LPS-Stressed Bovine Endometrial Cells upon Morulae in a Transwell Model of Embryo—Maternal Talk
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Uteri Collection and Endometrial Cell Isolation
2.2. Endometrial Epithelial Cell (EEC) Separation
2.3. Experiment 1: EEC Culture in Standard Plates
- (a)
- EEC Morphology
- (b)
- EEC viability by MTT test
2.4. Experiment 2: EEC Culture on Transwell System
- (a)
- TEER Test
- (b)
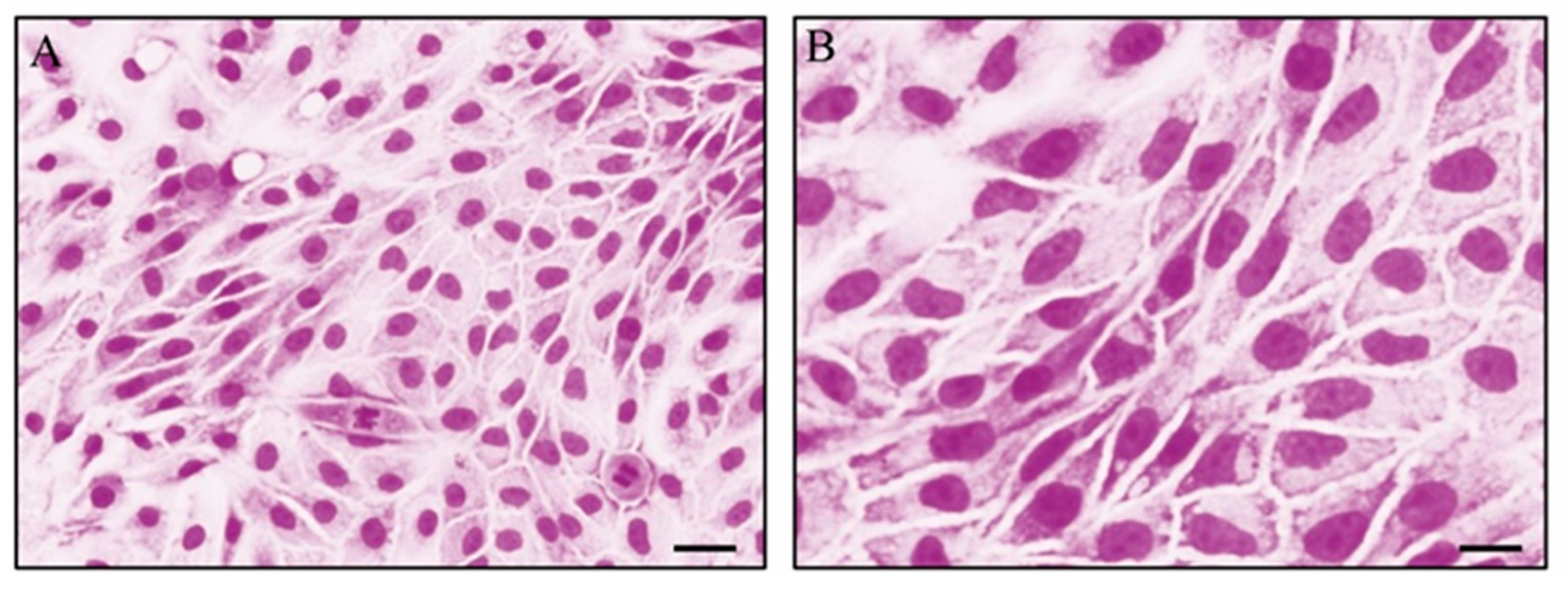
- Histological analysis of EECs on a transwell insert
2.5. In Vitro Embryo Production
2.6. Effect of LPS on Embryo Development
2.7. Embryo Rate Evaluation
2.8. Evaluation of Galectin-9 and LIF by ELISA
2.9. Isolation of EVs and Characterization by Nanosight, Western Blot, and Transmission Electron Microscopy
- (a)
- Nanosight
- (b)
- Western blotting
- (c)
- Transmission electron microscopy
2.10. Statistical Analysis
3. Results
3.1. Experiment 1: EEC Culture in Standard Plates
- (a)
- EEC morphology
- (b)
- EEC viability by MTT test
3.2. Experiment 2: EEC Culture on Transwell System
- (a)
- TEER test
- (b)
- Histological analysis of EECs on transwell insert
3.3. Effect of LPS on Embryo Development
3.4. Evaluation of Gal-9 and LIF by ELISA
3.5. Isolation and Characterization of EVs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CCOs | cumulus oocyte complex |
| EECs | epithelial endometrial cells |
| Evs | extracellular vesicles |
| Gal-9 | Galectin-9 |
| HG-DMEM | Dulbecco’s Modified Eagle’s Medium high glucose |
| IVC | in vitro culture |
| IVF | in vitro fertilization |
| IVM | in vitro maturation |
| LIF | leukaemia inhibitory factor |
| LPS | lipopolysaccharide |
| MTT | tetrazolium dye 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| TBST | tris-buffered saline |
| TEER | trans epithelial electrical resistance |
| TEM | transmission electron microscopy |
References
- Hansen, T.R.; Sinedino, L.D.P.; Spencer, T.E. Paracrine and endocrine actions of interferon tau (IFNT). Reproduction 2017, 154, 45–59. [Google Scholar] [CrossRef]
- Campanile, G.; Baruselli, P.S.; Limone, A.; D’Occhio, M.J. Local action of cytokines and immune cells in communication between the conceptus and uterus during the critical period of early embryo development, attachment and lipid mee Implications for embryo survival in cattle: A review. Theriogenology 2021, 167, 1–12. [Google Scholar] [CrossRef]
- EL Andaloussi, S.; Mäger, I.; Breakefield, X.O.; Wood, M.J.A. Extracellular vesicles: Biology and emerging therapeutic opportunities. Nat. Rev. Drug. Discov. 2013, 12, 347–357. [Google Scholar] [CrossRef]
- Lange-Consiglio, A.; Perrini, C.; Tasquier, R.; Deregibus, M.C.; Camussi, G.; Pascucci, L.; Marini, M.G.; Corradetti, B.; Bizzaro, D.; De Vita, B.; et al. Microvesicles secreted from equine amniotic-derived cells and their potential role in reducing inflammation in a tendon in vitro model. Stem Cells Dev. 2016, 25, 610–621. [Google Scholar] [CrossRef]
- Perrini, C.; Strillacci, M.G.; Bagnato, A.; Esposti, P.; Marini, M.G.; Corradetti, B.; Bizzaro, D.; Idda, A.; Ledda, S.; Capra, E.; et al. Microvesicles secreted from equine amniotic-derived cells and their potential role in reducing inflammation in endometrial cells in an in vitro model. Stem Cell Res. Ther. 2016, 7, 169. [Google Scholar] [CrossRef]
- Aguilera, C.; Wong, Y.S.; Gutierrez-Reinoso, M.A.; Velásquez, A.E.; Melo-Báez, B.; Cabezas, J.; Caamaño, D.; Navarrete, F.; Castro, F.O.; Rodriguez-Alvarez, L.L. Embryo-maternal communication mediated by extracellular vesicles in the early stages of embryonic development is modified by in vitro conditions. Theriogenology 2024, 214, 43–56. [Google Scholar] [CrossRef]
- Qamar, A.Y.; Mahiddine, F.Y.; Bang, S.; Fang, X.; Shin, S.T.; Kim, M.J.; Cho, J. Extracellular vesicle mediated crosstalk between the gametes, conceptus, and female reproductive tract. Front. Vet. Sci. 2020, 7, 589117. [Google Scholar] [CrossRef]
- Hart, A.R.; Khan, N.L.A.; Godaakumara, K.; Godakumara, K.; Dissanayake, K.; Piibor, J.; Muhandiram, S.; Eapen, S.; Heath, P.R.; Fazeli, A. The role of extracellular vesicles in endometrial receptivity and their potential in reproductive therapeutics and diagnosis. Reprod. Biol. 2022, 22, 100645. [Google Scholar] [CrossRef]
- Mazzarella, R.; Cajas, Y.N.; Gonzalez Martínez, M.E.; Rizos, D. Extracellular vesicles: Emerging paradigms in bovine embryo-maternal communication. Anim. Reprod. 2024, 26, e20240065. [Google Scholar] [CrossRef]
- Mishra, A.; Ashary, N.; Sharma, R.; Modi, D. Extracellular vesicles in embryo implantation and disorders of the endometrium. Am. J. Reprod. Immunol. 2021, 85, e13360. [Google Scholar] [CrossRef]
- Bhusane, K.; Bhutada, S.; Chaudhari, U.; Savardekar, L.; Katkam, R.; Sachdeva, G. Secrets of endometrial receptivity: Some are hidden in uterine secretome. Am. J. Reprod. Immunol. 2016, 75, 226–236. [Google Scholar] [CrossRef]
- Aplin, J.D.; Ruane, P.T. Embryo-epithelium interactions during implantation at a glance. J. Cell Sci. 2017, 130, 15–22. [Google Scholar] [CrossRef]
- Cajas, Y.N.; Cañón-Beltrán, K.; de la Blanca, M.G.M.; Sánchez, J.M.; Fernandez-Fuertes, B.; González, E.M.; Rizos, D. Role of reproductive fluids and extracellular vesicles in embryo–maternal interaction during early pregnancy in cattle. Reprod. Fertil. Dev. 2022, 34, 117–138. [Google Scholar] [CrossRef]
- Gilbert, R.O. Symposium review: Mechanisms of disruption of fertility by infectious diseases of the reproductive tract. J. Dairy Sci. 2019, 102, 3754–3766. [Google Scholar] [CrossRef]
- Guo, Y.; van Schaik, T.; Jhamat, N.; Niazi, A.; Chanrot, M.; Charpigny, G.; Valarcher, J.F.; Bongcam-Rudloff, E.; Andersson, G.; Humblot, P. Differential gene expression in bovine endometrial epithelial cells after challenge with LPS; specific implications for genes involved in embryo maternal interactions. PLoS ONE 2019, 14, e0222081. [Google Scholar] [CrossRef]
- Kasimanickam, R.K.; Kasimanickam, V.R.; Kumar, N.; Reisenauer, C. Day 7 embryo quality and suboptimal uterine environment influence morphometry of Day 16 conceptus in dairy cows. Theriogenology 2021, 163, 10–17. [Google Scholar] [CrossRef]
- Szelényi, Z.; Kovács, L.; Szenci, O.; Lopez-Gatius, F. The uterus as an influencing factor for late embryo/early fetal loss—A clinical update. Animals 2022, 12, 1873. [Google Scholar] [CrossRef]
- Jhamat, N.; Niazi, A.; Guo, Y.; Chanrot, M.; Ivanova, E.; Kelsey, G.; Bongcam-Rudloff, E.; Andersson, G.; Humblot, P. LPS-treatment of bovine endometrial epithelial cells causes differential DNA methylation of genes associated with inflammation and endometrial function. BMC Genom. 2020, 21, 385. [Google Scholar] [CrossRef]
- Tinning, H.; Edge, J.C.; DeBem, T.H.C.; Deligianni, F.; Giovanardi, G.; Pensabene, V.; Meirelles, F.V.; Forde, N. Review: Endometrial function in pregnancy establishment in cattle. Animals 2023, 17, 100751. [Google Scholar] [CrossRef]
- Husnain, A.; Arshad, U.; Zimpel, R.; Schmitt, E.; Dickson, M.J.; Perdomo, M.C.; Marinho, M.N.; Ashrafi, N.; Graham, S.F.; Bishop, J.V.; et al. Induced endometrial inflammation compromises conceptus development in dairy cattle. Biol. Reprod. 2023, 109, 415–431. [Google Scholar] [CrossRef]
- Brubel, R.; Bokor, A.; Pohl, A.; Schilli, G.K.; Szereday, L.; Bacher-Szamuel, R.; Rigo, J.; Polgar, B. Serum galectin-9 as a noninvasive biomarker for the detection of endometriosis and pelvic pain or infertility-related gynecologic disorders. Fertil. Steril. 2017, 108, 1016–1025. [Google Scholar] [CrossRef]
- Martin-Saldaña, S.; Chevalier, M.T.; Pandit, A. Therapeutic potential of targeting galectins—A biomaterials-focused perspective. Biomaterials 2022, 286, 121585. [Google Scholar] [CrossRef]
- Paiva, P.; Menkhorst, E.; Salamonsen, L.; Dimitriadis, E. Leukemia inhibitory factor and IL-11: Critical regulators in the establishment of pregnancy. Cytokine Growth Factor Rev. 2009, 20, 319–328. [Google Scholar] [CrossRef]
- Donofrio, G.; Franceschi, V.; Capocefalo, A.; Cavirani, S.; Sheldon, I.M. Bovine endometrial stromal cells display osteogenic properties. Reprod. Biol. Endocrinol. 2008, 6, 65. [Google Scholar] [CrossRef]
- Ghasemi, M.; Turnbull, T.; Sebastian, S.; Kempson, I. The MTT assay: Utility, limitations, pitfalls, and interpretation in bulk and single-cell analysis. Int. J. Mol. Sci. 2021, 22, 12827. [Google Scholar] [CrossRef]
- Benson, K.; Cramer, S.; Galla, H.J. Impedance-based cell monitoring: Barrier properties and beyond. Fluids Barriers CNS 2013, 10, 5. [Google Scholar] [CrossRef]
- Srinivasan, B.; Kolli, A.R.; Esch, M.B.; Abaci, H.E.; Shuler, M.L.; Hickman, J.J. TEER measurement techniques for in vitro barrier model systems. J. Lab. Autom. 2015, 20, 107–126. [Google Scholar] [CrossRef]
- Manna, V.; Caradonna, S. Isolation, expansion, differentiation, and histological processing of human nasal epithelial cells. STAR Protoc. 2021, 2, 100782. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (misev2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Welsh, J.A.; Goberdhan, D.C.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J. Extracell. Vesicles 2023, 13, e12451. [Google Scholar] [CrossRef]
- Turner, M.L.; Cronin, J.G.; Healey, G.D.; Sheldon, I.M. Epithelial and Stromal Cells of Bovine Endometrium Have Roles in Innate Immunity and Initiate Inflammatory Responses to Bacterial Lipopeptides In Vitro via Toll-Like Receptors TLR2, TLR1, and TLR6. Endocrinology 2014, 155, 1453–1465. [Google Scholar] [CrossRef]
- Chanrot, M.; Guo, Y.; Dalin, A.M.; Persson, E.; Båge, R.; Svensson, A.; Gustafsson, H.; Humblot, P. Dose related effects of LPS on endometrial epithelial cell populations from dioestrus cows. Anim. Reprod. Sci. 2017, 177, 12–24. [Google Scholar] [CrossRef]
- Ma, X.; Li, Y.; Shen, W.; Oladejo, O.A.; Yang, J.; Jiang, W.; Imam, B.H.; Wu, X.; Ding, X.; Yang, Y.; et al. LPS mediates endometrial epithelial cell pyroptosis directly through both NLRP3 classical and non-classical inflammasome pathways. Front. Immunol. 2021, 12, 676088. [Google Scholar] [CrossRef]
- Marini, M.G.; Perrini, C.; Esposti, P.; Corradetti, B.; Bizzaro, D.; Riccaboni, P.; Fantinato, E.; Urbani, G.; Gelati, G.; Cremonesi, F.; et al. Effects of platelet-rich plasma in a model of bovine endometrial inflammation in vitro. Reprod. Biol. Endocrinol. 2016, 14, 58. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki-Hozumi, H.; Chagan-Yasutan, H.; Ashino, Y.; Hattori, T. Blood levels of Galectin-9, an immuno-regulating molecule, reflect the severity for the acute and chronic infectious diseases. Biomolecules 2021, 11, 430. [Google Scholar] [CrossRef] [PubMed]
- Popovici, R.M.; Krause, M.S.; Germeyer, A.; Strowitzki, T.; von Wolff, M. Galectin-9: A new endometrial epithelial marker for the mid- and late-secretory and decidual phases in humans. J. Clin. Endocrinol. Metab. 2005, 90, 6170–6176. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, Y.; Kabir-Salmani, M.; Azadbakht, M.; Sugihara, K.; Sakai, K.; Iwashita, M. Expression and localization of galectin-9 in the human uterodome. Endocr. J. 2008, 55, 879–887. [Google Scholar] [CrossRef]
- Conquet, P.; Brulet, F. Developmental expression of myeloid leukaemia inhibitory factor gene in preimplantation blastocysts and in extraembryonic tissue of mouse embryos. Mol. Cell Biol. 1990, 10, 3801–3805. [Google Scholar] [CrossRef]
- Zollner, U.; Bischofs, S.; Lalic, I.; Zollner, K.P. LIF and TNF alpha concentrations in embryo culture media are predictive for embryo implantation in IVF. Asian Pac. J. Reprod. 2012, 1, 277–282. [Google Scholar] [CrossRef]
- Chen, H.F.; Shew, J.Y.; Ho, H.N.; Hsu, W.L.; Yang, Y.S. Expression of leukemia inhibitory factor and its receptor in preimplantation embryos. Fertil. Steril. 1999, 72, 713–719. [Google Scholar] [CrossRef]
- Dunglison, G.F.; Barlow, D.H.; Sargent, I.L. Leukaemia inhibitory factor signi®cantly enhances the blastocyst formation rates of human embryos cultured in serum-free medium. Hum. Reprod. 1996, 11, 191–196. [Google Scholar] [CrossRef]
- Kocyigit, A.; Cevik, M. Leucemia inhibitory factor; investigating the time-dependent effect on viability of vitrified bovine embryos. Reprod. Domest. Anim. 2017, 52, 1113–1119. [Google Scholar] [CrossRef]
- Oliver, M.A.; Alward, K.J.; Rhoads, M.L.; Ealy, A.D. Human recombinant interleukin-6 and keukemia inhibitory factor improve inner cell mass cell number but lack cryoprotective activities on in vitro-produced bovine blastocysts. Animals 2025, 15, 668. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, Y.; Li, H.; Jiang, W.; Liu, H.; Yan, J.; Chen, Z.J.; Li, W. Leukaemia inhibitory factor in serum and follicular fluid of women with polycystic ovary syndrome and its correlation with IVF outcome. Reprod. Biomed Online 2018, 36, 483–489. [Google Scholar] [CrossRef]
- Bhatt, H.; Brunet, L.J.; Stewart, C.A. Uterine expression of leukemia inhibitory factor coincides with the onset of blastocyst implantation. Proc. Natl. Acad. Sci. USA 1991, 88, 11408–11412. [Google Scholar] [CrossRef]
- Stewart, C.L. Leukaemia inhibitory factor and the regulation of pre-implantation development of the mammalian embryo. Mol. Reprod. Dev. 1994, 39, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Mazzarella, R.; Cañón-Beltrán, K.; Cajas, Y.N.; Hamdi, M.; González, E.M.; da Silveira, J.C.; Leal, C.L.V.; Rizos, D. Extracellular vesicles-coupled miRNAs from oviduct and uterus modulate signaling pathways related to lipid metabolism and bovine early embryo development. J. Anim. Sci. Biotechnol. 2024, 15, 69. [Google Scholar] [CrossRef] [PubMed]
- Machtinger, R.; Baccarelli, A.A.; Wu, H. Extracellular vesicles and female reproduction. J. Assist. Reprod. Gen. 2021, 38, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Dissanayake, K.; Nõmm, M.; Lättekivi, F.; Ressaissi, Y.; Godakumara, K.; Lavrits, A.; Midekessa, G.; Viil, J.; Bæk, R.; Jørgensen, M.M.; et al. Individually cultured bovine embryos produce extracellular vesicles that have the potential to be used as non-invasive embryo quality markers. Theriogenology 2020, 149, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Kusama, K.; Nakamura, K.; Bai, R.; Nagaoka, K.; Sakurai, T.; Imakawa, K. Intrauterine exosomes are required for bovine conceptus implantation. Biochem. Biophys. Res. Commun. 2018, 960, 1370–1375. [Google Scholar] [CrossRef]
- Giacomini, E.; Vago, R.; Sanchez, A.M.; Podini, P.; Zarovni, N.; Murdica, V. Secretome of in vitro cultured human embryos contains extracellular vesicles that are uptaken by the maternal side. Sci. Rep. 2017, 7, 5210. [Google Scholar] [CrossRef] [PubMed]
- Burns, G.W.; Brooks, K.E.; Spencer, T.E. Extracellular vesicles originate from the conceptus and uterus during early pregnancy in sheep. Biol. Reprod. 2016, 94, 56. [Google Scholar] [CrossRef]
- Aguilera, C.; Velásquez, A.E.; Gutierrez-Reinoso, M.A.; Wong, Y.S.; Melo-Baez, B.; Cabezas, J.; Caamaño, D.; Navarrete, F.; Rojas, D.; Riadi, G.; et al. Extracellular vesicles secreted by pre-hatching bovine embryos produced in vitro and in vivo alter the expression of IFNtau-stimulated genes in bovine endometrial cells. Int. J. Mol. Sci. 2023, 24, 7438. [Google Scholar] [CrossRef]
- Mazzarella, R.; Sánchez, J.M.; Egido, S.G.; McDonald, M.; Álvarez-Barrientos, A.; González, E.; Falcón-Pérez, J.M.; Azkargorta, M.; Elortza, F.; González, M.E.; et al. Deciphering the dialogue between the bovine blastocyst and the uterus: Embryo-induced alterations in extracellular vesicle protein content from an ex vivo model and the in vivo environment. J. Anim. Sci. Biotechnol. 2025, 24, 137. [Google Scholar] [CrossRef]
- Piibor, J.; Waldmann, A.; Dissanayake, K.; Andronowska, A.; Ivask, M.; Prasadani, M.; Kavak, A.; Kodithuwakku, S.; Fazeli, A. Uterine fluid extracellular vesicles proteome is altered during the estrous cycle. Mol. Cell Proteom. 2023, 22, 100642. [Google Scholar] [CrossRef]
- Chiaradia, E.; Tancini, B.; Emiliani, C.; Delo, F.; Pellegrino, R.M.; Tognoloni, A.; Urbanelli, L.; Buratta, S. Extracellular vesicles under oxidative stress conditions: Biological properties and physiological roles. Cells 2021, 10, 1763. [Google Scholar] [CrossRef]
- Senn, L.K.; Peterson, K.D.; Edwards, J.L.; Payton, R.R.; Mathew, D.J. Oviduct and endometrial epithelium improve in vitro produced bovine embryo developmental kinetics. Reproduction 2024, 167, e240008. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Banliat, C.; Mahé, C.; Lavigne, R.; Com, E.; Pineau, C.; Labas, V.; Guyonnet, B.; Mermillod, P.; Saint-Dizier, M. The proteomic analysis of bovine embryos developed in vivo or in vitro reveals the contribution of the maternal environment to early embryo. BMC Genom. 2022, 23, 839. [Google Scholar] [CrossRef] [PubMed]
- Lange-Consiglio, A.; Lazzari, B.; Pizzi, F.; Idda, A.; Cremonesi, F.; Capra, E. Amniotic microvesicles impact hatching and pregnancy percentages of in vitro bovine embryos and blastocyst microRNA expression versus in vivo controls. Sci. Rep. 2020, 10, 501. [Google Scholar] [CrossRef] [PubMed]







| Time (Days) | % Viability (IVC/DMEM) |
|---|---|
| 4 | 89.59 ± 5.56 a |
| 8 | 85.28 ± 8.83 a |
| 11 | 90.77 ± 6.12 a |
| Day of Culture | Embryo (% Blastocysts) | EECs+Embryo (% Blastocysts) | EECs+LPS+Embryo (% Blastocysts) |
|---|---|---|---|
| 7 | 69/208 = 33.17 ± 2.54 a | 58/166 = 34.94 ± 1.95 a | 119/360 = 33.06 ± 3.08 a |
| 9 | 71/192 = 36.98 ± 3.22 a | 61/182 = 33.52 ± 3.22 a | 47/196 = 23.98 ± 3.41 b |
| 11 | 41/164 = 25.00 ± 2.10 a | 35/152 = 23.03 ± 3.18 a | 0/166 = 0 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lange-Consiglio, A.; Gaspari, G.; Gagni, P.; Bosi, G.; Riccaboni, P.; Cremonesi, F. LPS-Stressed Bovine Endometrial Cells upon Morulae in a Transwell Model of Embryo—Maternal Talk. Animals 2026, 16, 38. https://doi.org/10.3390/ani16010038
Lange-Consiglio A, Gaspari G, Gagni P, Bosi G, Riccaboni P, Cremonesi F. LPS-Stressed Bovine Endometrial Cells upon Morulae in a Transwell Model of Embryo—Maternal Talk. Animals. 2026; 16(1):38. https://doi.org/10.3390/ani16010038
Chicago/Turabian StyleLange-Consiglio, Anna, Giulia Gaspari, Paola Gagni, Giampaolo Bosi, Pietro Riccaboni, and Fausto Cremonesi. 2026. "LPS-Stressed Bovine Endometrial Cells upon Morulae in a Transwell Model of Embryo—Maternal Talk" Animals 16, no. 1: 38. https://doi.org/10.3390/ani16010038
APA StyleLange-Consiglio, A., Gaspari, G., Gagni, P., Bosi, G., Riccaboni, P., & Cremonesi, F. (2026). LPS-Stressed Bovine Endometrial Cells upon Morulae in a Transwell Model of Embryo—Maternal Talk. Animals, 16(1), 38. https://doi.org/10.3390/ani16010038






