Unawareness of Apathy in Parkinson’s Disease: The Role of Executive Dysfunction on Symptom Recognition
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Neuropsychological Evaluation
2.2.1. Apathy
2.2.2. Cognitive Evaluation
2.3. Statistical Analysis
3. Results
3.1. Agreement between Patients’ Self-Report Apathy Scores and Caregivers’ Ratings
3.2. Clinical Predictors of Discrepancy between Patients’ Self-Report Apathy Scores and Caregivers’ Ratings
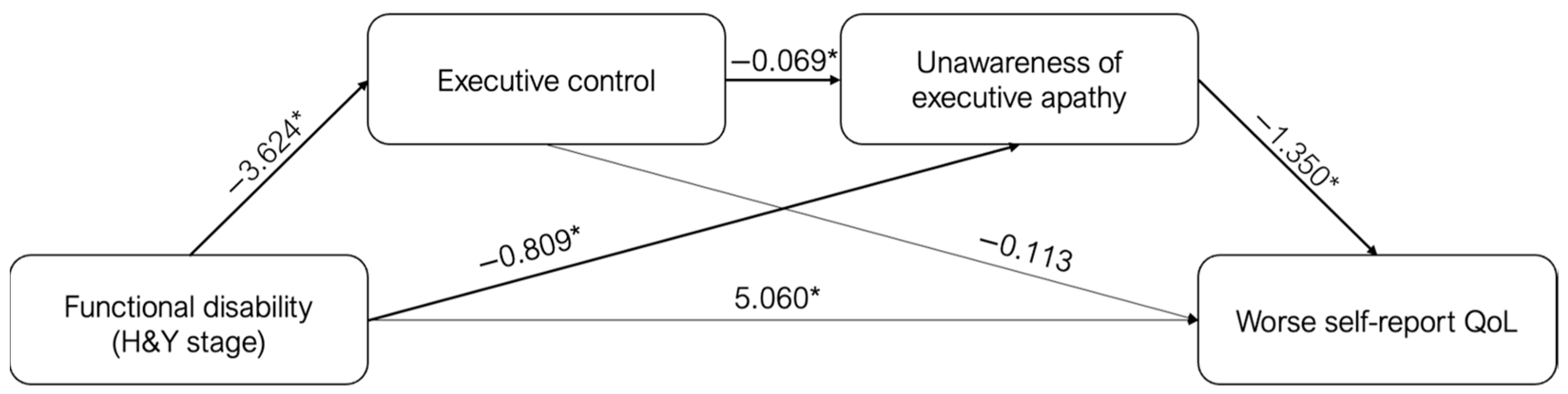
3.3. Effect of Discrepancy on Self-Report Quality of Life
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rosen, H.J. Anosognosia in Neurodegenerative Disease. Neurocase 2011, 17, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Prigatano, G.P. Anosognosia: Clinical and Ethical Considerations. Curr. Opin. Neurol. 2009, 22, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Shany-Ur, T.; Lin, N.; Rosen, H.J.; Sollberger, M.; Miller, B.L.; Rankin, K.P. Self-Awareness in Neurodegenerative Disease Relies on Neural Structures Mediating Reward-Driven Attention. Brain 2014, 137, 2368–2381. [Google Scholar] [CrossRef]
- Gray, J.R.; Braver, T.S.; Raichle, M.E. Integration of Emotion and Cognition in the Lateral Prefrontal Cortex. Proc. Natl. Acad. Sci. USA 2002, 99, 4115–4120. [Google Scholar] [CrossRef] [PubMed]
- Toglia, J.; Kirk, U. Understanding Awareness Deficits Following Brain Injury. NeuroRehabilitation 2000, 15, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Rosen, H.J.; Alcantar, O.; Zakrzewski, J.; Shimamura, A.P.; Neuhaus, J.; Miller, B.L. Metacognition in the Behavioral Variant of Frontotemporal Dementia and Alzheimer’s Disease. Neuropsychology 2014, 28, 436. [Google Scholar] [CrossRef] [PubMed]
- Maier, F.; Prigatano, G.P. Impaired Self-Awareness of Motor Disturbances in Parkinson’s Disease. Arch. Clin. Neuropsychol. 2017, 32, 802–809. [Google Scholar] [CrossRef]
- Pietracupa, S.; Latorre, A.; Berardelli, A.; Fabbrini, G. Parkinsonian Patients and Poor Awareness of Dyskinesias. Front. Neurol. 2014, 5, 32. [Google Scholar] [CrossRef]
- Pennington, C.; Duncan, G.; Ritchie, C. Altered Awareness of Cognitive and Neuropsychiatric Symptoms in Parkinson’s Disease and Dementia with Lewy Bodies: A Systematic Review. Int. J. Geriatr. Psychiatry 2021, 36, 15–30. [Google Scholar] [CrossRef]
- Marin, R.S. Apathy: A Neuropsychiatric Syndrome. J. Neuropsychiatry Clin. Neurosci. 1991, 3, 243–254. [Google Scholar]
- Santangelo, G.; D’Iorio, A.; Maggi, G.; Cuoco, S.; Pellecchia, M.T.; Amboni, M.; Barone, P.; Vitale, C. Cognitive Correlates of “Pure Apathy” in Parkinson’s Disease. Park. Relat. Disord. 2018, 53, 101–104. [Google Scholar] [CrossRef] [PubMed]
- D’Iorio, A.; Maggi, G.; Vitale, C.; Trojano, L.; Santangelo, G. “Pure Apathy” and Cognitive Dysfunctions in Parkinson’s Disease: A Meta-Analytic Study. Neurosci. Biobehav. Rev. 2018, 94, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.-S.; Lee, J.E.; Lee, P.H.; Kim, J.-S. Neuropsychiatric Symptoms in Parkinson’s Disease Dementia Are Associated with Increased Caregiver Burden. J. Mov. Disord. 2015, 8, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.; Dubois, B. Apathy and the Functional Anatomy of the Prefrontal Cortex-Basal Ganglia Circuits. Cerebral Cortex 2006, 16, 916–928. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.S.; Chung, S.J.; Lee, Y.H.; Ye, B.S.; Sohn, Y.H.; Lee, P.H. Cognitive Anosognosia Is Associated with Frontal Dysfunction and Lower Depression in Parkinson’s Disease. Eur. J. Neurol. 2020, 27, 951–958. [Google Scholar] [CrossRef]
- Maggi, G.; Cima Muñoz, A.M.; Obeso, I.; Santangelo, G. Neuropsychological, neuropsychiatric, and clinical correlates of affective and cognitive theory of mind in Parkinson’s disease: A meta-analysis. Neuropsychology 2022, 36, 483–504. [Google Scholar] [CrossRef]
- Maggi, G.; Di Meglio, D.; Vitale, C.; Amboni, M.; Obeso, I.; Santangelo, G. The impact of executive dysfunctions on Theory of Mind abilities in Parkinson’s disease. Neuropsychologia 2022, 176, 108389. [Google Scholar] [CrossRef]
- Mathias, J.L. Neurobehavioral Functioning of Persons with Parkinson’s Disease. Appl. Neuropsychol. 2003, 10, 57–68. [Google Scholar] [CrossRef]
- Valentino, V.; Iavarone, A.; Amboni, M.; Moschiano, F.; Picillo, M.; Petretta, V.; Cicarelli, G. Apathy in Parkinson’s Disease: Differences between Caregiver’s Report and Self-Evaluation. Funct. Neurol. 2018, 33, 31–35. [Google Scholar] [CrossRef]
- Schiehser, D.M.; Liu, L.; Lessig, S.L.; Song, D.D.; Obtera, K.M.; Burke, M.M.; Earl, S.R.; Vincent Filoteo, J. Predictors of Discrepancies in Parkinson’s Disease Patient and Caregiver Ratings of Apathy, Disinhibition, and Executive Dysfunction before and after Diagnosis. J. Int. Neuropsychol. Soc. 2013, 19, 295–304. [Google Scholar] [CrossRef]
- Santangelo, G.; Siciliano, M.; Pedone, R.; Vitale, C.; Falco, F.; Bisogno, R.; Siano, P.; Barone, P.; Grossi, D.; Santangelo, F.; et al. Normative Data for the Montreal Cognitive Assessment in an Italian Population Sample. Neurol. Sci. 2015, 36, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Maggi, G.; D’Iorio, A.; Aiello, E.N.; Poletti, B.; Ticozzi, N.; Silani, V.; Amboni, M.; Vitale, C.; Santangelo, G. Psychometrics and Diagnostics of the Italian Version of the Beck Depression Inventory-II (BDI-II) in Parkinson’s Disease. Neurol. Sci. 2023, 44, 1607–1612. [Google Scholar] [CrossRef] [PubMed]
- Radakovic, R.; Abrahams, S. Developing a New Apathy Measurement Scale: Dimensional Apathy Scale. Psychiatry Res. 2014, 219, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Santangelo, G.; Raimo, S.; Siciliano, M.; D’Iorio, A.; Piscopo, F.; Cuoco, S.; Bottone, M.; Trojsi, F.; Grossi, D.; Trojano, L. Assessment of Apathy Independent of Physical Disability: Validation of the Dimensional Apathy Scale in Italian Healthy Sample. Neurol. Sci. 2017, 38, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Santangelo, G.; D’Iorio, A.; Piscopo, F.; Cuoco, S.; Longo, K.; Amboni, M.; Baiano, C.; Tafuri, D.; Pellecchia, M.T.; Barone, P.; et al. Assessment of Apathy Minimising the Effect of Motor Dysfunctions in Parkinson’s Disease: A Validation Study of the Dimensional Apathy Scale. Qual. Life Res. 2017, 26, 2533–2540. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive Functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Enticott, P.G.; Ogloff, J.R.P. Elucidation of Impulsivity. Aust. Psychol. 2006, 41, 3–14. [Google Scholar] [CrossRef]
- Friedman, N.P.; Miyake, A. The Relations Among Inhibition and Interference Control Functions: A Latent-Variable Analysis. J. Exp. Psychol. Gen. 2004, 133, 101–135. [Google Scholar] [CrossRef]
- Landis, J.R.; Koch, G.G. The Measurement of Observer Agreement for Categorical Data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef]
- Preacher, K.J.; Hayes, A.F. Asymptotic and Resampling Strategies for Assessing and Comparing Indirect Effects in Multiple Mediator Models. Behav. Res. Methods 2008, 40, 879–891. [Google Scholar] [CrossRef]
- Alexander, G.E.; DeLong, M.R.; Strick, P.L. Parallel Organization of Functionally Segregated Circuits Linking Basal Ganglia and Cortex. Annu. Rev. Neurosci. 1986, 9, 357–381. [Google Scholar] [CrossRef] [PubMed]
- Masina, F.; Vallesi, A.; di Rosa, E.; Semenzato, L.; Mapelli, D. Possible Role of Dorsolateral Prefrontal Cortex in Error Awareness: Single-Pulse TMS Evidence. Front. Neurosci. 2018, 12, 179. [Google Scholar] [CrossRef] [PubMed]
- Magno, E.; Foxe, J.J.; Molholm, S.; Robertson, I.H.; Garavan, H. The Anterior Cingulate and Error Avoidance. J. Neurosci. 2006, 26, 4769–4773. [Google Scholar] [CrossRef] [PubMed]
- Carter, C.S.; van Veen, V. Anterior Cingulate Cortex and Conflict Detection: An Update of Theory and Data. Cogn. Affect Behav. Neurosci. 2007, 26, 4769–4773. [Google Scholar] [CrossRef]
- Carter, C.S.; Braver, T.S.; Barch, D.M.; Botvinick, M.M.; Noll, D.; Cohen, J.D. Anterior Cingulate Cortex, Error Detection, and the Online Monitoring of Performance. Science 1998, 280, 747–749. [Google Scholar] [CrossRef] [PubMed]
- Mograbi, D.C.; Brown, R.G.; Salas, C.; Morris, R.G. Emotional Reactivity and Awareness of Task Performance in Alzheimer’s Disease. Neuropsychologia 2012, 50, 2075–2084. [Google Scholar] [CrossRef]
- Mograbi, D.C.; Morris, R.G. On the Relation among Mood, Apathy, and Anosognosia in Alzheimer’s Disease. J. Int. Neuropsychol. Soc. 2014, 20, 2–7. [Google Scholar] [CrossRef]
- Radakovic, R.; Starr, J.M.; Abrahams, S. A Novel Assessment and Profiling of Multidimensional Apathy in Alzheimer’s Disease. J. Alzheimer’s Dis. 2017, 60, 57–67. [Google Scholar] [CrossRef]
- Vitale, C.; Pellecchia, M.T.; Grossi, D.; Fragassi, N.; Cuomo, T.; di Maio, L.; Barone, P. Unawareness of Dyskinesias in Parkinson’s and Huntington’s Diseases. Neurol. Sci. 2001, 22, 105–106. [Google Scholar] [CrossRef]
- Schrag, A.; Quinn, N. Dyskinesias and Motor Fluctuations in Parkinson’s Disease: A Community-Based Study. Brain 2000, 123, 2297–2305. [Google Scholar] [CrossRef]
- Amanzio, M.; Bartoli, M.; Cipriani, G.E.; Palermo, S. Executive Dysfunction and Reduced Self-Awareness in Patients With Neurological Disorders. A Mini-Review. Front. Psychol. 2020, 11, 1697. [Google Scholar] [CrossRef] [PubMed]
- Ownsworth, T.; Fleming, J.; Strong, J.; Radel, M.; Chan, W.; Clare, L. Awareness Typologies, Long-Term Emotional Adjustment and Psychosocial Outcomes Following Acquired Brain Injury. Neuropsychol. Rehabil. 2007, 17, 129–150. [Google Scholar] [CrossRef] [PubMed]
- Haber, S.N. Corticostriatal Circuitry. Dialogues Clin. Neurosci. 2016, 18, 21. [Google Scholar] [CrossRef] [PubMed]
- Kwak, Y.; Peltier, S.; Bohnen, N.I.; Müller, M.L.T.M.; Dayalu, P.; Seidler, R.D. Altered Resting State Cortico-Striatal Connectivity in Mild to Moderate Stage Parkinson’s Disease. Front. Syst. Neurosci. 2010, 4, 143. [Google Scholar] [CrossRef] [PubMed]
- Fitts, W.; Massimo, L.; Lim, N.; Grossman, M.; Dahodwala, N. Computerized Assessment of Goal-Directed Behavior in Parkinson’s Disease. J. Clin. Exp. Neuropsychol. 2016, 38, 1015–1025. [Google Scholar] [CrossRef]
- Esposito, A.; Raimo, G.; Maldonato, M.; Vogel, C.; Conson, M.; Cordasco, G. Behavioral Sentiment Analysis of Depressive States. In Proceedings of the 11th IEEE International Conference on Cognitive Infocommunications, CogInfoCom 2020—Proceedings; 2020, Mariehamn, Finland, 23–25 September 2020. [Google Scholar]

| Mean ± SD | |
|---|---|
| Age (ys) | 66.06 ± 8.03 |
| Education (ys) | 11.54 ± 4.48 |
| Gender (n) | M = 44; F = 23 |
| Disease Duration (ys) | 9.57 ± 5.93 |
| UPDRS-III | 14.84 ± 8.60 |
| Hoehn and Yahr | 2.35 ± 0.63 |
| LEDD | 732.21 ± 413.17 |
| MoCA | 19.82 ± 4.47 |
| BDI-II | 7.28 ± 6.79 |
| TMT: A | 73.88 ± 50.94 |
| TMT: B-A | 179.67 ± 124.90 |
| Phonological fluency | 28.64 ± 10.82 |
| Stroop Test-Color | 33.87 ± 12.79 |
| Stroop Test-Interference | 12.85 ± 8.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maggi, G.; Vitale, C.; Delle Curti, A.; Amboni, M.; Santangelo, G. Unawareness of Apathy in Parkinson’s Disease: The Role of Executive Dysfunction on Symptom Recognition. Brain Sci. 2023, 13, 964. https://doi.org/10.3390/brainsci13060964
Maggi G, Vitale C, Delle Curti A, Amboni M, Santangelo G. Unawareness of Apathy in Parkinson’s Disease: The Role of Executive Dysfunction on Symptom Recognition. Brain Sciences. 2023; 13(6):964. https://doi.org/10.3390/brainsci13060964
Chicago/Turabian StyleMaggi, Gianpaolo, Carmine Vitale, Alessia Delle Curti, Marianna Amboni, and Gabriella Santangelo. 2023. "Unawareness of Apathy in Parkinson’s Disease: The Role of Executive Dysfunction on Symptom Recognition" Brain Sciences 13, no. 6: 964. https://doi.org/10.3390/brainsci13060964
APA StyleMaggi, G., Vitale, C., Delle Curti, A., Amboni, M., & Santangelo, G. (2023). Unawareness of Apathy in Parkinson’s Disease: The Role of Executive Dysfunction on Symptom Recognition. Brain Sciences, 13(6), 964. https://doi.org/10.3390/brainsci13060964






