Poliovirus-Neutralizing Antibody Seroprevalence and Vaccine Habits in a Vaccine-Derived Poliovirus Outbreak Region in the Democratic Republic of Congo in 2018: The Impact on the Global Eradication Initiative
Abstract
1. Background
2. Methods
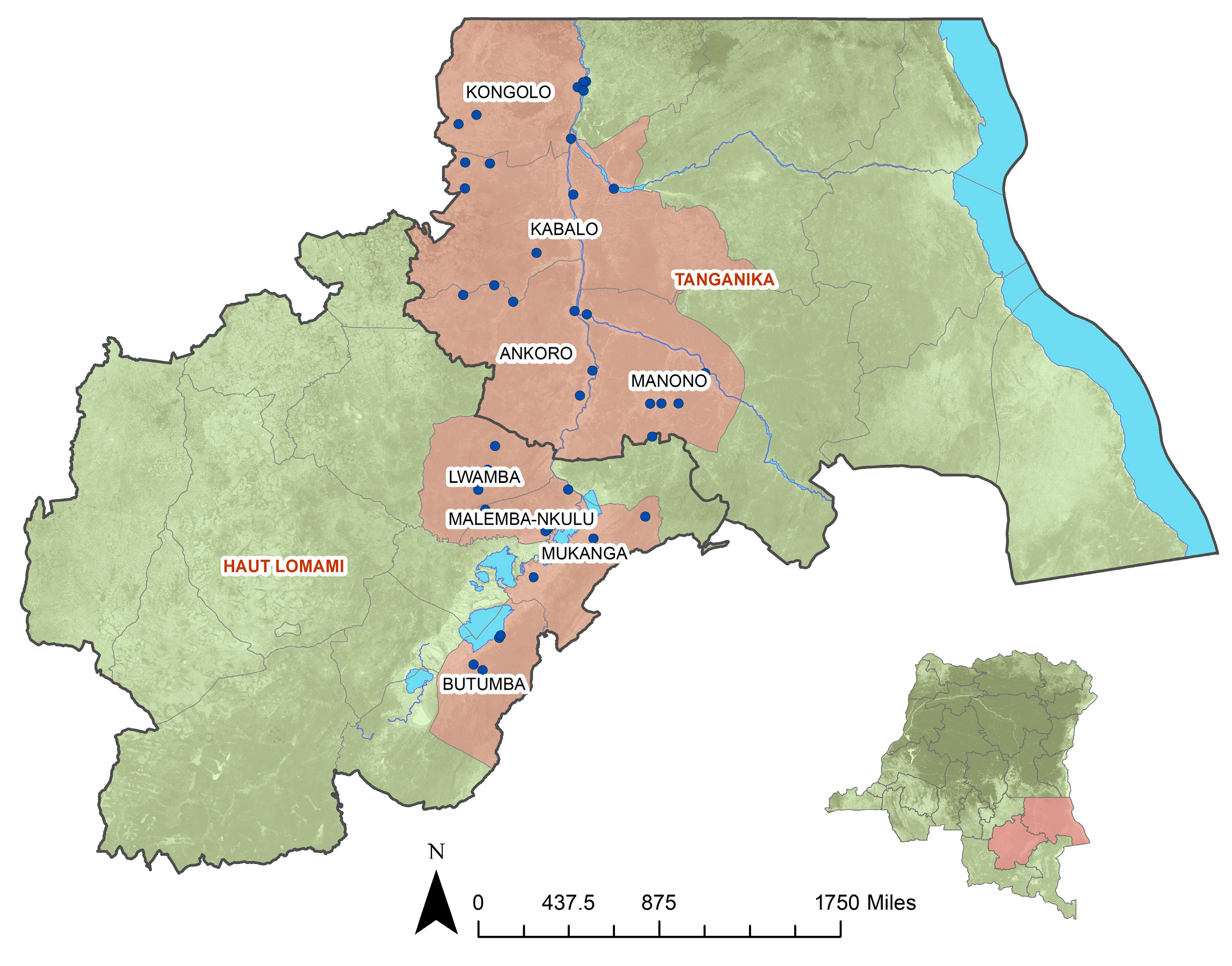
2.1. Study Sample
2.2. Laboratory Analysis
2.3. Statistical Analysis
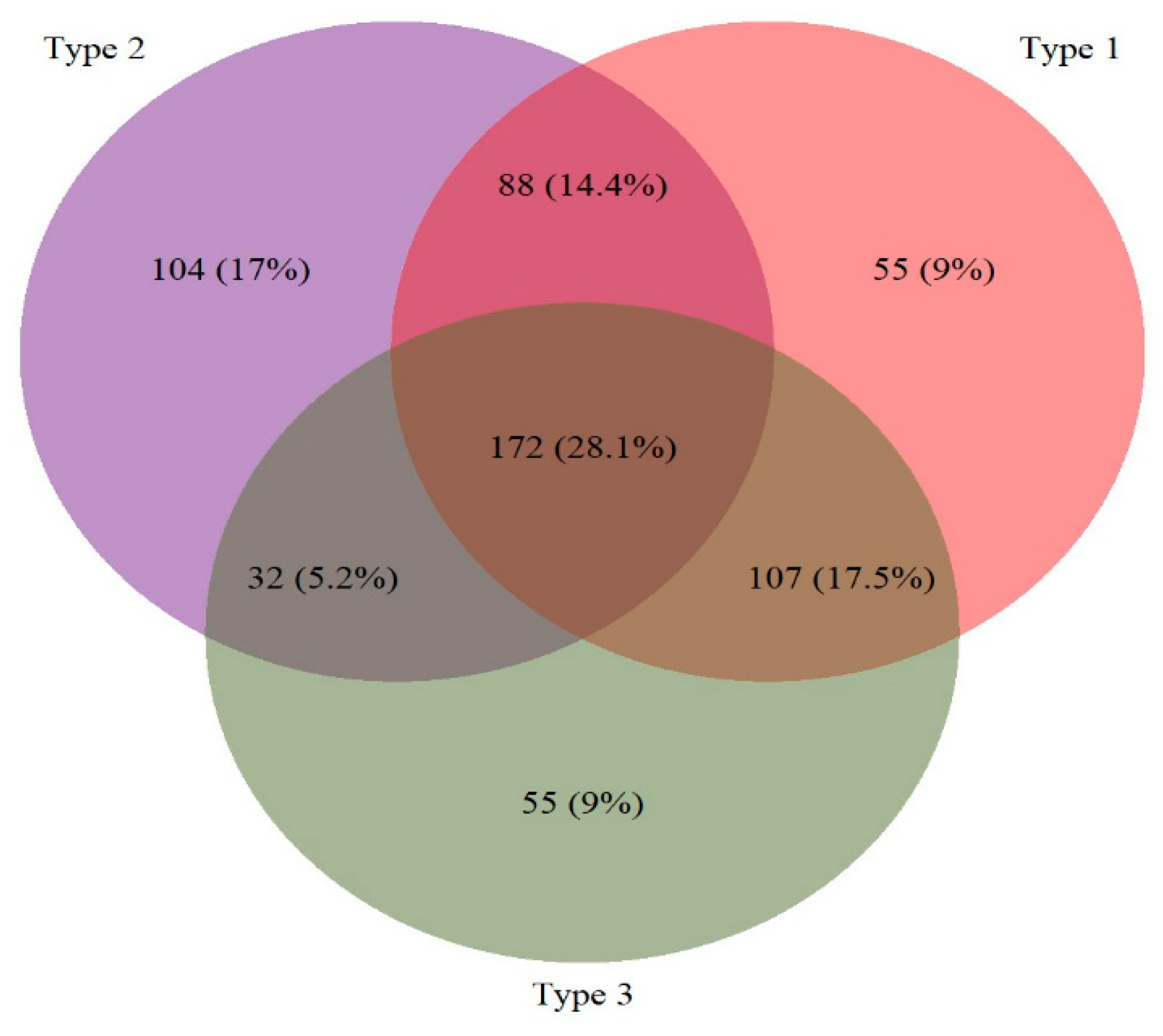
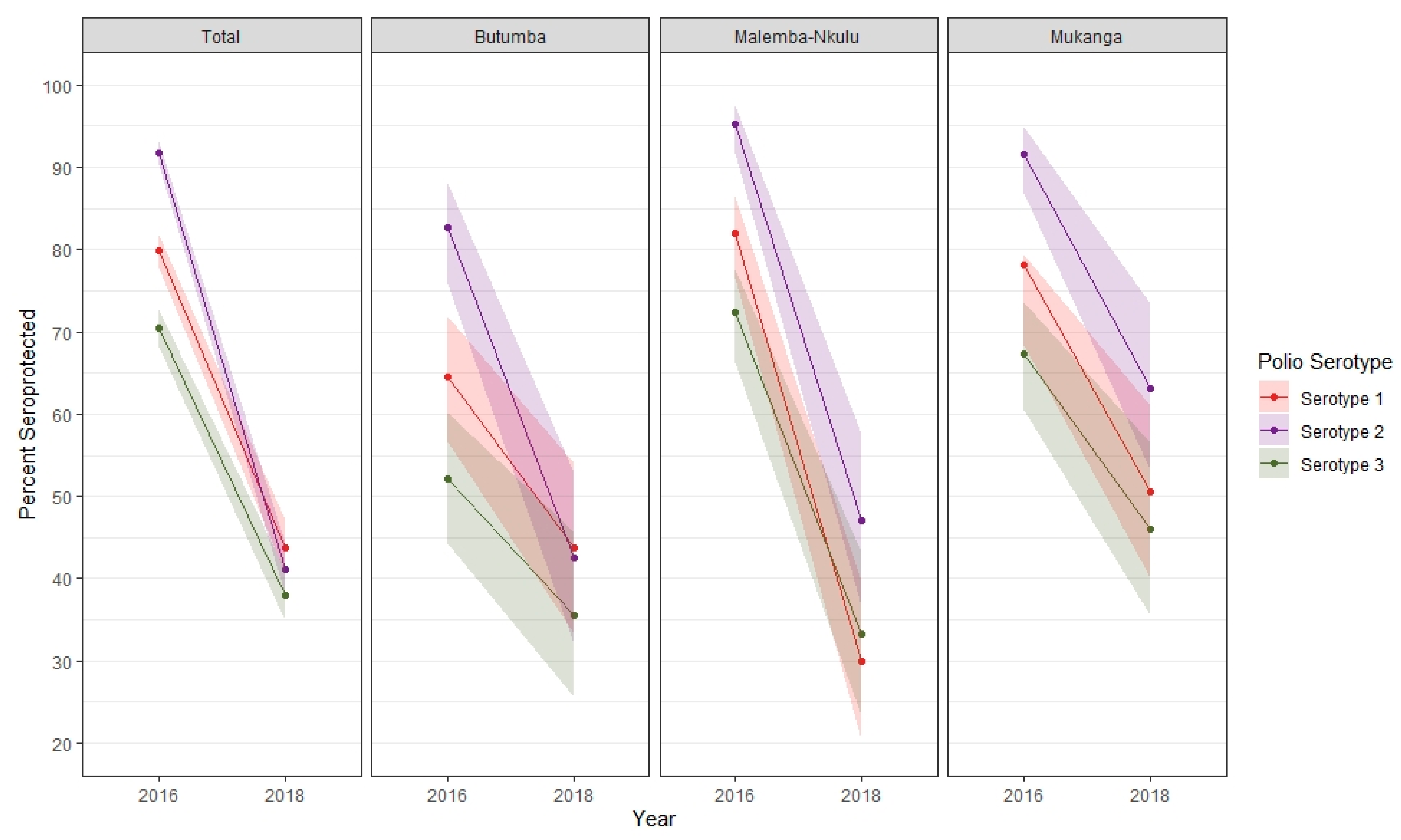
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kabir, M.; Afzal, M.S. Epidemiology of polio virus infection in Pakistan and possible risk factors for its transmission. Asian Pac. J. Trop. Med. 2016, 9, 1044–1047. [Google Scholar] [CrossRef]
- World Health Organization. Two Out of Three Wild Poliovirus Strains Eradicated; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- CDC. Wild Poliovirus Type 1 and Type 3 Importations—15 countries, Africa, 2008–2009. MMWR Morb. Mortal. Wkly. Rep. 2009, 58, 357–362. [Google Scholar]
- Gumede, N.; Jorba, J.; Deshpande, J.; Pallansch, M.; Yogolelo, R.; Muyembe-Tamfum, J.J.; Kew, O.; Venter, M.; Burns, C.C. Phylogeny of imported and reestablished wild polioviruses in the Democratic Republic of the Congo from 2006 to 2011. J. Infect. Dis. 2014, 210 (Suppl. 1), S361–S367. [Google Scholar] [CrossRef] [PubMed]
- Burns, C.C.; Diop, O.M.; Sutter, R.W.; Kew, O.M. Vaccine-Derived Polioviruses. J. Infect. Dis. 2014, 210, S283–S293. [Google Scholar] [CrossRef]
- Voorman, A.; Hoff, N.A.; Doshi, R.H.; Alfonso, V.; Mukadi, P.; Muyembe-Tamfum, J.-J.; Wemakoy, E.O.; Bwaka, A.; Weldon, W.; Gerber, S.; et al. Polio immunity and the impact of mass immunization campaigns in the Democratic Republic of the Congo. Vaccine 2017, 35, 5693–5699. [Google Scholar] [CrossRef] [PubMed]
- Alleman, M.M.; Chitale, R.; Burns, C.C.; Iber, J.; Dybdahl-Sissoko, N.; Chen, Q.; Andriamihantanirina, R. Vaccine-Derived Poliovirus Outbreaks and Events—Three Provinces, Democratic Republic of the Congo, 2017. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Bigouette, J.P.; Henderson, E.; Traoré, M.A.; Wassilak, S.G.; Jorba, J.; Mahoney, F.; Bolu, O.; Diop, O.M.; Burns, C.C. Update on vaccine-derived poliovirus outbreaks—Worldwide, January 2021–December 2022. Morb. Mortal. Wkly. Rep. 2023, 72, 366. [Google Scholar] [CrossRef]
- Jorba, J.; Diop, O.; Iber, J.; Henderson, E.; Zhao, K.; Quddus, A.; Sutter, R.; Vertefeuille, J.F.; Wenger, J.; Wassilak, S.G.F.; et al. Update on Vaccine-Derived Poliovirus Outbreaks—Worldwide, January 2018–June 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 1024–1028. [Google Scholar] [CrossRef] [PubMed]
- Fine, P.E.M.; Carneiro, I.A.M. Transmissibility and Persistence of Oral Polio Vaccine Viruses: Implications for the Global Poliomyelitis Eradication Initiative. Am. J. Epidemiol. 1999, 150, 1001–1021. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, K.M.; Lamoureux, C.; Molodecky, N.A.; Lyons, H.; Grassly, N.C.; Tallis, G. An assessment of the geographical risks of wild and vaccine-derived poliomyelitis outbreaks in Africa and Asia. BMC Infect. Dis. 2017, 17, 367. [Google Scholar] [CrossRef] [PubMed]
- Mbaeyi, C.; Alleman, M.M.; Ehrhardt, D.; Wiesen, E.; Burns, C.C.; Liu, H.; Ewetola, R.; Seakamela, L.; Mdodo, R.; Ndoutabe, M.; et al. Update on Vaccine-Derived Poliovirus Outbreaks—Democratic Republic of the Congo and Horn of Africa, 2017–2018. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 225–230. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Poliomyelitis—Democratic Republic of the Congo; Disease Outbreak, News; Emergencies Preparedness, Response; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Halbrook, M.; Alfonso, V.H.; Hoff, N.A.; Voorman, A.; Mukadi, P.; Weldon, W.C.; Gadoth, A.; Sinai, C.; Musene, K.; Spender, D. Vaccination and Serologic Response Against Poliovirus Among Children in an Outbreak Area of Vaccine Derived Poliovirus in the Democratic Republic of the Congo; University of California, Los Angeles (UCLA): Los Angeles, CA, USA, 2020. [Google Scholar]
- Weldon, W.C.; Oberste, M.S.; Pallansch, M.A. Standardized Methods for Detection of Poliovirus Antibodies. Methods Mol. Biol. 2016, 1387, 145–176. [Google Scholar] [PubMed]
- Reinheimer, C.; Friedrichs, I.; Rabenau, H.F.; Doerr, H.W. Deficiency of immunity to poliovirus type 3: A lurking danger? BMC Infect. Dis. 2012, 12, 24. [Google Scholar] [CrossRef] [PubMed]
- Nathanson, N.; Kew, O.M. From Emergence to Eradication: The Epidemiology of Poliomyelitis Deconstructed. Am. J. Epidemiol. 2010, 172, 1213–1229. [Google Scholar] [CrossRef] [PubMed]
- Miles, M.; Ryman, T.K.; Dietz, V.; Zell, E.; Luman, E.T. Validity of vaccination cards and parental recall to estimate vaccination coverage: A systematic review of the literature. Vaccine 2013, 31, 1560–1568. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO position paper on Haemophilus influenzae type b conjugate vaccines. (Replaces WHO position paper on Hib vaccines previously published in the Weekly Epidemiological Record. Wkly. Epidemiol. Rec. 2023, 98, 346–348. [Google Scholar]

| n | % | |
|---|---|---|
| Do you know what polio is? | 585 | 60.68 |
| Do you know how polio is spread? | 247 | 25.62 |
| If your child was to get sick with polio, what symptoms could they get? | ||
| Paralysis | 717 | 74.38 |
| Fever | 370 | 38.38 |
| Diarrhea | 314 | 32.57 |
| Don’t know | 141 | 14.63 |
| What would you do if your child suddenly was unable to walk? | ||
| Take them to a local health care practitioner | 452 | 46.89 |
| Take them to a doctor | 245 | 25.41 |
| Take them to a hospital | 133 | 13.8 |
| I don’t know | 61 | 6.33 |
| Do nothing or wait | 41 | 4.25 |
| Treat at home with over-the-counter medicines | 29 | 3.01 |
| Has someone ever come to your home to give you information about polio? | 765 | 79.36 |
| In the last year have you heard about any polio vaccine campaigns? | 853 | 88.49 |
| In what ways have received campaign information about polio? | ||
| Community/Village health volunteer | 182 | 18.88 |
| TV commercial | 890 | 92.32 |
| Radio Commercial | 948 | 98.34 |
| Poster | 247 | 25.62 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Halbrook, M.; Gadoth, A.; Mukadi, P.; Hoff, N.A.; Musene, K.; Dzogang, C.; Sinai, C.S.; Spencer, D.; Ngoie-Mwamba, G.; Tangney, S.; et al. Poliovirus-Neutralizing Antibody Seroprevalence and Vaccine Habits in a Vaccine-Derived Poliovirus Outbreak Region in the Democratic Republic of Congo in 2018: The Impact on the Global Eradication Initiative. Vaccines 2024, 12, 246. https://doi.org/10.3390/vaccines12030246
Halbrook M, Gadoth A, Mukadi P, Hoff NA, Musene K, Dzogang C, Sinai CS, Spencer D, Ngoie-Mwamba G, Tangney S, et al. Poliovirus-Neutralizing Antibody Seroprevalence and Vaccine Habits in a Vaccine-Derived Poliovirus Outbreak Region in the Democratic Republic of Congo in 2018: The Impact on the Global Eradication Initiative. Vaccines. 2024; 12(3):246. https://doi.org/10.3390/vaccines12030246
Chicago/Turabian StyleHalbrook, Megan, Adva Gadoth, Patrick Mukadi, Nicole A. Hoff, Kamy Musene, Camille Dzogang, Cyrus Shannon Sinai, D’Andre Spencer, Guillaume Ngoie-Mwamba, Sylvia Tangney, and et al. 2024. "Poliovirus-Neutralizing Antibody Seroprevalence and Vaccine Habits in a Vaccine-Derived Poliovirus Outbreak Region in the Democratic Republic of Congo in 2018: The Impact on the Global Eradication Initiative" Vaccines 12, no. 3: 246. https://doi.org/10.3390/vaccines12030246
APA StyleHalbrook, M., Gadoth, A., Mukadi, P., Hoff, N. A., Musene, K., Dzogang, C., Sinai, C. S., Spencer, D., Ngoie-Mwamba, G., Tangney, S., Salet, F., Nyembwe, M., Kambamba Nzaji, M., Tambu, M., Mbala, P., Fuller, T., Gerber, S. K., Kaba, D., Muyembe-Tamfum, J. J., & Rimoin, A. W. (2024). Poliovirus-Neutralizing Antibody Seroprevalence and Vaccine Habits in a Vaccine-Derived Poliovirus Outbreak Region in the Democratic Republic of Congo in 2018: The Impact on the Global Eradication Initiative. Vaccines, 12(3), 246. https://doi.org/10.3390/vaccines12030246






