Quantitative Flow Ratio Is Related to Anatomic Left Main Stem Lesion Parameters as Assessed by Intravascular Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Intravascular Imaging: Image Acquisition and Analysis
2.3. QFR Analysis
2.4. Statistical Analysis
3. Results
3.1. Feasibility of QFR in the Assessment of LMS Disease
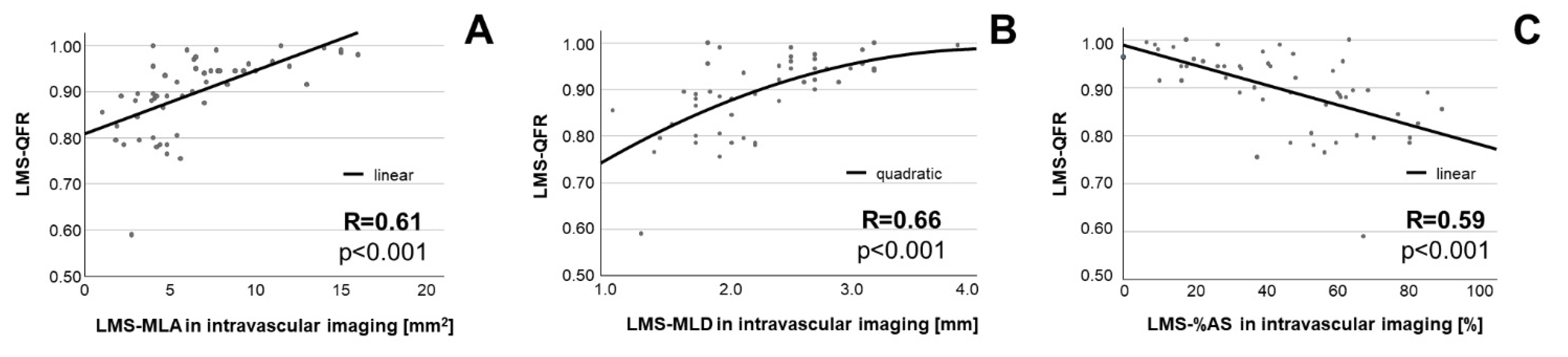
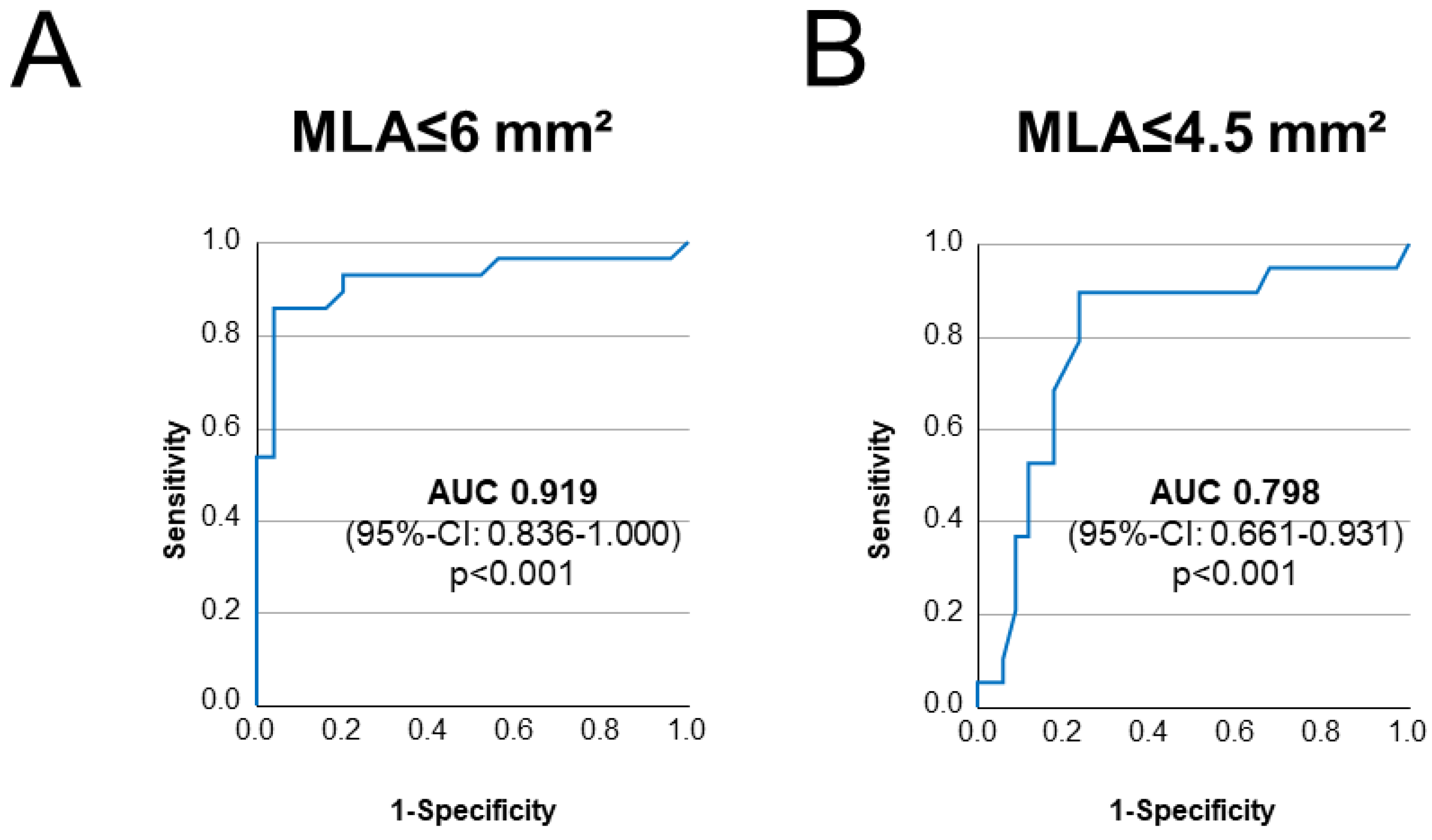
3.2. Association of QFR with Intravascular Imaging
3.3. Impact of Lesion Localization and Downstream Disease
4. Discussion
4.1. QFR Is Feasible in Assessing LMS Disease
4.2. Anatomic Severity of LMS Disease Shows Association with QFR
4.3. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Collet, C.; Capodanno, D.; Onuma, Y.; Banning, A.; Stone, G.W.; Taggart, D.P.; Sabik, J.; Serruys, P.W. Left main coronary artery disease: Pathophysiology, diagnosis, and treatment. Nat. Rev. Cardiol. 2018, 15, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Conley, M.J.; Ely, R.L.; Kisslo, J.; Lee, K.L.; McNeer, J.F.; Rosati, R.A. The prognostic spectrum of left main stenosis. Circulation 1978, 57, 947–952. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.M.; Roh, J.H.; Kim, Y.H.; Park, D.W.; Yun, S.C.; Lee, P.H.; Park, S.J. Randomized Trial of Stents Versus Bypass Surgery for Left Main Coronary Artery Disease: 5-Year Outcomes of the PRECOMBAT Study. J. Am. Coll. Cardiol. 2015, 65, 2198–2206. [Google Scholar] [CrossRef] [PubMed]
- Buszman, P.E.; Buszman, P.P.; Banasiewicz-Szkrobka, I.; Milewski, K.P.; Zurakowski, A.; Orlik, B.; Bochenek, A. Left Main Stenting in Comparison with Surgical Revascularization: 10-Year Outcomes of the (Left Main Coronary Artery Stenting) LE MANS Trial. JACC Cardiovasc. Interv. 2016, 9, 318–327. [Google Scholar] [CrossRef]
- Buszman, P.E.; Kiesz, S.R.; Bochenek, A.; Peszek-Przybyla, E.; Szkrobka, I.; Debinski, M.; Bialkowska, B.; Dudek, D.; Gruszka, A.; Zurakowski, A.; et al. Acute and late outcomes of unprotected left main stenting in comparison with surgical revascularization. J. Am. Coll. Cardiol. 2008, 51, 538–545. [Google Scholar] [CrossRef]
- Holm, N.R.; Makikallio, T.; Lindsay, M.M.; Spence, M.S.; Erglis, A.; Menown, I.B.A.; Trovik, T.; Kellerth, T.; Kalinauskas, G.; Mogensen, L.J.H.; et al. Percutaneous coronary angioplasty versus coronary artery bypass grafting in the treatment of unprotected left main stenosis: Updated 5-year outcomes from the randomised, non-inferiority NOBLE trial. Lancet 2020, 395, 191–199. [Google Scholar] [CrossRef]
- Makikallio, T.; Holm, N.R.; Lindsay, M.; Spence, M.S.; Erglis, A.; Menown, I.B.; Trovik, T.; Eskola, M.; Romppanen, H.; Kellerth, T.; et al. Percutaneous coronary angioplasty versus coronary artery bypass grafting in treatment of unprotected left main stenosis (NOBLE): A prospective, randomised, open-label, non-inferiority trial. Lancet 2016, 388, 2743–2752. [Google Scholar] [CrossRef]
- Park, D.W.; Seung, K.B.; Kim, Y.H.; Lee, J.Y.; Kim, W.J.; Kang, S.J.; Lee, S.-W.; Lee, C.W.; Park, S.-W.; Yun, S.-C.; et al. Long-term safety and efficacy of stenting versus coronary artery bypass grafting for unprotected left main coronary artery disease: 5-year results from the MAIN-COMPARE (Revascularization for Unprotected Left Main Coronary Artery Stenosis: Comparison of Percutaneous Coronary Angioplasty Versus Surgical Revascularization) registry. J. Am. Coll. Cardiol. 2010, 56, 117–124. [Google Scholar]
- Park, S.J.; Kim, Y.H.; Park, D.W.; Yun, S.C.; Ahn, J.M.; Song, H.G.; Lee, J.-Y.; Kim, W.-J.; Kang, S.-J.; Lee, S.-W.; et al. Randomized trial of stents versus bypass surgery for left main coronary artery disease. N. Engl. J. Med. 2011, 364, 1718–1727. [Google Scholar] [CrossRef]
- Stone, G.W.; Sabik, J.F.; Serruys, P.W.; Simonton, C.A.; Genereux, P.; Puskas, J.; Kandzari, D.E.; Morice, M.-C.; Lembo, N.; Brown, W.M.; et al. Everolimus-Eluting Stents or Bypass Surgery for Left Main Coronary Artery Disease. N. Engl. J. Med. 2016, 375, 2223–2235. [Google Scholar] [CrossRef]
- Hamilos, M.; Muller, O.; Cuisset, T.; Ntalianis, A.; Chlouverakis, G.; Sarno, G.; Nelis, O.; Bartunek, J.; Vanderheyden, M.; Wyffels, E.; et al. Long-term clinical outcome after fractional flow reserve-guided treatment in patients with angiographically equivocal left main coronary artery stenosis. Circulation 2009, 120, 1505–1512. [Google Scholar] [CrossRef]
- Fearon, W.F.; Yong, A.S.; Lenders, G.; Toth, G.G.; Dao, C.; Daniels, D.V.; De Bruyne, B. The impact of downstream coronary stenosis on fractional flow reserve assessment of intermediate left main coronary artery disease: Human validation. JACC Cardiovasc. Interv. 2015, 8, 398–403. [Google Scholar] [CrossRef]
- Yong, A.S.; Daniels, D.; De Bruyne, B.; Kim, H.S.; Ikeno, F.; Lyons, J.; Fearon, W.F. Fractional flow reserve assessment of left main stenosis in the presence of downstream coronary stenoses. Circ. Cardiovasc. Interv. 2013, 6, 161–165. [Google Scholar] [CrossRef]
- Neumann, F.J.; Sousa-Uva, M.; Ahlsson, A.; Alfonso, F.; Banning, A.P.; Benedetto, U.; Zembala, M.O. 2018 ESC/EACTS Guidelines on myocardial revascularization. Kardiol. Pol. 2018, 76, 1585–1664. [Google Scholar] [CrossRef]
- De la Torre Hernandez, J.M.; Hernandez Hernandez, F.; Alfonso, F.; Rumoroso, J.R.; Lopez-Palop, R.; Sadaba, M.; LITRO Study Group. Prospective application of pre-defined intravascular ultrasound criteria for assessment of intermediate left main coronary artery lesions results from the multicenter LITRO study. J. Am. Coll. Cardiol. 2011, 58, 351–358. [Google Scholar] [CrossRef]
- Park, S.J.; Ahn, J.M.; Kang, S.J.; Yoon, S.H.; Koo, B.K.; Lee, J.Y.; Kim, W.-J.; Park, D.-W.; Lee, S.-W.; Kim, Y.-H.; et al. Intravascular ultrasound-derived minimal lumen area criteria for functionally significant left main coronary artery stenosis. JACC Cardiovasc. Interv. 2014, 7, 868–874. [Google Scholar] [CrossRef]
- Johnson, T.W.; Raber, L.; Di Mario, C.; Bourantas, C.V.; Jia, H.; Mattesini, A.; Gonzalo, N.; Hernandez, J.M.D.L.T.; Prati, F.; Koskinas, K.C.; et al. Clinical use of intracoronary imaging. Part2: Acute coronary syndromes, ambiguous coronary angiography findings, and guiding interventional decision-making: An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. EuroIntervention 2019, 15, 434–451. [Google Scholar]
- Tu, S.; Barbato, E.; Koszegi, Z.; Yang, J.; Sun, Z.; Holm, N.R.; Reiber, J.H. Fractional flow reserve calculation from 3-dimensional quantitative coronary angiography and TIMI frame count: A fast computer model to quantify the functional significance of moderately obstructed coronary arteries. JACC Cardiovasc. Interv. 2014, 7, 768–777. [Google Scholar] [CrossRef]
- Tu, S.; Westra, J.; Yang, J.; von Birgelen, C.; Ferrara, A.; Pellicano, M.; FAVOR Pilot Trial Study Group. Diagnostic Accuracy of Fast Computational Approaches to Derive Fractional Flow Reserve from Diagnostic Coronary Angiography: The International Multicenter FAVOR Pilot Study. JACC Cardiovasc. Interv. 2016, 9, 2024–2035. [Google Scholar] [CrossRef]
- Westra, J.; Andersen, B.K.; Campo, G.; Matsuo, H.; Koltowski, L.; Eftekhari, A.; Liu, T.; Di Serafino, L.; Di Girolamo, D.; Escaned, J.; et al. Diagnostic Performance of In-Procedure Angiography-Derived Quantitative Flow Reserve Compared to Pressure-Derived Fractional Flow Reserve: The FAVOR II Europe-Japan Study. J. Am. Heart Assoc. 2018, 7, e009603. [Google Scholar] [CrossRef]
- Xu, B.; Tu, S.; Qiao, S.; Qu, X.; Chen, Y.; Yang, J.; Guo, L.; Sun, Z.; Li, Z.; Tian, F.; et al. Diagnostic Accuracy of Angiography-Based Quantitative Flow Ratio Measurements for Online Assessment of Coronary Stenosis. J. Am. Coll. Cardiol. 2017, 70, 3077–3087. [Google Scholar] [CrossRef]
- Lauri, F.M.; Macaya, F.; Mejia-Renteria, H.; Goto, S.; Yeoh, J.; Nakayama, M.; Quirós, A.; Liontou, C.; Pareek, N.; Fernández-Ortíz, A.; et al. Angiography-derived functional assessment of non-culprit coronary stenoses in primary percutaneous coronary intervention. EuroIntervention 2020, 15, e1594–e1601. [Google Scholar] [CrossRef]
- Sejr-Hansen, M.; Westra, J.; Thim, T.; Christiansen, E.H.; Eftekhari, A.; Kristensen, S.D.; Maeng, M. Quantitative flow ratio for immediate assessment of nonculprit lesions in patients with ST-segment elevation myocardial infarction-An iSTEMI substudy. Catheter. Cardiovasc. Interv. 2019, 94, 686–692. [Google Scholar] [CrossRef]
- Spitaleri, G.; Tebaldi, M.; Biscaglia, S.; Westra, J.; Brugaletta, S.; Erriquez, A.; Passarini, G.; Brieda, A.; Leone, A.M.; Picchi, A.; et al. Quantitative Flow Ratio Identifies Nonculprit Coronary Lesions Requiring Revascularization in Patients with ST-Segment-Elevation Myocardial Infarction and Multivessel Disease. Circ. Cardiovasc. Interv. 2018, 11, e006023. [Google Scholar] [CrossRef]
- Milzi, A.; Dettori, R.; Marx, N.; Reith, S.; Burgmaier, M. Quantitative flow ratio (QFR) identifies functional relevance of non-culprit lesions in coronary angiographies of patients with acute myocardial infarction. Clin. Res. Cardiol. 2021, 110, 1659–1667. [Google Scholar] [CrossRef]
- Milzi, A.; Dettori, R.; Burgmaier, K.; Marx, N.; Reith, S.; Burgmaier, M. Quantitative Flow Ratio Is Related to Intraluminal Coronary Stenosis Parameters as Assessed with Optical Coherence Tomography. J. Clin. Med. 2021, 10, 1856. [Google Scholar] [CrossRef]
- Raber, L.; Mintz, G.S.; Koskinas, K.C.; Johnson, T.W.; Holm, N.R.; Onuma, Y.; ESC Scientific Document Group Alfonso Fernando Bhindi Ravinay Ali Ziad Carter Rickey. Clinical use of intracoronary imaging. Part 1: Guidance and optimization of coronary interventions. An expert consensus document of the European Association of Percutaneous Cardiovascular Interventions. Eur. Heart J. 2018, 39, 3281–3300. [Google Scholar] [CrossRef]
- Simundic, A.M. Measures of Diagnostic Accuracy: Basic Definitions. EJIFCC 2009, 19, 203–211. [Google Scholar] [PubMed]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, A.; Chen, Y.; Zanchin, T.; Jones, D.A.; Rathod, K.; Jin, C.; Bourantas, C.V. Optical coherence tomography enables more accurate detection of functionally significant intermediate non-left main coronary artery stenoses than intravascular ultrasound: A meta-analysis of 6919 patients and 7537 lesions. Int. J. Cardiol. 2020, 301, 226–234. [Google Scholar] [CrossRef] [PubMed]

| n = 53 | |
|---|---|
| Male sex (n,%) | 37 (70%) |
| Age (years) | 70.8 ± 9.5 |
| NSTEMI at presentation (n,%) | 11 (21%) |
| T2DM (n,%) | 24 (45%) |
| Hypertension (n,%) | 41 (77%) |
| Nicotine use (n,%) | 14 (26%) |
| Packyears (PY) | 10.5 ± 15.0 |
| BMI (g/m2) | 28.8 ± 4.8 |
| LVEF (%) | 47.1 ± 9.5 |
| Lab values | |
| Cholesterol (mg/dL) | 174 ± 56 |
| LDL-c (mg/dL) | 109 ± 51 |
| HDL-c (mg/dL) | 45 ± 11 |
| Triglycerides (mg/dL) | 149 ± 76 |
| HbA1c (%) | 6.2 ± 1.1 |
| Lesion characteristics | |
| Lesion localization | |
| Proximal | 9 (17%) |
| Shaft | 8 (15.1%) |
| Distal | 34 (64.2%) |
| Diffuse | 2 (3.8%) |
| Medina classification | |
| 1, 0, 0 | 11 (20.8%) |
| 1, 1, 0 | 16 (30.2%) |
| 1, 0, 1 | 11 (20.8%) |
| 1, 1, 1 | 15 (28.3%) |
| MLA (mm2) | 6.5 ± 3.7 |
| MLD in intravascular imaging (mm) | 2.43 ± 0.73 |
| QCA-based MLD (mm) | 2.22 ± 0.69 |
| Percent diameter stenosis (%) | 34 ± 14 |
| Lesion length (mm) | 7.9 ± 3.3 |
| Average LMS-QFR | 0.90 ± 0.08 |
| LAD involvement (n,%) | 33 (62%) |
| LCx involvement (n,%) | 24 (45%) |
| LMS-QFR | LMS-MLA ≤ 6 mm2 | LMS-MLA ≤ 4.5 mm2 | ||
|---|---|---|---|---|
| PPV | NPV | PPV | NPV | |
| 0.75 | 1.00 | 0.48 | 1.00 | 0.65 |
| 0.80 | 1.00 | 0.58 | 0.70 | 0.72 |
| 0.85 | 1.00 | 0.62 | 0.69 | 0.75 |
| 0.90 | 0.92 | 0.85 | 0.65 | 0.93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milzi, A.; Dettori, R.; Lubberich, R.K.; Burgmaier, K.; Marx, N.; Reith, S.; Burgmaier, M. Quantitative Flow Ratio Is Related to Anatomic Left Main Stem Lesion Parameters as Assessed by Intravascular Imaging. J. Clin. Med. 2022, 11, 6024. https://doi.org/10.3390/jcm11206024
Milzi A, Dettori R, Lubberich RK, Burgmaier K, Marx N, Reith S, Burgmaier M. Quantitative Flow Ratio Is Related to Anatomic Left Main Stem Lesion Parameters as Assessed by Intravascular Imaging. Journal of Clinical Medicine. 2022; 11(20):6024. https://doi.org/10.3390/jcm11206024
Chicago/Turabian StyleMilzi, Andrea, Rosalia Dettori, Richard Karl Lubberich, Kathrin Burgmaier, Nikolaus Marx, Sebastian Reith, and Mathias Burgmaier. 2022. "Quantitative Flow Ratio Is Related to Anatomic Left Main Stem Lesion Parameters as Assessed by Intravascular Imaging" Journal of Clinical Medicine 11, no. 20: 6024. https://doi.org/10.3390/jcm11206024
APA StyleMilzi, A., Dettori, R., Lubberich, R. K., Burgmaier, K., Marx, N., Reith, S., & Burgmaier, M. (2022). Quantitative Flow Ratio Is Related to Anatomic Left Main Stem Lesion Parameters as Assessed by Intravascular Imaging. Journal of Clinical Medicine, 11(20), 6024. https://doi.org/10.3390/jcm11206024





