Simple Summary
Modern poultry production is about converting growth performance into commercial value through improved feed efficiency and carcass quality. This can be achieved only with the use of specific feed additives and probiotics have emerged as one of the most promising alternatives to antibiotics in broiler nutrition. After conducting an experiment where the diets of broilers have been supplemented with a mixture of two probiotics, the results showed that they can enhance growth performance and improve carcass composition, even under challenging rearing conditions. Moreover, the probiotics supported the immune system of broilers and protected the intestine from epithelial injury. Therefore, according to these results, their efficacy is confirmed and when their use is coupled with precision nutrition approaches, their adoption by the poultry industry will certainly increase in the future.
Abstract
Probiotics have emerged as an important strategy to achieve improved feed efficiency and carcass quality. To evaluate the effects of a probiotic combination based on Weizmannia faecalis (formerly Bacillus coagulans) and Bacillus licheniformis on broiler performance, carcass, and intestinal health, a study was conducted. As-hatched ROSS 308 broilers were purchased from a local hatchery at day 0 and were randomly allocated to two treatments (160 birds per treatment; 8 replicates of 20 birds each): the control, which was fed a standard commercial diet throughout the experiment, and the probiotics group, where the standard diet was further supplemented with the probiotic combination. Feed and water were offered for ad libitum consumption while the feeding schedule was as follows: Starter, 1–10 days, mash; Grower, 11–24 days, mash; Finisher, 25–42, mash. The birds were challenged using re-used litter as bedding and the application of increased stocking density and mild heat stress. The final body weight of the probiotics’ supplemented group was higher than the control at 42 days of age (2822.7 g vs. 2575.4 g, respectively) (p < 0.05), and the overall feed conversion ratio was significantly reduced. The weight of all the commercial parts increased, along with the thigh and drumstick yield, thus indicating an improvement of carcass composition (p < 0.05). The European Poultry Efficiency Factor significantly improved following the probiotic dietary supplementation (409.7 vs. 344.9 of the control), while the probiotic fed birds had higher antibody titers for Bursal disease at 42 days and lower serum concentration of fatty acid binding protein 2 at 24 days (p < 0.05). Overall, the dietary supplementation of broilers with the probiotic mixture, under challenging rearing conditions, enhanced growth performance and improved carcass composition.
1. Introduction
Achieving optimal growth in broilers is not only about reaching higher final weights, but also about converting that growth into commercial value through improved feed efficiency and carcass quality. Historically, such performance gains in poultry production were strongly supported by antibiotic growth promoters (AGPs), which improved gut health, growth rate, and ultimately carcass development [1]. After their EU ban in 2006, due to concerns about antimicrobial resistance and consumer expectations, therapeutic antibiotic use has remained common only to manage disease, highlighting the ongoing need for effective non-pharmaceutical solutions [2].
Probiotics are defined by the WHO as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” [3] and have emerged as one of the most promising AGP alternatives in broiler nutrition and as an important preventive strategy to reduce antibiotic interventions [4]. The term microorganisms refers to bacteria (mainly belonging to the genera Bacillus, Lactobacillus, Enterococcus, and Bifidobacterium), but certain yeasts and fungal probiotics have also been used, e.g., Aspergillus oryzae, Candida pintolopesii, Saccharomyces and Saccharomyces cerevisiae.
Evidence indicates that probiotics can change the microbial population dynamics in the gastrointestinal tract by enhancing the multiplication of beneficial microbes and suppressing the harmful ones [5]. Moreover, they increase the digestion and absorption of nutrients [6] and prevent the chronic inflammation of the gastrointestinal tract through stimulation of innate immunity in the epithelium [7], overall resulting in enhanced performance and feed conversion.
Weizmannia faecalis DSM 32,016 (formerly classified as Bacillus coagulans) is a Gram-positive, facultative aerobic, and spore-forming bacterium with unique properties. It is a homofermentative lactic acid producer [8,9], performs very well in harsh environments (high temperatures and low pH) [10], and produces compounds with antimicrobial activity against Gram-positive and Gram-negative food pathogens [11]. These properties contribute to its wide use in supporting gut and overall health in both humans and broilers, as well as other farm animals [12,13].
Similarly, Bacillus licheniformis is a spore-forming, Gram-positive, facultatively aerobic bacterium that exerts heat and pH resistance and produces bacteriocins that mainly fight Gram-positive pathogens. It has been proven to enhance broilers’ growth performance [14] and to maintain gut microbial population balance [15]. It can also assist in preventing the occurrence of necrotic enteritis in chickens [16] and has been recognized by the European Food Safety Authority (EFSA) as safe for animal dietary use.
Considering the proven efficacy of dietary supplementation with the probiotic microorganisms Weizmannia faecalis DSM 32,016 and Bacillus licheniformis DSM 33,806 under non-challenged conditions [17], the aim of the present study was to evaluate the effects of this probiotic combination on broiler performance, carcass quality, and intestinal health under the challenges of increased stocking density, reused litter, and mild heat stress, with a particular focus on carcass optimization.
2. Materials and Methods
As-hatched ROSS 308 broilers (n = 320) were purchased from a local hatchery and were brought to the experimental facilities of the Research Institute of Animal Science in Paralimni Giannitson, which comprise solid floored pens (100 × 100 cm) featuring chopped wheat straw bedding. Upon arrival, the chicks were individually weighed and randomly allocated to two treatments, each consisting of 8 replicates of 20 birds each (160 birds per treatment). The experimental treatments consisted of the control group, which was fed a standard commercial diet throughout the experiment (C), and the probiotics group, where the standard diet was further supplemented with Weizmannia faecalis DSM 32,016 and Bacillus licheniformis DSM 33,806 (Technocare, Biochem, Lohne, Germany) at a rate of 200 g/t (Pr), according to product specifications. Feed and water were offered in bell feeders and nipple drinkers, respectively, for ad libitum consumption. The birds were provided with a mash diet across three phases: Starter (days 1–10), Grower (days 11–24), and Finisher (days 25–42). The composition and calculated analysis of experimental diets are presented in Table 1. To determine total tract nutrient digestibility (TTD), titanium dioxide was incorporated into the diets at a concentration of 5 g/kg to serve as an indigestible marker.

Table 1.
Composition and calculated chemical analysis of experimental diets.
The lighting regimen consisted of continuous illumination (24 h) for the first three days, followed by a 23L:1D cycle until the end of the third week, and 20L:4D for the remainder of the study. Ambient temperature gradually decreased from 32 °C on day 0 to 19 °C on day 42 (except from the hours when heat stress was applied). The chicks were vaccinated at the hatchery for infectious bronchitis and Newcastle disease, and a second application of the vaccines was performed at 14 days of age. No other supplement was given to the birds throughout the experiment.
To evaluate the efficacy of the probiotic dietary supplementation in challenged birds, three challenges were applied during the experiment: used litter from a previous experiment was used as bedding, elevated stocking density was applied (>39 kg/m2 from day 30 and onwards, above the upper limit of stocking density permitted by EU regulations), and mild heat stress (28 °C, for 3 h at midday, days 28–38, 25–35% above suggested room temperature according to the supplier’s management handbook [18]).
The duration of the experiment was 42 days. On day 42, 24 birds per group, sex-balanced (3 per pen), were slaughtered for the recording of carcass measurements. The birds were fastened 12 h before slaughter. They were sacrificed via decapitation and allowed to exsanguinate for approximately 1 min. Carcasses were then scalded (60 °C for 2 min), mechanically plucked (2 min), and manually eviscerated. Following processing, carcasses were chilled at 4 °C for 24 h.
Body weight (BW) and feed intake (FI) were recorded on a per-pen basis at the conclusion of each feeding phase (days 10, 24, and 42). Mortality was monitored daily throughout the experimental period. Flock uniformity was assessed by calculating the coefficient of variation (CV) of body weights within each pen. These primary data were used to determine average daily gain (ADG), feed conversion ratio (FCR), and the European Poultry Efficiency Factor (EPEF).
For the measurement of total tract digestibility, excreta were collected per pen at the end of each feeding phase (days 10, 24 and 42). Excreta samples were subsequently analyzed for dry matter (DM), crude protein (CP), fat content (FC) and fiber content (FiC) using the routine procedures [19]. The total tract digestibility of nutrients was calculated using the following equation:
where N and T are the nutrient and titanium concentrations, respectively, while the markers f or d represent the subsequent quantity in the feces and diet, respectively (% DM).
TTD = 1 − [(Td/Tf) × (Nf/Nd)] × 100
At slaughter, all carcasses were scored for footpad lesions, according to a five-point scale developed by Riber et al. [20]. After slaughter (24 h), the weights of cold carcass, thigh, drumstick, liver, abdominal fat and carcass yield (on the basis of cold carcass to live BW) were also recorded. The weight and length of the tibiotarsus bone were also recorded and the Seedor index (weight/length) calculated [21].
At the ages of 10, 24 and 42 days, blood samples were collected from 3 birds per pen (24 per group) in 15 mL falcon tubes without coagulant, after slaughter. The samples were centrifuged at 3200× g for 1 h, and the collected serum was stored at −80 °C until the determination antibodies titers and FABP2 concentration. Serum antibody titers for Infectious Bursal Disease (IBD), Infectious Bronchitis Virus (IBV), and Newcastle Disease Virus (NDV) were determined using commercial ELISA kits (CK113, CK119, and CK116, respectively; BioChek BV, Reeuwijk, The Netherlands) following the manufacturer’s instructions. Briefly, serum samples (1:500 dilution) were processed and read at 405 nm. Antibody status was defined by Sample-to-Positive (S/P) ratios, with positivity thresholds set at ≥0.35 for NDV and ≥0.20 for IBD and IBV. Antibody titers were calculated using the following kit-specific equations:
NDV: Log10 Titer = 1.0 × Log10(S/P) + 3.52
IBV: Log10 Titer = 1.0 × Log10(S/P) + 3.62
IBD: Log10 Titer = 1.1 × Log10(S/P) + 3.361 Serum FABP2 (fatty acid binding protein 2) concentrations were measured using a commercial sandwich ELISA kit specific to chicken FABP2 (SEA559Ga; Cloud-Clone Corp., Katy, TX, USA). The ELISA detection range was 0.78–50 ng/mL, and the LLD was <0.29 ng/mL, as reported by the manufacturer. Serum samples were thawed on ice, gently mixed, and assayed in duplicate without dilution. The ELISA procedure was performed exactly as described by the manufacturer: 100 μL of standards, blank, and samples were added to the pre-coated wells and incubated for 1 h at 37 °C, followed by incubation with Detection Reagent A (1 h at 37 °C), washing, incubation with Detection Reagent B (30 min at 37 °C), further washing, and color development with TMB substrate for 10–20 min. The reaction was stopped with 50 μL stop solution, and absorbance was measured at 450 nm.
Performance data (BW, FI, antibody titers, and FABP2) were analyzed via two-way ANOVA with group, age, and their interaction as fixed effects. Carcass traits, bone properties, and footpad scores were analyzed via one-way ANOVA with group as the fixed effect. The pen served as the experimental unit. Data was screened for outliers based on plausibility and verified for normality and homoscedasticity. Results are expressed as least square means ± pooled SE. Mean comparisons were performed using the Student–Newman–Keuls (SNK) test, with significance defined at p < 0.05. All analyses were conducted using STATGRAPHICS (v18, Windows).
3. Results
3.1. Growth Parameters and Total Tract Digestibility of Nutrients
Concerning the body weights, no significant differences were recorded for days 0, 10 and 24, while at the end of the experiment (D42), the final body weight of probiotic-supplemented broilers was higher than the control group (p < 0.05) (Table 2). The average daily gain was comparable between the treatments for the starting and middle period of growth, while for the third phase (D25–42), the probiotic group had the highest ADG (p < 0.05). Feed intake was not different among groups at any stage of growth (p > 0.05). On the other hand, FCR was lower at D42 in the probiotic group (p < 0.05), indicating that the probiotic supplementation significantly improved feed conversion. Regarding the uniformity of the flocks, the coefficient variation (CV) of the body weights per feeding period was not different between the experimental groups, and it ranged between 8 and 10% (Table 2).

Table 2.
Growth parameters and total tract digestibility of the experimental groups per feeding period.
In the starter period, the probiotic supplementation improved the digestibility of all three nutrients (p < 0.05) (Table 3). In the growing period, the differences persisted for fat and fiber content, while the digestibility of crude protein tended to be higher in the probiotics group (p < 0.10). In the finisher period, no statistical differences were recorded between the two groups (p > 0.05) (Table 3).

Table 3.
Nutrient total tract digestibility (%) of the experimental groups per feeding period.
3.2. Carcass Parameters, Bone Properties and EPEF
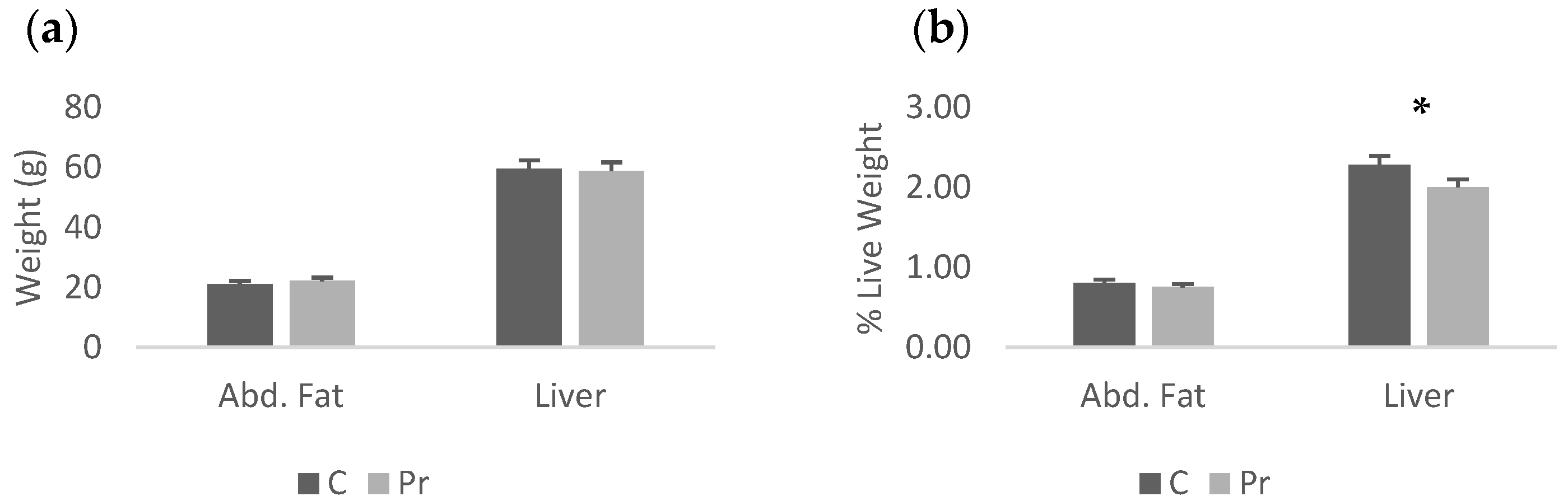
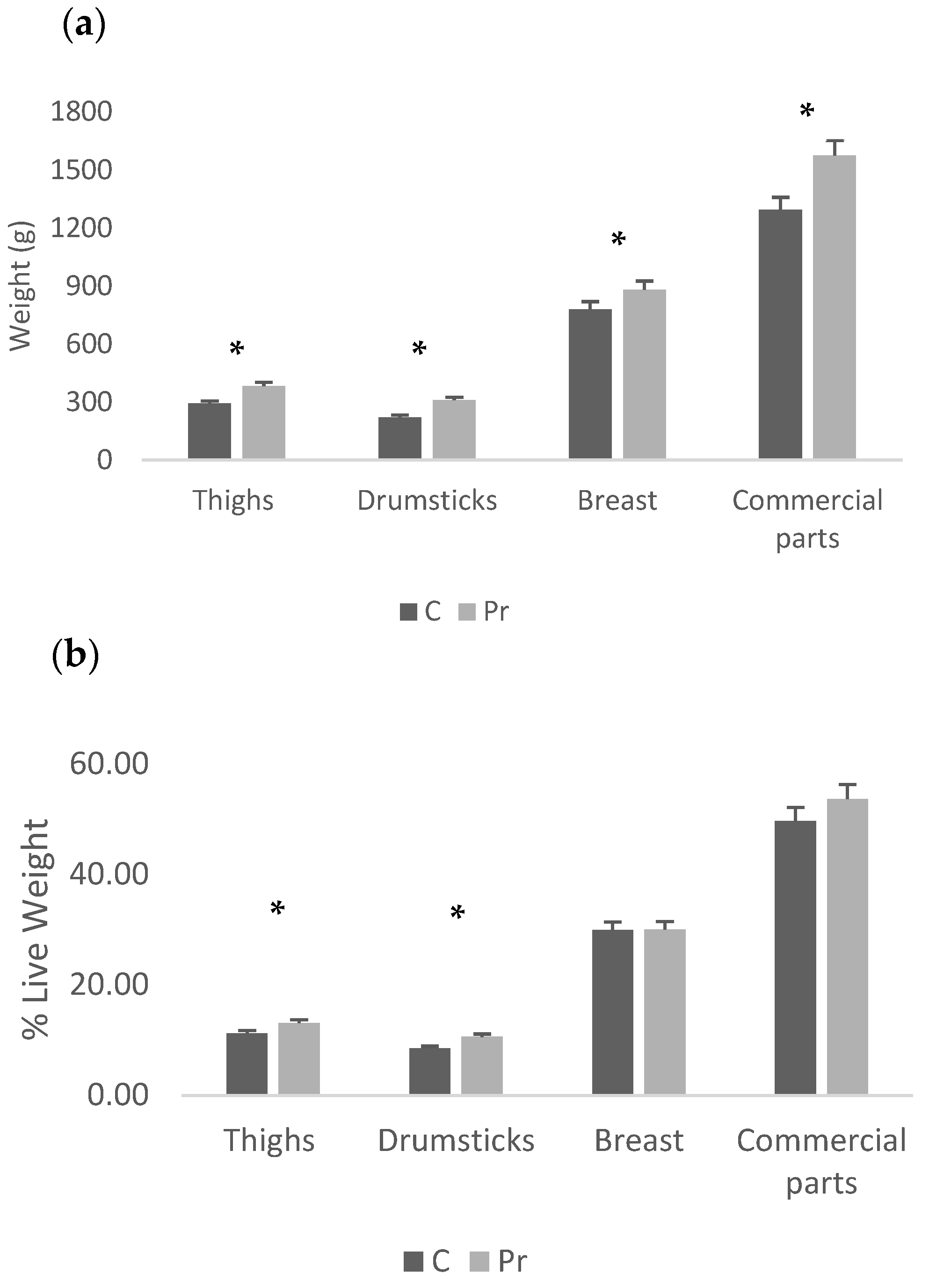
The probiotics group had higher live and cold carcass weight, while carcass yield was not different between groups (Table 4). In terms of absolute weights of the abdominal fat and liver, there was no statistically significant difference between the experimental groups, while in terms of proportions, the probiotics group had a significantly lighter liver than the control (Figure 1a,b). Moreover, regarding the absolute weights of the main carcass parts, the probiotics group had heavier thigh, drumstick, breast and the commercial parts (two feet plus the breast (p < 0.05) (Figure 2a)). In fact, the probiotic supplementation increased thigh weight by 31.1%, drumstick weight by 40.3%, breast weight by 13.1% and commercial parts weight by 21.8%. In proportion to the live weight, significant differences were recorded only for the thigh and drumstick (Figure 2b). Nevertheless, the relevant increases were 16.4% and 24.5% for the thigh and drumstick, respectively, as well as 0.3% for the breast and 8.1% for the commercial parts.

Table 4.
Carcass parameters, footpad scoring, litter moisture and bone properties of experimental groups.
Figure 1.
Abdominal fat and liver of the experimental groups presented as absolute weights (a) and proportions of live weight (b) (* = p < 0.05).
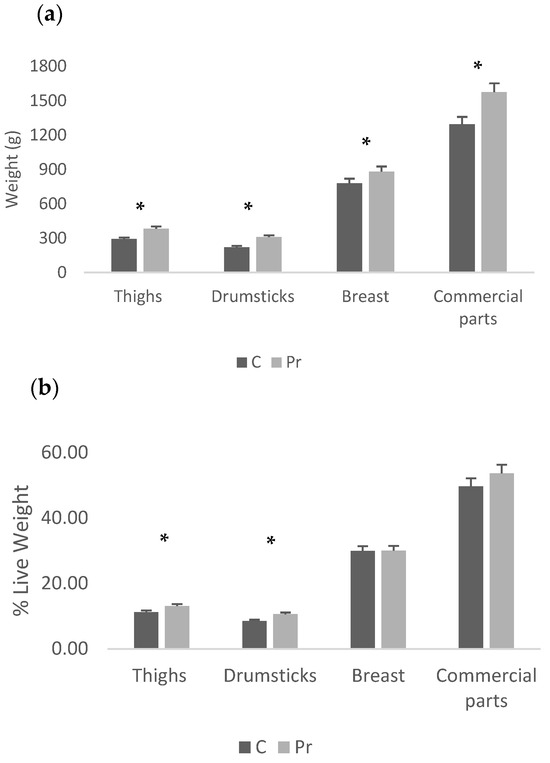
Figure 2.
Carcass parts of the experimental groups presented as absolute weights (a) and proportions of live weight (b) (* = p < 0.05).
Tibiotarsus weight and the Seedor index were not different among groups, but the probiotics group had a higher average length of that bone (p < 0.05) (Table 4). Footpad scoring, on the other hand, was comparable for both groups at the two ages examined. In terms of EPEF, the index that considers body weight gain, feed conversion and mortality (1.7 and 1.2 for the control and probiotics group, respectively), it was significantly higher in the probiotics group, indicating a positive effect of the probiotic supplementation on the overall performance of broilers (Table 4).
3.3. Antibodies Titer and FABP2 Concentrations
For the first two diseases, no differences were recorded between the experimental groups, at any age (Table 5). For IBD, while the antibody titer was comparable between the two groups at 10 and 24 days of age, the probiotics group exhibited a higher antibody titer than the control at 42 days of age (p < 0.05) (Table 5).

Table 5.
Antibodies titer for NDV, IBV and IBD at 10, 24 and 42 days of age.
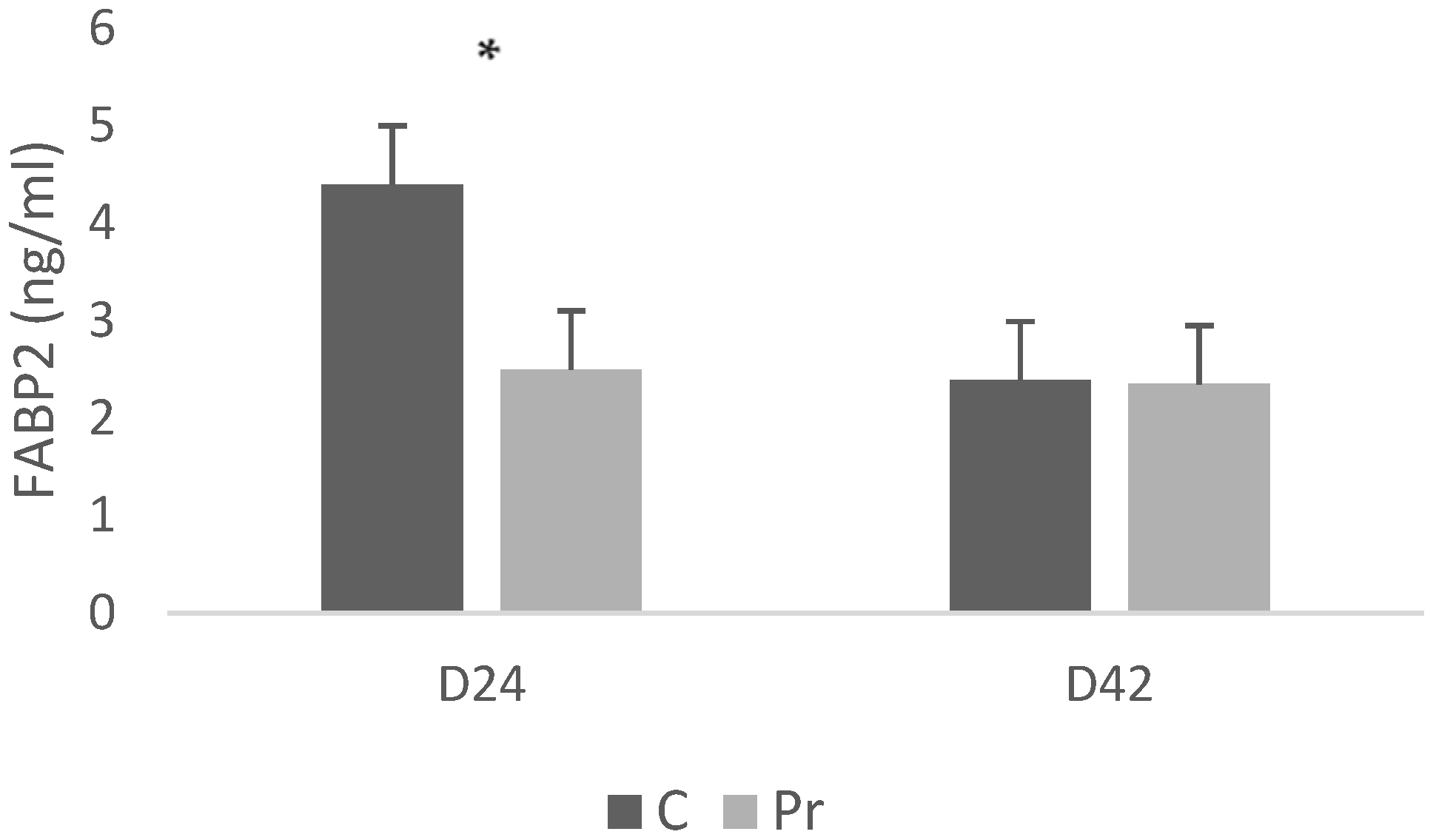
With respect to the FABP2 concentrations, the control group exhibited significantly higher serum FABP2 concentrations (4.4 ng/mL) compared with the birds that were receiving the probiotics (2.5 ng/mL) (Figure 3). Nevertheless, at 42 days, FABP2 concentrations were similar between treatments, with both groups averaging approximately 2.3 ng/mL.
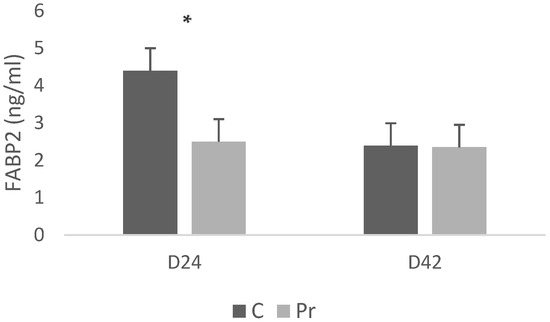
Figure 3.
FABP2 (fatty acid binding protein 2) serum concentrations of experimental groups at 24 and 42 days of age (* = p < 0.05).
4. Discussion
It is well documented that the addition of probiotics in broilers’ diets improves growth performance [22,23], mainly in terms of absolute body weight and weight gain [6,24], an effect that was also observed in this study. With respect to the probiotic microorganisms used in this study, other authors have observed the same positive effects either by using single cultures of Weizmannia faecalis (Bacillus coagulans) [13,25,26], single cultures of Bacillus licheniformis [27,28] or a combination of both probiotics [17]. In terms of feed intake, most researchers agree that it is not affected by the dietary supplementation of probiotics [15,25,29], and, therefore, the increased growth is a result of a better utilization of nutrients by the birds.
The previous assumption is verified by the digestibility results of this study, which have shown that the total tract digestibility of crude protein, fat and fiber are elevated in the birds receiving the combination of probiotics, for the first two feeding phases. Probiotic supplementation significantly improved protein, fat, and fiber digestibility during these early phases, when endogenous enzyme secretion, absorptive capacity, and microbial functionality are still immature and limit nutrient utilization [30]. Although not directly demonstrated in the present study, it can be hypothesized that the probiotic may support the development of a more efficient gastrointestinal tract, potentially enhancing nutrient utilization early in life. This assumption is supported by previous literature reporting that probiotics can promote intestinal morphological development, including increases in villus height and absorptive surface area [31,32]. By the finisher phase, digestive capacity is largely mature and operates close to its physiological maximum, leading to a ceiling effect.
In general, probiotics are widely reported to improve nutrient digestibility in broilers [33,34], mainly through an enhancement of digestive enzyme activities (e.g., trypsin, amylase, protease) [35,36], an improvement of gut morphology [35,37] and the modulation of gut microbiota towards the beneficial bacteria [6]. Specifically, the more pronounced effects have been observed in the improvement of digestibility of dry matter [38], crude protein and selected amino acids [34] and gross energy [39] and less for fat and starch [33,39]. Overall, irrespective of the proposed mechanisms, the combination of increased body weight and unaffected feed intake that was recorded in this study leads to a reduced feed conversion ratio [10,17], an increased EPEF [36,40] and collectively a better economic output of poultry production.
During this study, three challenges were applied to the birds to better evaluate the efficacy of the probiotic dietary supplementation: used litter, elevated stocking density and mild heat stress. The results proved that the challenges were successful, since the final weight of the control group and feed conversion were lower and higher, respectively, to the broiler objectives demonstrated by Aviagen for Ross 308 [18], while the probiotic supplementation helped alleviate these negative effects. Although the present study did not include an unchallenged baseline group, the reduced performance of the control birds compared with breeder objectives indicates that the flock was exposed to moderate production stress. Future studies including both challenged and unchallenged controls would allow a more precise quantification of the challenge severity and the magnitude of probiotic-mediated mitigation. Nunes et al. [41] also found that a combination of probiotic microorganisms (Lactobacillus acidophilus, Enterococcus faecium and Bifidubacterium bifidum) improved the performance of broilers raised on re-used litter. Moreover, it has been reported that dietary supplementation with probiotics can help broilers cope with the adverse effects of high stocking density [42,43,44] or heat stress [45,46,47].
Carcass composition is largely dependent on growth performance and live weight. In this study, the increase in body weight recorded after the dietary supplementation with probiotics had apparent effects on carcass composition, increasing the weight of all the commercial parts. Obviously, if body weight was not affected, the same would apply for the major carcass parts, as it has already been reported elsewhere [48,49]. In terms of the parts’ yield in relation to live weight, there are quite diverse results in the literature. Khan et al. [50] found that the addition of probiotics increased the breast and drumstick yield, Kaushal et al. [51] reposted an increase for breast only with no differences for thigh and drumstick, while Pelicano et al. [52] reported a significant difference only for the thigh. Finally, lower liver yield was also reported by Giang et al. [53]. These diverse results are justified due to the variation in diets, probiotics and management practices used in the related papers.
Probiotics, through the modification of gut microbiota and several physiological pathways, have the ability to influence bone formation and properties in broilers and other animal species [54]. The combination of probiotics used in this study increased tibiotarsus length but did not affect either the weight of the bone or the weight/length index. Javid et al. [55] also reported that a probiotic supplementation increased tibia length, but in their study, bone weight and weight/length index were also higher. On the contrary, Mutus et al. [56] did not find any effect of a probiotic supplementation with Bacillus licheniformis and Bacillus subtilis on bone properties. Other studies have reported more prominent effects, e.g., an increase in mineral content [57] as well as an increase in the content of Ca and P in the bone [58].
Footpad dermatitis causes necrotic lesions on the footpads of broilers and its prevalence if affected by a lot of factors, with litter moisture being reported as the most prominent factor [59]. Some researchers suggested that the addition of probiotics decreases the prevalence of the disease in broilers [60,61], while others have reported that such an effect is not significant [62]. In this study, there was no difference in footpad scoring for dermatitis between the two groups, while litter moisture was significantly lower at 24 days of age and comparable at 42 days. It must be also noted that, in general, the recorded scores were relatively low, indicating good management practices for the flock.
Probiotics are generally reported to increase antibody titers in poultry [63,64], although this is not always consistent [49,65]. The positive effects likely reflect a complex modulation of the immune system, rather than a simple enhancement of antibody production. Consequently, antibody titers do not always directly indicate the efficiency of the immune response. In this study, the lower antibody titer observed in probiotic-fed broilers 10 days after the second vaccination may reflect an early modulation of immunity, where probiotics support immune cell maturation and regulation in response to vaccination, rather than immediately boosting circulating antibodies as seen in the control animals. Over time, improved gut health, nutrient absorption, and the development of immunological memory likely contributed to a stronger and more sustained humoral response, resulting in higher antibody titers against NDV and IBV by day 42. Interestingly, in the case of IBD, where broilers were vaccinated only at the hatchery, the final antibody titer at day 42 was significantly increased in probiotic-fed birds, a result also reported by Rehman et al. [49]. This stronger effect is likely because the unvaccinated control birds had a relatively weak and inefficient baseline immune response, making the stimulatory effect of probiotics on antibody production more pronounced. Given that growth performance and feed conversion were significantly improved in probiotic-fed broilers and no signs of infection were observed despite challenges, these results suggest that the probiotic combination may promote a more efficient and durable immune response, favoring long-term protection rather than an early acute rise in antibodies.
The probiotic supplementation resulted in lower FABP2 concentrations at 24 days of age in relation to the control group. FABP2 (I-FABP) is a cytosolic protein released during enterocyte damage, widely validated as a circulating biomarker of intestinal epithelial injury and increased permeability in humans [66,67]. Although serum FABP2 has not yet been extensively characterized in poultry, its biological function is conserved, and FABP2 has been well studied in chicken intestinal physiology and gene expression [68,69]. Thus, lower FABP2 in probiotic-fed birds at 24 days is consistent with reduced epithelial stress at early stages of growth. FABP2 values in probiotic-fed birds at 42 days were comparable to those of the control group, indicating that late-phase elevations remained within the physiological range observed in non-supplemented birds. A plausible interpretation is that the probiotic combination exerts its influence primarily during the early developmental phase, when the intestine is more susceptible to epithelial disruption, but this effect does not extend into the finisher period. This notion agrees with previous work showing that probiotic effects on gut morphology and permeability tend to be strongest during early growth and can diminish as the gastrointestinal tract matures [70,71]. Although circulating FABP2 is not yet widely used in poultry research, its mechanistic relevance and established diagnostic value in mammals support its potential as an emerging biomarker for assessing enterocyte integrity in broilers. Combining serum FABP2 with established permeability markers (such as DAO, D-lactate, and endotoxin) and histological analysis would strengthen future evaluations of probiotic effects on gut barrier function.
5. Conclusions
The beneficial effects of probiotics are well documented in the relevant literature. In this study, the dietary supplementation of broilers with a probiotic combination based on Weizmannia faecalis (formerly Bacillus coagulans) and Bacillus licheniformis, under challenging rearing conditions, enhanced growth performance and increased the total tract digestibility of protein, fat and fiber, without affecting feed intake. In terms of carcass composition, the weight of the commercial parts was increased while carcass yield was not affected by the probiotic dietary supplementation. Finally, the antibody production against IBD at 42 days of age was increased and the FABP2 concentrations at 24 days of age decreased in the supplemented group in relation to the control group. Further research would be useful in terms of clarifying the appropriate dose as well as shedding light on the specific changes of the intestinal microbiota of broilers. Nevertheless, as the probiotic delivery systems continue to innovate parallel to the adoption of precision nutrition approaches, it is likely that the effectiveness and adoption of probiotics by the poultry industry will increase in the future.
Author Contributions
Conceptualization, G.K.S., L.Z. and A.S.; methodology, G.K.S., I.G., P.S., D.K. (Despoina Karatosidi) and V.D.; validation, I.G., P.S., V.D., D.V., N.L., D.K. (Dimitrios Koutsianos) and D.K. (Despoina Karatosidi); formal analysis, G.K.S.; investigation, D.V., N.L., D.K. (Dimitrios Koutsianos), G.T., M.G. and E.D.; writing—original draft preparation, G.K.S., L.Z., A.S., G.T., M.G. and E.D.; writing—review and editing, G.K.S., L.Z., A.S., I.G., P.S., D.K. (Despoina Karatosidi), V.D., G.T., M.G., E.D., D.V., N.L. and D.K. (Dimitrios Koutsianos); project administration, G.K.S.; All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Biochem Zusatzstoffe Handels- und Produktionsgesellschaft mbH, Lohne, Germany.
Institutional Review Board Statement
The study was conducted in accordance with the guidelines of “Council Directive 86/609/EEC regarding the protection of animals used for experimental and other scientific purposes” and approved by the Ethics Committee of ELGO-DIMITRA (protocol code 48966-26/09/2024).
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
Authors Lydia Zeibich, Alexandra Schlagheck, Dimitrios Verros and Nikolaos Lykos were employed by the company Biochem Zusatzstoffe Handels- und Produktionsgesellschaft mbH, and Dimitris Koutsianos was employed by the company Vet analysis. All authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The funders had no role in the design of the study or in the collection, analyses, or interpretation of data. The publication of the results has been approved by the funders.
References
- Ajuwon, K.M. Toward a Better Understanding of Mechanisms of Probiotics and Prebiotics Action in Poultry Species. J. Appl. Poult. Res. 2016, 25, 277–283. [Google Scholar] [CrossRef]
- Ban on Antibiotics as Growth Promoters in Animal Feed Enters into Effect. Available online: https://ec.europa.eu/commission/presscorner/detail/en/ip_05_1687 (accessed on 2 March 2026).
- FAO/WHO. Probiotics in Food Health and Nutritional Properties and Guidelines for Evaluation. Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria, Cordoba, Argentina, 1–4 October 2001 [and] Report of a Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food, London, Ontario, Canada, 30 April–1 May 2002; FAO Food and Nutrition Paper 85; Food and Agriculture Organization of the United Nations, World Health Organization: Rome, Italy, 2006. Available online: https://www.scirp.org/reference/referencespapers?referenceid=2833926 (accessed on 8 September 2025).
- Yaqoob, M.U.; Wang, G.; Wang, M. An Updated Review on Probiotics as an Alternative of Antibiotics in Poultry—A Review. Anim. Biosci. 2022, 35, 1109–1120. [Google Scholar] [CrossRef] [PubMed]
- Mountzouris, K.C.; Balaskas, C.; Xanthakos, I.; Tzivinikou, A.; Fegeros, K. Effects of a Multi-Species Probiotic on Biomarkers of Competitive Exclusion Efficacy in Broilers Challenged with Salmonella Enteritidis. Br. Poult. Sci. 2009, 50, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Kim, I.H. Effects of Multistrain Probiotics on Growth Performance, Apparent Ileal Nutrient Digestibility, Blood Characteristics, Cecal Microbial Shedding, and Excreta Odor Contents in Broilers. Poult. Sci. 2014, 93, 364–370. [Google Scholar] [CrossRef]
- Pagnini, C.; Saeed, R.; Bamias, G.; Arseneau, K.O.; Pizarro, T.T.; Cominelli, F. Probiotics Promote Gut Health through Stimulation of Epithelial Innate Immunity. Proc. Natl. Acad. Sci. USA 2010, 107, 454–459. [Google Scholar] [CrossRef]
- Payot, T.; Chemaly, Z.; Fick, M. Lactic Acid Production by Bacillus coagulans—Kinetic Studies and Optimization of Culture Medium for Batch and Continuous Fermentations. Enzym. Microb. Technol. 1999, 24, 191–199. [Google Scholar] [CrossRef]
- De Clerck, E.; Rodriguez-Diaz, M.; Forsyth, G.; Lebbe, L.; Logan, N.A.; DeVos, P. Polyphasic Characterization of Bacillus coagulans Strains, Illustrating Heterogeneity within This Species, and Emended Description of the Species. Syst. Appl. Microbiol. 2004, 27, 50–60. [Google Scholar] [CrossRef]
- Zhou, Y.; Zeng, Z.; Xu, Y.; Ying, J.; Wang, B.; Majeed, M.; Majeed, S.; Pande, A.; Li, W. Application of Bacillus coagulans in Animal Husbandry and Its Underlying Mechanisms. Animals 2020, 10, 454. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Lee, J.; Heo, S.; Lee, J.-H.; Jeong, D.-W. Technology and Safety Evaluation of Bacillus coagulans Exhibiting Antimicrobial Activity for Starter Development. LWT 2021, 137, 110464. [Google Scholar] [CrossRef]
- Cao, J.; Yu, Z.; Liu, W.; Zhao, J.; Zhang, H.; Zhai, Q.; Chen, W. Probiotic Characteristics of Bacillus coagulans and Associated Implications for Human Health and Diseases. J. Funct. Foods 2020, 64, 103643. [Google Scholar] [CrossRef]
- Zhang, B.; Zhang, H.; Yu, Y.; Zhang, R.; Wu, Y.; Yue, M.; Yang, C. Effects of Bacillus coagulans on Growth Performance, Antioxidant Capacity, Immunity Function, and Gut Health in Broilers. Poult. Sci. 2021, 100, 101168. [Google Scholar] [CrossRef]
- Xu, Y.; Yu, Y.; Shen, Y.; Li, Q.; Lan, J.; Wu, Y.; Zhang, R.; Cao, G.; Yang, C. Effects of Bacillus subtilis and Bacillus licheniformis on Growth Performance, Immunity, Short Chain Fatty Acid Production, Antioxidant Capacity, and Cecal Microflora in Broilers. Poult. Sci. 2021, 100, 101358. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Yu, Y.-H. Bacillus licheniformis–Fermented Products Improve Growth Performance and the Fecal Microbiota Community in Broilers. Poult. Sci. 2020, 99, 1432–1443. [Google Scholar] [CrossRef]
- Xu, S.; Lin, Y.; Zeng, D.; Zhou, M.; Zeng, Y.; Wang, H.; Zhou, Y.; Zhu, H.; Pan, K.; Jing, B.; et al. Bacillus licheniformis Normalize the Ileum Microbiota of Chickens Infected with Necrotic Enteritis. Sci. Rep. 2018, 8, 1744. [Google Scholar] [CrossRef]
- Elleithy, E.M.M.; Bawish, B.M.; Kamel, S.; Ismael, E.; Bashir, D.W.; Hamza, D.; Fahmy, K.N.E.-D. Influence of Dietary Bacillus coagulans and/or Bacillus licheniformis-Based Probiotics on Performance, Gut Health, Gene Expression, and Litter Quality of Broiler Chickens. Trop. Anim. Health Prod. 2023, 55, 38. [Google Scholar] [CrossRef]
- Ross. Available online: https://aviagen.com/eu/brands/ross/ (accessed on 2 March 2026).
- AOAC. Official Methods of Analysis, 14th ed.; Association of Analytical Chemists: Washington, DC, USA, 1984; Available online: https://www.scirp.org/reference/referencespapers?referenceid=1882886 (accessed on 19 January 2026).
- Riber, A.B.; Rangstrup-Christensen, L.; Hansen, M.S.; Hinrichsen, L.K.; Herskin, M.S. Characterisation of Footpad Lesions in Organic and Conventional Broilers. Animal 2020, 14, 119–128. [Google Scholar] [CrossRef]
- Seedor, J.G.; Quartuccio, H.A.; Thompson, D.D. The Bisphosphonate Alendronate (MK-217) Inhibits Bone Loss Due to Ovariectomy in Rats. J. Bone Min. Res. 1991, 6, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Ramlucken, U.; Ramchuran, S.O.; Moonsamy, G.; Lalloo, R.; Thantsha, M.S.; Jansen van Rensburg, C. A Novel Bacillus Based Multi-Strain Probiotic Improves Growth Performance and Intestinal Properties of Clostridium perfringens Challenged Broilers. Poult. Sci. 2020, 99, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Wang, R.; Liu, J.; Wang, W.; Chen, Y.; Cai, W. Effects of Novel Microecologics Combined with Traditional Chinese Medicine and Probiotics on Growth Performance and Health of Broilers. Poult. Sci. 2022, 101, 101412. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, R.; Jia, H.; Zhu, Z.; Li, H.; Ma, Y. Supplementation of Probiotics in Water Beneficial Growth Performance, Carcass Traits, Immune Function, and Antioxidant Capacity in Broiler Chickens. Open Life Sci. 2021, 16, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Khajeh Bami, M.; Afsharmanesh, M.; Ebrahimnejad, H. Effect of Dietary Bacillus coagulans and Different Forms of Zinc on Performance, Intestinal Microbiota, Carcass and Meat Quality of Broiler Chickens. Probiotics Antimicrob. Proteins 2020, 12, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Zhen, W.; Shao, Y.; Gong, X.; Wu, Y.; Geng, Y.; Wang, Z.; Guo, Y. Effect of Dietary Bacillus coagulans Supplementation on Growth Performance and Immune Responses of Broiler Chickens Challenged by Salmonella enteritidis. Poult. Sci. 2018, 97, 2654–2666. [Google Scholar] [CrossRef]
- Dumitru, M.; Lefter, N.A.; Ciurescu, G.; Draghici, R. Effect of Bacillus licheniformis on Growth, Bone Mineralization, and Intestinal Microbiota in Broilers Fed Cowpea Diets. Agriculture 2024, 14, 2013. [Google Scholar] [CrossRef]
- Qin, S.; Xiao, X.; Dai, Z.; Zhao, G.; Cui, Z.; Wu, Y.; Yang, C. Effects of Bacillus licheniformis on Growth Performance, Immune and Antioxidant Functions, and Intestinal Microbiota of Broilers. Poult. Sci. 2024, 103, 103210. [Google Scholar] [CrossRef]
- Tayeri, V.; Seidavi, A.; Asadpour, L.; Phillips, C.J.C. A Comparison of the Effects of Antibiotics, Probiotics, Synbiotics and Prebiotics on the Performance and Carcass Characteristics of Broilers. Vet. Res. Commun. 2018, 42, 195–207. [Google Scholar] [CrossRef]
- Ravindran, V.; Abdollahi, M.R. Nutrition and Digestive Physiology of the Broiler Chick: State of the Art and Outlook. Animals 2021, 11, 2795. [Google Scholar] [CrossRef]
- Awad, W.A.; Ghareeb, K.; Abdel-Raheem, S.; Böhm, J. Effects of Dietary Inclusion of Probiotic and Synbiotic on Growth Performance, Organ Weights, and Intestinal Histomorphology of Broiler Chickens. Poult. Sci. 2009, 88, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Bogucka, J.; Ribeiro, D.M.; Bogusławska-Tryk, M.; Dankowiakowska, A.; da Costa, R.P.R.; Bednarczyk, M. Microstructure of the Small Intestine in Broiler Chickens Fed a Diet with Probiotic or Synbiotic Supplementation. J. Anim. Physiol. Anim. Nutr. (Berl.) 2019, 103, 1785–1791. [Google Scholar] [CrossRef]
- Singh, A.K.; Tiwari, U.P.; Berrocoso, J.D.; Dersjant-Li, Y.; Awati, A.; Jha, R. Effects of a Combination of Xylanase, Amylase and Protease, and Probiotics on Major Nutrients Including Amino Acids and Non-Starch Polysaccharides Utilization in Broilers Fed Different Level of Fibers. Poult. Sci. 2019, 98, 5571–5581. [Google Scholar] [CrossRef] [PubMed]
- Li, T.X.; Eom, J.; Kim, H.K.; Beak, S.H.; Jo, A.R.; Kim, I.H. Dual Effects of Dietary Protein Restriction and Probiotic Supplementation on Broiler Performance and Fecal Gas Emission. Poult. Sci. 2025, 104, 105416. [Google Scholar] [CrossRef] [PubMed]
- Goodarzi Boroojeni, F.; Vahjen, W.; Männer, K.; Blanch, A.; Sandvang, D.; Zentek, J. Bacillus subtilis in Broiler Diets with Different Levels of Energy and Protein. Poult. Sci. 2018, 97, 3967–3976. [Google Scholar] [CrossRef]
- Bromfield, J.I.; Niknafs, S.; Chen, X.; von Hellens, J.; Horyanto, D.; Sun, B.; Yu, L.; Tran, V.H.; Navarro, M.; Roura, E. The Evaluation of Next-Generation Probiotics on Broiler Growth Performance, Gut Morphology, Gut Microbiome, Nutrient Digestibility, in Addition to Enzyme Production of Bacillus Spp. in Vitro. Anim. Nutr. 2024, 18, 133–144. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.-M.E.; Ali, S.A.M.; Sallam, M.G.; Elbaz, A.M.; Mesalam, N.M.; Mohamed, Z.S.; Abdelhady, A.Y.; Yang, B.; Elsadek, M.F. Effects of Cold-Pressed Wheat Germ Oil and Bacillus subtilis on Growth Performance, Digestibility, Immune Status, Intestinal Microbial Enumeration, and Gene Expression of Broilers under Heat Stress. Poult. Sci. 2025, 104, 104708. [Google Scholar] [CrossRef]
- Song, D.; Li, A.; Wang, Y.; Song, G.; Cheng, J.; Wang, L.; Liu, K.; Min, Y.; Wang, W. Effects of Synbiotic on Growth, Digestibility, Immune and Antioxidant Performance in Broilers. Animal 2022, 16, 100497. [Google Scholar] [CrossRef]
- Abdelqader, A.; Mahasneh, Z.M.H.; Van Hoeck, V.; Abuajamieh, M.; Abedal-Majed, M.; Al-Qaisi, M.; Irshaid, R.; Al-Khaza’leh, J. Exogenous Enzymes Unlock Prebiotic Potential of Ulva lactuca: Boosting Gut Health and Growth Performance in Broilers. Poult. Sci. 2025, 104, 105834. [Google Scholar] [CrossRef] [PubMed]
- Golshahi, A.; Shams Shargh, M.; Dastar, B.; Rahmatnejad, E. The Effect of Thymus Vulgaris Extract and Probiotic on Growth Performance, Blood Parameters, Intestinal Morphology, and Litter Quality of Broiler Chickens Fed Low-Protein Diets. Poult. Sci. 2024, 104, 104554. [Google Scholar] [CrossRef]
- Nunes, R.V.; Scherer, C.; Pozza, P.C.; Eyng, C.; Bruno, L.D.G.; Vieites, F.M. Use of Probiotics to Replace Antibiotics for Broilers. Rev. Bras. Zootec. 2012, 41, 2219–2224. [Google Scholar] [CrossRef]
- Rezapoor, R.; Hosseinian, S.A.; Shekarforoush, S.S. Impact of Bacillus and Lactobacillus Probiotics on Growth Performance, Stress Indices, and Ileal Histomorphology in Broilers under High Stocking Density. Vet. México OA 2025, 12. [Google Scholar] [CrossRef]
- Cengiz, Ö.; Köksal, B.H.; Tatlı, O.; Sevim, Ö.; Ahsan, U.; Üner, A.G.; Ulutaş, P.A.; Beyaz, D.; Büyükyörük, S.; Yakan, A.; et al. Effect of Dietary Probiotic and High Stocking Density on the Performance, Carcass Yield, Gut Microflora, and Stress Indicators of Broilers. Poult. Sci. 2015, 94, 2395–2403. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, R.M.; El-Rayes, T.K. Effect of Stocking Density and Probiotic Supplementation on Broiler Performance. J. Anim. Poult. Prod. 2016, 7, 491–497. [Google Scholar] [CrossRef]
- Sumanu, V.O.; Naidoo, V.; Oosthuizen, M.C.; Chamunorwa, J.P. Evaluating the Efficacy of Probiotics and Ascorbic Acid as Anti-Stress Agents against Heat Stress in Broiler Chickens. Front. Vet. Sci. 2024, 11, 1482134. [Google Scholar] [CrossRef]
- Hernández-Coronado, A.C.; Cervantes, M.; González, F.; Valle, A.; Arce, N.; Vásquez, N.; Bernal, H.; Morales, A. Effect of Probiotic Supplementation on Productive Performance and Epithelial Intestinal Integrity of Broiler Chickens Exposed to Heat Stress. Trop. Anim. Health Prod. 2025, 57, 235. [Google Scholar] [CrossRef] [PubMed]
- Aydin, S.S.; Hatipoglu, D. Probiotic Strategies for Mitigating Heat Stress Effects on Broiler Chicken Performance. Int. J. Biometeorol. 2024, 68, 2153–2171. [Google Scholar] [CrossRef] [PubMed]
- Junaid, N.; Biswas, A.; Kumawat, M.; Mandal, A.B. Production Performance, Immune Response and Carcass Traits of Broiler Chickens Fed Diet Incorporated with Probiotics. Indian J. Anim. Res. 2018, 52, 1597–1602. [Google Scholar] [CrossRef]
- Rehman, A.; Arif, M.; Sajjad, N.; Al-Ghadi, M.Q.; Alagawany, M.; Abd El-Hack, M.E.; Alhimaidi, A.R.; Elnesr, S.S.; Almutairi, B.O.; Amran, R.A.; et al. Dietary Effect of Probiotics and Prebiotics on Broiler Performance, Carcass, and Immunity. Poult. Sci. 2020, 99, 6946–6953. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.; Rahman, S.M.; Khan, S.; Roy, S.; Hossain, K.M. Effects of Probiotics on Productive Performances and Serum Lipid Profile of Broiler as Substitute of Antibiotics. Sci. Prog. 2024, 107, 00368504241276259. [Google Scholar] [CrossRef]
- Kaushal, S.; Sharma, R.K.; Singh, D.V.; Shukla, S.K.; Kumar, S.; Palod, J.; Singh, M.K. Performance, Carcass Characteristics and Economics of Broiler Chickens Fed Dietary Enzymes and Probiotic. Iran. J. Vet. Res. 2019, 20, 293–298. [Google Scholar]
- Pelicano, E.R.L.; Souza, P.d.; Souza, H.d.; Oba, A.; Norkus, E.A.; Kodawara, L.M.; Lima, T.d. Effect of Different Probiotics on Broiler Carcass and Meat Quality. Braz. J. Poult. Sci. 2003, 5, 207–214. [Google Scholar] [CrossRef]
- Growth Performance and Carcass Characteristics of Tam Hoang Chickens Supplemented with Probiotic Lactobacillus sp. Available online: https://www.lrrd.org/lrrd32/8/ntgiang32134.html (accessed on 16 December 2025).
- Chen, P.; Xu, T.; Zhang, C.; Tong, X.; Shaukat, A.; He, Y.; Liu, K.; Huang, S. Effects of Probiotics and Gut Microbiota on Bone Metabolism in Chickens: A Review. Metabolites 2022, 12, 1000. [Google Scholar] [CrossRef]
- Javid, M.A.; Masood, S.; Zaneb, H.; Rehman, H.U.; Nazir, M.M.; Waqas, M.Y.; Asif, M.; Basit, M.A.; Abbas, G.; Sufyan, A.B. Effect of Dietary Growth Promoter Supplementations on the Serum Biochemistry and Morphometric Characteristics of Tibia Bone in Broiler Chicken. Braz. J. Poult. Sci. 2022, 24, eRBCA. [Google Scholar] [CrossRef]
- Mutuş, R.; Kocabağli, N.; Alp, M.; Acar, N.; Eren, M.; Gezen, Ş.Ş. The Effect of Dietary Probiotic Supplementation on Tibial Bone Characteristics and Strength in Broilers. Poult. Sci. 2006, 85, 1621–1625. [Google Scholar] [CrossRef]
- Yan, F.-F.; Wang, W.-C.; Cheng, H.-W. Bacillus subtilis-Based Probiotic Promotes Bone Growth by Inhibition of Inflammation in Broilers Subjected to Cyclic Heating Episodes. Poult. Sci. 2020, 99, 5252–5260. [Google Scholar] [CrossRef]
- Liu, F.; Kong, A.; Fu, P.; Cao, Q.-Q.; Tao, K.-S.; Liu, D.-Y.; Wang, X.-B.; Tong, Z.-X.; Rehman, M.U.; Huang, S.-C. Lactobacillus Rhamnosus JYLR-005 Prevents Thiram-Induced Tibial Dyschondroplasia by Enhancing Bone-Related Growth Performance in Chickens. Probiotics Antimicrob. Proteins 2021, 13, 19–31. [Google Scholar] [CrossRef]
- Shepherd, E.M.; Fairchild, B.D. Footpad Dermatitis in Poultry. Poult. Sci. 2010, 89, 2043–2051. [Google Scholar] [CrossRef]
- de Oliveira, M.J.K.; Sakomura, N.K.; de Paula Dorigam, J.C.; Doranalli, K.; Soares, L.; Viana, G.d.S. Bacillus Amyloliquefaciens CECT 5940 Alone or in Combination with Antibiotic Growth Promoters Improves Performance in Broilers under Enteric Pathogen Challenge. Poult. Sci. 2019, 98, 4391–4400. [Google Scholar] [CrossRef]
- Park, C.J.; Sun, S.S. Effect of Dietary Metallo-Protease and Bacillus Velezensis CE 100 Supplementations on Growth Performance, Footpad Dermatitis and Manure Odor in Broiler Chickens. Anim. Biosci. 2022, 35, 1628–1634. [Google Scholar] [CrossRef]
- Flores, C.; Williams, M.; Pieniazek, J.; Dersjant-Li, Y.; Awati, A.; Lee, J.T. Direct-Fed Microbial and Its Combination with Xylanase, Amylase, and Protease Enzymes in Comparison with AGPs on Broiler Growth Performance and Foot-Pad Lesion Development. J. Appl. Poult. Res. 2016, 25, 328–337. [Google Scholar] [CrossRef]
- Kőrösi Molnár, A.; Podmaniczky, B.; Kürti, P.; Glávits, R.; Virág, G.; Szabó, Z.; Farkas, Z. Effect of Different Concentrations of Bacillus subtilis on Immune Response of Broiler Chickens. Probiotics Antimicrob. Proteins 2011, 3, 8–14. [Google Scholar] [CrossRef]
- Khaksefidi, A.; Ghoorchi, T. Effect of Probiotic on Performance and Immunocompetence in Broiler Chicks. J. Poult. Sci. 2006, 43, 296–300. [Google Scholar] [CrossRef]
- Nguyen, T.L.V.; Ho, T.K.H.; Nguyen, V.N.; Le, H.N.; Tran, H.T.; Duong, C.M. Effects of Dietary Supplementation with Antibiotic, Organic Acid, Probiotic and Prebiotic on the Intestinal Morphology and Newcastle Disease Virus Titers of Broilers in Commercial Farms. J. Agric. Dev. 2022, 21, 32–39. [Google Scholar] [CrossRef]
- Kocsis, D.; Papp, M.; Tornai, T.; Tulassay, Z.; Herszényi, L.; Tóth, M.; Juhász, M. Intestinal fatty acid binding protein: Marker of enterocyte damage in acute and chronic gastroenterological diseases. Orv. Hetil. 2016, 157, 59–64. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Voth, M.; Duchene, M.; Auner, B.; Lustenberger, T.; Relja, B.; Marzi, I. I-FABP Is a Novel Marker for the Detection of Intestinal Injury in Severely Injured Trauma Patients. World J. Surg. 2017, 41, 3120–3127. [Google Scholar] [CrossRef] [PubMed]
- Banaszak, L.; Winter, N.; Xu, Z.; Bernlohr, D.A.; Cowan, S.; Jones, T.A. Lipid-Binding Proteins: A Family of Fatty Acid and Retinoid Transport Proteins. Adv. Protein Chem. 1994, 45, 89–151. [Google Scholar] [CrossRef] [PubMed]
- Cahyaningsih, U.; Satyaningtijas, A.S.; Tarigan, R.; Nugraha, A.B. Chicken I-FABP as Biomarker of Chicken Intestinal Lesion Caused by Coccidiosis. IOP Conf. Ser. Earth Environ. Sci. 2018, 196, 012032. [Google Scholar] [CrossRef]
- Giannenas, I.; Papadopoulos, E.; Tsalie, E.; Triantafillou, E.; Henikl, S.; Teichmann, K.; Tontis, D. Assessment of Dietary Supplementation with Probiotics on Performance, Intestinal Morphology and Microflora of Chickens Infected with Eimeria tenella. Vet. Parasitol. 2012, 188, 31–40. [Google Scholar] [CrossRef]
- Mountzouris, K.C.; Tsitrsikos, P.; Palamidi, I.; Arvaniti, A.; Mohnl, M.; Schatzmayr, G.; Fegeros, K. Effects of Probiotic Inclusion Levels in Broiler Nutrition on Growth Performance, Nutrient Digestibility, Plasma Immunoglobulins, and Cecal Microflora Composition1. Poult. Sci. 2010, 89, 58–67. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.