Abstract
Background/Objectives: Forest bathing (Shinrin-yoku) is a nature-based approach with potential preventive health relevance. This review summarizes evidence on its effects on immune function, stress physiology, and neuroprotective pathways. Methods: A narrative review of peer-reviewed studies was conducted using major scientific databases, including observational and interventional research assessing physiological or neurocognitive outcomes following forest exposure. Results: Forest bathing is associated with enhanced natural killer (NK) cell activity, modulation of inflammatory cytokine profiles, reductions in cortisol levels, and shifts toward parasympathetic autonomic dominance. Evidence also suggests a contributory role of tree-derived biogenic volatile organic compounds and phytoncides in immune and stress-regulatory effects. Emerging findings indicate potential benefits for cognitive restoration, emotional regulation, and neurotrophic signaling; however, substantial heterogeneity in study design, exposure characteristics, and outcome measures limits direct comparability and causal inference. Conclusions: Current evidence supports forest bathing as a promising, low-risk strategy for supporting immune resilience, stress regulation, and neurocognitive well-being within a preventive health framework. Preliminary findings also suggest potential benefits in chronic neurological conditions, supporting its neuroprotective role within multimodal neurorehabilitation strategies. Standardized intervention protocols, mechanistic biomarkers, and longitudinal studies are required to strengthen clinical relevance and guide evidence-based integration into public health and lifestyle medicine.
1. Introduction
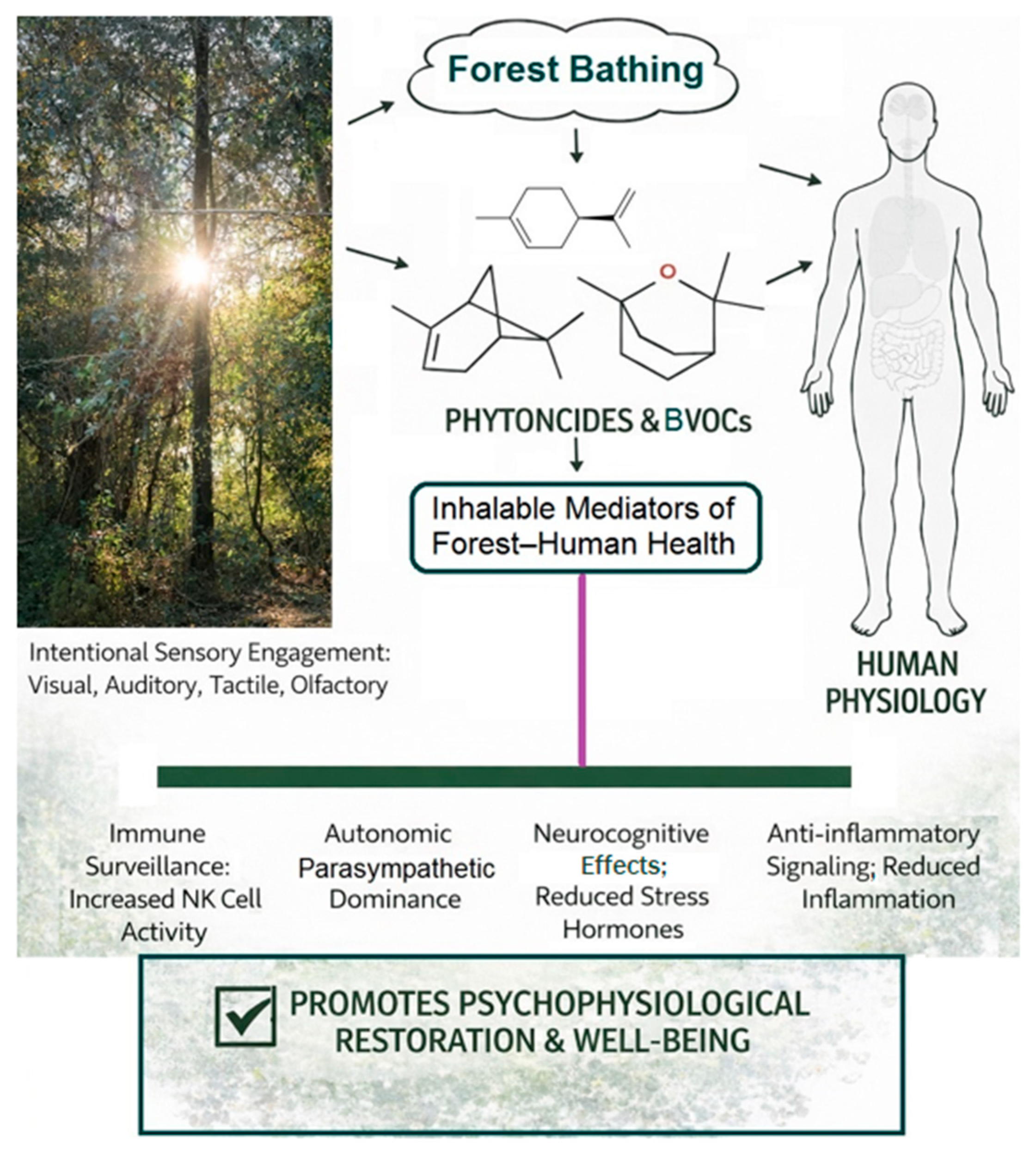
The increasing global prevalence of stress-related disorders [1], immune dysregulation [2], and neurocognitive decline [3] has intensified the search for accessible, non-pharmacological strategies capable of supporting preventive maintenance. Modern lifestyles characterized by urbanization, environmental overstimulation, and reduced contact with natural environments [3] have contributed to physiological burdens that accumulate across the lifespan. Within this context, forest bathing, or Shinrin-yoku [4], has emerged as a structured nature-based practice with potential to mitigate some of these effects. More specifically, forest bathing refers to immersion in the forest atmosphere through multisensory engagement rather than physical exercise, as it involves simply being present in nature and connecting with it. Interestingly, in Japan, Shinrin-yoku is prescribed as a medical intervention in a manner similar to conventional treatments, with structured and repeated sessions administered over time [5]. Originally conceptualized as a public health initiative, it involves slow, mindful immersion in green environments with the aim of engaging the mentioned multiple sensory pathways and promoting psychophysiological restoration [4,6,7]. Forest bathing encourages attentive interaction with green surroundings and is distinct from general outdoor recreation because it emphasizes intentional sensory engagement, reduced cognitive load, and a contemplative pace. The forest environment provides a complex array of stimuli [8], including visual fractal patterns, natural sounds, organic scents, and biogenic volatile organic compounds [9,10] (BVOCs, Scheme 1).
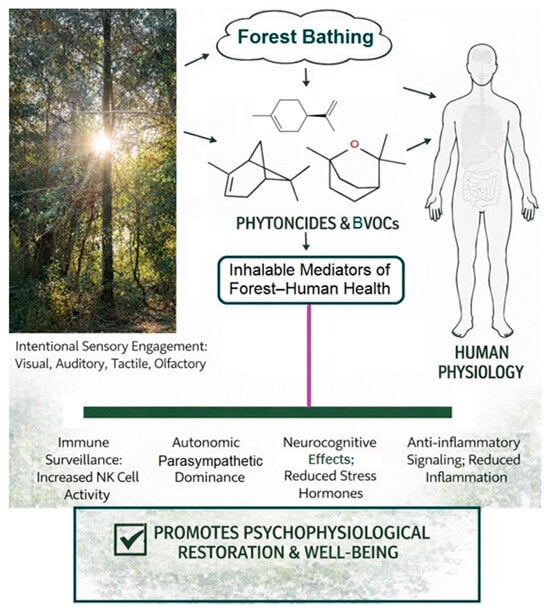
Scheme 1.
Schematic representation of the psychophysiological pathways of Forest Bathing (Shinrin-yoku) and the role of biogenic volatile organic compounds (Green photograph courtesy of G.N. Roviello).
These environmental elements are thought to elicit physiological responses through multiple pathways (Scheme 1), potentially mediated by bi-directional neuro-immune and metabolic–endocrine interactions, a mechanism well supported in the literature for these regulatory systems [11,12]. Remarkably, the clinical field study of Yu et al. suggested that forest exposure can improve cardiovascular function and psychological well-being, reducing heart rate, blood pressure, and negative mood while increasing positive emotion [13]. In parallel, emerging evidence from the same study, along with findings from the intervention study by Li et al., indicated that forest exposure can enhance cognitive restoration and recovery from fatigue, reflected in increased vigor, decreased fatigue, improved subjective sleep quality, reductions in tension, anger, depression, and confusion, and lower anxiety levels [13,14]. Previous studies have already addressed the relevance of forest bathing in preventing respiratory diseases and mitigating the impact of pandemics such as COVID-19 [15,16,17,18], as well as the necessity of protecting natural environments, including riparian [19,20] and coastal vegetation [21], to ensure that populations can benefit from the health-enhancing effects of nature, including the passive inhalation of tree-emitted volatile organic compounds. These contributions highlight the broader ecological and public health implications of maintaining biodiverse [22], intact natural landscapes capable of supporting human physiological resilience [22,23]. Despite growing interest, the scientific literature on forest bathing remains heterogeneous. Studies vary widely in exposure duration, environmental characterization, participant demographics, and biomarker selection [24]. Many investigations employ small sample sizes or short-term designs, limiting the ability to draw firm conclusions about long-term health impacts or underlying mechanisms [24]. Furthermore, the relative contributions of sensory, psychological, and biochemical components of forest environments remain insufficiently delineated [25]. These gaps highlight the need for a comprehensive synthesis that integrates findings across immune, endocrine, autonomic, and neurocognitive domains in relation to forest bathing [26]. In this context, the purpose of this narrative review is to examine forest bathing as a potential component of preventive medicine, with particular emphasis on aspects including immune modulation, stress physiology, and neuroprotective mechanisms [3]. By consolidating current evidence, including observed improvements in immune profiles, stress markers, and hematological parameters, alongside the identification of key variables like forest features and sensorial dimensions [27,28], this work aims to clarify the biological plausibility of forest-based interventions, identifying consistent patterns across studies, and ultimately highlighting methodological limitations that must be addressed to advance the field. In doing so, it seeks to provide a scientific foundation for the integration of forest bathing, supported by evidence of improved cardiovascular, metabolic, and antioxidant indexes and its recent recognition in clinical guidelines for hypertension [29,30], into broader preventive health strategies while outlining priorities for future research to strengthen clinical relevance.
Methodology
To ensure a comprehensive overview of the current scientific landscape regarding forest bathing and its physiological impacts, a comprehensive literature search was conducted across multiple scientific databases, including Google Scholar, PubMed, Scopus, and Web of Science. The search strategy was performed using combinations of terms, including “forest bathing”, “Shinrin-yoku”, “forest therapy”, “nature-based interventions”, “forest medicine”, “immune modulation”, and “neuroprotection”. The search strategy focused on peer-reviewed articles exploring the intersection of factors such as forest-related practices and their links to immune modulation and psychophysiological processes with stress physiology, neuroprotective and neurocognitive outcomes. An initial search yielded 346 results. These records were subsequently screened based on specific inclusion criteria: studies published in English in peer-reviewed journals; research providing primary data or comprehensive syntheses on physiological, immunological, neuroprotective, or neurocognitive outcomes related to forest exposure; and studies involving both observational and interventional designs across diverse demographic groups. Additional books and theses were consulted where relevant to the thematic scope of this work. Following the removal of duplicates, retracted articles, and papers that were not peer-reviewed, 116 references were formally included in the bibliography. Although the full text cites 116 references, 30 are used to provide general context, while the core synthesis draws on more than 80 studies and further elaborates on selected literature found in the Introduction. Notably, a substantial portion of the literature cited in our work is predominantly recent; the papers published within the last five years represent about 70% of the total included references, revealing a surging contemporary interest in forest bathing for supporting immune regulation, stress reduction, neuroprotection, and neurocognitive resilience.
2. Biologic Responses and Physiological Outcomes of Forest Bathing
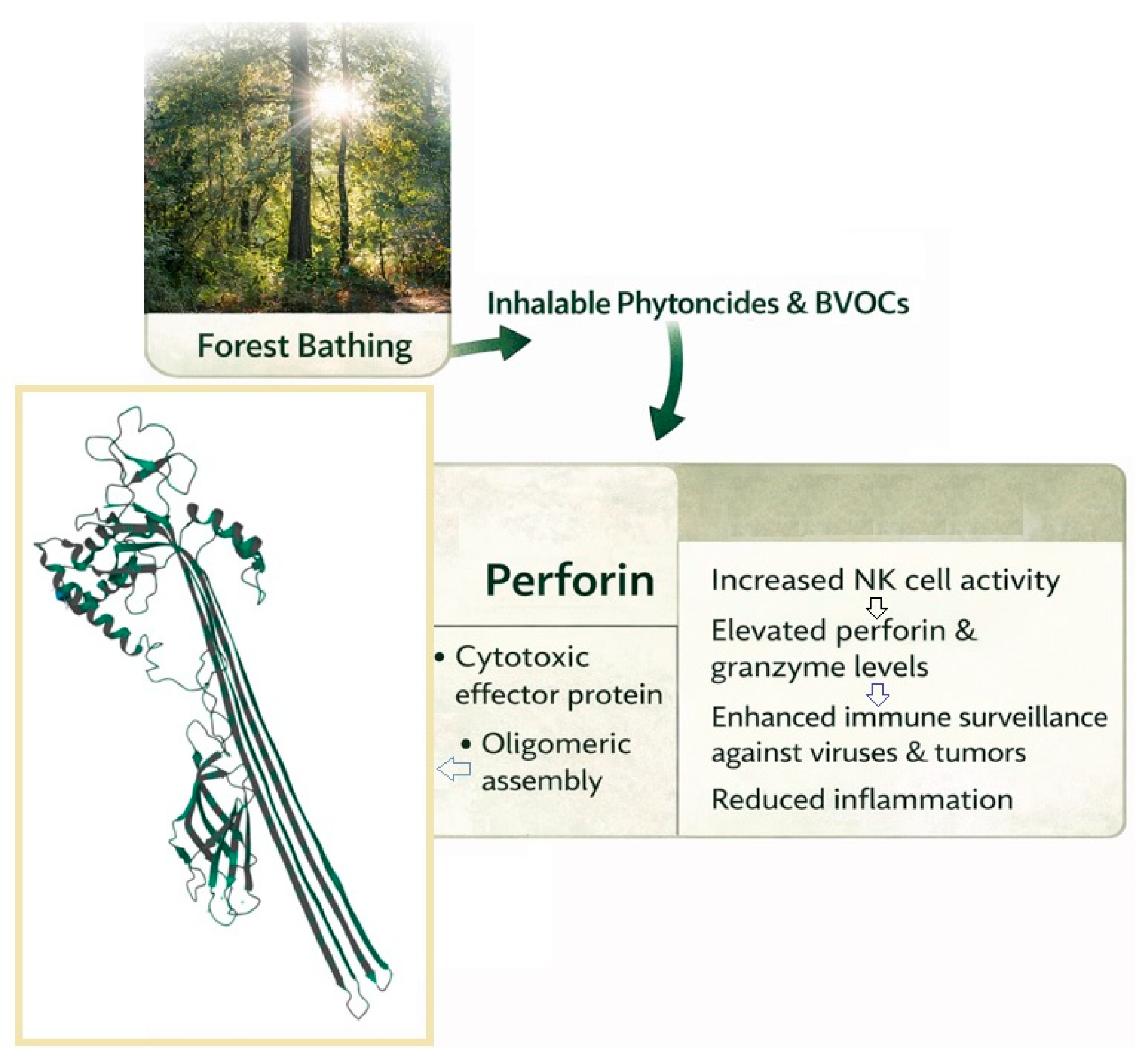
Human interaction with forest environments has historically been associated with feelings of relaxation and well-being, largely due to sensory factors such as visual greenery, natural sounds, clean air, and forest-derived aromas [15]. In Japan, this long-standing cultural practice was formally investigated through scientific research beginning in the early 2000s, leading to the emergence of Forest Medicine as a distinct interdisciplinary field [31]. Forest medicine draws upon concepts from environmental science, preventive medicine, and complementary health approaches to examine how forest exposure influences human physiological and psychological health [32]. Research in this area consistently indicates that forest bathing supports health maintenance not only by reducing stress and regulating autonomic nervous system activity, but also improving sleep quality, and enhancing emotional well-being. Together, these outcomes suggest that forest-based interventions may contribute meaningfully to the prevention of chronic, stress-related non-communicable diseases [29]. Mechanistically, a substantial body of evidence highlights immune modulation as a key biological effect of forest bathing [33]. Controlled observational studies involving short forest stays, and consequent exposure to BVOCs and phytoncides, have demonstrated increased activity and abundance of natural killer (NK) cells, alongside elevated levels of intracellular cytotoxic molecules, specifically effector proteins such as perforin (Scheme 2) and granzymes, which serve as a primary defense mechanism in immune surveillance against compromised or virally infected cells [34].
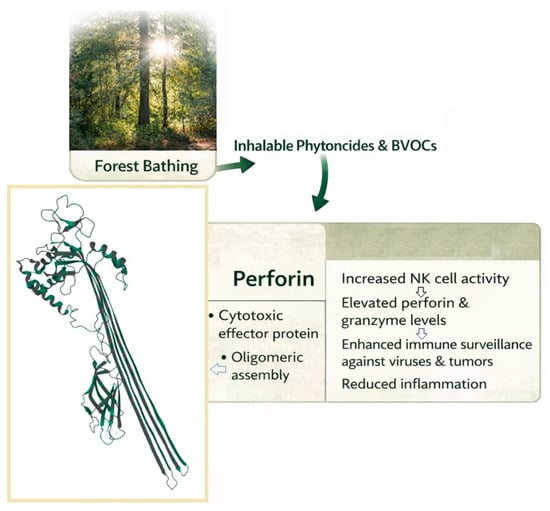
Scheme 2.
Schematic illustrating how forest bathing-derived inhalable compounds (phytoncides and BVOCs) may enhance immune surveillance. Exposure is associated with increased NK cell activity and elevated cytotoxic effector molecules, exemplified by perforin, shown here as a cryo-electron microscopy-derived three-dimensional structure (PDB ID: 7PAG [35]; https://www.rcsb.org/structure/7PAG (accessed on 9 January 2026)).
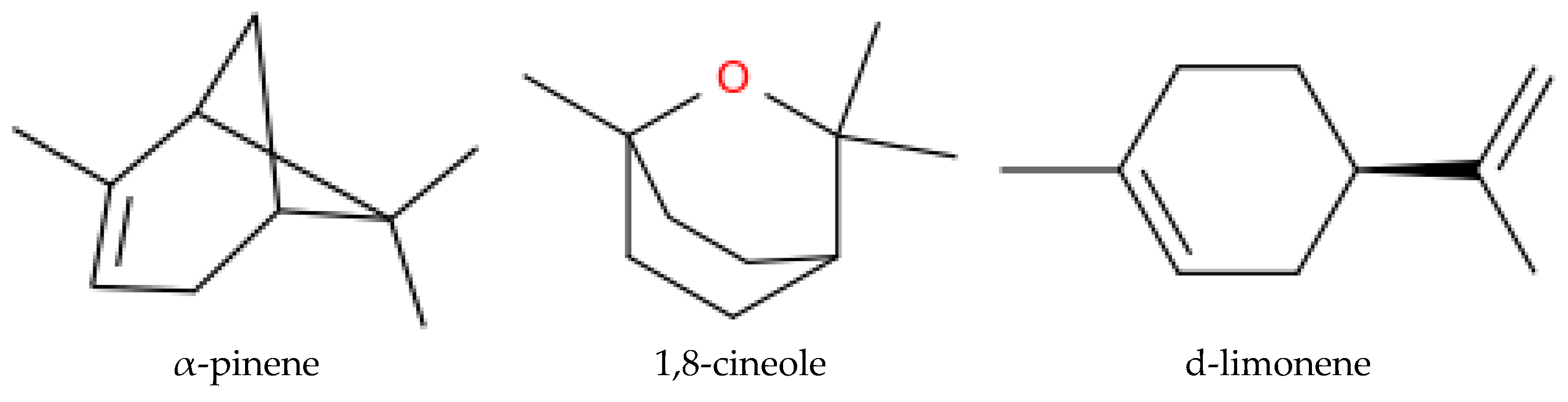
These immune enhancements are typically accompanied by reductions in stress-associated catecholamines and glucocorticoids, indicating a link between neuroendocrine regulation and immune function. Importantly, the persistence of elevated immune activity for several weeks following forest exposure suggests that even periodic engagement with forest environments may produce lasting biological effects [36,37]. Comparative studies further show that similar durations of urban exposure (e.g., walking in a city center or high-traffic areas) do not elicit these immune responses, revealing the distinct physiological influence of forest settings [38,39,40]. Growing mechanistic evidence suggests that phytoncides, including volatile monoterpenes and essential-oil constituents such as α-pinene, 1,8-cineole, d-limonene (Figure 1), and complex mixtures derived from Chamaecyparis (hinoki), Thujopsis dolabrata (hiba), and Cryptomeria japonica (sugi) stem oils, exert immunomodulatory activity by enhancing intracellular activation programs in human cytotoxic lymphocytes, thereby providing a plausible molecular basis for the health-supporting effects associated with forest environments [41].
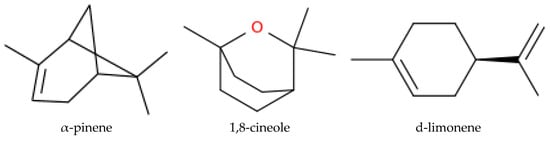
Figure 1.
Chemical structures of the three purified phytoncides, α-pinene, 1,8-cineole, and d-limonene, examined alongside the complex plant stem oils reported in [41].
Interestingly, experimental studies conducted in non-forest environments using controlled phytoncide exposure have reported immune activation patterns comparable to those observed during forest bathing, including enhanced immune activity and reduced stress hormone levels [37,42]. Beyond immunological outcomes, forest bathing has been associated with cardiovascular regulation, including lowered blood pressure, reduced heart rate, and improved heart rate variability. Psychological benefits, such as reduced anxiety, depression, fatigue, and improved mood and sleep quality, further reinforce the role of forest bathing as a preventive health intervention [29,43].
The randomized controlled field study of Ochiai et al. provides growing evidence that forest bathing exerts measurable preventive health effects through combined immunological, psychological, and stress-regulatory pathways [44]. Moreover, the randomized controlled crossover laboratory study by Longman et al. demonstrated that brief exposure to forest acoustic environments, compared to urban or industrial soundscapes, significantly enhances positive affect in adults, perceived restoration, and cognitive performance [45]. The immune changes following forest bathing sessions appear closely linked to concurrent shifts in mood and stress-related responses, supporting a psychoneuroimmunological mechanism underlying forest-based interventions [44,46]. Importantly, evidence suggests that even low-intensity and low-dose forest bathing sessions, such as two to three standardized exposures or short guided visits during work breaks, can yield benefits comparable to those observed in longer or more intensive interventions for mental well-being and stress resilience [47]. Real-world workplace studies further highlight the feasibility of integrating forest exposure into daily routines, with observed improvements in immune markers, and work-related attitudes among urban employees [47,48]. At the same time, conceptual analyses of the forest bathing literature emphasize the need to better characterize human–forest interactions, environmental features, and experiential conditions that shape health outcomes [48,49]. Together, these findings reinforce forest bathing as a practical, scalable preventive health strategy while demonstrating the importance of refined methodological frameworks to support clinical translation and evidence-based implementation [29,50,51]. To clarify the multidimensional nature of the above-mentioned health benefits, the table below provides a systematic overview of key studies investigating the physiological and psychological impacts of forest bathing, categorized by health domain, intervention characteristics, and primary outcomes (Table 1).

Table 1.
Physiological and psychological effects of forest bathing.
3. Integrated Systemic Regulation Underlying Forest-Based Health Effects
In this section we examine the physiological mechanisms through which forest environments exert systemic effects on human health and investigate how exposure to natural settings modulates immune function, attenuates inflammatory activity, and influences neuroendocrine and autonomic regulation. In particular, we focused on the multisensory and biochemical inputs characteristic of forest ecosystems, which have been shown to reduce stress reactivity, alter cytokine and hormonal profiles, and promote homeostatic stability. Together, these processes illustrate the capacity of forest environments to act as complex bioregulatory stimuli, contributing to improved physiological resilience and supporting both physical and psychological well-being.
3.1. Immunological Resilience and Inflammatory Regulation
Immersive exposure to natural forest environments has been shown to influence key physiological processes that maintain immune balance and contribute to lowering blood pressure [59]. Immersive exposure to low-latitude evergreen broad-leaved forests has been shown to improve sleep quality, mood, and immune function, supporting its role as a non-pharmacological intervention for holistic well-being [27]. In addition, compounds released by trees, such as the already mentioned phytoncides, may enhance the activity and cytotoxic function of CD8+ T and NKT cells, strengthening the body’s defenses against infections and abnormal cell growth [32,60]. Forest exposure also appears to modulate the expression of activating and inhibitory receptors on immune cells, supporting both immune activation and regulatory balance. Alongside these cellular effects, nature immersion can shift cytokine profiles toward an anti-inflammatory state, helping reduce systemic inflammation [61,62,63]. Overall, these interactions suggest that forest environments support immune resilience, promote inflammatory regulation, and offer a non-pharmacological strategy to improve overall health and prevent chronic disease [59,64]. The convergence of immunological modulation and neuroendocrine stabilization supports the concept of forest bathing as a systemic intervention operating through the psycho-neuro-endocrino-immune network, with implications for the regulation of physiological homeostasis [65]. Rather than merely suppressing or stimulating a single pathway, the multi-sensory and chemical input from the forest environment appears to facilitate a state of biological readiness, where the synchronization of cellular defenses and inflammatory control provides a foundation for the broader neurocognitive resilience currently being explored in clinical research [30]. This integrative perspective is further supported by emerging evidence from natural-product-based and biomolecular research, highlighting how bioactive compounds, peptides, and nature-inspired molecules can modulate immune defense, inflammatory signaling, metabolic pathways, and neurocognitive resilience across infectious, inflammatory, and neurodegenerative contexts [66,67,68,69,70,71,72,73].
3.2. Systemic Bioregulation: Autonomic Balance, Neuroendocrine Shifts, and Environmental Specificity
Field-based and review evidence consistently demonstrates that forest bathing is associated with measurable reductions in physiological stress and promotes autonomic nervous system regulation [74]. Controlled crossover experiments conducted across multiple forest sites in Taiwan and Japan show that short periods of walking or passive viewing in forest environments lead to lower salivary stress-related hormone levels, reduced blood pressure and pulse rate when compared with urban settings [13,53]. These physiological changes indicate a shift toward a relaxed and adaptive stress response, reflected by improved sympathovagal balance [75] and the previously mentioned harmonizing effects of natural environments across cardiovascular, endocrine, neural, and immune systems may play a particularly important role in stress-related outcomes [76,77]. Although methodological heterogeneity and small sample sizes limit causal inference, the convergence of evidence suggests that forest environments reduce allostatic load and enhance physiological resilience to stress [78]. In other words, these findings position Shinrin-yoku as a promising nature-based strategy for stress management, health promotion, and disease prevention within broader preventive-medicine and green public-health frameworks, while highlighting the need for larger, longitudinal, and methodologically robust studies [79,80,81]. Importantly, forest bathing has been increasingly studied for its ability to reduce stress and improve mental well-being [24], with research showing that spending time in forest environments can lower physiological markers of stress, including not only salivary but also serum cortisol, compared to urban settings, indicating a calming effect on the body’s stress-response system [52]. The intervention study of Li et al., which used a randomized crossover design, has also reported improvements in emotional well-being, with participants exhibiting reduced fatigue, better sleep quality, and increased positive feelings after forest exposure [14]. Biological changes, including elevated levels of serotonin, oxytocin, and insulin-like growth factor-1 [54], suggest that forest bathing may influence neuroendocrine pathways, contributing to both mental and physical health benefits [29]. Broader reviews on natural environments confirm that exposure to greenery generally supports stress reduction, in line with theories such as the Biophilia Hypothesis and Stress Recovery Theory [82,83,84], which posit an innate human affinity for life-sustaining landscapes and an evolved capacity for rapid physiological recovery when perceiving natural cues as safety signals [85]. These frameworks suggest that the restorative effects of the forest arise from evolved neural mechanisms that favor resource-rich environments, reducing modern stress responses through basic evolutionary tendencies [86]. Moreover, forest-based interventions have shown potential in managing chronic health conditions like hypertension in older adults [28]. This evidence has catalyzed an international shift toward formalizing forest-based health strategies, moving beyond individual well-being to address the “civilization diseases” associated with sedentary, urban lifestyles [87,88]. Comparative analyses of forest programs, particularly between Asian pioneers and European countries, demonstrate that integrating these natural assets into national preventive healthcare frameworks and social forestry dimensions can significantly improve the quality of life on a population level [28,89]. Beyond the generalized restorative effects of greenery, the clinical efficacy of forest immersion is increasingly viewed as a function of specific ecological characteristics that target physiological and psychological vulnerabilities [90]. Empirical data from diverse forest landscapes further suggests that these environments act as complex bioregulators, capable of stabilizing hemodynamic parameters and modulating the neurochemical substrates of affect. Seasonal variations and forest types appear to influence these effects significantly; for instance, visits to Cinnamomum camphora forests were found to reduce systolic and diastolic blood pressure and increase oxygen saturation especially in summer and autumn [56,91]. Similarly, different plant communities, such as mixed coniferous forests or mixed broad-leaved forests, exhibit distinct impacts on stress relief, emotional well-being, and physiological indicators like salivary stress-related hormones. These findings highlight the potential of thoughtfully designed urban green spaces and diverse plant communities to enhance public health, offering a scientific foundation for planning and optimizing natural environments for human well-being as further supported by the on-site experimental study of Zhang et al. [56]. The dynamic interplay between specific forest compositions and the resulting neuroendocrine shifts suggests that the “dose” of nature is not a generic variable, but one modulated by botanical diversity and seasonal bioactivity. This complexity seems to indicate that the body not only reacts to forest-derived sensory and biochemical inputs, but also undergoes integrated physiological shifts that may contribute to cognitive and neuroprotective outcomes.
4. Neurocognitive and Potential Neuroprotective Effects of Forest and Tree-Based Nature Exposure, with Associated Psychophysiological Responses
Forest environments are increasingly viewed as neurobiological systems that influence central nervous system function through integrated sensory, psychological, and biophysical inputs. Through multisensory cues forest bathing engages stress-responsive neural circuits, supports adaptive neuroplasticity, and may contribute to cognitive regulation by modulating prefrontal, limbic, and hippocampal networks involved in attention, emotional regulation, memory, and stress processing. The pragmatic controlled trial of McEwan et al. demonstrated that forest bathing improves psychological wellbeing and autonomic regulation, showing equivalence to an established wellbeing intervention [92]. Studies show that interacting with natural environments is associated with improvements in cognitive function, emotional regulation, memory, and sensorimotor processing, often mediated through region-specific changes in the brain, including stress-sensitive areas such as the amygdala and subiculum [93,94]. Additionally, exposure to natural environments has been shown to enhance heart rate variability and also reduce rumination, a key cognitive marker of psychological distress characterized by repetitive, self-referential negative thought patterns, indicating improved autonomic regulation and stress resilience [95]. Controlled trials, including the first pragmatic study in the United Kingdom, indicate that exposure to natural environments can provide wellbeing improvements comparable to established psychological interventions, with sustained effects on mood, nature connection, and physiological markers of neurocognitive health [95]. Systematic reviews further suggest that forests exert stronger neuroplastic effects than other types of green spaces, while residential greenness within proximal distances also offers measurable benefits [96]. These findings highlight the potential of forest and tree-mediated interventions to support adaptive neuroplasticity, mental health, and long-term brain resilience, suggesting the importance of integrating green spaces into public health strategies and sustainable urban design [92,97,98]. These integrated effects also align with emerging evidence suggesting potential neuroprotective benefits of forest and tree-based environments, as further outlined in Table 2 together with their cognitive implications.
Cognitive and Neural Effects of Forest Environments
Recent research highlights the significant impact of forests on human cognitive function and brain health across the lifespan [99], with structural and functional neuroimaging studies identifying the prefrontal cortex, amygdala, and hippocampus as regions consistently influenced by environmental exposure and showing that greener, less urbanized settings are associated with enhanced structural integrity of these regions and reduced stress-related neural activity [100,101]. The controlled field study by Ramanpong et al. demonstrated that, in elderly populations, both structured and self-guided forest bathing significantly improved key cognitive domains, including attention, working memory, and creativity [26], as evidenced by performance gains in tasks such as the Stroop test, Forward Digit Span Task, and Remote Associates Test [102]. Notably, the benefits of forest bathing appear more strongly linked to the frequency of forest engagement rather than the total time spent or distance covered, suggesting that regular, repeated exposure is more critical than prolonged sessions [26,57,58]. The cognitive improvements are likely mediated by multiple mechanisms, including stress reduction, restoration of attentional resources (as proposed by Attention Restoration Theory), and multisensory stimulation that enhances cognitive flexibility and creative thinking [26,103]. Epidemiological studies further indicate that increased residential green space may slightly slow cognitive decline and support brain health, potentially through promoting physical activity, providing restorative environments, and reducing exposure to air pollution [104]. While some complex cognitive measures, such as backward digit span performance, may be less responsive in older adults, overall evidence reveals the potential of forest-based and green space interventions as accessible, low-cost strategies for maintaining cognitive function and promoting neurocognitive resilience in aging populations [26]. Complementing these findings, evidence from neuroimaging [93,94], environmental exposure studies [105], and mechanistic frameworks [106] indicates consistent modulation of stress-related neural circuits and associated cognitive benefits [107] (Table 2).

Table 2.
Neurocognitive and potential neuroprotective effects of forest bathing and green space exposure.
Table 2.
Neurocognitive and potential neuroprotective effects of forest bathing and green space exposure.
| Study/Focus | Intervention/Exposure | Key Cognitive/Neural Outcomes | Ref. |
|---|---|---|---|
| Environmental and Neurophysiological Studies | Urban vs. rural/green environments | Modulation of amygdala activity, hippocampal subfields, and physiological stress-recovery markers | [93,94] |
| Self-Guided and Structured Forest Bathing Programs | Multiple short forest visits or multi-session forest-healing programs over several weeks | Improvements in attention, working memory, creativity, and global cognitive function; enhanced memory, orientation, and emotional stability; frequency-dependent gains in attentional control | [26,108] |
| Forest Exposure and Alzheimer’s Disease and Related Dementias (ADRD) Risk | Green space exposure defined by duration, proximity, and frequency | Potential deceleration of cognitive decline. Preserved regional brain volumes and cortical thickness; modulation of neuroplasticity and inflammatory markers. | [96,99,105] |
| Theoretical and Mechanistic Studies | Forest bathing and green space exposure | Enhanced affective recovery, attentional restoration, and domain-specific cognitive resilience, supported by multisensory, stress-reduction, and neuroplasticity-related mechanisms | [82,83,84,106] |
| Forest walking or structured forest-therapy programs in middle-aged and older adults with elevated blood pressure | Forest walking or structured forest-therapy programs in middle-aged and older adults with elevated blood pressure | Improved mood, reduced anxiety, enhanced autonomic regulation, and significant blood-pressure reduction; psychophysiological pathways associated with lower neurocognitive decline and neurodegenerative risks | [13,29,53,56] |
| Chronic Neurological and Neurodegenerative Conditions (e.g., Stroke, Parkinson’s Disease, Dementia, Multiple Sclerosis, and age-related cognitive decline) | Forest therapy and nature-based rehabilitation programs; observational and interventional studies across neurological populations | Limited but suggestive benefits, including improvements in depressive and anxiety symptoms, functional recovery, autonomic regulation, quality of life, and domain-specific cognitive outcomes relevant to neurorehabilitation | [92,95,107] |
In other terms, forests may act as complex bio-social environments capable of modulating neural circuits, immune pathways, and cognitive processes in concert. They are hypothesized to function as dynamic neuroecological systems, where psychological, sensory, and biophysical cues jointly influence neural plasticity and cognitive performance, with this integrative view raising the possibility that distinct forest attributes, ranging from tree canopy chemistry to spatial structure, could be mapped onto specific neurocognitive outcomes. Collectively, the findings of our work support the integration of forest-based therapies into preventive medicine frameworks, while also emphasizing the need for rigorously designed randomized controlled trials to strengthen causal inference and clinical translation [32,43,109,110].
5. Conclusions
Shinrin-yoku, or forest bathing, is a nature-based practice that supports preventive health by positively influencing the brain function, stress responses, and immune system [111]. Spending time in forests has been shown to increase immune activity, balance inflammatory signals, and lower stress hormones like cortisol, promoting immune resilience and reducing chronic inflammation [74]. It also enhances autonomic nervous system function, improving heart rate variability, lowering blood pressure, and fostering relaxation. Additionally, exposure to natural environments can support cognitive and emotional health, improving attention, memory, and creativity, likely through sensory engagement and stress reduction. These benefits highlight forest bathing as a practical, low-cost approach for maintaining physical and mental well-being, with potential applications in lifestyle medicine and public health initiatives [29]. Forest bathing represents a scientifically grounded, ecologically sustainable approach to preventive medicine, capable of influencing multiple physiological systems relevant to modern chronic disease burdens [31,112]. Regular immersion in forest environments may modulate immune pathways, a process closely linked to microglial-mediated immune surveillance in the brain [113], reduce systemic stress responses, and support neuroprotective pathways that are linked to cognitive resilience and emotional well-being [114]. Studies have examined the potential of forest therapy in chronic neurological conditions such as stroke (rehabilitation phase), Parkinson’s disease, dementia, and multiple sclerosis [107]; although evidence-based results are lacking, preliminary findings suggest possible benefits, supporting its proposed integration as a complementary component of multimodal neurorehabilitation [107]. Integrating forest-based practices into lifestyle medicine, urban planning, and community health programs may offer a practical strategy to counteract the health impacts of urbanization, sedentary behavior, and chronic stress [28,89]. As the field advances, interdisciplinary collaboration will be essential to fully elucidate the therapeutic potential of nature-based interventions and to position forest bathing as a validated component of preventive healthcare [22,115,116]. Importantly, forest bathing should be viewed not as a stand-alone therapeutic intervention, but as a complementary, low-risk strategy within lifestyle medicine and public health prevention frameworks. The convergence of immune, autonomic, and neurocognitive effects supports its biological plausibility as a means of enhancing physiological resilience in the context of chronic stress, urbanization, and aging populations. Future research priorities should include the development of standardized intervention protocols, improved environmental characterization of forest settings, and the use of robust mechanistic biomarkers across immune, endocrine, and neural systems. Additional efforts should also focus on the design and implementation of both healing gardens and indoor forest-bathing environments within public buildings such as schools, hospitals, churches and other places of worship, as well as other community facilities. Longitudinal and controlled studies on forest bathing will be essential to clarify dose–response relationships, durability of effects, and population-specific benefits. As interdisciplinary collaboration advances, forest bathing may become an evidence-informed component of preventive healthcare strategies that integrate ecological, physiological, and public-health perspectives.
Author Contributions
A.B., S.S. and G.N.R. have contributed substantially to all stages of the work, including the preparation of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No data available from this literature review.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ADRD | Alzheimer’s Disease and Related Dementias |
| ANS | Autonomic Nervous System |
| BVOCs | Biogenic Volatile Organic Compounds |
| CD8+ T | Cytotoxic T cells |
| DMN | Default Mode Network |
| FDST | Forward Digit Span Task |
| HPA | Hypothalamic–Pituitary–Adrenal |
| HRV | Heart Rate Variability |
| IGF-1 | Insulin-like Growth Factor-1 |
| NK | Natural Killer |
| NKT | Natural Killer T |
| RAT | Remote Associates Test |
| sgPFC | Subgenual Prefrontal Cortex |
| sIgA | Salivary Immunoglobulin A |
| SRT | Stress Recovery Theory |
| VOCs | Volatile Organic Compounds |
References
- Werner, F.; Schumacher, F.; Mühle, C.; Adler, W.; Schug, C.; Schäflein, E.; Morawa, E.; Kleuser, B.; Kornhuber, J.; Erim, Y. Psychosomatic-psychotherapeutic treatment of stress-related disorders impacts the sphingolipid metabolism towards increased sphingosine and sphingosine-1-phosphate levels. Eur. Arch. Psychiatry Clin. Neurosci. 2025, 275, 2049–2058. [Google Scholar] [CrossRef]
- Henderson, L.A.; Abraham, R.S.; Ahmed, A.; Blount, L.; Canna, S.W.; Chaimowitz, N.S.; Chandrakasan, S.; Coates, B.; Connelly, J.A.; Cooper, M.A. Multidisciplinary approach to treating complex immune dysregulation disorders: An adaptive model for institutional implementation. Front. Immunol. 2025, 16, 1519955. [Google Scholar] [CrossRef]
- Xia, M.; Ntim, M.; Wang, B. Neurovascular Health Insights: A Powerful Tool to Understand and Prognose Neurocognitive Decline; Frontiers Media SA: Lausanne, Switzerland, 2025; Volume 17, p. 1584895. [Google Scholar]
- Kotera, Y.; Richardson, M.; Sheffield, D. Effects of shinrin-yoku (forest bathing) and nature therapy on mental health: A systematic review and meta-analysis. Int. J. Ment. Health Addict. 2022, 20, 337–361. [Google Scholar] [CrossRef]
- Brams, J. The Nature Embedded Mind: How the Way We Think Can Heal Our Planet and Ourselves; John Hunt Publishing: Hampshire, UK, 2025. [Google Scholar]
- Vårhammar, A. Coming Back to Our Senses: Exploring the Potential of Guided Forest Bathing as an Intervention for Human-Nature Connection. Master’s Thesis, Stockholm University, Stockholm, Sweden, 2021. [Google Scholar]
- Keller, J. Forest Bathing Increases Adolescent Mental Well-Being and Connection to Nature: A Transformative Mixed Methods Study. Ph.D. Thesis, Antioch University New England, Keene, NH, USA, 2023. [Google Scholar]
- Hansen, M.M.; Jones, R.; Tocchini, K. Shinrin-yoku (forest bathing) and nature therapy: A state-of-the-art review. Int. J. Environ. Res. Public Health 2017, 14, 851. [Google Scholar] [CrossRef] [PubMed]
- Lopes, M.K.; Falk, T.H. Audio-visual-olfactory immersive digital nature exposure for stress and anxiety reduction: A systematic review on systems, outcomes, and challenges. Front. Virtual Real. 2024, 5, 1252539. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Q. Investigating the Impact of Garden Plant Smellscapes on Human Well-Being: A Case Study of Pine Forests. Forests 2024, 15, 1794. [Google Scholar] [CrossRef]
- Besedovsky, H.O.; Del Rey, A. Immune-neuro-endocrine interactions: Facts and hypotheses. Endocr. Rev. 1996, 17, 64–102. [Google Scholar] [CrossRef]
- Procaccini, C.; Pucino, V.; De Rosa, V.; Marone, G.; Matarese, G. Neuro-endocrine networks controlling immune system in health and disease. Front. Immunol. 2014, 5, 143. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-P.; Lin, C.-M.; Tsai, M.-J.; Tsai, Y.-C.; Chen, C.-Y. Effects of short forest bathing program on autonomic nervous system activity and mood states in middle-aged and elderly individuals. Int. J. Environ. Res. Public Health 2017, 14, 897. [Google Scholar] [CrossRef]
- Li, Q.; Ochiai, H.; Ochiai, T.; Takayama, N.; Kumeda, S.; Miura, T.; Aoyagi, Y.; Imai, M. Effects of forest bathing (shinrin-yoku) on serotonin in serum, depressive symptoms and subjective sleep quality in middle-aged males. Environ. Health Prev. Med. 2022, 27, 44. [Google Scholar] [CrossRef]
- Li, Q.; Takayama, N.; Kimura, Y.; Takayama, H.; Kumeda, S.; Miura, T.; Kitagawa, T.; Aoyagi, Y.; Imai, M. Forest bathing improves inflammatory markers, SpO2, and subjective symptoms related to chronic obstructive pulmonary disease (COPD) in male subjects at risk of developing COPD. J. Occup. Health 2025, 67, uiaf041. [Google Scholar] [CrossRef] [PubMed]
- Roviello, V.; Gilhen-Baker, M.; Vicidomini, C.; Roviello, G.N. Forest-bathing and physical activity as weapons against COVID-19: A review. Environ. Chem. Lett. 2022, 20, 131–140. [Google Scholar] [CrossRef]
- Roviello, V.; Roviello, G.N. Less COVID-19 deaths in southern and insular Italy explained by forest bathing, Mediterranean environment, and antiviral plant volatile organic compounds. Environ. Chem. Lett. 2022, 20, 7–17. [Google Scholar] [CrossRef]
- Baker, S.; Gilhen-Baker, M.; Roviello, G.N. The role of nutrition and forest-bathing in the physical rehabilitation of physically inactive patients: From the molecular aspects to new nature-inspired techniques. Int. J. Environ. Res. Public Health 2022, 20, 793. [Google Scholar] [CrossRef]
- Roviello, V.; Gilhen-Baker, M.; Roviello, G.N.; Lichtfouse, E. River therapy. Environ. Chem. Lett. 2022, 20, 2729–2734. [Google Scholar] [CrossRef]
- Roviello, G.N. The Multifunctional Role of Salix spp.: Linking Phytoremediation, Forest Therapy, and Phytomedicine for Environmental and Human Benefits. Forests 2025, 16, 1808. [Google Scholar] [CrossRef]
- Costanzo, M.; De Giglio, M.A.R.; Gilhen-Baker, M.; Roviello, G.N. The chemical basis of seawater therapies: A review. Environ. Chem. Lett. 2024, 22, 2133–2149. [Google Scholar] [CrossRef]
- Rodtook, P.; Simpattanawong, D.; Ruenbanthoeng, T.; Nongkhai, K.N. Forest bathing activity as a tool to promote wellness tourism in Thailand. Humanit. Arts Soc. Sci. Stud. 2024, 24, 428–440. [Google Scholar] [CrossRef]
- Lazo Álvarez, A.C.; Ednie, A.; Gale-Detrich, T. Contributions of Nature Bathing to Resilience and Sustainability. In Tourism and Conservation-Based Development in the Periphery: Lessons from Patagonia for a Rapidly Changing World; Red Wheel/Weiser: Newburyport, MA, USA, 2023; pp. 389–408. [Google Scholar]
- Antonelli, M.; Barbieri, G.; Donelli, D. Effects of forest bathing (shinrin-yoku) on levels of cortisol as a stress biomarker: A systematic review and meta-analysis. Int. J. Biometeorol. 2019, 63, 1117–1134. [Google Scholar] [CrossRef]
- Mao, G.-X.; Cao, Y.-B.; Lan, X.-G.; He, Z.-H.; Chen, Z.-M.; Wang, Y.-Z.; Hu, X.-L.; Lv, Y.-D.; Wang, G.-F.; Yan, J. Therapeutic effect of forest bathing on human hypertension in the elderly. J. Cardiol. 2012, 60, 495–502. [Google Scholar] [CrossRef]
- Ramanpong, J.; Tsao, C.; Yin, J.; Wu, C.-D.; Huang, Y.-C.; Yu, C.-P. Effects of forest bathing and the influence of exposure levels on cognitive health in the elderly: Evidence from a suburban forest recreation area. Urban For. Urban Green. 2025, 104, 128667. [Google Scholar] [CrossRef]
- Dai, X.; Liao, D.; Ouyang, J.; Xiao, H.; Tian, Z.; Qiu, L.; Yang, J.; Wang, H.; Gong, D.; Li, Z.; et al. Forest bathing enhances sleep, mood, and immunity: Insights from low-latitude evergreen broad-leaved forests. Front. For. Glob. Change 2025, 8, 1619569. [Google Scholar] [CrossRef]
- Doimo, I.; Masiero, M.; Gatto, P. Forest and wellbeing: Bridging medical and forest research for effective forest-based initiatives. Forests 2020, 11, 791. [Google Scholar] [CrossRef]
- Wen, Y.; Yan, Q.; Pan, Y.; Gu, X.; Liu, Y. Medical empirical research on forest bathing (Shinrin-yoku): A systematic review. Environ. Health Prev. Med. 2019, 24, 70. [Google Scholar] [CrossRef] [PubMed]
- Racz, A.; Marinović Glavić, M.; Juraga, D.; Immich, G.; Armano, L.; Vasiljev, V. The potential to integrate Forest therapy and Forest bathing (Shinrin-Yoku) with Nature-based mindfulness into public health programs. Rad Hrvat. Akad. Znan. I Umjetnosti. Med. Znan. 2024, 563, 26–37. [Google Scholar] [CrossRef]
- Li, Q. Effects of forest environment (Shinrin-yoku/Forest bathing) on health promotion and disease prevention—The Establishment of “Forest Medicine”. Environ. Health Prev. Med. 2022, 27, 43. [Google Scholar] [CrossRef]
- Liu, Q. Effect of forest bathing trips on human immune function. Environ. Health Prev. Med. 2010, 15, 9–17. [Google Scholar] [CrossRef]
- Li, Q.; Kobayashi, M.; Wakayama, Y.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Shimizu, T.; Kawada, T.; Park, B.; et al. Effect of phytoncide from trees on human natural killer cell function. Int. J. Immunopathol. Pharmacol. 2009, 22, 951–959. [Google Scholar] [CrossRef]
- Lew, T.; Fleming, K.J. Phytoncides and immunity from forest to facility: A systematic review and meta-analysis. Pharmacol. Res.-Nat. Prod. 2024, 4, 100061. [Google Scholar] [CrossRef]
- Ivanova, M.E.; Lukoyanova, N.; Malhotra, S.; Topf, M.; Trapani, J.A.; Voskoboinik, I.; Saibil, H.R. The pore conformation of lymphocyte perforin. Sci. Adv. 2022, 8, eabk3147. [Google Scholar] [CrossRef]
- Chae, Y.; Lee, S.; Jo, Y.; Kang, S.; Park, S.; Kang, H. The effects of forest therapy on immune function. Int. J. Environ. Res. Public Health 2021, 18, 8440. [Google Scholar] [CrossRef]
- Li, Q. Preventive Effects of Forest Bathing/Shinrin-Yoku on Cardiovascular Diseases: A Review of Mechanistic Evidence. Forests 2025, 16, 310. [Google Scholar] [CrossRef]
- Denche-Zamorano, A.; Rodríguez-Redondo, Y.; Rojo-Ramos, J.; Miguel-Barrado, V.; Sánchez-Leal, A.; Pérez-Calderon, E. Global Review of Literature on Forest Bathing. Austrian J. For. Sci. Cent. Für Das Gesamte Forstwes. 2024, 141, 145–170. [Google Scholar]
- Shang, X.; Lu, J.; Tao, M.; Fei, C.; Fei, J. Benefits of forest therapy for adult mental health: A systematic review and meta-analysis based on the Profile of Mood States (POMS). Front. Psychol. 2025, 16, 1670804. [Google Scholar] [CrossRef]
- Szitás, D.; Halamová, J.; Ottingerová, L.; Schroevers, M. The effects of forest bathing on self-criticism, self-compassion, and self-protection: A systematic review. J. Environ. Psychol. 2024, 97, 102372. [Google Scholar] [CrossRef]
- Li, Q.; Nakadai, A.; Matsushima, H.; Miyazaki, Y.; Krensky, A.M.; Kawada, T.; Morimoto, K. Phytoncides (wood essential oils) induce human natural killer cell activity. Immunopharmacol. Immunotoxicol. 2006, 28, 319–333. [Google Scholar] [CrossRef]
- Putra, R.R.; Veridianti, D.D.; Nathalia, E.; Brilliant, D.; Rosellinny, G.; Suarez, C.G.; Sumarpo, A. Immunostimulant effect from phytoncide of forest bathing to prevent the development of cancer. Adv. Sci. Lett. 2018, 24, 6653–6659. [Google Scholar] [CrossRef]
- Chen, H.; Meng, Z.; Luo, J. Is forest bathing a panacea for mental health problems? A narrative review. Front. Public Health 2025, 13, 1454992. [Google Scholar] [CrossRef]
- Ochiai, H.; Inoue, S.; Masuda, G.; Amagasa, S.; Sugishita, T.; Ochiai, T.; Yanagisawa, N.; Nakata, Y.; Imai, M. Randomized controlled trial on the efficacy of forest walking compared to urban walking in enhancing mucosal immunity. Sci. Rep. 2025, 15, 3272. [Google Scholar] [CrossRef] [PubMed]
- Longman, D.P.; Van Hedger, S.C.; McEwan, K.; Griffin, E.; Hannon, C.; Harvey, I.; Kikuta, T.; Nickels, M.; O’Donnell, E.; Pham, V.A.-V.; et al. Forest soundscapes improve mood, restoration and cognition, but not physiological stress or immunity, relative to industrial soundscapes. Sci. Rep. 2025, 15, 33967. [Google Scholar] [CrossRef] [PubMed]
- Meinköhn, M.; Kessler, C.S.; Kandil, F.I.; Kuballa, L.; Schweininger, S.; von Scheidt, C.; Paul, A.; Haller, H.; Cramer, H.; Joachim, S.; et al. Effects of Forest Therapy on Participant-Reported Outcomes: A Randomized Controlled Dose-Comparison Trial Among the General Population in Germany. J. Integr. Complement. Med. 2025, 31, 826–843. [Google Scholar] [CrossRef]
- Longman, D.P.; Todorova, Y.; Bishop, N.C.; Coates, J.K.; James, L.J.; Lintuluoto, A.; Shaw, C.N. Forest breaks improve employees’ health and attitudes towards work. Urban For. Urban Green. 2025, 114, 129142. [Google Scholar] [CrossRef]
- Gobster, P.H.; Schultz, C.L.; Kruger, L.E.; Henderson, J.R. Forest therapy trails: A conceptual framework and scoping review of research. Forests 2022, 13, 1613. [Google Scholar] [CrossRef]
- Yi, Y.; Seo, E.; An, J. Does forest therapy have physio-psychological benefits? A systematic review and meta-analysis of randomized controlled trials. Int. J. Environ. Res. Public Health 2022, 19, 10512. [Google Scholar] [CrossRef]
- Cheng, X.; Liu, J.; Liu, H.; Lu, S. A systematic review of evidence of additional health benefits from forest exposure. Landsc. Urban Plan. 2021, 212, 104123. [Google Scholar] [CrossRef]
- Kang, M.-J.; Kim, H.-S.; Kim, J.-Y. Effects of forest-based interventions on mental health: A meta-analysis of randomized controlled trials. Int. J. Environ. Res. Public Health 2022, 19, 4884. [Google Scholar] [CrossRef]
- Qiu, Q.; Yang, L.; He, M.; Gao, W.; Mar, H.; Li, J.; Wang, G. The effects of forest therapy on the blood pressure and salivary cortisol levels of urban residents: A meta-analysis. Int. J. Environ. Res. Public Health 2022, 20, 458. [Google Scholar] [CrossRef] [PubMed]
- Yau, K.K.-Y.; Loke, A.Y. Effects of forest bathing on pre-hypertensive and hypertensive adults: A review of the literature. Environ. Health Prev. Med. 2020, 25, 23. [Google Scholar] [CrossRef]
- Li, Q.; Takayama, N.; Katsumata, M.; Takayama, H.; Kimura, Y.; Kumeda, S.; Miura, T.; Ichimiya, T.; Tan, R.; Shimomura, H.; et al. Effects of Forest Bathing (Shinrin-Yoku) in Female Subjects with Depression/Depressive Tendencies. Preprints 2024. [Google Scholar] [CrossRef]
- Farrow, M.R.; Washburn, K. A review of field experiments on the effect of forest bathing on anxiety and heart rate variability. Glob. Adv. Health Med. 2019, 8, 2164956119848654. [Google Scholar] [CrossRef]
- Huang, R.; Li, A.; Li, Z.; Chen, Z.; Zhou, B.; Wang, G. Adjunctive therapeutic effects of forest bathing trips on geriatric hypertension: Results from an on-site experiment in the Cinnamomum camphora forest environment in four seasons. Forests 2022, 14, 75. [Google Scholar] [CrossRef]
- Bettmann, J.E.; Speelman, E.; Jolley, A.; Casucci, T. A Systematic Review and Meta-Analysis on the Effect of Nature Exposure Dose on Adults with Mental Illness. Behav. Sci. 2025, 15, 153. [Google Scholar] [CrossRef]
- Lee, M.; Lee, J. Analysis of Forest Utilization Patterns to Improve Life Satisfaction and Policy Directions. Sustainability 2025, 17, 3689. [Google Scholar] [CrossRef]
- Peterfalvi, A.; Meggyes, M.; Makszin, L.; Farkas, N.; Miko, E.; Miseta, A.; Szereday, L. Forest Bathing Always Makes Sense: Blood Pressure-Lowering and Immune System-Balancing Effects in Late Spring and Winter in Central Europe. Int. J. Environ. Res. Public Health 2021, 18, 2067. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.; Corazon, S.S.; Stigsdotter, U.K. Nature exposure and its effects on immune system functioning: A systematic review. Int. J. Environ. Res. Public Health 2021, 18, 1416. [Google Scholar] [CrossRef]
- Chen, Z.; Bozec, A.; Ramming, A.; Schett, G. Anti-inflammatory and immune-regulatory cytokines in rheumatoid arthritis. Nat. Rev. Rheumatol. 2019, 15, 9–17. [Google Scholar] [CrossRef]
- Opal, S.M.; DePalo, V.A. Anti-inflammatory cytokines. Chest 2000, 117, 1162–1172. [Google Scholar] [CrossRef]
- Cicchese, J.M.; Evans, S.; Hult, C.; Joslyn, L.R.; Wessler, T.; Millar, J.A.; Marino, S.; Cilfone, N.A.; Mattila, J.T.; Linderman, J.J.; et al. Dynamic balance of pro-and anti-inflammatory signals controls disease and limits pathology. Immunol. Rev. 2018, 285, 147–167. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.; Cao, Y.; Wang, B.; Wang, S.; Chen, Z.; Wang, J.; Xing, W.; Ren, X.; Lv, X.; Dong, J.; et al. The Salutary Influence of Forest Bathing on Elderly Patients with Chronic Heart Failure. Int. J. Environ. Res. Public Health 2017, 14, 368. [Google Scholar] [CrossRef]
- Li, Q. New concept of forest medicine. Forests 2023, 14, 1024. [Google Scholar] [CrossRef]
- Fontanella, F.; D’Alessandro, T.; Nardone, E.; De Stefano, C.; Vicidomini, C.; Roviello, G.N. Artificial Intelligence for Natural Products Drug Discovery in Neurodegeneration Therapies: A Review. Biomolecules 2026, 16, 129. [Google Scholar] [CrossRef]
- Stepanyan, L.; Israyelyan, M.; Gori, A.; Tsaturyan, A.; Saribekyan, Z.; Hovsepyan, K.; Sargsyan, T.; Pastore, R.; De Luca, A.; Roviello, G.N. Natural and Synthetic Peptides as Alternatives to Antibiotics in Intestinal Infections—A Review. Antibiotics 2026, 15, 68. [Google Scholar] [CrossRef] [PubMed]
- Najafi, S.; Jojani, M.; Najafi, K.; Costanzo, V.; Vicidomini, C.; Roviello, G.N. West Nile Virus: Epidemiology, Surveillance, and Prophylaxis with a Comparative Insight from Italy and Iran. Vaccines 2026, 14, 57. [Google Scholar] [CrossRef] [PubMed]
- Roviello, G.N. Nature-Inspired Pathogen and Cancer Protein Covalent Inhibitors: From Plants and Other Natural Sources to Drug Development. Pathogens 2025, 14, 1153. [Google Scholar] [CrossRef] [PubMed]
- Sargsyan, T.; Hakobyan, H.; Simonyan, H.; Soghomonyan, T.; Tsaturyan, A.; Hovhannisyan, A.; Sardaryan, S.; Saghyan, A.; Roviello, G.N. Biomacromolecular interactions and antioxidant properties of novel synthetic amino acids targeting DNA and serum albumin. J. Mol. Liq. 2025, 440, 128700. [Google Scholar] [CrossRef]
- Sargsyan, T.; Stepanyan, L.; Tsaturyan, A.; Palumbo, R.; Vicidomini, C.; Roviello, G.N. Intracellular Parasitic Infections Caused by Plasmodium falciparum, Leishmania spp., Toxoplasma gondii, Echinococcus multilocularis, Among Key Pathogens: Global Burden, Transmission Dynamics, and Vaccine Advances—A Narrative Review with Contextual Insights from Armenia. Vaccines 2025, 13, 1082. [Google Scholar] [CrossRef]
- Costanzo, M.; Roviello, G.N. Precision Therapeutics Through Bioactive Compounds: Metabolic Reprogramming, Omics Integration, and Drug Repurposing Strategies. Int. J. Mol. Sci. 2025, 26, 10047. [Google Scholar] [CrossRef]
- Vicidomini, C.; Roviello, G.N. Therapeutic Convergence in Neurodegeneration: Natural Products, Drug Repurposing, and Biomolecular Targets. Biomolecules 2025, 15, 1333. [Google Scholar] [CrossRef]
- Park, B.J.; Tsunetsugu, Y.; Kasetani, T.; Kagawa, T.; Miyazaki, Y. The physiological effects of Shinrin-yoku (taking in the forest atmosphere or forest bathing): Evidence from field experiments in 24 forests across Japan. Environ. Health Prev. Med. 2010, 15, 18–26. [Google Scholar] [CrossRef]
- Queirolo, L.; Fazia, T.; Roccon, A.; Pistollato, E.; Gatti, L.; Bernardinelli, L.; Zanette, G.; Berrino, F. Effects of forest bathing (Shinrin-yoku) in stressed people. Front. Psychol. 2024, 15, 1458418. [Google Scholar] [CrossRef]
- Bottaccioli, A.G.; Bottaccioli, F.; Minelli, A. Stress and the psyche–brain–immune network in psychiatric diseases based on psychoneuroendocrineimmunology: A concise review. Ann. N. Y. Acad. Sci. 2019, 1437, 31–42. [Google Scholar] [CrossRef]
- Esch, T.; Stefano, G.B.; Fricchione, G.L.; Benson, H. Stress in cardiovascular diseases. Signature 2002, 8, 101. [Google Scholar]
- Haluza, D.; Schönbauer, R.; Cervinka, R. Green perspectives for public health: A narrative review on the physiological effects of experiencing outdoor nature. Int. J. Environ. Res. Public Health 2014, 11, 5445–5461. [Google Scholar] [CrossRef]
- Kobayashi, H.; Song, C.; Ikei, H.; Park, B.-J.; Lee, J.; Kagawa, T.; Miyazaki, Y. Forest walking affects autonomic nervous activity: A population-based study. Front. Public Health 2018, 6, 278. [Google Scholar] [CrossRef]
- Oliveros, E.; Patel, H.; Kyung, S.; Fugar, S.; Goldberg, A.; Madan, N.; Williams, K.A. Hypertension in older adults: Assessment, management, and challenges. Clin. Cardiol. 2020, 43, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Baban, K.A.; Morton, D.P. Lifestyle medicine and stress management. J. Fam. Pract. 2022, 71, S24–S29. [Google Scholar] [CrossRef] [PubMed]
- Berto, R. The role of nature in coping with psycho-physiological stress: A literature review on restorativeness. Behav. Sci. 2014, 4, 394–409. [Google Scholar] [CrossRef]
- Yao, W.; Zhang, X.; Gong, Q. The effect of exposure to the natural environment on stress reduction: A meta-analysis. Urban For. Urban Green. 2021, 57, 126932. [Google Scholar] [CrossRef]
- Li, H.; Zhang, G. How can plant-enriched natural environments benefit human health: A narrative review of relevant theories. Int. J. Environ. Health Res. 2024, 34, 1241–1254. [Google Scholar] [CrossRef]
- Clifford, M.A. Your Guide to Forest Bathing (Expanded Edition): Experience the Healing Power of Nature; Red Wheel/Weiser: Newburyport, MA, USA, 2021. [Google Scholar]
- Willis, K. Good Nature: Why Seeing, Smelling, Hearing and Touching Plants is Good for Our Health; Simon and Schuster: New York, NY, USA, 2024. [Google Scholar]
- Janeczko, E.; Bielinis, E.; Wójcik, R.; Woźnicka, M.; Kędziora, W.; Łukowski, A.; Elsadek, M.; Szyc, K.; Janeczko, K. When urban environment is restorative: The effect of walking in suburbs and forests on psychological and physiological relaxation of young Polish adults. Forests 2020, 11, 591. [Google Scholar] [CrossRef]
- Meyer, K.; Botsch, K. Do forest and health professionals presume that forests offer health benefits, and is cross-sectional cooperation conceivable? Urban For. Urban Green. 2017, 27, 127–137. [Google Scholar] [CrossRef]
- Meyer, K.; Buerger-Arndt, R. How forests foster human health–Present state of research-based knowledge (in the field of Forests and Human Health). Int. For. Rev. 2014, 16, 421–446. [Google Scholar] [CrossRef]
- Yao, W.; Luo, Q.; Zhang, X.; Zhuo, C.; Mi, L. Exploring the effect of different typical plant community on human stress reduction: A field experiment. Sci. Rep. 2024, 14, 5600. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Huang, Q.; Chen, Z.; Cao, Y.; Mao, G.; Dong, J.; Wang, S.; Lv, X.; Wang, G. Effects of Cinnamomum camphora Forest Environment on Elderly Patients with Hypertension: Implications for Adjunctive Therapy; Atlantis Press: Dordrecht, The Netherlands, 2019; pp. 203–209. [Google Scholar]
- McEwan, K.; Giles, D.; Clarke, F.J.; Kotera, Y.; Evans, G.; Terebenina, O.; Minou, L.; Teeling, C.; Basran, J.; Wood, W.; et al. A pragmatic controlled trial of forest bathing compared with compassionate mind training in the UK: Impacts on self-reported wellbeing and heart rate variability. Sustainability 2021, 13, 1380. [Google Scholar] [CrossRef]
- Sudimac, S.; Kühn, S. Can a nature walk change your brain? Investigating hippocampal brain plasticity after one hour in a forest. Environ. Res. 2024, 262, 119813. [Google Scholar] [CrossRef]
- Sudimac, S. Exposure to Natural Versus Urban Environments: Short-Term Effects on Stress, Stress-Related Brain Function, and Hippocampal Structure; Freie Universitaet Berlin: Berlin, Germany, 2024. [Google Scholar]
- Jimenez, M.P.; DeVille, N.V.; Elliott, E.G.; Schiff, J.E.; Wilt, G.E.; Hart, J.E.; James, P. Associations between nature exposure and health: A review of the evidence. Int. J. Environ. Res. Public Health 2021, 18, 4790. [Google Scholar] [CrossRef]
- Khalil, M.H. Green Environments for Sustainable Brains: Parameters Shaping Adaptive Neuroplasticity and Lifespan Neurosustainability—A Systematic Review and Future Directions. Int. J. Environ. Res. Public Health 2025, 22, 690. [Google Scholar] [CrossRef] [PubMed]
- Kerimova, N.; Akimov, P.; Ntoumanis, I.; Blank, I.B.; Moiseeva, V.; Klucharev, V. Green urban environments enhance brain-to-brain synchrony. Sci. Rep. 2025, 15, 43524. [Google Scholar] [CrossRef]
- Rhee, J.H.; Schermer, B.; Han, G.; Park, S.Y.; Lee, K.H. Effects of nature on restorative and cognitive benefits in indoor environment. Sci. Rep. 2023, 13, 13199. [Google Scholar] [CrossRef]
- Oh, B.; Lee, K.J.; Zaslawski, C.; Yeung, A.; Rosenthal, D.; Larkey, L.; Back, M. Health and well-being benefits of spending time in forests: Systematic review. Environ. Health Prev. Med. 2017, 22, 71. [Google Scholar] [CrossRef]
- McEwen, B.S.; Nasca, C.; Gray, J.D. Stress effects on neuronal structure: Hippocampus, amygdala, and prefrontal cortex. Neuropsychopharmacology 2016, 41, 3–23. [Google Scholar] [CrossRef]
- Albadawi, E.A. Structural and functional changes in the hippocampus induced by environmental exposures. Neurosci. J. 2025, 30, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Edl, S.; Benedek, M.; Papousek, I.; Weiss, E.M.; Fink, A. Creativity and the Stroop interference effect. Personal. Individ. Differ. 2014, 69, 38–42. [Google Scholar] [CrossRef]
- T.S, S.; Bhanavi, K. Travelling to the Woods to Enhance Creativity: Effectiveness of Connecting with Nature for Enhancing Divergent Thinking in Employees. 24 June 2025. Available online: https://ssrn.com/abstract=5318718 (accessed on 12 February 2026).
- Jennings, V.; Browning, M.H.; Rigolon, A. Urban Green Spaces: Public Health and Sustainability in the United States; Springer: Berlin/Heidelberg, Germany, 2019; Volume 8. [Google Scholar]
- Besser, L. Outdoor green space exposure and brain health measures related to Alzheimer’s disease: A rapid review. BMJ Open 2021, 11, e043456. [Google Scholar] [CrossRef]
- Ascone, L.; Mostajeran, F.; Mascherek, A.; Tawil, N.; Knaust, T.; Samaan, L.; Kühn, S. Multi-vs. Unimodal Forest-Bathing in VR to Enhance Affective and Cognitive Recovery after Acute Stress. J. Environ. Psychol. 2025, 105, 102637. [Google Scholar] [CrossRef]
- Heckmann, J.G.; Kiem, M.; Immich, G. Forest Therapy as a Nature-Based Intervention: An Option for Neurological Rehabilitation? Complement. Med. Res. 2024, 31, 56–63. [Google Scholar] [CrossRef]
- Park, J.; Wang, S.-M.; Kang, D.W.; Lee, B.; Choi, H. Effect of Anti-Aging Standard Forest Healing Program With Multiple Visits to a Forest Facility on Cognition in Older Age Patients. Dement. Neurocognitive Disord. 2024, 23, 44–53. [Google Scholar] [CrossRef]
- Krala-Szkaradowska, M.; Stencel, N.J.; Skrzypczak, K.O.; Stuczyński, S.K.; Konczewska, L. Effect of forest bathing (‘shinrin-yoku’) on human health–a literature review. Med. Sr. Environ. Med. 2024, 27, 12–17. [Google Scholar] [CrossRef]
- Mazzoleni, E.; Donelli, D.; Zabini, F.; Meneguzzo, F.; Antonelli, M. Forest therapy research in Europe: A scoping review of the scientific literature. Forests 2024, 15, 848. [Google Scholar] [CrossRef]
- Li, Q.; Morimoto, K.; Nakadai, A.; Inagaki, H.; Katsumata, M.; Shimizu, T.; Hirata, Y.; Hirata, K.; Suzuki, H.; Miyazaki, Y.; et al. Forest bathing enhances human natural killer activity and expression of anti-cancer proteins. Int. J. Immunopathol. Pharmacol. 2007, 20, 3–8. [Google Scholar] [CrossRef]
- Craig, J.M.; Logan, A.C.; Prescott, S.L. Natural environments, nature relatedness and the ecological theater: Connecting satellites and sequencing to shinrin-yoku. J. Physiol. Anthropol. 2016, 35, 1. [Google Scholar] [CrossRef] [PubMed]
- Turrin, N.P.; Rivest, S. Molecular and cellular immune mediators of neuroprotection. Mol. Neurobiol. 2006, 34, 221–242. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.K. The Handbook of Neuroprotection; Humana: Totowa, NJ, USA, 2011. [Google Scholar]
- Pichler, C.; Freidl, J.; Bischof, M.; Kiem, M.; Weisböck-Erdheim, R.; Huber, D.; Squarra, G.; Murschetz, P.C.; Hartl, A. Mountain hiking vs. Forest therapy: A study protocol of novel types of nature-based intervention. Int. J. Environ. Res. Public Health 2022, 19, 3888. [Google Scholar] [CrossRef]
- Tomova, T.; Popova-Dobreva, D. Shinrin-yoku/Forest bathing: A natural way to promote health, prevention and cure diseases. The heritage of Master Peter Dunov in Forest Medicine. Acad. Rep.-Bull. Nasled. BG 2024, 32–41. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.