Cellular Damage of Bacteria Attached to Senescent Phytoplankton Cells as a Result of the Transfer of Photochemically Produced Singlet Oxygen: A Review
Abstract
1. Introduction
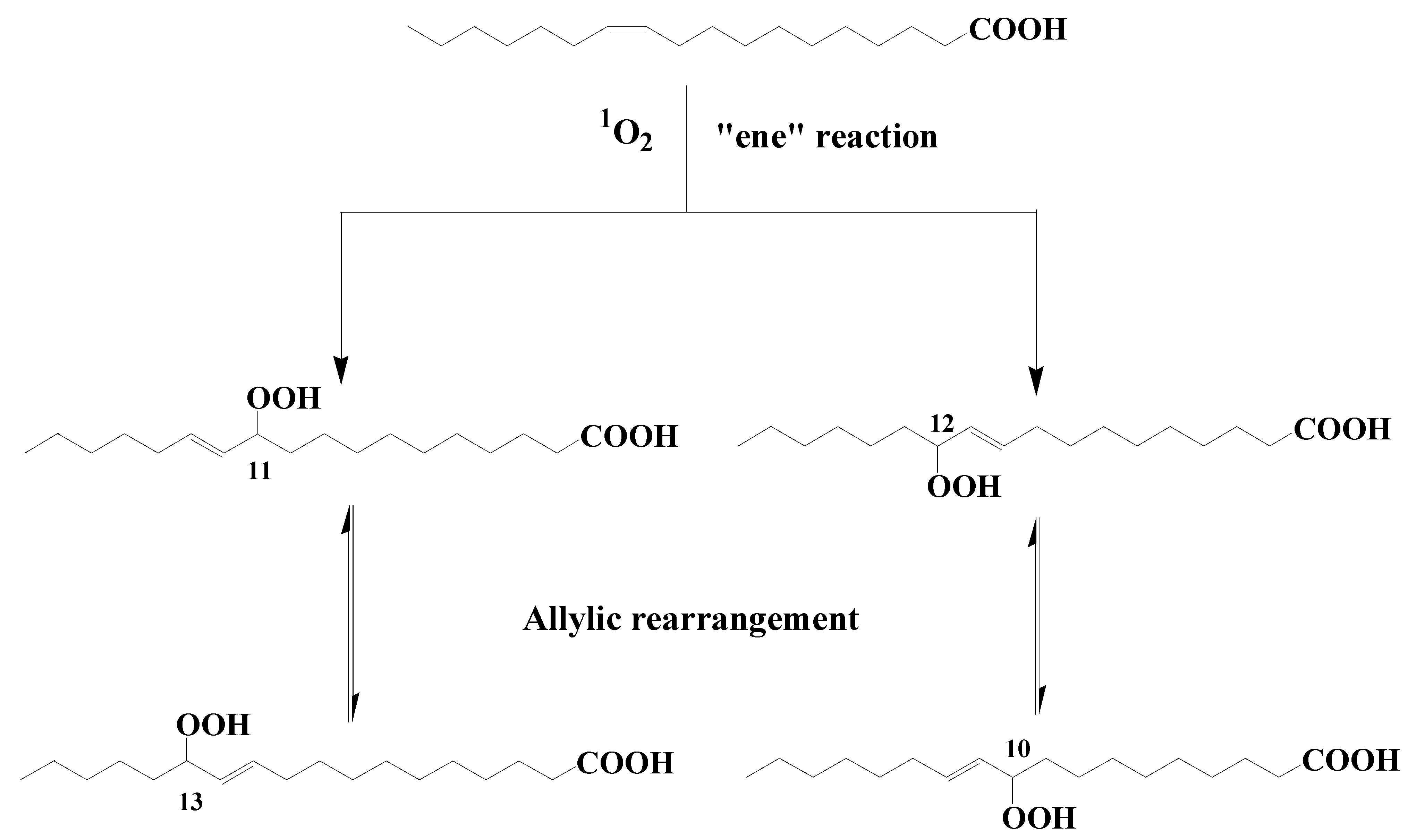
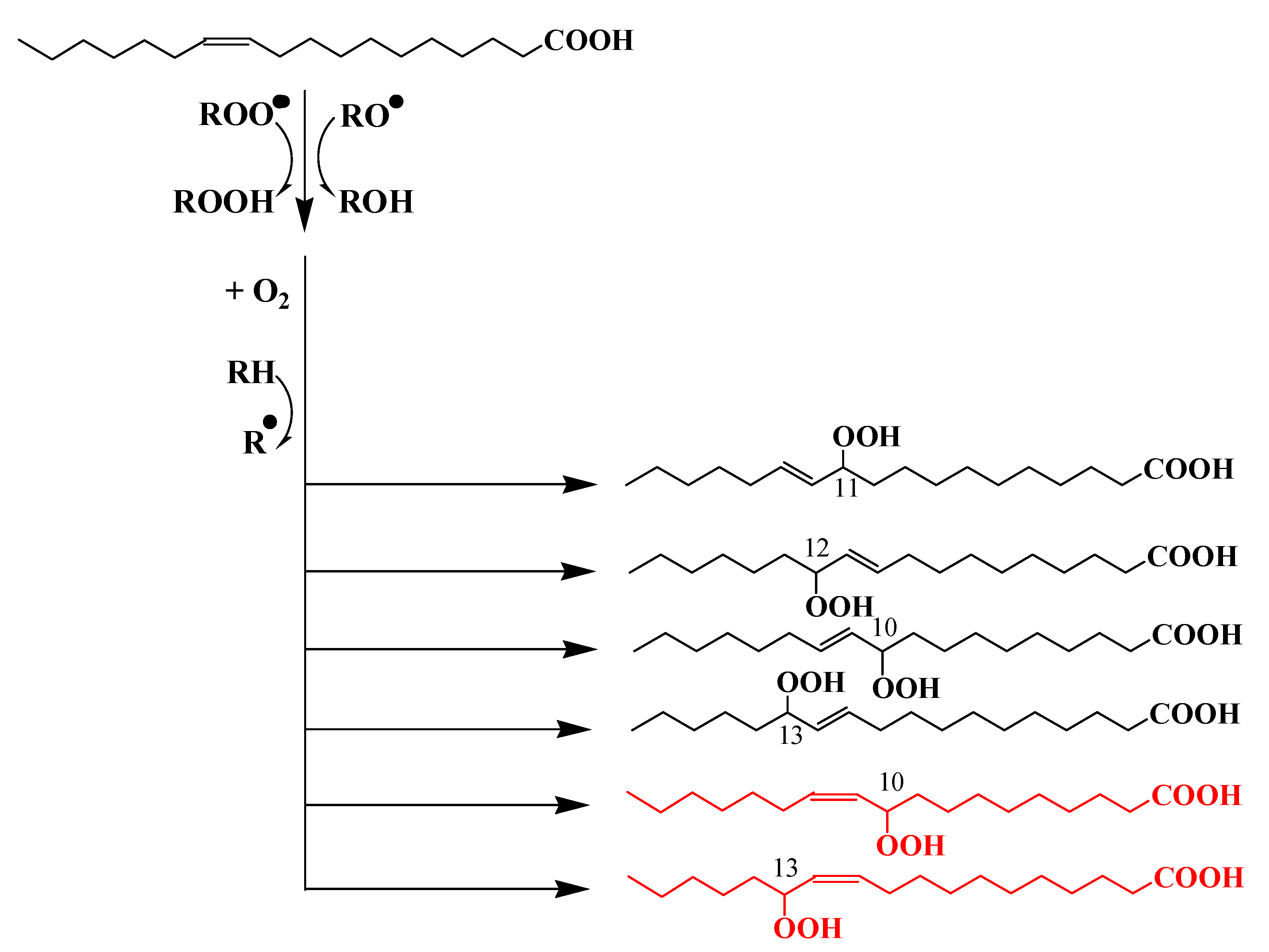
2. Potential Sources of Photoproducts of cis-Vaccenic Acid in Oceans
2.1. Photooxidation of Aerobic Anoxygenic Phototrophic Bacteria (AAPB)
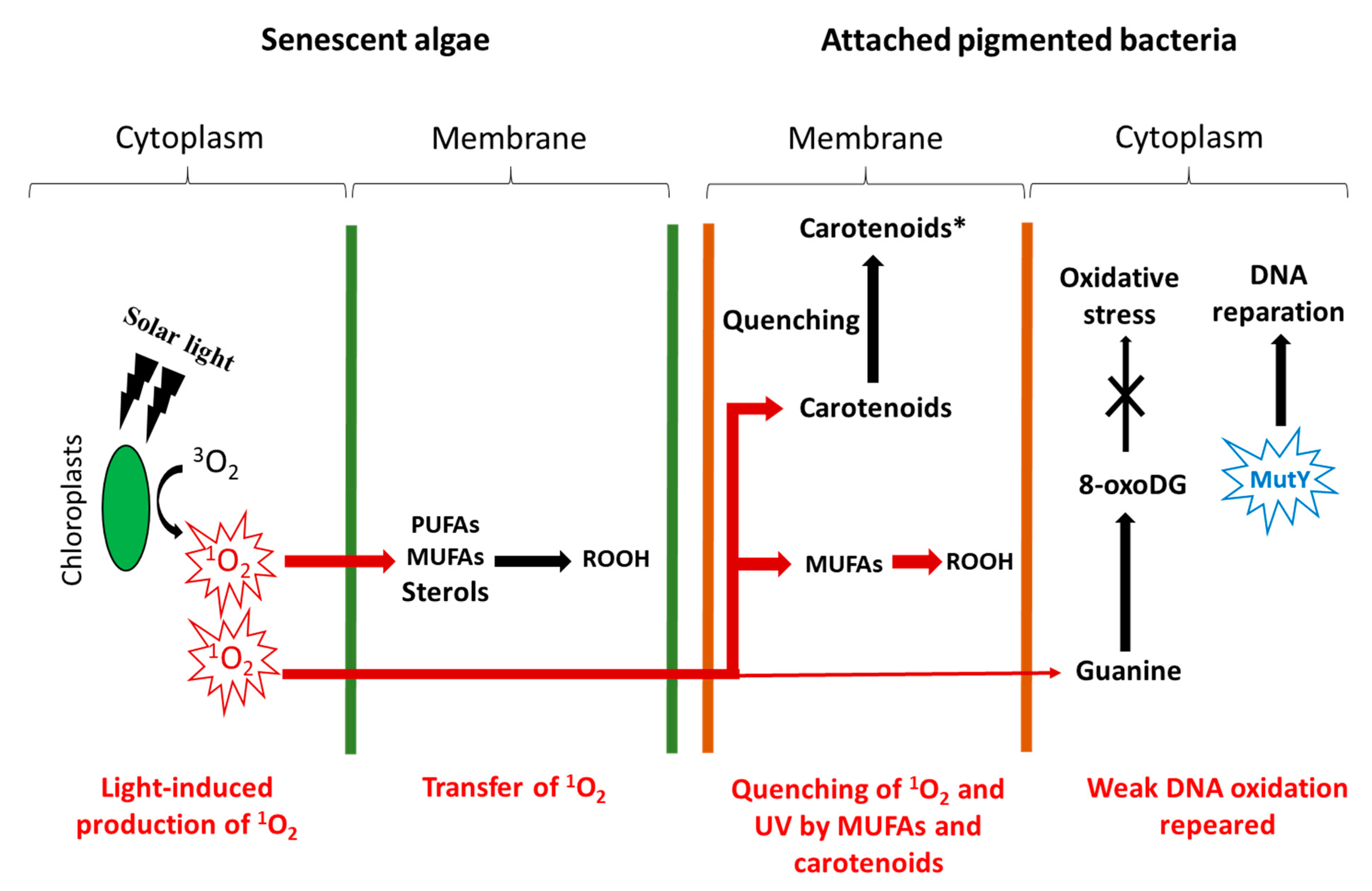
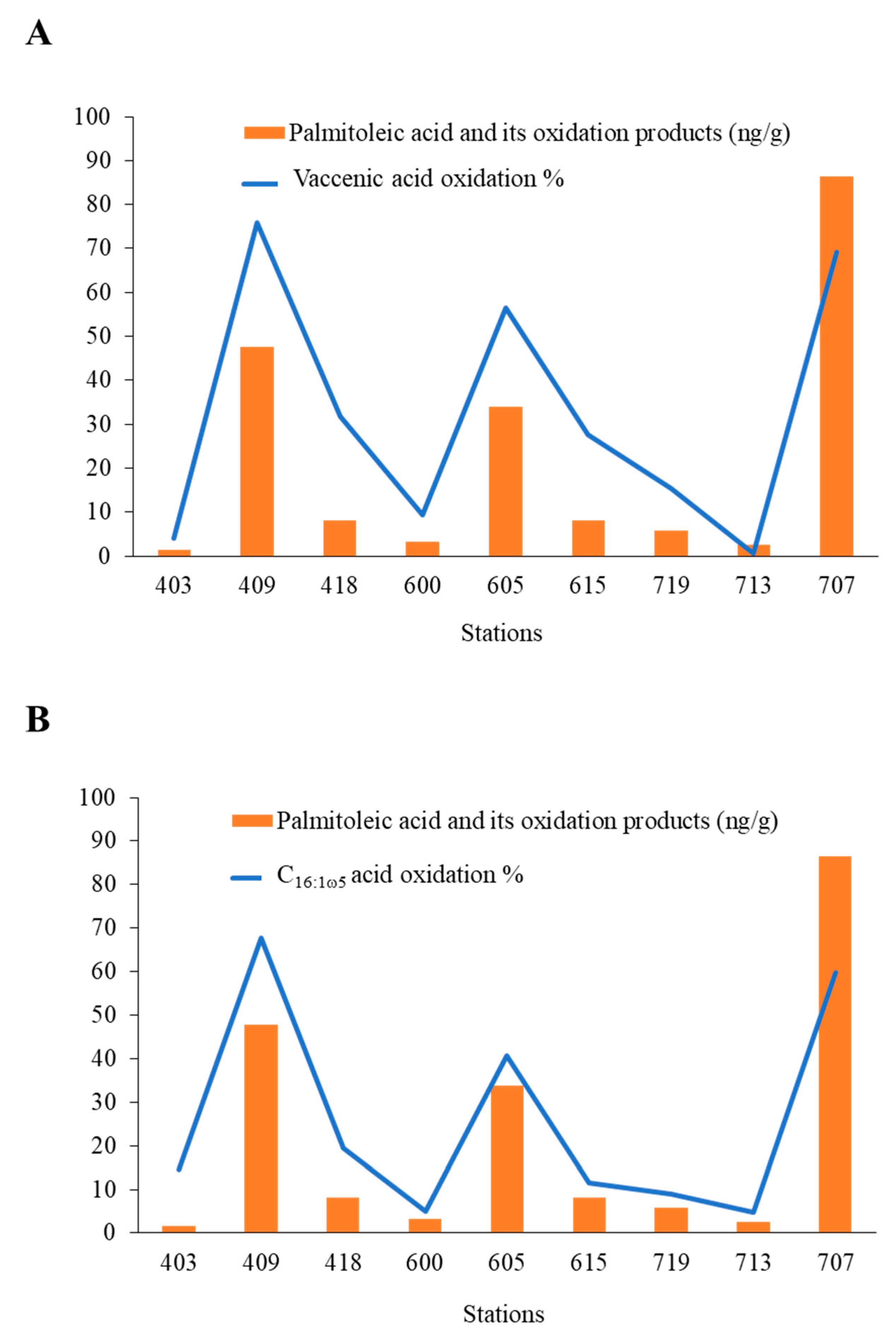
2.2. Transfer of Photochemically Produced 1O2 from Senescent Phytoplanktonic Cells to Their Attached Heterotrophic Bacteria
3. Effect of Polar Matrices Surrounding Phytoplankton Cells on the Transfer of 1O2 from Irradiated Phytodetritus to Their Attached Bacteria
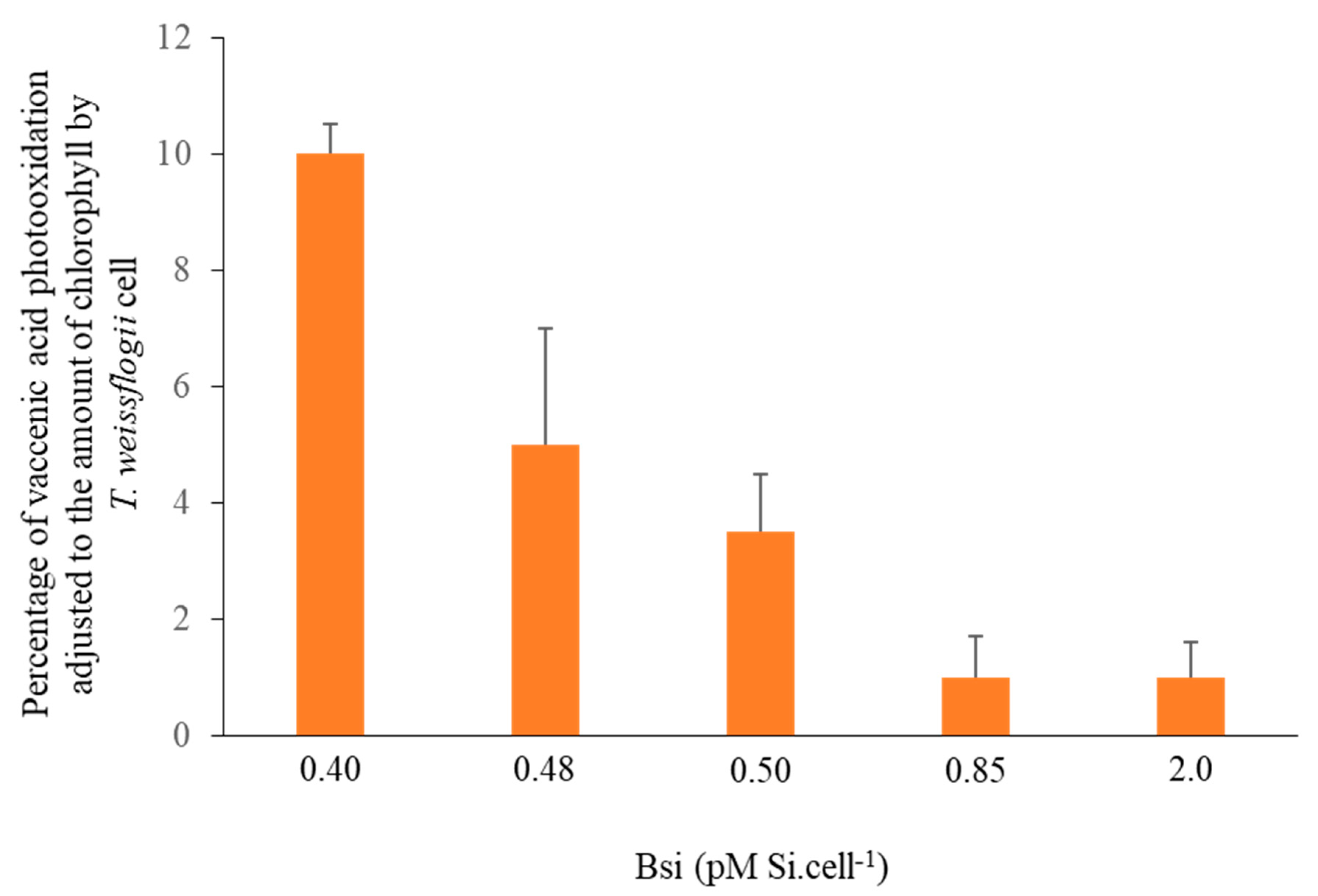
3.1. Silica and Carbonaceous Charged Mineral Surfaces
3.2. Exopolymeric Substances (EPS)
4. Effect of Solar Irradiance Intensity on the Transfer of 1O2 from Irradiated Phytodetritus to Their Attached Bacteria
5. Effect of Bacterial Carotenoid Content on the 1O2 Transfer from Phytodetritus to Attached Bacteria
6. Effect of the Production of 1O2 by Irradiated Phytodetritus on Motile Bacteria
7. Induction of Autoxidative Processes in Bacteria: A Consequence of Photooxidation Processes
8. Impact of the Oxidation of Bacteria Attached to Microalgal Material on Algal Preservation: A Focus on the Arctic
9. Future Research Developments
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Knox, J.P.; Dodge, A.D. Singlet Oxygen and Plants. Phytochemistry 1985, 24, 889–896. [Google Scholar] [CrossRef]
- Foote, C.S. Photosensitized Oxidation and Singlet Oxygen: Consequences in Biological Systems. In Free Radicals in Biology; Elsevier: Amsterdam, The Netherlands, 1976; Volume 2, p. 85. [Google Scholar]
- Halliwell, B. Oxidative Damage, Lipid Peroxidation and Antioxidant Protection in Chloroplasts. Chem. Phys. Lipids 1987, 44, 327–340. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Hendry, G.A. Free Radical Metabolism, Pigment Degradation and Lipid Peroxidation in Leaves during Senescence. Proc. R. Soc. Edinb. Sect. B Biol. Sci. 1994, 102, 459–471. [Google Scholar] [CrossRef]
- Rontani, J.-F. Visible Light-Dependent Degradation of Lipidic Phytoplanktonic Components during Senescence: A Review. Phytochemistry 2001, 58, 187–202. [Google Scholar] [CrossRef]
- Dmitrieva, V.A.; Tyutereva, E.V.; Voitsekhovskaja, O.V. Singlet Oxygen in Plants: Generation, Detection, and Signaling Roles. Int. J. Mol. Sci. 2020, 21, 3237. [Google Scholar] [CrossRef]
- Rontani, J.-F.; Charriere, B.; Forest, A.; Heussner, S.; Vaultier, F.; Petit, M.; Delsaut, N.; Fortier, L.; Sempere, R. Intense Photooxidative Degradation of Planktonic and Bacterial Lipids in Sinking Particles Collected with Sediment Traps across the Canadian Beaufort Shelf (Arctic Ocean). Biogeosciences 2012, 9, 4787–4802. [Google Scholar] [CrossRef]
- Rontani, J.-F.; Amiraux, R.; Smik, L.; Wakeham, S.G.; Paulmier, A.; Vaultier, F.; Sun-Yong, H.; Jun-Oh, M.; Belt, S.T. Type II Photosensitized Oxidation in Senescent Microalgal Cells at Different Latitudes: Does Low under-Ice Irradiance in Polar Regions Enhance Efficiency? Sci. Total Environ. 2021, 779, 146363. [Google Scholar] [CrossRef]
- Rontani, J.-F.; Belt, S.T. Photo-and Autoxidation of Unsaturated Algal Lipids in the Marine Environment: An Overview of Processes, Their Potential Tracers, and Limitations. Org. Geochem. 2020, 139, 103941. [Google Scholar] [CrossRef]
- Frimer, A.A. The Reaction of Singlet Oxygen with Olefins: The Question of Mechanism. Chem. Rev. 1979, 79, 359–387. [Google Scholar] [CrossRef]
- Frankel, E. Lipid Oxidation; The Oily Press LTD.: Dundee, UK, 1998. [Google Scholar]
- Frankel, E.N.; Neff, W.; Bessler, T. Analysis of Autoxidized Fats by Gas Chromatography-Mass Spectrometry: V. Photosensitized Oxidation. Lipids 1979, 14, 961–967. [Google Scholar] [CrossRef]
- Rontani, J.-F. Use of Gas Chromatography-Mass Spectrometry Techniques (GC-MS, GC-MS/MS and GC-QTOF) for the Characterization of Photooxidation and Autoxidation Products of Lipids of Autotrophic Organisms in Environmental Samples. Molecules 2022, 27, 1629. [Google Scholar] [CrossRef] [PubMed]
- Marchand, D.; Rontani, J.-F. Characterisation of Photo-Oxidation and Autoxidation Products of Phytoplanktonic Monounsaturated Fatty Acids in Marine Particulate Matter and Recent Sediments. Org. Geochem. 2001, 32, 287–304. [Google Scholar] [CrossRef]
- Christodoulou, S.; Marty, J.-C.; Miquel, J.-C.; Volkman, J.K.; Rontani, J.-F. Use of Lipids and Their Degradation Products as Biomarkers for Carbon Cycling in the Northwestern Mediterranean Sea. Mar. Chem. 2009, 113, 25–40. [Google Scholar] [CrossRef]
- Perry, G.; Volkman, J.; Johns, R.; Bavor Jr, H. Fatty Acids of Bacterial Origin in Contemporary Marine Sediments. Geochim. Cosmochim. Acta 1979, 43, 1715–1725. [Google Scholar] [CrossRef]
- Volkman, J.; Johns, R.; Gillan, F.; Perry, G.; Bavor Jr, H. Microbial Lipids of an Intertidal Sediment—I. Fatty Acids and Hydrocarbons. Geochim. Cosmochim. Acta 1980, 44, 1133–1143. [Google Scholar] [CrossRef]
- Sicre, M.-A.; Paillasseur, J.-L.; Marty, J.-C.; Saliot, A. Characterization of Seawater Samples Using Chemometric Methods Applied to Biomarker Fatty Acids. Org. Geochem. 1988, 12, 281–288. [Google Scholar] [CrossRef]
- Keweloh, H.; Heipieper, H.J. Trans Unsaturated Fatty Acids in Bacteria. Lipids 1996, 31, 129–137. [Google Scholar] [CrossRef]
- Westbroek, P.; De Jong, E.; Van der Wal, P.; Borman, A.; De Vrind, J.; Kok, D.; De Bruijn, W.; Parker, S. Mechanism of Calcification in the Marine Alga Emiliania Huxleyi. Philos. Trans. R. Soc. London. B Biol. Sci. 1984, 304, 435–444. [Google Scholar]
- Zheng, Q.; Liu, Y.; Jeanthon, C.; Zhang, R.; Lin, W.; Yao, J.; Jiao, N. Geographic Impact on Genomic Divergence as Revealed by Comparison of Nine Citromicrobial Genomes. Appl. Environ. Microbiol. 2016, 82, 7205–7216. [Google Scholar] [CrossRef][Green Version]
- Saini, A.; Panwar, D.; Panesar, P.S.; Bera, M.B. Encapsulation of Functional Ingredients in Lipidic Nanocarriers and Antimicrobial Applications: A Review. Environ. Chem. Lett. 2021, 19, 1107–1134. [Google Scholar] [CrossRef]
- Thiel, V.; Tank, M.; Bryant, D.A. Diversity of Chlorophototrophic Bacteria Revealed in the Omics Era. Annu. Rev. Plant Biol. 2018, 69, 21–49. [Google Scholar] [CrossRef]
- Kolber, Z.S.; Gerald, F.; Plumley; Lang, A.S.; Beatty, J.T.; Blankenship, R.E.; VanDover, C.L.; Vetriani, C.; Koblizek, M.; Rathgeber, C. Contribution of Aerobic Photoheterotrophic Bacteria to the Carbon Cycle in the Ocean. Science 2001, 292, 2492–2495. [Google Scholar] [CrossRef] [PubMed]
- Daniel, C. Regrowth of Subtropical Aerobic Anoxygenic Photoheterotrophic Bacteria upon Grazer Removal: A Case Study of Surface Waters Collected from the Southern Pensacola Bay. Master’s Thesis, University of West Florida, Pensacola, FL, USA, 2022. [Google Scholar]
- Kolber, Z.S.; Van Dover, C.; Niederman, R.; Falkowski, P. Bacterial Photosynthesis in Surface Waters of the Open Ocean. Nature 2000, 407, 177–179. [Google Scholar] [CrossRef] [PubMed]
- Lehours, A.-C.; Enault, F.; Boeuf, D.; Jeanthon, C. Biogeographic Patterns of Aerobic Anoxygenic Phototrophic Bacteria Reveal an Ecological Consistency of Phylogenetic Clades in Different Oceanic Biomes. Sci. Rep. 2018, 8, 4105. [Google Scholar] [CrossRef] [PubMed]
- Gazulla, C.R.; Auladell, A.; Ruiz-González, C.; Junger, P.C.; Royo-Llonch, M.; Duarte, C.M.; Gasol, J.M.; Sánchez, O.; Ferrera, I. Global Diversity and Distribution of Aerobic Anoxygenic Phototrophs in the Tropical and Subtropical Oceans. Environ. Microbiol. 2022, 24, 2222–2238. [Google Scholar] [CrossRef]
- Yang, C.-H.; Huang, K.-S.; Wang, Y.-T.; Shaw, J.-F. A Review of Bacteriochlorophyllides: Chemical Structures and Applications. Molecules 2021, 26, 1293. [Google Scholar] [CrossRef]
- Rontani, J.-F.; Christodoulou, S.; Koblizek, M. GC-MS Structural Characterization of Fatty Acids from Marine Aerobic Anoxygenic Phototrophic Bacteria. Lipids 2005, 40, 97–108. [Google Scholar] [CrossRef]
- Kopejtka, K.; Zeng, Y.; Kaftan, D.; Selyanin, V.; Gardian, Z.; Tomasch, J.; Sommaruga, R.; Koblížek, M. Characterization of the Aerobic Anoxygenic Phototrophic Bacterium Sphingomonas sp. AAP5. Microorganisms 2021, 9, 768. [Google Scholar] [CrossRef] [PubMed]
- Rontani, J.; Koblížek, M.; Beker, B.; Bonin, P.; Kolber, Z.S. On the Origin of Cis-vaccenic Acid Photodegradation Products in the Marine Environment. Lipids 2003, 38, 1085–1092. [Google Scholar] [CrossRef]
- Porter, N.A.; Caldwell, S.E.; Mills, K.A. Mechanisms of Free Radical Oxidation of Unsaturated Lipids. Lipids 1995, 30, 277–290. [Google Scholar] [CrossRef]
- Marchand, D.; Rontani, J.-F. Visible Light-Induced Oxidation of Lipid Components of Purple Sulfur Bacteria: A Significant Process in Microbial Mats. Org. Geochem. 2003, 34, 61–79. [Google Scholar] [CrossRef]
- Unanue, M.; Azúa, I.; Arrieta, J.; Labirua-Iturburu, A.; Egea, L.; Iriberri, J. Bacterial Colonization and Ectoenzymatic Activity in Phytoplankton-Derived Model Particles: Cleavage of Peptides and Uptake of Amino Acids. Microb. Ecol. 1998, 35, 136–146. [Google Scholar] [CrossRef]
- Kiørboe, T.; Grossart, H.-P.; Ploug, H.; Tang, K. Mechanisms and Rates of Bacterial Colonization of Sinking Aggregates. Appl. Environ. Microbiol. 2002, 68, 3996–4006. [Google Scholar] [CrossRef]
- Ogilby, P.R. Singlet Oxygen: There Is Indeed Something New under the Sun. Chem. Soc. Rev. 2010, 39, 3181–3209. [Google Scholar] [CrossRef]
- Klaper, M.; Fudickar, W.; Linker, T. Role of Distance in Singlet Oxygen Applications: A Model System. J. Am. Chem. Soc. 2016, 138, 7024–7029. [Google Scholar] [CrossRef] [PubMed]
- Murotomi, K.; Umeno, A.; Shichiri, M.; Tanito, M.; Yoshida, Y. Significance of Singlet Oxygen Molecule in Pathologies. Int. J. Mol. Sci. 2023, 24, 2739. [Google Scholar] [CrossRef]
- Skovsen, E.; Snyder, J.W.; Lambert, J.D.; Ogilby, P.R. Lifetime and Diffusion of Singlet Oxygen in a Cell. J. Phys. Chem. B 2005, 109, 8570–8573. [Google Scholar] [CrossRef]
- Burot, C. Etude de La Dégradation Des Algues de Glace et Du Phytoplancton d’eau Libre En Zone Arctique: Impact de l’état de Stress Des Bactéries Associées à Ce Matériel Sur Sa Préservation et Sa Contribution Aux Sédiments. Ph.D. Thesis, Aix-Marseille University, Marseille, France, 2022. [Google Scholar]
- Petit, M.; Sempéré, R.; Vaultier, F.; Rontani, J.-F. Photochemical Production and Behavior of Hydroperoxyacids in Heterotrophic Bacteria Attached to Senescent Phytoplanktonic Cells. Int. J. Mol. Sci. 2013, 14, 11795–11815. [Google Scholar] [CrossRef] [PubMed]
- Suwa, K.; Kimura, T.; Schaap, A.P. Reactivity of Singlet Molecular Oxygen with Cholesterol in a Phospholipid Membrane Matrix. A Model for Oxidative Damage of Membranes. Biochem. Biophys. Res. Commun. 1977, 75, 785–792. [Google Scholar] [CrossRef]
- Hurst, J.R.; Schuster, G.B. Nonradiative Relaxation of Singlet Oxygen in Solution. J. Am. Chem. Soc. 1983, 105, 5756–5760. [Google Scholar] [CrossRef]
- Gehlen, M.; Beck, L.; Calas, G.; Flank, A.-M.; Van Bennekom, A.; Van Beusekom, J. Unraveling the Atomic Structure of Biogenic Silica: Evidence of the Structural Association of Al and Si in Diatom Frustules. Geochim. Cosmochim. Acta 2002, 66, 1601–1609. [Google Scholar] [CrossRef]
- Yuan, P.; Liu, D.; Zhou, J.; Tian, Q.; Song, Y.; Wei, H.; Wang, S.; Zhou, J.; Deng, L.; Du, P. Identification of the Occurrence of Minor Elements in the Structure of Diatomaceous Opal Using FIB and TEM-EDS. Am. Mineral. 2019, 104, 1323–1335. [Google Scholar] [CrossRef]
- Tian, Q.; Liu, D.; Yuan, P.; Li, M.; Yang, W.; Zhou, J.; Wei, H.; Zhou, J.; Guo, H. Occurrence of Structural Aluminium (Al) in Marine Diatom Biological Silica: Visible Evidence from Microscopic Analysis. Ocean Sci. 2022, 18, 321–329. [Google Scholar] [CrossRef]
- Walker, J.; Langer, G. Coccolith Crystals: Pure Calcite or Organic-Mineral Composite Structures? Acta Biomater. 2021, 125, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Petit, M.; Suroy, M.; Sempere, R.; Vaultier, F.; Volkman, J.K.; Goutx, M.; Rontani, J.-F. Transfer of Singlet Oxygen from Senescent Irradiated Phytoplankton Cells to Attached Heterotrophic Bacteria: Effect of Silica and Carbonaceous Matrices. Mar. Chem. 2015, 171, 87–95. [Google Scholar] [CrossRef]
- Moore, T.S.; Dowell, M.D.; Franz, B.A. Detection of Coccolithophore Blooms in Ocean Color Satellite Imagery: A Generalized Approach for Use with Multiple Sensors. Remote Sens. Environ. 2012, 117, 249–263. [Google Scholar] [CrossRef]
- Perrot, L.; Gohin, F.; Ruiz-Pino, D.; Lampert, L.; Huret, M.; Dessier, A.; Malestroit, P.; Dupuy, C.; Bourriau, P. Coccolith-Derived Turbidity and Hydrological Conditions in May in the Bay of Biscay. Prog. Oceanogr. 2018, 166, 41–53. [Google Scholar] [CrossRef]
- Suh, H.; Lee, H.; Jung, J. Mycosporine Glycine Protects Biological Systems Against Photodynamic Damage by Quenching Singlet Oxygen with a High Efficiency. Photochem. Photobiol. 2003, 78, 109–113. [Google Scholar] [CrossRef]
- Torres, P.; Santos, J.P.; Chow, F.; Ferreira, M.J.P.; dos Santos, D.Y. Comparative Analysis of in Vitro Antioxidant Capacities of Mycosporine-like Amino Acids (MAAs). Algal Res. 2018, 34, 57–67. [Google Scholar] [CrossRef]
- Ingalls, A.E.; Whitehead, K.; Bridoux, M.C. Tinted Windows: The Presence of the UV Absorbing Compounds Called Mycosporine-like Amino Acids Embedded in the Frustules of Marine Diatoms. Geochim. Cosmochim. Acta 2010, 74, 104–115. [Google Scholar] [CrossRef]
- Weiss, E.L.; Cape, M.R.; Pan, B.J.; Vernet, M.; James, C.C.; Smyth, T.J.; Ha, S.-Y.; Iriarte, J.L.; Mitchell, B.G. The Distribution of Mycosporine-like Amino Acids in Phytoplankton across a Southern Ocean Transect. Front. Mar. Sci. 2022, 9, 2133. [Google Scholar] [CrossRef]
- Xiao, R.; Zheng, Y. Overview of Microalgal Extracellular Polymeric Substances (EPS) and Their Applications. Biotechnol. Adv. 2016, 34, 1225–1244. [Google Scholar] [CrossRef] [PubMed]
- Decho, A.W.; Gutierrez, T. Microbial Extracellular Polymeric Substances (EPSs) in Ocean Systems. Front. Microbiol. 2017, 8, 922. [Google Scholar] [CrossRef] [PubMed]
- Roux, P.; Siano, R.; Collin, K.; Bilien, G.; Sinquin, C.; Marchand, L.; Zykwinska, A.; Delbarre-Ladrat, C.; Schapira, M. Bacteria Enhance the Production of Extracellular Polymeric Substances by the Green Dinoflagellate Lepidodinium Chlorophorum. Sci. Rep. 2021, 11, 4795. [Google Scholar] [CrossRef] [PubMed]
- Amiraux, R.; Belt, S.T.; Vaultier, F.; Galindo, V.; Gosselin, M.; Bonin, P.; Rontani, J.-F. Monitoring Photo-Oxidative and Salinity-Induced Bacterial Stress in the Canadian Arctic Using Specific Lipid Tracers. Mar. Chem. 2017, 194, 89–99. [Google Scholar] [CrossRef]
- Volkman, J. Sterols in Microorganisms. Appl. Microbiol. Biotechnol. 2003, 60, 495–506. [Google Scholar] [CrossRef]
- Amiraux, R.; Jeanthon, C.; Vaultier, F.; Rontani, J. Paradoxical Effects of Temperature and Solar Irradiance on the Photodegradation State of Killed Phytoplankton. J. Phycol. 2016, 52, 475–485. [Google Scholar] [CrossRef]
- Lund-Hansen, L.C.; Bjerg-Nielsen, M.; Stratmann, T.; Hawes, I.; Sorrell, B.K. Upwelling Irradiance below Sea Ice—PAR Intensities and Spectral Distributions. J. Mar. Sci. Eng. 2021, 9, 830. [Google Scholar] [CrossRef]
- Hashimoto, H.; Uragami, C.; Cogdell, R.J. Carotenoids and Photosynthesis. Carotenoids Nat. Biosynth. Regul. Funct. 2016, 79, 111–139. [Google Scholar]
- Wu, M.; Zhu, R.; Lu, J.; Lei, A.; Zhu, H.; Hu, Z.; Wang, J. Effects of Different Abiotic Stresses on Carotenoid and Fatty Acid Metabolism in the Green Microalga Dunaliella Salina Y6. Ann. Microbiol. 2020, 70, 48. [Google Scholar] [CrossRef]
- Sun, T.; Rao, S.; Zhou, X.; Li, L. Plant Carotenoids: Recent Advances and Future Perspectives. Mol. Hortic. 2022, 2, 3. [Google Scholar] [CrossRef]
- Cogdell, R.J.; Frank, H.A. How Carotenoids Function in Photosynthetic Bacteria. Biochim. Biophys. Acta (BBA)-Rev. Bioenerg. 1987, 895, 63–79. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Tselios, C.; Varotsis, C. Photoreduction of Carotenoids in the Aerobic Anoxygenic Photoheterotrophs Probed by Real Time Raman Spectroscopy. J. Photochem. Photobiol. B Biol. 2020, 213, 112069. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, T. Nature and Distribution of Carotenoids. Food Chem. 1980, 5, 3–13. [Google Scholar] [CrossRef]
- Hundle, B.; Alberti, M.; Nievelstein, V.; Beyer, P.; Kleinig, H.; Armstrong, G.; Burke, D.; Hearst, J. Functional Assignment of Erwinia Herbicola Eho10 Carotenoid Genes Expressed in Escherichia Coli. Mol. Gen. Genet. MGG 1994, 245, 406–416. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, P.C. Functional Expression and Extension of Staphylococcal Staphyloxanthin Biosynthetic Pathway in Escherichia Coli. J. Biol. Chem. 2012, 287, 21575–21583. [Google Scholar] [CrossRef]
- Petit, M.; Bonin, P.; Amiraux, R.; Michotey, V.; Guasco, S.; Armitano, J.; Jourlin-Castelli, C.; Vaultier, F.; Méjean, V.; Rontani, J.-F. Dynamic of Bacterial Communities Attached to Lightened Phytodetritus. Environ. Sci. Pollut. Res. 2015, 22, 13681–13692. [Google Scholar] [CrossRef]
- Dahl, T.; Midden, W.R.; Neckers, D. Comparison of Photodynamic Action by Rose Bengal in Gram-positive and Gram-negative Bacteria. Photochem. Photobiol. 1988, 48, 607–612. [Google Scholar] [CrossRef]
- Glaeser, S.P.; Grossart, H.; Glaeser, J. Singlet Oxygen, a Neglected but Important Environmental Factor: Short-term and Long-term Effects on Bacterioplankton Composition in a Humic Lake. Environ. Microbiol. 2010, 12, 3124–3136. [Google Scholar] [CrossRef]
- Lalucat, J.; Bennasar, A.; Bosch, R.; García-Valdés, E.; Palleroni, N.J. Biology of Pseudomonas Stutzeri. Microbiol. Mol. Biol. Rev. 2006, 70, 510–547. [Google Scholar] [CrossRef]
- Biebl, H.; Allgaier, M.; Tindall, B.J.; Koblizek, M.; Lünsdorf, H.; Pukall, R.; Wagner-Döbler, I. Dinoroseobacter Shibae Gen. Nov., Sp. Nov., a New Aerobic Phototrophic Bacterium Isolated from Dinoflagellates. Int. J. Syst. Evol. Microbiol. 2005, 55, 1089–1096. [Google Scholar] [CrossRef]
- Di Mascio, P.; Martinez, G.R.; Miyamoto, S.; Ronsein, G.E.; Medeiros, M.H.; Cadet, J. Singlet Molecular Oxygen Reactions with Nucleic Acids, Lipids, and Proteins. Chem. Rev. 2019, 119, 2043–2086. [Google Scholar] [CrossRef] [PubMed]
- Aerssens, D.; Cadoni, E.; Tack, L.; Madder, A. A Photosensitized Singlet Oxygen (1O2) Toolbox for Bio-Organic Applications: Tailoring 1O2 Generation for DNA and Protein Labelling, Targeting and Biosensing. Molecules 2022, 27, 778. [Google Scholar] [CrossRef] [PubMed]
- Ravanat, J.; Dumont, E. Reactivity of Singlet Oxygen with DNA, an Update. Photochem. Photobiol. 2022, 98, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Ding, P.; Xie, C.; Ye, C.; Ye, M.; Pan, C.; Cao, X.; Zhang, S.; Zheng, S. Potential Application of the Oxidative Nucleic Acid Damage Biomarkers in Detection of Diseases. Oncotarget 2017, 8, 75767. [Google Scholar] [CrossRef] [PubMed]
- Malyarchuk, S.; Youngblood, R.; Landry, A.M.; Quillin, E.; Harrison, L. The Mutation Frequency of 8-Oxo-7,8-Dihydroguanine (8-OxodG) Situated in a Multiply Damaged Site: Comparison of a Single and Two Closely Opposed 8-OxodG in Escherichia Coli. DNA Repair 2003, 2, 695–705. [Google Scholar] [CrossRef]
- Glaeser, J.; Nuss, A.; Berghoff, B.; Klug, G. Singlet Oxygen Stress in Microorganisms. Adv. Microb. Physiol. 2011, 58, 141–173. [Google Scholar]
- Ighodaro, O.; Akinloye, O. First Line Defence Antioxidants-Superoxide Dismutase (SOD), Catalase (CAT) and Glutathione Peroxidase (GPX): Their Fundamental Role in the Entire Antioxidant Defence Grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Michaels, M.; Miller, J.H. The GO System Protects Organisms from the Mutagenic Effect of the Spontaneous Lesion 8-Hydroxyguanine (7,8-Dihydro-8-Oxoguanine). J. Bacteriol. 1992, 174, 6321–6325. [Google Scholar] [CrossRef]
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA Damage, Repair, and Mutagenesis. Environ. Mol. Mutagen. 2017, 58, 235–263. [Google Scholar] [CrossRef]
- Conn, P.F.; Schalch, W.; Truscott, T.G. The Singlet Oxygen and Carotenoid Interaction. J. Photochem. Photobiol. B Biol. 1993, 17, 89. [Google Scholar] [CrossRef]
- Ezraty, B.; Gennaris, A.; Barras, F.; Collet, J.-F. Oxidative Stress, Protein Damage and Repair in Bacteria. Nat. Rev. Microbiol. 2017, 15, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Fasnacht, M.; Polacek, N. Oxidative Stress in Bacteria and the Central Dogma of Molecular Biology. Front. Mol. Biosci. 2021, 8, 671037. [Google Scholar] [CrossRef] [PubMed]
- Lim, P.; Wuenschell, G.E.; Holland, V.; Lee, D.-H.; Pfeifer, G.P.; Rodriguez, H.; Termini, J. Peroxyl Radical Mediated Oxidative DNA Base Damage: Implications for Lipid Peroxidation Induced Mutagenesis. Biochemistry 2004, 43, 15339–15348. [Google Scholar] [CrossRef]
- Cadet, J.; Wagner, J.R. DNA Base Damage by Reactive Oxygen Species, Oxidizing Agents, and UV Radiation. Cold Spring Harb. Perspect. Biol. 2013, 5, a012559. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.-G.; Meng, Y.; Johnson, J.; Szabó, P.E.; Pfeifer, G.P. Concordance of Hydrogen Peroxide–Induced 8-Oxo-Guanine Patterns with Two Cancer Mutation Signatures of Upper GI Tract Tumors. Sci. Adv. 2022, 8, eabn3815. [Google Scholar] [CrossRef]
- Omar, N.M.; Prášil, O.; McCain, J.S.P.; Campbell, D.A. Diffusional Interactions among Marine Phytoplankton and Bacterioplankton: Modelling H2O2 as a Case Study. Microorganisms 2022, 10, 821. [Google Scholar] [CrossRef] [PubMed]
- Omar, N.M.; Fleury, K.; Beardsall, B.; Prášil, O.; Campbell, D.A. Genomic Capacities for Reactive Oxygen Species Metabolism across Marine Phytoplankton. PLoS ONE 2023, 18, e0284580. [Google Scholar] [CrossRef]
- Hahnke, S.; Tindall, B.J.; Schumann, P.; Sperling, M.; Brinkhoff, T.; Simon, M. Planktotalea Frisia Gen. Nov., Sp. Nov., Isolated from the Southern North Sea. Int. J. Syst. Evol. Microbiol. 2012, 62, 1619–1624. [Google Scholar] [CrossRef]
- Zou, S.; Zhang, Q.; Zhang, X.; Dupuy, C.; Gong, J. Environmental Factors and Pollution Stresses Select Bacterial Populations in Association with Protists. Front. Mar. Sci. 2020, 7, 659. [Google Scholar] [CrossRef]
- Shin, H.; Lee, E.; Shin, J.; Ko, S.-R.; Oh, H.-S.; Ahn, C.-Y.; Oh, H.-M.; Cho, B.-K.; Cho, S. Elucidation of the Bacterial Communities Associated with the Harmful Microalgae Alexandrium Tamarense and Cochlodinium Polykrikoides Using Nanopore Sequencing. Sci. Rep. 2018, 8, 5323. [Google Scholar] [CrossRef] [PubMed]
- Barak-Gavish, N.; Frada, M.J.; Ku, C.; Lee, P.A.; DiTullio, G.R.; Malitsky, S.; Aharoni, A.; Green, S.J.; Rotkopf, R.; Kartvelishvily, E. Bacterial Virulence against an Oceanic Bloom-Forming Phytoplankter Is Mediated by Algal DMSP. Sci. Adv. 2018, 4, eaau5716. [Google Scholar] [CrossRef] [PubMed]
- Ramanan, R.; Kim, B.-H.; Cho, D.-H.; Oh, H.-M.; Kim, H.-S. Algae–Bacteria Interactions: Evolution, Ecology and Emerging Applications. Biotechnol. Adv. 2016, 34, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Dang, H.; Lovell, C.R. Bacterial Primary Colonization and Early Succession on Surfaces in Marine Waters as Determined by Amplified RRNA Gene Restriction Analysis and Sequence Analysis of 16S RRNA Genes. Appl. Environ. Microbiol. 2000, 66, 467–475. [Google Scholar] [CrossRef]
- Wagner-Döbler, I.; Thiel, V.; Eberl, L.; Allgaier, M.; Bodor, A.; Meyer, S.; Ebner, S.; Hennig, A.; Pukall, R.; Schulz, S. Discovery of Complex Mixtures of Novel Long-chain Quorum Sensing Signals in Free-living and Host-associated Marine Alphaproteobacteria. ChemBioChem 2005, 6, 2195–2206. [Google Scholar] [CrossRef]
- Long, R.A.; Azam, F. Microscale Patchiness of Bacterioplankton Assemblage Richness in Seawater. Aquat. Microb. Ecol. 2001, 26, 103–113. [Google Scholar] [CrossRef]
- Wadhwa, N.; Berg, H.C. Bacterial Motility: Machinery and Mechanisms. Nat. Rev. Microbiol. 2022, 20, 161–173. [Google Scholar] [CrossRef]
- Palma, V.; Gutiérrez, M.S.; Vargas, O.; Parthasarathy, R.; Navarrete, P. Methods to Evaluate Bacterial Motility and Its Role in Bacterial–Host Interactions. Microorganisms 2022, 10, 563. [Google Scholar] [CrossRef]
- Amin, S.A.; Parker, M.S.; Armbrust, E.V. Interactions between Diatoms and Bacteria. Microbiol. Mol. Biol. Rev. 2012, 76, 667–684. [Google Scholar] [CrossRef]
- Mayali, X. Metabolic Interactions between Bacteria and Phytoplankton. Front. Microbiol. 2018, 9, 727. [Google Scholar] [CrossRef]
- Cirri, E.; Pohnert, G. Algae− Bacteria Interactions That Balance the Planktonic Microbiome. New Phytol. 2019, 223, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Sonnenschein, E.C.; Syit, D.A.; Grossart, H.-P.; Ullrich, M.S. Chemotaxis of Marinobacter Adhaerens and Its Impact on Attachment to the Diatom Thalassiosira Weissflogii. Appl. Environ. Microbiol. 2012, 78, 6900–6907. [Google Scholar] [CrossRef] [PubMed]
- Boyeldieu, A.; Poli, J.; Ali Chaouche, A.; Fierobe, H.; Giudici-Orticoni, M.; Méjean, V.; Jourlin-Castelli, C. Multiple Detection of Both Attractants and Repellents by the DCache-chemoreceptor SO_1056 of Shewanella Oneidensis. FEBS J. 2022, 289, 6752–6766. [Google Scholar] [CrossRef]
- Li, S.; Chu, Y.; Xie, P.; Xie, Y.; Chang, H.; Ho, S.-H. Insights into the Microalgae-Bacteria Consortia Treating Swine Wastewater: Symbiotic Mechanism and Resistance Genes Analysis. Bioresour. Technol. 2022, 349, 126892. [Google Scholar] [CrossRef]
- Ożog, Ł.; Aebisher, D. Singlet Oxygen Lifetime and Diffusion Measurements. Eur. J. Clin. Exp. Med. 2018, 16, 123–126. [Google Scholar] [CrossRef]
- Cosa, G. Singlet Oxygen: Applications in Biosciences and Nanosciences; Royal Society of Chemistry: London, UK, 2016; ISBN 1-78262-220-9. [Google Scholar]
- Schaich, K.; Shahidi, F. Bailey’s Industrial Oil and Fat Products; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Rontani, J.-F.; Lalande, C.; Vilgrain, L.; Vaultier, F.; Amiraux, R. Control of the Preservation of Sympagic Algal Material in Surficial Sediments of Central and Eastern Baffin Bay by Bactericidal Hydroperoxides and Free Fatty Acids. Mar. Chem. 2022, 247, 104177. [Google Scholar] [CrossRef]
- Pretorius, C.J.; Zeiss, D.R.; Dubery, I.A. The Presence of Oxygenated Lipids in Plant Defense in Response to Biotic Stress: A Metabolomics Appraisal. Plant Signal. Behav. 2021, 16, 1989215. [Google Scholar] [CrossRef]
- Anderson, S.; Barton, A.; Clayton, S.; Dutkiewicz, S.; Rynearson, T. Marine Phytoplankton Functional Types Exhibit Diverse Responses to Thermal Change. Nat. Commun. 2021, 12, 6413. [Google Scholar] [CrossRef]
- Ducklow, H.; Kirchman, D.; Quinby, H.; Carlson, C.; Dam, H. Stocks and Dynamics of Bacterioplankton Carbon during the Spring Bloom in the Eastern North Atlantic Ocean. Deep Sea Res. Part II Top. Stud. Oceanogr. 1993, 40, 245–263. [Google Scholar] [CrossRef]
- Claustre, H.; Legendre, L.; Boyd, P.W.; Levy, M. The Oceans’ Biological Carbon Pumps: Framework for a Research Observational Community Approach. Front. Mar. Sci. 2021, 8, 780052. [Google Scholar] [CrossRef]
- Burdige, D.J. Preservation of Organic Matter in Marine Sediments: Controls, Mechanisms, and an Imbalance in Sediment Organic Carbon Budgets? Chem. Rev. 2007, 107, 467–485. [Google Scholar] [CrossRef] [PubMed]
- Zonneveld, K.A.; Versteegh, G.J.; Kasten, S.; Eglinton, T.I.; Emeis, K.-C.; Huguet, C.; Koch, B.P.; de Lange, G.J.; de Leeuw, J.W.; Middelburg, J.J. Selective Preservation of Organic Matter in Marine Environments; Processes and Impact on the Sedimentary Record. Biogeosciences 2010, 7, 483–511. [Google Scholar] [CrossRef]
- Faust, J.C.; Ascough, P.; Hilton, R.G.; Stevenson, M.A.; Hendry, K.R.; März, C. New Evidence for Preservation of Contemporary Marine Organic Carbon by Iron in Arctic Shelf Sediments. Environ. Res. Lett. 2022, 18, 014006. [Google Scholar] [CrossRef]
- Hartnett, H.E.; Keil, R.G.; Hedges, J.I.; Devol, A.H. Influence of Oxygen Exposure Time on Organic Carbon Preservation in Continental Margin Sediments. Nature 1998, 391, 572–575. [Google Scholar] [CrossRef]
- Hou, Y.; Torres, M.A. Autogenic Signals in the Sedimentary Record of Organic Carbon Preservation. Geophys. Res. Lett. 2022, 49, e2021GL097654. [Google Scholar] [CrossRef]
- Hemingway, J.D.; Rothman, D.H.; Grant, K.E.; Rosengard, S.Z.; Eglinton, T.I.; Derry, L.A.; Galy, V.V. Mineral Protection Regulates Long-Term Global Preservation of Natural Organic Carbon. Nature 2019, 570, 228–231. [Google Scholar] [CrossRef]
- Amiraux, R.; Patricia, B.; Christopher, B.; Jean-François, R. Use of Stress Signals of Their Attached Bacteria to Monitor Sympagic Algae Preservation in Canadian Arctic Sediments. Microorganisms 2021, 9, 2626. [Google Scholar] [CrossRef]
- Boetius, A.; Albrecht, S.; Bakker, K.; Bienhold, C.; Felden, J.; Fernández-Méndez, M.; Hendricks, S.; Katlein, C.; Lalande, C.; Krumpen, T. Export of Algal Biomass from the Melting Arctic Sea Ice. Science 2013, 339, 1430–1432. [Google Scholar] [CrossRef]
- Yunda-Guarin, G.; Brown, T.A.; Michel, L.N.; Saint-Beat, B.; Amiraux, R.; Nozais, C.; Archambault, P. Reliance of Deep-Sea Benthic Macrofauna on Ice-Derived Organic Matter Highlighted by Multiple Trophic Markers during Spring in Baffin Bay, Canadian Arctic. Elem. Sci. Anthr. 2020, 8, 47. [Google Scholar] [CrossRef]
- Riebesell, U.; Schloss, I.; Smetacek, V. Aggregation of Algae Released from Melting Sea Ice: Implications for Seeding and Sedimentation. Polar Biol. 1991, 11, 239–248. [Google Scholar] [CrossRef]
- Marmillot, V.; Parrish, C.C.; Tremblay, J.-É.; Gosselin, M.; MacKinnon, J.F. Environmental and Biological Determinants of Algal Lipids in Western Arctic and Subarctic Seas. Front. Environ. Sci. 2020, 8, 538635. [Google Scholar] [CrossRef]
- Guezennec, J.; Fiala-Medioni, A. Bacterial Abundance and Diversity in the Barbados Trench Determined by Phospholipid Analysis. FEMS Microbiol. Ecol. 1996, 19, 83–93. [Google Scholar] [CrossRef]
- Blumenberg, M.; Seifert, R.; Nauhaus, K.; Pape, T.; Michaelis, W. In Vitro Study of Lipid Biosynthesis in an Anaerobically Methane-Oxidizing Microbial Mat. Appl. Environ. Microbiol. 2005, 71, 4345–4351. [Google Scholar] [CrossRef] [PubMed]
- Girotti, A.W. Lipid Hydroperoxide Generation, Turnover, and Effector Action in Biological Systems. J. Lipid Res. 1998, 39, 1529–1542. [Google Scholar] [CrossRef]
- Tamburini, C.; Boutrif, M.; Garel, M.; Colwell, R.R.; Deming, J.W. Prokaryotic Responses to Hydrostatic Pressure in the Ocean–a Review. Environ. Microbiol. 2013, 15, 1262–1274. [Google Scholar] [CrossRef]
- Rapp, J.Z.; Fernández-Méndez, M.; Bienhold, C.; Boetius, A. Effects of Ice-Algal Aggregate Export on the Connectivity of Bacterial Communities in the Central Arctic Ocean. Front. Microbiol. 2018, 9, 1035. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Hyun, D.-W.; Lee, J.-Y.; Kim, P.S.; Whon, T.W.; Kang, W.; Bae, J.-W. Sedimentitalea Todarodis Sp. Nov., Isolated from the Intestinal Tract of a Japanese Flying Squid. Int. J. Syst. Evol. Microbiol. 2016, 66, 3293–3298. [Google Scholar] [CrossRef] [PubMed]
- Deboever, E.; Lins, L.; Ongena, M.; De Clerck, C.; Deleu, M.; Fauconnier, M.-L. Linolenic Fatty Acid Hydroperoxide Acts as Biocide on Plant Pathogenic Bacteria: Biophysical Investigation of the Mode of Action. Bioorg. Chem. 2020, 100, 103877. [Google Scholar]
- Deboever, E.; Deleu, M.; Mongrand, S.; Lins, L.; Fauconnier, M.-L. Plant–Pathogen Interactions: Underestimated Roles of Phyto-Oxylipins. Trends Plant Sci. 2020, 25, 22–34. [Google Scholar] [CrossRef]
- Desbois, A.P.; Smith, V.J. Antibacterial Free Fatty Acids: Activities, Mechanisms of Action and Biotechnological Potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rontani, J.-F.; Bonin, P. Cellular Damage of Bacteria Attached to Senescent Phytoplankton Cells as a Result of the Transfer of Photochemically Produced Singlet Oxygen: A Review. Microorganisms 2023, 11, 1565. https://doi.org/10.3390/microorganisms11061565
Rontani J-F, Bonin P. Cellular Damage of Bacteria Attached to Senescent Phytoplankton Cells as a Result of the Transfer of Photochemically Produced Singlet Oxygen: A Review. Microorganisms. 2023; 11(6):1565. https://doi.org/10.3390/microorganisms11061565
Chicago/Turabian StyleRontani, Jean-François, and Patricia Bonin. 2023. "Cellular Damage of Bacteria Attached to Senescent Phytoplankton Cells as a Result of the Transfer of Photochemically Produced Singlet Oxygen: A Review" Microorganisms 11, no. 6: 1565. https://doi.org/10.3390/microorganisms11061565
APA StyleRontani, J.-F., & Bonin, P. (2023). Cellular Damage of Bacteria Attached to Senescent Phytoplankton Cells as a Result of the Transfer of Photochemically Produced Singlet Oxygen: A Review. Microorganisms, 11(6), 1565. https://doi.org/10.3390/microorganisms11061565





