Abstract
Granitic soils in the Highlands support the cultivation of Arabica coffee in northern Thailand; however, their geochemical and radiological properties are inadequately defined. This study examined major oxides, trace elements, natural radionuclides, and extractable phosphorus in granitic-derived coffee soils from the Agricultural Innovation Research, Integration, Demonstration, and Training Center (AIRID) in Chiang Mai. Twenty soil samples were obtained from 10 locations at two depth intervals (0–30 cm and 30–60 cm). Major and trace elements were analyzed via X-ray fluorescence (XRF), natural radionuclides were analyzed through high-purity germanium (HPGe) gamma spectrometry, and extractable phosphorus was determined using the Bray II method. The soils demonstrate remarkably high 40K activity concentrations (1.2–1.9 kBq kg−1) and increased K2O contents (4.9–7.8 wt%), about three to five times more than worldwide soil averages according to Reimann & de Caritat, indicating enrichment from potassium-rich granitic rocks. Major oxide compositions suggest extensive tropical weathering, characterized by elevated SiO2 (>60 wt%) and Al2O3 (>14 wt%), alongside significant depletion of CaO and MgO (<1 wt%). In topsoil, Bray II–extractable phosphorus constitutes 10–25% of total phosphorus and has a robust positive connection with P2O5 (R2 = 0.95, p < 0.001), signifying surface accumulation and restricted vertical mobility. Multivariate analysis indicates lithogenic grouping of trace elements with negligible vertical redistribution. These findings establish a geochemical and radiological baseline for highland coffee soils in northern Thailand, with implications for soil fertility assessment, soil–plant transfer research, and evaluations of natural radioactive exposure related to coffee production.
1. Introduction
Soil fertility in high-elevation coffee plantations is strongly controlled by the underlying geological parent material [1]. In the granitic terrains that dominate the northern highlands of Thailand, soils are derived primarily from quartz, K-feldspar, plagioclase, biotite, and amphibole. During progressive chemical weathering, trace elements are released from these primary minerals in a predictable sequence. Transition metals such as Co, Ni, Cr, V, Cu, Ga, Zn, and Pb are mainly supplied by the gradual decomposition of mafic minerals (biotite and amphibole), whereas elements such as Th, U, Y, Nb, and Sr are largely associated with accessory minerals (e.g., monazite, xenotime, apatite, and zircon) and with secondary Fe- and Al-oxide phases [1,2,3]. These mineral-derived geochemical signatures can persist in tropical, well-drained upland soils and provide a robust framework for interpreting nutrient dynamics and trace element behavior. Potassium (K) is one of the most important macronutrients controlling the growth and productivity of Coffea arabica in upland environments. Unlike nitrogen and phosphorus, potassium is not structurally incorporated into organic matter and is therefore retained mainly in exchangeable pools derived from the weathering of K-bearing minerals, such as K-feldspar and micas. Potassium availability is further regulated by adsorption onto Fe–Al oxides and by leaching losses under high rainfall conditions [3]. Coffee cultivators in northern Thailand grow Arabica coffee on elevated granitic soils that have had significant tropical weathering, leading to acidic conditions, poor base saturation, and a reduction in exchangeable calcium and magnesium due to extensive leaching from monsoon precipitation [4]. Despite granitic parent materials providing significant potassium via the weathering of K-feldspar and mica, this frequently results in nutrient imbalance rather than optimal fertility, as increased K availability may be associated with deficiencies in Ca, Mg, and micronutrients, negatively impacting nutrient uptake efficiency and yield stability [5,6]. Phosphorus supply is a continual limitation as phosphate is significantly adsorbed onto iron and aluminum oxides generated through extensive weathering, diminishing fertilizer efficacy and elevating production expenses [7,8,9]. The issues are intensified by steep gradients and substantial precipitation, which facilitate erosion and nutrient depletion in upland coffee systems [10]. Comprehending the geochemical and radiological properties of these soils is essential for differentiating lithogenic nutrient sources from human contributions, elucidating the factors influencing nutrient mobility and trace-element dynamics [11,12], and determining baseline activity concentrations of naturally occurring radionuclides, including 40K, U, and Th, in granitic terrains [13]. This fundamental information supports soil–plant transfer studies, promotes sustainable coffee production, and provides reference radionuclide activity concentrations to support external exposure assessment and future soil–plant transfer investigations. Consequently, the natural K status of granitic soils can either reduce the need for external fertilization or, if depleted, become a major limitation to sustainable coffee production. Despite the agronomic importance of potassium, quantitative data on K concentrations and associated natural radioactivity in Thai highland soils remain limited. Granitic rocks are also naturally enriched in the radioactive isotope 40K, as well as in uranium- and thorium-series radionuclides [14,15]. Weathering and pedogenesis can redistribute these radionuclides within the soil profile, where they may become associated with secondary oxide and clay mineral phases. Northern Thailand, particularly Chiang Mai Province, has been recognized as an area of relatively high natural background radiation compared with other regions of the country [16]. However, little information is available on the distribution of natural radionuclides specifically within agricultural soils used for coffee cultivation and on their relationships with soil mineralogy and geochemistry. While research specifically focused on radionuclide distributions in soils cultivated with coffee is limited, several peer-reviewed studies of agricultural soils in coffee-producing or geologically similar areas indicate that the activity concentrations of natural radionuclides are predominantly influenced by parent material, soil mineralogy, and geochemical processes. In southern Ecuador, a significant Andean coffee-growing area, Ramos-Lerate et al. demonstrated that the activities of 226Ra, 232Th, and 40K in both cultivated and uncultivated agricultural soils are predominantly governed by geological background, with minimal impact of farm practices, underscoring the preeminence of lithogenic factors on soil radioactivity [17]. Comparable findings were documented for agricultural soils in Yemen, a conventional coffee-exporting nation, where Hussien et al. noted that the geographical variability in activity concentrations of 226Ra, 232Th, and 40K is indicative of soil-forming processes and parent lithology, rather than land use alone [18]. In Brazil, a leading global coffee producer, the distribution of natural radionuclides in cultivated soils is intricately associated with pedological attributes and mineralogical composition, notably the prevalence of potassium-rich minerals and thorium-enriched accessory phases [19,20]. Collectively, these studies demonstrate that radionuclide concentrations in agricultural soils of coffee-growing areas are primarily geogenic and closely related to soil mineralogy and geochemistry, underscoring the necessity for site-specific characterization in evaluating radionuclide dynamics within coffee cultivation systems. In addition to potassium and natural radionuclides, phosphorus (P) behavior in highly weathered tropical soils is strongly influenced by Fe–Al oxide phases, which can immobilize phosphate through specific adsorption reactions. As a result, surface accumulation of P from repeated fertilizer application is common, whereas vertical mobility is typically limited. Understanding the coupled behavior of P, K, trace elements, and natural radionuclides in granitic-derived soils is therefore essential for evaluating long-term soil fertility, nutrient-use efficiency, and environmental geochemical baselines in upland agroecosystems. This study aims to: (i) quantify the major and trace element composition of granitic-derived highland coffee soils in Chiang Mai, Thailand; (ii) determine the activity concentrations of the natural radionuclides 40K, 238U, and 232Th in soil profiles and analyze their correlation with K content; (iii) assess vertical variations in extractable and total P; and (iv) identify the predominant lithogenic and pedogenic influences on element distributions through multivariate statistical analysis.
2. Materials and Methods
2.1. Study Area and Sampling
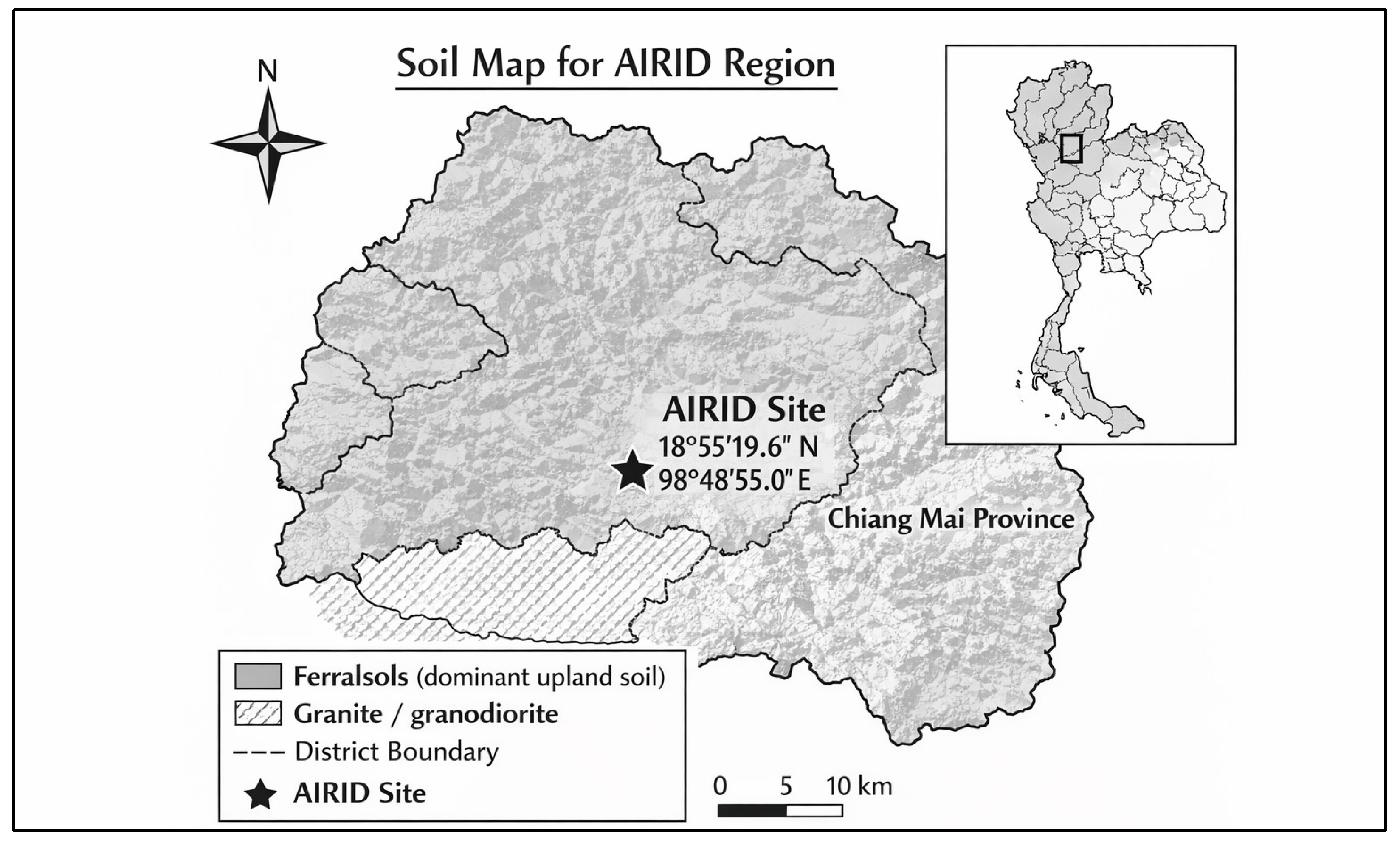
The study was conducted in the Agricultural Innovation Research, Integration, Demonstration, and Training Center (AIRID; 18°55′19.6″ N, 98°48′55.0″ E, Figure 1) located within the Nhong Hoi Highland Agricultural Research Station, Pong Yang Sub-district, Mae Rim District, Chiang Mai Province, Thailand. The site lies at an elevation of 850–900 m above sea level, with an average slope of 18–25° [21]. The climate is tropical, characterized by three seasons influenced by Pacific typhoons and the southwest monsoon; mean annual precipitation is 1354 mm (mainly April–August) and mean annual temperature is 28.5 °C (range: 10.3 °C in January to 39.8 °C in July–August). Geologically, the area belongs to the Central Granitic Belt and is underlain by granodiorite and monzogranite intrusions rich in K-feldspar, quartz, and biotite (Petrographic analysis, Ref. [22]). These lithologies give rise to Ferralsols (WRB classification, Ref. [23]) with naturally high potassium concentrations. At elevations around 1000 m, fertilization is typically applied in May, August, and October, with a harvest period of approximately nine months. At AIRID and its associated demonstration sites, research encompasses seedling production, planting on sloped highland terrain, fertilization scheduling, pest and disease management, and post-harvest evaluation, often integrated into community outreach and agritourism programs such as ‘from seed to cup’ coffee educational experiences [24]. Coffee is grown in areas with Ferralsols originating from granitic parent materials characteristic of the Central Granitic Belt in northern Thailand, influencing soil fertility, nutrient composition, and hydrological dynamics [24]. The distribution of soil types in the broader region can be obtained from national and international soil survey databases, facilitating the development of schematic soil maps to depict AIRID’s edaphic context [24].
Root distribution studies on Coffea spp. have shown that 30–55% of total root biomass occurs within the upper 10–30 cm of soil, and approximately 95% of roots are confined to the top 30 cm in clayey substrates [25,26,27,28]. Accordingly, soil samples were collected at two depth intervals—0–30 cm (root-active zone) and 30–60 cm (sub-surface layer)—to capture vertical variations in elements.
On 13 July 2023, ten sampling points were established across a relatively homogeneous portion of the coffee farm. At each point, an auger was used to retrieve composite samples from the two depth intervals described above. The locations were selected to represent the range of topographic positions (e.g., slope aspect and curvature) and management practices present within the study area. This sampling design follows a stratified random approach commonly used in agronomic soil surveys to depict the spatial diversity of soil parameters within the study area. Before sampling, the coffee farm was categorized into distinct strata according to observable site factors that affect soil growth and nutrient distribution. Sampling locations within each stratum were selected at random while ensuring a minimum distance to prevent spatial clustering. A total of ten sampling points were selected within the study area to ensure representative coverage of the observed variability in soil properties (see Figure 1).
Figure 1.
The Agricultural Innovation Research, Integration, Demonstration, and Training (AIRID) site (18°55′19.6″ N, 98°48′55.0″ E) within the Nhong Hoi Highland Agricultural Research Station is marked [29].
Figure 1.
The Agricultural Innovation Research, Integration, Demonstration, and Training (AIRID) site (18°55′19.6″ N, 98°48′55.0″ E) within the Nhong Hoi Highland Agricultural Research Station is marked [29].
2.2. Radiological Analysis
The soil samples were dried at 110 °C and stored in pre-weighed polystyrene containers (U8; outer diameter = 50 mm, inner diameter = 48 mm, height = 62 mm), following the method described in Hosoda et al. (2013) and then left to reach radioactive stored for >28 days prior to measurement to ensure secular equilibrium between 226Ra and its short-lived progeny 222Rn [30]. The natural radionuclide measurements were conducted using an HPGe detector (EGPC 100-P15, CANBERRA, Meriden, CT, USA). Each measurement was run for 80,000 s. Radionuclide concentrations were decay-corrected to the sampling date. Calibration was performed using standard gamma volume sources (MX033U8PP; Japan Radioisotope Association, Tokyo, Japan), which contained nine mixed nuclides (109Cd, 57Co, 139Ce, 51Cr, 85Sr, 137Cs, 54Mn, 88Y, and 60Co) with five different thicknesses (5, 10, 20, 30, and 50 mm) according to the Japanese Nuclear Regulation Authority. Minimum detectable activities (MDAs) were evaluated based on measurements conducted with a counting time of 80,000 s and were derived from the analysis software (Spectra Explorer software version 2.13), which reported detection limits for the gamma lines used in activity determination. Under these measurement conditions, MDAs were on the order of (1.2–1.5) × 10−2 Bq g−1 for 40K (1460.8 keV), (2.0–2.6) × 10−3 Bq g−1 for the 238U decay series (assessed via 214Pb and 214Bi assuming secular equilibrium), and (4.0–5.0) × 10−3 Bq g−1 for the 232Th decay series (via 228Ac at 911.2 keV). Variability among samples reflects differences in sample mass, detector efficiency at specific gamma energies, and background levels, rather than analytical inconsistency. The activity concentrations of 40K were ascertained utilizing the 1460.75 keV gamma line. The decay series of 238U was calculated indirectly by gamma emissions from 214Pb (295.22 and 351.99 keV) and 214Bi (609.31, 1120.29, and 1238.11 keV), whereas the decay series of 232Th was quantified using 228Ac (338.70, 911.20, and 968.80 keV), 212Pb (238.63 keV), and 208Tl (583.14 and 860.37 keV). Measurement uncertainties are stated to vary from several percent to under 10%. Annual effective doses were estimated for an outdoor agricultural worker exposure scenario using internationally recommended conversion coefficients and an outdoor occupancy factor of 0.2.
Absorbed dose rates in air (D, nGy h−1) were estimated from the measured activity concentrations of the 238U series, 232Th series, and 40K using internationally recommended conversion coefficients. The absorbed dose rate was calculated using the following equation:
where , , and represent the activity concentrations (Bq kg−1) of the 238U series, 232Th series, and 40K, respectively. The coefficients (nGy h−1 per Bq kg−1) were adopted from UNSCEAR (2000) [13].
2.3. Geochemical Analysis
Soil samples were combusted at 800 °C for 8 h to remove organic matter and carbonates prior to geochemical analysis. Following combustion, 0.5 g of each sample was fused with 5.0 g of flux to produce a borate glass bead using an electrical fusion system (Bead & Fuse Sampler TK-4100, Amena Tech, Yokohama, Japan) at 1141 °C. The flux consisted of a lithium borate mixture containing 67% Li2B4O7 and 33% LiBO2. Fusion was performed in Pt–Au crucibles (95% Pt, 5% Au). After fusion, the melt was rapidly quenched to obtain a homogeneous glass bead suitable for XRF analysis. Elemental concentrations were measured using a wavelength-dispersive X-ray fluorescence (WD-XRF) spectrometer (ZSX Primus IV, Rigaku, Japan) operated under vacuum conditions, with calibration performed using certified geological reference materials.
Major and trace elements were quantified by X-ray fluorescence spectrometry following borate fusion. Major oxides determined included SiO2, Al2O3, Fe2O3, MgO, CaO, MnO, Na2O, K2O, and P2O5. Trace elements (Ba, Ce, Co, Cr, Cu, Ga, Nb, Ni, Pb, Rb, Sr, Th, U, V, W, Y, Zn, Zr, and As) were measured to support the interpretation of weathering intensity, lithogenic controls, and potential anthropogenic contributions.
To compare XRF-derived potassium concentrations with HPGe-measured 40K activities, 40K was independently estimated from K2O values using a mass-basis conversion. Elemental potassium was first derived from K2O using the molar-mass ratio (2M K/1M K2O = 0.83015). The potassium concentration was then converted from wt% to g kg−1 (1 wt% = 10 g kg−1), and 40K activity was calculated assuming a specific activity of 31.1 Bq g−1 for natural potassium (40K (Bq kg−1) = K2O (wt%) × 258.3).
All K2O concentrations are reported on an oven-dry basis (110 °C), and the calculated 40K activities therefore correspond to the same oven-dry mass basis. No LOI correction or normalization to 100% was applied to the oxide data; thus, no dry-versus-ignited inconsistency is introduced in the conversion.
The data processing, statistical analysis, and visualization were conducted using Python 3.12 within a Google Colab environment. Data manipulation and numerical operations were performed using pandas (v2.2.2) and numpy (v2.0.2). Visualization employed matplotlib (v3.10.0) and seaborn (v0.13.2). Prior to multivariate analysis, major oxide data were centered and scaled using Z-score normalization. Principal component analysis (PCA) was applied to the standardized dataset to identify dominant geochemical patterns. K-means clustering was subsequently performed on the PCA scores of the first two principal components, resulting in the classification of samples into three compositional clusters [31]. Hierarchical clustering and additional statistical routines were implemented using scipy (v1.16.3). Major oxide concentrations are compositional data constrained by constant-sum effects. In this study, Z-score standardization was applied to explore relative variation patterns; however, no log-ratio transformation was performed. Therefore, PCA and clustering results are interpreted descriptively as visualization tools for relative geochemical gradients rather than as formal compositional-data analysis. Interpretations are supported by independent elemental trends and geological reasoning.
2.4. Bray II Extraction
A 2.5 g aliquot of sieved soil (<2 mm) was placed into a 50 mL bottle, to which 25 mL of Bray II extractant (0.03 N NH4F, 0.1 N HCl) was added. The mixture was shaken for 1 min and subsequently filtered. An aliquot (1–5 mL) of the clear filtrate was transferred into a 25 mL volumetric flask, followed by the addition of 5 mL of molybdenum color reagent (MCR). The volume was adjusted to 25 mL with distilled water, mixed thoroughly, and allowed to develop color for 10 min. Absorbance was measured at 882 nm using a UV–Vis spectrophotometer. Phosphorus concentrations were quantified using an external calibration curve prepared from potassium dihydrogen phosphate (KH2PO4) standard solutions. Calibration standards with concentrations of 0, 0.2, 0.4, 0.6, 0.8, and 1.0 mg P L−1 were prepared by serial dilution and processed using the same extraction and color-development procedures as the soil samples. The calibration curve was constructed by linear regression of absorbance measured at 882 nm versus phosphorus concentration and was verified to be linear prior to sample analysis.
Extractable phosphorus was measured using the Bray II method. A subsample of soil was shaken with Bray II extractant, filtered, and analyzed colorimetrically. Total phosphorus measured via XRF was compared with extractable fractions to evaluate accumulation and mobility.
3. Results
3.1. Natural Radionuclide 40K Enrichment
The activity concentrations of 40K measured by HPGe gamma spectrometry ranged from 1.2 ± 0.1 to 1.9 ± 0.2 kBq kg−1 (n = 20). The 40K activities estimated from K2O using the stated conversion factor (40K = 258.3 × K2O, where K2O is in wt%, oven-dry basis) ranged from 1.2 to 1.9 kBq kg−1 and showed strong agreement with HPGe-measured values (Supplementary Figure S1), confirming internal analytical consistency and mass-basis alignment between methods. This difference can be attributed to contrasting geological settings. The present study area is developed on granitic parent material characterized by abundant K-bearing feldspars and mica minerals, which naturally elevate potassium and associated 40K levels. In contrast, soils in the Kong Khaek subdistrict are derived from non-granitic lithologies with lower primary potassium content, resulting in lower background 40K activity concentrations. The observed differences, therefore, reflect lithogenic controls rather than anthropogenic inputs [16]. Table 1 summarizes representative major oxide concentrations from the studied soils. Median K2O contents reached 5.3% (range 4.9–7.8%).

Table 1.
Comparison of major oxide concentrations (oven-dry basis) from the studied soils and previous studies of Thai paddy soil and the global median [32].
The 238U–series activity concentrations, determined from 214Pb and 214Bi under equilibrium conditions, ranged from approximately 0.07 to 0.10 kBq kg−1, with a mean value of approximately 0.085 kBq kg−1. The consistency between daughter radionuclides within counting uncertainties indicates stable equilibrium and reliable measurement conditions. These values fall within typical background levels reported for granitic soils.
The 232Th–series activities, derived from 228Ac, 212Pb, and 208Tl, ranged from approximately 0.10 to 0.13 kBq kg−1, with a mean value of approximately 0.1 kBq kg−1. Agreement among daughter radionuclides confirms equilibrium within the thorium decay series. Compared with global soil averages, the measured uranium- and thorium-series activities are within expected ranges for granitic terrains and do not indicate anomalous enrichment.
Overall, the radionuclide distributions demonstrate that the radiological characteristics of the AIRID soils are controlled primarily by granitic parent material, with pronounced enrichment in 40K and typical background levels of uranium and thorium.
Radiation Dose Assessment
Absorbed dose rates in air were estimated using internationally recommended conversion coefficients for the 238U–series, 232Th–series, and 40K based on the measured activity concentrations according to Equation (1). The total absorbed dose rates ranged from approximately 65 to 95 nGy h−1, with a mean value of approximately 80 nGy h−1 across the study area. The contribution of 40K dominated the total dose due to its elevated activity concentrations (1.2–1.9 kBq kg−1), while the uranium- and thorium-series radionuclides provided smaller but measurable contributions.
To define a realistic exposure condition, annual effective doses were estimated assuming an outdoor agricultural worker exposed to external gamma radiation from soil. An outdoor occupancy factor of 0.2 was applied in accordance with UNSCEAR recommendations for individuals engaged in agricultural activities [13]. Under this scenario, annual effective doses ranged from approximately 0.08 to 0.12 mSv yr−1, with a mean value of approximately 0.1 mSv yr−1.
Although the absorbed dose rates are slightly higher than the global median outdoor terrestrial value (~59 nGy h−1), the calculated annual effective doses remain well below the worldwide average natural radiation exposure of 2.4 mSv yr−1 reported by UNSCEAR. The elevated dose rates are therefore attributable to the naturally high potassium content of the granitic parent material rather than anthropogenic contamination, and they do not indicate radiological concern for external exposure of agricultural workers in the study area [13].
3.2. Controls on Element and Radionuclide Mobility in Granitic-Derived Soils
Radionuclide mobility in the examined soils is primarily governed by lithogenic and pedogenic factors, with soil mineralogy and oxide composition exerting strong control over element retention and dispersion [33]. The granitic parent material of the study area contributes to elevated background levels of naturally occurring radionuclides and trace elements, consistent with previous findings that radionuclide behavior in soils is largely dictated by mineral-associated sorption and immobilization processes [33]. Iron and aluminum oxide phases, which are abundant in strongly weathered tropical soils, possess high specific surface areas that promote effective adsorption of trace elements and radionuclides, thereby limiting their mobility [34]. These oxides and oxyhydroxides play a central role in adsorption, ion transport, and sorption stability, acting as major sinks that restrict vertical migration of both lithogenic and fertilizer-derived elements [34]. Radionuclide studies further demonstrate that relatively immobile species frequently associate with weakly crystalline Fe–Al minerals and organic matter under typical soil conditions, further impeding downward transport within the soil profile [35]. Agricultural management practices may enhance localized accumulation of nutrient-related elements, particularly phosphorus and potassium; however, these inputs generally do not override dominant pedogenic controls on element mobility [36,37,38,39]. The presence of Fe–Al oxides and clay minerals in these granitic soils substantially increases sorption capacity, restricting vertical redistribution even for elements introduced through fertilization [40,41,42]. Overall, the integrated geochemical and radiological evidence indicates that element mobility in these granitic-derived soils is limited, with mineralogical and oxide controls prevailing over simple leaching or vertical transport mechanisms [43]. Major oxide concentrations on an oven-dry (110 °C) basis are summarized in Table 2.

Table 2.
Comparison of major oxide concentrations (%) on an oven-dry basis reported in the literature, including data for granitic-derived soils and Thai paddy soils [32,44,45].
3.3. Multivariate Analysis of Trace Elements
3.3.1. Correlation Structure of Trace Elements
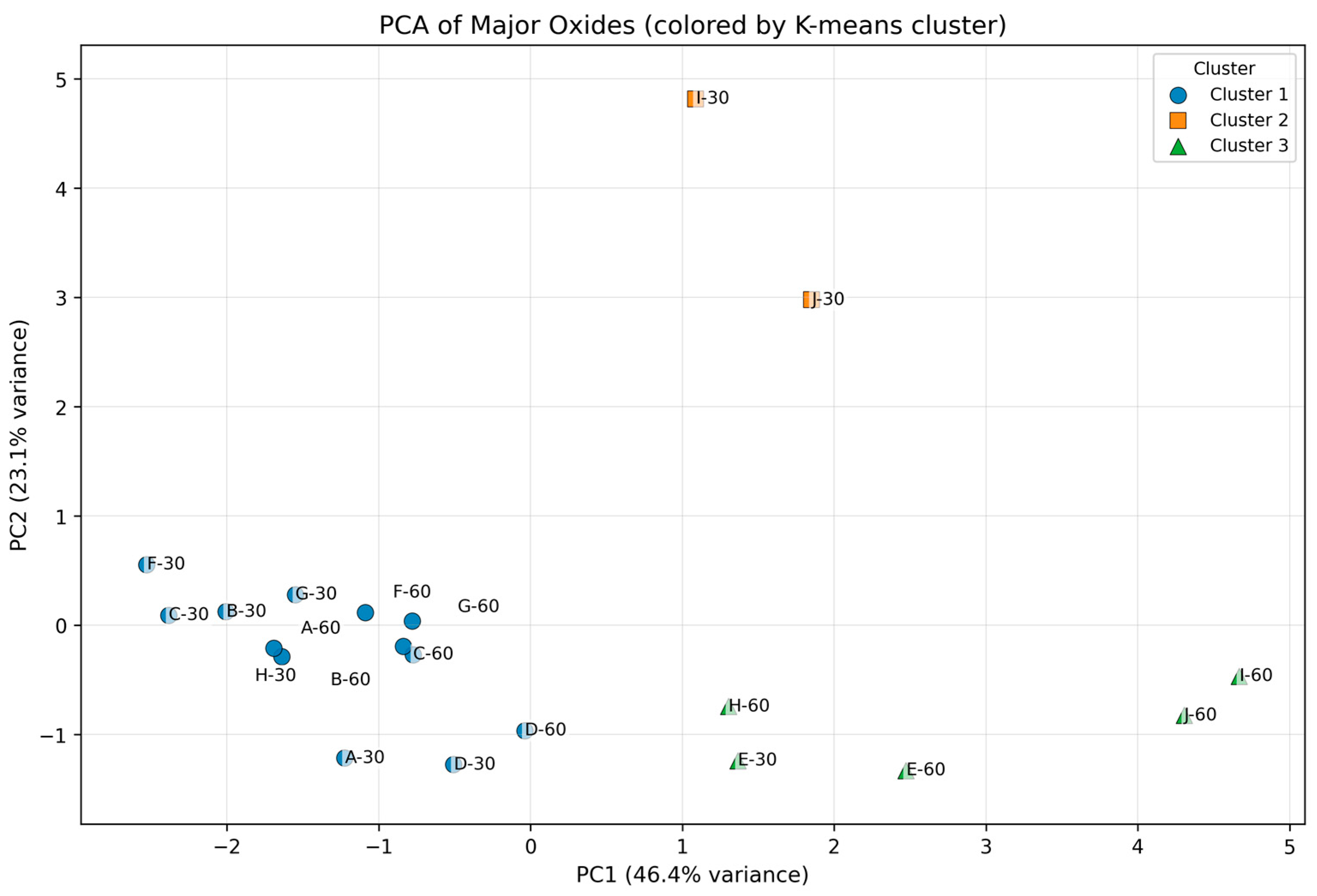
Principal Component Analysis (PCA) was employed as an exploratory multivariate approach to reduce dimensionality and mitigate multicollinearity among major and trace elements, thereby facilitating identification of the dominant geochemical gradients influencing soil composition. Prior to PCA, all variables were centered and scaled using Z-score normalization to ensure equal weighting of elements with differing units and variances. K-means clustering was subsequently applied to PCA scores to classify samples within a standardized, low-dimensional space highlighting meaningful compositional variation. Clustering was performed using the scores of the first two principal components (PC1 and PC2), which together explain the majority of variance in the major-oxide dataset (Figure 2).
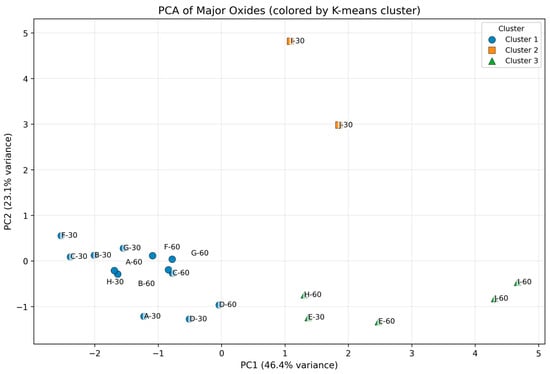
Figure 2.
Principal Component Analysis (PCA) of Z-score–standardized major oxides (SiO2, Al2O3, Fe2O3, MgO, CaO, Na2O, K2O, TiO2, MnO). Samples are grouped using k-means clustering (k = 3) based on PC1–PC2 scores.
The number of clusters (k = 3) was selected based on convergence of the within-cluster sum of squares and the geochemical interpretability of the resulting groupings. Rather than relying on formal cluster optimality diagnostics, the clustering solution is supported by its consistency with independent geochemical evidence, including major-oxide distributions, trace-element correlation patterns, depth-related trends, and hierarchical clustering results. Accordingly, the multivariate analysis is interpreted descriptively, with emphasis on robust compositional structure rather than inferential cluster validation.
Multivariate analysis integrating both major and trace elements revealed coherent and interpretable geochemical groupings across the AIRID soils. Cluster 1 comprises most samples and is characterized by slightly higher standardized Na2O and K2O and comparatively lower Al2O3–Fe2O3–TiO2, indicating relatively alkali-richer compositions within the dataset. Cluster 2 contains only I-30 and J-30 and shows strong SiO2 enrichment coupled with pronounced depletion of Na2O and K2O, separating these silica-rich samples from the main population. Cluster 3 is enriched in Al2O3, Fe2O3, TiO2, MnO, and MgO and depleted in SiO2 and K2O, consistent with a more oxide/residual-mineral–influenced composition.
Standardized values represent deviations from the dataset mean expressed in units of standard deviation and indicate relative enrichment or depletion rather than absolute concentration differences. When interpreted together with PCA loadings, these values define cluster positions along dominant geochemical gradients and link cluster differentiation primarily to mineralogical composition and weathering processes rather than concentration magnitude alone, as illustrated in Figure 2.
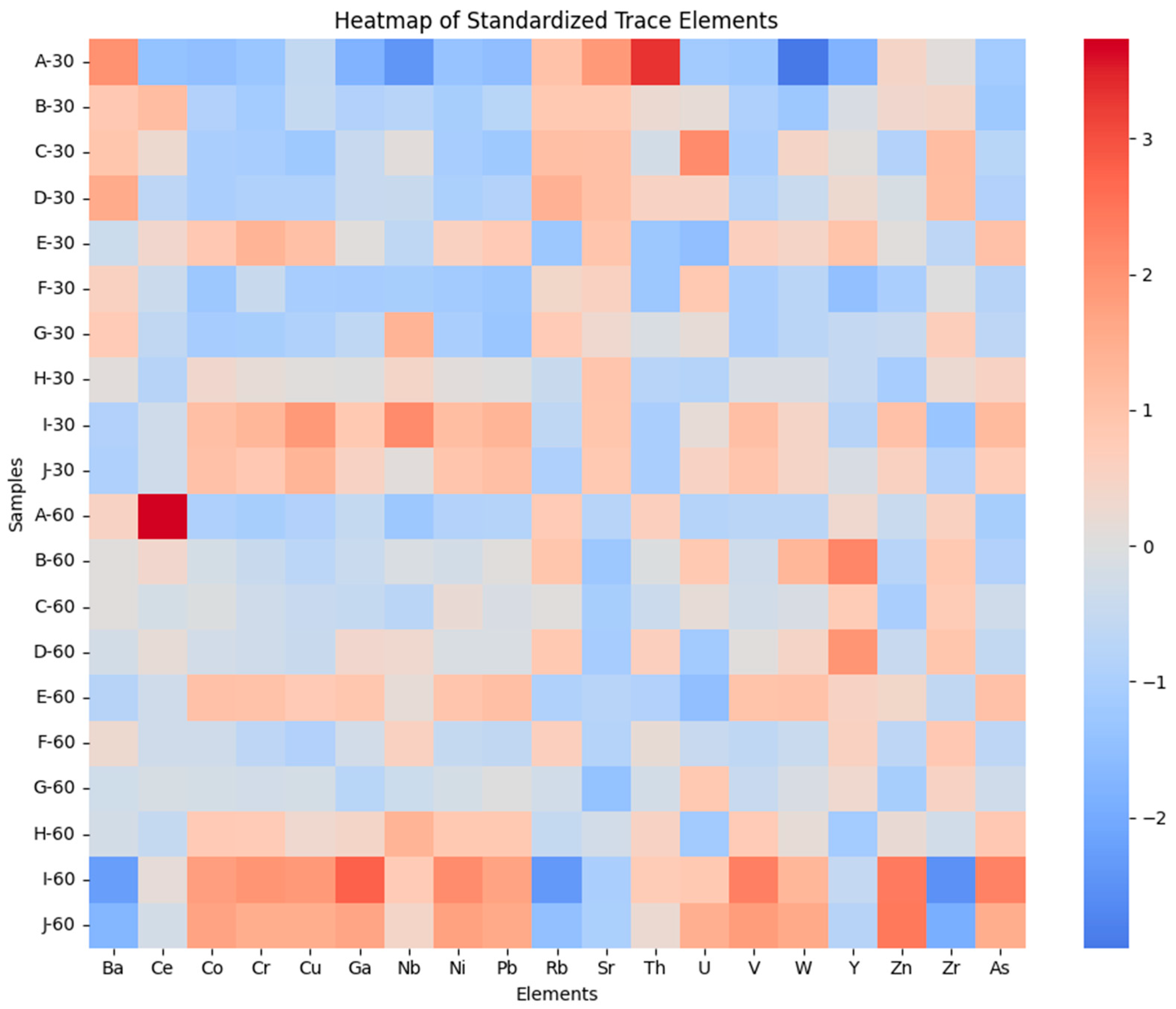
Trace-element distributions closely mirror the major-element clustering structure. As shown in the standardized trace-element heatmap (Figure 3), mafic-associated elements.
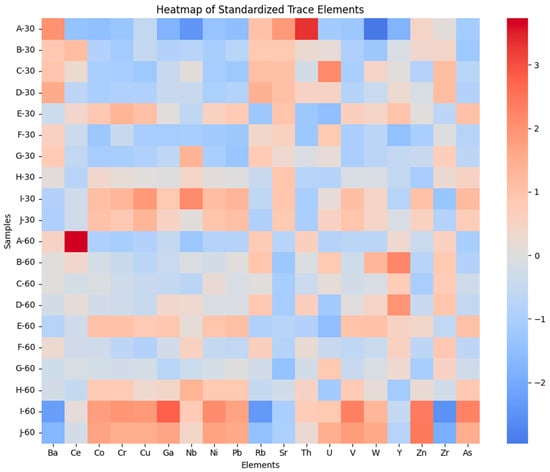
Figure 3.
Heatmap of Z-score–standardized trace-element concentrations across AIRID soil samples. Rows represent individual samples (0–30 cm and 30–60 cm), and columns represent trace elements. Values are standardized to the dataset mean and standard deviation (Z-scores), where positive values indicate relative enrichment and negative values indicate relative depletion.
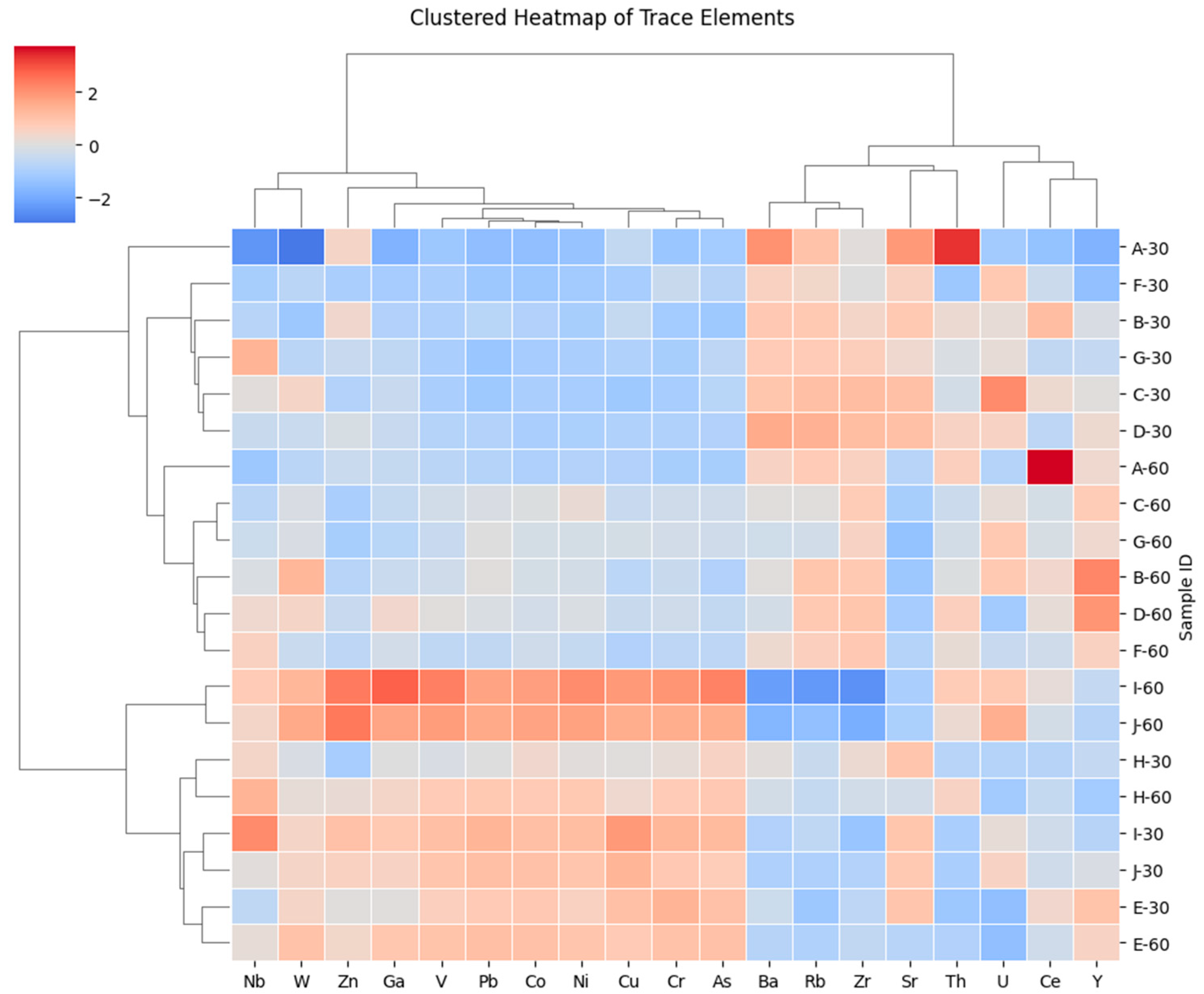
Hierarchical clustering of standardized trace-element data in Figure 4 further supports these relationships, separating trace elements into mafic-derived, felsic-derived, and accessory-mineral-associated groups, while samples cluster primarily according to geochemical similarity rather than soil depth. This pattern indicates that parent-material inheritance exerts a stronger control on trace-element distributions than vertical redistribution processes.
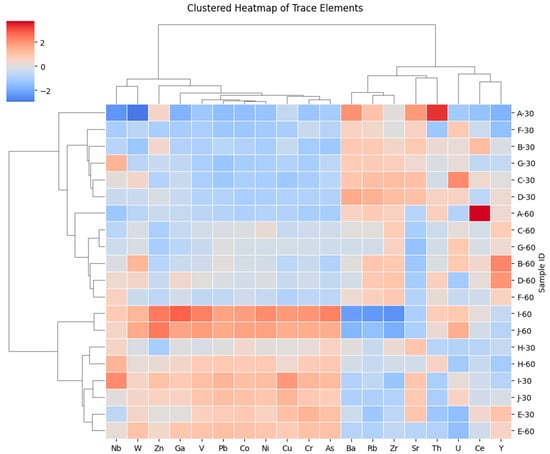
Figure 4.
Hierarchically clustered heatmap of Z-score–standardized trace-element concentrations. Dendrograms indicate similarity-based clustering of samples and elements using standardized concentrations (Z-scores), highlighting groups of elements that co-vary across the dataset and samples with similar trace-element signatures.
The supplementary Figure S2 provided a strong positive relationship observed between total phosphorus (P2O5 determined by XRF) and Bray II–extractable phosphorus in surface soils (Pearson correlation, r = 0.97, p < 0.001). Paired t-tests comparing surface (0–30 cm) and subsurface (30–60 cm) soils were conducted for major oxides and trace elements. To account for multiple comparisons, p-values were adjusted using the Benjamini–Hochberg false discovery rate (FDR) procedure. After adjustment, P2O5 remained significantly different between depths (FDR-adjusted p < 0.05), whereas all other elements showed no statistically significant depth-related differences (adjusted p > 0.05).
3.3.2. Implications for Soil Genesis
The multivariate geochemical patterns indicate that the highland soils of Chiang Mai retain strong mineralogical signatures inherited from their granitic parent materials. Coherent mafic-associated signals reflect limited mobility of transition metals, while the distinct behavior of Ba and Rb highlights persistent contributions from K-bearing minerals relevant to potassium availability in coffee agroecosystems. Accessory-element patterns suggest ongoing weathering of trace minerals coupled with sorption onto Fe–Al oxide surfaces.
Chiang Mai Province is recognized for its elevated natural background radiation levels in Thailand [16], with reported average soil 40K activities of approximately 0.7 kBq kg−1, exceeding global averages [16,46]. In the present study, soils from the Chiang Mai coffee field exhibit exceptionally high 40K activity concentrations, ranging from 1.2 to 1.9 kBq kg−1, corresponding to approximately three to five times the global average soil value (~0.4 kBq kg−1) [13,22].
4. Discussion
4.1. Soil Geochemical Characteristics and Fertility Implications
The increased concentrations of SiO2 and Al2O3 present in the examined soils substantially influence soil fertility and physical properties. Elevated SiO2 levels signify the dominance of quartz and silica-rich sand and silt constituents, which are chemically inert and contribute negligibly to nutrient availability [1]. Thus, soils with high SiO2 concentrations typically exhibit poor inherent nutrient stocks and limited buffering capacity, making crop yields largely dependent on external fertilizer applications and organic matter fluctuations [47]. Elevated Al2O3 concentrations signify the prevalence of aluminosilicate clay minerals and aluminum-rich secondary phases generated by extensive weathering [48]. Although these minerals enhance soil structure and aggregation, they are typically linked to a poor cation exchange capacity per unit mass and a high sorption of phosphorus, hence diminishing its availability to plants. Moreover, aluminum-rich soils frequently display acidic conditions, which can further restrict nutrient availability and root development if inadequately handled [34]. The combined presence of quartz-rich coarse fractions and clay-rich aluminum-bearing phases results in soils that are generally well drained but susceptible to erosion and characterized by moderate to poor water-holding capacity. These properties are typical of strongly weathered tropical soils derived from granitic parent material and highlight the need for careful soil management practices to sustain long-term soil fertility and agricultural productivity.
The amalgamation of quartz-rich coarse fractions and clay-rich aluminum-bearing phases results in well-drained yet erosion-susceptible soils with moderate to poor water retention capacity [49]. These characteristics are indicative of extensively worn tropical soils originating from granitic parent material and necessitate meticulous management, including the incorporation of organic matter and erosion mitigation, to maintain enduring soil fertility and agricultural output [49].
4.2. Potassium Content and 40K Activity Concentrations
The AIRID soils exhibit substantially higher than reported medians for Thai paddy soils (0.8%) and global averages (1.7%) [32]. Other major oxides (SiO2, Fe2O3, CaO, MgO) fall within ranges typical of strongly weathered granitic soils in the region. The measured activities are higher than those reported for soils in the Kong Khaek subdistrict of Chiang Mai (0.2–1.1 kBq kg−1; Autsavapromporn et al., 2024) [16]. The elevated K2O contents and corresponding 40K activities indicate that the AIRID soils possess a naturally high potassium reservoir, which may reduce the need for supplemental K fertilization in Arabica coffee cultivation. The average specific activity of 40K in soil: 0.4 ± 0.024 kBq kg−1 [13,46]. These values exceed global terrestrial 40K averages but remain well below the worldwide average natural radiation exposure of 2.4 mSv yr−1 under the defined outdoor exposure scenario, indicating elevated natural background but no radiological concern for agricultural workers [13]. The results are interpreted cautiously with emphasis on consistent geochemical patterns rather than isolated significance values. Full major oxide data for all samples are provided in Supplementary Table S1.
In addition to 40K, the activity concentrations of 238U- and 232Th-series radionuclides were evaluated to provide a comprehensive radiological characterization. While measurable, their activities fall within typical background ranges for granitic terrains and contribute less to the total absorbed dose compared with 40K. The relative dose contribution is dominated by 40K, followed by the 232Th-series and minor contributions from the 238U-series, a distribution characteristic of highly weathered granitic systems in tropical environments.
The combined geochemical and radiological evidence therefore indicates that radionuclide activities are predominantly lithogenic in origin, reflecting mineral inheritance from the granitic parent material rather than agricultural inputs or anthropogenic contamination.
4.3. Phosphorus Dynamics
Phosphorus data indicate that total P content is a strong predictor of plant-available P in the surface soil layer. The surface-to-subsurface P2O5 ratio of 2.5 demonstrates marked phosphorus enrichment in the upper 0–30 cm, consistent with surface fertilizer application, organic matter turnover, and the well-documented low vertical mobility of phosphorus in humid tropical soils. These results highlight the dominant influence of surface management practices on phosphorus availability within the soil profile.
The distribution of P2O5 between soil depths, together with its relationship to extractable phosphorus, indicates a system characterized by strong phosphorus retention dominated by Fe-oxide interactions. This pattern is consistent with ferrallitic weathering processes typical of granitic highland soils, where intense leaching and secondary mineral formation promote the accumulation of Fe–Al oxides capable of strongly adsorbing phosphate [50]. The pronounced enrichment of extractable P in surface soils is consistent with repeated fertilizer applications, a common phenomenon in managed tropical cropping systems [51]. The positive correlation between total P and extractable P suggests progressive accumulation at the surface with limited downward transport, reflecting the low mobility of phosphate in highly weathered soils [52]. The dominance of Fe and Al oxides—particularly goethite, hematite, and gibbsite—enhances phosphate adsorption through ligand exchange mechanisms, thereby restricting vertical movement but permitting gradual long-term buildup in the upper horizons [53]. This immobilization behavior has implications for soil fertility, as strongly adsorbed P may become less plant-available over time, while excess surface accumulation can increase the risk of P loss through erosion or runoff under high-rainfall conditions [54,55,56,57,58].
In ferrallitic weathering conditions characteristic of granitic highland soils, vigorous leaching facilitates the development and buildup of secondary Fe and Al oxides, including goethite, hematite, and gibbsite, which demonstrate a strong affinity for phosphate [59]. Phosphate is predominantly immobilized by inner-sphere ligand exchange processes, in which phosphate anions substitute surface hydroxyl groups on Fe–Al oxide minerals, resulting in robust and frequently irreversible surface complexes [60].
This mechanism limits vertical phosphorus mobility while facilitating surface accumulation, aligning with the observed concentration of extractable phosphorus in topsoil layers and the positive association between total and extractable phosphorus [61]. From a fertility standpoint, robust phosphate sorption diminishes immediate phosphorus availability to plants, hence heightening need on fertilizer applications to sustain sufficient soil solution phosphorus concentrations [61]. Over time, repeated fertilizer applications can result in the gradual accumulation of phosphorus in surface horizons, where heavily adsorbed phosphorus may function as a slow-release reservoir. Conversely, significant surface buildup elevates the danger of phosphorus loss by erosion or runoff during high-rainfall events, despite minimal leaching, underscoring the necessity for meticulous nutrient management in highly worn tropical soils [62].
4.4. Element Interpretation
Element mobility in the examined soils is controlled by lithogenic and pedogenic variables, with soil mineralogy and oxide concentration playing important roles in regulating contaminant dispersion and retention [33]. Granitic parent material contributes to heightened background levels of naturally occurring radionuclides and trace elements, corroborating findings that soil mineralogy significantly affects radionuclide behavior and retention in natural systems, as radionuclide interactions with soil constituents are governed by sorption and immobilization mechanisms linked to minerals [33]. Iron and aluminum oxide phases are prevalent in extensively worn tropical soils and have substantial specific surface areas that effectively adsorb trace elements and radionuclides, hence restricting their mobility [34]. Iron and aluminum oxides and oxyhydroxides affect adsorption processes, ion transport, and the stability of element sorption in soils, serving as significant sinks for elements and diminishing their vertical mobility, according to accepted soil geochemistry principles [34]. Moreover, radionuclide investigations have demonstrated that immobile species frequently correlate with weakly crystalline Fe/Al minerals and organic matter under standard soil conditions, hence further impeding their mobility within the profile [35].
Agricultural management approaches enhance the localized accumulation of nutrient-related elements (e.g., P, K) but typically do not surpass the pedogenic influence on contaminant spread [36,37,38,39]. The existence of Fe–Al oxides and clay minerals in these granitic soils increases sorption capacity, thereby restricting downward transit, even for elements introduced via fertilization [40,41,42]. The integrated geochemical and radiological data suggest that the mobility of elements in these granitic-derived soils is minimal, with mineralogical and oxide factors prevailing over basic leaching or vertical transport mechanisms [43].
4.5. Implications for Coffee Agronomy
Elemental distributions, especially K, Ca, Mg, and P behavior, are directly relevant to nutrient availability for coffee plants. High natural K reservoirs reduce dependency on fertilizer inputs, while P retention indicates potential need for P management strategies. A higher surface-to-subsurface P2O5 ratio indicates that phosphorus is more concentrated in the surface soil layer compared to deeper layers. This pattern reflects the accumulation of phosphorus, which might be due to surface applications of fertilizers, organic matter decomposition, and limited vertical movement of phosphorus in the soil profile.
4.6. Trace Element Behavior and Geochemical Implications
Trace element distributions in the studied soils reveal a strong lithogenic imprint that reflects the mineralogical composition of the granitic parent material. Elements such as Co, Ni, Cr, V, Cu, Ga, Pb, Zn, and As form a coherent group and consistently show strong positive relationships across both depths. These trace metals are primarily released during the weathering of biotite and amphibole, minerals that host transition metals within their octahedral layers. Their close inter-element associations and limited mobility in acidic, well-drained tropical soils allow these lithogenic signatures to be preserved even after long-term weathering [63].
In contrast, Ba, Rb, and Zr indicate the influence of felsic mineral components. Ba and Rb derive from K-feldspar and mica, while Zr originates almost exclusively from zircon, a highly resistant accessory mineral. The chemical durability of zircon explains the stability of Zr concentrations across depths and its well-established role as an indicator of cumulative weathering and mineral residue enrichment [64]. The opposite behavior of this felsic-associated group relative to the mafic-derived elements highlights the contrasting contributions of different mineral phases within the parent rock.
A third trace element association, including Th, U, Y, Nb, Sr, and W, reflects inputs from accessory minerals such as monazite, xenotime, apatite, and zircon, together with strong sorption by Fe- and Al-oxide phases. These oxide minerals are abundant in highly weathered granitic soils and play a key role in immobilizing many trace metals. In particular, the low mobility and oxide affinity of Th and U indicate strong retention within the soil matrix, with minimal leaching under current environmental conditions [65].
The distribution of trace elements in the AIRID soils demonstrates significant lithogenic and pedogenic influence with restricted vertical mobility. Consistent associations of Co, Ni, Cr, V, Cu, Zn, Pb, and As at both depths, along with their robust inter-element correlations, indicate a shared mineralogical source related to the weathering of biotite and amphibole. The lack of notable depth-related variations signifies robust retention by clay minerals and Fe–Al oxides in acidic tropical environments [66].
Numerous elements, such as Cu and Zn, are vital micronutrients for coffee plants; yet, their significant interaction with oxide and clay phases suggests restricted bioavailability despite low overall concentrations [67,68]. The effects of fertilizers seem subordinate to mineralogical factors, as trace elements exhibit negligible vertical redistribution and are predominantly retained in surface soils [69]. The findings indicate minimal trace-element mobility and a moderate toxicity risk, while emphasizing the possibility of micronutrient deficiencies in prolonged coffee cultivation due to robust sorption mechanisms. Depth-wise comparisons of trace element concentrations are summarized in Table 3.

Table 3.
Comparison of trace element concentrations between surface (0–30 cm) and subsurface (30–60 cm) soils.
Depth variations in trace elements support the influence of fertilizer application on the upper soil horizon. Surface enrichment of Sr, U, Ba, and Rb is consistent with repeated inputs of P-containing fertilizers and nutrient cycling in the biologically active surface layer. Conversely, lithogenic elements (Cr, Ni, Co, V, Pb, Ga, Th, Y, Zn) exhibited slightly higher concentrations at 60 cm, reflecting their origin from parent material weathering and minimal vertical mobility. Overall, the depth distributions indicate that fertilizer-responsive elements accumulate near the surface, while most trace elements remain controlled by the granitic parent material.
These patterns have direct implications for soil fertility in highland coffee systems. High natural K-bearing mineral content provides an intrinsic reservoir of potassium, reducing dependence on external fertilizer inputs. At the same time, strong P fixation by Fe- and Al-oxides may constrain phosphorus availability despite moderate total P concentrations [70]. Furthermore, the predictable release of micronutrients from mafic minerals, such as Ni, Zn, and Cu, supports a stable micronutrient supply for coffee growth. Overall, the trace element distributions reflect a soil environment in which mineral inheritance outweighs pedogenic redistribution, offering valuable insights for guiding nutrient management and sustaining coffee production in granitic-derived highland ecosystems.
5. Conclusions
This study illustrates that the geochemical properties of the AIRID highland coffee soils are predominantly dictated by granitic parent material, with mineralogical inheritance having a more significant impact than vertical pedogenic redistribution. Major oxide and trace element distributions indicate extensive tropical weathering, marked by elevated levels of SiO2, Al2O3, and K2O, and a reduction in CaO and MgO. Consistent trace element relationships further validate lithogenic control, exhibiting restricted mobility throughout soil depths. The behavior of phosphorus suggests significant fixation by soils rich in Fe–Al oxides, which may limit phosphorus availability, while the presence of copious potassium-bearing minerals offers a considerable natural reserve of potassium that could diminish fertilizer needs. Collectively, our findings underscore the pivotal influence of parent material on nutrient availability and soil fertility in granitic highland coffee systems.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/geosciences16030110/s1. Figure S1: Comparison of measured and calculated activity concentrations of ⁴⁰K in soil samples from the AIRID coffee field, Chiang Mai, Thailand; Figure S2: Correlation between extractable phosphorus (Bray II P) and total phosphorus (P₂O₅) in soil samples from the AIRID coffee field, Chiang Mai, Thailand; Table S1: Major oxide composition (wt%, dried) of soil samples from the AIRID coffee field, Chiang Mai, Thailand, determined by XRF at depths of 30 cm and 60 cm.
Author Contributions
Conceptualization, K.K. (Khemruthai Kheamsiri) and N.A.; methodology, N.A., I.R., H.T., Y.U. and A.T.; software, K.K. (Khemruthai Kheamsiri) and A.T.; validation, Y.U. and A.T.; formal analysis, K.K. (Khemruthai Kheamsiri), Y.U. and A.T.; investigation, K.K. (Khemruthai Kheamsiri), N.A., C.K., T.T., H.K., K.K. (Krit Khetanun) and N.Y.; resources, N.A.; data curation, K.K. (Khemruthai Kheamsiri); writing—original draft preparation, K.K. (Khemruthai Kheamsiri); writing—review and editing, N.A., C.K., H.T., T.T., I.R., H.K., K.K. (Krit Khetanun), N.Y., Y.U. and A.T.; visualization, N.A., Y.U. and A.T.; supervision, N.A., Y.U. and A.T.; project administration, N.A.; funding acquisition, N.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the Sasakawa Scientific Research Grant, administered by the Sasakawa Peace Foundation, Grant No. 2024-3026, and by JST SPRING, Grant Number JPMJSP2152. Additionally, the research materials were imported from overseas under the Ministerial Licensing System.
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding authors.
Acknowledgments
The authors used ChatGPT (OpenAI, USA) as a supporting tool for data organization, preliminary statistical exploration, and improvement of English language clarity. All final interpretations, scientific judgments, and conclusions were made by the authors.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| HPGe | High-Purity Germanium (gamma ray detector) |
| XRF | X-ray Fluorescence |
| MCR | Molybdenum Color Reagent |
| WRB | World Reference Base for Soil Resources |
| AIRID | Agricultural Innovation Research, Integration, Demonstration, and Training Center |
References
- De Bauw, P.; Van Asten, P.; Jassogne, L.; Merckx, R. Soil fertility gradients and production constraints for coffee and banana on volcanic mountain slopes in the East African Rift: A case study of Mt. Elgon. Agric. Ecosyst. Environ. 2016, 231, 166–175. [Google Scholar] [CrossRef]
- Liang, W. Petrology and Multimineral Fingerprinting of Modern Sand Derived from the Himalayan Orogen. Ph.D. Thesis, Università degli Studi di Milano-Bicocca, Milan, Italy, 2020. [Google Scholar]
- DaMatta, F.M.; Ronchi, C.P.; Maestri, M.; Barros, R.S. Ecophysiology of coffee growth and production. Braz. J. Plant Physiol. 2007, 19, 485–510. [Google Scholar] [CrossRef]
- Jahn, R.; Blume, H.-P.; Asio, V.B.; Spaargaren, O.; Schad, P. Guidelines for Soil Description, 4th ed.; FAO: Rome, Italy, 2006. [Google Scholar]
- Da Silva, Y.J.A.B.; Do Nascimento, C.W.A.; Biondi, C.M.; Van Straaten, P.; De Souza, V.S.; Ferreira, T.O. Geochemical signatures and weathering rates in soils derived from different granites in contrasting climatic locations. Acta Sci. Agron. 2019, 41, e39708. [Google Scholar] [CrossRef]
- Wang, W.; Feng, J.; Qiu, M. Mineral weathering and element migration in granite weathering pits (gnammas): A case study in eastern China. Minerals 2023, 13, 70. [Google Scholar] [CrossRef]
- El Attar, I.; Hnini, M.; Taha, K.; Aurag, J. Phosphorus availability and its sustainable use. J. Soil Sci. Plant Nutr. 2022, 22, 5036–5048. [Google Scholar] [CrossRef]
- Ogwu, M.C.; Patterson, M.E.; Senchak, P.A. Phosphorus mining and bioavailability for plant acquisition: Environmental sustainability perspectives. Environ. Monit. Assess. 2025, 197, 572. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.; Iqbal, M.; Tang, Y.-T.; Khan, S.; Guan, D.-X.; Li, G. Phosphorus mobilization in plant–soil environments and inspired strategies for managing phosphorus: A review. Agronomy 2022, 12, 2539. [Google Scholar] [CrossRef]
- Kumar, S.; David Raj, A.; Kalambukattu, J.G.; Chatterjee, U. Climate change impact on land degradation and soil erosion in hilly and mountainous landscape: Sustainability issues and adaptation strategies. In Ecological Footprints of Climate Change: Adaptive Approaches and Sustainability; Springer: Cham, Switzerland, 2022; pp. 119–155. [Google Scholar] [CrossRef]
- Brenko, T.; Ružičić, S.; Radonić, N.; Puljko, M.; Cvetković, M. Geochemical factors as a tool for distinguishing geogenic from anthropogenic sources of potentially toxic elements in the soil. Land 2024, 13, 434. [Google Scholar] [CrossRef]
- Chandra, K.; Proshad, R.; Dey, H.C.; Idris, A.M. A review on radionuclide pollution in global soils with environmental and health hazards evaluation. Environ. Geochem. Health 2023, 45, 9245–9266. [Google Scholar] [CrossRef]
- UNSCEAR. Sources and Effects of Ionizing Radiation. In UNSCEAR 2000 Report to the General Assembly with Scientific Annexes, Volume I: Sources; United Nations: New York, NY, USA, 2000. [Google Scholar]
- Abdel Karim, M.; Gafaar, I.; Abd El-Halim, E.; Hanfi, M.; Walley El-Dine, N. Natural radioactivity and radiological implications of granite rocks, El-Sela area, southeastern desert, Egypt. J. Radioanal. Nucl. Chem. 2021, 330, 707–720. [Google Scholar] [CrossRef]
- Heikal, M.T.S.; Kamar, M.S.; Azer, M.K.; Ibrahim, M.O.; Monsef, M.A. Natural radioactivity levels (238U, 232Th, 40K) and geochemical characteristics of granitic rocks from Nuweibi area, Egyptian Nubian Shield. Radiochemistry 2023, 65, 497–509. [Google Scholar] [CrossRef]
- Autsavapromporn, N.; Kranrod, C.; Kritsananuwat, R.; Sola, P.; Klunklin, P.; Chitapanarux, I.; Jaikang, C.; Monum, T.; Hosoda, M.; Tokonami, S. Health impacts of natural background radiation in high air pollution area of Thailand. Toxics 2024, 12, 428. [Google Scholar] [CrossRef]
- Ramos-Lerate, I.; Lozano-Bermejo, R.; Burneo, J.I.; Armijos, C.; Piñero, M.; Bolívar, J.P.; Gázquez, M.J. Radiological assessment of both unperturbed and agricultural soils from southern Ecuador. J. Agric. Food Res. 2024, 17, 101236. [Google Scholar] [CrossRef]
- Taher Hussien, M.; Salaheldin, G.; Salaheldin Mohamed, H.; Mansour, H. Distribution of natural radionuclides and their radiological risks on agricultural soil samples collected from Yemen. Pollution 2023, 9, 195–210. [Google Scholar]
- Coltro, L.; Mourad, A.L.; Oliveira, P.A.P.L.V.; Baddini, J.P.O.A.; Kletecke, R.M. Environmental profile of Brazilian green coffee. Int. J. Life Cycle Assess. 2006, 11, 16–21. [Google Scholar] [CrossRef]
- Leal, A.L.D.C.; Lauria, D.d.C.; Ribeiro, F.C.; Viglio, E.P.; Franzen, M.; Lima, E.d.A.M. Spatial distributions of natural radionuclides in soils of the state of Pernambuco, Brazil: Influence of bedrocks, soils types and climates. J. Environ. Radioact. 2020, 211, 106046. [Google Scholar] [CrossRef]
- Soilueang, P.; Jaikrasen, K.; Chromkaew, Y.; Buachun, S.; Yimyam, N.; Sanjunthong, W.; Kullachonphuri, S.; Wicharuck, S.; Mawan, N.; Khongdee, N. Dynamics of soil nitrogen availability following conversion of natural forests to various coffee cropping systems in northern Thailand. Heliyon 2023, 9, e22988. [Google Scholar] [CrossRef]
- Jundee, P.K.; Chaiwchan, S.; Inthacharoensarn, S.; Phajuy, B.; Nontarak, P. Petrochemistry and tectonic setting of the Phroa–Wiang Pa Pao granitic rocks, Chiang Mai, and Chiang Rai provinces, central granitic belt of Thailand. Trends Sci. 2024, 21, 8055. [Google Scholar] [CrossRef]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015; FAO: Rome, Italy, 2015. [Google Scholar]
- Agricultural Innovation Research, Integration, Demonstration and Training Center (AIRID). Highland Coffee Research and Training Programs. Available online: https://www.agri.cmu.ac.th/2017/webs/mainmenu_detail/3/383/3 (accessed on 19 January 2026).
- Guiscafré-Arrillaga, J.; Gómez, L.A. Studies of the root system of Coffea arabica L. Part I. Environmental conditions affecting the distribution of coffee roots in coloso clay. J. Agric. 1938, 22, 227–262. [Google Scholar]
- Schmidt, R.; Silva, L.O.E.; Ferreira, A.; Rodrigues, W.P.; Vieira, H.D.; Tomaz, M.A.; Partelli, F.L. Root system of Coffea canephora cv. Robusta genotypes cultivated in the Brazilian Amazon. Discover Agric. 2024, 2, 32. [Google Scholar] [CrossRef]
- Defrenet, E.; Roupsard, O.; den Meersche, K.; Charbonnier, F.; Pérez-Molina, J.; Khac, E.; Prieto, I.; Stokes, A.; Roumet, C.; Rapidel, B.; et al. Root biomass, turnover and net primary productivity of a coffee agroforestry system in Costa Rica: Effects of soil depth, shade trees, distance to row and coffee age. Ann. Bot. 2016, 118, 833–851. [Google Scholar] [CrossRef]
- Silva, L.O.E.; Schmidt, R.; Valani, G.P.; Ferreira, A.; Ribeiro-Barros, A.I.; Partelli, F.L. Root trait variability in Coffea canephora genotypes and its relation to plant height and crop yield. Agronomy 2020, 10, 1394. [Google Scholar] [CrossRef]
- Ministry of Agriculture and Cooperatives, Land Development Department (LDD), Thailand. Available online: https://dinonline.ldd.go.th/Default.aspx (accessed on 18 January 2026).
- Hosoda, M.; Tokonami, S.; Tazoe, H.; Sorimachi, A.; Monzen, S.; Osanai, M.; Akata, N.; Kakiuchi, H.; Omori, Y.; Ishikawa, T. Activity concentrations of environmental samples collected in Fukushima Prefecture immediately after the Fukushima nuclear accident. Sci. Rep. 2013, 3, 2283. [Google Scholar] [CrossRef]
- Jolliffe, I.T.; Cadima, J. Principal component analysis: A review and recent developments. Philos. Trans. R. Soc. A 2016, 374, 20150202. [Google Scholar] [CrossRef] [PubMed]
- de Caritat, P.; Reimann, C.; NGSA Team; GEMAS Team. Comparing results from two continental geochemical surveys to world soil composition and deriving predicted empirical global soil (PEGS2) reference values. Earth Planet. Sci. Lett. 2012, 319–320, 269–276. [Google Scholar] [CrossRef]
- Guo, Y.; Fei, X. Insights on experimental methodologies and theoretical models for microplastics transport in soils and sediments based on meta-analysis. Acta Geotech. 2023, 18, 4477–4492. [Google Scholar] [CrossRef]
- Kirsten, M.; Mikutta, R.; Vogel, C.; Thompson, A.; Mueller, C.W.; Kimaro, D.N.; Bergsma, H.L.T.; Feger, K.-H.; Kalbitz, K. Iron oxides and aluminous clays selectively control soil carbon storage and stability in the humid tropics. Sci. Rep. 2021, 11, 5076. [Google Scholar] [CrossRef]
- Iurian, A.R.; Phaneuf, M.O.; Mabit, L. Mobility and bioavailability of radionuclides in soils. In Radionuclides in the Environment: Influence of Chemical Speciation and Plant Uptake on Radionuclide Migration; Springer: Cham, Switzerland, 2015; pp. 37–59. [Google Scholar] [CrossRef]
- Jia, L. Soil and water management: Practices to mitigate nutrient losses in agricultural watersheds. Water 2024, 16, 2269. [Google Scholar] [CrossRef]
- Biswas, S.; Singh, S.; Bera, A. Regenerative nutrient management practices for enhancing plant nutrition and soil health. In Regenerative Agriculture for Sustainable Food Systems; Springer: Singapore, 2024; pp. 303–339. [Google Scholar] [CrossRef]
- Liang, X.; Yu, S.; Ju, Y.; Wang, Y.; Yin, D. Integrated management practices foster soil health, productivity, and agroecosystem resilience. Agronomy 2025, 15, 1816. [Google Scholar] [CrossRef]
- Paramesh, V.; Mohan Kumar, R.; Rajanna, G.A.; Gowda, S.; Nath, A.J.; Madival, Y.; Jinger, D.; Bhat, S.; Toraskar, S. Integrated nutrient management for improving crop yields, soil properties, and reducing greenhouse gas emissions. Front. Sustain. Food Syst. 2023, 7, 1173258. [Google Scholar] [CrossRef]
- Van Groeningen, N.; Thomasarrigo, L.K.; Byrne, J.M.; Kappler, A.; Christl, I.; Kretzschmar, R. Interactions of ferrous iron with clay mineral surfaces during sorption and subsequent oxidation. Environ. Sci. Process. Impacts 2020, 22, 1607–1620. [Google Scholar] [CrossRef]
- Oyebiyi, O.O. Distribution and forms of iron and aluminium oxides in tropical soils of central southwestern Nigeria. Environ. Earth Sci. 2024, 83, 552. [Google Scholar] [CrossRef]
- Kirsten, M.; Mikutta, R.; Kimaro, D.N.; Feger, K.-H.; Kalbitz, K. Aluminous clay and pedogenic Fe oxides modulate aggregation and related carbon contents in soils of the humid tropics. SOIL 2021, 7, 363–382. [Google Scholar] [CrossRef]
- Beaver, R.C.; Neufeld, J.D. Microbial ecology of the deep terrestrial subsurface. ISME J. 2024, 18, wrae091. [Google Scholar] [CrossRef]
- Prakongkep, N.; Suddhiprakarn, A.; Kheoruenromne, I.; Smirk, M.; Gilkes, R. The geochemistry of Thai paddy soils. Geoderma 2008, 144, 310–324. [Google Scholar] [CrossRef]
- Hirose, M.; Yanai, J.; Tanaka, S.; Nakao, A.; Dejbhimon, K.; Sriprachote, A.; Kanyawongha, P.; Lattirasuvan, T.; Abe, S.S.; Nawata, E. Changes in paddy soil fertility in Thailand in relation to total content of elements and micronutrient status under the Green Revolution from the 1960s to the 2010s. Trop. Agric. Dev. 2022, 66, 33–43. [Google Scholar]
- UNSCEAR. Sources and Effects of Ionizing Radiation. In UNSCEAR 2008 Report to the General Assembly with Scientific Annexes, Volume I; United Nations: New York, NY, USA, 2008. [Google Scholar]
- Bashir, O.; Ali, T.; Baba, Z.A.; Rather, G.H.; Bangroo, S.A.; Mukhtar, S.D.; Naik, N.; Mohiuddin, R.; Bharati, V.; Bhat, R.A. Soil organic matter and its impact on soil properties and nutrient status. In Microbiota and Biofertilizers; Springer: Cham, Switzerland, 2021; Volume 2, pp. 129–159. [Google Scholar] [CrossRef]
- Maj, I.; Matus, K. Aluminosilicate clay minerals: Kaolin, bentonite, and halloysite as fuel additives for thermal conversion of biomass and waste. Energies 2023, 16, 4359. [Google Scholar] [CrossRef]
- Jiao, W.; Huang, Y.; Zhao, D.; Zhang, H.; Wen, L.; Zhang, Y.; Guo, P.; Zhang, J. Characterization and application of quartz from different sources in typical shale reservoirs. Chem. Technol. Fuels Oils 2024, 60, 440–450. [Google Scholar] [CrossRef]
- Earle, S. Physical Geology; BCcampus: Victoria, BC, Canada, 2015. [Google Scholar]
- Doydora, S.; Gatiboni, L.; Grieger, K.; Hesterberg, D.; Jones, J.L.; McLamore, E.S.; Peters, R.; Sozzani, R.; Van den Broeck, L.; Duckworth, O.W. Accessing legacy phosphorus in soils. Soil Syst. 2020, 4, 74. [Google Scholar] [CrossRef]
- Herrera, D.; Mylavarapu, R.S.; Harris, W.G.; Colee, J. Soil phosphorus sources and their relative water solubility and extractability. Commun. Soil Sci. Plant Anal. 2022, 53, 2120–2137. [Google Scholar] [CrossRef]
- Torrent, J.; Barrón, V.; Schwertmann, U. Phosphate adsorption and desorption by goethites differing in crystal morphology. Soil Sci. Soc. Am. J. 1990, 54, 1007–1012. [Google Scholar] [CrossRef]
- Nur, M.A.; Kamruzzaman, M.; Amin, M.S. Microbial immobilization and phosphorus transformation in saline soil: Effects of organic amendments. J. Soil Sci. Plant Nutr. 2025, 25, 1387–1400. [Google Scholar] [CrossRef]
- Jindo, K.; Audette, Y.; Olivares, F.L.; Canellas, L.P.; Smith, D.S.; Voroney, R.P. Biotic and abiotic effects of soil organic matter on the phytoavailable phosphorus in soils: A review. Chem. Biol. Technol. Agric. 2023, 10, 29. [Google Scholar] [CrossRef]
- Zhou, S.; Li, N.; Margenot, A.J. Soil meets stream: Vertical distribution of soil phosphorus in streambanks. Geoderma 2022, 424, 115989. [Google Scholar] [CrossRef]
- Annappa, N.N.; Murthy, R.K.; Bhavya, N.; Govinda, K.; Kumar, S.N.U. Soil phosphorus distribution across diverse land use systems: A comprehensive review. J. Sci. Res. Rep. 2024, 30, 352–364. [Google Scholar] [CrossRef]
- Yang, X.; Post, W.M.; Thornton, P.E.; Jain, A. The distribution of soil phosphorus for global biogeochemical modeling. Biogeosciences 2013, 10, 2525–2537. [Google Scholar] [CrossRef]
- Van Ranst, E.; Mees, F.; De Grave, E.; Ye, L.; Cornelis, J.T.; Delvaux, B. Impact of andosolization on pedogenic Fe oxides in ferrallitic soils. Geoderma 2019, 347, 174–184. [Google Scholar] [CrossRef]
- Zhang, Z.; Yu, H.; Zhu, R.; Zhang, X.; Yan, L. Phosphate adsorption performance and mechanisms by nanoporous biochar–iron oxides from aqueous solutions. Environ. Sci. Pollut. Res. 2020, 27, 32574–32586. [Google Scholar] [CrossRef]
- Lei, J.; Yin, J.; Chen, S.; Fenton, O.; Liu, R.; Chen, Q.; Fan, B.; Zhang, S. Understanding phosphorus mobilization mechanisms in acidic soil amended with calcium–silicon–magnesium–potassium fertilizer. Sci. Total Environ. 2024, 916, 170294. [Google Scholar] [CrossRef]
- Mackey, K.R.M.; Van Mooy, B.A.S.; Cade-Menun, B.J.; Paytan, A. Phosphorus dynamics in the environment. In Reference Module in Life Sciences; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar] [CrossRef]
- Kaizer, J.; Hirose, K.; Povinec, P.P. Assessment of environmental impacts from authorized discharges of tritiated water from the Fukushima site to coastal and offshore regions. J. Environ. Radioact. 2024, 278, 107507. [Google Scholar] [CrossRef]
- Carrasco-Godoy, C.; Campbell, I.H.; Cajal, Y. Quantifying the criteria used to identify zircons from ore-bearing and barren systems in porphyry copper exploration. Econ. Geol. 2024, 119, 1035–1058. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, C. Trace element geochemistry during chemical weathering. Chin. Sci. Bull. 1999, 44, 2260–2263. [Google Scholar] [CrossRef]
- Steadman, J.A.; Large, R.R.; Olin, P.H.; Danyushevsky, L.V.; Meffre, S.; Huston, D.; Fabris, A.; Lisitsin, V.; Wells, T. Pyrite trace element behavior in magmatic–hydrothermal environments: An LA-ICP-MS imaging study. Ore Geol. Rev. 2021, 128, 103878. [Google Scholar] [CrossRef]
- Imtiaz, M.; Rashid, A.; Khan, P.; Memon, M.Y.; Aslam, M. The role of micronutrients in crop production and human health. Pak. J. Bot. 2010, 42, 2565–2578. [Google Scholar]
- Afzal, S.; Sirohi, P.; Sharma, D.; Singh, N.K. Micronutrient movement and signalling in plants from a biofortification perspective. In Plant Micronutrients: Deficiency and Toxicity Management; Springer: Cham, Switzerland, 2020; pp. 93–115. [Google Scholar] [CrossRef]
- Niu, Y.; Zhang, M.; Bai, S.H.; Xu, Z.; Liu, Y.; Chen, F.; Guo, X.; Zhang, L.; Luo, H.; Zhang, Q. Mineral fertilization and soil depth slightly affected aggregate structures despite significantly altered microbial properties in surface forest soils. J. Soils Sediments 2020, 20, 4184–4198. [Google Scholar] [CrossRef]
- Violante, A.; Cozzolino, V.; Perelomov, L.; Caporale, A.G.; Pigna, M. Mobility and bioavailability of heavy metals and metalloids in soil environments. J. Soil Sci. Plant Nutr. 2010, 10, 268–292. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.