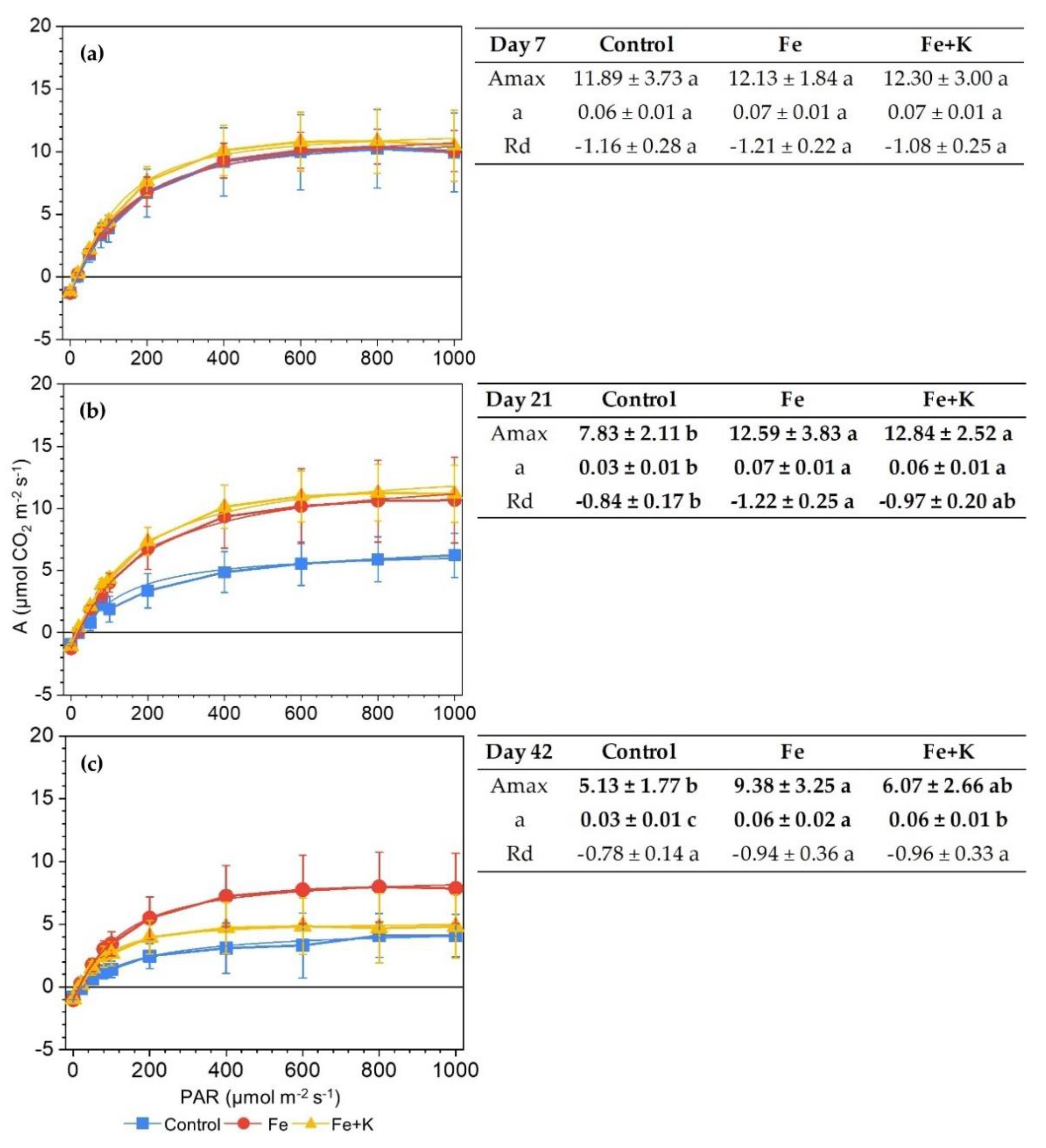
2.6.1. Gas Exchange and Light Response Curves
2.6.2. Fluorescence of Chlorophyll a In Vivo
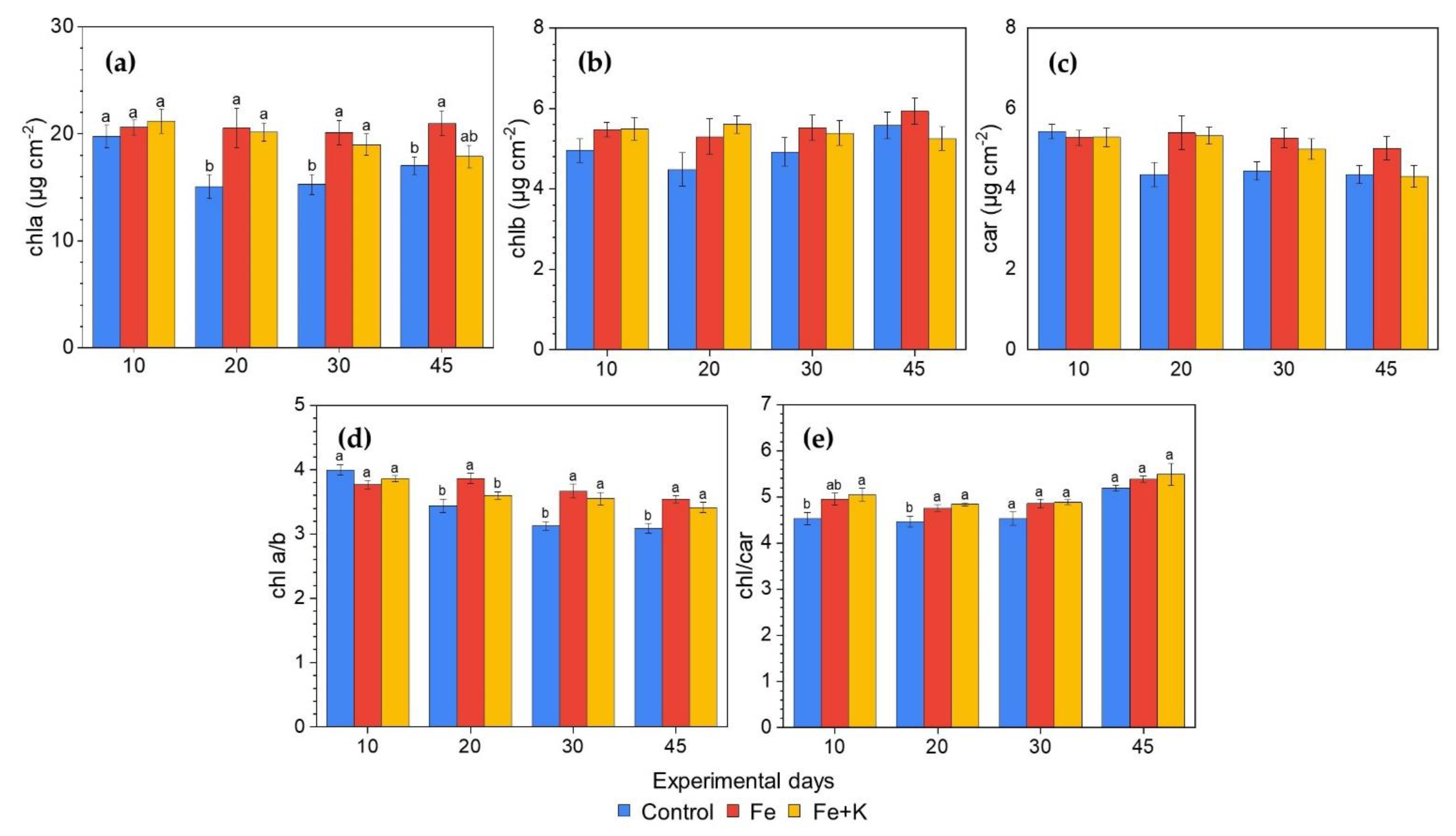
2.6.3. Photosynthetic Pigments Content
2.6.4. Elemental Tissue Analysis
2.6.5. Antioxidant Activity
2.7. Plant Growth Parameters
2.8. DNA Extraction and Isolation
2.9. Statistical Analysis
3. Results
3.1. Water Physicochemical and Quality Parameters
3.2. Fish Growth Performance
3.3. Growth and Physiological Parameters of Lettuce
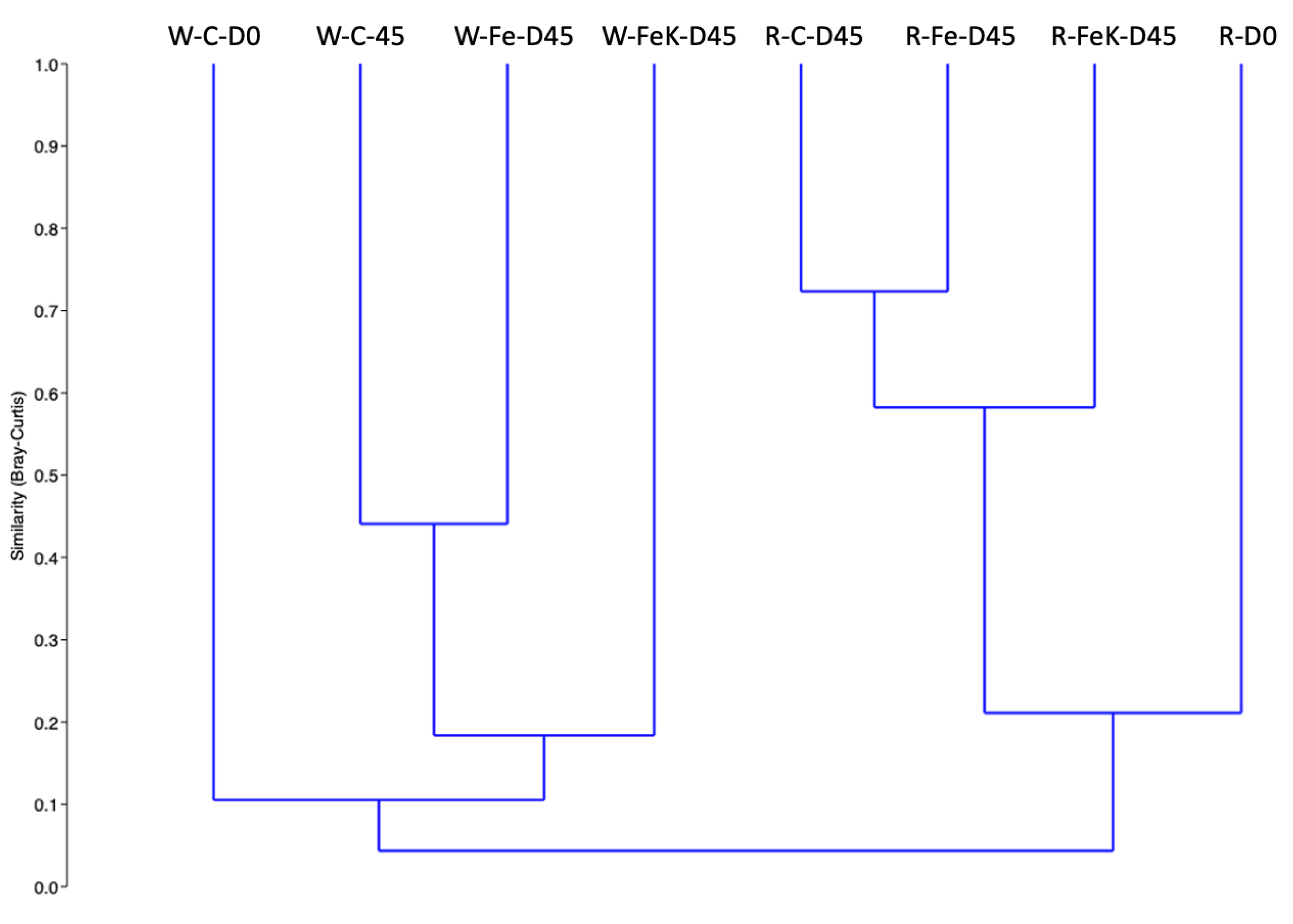
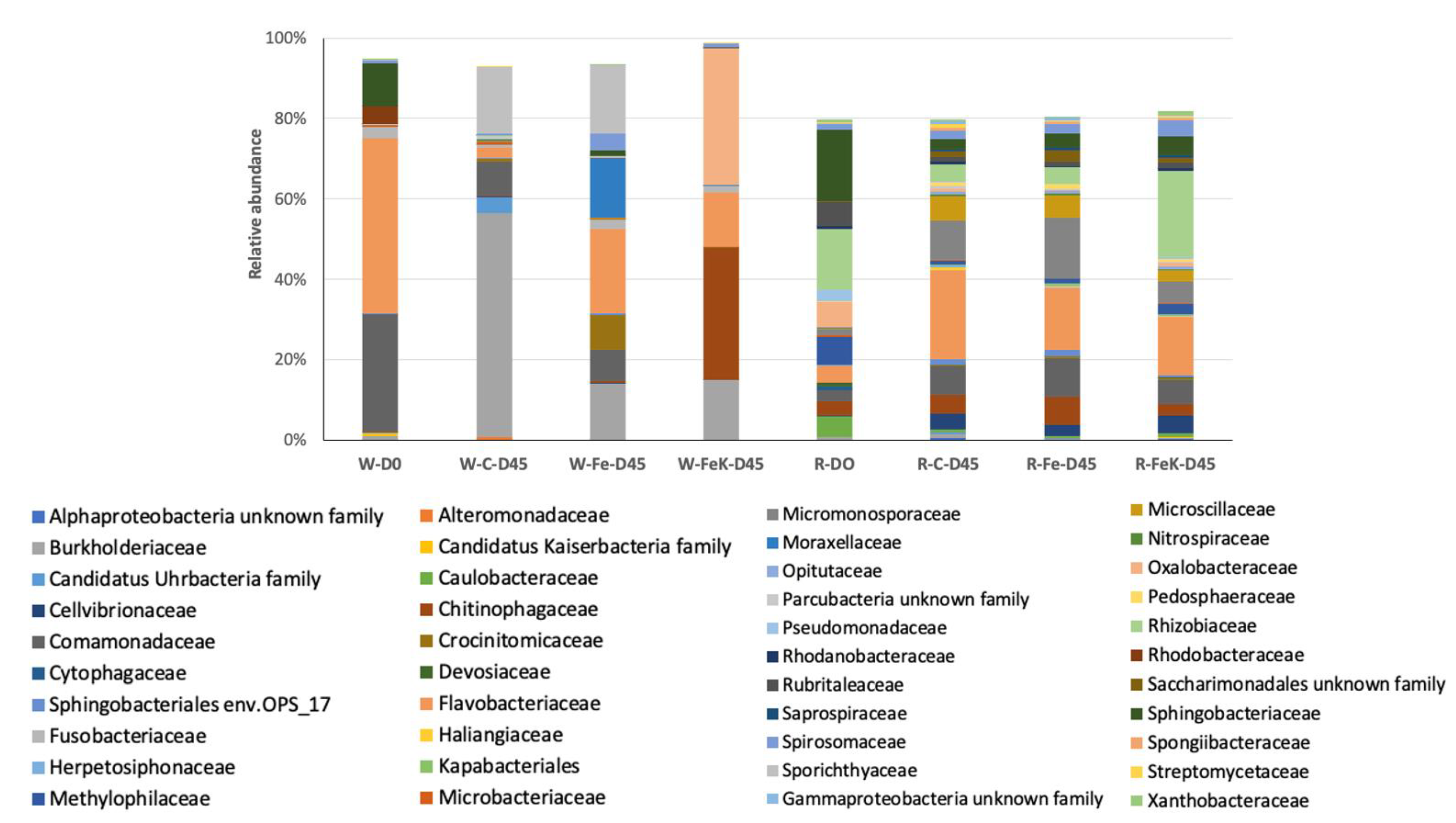
3.4. Root and Water Bacterial Community Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rakocy, J.E.; Bailey, D.S.; Shultz, R.C.; Thoman, E.S. Update on Tilapia and Vegetable Production in the UVI Aquaponic System. In New Dimensions on Farmed Tilapia, Proceedings of the Sixth International Symposium on Tilapia in Aquaculture, Manila, Philippines, 12–16 September 2004; Creative Unlimited: Cham, Switzerland, 2004. [Google Scholar]
- Delaide, B.; Delhaye, G.; Dermience, M.; Gott, J.; Soyeurt, H.; Jijakli, M.H. Plant and Fish Production Performance, Nutrient Mass Balances, Energy and Water Use of the PAFF Box, a Small-Scale Aquaponic System. Aquac. Eng. 2017, 78, 130–139. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture; Food and Agricultural Organization: Rome, Italy, 2014. [Google Scholar]
- Robaina, L.; Pirhonen, J.; Mente, E.; Sánchez, J.; Goosen, N. Fish Diets in Aquaponics. In Aquaponics Food Production Systems; Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 333–352. ISBN 978-3-030-15942-9. [Google Scholar]
- Endut, A.; Jusoh, A.; Ali, N.; Wan Nik, W.B.; Hassan, A. A Study on the Optimal Hydraulic Loading Rate and Plant Ratios in Recirculation Aquaponic System. Bioresour. Technol. 2010, 101, 1511–1517. [Google Scholar] [CrossRef] [PubMed]
- Graber, A.; Junge, R. Aquaponic Systems: Nutrient Recycling from Fish Wastewater by Vegetable Production. Desalination 2009, 246, 147–156. [Google Scholar] [CrossRef]
- Rafiee, G.R.; Ros Saad, C.; Kamarudin, M.S.; Ismail, M.R.; Sijam, K. Effects of Supplementary Nutrient in an Aquaponic System for Production of Ornamental Red Tilapia (Oreochromis sp.) and Lettuce (Lactuca Sativa var Longifolia). Surv. Fish. Sci. 2019, 5, 65–75. [Google Scholar] [CrossRef]
- Rakocy, J.E. Aquaponics:Integrating Fish and Plant Culture. Aquac. Prod. Syst. 2012, 1, 344–386. [Google Scholar]
- Roosta, H.R. Effects of Foliar Spray of K on Mint, Radish, Parsley and Coriander Plants in Aquaponic System. J. Plant Nutr. 2014, 37, 2236–2254. [Google Scholar] [CrossRef]
- Kasozi, N.; Tandlich, R.; Fick, M.; Kaiser, H.; Wilhelmi, B. Iron Supplementation and Management in Aquaponic Systems: A Review. Aquac. Rep. 2019, 15, 100221. [Google Scholar] [CrossRef]
- Molassiotis, A.; Tanou, G.; Diamantidis, G.; Patakas, A.; Therios, I. Effects of 4-Month Fe Deficiency Exposure on Fe Reduction Mechanism, Photosynthetic Gas Exchange, Chlorophyll Fluorescence and Antioxidant Defense in Two Peach Rootstocks Differing in Fe Deficiency Tolerance. J. Plant Physiol. 2006, 163, 176–185. [Google Scholar] [CrossRef]
- Delaide, B.; Goddek, S.; Gott, J.; Soyeurt, H.; Jijakli, M. Lettuce (Lactuca Sativa L. var. Sucrine) Growth Performance in Complemented Aquaponic Solution Outperforms Hydroponics. Water 2016, 8, 467. [Google Scholar] [CrossRef]
- Nicoletto, C.; Maucieri, C.; Mathis, A.; Schmautz, Z.; Komives, T.; Sambo, P.; Junge, R. Extension of Aquaponic Water Use for NFT Baby-Leaf Production: Mizuna and Rocket Salad. Agronomy 2018, 8, 75. [Google Scholar] [CrossRef]
- Roosta, H.R.; Hamidpour, M. Mineral Nutrient Content of Tomato Plants in Aquaponic and Hydroponic Systems: Effect of Foliar Application of Some Macro- and Micro-Nutrients. J. Plant Nutr. 2013, 36, 2070–2083. [Google Scholar] [CrossRef]
- Vandam, D.; Anderson, T.; de Villiers, D.; Timmons, M. Growth and Tissue Elemental Composition Response of Spinach (Spinacia Oleracea) to Hydroponic and Aquaponic Water Quality Conditions. Horticulturae 2017, 3, 32. [Google Scholar] [CrossRef]
- Buzby, K.M.; Waterland, N.L.; Semmens, K.J.; Lin, L.-S. Evaluating Aquaponic Crops in a Freshwater Flow-through Fish Culture System. Aquaculture 2016, 460, 15–24. [Google Scholar] [CrossRef]
- Ru, D.; Liu, J.; Hu, Z.; Zou, Y.; Jiang, L.; Cheng, X.; Lv, Z. Improvement of Aquaponic Performance through Micro- and Macro-Nutrient Addition. Environ. Sci. Pollut. Res. 2017, 24, 16328–16335. [Google Scholar] [CrossRef]
- Sonneveld, C.; Straver, N. Nutrient Solutions for Vegetables and Flowers Grown in Water or Substrates. Voedingspoloss. Glas. 1994, 8, 33. [Google Scholar]
- Hirayama, K. Water Control by Filtration in Closed Culture Systems. Aquaculture 1974, 4, 369–385. [Google Scholar] [CrossRef]
- Markos, N.; Kyparissis, A. Ecophysiological Modelling of Leaf Level Photosynthetic Performance for Three Mediterranean Species with Different Growth Forms. Funct. Plant Biol. 2011, 38, 314–326. [Google Scholar] [CrossRef] [PubMed]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The Fluorescence Transient as a Tool to Haracterize and Screen Photosynthetic Samples. In Probing Photosynthesis: Mechanisms, Regulation and Adaptation; Taylor & Francis: Abingdon, UK, 2000; pp. 445–483. ISBN 978-0-12-384905-2. [Google Scholar]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of Total Carotenoids and Chlorophylls a and b of Leaf Extracts in Different Solvents. Biochem. Soc. Trans. 1983, 11, 591–592. [Google Scholar] [CrossRef]
- Avdouli, D.; Max, J.F.J.; Katsoulas, N.; Levizou, E. Basil as Secondary Crop in Cascade Hydroponics: Exploring Salinity Tolerance Limits in Terms of Growth, Amino Acid Profile, and Nutrient Composition. Horticulturae 2021, 7, 203. [Google Scholar] [CrossRef]
- Goupy, P.; Hugues, M.; Boivin, P.; Amiot, M.J. Antioxidant Composition and Activity of Barley (Hordeum Vulgare) and Malt Extracts and of Isolated Phenolic Compounds. J. Sci. Food Agric. 1999, 79, 1625–1634. [Google Scholar] [CrossRef]
- Hayes, J.E.; Allen, P.; Brunton, N.; O’Grady, M.N.; Kerry, J.P. Phenolic Composition and in Vitro Antioxidant Capacity of Four Commercial Phytochemical Products: Olive Leaf Extract (Olea Europaea L.), Lutein, Sesamol and Ellagic Acid. Food Chem. 2011, 126, 948–955. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and Next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing Mothur: Open-Source, Platform-Independent, Community-Supported Software for Describing and Comparing Microbial Communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Gevers, D.; Westcott, S.L. Reducing the Effects of PCR Amplification and Sequencing Artifacts on 16S RRNA-Based Studies. PLoS ONE 2011, 6, e27310. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA Ribosomal RNA Gene Database Project: Improved Data Processing and Web-Based Tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Yilmaz, P.; Parfrey, L.W.; Yarza, P.; Gerken, J.; Pruesse, E.; Quast, C.; Schweer, T.; Peplies, J.; Ludwig, W.; Glöckner, F.O. The SILVA and “All-Species Living Tree Project (LTP)” Taxonomic Frameworks. Nucleic Acids Res. 2014, 42, D643–D648. [Google Scholar] [CrossRef]
- Sonneveld, C.; Voogt, W. Plant Nutrition of Greenhouse Crops; Springer: Dordrecht, NY, USA, 2009; ISBN 978-90-481-2531-9. [Google Scholar]
- Cai, J.; Leung, P.; Luo, Y.; Yuan, X.; Yuan, Y. Food and Agriculture Organization of the United Nations. In Improving the Performance of Tilapia Farming under Climate Variation: Perspective from Bioeconomic Modelling; FAO: Rome, Italy, 2018; ISBN 978-92-5-130162-3. [Google Scholar]
- Somerville, C.; Cohen, M.; Pantanella, E.; Stankus, A.; Lovatelli, A. Food and Agriculture Organization of the United Nations. In Small-Scale Aquaponic Food Production: Integrated Fish and Plant Farming; FAO: Rome, Italy, 2014; ISBN 978-92-5-108533-2. [Google Scholar]
- Maucieri, C.; Nicoletto, C.; Junge, R.; Schmautz, Z.; Sambo, P.; Borin, M. Hydroponic Systems and Water Management in Aquaponics: A Review. Ital. J. Agron. 2018, 13, 1–11. [Google Scholar] [CrossRef]
- Rakocy, J.E.; Masser, M.P.; Losordo, T. Recirculating Aquaculture Tank Production Systems: Aquaponics-Integrating Fish and Plant Culture; SRAC Publication: Washington, DC, USA, 2006. [Google Scholar]
- Schneider, O.; Sereti, V.; Eding, E.H.; Verreth, J.A.J. Analysis of Nutrient Flows in Integrated Intensive Aquaculture Systems. Aquac. Eng. 2005, 32, 379–401. [Google Scholar] [CrossRef]
- Randall, D.J.; Tsui, T.K.N. Ammonia Toxicity in Fish. Mar. Pollut. Bull. 2002, 45, 17–23. [Google Scholar] [CrossRef]
- Pantanella, E.; Cardarelli, M.; Colla, G.; Rea, E.; Marcucci, A. Aquaponics vs. Hydroponics: Production and Quality of Lettuce Crop. Acta Hortic. 2012, 927, 887–893. [Google Scholar] [CrossRef]
- Resh, H.M. Hydroponic Food Production: A Definitive Guidebook for the Advanced Home Gardener and the Commercial Hydroponic Grower, 7th ed.; CRC Press: Boca Raton, FL, USA, 2013; ISBN 978-1-4398-7867-5. [Google Scholar]
- da Silva, M.A.; de Alvarenga, R.; da Costa, F.F.B.; Turra, E.M.; Alves, G.F.D.O.; Manduca, L.G.; de Sales, S.C.M.; Leite, N.R.; Bezerra, V.M.; Moraes, S.G.D.S.; et al. Feeding Management Strategies to Optimize the Use of Suspended Feed for Nile Tilapia (Oreochromis Niloticus) Cultivated in Bioflocs. Aquac. Res. 2020, 51, 605–615. [Google Scholar] [CrossRef]
- Stathopoulou, P.; Tsoumalakou, E.; Levizou, E.; Vanikiotis, T.; Zaoutsos, S.; Berillis, P. Iron and Potassium Fertilization Improve Rocket Growth without Affecting Tilapia Growth and Histomorphology Characteristics in Aquaponics. Appl. Sci. 2021, 11, 5681. [Google Scholar] [CrossRef]
- Lennard, W. Aquaponic System Design Parameters: Fish to Plant Ratios (Feeding Rate Ratios). Aquaponic Solut. 2012, 3, 1–11. [Google Scholar]
- Nozzi, V.; Graber, A.; Schmautz, Z.; Mathis, A.; Junge, R. Nutrient Management in Aquaponics: Comparison of Three Approaches for Cultivating Lettuce, Mint and Mushroom Herb. Agronomy 2018, 8, 27. [Google Scholar] [CrossRef]
- Yang, T.; Kim, H.-J. Nutrient Management Regime Affects Water Quality, Crop Growth, and Nitrogen Use Efficiency of Aquaponic Systems. Sci. Hortic. 2019, 256, 108619. [Google Scholar] [CrossRef]
- Marschner, H.; Marschner, P. (Eds.) Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Elsevier: London, UK; Academic Press: Waltham, MA, USA, 2012; ISBN 978-0-12-384905-2. [Google Scholar]
- Larbi, A.; Abadía, A.; Abadía, J.; Morales, F. Down Co-Regulation of Light Absorption, Photochemistry, and Carboxylation in Fe-Deficient Plants Growing in Different Environments. Photosynth. Res. 2006, 89, 113–126. [Google Scholar] [CrossRef]
- Morales, F.; Belkhodja, R.; Abadía, A.; Abadía, J. Photosystem II Efficiency and Mechanisms of Energy Dissipation in Iron-Deficient, Field-Grown Pear Trees (Pyrus Communis L.). Photosynth. Res. 2000, 63, 9–21. [Google Scholar] [CrossRef]
- Roosta, H.R.; Estaji, A.; Niknam, F. Effect of Iron, Zinc and Manganese Shortage-Induced Change on Photosynthetic Pigments, Some Osmoregulators and Chlorophyll Fluorescence Parameters in Lettuce. Photosynthetica 2018, 56, 606–615. [Google Scholar] [CrossRef]
- Zhao, D.; Oosterhuis, D.M.; Bednarz, C.W. Influence of Potassium Deficiency on Photosynthesis, Chlorophyll Content, and Chloroplast Ultrastructure of Cotton Plants. Photosynthetica 2001, 39, 103–109. [Google Scholar] [CrossRef]
- Roosta, H.R.; Mohsenian, Y. Effects of Foliar Spray of Different Fe Sources on Pepper (Capsicum Annum L.) Plants in Aquaponic System. Sci. Hortic. 2012, 146, 182–191. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Oukarroum, A.; Alexandrov, V.; Kouzmanova, M.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Allakhverdiev, S.I.; Goltsev, V. Identification of Nutrient Deficiency in Maize and Tomato Plants by in Vivo Chlorophyll a Fluorescence Measurements. Plant Physiol. Biochem. 2014, 81, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Rai, S.; Singh, P.K.; Mankotia, S.; Swain, J.; Satbhai, S.B. Iron Homeostasis in Plants and Its Crosstalk with Copper, Zinc, and Manganese. Plant Stress 2021, 1, 100008. [Google Scholar] [CrossRef]
- Moosavi, A.A.; Ronaghi, A. Influence of Foliar and Soil Applications of Iron and Manganese on Soybean Dry Matter Yield and Iron-Manganese Relationship in a Calcareous Soil. Aust. J. Crop Sci. 2011, 5, 1550–1556. [Google Scholar] [CrossRef]
- Kabir, A.H.; Debnath, T.; Das, U.; Prity, S.A.; Haque, A.; Rahman, M.M.; Parvez, M.S. Arbuscular Mycorrhizal Fungi Alleviate Fe-Deficiency Symptoms in Sunflower by Increasing Iron Uptake and Its Availability along with Antioxidant Defense. Plant Physiol. Biochem. 2020, 150, 254–262. [Google Scholar] [CrossRef]
- Patel, M.; Fatnani, D.; Parida, A.K. Potassium Deficiency Stress Tolerance in Peanut (Arachis Hypogaea) through Ion Homeostasis, Activation of Antioxidant Defense, and Metabolic Dynamics: Alleviatory Role of Silicon Supplementation. Plant Physiol. Biochem. 2022, 182, 55–75. [Google Scholar] [CrossRef] [PubMed]
- Kasozi, N.; Kaiser, H.; Wilhelmi, B. Effect of Bacillus spp. on Lettuce Growth and Root Associated Bacterial Community in a Small-Scale Aquaponics System. Agronomy 2021, 11, 947. [Google Scholar] [CrossRef]
- Schmautz, Z.; Graber, A.; Jaenicke, S.; Goesmann, A.; Junge, R.; Smits, T.H.M. Microbial Diversity in Different Compartments of an Aquaponics System. Arch. Microbiol. 2017, 199, 613–620. [Google Scholar] [CrossRef]
- Ling, N.; Wang, T.; Kuzyakov, Y. Rhizosphere Bacteriome Structure and Functions. Nat. Commun. 2022, 13, 836. [Google Scholar] [CrossRef]
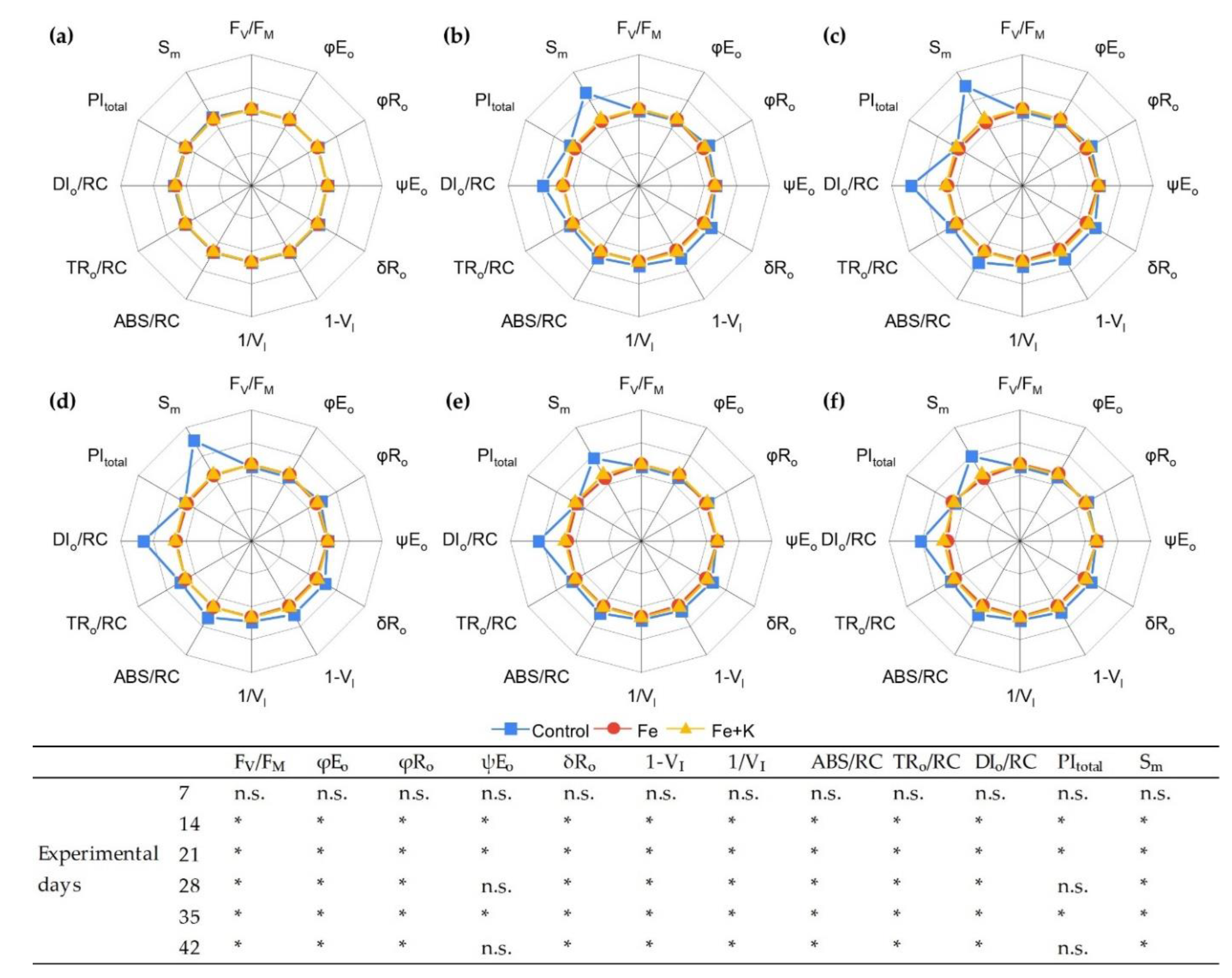

| Parameters | Explanations |
|---|---|
| Fo = F50μs | The initial value of the fluorescence. This is the first reliable fluorescence value after the illumination |
| F300μs, FJ, FI | The fluorescence values at 300 μs, 2 ms and 30 ms respectively |
| FM | The maximum value of fluorescence. This is the maximum level of OJIP curve |
| Area | Area between from fluorescence induction (OJIP) curve to maximal fluorescence FM |
| Sm = Area − FV | Normalized total complementary area above the OJIP transient |
| FV = FM − Fο | Variable chl fluorescence |
| FV/FM | Maximal quantum yield of PSII photochemistry |
| Mo = 4 (F300 μs− Fο)/(FM − Fο) | The initial slope (in ms−1) of relative variable chl fluorescence curve (for Fo = F50μs) |
| VJ = (F2 ms − Fο)/(FM − Fο) | Relative variable fluorescence at 2 ms—point J of OJIP curve |
| VI = (F30 ms − Fο)/(FM − Fο) | Relative variable fluorescence at 30 ms—point I of OJIP curve |
| φPo = TRο/ABS = FV/FM = 1 − Fο/FM | Maximum quantum yield of primary photochemistry (at t = 0) |
| φEo = ETο/ABS = φPo·ψEο = 1 − FJ/FM | Quantum yield of electron transport to intermediate acceptors |
| φRo = φPο·ψEο·δRο = 1 − FI/FM | Quantum yield of electron transport to final acceptors |
| ψEο = ETο/TRο = 1 − VJ | Probability that a trapped exciton moves an electron into the electron transport chain to intermediate acceptors |
| δRo = REο/ETο = (1 − VI)/(1 − VJ) | Probability that a trapped exciton moves an electron into the electron transport chain from intermediate receptors to final acceptors of PSI |
| 1 − VI | Yield of reactive centers of PSI |
| 1/VI | Relative yield of final acceptors e− of PSI |
| ABS/RC = (Mo/VJ)·FM/(FM − Fo) | Absorption flux (for PSII antenna chls) per reaction center (RC) |
| TRo/RC = Mo/VJ | Trapped energy flux per RC |
| DIo/RC = (Mo/VJ)·(Fo/FV) | Dissipated energy flux per RC |
| PItotal = (RC/ABS)·(φPo/1 − φPo) (ψEο/1 − ψEο)·(δRo/1 − δRo) | Index of total photosynthetic efficiency |
| Control | Fe | Fe+K | |
|---|---|---|---|
| pH | 7.04 ± 0.04 a | 7.01 ± 0.05 a | 7.08 ± 0.04 a |
| O2 (mg L−1) | 8.22 ± 0.09 a | 8.05 ± 0.08 a | 8.05 ± 0.08 a |
| EC (μS cm−1) | 924.35 ± 25.35 b | 950.29 ± 25.61 b | 1628.44 ± 61.85 a |
| NH3 (mg L−1) | 0.06 ± 0.01 a | 0.07 ± 0.01 a | 0.08 ± 0.01 a |
| NO2− (mg L−1) | 0.04 ± 0.004 a | 0.03 ± 0.004 a | 0.03 ± 0.004 a |
| NO3− (mg L−1) | 111.09 ± 11.89 a | 100.64 ± 11.05 a | 95.11 ± 10.22 a |
| PO43− (mg L−1) | 41.64 ± 2.44 a | 38.81 ± 2.03 a | 45.41 ± 3.41 a |
| SO42− (mg L−1) | 41.13 ± 1.74 b | 44.17 ± 1.38 b | 319.62 ± 28.25 a |
| Fe (mg L−1) | 0.11 ± 0.04 b | 1.67 ± 0.17 a | 1.65 ± 0.17 a |
| K (mg L−1) | 10.65 ± 1.45 b | 9.81 ± 1.35 b | 278.99 ± 25.99 a |
| Ca2+ (mg L−1) | 22.57 ± 1.92 b | 24.34 ± 2.37 b | 32.09 ± 2.92 a |
| Na (mg L−1) | 96.46 ± 6.74 a | 101.67 ± 6.36 a | 91.15 ± 5.29 a |
| Control | Fe | Fe+K | |
|---|---|---|---|
| Initial weight (g) | 15.97 ± 0.61 a | 16.06 ± 0.62 a | 16.07 ± 0.63 a |
| Initial lenght (cm) | 9.98 ± 0.14 a | 9.93 ± 0.15 a | 9.95 ± 0.15 a |
| Final weight (g) | 52.32 ± 1.62 a | 57.23 ± 2.15 a | 57.48 ± 2.11 a |
| Final lenght (cm) | 14.42 ± 0.14 a | 14.73 ± 0.17 a | 14.72 ± 0.18 a |
| SGR (% day−1) | 2.55 ± 0.04 b | 2.67 ± 0.05 a | 2.73 ± 0.04 a |
| Daily feed consumption (g day−1) | 21.84 ± 0.68 a | 23.30 ± 0.74 a | 24.22 ± 0.68 a |
| FCR | 0.93 ± 0.04 a | 0.86 ± 0.04 a | 0.84 ± 0.04 a |
| Survival (%) | 90.00 a | 88.89 a | 88.89 a |
| Lettuce Growth Performance (Mean ± SEM) | Control | Fe | Fe+K |
|---|---|---|---|
| Aerial part fresh weight (g) | 148.98 ± 20.14 b | 190.35 ± 21.97 b | 322.99 ± 42.51 a |
| Aerial part dry weight (g) | 6.77 ± 0.71 c | 9.18 ± 0.95 b | 14.26 ± 1.65 a |
| Root dry weight (g) | 1.19 ± 0.35 b | 2.05 ± 0.57 a | 2.65 ± 0.66 a |
| Root to shoot ratio | 0.15 ± 0.03 a | 0.19 ± 0.03 a | 0.17 ± 0.02 a |
| Number of leaves | 32.54 ± 1.50 b | 34.83 ± 1.53 b | 41.96 ± 1.88 a |
| Leaf area (cm2) | 2230.80 ± 275.15 b | 2809.52 ± 320.01 ab | 4205.98 ± 527.83 a |
| Control | Fe | Fe+K | ||
|---|---|---|---|---|
| % | N | 3.39 ± 0.22 a | 3.28 ± 0.03 a | 3.39 ± 0.26 a |
| P | 0.70 ± 0.05 a | 0.77 ± 0.04 a | 0.76 ± 0.02 a | |
| K | 6.87 ± 0.28 b | 5.93 ± 0.22 b | 7.56 ± 0.53 a | |
| Ca | 1.46 ± 0.11 a | 1.36 ± 0.10 a | 1.20 ± 0.12 b | |
| Mg | 0.37 ± 0.04 b | 0.43 ± 0.01 a | 0.30 ± 0.02 c | |
| ppm | Fe | 54.70 ± 6.52 b | 82.99 ± 7.02 a | 90.01 ± 15.00 a |
| Zn | 19.14 ± 2.30 a | 23.01 ± 1.56 a | 23.63 ± 2.47 a | |
| Mn | 7.00 ± 0.68 b | 24.44 ± 1.35 a | 23.96 ± 2.86 a | |
| Cu | 5.31 ± 0.26 a | 5.59 ± 0.68 a | 7.04 ± 1.76 a | |
| Treatment | No. of OTUs | Simpson 1-D ± SD | Relative Abundance of the Most Abundant OTU | No. of Dominant OTUs * |
|---|---|---|---|---|
| W-D0 | 394 | 0.73 | Flavobacterium sp. 41.7% | 4 |
| W-C-D45 | 367 | 0.65 | Polynucleobacter sp. 55.6% | 3 |
| W-Fe-D45 | 379 | 0.89 | Flavobacterium sp. 18.5% | 8 |
| W-FeK-D45 | 215 | 0.74 | Oxalobacteraceae 34.0% | 3 |
| R-D0 | 581 | 0.98 | Rhizobiaceae 8.3% | 67 |
| R-C-D45 | 845 | 0.96 | Flavobacterium sp. 12.0% | 91 |
| R-Fe-D45 | 791 | 0.95 | Actinoplanes sp. 15.0% | 75 |
| R-FeK-D45 | 676 | 0.97 | Rhizobiaceae 11.0% | 78 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsoumalakou, E.; Mente, E.; Kormas, K.A.; Katsoulas, N.; Vlahos, N.; Kapsis, P.; Levizou, E. Precise Monitoring of Lettuce Functional Responses to Minimal Nutrient Supplementation Identifies Aquaponic System’s Nutrient Limitations and Their Time-Course. Agriculture 2022, 12, 1278. https://doi.org/10.3390/agriculture12081278
Tsoumalakou E, Mente E, Kormas KA, Katsoulas N, Vlahos N, Kapsis P, Levizou E. Precise Monitoring of Lettuce Functional Responses to Minimal Nutrient Supplementation Identifies Aquaponic System’s Nutrient Limitations and Their Time-Course. Agriculture. 2022; 12(8):1278. https://doi.org/10.3390/agriculture12081278
Chicago/Turabian StyleTsoumalakou, Evangelia, Eleni Mente, Konstantinos A. Kormas, Nikolaos Katsoulas, Nikolaos Vlahos, Panagiotis Kapsis, and Efi Levizou. 2022. "Precise Monitoring of Lettuce Functional Responses to Minimal Nutrient Supplementation Identifies Aquaponic System’s Nutrient Limitations and Their Time-Course" Agriculture 12, no. 8: 1278. https://doi.org/10.3390/agriculture12081278
APA StyleTsoumalakou, E., Mente, E., Kormas, K. A., Katsoulas, N., Vlahos, N., Kapsis, P., & Levizou, E. (2022). Precise Monitoring of Lettuce Functional Responses to Minimal Nutrient Supplementation Identifies Aquaponic System’s Nutrient Limitations and Their Time-Course. Agriculture, 12(8), 1278. https://doi.org/10.3390/agriculture12081278









