Abstract
(1) Introduction: Polycaprolactone (PCL) materials have been developed with components that promote bone growth. The main objective of this work was to evaluate the biocompatibility and cytotoxic effects that different combinations of PCL with nanohydroxyapatite and strontium could produce on periodontal ligament stem cells (PDLSC). (2) Materials and Methods: PDLSCs were seeded in six 96-well plates. Three plates were used for the MTT test, and three were used for the Hoechst 33342 test. In each of the plates, three samples of different concentrations of PCL were introduced (PCL 100%, PCL 95% combined with nanohydroxyapatite functionalized with strontium, and PCL 90% with nanohydroxyapatite). Apoptosis was analyzed using Hoechst and cell viability combined with MTT at 24, 48, and 72 h. (3) Results: No statistically significant differences (p < 0.05) were found between the different concentrations of PCL or regarding the duration for which the cells were subjected to elution. (4) Conclusions: Pure PCL and both PCL combined with nanohydroxyapatite/strontium and nanohydroxyapatite are biocompatible materials, and there are no significant differences between them after apoptosis and in cell viability assays.
1. Introduction
The periodontal ligament is a connective tissue that is located between the root cementum and the internal cortical of the alveolus. In this tissue, stem cells (PDLSCs) with the ability to differentiate into cells of the periodontal ligament are present and have different functions such as dental support, supplying nutrients and maintaining the homeostasis of the teeth through continuous regeneration [1,2,3]. On the other hand, the alveolar bone is a mineralized connective tissue made up of three types of cell lines: osteoblasts, osteoclasts, and osteocytes. It is a dynamic tissue that undergoes constant remodeling. This regeneration is carried out through three phases: the first consists of resorption and is carried out through osteoclasts; the second phase of bone formation is the transition period, in which bone formation and destruction take place at the same time; and the third phase is that of bone formation only through osteoblasts [3]. This regeneration cannot always be carried out, and therefore, the bone cortical is destroyed. Bone loss of the upper and lower jaws is mainly caused by pathogenic microorganisms, environmental factors, bad habits such as smoking, genetic factors, or a lack of replacement after tooth loss. This issue of bone loss has led to the development of different techniques to promote osteogenesis and thus combat the defect, since bone is a dynamic tissue. Some of these techniques include bone grafting and the use of scaffolds, stem cells, growth factors, and other biomaterials [4,5].
The primary function of the material in these techniques is to stimulate the regeneration of bone tissue in areas that are affected by defects (such as bone loss in periodontitis) by degrading in the same place to facilitate the replacement of the material by newly formed bone tissue. Nanocomposite biomaterials are relatively novel and consist of a polymeric and biodegradable structure that includes nano-sized materials with a bioactive function [6].
The biomaterial characteristics that must be considered are its chemical composition; biological characteristics, such as providing osteoinduction and biocompatibility; biodegradation; and manufacturing process. Biomaterials that are used in bone tissue engineering can be degradable or non-degradable and synthetic or natural. Several materials are used today.
Natural polymers: these are polymers obtained from natural substances. The main characteristic of these types of materials is their great biocompatibility; however, they have different disadvantages, such as procurement difficulties, production limitations, and their high cost. These types of polymers include collagen, gelatin, fibrinogen, or polysaccharide polymers [7].
Synthetic polymers: these are polymers that are artificially formed through chemical processes. These polymers have a longer storage time and higher production capacity and can achieve physical properties that are very similar to those of bone. Some polymers, such as polylactic acid (PLA), polycaprolactone (PCL), and polyglycolic acid (PGA) [8], are modified with other substances, such as hydroxyapatite. PCL is a linear synthetic polymer and used as a biomaterial in the field of health due to its beneficial properties such as porosity, slow degradation (thus offering greater load support), high biocompatibility, and lower release of acidic products compared with other polymers. However, pure PCL has some disadvantages. Because it is a hydrophobic material, it lacks the osteogenic potential for the formation of bone tissue; for this reason, PCL has been combined with different inorganic materials to improve its weaknesses. This is how the combination of PCL with hydroxyapatite (HA) nanocrystals emerged. This mixture of both materials offers mechanical and biological improvements to reduce its limitations, making it a promising material in scaffolds [9]. Strontium has also been used in combination with PCL due to the improvement in biological and osteoinductive properties that it offers for bone formation [10].
Biocompatibility is defined as the ability of a material to adapt to act favorably on a biological tissue. On the other hand, cytotoxicity is defined as the ability of a material to cause cell damage. To evaluate cell toxicity and viability, one of the assays that can be performed is the MTT, which is highly reliable and allows us to know the cell proliferation and cytotoxicity that are produced by both materials and drugs in cell cultures [11,12]. The main disadvantage of this type of assay is that it does not allow for the assessment of morphological changes in cells; therefore, it is combined with apoptosis or cell death assays such as Hoechst 33342. This technique consists of a fluorescent dye that can cross the cell membrane of the cell, staining the nucleus of living cells and allowing for their subsequent measurement under an optical microscope [13]. Periodontal ligament stem cells (PDLSCs) are capable of performing important functions in the development and regeneration of periodontal tissues. These are cells with a great capacity for development and proliferation in suitable environments [14]. As they are cells with a great proliferative capacity, they have been used in a multitude of studies, in which in vitro research was carried out on this cell lineage [15].
The aim of this work was to test the cytotoxic effects of various concentrations of polycaprolactone and other associated materials, such as strontium and hydroxyapatite, on periodontal ligament cells (PDLSCs).
2. Materials and Methods
2.1. Cell Culture
To perform the cell culture, PDLSCs were stored at −196 °C in liquid nitrogen, and thawed at the cell culture service laboratory of the University of Murcia. After thawing, the culture was carried out in three flasks (two 75 cm2 flasks and one 25 cm2 flask). To achieve this, a DMEM cell culture medium containing 10% phenol red bovine serum (SBF) in volume was used, as well as 4.5 g/L glucose, 4 mM glutamine, and 1% penicillin. Once combined, the cells and medium were stored in an incubator at 37 °C, 7.5% CO2, and 85% relative humidity, favoring cell development and growth adhering to the lower part of the culture dish.
From these culture vials, 3 subcultures were performed when the cells were close to reaching the confluence state occupying 85% of the vial base. To carry out this subculture, the medium was changed out every 4 days, and the cells were also subjected to a trypsinization process. To perform the cell count and ensure that the subculture was correct, a TC20TM automatic cell counter was used using trypan blue. This coloring substance can enter dead cells by staining them blue, while the living cells do not receive any type of staining due to the plasma membrane that prevents the passage of staining, thus indicating cell viability and the total number of cells, and allowing for the calculation of the cell solution.
2.2. Sample Preparation
The experimental hybrid inks were manufactured by the Polymers Research Group (CSIC, Madrid, Spain), following the subsequent stages:
The synthesis of strontium-free and substituted hydroxyapatites involved adding 500 mL of 0.24 M di-ammonium hydrogen phosphate solution and 500 mL of 0.40 M of analytical-grade calcium acetate or calcium acetate plus a strontium acetate solution simultaneously in a closed polypropylene reaction vessel. The solutions were added in 10 mL aliquots with a 700 Dosino autoburette (Metrohm, Herisau, Switzerland), at a rate of 10 mL/min, until total addition. Ca/Sr ratios and sample codes of the prepared samples can be found in Table A1, Appendix A. The pH of the mixed solution was set to a value of 9.40 and it was maintained at this level throughout the reaction with a pH stat (Titrino 736, Metrohm, Switzerland) with the addition of 3.3 vol% ammonia solution. The temperature was set at 40 °C. After finishing the addition, the reaction was aged for 17 h. The obtained powders were filtered, washed with deionized water, and dried at 60 °C. Powders were calcined at 600 °C for 5 h using a 10 °C/min heating rate. Some of the powders were also calcined at 900 °C for further characterization (Appendix A). The synthesis reaction is:
where for Sr0, x = 0; for Sr10, x = 1
(10 − x)Ca(CH3COO)2 + x Sr(CH3COO)2 + 6(NH4)2 HPO4 + OH−
⟶Ca(10−x) Srx(PO4)6(OH)2
⟶Ca(10−x) Srx(PO4)6(OH)2
Characterization results can be found in Appendix A, and references are provided there.
The polymer was mixed with ceramic fillers in different percentages until the maximum possible load was optimized. Once all polymer–ceramic mixtures were obtained, a micro-extruder (Thermo-Haake Minilab system, Thermo Fisher Scientific, Waltham, MA, USA) was used to obtain a printable filament. Pore size values of 300 microns with 100% interconnectivity were used as reference values for the internal face of the graft (oriented towards the bone substrate). On the contrary, the external face had porosities <50 μm to prevent soft tissue invasion. Parametric design software, OpenScad v2021.01, was used for the design of the scaffolds. A fuse filament printer based on a Hephestos 2 (Prusa i3-BQ, Madrid, Spain) was used for the printing of 3D models. The porosity was designed directly into the slicing program, Cura (UltiMaker, Utrecht, The Netherlands). Two types of experimental, easily printable, hybrid filaments were obtained, i.e., PCL90 and PCL95. The PCL90 was composed of 90% of PCL (Sigma-Aldrich, St. Louis, MO, USA, Mn 80,000 g/mol) and 10% of hydroxyapatite, whereas PCL95 was made of 95% of PCL and 5% of strontium-loaded hydroxyapatite. In the preclinical testing, a control polymer made of 100% PCL was used (3D4makers.com, Haarlem, The Netherlands, Mw: 84,500). Synthesis reactions have been added to the Appendix A.
The first material weighed 0.0837 g, with a pure PCL concentration of 100%; the second material had a weight of 0.3186 g and a PCL concentration of 95% of PCL and 5% of strontium-loaded hydroxyapatite; finally, the third PCL material had a PCL concentration of 90% and 10% hydroxyapatite and a weight of 0.2034 g (Figure 1).

Figure 1.
Samples of PCL. PCL 100% (left), PCL 95% (center), and PCL 90% (right).
These samples were introduced into empty 5 mL tubes and sterilized twice for 30′ in a laminar UV/C flow cabinet at 254 nm. Once sterilization was completed, different amounts of DMEM culture medium without phenol red were added to each tube following ISO 10993-12 standards [16] and according to the weight of the material, resulting in 1.1 mL in the first tube, 3.186 mL in the second, and 2.34 mL in the third. The samples were stored for 24 h in the CO2 incubator at 7.5%, 37 °C, and a relative humidity of 85%. After 24 h, the liquid resulting from incubation was used for the cell viability assay.
2.3. Plate Seeding
Cell seeding was performed in a type II biological safety cabinet with an HEPA filter, using 6 plates of 96 wells; 3 of them were used for MTT, and the remaining 3 for Hoechst staining, with each well being seeded, having a cell density of 4000 cells, containing 250 μL of a DMEM culture medium with phenol red, and being incubated for 24 h under the following conditions: 37 °C, 7.5% CO2, and 85% humidity.
To perform the MTT, 3 plates were obtained, in which 3 wells in each column were dedicated to each substance, with 3 being blank (no presence of cells), 6 being positive controls, 3 consisting of material 1 (PCL 100), 3 consisting of material 2 (PCL 95), 3 consisting of material 3 (PCL 90), and 3 being negative controls guaranteeing cell death through the presence of a toxic medium such as DMSO. To perform Hoechst staining, the same seeding procedure was followed as for the MTT test, except for the pure PCL, since previous studies concluded that it did not induce apoptosis.
2.4. Cell Viability Assay (MTT)
The MTT is based on the enzymatic reduction of salt from yellowish tetrazolium to purple formazan. This occurs because the chemical compound can enter the mitochondria of living cells through its positive charge, leading to purple coloration and subsequent transformation into formazan. Then, the amount of reduced MTT can be determined using an absorbance reader. This type of assay is used to understand the cell viability of a substance, i.e., the capacity of each well in the plate for cell activity and proliferation [17]. This experiment was performed in the same way after 24, 48, and 72 h.
2.5. Apoptosis Assay
To determine the cellular apoptosis, the Hoechst 33342 method was used, which can detect and stain the nuclei of cells, emitting blue fluorescence when excited by ultraviolet light. This allows for a total cell count to be performed, since there is a difference in the intensity of the luminescence of living cells and those that have died [18]. The following procedure was performed on the 3 plates after 24, 48, and 72 h, respectively. After the cell incubation period in the wells and under conditions of 37 °C, 5% CO2, and 95% humidity, 100 μL was removed from the culture medium, and the cells were fixed by adding 100 μL of Casnoy’s fixative solution (3/1 proportion of absolute methanol and glacial acetic acid) for 5 min. After 5 min, the medium and fixative were removed from the wells. Next, 200 μL of fixative solution was added to each well, and we waited for 10 min. The fixative solution was removed and allowed to air dry for 30–60 min. Once fixed and dry, in dark conditions in the cabin, 200 μL of Hoechst solution with a concentration of 1 μg/mL in PBS was added. They were incubated in darkness at room temperature for 20–30 min (the plate was covered with aluminum foil). After some time, the coloring substance was removed and washed 3 times with distilled water. The excess water was left to dry in the air. The sample was taken to a Nikon Eclipse TE2000-U inverted microscope at a 50 μm scale for cell counting using 3 photographs of each well, ensuring that there were at least 200 stained cores per well.
2.6. Statistical Method
Statistical analysis was carried out using Stata V14 statistical software (Statacorp LLC, College Station, TX, USA) for both cell viability and cell death assays.
First, a descriptive analysis of the variables of interest was carried out using the mean and standard deviation, median and interquartile range, and minimum and maximum. Once these data were obtained, the normality of the variables was studied, and the non-parametric Kruskal–Wallis contrast was used, allowing us to compare the different concentrations of PCL in relation to time, and establishing a level of significance of α = 0.5 so that any value lower than p < 0.05 was considered significant.
3. Results
3.1. MTT Cell Viability Assay
To analyze the cell viability, we used an MTT to measure the absorbance achieved by each sample. A lower absorbance means a decrease in metabolic activity, meaning that high values are related to an increase in cell activity, and therefore a higher viability rate. The results obtained for the absorbance value in each of the materials were analyzed using the Kruskal–Wallis test to study whether there were differences both between the three materials and the different durations of time. The analysis relating the absorbance with the PCL concentration and control presented a value of p = 0.6217. On the other hand, the analysis focusing on the relationship between the absorbance of the PCL and the elapsed time resulted in p = 0.2019. The results for the absorbance of each material when using the MTT test, also considering the immersion time, are presented in the graph below (Figure 2) (Table A2, Appendix A).
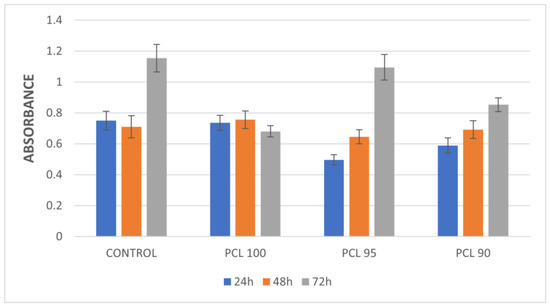
Figure 2.
Absorbance vs. materials and time after MTT.
3.2. Apoptosis or Cell Death Assay
The apoptosis assay was performed using Hoechst staining, since this method allows for a cell count of living and dead cells to be performed, because the cell nuclei are stained blue (Figure 3). The photographs of the stained cells were input into the Fiji Sc program [19], and using the Cell Counting.ijm macro (Figure 4), we determined the number of nuclei red and dead cells according to their anatomy.
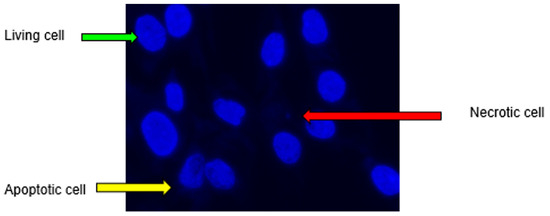
Figure 3.
Image of living, apoptotic, and necrotic cells.
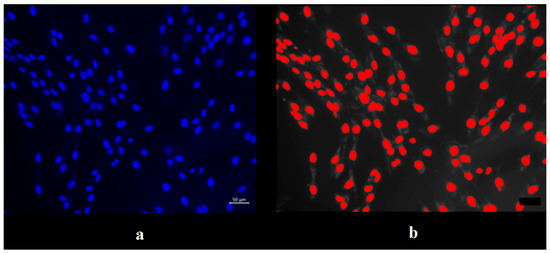
Figure 4.
(a). Cells stained with Hoechst. (b). Image using Fiji program.
In this study, the Kruskal–Wallis test was also performed to observe if there were significant differences. First, the number of live cells and the concentration of PCL were compared to the control, with p = 0.8371. Next, the number of living cells in which PCL was present was investigated in relation to time, leading to a p = 0.5647. Finally, the p-value for the relationship of dead cells with the concentration was 0.8655, and that for the relationship between the number of dead cells in the materials and the duration was 0.1801 (Table 1).

Table 1.
Number of living cells/well (mean ± standard deviation).
4. Discussion
After analyzing the results obtained in the cell viability assay (MTT), no significant differences were found between the materials; the results for the different samples according to absorbance and elapsed time were very similar, giving rise to values that were well above p < 0.05. The p-value delimits whether there are significant differences between samples. The results obtained in terms of absorbance according to the concentration of PCL turned out to be p = 0.6217, and according to the absorbance and number of hours elapsed, p = 0.2019, so the differences were not considered significant. These results were compared with each other and with the control group, guaranteeing that the materials under study did not cause any cell damage to the cultures apart from that which occurred in the control.
The cell viability results were positive, with a high rate of live cells per material, thus agreeing with different studies in which the biocompatibility of PCL was analyzed, such as that by Ghaedamini et al., in which it was concluded that PCL is a material with a high rate of fibroblast proliferation due to its high biocompatibility, which gives it favorable properties as a scaffolding material in bone regeneration [20]. Another study with favorable results with regard to the biocompatibility of polycaprolactone in periodontal ligament cells is the study by Safi et al., in which it was observed that the material was biocompatible and did not present harmful effects on the tissues supporting the tooth. In this research, the use of PCL in clinical applications such as dental implants was also analyzed, concluding that the materials characteristics are favorable for the union and growth of periodontal cells around it [21].
In the research carried out by Bazgir et al., the authors also concluded that synthetic polymers such as polycaprolactone demonstrated favorable results in terms of cell viability, demonstrating that they are non-toxic and exhibited high biocompatibility. The novelty of this study was that a synthetic material, i.e., PCL, was combined with natural materials such as collagen membranes, leading to satisfactory results for the development of cell lines in tissue engineering [22].
In the present in vitro research, PCL combined with strontium was used as one of the materials along with hydroxyapatite; this combination of components has already been analyzed thoroughly in other preclinical studies. In an article published by Kontogianni et al., both the toxicity and osteogenic capacity in bone regeneration of both materials when combined were analyzed. Nanohydroxyapatite is the main mineral component of bone, which, when combined with strontium ions, has been shown to have better biological and mechanical capabilities. The results of this study showed that strontium-ionized hydroxyapatite has higher levels of osteocalcin, a hormone related to bone production [23]. In another similar study carried out by Ran et al., it was shown that the addition of Sr to HA leads to an improvement in osteogenic capacity. As HA has good biocompatibility and osteoconductivity, when combined with Sr, it can enhance both properties to initiate osteogenesis [24].
To evaluate cell viability, it was decided to use the MTT test, which is a safe, reliable and widely accepted method in this field of research. However, this assay has a limitation when it comes to performing cell viability counting based on absorbance, because the MTT reagent tends to be introduced in the initial stages, during which the cell is entering the apoptosis phase. This can lead to false positive results, as metabolic activity can be falsely observed due to cells in the apoptosis phase being detected as living cells. For this reason, the assay was used alongside Hoechst 33342, a complementary technique because it allows for the identification of cells that have nuclei in an apoptotic state [25]. The results of the apoptosis assay showed that there were no significant differences between the materials in terms of the number of dead cells in each sample, presenting values that were very similar to each other and a value of p = 0.8371; this is well above p < 0.05, indicating that the numbers of live and dead cells for each material were very similar. According to Jeong et al., polycaprolactone is a polymer with a porous internal structure that allows for the penetration of cells from surrounding tissues such as periodontal ligament cells, thus increasing the capacity for biocompatibility and favoring bone regeneration. In addition, a study has shown that PCL combined with tricalcium beta phosphate (β-TCP) presented better mechanical and biological results, as it favored the induction of osteogenic cells, achieving a higher degree of mineralization [26].
This study analyzed the biocompatibility of PCL in its pure form or combined with other materials, and no significant differences were found between the different concentrations. Based on these results and after testing the biocompatibility of the three materials, future research can be carried out to evaluate the individual osteoinductive capacity of each of these materials to later use them in the field of guided bone regeneration. Regarding the limitations of this study, this in vitro test does not consider the evaluation of mechanical properties, as well as other factors such as the observation of cell adhesion to materials using SEM. A long-term biological study would also be necessary to analyze factors such as osteogenic differentiation.
The results of this in vitro study demonstrate that pure or combined PCL with strontium or nanohydroxyapatite are biocompatible materials that should be considered for use, since in addition to not producing toxic effects on the cells of the periodontal ligament, they seem to enhance osteogenic effects favoring bone regeneration. Once its biocompatibility has been proven, this emerging material should be analyzed in detail in future research to fully understand its regenerative potential, guaranteeing safety in treatments and efficacy in clinical practice.
5. Conclusions
The determined viability and cell proliferation are similar at different concentrations of polycaprolactone and after different durations of time (24, 48, and 72 h), as well as between the samples and the control group. The degree of apoptosis or cell death produced at different concentrations of polycaprolactone at different time points is similar to that of the negative control group, meaning that they are suitable for dental use. Polycaprolactone is a biocompatible material at different concentrations and in combination with other materials such as hydroxyapatite and strontium.
Author Contributions
Conceptualization, I.S.-B., J.M. and A.M.-C.; methodology, I.S.-B., L.M.R.-L., N.C.-M. and A.L.-N.; software, Á.R.-M.; validation, I.S.-B. and A.M.-C.; formal analysis, V.P.-F.; investigation, Á.R.-M.; resources, I.S.-B., L.M.R.-L., N.C.-M. and A.L.-N.; data curation, Á.R.-M.; writing—original draft preparation, Á.R.-M.; writing—review and editing, A.M.-C. and L.M.R.-L.; visualization, A.M.-C.; supervision, I.S.-B. and J.M.; project administration, I.S.-B. and J.M.; funding acquisition, J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially supported by the Research Group named “Avances en Salud Oral” of the University of Salamanca.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of the University of Murcia (CBE 599).
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is contained within the article.
Acknowledgments
We acknowledge support from the UA Bio2-ARIES and PTI-FAB3D, Consejo Superior de Investigaciones Científicas (CSIC), Spain.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| PLDSC | Periodontal ligament stem cells |
| PCL | Polycaprolactone |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| DMEM | Dulbecco’s modified Eagle medium |
| DMSO | Dimethyl sulfoxide |
Appendix A
Preparation and characterization of strontium-substituted hydroxyapatites:
Monohydrated calcium acetate, strontium acetate, dibasic ammonium phosphate, and ammonium hydroxide were purchased from Sigma-Aldrich and used without modification. The synthesis reaction has been included in the main body of this manuscript.
The products obtained were characterized by X-ray diffraction (XRD), and patterns were registered in a Bruker D8 Advance set-up with a Cu Kα radiation between 2θ 10 and 60 using a 0.024 step size and 0.2 s per step. Figure A1 left and right show that both selected compositions are made of a single-phase poorly crystalized hydroxyapatite. The crystal size was calculated using the Scherrer equation. Typical standard deviations are +10 A. Cell parameters have been determined using a whole pattern-fitting structure refinement. The Ca/P ratio of the powders was calculated from a multi-phase whole pattern analysis after a thermal treatment at 900 °C as follows:
Ca/P = (1.5% TCP + 1.667% OHAp)/100
Calcium and strontium content was determined by inductively coupled plasma (ICP) in a Perkin Elmer Optima 4300 DV. The specific surface area of the powders was calculated from a N2 isotherm using the BET method with a MS-13, Quantachrome Autosorb, Boynton Beach, FL, USA. Full details can be found in Angélica M Lara Nuñez, Universidad de Salamanca, PhD thesis (2017): http://hdl.handle.net/10261/390525 (accessed on 17 March 2024).

Table A1.
Ca/Sr ratios and samples codes of the prepared samples.
Table A1.
Ca/Sr ratios and samples codes of the prepared samples.
| Ca/P Ratio | Ca-Sr Content | a Lattice p | c Lattice p | Crystal Size (A) | SSA m2/g | |
|---|---|---|---|---|---|---|
| Sr0 | 1.505 | - | 9.439(3) | 6.885(2) | 807 | 95 |
| Sr10 | 1.505 | Ca7.93Sr1.07 | 9.462(6) | 6.906(4) | 1045 | 95 |
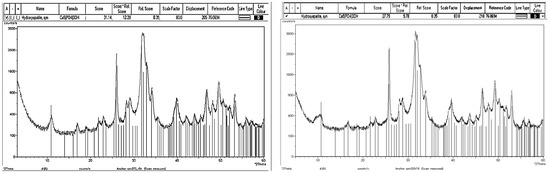
Figure A1.
XRD patterns of (left): Sr-free apatite (Sr0); (right) 10% Sr-enriched apatite (Sr10). Assignation to JCPDD 76−0694 hydroxyapatite syn is included.

Table A2.
Absorbance vs. materials and time after MTT.
Table A2.
Absorbance vs. materials and time after MTT.
| Immersion Time | Materials | Absorbance (Average) | Standard Deviation |
|---|---|---|---|
| 24 h | CONTROL | 0.750 | 0.061 |
| PCL 100 | 0.736 | 0.048 | |
| PCL 95 | 0.497 | 0.032 | |
| PCL 90 | 0.590 | 0.048 | |
| 48 h | CONTROL | 0.711 | 0.072 |
| PCL 100 | 0.756 | 0.057 | |
| PCL 95 | 0.646 | 0.045 | |
| PCL 90 | 0.693 | 0.058 | |
| 72 h | CONTROL | 1.155 | 0.089 |
| PCL 100 | 0.681 | 0.036 | |
| PCL 95 | 1.095 | 0.083 | |
| PCL 90 | 0.853 | 0.044 |
References
- Tomokiyo, A.; Wada, N.; Maeda, H. Periodontal Ligament Stem Cells: Regenerative Potency in Periodontium. Stem Cells Dev. 2019, 28, 974–9851. [Google Scholar] [CrossRef]
- Trejo Iriarte, C.G.; Ramírez Ramírez, O.; Muñoz García, A.; Verdín Terán, S.L.; Gómez Clavel, J.F. Isolation of periodontal ligament stem cells from extracted premolars. Simplified method. Rev. Odontológica Mex. 2017, 21, e12–e20. [Google Scholar] [CrossRef]
- Florencio-Silva, R.; Sasso, G.R.S.; Sasso-Cerri, E.; Simões, M.J.; Cerri, P.S. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. BioMed Res. Int. 2015, 2015, 4217463. [Google Scholar] [CrossRef]
- Liu, J.; Ruan, J.; Weir, M.D.; Ren, K.; Schneider, A.; Wang, P.; Oates, T.W.; Chang, X.; Xu, H.H.K. Periodontal Bone-Ligament-Cementum Regeneration via Scaffolds and Stem Cells. Cells 2019, 8, 5374. [Google Scholar] [CrossRef]
- Pardal-Peláez, B.; Gómez-Polo, C.; Flores-Fraile, J.; Quispe-López, N.; Serrano-Belmonte, I.; Montero, J. Three-Dimensional Scaffolds Designed and Printed Using CAD/CAM Technology: A Systematic Review. Appl. Sci. 2024, 14, 98775. [Google Scholar] [CrossRef]
- Murugan, S.; Parcha, S.R. Fabrication techniques involved in developing the composite scaffolds PCL/HA nanoparticles for bone tissue engineering applications. J. Mater. Sci. Mater. Med. 2021, 32, 936. [Google Scholar] [CrossRef]
- Bharadwaz, A.; Jayasuriya, A.C. Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater. Sci. Eng. C 2020, 110, 1106987. [Google Scholar] [CrossRef]
- Venkatesan, J.; Kim, S.K. Nano-Hydroxyapatite Composite Biomaterials for Bone Tissue Engineering—A Review. J. Biomed. Nanotechnol. 2014, 10, 3124–31408. [Google Scholar] [CrossRef]
- Stafin, K.; Śliwa, P.; Piątkowski, M. Towards Polycaprolactone-Based Scaffolds for Alveolar Bone Tissue Engineering: A Biomimetic Approach in a 3D Printing Technique. Int. J. Mol. Sci. 2023, 24, 161809. [Google Scholar] [CrossRef]
- Lino, A.B.; McCarthy, A.D.; Fernández, J.M. Evaluation of Strontium-Containing PCL-PDIPF Scaffolds for Bone Tissue Engineering: In Vitro and In Vivo Studies. Ann. Biomed. Eng. 2019, 47, 902–91210. [Google Scholar] [CrossRef]
- Prakash, J.; Shenoy, M.; Alhasmi, A.; Al Saleh, A.A.; C, S.G.; Shivakumar, S. Biocompatibility of 3D-Printed Dental Resins: A Systematic Review. Cureus 2024, 12, e12345. [Google Scholar] [CrossRef]
- Ghasemi, M.; Turnbull, T.; Sebastian, S.; Kempson, I. The MTT Assay: Utility, Limitations, Pitfalls, and Interpretation in Bulk and Single-Cell Analysis. Int. J. Mol. Sci. 2021, 22, 1282712. [Google Scholar] [CrossRef]
- Cordeiro, M.M.; Filipe, H.A.L.; Santos, P.; Samelo, J.; Ramalho, J.P.P.; Loura, L.M.S.; Moreno, M.J. Interaction of Hoechst 33342 with POPC Membranes at Different pH Values. Molecules 2023, 28, 564013. [Google Scholar] [CrossRef]
- Zhang, D.; Lin, W.; Jiang, S.; Deng, P.; Liu, L.; Wang, Q.; Sheng, R.; Shu, H.S.; Wang, L.; Zou, W.; et al. Lepr-Expressing PDLSCs Contribute to Periodontal Homeostasis and Respond to Mechanical Force by Piezo1. Adv. Sci. 2023, 10, 230329114. [Google Scholar] [CrossRef]
- Abuarqoub, D.A.; Aslam, N.; Barham, R.B.; Ababneh, N.A.; Shahin, D.A.; Al-oweidi, A.A.; Jafar, H.D.; Al-Salihi, M.A.; Awidi, A.S. The effect of platelet lysate in culture of PDLSCs: An in vitro comparative study. PeerJ 2019, 7, e746515. [Google Scholar] [CrossRef]
- DIN EN ISO 10993–12:2021; Biological Evaluation of Medical Devices—Part 12: Sample Preparation and Reference Materials. Beuth Verlag GmbH: Berlin, Germany, 2021.
- Benov, L. Improved Formazan Dissolution for Bacterial MTT Assay. Microbiol. Spectr. 2021, 9, e0163721. [Google Scholar] [CrossRef]
- Arvidsson, M.; Rashed, S.K.; Aits, S. An annotated high-content fluorescence microscopy dataset with Hoechst 33342-stained nuclei and manually labelled outlines. Data Brief 2023, 46, 10876917. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K. WNIH Image to ImageJ: 25 years of image analysis. Nature Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Ghaedamini, S.; Karbasi, S.; Hashemibeni, B.; Honarvar, A.; Rabiei, A. PCL/Agarose 3D-printed scaffold for tissue engineering applications: Fabrication, characterization, and cellular activities. Res. Pharm. Sci. 2023, 18, 566–579. [Google Scholar] [CrossRef]
- Safi, I.N.; Al-Shammari, A.M.; Ul-Jabbar, M.A.; Hussein, B.M.A. Preparing polycaprolactone scaffolds using electrospinning technique for construction of artificial periodontal ligament tissue. J. Taibah Univ. Med. Sci. 2020, 15, 363–37319. [Google Scholar] [CrossRef]
- Bazgir, M.; Saeinasab, M.; Zhang, W.; Zhang, X.; Min Tsui, K.; Maasoumi Sarvestani, A.; Nawaz, S.; Coates, P.; Youseffi, M.; Elies, J.; et al. Investigation of Cell Adhesion and Cell Viability of the Endothelial and Fibroblast Cells on Electrospun PCL, PLGA and Coaxial Scaffolds for Production of Tissue Engineered Blood Vessel. J. Funct. Biomater. 2022, 13, 28220. [Google Scholar] [CrossRef]
- Kontogianni, G.I.; Coelho, C.; Gauthier, R.; Fiorilli, S.; Quadros, P.; Vitale-Brovarone, C.; Chatzinikolaidou, M. Osteogenic Potential of Nano-Hydroxyapatite and Strontium-Substituted Nano-Hydroxyapatite. Nanomaterials 2023, 13, 188121. [Google Scholar] [CrossRef]
- Ran, L.; Liu, L.; Gao, J.; Pan, Y.; Ramalingam, M.; Du, X.; Liu, Y.; Cheng, L.; Shi, Z. Strontium-doped hydroxyapatite and its role in osteogenesis and angiogenesis. Int. J. Dev. Biol. 2023, 67, 137–14622. [Google Scholar] [CrossRef]
- Karakaş, D.; Ari, F.; Ulukaya, E. The MTT viability assay yields strikingly false-positive viabilities although the cells are killed by some plant extracts. Turk. J. Biol. 2017, 41, 919–92523. [Google Scholar] [CrossRef]
- Jeong, W.S.; Kim, Y.C.; Min, J.C.; Park, H.J.; Lee, E.J.; Shim, J.H.; Choi, J.W. Clinical Application of 3D-Printed Patient-Specific Polycaprolactone/Beta Tricalcium Phosphate Scaffold for Complex Zygomatico-Maxillary Defects. Polymers 2022, 14, 74024. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).