The Technological Design of Geometrically Complex Ti-6Al-4V Parts by Metal Injection Molding
Abstract
:1. Introduction
2. Materials and Methods
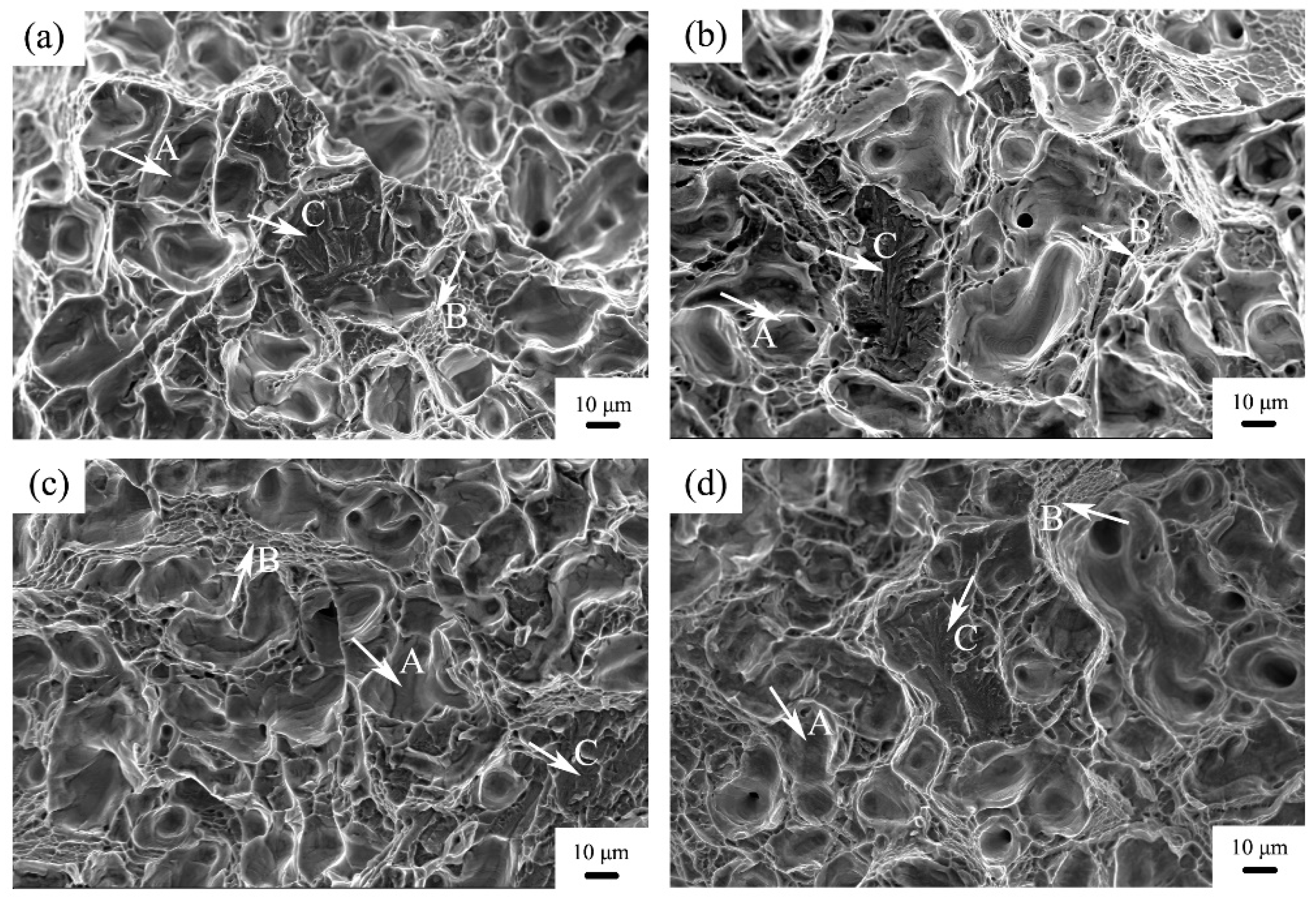
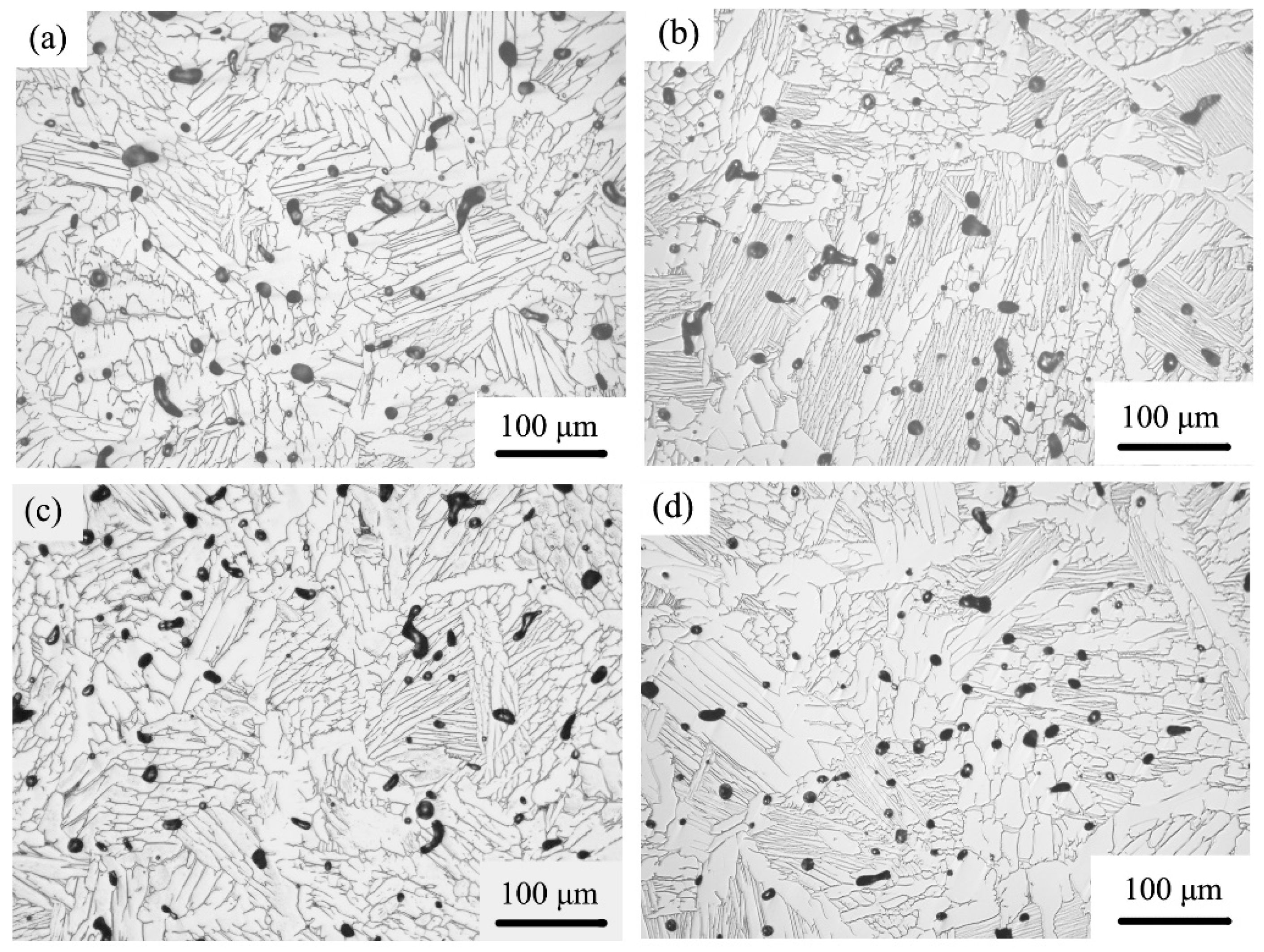
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leyens, C.; Peters, M. Titanium and Titanium Alloys Fundamentals and Applications, 1st ed.; Wiley-VCH: Weinheim, Germany, 2003; pp. 1–36. [Google Scholar] [CrossRef]
- Froes, F.H. Titanium alloys. In Handbook of Advanced Materials: Enabling New Designs, 1st ed.; Wessel, J.K., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004; pp. 271–319. [Google Scholar] [CrossRef]
- Froes, F.H.; Qian, M. Titanium background, alloying behavior and advanced fabrication techniques—An overview. In Titanium in Medical and Dental Applications, 1st ed.; Froes, F.H., Qian, M., Eds.; Woodhead Publishing: Duxford, UK, 2018; pp. 23–37. [Google Scholar] [CrossRef]
- Ezugwu, E.O.; Wang, Z.M. Titanium alloys and their machinability—A review. J. Mater. Process. Technol. 1997, 68, 262–274. [Google Scholar] [CrossRef]
- German, R.M. Progress in Titanium Metal Powder Injection Molding. Materials 2013, 6, 3641–3662. [Google Scholar] [CrossRef] [PubMed]
- Ebel, T. Metal injection molding (MIM) of titanium and titanium alloys. In Handbook of Metal Injection Molding, 1st ed.; Heaney, D.F., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2012; pp. 415–445. [Google Scholar] [CrossRef]
- Ebel, T.; Friederici, V.; Imgrund, P.; Hartwig, T. Metal Injection Molding of Titanium. In Titanium Powder Metallurgy Science, Technology and Applications, 1st ed.; Qian, M., Froes, F.H., Eds.; Butterworth-Heinemann: Oxford, UK, 2015; pp. 337–360. [Google Scholar] [CrossRef]
- Dehghan-Manshadi, A.; Bermingham, M.J.; Dargusch, M.S.; StJohn, D.H.; Qian, M. Metal injection moulding of titanium and titanium alloys: Challenges and recent development. Powder Technol. 2017, 319, 289–301. [Google Scholar] [CrossRef]
- Sun, P.; Fang, Z.Z.; Zhang, Y.; Xia, Y. Review of the Methods for Production of Spherical Ti and Ti Alloy Powder. JOM 2017, 69, 1853–1860. [Google Scholar] [CrossRef]
- Wen, G.; Cao, P.; Gabbitas, B.; Zhang, D.; Edmonds, N. Development and Design of Binder Systems for Titanium Metal Injection Molding: An Overview. Metall. Mater. Trans. A 2012, 44, 1530–1547. [Google Scholar] [CrossRef]
- Nyberg, E.; Miller, M.; Simmons, K.; Weil, K.S. Microstructure and mechanical properties of titanium components fabricated by a new powder injection molding technique. Mater. Sci. Eng. C 2005, 25, 336–342. [Google Scholar] [CrossRef]
- Shibo, G.; Xuanhui, Q.; Xinbo, H.; Ting, Z.; Bohua, D. Powder injection molding of Ti–6Al–4V alloy. J. Mater. Process. Technol. 2006, 173, 310–314. [Google Scholar] [CrossRef]
- Obasi, G.C.; Ferri, O.M.; Ebel, T.; Bormann, R. Influence of processing parameters on mechanical properties of Ti-6Al-4V alloy fabricated by MIM. Mater. Sci. Eng. A 2010, 527, 3929–3935. [Google Scholar] [CrossRef]
- Ergül, E.; Özkan Gülsoy, H.; Günay, V. Effect of sintering parameters on mechanical properties of injection moulded Ti–6Al–4V alloys. Powder Metall. 2013, 52, 65–71. [Google Scholar] [CrossRef]
- Ferri, O.M.; Ebel, T.; Bormann, R. Influence of surface quality and porosity on fatigue behaviour of Ti-6Al-4V components processed by MIM. Mater. Sci. Eng. A 2010, 527, 1800–1805. [Google Scholar] [CrossRef]
- Ferri, O.M.; Ebel, T.; Bormann, R. High cycle fatigue behaviour of Ti–6Al–4V fabricated by metal injection moulding technology. Mater. Sci. Eng. A 2009, 504, 107–113. [Google Scholar] [CrossRef]
- Ebel, T.; Blawert, C.; Willumeit, R.; Luthringer, B.J.C.; Ferri, O.M.; Feyerabend, F. Ti–6Al–4V–0.5B—A Modified Alloy for Implants Produced by Metal Injection Molding. Adv. Eng. Mater. 2011, 13, B440–B453. [Google Scholar] [CrossRef]
- Oger, B.; Ebel, T.; Limberg, W. The manufacture of highly-ductile and geometrically complex MIM-parts based on TiAl6V4. In Proceedings of the Euro PM2006, Ghent, Belgium, 23–25 October 2006; Volume 2, pp. 191–196. [Google Scholar]
- Gökelma, M.; Celik, D.; Tazegul, O.; Cimenoglu, H.; Friedrich, B. Characteristics of Ti6Al4V Powders Recycled from Turnings via the HDH Technique. Metals 2018, 8, 336. [Google Scholar] [CrossRef]
- Dunstan, M.K.; Gordon, A.; Paramore, J.D.; Butler, B.G. Feasibility of Using Titanium Machine Turnings in Powder Metallurgy Processes. JOM 2019. [Google Scholar] [CrossRef]
- Lin, D.; Sanetrnik, D.; Cho, H.; Chung, S.T.; Kwon, Y.S.; Kate, K.H.; Hausnerova, B.; Atre, S.V.; Park, S.J. Rheological and thermal debinding properties of blended elemental Ti-6Al-4V powder injection molding feedstock. Powder Technol. 2017, 311, 357–363. [Google Scholar] [CrossRef]
- Li, Y.; Chou, X.M.; Yu, L. Dehydrogenation debinding process of MIM titanium alloys by TiH2powder. Powder Metall. 2013, 49, 236–239. [Google Scholar] [CrossRef]
- Ye, S.; Mo, W.; Lv, Y.; Li, X.; Kwok, C.T.; Yu, P. Metal Injection Molding of Thin-Walled Titanium Glasses Arms: A Case Study. JOM 2018, 70, 616–620. [Google Scholar] [CrossRef]
- Park, S.-J.; Wu, Y.; Heaney, D.F.; Zou, X.; Gai, G.; German, R.M. Rheological and Thermal Debinding Behaviors in Titanium Powder Injection Molding. Metall. Mater. Trans. A 2008, 40, 215–222. [Google Scholar] [CrossRef]
- Lin, D.; Kang, T.G.; Han, J.S.; Park, S.J.; Chung, S.T.; Kwon, Y.-S. Experimental and Numerical Analysis of Injection Molding of Ti-6Al-4V Powders for High-Performance Titanium Parts. JOM 2018, 70, 621–625. [Google Scholar] [CrossRef]
- He, Y.H.; Jiang, Y.; Xu, N.P.; Zou, J.; Huang, B.Y.; Liu, C.T.; Liaw, P.K. Fabrication of Ti–Al Micro/ Nanometer-Sized Porous Alloys through the Kirkendall Effect. Adv. Mater. 2007, 19, 2102–2106. [Google Scholar] [CrossRef]
- Ye, S.; Hao, H.; Mo, W.; Yu, K.; Liu, L.; Deng, C.; Yu, P. Effects of cold compacting pressure on the expansion behavior of Ti-48Al during sintering. J. Alloys Compd. 2016, 673, 399–404. [Google Scholar] [CrossRef]
- Frees, F.H. Powder Injection Molding (Pim) Of Titanium Alloys—Ripe For Expansion. Mater. Technol. 2016, 15, 295–299. [Google Scholar] [CrossRef]

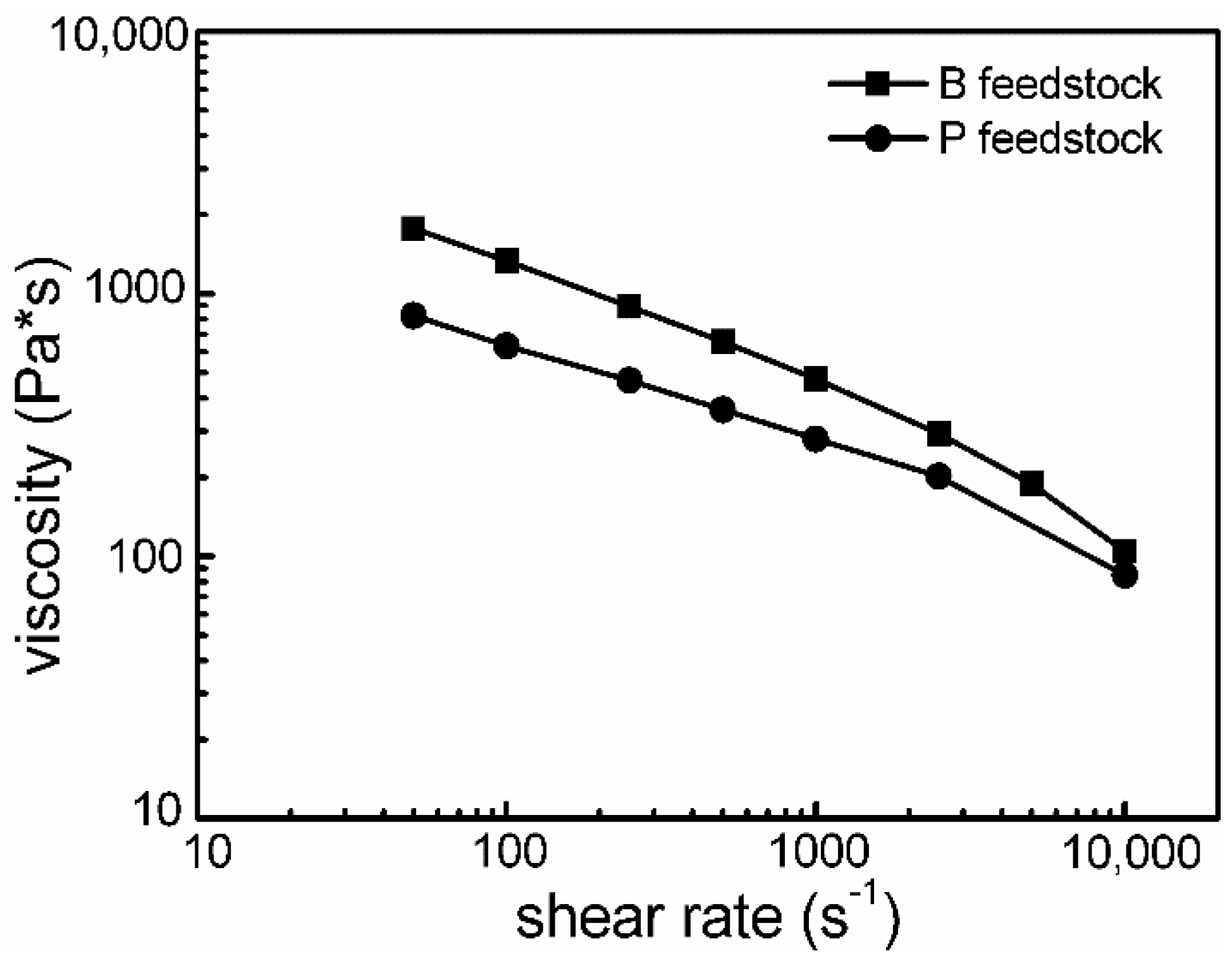

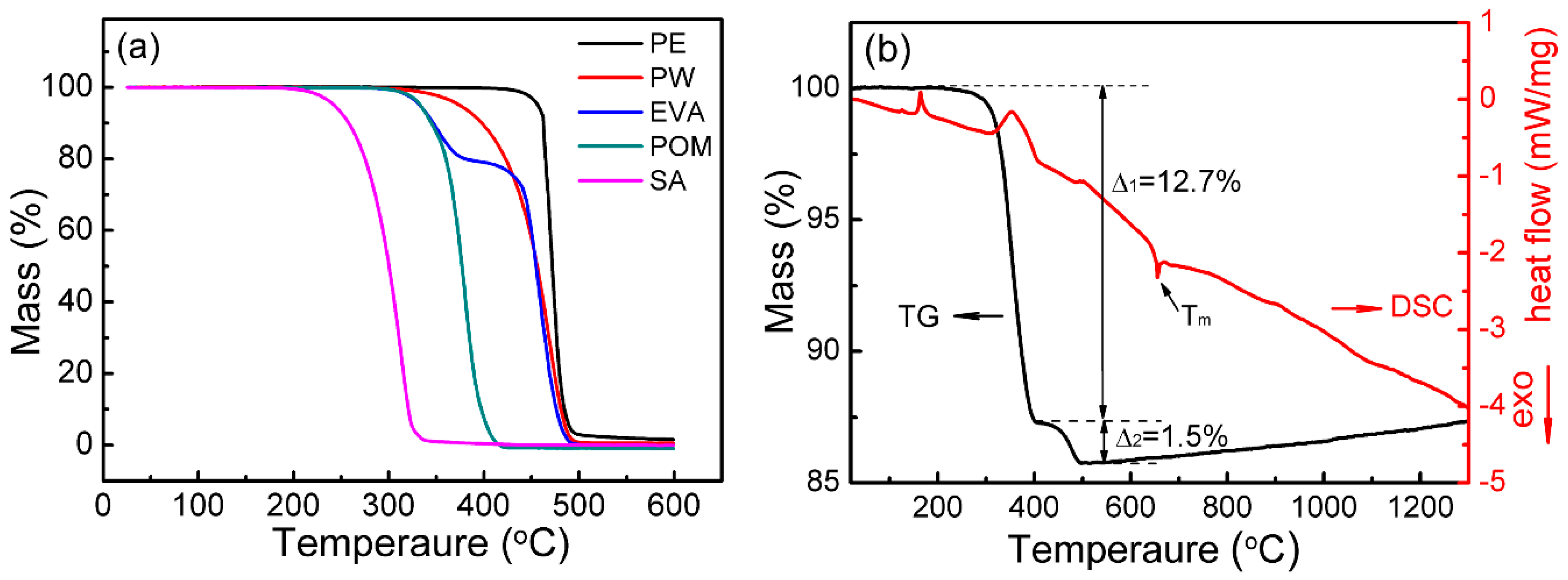
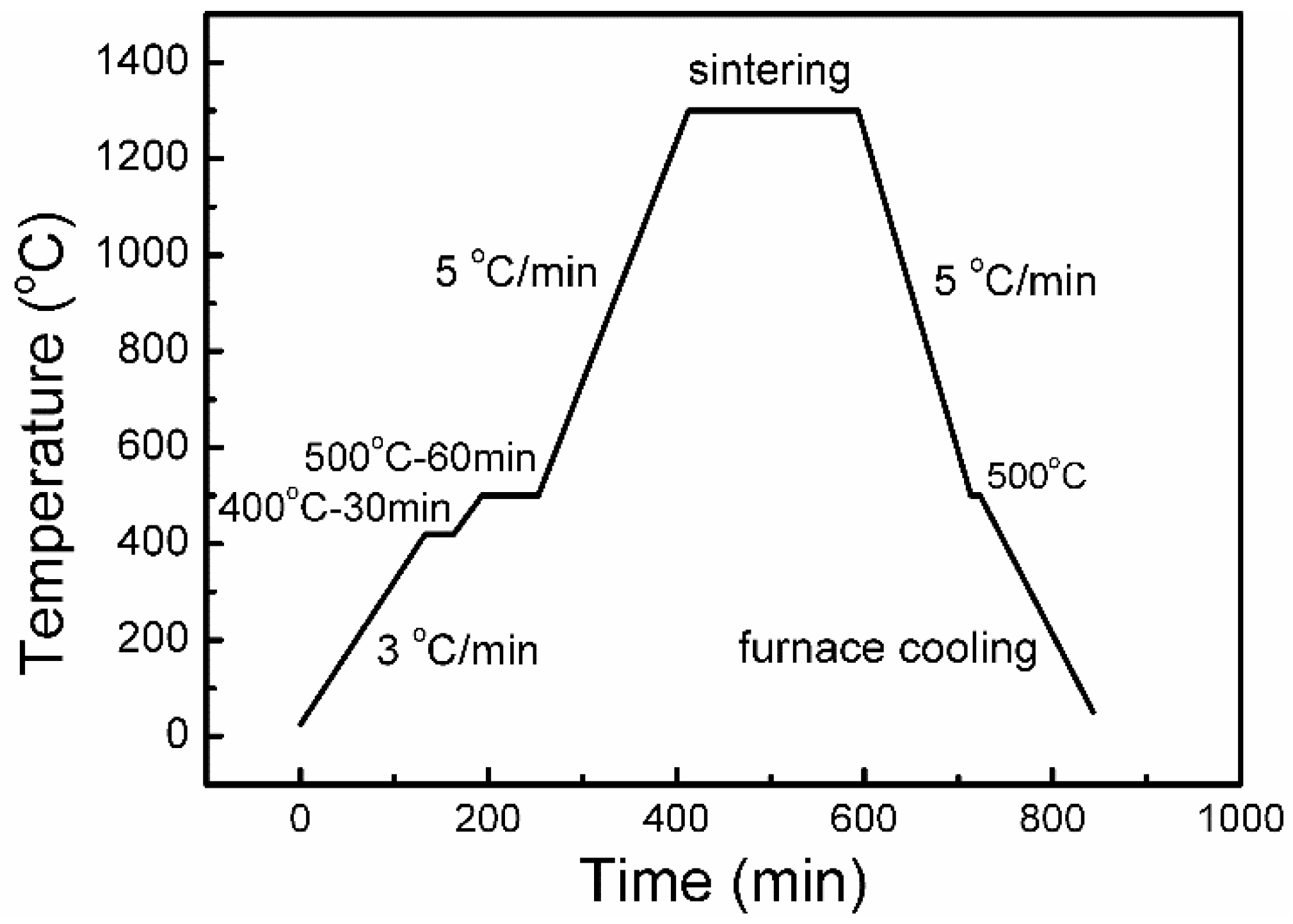

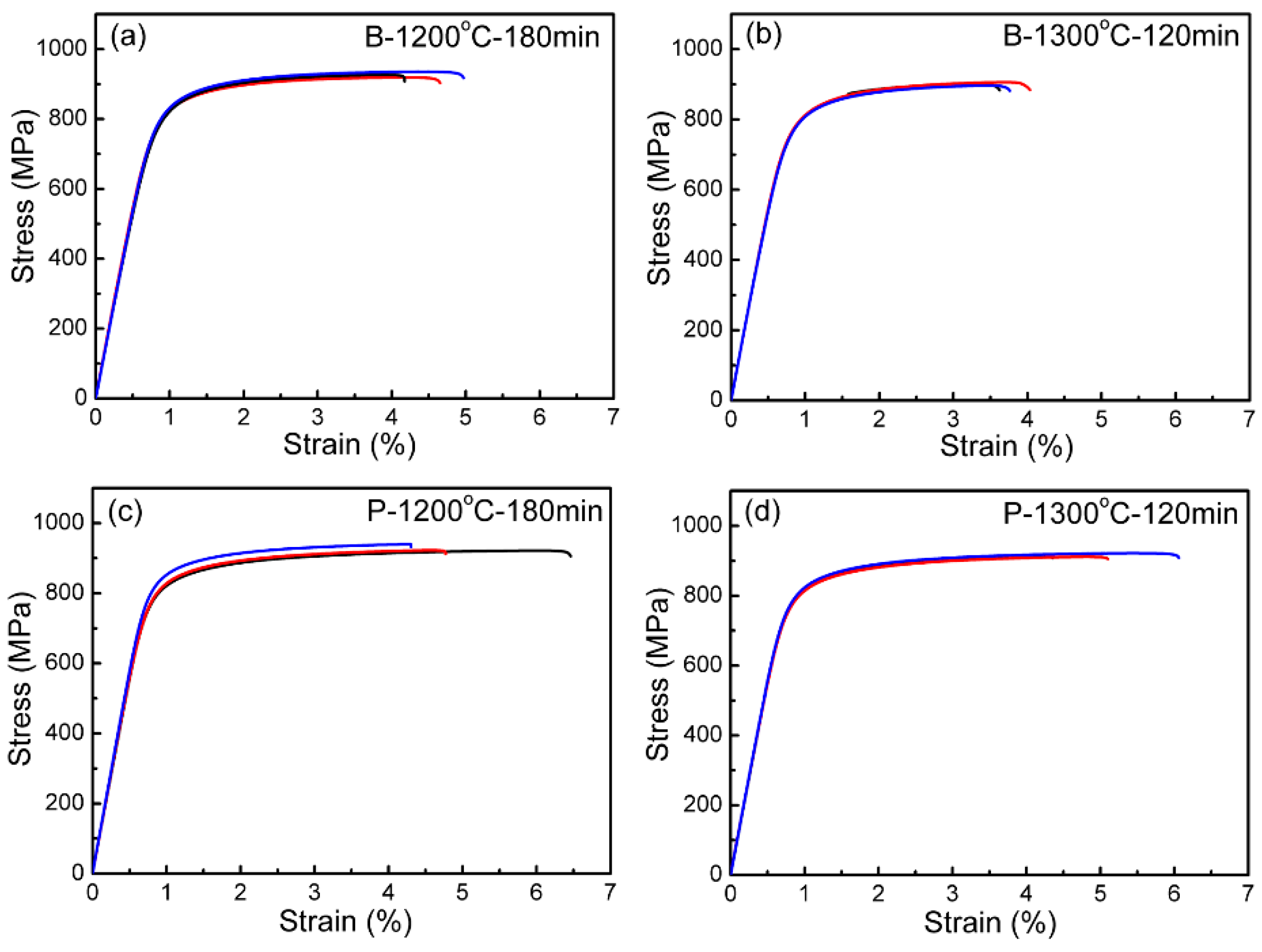

| Powder | O (wt.%) | C (wt.%) | N (wt.%) | Price (USD/kg) |
|---|---|---|---|---|
| GA Ti | 0.123 | 0 | 0.009 | 220 |
| Al | 0.220 | - | - | 30 |
| 35Al65V | 0.370 | 0.027 | 0.180 | 60 |
| GA Ti-6Al-4V | 0.176 | 0.004 | 0.025 | 520 |
| Specimens | O (wt.%) | C (wt.%) | N (wt.%) | Density (g/cm3) | Yield Stress (MPa) | UTS (MPa) | Elongation (%) |
|---|---|---|---|---|---|---|---|
| B-1200 °C-180 min | 0.318 | 0.071 | 0.028 | 4.26 | 810 | 927 | 4.6 |
| B-1300 °C-120 min | 0.326 | 0.077 | 0.031 | 4.26 | 790 | 900 | 3.9 |
| P-1200 °C-180 min | 0.250 | 0.076 | 0.034 | 4.26 | 818 | 928 | 5.2 |
| P-1300 °C-120 min | 0.312 | 0.070 | 0.034 | 4.28 | 800 | 913 | 5.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, S.; Mo, W.; Lv, Y.; Wang, Z.; Kwok, C.T.; Yu, P. The Technological Design of Geometrically Complex Ti-6Al-4V Parts by Metal Injection Molding. Appl. Sci. 2019, 9, 1339. https://doi.org/10.3390/app9071339
Ye S, Mo W, Lv Y, Wang Z, Kwok CT, Yu P. The Technological Design of Geometrically Complex Ti-6Al-4V Parts by Metal Injection Molding. Applied Sciences. 2019; 9(7):1339. https://doi.org/10.3390/app9071339
Chicago/Turabian StyleYe, Shulong, Wei Mo, Yonghu Lv, Zhanhua Wang, Chi Tat Kwok, and Peng Yu. 2019. "The Technological Design of Geometrically Complex Ti-6Al-4V Parts by Metal Injection Molding" Applied Sciences 9, no. 7: 1339. https://doi.org/10.3390/app9071339
APA StyleYe, S., Mo, W., Lv, Y., Wang, Z., Kwok, C. T., & Yu, P. (2019). The Technological Design of Geometrically Complex Ti-6Al-4V Parts by Metal Injection Molding. Applied Sciences, 9(7), 1339. https://doi.org/10.3390/app9071339




