Abstract
Comprehensive epidemiological reports on the incidence and pathogenicity of Yersinia spp., specifically Yersinia pseudotuberculosis and Yersinia enterocolitica, in rhesus macaques (Macaca mulatta) are not prevalent. Here we report findings from a retrospective analysis of microbial culture results, necropsy reports, and histology records collected over 24 years (2000–2024) in a large captive rhesus macaque colony at the Oregon National Primate Research Center (ONPRC). Findings are compared between animals infected with either Y. pseudotuberculosis or Y. enterocolitica to evaluate the prevalence of infection within the population and determine whether Y. pseudotuberculosis is more likely to be associated with systemic disease and higher mortality than Y. enterocolitica. Among 101 affected animals, Y. pseudotuberculosis was the predominant species (75.2%), followed by Y. enterocolitica (22.7%) and Y. kristensenii (2.0%). Overall mortality among animals with confirmed yersiniosis was 25.3%, with comparable mortality rates for Y. pseudotuberculosis (23.7%) and Y. enterocolitica (30.4%) infections. Y. pseudotuberculosis was most frequently associated with systemic illness, spontaneous death, or significant clinical decline. Overall, these findings emphasize the importance of continued surveillance and targeted management strategies to mitigate the impact of Yersinia infections in captive nonhuman primate colonies.
1. Introduction
Yersinia enterocolitica and Yersinia pseudotuberculosis are Gram-negative, facultatively anaerobic coccobacilli in the family Enterobacteriaceae. Of the 26 species of Yersinia, enterocolitica and pseudotuberculosis are commonly recognized zoonotic pathogens capable of causing gastrointestinal and systemic disease in a variety of species, including nonhuman primates (NHPs) and humans [1,2]. In both human and NHP populations, these enteropathogenic Yersiniae are commonly identified causative agents of yersiniosis [3,4]. Old and New World monkey species, including squirrel monkeys (Samiri spp.), marmosets (Callithrix spp.), brown-headed spider monkeys (Ateles fusciceps), owl monkeys (Aotus spp.), patas monkeys (Erythrocebus patas), vervets (Chlorocebus spp.), mangabeys (Cercocebus spp.), tamarins (Saguinus spp.), and macaques (Macaca fascicularis and Macaca mulatta), are reported to be sensitive to Yersinia infection and express variable morbidity and mortality [5,6,7].
Notable similarities and differences exist between humans and NHPs with yersiniosis. In humans, Y. pseudotuberculosis infections are less common than Y. enterocolitica but tend to cause more mesenteric lymphadenitis and appendicitis-like presentations, sometimes progressing to systemic disease, notably in vulnerable populations such as children or those with underlying conditions. Approximately 75% of patients infected with Y. pseudotuberculosis are between the ages of five and 15 years [8]. Y. pseudotuberculosis is considered the evolutionary ancestor of Y. pestis, highlighting its historic relevance as a zoonotic agent [9]. In contrast, Y. enterocolitica is more frequently associated with gastroenteritis, including diarrhea, abdominal pain, and fever, and it more often leads to pseudoappendicitis. Transmission typically occurs after consumption of contaminated food, with pork being a common source [10,11]. Certain Y. enterocolitica serotypes (O:3, O:9, O:8, and O:5,27) are most commonly associated with severe gastrointestinal disease [12]. Sepsis in infants and immunocompromised patients is associated with high mortality rates [11]. While both pathogens can cause extraintestinal complications like reactive arthritis or erythema nodosum in humans, Y. pseudotuberculosis is more commonly linked to systemic involvement, while Y. enterocolitica is notable for its higher incidence, prolonged fecal shedding, and the potential for severe outcomes, such as sepsis, in immunocompromised individuals [8,11].
In captive NHPs, including rhesus macaques (Macaca mulatta), both species of enteropathogenic Yersinia have been described as causing comparable clinical symptoms [13,14]. Infections with Y. pseudotuberculosis and Y. enterocolitica are often associated with enterocolitis and manifest clinically as self-limiting diarrhea (with or without blood), dehydration, anorexia, depression, and weight loss. Less frequently, these pathogens can cause systemic infection and septicemia, which carry significant morbidity and mortality [15,16]. In these cases, gross lesions may include ulcerative, necrotizing enterocolitis, with discrete multifocal mucosal ulcers within the small intestine and colon, and enlarged, edematous mesenteric lymph nodes. Bacterial dissemination to extraintestinal organs, especially the liver and spleen, may occur [6]. The likelihood of disease expression may be influenced by predisposing factors such as physiological stress, immunosuppression, and concurrent infections. Coinfections with other enteropathogens (e.g., Campylobacter spp., Shigella spp., intestinal parasites) may exacerbate clinical disease [7,17]. While both organisms produce similar clinical conditions, Y. enterocolitica tends to remain localized to the gastrointestinal tract, while Y. pseudotuberculosis is more frequently associated with extraintestinal dissemination [7,18]. Reproductive effects, including abortion and stillbirths, secondary to Y. pseudotuberculosis infection have also been described [13].
Diagnosis of yersiniosis typically relies on a combination of clinical signs, microbiological culture, and histopathologic evaluation. Culture obtained from feces, intestinal tissue, blood, or mesenteric lymph nodes remains the gold standard. Yersinia spp. grow on blood, chocolate, MacConkey, and other laboratory media between 4 °C and 43 °C. The slow-growing nature of Yersinia spp. can present challenges, especially when there is competition with commensal flora [6,15]. Leukocytosis secondary to neutrophilia, hyponatremia, hypochloremia, prerenal azotemia, and moderate hyperfibrinogenemia have been reported [19].
Transmission typically occurs via the fecal-oral route. Natural reservoirs include wild rodents, birds, and contaminated water sources [14]. Asymptomatic carriage and intermittent shedding in NHP colonies are suspected but poorly characterized, complicating eradication and surveillance strategies [13,20]. Subclinical carriers may serve as reservoirs of infection, intermittently introducing Yersinia spp. into social groups without overt clinical signs. This has been documented in other enteric pathogens of NHPs, where stress, dietary changes, or concurrent infections can trigger bacterial shedding and facilitate transmission [13,21,22]. Environmental persistence also plays a role in outbreaks, as both Y. pseudotuberculosis and Y. enterocolitica are psychrotrophic organisms with a known resistance to cold temperatures and the ability to survive for prolonged periods in soil and water [8,23]. Clinical disease is most commonly observed in outdoor-housed NHPs during cooler months, particularly winter and early spring [18].
The significance of these pathogens has been well documented in veterinary and public health contexts [24,25,26,27], and it is widely acknowledged that, in NHP facilities, outbreaks can compromise research programs, animal welfare, and breeding efforts, while posing zoonotic risks to animal care staff and researchers [4,16,26,27]. Despite this importance, comprehensive epidemiological data characterizing the incidence, clinical presentation, and pathogenic outcomes of yersiniosis in captive rhesus macaques remain limited, leaving critical gaps in understanding of disease dynamics and control in NHP populations.
In this study, we retrospectively analyze 24 years of microbiological, clinical, and histopathology data from the Oregon National Primate Research Center (ONPRC) to better understand the epidemiology and pathogenic variation in Yersinia infections in a large colony of rhesus macaques. Specifically, we aim to compare the frequency and clinical severity of infections caused by Y. pseudotuberculosis versus Y. enterocolitica, with particular attention to differences in clinical signs, necropsy findings, and histopathologic outcomes. This work seeks to enhance our understanding of the prevalence and severity of these important pathogens in captive primate populations.
2. Materials and Methods
All animals from this study were housed at the ONPRC, an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALACi) accredited facility. This was a retrospective study utilizing clinical and diagnostic records generated at the ONPRC. No animals were experimentally infected for the purposes of this study. All colony management practices were overseen by the ONPRC Institutional Animal Care and Use Committee (IACUC).
Microbial culture results, animal demographic and clinical information, and necropsy records were obtained from the ONPRC electronic health record system, Primate Records and Information Management (PRIMe). The PRIMe database is a centralized, searchable electronic system that contains clinical exam notes, laboratory test results, diagnostic imaging, housing assignments, and necropsy reports. Data is entered by veterinary staff and pathologists and undergoes periodic quality review. Health and colony management records for 21,510 rhesus macaques within the designated study timeframe (January 2000 through December 2024) were available for review. The colony included animals of both sexes and a broad age distribution, maintained in a variety of housing environments including indoor, outdoor, and indoor/outdoor facilities. Age categories were defined as neonatal, (0–28 days), infant (29–364 days), juvenile (1 year to 3 years), adult (4 years to 17 years), and geriatric (18 years or older). Animals were socially housed whenever possible, and husbandry practices followed standard ONPRC protocols for primate care, feeding, and sanitation.
To identify cases of Yersinia spp. infection, microbiology records in PRIMe were queried for all cultures collected between 2000 and 2024 that were positive for Yersinia. Available results included cultures positive for Y. enterocolitica, Y. pseudotuberculosis, and Y. kristensenii. Culture results from animals treated clinically were obtained from fresh fecal swabs. Cultures collected at time of necropsy included gastrointestinal contents, and sectioned surfaces of lymph nodes, and other viscera. Each culture had been submitted to the ONPRC Clinical Pathology Laboratory as part of routine veterinary diagnostics performed on live, clinically healthy and ill animals, or necropsy evaluations. Cultures were incubated on MacConkey agar at room temperature for 24–48 h to limit overgrowth by non-target bacteria. Subcultures of all non-lactose fermenting colonies, identified within 48 h, were incubated on blood agar plates at 37 °F for 24 h. Finally, a urease tube was inoculated with the pure culture using aseptic technique. Identification of Yersinia species was performed on colonies appearing within 4–24 h, using the API® 20E bacterial identification system, developed by BioMerieux (Marcy-l’Étoile, France). Due to the retrospective nature of this study, the use of molecular techniques to further characterize the isolated Yersinia strains was not available. Duplicate culture results from the same specimen were excluded, and when multiple positive cultures were identified for a single animal, all were recorded and linked under the unique animal identifier.
For each animal with a positive culture, associated clinical records were reviewed to extract demographic data (sex, age, housing type, season at time of microbial culture), presenting clinical signs, diagnostic procedures, and treatment information when available. Necropsy reports were also reviewed for gross and histologic findings consistent with Yersinia infection. All pathology reports were authored or reviewed by diplomates of the American College of Veterinary Pathologists. Full necropsies and standard tissue collection of major organs for each animal had been performed. Tissues were preserved in 10% neutral buffered formalin and routinely processed into paraffin, sectioned at 4–6 µm, and stained with hematoxylin and eosin (H&E).
Differences in mortality, rate of spontaneous death (animals found deceased without prior clinical signs), and incidence of co-infection between rhesus macaques infected with Y. pseudotuberculosis and Y. enterocolitica were assessed using two-sided Fisher’s exact test. Relative risk (RR) and odds ratio (OR) with 95% confidence intervals (CI) were calculated. p < 0.05 was considered statistically significant. All analyses were performed using the GraphPad QuickCalcs online contingency table calculator (GraphPad Software, La Jolla, CA, USA; https://www.graphpad.com, accessed on 9 December 2025).
3. Results
Between the years 2000 and 2024, there were 113 positive samples for Yersinia spp. from 101 individual rhesus macaques at the ONPRC. Samples were either collected upon presentation to the colony clinic or at necropsy. Of these 101 animals, 76 (75.2%) animals cultured positive for Y. pseudotuberculosis and 23 (22.7%) for Y. enterocolitica. Two animals cultured positive for Y. kristensenii (1.98%).
All positive culture results originated from animals housed in outdoor or indoor/outdoor facilities. There were seven non-consecutive years that no Yersinia spp. were cultured in rhesus macaques at the ONPRC. This included 2003–2006, 2010, 2014, and 2018. Totals for all other years ranged from one case to 24 cases per year. Most cases occurred in the winter months, defined as December through February (n = 58, 57.4%), followed by the spring months, defined as March through May (n = 33, 32.7%). Fewer cases were detected in the summer months, defined as June through August (n = 1, 0.99%), and the fall months, defined as September through November (n = 9, 8.9%). Mortality rates were highest in the winter (n = 14, 13.9%) with Y. pseudotuberculosis cases presenting to necropsy 10 times and Y. enterocolitica cases presenting to necropsy 4 times throughout the winter months. Clinical cases (those animals treated for infection and successfully released from care) presenting with Y. pseudotuberculosis occurred most commonly during the winter (n = 35, 34.7%), while clinical cases presenting with Y. enterocolitica occurred more frequently during the spring months (n = 9, 8.9%). A summary of all culture results organized by year can be found in Table A2 of Appendix B.
Clinical Presentation:
During this 24-year period, Yersinia spp. were isolated from 76 animals that were treated in the ONPRC colony clinic and successfully released from care. Animals that did not survive are addressed in the following section. Yersinia pseudotuberculosis was isolated from 58 (76.3%) of these animals, while Y. enterocolitica was isolated from 16 (21.1%). Y. kristensenii was isolated from two animals (2.6%). Due to its low prevalence and absence of clinical significance, limited attention was given to Y. kristensenii during this study, and it was excluded from all statistical analyses. Of these rhesus macaques, 43 (56.6%) were females, and 33 were males (43.4%), ranging in age from 13 days to 16 years. The age categories of those affected included neonatal (n = 1), infant (n = 37), juvenile, 1 year to 3 years (n = 30), and adult (n = 8). No geriatric animals were identified in the group that was treated in the ONPRC colony clinic and successfully released from clinical care. Table 1 summarizes the number of animals, presenting either for clinical treatment or necropsy, that cultured positive for Y. pseudotuberculosis, Y. enterocolitica, or Y. kristensenii, according to age, sex, and season at time of microbial culture. Table 2 notes the clinical signs identified at initial presentation in order of frequency of occurrence for animals infected with Y. pseudotuberculosis, Y. enterocolitica, or Y. kristensenii and successfully released from clinical care. The most common findings included diarrhea, dehydration (determined by decrease in skin turgor, dry/tacky mucous membranes and/or sunken eyes), and azotemia.

Table 1.
Demographics of Rhesus Macaques Culturing Positive for Yersinia spp. (Clinical vs. Necropsy Cases).

Table 2.
Common Clinical Presentations for Rhesus Macaques Treated and Released from the Colony Hospital.
Necropsy Findings:
Yersinia spp. were isolated at time of necropsy in 25 animals between the years 2000 and 2024. Y. pseudotuberculosis was isolated from 18 (72.0%) of these cases, while Y. enterocolitica was isolated from 7 (28.0%). Of the cases submitted for necropsy, 17 were females (68.0%), and 8 were males (32.0%), ranging in age from 1 day to 19 years. The age groupings included neonatal (n = 1), infant (n = 12), juvenile (n = 9), adult (n = 2), and geriatric (n = 1). Table 1 contains a summary of animals in each group (culture positive for Y. pseudotuberculosis vs. Y. enterocolitica) according to age, sex, and seasonality at time of microbial culture and necropsy.
Half of the necropsied animals culturing positive for Y. pseudotuberculosis were submitted following spontaneous death. An additional 5% succumbed to the disease within 48 h of clinical presentation, despite supportive and therapeutic care. Co-organisms, including Campylobacter coli, Campylobacter jejuni, Trichuris sp., flagellates, and Balantidium coli were identified in 50% (n = 9) of these cases. 14% of the cases that cultured positive for Y. enterocolitica at time of necropsy had been found deceased. All other animals infected with Y. enterocolitica presented to the clinic with diarrhea prior to necropsy. Two of the clinical cases (33%) died spontaneously following initial clinical care. Co-organisms were identified following necropsy in 85% of Y. enterocolitica cases, including Campylobacter coli (71%), Campylobacter jejuni, and Shigella flexneri.
Systems and organs affected:
Of those cases presented to necropsy, Y. pseudotuberculosis was isolated on microbial culture in the small intestine (n = 7, 38.9%), colon (n = 8, 44.4%), liver (n = 4, 22.2%), blood (n = 2, 11.1%), feces (n = 2, 11.1%), mesenteric lymph nodes (n = 1, 5.6%), and lung (n = 1, 5.6%). Y. enterocolitica was isolated on microbial culture in the small intestine (n = 2, 28.6%), colon (n = 4, 57.1%), and feces (n = 2, 28.6%). Grossly, lesions varied from extensive involvement of multiple organs to minimal lesions in the small or large intestine. In the gastrointestinal tract, mucosal hyperemia, edema and multifocal erosions and ulcerations were observed (Figure 1A,B). Hepatic involvement was usually manifested as numerous pale foci (Figure 1C).
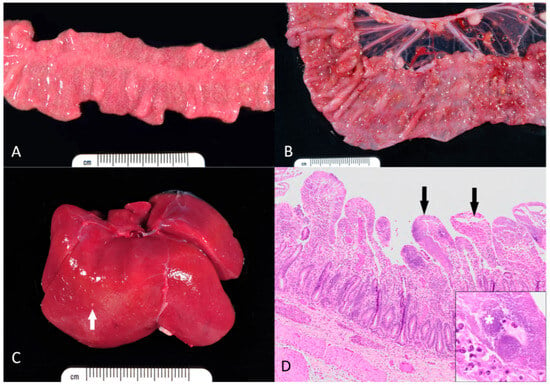
Figure 1.
Gross and microscopic appearance of yersiniosis in rhesus macaques. (A) Small intestine. The mucosa is diffusely edematous and contains myriads of pale necrotic and hemorrhagic foci. (B) Colon. Within the mucosa, there are numerous necroulcerative foci covered with tan fibrin and rimmed by hemorrhage. (C) Liver. Numerous pale microabscesses (white arrow) are scattered throughout the hepatic parenchyma. (D) Small intestine. The lamina propria of most villi is effaced by large aggregates of coccobacilli, neutrophils, and necrotic debris (black arrows). Inset: Dense colonies of Yersinia bacilli (asterisk). Hematoxylin and eosin.
Histologically, coccobacilli (Y. pseudotuberculosis based on culture results) were noted in the small intestine (n = 12, 66.7%), colon (n = 10, 55.6%), liver (n = 3, 16.7%), stomach (n = 2, 11.1%), spleen (n = 1, 5.6%), and mesenteric lymph nodes (n = 1, 5.6%). Histologically, coccobacilli (Y. enterocolitica based on culture results) were noted in the colon (n = 6, 85.7%) and small intestine (n = 4, 57.1%). Common histopathological findings included large foci of mucosal necrosis and local infiltration of inflammatory cells, especially neutrophils, forming abscesses in the lamina propria, often near Peyer’s patches. In the intestines, colonies of Gram-negative coccobacilli were characteristic (Figure 1C). Additional findings included multifocal, acute and necrotizing hepatitis, splenitis, and lymphadenitis Figure 1D. A summary of all histologic findings, in order by frequency of occurrence, can be found in Table A1 of Appendix A.
Statistical Analysis:
Mortality occurred in 25 of 99 cases with confirmed yersiniosis (25.3%). The mortality rate was 23.7% (18/76) for those with Y. pseudotuberculosis infections and 30.4% (7/23) for those with Y. enterocolitica infections. Patients with Y. enterocolitica had a 28% higher risk of death compared with those infected by Y. pseudotuberculosis (RR 1.28, 95% CI 0.60–2.73). By two-sided Fisher’s exact test, the difference in mortality between the two species was not statistically significant (p = 0.589). The corresponding odds ratio was 1.41 (95% CI 0.53–3.76).
Among the cases presented for necropsy, spontaneous death occurred in 10 animals. The spontaneous death rate was higher in animals with Y. pseudotuberculosis than Y. enterocolitica (50.0% [9/18] vs. 14.3% [1/7]). Rhesus macaques with Y. enterocolitica had a 78% lower risk of spontaneous death compared with those infected with Y. pseudotuberculosis (RR 0.22, 95% CI 0.03–1.49). By two-sided Fisher’s exact test, this difference was not statistically significant (p = 0.204). The corresponding odds ratio was 0.17 (95% CI 0.02–1.54).
Co-infection with other pathogens was documented in 15 of the 25 cases presented for necropsy (60.0%). The frequency of co-infection was higher in animals with Y. enterocolitica than Y. pseudotuberculosis (85.7% [6/7] vs. 50.0% [9/18]). Cases with Y. enterocolitica had a 71% higher risk of co-infection compared with those infected with Y. pseudotuberculosis (RR 1.71, 95% CI 0.99–2.98). By two-sided Fisher’s exact test, this difference was not statistically significant (p = 0.179). The corresponding odds ratio was 6.00 (95% CI 0.60–60.38).
4. Discussion
We identified 101 cases of culture-positive yersiniosis in rhesus macaques at the ONPRC between 2000 and 2024. Of these 101 animals, Yersinia pseudotuberculosis accounted for the majority (n = 76, 75.2%), while Y. enterocolitica was less common (n = 23, 22.8%). Based on previous data identifying a higher prevalence of Y. pseudotuberculosis versus Y. enterocolitica in our rhesus macaque colony, we suspected that morbidity and mortality would be higher in those animals infected with Y. pseudotuberculosis versus Y. enterocolitica. However, despite its higher prevalence, Y. pseudotuberculosis infections resulted in a slightly lower mortality rate than Y. enterocolitica infections (23.7% [18/76] vs. 30.4% [7/23]; p = 0.589). The overall mortality among the 99 culture-positive cases was 25.3%.
Y. kristensenii is commonly categorized as a Y. enterocolitica-like species. While it has been recently studied due to its role in foodborne illness and zoonotic potential, it is widely considered non-pathogenic [28]. Because the two rhesus macaques in our dataset that cultured positive for Y. kristensenii presented with limited clinical signs and survived to release without complication, its implications in the ONPRC colony were not evaluated for this study. However, some new research has been performed to better characterize the genotypic and phenotypic virulence potential of Y. enterocolitica-like species (Y. kristensenii, Y. frederiksenii, and Y. intermedia). While typically regarded as commensal or opportunistic species, the possibility of strain-specific pathogenicity could not be excluded. Some analyses suggest that Y. kristensenii possesses several virulence-associated genes, though their functional expression and clinical relevance remain uncertain [29,30]. Continued monitoring and characterization of isolates will be important to determine whether Y. kristensenii plays a more meaningful role in the pathogenicity of yersiniosis in NHPs than previously appreciated.
In our population of rhesus macaques at the ONPRC, the clinical presentations of animals infected with Y. pseudotuberculosis or Y. enterocolitica included signs of chronic enteric disease (dehydration, diarrhea, weight loss). Azotemia was noted clinically, and renal tubular necrosis was identified on microscopic examination in most cases. These findings were attributed to diarrhea-related dehydration and renal hypoperfusion. Y. pseudotuberculosis was cultured outside of the GI tract, including the liver, blood, and mesenteric lymph nodes, indicating a higher likelihood of systemic lesions compared to Y. enterocolitica, which was only cultured from the small intestine and colon. Histologically, coccobacilli (Y. pseudotuberculosis based on culture) were identified multiple tissues, including the small intestine, colon, liver, stomach, spleen, and mesenteric lymph nodes, in contrast to Y. enterocolitica, which could only be identified histologically in the small intestine and colon. Spontaneous death (animals found deceased without prior clinical presentation) was uncommon overall but occurred more often in macaques infected with Y. pseudotuberculosis than Y. enterocolitica (50.0% vs. 14.3%). Although Y. enterocolitica infections showed a lower relative risk of spontaneous death (RR 0.22; 95% CI 0.03–1.49) and a lower odds ratio (OR 0.17; 95% CI 0.02–1.54), the finding was not statistically significant (p = 0.204). Overall, the data suggest a trend toward higher spontaneous mortality with Y. pseudotuberculosis, but the small sample size limits firm conclusions.
Our findings highlight important differences in disease patterns between the two primary Yersinia spp. Y. pseudotuberculosis was strongly associated with systemic infection: isolates were recovered from extra-intestinal sites including the liver, blood, and mesenteric lymph nodes, and histologic examination confirmed widespread dissemination of coccobacilli. These infections were frequently identified in animals found dead or succumbing rapidly after clinical presentation, supporting its role as a potential cause of spontaneous (sudden) death in rhesus macaques. In contrast, Y. enterocolitica was strictly isolated from the gastrointestinal tract and produced a more localized enteric disease pattern, often characterized by diarrhea, dehydration, and weight loss. Although Y. enterocolitica cases had a slightly higher mortality rate, this trend did not reach statistical significance. This could be due to the limited number of affected animals, but may also be multifactorial.
Coinfections, especially Campylobacter spp., were frequent, particularly in cases of Y. enterocolitica. These were identified in 85% of Y. enterocolitica-positive necropsy cases, compared with 50% of Y. pseudotuberculosis cases. However, the lack of statistical significance (p = 0.179) indicates that these findings should be interpreted cautiously, as the small number of Y. enterocolitica cases may inaccurately amplify perceived differences and prevent definitive conclusions. This disparity may suggest that Y. enterocolitica can act as an opportunistic pathogen or that gastrointestinal coinfections exacerbate disease severity, but further investigation with a larger study group is warranted [31,32,33,34]. It is also important to note that several of the enteric pathogens identified as co-infections are more commonly detected within the ONPRC colony than Yersinia spp. For example, over the study period, C. coli was isolated from 8232 rhesus macaque samples, and C. jejuni was isolated from 888 rhesus macaque samples. These background detection rates suggest that the presence of co-infection may reflect common microbial carriage rather than a direct causal relationship with disease severity.
To summarize, several conclusions can be drawn from the data gathered, each addressing different aspects of infection severity and prevalence associated with Y. pseudotuberculosis and Y. enterocolitica in rhesus macaques. When considering infection severity, we recognized that, while Y. pseudotuberculosis was isolated more commonly overall, a non-significant trend towards lower mortality was observed. Although not statistically significant, Y. pseudotuberculosis infections were associated with a higher proportion of animals dying spontaneously before exhibiting clinical signs, whereas Y. enterocolitica cases showed a trend toward higher overall mortality but tended to develop signs of illness that warranted clinical intervention. Multiorgan involvement may explain the trend towards higher rates of spontaneous death associated with Y. pseudotuberculosis infection [35]. Coinfections, particularly with Campylobacter spp., were more frequent in animals culturing positive for Y. enterocolitica than with Y. pseudotuberculosis. The trend towards higher mortality in Y. enterocolitica cases could be partly attributed to the combined pathogenic impact of mixed infections. This may imply that Y. enterocolitica is opportunistic or requires cofactors to cause severe disease [33]. The clinical and diagnostic implications of these findings suggest that Y. pseudotuberculosis may need to be considered in cases of unexplained sudden death or multisystemic signs. Additionally, animals with severe clinical signs associated with Y. enterocolitica infections might warrant investigation for coinfection, particularly when gastrointestinal signs are present.
The influence of age and immune maturity should also be considered. Infants and juveniles represented the largest portion of affected animals in this study. Young rhesus macaques may be particularly vulnerable due to immature immune defenses, social contact within age-matched juvenile groups, and increased exposure due to environmental exploration, all of which elevate the likelihood of exposure and clinical disease. While it is uncertain, due to the retrospective nature of this study, transmission from subclinical dams to their infants may have played a role in the increased rate of infection in the neonatal and infant age groups. Adults and geriatric animals were comparatively underrepresented, suggesting some degree of acquired immunity or reduced exposure risk [21,31,36].
The environmental and seasonal trends identified in this study are consistent with the psychrotrophic nature of Yersinia spp., which are capable of persisting in soil, water, and food sources at low temperatures [31,32]. Outdoor housing, where animals are exposed to fluctuating weather and potential wildlife reservoirs, appears to represent a key risk factor for transmission [37,38]. Specifically, the cold temperatures and heavy rainfall, common during Oregon winters, can lead to longer exposure to wet ground cover [39,40]. Seasonal peaks may also coincide with changes in NHP behavior, such as increased clustering for warmth, which can facilitate pathogen spread within social groups [36,41]. Additionally, rodent and avian populations, recognized reservoirs of Yersinia, often increase their interactions with outdoor enclosures during cooler months, further elevating exposure risk [21,31]. These observations highlight the need for continued disease surveillance that considers not only colony management practices but also local wildlife and environmental monitoring.
While our findings suggest that Y. pseudotuberculosis may be more prevalent and commonly associated with systemic disease in rhesus macaques compared to Y. enterocolitica, several limitations must be considered when interpreting these results. Isolating and culturing Yersinia spp. from clinical samples can be challenging. The phenotypic API20E method for species identification is not the most reliable system for differentiating between Yersinia spp., especially non-enterocolitica Yersinia [42,43] [Varettas and Mancini]. False-negative culture results may occur, especially in cases with a low bacterial load or bacterial loads that fluctuate due to intermittent shedding, when Yersinia is outcompeted by other microbial flora, or when samples are obtained after antibiotic administration [7,44]. Previous studies have demonstrated variable sensitivity of Yersinia cultures depending on methodology and timing of specimen collection [45,46].
Additionally, histopathologic evidence suggestive of Yersinia infection was identified in several cases in the absence of confirmatory microbial culture. These cases often included gross lesions consistent with Yersinia infection (e.g., mesenteric lymphadenitis, enterocolitis) and coccobacilli observed microscopically; however, definitive microbial identification was not obtained. The identification of histopathologic evidence suggestive of Yersinia infection in the absence of confirmatory microbial culture has several important consequences. First, it highlights the diagnostic limitations of relying solely on culture-based methods. Yersinia spp. can be difficult to isolate due to intermittent shedding, competition with commensal flora, or prior antimicrobial therapy, as previously mentioned [38,47]. As a result, some true cases of yersiniosis may have been excluded from our dataset, potentially skewing calculations. From a colony health management perspective, under-recognition of Yersinia infections could delay or complicate outbreak responses. Animals with suggestive clinical signs or lesions but no culture confirmation might not have been classified as cases, potentially obscuring patterns of transmission within the colony. This potential underestimation of disease prevalence could lead to an underappreciation of the pathogen’s role in morbidity, mortality, or reproductive loss.
Finally, the retrospective nature of this study introduces several important limitations that influence interpretation of the findings [48,49]. Because diagnostic approaches evolved over the 24-year study period, the accuracy in identifying Yersinia infections was likely inconsistent. For example, earlier records may have relied more heavily on gross pathology or culture under suboptimal conditions, while later years benefited from improved microbiological techniques and record-keeping. This variation creates the possibility of temporal bias, in which the apparent prevalence of yersiniosis reflects changes in diagnostic capability rather than true shifts in disease burden. Inconsistent sample collection must also be considered [48]. Some animals may have undergone full diagnostic workups, while others may have been sampled more selectively depending on clinical presentation or logistical constraints. This non-uniformity can lead to underdiagnosis in mildly affected or subclinical animals and overrepresentation of severe cases. In the future, the epidemiology and clinical significance of yersiniosis could be better characterized by utilizing data that have been gathered following standardized diagnostic criteria and uniform sampling protocols within the NHP colony [48,49].
5. Conclusions
This retrospective study offers insights into the epidemiology and pathogenicity of Yersinia infections in a captive rhesus macaque colony. Y. pseudotuberculosis was more frequently isolated and more often linked to systemic disease and spontaneous death, while Y. enterocolitica exhibited a slightly higher, although statistically non-significant, overall mortality rate and was more commonly associated with concurrent infections, particularly Campylobacter spp. These findings underscore the need for continued diagnostic surveillance and prospective research to better understand the role of Y. pseudotuberculosis in sudden, multisystemic disease and the contribution of coinfections as they pertain to the severity of Y. enterocolitica infections.
From a laboratory animal medicine perspective, these results emphasize the importance of ongoing diagnostic surveillance, especially in outdoor-housed primates during cooler seasons when Yersinia infections are more prevalent. The identification of culture-negative but histologically consistent cases also highlights the limitations of culture-based diagnostics. Molecular assays such as multiplex PCR have shown promise in improving detection rates and differentiating pathogenic strains and could be considered in the future depending on availability [38]. Beyond colony health, the findings have broader relevance to zoonotic risk assessment and comparative medicine. Many of the clinical signs reported in NHPs mirror patterns reported in human yersiniosis, supporting the rhesus macaque as a valuable model for understanding disease dynamics across species [7,35,50]. Finally, this study underscores the need for prospective research incorporating standardized diagnostic protocols, pathogen detection, and systematic evaluation of potential risk factors. Such approaches will be critical for refining disease control strategies, protecting colony health and research integrity, and clarifying the zoonotic significance of Yersinia spp. in NHP populations.
Author Contributions
S.H., A.D.L. and H.A.L.: Conceptualized and designed the study. S.H.: Wrote the original draft. A.D.L. and H.A.L.: Reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Institutes of Health (P51OD011092 to ONPRC). The funding sources had no involvement in study design, the collection, analysis and interpretation of data, or the writing of the report.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to thank Lois Colgin, Amanda Johnson, Rebecca Ducore, ONPRC Pathology Services Unit staff managed by Wendy Price, and the ONPRC Division of Comparative Medicine clinical veterinary staff for their contributions to the vast archive of pathology and clinical data used for this study.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ONPRC | Oregon National Primate Research Center |
| NHP | Nonhuman Primate |
Appendix A

Table A1.
Common Histologic Findings in Rhesus Macaques Submitted for Necropsy Following Infection with Y. pseudotuberculosis or Y. enterocolitica.
Table A1.
Common Histologic Findings in Rhesus Macaques Submitted for Necropsy Following Infection with Y. pseudotuberculosis or Y. enterocolitica.
| Organ/System | Histologic Findings | Organism | Total | |
|---|---|---|---|---|
| Y. pseudotuberculosis | Y. enterocolitica | |||
| Small Intestine | Inflammation | 10 | 4 | 14 |
| Neutrophilic infiltration | 7 | 2 | 9 | |
| GALT depletion | 3 | 1 | 4 | |
| GALT hyperplasia | 2 | 0 | 2 | |
| Crypt hyperplasia | 2 | 0 | 2 | |
| Amyloid | 2 | 6 | 8 | |
| Crypt abscess | 0 | 1 | 1 | |
| Colon | Inflammation | 13 | 7 | 20 |
| Neutrophilic infiltration | 8 | 2 | 10 | |
| Amyloid | 1 | 0 | 1 | |
| Crypt abscess | 0 | 2 | 2 | |
| Cecum | Inflammation | 10 | 7 | 17 |
| Kidney | Hyaline casts | 2 | 2 | 4 |
| Tubular necrosis | 12 | 4 | 16 | |
| Inflammatory infiltrate | 1 | 1 | 2 | |
| Amyloid | 1 | 0 | 1 | |
| Mesenteric LN | Inflammation | 4 | 0 | 4 |
| Lymph follicle depletion | 4 | 1 | 5 | |
| Lymphoid hyperplasia | 3 | 1 | 4 | |
| Fibrin deposition | 1 | 0 | 1 | |
| Amyloid | 1 | 1 | 2 | |
| Stomach | Inflammation | 5 | 1 | 6 |
| Neutrophilic infiltration | 2 | 0 | 2 | |
| Amyloid | 1 | 4 | 5 | |
| Thymus | Depletion | 10 | 3 | 12 |
| Amyloid | 1 | 0 | 1 | |
| Spleen | Inflammation | 2 | 0 | 2 |
| Lymph follicle depletion | 4 | 1 | 5 | |
| Amyloid | 1 | 4 | 5 | |
| Lymphoid follicle hyperplasia | 1 | 0 | 1 | |
| Liver | Inflammation | 6 | 0 | 6 |
| Micro abscess | 1 | 0 | 1 | |
| Inflammatory infiltrate, portal tract | 1 | 0 | 1 | |
| Amyloid | 2 | 1 | 3 | |
| Peripheral LN | Lymph follicle depletion | 4 | 2 | 6 |
| Hyperplasia | 1 | 1 | 2 | |
| Inflammation | 1 | 0 | 1 | |
| Amyloid | 1 | 1 | 2 | |
| Joint | Synovial inflammation | 3 | 1 | 4 |
| Lung | Neutrophilic infiltration | 2 | 0 | 2 |
| Edema, congestion | 2 | 0 | 2 | |
| Adrenal gland | Congestion, cortex | 2 | 0 | 2 |
| Amyloid | 1 | 0 | 1 | |
| Hemorrhage | 1 | 0 | 1 | |
| Cerebrum | Leptomeningeal edema | 3 | 0 | 3 |
| Heart | Amyloid | 1 | 0 | 1 |
| Inflammation | 1 | 0 | 1 | |
| Gallbladder & Bile Ducts | Inflammation | 1 | 0 | 1 |
Appendix B

Table A2.
Yersinia spp. Culture Results by Year (2000–2024).
Table A2.
Yersinia spp. Culture Results by Year (2000–2024).
| Necropsy Y. pseudotuberculosis | Necropsy Y. enterocolitica | Clinical Y. pseudotuberculosis | Clinical Y. enterocolitica | Clinical Y. kristensenii | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year | TOTAL | Winter | Spring | Summer | Fall | Winter | Spring | Summer | Fall | Winter | Spring | Summer | Fall | Winter | Spring | Summer | Fall | Spring |
| 2000 | 1 | 1 | ||||||||||||||||
| 2001 | 3 | 1 | 1 | 1 | ||||||||||||||
| 2002 | 4 | 2 | 1 | 1 | ||||||||||||||
| 2007 | 5 | 1 | 4 | |||||||||||||||
| 2009 | 4 | 1 | 3 | |||||||||||||||
| 2011 | 3 | 1 | 1 | 1 | ||||||||||||||
| 2012 | 3 | 2 | 1 | |||||||||||||||
| 2013 | 8 | 2 | 2 | 1 | 1 | 2 | ||||||||||||
| 2015 | 4 | 2 | 1 | 1 | ||||||||||||||
| 2016 | 2 | 1 | 1 | |||||||||||||||
| 2017 | 3 | 1 | 1 | 1 | ||||||||||||||
| 2019 | 3 | 1 | 1 | 1 | ||||||||||||||
| 2020 | 6 | 1 | 4 | 1 | ||||||||||||||
| 2021 | 16 | 1 | 9 | 5 | 1 | |||||||||||||
| 2022 | 6 | 5 | 1 | |||||||||||||||
| 2023 | 24 | 7 | 15 | 2 | ||||||||||||||
| 2024 | 6 | 1 | 5 | |||||||||||||||
| TOTALS | 101 | 10 | 5 | 0 | 3 | 4 | 1 | 1 | 1 | 35 | 20 | 0 | 3 | 4 | 9 | 1 | 2 | 2 |
References
- Balamayooran, G.; Atkins, H.M.; Andrews, R.N.; Michalson, K.T.; Hutchison, A.R.; LeGrande, A.C.; Wilson, Q.N.; Gee, M.K.; Aycock, S.T.; Jorgensen, M.J.; et al. Epizootic Yersinia enterocolitica in captive African green monkeys (Chlorocebus aethiops sabaeus). Front. Vet. Sci. 2022, 9, 922961. [Google Scholar] [CrossRef]
- Bottone, E.J. Yersinia enterocolitica: The charisma continues. Clin. Microbiol. Rev. 1997, 10, 257–276. [Google Scholar] [CrossRef]
- Straley, S.C.; Perry, R.D. Environmental modulation of gene expression and pathogenesis in Yersinia. Trends Microbiol. 1995, 3, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Nishiura, H.; Yamazaki, A.; Wakakuri, K.; Sasaki, J.; Terajima, J.; Ochiai, K. Yersinia infection in two captive guereza colobus monkeys (Colobus guereza). J. Vet. Med. Sci. 2019, 81, 1201–1204. [Google Scholar] [CrossRef] [PubMed]
- Iwata, T.; Une, Y.; Okatani, A.T.; Kato, Y.; Nakadai, A.; Lee, K.I.; Watanabe, M.; Taniguchi, T.; Elhelaly, A.E.; Hirota, Y.; et al. Virulence characteristics of Yersinia pseudotuberculosis isolated from breeding monkeys in Japan. Vet. Microbiol. 2008, 129, 404–409. [Google Scholar] [CrossRef]
- Simmons, J.; Gibson, S. Chapter 2—Bacterial and Mycotic Diseases of Nonhuman Primates. In Nonhuman Primates in Biomedical Research, 2nd ed.; Abee, C.R., Mansfield, K., Tardif, S., Morris, T., Eds.; Academic Press: Boston, MA, USA, 2012; pp. 105–172. [Google Scholar]
- Nederlof, R.A.; Bruins-van Sonsbeek, L.G.R.; Stumpel, J.B.G.; van Bolhuis, H.; Broens, E.M.; IJzer, J.; Bakker, J. Yersinia pseudotuberculosis in Non-Domesticated Mammals and Birds in Captivity. Vet. Sci. 2025, 12, 161. [Google Scholar] [CrossRef] [PubMed]
- Brady, M.F.; Yarrarapu, S.N.S.; Anjum, F. Yersinia pseudotuberculosis. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: http://www.ncbi.nlm.nih.gov/books/NBK430717/ (accessed on 11 September 2025).
- McNally, A.; Thomson, N.R.; Reuter, S.; Wren, B.W. “Add, stir and reduce”: Yersinia spp. as model bacteria for pathogen evolution. Nat. Rev. Microbiol. 2016, 14, 177–190. [Google Scholar] [CrossRef]
- Dube, P. Interaction of Yersinia with the gut: Mechanisms of pathogenesis and immune evasion. Curr. Top. Microbiol. Immunol. 2009, 337, 61–91. [Google Scholar]
- Aziz, M.; Yelamanchili, V.S. Yersinia enterocolitica. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2025. Available online: http://www.ncbi.nlm.nih.gov/books/NBK499837/ (accessed on 11 September 2025).
- Hu, B.; Wang, J.; Li, L.; Wang, Q.; Qin, J.; Chi, Y.; Yan, J.; Sun, W.; Cao, B.; Guo, X. Functional Identification and Genetic Analysis of O-Antigen Gene Clusters of Food-Borne Pathogen Yersinia enterocolitica O:10 and Other Uncommon Serotypes, Further Revealing Their Virulence Profiles. J. Microbiol. Biotechnol. 2024, 34, 1599–1608. [Google Scholar] [CrossRef]
- Brady, A.G.; Carville, A.A.L. Chapter 12—Digestive System Diseases of Nonhuman Primates. In Nonhuman Primates in Biomedical Research, 2nd ed.; Abee, C.R., Mansfield, K., Tardif, S., Morris, T., Eds.; Academic Press: Boston, MA, USA, 2012; pp. 589–627. [Google Scholar]
- Mätz-Rensing, K.; Lowenstine, L.J. Chapter 14—New World and Old World Monkeys. In Pathology of Wildlife and Zoo Animals; Terio, K.A., McAloose, D., Leger, J.S., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 343–374. [Google Scholar]
- Moxley, R.A. Family Yersiniaceae. In Veterinary Microbiology, 4th ed.; McVey, D.S., Kennedy, M., Chengappa, M.M., Wilkes, R., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2022; pp. 88–99. [Google Scholar]
- Vore, S.J.; Peele, P.D.; Barrow, P.A.; Bradfield, J.F.; Pryor, W.H. A prevalence survey for zoonotic enteric bacteria in a research monkey colony with specific emphasis on the occurrence of enteric Yersinia. J. Med. Primatol. 2001, 30, 20–25. [Google Scholar] [CrossRef]
- Taffs, L.F.; Dunn, G. An outbreak of Yersinia pseudotuberculosis infection in a small indoor breeding colony of red-bellied (Saguinus labiatus) tamarins. Lab. Anim. 1983, 17, 311–320. [Google Scholar] [CrossRef]
- Bronson, R.T.; May, B.D.; Ruebner, B.H. An outbreak of infection by Yersinia pseudotuberculosis in nonhuman primates. Am. J. Pathol. 1972, 69, 289–308. [Google Scholar] [PubMed]
- Bernacky, B.J.; Gibson, S.V.; Keeling, M.E.; Abee, C. Chapter 16—Nonhuman Primates. In Laboratory Animal Medicine, 2nd ed.; Fox, J.G., Anderson, L.C., Loew, F.M., Quimby, F.W., Eds.; Academic Press: San Diego, CA, USA, 2002; pp. 676–791. [Google Scholar]
- MacArthur, J.A.; Wood, M. Yersiniosis in a breeding unit of Macaca fascicularis (cynomolgus monkeys). Lab. Anim. 1983, 17, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Compo, N.R.; Mieles-Rodriguez, L.; Gomez, D.E. Fecal Bacterial Microbiota of Healthy Free-Ranging, Healthy Corralled, and Chronic Diarrheic Corralled Rhesus Macaques (Macaca mulatta). Comp. Med. 2021, 71, 152–165. [Google Scholar] [CrossRef]
- Banish, L.D.; Sims, R.; Sack, D.; Montali, R.J.; Phillips, L., Jr.; Bush, M. Prevalence of shigellosis and other enteric pathogens in a zoologic collection of primates. J. Am. Vet. Med. Assoc. 1993, 203, 126–132. [Google Scholar] [CrossRef]
- Seabaugh, J.A.; Anderson, D.M. Pathogenicity and virulence of Yersinia. Virulence 2024, 15, 2316439. [Google Scholar] [CrossRef]
- Martins, B.T.F.; Camargo, A.C.; Tavares Rde, M.; Nero, L.A. Relevant foodborne bacteria associated to pork production chain. Adv. Food Nutr. Res. 2025, 113, 181–218. [Google Scholar] [PubMed]
- Carniel, E.; Mollaret, H.H. Yersiniosis. Comp. Immunol. Microbiol. Infect. Dis. 1990, 13, 51–58. [Google Scholar] [CrossRef]
- Fredriksson-Ahomaa, M.; Naglic, T.; Turk, N.; Seol, B.; Grabarević, Z.; Bata, I.; Perkovic, D.; Stolle, A. Yersiniosis in zoo marmosets (Callitrix jacchuss) caused by Yersinia enterocolitica 4/O:3. Vet. Microbiol. 2007, 121, 363–367. [Google Scholar] [CrossRef]
- Soto, E.; Loftis, A.; Boruta, D.; Rostad, S.; Beierschmitt, A.; McCoy, M.; Francis, S.; Berezowski, J.; Illanes, O.; Recinos, D.; et al. Multispecies Epidemiologic Surveillance Study after an Outbreak of Yersiniosis at an African Green Monkey Research Facility. Comp. Med. 2015, 65, 526–531. [Google Scholar]
- Lü, Z.; Su, L.; Han, M.; Wang, X.; Li, M.; Wang, S.; Cui, S.; Chen, J.; Yang, B. Genomic characteristics and virulence of common but overlooked Yersinia intermedia, Y. frederiksenii, and Y. kristensenii in food. Int. J. Food Microbiol. 2025, 430, 111052. [Google Scholar] [CrossRef]
- Vilela, F.P.; Imori, P.F.M.; Allard, M.W.; Falcão, J.P. Insights into the genomic traits of Yersinia frederiksenii, Yersinia intermedia and Yersinia kristensenii isolated from diverse sources in Brazil. Antonie Van Leeuwenhoek 2024, 117, 86. [Google Scholar] [CrossRef] [PubMed]
- Joutsen, S.; Johansson, P.; Laukkanen-Ninios, R.; Björkroth, J.; Fredriksson-Ahomaa, M. Two copies of the ail gene found in Yersinia enterocolitica and Yersinia kristensenii. Vet. Microbiol. 2020, 247, 108798. [Google Scholar] [CrossRef] [PubMed]
- Prongay, K.; Park, B.; Murphy, S.J. Risk Factor Analysis May Provide Clues to Diarrhea Prevention in Outdoor-Housed Rhesus Macaques (Macaca mulatta). Am. J. Primatol. 2013, 75, 872–882. [Google Scholar] [CrossRef]
- Valentini, D.; Vittucci, A.C.; Grandin, A.; Tozzi, A.E.; Russo, C.; Onori, M.; Menichella, D.; Bartuli, A.; Villani, A. Coinfection in acute gastroenteritis predicts a more severe clinical course in children. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 2013, 32, 909–915. [Google Scholar] [CrossRef]
- Wang, G.; He, Y.; Jin, X.; Zhou, Y.; Chen, X.; Zhao, J.; Zhang, H.; Chen, W. The Effect of Co-infection of Food-Borne Pathogenic Bacteria on the Progression of Campylobacter jejuni Infection in Mice. Front. Microbiol. 2018, 9, 1977. [Google Scholar] [CrossRef] [PubMed]
- Bacon, R.L.; Hodo, C.L.; Wu, J.; Welch, S.; Nickodem, C.; Vinasco, J.; Threadgill, D.; Gray, S.B.; Norman, K.N.; Lawhon, S.D. Diversity of Campylobacter spp. circulating in a rhesus macaque (Macaca mulatta) breeding colony using culture and molecular methods. mSphere 2024, 9, e00560-24. [Google Scholar] [CrossRef]
- Riede, L.L.; Knauf-Witzens, T.; Westerhüs, U.; Bonke, R.; Schlez, K.; Büttner, K.; Rau, J.; Fischer, D.; Grund, L.; Roller, M.; et al. Occurrence and Monitoring of the Zoonotic Pathogen Yersinia pseudotuberculosis in Various Zoo Animal Species. Microorganisms 2025, 13, 516. [Google Scholar] [CrossRef]
- Elmore, D.B.; Anderson, J.H.; Hird, D.W.; Sanders, K.D.; Lerche, N.W. Diarrhea rates and risk factors for developing chronic diarrhea in infant and juvenile rhesus monkeys. Lab. Anim. Sci. 1992, 42, 356–359. [Google Scholar]
- Peel, M.; Hardgrove, E.; Swenson, J.; Clancy, M.; Marinkovich, M.; Garner, M.M.; Haefele, H.; Hernández, B.A.; Kubiski, S.V. Clinical and Pathologic Presentation of Yersiniosis in Various Nondomestic Species: An Investigation of Yersinia pseudotuberculosis Outbreaks from Four North American Zoological Institutions. J. Zoo. Wildl. Med. Off. Publ. Am. Assoc. Zoo. Vet. 2024, 55, 1095–1103. [Google Scholar] [CrossRef]
- Haertel, A.J.; McCoy, S.; McCoy, C.S.; Reyes, M.D.; Palmer, H.; Sosa, P.M.; Burke, M.C.; Melendez, M.; Nham, P.B.; Timmel, G.; et al. Multicenter analysis of the attributable diarrhea risk and odds ratios of pathogens in rhesus macaques (Macaca mulatta) using multiplex PCR gastrointestinal panel testing and conventional fecal culture. J. Am. Assoc. Lab. Anim. Sci. 2025, 64, 873–881. [Google Scholar] [CrossRef]
- Asadishad, B.; Ghoshal, S.; Tufenkji, N. Role of Cold Climate and Freeze–Thaw on the Survival, Transport, and Virulence of Yersinia enterocolitica. Environ. Sci. Technol. 2013, 47, 14169–14177. [Google Scholar] [CrossRef] [PubMed]
- Western Regional Climate Center. Available online: https://wrcc.dri.edu (accessed on 14 January 2026).
- Sha, J.C.M.; Du, H.; Deng, J.; Chen, Z.; Wu, Q.; Chen, W. Differential responses of non-human primates to seasonal temperature fluctuations. Primates 2020, 61, 455–464. [Google Scholar] [CrossRef]
- Mancini, M.E.; Beverelli, M.; Donatiello, A.; Didonna, A.; Dattoli, L.; Faleo, S.; Occhiochiuso, G.; Galante, D.; Rondinone, V.; Del Sambro, L.; et al. Isolation and characterization of Yersinia enterocolitica from foods in Apulia and Basilicata regions (Italy) by conventional and modern methods. PLoS ONE 2022, 17, e0268706. [Google Scholar] [CrossRef]
- Varettas, K.; Mukerjee, C.; Schmidt, M. A comparative study of the BBL crystal enteric/nonfermenter identification system and the biomerieux API20E and API20NE identification systems after overnight incubation. Pathology 1995, 27, 358–361. [Google Scholar] [CrossRef]
- Bottone, E.J. Yersinia enterocolitica: Overview and epidemiologic correlates. Microbes Infect. 1999, 1, 323–333. [Google Scholar] [CrossRef]
- Shoaib, M.; Shehzad, A.; Raza, H.; Niazi, S.; Khan, I.M.; Akhtar, W.; Safdar, W.; Wang, Z. A comprehensive review on the prevalence, pathogenesis and detection of Yersinia enterocolitica. RSC Adv. 2019, 9, 41010–41021. [Google Scholar] [CrossRef]
- Bui, T.H.; Ikeuchi, S.; O’Brien, Y.S.; Niwa, T.; Hara-Kudo, Y.; Taniguchi, T.; Hayashidani, H. Multiplex PCR method for differentiating highly pathogenic Yersinia enterocolitica and low pathogenic Yersinia enterocolitica, and Yersinia pseudotuberculosis. J. Vet. Med. Sci. 2021, 83, 1982–1987. [Google Scholar] [CrossRef] [PubMed]
- Savin, C.; Leclercq, A.; Carniel, E. Evaluation of a Single Procedure Allowing the Isolation of Enteropathogenic Yersinia along with Other Bacterial Enteropathogens from Human Stools. PLoS ONE 2012, 7, e41176. [Google Scholar] [CrossRef] [PubMed]
- Talari, K.; Goyal, M. Retrospective studies—Utility and caveats. J. R. Coll. Physicians Edinb. 2020, 50, 398–402. [Google Scholar] [CrossRef]
- Vassar, M.; Holzmann, M. The retrospective chart review: Important methodological considerations. J. Educ. Eval. Health Prof. 2013, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Ohta, E. Pathologic characteristics of infectious diseases in macaque monkeys used in biomedical and toxicologic studies. J. Toxicol. Pathol. 2023, 36, 95–122. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.