The Apoplastic and Symplastic Antioxidant System in Onion: Response to Long-Term Salt Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Treatments and Experimental Design
2.3. Sample Selection
2.4. Histochemical ROS Detection
2.5. Oxidative Stress Parameters
2.5.1. Electrolyte Leakage Measurement
2.5.2. Lipid Peroxidation
2.6. Apoplast Extraction
2.7. Leaf Enzyme Extraction and Enzymatic Analyses
2.8. Ascorbate and Glutathione Measurements
2.9. Statistical Analysis
3. Results
3.1. Effect of Salt Stress on Oxidative Stress Parameters
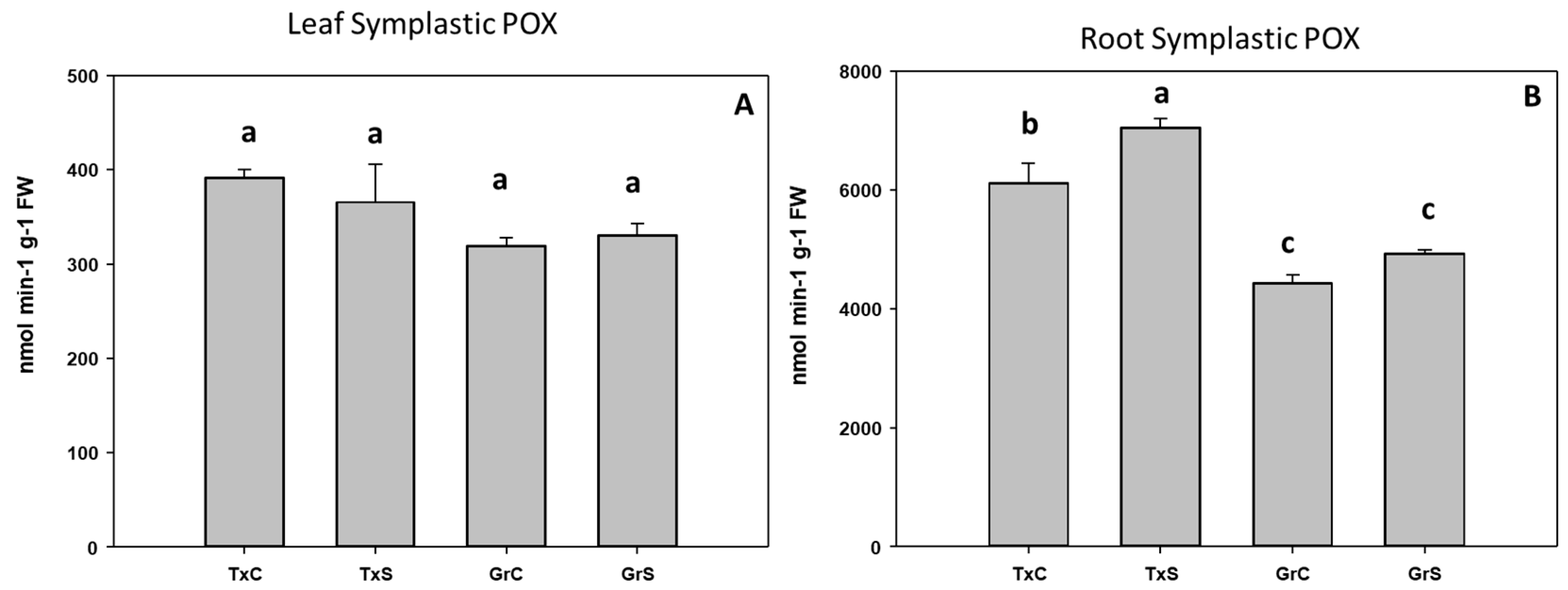
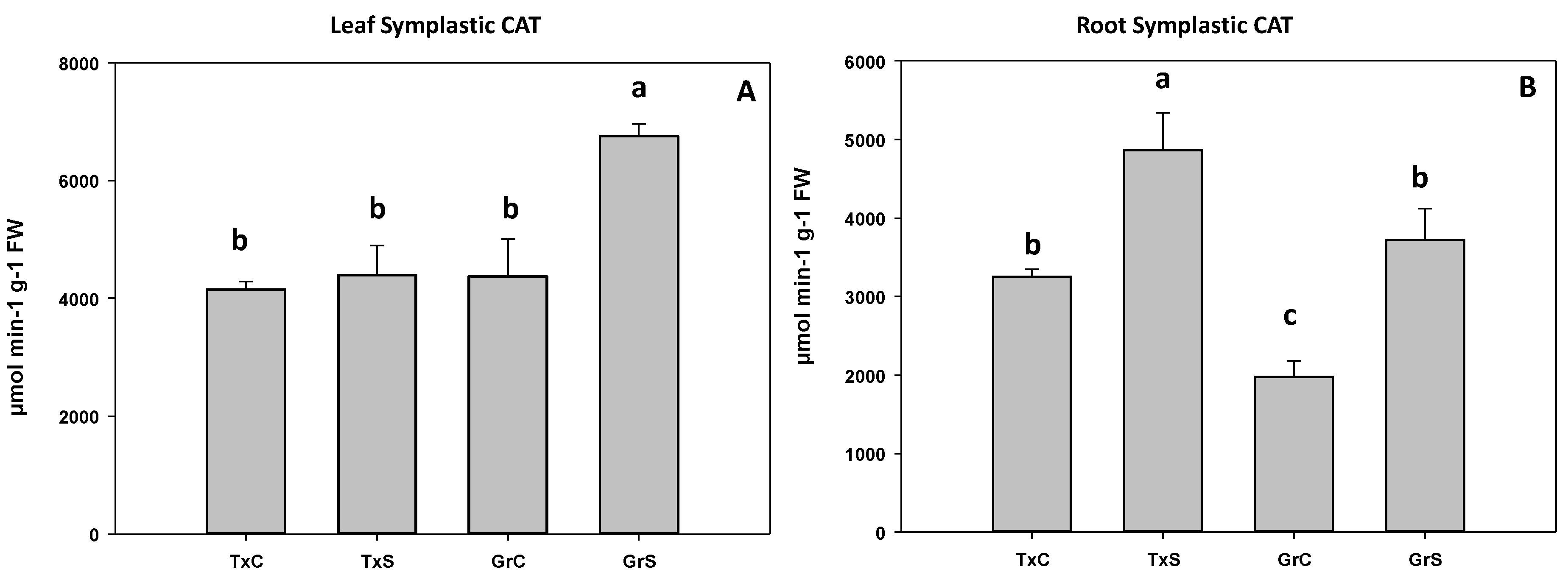
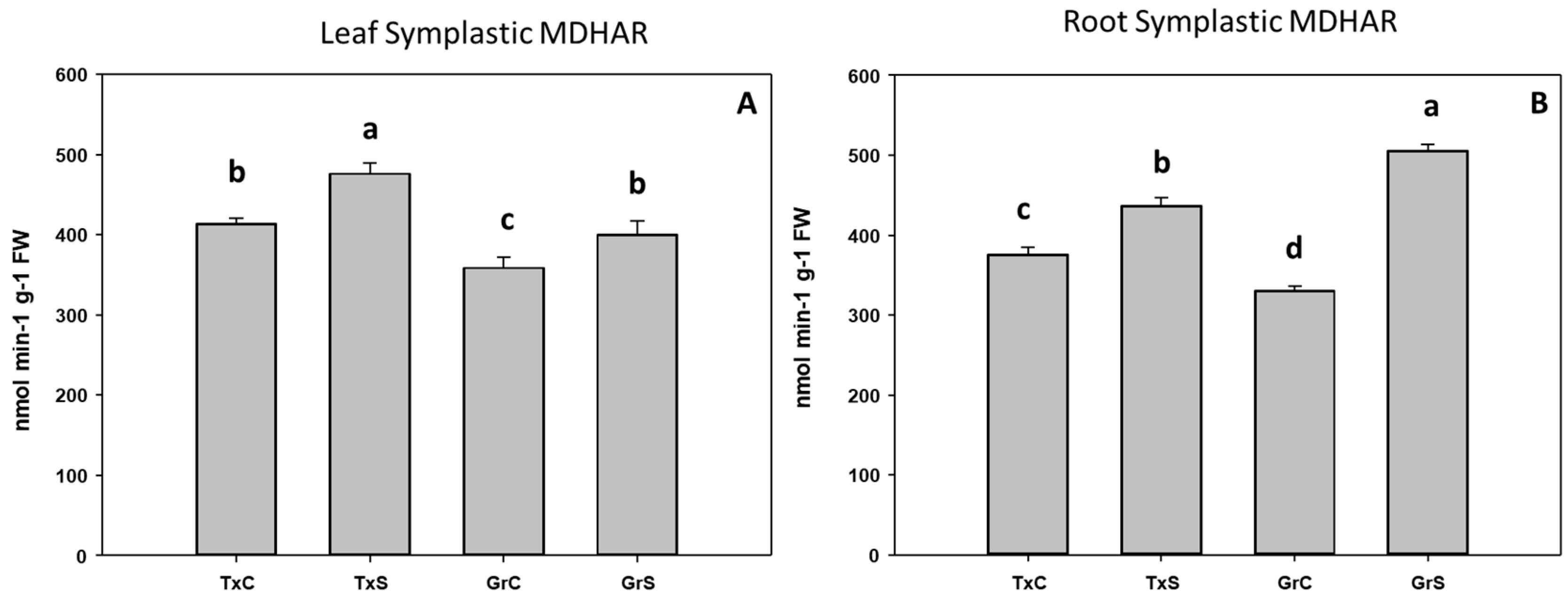
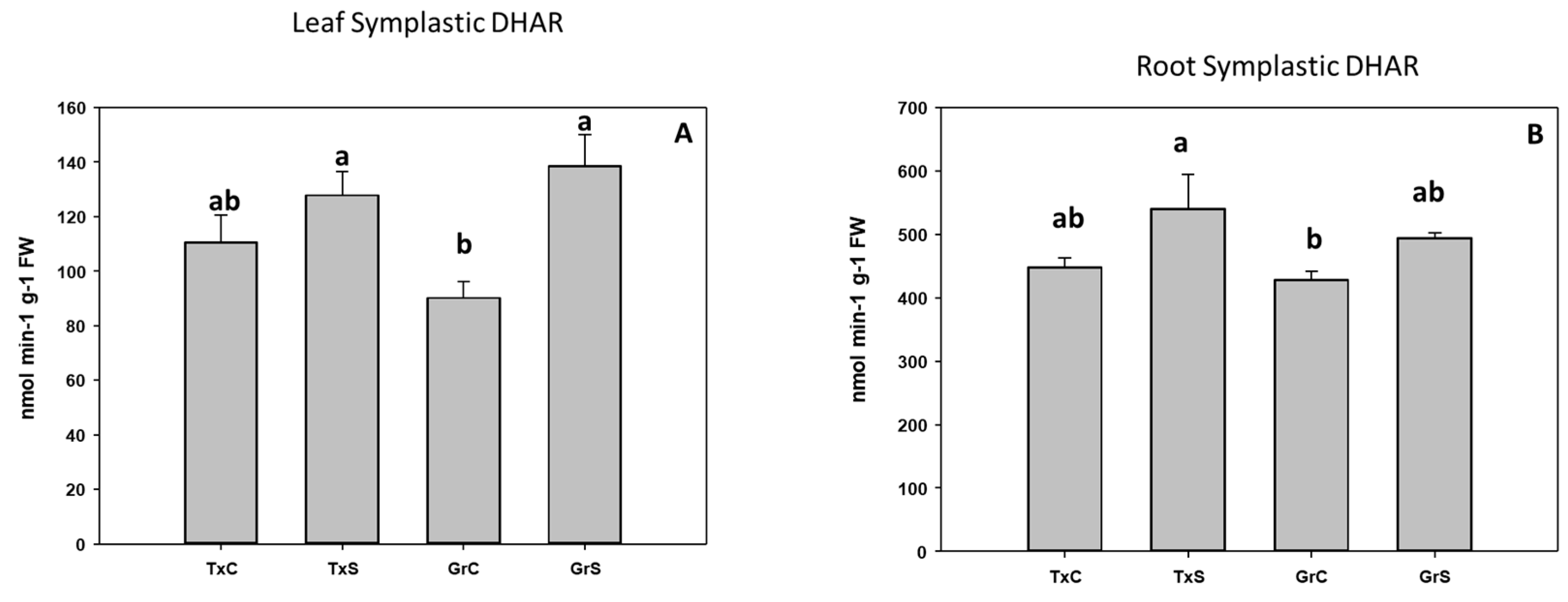
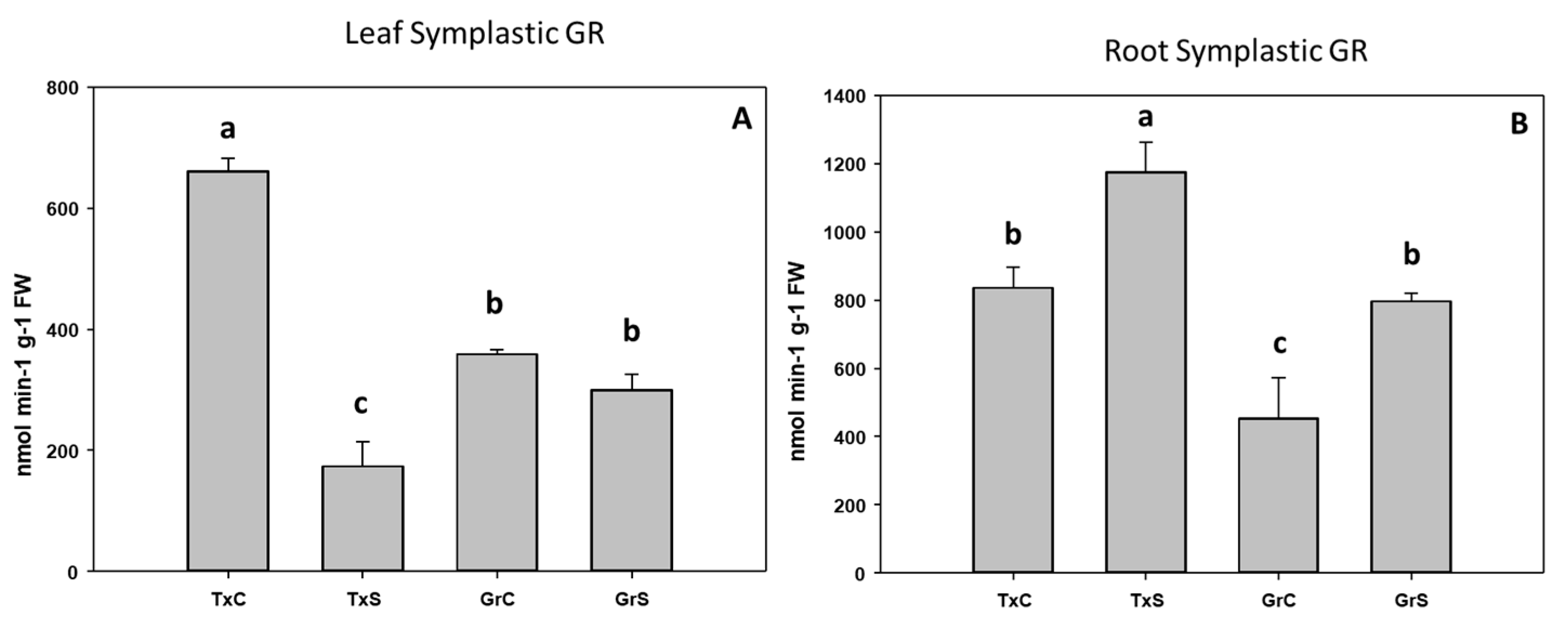
3.2. Antioxidant Enzymatic Activities
3.3. Non-Enzymatic Antioxidants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ashraf, M.; Harris, J.C. Potential biochemical indicators of salinity tolerance in plants. Plant Sci. 2004, 166, 3–16. [Google Scholar] [CrossRef]
- Sairam, R.K.; Tyagi, A. Physiology and molecular biology of salinity stress tolerance in plants. Curr. Sci. 2004, 86, 407–427. [Google Scholar]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef]
- Acosta-Motos, J.R.; Ortuño, M.F.; Bernal-Vicente, A.; Diaz-Vivancos, P.; Sanchez-Blanco, M.J.; Hernandez, J.A. Plant responses to salt stress: Adaptive mechanisms. Agronomy 2017, 7, 18. [Google Scholar] [CrossRef]
- AbdElgawad, H.; Zinta, G.; Hegab, M.M.; Pandey, R.; Asard, H.; Abuelsoud, W. High salinity induces different oxidative stress and antioxidant responses in maize seedlings organs. Front. Plant Sci. 2016, 7, 276. [Google Scholar] [CrossRef]
- Kumar, M.; Kumar, R.; Jain, V.; Jain, S. Differential behavior of the antioxidant system in response to salinity induced oxidative stress in salt-tolerant and salt-sensitive cultivars of Brassica juncea L. Biocatal. Agric. Biotechnol. 2018, 13, 12–19. [Google Scholar] [CrossRef]
- Khan, M.H.; Panda, S.K. Alterations in root lipid peroxidation and antioxidative responses in two rice cultivars under NaCl-salinity stress. Acta Physiol. Plant. 2008, 30, 81. [Google Scholar] [CrossRef]
- Choudhury, F.K.; Rivero, R.M.; Blumwald, E.; Mittler, R. Reactive oxygen species, abiotic stress and stress combination. Plant J. 2017, 90, 856–867. [Google Scholar] [CrossRef]
- El-baky, A.; Hanaa, H.; Hussein, M.M.; Amal, M.A. Influence of Salinity on Lipid Peroxidation, Antioxidant Enzymes and Electrophoretic Patterns of Protein and Isoenzymes in Leaves of Some Onion Cultivars. Asian J. Plant Sci. 2003, 2, 633–638. [Google Scholar]
- Hernandez, J.A.; Ferrer, M.A.; Jimenez, A.; Barcelo, A.R.; Sevilla, F. Antioxidant systems and O2•−/H2O2 production in the apoplast of pea leaves. Its relation with salt-induced necrotic lesions in minor veins. Plant Physiol. 2001, 127, 817–831. [Google Scholar] [CrossRef]
- Farooq, M.; Hussain, M.; Wakeel, A.; Siddique, K.H.M. Salt stress in maize: Effects, resistance mechanisms, and management. A review. Agron. Sustain. Dev. 2015, 35, 461–481. [Google Scholar] [CrossRef]
- Noctor, G.; Foyer, C.H. Ascorbate and Glutathione: Keeping Active Oxygen Under Control. Annu. Rev. Plant Biol. 1998, 49, 249–279. [Google Scholar] [CrossRef]
- Bartels, D.; Sunkar, R. Drought and salt tolerance in plants. CRC. Crit. Rev. Plant Sci. 2005, 24, 23–58. [Google Scholar] [CrossRef]
- Das, K.; Roychoudhury, A. Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 53. [Google Scholar] [CrossRef]
- Kibria, M.G.; Hossain, M.; Murata, Y.; Hoque, M.A. Antioxidant Defense Mechanisms of Salinity Tolerance in Rice Genotypes. Rice Sci. 2017, 24, 155–162. [Google Scholar] [CrossRef]
- Guerra-Guimarães, L.; Pinheiro, C.; Chaves, I.; Barros, D.R.; Ricardo, C.P. Protein dynamics in the plant extracellular space. Proteomes 2016, 4, 22. [Google Scholar] [CrossRef]
- Diaz-Vivancos, P.; Rubio, M.; Mesonero, V.; Periago, P.M.; Barcelo, A.R.; Martinez-Gomez, P.; Hernandez, J.A. The apoplastic antioxidant system in Prunus: Response to long-term plum pox virus infection. J. Exp. Bot. 2006, 57, 3813–3824. [Google Scholar] [CrossRef]
- Podgórska, A.; Burian, M.; Szal, B. Extra-cellular but extra-ordinarily important for cells: Apoplastic reactive oxygen species metabolism. Front. Plant Sci. 2017, 8, 1353. [Google Scholar] [CrossRef]
- Villafañe, R.; Azpúrua, M.; Ruiz, T.; Dugarte, J.; Abarca, O. Distribución espacial de la salinidad en los suelos de Qíbor y su relación con las limitaciones de drenaje y la calidad del agua. Bioagro 1999, 11, 43–50. [Google Scholar]
- Torres, D.; Álvarez, J.; Contreras, J.; Henríquez, M.; Hernández, W.; Lorbes, J.; Mogollón, J.P. Identificación De potencialidades y limitaciones de suelos agrícolas Del Estado Lara, Venezuela. Bioagro 2017, 29, 207–218. [Google Scholar]
- García, G.; García, M.; Ramírez, H. Comportamiento de siete cultivares de Allium cepa L. ante diferentes niveles de estrés salino. Bioagro 2015, 27, 93–102. [Google Scholar]
- Maia, J.M.; de Macedo, C.E.C.; Voigt, E.L.; Freitas, J.B.S.; Silveira, J.A.G. Antioxidative enzymatic protection in leaves of two contrasting cowpea cultivars under salinity. Biol. Plant. 2010, 54, 159–163. [Google Scholar] [CrossRef]
- Hernández, J.A.; Almansa, M.S. Short-term effects of salt stress on antioxidant systems and leaf water relations of pea leaves. Physiol. Plant. 2002, 115, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Barba-Espin, G.; Clemente-Moreno, M.J.; Alvarez, S.; Garcia-Legaz, M.F.; Hernandez, J.A.; Diaz-Vivancos, P. Salicylic acid negatively affects the response to salt stress in pea plants. Plant Biol. 2011, 13, 909–917. [Google Scholar] [CrossRef] [PubMed]
- Weisiger, R.A.; Fridovich, I. Mitochondrial superoxide dismutase. Site of synthesis and intramitochondrial localization. J. Biol. Chem. 1973, 248, 4793–4796. [Google Scholar] [PubMed]
- Gomez, J.M.; Hernandez, J.A.; Jimenez, A.; del Rio, L.A.; Sevilla, F. Differential response of antioxidative enzymes of chloroplasts and mitochondria to long-term NaCl stress of pea plants. Free Radic. Res. 1999, 31 (Suppl. 1), 11–18. [Google Scholar] [CrossRef]
- Hernández, J.A.; Jiménez, A.; Mullineaux, P.; Sevilla, F. Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defences. Plant Cell Environ. 2000, 23, 853–862. [Google Scholar] [CrossRef]
- Vanacker, H.; Harbinson, J.; Ruisch, J.; Carver, T.L.W.; Foyer, C.H. Antioxidant defences of the apoplast. Protoplasma 1998, 205, 129–140. [Google Scholar] [CrossRef]
- Law, M.Y.; Charles, S.A.; Halliwell, B. Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts. The effect of hydrogen peroxide and of Paraquat. Biochem. J. 1983, 210, 899–903. [Google Scholar] [CrossRef]
- Zhang, J.; Kirkham, M.B. Enzymatic responses of the ascorbate-glutathione cycle to drought in sorghum and sunflower plants. Plant Sci. 1996, 113, 139–147. [Google Scholar] [CrossRef]
- Vanacker, H.; Carver, T.L.W.; Foyer, C.H. Pathogen-induced changes in the antioxidant status of the apoplast in barley leaves. Plant Physiol. 1998, 117, 1103–1114. [Google Scholar] [CrossRef] [PubMed]
- Pignocchi, C.; Foyer, C.H. Apoplastic ascorbate metabolism and its role in the regulation of cell signalling. Curr. Opin. Plant Biol. 2003, 6, 379–389. [Google Scholar] [CrossRef]
- Clemente-Moreno, M.J.; Gago, J.; Díaz-Vivancos, P.; Bernal, A.; Miedes, E.; Bresta, P.; Liakopoulos, G.; Fernie, A.R.; Hernández, J.A.; Flexas, J. The apoplastic antioxidant system and altered cell wall dynamics influence mesophyll conductance and the rate of photosynthesis. Plant J. 2019, 99, 1031–1046. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Ali, Q. Relative membrane permeability and activities of some antioxidant enzymes as the key determinants of salt tolerance in canola (Brassica napus L.). Environ. Exp. Bot. 2008, 63, 266–273. [Google Scholar] [CrossRef]
- Bustos, D.; Lascano, R.; Villasuso, A.L.; Machado, E.; Senn, M.E.; Córdoba, A.; Taleisnik, E. Reductions in maize root-tip elongation by salt and osmotic stress do not correlate with apoplastic O2—Levels. Ann. Bot. 2008, 102, 551–559. [Google Scholar] [CrossRef]
- Córdoba-Pedregosa, M.D.C.; Córdoba, F.; Villalba, J.M.; González-Reyes, J.A. Zonal changes in ascorbate and hydrogen peroxide contents, peroxidase, and ascorbate-related enzyme activities in onion roots. Plant Physiol. 2003, 131, 697–706. [Google Scholar] [CrossRef]
- Ikbal, F.E.; Antonio Hernandez, J.; Barba-Espin, G.; Koussa, T.; Aziz, A.; Faize, M.; Diaz-Vivancos, P. Enhanced salt-induced antioxidative responses involve a contribution of polyamine biosynthesis in grapevine plants. J. Plant Physiol. 2014, 171, 779–788. [Google Scholar] [CrossRef]
- Cantabella, D.; Piqueras, A.; Acosta-Motos, J.R.; Bernal-Vicente, A.; Hernández, J.A.; Díaz-Vivancos, P. Salt-tolerance mechanisms induced in Stevia rebaudiana Bertoni: Effects on mineral nutrition, antioxidative metabolism and steviol glycoside content. Plant Physiol. Biochem. 2017, 115, 484–496. [Google Scholar] [CrossRef]
- Acosta-Motos, J.-R.; Diaz-Vivancos, P.; Alvarez, S.; Fernandez-Garcia, N.; Jesus Sanchez-Blanco, M.; Antonio Hernandez, J. Physiological and biochemical mechanisms of the ornamental Eugenia myrtifolia L. plants for coping with NaCl stress and recovery. Planta 2015, 242, 829–846. [Google Scholar] [CrossRef]
- Acosta-Motos, J.R.; Diaz-Vivancos, P.; Alvarez, S.; Fernandez-Garcia, N.; Jesus Sanchez-Blanco, M.; Hernandez, J.A. NaCl-induced physiological and biochemical adaptative mechanisms in the ornamental Myrtus communis L. plants. J. Plant Physiol. 2015, 183, 41–51. [Google Scholar] [CrossRef]
- Brisson, L.F.; Zelitch, I.; Havir, E.A. Manipulation of catalase levels produces altered photosynthesis in transgenic tobacco plants. Plant Physiol. 1998, 116, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Ros-Barceló, A.; Gómez-Ros, L.V.; Ferrer, M.A.; Hernández, J.A. The apoplastic antioxidant enzymatic system in the wood-forming tissues of trees. Trees Struct. Funct. 2006, 20, 145. [Google Scholar] [CrossRef]
- Polle, A.; Chakrabarti, K.; Schürmann, W.; Rennenberg, H. Composition and properties of hydrogen peroxide decomposing systems in extracellular and total extracts from needles of Norway spruce (Picea abies L., karst.). Plant Physiol. 1990, 94, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Gueta-Dahan, Y.; Yaniv, Z.; Zilinskas, B.A.; Ben-Hayyim, G. Salt and oxidative stress: Similar and specific responses and their relation to salt tolerance in citrus. Planta 1997, 203, 460–469. [Google Scholar] [CrossRef] [PubMed]
- López-Climent, M.F.; Arbona, V.; Pérez-Clemente, R.M.; Gómez-Cadenas, A. Relationship between salt tolerance and photosynthetic machinery performance in citrus. Environ. Exp. Bot. 2008, 62, 176–184. [Google Scholar] [CrossRef]
- Hernández, J.A.; Aguilar, A.B.; Portillo, B.; López-Gómez, E.; Beneyto, J.M.; García-Legaz, M.F. The effect of calcium on the antioxidant enzymes from salt-treated loquat and anger plants. Funct. Plant Biol. 2003, 30, 1127–1137. [Google Scholar] [CrossRef]
- Mittova, V.; Tal, M.; Volokita, M.; Guy, M. Up-regulation of the leaf mitochondrial and peroxisomal antioxidative systems in response to salt-induced oxidative stress in the wild salt-tolerant tomato species Lycopersicon pennellii. Plant Cell Environ. 2003, 26, 845–856. [Google Scholar] [CrossRef]
- Hernández, J.A.; Barba-Espín, G.; Clemente-Moreno, M.J.; Díaz-Vivancos, P. Plant Responses to Salinity Through an Antioxidative Metabolism and Proteomic Point of View; Springer International Publishing: Switzerland, 2016; ISBN 9783319421834. [Google Scholar]
- Asada, K. Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol. 2006, 141, 391–396. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Adams, W.W. The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci. 1996, 1, 21–26. [Google Scholar] [CrossRef]
- Taïbi, K.; Taïbi, F.; Ait Abderrahim, L.; Ennajah, A.; Belkhodja, M.; Mulet, J.M. Effect of salt stress on growth, chlorophyll content, lipid peroxidation and antioxidant defence systems in Phaseolus vulgaris L. S. Afr. J. Bot. 2016, 105, 306–312. [Google Scholar] [CrossRef]
- Di Baccio, D.; Navari-Izzo, F.; Izzo, R. Seawater irrigation: Antioxidant defence responses in leaves and roots of a sunflower (Helianthus annuus L.) ecotype. J. Plant Physiol. 2004, 161, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Aliniaeifard, S.; Hajilou, J.; Tabatabaei, S.J.; Sifi-Kalhor, M. Effects of Ascorbic Acid and Reduced Glutathione on the Alleviation of Salinity Stress in Olive Plants. Int. J. Fruit Sci. 2016, 19, 395–409. [Google Scholar] [CrossRef]



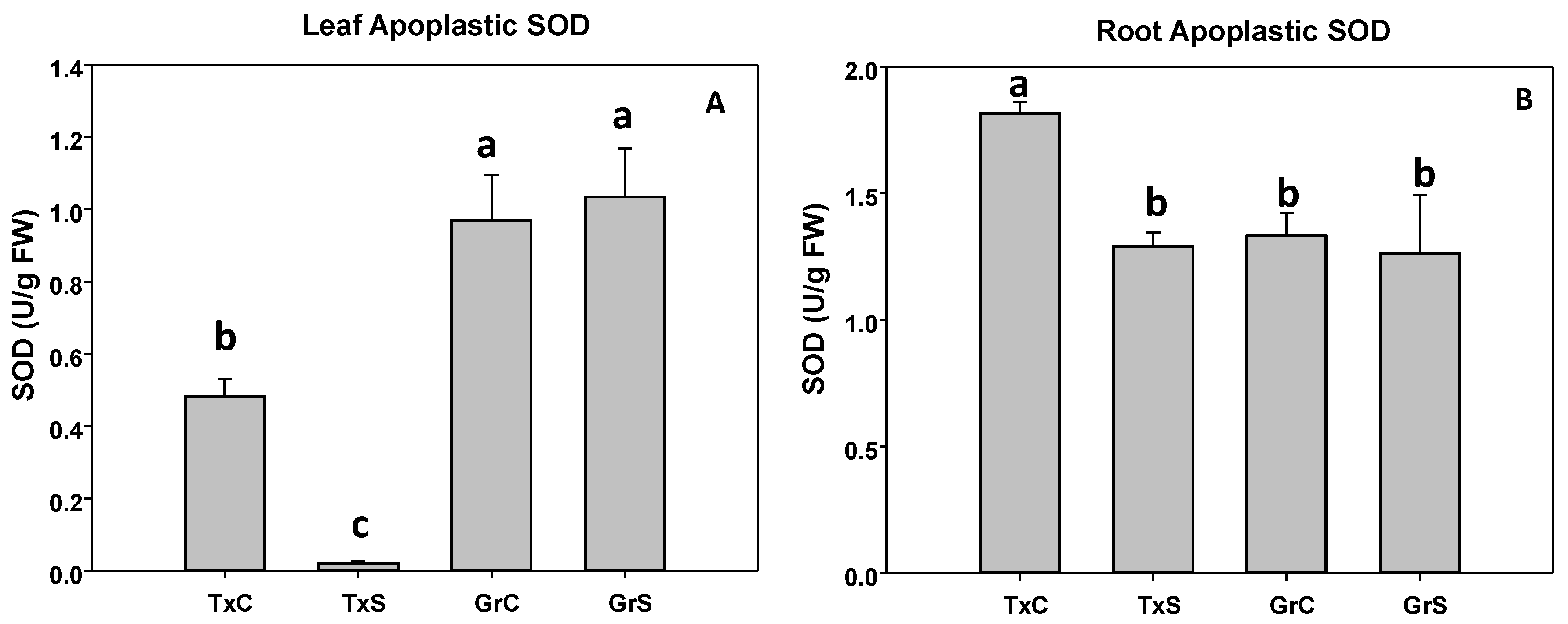
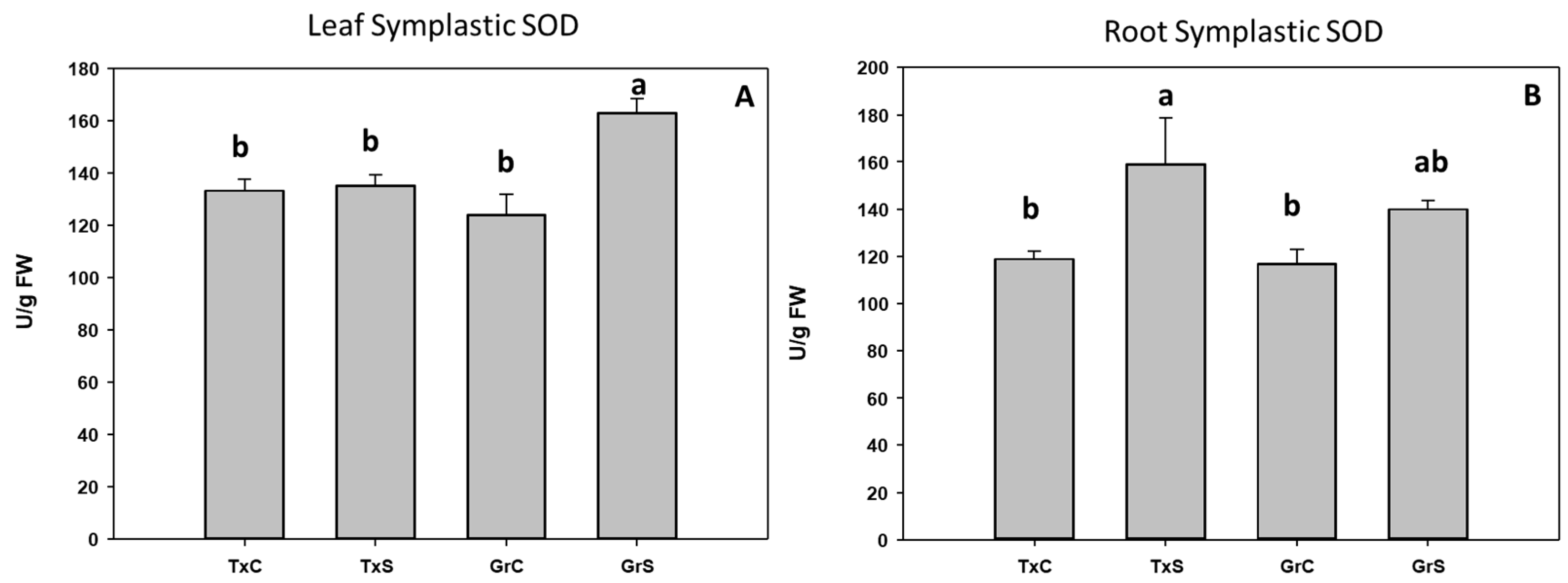
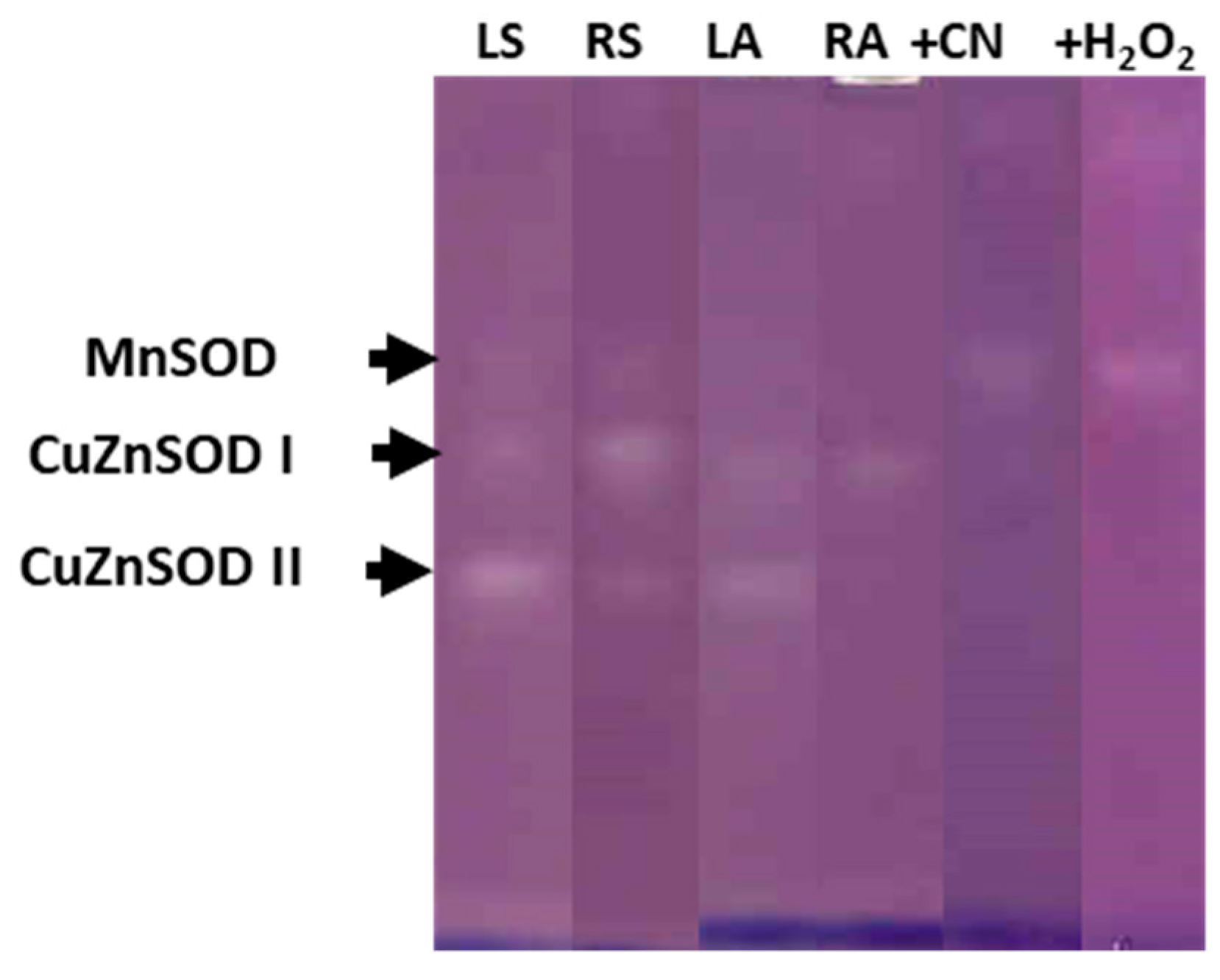
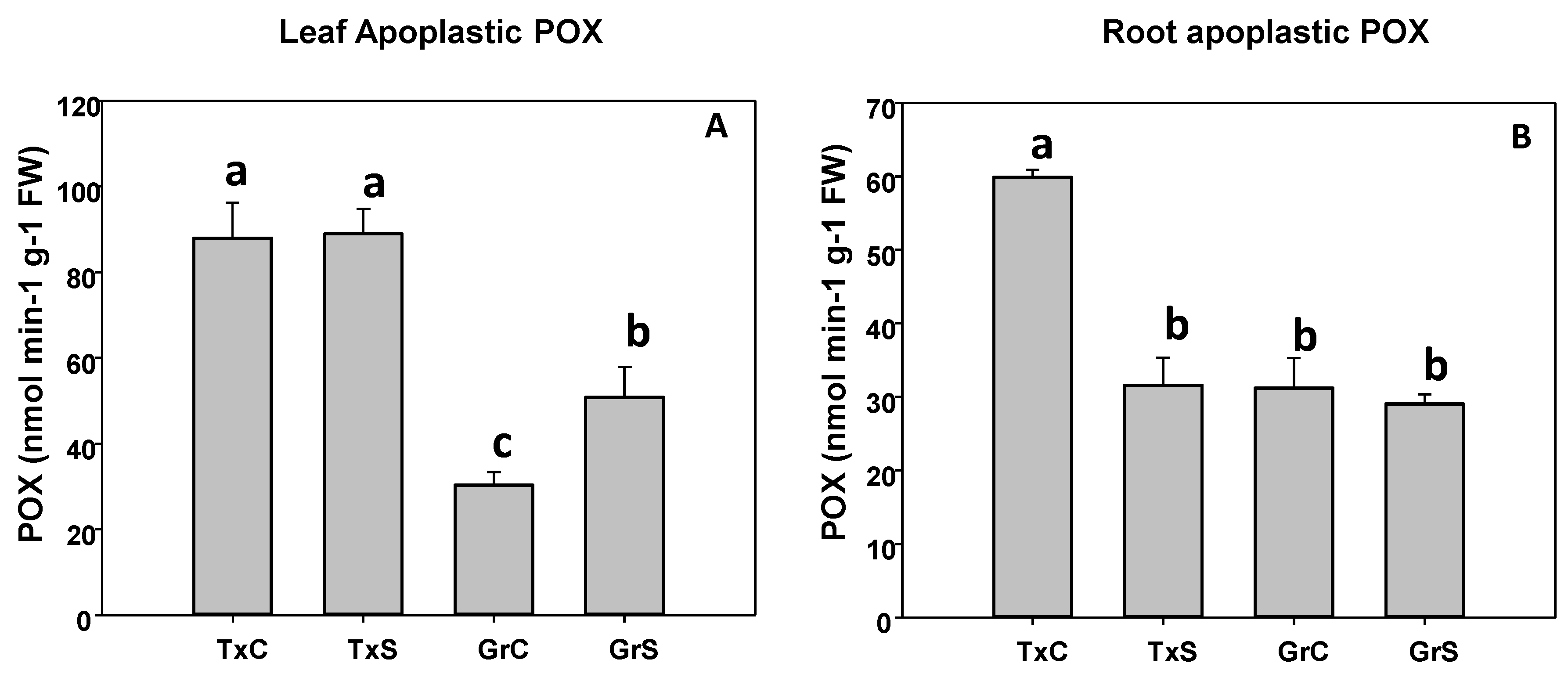
| Genotype | Treatment | ASC | DHA | Redox State | |
|---|---|---|---|---|---|
| (nmol g−1 FW) | (%) | ||||
| ‘Texas 502’ | Control | 1020.9 b,c | 523.4 a | 66.1 | |
| Leaves | Salinity | 1166.1 a | 536.7 a | 68.7 | |
| ‘Granex 429’ | Control | 986.2 c | 494.2 a | 66.6 | |
| Salinity | 1151.3 a,b | 451.8 a | 71.8 | ||
| ‘Texas 502’ | Control | 893.5 a,b | 602.3 a | 59.7 | |
| Roots | Salinity | 1030.2 a | 462.3 b | 69.0 | |
| ‘Granex 429’ | Control | 961.8 a,b | 503.3 b | 65.6 | |
| Salinity | 808.1 b | 262.5 c | 75.5 | ||
| Genotype | Treatment | GSH | GSSG | Redox State | |
|---|---|---|---|---|---|
| (nmol g−1 FW) | (%) | ||||
| ‘Texas 502’ | Control | 11.59 a | 2.66 c | 81.3 | |
| Leaves | Salino | 5.84 b | 3.94 b | 59.7 | |
| ‘Granex 429’ | Control | 6.21 b | 4.42 b | 58.4 | |
| Salino | 3.00 b | 5.76 a | 34.2 | ||
| ‘Texas 502’ | Control | 52.43 a | 9.16 a | 85.1 | |
| Roots | Salino | 56.82 a | 9.20 a | 86.1 | |
| ‘Granex 429’ | Control | 34.97 b | 10.30 a | 77.2 | |
| Salino | 48.1 a,b | 11.55 a | 80.6 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García, G.; Clemente-Moreno, M.J.; Díaz-Vivancos, P.; García, M.; Hernández, J.A. The Apoplastic and Symplastic Antioxidant System in Onion: Response to Long-Term Salt Stress. Antioxidants 2020, 9, 67. https://doi.org/10.3390/antiox9010067
García G, Clemente-Moreno MJ, Díaz-Vivancos P, García M, Hernández JA. The Apoplastic and Symplastic Antioxidant System in Onion: Response to Long-Term Salt Stress. Antioxidants. 2020; 9(1):67. https://doi.org/10.3390/antiox9010067
Chicago/Turabian StyleGarcía, Grisaly, María José Clemente-Moreno, Pedro Díaz-Vivancos, Marina García, and José Antonio Hernández. 2020. "The Apoplastic and Symplastic Antioxidant System in Onion: Response to Long-Term Salt Stress" Antioxidants 9, no. 1: 67. https://doi.org/10.3390/antiox9010067
APA StyleGarcía, G., Clemente-Moreno, M. J., Díaz-Vivancos, P., García, M., & Hernández, J. A. (2020). The Apoplastic and Symplastic Antioxidant System in Onion: Response to Long-Term Salt Stress. Antioxidants, 9(1), 67. https://doi.org/10.3390/antiox9010067







