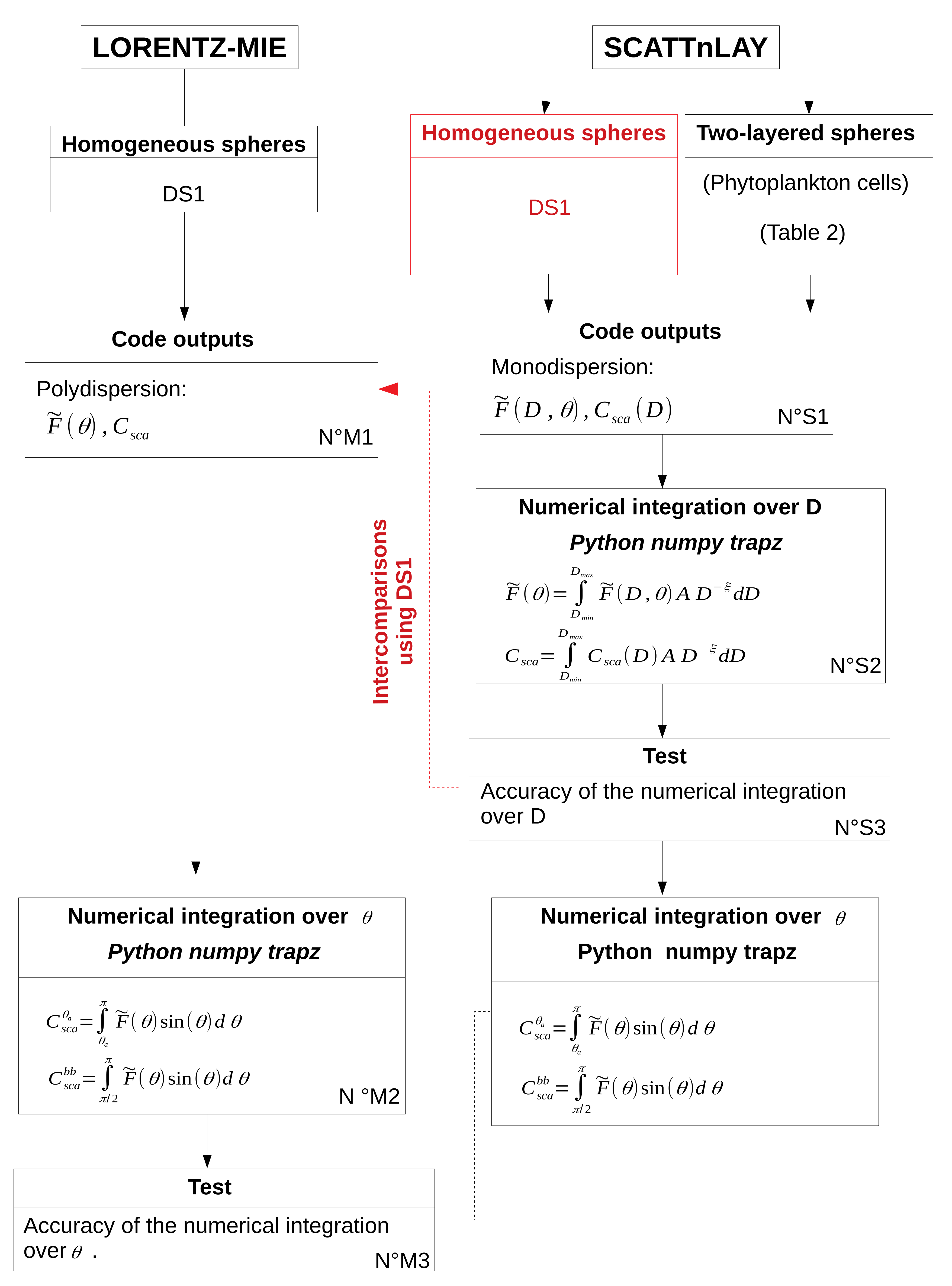
5.1. Accuracy of Numerical Computations
A numerical integration over
is required to derive
and
from the normalized phase function (
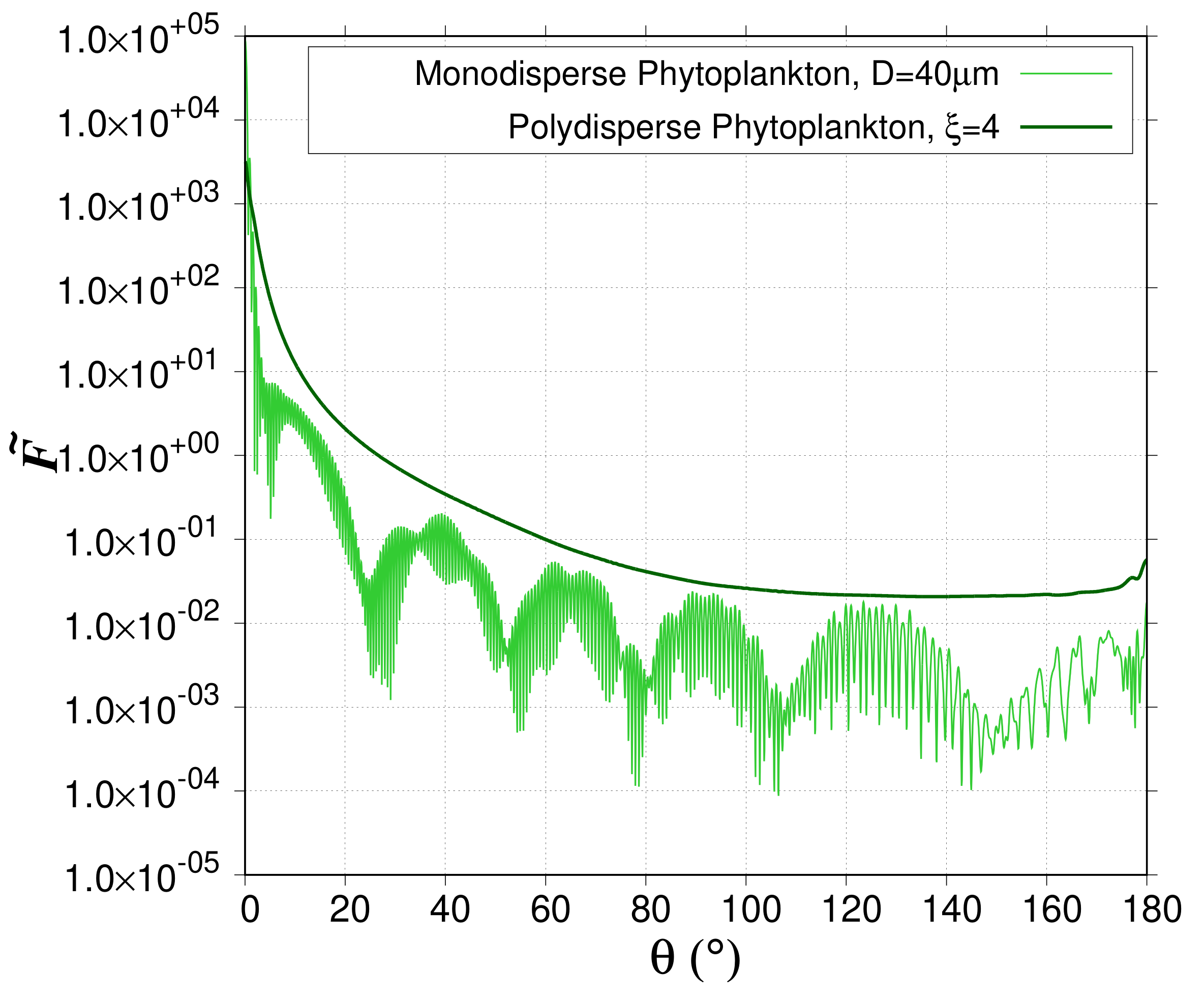
Section 2). Due to the sharp increase of the normalized phase function in the forward scattering directions (
Figure 3), the selection of the relevant angular step for the numerical integration is crucial. For that purpose, the impact of angular step (
) on the calculation of
is studied using Lorentz-Mie simulations in DS1 (
Figure 1, N
oM2, M3). The normalized phase function of polydisperse particles
exhibits a maximum around
= 0
o [
26]. For small
value, that is when the proportion of large-sized particles compared to smaller particles increases, the forward peak is sharper. Indeed, for particles with a large diameter as compared to the wavelength,
displays a sharp forward peak [
26] due the concentration of light near
= 0
o caused by diffraction. The presence of the peak in
requires several integration points large enough to provide the desired numerical accuracy. The numerical integration over
(
Figure 1, N
oM2) is performed using the “Trapz” function from the Numpy package with Python. The “Trapz” function performs an integration along the given axis using the composite trapezoidal rule. To test the accuracy of the integration and to find the correct integration step,
, we compare the result of the numerical integration of
between 0 and
to its theoretical value (=2) (
Figure 1, N
oM3). When
= 0.05
o, corresponding to a total number of integration steps (N
) of 3600, the numerical integration value of
is in the range [1.999–2.000] for small
. For larger
, it is in the range [1.800–1.999]. When the value of the numerical integration is in the range [1.800–1.999], a renormalization factor is applied to
to ensure that the result of the numerical integration is exactly 2. We could also increase the number of integration points, but it would increase the computation time. Using a renormalization factor for large
is a good compromise to guarantee the accuracy and save computation time.
For two-layered spheres (i.e., phytoplankton cells), the ScattnLay code provides only normalized phase functions for monodisperse particles (
Figure 1, N
oS1), so the numerical integration over the particle diameter range (Equation (
2)) is realized as a separate calculation with the Python “Trapz” function (
Figure 1, N
oS2). For monodisperse particles, the normalized phase function displays a forward peak as explained above but can also display a sequence of maxima and minima due to interference and resonance features [
26,
42]. The frequency of the maxima and minima over the range of
increases with both increasing
and size parameter (=
). To test the accuracy of the numerical integration over the particle diameter range (
Figure 1, N
oS3), we ran the ScattnLay code for DS1 case studies and compared
and
rebuilt from Equation (
2) with Lorentz-Mie computations as the Lorentz-Mie code provides the polydisperse phase functions and cross section as outputs (
Figure 1, N
oM1). Note that even a narrow polydispersion washes out the interference and resonance features, which explains why most natural particulate assemblages do not exhibit such patterns [
26,
42] (
Figure 3). A perfect match is obtained between the ScattnLay-rebuilt-polydisperse and Lorentz-Mie-polydisperse
and
values when the integration step (
) is set to 0.01
m for
D in the range [0.03, 2
m]; 0.1
m for
D in the range [2, 20
m]; 2.0
m for
D in the range [20, 200
m]; and 10.0
m for
D in the range [200, 500
m].
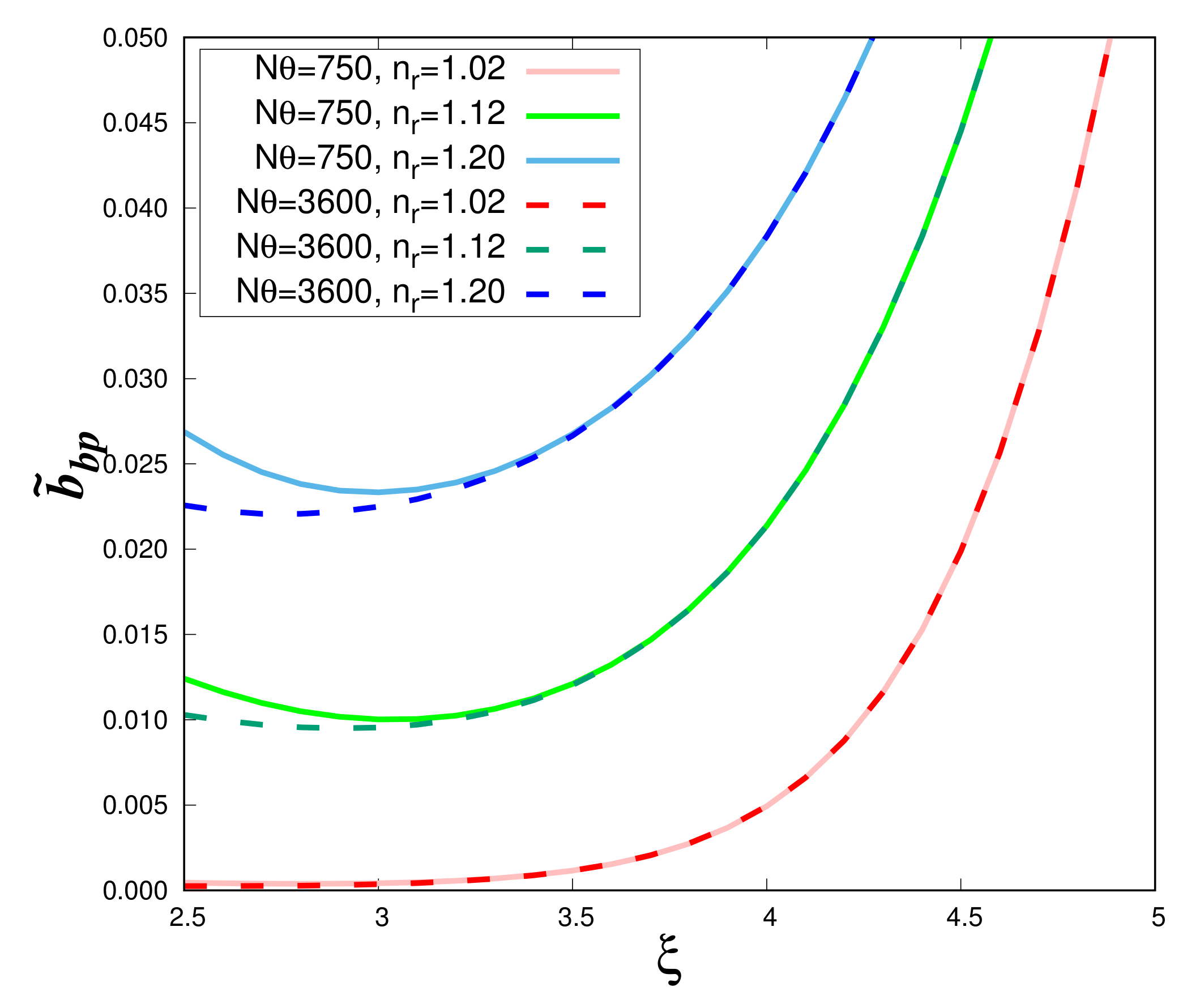
Using the DS1 data set, the impact of the angular integration on the backscattering ratio
is examined as a function of the hyperbolic slope
for different values of the real refractive index and two values of total angular steps (i.e., N
= 750 and 3600) (
Figure 4). The impact of the integration is noticeable only for
values lower than about 3 and relatively high
values. When the number of angular steps increases, the curves become flatter at low
values. Differences in the curve shape are reduced if we increase the angular step. For
= 0.24
o (N
= 750), the present results of the Lorentz-Mie calculations (solid lines in
Figure 4) perfectly match those previously obtained by Twardowski et al. [
5] (not shown). However, in this case (N
= 750), the numerical integration is not accurate enough as the integration of Equation (
1) gives values between 1.999 (
= 4.9) and 1.04 (
). In the following,
is set to 0.05
o (N
= 3600) and
Figure 4 (dashed lines) will be the reference figure for homogeneous spheres.
5.2. Impact of the Structural Heterogeneity of Phytoplankton Cells on the Bulk Particulate Backscattering Ratio
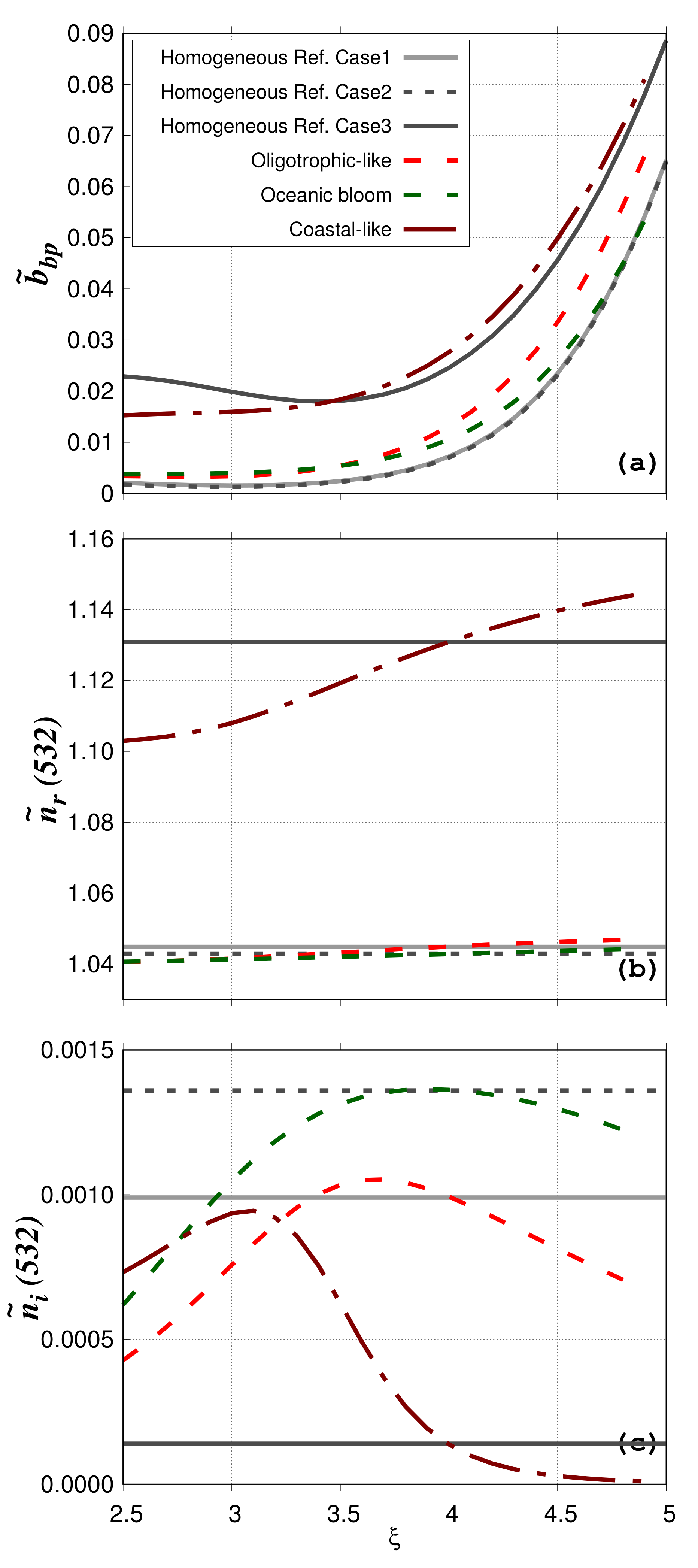
The impact of phytoplankton cell structural heterogeneity on
is examined as a function of
for the three previously described water bodies (oligotrophic-like, phytoplankton bloom, coastal-like) considering the 80%–20% phytoplankton morphological model (
Figure 5a). The real and imaginary bulk refractive indices for oligotrophic-like, phytoplankton bloom, and coastal-like case studies, vary with
as the relative proportions of the different particle components, having different
and
, vary with
(
Table 1,
Table 2,
Table 3,
Table 4 and
Table 5 and
Figure 5b,c). For the no-mineral water bodies (oligotrophic-like and phytoplankton bloom),
stays around 1.04 ± 0.007 (
Figure 5b). In contrast,
shows large variation with
for both oligotrophic-like and phytoplankton bloom water bodies (
Figure 5c). In bloom conditions,
increases as the relative proportion of phytoplankton increases as compared to the no bloom conditions. In agreement with typical values of the oceanic bulk imaginary refractive index [
43], the
values for the particulate populations considered here are always lower than 0.002. In the presence of mineral particles (coastal-like),
increases as MIN have a higher
than VIR, BAC, PHY and DET. Its values are between 1.103 (
) and 1.145 (
). Values of
vary between 9.79 × 10
and 9.44 × 10
.
The impact of the structural heterogeneity of phytoplankton cells is evaluated by comparison with Lorentz-Mie calculations (particulate assemblages composed of homogeneous spheres only, regardless of the particle group) performed for low and high bulk refractive index. These case studies with homogeneous spheres only are named “Homogeneous reference cases”. The real and imaginary values of the bulk refractive indices are 1.045 and 9.93 × 10
for the “Homogeneous reference case 1”, 1.043 and 1.36 × 10
for the “Homogeneous reference case 2”, and 1.131 and 1.37 × 10
for the “Homogeneous reference case 3”, respectively (
Figure 5b,c). These values of
and
were chosen to be equal to values of
and
obtained for the oligotrophic-like, phytoplankton bloom, and the coastal-like case study when
= 4. “Homogeneous reference cases 1 and 2” with low
represent phytoplankton-dominated Case 1 waters and are compared with the oligotrophic-like and phytoplankton bloom water body, respectively. “Homogeneous reference case 3” with high
represents mineral-dominated Case 2 waters and is compared with the coastal-like water body. The variation of
due to structural heterogeneity of phytoplankton cells is evaluated using the relative absolute difference calculated between the homogeneous reference cases (named
x in Equation (
13)) and oligotrophic, phytoplankton bloom or coastal-like water bodies (named
y in Equation (
13)):
Even if the numerical relative abundance of phytoplankton is very small for the oligotrophic-like water body (=8.4 × 10
%), the structural heterogeneity increases the
value by 58% compared to the homogeneous case (“Homogeneous reference case 1”). This is consistent with previous studies showing the large contribution of coated spheres to the backscattering signal [
15,
16,
18,
19,
22,
44]. The value of
calculated between the oligotrophic-like and phytoplankton bloom water bodies is smaller (=22% at
= 4) even if
is different (9.93 × 10
for oligotrophic-like against 1.36 × 10
for phytoplankton bloom). This latter pattern provides evidence that the structural heterogeneity (coated-sphere model) has a greater impact on the particulate backscattering ratio than the tested increase in the bulk imaginary refractive index. The relative absolute differences between the phytoplankton bloom and “Homogeneous reference case 2” is 41%. When mineral particles are taken into account,
is 12% between the “Homogeneous reference case 3” and the coastal-like water body. This smaller difference is because phytoplankton have a smaller impact on the bulk scattering when highly scattering particles such as minerals are added.
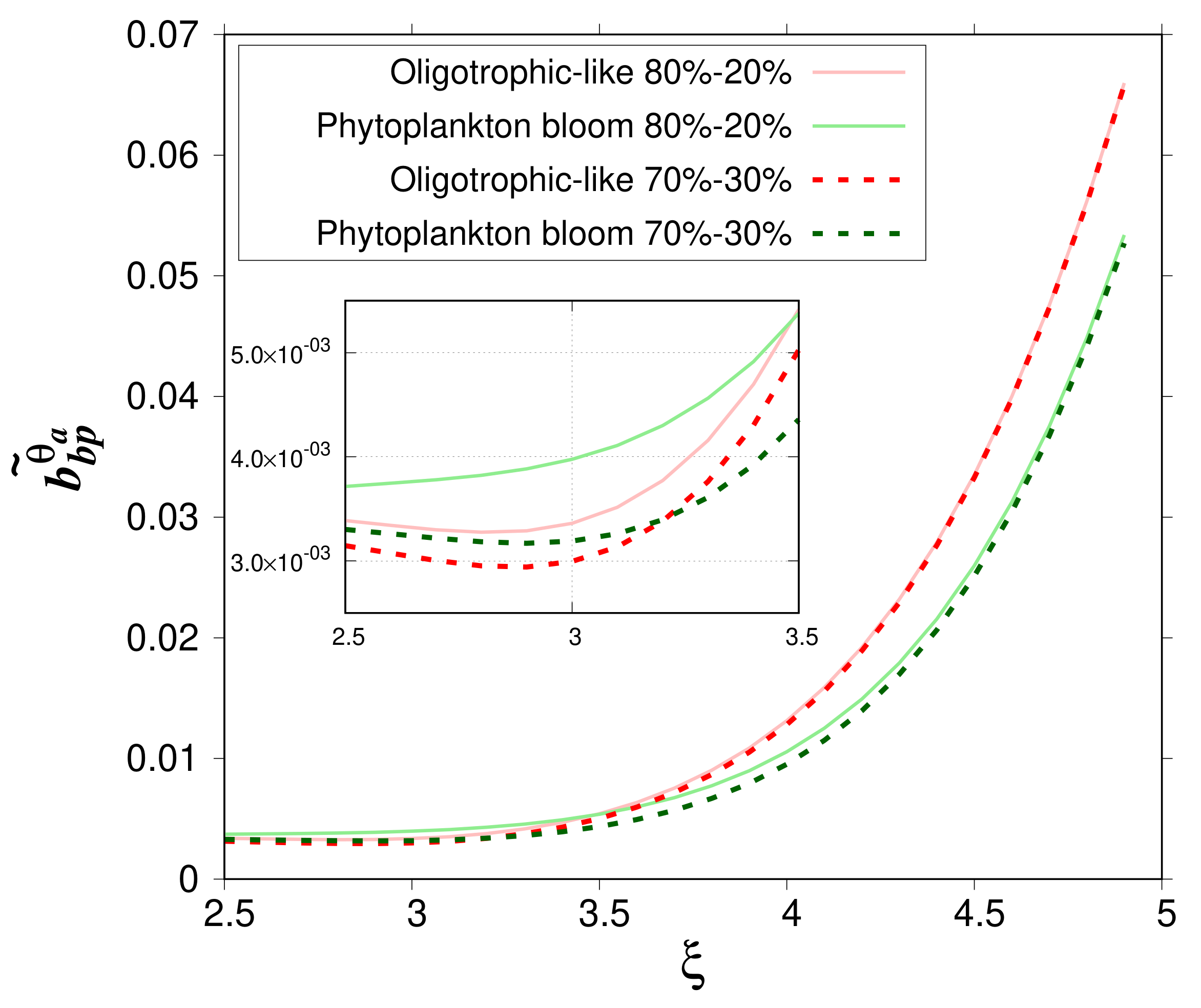
The impact of the relative volume of the cytoplasm on
is now evaluated by comparing the change of
as a function of
for the 80%–20% and 70%–30% models for the oligotrophic-like and phytoplankton bloom water bodies (
Figure 6). The mean relative difference in
is about 5.41% with a maximum value of 11.5 % (
= 3) for oligotrophic-like case study. In bloom conditions, the mean relative difference reaches 13.0% with a maximum value of 23.5% (
= 3.2).
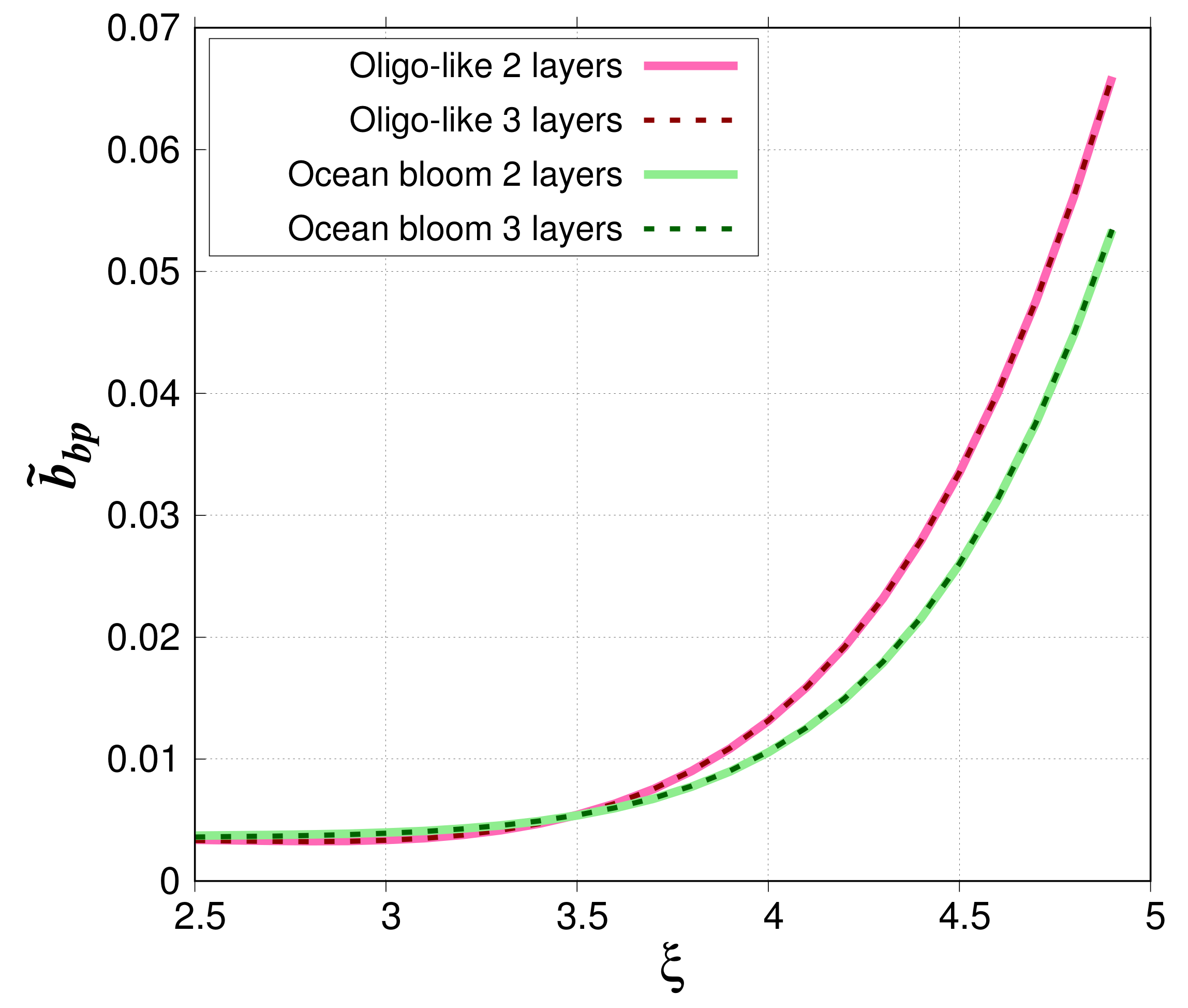
Figure 7 compares simulated
when phytoplankton cells are modeled as two-layered spheres (80%–20%) or three-layered spheres (80%–18.5%–1.5%). For the oligotrophic-like waters, relative absolute differences are small. They range between 0.0174% and 1.81% with a mean value of 0.444%. For phytoplankton bloom case study, they are between 9.84 × 10
and 2.86% with a mean value of 0.894%.
Regardless of the morphological model used to optically simulate phytoplankton cells, the
reaches an asymptote when
decreases for phytoplankton bloom water bodies (
= 0.005 for
= 3.5 and 0.004 for
= 2.5). The value of the asymptote is consistent with previous observations [
5], which showed the lowest backscattering ratio (about 0.005) in waters with high chlorophyll-a concentration.
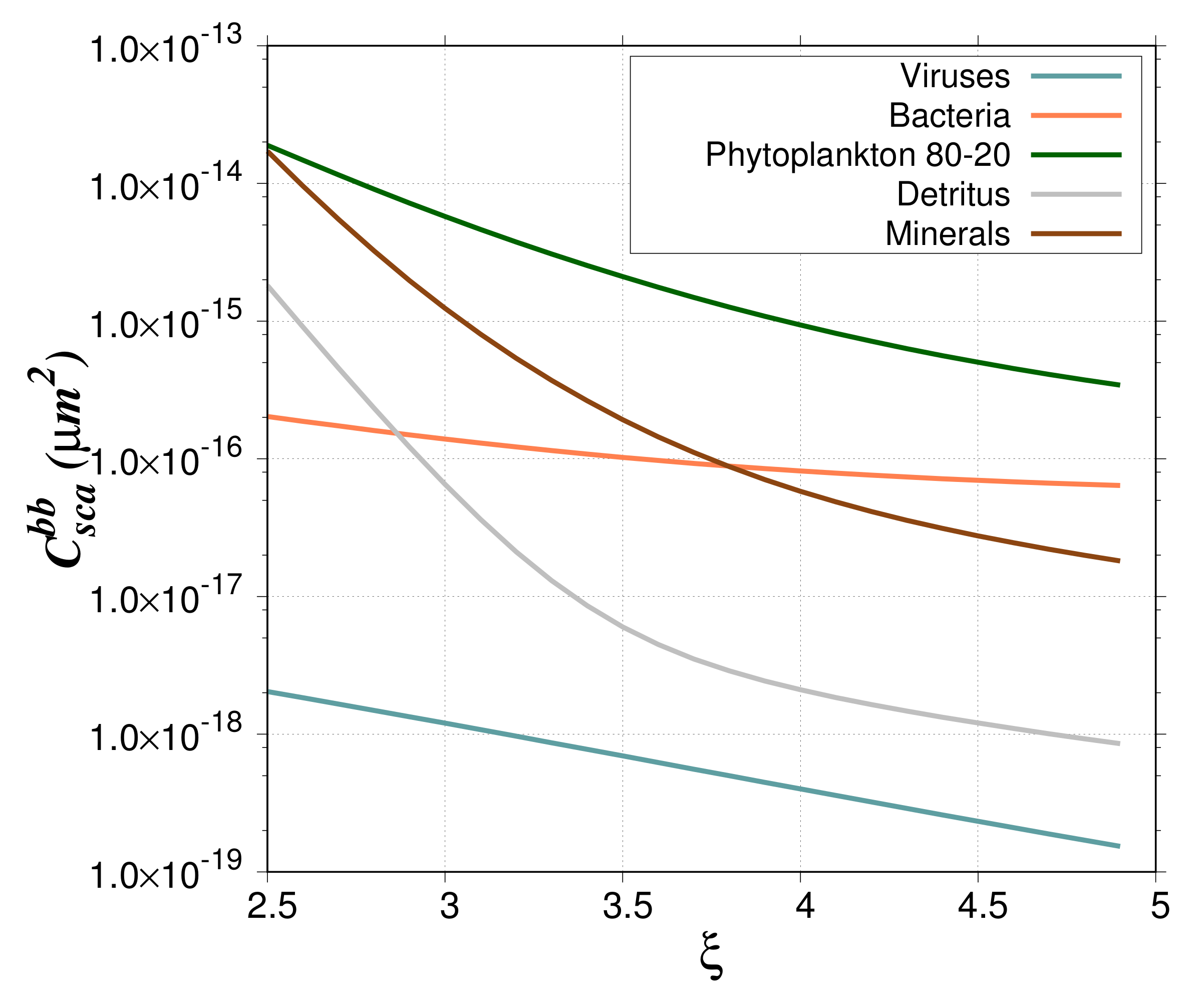
The contribution of the different particle groups to the backscattering ratio is presented in
Figure 8 for the 80%–20% model and
= 4. For coastal-like waters, the minerals contribute more than 80% of the total
, whereas they contribute only 6.5% to the total particulate abundance. This percentage agrees with the results of Stramski et al. [
25] (Figure 12 in their paper). Such high contribution to backscattering is due to the high real refractive index of minerals. As in Stramski et al. [
25], these results show the important role of minerals even when they are less abundant than organic living and non-living particles. In oligotrophic-like waters, the contribution of heterotrophic bacteria ranges from a few to about 30% with a maximum for
between 3.5 and 4, which agrees with Stramski and Kiefer [
24]. The contribution of viruses is quite high, about 40–60% for
between 4 and 5. This high contribution is explained by the extreme value of viral abundance (around 1 × 10
particles per cubic meter) used in this study [
24]. As for bacteria, the contribution of phytoplankton ranges between a few and 30% with a maximum around
= 3. For a
value of 4, typical of oligotrophic waters, the contribution is around 10%. For the phytoplankton bloom study case, the contribution of phytoplankton cells is between 10% and 60%; maximum values are reached for a PSD slope between 3 and 3.5. Such high percentages are due to the higher backscattering cross section of phytoplankton as compared to the other particles (
Figure 9). The low phytoplankton abundance is offset by the high
so that the backscattering coefficient of phytoplankton represents a significant contribution to the total backscattering.