Abstract
Mycoplasma hominis is a habitual colonizing microorganism of the lower genital tract but can exceptionally be the causal agent of blood culture-negative infective endocarditis (IE). Only 11 cases of this entity have been published to date. The study objectives were to describe the first case diagnosed in our center of IE by M. hominis on pacemaker lead and to carry out a narrative review. Among published cases of IE by this microorganism, 72.7% were male, with a mean age of 45 years and a history of valve surgery; the diagnosis was by culture (54.5%) or molecular technique (45.5%), and the prognosis was favorable in 72.7% of cases. The most frequently prescribed antibiotics were doxycycline, quinolones, and clindamycin.
1. Introduction
The Mycoplasma genus belongs to the Mycoplasmataceae family in the Mollicutes class. It includes a group of slow-growing bacteria species with no cell wall, characterized by a small genome and limited biosynthetic capacity. They are beta-lactam-resistant due to the absence of a cell wall, and their high nutritional demand makes them difficult to culture [1,2], although this problem has recently been solved by applying molecular techniques. They are often found as colonizers of mucosae, mainly respiratory and genitourinary [3,4]. Mycoplasma hominis was the first of various human pathogen species to be isolated. It colonizes the human genital tract and is transmitted by sexual contact [1]. It is not a pathogen in the adult vagina, being present in the vaginal/cervical secretions of around 50% of adult women [5]. However, it has increasingly been attributed to pathogenic action mechanisms at different levels and can be the cause of joint (periprosthetic, septic arthritis) [6], nervous system (drainage-related ventriculitis, neonatal meningitis) [7], respiratory [8], mediastinal [9], genitourinary (pyelonephritis, urethritis, bacterial vaginosis) [10], and even endovascular [11] infection. Reported pathogenicity mechanisms include enhanced adherence to eukaryotic cells (P50, P100), cell dissemination via certain proteins (e.g., Vaa), and local toxicity. Polymerase chain reaction (PCR) is frequently utilized for the microbiological identification of M. hominis, which is commonly susceptible to doxycycline and moxifloxacin [12].
We present the first reported case of M. hominis infective endocarditis (IE) on pacemaker lead and carry out a narrative review of previous reports on IE by this microorganism.
2. Materials and Methods
A search was performed of the PubMed MEDLINE database up to 31 December 2021, including studies written in English or Spanish. Studies of other mycoplasma species and reports on cases without a confirmed diagnosis were excluded. Search terms were: “mycoplasma and endocarditis” (63 results), “mycoplasma and pacemaker” (5 results), “mycoplasma and prosthetic valve infection” (10 results), and “mycoplasma and endovascular infection” (5 results). The final review included 12 eligible studies describing 11 cases. Figure 1 depicts the study selection process.
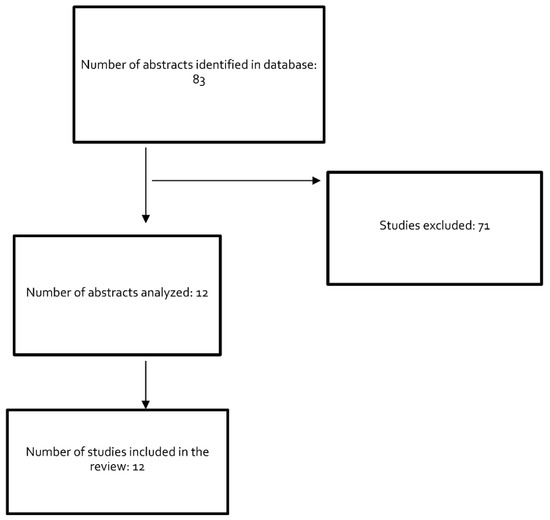
Figure 1.
Flowchart of study selection for the narrative review.
3. Case Report
We report a case of blood culture-negative infective endocarditis (BCNIE) by Mycoplasma hominis in a 67-year-old male farmer with no family history of interest (early cardiovascular disease or sudden death) but a personal history of essential arterial hypertension, hypercholesterolemia, and asymptomatic hyperuricemia. He was under treatment with enalapril (20 mg/day), hydrochlorothiazide (50 mg)/amiloride (5 mg)/day, atorvastatin (80 mg/day), and apixaban (5 mg/12 h). He reported being an ex-smoker (CSI: 60 packs/year) and alcohol consumer (3 SDUs/day) with no known drug allergies.
On 28 April 2021, he was admitted to the cardiology department for sustained monomorphic ventricular tachycardia secondary to structural heart disease (inferolateral scar tissue in the left ventricle with no angiographic findings of significant coronary artery lesions). An implantable cardioverter-defibrillator (ICD) was placed after endocardial ablation, and he was discharged after three days.
On 7 May, 10 days later, he visited the emergency department for oppressive central chest pain radiating to the jaw, accompanied by vegetative symptoms. Physical examination evidenced blood pressure of 74/52 mmHg; heart rate of 104 bpm; respiratory rate of 22 rpm; peripheral oxygen saturation (SpO2) of 92% with inspired oxygen fraction (FiO2) of 21%; absence of fever; poor general state with mucocutaneous pallor; auscultated rhythmic heart sounds without murmurs and preserved vesicular murmur with no added sounds; soft, depressible abdomen non-painful to deep palpation; no megalies or signs of peritonism; preserved hydro-aerial sounds; no lower limb edemas; symmetrical pedal pulses; and no signs of deep venous thrombosis. Electrocardiogram (ECG) results were: sinus rhythm at 90 bpm; axis +30°; narrow QRS; Q wave in D III and aVF; and flattened T wave I, aVL, II, III, aVF, V4 to V6 (with no changes in results obtained at hospital discharge one week earlier). Venous blood gas values were pH 7.43, 23 mmHg pCO2, 15 mmol/L HCO3, and 2.5 mmol/L lactic acid. Blood analysis results are summarized in Table 1. There were no urine sedimentation abnormalities. Anteroposterior chest X-ray showed bilateral pleural effusion, more marked in the left hemithorax. Chest CT scan with intravenous contrast ruled out pulmonary thromboembolism, revealing mild bilateral pleural effusion (more marked on the left), moderate pericardial effusion of normal appearance, no evident thickening or enhancement of pericardial layers suggesting complications, and a slight increase in the density of subcutaneous cell tissue fat at the lower pacemakerpole. The patient was then transferred to the intensive care unit for suspicion of sepsis secondary to infection of the ICD generator pocket. Blood was drawn for culture in the absence of fever, and he was started on empirical treatment with meropenem (2 g/iv/8 h), daptomycin (10 mg/kg/iv/24 h), and clindamycin (600 mg/iv/8 h) alongside noradrenaline (target MAP of 65 mmHg) and rehydration therapy. Diagnostic thoracocentesis yielded 400 cc of serous fluid compatible with exudate according to Light’s criteria [pH 7.481, 9000 red blood cells, 1328 leukocytes with 80% polymorphonuclears, glucose 147 mg/dL, total proteins 3.7 g/dL (plasma 6.6 g/dL), and LDH 472 mg/dL (plasma 373 mg/dL)]. He then underwent cardiovascular surgery for ICD explantation, pericardiocentesis (aspirating 900 cc of exudate-like serohematic fluid), and debridement of the generator pocket. The pericardial fluid contained 580,000 red blood cells, 7400 leukocytes with 60% polymorphonuclears, glucose 0.1 mg/dL (plasma 116 mg/dL), and total proteins 5.6 g/dL (plasma 6.2 g/dL). The fluid was studied in the microbiology laboratory using SeptiFast (LightCycler® SeptiFast Test Mgrade; Roche Diagnostics GmbH, Mannheim, Germany), a PCR test for bacteria and fungi identification, and was found to be positive for Mycoplasma sp. Accordingly, the antibiotic regimen was changed to moxifloxacin (400 mg/24 h) and doxycycline (100 mg/12 h) intravenously for the first week and then orally.

Table 1.
Patient analytics at infection onset and after 12 days of antibiotic treatment.
Four sets of hemocultures were obtained at different times, and SeptiFast identified M. hominis in one of these. Pleural fluid culture was negative. Cultures of pericardial fluid, “pocket” wound exudate, and ICD lead samples all evidenced the growth of M. hominis, which was resistant to erythromycin and clindamycin and susceptible to levofloxacin, ciprofloxacin, tetracycline, and doxycycline. Negative results were obtained for methicillin-resistant S. aureus colonization from the nasal exudate samples and for multi-drug resistant bacteria from the culture of perianal exudate samples. Transthoracic echocardiography study at five days post-pericardiocentesis evidenced no anatomical or functional valvular abnormalities but the persistence of the mild pericardial effusion, while the right cavities showed no sign of hemodynamic compromise.
A new ICD was implanted after the patient achieved hemodynamic stabilization and normalization of acute phase reactants 12 days after the start of antibiotic therapy (Table 1). There were no complications, and the patient had a favorable outcome, being discharged at two weeks post-implantation with no symptoms (NYHA functional class I) and complete resolution of the pericardial effusion. The combined antibiotic treatment was continued for a further two days post-discharge. At 10 months post-discharge, the patient remained asymptomatic with no new ICD-related incidents.
4. Discussion
The narrative review yielded 11 cases of IE by M. hominis. The data summarized in Table 2 show that 72.7% of the patients were male with a mean age of 45 years (range, 4–74 years) and out of the 11 patients, 10 had a history of heart surgery for valve replacement, valve repair, or congenital malformation [13,14,15,16,17,18,19,20,21,22,23], as in the present case.

Table 2.
Cases of endocarditis and M. hominis documented in the literature.
The underlying pathogenic mechanism of endocarditis by M. hominis appears to be nosocomial, possibly secondary to bacteremia caused by the instrumentalization of colonized areas before, during, or after surgery. The median time interval between heart surgery and symptom onset was six months (range, 1–12 months).
The most frequent clinical signs/symptoms in the published cases were fever and valve insufficiency [13,14,15,16,17,18,19,20,21,22,23]. Serial blood cultures were negative in all 11 patients. M. hominis was identified by culture in six cases (in combination with molecular analysis in three of these) and by molecular analysis (16 s ribosomal DNA PCR) of the explanted valve in the remaining five [13,14,15,16,17,18,19,20,21,22,23].
Blood culture plays an essential role in the diagnosis and treatment of IE, and a major diagnostic challenge is therefore posed by blood culture-negative infective endocarditis (BCNIE) [24], reported to represent between 2.5 and 70% of all endocarditis cases in different countries and series [25]. It is essential to consider other causes in patients with IE and negative blood cultures after ruling out an infectious etiology [26]. Besides serology for the detection of fastidious agents such as Coxiella burnetii and Bartonella spp. [26], valve biopsy is the most useful diagnostic approach, especially with the application of histologic and broad-range PCR techniques, when available [27].
Mycoplasma hominis does not Gram stain, it has limited biosynthetic capacity, and it is difficult to isolate in culture medium1. In addition, the characteristic “fried egg” (0.3–0.6 nm) appearance of colonies in culture hampers their ready visualization [25]. Currently, PCR is successfully used to study the extracted valve, offering elevated specificity and sensitivity values [27]. In the present case, the study of blood cultures with real-time multiplex PCR (Septifast) yielded a diagnosis only a few hours after establishing clinical suspicion, allowing the early administration of targeted antibiotic therapy.
After blood samples had been drawn for culture, all patients received empirical antibiotic treatment. Seven (63.6%) were prescribed combined antibiotic treatment with three drugs, most frequently associating a glycopeptide or lipopeptide with beta-lactam and aminoglycoside [13,16,17,18,21,22,23]. The other four received a combination of glycopeptide with aminoglycoside, beta-lactam with aminoglycoside, glycopeptide with beta-lactam, or glycopeptide with lipopeptide [14,16,19,20]. The present patient was treated with a combination of beta-lactam, lipopeptide, and macrolide.
After the detection of M. hominis, nine of the eleven patients (81.8%) [13,14,15,17,18,19,20,21] were prescribed targeted antibiotic treatment, one died during the immediate postoperative period, and the remaining patient was lost to the follow-up [16,22]. Only one of the empirical treatments initially received by the patients was active against M. hominis [16]. The antibiotics most frequently administered in targeted therapy were doxycycline (7/9 cases, 77.8%) [13,15,17,18,19,20,21], quinolones (6/9 cases, 66.7%) [16,17,19,20,21,23], and clindamycin (4/9 cases, 44.4%) [13,16,18,20]. All patients were treated with a combination of antibiotics except for one prescribed monotherapy with doxycycline15.
The duration of treatment ranged between 4 and 10 weeks (median of 8 weeks, mean of 9.5 weeks). Eight patients (72.7%) had a favorable outcome, including two who required heart transplantation [13,23], two patients (18.2%) died [14,16], and one was lost to the follow-up [22].
The intrinsic structural characteristics of M. hominis render it resistant to the antibiotics usually administered against IE, including macrolides, aminoglycosides, beta-lactams, and cotrimoxazole. The treatment of choice is doxycycline (100 mg/12 h), quinolones (moxifloxacin [400 mg/day], levofloxacin [500 mg/day]), or clindamycin (300 mg/8 h) [12]. The optimal antibiotic regimen for endocarditis by M. hominis has not been established, although most authors recommend a single antibiotic therapy cycle of 6–8 weeks alongside valve replacement [13,14,15,16,17,18,19,20,21,22,23]. One patient who received doxycycline for 8 weeks required heart transplantation for acute heart failure due to recurrent perivalvular insufficiency [13].
In conclusion, M. hominis is rarely the cause of IE but should be contemplated in cases of BCNIE, especially in individuals with a recent history of cardiac surgery or device implantation. Although the clinical manifestations are non-specific, this entity should be considered in such patients in the presence of fever, clinical bacteremia, and/or signs/symptoms of heart failure not previously experienced. Clinical suspicion is also strengthened by the presence of elevated acute phase reactants (C-reactive protein, procalcitonin, leukocytosis) early after cardiac surgery with no evident focus of infection, especially when blood cultures are negative [19].
We highlight the diagnostic usefulness of molecular techniques, initially applied in blood samples, allowing the identification and recovery of a proportion of cases with negative blood cultures. Nevertheless, the definitive diagnosis is usually obtained through the identification of M. hominis in samples of valves or endovascular prosthetic material [18].
A more rapid diagnosis permits the earlier administration of targeted treatment, with doxycycline and quinolones as the drugs of choice. This has important prognostic implications, given the intrinsic resistance of M. hominis to the antibiotics habitually prescribed against IE produced by staphylococci and streptococci [14].
Author Contributions
Conceptualization, A.B.-M. and A.R.-C.; methodology, A.R.-C.; software, A.B.-M.; validation, A.B.-M., A.R.-C. and C.H.-T.; formal analysis, F.C.; investigation, C.H.-T.; resources, F.C.; data curation, C.H.-T.; writing—original draft preparation, A.B.-M.; writing—review and editing, A.R.-C.; visualization, F.C.; supervision, C.H.-T.; project administration, C.H.-T.; funding acquisition, C.H.-T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available in the main text.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ahmed, J.; Rawre, J.; Dhawan, N.; Khanna, N.; Dhawan, B. Mycoplasma hominis: An under recognized pathogen. Indian J. Med. Microbiol. 2021, 39, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Gonzalo de Liria, C.R.; Méndez Hernández, M. Infecciones causadas por Mycoplasma pneumoniae. An. Pediatr. Contin. 2013, 11, 23–29. [Google Scholar] [CrossRef]
- Gómez Rufo, D.; García Sánchez, E.; García Sánchez, J.E.; García Moro, M. Implicaciones clínicas de las especies del género Mycoplasma [Clinical implications of the genus Mycoplasma]. Rev. Esp. Quimioter. 2021, 34, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Farhadifar, F.; Khodabandehloo, M.; Ramazanzadeh, R.; Rouhi, S.; Ahmadi, A.; Ghaderi, E. Survey on association between Mycoplasma hominis endocervical infection and spontaneous abortion using Polymerase Chain Reaction. Int. J. Reprod. Biomed. 2016, 14, 181–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor-Robinson, D. Mollicutes in vaginal microbiology: Mycoplasma hominis, Ureaplasma urealyticum, Ureaplasma parvum and Mycoplasma genitalium. Res. Microbiol. 2017, 168, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Lu, B. Infection due to Mycoplasma hominis after left hip replacement: Case report and literature review. BMC Infect. Dis. 2019, 19, 50. [Google Scholar] [CrossRef] [PubMed]
- Hata, A.; Honda, Y.; Asada, K.; Sasaki, Y.; Kenri, T.; Hata, D. Mycoplasma hominis meningitis in a neonate: Case report and review. J. Infect. 2008, 57, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Sáez, A.; Monteagudo, I.; Miñambres, E.; Ots, E.; Rodríguez-Borregán, J.C.; García, C. Pneumonia due to Mycoplasma hominis in a healthy adult. Scand. J. Infect. Dis. 2003, 35, 282–284. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, H.; Shimizu, H.; Katayama, K.; Tadera, K.; Nomura, T.; Omori, K.; Shigemoto, N.; Takasaki, T.; Ohge, H.; Takahashi, S. Postoperative mediastinitis after cardiac surgery caused by Mycoplasma hominis: A case report. Surg. Case Rep. 2021, 7, 248. [Google Scholar] [CrossRef] [PubMed]
- Küchle, C.; Abele-Horn, M.; Menninger, M.; Held, E.; Heesemann, J. Mycoplasma hominis. A rare causative agent of acute pyelonephritis. Dtsch. Med. Wochenschr. 1997, 122, 542–544. [Google Scholar] [CrossRef]
- Cohen, J.I.; Sloss, L.J.; Kundsin, R.; Golightly, L. Prosthetic valve endocarditis caused by Mycoplasma hominis. Am. J. Med. 1989, 86, 819–821. [Google Scholar] [CrossRef]
- Krausse, R.; Schubert, S. In-vitro activities of tetracyclines, macrolides, fluoroquinolones and clindamycin against Mycoplasma hominis and Ureaplasma ssp. isolated in Germany over 20 years. Clin. Microbiol. Infect. 2010, 16, 1649–1655. [Google Scholar] [CrossRef] [PubMed]
- DiSesa, V.J.; Sloss, L.J.; Cohn, L.H. Heart transplantation for intractable prosthetic valve endocarditis. J. Heart Transplant. 1990, 9, 142–143. [Google Scholar] [PubMed]
- Blasco, M.; Torres, L.; Marco, M.L.; Moles, B.; Villuendas, M.C.; García Moya, J.B. Prosthetic valve endocarditis caused by Mycoplasma hominis. Eur. J. Clin. Microbiol. Infect. Dis. 2000, 19, 638–640. [Google Scholar] [CrossRef]
- Fenollar, F.; Gauduchon, V.; Casalta, J.P.; Lepidi, H.; Vandenesch, F.; Raoult, D. Mycoplasma endocarditis: Two case reports and a review. Clin. Infect. Dis. 2004, 38, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, S.R.; Littlehorn, C.; Nyquist, A.C. Mycoplasma hominis endocarditis in a child with a complex congenital heart defect. Pediatr. Infect. Dis. J. 2006, 25, 851–852. [Google Scholar] [CrossRef]
- Hidalgo-Tenorio, C.; Pasquau, J.; López-Checa, S.; López-Ruz, M.A. Endocarditis por Mycoplasma hominis [Endocarditis due to Mycoplasma hominis]. Enferm. Infecc. Microbiol. Clin. 2006, 24, 470–471. [Google Scholar] [CrossRef]
- Jamil, H.A.; Sandoe, J.A.T.; Gascoyne-Binzi, D.; Chalker, V.J.; Simms, A.D.; Munsch, C.M.; Baig, M.W. Late-onset prosthetic valve endocarditis caused by Mycoplasma hominis, diagnosed using broad-range bacterial PCR. J. Med. Microbiol. 2012, 61, 300–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hussain, S.T.; Gordon, S.M.; Tan, C.D.; Smedira, N.G. Mycoplasma hominis prosthetic valve endocarditis: The value of molecular sequencing in cardiac surgery. J. Thorac. Cardiovasc. Surg. 2013, 146, 7–9. [Google Scholar] [CrossRef] [Green Version]
- Gagneux-Brunon, A.; Grattard, F.; Morel, J.; Suy, F.; Fuzellier, J.-F.; Verhoeven, P.; Cazorla, C.; Guglielminotti, C.; Fresard, A.; Lucht, F.; et al. Mycoplasma hominis, a Rare but True Cause of Infective Endocarditis. J. Clin. Microbiol. 2015, 53, 3068–3071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romeu Prieto, J.M.; Lizcano Lizcano, A.M.; López de Toro Martín Consuegra, I.; Largo Pau, J.; López Almodóvar, L.F.; García Camacho, E. Culture-negative Endocarditis: Mycoplasma hominis Infection. Rev. Esp. Cardiol. (Engl. Ed.) 2015, 68, 1037–1038. [Google Scholar] [CrossRef] [PubMed]
- Kotaskova, I.; Nemec, P.; Vanerkova, M.; Malisova, B.; Tejkalova, R.; Orban, M. First report of Sneathia sanguinegens together with Mycoplasma hominis in postpartum prosthetic valve infective endocarditis: A case report. BMC Infect. Dis. 2017, 17, 563. [Google Scholar] [CrossRef] [PubMed]
- Givone, F.; Peghin, M.; Vendramin, I.; Carletti, S.; Tursi, V.; Pasciuta, R.; Livi, U.; Bassetti, M. Salvage heart transplantation for Mycoplasma hominis prosthetic valve endocarditis: A case report and review of the literature. Transpl. Infect. Dis. 2020, 22, e13249. [Google Scholar] [CrossRef] [PubMed]
- Brouqui, P.; Raoult, D. Endocarditis due to rare and fastidious bacteria. Clin. Microbiol. Rev. 2001, 14, 177–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fournier, P.E.; Thuny, F.; Richet, H.; Lepidi, H.; Casalta, J.P.; Arzouni, J.P.; Maurin, M.; Celard, M.; Mainardi, J.L.; Caus, T.; et al. Comprehensive diagnostic strategy for blood culture-negative endocarditis: A prospective study of 819 new cases. Clin. Infect. Dis. 2010, 51, 131–140. [Google Scholar] [CrossRef] [Green Version]
- Hurrell, H.; Roberts-Thomson, R.; Prendergast, B.D. Non-infective endocarditis. Heart 2020, 106, 1023–1029. [Google Scholar] [CrossRef]
- Marín, M.; Muñoz, P.; Sánchez, M.; Del Rosal, M.; Alcalá, L.; Rodríguez-Créixems, M.; Bouza, E.; on behalf of the Group for the Management of Infective Endocarditis of the Gregorio Marañón Hospital. Molecular diagnosis of infective endocarditis by real- time broad-range polymerase chain reaction (PCR) and sequencing directly from heart valve tissue. Medicine 2007, 86, 195–202. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).