Abstract
There are no two main-group elements that exhibit more similar physical and chemical properties than sulfur and selenium. Nonetheless, Nature has deemed both essential for life and has found a way to exploit the subtle unique properties of selenium to include it in biochemistry despite its congener sulfur being 10,000 times more abundant. Selenium is more easily oxidized and it is kinetically more labile, so all selenium compounds could be considered to be “Reactive Selenium Compounds” relative to their sulfur analogues. What is furthermore remarkable is that one of the most reactive forms of selenium, hydrogen selenide (HSe− at physiologic pH), is proposed to be the starting point for the biosynthesis of selenium-containing molecules. This review contrasts the chemical properties of sulfur and selenium and critically assesses the role of hydrogen selenide in biological chemistry.
1. Overview of Chalcogens in Biology
Chalcogens are the chemical elements in group 16 of the periodic table. This group, which is also known as the oxygen family, consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive element polonium (Po). O, S, and Se are essential for life, although Se is only required in trace amounts of about 15 mg in a typical 70 kg adult human [1]. The conventional roles of oxygen in aerobic respiration and photosynthesis (O2), water (~65 wt % of the human body), and a myriad of oxygen-containing bioorganic molecules (amino acids, nucleotides, etc.), may be contrasted with the sometimes deleterious properties of biological reactive oxygen species (ROS: OH•, O2−•, H2O2, etc.) [2,3,4]. Similarly, sulfur serves a conventional role in the amino acids (e.g., Cys and Met) and in biological reactive sulfur species (RSS: H2S, OSCN− [5,6], polysulfides, etc.) [7,8,9,10,11,12,13]. Although it is toxic in large doses, selenium is an essential micronutrient for animals [14,15,16,17]. In plants, it sometimes occurs in toxic amounts as forage, e.g., locoweed [18]. Selenium is a component of the amino acids selenocysteine (Sec, the “21st amino acid”) [19] and selenomethionine (SeM) [20]. In humans, according to proteomics, Sec is incorporated into at least 25 different proteins [21,22,23]. The identified proteins, including glutathione peroxidases and certain forms of thioredoxin reductase, incorporate Sec into their active sites, whereas SeM is randomly incorporated into proteins [21,22,24,25,26]. As selenium compounds are generally more reactive than the corresponding sulfur derivatives (vide infra), one might argue that all biologically relevant selenium compounds are “Reactive Selenium Species”. The present review focuses on hydrogen selenide (H2Se), typically the least stable oxidation state for selenium in a biological setting and the putative reactant that leads to all known selenium-containing biomolecules.
2. Chemistry of Sulfur vs. Selenium
Sulfur is approximately 105 times more abundant in the human body than selenium, but the latter element is selected for certain biological functions [27]. It is particularly remarkable that this is achieved, as there are no other two main-group elements that exhibit more similar physical and chemical properties [28]. Nonetheless, the selective use of selenium must eventually rely upon the unique chemical properties of the element. Despite the fact that the average atomic mass of Se is more than twice that of S, the atomic radii are remarkable similar, as a consequence of d-block contraction [29], as are the bond lengths that are observed in main-group compounds (Table 1). Furthermore, the electronegativities of the two elements are comparable, and both are more similar to carbon (χ = 2.55) than any other elements of the periodic table. However, important differences exist between the two elements. S forms stronger covalent bonds relative to Se (Table 1). Additionally, it is easier to oxidize Se than S (Table 1). Moreover, selenols are more acidic than thiols with acid dissociation constants that are typically 3–4 orders of magnitude larger (Table 1).

Table 1.
General properties of sulfur and selenium.
2.1. Selenium Compounds Are More Reactive
Given that free energy relationships frequently exist between thermodynamics and kinetics [49,50], it is unsurprising that Se compounds tend to be more reactive than S compounds. For example, for the exchange of thiols and selenolates (note the proton state of each [51]) with disulfides, diselenides, and mixed chalcogenides, reaction rates of selenium as a nucleophile and as an electrophile are 2–3 and 4 orders of magnitude higher, respectively, than those of sulfur at neutral pH [52]:
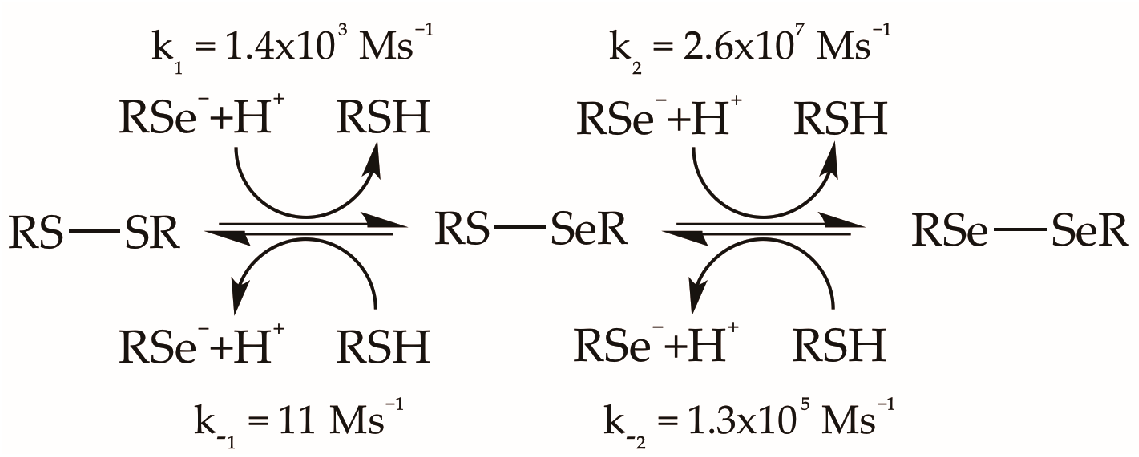
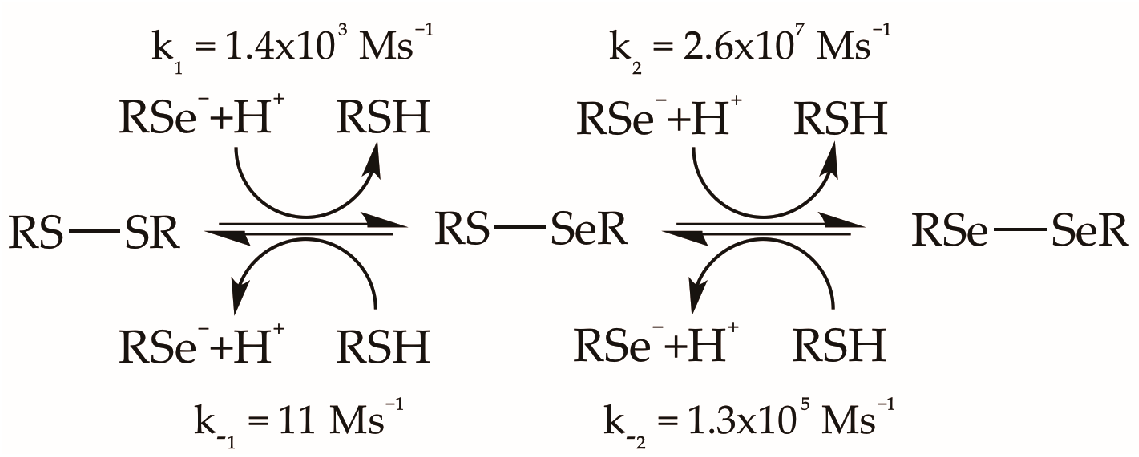
From left to right, the greater nucleophilicity of Sec relative to Cys may be attributed to the fact that Se is deprotonated and S is protonated at neutral pH, and to the fact that a S–H covalent bond is formed when selenolate reacts and Cys is liberated. In the other direction, the greater electrophilicity of Se in this particular reaction is attributed to properties of the S–S vs. Se–Se vs. Se–S bonds, and not the nature of the leaving group. From these rate constants, the ratio of cystine (RSSR) to selenocystine (RSeSeR) is given by: K = K1 × K2 = k1 × k2/k−1 × k−2 = 2.5 × 104 for [Cys] = [Sec]. While this result may at first appear inconsistent with the X–X bond strengths of Table 1, the equilibrium of Equation (2) in fact reflects a very important difference between S and Se, selenolates are more stable than thiolates, as reflected in the pKa values of Table 1:
The difference in pKa between Cys and Sec (Equation (5)) drives the formation of the unfavorable Se–Se bond at pH 7. This difference in pKa is largely due to the greater polarizability (not electronegativity) of Se [40,41] that delocalizes charges and stabilizes the selenolate (Table 1). The example of Equations (3)–(5) illustrates how subtle differences in the chemical properties between S and Se can be exploited to drive otherwise unfavorable reactions.
2.2. Selenium Favors Higher Oxidation States
The ionization energies of Table 1 suggest Se is easier to oxidize than S. This fact is further evidenced in the Frost diagrams of Figure 1 [53,54]. Thus, for a particular pH (0 or 14) and a given oxidation state (−II to +VI), Se is more easily oxidized than S. Note the general trend that oxidation potentials increase with oxidation states at low pH and they decrease with oxidation state at high pH. These trends are due to the effects of the proton states of the species involved. Finally, note that the most stable oxidation state at pH 0 for S and Se are −II and 0, respectively.
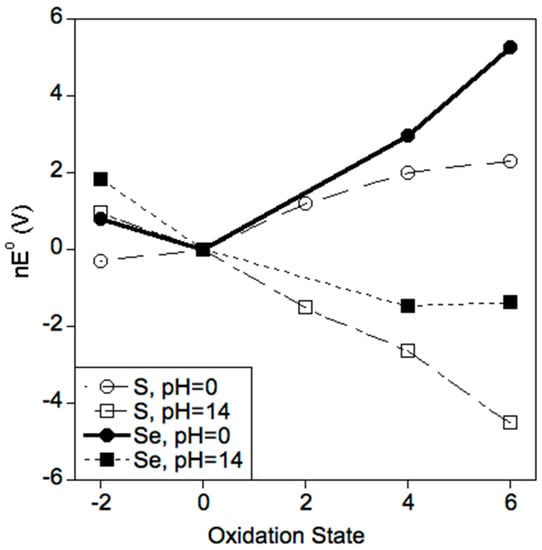
Figure 1.
Frost diagrams for sulfur and selenium at pH 0 and 14. The nE° = 0 value is arbitrarily assigned to the zero-valent state. Note that S(−II) and Se(0) are the most stable oxidation states at pH 0, and that S(VI) and Se(VI) are the most stable at pH 14.
3. Selenium in Biology
Se is an essential trace element and a potential toxin. It is the only trace element found in proteins that is genetically encoded. Selenocysteine (Sec) [19], the “21st amino acid”, is incorporated into selenoproteins by a co-translational process that uses translation machinery that redefines UGA (uracil/guanine/Adenine) codons to encode Sec [56,57]. Many biological functions are performed by selenoproteins, all of which contain Sec residues in their primary structures [58]. The roles of Sec in proteins has been thoroughly reviewed [21,23,24,59,60].
Although an essential trace element, Se is toxic if taken in excess. Exceeding the Tolerable Upper Intake Level (UL) of 400 micrograms per day can lead to selenosis [61]. Selenomethionine (SeM), selenocysteine (Sec), selenite (SeO32−), and selenate (SeO42−) account for almost all the Se in diets, and all these forms are absorbed without regulation. However, SeM is the principal chemical form of Se in most mammalian diets. It is synthesized by plants and incorporated randomly into plant proteins at Met positions. Once absorbed, SeM is transported and incorporated randomly into mammalian proteins using the Met system.
3.1. Which Oxidation States of Sulfur and Selenium Are (Most) Stable in Biology?
Figure 2 illustrates the Pourbaix diagrams for S and Se [62]. The diagrams describe the thermodynamically preferred oxidation states of the elements as a function of applied potential and pH [63,64,65]. Note these propensity diagrams are concentration-dependent [66]. Two examples for each element are illustrated: standard conditions (top, total concentration of chalcogen equal to 1 M), and more dilute conditions (bottom, 1 μM). The dashed red lines encompass the stability region of water. Potentials above the top line would oxidize water to produce O2, and potentials below the bottom line would reduce water to produce H2. Any species that overlaps the stability region of water would be thermodynamically stable under the E/pH conditions of the overlap. For S at high concentrations (top left), the −II, 0, and +VI oxidations states are stable, although S(0) disproportionate to S(−II) and S(+IV) under alkaline conditions. However, at lower concentrations of S, only the −II and +VI oxidations states are thermodynamically stable (bottom left). In contrast to S, Se in the −II, 0, +IV, and +VI oxidations states are stable in water, although H2Se is capable of reducing water at high concentrations under acidic conditions (top right), but not once the H2Se is sufficiently diluted (bottom right). We can conclude that selenide is thermodynamically stable with respect to water under anaerobic conditions, but what about oxygen-rich environments? Selenide solutions above 10−6 M are known to be oxidized under atmospheric pressures of oxygen in less than three minutes at pH 7 [67]. Air-saturated water contains 2.7 × 10−4 M dissolved O2. Since it has been determined that the reaction is zero-order in oxygen for [O2] > 10−4 M and first-order in [H2Se], we can conclude that the half-life for oxidation of selenide to colloidal elemental selenium in air-saturated water (in the absence of other reactants) is about 30 s at pH 7, and that the rate is independent of the concentration of selenide [67]. By comparison, solutions of H2S are oxidized over a period of several hours under similar conditions.
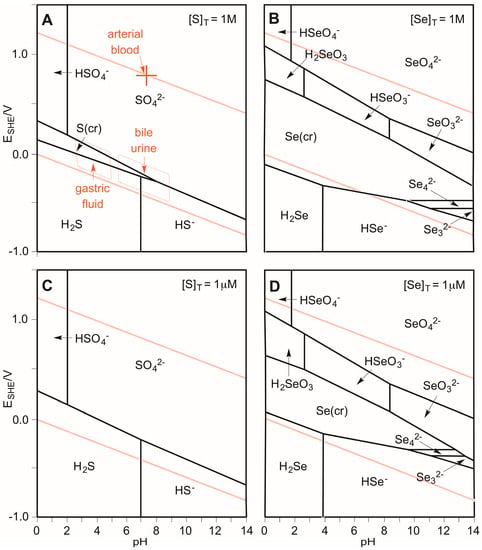
Figure 2.
Pourbaix diagrams for S (A and C) and Se (B and D) at 1M total chalcogen (A and B) and 1 μM total chalcogen (C and D) at 25 °C. The red lines define the stability region of water. Various physiological fluids are illustrated (A) [55].
Three important conclusions can be drawn from Figure 1 and Figure 2: (1) Se is more easily oxidized relative to S (Figure 1); (2) Se exhibits more stable oxidation states in water relative to S (Figure 2); and (3) Se(0) and S(+VI) are the most thermodynamically stable oxidation states for Se and S, respectively, in an aqueous environment in the absence of an applied SHE potential (Figure 1 and Figure 2). For tissues that are perfused with arterial blood at pH 7.37 (an equivalent potential of +0.782 V), the thermodynamically most stable oxidation state for S and Se at all concentrations is +VI (Figure 2). In contrast, the −II oxidation state becomes more stable under the reducing conditions of gastric fluid, bile, and urine (Figure 2). Importantly, this discussion of thermodynamics does not address the issue of whether a given reaction is kinetically viable. Indeed, biological redox systems are typically not in thermodynamic equilibrium [68]. For example, the thiol and disulfide forms of glutathione (GSH in its reduced and GSSG in its oxidized forms) and cysteine (Cys in its reduced and CySSCy in its oxidized forms) that are measured in human plasma suggest that the GSSG/2GSH and the CySSCy/2Cys pools are not in redox equilibrium [69].
Note that in many biological settings, H2Se is thermodynamically unstable, for example, in the oxygenated intercellular fluids of eukaryotic cells. However, those cells contain millimolar concentrations of GSH, most of which is kept in the reduced state by glutathione reductase. Furthermore, SeO32− is known to react spontaneously with GSH [70,71,72,73,74] to initially produce selenodiglutathione (GS-Se-SG), Equation (6). In the presence of excess GSH, GSSeSG is further reduced to glutathioselenol (GSSeH), Equation (7). GSSeH either spontaneously dismutates into Se(0) and GSH, Equation (8), or is further reduced by GSH to yield H2Se, Equation (9). As discussed before, H2Se is readily oxidized by O2 into Se(0), Equation (10).
However, excess GSH can protect H2Se from oxidation. Accordingly, while Se(−II) may be thermodynamically unstable in a biological setting, it may be kinetically stabilized, Equation (11).
Indeed, it is precisely the role of GSH to protect functional groups from oxidation.
3.2. Role of Selenide In Vivo
Before discussing H2Se, it is valuable to review the mechanisms by which its S congener is released in vivo: (1) H2S is immediately released after its production by enzymes (e.g., cystathionine β-synthase [75], cystathionine γ-lyase [76], 3-mercaptopyruvate sulfurtransferase [77], and cysteine aminotransferase [78]); and (2) it can be released via non-enzymatic processes from stored labile sulfur [79]. Two forms of sulfur stores in cells that have been identified are [80,81]: (1) acid-labile sulfur [82]; and (2) bound sulfane sulfur (RS(S)n−, which releases H2S under reducing conditions) [83]. Acid-labile sulfur is mainly sulfur atoms in the iron–sulfur proteins (e.g., ferredoxins), which play a critical role in a wide range of redox reactions. However, H2S is only released from acid-labile sulfur below pH 5.4 [82], which is well below most physiologic fluids. Like Cys, H2S exhibits both anti- and pro-oxidant behavior [84].
In the previous section we discussed the various oxidation states of Se that exhibit thermodynamic stability with respect to water and molecular oxygen (for certain [Se], [H+], and [O2]). However, even under conditions in which H2Se exhibits thermodynamic stability, it remains a powerful reductant, with a reduction potential close to that of H2. Not surprisingly, H2Se has not been directly identified in vivo. Nonetheless, it is widely held that H2Se exists at the intersection of various metabolic pathways (Figure 3). The principle dietary sources of selenium are SeM and SeO32−, and both are apparently sources of H2Se. Two metabolic pathways have been identified that convert SeM to H2Se. SeM is converted to methylselenol (MeSeH) [85] by γ-lyases [86], which is subsequently demethylated to H2Se [87]. MeSeH is also produced from methylselenocysteine (MeSeCys) and cysteine β-lyases [88,89]. Alternatively, a transselenation pathway yields Sec, which is subsequently reduced by β-lyases to H2Se [90]. The reduction of SeO32− to H2Se can either be carried out directly by thioredoxin and thioredoxin reductases (TrxR) [91], or by glutathione reductases (GR) via the intermediate selenodiglutathione (GSSeSG) [92]. There is one known productive use of H2Se, as the source of selenium for selenophosphate synthetase (SPS) [93]. The product selenophosphate, SePO33−, is subsequently used in the synthesis of Sec. H2Se can be excreted by two metabolic pathways, as selenosugars or as methylated derivatives. The most common elimination products for Se are selenium-containing carbohydrates [94] such as methyl-2-acetamido-2-deoxy-1-seleno-β-d-galactopyranoside (SeGalNAc). However, H2Se is eliminated after methylation in the form of dimethylselenide (Me2Se) via breath [95] or trimethylselenonium (Me3Se+) ion via urine [96]. We will next systematically examine the evidence for involvement of H2Se in each of these reactions.
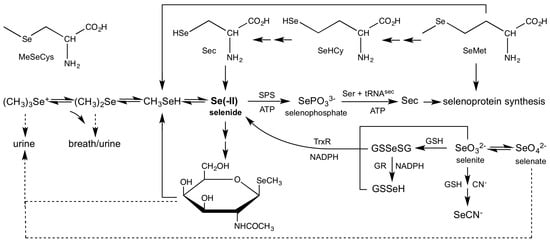
Figure 3.
Metabolic pathways for dietary selenium compounds.
3.3. Mechanism of Selenophosphate Synthetase (SPS)
The synthesis of Sec is the only known productive use of H2Se in a biological setting. The first step of the biosynthesis involves production of the intermediate SePO33−. SPS requires some form of reduced Se (such as H2Se) and ATP (adenosine triphosphate) as substrates in order to generate a stoichiometric amount of SePO33−, AMP (adenosine monophosphate), and orthophosphate [93]. Early studies by Stadtman et al. established that a SPS enzyme preparation from Salmonella typhimurium produced “a compound containing selenium bonded to phosphorus” only in the presence of NaSeH, MgCl2, PO43−, and ATP [97,98]. The compound formed was later identified as SePO33− [99]. Omitting any of the components of the system eliminated production of SePO33−, although SeO32− + dithiothreitol (DTT) could replace NaSeH, whereby Sec produced lesser amounts of SePO33− that were attributed to contamination of the Sec by NaSeH. While this is not direct detection of H2Se, we find these observations the most compelling evidence for its involvement in biochemistry.
3.4. Reduction of Inorganic Selenium
SeO32− and SeO42− are the primary environmental inorganic sources of Se. To be incorporated into amino acids, a reduction must occur from Se(IV) and Se(VI) to Se(−II), a process that involves 6 and 8 e−, respectively. While one- and two-electron reduction mechanisms are commonplace, it is very improbable that the elementary reaction steps involve 4–6 e− processes. Consequently, Se(0) is likely to be an intermediate, albeit not necessary in elemental form. Multi-electron biochemical reductions frequently involve unstable intermediates, for example, in fixation, an 8 e− process. Unstable intermediates are sometimes protected at the active sites at which they are produced, and in other cases, they are converted to relatively stable free molecules prior to further reaction. It is noteworthy that a number of derivatives of Se in the formal −II oxidation state have been identified in a biological setting. Besides H2Se, such species include RSeH (MeSeH [87,100], Sec [101], SeHCys [86], etc.), SePO33− [93], and SeCN− [102]. Many of these compounds are on the pathway to/from H2Se, but there exists very little direct evidence that H2Se is involved. Nonetheless, given the significant evidence that SPS employs H2Se as a substrate, it has been shown that Se delivered from Sec by an Escherichia coli cysteine desulfurase-like protein can replace H2Se in the in vitro SPS assay for SePO33− formation [103,104]. These and related studies have demonstrated, in the presence of supporting enzymes, that Se(−II) can be mobilized from alternative sources.
3.5. Mechanism of Selenocysteine β-Lyase
Sec is incorporated into proteins co-translationally, as directed by the UGA codon. This is achieved by employing a special tRNA that has an anticodon complimentary to UGA (tRNASec). In many species of bacteria, this tRNASec is first amino acylated with serine to yield seryl-tRNASec, which is subsequently converted to selenocysteyl-tRNASec. SePO33− serves as the Se donor in the known systems that produce selenocysteyl-tRNASec [19]. Selenocysteine β-lyase catalyzes the chemical reaction: Sec + reduced acceptor → H2Se + Ala + acceptor [90,97]. The enzyme was first purified from pig liver in 1982 [90]. The reducing agent in laboratory studies is typically DTT. A subsequent, more detailed investigation of the analogous enzyme isolated from Citrobacter feundii suggested elemental Se(0) and not H2Se was produced in the absence of DTT [105]. A decade later, the nifs gene product was found to be a pyridoxal phosphate binding enzyme that catalyzes the desulfurization of Cys to yield Ala and S(0). Relevant to the present topic, as mentioned previously, these so-called NIFS proteins (or cysteine desulfurase) were found to employ Sec as a source of Se for SPS to produce SePO33− [103,104]. Importantly, transfer of Se from free Sec by the NIFS protein to SPS occurred in aqueous solution without equilibration with a solvent [104]. Thus, a complex between the NIFS and SPS enzymes may take place that facilitates the direct transfer of Se(−II). In addition to allowing for greater differentiation between S and Se in vivo, such a hands-off mechanism could serve to stabilize reactive intermediates (cf. nitrogenase).
3.6. Demethylation of Methylselenol
There are several pathways that lead to MeSeH. Once formed, MeSeH can be sequentially dimethylated to give the diselenide (Me)2Se, and trimethylated to give (Me)3Se+. The suggestion that H2Se might be an intermediate in the production of MeSeH from SeO32− was first made in 1966 [106]. However, the focus here is on the demethylation of MeSeH to give H2Se [87], which was first proposed forty years later [107,108,109]. To date, H2Se has not been directly observed by the demethylation of MeSeH. However, the Se liberated by rat organ supernatants and homogenates was quantified as its oxidation product SeO32− [107]. Without the identification of specific demethylation pathways and the exclusion of alternative processes, it will not be possible to unambiguously identify demethylation of MeSeH as a direct pathway to H2Se.
3.7. Incorporation of Selenide into Selenosugars
Selenosugars are major urinary metabolites for Se. [110]. While many of the published metabolic pathways of Se suggest selenosugars are derived from H2Se, there is in fact very little information on their biosynthetic pathways, and no sound evidence that Se(−II) is directly involved [111,112].
4. Unanswered Questions
4.1. Is Se an Anti-oxidant or a Pro-Oxidant?
The answer is probably both, but the underlying mechanisms remain unresolved. The potential anti-oxidant properties of Se-containing compounds are self-evident given the chemical properties of the element, and indeed natural and synthetic Se-containing small molecules are frequently touted as potential therapies for oxidative stress [113]. Furthermore, the SeM that is randomly incorporated into proteins is thought to protect nearby residues from oxidative damage [114,115] and SeM-containing proteins are themselves thought to be anti-oxidants [113]. However, it is likely that many of the mechanisms by which Se influences redox status are not intuitive. By way of illustration, Cys serves a key role within the tripeptide glutathione (GSH) as the primary anti-oxidant in eukaryotic cells, but free Cys also acts as a reductant of Fe(III) that supports deleterious Fenton chemistry, thereby serving as a pro-oxidant [116]. Consequently, GSH is naturally maintained in a reduced state, whereas Cys is kept in an oxidized state in vivo. However, the oxidation potential of GSH is very similar to Cys [117]. Why, then, are living cells that contain millimolar concentrations of GSH oxidatively stressed upon addition of Cys [118,119,120]? One explanation could be that the kinetics of the reduction of Cys are unfavorable. Indeed, as mentioned earlier, there is evidence that the GSSG/2GSH and the CySSCy/2Cys pools are not in redox equilibrium [69]. Enter the role of Se as a chalcogen that is more kinetically labile. Selenols are known to catalyze the interchange reactions of thiols and disulfides [121]. Therefore, one can envisage a mechanism whereby Se compounds could introduce a redox imbalance and oxidative stress in vivo, e.g., via release of Cys by H2Se and/or selenols:
Alternatively, Se compounds that are capable of effecting Rxn 12 may also be capable of directly reducing Fe(III), thereby propagating the Fenton cycle.
4.2. Why Is Inorganic Se More Toxic Than Organic Se?
The molecular mechanism of Se toxicity remains unclear. However, there is anecdotal evidence that excess Se influences redox homeostasis, especially when the source is inorganic Se [122,123,124,125,126]. Sec and SeM, particularly in their L-enantiomers, are less toxic than SeO32− [127] Indeed, it is widely held that the replacement of Met with SeM in proteins does not markedly influence their structures or functions. However, as a consequence of weak Se–Se and Se–S bonds, replacement of Cys with Sec at sites of dichalogenide bonds would likely result in structural instability. Furthermore, the replacement of Cys with Sec at the active sites of enzymes is expected to influence function as a consequence of the different redox and acid/base properties of the two residues. Consistent with these potential deleterious effects of random incorporation of Sec, Cys substitution with Sec in natural selenoproteins is generally believed to be genetically encoded, although there has been a recent report of incorporation of Sec into yeast in the absence of the UGA codon [128].
So what causes the toxicity of Se? Some insight into the toxicology of Se may be garnered from studies of the anticancer properties of Se compounds [129,130,131,132,133]. Oxidative stress and apoptosis occurs when mammalian cells are treated with SeO32− [124,134,135,136,137,138,139,140,141,142,143]. Superoxide (O2−•) is known to be produced when SeO32− reacts with GSH [144]. In Vitro evidence demonstrated that non-enzymatic reduction of SeO32− by GSH rapidly yields GSSeSG and O2−• [135,144,145,146,147]. Additionally, apoptosis is known to be induced by ROS like O2−• [3]. While the precise mechanism by which O2−• is produced when SeO32− is reduced by GSH is not clear, the following stoichiometry has been proposed [147]:
In contrast, SeM is not known to produce ROS [124], and while Sec can produce ROS [124], it is more tightly regulated (vide supra). Although the release of ROS during the oxidation of H2Se has apparently not been investigated, the reaction is believed to proceed via a chain-radical mechanism, which would likely involve one-electron processes and the formation of HSe. and ROS like O2−• [67]. In fact, H2Se breaks DNA phosphodiester bonds in vitro under aerobic conditions, and chromosome fragmentation has been observed in vivo [148,149,150], behavior that is consistent with the formation of ROS. Similar effects may be observed for SeO32− in the presence of reducing agents like GSH.
Besides the direct generation of ROS, it is conceivable that Se has indirect effects, for example by inhibiting thiol proteins that have roles in the management of oxidative stress. Furthermore, while the replacement of inorganic S2− with Se2− in vivo, for example in iron-sulfur proteins (e.g., ferredoxins), is without literature precedence, it seems logical that such substitution might be more readily achieved with inorganic sources of Se rather than organic sources like Sec or SeM. It is noteworthy that in vitro substitution of Se2− for S2− in ferredoxins have produced less stable proteins [151]. Since iron-sulfur proteins play critical roles in energy-related electron transfer pathways, it is possible that such substitutions might affect Fe–S/Se protein function. Surprisingly, thus far in vitro substitution of Se2− for S2− in Fe–S proteins has only marginally modified their properties [151]. It is noteworthy that early studies did not reveal significant differentiation between S2− for Se2− during chalcogenide incorporation into Fe-S proteins [151], but a recent reaction of nitrogenase with selenocyanate (vida infra) revealed highly selective incorporation of Se2− into the FeMoCo cluster [152]. Although the current literature does not suggest that the incorporation of inorganic Se into Fe–S clusters is a major source of Se toxicity, further investigation is warranted.
4.3. Newly Discovered and Undiscovered Biological Selenium Compounds
It has been recently discovered that selenocyanate (SeCN−) is present in human fluids [102]. It is useful to discuss the possible biological significance of SeCN− in parallel with its S congener thiocyanate (SCN−). SCN− is produced endogenously as a detoxification product of the rhodanase-catalyzed reaction between cyanide (CN−) and thiosulfate (S2O32−) in the liver [153]. In contrast, the production of SeCN− apparently does not require rhodanase [102], which is consistent with the higher reactivity of selenium (vide supra). It has been observed that in the presence of SeO32−, CN−, and GSH, SeCN− is produced [102]. Higher chemical yields of SeCN− are produced if SeO32− and GSH are replaced with selenosulfate (SeSO32−) [102]. Given the mechanism by which rhodanase operates (reaction of Cys-247 with S2O32− to produce a persulfide, CySSH, followed by reaction with CN− to regenerate Cys-247 and produce SCN−) [154,155], and given the higher reactivity of Se, we imagine that species like GSSeH (Equation (7)) may react with CN− in vivo to produce SeCN− [156]. There are many endogenous and exogenous sources of CN−, including the metabolism of vitamin B12 and certain foods containing cyanogenic glucosides, for example, nuts (especially almonds) and cruciferous vegetables (e.g., the Brassica) [157]. Nonmetabolic sources of cyanide in humans include tobacco and occupationally derived smoke (HCN, e.g., >200 mg/cigarette) [158], chlorination of glycine by human myeloperoxidase (MPO) during inflammation [159], and cyanogenesis (the biochemical production of CN−) by Pseudomonas aeruginosa (an opportunistic pathogen that infects wounds and the lungs of immune-compromised individuals) [160,161,162,163,164]. SCN− is abundant in all physiologic fluids and especially in those that are derived from the mucosae (e.g., saliva, lachrymal fluids, breast milk, and the mucosal layer of the lung), where the concentration of SCN− is one or two orders of magnitude larger than in blood plasma as a consequence of its active transport [165]. We note that the same transport mechanisms function with SeCN− [166,167], so SeCN− is apparently also concentrated in exocrine fluids [167]. Unlike many other inorganic ions, such as Ca2+ (biological messenger and structural component of bone), K+ (osmotic regulator), CO32− (pH buffering), and PO42− (ATP, DNA, etc.), SCN− is not generally considered to be a biologically functional ion. This is largely because SCN− is not chemically reactive (although it forms coordination-covalent bonds with metals). However, in addition to being a product of detoxification of CN−, the SCN− ion plays an important role as a substrate for human defensive peroxidases, components of the human innate defense stratagem that include lactoperoxidase (LPO), salivary peroxidase (SPO), myeloperoxidase (MPO), and eosinophil peroxidase (EPO). The product of oxidation of SCN− by the defensive peroxidases is hypothiocyanite (OSCN−), a non-antibiotic antimicrobial. While it is not known whether SeCN− is a substrate for these aforementioned peroxidases, and while the corresponding oxidized species hyposelenocyanite (OSeCN−) is not known, importantly, in contrast to SCN−, SeCN− is apparently chemically reactive, and it has been suggested that it is part of the intrinsic Se pool [102].
5. Conclusions
While related by the periodic table and similar chemical properties, S and Se compounds exhibit markedly different behavior in a biological setting. In general, S chemistry is regulated in vivo, whereas Se chemistry is not. Many of the chemical transformation that require enzymatic catalysts to effect for S, proceed via uncatalyzed reactions for Se. The present review has focused on the role of H2Se, the putative linchpin of Se biology. While it is clear that H2Se plays a critical role in biology, exploration of its function is hindered by low relative concentrations, high reactivity, and the fact that tell-tale cellular machinery is not required to carry out many biotransformations that likely involve H2Se. There is a need for a better general understanding of the chemistry of H2Se, and for the development of specific assays that will facilitate experimentation, including colorimetric probes of Se-containing biomolecules [8,168]
Perhaps researchers should take a cue from Nature, which has found a way to exploit the subtle physical and chemical differences of S and Se compounds, in developing novel methods to explore the biology of H2Se and other Se compounds.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Schroeder, H.A.; Frost, D.V.; Balassa, J.J. Essential trace metals in man: Selenium. J. Chronic Dis. 1970, 23, 227–243. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Ann. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [PubMed]
- Simon, H.U.; Haj-Yehia, A.; Levi-Schaffer, F. Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 2000, 5, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Ashby, M.T. Hypothiocyanite. Adv. Inorg. Chem. 2012, 64, 263–303. [Google Scholar]
- Ashby, M.T. Inorganic chemistry of defensive peroxidases in the human oral cavity. J. Dent. Res. 2008, 87, 900–914. [Google Scholar] [CrossRef] [PubMed]
- Mishanina, T.V.; Libiad, M.; Banerjee, R. Biogenesis of reactive sulfur species for signaling by hydrogen sulfide oxidation pathways. Nat. Chem. Biol. 2015, 11, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Lin, V.S.; Chen, W.; Xian, M.; Chang, C.J. Chemical probes for molecular imaging and detection of hydrogen sulfide and reactive sulfur species in biological systems. Chem. Soc. Rev. 2015, 44, 4596–4618. [Google Scholar] [CrossRef] [PubMed]
- Gruhlke, M.C.H.; Slusarenko, A.J. The biology of reactive sulfur species (RSS). Plant Physiol. Biochem. 2012, 59, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Jacob, C.; Anwar, A. The chemistry behind redox regulation with a focus on sulphur redox systems. Physiol. Plant. 2008, 133, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Jacob, C.; Lancaster, J.R.; Giles, G.I. Reactive sulphur species in oxidative signal transduction. Biochem. Soc. Trans. 2004, 32, 1015–1017. [Google Scholar] [CrossRef] [PubMed]
- Giles Gregory, I.; Jacob, C. Reactive sulfur species: An emerging concept in oxidative stress. Biol. Chem. 2002, 383, 375–388. [Google Scholar]
- Giles, G.I.; Tasker, K.M.; Jacob, C. Hypothesis: The role of reactive sulfur species in oxidative stress. Free Radic. Biol. Med. 2001, 31, 1279–1283. [Google Scholar] [CrossRef]
- Duntas, L.H.; Benvenga, S. Selenium: An element for life. Endocrine 2015, 48, 756–775. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohe, R. The evolving versatility of selenium in biology. Antioxid. Redox Signal. 2015, 23, 757–760. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics Integr. Biomet. Sci. 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Terry, N.; Zayed, A.M.; De Souza, M.P.; Tarun, A.S. Selenium in higher plants. Ann. Rev. Plant Physiol. Plant Mol. Biol. 2000, 51, 401–432. [Google Scholar] [CrossRef] [PubMed]
- Boeck, A.; Thanbichler, M. Selenocysteine. EcoSal Plus 2014, 1–12. [Google Scholar]
- Block, E.; Birringer, M.; Jiang, W.; Nakahodo, T.; Thompson, H.J.; Toscano, P.J.; Uzar, H.; Zhang, X.; Zhu, Z. Allium chemistry: Synthesis, natural occurrence, biological activity, and chemistry of Se-alk(en)ylselenocysteines and their γ-glutamyl derivatives and oxidation products. J. Agric. Food Chem. 2001, 49, 458–470. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Holmgren, A. Selenoproteins. J. Biol. Chem. 2009, 284, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Papp, L.V.; Lu, J.; Holmgren, A.; Khanna, K.K. From selenium to selenoproteins: Synthesis, identity, and their role in human health. Antioxid. Redox Signal. 2007, 9, 775–806. [Google Scholar] [CrossRef] [PubMed]
- Gromer, S.; Eubel, J.K.; Lee, B.L.; Jacob, J. Human selenoproteins at a glance. Cell. Mol. Life Sci. 2005, 62, 2414–2437. [Google Scholar] [CrossRef] [PubMed]
- Steinbrenner, H.; Speckmann, B.; Klotz, L.-O. Selenoproteins: Antioxidant selenoenzymes and beyond. Arch. Biochem. Biophys. 2016, 595, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.M.; Arthur, J.R. Selenium, selenoproteins and human health: A review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Holben, D.H.; Smith, A.M. The diverse role of selenium within selenoproteins: A review. J. Am. Diet. Assoc. 1999, 99, 836–843. [Google Scholar] [CrossRef]
- Johansson, A.-L.; Collins, R.; Arner Elias, S.J.; Brzezinski, P.; Hogbom, M. Biochemical discrimination between selenium and sulfur 2: Mechanistic investigation of the selenium specificity of human selenocysteine lyase. PLoS ONE 2012, 7, e30528. [Google Scholar] [CrossRef] [PubMed]
- Haynes, W.M.; Lide, D.R.; Bruno, T.J. Crc Handbook of Chemistry and Physics, 94th ed.; CRC Press: Boca Raton, FL, USA, 2013; p. 2668. [Google Scholar]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements, 2nd ed.; Elsevier: New York, NY, USA, 1997. [Google Scholar]
- Slater, J.C. Atomic radii in crystals. J. Chem. Phys. 1964, 41, 3199–3204. [Google Scholar] [CrossRef]
- Bondi, A. Van der waals volumes and radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]
- Allen, F.H. The cambridge structural database: A quarter of a million crystal structures and rising. Acta Crystallogr. B Struct. Sci. 2002, B58, 380–388. [Google Scholar] [CrossRef]
- Nicovich, J.M.; Kreutter, K.D.; van Dijk, C.A.; Wine, P.H. Temperature-dependent kinetics studies of the reactions bromine atom(2p3/2) + hydrogen sulfide ↔ mercapto + hydrogen bromide and bromine atom(2p3/2) + methanethiol ↔ methylthiol + hydrogen bromide. Heats of formation of mercapto and methylthio radicals. J. Phys. Chem. 1992, 96, 2518–2528. [Google Scholar] [CrossRef]
- Berkowitz, J.; Ellison, G.B.; Gutman, D. Three methods to measure RH bond energies. J. Phys. Chem. 1994, 98, 2744–2765. [Google Scholar] [CrossRef]
- Guziec, F.S., Jr. Seleno- and telluro-carbonyl compounds. Chem. Org. Selenium Tellurium Compd. 1987, 2, 215–273. [Google Scholar]
- Gonzales, J.M.; Musaev, D.G.; Morokuma, K. Theoretical studies of oxidative addition of E–E bonds (E = S, Se, Te) to palladium(0) and platinum(0) complexes. Organometallics 2005, 24, 4908–4914. [Google Scholar] [CrossRef]
- Moore, C.E. Ionization Potentials and Ionization Limits Derived from the Analyses of Optical Spectra; National Bureau of Standards: Washington, DC, USA, 1970; p. 22. [Google Scholar]
- Hotop, H.; Lineberger, W.C. Binding energies in atomic negative ions. J. Phys. Chem. Ref. Data 1975, 4, 539–576. [Google Scholar] [CrossRef]
- Allred, A.L. Electronegativity values from thermochemical data. J. Inorg. Nucl. Chem. 1961, 17, 215–221. [Google Scholar] [CrossRef]
- Andersson, K.; Sadlej, A.J. Electric dipole polarizabilities of atomic valence states. Phys. Rev. A Atomic Mol. Opt. Phys. 1992, 46, 2356–2362. [Google Scholar] [CrossRef]
- Alpher, R.A.; White, D.R. Optical refractivity of high-temperature gases. I. Effects resulting from dissociation of diatomic gases. Phys. Fluids 1959, 2, 153–161. [Google Scholar] [CrossRef]
- Widmer, M.; Schwarzenbach, G. Acidity of the hydrosulfide ion HS. Helv. Chim. Acta 1964, 47, 266–271. [Google Scholar] [CrossRef]
- Hagisawa, H. The glass electrode and its applications. XII. Dissociation constants of hydrogen selenide. Rikagaku Kenkyusho Iho 1941, 20, 384–389. [Google Scholar]
- Levy, D.E.; Myers, R.J. Spectroscopic determination of the second dissociation constant of hydrogen selenide and the activity coefficients and spectral shifts of its ions. J. Phys. Chem. 1990, 94, 7842–7847. [Google Scholar] [CrossRef]
- Byun, B.J.; Kang, Y.K. Conformational preferences and PKA value of selenocysteine residue. Biopolymers 2011, 95, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Nagy, P.; Ashby, M.T. Reactive sulfur species: Kinetics and mechanisms of the oxidation of cysteine by hypohalous acid to give cysteine sulfenic acid. J. Am. Chem. Soc. 2007, 129, 14082–14091. [Google Scholar] [CrossRef] [PubMed]
- Tajc, S.G.; Tolbert, B.S.; Basavappa, R.; Miller, B.L. Direct determination of thiol PKA by isothermal titration microcalorimetry. J. Am. Chem. Soc. 2004, 126, 10508–10509. [Google Scholar] [CrossRef] [PubMed]
- Arnold, A.P.; Tan, K.S.; Rabenstein, D.L. Nuclear magnetic resonance studies of the solution chemistry of metal complexes. 23. Complexation of methylmercury by selenohydryl-containing amino acids and related molecules. Inorg. Chem. 1986, 25, 2433–2437. [Google Scholar] [CrossRef]
- Greig, I.R. The analysis of enzymic free energy relationships using kinetic and computational models. Chem. Soc. Rev. 2010, 39, 2272–2301. [Google Scholar] [CrossRef] [PubMed]
- Schmid, R.; Sapunov, V.N. Monographs in Modern Chemistry, Volume 14: Non-Formal Kinetics in Search for Chemical Reaction Pathways; Verlag Chemie: Weinheim, Germany, 1982; p. 199. [Google Scholar]
- Huber, R.; Criddle, R.S. Comparison of the chemical properties of selenocysteine and selenocystine with their sulfur analogs. Arch. Biochem. Biophys. 1967, 122, 164–173. [Google Scholar] [CrossRef]
- Steinmann, D.; Nauser, T.; Koppenol, W.H. Selenium and sulfur in exchange reactions: A comparative study. J. Org. Chem. 2010, 75, 6696–6699. [Google Scholar] [CrossRef] [PubMed]
- Seby, F.; Potin-Gautier, M.; Giffaut, E.; Borge, G.; Donard, O.F.X. A critical review of thermodynamic data for selenium species at 25 °C. Chem. Geol. 2001, 171, 173–194. [Google Scholar] [CrossRef]
- Frost, A.A. Oxidation potential-free energy diagrams. J. Am. Chem. Soc. 1951, 73, 2680–2682. [Google Scholar] [CrossRef]
- Black, J. Biological Performance of Materials: Fundamentals of Biocompatibility, 3rd ed.; Marcel Dekker: New York, NY, USA, 1999; p. 463. [Google Scholar]
- Yoshizawa, S.; Boeck, A. The many levels of control on bacterial selenoprotein synthesis. Biochim. Biophys. Acta Gen. Subj. 2009, 1790, 1404–1414. [Google Scholar] [CrossRef] [PubMed]
- Allmang, C.; Wurth, L.; Krol, A. The selenium to selenoprotein pathway in eukaryotes: More molecular partners than anticipated. Biochim. Biophys. Acta Gen. Subj. 2009, 1790, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Kryukov, G.V.; Castellano, S.; Novoselov, S.V.; Lobanov, A.V.; Zehtab, O.; Guigo, R.; Gladyshev, V.N. Characterization of mammalian selenoproteomes. Science 2003, 300, 1439–1443. [Google Scholar] [CrossRef] [PubMed]
- Raman, A.V.; Berry, M.J. Selenoproteins in cellular redox regulation and signaling. Oxid. Stress Vertebr. Invertebr. 2012, 195–208. [Google Scholar]
- Steinbrenner, H.; Sies, H. Protection against reactive oxygen species by selenoproteins. Biochim. Biophys. Acta Gen. Subj. 2009, 1790, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- MacFarquhar Jennifer, K.; Broussard Danielle, L.; Melstrom, P.; Hutchinson, R.; Wolkin, A.; Martin, C.; Burk Raymond, F.; Dunn John, R.; Green Alice, L.; Hammond, R.; et al. Acute selenium toxicity associated with a dietary supplement. Arch. Intern. Med. 2010, 170, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Puigdomenech, I. Hydra/medusa Chemical Equilibrium Database and Plotting Software. Available online: http://www.kth.se/che/medusa/ (accessed on 14 November 2016).
- Barnum, D.W. Potential-pH diagrams. J. Chem. Educ. 1982, 59, 809–812. [Google Scholar] [CrossRef]
- Campbell, J.A.; Whiteker, R.A. Periodic table based on potential-pH diagrams. J. Chem. Educ. 1969, 46, 90–92. [Google Scholar] [CrossRef]
- Delahay, P.; Pourbaix, M.; Van Rysselberghe, P. Potential-pH diagrams. J. Chem. Educ. 1950, 27, 683–688. [Google Scholar] [CrossRef]
- Pesterfield, L.L.; Maddox, J.B.; Crocker, M.S.; Schweitzer, G.K. Pourbaix (E-pH-M) diagrams in three dimensions. J. Chem. Educ. 2012, 89, 891–899. [Google Scholar] [CrossRef]
- Nuttall, K.L.; Allen, F.S. Kinetics of the reaction between hydrogen selenide ion and oxygen. Inorg. Chim. Acta 1984, 91, 243–246. [Google Scholar] [CrossRef]
- Kemp, M.; Go, Y.-M.; Jones, D.P. Nonequilibrium thermodynamics of thiol/disulfide redox systems: A perspective on redox systems biology. Free Radic. Biol. Med. 2008, 44, 921–937. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P.; Carlson, J.L.; Mody, V.C.; Cai, J.; Lynn, M.J.; Sternberg, P. Redox state of glutathione in human plasma. Free Radic. Biol. Med. 2000, 28, 625–635. [Google Scholar] [CrossRef]
- Painter, E.P. The chemistry and toxicity of selenium compounds, with special reference to the selenium problem. Chem. Rev. 1941, 28, 179–213. [Google Scholar] [CrossRef]
- Ganther, H.E. Selenotrisulfides. Formation by the reaction of thiols with selenious acid. Biochemistry 1968, 7, 2898–2905. [Google Scholar] [CrossRef] [PubMed]
- Ganther, H.E. Reduction of the selenotrisulfide derivative of glutathione to a persulfide analog by glutathione reductase. Biochemistry 1971, 10, 4089–4098. [Google Scholar] [CrossRef] [PubMed]
- Kice, J.L.; Lee, T.W.S.; Pan, S.-T. Mechanism of the reaction of thiols with selenite. J. Am. Chem. Soc. 1980, 102, 4448–4455. [Google Scholar] [CrossRef]
- Tarze, A.; Dauplais, M.; Grigoras, I.; Lazard, M.; Ha-Duong, N.-T.; Barbier, F.; Blanquet, S.; Plateau, P. Extracellular production of hydrogen selenide accounts for thiol-assisted toxicity of selenite against saccharomyces cerevisiae. J. Biol. Chem. 2007, 282, 8759–8767. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Jhee, K.-H.; Kruger, W.D. Production of the neuromodulator H2S by cystathionine β-synthase via the condensation of cysteine and homocysteine. J. Biol. Chem. 2004, 279, 52082–52086. [Google Scholar] [CrossRef] [PubMed]
- Chiku, T.; Padovani, D.; Zhu, W.; Singh, S.; Vitvitsky, V.; Banerjee, R. H2S biogenesis by human cystathionine γ-lyase leads to the novel sulfur metabolites lanthionine and homolanthionine and is responsive to the grade of hyperhomocysteinemia. J. Biol. Chem. 2009, 284, 11601–11612. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Toyofuku, Y.; Koike, S.; Shibuya, N.; Nagahara, N.; Lefer, D.; Ogasawara, Y.; Kimura, H. Identification of H2S3 and H2S produced by 3-mercaptopyruvate sulfurtransferase in the brain. Sci. Rep. 2015, 5, 14774. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, R.; Otsuguro, K.-I.; Yamaguchi, S.; Ito, S. Contribution of cysteine aminotransferase and mercaptopyruvate sulfurtransferase to hydrogen sulfide production in peripheral neurons. J. Neurochem. 2014, 130, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H. Hydrogen sulfide: Its production, release and functions. Amino Acids 2011, 41, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, Y.; Ishii, K.; Togawa, T.; Tanabe, S. Determination of bound sulfur in serum by gas dialysis/high-performance liquid chromatography. Anal. Biochem. 1993, 215, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Toohey, J.I. Sulphane sulphur in biological systems: A possible regulatory role. Biochem. J. 1989, 264, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Ishigami, M.; Hiraki, K.; Umemura, K.; Ogasawara, Y.; Ishii, K.; Kimura, H. A source of hydrogen sulfide and a mechanism of its release in the brain. Antioxid. Redox Signal. 2009, 11, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, Y.; Isoda, S.; Tanabe, S. Tissue and subcellular distribution of bound and acid-labile sulfur, and the enzymic capacity for sulfide production in the rat. Biol. Pharm. Bull. 1994, 17, 1535–1542. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Moore, P.K. Putative biological roles of hydrogen sulfide in health and disease: A breath of not so fresh air? Trends Pharmacol. Sci. 2008, 29, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Itoh, M.; Suzuki, K.T. Effects of dose on the methylation of selenium to monomethylselenol and trimethylselenonium ion in rats. Arch. Toxicol. 1997, 71, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Okuno, T.; Hasegawa, T.; Ueno, H.; Nakamuro, K. Study on detoxification and utilization of selenomethionine: A role of cystathionine γ-lyase in selenomethionine metabolism. Biomed. Res. Trace Elem. 2007, 18, 221–230. [Google Scholar]
- Ohta, Y.; Suzuki, K.T. Methylation and demethylation of intermediates selenide and methylselenol in the metabolism of selenium. Toxicol. Appl. Pharmacol. 2008, 226, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Lisk, D.; Block, E.; Ip, C. Characterization of the biological activity of γ-glutamyl-se-methylselenocysteine: A novel, naturally occurring anticancer agent from garlic. Cancer Res. 2001, 61, 2923–2928. [Google Scholar] [PubMed]
- Andreadou, I.; Menge, W.M.P.B.; Commandeur, J.N.M.; Worthington, E.A.; Vermeulen, N.P.E. Synthesis of novel Se-substituted selenocysteine derivatives as potential kidney selective prodrugs of biologically active selenol compounds: Evaluation of kinetics of β-elimination reactions in rat renal cytosol. J. Med. Chem. 1996, 39, 2040–2046. [Google Scholar] [CrossRef] [PubMed]
- Esaki, N.; Nakamura, T.; Tanaka, H.; Soda, K. Selenocysteine lyase, a novel enzyme that specifically acts on selenocysteine. Mammalian distribution and purification and properties of pig liver enzyme. J. Biol. Chem. 1982, 257, 4386–4391. [Google Scholar] [PubMed]
- Lu, J.; Berndt, C.; Holmgren, A. Metabolism of selenium compounds catalyzed by the mammalian selenoprotein thioredoxin reductase. Biochim. Biophys. Acta Gen. Subj. 2009, 1790, 1513–1519. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, H.S.; Ganther, H.E. Biosynthesis of dimethyl selenide from sodium selenite in rat liver and kidney cell-free systems. Biochim. Biophys. Acta Gen. Subj. 1977, 497, 205–217. [Google Scholar] [CrossRef]
- Wolfe, M.D. Selenophosphate synthetase. EcoSal Plus 2014, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Francesconi Kevin, A.; Pannier, F. Selenium metabolites in urine: A critical overview of past work and current status. Clin. Chem. 2004, 50, 2240–2253. [Google Scholar] [CrossRef] [PubMed]
- Kremer, D.; Ilgen, G.; Feldmann, J. GC-ICP-MS determination of dimethylselenide in human breath after ingestion of 77Se-enriched selenite: Monitoring of in vivo methylation of selenium. Anal. Bioanal. Chem. 2005, 383, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Lajin, B.; Kuehnelt, D.; Francesconi, K.A. Exploring the urinary selenometabolome following a multi-phase selenite administration regimen in humans. Metallomics Integr. Biomet. Sci. 2016, 8, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, T.C. Selenocysteine lyase. EcoSal Plus 2014, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Veres, Z.; Tsai, L.; Scholz, T.D.; Politino, M.; Balaban, R.S.; Stadtman, T.C. Synthesis of 5-methylaminomethyl-2-selenouridine in tRNAs: Phosphorus-31 NMR studies show the labile selenium donor synthesized by the seld gene product contains selenium bonded to phosphorus. Proc. Natl. Acad. Sci. USA 1992, 89, 2975–2979. [Google Scholar] [CrossRef] [PubMed]
- Glass, R.S.; Singh, W.P.; Jung, W.; Veres, Z.; Scholz, T.D.; Stadtman, T. Monoselenophosphate: Synthesis, characterization, and identity with the prokaryotic biological selenium donor, compound sepx. Biochemistry 1993, 32, 12555–12559. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.P.; Wallenberg, M.; Gandin, V.; Misra, S.; Tisato, F.; Marzano, C.; Rigobello, M.P.; Kumar, S.; Bjoernstedt, M. Methylselenol formed by spontaneous methylation of selenide is a superior selenium substrate to the thioredoxin and glutaredoxin systems. PLoS ONE 2012, 7, e50727. [Google Scholar] [CrossRef] [PubMed]
- Stadtman, T.C. Selenocysteine. Ann. Rev. Biochem. 1996, 65, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Anan, Y.; Kimura, M.; Hayashi, M.; Koike, R.; Ogra, Y. Detoxification of selenite to form selenocyanate in mammalian cells. Chem. Res. Toxicol. 2015, 28, 1803–1814. [Google Scholar] [CrossRef] [PubMed]
- Lacourciere, G.M. Selenium is mobilized in vivo from free selenocysteine and is incorporated specifically into formate dehydrogenase H and tRNA nucleosides. J. Bacteriol. 2002, 184, 1940–1946. [Google Scholar] [CrossRef] [PubMed]
- Lacourciere, G.M.; Stadtman, T.C. Utilization of selenocysteine as a source of selenium for selenophosphate biosynthesis. BioFactors 2001, 14, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Chocat, P.; Esaki, N.; Tanizawa, K.; Nakamura, K.; Tanaka, H.; Soda, K. Purification and characterization of selenocysteine beta-lyase from citrobacter freundii. J. Bacteriol. 1985, 163, 669–676. [Google Scholar] [PubMed]
- Ganther, H.E. Enzymic synthesis of dimethyl selenide from sodium selenite in mouse liver extracts. Biochemistry 1966, 5, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.T.; Kurasaki, K.; Suzuki, N. Selenocysteine β-lyase and methylselenol demethylase in the metabolism of Se-methylated selenocompounds into selenide. Biochim. Biophys. Acta Gen. Subj. 2007, 1770, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.T.; Kurasaki, K.; Ogawa, S.; Suzuki, N. Metabolic transformation of methylseleninic acid through key selenium intermediate selenide. Toxicol. Appl. Pharmacol. 2006, 215, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.T.; Doi, C.; Suzuki, N. Metabolism of 76Se-methylselenocysteine compared with that of 77Se-selenomethionine and 82Se-selenite. Toxicol. Appl. Pharmacol. 2006, 217, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Ogra, Y.; Ishiwata, K.; Takayama, H.; Aimi, N.; Suzuki, K.T. Selenosugars are key and urinary metabolites for selenium excretion within the required to low-toxic range. Proc. Natl. Acad. Sci. USA 2002, 99, 15932–15936. [Google Scholar] [CrossRef] [PubMed]
- Ogra, Y. Selenium metabolism. In Diversity of Selenium Functions in Health and Disease; Regina, B.-F., Sies, H., Eds.; CRC Press: Boca Raton, FL, USA, 2016; pp. 19–30. [Google Scholar]
- Weekley, C.M.; Harris, H.H. Which form is that? The importance of selenium speciation and metabolism in the prevention and treatment of disease. Chem. Soc. Rev. 2013, 42, 8870–8894. [Google Scholar] [CrossRef] [PubMed]
- Rahmanto Aldwin, S.; Davies Michael, J. Selenium-containing amino acids as direct and indirect antioxidants. IUBMB Life 2012, 64, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Rahmanto, A.S.; Davies, M.J. Catalytic activity of selenomethionine in removing amino acid, peptide, and protein hydroperoxides. Free Radic. Biol. Med. 2011, 51, 2288–2299. [Google Scholar] [CrossRef] [PubMed]
- Krause, R.J.; Elfarra, A.A. Reduction of l-methionine selenoxide to seleno-l-methionine by endogenous thiols, ascorbic acid, or methimazole. Biochem. Pharmacol. 2009, 77, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Nagy, P.; Becker, J.D.; Mallo, R.C.; Ashby, M.T. The jekyll and hyde roles of cysteine derivatives during oxidative stress. ACS Symp. Ser. 2007, 967, 193–212. [Google Scholar]
- Buettner, G.R. The pecking order of free radicals and antioxidants: Lipid peroxidation, alpha-tocopherol, and ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, G.V.; Muzyka, N.G.; Oktyabrsky, O.N. Effects of cystine and hydrogen peroxide on glutathione status and expression of antioxidant genes in escherichia coli. Biochemistry 2005, 70, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Imlay, J.A. High levels of intracellular cysteine promote oxidative DNA damage by driving the fenton reaction. J. Bacteriol. 2003, 185, 1942–1950. [Google Scholar] [CrossRef] [PubMed]
- Berglin, E.H.; Edlund, M.B.K.; Nyberg, G.K.; Carlsson, J. Potentiation by l-cysteine of the bactericidal effect of hydrogen peroxide in escherichia coli. J. Bacteriol. 1982, 152, 81–88. [Google Scholar] [PubMed]
- Singh, R.; Whitesides, G.M. Selenols catalyze the interchange reactions of dithiols and disulfides in water. J. Org. Chem. 1991, 56, 6931–6933. [Google Scholar] [CrossRef]
- Hafeman, D.G.; Sunde, R.A.; Hoekstra, W.G. Effect of dietary selenium on erythrocyte and liver glutathione peroxidase in the rat. J. Nutr. 1974, 104, 580–587. [Google Scholar] [PubMed]
- Csallany, A.S.; Menken, B.Z. Effect of dietary selenite on hepatic organic solvent-soluble lipofuscin pigments. J. Am. Coll. Toxicol. 1986, 5, 79–85. [Google Scholar] [CrossRef]
- Spallholz, J.E. Free radical generation by selenium compounds and their prooxidant toxicity. Biomed. Environ. Sci. 1997, 10, 260–270. [Google Scholar] [PubMed]
- Terada, A.; Yoshida, M.; Seko, Y.; Kobayashi, T.; Yoshida, K.; Nakada, M.; Nakada, K.; Echizen, H.; Ogata, H.; Rikihisa, T. Active oxygen species generation and cellular damage by additives of parenteral preparations: Selenium and sulfhydryl compounds. Nutrition 1999, 15, 651–655. [Google Scholar] [CrossRef]
- Raisbeck, M.F. Selenosis. Vet. Clin. N. Am. Food Anim. Pract. 2000, 16, 465–480. [Google Scholar] [CrossRef]
- Spallholz, J.E. On the nature of selenium toxicity and carcinostatic activity. Free Radic Biol Med 1994, 17, 45–64. [Google Scholar] [CrossRef]
- Bierla, K.; Bianga, J.; Ouerdane, L.; Szpunar, J.; Yiannikouris, A.; Lobinski, R. A comparative study of the Se/S substitution in methionine and cysteine in Se-enriched yeast using an inductively coupled plasma mass spectrometry (ICP MS)-assisted proteomics approach. J. Proteom. 2013, 87, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Ip, C. Lessons from basic research in selenium and cancer prevention. J. Nutr. 1998, 128, 1845–1854. [Google Scholar] [PubMed]
- Combs, G.F., Jr.; Gray, W.P. Chemopreventive agents: Selenium. Pharmacol. Ther. 1998, 79, 179–192. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium in cancer prevention: A review of the evidence and mechanism of action. Proc. Nutr. Soc. 2005, 64, 527–542. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Combs, G.F., Jr. Selenium as an anticancer nutrient: Roles in cell proliferation and tumor cell invasion. J. Nutr. Biochem. 2008, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Schrauzer, G.N. Anticarcinogenic effects of selenium. Cell. Mol. Life Sci. 2000, 57, 1864–1873. [Google Scholar] [CrossRef] [PubMed]
- Weekley, C.M.; Jeong, G.; Tierney, M.E.; Hossain, F.; Maw, A.M.; Shanu, A.; Harris, H.H.; Witting, P.K. Selenite-mediated production of superoxide radical anions in A549 cancer cells is accompanied by a selective increase in SOD1 concentration, enhanced apoptosis and Se-Cu bonding. J. Biol. Inorg. Chem. 2014, 19, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-J.; Boylan, L.M.; Wu, C.-K.; Spallholz, J.E. Oxidation of glutathione and superoxide generation by inorganic and organic selenium compounds. BioFactors 2007, 31, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.-M.; Yang, C.-F.; Ong, C.-N. Sodium selenite-induced oxidative stress and apoptosis in human hepatoma HepG2 cells. Int. J. Cancer 1999, 81, 820–828. [Google Scholar] [CrossRef]
- Stewart, M.S.; Spallholz, J.E.; Neldner, K.H.; Pence, B.C. Selenium compounds have disparate abilities to impose oxidative stress and induce apoptosis. Free Radic. Biol. Med. 1998, 26, 42–48. [Google Scholar] [CrossRef]
- Kim, E.H.; Sohn, S.; Kwon, H.J.; Kim, S.U.; Kim, M.-J.; Lee, S.-J.; Choi, K.S. Sodium selenite induces superoxide-mediated mitochondrial damage and subsequent autophagic cell death in malignant glioma cells. Cancer Res. 2007, 67, 6314–6324. [Google Scholar] [CrossRef] [PubMed]
- Zhou, N.; Xiao, H.; Li, T.-K.; Nur-E-Kamal, A.; Liu, L.F. DNA damage-mediated apoptosis induced by selenium compounds. J. Biol. Chem. 2003, 278, 29532–29537. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.J.; Wilson, A.; Lu, J.; Singh, M.; Jiang, C.; Upadhyaya, P.; El-Bayoumy, K.; Ip, C. Comparison of the effects of an organic and an inorganic form of selenium on a mammary carcinoma cell line. Carcinogenesis 1994, 15, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Drake, E.N. Cancer chemoprevention: Selenium as a prooxidant, not an antioxidant. Med. Hypotheses 2006, 67, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.M.; Yang, C.F.; Ding, W.X.; Liu, J.; Ong, C.N. Superoxide radical-initiated apoptotic signalling pathway in selenite-treated HepG2 cells: Mitochondria serve as the main target. Free Radic. Biol. Med. 2001, 30, 9–21. [Google Scholar] [CrossRef]
- Shen, H.M.; Yang, C.F.; Liu, J.; Ong, C.N. Dual role of glutathione in selenite-induced oxidative stress and apoptosis in human hepatoma cells. Free Radic. Biol. Med. 2000, 28, 1115–1124. [Google Scholar] [CrossRef]
- Fisher, B.; Yarmolinsky, D.; Abdel-Ghany, S.; Pilon, M.; Pilon-Smits, E.A.; Sagi, M.; Van Hoewyk, D. Superoxide generated from the glutathione-mediated reduction of selenite damages the iron-sulfur cluster of chloroplastic ferredoxin. Plant Physiol. Biochem. 2016, 106, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Seko, Y.; Saito, Y.; Kitahara, J.; Imura, N. Active oxygen generation by the reaction of selenite with reduced glutathione in vitro. In Selenium in Biology and Medicine; Springer: Berlin, Germany, 1989; pp. 70–73. [Google Scholar]
- Pilon, M.; Owen Jennifer, D.; Garifullina Gulnara, F.; Kurihara, T.; Mihara, H.; Esaki, N.; Pilon-Smits Elizabeth, A.H. Enhanced selenium tolerance and accumulation in transgenic arabidopsis expressing a mouse selenocysteine lyase. Plant Physiol. 2003, 131, 1250–1257. [Google Scholar] [CrossRef] [PubMed]
- Kessi, J.; Hanselmann, K.W. Similarities between the abiotic reduction of selenite with glutathione and the dissimilatory reaction mediated by rhodospirillum rubrum and escherichia coli. J. Biol. Chem. 2004, 279, 50662–50669. [Google Scholar] [CrossRef] [PubMed]
- Peyroche, G.; Saveanu, C.; Dauplais, M.; Lazard, M.; Beuneu, F.; Decourty, L.; Malabat, C.; Jacquier, A.; Blanquet, S.; Plateau, P. Sodium selenide toxicity is mediated by O2-dependent DNA breaks. PLoS ONE 2012, 7, e36343. [Google Scholar] [CrossRef] [PubMed]
- Herrero, E.; Weilinger, R.E. Yeast as a model system to study metabolic impact of selenium compounds. Microb. Cell 2015, 2, 139–149. [Google Scholar] [CrossRef]
- Bockhorn, J.; Balar, B.; He, D.; Seitomer, E.; Copeland, P.R.; Kinzy, T.G. Genome-wide screen of saccharomyces cerevisiae null allele strains identifies genes involved in selenomethionine resistance. Proc. Natl. Acad. Sci. USA 2008, 105, 17682–17687. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.; Moulis, J.M.; Gaillard, J.; Lutz, M. Replacement of sulfur by selenium in iron-sulfur proteins. Adv. Inorg. Chem. 1992, 38, 73–115. [Google Scholar]
- Spatzal, T.; Rees Douglas, C.; Spatzal, T.; Perez Kathryn, A.; Howard James, B.; Rees Douglas, C.; Howard James, B. Catalysis-dependent selenium incorporation and migration in the nitrogenase active site iron-molybdenum cofactor. eLife 2015, 4, e11620. [Google Scholar] [CrossRef] [PubMed]
- Wrobel, M.; Jurkowska, H.; Sliwa, L.; Srebro, Z. Sulfurtransferases and cyanide detoxification in mouse liver, kidney, and brain. Toxicol. Mech. Methods 2004, 14, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Bordo, D.; Bork, P. The rhodanese/Cdc25 phosphatase superfamily. Sequence-structure-function relations. EMBO Rep. 2002, 3, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Westley, J. Rhodanese. Adv. Enzymol. Relat. Areas Mol. Biol. 1973, 39, 327–368. [Google Scholar] [PubMed]
- Mueller, E.G. Trafficking in persulfides: Delivering sulfur in biosynthetic pathways. Nat. Chem. Biol. 2006, 2, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Weuffen, W.; Franzke, C.; Thuerkow, B. Alimentary ingestion, analysis and biological significance of thiocyanate. Nahrung 1984, 28, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Moir, D.; Rickert, W.S.; Levasseur, G.; Larose, Y.; Maertens, R.; White, P.; Desjardins, S. A comparison of mainstream and sidestream marijuana and tobacco cigarette smoke produced under two machine smoking conditions. Chem. Res. Toxicol. 2008, 21, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Zgliczynski, J.M.; Stelmaszynska, T. Hydrogen cyanide and cyanogen chloride formation by the myeloperoxidase hydrogen peroxide-chloride ion system. Biochim. Biophys. Acta Enzymol. 1979, 567, 309–314. [Google Scholar] [CrossRef]
- Pessi, G.; Haas, D. Transcriptional control of the hydrogen cyanide biosynthetic genes hcnABC by the anaerobic regulator ANR and the quorum-sensing regulators LasR and RhlR in pseudomonas aeruginosa. J. Bacteriol. 2000, 182, 6940–6949. [Google Scholar] [CrossRef] [PubMed]
- Ryall, B.; Davies, J.C.; Wilson, R.; Shoemark, A.; Williams, H.D. Pseudomonas aeruginosa, cyanide accumulation and lung function in CF and non-CF bronchiectasis patients. Eur. Respir. J. 2008, 32, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Broderick, K.E.; Chan, A.; Balasubramanian, M.; Feala, J.; Reed, S.L.; Panda, M.; Sharma, V.S.; Pilz, R.B.; Bigby, T.D.; Boss, G.R. Cyanide produced by human isolates of pseudomonas aeruginosa contributes to lethality in drosophila melanogaster. J. Infect. Dis. 2007, 197, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Askeland, R.A.; Morrison, S.M. Cyanide production by pseudomonas fluorescens and pseudomonas aeruginosa. Appl. Environ. Microbiol. 1983, 45, 1802–1807. [Google Scholar] [PubMed]
- Conner, G.E.; Wijkstrom-Frei, C.; Randell, S.H.; Fernandez, V.E.; Salathe, M. The lactoperoxidase system links anion transport to host defense in cystic fibrosis. FEBS Lett. 2007, 581, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Fragoso, M.A.; Fernandez, V.; Forteza, R.; Randell, S.H.; Salathe, M.; Conner, G.E. Transcellular thiocyanate transport by human airway epithelia. J. Physiol. 2004, 561, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Eskandari, S.; Loo, D.D.F.; Dai, G.; Levy, O.; Wright, E.M.; Carrasco, N. Thyroid NA+/I− symporter. Mechanism, stoichiometry, and specificity. J. Biol. Chem. 1997, 272, 27230–27238. [Google Scholar] [CrossRef] [PubMed]
- Wolff, J.; Maurey, J.R. Thyroidal iodide transport. IX. The accumulation and metabolism of selenocyanate. Endocrinology 1966, 79, 795–800. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-L.; Zhao, Y.-B. CdS quantum dots as fluorescence probes for the sensitive and selective detection of highly reactive HSe− ions in aqueous solution. Anal. Bioanal. Chem. 2007, 388, 717–722. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).