Outer Membrane Vesicles Attenuate Klebsiella pneumoniae Infection Injury by Affecting Macrophage Polarisation and Helper T Cell Differentiation
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial and Cell Culture
2.2. Extraction of OMVs
2.3. Characterization and Analysis of K. pneumoniae_OMVs
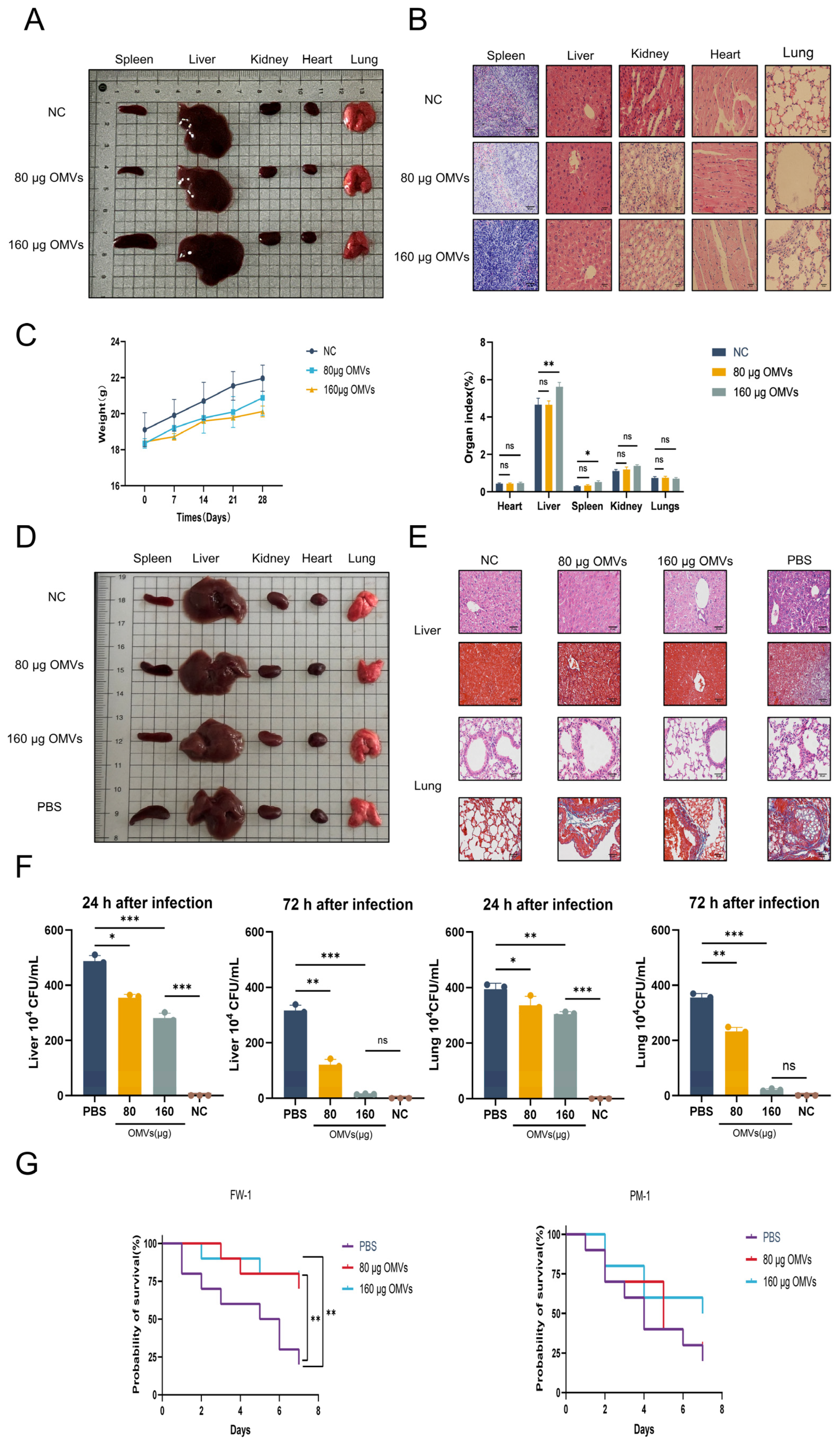
2.4. Mouse Immunisation and Infection
2.5. Organ Coefficient and Bacterial Load
2.6. Hematoxylin and Eosin Staining (HE) and Masson
2.7. RNA Isolation and Library Preparation and RNA Sequencing
2.8. Assessment of Cell Viability and Cell Grouping
2.9. qRT-PCR
2.10. ELISA
2.11. Western Blotting
2.12. IHC
2.13. Statistical Analysis
3. Results
3.1. Characterization of K. pneumoniae_OMVs
3.2. Immunological Safety and Protective Effects of K. pneumoniae_OMVs
3.3. Analysis of Transcriptome Data
3.4. Effects of K. pneumoniae_OMVs on Raw264.7 Proliferation and Polarization
3.5. K. pneumoniae_OMVs Induced Th Cells Differentiation in Mice
3.6. K. pneumoniae_OMVs Induce Macrophage Polarization and Helper T Cell Differentiation to Protect K. pneumoniae-Infected Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bassetti, M.; Righi, E.; Carnelutti, A.; Graziano, E.; Russo, A. Multidrug-resistant Klebsiella pneumoniae: Challenges for treatment, prevention and infection control. Expert Rev. Anti-Infect. Ther. 2018, 16, 749–761. [Google Scholar] [CrossRef]
- Martin, R.M.; Bachman, M.A. Colonization, Infection, and the Accessory Genome of Klebsiella pneumoniae. Front. Cell. Infect. Microbiol. 2018, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Sayed, M.G.; Mansy, M.S.; El Borhamy, M.I.; Elsherif, H.M. Exploring virulence factors, virulome, and multidrug resistance of Klebsiella pneumoniae strains isolated from patients with central Line-associated bloodstream infections. Sci. Rep. 2025, 15, 20230. [Google Scholar] [CrossRef]
- Tiku, V.; Tan, M.W. Host immunity and cellular responses to bacterial outer membrane vesicles. Trends Immunol. 2021, 42, 1024–1036. [Google Scholar] [CrossRef]
- Zhang, X.; Qian, C.; Tang, M.; Zeng, W.; Kong, J.; Fu, C.; Xu, C.; Ye, J.; Zhou, T. Carbapenemase-loaded outer membrane vesicles protect Pseudomonas aeruginosa by degrading imipenem and promoting mutation of antimicrobial resistance gene. Drug Resist. Updates 2023, 68, 100952. [Google Scholar] [CrossRef] [PubMed]
- Jalalifar, S.; Morovati Khamsi, H.; Hosseini-Fard, S.R.; Karampoor, S.; Bajelan, B.; Irajian, G.; Mirzaei, R. Emerging role of microbiota derived outer membrane vesicles to preventive, therapeutic and diagnostic proposes. Infect. Agents Cancer 2023, 18, 3. [Google Scholar] [CrossRef]
- Pizza, M.; Bekkat-Berkani, R.; Rappuoli, R. Vaccines against Meningococcal Diseases. Microorganisms 2020, 8, 1521. [Google Scholar] [CrossRef] [PubMed]
- Garling, A.; Goursat, C.; Seguy, C.; Martin, P.; Goman, A.; Nougayrède, J.P.; Oswald, É.; Auvray, F.; Branchu, P. Development of intimin-enriched outer membrane vesicles (OMVs) as a vaccine to control intestinal carriage of Enterohemorrhagic Escherichia coli. Vaccine 2025, 52, 126899. [Google Scholar] [CrossRef]
- Liu, Q.; Li, B.; Ma, J.; Lei, X.; Ma, J.; Da, Y.; Zhou, Z.; Tao, J.; Ren, X.; Zeng, T.; et al. Development of a Recombinant Outer Membrane Vesicles (OMVs)-Based Vaccine Against Helicobacter pylori Infection in Mice. J. Extracell. Vesicles 2025, 14, e70085. [Google Scholar] [CrossRef]
- Liu, Z.; Ou, Y.; He, X.; Yuan, T.; Li, M.; Long, Y.; Li, Y.; Tan, Y. Guardians of the Lung: The Multifaceted Roles of Macrophages in Cancer and Infectious Disease. DNA Cell Biol. 2025, 44, 249–262. [Google Scholar] [CrossRef]
- Glass, C.K.; Natoli, G. Molecular control of activation and priming in macrophages. Nat. Immunol. 2016, 17, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.X.; Xu, X.H.; Jin, L. Effects of Metabolism on Macrophage Polarization Under Different Disease Backgrounds. Front. Immunol. 2022, 13, 880286. [Google Scholar] [CrossRef] [PubMed]
- Hansakon, A.; Png, C.W.; Zhang, Y.; Angkasekwinai, P. Macrophage-Derived Osteopontin Influences the Amplification of Cryptococcus neoformans-Promoting Type 2 Immune Response. J. Immunol. 2021, 207, 2107–2117. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wang, J.; Gan, L.; Wu, Z.; Wu, T.; Li, F.; Xu, X.; Lei, L.; Li, N. Carbon dot-based treatment for bacterial pneumonia by promoting a PI3K-mediated M1 polarization of macrophages. J. Nanobiotechnol. 2025, 23, 315. [Google Scholar] [CrossRef]
- Wang, X.; Sun, Y.; Guo, H.; Yan, X.; Ma, S.; Yang, B.; Jiang, L. Xylose utilization promotes Salmonella replication within macrophages and systemic infection in mice. Virulence 2024, 15, 2435381. [Google Scholar] [CrossRef]
- Shang, Q.; Yu, X.; Sun, Q.; Li, H.; Sun, C.; Liu, L. Polysaccharides regulate Th1/Th2 balance: A new strategy for tumor immunotherapy. Biomed. Pharmacother. 2024, 170, 115976. [Google Scholar] [CrossRef]
- Vareille-Delarbre, M.; Miquel, S.; Garcin, S.; Bertran, T.; Balestrino, D.; Evrard, B.; Forestier, C. Immunomodulatory Effects of Lactobacillus plantarum on Inflammatory Response Induced by Klebsiella pneumoniae. Infect. Immun. 2019, 87, e00570-19. [Google Scholar] [CrossRef]
- Wojciechowski, W.; Harris, D.P.; Sprague, F.; Mousseau, B.; Makris, M.; Kusser, K.; Honjo, T.; Mohrs, K.; Mohrs, M.; Randall, T.; et al. Cytokine-producing effector B cells regulate type 2 immunity to H. polygyrus. Immunity 2009, 30, 421–433. [Google Scholar] [CrossRef]
- Huang, Y.; Nan, L.; Xiao, C.; Dong, J.; Li, K.; Cheng, J.; Ji, Q.; Wei, Q.; Bao, G.; Liu, Y. Outer Membrane Vesicles Coating Nano-Glycyrrhizic Acid Confers Protection Against Borderella bronchiseptica Through Th1/Th2/Th17 Responses. Int. J. Nanomed. 2022, 17, 647–663. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, F.; Zou, J.; Wu, W.; Jing, H.; Gou, Q.; Li, H.; Gu, J.; Zou, Q.; Zhang, J. Immunization with Pseudomonas aeruginosa outer membrane vesicles stimulates protective immunity in mice. Vaccine 2018, 36, 1047–1054. [Google Scholar] [CrossRef]
- Wu, G.; Ji, H.; Guo, X.; Li, Y.; Ren, T.; Dong, H.; Liu, J.; Liu, Y.; Shi, X.; He, B. Nanoparticle reinforced bacterial outer-membrane vesicles effectively prevent fatal infection of carbapenem-resistant Klebsiella pneumoniae. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102148. [Google Scholar] [CrossRef]
- Jiang, S.; Li, H.; Zhang, L.; Mu, W.; Zhang, Y.; Chen, T.; Wu, J.; Tang, H.; Zheng, S.; Liu, Y.; et al. Generic Diagramming Platform (GDP): A comprehensive database of high-quality biomedical graphics. Nucleic Acids Res. 2025, 53, D1670–D1676. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Chen, M.; Huang, X.; Zhang, G.; Zeng, L.; Zhang, G.; Wu, S.; Wang, Y. SRplot: A free online platform for data visualization and graphing. PLoS ONE 2023, 18, e0294236. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wu, Y. Potential of bacterial outer membrane vesicles in tumor vaccine: Characteristics, advancements, and future directions. Essays Biochem. 2025, 69, EBC20253004. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Antenucci, F.; Villumsen, K.R.; Bojesen, A.M. Bacterial Outer Membrane Vesicles as a Versatile Tool in Vaccine Research and the Fight against Antimicrobial Resistance. mBio 2021, 12, e0170721. [Google Scholar] [CrossRef]
- Lu, L.; Zhai, L.; Ou, Q.; Sang, S.; Cao, C.; Guan, Y.; Mao, Y.; Zhai, Y.; Li, K.; Yu, R.; et al. Engineered Outer Membrane Vesicles for Antigen Delivery: Exploratory Study on Adjuvant Activity and Systemic Reactogenicity. Vaccines 2025, 13, 552. [Google Scholar] [CrossRef]
- Wang, B.; Gu, A.; Yan, J.; Zhang, Y.; Liu, J.; Sun, C.; Wei, Y.; Gu, C.; Wang, Y. Kaempferol alleviates sepsis related acute lung injury by inhibiting the activation of alveolar macrophages mediated by extracellular vesicles from alveolar epithelial cells. Int. Immunopharmacol. 2025, 162, 115130. [Google Scholar] [CrossRef]
- Lu, Z.X.; Liu, L.X.; Fu, Z.; Wang, S.N.; Sun, C.N.; Yu, W.G.; Lu, X.Z. Chitosan oligosaccharides alleviate macrophage pyroptosis and protect sepsis mice via activating the Nrf2/GPX4 pathway. Int. J. Biol. Macromol. 2024, 277 Pt 1, 133899. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, H.; Peng, X.; Li, T.; Fan, X.; Shen, Q.; Zhang, G.; Li, Q.; Boireau, P.; Jiang, H.; et al. Biological nanoparticles from Brucella abortusΔeipB∆perΔwadC elicit protective immunity against brucellosis. J. Nanobiotechnol. 2025, 23, 727. [Google Scholar] [CrossRef]
- Bjånes, E.; Krishnan, N.; Koh, T.; Ngo, A.T.; Cole, J.; Olson, J.; Cornax, I.; Chen, C.H.; Chavarria, N.; Dahesh, S.; et al. STING-adjuvanted outer membrane vesicle nanoparticle vaccine against Pseudomonas aeruginosa. JCI Insight 2025, 10, e188105. [Google Scholar] [CrossRef]
- Xiang, S.; Khan, A.; Yao, Q.; Wang, D. Recent advances in bacterial outer membrane vesicles: Effects on the immune system, mechanisms and their usage for tumor treatment. J. Pharm. Anal. 2024, 14, 101049. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Miao, Y.; Chen, H.; Wang, K.; Wang, S.; Wang, R.; Wu, Z.; Li, J. Revealing the mechanism: The influence of Baicalin on M1/M2 and Th1/Th2 imbalances in mycoplasma gallisepticum infection. Poult. Sci. 2024, 103, 104145. [Google Scholar] [CrossRef]
- Bierwagen, J.; Wiegand, M.; Laakmann, K.; Danov, O.; Limburg, H.; Herbel, S.M.; Heimerl, T.; Dorna, J.; Jonigk, D.; Preußer, C.; et al. Bacterial vesicles block viral replication in macrophages via TLR4-TRIF-axis. Cell Commun. Signal. 2023, 21, 65. [Google Scholar] [CrossRef]
- Ryu, S.; Ni, K.; Wang, C.; Sivanantham, A.; Carnino, J.M.; Ji, H.L.; Jin, Y. Bacterial Outer Membrane Vesicles Promote Lung Inflammatory Responses and Macrophage Activation via Multi-Signaling Pathways. Biomedicines 2023, 11, 568. [Google Scholar] [CrossRef]
- Lee, W.H.; Choi, H.I.; Hong, S.W.; Kim, K.S.; Gho, Y.S.; Jeon, S.G. Vaccination with Klebsiella pneumoniae-derived extracellular vesicles protects against bacteria-induced lethality via both humoral and cellular immunity. Exp. Mol. Med. 2015, 47, e183. [Google Scholar] [CrossRef] [PubMed]
- Martinez, F.O.; Sica, A.; Mantovani, A.; Locati, M. Macrophage activation and polarization. Front. Biosci. 2008, 13, 453–461. [Google Scholar] [CrossRef]
- Garling, A.; Auvray, F.; Epardaud, M.; Oswald, É.; Branchu, P. Outer Membrane Vesicles as a Versatile Platform for Vaccine Development: Engineering Strategies, Applications and Challenges. J. Extracell. Vesicles 2025, 14, e70150. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Qiu, Y.; Liu, B.; Zhen, X.; Li, R. Engineered nano-bacteria hybrids for precision cancer immunotherapy. Mater. Today Bio 2025, 35, 102484. [Google Scholar] [CrossRef]
- Zindel, J.; Kubes, P. DAMPs, PAMPs, and LAMPs in Immunity and Sterile Inflammation. Annu. Rev. Pathol. 2020, 15, 493–518. [Google Scholar] [CrossRef]
- Yunna, C.; Mengru, H.; Lei, W.; Weidong, C. Macrophage M1/M2 polarization. Eur. J. Pharmacol. 2020, 877, 173090. [Google Scholar] [CrossRef]
- Bertheloot, D.; Latz, E.; Franklin, B.S. Necroptosis, pyroptosis and apoptosis: An intricate game of cell death. Cell. Mol. Immunol. 2021, 18, 1106–1121. [Google Scholar] [CrossRef]
- Wei, X.; Xie, F.; Zhou, X.; Wu, Y.; Yan, H.; Liu, T.; Huang, J.; Wang, F.; Zhou, F.; Zhang, L. Role of pyroptosis in inflammation and cancer. Cell. Mol. Immunol. 2022, 19, 971–992. [Google Scholar] [CrossRef]
- Li, Y.; Li, X.; Wu, W.; Liu, P.; Liu, J.; Jiang, H.; Deng, L.; Ni, C.; Wu, X.; Zhao, Y.; et al. Insights into the roles of macrophages in Klebsiella pneumoniae infections: A comprehensive review. Cell. Mol. Biol. Lett. 2025, 30, 34. [Google Scholar] [CrossRef]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, W.; Wang, W.; Kong, L.; Chen, S.; Zhang, X.; Zhai, Y.; Zhang, B.; Wang, Y.; Zhao, D.; Tang, X.; et al. Outer Membrane Vesicles Attenuate Klebsiella pneumoniae Infection Injury by Affecting Macrophage Polarisation and Helper T Cell Differentiation. Microorganisms 2025, 13, 2849. https://doi.org/10.3390/microorganisms13122849
Fan W, Wang W, Kong L, Chen S, Zhang X, Zhai Y, Zhang B, Wang Y, Zhao D, Tang X, et al. Outer Membrane Vesicles Attenuate Klebsiella pneumoniae Infection Injury by Affecting Macrophage Polarisation and Helper T Cell Differentiation. Microorganisms. 2025; 13(12):2849. https://doi.org/10.3390/microorganisms13122849
Chicago/Turabian StyleFan, Wei, Wei Wang, Lin Kong, Shifan Chen, Xinyu Zhang, Yilu Zhai, Bo Zhang, Yan Wang, Dazhuo Zhao, Xiukai Tang, and et al. 2025. "Outer Membrane Vesicles Attenuate Klebsiella pneumoniae Infection Injury by Affecting Macrophage Polarisation and Helper T Cell Differentiation" Microorganisms 13, no. 12: 2849. https://doi.org/10.3390/microorganisms13122849
APA StyleFan, W., Wang, W., Kong, L., Chen, S., Zhang, X., Zhai, Y., Zhang, B., Wang, Y., Zhao, D., Tang, X., Fu, J., & Sun, F. (2025). Outer Membrane Vesicles Attenuate Klebsiella pneumoniae Infection Injury by Affecting Macrophage Polarisation and Helper T Cell Differentiation. Microorganisms, 13(12), 2849. https://doi.org/10.3390/microorganisms13122849





