From Metabolism to Vitality: Uncovering Riboflavin’s Importance in Poultry Nutrition
Abstract
Simple Summary
Abstract
1. Introduction
- Biochemical Fundamentals of Riboflavin;
- Riboflavin Metabolism in Poultry;
- Riboflavin and Poultry Growth;
- Oxidative Stress Defense;
- Reproductive Performance and Hatchability;
- Riboflavin Requirements for Poultry;
- Future Prospects and Research Avenues.
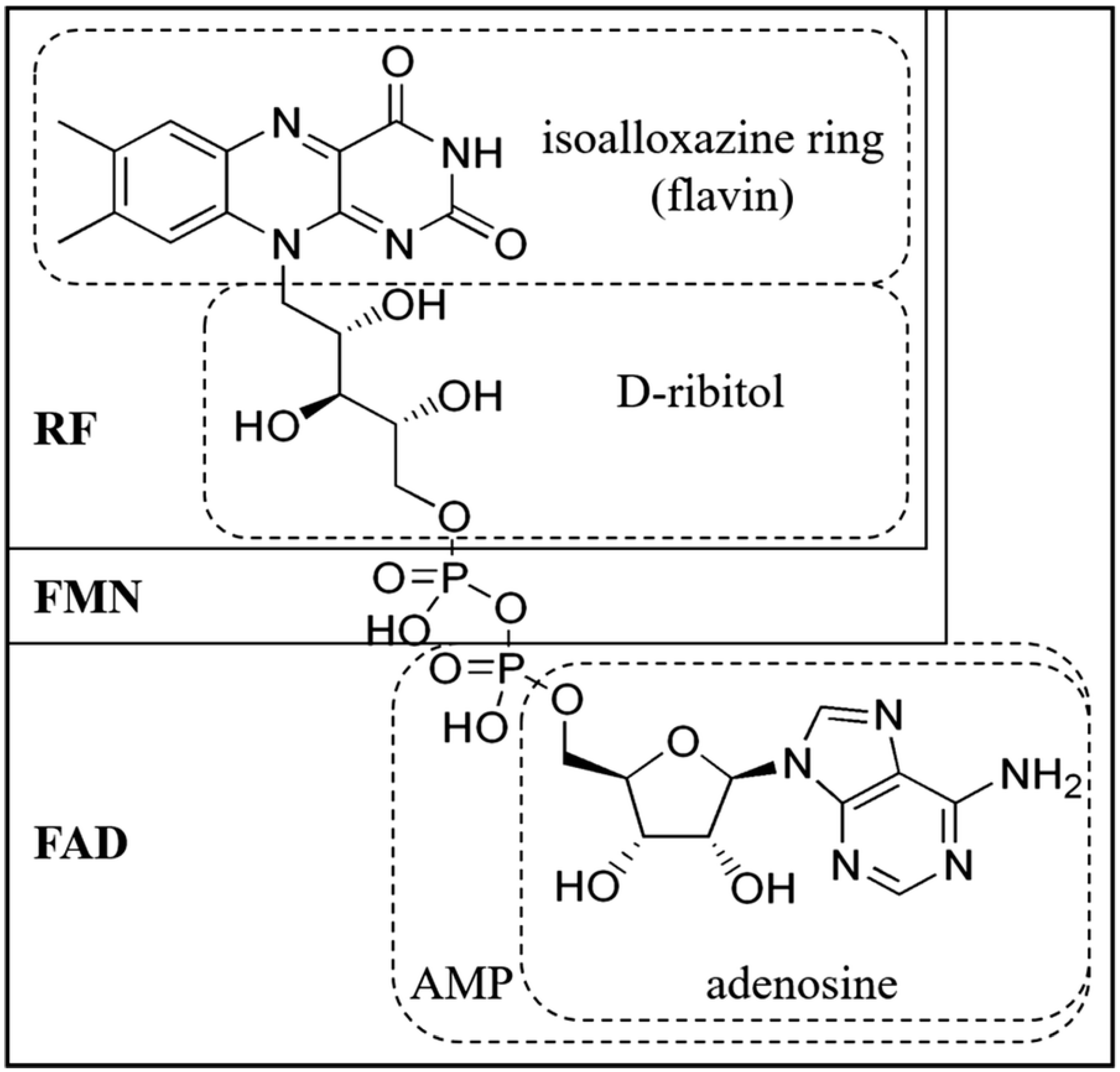
2. Biochemical Fundamentals of Riboflavin
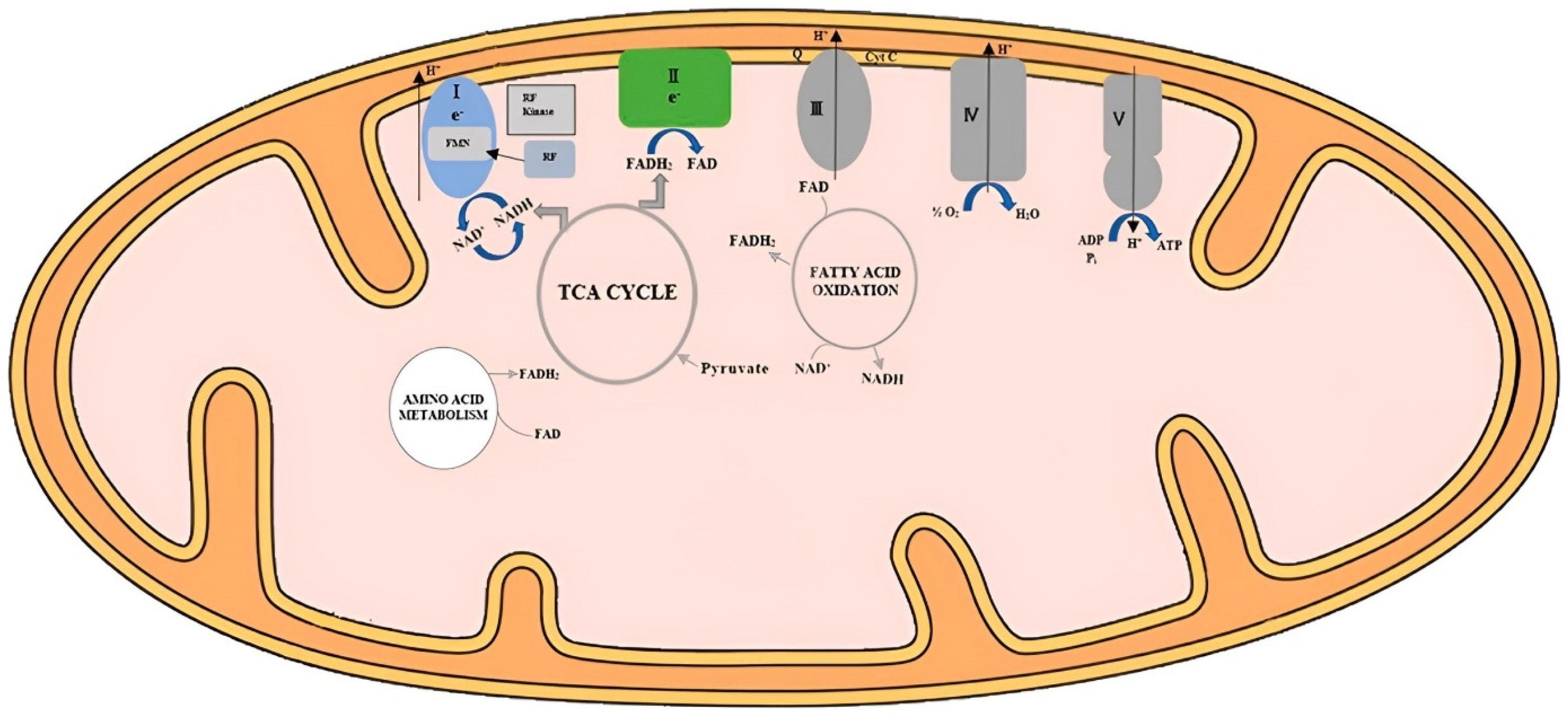
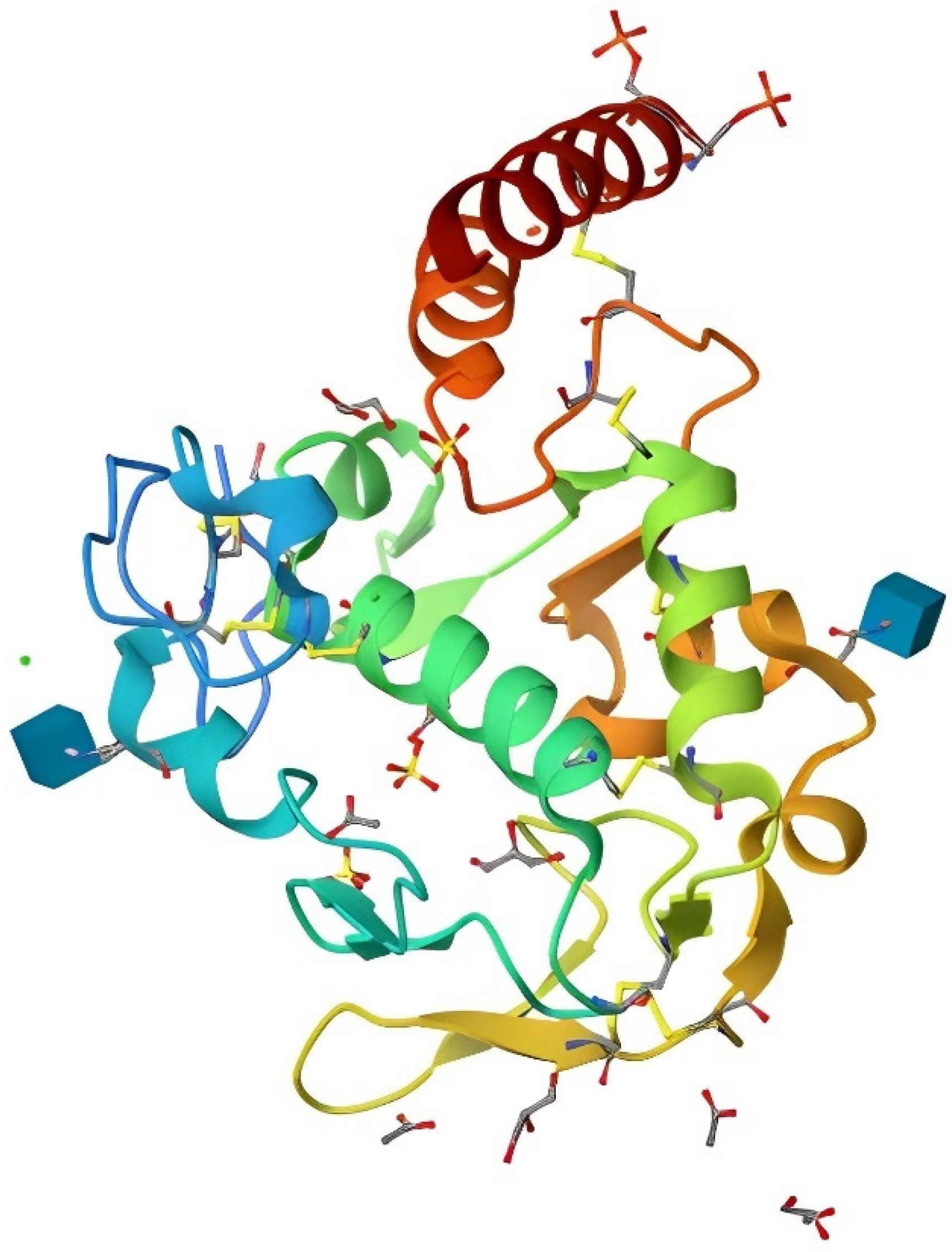
3. Riboflavin Metabolism in Poultry
4. Riboflavin and Poultry Growth
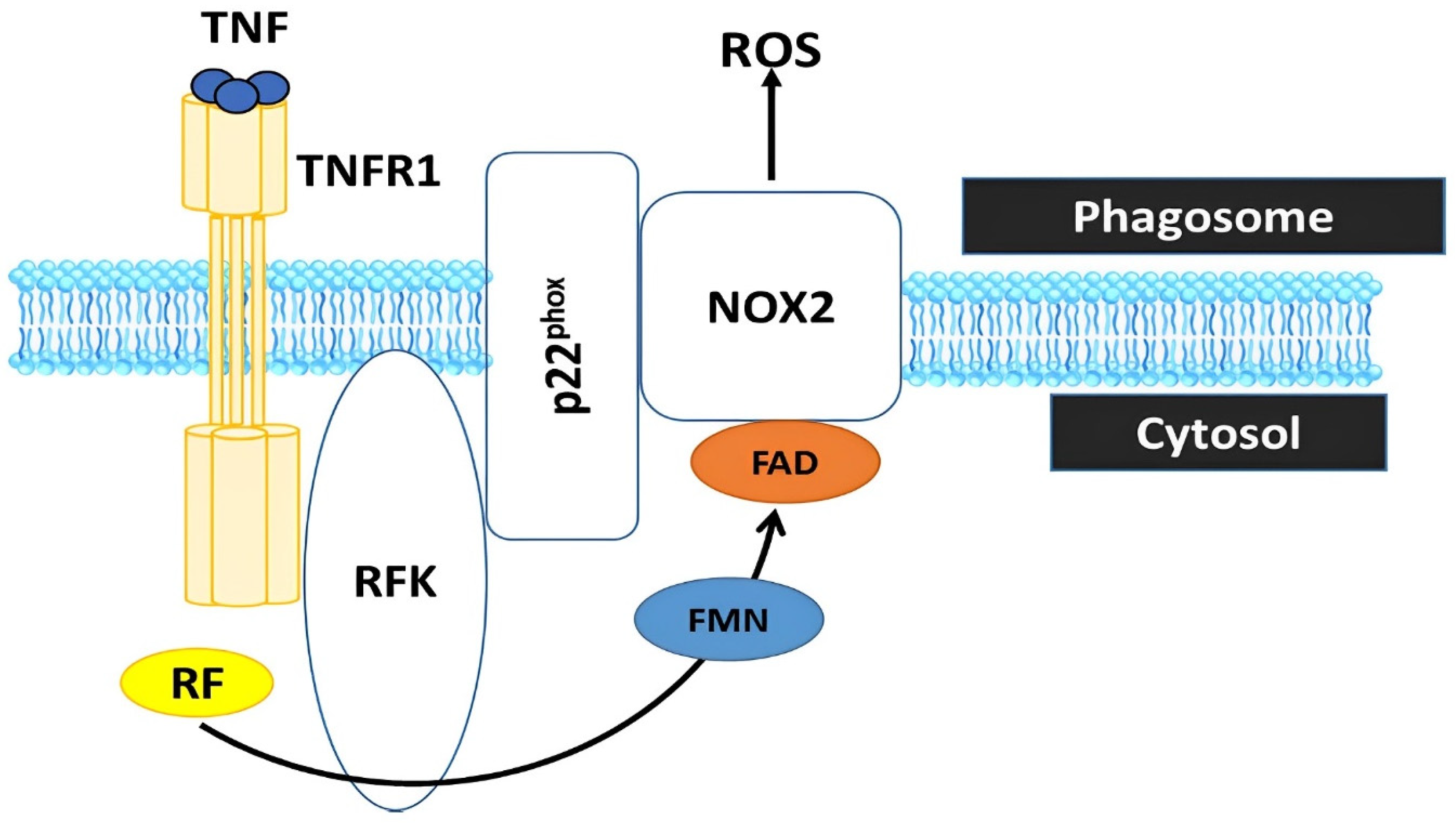
5. Oxidative Stress Defense
6. Reproductive Performance and Hatchability
7. Riboflavin Requirements for Poultry
8. Future Prospects and Research Avenues
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AMP | Adenosine monophosphate |
| ATP | Adenosine triphosphate |
| COX | Cytochrome c oxidase |
| ETC | Electron transport chain |
| FAD | Flavin adenine dinucleotide |
| FADH2 | Dinucleotide adenine flavine molecule |
| FAO | The Food and Agriculture Organization of the United Nations |
| FCR | Feed conversion ratio |
| FMN | Flavin mononucleotide |
| G-6P-D | Glucose-6-phosphate dehydrogenase |
| GSH | Reduced form of glutathione |
| GSSG | Oxidized glutathione |
| Irg1 | The immune responsive gene 1 protein |
| IUPAC | The International Union of Pure and Applied Chemistry |
| NADH | Dinucleotide nicotinamide molecules |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| Nox2 | NADPH oxidase 2 |
| PDH | Pyruvate dehydrogenase |
| Q | Coenzyme Q10 |
| RBP | Riboflavin-binding protein |
| RF | Riboflavin |
| RFK | Riboflavin kinase |
| ROS | Reactive oxygen species |
| TCA | Tricarboxylic Acid cycle |
| TNF | Tumor necrosis factor |
| TNFR1 | Tumor necrosis factor receptor 1 |
References
- FAO (The Food and Agriculture Organization of the United Nations). OECD-FAO Agricultural Outlook 2022–2031: 6. Meat. 2022. Available online: https://www.oecd-ilibrary.org/sites/ab129327-en/index.html?itemId=/content/component/ab129327-en#:~:text=Poultry%20meat%20is%20projected%20to,by%20pig%2C%20sheep%20and%20bovine (accessed on 23 August 2023).
- Roth-Maier, D.A.; Paulicks, B.R. Effects of a suboptimal dietary intake of particular B-vitamins on the growth of fattening chicken. Arch. Geflügelkunde 2002, 66, 201–205. [Google Scholar]
- McDowell, L.R. Vitamin nutrition of livestock animals: Overview from vitamin discovery to today. Can. J. Anim. Sci. 2006, 86, 171–179. [Google Scholar] [CrossRef]
- Jortner, B.S.; Cherry, J.; Lidsky, T.I.; Manetto, C.; Shell, L. Peripheral Neuropathy of Dietary Riboflavin Deficiency in Chickens. J. Neuropathol. Exp. Neurol. 1987, 46, 544–555. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.D.; Storts, R.W. Peripheral Neuropathy Associated with Dietary Riboflavin Deficiency in the Chicken I. Light Microscopic Study. Vet. Pathol. 1988, 25, 9–16. [Google Scholar] [CrossRef]
- Cai, Z.; Finnie, J.W.; Blumbergs, P.C. Avian Riboflavin Deficiency: An Acquired Tomaculous Neuropathy. Vet. Pathol. 2006, 43, 780–781. [Google Scholar] [CrossRef]
- Leiber, F.; Amsler, Z.; Bieber, A.; Quander-Stoll, N.; Maurer, V.; Lambertz, C.; Früh, B.; Ayrle, H. Effects of riboflavin supplementation level on health, performance, and fertility of organic broiler parent stock and their chicks. Animal 2022, 16, 100433. [Google Scholar] [CrossRef]
- Asplin, F. Riboflavin Deficiency in Poultry. Vet. J. 1941, 97, 16–26. [Google Scholar] [CrossRef]
- Cai, Z.; Finnie, J.; Manavis, J.; Blumbergs, P. Avian riboflavin deficiency causes reliably reproducible peripheral nerve demyelination and, with vitamin supplementation, rapid remyelination. Hum. Exp. Toxicol. 2023, 42. [Google Scholar] [CrossRef]
- Suwannasom, N.; Kao, I.; Pruß, A.; Georgieva, R.; Bäumler, H. Riboflavin: The Health Benefits of a Forgotten Natural Vitamin. Int. J. Mol. Sci. 2020, 21, 950. [Google Scholar] [CrossRef]
- Zhang, B.; Zhao, R.; Fouad, A.; Wu, Y.; Sun, P.; Wei, J.; Huang, W.; Xie, M.; Tang, J.; Hou, S. Research Note: Effects of riboflavin on reproductive performance and antioxidant status of duck breeders. Poult. Sci. 2020, 99, 1564–1570. [Google Scholar] [CrossRef]
- Tang, J.; Hu, J.; Xue, M.; Guo, Z.; Xie, M.; Zhang, B.; Zhou, Z.; Huang, W.; Hou, S. Maternal diet deficient in riboflavin induces embryonic death associated with alterations in the hepatic proteome of duck embryos. Nutr. Metab. 2019, 16, 19. [Google Scholar] [CrossRef]
- Lambertz, C.; Leopold, J.; Damme, K.; Vogt-Kaute, W.; Ammer, S.; Leiber, F. Effects of a riboflavin source suitable for use in organic broiler diets on performance traits and health indicators. Animal 2020, 14, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Cherian, G. Nutrition and metabolism in poultry: Role of lipids in early diet. J. Anim. Sci. Biotechnol. 2015, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Abbas, C.A.; Sibirny, A.A. Genetic Control of Biosynthesis and Transport of Riboflavin and Flavin Nucleotides and Construction of Robust Biotechnological Producers. Microbiol. Mol. Biol. Rev. 2011, 75, 321–360. [Google Scholar] [CrossRef] [PubMed]
- Udhayabanu, T.; Manole, A.; Rajeshwari, M.; Varalakshmi, P.; Houlden, H.; Ashokkumar, B. Riboflavin Responsive Mitochondrial Dysfunction in Neurodegenerative Diseases. J. Clin. Med. 2017, 6, 52. [Google Scholar] [CrossRef] [PubMed]
- Brooks, A.; Martin, E. Riboflavin Deficiency in Broiler Chickens. AHL Newsletter 27(1):20 (Animal Health Laboratory, University of Guelph, Ontario, Canada). Available online: https://www.uoguelph.ca/ahl/riboflavin-deficiency-broiler-chickens (accessed on 23 August 2023).
- Cook, M.E.; Springer, W.T. Effect of Reovirus Infection and Dietary Levels of Selected Vitamins on Immunocompetence of Chickens. Avian Dis. 1983, 27, 367–377. [Google Scholar] [CrossRef]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef]
- Scott, M.L.; Holm, E.R.; Reynolds, R.E. Studies on the Niacin, Riboflavin, Choline, Manganese and Zinc Requirements of Young Ringnecked Pheasants for Growth, Feathering and Prevention of Leg Disorders. Poult. Sci. 1959, 38, 1344–1350. [Google Scholar] [CrossRef]
- Wyatt, R.D.; Tung, H.T.; Donaldson, W.E.; Hamilton, P.B. A New Description of Riboflavin Deficiency Syndrome in Chickens. Poult. Sci. 1973, 52, 237–244. [Google Scholar] [CrossRef]
- Serafin, J. Studies on the Riboflavin, Niacin, Pantothenic Acid and Choline Requirements of Young Bobwhite Quail. Poult. Sci. 1974, 53, 1522–1532. [Google Scholar] [CrossRef]
- Lee, D.J.W. Growth, erythrogyte glutathione redugtase and liver flavin as indicators of riboflavin status in Turkey poults. Br. Poult. Sci. 1982, 23, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Roth-Maier, D.A.; Kirchgessner, M. Investigations on riboflavin requirement of fattening chickens. Arch. Geflügelkunde 1997, 61, 14–16. [Google Scholar]
- Olkowski, A.; Classen, H.L. The study of riboflavin requirement in broiler chickens. Int. J. Vitam. Nutr. Res. 1998, 68, 316–327. [Google Scholar] [PubMed]
- Cogburn, L.A.; Smarsh, D.N.; Wang, X.; Trakooljul, N.; Carré, W.; White, H.B. Transcriptional profiling of liver in riboflavin-deficient chicken embryos explains impaired lipid utilization, energy depletion, massive hemorrhaging, and delayed feathering. BMC Genom. 2018, 19, 177. [Google Scholar] [CrossRef]
- Gržinić, G.; Piotrowicz-Cieślak, A.; Klimkowicz-Pawlas, A.; Górny, R.L.; Ławniczek-Wałczyk, A.; Piechowicz, L.; Olkowska, E.; Potrykus, M.; Tankiewicz, M.; Krupka, M.; et al. Intensive poultry farming: A review of the impact on the environment and human health. Sci. Total Environ. 2023, 858 Pt 3, 160014. [Google Scholar] [CrossRef]
- Balasubramaniam, S.; Yaplito-Lee, J. Riboflavin metabolism: Role in mitochondrial function. J. Transl. Genet. Genom. 2020, 4, 285–306. [Google Scholar] [CrossRef]
- Poudel, S.; Tabler, G.T.; Lin, J.; Zhai, W.; Zhang, L. Riboflavin and Bacillus subtilis effects on growth performance and woody-breast of Ross 708 broilers with or without Eimeria spp. challenge. J. Anim. Sci. Technol. 2022, 64, 443–461. [Google Scholar] [CrossRef]
- Ashoori, M.; Saedisomeolia, A. Riboflavin (vitamin B2) and oxidative stress: A review. Br. J. Nutr. 2014, 111, 1985–1991. [Google Scholar] [CrossRef]
- Olfat, N.; Ashoori, M.; Saedisomeolia, A. Riboflavin is an antioxidant: A review update. Br. J. Nutr. 2022, 128, 1887–1895. [Google Scholar] [CrossRef]
- Alagawany, M.; Elnesr, S.S.; Farag, M.R.; Tiwari, R.; Yatoo, M.I.; Karthik, K.; Michalak, I.; Dhama, K. Nutritional significance of amino acids, vitamins and minerals as nutraceuticals in poultry production and health—A comprehensive review. Vet. Q. 2020, 41, 1–29. [Google Scholar] [CrossRef]
- Cai, Z.; Blumbergs, P.C.; Finnie, J.W.; Manavis, J.; Thompson, P.D. Selective vulnerability of peripheral nerves in avian riboflavin deficiency demyelinating polyneuropathy. Vet. Pathol. 2009, 46, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.; Bacher, A. Biosynthesis of vitamin B2 in plants. Physiol. Plant. 2006, 126, 304–331. [Google Scholar] [CrossRef]
- Kleyn, R.; Chrystal, P. Vitamins. In Broiler Nutrition: Masterclass; Context Products Ltd.: Leicestershire, UK, 2020; pp. 129–142. [Google Scholar]
- Witten, S. Characterisation of Organic Cereals and Grain Legumes as Feedstuffs for Monogastric Animals: Effects of Variety and Environmental Conditions on the Contents of Crude Nutrients, Amino Acids, Thiamine, Riboflavin, and In Vitro Digestibility of Crude Protein and Amino Acids. Ph.D. Thesis, Georg-August-Universität Göttingen, Göttingen, Germany, 2018. [Google Scholar]
- Szczuko, M.; Ziętek, M.; Kulpa, D.; Seidler, T. Riboflavin-properties, occurrence and its use in medicine. Pteridines 2019, 30, 33–47. [Google Scholar] [CrossRef]
- Liu, S.; Hu, W.; Wang, Z.; Chen, T. Production of riboflavin and related cofactors by biotechnological processes. Microb. Cell Factories 2020, 19, 31. [Google Scholar] [CrossRef] [PubMed]
- Rivero, M.; Boneta, S.; Novo, N.; Velázquez-Campoy, A.; Polo, V.; Medina, M. Riboflavin kinase and pyridoxine 5′-phosphate oxidase complex formation envisages transient interactions for FMN cofactor delivery. Front. Mol. Biosci. 2023, 10, 1167348. [Google Scholar] [CrossRef] [PubMed]
- Huerta, C.; Borek, D.; Machius, M.; Grishin, N.V.; Zhang, H. Structure and Mechanism of a Eukaryotic FMN Adenylyltransferase. J. Mol. Biol. 2009, 389, 388–400. [Google Scholar] [CrossRef]
- Friedmann, H.C. Flavin Mononucleotide. In Methods of Enzymatic Analysis, 2nd ed.; Bergmeyer, H.U., Ed.; Academic Press: Cambridge, MA, USA, 1974; pp. 2179–2181. [Google Scholar] [CrossRef]
- Pinto, J.T.; Zempleni, J. Riboflavin. Adv. Nutr. Int. Rev. J. 2016, 7, 973–975. [Google Scholar] [CrossRef]
- Oprian, D.D.; Coon, M.J. Oxidation-reduction states of FMN and FAD in NADPH-cytochrome P-450 reductase during reduction by NADPH. J. Biol. Chem. 1982, 257, 8935–8944. [Google Scholar] [CrossRef]
- Mansoorabadi, S.O.; Thibodeaux, C.J.; Liu, H.-W. The Diverse Roles of Flavin Coenzymes—Nature’s Most Versatile Thespians. J. Org. Chem. 2007, 72, 6329–6342. [Google Scholar] [CrossRef]
- Toyomizu, M.; Kikusato, M.; Kawabata, Y.; Azad, A.K.; Inui, E.; Amo, T. Meat-type chickens have a higher efficiency of mitochondrial oxidative phosphorylation than laying-type chickens. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2011, 159, 75–81. [Google Scholar] [CrossRef]
- Hubert, S.; Athrey, G. Transcriptomic signals of mitochondrial dysfunction and OXPHOS dynamics in fast-growth chicken. PeerJ 2022, 10, e13364. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.L.; Nesheim, M.C.; Young, R.S. Riboflavin. In Nutrition of the Chicken; Scott, M.L., Ed.; Associate Publishing: Ithaca, NY, USA, 1982; pp. 196–204. [Google Scholar]
- Jastroch, M.; Divakaruni, A.S.; Mookerjee, S.; Treberg, J.R.; Brand, M.D. Mitochondrial proton and electron leaks. Essays Biochem. 2010, 47, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.; Mourokh, L.; Vittadello, M. Mechanism of Proton Pumping in Complex I of the Mitochondrial Respiratory Chain. Quantum Rep. 2021, 3, 425–434. [Google Scholar] [CrossRef]
- Teixeira, F.K.; Sanchez, C.G.; Hurd, T.R.; Seifert, J.R.K.; Czech, B.; Preall, J.B.; Hannon, G.J.; Lehmann, R. ATP synthase promotes germ cell differentiation independent of oxidative phosphorylation. Nature 2015, 17, 689–696. [Google Scholar] [CrossRef]
- Qin, Y.; Zhou, J.; Xiong, X.; Huang, J.; Li, J.; Wang, Q.; Yang, H.; Yin, Y. Effect of riboflavin on intestinal development and intestinal epithelial cell function of weaned piglets. J. Anim. Physiol. Anim. Nutr. 2022, 107, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.P.; Schrauwen, P.; Somm, E.; Gastaldi, G.; Hesselink, M.K.C.; Schaart, G.; Kornips, E.; Lo, S.K.; Bufano, D.; Giacobino, J.-P.; et al. Decreased Fatty Acid β-Oxidation in Riboflavin-Responsive, Multiple Acylcoenzyme A Dehydrogenase-Deficient Patients Is Associated with an Increase in Uncoupling Protein-3. J. Clin. Endocrinol. Metab. 2003, 88, 5921–5926. [Google Scholar] [CrossRef][Green Version]
- Parsons, H.G.; Dias, V.C. Intramitochondrial fatty acid metabolism: Riboflavin deficiency and energy production. Biochem. Cell Biol. 1991, 69, 490–497. [Google Scholar] [CrossRef]
- Ruiz, N.; Harms, R.H. Conversion of Tryptophan into Niacin in the Turkey (Meleagris gallipavos). Poult. Sci. 1990, 69, 446–450. [Google Scholar] [CrossRef]
- FNB (Food and Nutrition Board, Institute of Medicine, National Academy of Sciences). Dietary Reference Intakes: Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academy Press: Washington, DC, USA, 1998. [Google Scholar]
- Naber, E.C.; Squires, M.W. Research Note: Early Detection of the Absence of a Vitamin Premix in Layer Diets by Egg Albumen Riboflavin Analysis. Poult. Sci. 1993, 72, 1989–1993. [Google Scholar] [CrossRef]
- Wang, Z.; Brannick, E.; Abasht, B. Integrative transcriptomic and metabolomic analysis reveals alterations in energy metabolism and mitochondrial functionality in broiler chickens with wooden breast. Sci. Rep. 2023, 13, 4747. [Google Scholar] [CrossRef]
- Shastak, Y.; Gordillo, A.; Pelletier, W. The relationship between vitamin A status and oxidative stress in animal production. J. Appl. Anim. Res. 2023, 51, 546–553. [Google Scholar] [CrossRef]
- Cordona, N.; Payne, I. Absorption of Riboflavin in Chickens. Poult. Sci. 1967, 46, 1176–1179. [Google Scholar] [CrossRef]
- Ruiz, N.; Harms, R. Riboflavin Requirement of Broiler Chicks Fed a Corn-Soybean Diet. Poult. Sci. 1988, 67, 794–799. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, N.; Harms, R. Riboflavin Requirement of Turkey Poults Fed a Corn-Soybean Meal Diet from 1 to 21 Days of Age. Poult. Sci. 1989, 68, 715–718. [Google Scholar] [CrossRef]
- Banaszkiewicz, T. Nutritional Value of Soybean Meal. In Soybean and Nutrition; El-Shemy, H., Ed.; IntechOpen: Rijeka, Croatia, 2011; pp. 1–20. Available online: https://www.intechopen.com/books/soybean-and-nutrition/nutritional-value-of-soybeanmeal (accessed on 26 August 2023).
- Merrill, A.H.; Lambeth, J.D.; Edmondson, D.E.; McCormick, D.B. Formation and mode of action of flavoproteins. Annu. Rev. Nutr. 1981, 1, 281–317. [Google Scholar] [CrossRef]
- Chung, T.K.; Baker, D.H. Riboflavin Requirement of Chicks Fed Purified Amino Acid and Conventional Corn-Soybean Meal Diets. Poult. Sci. 1990, 69, 1357–1363. [Google Scholar] [CrossRef]
- Dove, R.; Cook, D.A. Water-Soluble Vitamins in Swine Nutrition. In Swine Nutrition; Lewis, A.J., Southern, L.L., Eds.; CRC Press: New York, NY, USA, 2000; pp. 315–356. [Google Scholar]
- Sheraz, M.A.; Kazi, S.H.; Ahmed, S.; Anwar, Z.; Ahmad, I. Photo, thermal and chemical degradation of riboflavin. Beilstein J. Org. Chem. 2014, 10, 1999–2012. [Google Scholar] [CrossRef]
- Hynd, P.I. Digestion in the mono-gastric animal. In Animal Nutrition: From Theory to Practice; Hynd, P.I., Ed.; CSIRO Publishing: Clayton, Australia, 2019; pp. 42–63. [Google Scholar]
- White, H.B., III; Merrill, A.H., Jr. Riboflavin-Binding Proteins. Annu. Rev. Nutr. 1988, 8, 279–299. [Google Scholar] [CrossRef]
- M’Clelland, D.A. The Refolding of Riboflavin Binding Protein. Ph.D. Thesis, Department of Biological and Molecular Sciences, University of Stirling, Stirling, UK, 1996. [Google Scholar]
- EFSA (the European Food Safety Authority). Tolerable Upper Intake Levels for Vitamins and Minerals. Scientific Committee on Food, Scientific Panel on Dietetic Products, Nutrition and Allergies. 2006. Available online: https://www.efsa.europa.eu/sites/default/files/efsa_rep/blobserver_assets/ndatolerableuil.pdf (accessed on 28 August 2023).
- Shastak, Y.; Pelletier, W. Delving into vitamin A supplementation in poultry nutrition: Current knowledge, functional effects, and practical implications. World’s Poult. Sci. J. 2023, 1–23. [Google Scholar] [CrossRef]
- McCormick, D.B. Riboflavin. In Present Knowledge in Nutrition, 10th ed.; Erdman, J.W., Macdonald, I.A., Zeisel, S.H., Eds.; Wiley-Blackwell: Washington, DC, USA, 2012; pp. 280–292. [Google Scholar]
- Zheng, D.B.; Lim, H.M.; Pène, J.J.; White, H.B., 3rd. Chicken riboflavin-binding protein. cDNA sequence and homology with milk folate-binding protein. J. Biol. Chem. 1988, 263, 11126–11129. [Google Scholar] [CrossRef]
- Monaco, H.L. Crystal structure of chicken riboflavin-binding protein. EMBO J. 1997, 16, 1475–1483. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.S.; Benore-Parsons, M.; White, H.B. Dephosphorylation of chicken riboflavin-binding protein and phosvitin decreases their uptake by oocytes. J. Biol. Chem. 1982, 257, 6818–6824. [Google Scholar] [CrossRef] [PubMed]
- Mac Lachlan, I.; Nimpf, J.; Schneider, W. Avian riboflavin binding protein binds to lipoprotein receptors in association with vitellogenin. J. Biol. Chem. 1994, 269, 24127–24132. [Google Scholar] [CrossRef] [PubMed]
- Loch, J.I.; Lipowska, J.; Lewinski, K. Crystal Structure of Chicken Riboflavin Binding Protein in “Apo” Form at 2.5 A Resolution; Protein Data Bank, Brookhaven National Laboratory: New York, NY, USA, 2018. [Google Scholar] [CrossRef]
- Combs, G.F.; McClung, J.P. Riboflavin. In The Vitamins; Combs, G.F., McClung, J.P., Eds.; Academic Press: London, UK, 2017; pp. 110–159, 315–329. [Google Scholar]
- Norioka, N.; Okada, T.; Hamazume, Y.; Mega, T.; Ikenaka, T. Comparison of the Amino Acid Sequences of Hen Plasma-, Yolk-, and White-Riboflavin Binding Proteins. J. Biochem. 1985, 97, 19–28. [Google Scholar] [CrossRef]
- Hamazume, Y.; Mega, T.; Ikenaka, T. Characterization of Hen Egg White- and Yolk-Riboflavin Binding Proteins and Amino Acid Sequence of Egg White-Riboflavin Binding Protein. J. Biochem. 1984, 95, 1633–1644. [Google Scholar] [CrossRef]
- Hamazume, Y.; Mega, T.; Ikenaka, T. Positions of disulfide bonds in riboflavin-binding protein of hen egg white. J. Biochem. 1987, 101, 217–223. [Google Scholar] [CrossRef]
- Kirchhausen, T.; Owen, D.; Harrison, S.C. Molecular Structure, Function, and Dynamics of Clathrin-Mediated Membrane Traffic. Cold Spring Harb. Perspect. Biol. 2014, 6, a016725. [Google Scholar] [CrossRef]
- Carlsson, E.V.; Sherman, H.C. Riboflavin and a Further Growth Essential in the Tissues: Quantitative Distribution and the Influence of the Food, Two Figures. J. Nutr. 1938, 15, 57–65. [Google Scholar] [CrossRef]
- Hodson, A.Z. The Influence of Dietary Riboflavin on the Content of This Vitamin in Chicken Tissue. J. Nutr. 1940, 20, 377–382. [Google Scholar] [CrossRef]
- Leonhardt, M.; Wenk, C. Animal species and muscle related differences in thiamine and riboflavin contents of Swiss meat. Food Chem. 1997, 59, 449–452. [Google Scholar] [CrossRef]
- Rivlin, R.S. Hormonal Regulation of Riboflavin Metabolism. In Riboflavin; Rivlin, R.S., Ed.; Springer: Boston, MA, USA, 1975. [Google Scholar] [CrossRef]
- McCormick, D.B. Riboflavin. In Encyclopedia of Toxicology, 3rd ed.; Wexler, P., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 128–129. [Google Scholar] [CrossRef]
- Fischer, M.; Bacher, A. Riboflavin Biosynthesis. In Comprehensive Natural Products II; Liu, H.-W., Mander, L., Eds.; Elsevier: Singapore, 2010; pp. 3–36. [Google Scholar] [CrossRef]
- Karthikeyan, S.; Zhou, Q.; Mseeh, F.; Grishin, N.V.; Osterman, A.L.; Zhang, H. Crystal Structure of Human Riboflavin Kinase Reveals a β Barrel Fold and a Novel Active Site Arch. Structure 2003, 11, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Norris, L.; Heuser, C.G.F.; Wilgus, H.S. Is the chief value of milk for feeding poultry due to the presence of a new vitamin? Poult. Sci. 1930, 9, 133–140. [Google Scholar] [CrossRef]
- Lepkovsky, S.; Jukes, T.H. The Response of Rats, Chicks and Turkey Poults to Crystalline Vitamin G (Flavin). J. Nutr. 1936, 12, 515–526. [Google Scholar] [CrossRef]
- Norris, L.; Wilgus, C.H.S.; Ringrose, A.T.; Heiman, V.; Heuser, G.F. The vitamin G requirements of poultry. Cornell Agr. Exp. Stn. Bull. 1936, 600, 1. [Google Scholar]
- Bethke, R.M.; Record, P.R.; Wilder, O.H.M. Further studies on vitamin G in chick nutrition with special reference to flavins. Poult. Sci. 1937, 16, 175–182. [Google Scholar] [CrossRef]
- Heuser, G.; Wilgus, H.; Norris, L. The Quantitative Vitamin-G Requirement of Chicks. Poult. Sci. 1938, 17, 105–108. [Google Scholar] [CrossRef]
- Bethke, R.; Record, P. The Relation of Riboflavin to Growth and Curled-toe Paralysis in Chicks. Poult. Sci. 1942, 21, 147–154. [Google Scholar] [CrossRef]
- Biagi, E.; Mengucci, C.; Barone, M.; Picone, G.; Lucchi, A.; Celi, P.; Litta, G.; Candela, M.; Manfreda, G.; Brigidi, P.; et al. Effects of Vitamin B2 Supplementation in Broilers Microbiota and Metabolome. Microorganisms 2020, 8, 1134. [Google Scholar] [CrossRef]
- Northrop-Clewes, C.A.; Thurnham, D.I. The Discovery and Characterization of Riboflavin. Ann. Nutr. Metab. 2012, 61, 224–230. [Google Scholar] [CrossRef]
- Chou, S.T.; Sell, J.L.; Kondra, P.A. Interrelationships between riboflavin and dietary energy and protein utilisation in growing chicks. Br. J. Nutr. 1971, 26, 323–333. [Google Scholar] [CrossRef]
- De Oliveira, J.E.; Uni, Z.; Ferket, P.R. Important metabolic pathways in poultry embryos prior to hatch. World’s Poult. Sci. J. 2008, 64, 488–499. [Google Scholar] [CrossRef]
- Van Every, H.A.; Schmidt, C.J. Transcriptomic and metabolomic characterization of post-hatch metabolic reprogramming during hepatic development in the chicken. BMC Genom. 2021, 22, 380. [Google Scholar] [CrossRef] [PubMed]
- Tu, B.P.; Ho-Schleyer, S.C.; Travers, K.J.; Weissman, J.S. Biochemical Basis of Oxidative Protein Folding in the Endoplasmic Reticulum. Science 2000, 290, 1571–1574. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Cao, J.-T.; Wu, Y.-B.; Gao, K.-X.; Xie, M.; Zhou, Z.-K.; Tang, J.; Hou, S.-S. Riboflavin (Vitamin B2) Deficiency Induces Apoptosis Mediated by Endoplasmic Reticulum Stress and the CHOP Pathway in HepG2 Cells. Nutrients 2022, 14, 3356. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Nakamura, M. Effects of riboflavin deficiency on the lipids of rat liver. J. Nutr. Sci. Vitaminol. 1976, 22, 135–146. [Google Scholar] [CrossRef][Green Version]
- Manthey, K.C.; Chew, Y.C.; Zempleni, J. Riboflavin Deficiency Impairs Oxidative Folding and Secretion of Apolipoprotein B-100 in HepG2 Cells, Triggering Stress Response Systems. J. Nutr. 2005, 135, 978–982. [Google Scholar] [CrossRef][Green Version]
- Xin, Z.; Pu, L.; Gao, W.; Wang, Y.; Wei, J.; Shi, T.; Yao, Z.; Guo, C. Riboflavin deficiency induces a significant change in proteomic profiles in HepG2 cells. Sci. Rep. 2017, 7, srep45861. [Google Scholar] [CrossRef]
- Lambertz, C.; Leopold, J.; Ammer, S.; Leiber, F.; Thesing, B.; Wild, C.; Damme, K. Demand-oriented riboflavin supply of organic broiler using a feed material from fermentation of Ashbya gossypii. Animal 2021, 15, 100003. [Google Scholar] [CrossRef]
- Patrick, H.; Darrow, M.; Morgan, C. The Role of Riboflavin in Turkey Poult Nutrition. Poult. Sci. 1944, 23, 146–148. [Google Scholar] [CrossRef]
- Thesing, B.; Göppel, S.; Weindl, P.; Lambertz, C.; Damme, K.; Bellof, G. Efficiency of an Organic Farming Compatible Yeast Product to Ensure the Riboflavin-Supply of Organically raised B.U.T. 6 Turkey Poults–Effects on Animal Performance and Health; Verlag Eugen Ulmer: Stuttgart, Germany, 2021. [Google Scholar]
- Lepkovsky, S.; Taylor, L.W.; Jukes, T.H.; Almquist, H.J. The effect of riboflavin and the filtrate factor on egg production and hatchability. Hilgardia 1938, 11, 559–591. [Google Scholar] [CrossRef][Green Version]
- Jukes, T.H.; Stokstad, E.L.R.; Belt, M. Deficiencies of Certain Vitamins as Studied with Turkey Poults on a Purified Diet: I. Pteroylglutamic Acid, Riboflavin, Niacin and Inositol: Six Figures. J. Nutr. 1947, 33, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Coles, R.; Cumber, F. Observations on the relationship between riboflavin, hatchability and clubbed down. J. Agric. Sci. 1955, 46, 191–198. [Google Scholar] [CrossRef]
- Haves, R.O.; Buss, E.G. The use of the ribollavinless gene (rd) in determining the cause of clubbed down. Poultry Sci. 1965, 44, 773–778. [Google Scholar]
- Brean, S.E. Dietary Riboflavin and the Production of Clubbed Down in Chick Embryo. Master’s Thesis, Oregon State University, Department of Poultry Science, Corvallis, OR, USA, 1989. [Google Scholar]
- Alibardi, L.; Toni, M. Cytochemical and molecular characteristics of the process of cornification during feather morphogenesis. Prog. Histochem. Cytochem. 2008, 43, 1–69. [Google Scholar] [CrossRef]
- Kleiber, M.; Jukes, T.H. Metabolism and Food Utilization of Riboflavin-Deficient Chicks. Proc. Soc. Exp. Biol. Med. 1942, 49, 34–37. [Google Scholar] [CrossRef]
- Tang, J.; Hegeman, M.A.; Hu, J.; Xie, M.; Shi, W.; Jiang, Y.; de Boer, V.; Guo, Y.; Hou, S.; Keijer, J. Severe riboflavin deficiency induces alterations in the hepatic proteome of starter Pekin ducks. Br. J. Nutr. 2017, 118, 641–650. [Google Scholar] [CrossRef]
- Summers, J.D.; Shen, H.; Leeson, S.; Julian, R.J. Influence of Vitamin Deficiency and Level of Dietary Protein on the Incidence of Leg Problems in Broiler Chicks. Poult. Sci. 1984, 63, 1115–1121. [Google Scholar] [CrossRef]
- Squires, M.W.; Naber, E.C. Vitamin Profiles of Eggs as Indicators of Nutritional Status in the Laying Hen: Riboflavin Study. Poult. Sci. 1993, 72, 483–494. [Google Scholar] [CrossRef]
- Forrester, S.J.; Kikuchi, D.S.; Hernandes, M.S.; Xu, Q.; Griendling, K.K. Reactive Oxygen Species in Metabolic and Inflammatory Signaling. Circ. Res. 2018, 122, 877–902. [Google Scholar] [CrossRef]
- Donaldson, W. Interaction of lead toxicity and riboflavin status in chicks (Gallus domesticus). Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1986, 85, 1–3. [Google Scholar] [CrossRef]
- Deyhim, F.; Belay, T.; Teeter, R.G. An evaluation of dietary riboflavin supplementation on growth rate, feed efficiency, ration metabolizable energy content, and glutathione reductase activity of broilers. Nutr. Res. 1992, 12, 1123–1130. [Google Scholar] [CrossRef]
- Stepuro, I.I.; Adamchuk, R.I.; Stepuro, A.I. Interaction of riboflavin and hemoproteins with organic free radicals and superoxide anions generated in the ultrasound field. Biofizika 2002, 47, 977–988. [Google Scholar] [PubMed]
- Mishra, B.; Jha, R. Oxidative Stress in the Poultry Gut: Potential Challenges and Interventions. Front. Vet. Sci. 2019, 6, 60. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Kidd, M.T. Antioxidant Defence Systems and Oxidative Stress in Poultry Biology: An Update. Antioxidants 2019, 8, 235. [Google Scholar] [CrossRef]
- Shastak, Y.; Obermueller-Jevic, U.; Pelletier, W. A Century of Vitamin E: Early Milestones and Future Directions in Animal Nutrition. Agriculture 2023, 13, 1526. [Google Scholar] [CrossRef]
- Alamin, B.M.; Ibrahim, Y.; Salih, N.K.M. Effect of some vitamins and electrolytes on chickens infected with Infectious Bursal Disease. World Appl. Sci. J. 2009, 7, 366–370. [Google Scholar]
- King’ori, A.M. Review of the factors that influence egg fertility and hatchability in poultry. Int. J. Poult. Sci. 2011, 10, 483–484. [Google Scholar] [CrossRef]
- Davis, H.J.; Norris, L.C.; Heuser, G.F. The Rôle of Vitamin G in Reproduction in Poultry. Poult. Sci. 1938, 17, 81–86. [Google Scholar] [CrossRef]
- Schumacher, A.; Heuser, G. The Importance of Riboflavin in Reproduction in Poultry. Poult. Sci. 1939, 18, 369–374. [Google Scholar] [CrossRef]
- Abrams, V.A.; Han, C.-C.; White, H.B. Riboflavin-deficient chicken embryos: Hypoglycemia without dicarboxylic aciduria. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 1995, 111, 233–241. [Google Scholar] [CrossRef]
- Benore-Parsons, M. The Transport of Riboflavin-Binding Protein to the Hen Oocyte. Ph.D. Thesis, University of Delaware, Newark, DE, USA, 1986. [Google Scholar]
- Engel, R.; Phillips, P.; Halpin, J.G. The Effect of a Riboflavin Deficiency in the Hen upon Embryonic Development of the Chick. Poult. Sci. 1940, 19, 135–142. [Google Scholar] [CrossRef]
- Tuite, P.; Austic, R. Studies on a Possible Interaction between Riboflavin and Vitamin B12 as it Affects Hatchability of the Hen’s Egg. Poult. Sci. 1974, 53, 2125–2136. [Google Scholar] [CrossRef] [PubMed]
- Juriloff, D.; Roberts, C. Genetics of Cleft Palate in Chickens and the Relationship Between the Occurrence of the Trait and Maternal Riboflavin Deficiency. Poult. Sci. 1975, 54, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Anisha, M.; Karnani, S. Choudhary and Manju. Nutritional Factors: Affecting Egg Quality and Hatchability in Poultry. Pashu Sandesh. Available online: https://pashusandesh.com/Nutritional-Factors-Affecting-Egg-Quality- (accessed on 28 August 2023).
- Wilson, H.R. Effects of maternal nutrition on hatchability. Poult. Sci. 1997, 76, 134–143. [Google Scholar] [CrossRef]
- Folmes, C.D.L.; Terzic, A. Metabolic determinants of embryonic development and stem cell fate. Reprod. Fertil. Dev. 2015, 27, 82–88. [Google Scholar] [CrossRef]
- Givisiez, P.E.N.; Moreira Filho, A.L.B.; Santos, M.R.B.; Oliveira, H.B.; Ferket, P.R.; Oliveira, C.J.B.; Malheiros, R.D. Chicken embryo development: Metabolic and morphological basis for in ovo feeding technology. Poult. Sci. 2020, 99, 6774–6782. [Google Scholar] [CrossRef]
- Scanes, C.G.; Christensen, K.D. Fundamentals in Poultry Nutrition. In Poultry Science; Waveland Press, Inc.: Long Grove, IL, USA, 2020; pp. 109–131. [Google Scholar]
- Loetscher, Y.; Hospenthal, A.R.; Kreuzer, M.; Messikommer, R.E. Do antioxidants induce feed selection behavior in laying hens? Preference for, and palatability of, different antioxidant sources. Eur. Poult. Sci. 2014, 78, 1–11. [Google Scholar] [CrossRef]
- Yigit, A.; Panda, A.; Cherian, G. The avian embryo and its antioxidant defence system. World’s Poult. Sci. J. 2014, 70, 563–574. [Google Scholar] [CrossRef]
- Vieira, S. Chicken embryo utilization of egg micronutrients. Braz. J. Poult. Sci. 2007, 9, 1–8. [Google Scholar] [CrossRef]
- McDowell, L.R. Riboflavin. In Vitamins in Animal and Human Nutrition; McDowell, L.R., Ed.; Iowa State University Press: Ames, IA, USA, 2000; pp. 331–346. [Google Scholar]
- Anonymous. Riboflavin No. 1170; Hoffmann-La Roche: Basel, Switzerland, 1969. [Google Scholar]
- Zhang, B.; Tang, J.; Wu, Y.; Cao, J.; Xing, G.; Sun, P.; Huang, W.; Xie, M.; Hou, S. Effects of riboflavin deficiency on the lipid metabolism of duck breeders and duck embryos. Poult. Sci. 2021, 100, 101342. [Google Scholar] [CrossRef]
- Onwudike, O.; Adegbola, A. Riboflavin requirement of laying hens for egg production and reproduction in the humid tropics. Trop. Agric. 1984, 61, 205–2007. [Google Scholar]
- Leeson, S.; Reinhart, B.S.; Summers, J.D. Response of white leghorn and rhode island red breeder hens to dietary deficiencies of synthetic vitamins. 2. embryo mortality and abnormalities. Can. J. Anim. Sci. 1979, 59, 569–575. [Google Scholar] [CrossRef]
- Ribeiro, M.; Bittencourt, L.; Hermes, R.; Rönnau, M.; Rorig, A.; Lima, F.; Fernandes, J. Mineral Source and Vitamin Level in Broiler Diets: Effects on Performance, Yield, and Meat Quality. Braz. J. Poult. Sci. 2020, 22, 1–14. [Google Scholar] [CrossRef]
- NASEM (National Academies of Sciences, Engineering, and Medicine). Nutrient Requirements of Poultry, 9th ed.; National Academy Press: Washington, DC, USA, 1994. [Google Scholar]
- GfE (Gesellschaft für Ernährungsphysiologie). Empfehlungen zur Energie- und Nährstoffversorgung von Legehennen und Masthühnern (Broiler); Gesellschaft für Ernährungsphysiologie, DLG-Verlag: Frankfurt, Germany, 1999. [Google Scholar]
- Rostagno, H.S. (Ed.) Brazilian Tables for Poultry and Swine. In Composition of Feedstuffs and Nutritional Requirements, 3rd ed.; Universidade Federal de Viçosa—Departamento de Zootecnia: Viçosa, Brazil, 2011. [Google Scholar]
- FEDNA (Fundacion Espanola Deserrollo Nutricion Animal). Necesidades Nutricionales para Avicultura, 2nd ed.; Normas FEDNA; Santoma, G., Mateos, G.G., Eds.; Fundacion Espanola para el Desarrollo Nutricion Animal: Madrid, Spain, 2018. [Google Scholar]
- Cobb 500 Broiler Performance & Nutrition Supplement. 2022. Available online: https://www.cobb-vantress.com/assets/Cobb-Files/product-guides/5502e86566/2022-Cobb500-Broiler-Performance-Nutrition-Supplement.pdf (accessed on 28 July 2023).
- ROSS Broiler: Nutrition Specifications. 2022. Available online: https://aviagen.com/eu/brands/ross/products/ross-308 (accessed on 28 July 2023).
- Nicholas and, B.U.T. Heavy Lines Feeding Guidelines. 2015. Available online: https://www.aviagenturkeys.com/uploads/2015/11/20/NU06%20Feeding%20Guidelines%20for%20Nicholas%20&%20BUT%20Heavy%20Lines%20EN.pdf (accessed on 28 July 2023).
- Hubbard Management Guide. 2006. Available online: https://www.hubbardbreeders.com/media/manual_broiler_management_en___013796700_1441_27062016.pdf (accessed on 28 July 2023).
- Hybrid Turkeys Vitamin and Trace Mineral Supplementation. 2016. Available online: https://www.hybridturkeys.com/en/resources/commercial-management/feed-and-water/vitamin-and-trace-mineral-supplementation/ (accessed on 28 July 2023).
- Lohmann LSL-Lite Management Guide. 2014. Available online: https://lohmann-breeders.com/media/strains/cage/management/LOHMANN-LSL-Lite-Cage-1.pdf (accessed on 25 August 2016).
- Hy-Line W-36 Commercial Layers. 2020. Available online: https://www.hyline.com/filesimages/Hy-Line-Products/Hy-Line-Product-PDFs/W-36/36%20COM%20ENG.pdf (accessed on 25 July 2023).
- H&N Management Guide. 2016. Available online: https://www.hngb.co.uk/uploadedresources/1652881742-use_this_one_-_brown_management_guide_feb_22__version_6.pdf (accessed on 23 February 2018).
- ISA Nutrition Management Guide. 2018. Available online: https://www.isa-poultry.com/en/product/isa-brown/ (accessed on 22 February 2018).
- Dekalb Nutrition Management Guide. 2017. Available online: https://www.dekalb-poultry.com/ (accessed on 22 February 2018).
- Babcock Nutrition Management Guide. 2017. Available online: https://www.babcock-poultry.com/en/news/laying-hens-management-guide-alternative-systems/ (accessed on 22 February 2018).
- Hisex Nutrition Management Guide. 2017. Available online: https://www.hisex.com/en/news/laying-hens-management-guide-alternative-systems/ (accessed on 22 February 2018).
- Poudel, S.; Zhang, L.; Tabler, G.T.; Lin, J.; Zhai, W. Effects of riboflavin and Bacillus subtilis on internal organ development and intestinal health of Ross 708 male broilers with or without coccidial challenge. Poult. Sci. 2020, 100, 100973. [Google Scholar] [CrossRef]
- Leeson, S.; Summers, J.D. Commercial Poultry Nutrition. In Scott’s Nutrition of the Chicken, 3rd ed.; Leeson, S., Ed.; Nottingham University Press: Nottingham, UK, 2001; p. 398. [Google Scholar]
- Motyl, K.J.; Guntur, A.R.; Carvalho, A.L.; Rosen, C.J. Energy Metabolism of Bone. Toxicol. Pathol. 2017, 45, 887–893. [Google Scholar] [CrossRef]
- Costello, L.C.; Franklin, R.B.; Reynolds, M.A.; Chellaiah, M. The Important Role of Osteoblasts and Citrate Production in Bone Formation: “Osteoblast Citration” as a New Concept for an Old Relationship. Open Bone J. 2012, 4, 27–34. [Google Scholar] [CrossRef]
- Witten, S.; Aulrich, K. Exemplary calculations of native thiamine (vitamin B1) and riboflavin (vitamin B2) contents in common cereal-based diets for monogastric animals. Org. Agric. 2018, 9, 155–164. [Google Scholar] [CrossRef]
- Burgos, S.; Bohorquez, D.V.; Burgos, S.A. Vitamin Deficiency-Induced Neurological Diseases of Poultry. Int. J. Poult. Sci. 2006, 5, 804–807. [Google Scholar]
- Van Limbergen, T.; Ronsmans, S.; Maes, D.; Van Erum, J.; Van Ginderachter, J.; Verlinden, M.; Boel, F.; Garmyn, A.; Dewulf, J.; Ducatelle, R. A case of clubbed down syndrome in broilers. Avian Pathol. 2020, 50, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Hanna, M.; Jaqua, E.; Nguyen, V.; Clay, J. B Vitamins: Functions and Uses in Medicine. Perm. J. 2022, 26, 89–97. [Google Scholar] [CrossRef]
- Pedrolli, D.B.; Jankowitsch, F.; Schwarz, J.; Langer, S.; Nakanishi, S.; Frei, E.; Mack, M. Riboflavin Analogs as Antiinfectives: Occurrence, Mode of Action, Metabolism and Resistance. Curr. Pharm. Des. 2013, 19, 2552–2560. [Google Scholar] [CrossRef] [PubMed]
- Hemasa, A.L.; Mack, M.; Saliba, K.J. Roseoflavin, a Natural Riboflavin Analogue, Possesses In Vitro and In Vivo Antiplasmodial Activity. Antimicrob. Agents Chemother. 2022, 66, e0054022. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.W.; Brown, J.E.; Ashton, W.T.; Brown, R.D.; Rogers, E.F. Anticoccidial riboflavine antagonists. Cell. Mol. Life Sci. 1977, 33, 1274–1276. [Google Scholar] [CrossRef]
- Mansjö, M.; Johansson, J. The Riboflavin analog Roseoflavin targets an FMN-riboswitch and blocks Listeria monocytogenes growth, but also stimulates virulence gene-expression and infection. RNA Biol. 2011, 8, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Farah, N.; Chin, V.K.; Chong, P.P.; Lim, W.F.; Lim, C.W.; Basir, R.; Chang, S.K.; Lee, T.Y. Riboflavin as a promising antimicrobial agent? A multi-perspective review. Curr. Res. Microb. Sci. 2022, 3, 100111. [Google Scholar] [CrossRef]
- Moss, A.F.; Chrystal, P.V.; Cadogan, D.J.; Wilkinson, S.J.; Crowley, T.M.; Choct, M. Precision feeding and precision nutrition: A paradigm shift in broiler feed formulation? Anim. Biosci. 2021, 34, 354–362. [Google Scholar] [CrossRef]
- Patel, H.; Sana, A. Role of Computer Science (Artificial Intelligence) In Poultry Management. Devot. J. Res. Community Serv. 2022, 3, 2068–2088. [Google Scholar] [CrossRef]
- Mohamadi, M.A.; Farshi, P.; Ahmadi, P.; Ahmadi, A.; Yousefi, M.; Ghorbani, M.; Hosseini, S.M. Encapsulation of Vitamins Using Nanoliposome: Recent Advances and Perspectives. Adv. Pharm. Bull. 2023, 13, 48–68. [Google Scholar] [CrossRef]
- Date, A.A.; Hanes, J.; Ensign, L.M. Nanoparticles for oral delivery: Design, evaluation and state-of-the-art. J. Control. Release 2016, 240, 504–526. [Google Scholar] [CrossRef]
- Hirakawa, R.; Nurjanah, S.; Furukawa, K.; Murai, A.; Kikusato, M.; Nochi, T.; Toyomizu, M. Heat Stress Causes Immune Abnormalities via Massive Damage to Effect Proliferation and Differentiation of Lymphocytes in Broiler Chickens. Front. Vet. Sci. 2020, 7, 46. [Google Scholar] [CrossRef]
- Akinyemi, F.; Adewole, D. Environmental Stress in Chickens and the Potential Effectiveness of Dietary Vitamin Supplementation. Front. Anim. Sci. 2021, 2, 1–21. [Google Scholar] [CrossRef]
- Mosegaard, S.; Dipace, G.; Bross, P.; Carlsen, J.; Gregersen, N.; Olsen, R.K.J. Riboflavin Deficiency—Implications for General Human Health and Inborn Errors of Metabolism. Int. J. Mol. Sci. 2020, 21, 3847. [Google Scholar] [CrossRef] [PubMed]

| Function | Description | Reference |
|---|---|---|
| Redox reactions and energy production | Riboflavin is essential for producing energy via two key coenzymes, flavin mononucleotide (FMN) and flavin adenine dinucleotide (FAD). | [28,29] |
| Antioxidant capacity | Riboflavin possesses indirect antioxidant properties, aiding in the neutralization of harmful free radicals within the body. | [11,30,31] |
| Metabolism of fats, drugs, and steroids | Riboflavin participates in the enzymatic reactions associated with the metabolism of lipids, xenobiotic substances, and steroid compounds. | [32] |
| Cellular function, growth, and development | Riboflavin plays a fundamental role in the regulation of cellular functions, growth, and developmental processes. | [26] |
| Reproductive functions | Riboflavin is essential for the reproductive performance of poultry. It affects fertility, embryonic development, and hatchability. | [11,26] |
| Nerve function | Riboflavin deficiency has been associated with peripheral nerve demyelination in poultry, resulting in symptoms such as leg weakness and curled toe paralysis. | [5,33] |
| Poultry Species | Dietary Supplemental Riboflavin Levels in Feed | Effects of Riboflavin Supplementation on Performance and Leg Abnormalities | Reference |
|---|---|---|---|
| Broiler chicken | 0.0, 0.9, 2.0, 2.8, 3.6, 4.4 mg/kg | Improved body weight, feed intake, FCR, and reduced occurrence of leg paralysis | [60] |
| Broiler chicken | 2.75, 2.78, 3.05, 3.40, 3.71 mg/kg | Improved body weight, feed intake, FCR, and reduced occurrence of curled-toe paralysis | [98] |
| Turkey poults | 0.0, 0.6, 1.1, 1.7, 3.1, 4.4 mg/kg | Improved body weight, feed intake, FCR, and reduced occurrence of leg paralysis | [61] |
| Broiler chicken | 1.7, 3.7, 11.7 mg/kg | Bilateral leg weakness and rotation of the metatarsus with flexion of the digits and hock lesions in “the 1.7 mg/kg group” as well as leg weakness in “the 3.7 mg/kg group” compared to the “11.7 mg/kg group”. | [5] |
| Broiler chicken | 1.0–5.0 mg/kg | Improved body weight and FCR | [25] |
| Broiler chicken | 4.0-10.4 mg/kg | Improved daily weight gain, FCR, and European broiler index 1 | [106] |
| Pekin ducks | 0.0 and 10.0 mg/kg | Lower mortality, improved average daily gain, feed intake, and gain/feed ratio | [116] |
| Broiler chicken | 0.0, 1.0, 2.0, 3.0, 4.0, 8.0 mg/kg | Lower mortality, improved body weight, feed intake, FCR, and reduced occurrence of leg paralysis | [24] |
| Broiler chicken | 0.0, 0.2, 0.5, 0.9, 4.5 mg/kg | Lower mortality, improved body weight, and reduced occurrence of leg paralysis | [21] |
| Broiler chicken | 0.0, 9.0 mg/kg | Improved weight gain and FCR and reduced occurrence of leg paralysis | [117] |
| Bobwhite quail | 0.0, 0.8, 1.5, 2.5, 3.5, 5.0 mg/kg | Lower mortality, improved body weight and FCR | [22] |
| Ringnecked pheasants | 0.0, 0.4, 0.9, 1.3, 1.8, 2.4 mg/kg | Improved weight gain and reduced occurrence of leg abnormalities | [20] |
| Broiler chicken | 0.8, 6.6, 20.0 mg/kg | Improved FCR | [29] |
| Laying hen | 0.0 and 2.9 mg/kg | Improved egg weight | [56] |
| Broiler breeders | 2.5 and 4.0 mg/kg | No effect | [7] |
| Broiler chicken | 2.5 and 4.0 mg/kg | Improved growth rate and feed consumption | [7] |
| Turkey poults | 0.0, 2.0, 4.0 or 8.0 mg/kg | Higher body weight | [23] |
| Laying hen | 1.55, 2.20, 4.40, and 8.80 mg/kg | Improved egg production and egg weight | [118] |
| Poultry Species | Dietary Supplemental Riboflavin Levels in Feed | Effects of Riboflavin Supplementation on Reproductive Performance and Hatchability | Reference |
|---|---|---|---|
| Laying hen | 0.0 and 2.9 mg/kg | Improved hatchability | [56] |
| Laying hen | 1.55, 2.20, 4.40, and 8.80 mg/kg | Improved egg production, egg weight, hatchability, and hen weight as well as reduced incidence of hemorrhagic embryos and clubbed down | [118] |
| Duck breeder | 0.0 and 10.0 mg/kg | Improved hatchability | [12] |
| Duck breeder | 0.0 and 16.5 mg/kg | Improved hatchability and embryo weight | [145] |
| Broiler breeders | 2.5 and 4.0 mg/kg | No effect | [7] |
| Duck breeder | 0, 2.5, 5, 10, and 15 mg/kg | Improved hatchability | [11] |
| Laying hen | 0.9–8.1 mg/kg | Improved egg production and hatchability | [146] |
| White leghorn and Rhode island red breeder hens | 1.0 and 2.5 mg/kg | Reduced embryo mortality and number of malpositioned embryos | [147] |
| Source | Vitamin A Requirement, mg/kg Feed | |||
|---|---|---|---|---|
| Broilers | Laying Hens | Broiler Breeders | Turkeys | |
| NASEM [149] | 3.6 | 2.1 4 | n/a | 2.5–4.0 |
| GfE [150] | 2.9 | 2.5 | 2.5 | n/a |
| Brazilian tables [151] | 4.7–9.1 | 4.8 4 | 8.0 | n/a |
| FEDNA [152] | 3.1–6.5 | 4.0 | 7.0 | 5.5–10.0 |
| Cobb [153] | 6.0–9.0 | n/a | 13.0 | n/a |
| Aviagen [154,155] 1 | 7.0–9.0 | n/a | 10.0–16.0 | 4.0–10.0 |
| Hubbard [156] | 6.0–8.0 | n/a | 12 | n/a |
| Hendrix (Hybrid turkeys) [157] | n/a | n/a | n/a | 8.0–15.0 |
| EW Group (laying hens) 2 [158,159,160] | n/a | 4.0–6.6 | n/a | n/a |
| Hendrix (laying hens) 3 [161,162,163,164] | n/a | 5.0 | n/a | n/a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shastak, Y.; Pelletier, W. From Metabolism to Vitality: Uncovering Riboflavin’s Importance in Poultry Nutrition. Animals 2023, 13, 3554. https://doi.org/10.3390/ani13223554
Shastak Y, Pelletier W. From Metabolism to Vitality: Uncovering Riboflavin’s Importance in Poultry Nutrition. Animals. 2023; 13(22):3554. https://doi.org/10.3390/ani13223554
Chicago/Turabian StyleShastak, Yauheni, and Wolf Pelletier. 2023. "From Metabolism to Vitality: Uncovering Riboflavin’s Importance in Poultry Nutrition" Animals 13, no. 22: 3554. https://doi.org/10.3390/ani13223554
APA StyleShastak, Y., & Pelletier, W. (2023). From Metabolism to Vitality: Uncovering Riboflavin’s Importance in Poultry Nutrition. Animals, 13(22), 3554. https://doi.org/10.3390/ani13223554





