Comprehensive Overview of Toxoplasma gondii-Induced and Associated Diseases
Abstract
1. Introduction
2. Toxoplasma gondii Pathogenesis
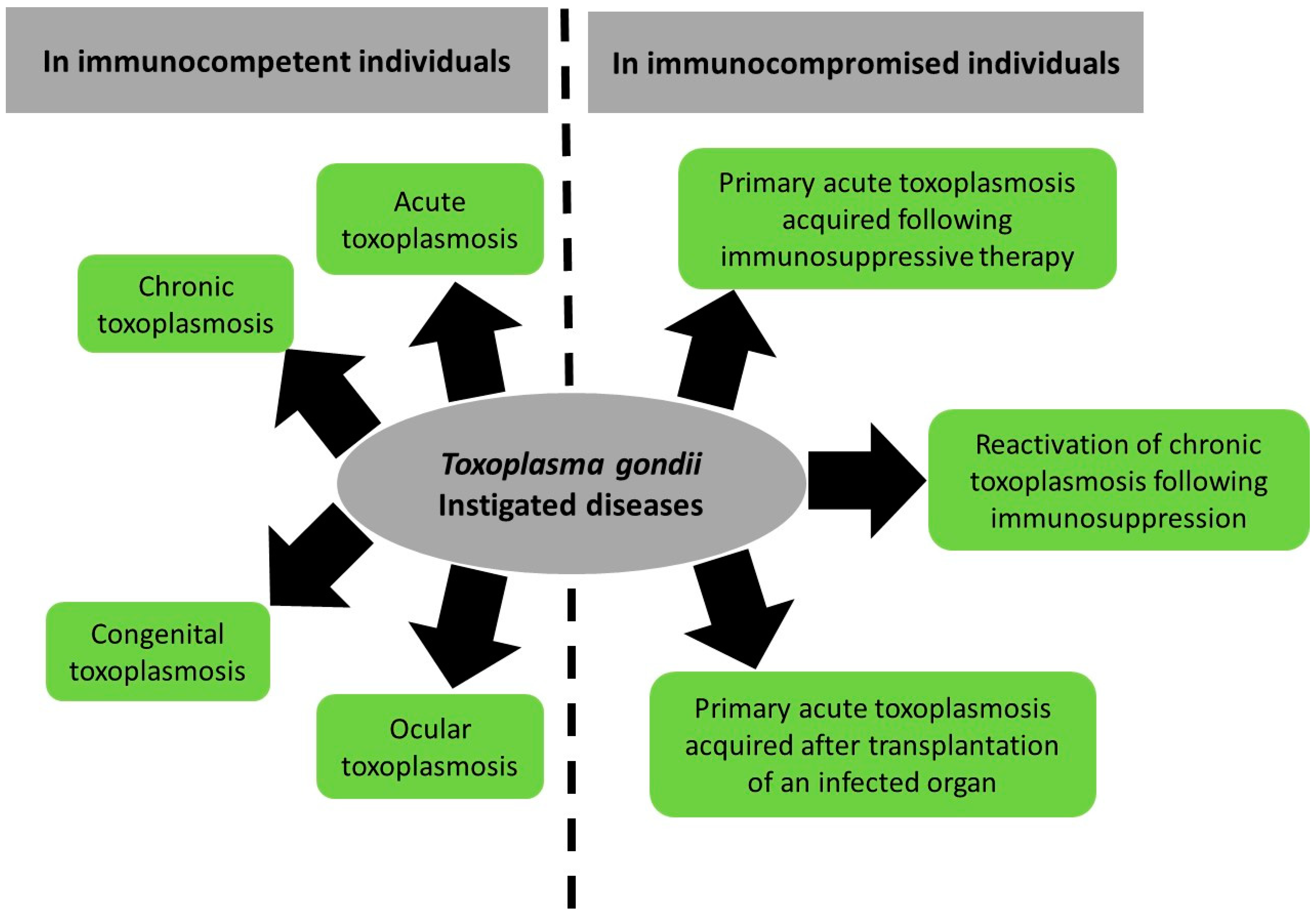
2.1. Toxoplasmosis in Immunocompetent Patients
2.1.1. Acute Toxoplasmosis
2.1.2. Congenital Toxoplasmosis
2.1.3. Ocular Toxoplasmosis
2.1.4. Chronic Toxoplasmosis
2.2. Toxoplasmosis in Immunocompromised Patients
3. Toxoplasma gondii-Associated Diseases
3.1. Toxoplasma gondii and Primary Neuropathies
3.1.1. Toxoplasmosis and Multiple Sclerosis
3.1.2. Toxoplasmosis and Epilepsy
3.1.3. Toxoplasmosis and Parkinson’s and Alzheimer’s Neuropathies
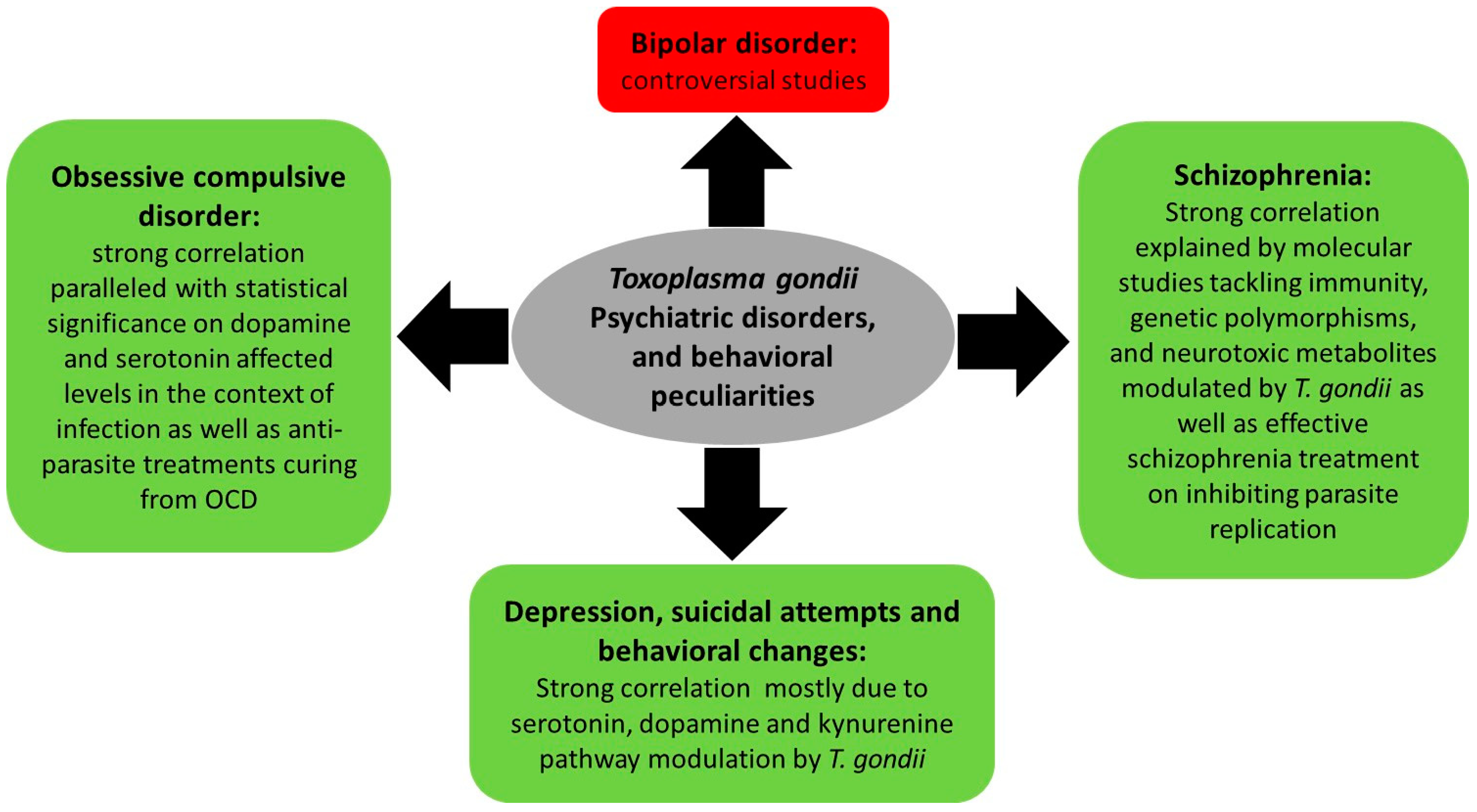
3.2. Toxoplasma gondii, Psychiatric and Behavioral Disorders
3.2.1. Toxoplasmosis, Depression, and Behavioral Changes
3.2.2. Toxoplasmosis and Schizophrenia
3.2.3. Toxoplasmosis and Bipolar Disorder
3.2.4. Toxoplasmosis and Obsessive Compulsive Disorder
3.3. Toxoplasma gondii and Cancers: Modulation of miRNAs as One Molecular Explanation of Toxoplasma-Associated Brain Cancers
4. Current Treatments
5. Toxoplasmosis and Prophylaxis: Available and Potential Vaccine Strategies
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Skariah, S.; McIntyre, M.K.; Mordue, D.G. Toxoplasma gondii: Determinants of tachyzoite to bradyzoite conversion. Parasitol. Res. 2010, 107, 253–260. [Google Scholar] [CrossRef]
- Robert-Gangneux, F.; Dardé, M.-L. Epidemiology of and Diagnostic Strategies for Toxoplasmosis. Clin. Microbiol. Rev. 2012, 25, 264. [Google Scholar] [CrossRef]
- Ben-Harari, R.R.; Connolly, M.P. High burden and low awareness of toxoplasmosis in the United States. Postgrad. Med. 2019, 131, 103–108. [Google Scholar] [CrossRef]
- Montoya, J.G.; Liesenfeld, O. Toxoplasmosis. Lancet 2004, 363, 1965–1976. [Google Scholar] [CrossRef]
- Reza Yazdani, M. Frequency of sero-positivity in household members of the patients with positive toxoplasma serology. Rev. Esp. Quimioter. 2018, 31, 506–510. [Google Scholar] [PubMed]
- Sibley, L.D.; Boothroyd, J.C. Virulent strains of Toxoplasma gondii comprise a single clonal lineage. Nature 1992, 359, 82–85. [Google Scholar] [CrossRef]
- Mendez, O.A.; Koshy, A.A. Toxoplasma gondii: Entry, association, and physiological influence on the central nervous system. PLoS Pathog. 2017, 13, e1006351. [Google Scholar] [CrossRef] [PubMed]
- Lambert, H. Induction of dendritic cell migration upon Toxoplasma gondii infection potentiates parasite dissemination. Cell Microbiol. 2006, 8, 1611–1623. [Google Scholar] [CrossRef] [PubMed]
- Lachenmaier, S.M.; Deli, M.A.; Meissner, M.; Liesenfeld, O. Intracellular transport of Toxoplasma gondii through the blood-brain barrier. J. Neuroimmunol. 2011, 232, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Ueno, N.; Harker, K.S.; Clarke, E.C.; McWhorter, F.Y.; Liu, W.F.; Tenner, A.J.; Lodoen, M.B. Real-time imaging of Toxoplasma-infected human monocytes under fluidic shear stress reveals rapid translocation of intracellular parasites across endothelial barriers. Cell Microbiol. 2014, 16, 580–595. [Google Scholar] [CrossRef] [PubMed]
- Courret, N.; Darche, S.; Sonigo, P.; Milon, G.; Buzoni-Gâtel, D. Isabelle Tardieux CD11c- and CD11b-expressing mouse leukocytes transport single Toxoplasma gondii tachyzoites to the brain. Blood 2006, 107, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Dobrowolski, J.M.; Sibley, L.D. Toxoplasma invasion of mammalian cells is powered by the actin cytoskeleton of the parasite. Cell 1996, 84, 933–939. [Google Scholar] [CrossRef]
- Lindsay, D.S.; Dubey, J.P. Toxoplasma gondii: The changing paradigm of congenital toxoplasmosis. Parasitology 2011, 138, 1829–1831. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, L.; Targa, L.S.; Sumita, L.M.; Shimokawa, P.T.; Rodrigues, J.C.; Kanunfre, K.A.; Okay, T.S. Association of Parasite Load Levels in Amniotic Fluid With Clinical Outcome in Congenital Toxoplasmosis. Obstet. Gynecol. 2017, 130, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Robert-Gangneux, F.; Murat, J.B.; Fricker-Hidalgo, H.F.; Brenier-Pincat, M.-P.; Gangneux, J.-P.; Pelloux, H. The placenta: A main role in congenital toxoplasmosis? Trends Parasitol. 2011, 27, 530–536. [Google Scholar] [CrossRef] [PubMed]
- McAuley, J.B. Congenital Toxoplasmosis. J. Pediatric Infect. Dis. Soc. 2014, 3, S30–S35. [Google Scholar] [CrossRef]
- Singh, S. Congenital toxoplasmosis: Clinical features, outcomes, treatment, and prevention. Trop. Parasitol. 2016, 6, 113–122. [Google Scholar] [CrossRef]
- Weiss, L.M.; Dubey, J.P. Toxoplasmosis: A history of clinical observations. Int. J. Parasitol. 2009, 39, 895–901. [Google Scholar] [CrossRef]
- Nowakowska, D.; Stray-Pedersen, B.; Spiewak, E.; Sobala, W.; Małafiej, E.; Wilczyński, J. Prevalence and estimated incidence of Toxoplasma infection among pregnant women in Poland: A decreasing trend in the younger population. Clin. Microbiol. Infect. 2006, 12, 913–917. [Google Scholar] [CrossRef]
- Nahouli, H.; Arnaout, N.E.; Chalhoub, E.; Anastadiadis, E.; Hajj, H.E. Seroprevalence of Anti-Toxoplasma gondii Antibodies Among Lebanese Pregnant Women. Vector Borne Zoonotic Dis. 2017, 17, 785–790. [Google Scholar] [CrossRef]
- Galal, L.; Sarry, A.; Cuny, T.; Brouat, C.; Coulibaly, F.; Sembène, M.; Diagne, M.; Diallo, M.; Sow, A.; Hamidović, A.; et al. The introduction of new hosts with human trade shapes the extant distribution of Toxoplasma gondii lineages. PLoS Negl. Trop. Dis. 2019, 13, e0007435. [Google Scholar] [CrossRef] [PubMed]
- Delhaes, L.; Ajzenberg, D.; Sicot, B.; Bourgeot, P.; Dardé, M.-U.; Dei-Cas, E.; Houfflin-Debarge, V. Severe congenital toxoplasmosis due to a Toxoplasma gondii strain with an atypical genotype: Case report and review. Prenat. Diagn. 2010, 30, 902–905. [Google Scholar] [CrossRef] [PubMed]
- Vallochi, A.L.; Goldberg, A.C.; Angela Falcai, A.; Ramasawmy, R.; Jorge Kalil, J.; Silveira, C.; Belfort, R.; Rizzo, L.V. Molecular markers of susceptibility to ocular toxoplasmosis, host and guest behaving badly. Clin. Ophthalmol. 2008, 2, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Atmaca, L.S.; Simsek, T.; Batioglu, F. Clinical features and prognosis in ocular toxoplasmosis. Jpn. J. Ophthalmol. 2004, 48, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Delair, E.; Latkany, P.; Noble, A.G.; Rabiah, P.; McLeod, R.; Brezin, A. Clinical manifestations of ocular toxoplasmosis. Ocul. Immunol. Inflamm. 2011, 19, 91–102. [Google Scholar] [CrossRef]
- Vasconcelos-Santos, D.V.; Dodds, E.M.; Orefice, F. Review for disease of the year: Differential diagnosis of ocular toxoplasmosis. Ocul. Immunol. Inflamm. 2011, 19, 171–179. [Google Scholar] [CrossRef]
- Sauer, A.; Rochet, E.; Lahmar, I.; Brunet, J.; Sabou, M.; Bourcier, T.; Candolfi, E.; Pfaff, A.W. The local immune response to intraocular Toxoplasma re-challenge: Less pathology and better parasite control through Treg/Th1/Th2 induction. Int. J. Parasitol. 2013, 43, 721–728. [Google Scholar] [CrossRef]
- Schlüter, D.; Barragan, A. Advances and Challenges in Understanding Cerebral Toxoplasmosis. Front. Immunol. 2019, 10, 242. [Google Scholar] [CrossRef]
- Blanchard, N.; Dunay, I.R.; Schlüter, D. Persistence of Toxoplasma gondii in the central nervous system: A fine-tuned balance between the parasite, the brain and the immune system. Parasite Immunol. 2015, 37, 150–158. [Google Scholar] [CrossRef]
- Matta, S.K.; Rinkenberger, N.; Dunay, I.R.; Sibley, L.D. Toxoplasma gondii infection and its implications within the central nervous system. Nat. Rev. Microbiol. 2021, 19, 467–480. [Google Scholar] [CrossRef]
- Harker, K.S.; Ueno, N.; Wang, T.; Bonhomme, C.; Liu, W.; Lodoen, M.B. Toxoplasma gondii modulates the dynamics of human monocyte adhesion to vascular endothelium under fluidic shear stress. J. Leukoc. Biol. 2013, 93, 789–800. [Google Scholar] [CrossRef]
- Torgerson, P.R.; Devleesschauwer, B.; Praet, N.; Speybroeck, N.; Willingham, A.L.; Kasuga, F.; Rokni, M.B.; Zhou, X.; Fèvre, E.M.; Sripa, B.; et al. World Health Organization Estimates of the Global and Regional Disease Burden of 11 Foodborne Parasitic Diseases, 2010: A Data Synthesis. PLoS Med. 2015, 12, e1001920. [Google Scholar] [CrossRef] [PubMed]
- Cekanaviciute, E.; Dietrich, H.K.; Axtell, R.C.; Williams, A.M.; Egusquiza, R.; Wai, K.M.; Koshy, A.A.; Buckwalter, M.S. Astrocytic TGF-beta signaling limits inflammation and reduces neuronal damage during central nervous system Toxoplasma infection. J. Immunol. 2014, 193, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Halonen, S.K.; Chiu, F.; Weiss, L.M. Effect of cytokines on growth of Toxoplasma gondii in murine astrocytes. Infect. Immun. 1998, 66, 4989–4993. [Google Scholar] [CrossRef]
- Miller, K.D.; Schnell, M.J.; Rall, G.F. Keeping it in check: Chronic viral infection and antiviral immunity in the brain. Nat. Rev. Neurosci. 2016, 17, 766–776. [Google Scholar] [CrossRef]
- Schluter, D.; Deckert, M.; Hof, H.; Frei, K. Toxoplasma gondii infection of neurons induces neuronal cytokine and chemokine production, but gamma interferon and tumor necrosis factor-stimulated neurons fail to inhibit the invasion and growth of T. gondii. Infect. Immun. 2001, 69, 7889–7893. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Conley, F.K.; Remington, J.S. Importance of endogenous IFN-gamma for prevention of toxoplasmic encephalitis in mice. J. Immunol. 1989, 143, 2045–2050. [Google Scholar]
- Evans, A.K.; Strassmann, P.S.; Lee, I.P.; Sapolsky, R.M. Patterns of Toxoplasma gondii cyst distribution in the forebrain associate with individual variation in predator odor avoidance and anxiety-related behavior in male Long-Evans rats. Brain Behav. Immun. 2014, 37, 122–133. [Google Scholar] [CrossRef]
- Hermes, G.; Ajioka, J.W.; Kelly, K.A.; Mui, E.; Roberts, F.; Kasza, K.; Mayr, T.; Kirisits, M.J.; Wollmann, R.; Ferguson, D.J.P.; et al. Neurological and behavioral abnormalities, ventricular dilatation, altered cellular functions, inflammation, and neuronal injury in brains of mice due to common, persistent, parasitic infection. J. Neuroinflamm. 2008, 5, 48. [Google Scholar] [CrossRef]
- Xiao, J.; Li, Y.; Gressitt, K.L.; He, H.; Kannan, G.; Schultz, T.L.; Svezhova, N.; Carruthers, V.B.; Pletnikov, M.V.; Yolken, R.H.; et al. Cerebral complement C1q activation in chronic Toxoplasma infection. Brain Behav. Immun. 2016, 58, 52–56. [Google Scholar] [CrossRef]
- David, C.N.; Frias, E.S.; Szu, J.I.; Vieira, P.A.; Hubbard, J.A.; Lovelace, J.; Michael, M.; Worth, D.; McGovern, K.E.; Ethell, I.M.; et al. GLT-1-Dependent Disruption of CNS Glutamate Homeostasis and Neuronal Function by the Protozoan Parasite Toxoplasma gondii. PLoS Pathog. 2016, 12, e1005643. [Google Scholar] [CrossRef]
- Parlog, A.; Harsan, L.A.; Zagrebelsky, M.; Weller, M.; Elverfeldt, D.V.; Mawrin, C.; Korte, M.; Dunay, I.R. Chronic murine toxoplasmosis is defined by subtle changes in neuronal connectivity. Dis. Models Mech. 2014, 7, 459. [Google Scholar] [CrossRef]
- Johnson, H.J.; Koshy, A.A. Latent Toxoplasmosis Effects on Rodents and Humans: How Much is Real and How Much is Media Hype? mBio 2020, 11. [Google Scholar] [CrossRef]
- Johnson, S.K.; Johnson, P.T.J. Toxoplasmosis: Recent Advances in Understanding the Link Between Infection and Host Behavior. Annu. Rev. Anim. Biosci. 2021, 9, 249–264. [Google Scholar] [CrossRef]
- Gharamti, A.A.; Rao, A.; Pecen, P.E.; Henao-Martínez, A.F.; Franco-Paredes, C.; Montoya, J.G. Acute Toxoplasma Dissemination With Encephalitis in the Era of Biological Therapies. Open Forum. Infect. Dis. 2018, 5, 256–259. [Google Scholar] [CrossRef]
- Bannoura, S.; El Hajj, R.; Khalifeh, I.; El Hajj, H. Acute disseminated encephalomyelitis and reactivation of cerebral toxoplasmosis in a child: Case report. IDCases 2018, 13, e00434. [Google Scholar] [CrossRef]
- Basavaraju, A. Toxoplasmosis in HIV infection: An overview. Trop. Parasitol. 2016, 6, 129–135. [Google Scholar] [CrossRef]
- Kodym, P.; Malý, M.; Beran, O.; Jilich, D.; Rozsypal, H.; Machala, L.; Holub, M. Incidence, immunological and clinical characteristics of reactivation of latent Toxoplasma gondii infection in HIV-infected patients. Epidemiol. Infect. 2015, 143, 600–607. [Google Scholar] [CrossRef]
- Gay, J.; Gendron, N.; Verney, C.; Joste, V.; Dardé, M.L.; Loheac, C.; Vrtovsnik, F.; Argy, N.; Houze, S. Disseminated toxoplasmosis associated with hemophagocytic syndrome after kidney transplantation: A case report and review. Transpl. Infect. Dis. 2019, 21, e13154. [Google Scholar] [CrossRef]
- Kollu, V.; Magalhaes-Silverman, M.; Tricot, G.; Ince, D. Toxoplasma Encephalitis following Tandem Autologous Hematopoietic Stem Cell Transplantation: A Case Report and Review of the Literature. Case Rep. Infect. Dis. 2018. [Google Scholar] [CrossRef]
- Paccoud, O.; Guitard, J.; Labopin, M.; Surgers, L.; Malard, F.; Battipaglia, G.; Duléry, R.; Hennequin, C.; Mohty, M.; Brissot, E. Features of Toxoplasma gondii reactivation after allogeneic hematopoietic stem-cell transplantation in a high seroprevalence setting. Bone Marrow Transpl. 2020, 55, 93–99. [Google Scholar] [CrossRef]
- Ramanan, P.; Scherger, S.; Benamu, E.; Bajrovic, V.; Jackson, W.; Hage, C.A.; Hakki, M.; Baddley, J.W.; Abidi, M.Z. Toxoplasmosis in non-cardiac solid organ transplant recipients: A case series and review of literature. Transpl. Infect. Dis. 2020, 22, e13218. [Google Scholar] [CrossRef] [PubMed]
- Ramchandar, N.; Pong, A.; Anderson, E. Identification of disseminated toxoplasmosis by plasma next-generation sequencing in a teenager with rapidly progressive multiorgan failure following haploidentical stem cell transplantation. Pediatr. Blood Cancer 2020, 67, e28205. [Google Scholar] [CrossRef] [PubMed]
- Robert-Gangneux, F.; Meroni, V.; Dupont, D.; Botterel, F.; Garcia, J.M.A.; Brenier-Pinchart, M.P.; Accoceberry, I.; Akan, H.; Abbate, I.; Boggian, K.; et al. Toxoplasmosis in Transplant. Recipients, Europe. 2010–2014. Emerg. Infect. Dis. 2018, 24, 1497–1504. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, S.; Weeratunga, P.; Rodrigo, C.; Lakshitha de Silva, N.; Fernando, S.D. Prophylaxis of human toxoplasmosis: A systematic review. Pathog. Glob. Health 2017, 111, 333–342. [Google Scholar] [CrossRef]
- Li, Y.; Zeng, Y.-M.; Liu, M.; Lu, Y.-Q.; Liu, X.-Y.; Zhang, Y.-L.; Jiang, Z.-S.; Yang, T.-T.; Sun, Y.; Lan, K.; et al. Development of a risk scoring system for prognostication in HIV-related toxoplasma encephalitis. BMC Infect. Dis. 2020, 20, 923. [Google Scholar] [CrossRef]
- Holland, M.S.; Sharma, K.; Lee, B.C. Cerebral toxoplasmosis after rituximab therapy for splenic marginal zone lymphoma: A case report and review of the literature. JMM Case Rep. 2015, 2. [Google Scholar] [CrossRef]
- Lee, E.B.; Ayoubi, N.; Albayram, M.; Kariyawasam, V.; Motaparthi, K. Cerebral toxoplasmosis after rituximab for pemphigus vulgaris. JAAD Case Rep. 2019, 6, 37–41. [Google Scholar] [CrossRef]
- Morjaria, S.; Epstein, D.J.; Romero, F.A.; Taur, Y.; Seo, S.K.; Papanicolaou, G.A.; Hatzoglou, V.; Rosenblum, M.; Perales, M.A.; Scordo, M.; et al. Toxoplasma Encephalitis in Atypical Hosts at an Academic Cancer Center. Open Forum Infect. Dis. 2016, 3. [Google Scholar] [CrossRef]
- Safa, G.; Darrieux, L. Cerebral Toxoplasmosis After Rituximab Therapy. JAMA Intern. Med. 2013, 173, 924–926. [Google Scholar] [CrossRef][Green Version]
- La Hoz, R.M.; Morris, M.I. Infectious Diseases Community of Practice of the American Society of, Tissue and blood protozoa including toxoplasmosis, Chagas disease, leishmaniasis, Babesia, Acanthamoeba, Balamuthia, and Naegleria in solid organ transplant recipients-Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin Transpl. 2019, 33, e13546. [Google Scholar]
- Adekunle, R.O.; Sherman, A.; Spicer, J.O.; Messina, J.A.; Steinbrink, J.M.; Sexton, M.E.; Lyon, G.M.; Mehta, A.K.; Phadke, V.K.; Woodworth, M.H. Clinical characteristics and outcomes of toxoplasmosis among transplant recipients at two US academic medical centers. Transpl. Infect Dis. 2021, 23, e13636. [Google Scholar] [CrossRef] [PubMed]
- Madireddy, S.; Rivas Chacon, E.D.; Mangat, R. Toxoplasmosis, in StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Ngo, H.M.; Zhou, Y.; Lorenzi, H.; Wang, K.; Kim, T.K.; Zhou, Y.; El Bissati, K.; Mui, E.; Fraczek, L.; Rajagopala, S.V.; et al. Toxoplasma Modulates Signature Pathways of Human Epilepsy, Neurodegeneration and Cancer. Sci. Rep. 2017, 7, 11496. [Google Scholar] [CrossRef]
- Saberi, R.; Sharif, M.; Sarvi, S.; Aghayan, S.A.; Hosseini, S.A.; Anvari, D.; Chegeni, T.N.; Hosseininejad, Z.; Daryani, A. Is Toxoplasma gondii playing a positive role in multiple sclerosis risk? A systematic review and meta-analysis. J. Neuroimmunol. 2018, 322, 57–62. [Google Scholar] [CrossRef]
- Nicoletti, A.; Cicero, C.E.; Giuliano, L.; Todaro, V.; Lo Fermo, S.; Chisari, C.; D’Amico, E.; Paradisi, V.; Mantella, A.; Bartoloni, A.; et al. Toxoplasma gondii and multiple sclerosis: A population-based case—Control study. Sci. Rep. 2020, 10, 18855. [Google Scholar] [CrossRef]
- Cicero, C.E.; Allibrio, F.E.; Giuliano, L.; Luna, J.; Preux, P.M.; Nicoletti, A. Toxoplasma gondii and multiple sclerosis: A systematic review and meta-analysis. Eur. J. Neurol. 2021. [Google Scholar] [CrossRef]
- Ngoungou, E.B.; Bhalla, D.; Nzoghe, A.; Dardé, M.L.; Preux, P.M. Toxoplasmosis and epilepsy--systematic review and meta analysis. PLoS Negl. Trop. Dis. 2015, 9, e0003525. [Google Scholar] [CrossRef]
- Bazilevich, S.N. Cryptogenic epilepsy in adults: “hidden problems of structural well-being”. Zh. Nevrol. Psikhiatr. Im. S S Korsakova 2013, 113, 10–19. [Google Scholar] [PubMed]
- Yazar, S.; Arman, F.; Yalçin, S.; Demirtaş, F.; Yaman, O.; Sahin, I. Investigation of probable relationship between Toxoplasma gondii and cryptogenic epilepsy. Seizure 2003, 12, 107–109. [Google Scholar] [CrossRef]
- Stommel, E.W.; Seguin, R.; Thadani, V.M.; Schwartzman, J.D.; Gilbert, K.; Ryan, K.A.; Tosteson, T.D.; Kasper, L.H. Cryptogenic Epilepsy: An. Infectious Etiology? Epilepsia 2001, 42, 436–438. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, M.; Riahi, S.M.; Mohammadi, M.; Saber, V.; Aghamolaie, S.; Moghaddam, S.A.; Aghaei, S.; Javanian, M.; Gamble, H.R.; Rostami, A. An updated meta-analysis of the association between Toxoplasma gondii infection and risk of epilepsy. Trans. R. Soc. Trop Med. Hyg. 2019, 113, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Bayani, M.; Riahi, S.M.; Bazrafshan, N.; Gamble, H.R.; Rostami, A. Toxoplasma gondii infection and risk of Parkinson and Alzheimer diseases: A systematic review and meta-analysis on observational studies. Acta Trop. 2019, 196, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Mahami Oskouei, M.; Hamidi, F.; Talebi, M.; Farhoudi, M.; Taheraghdam, A.A.; Kazemi, T.; Sadeghi-Bazargani, H.; Fallah, E. The correlation between Toxoplasma gondii infection and Parkinson’s disease: A case-control study. J. Parasit. Dis. 2016, 40, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Kusbeci, O.Y.; Miman, O.; Yaman, M.; Aktepe, O.C.; Yazar, S. Could Toxoplasma gondii have any role in Alzheimer disease? Alzheimer Dis. Assoc. Disord. 2011, 25, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Perry, V.H.; Cunningham, C.; Holmes, C. Systemic infections and inflammation affect chronic neurodegeneration. Nat. Rev. Immunol. 2007, 7, 161–167. [Google Scholar] [CrossRef]
- Wyman, C.P.; Gale, S.D.; Hedges-Muncy, A.; Erickson, L.D.; Wilson, E.; Hedges, D.W. Association between Toxoplasma gondii seropositivity and memory function in nondemented older adults. Neurobiol. Aging 2017, 53, 76–82. [Google Scholar] [CrossRef]
- Perry, C.E.; Gale, S.D.; Erickson, L.; Wilson, E.; Nielsen, B.; Kauwe, J.; Hedges, D.W. Seroprevalence and Serointensity of Latent Toxoplasma gondii in a Sample of Elderly Adults With and Without Alzheimer Disease. Alzheimer Dis. Assoc. Disord. 2016, 30, 123–126. [Google Scholar] [CrossRef]
- Carter, C.J. Toxoplasmosis and Polygenic Disease Susceptibility Genes: Extensive Toxoplasma gondii Host/Pathogen Interactome Enrichment in Nine Psychiatric or Neurological Disorders. J. Pathog. 2013, 2013, 965046. [Google Scholar] [CrossRef]
- Mahmoudi, S.; Mamishi, S.; Suo, X.; Keshavarz, H. Early detection of Toxoplasma gondii infection by using a interferon gamma release assay: A review. Exp. Parasitol. 2017, 172, 39–43. [Google Scholar] [CrossRef]
- Torres, L.; Robinson, S.A.; Kim, D.G.; Yan, A.; Cleland, T.A.; Bynoe, M.S. Toxoplasma gondii alters NMDAR signaling and induces signs of Alzheimer’s disease in wild-type, C57BL/6 mice. J. Neuroinflamm. 2018, 15, 57. [Google Scholar] [CrossRef]
- Mohle, L.; Israel, N.; Paarmann, K.; Krohn, M.; Pietkiewicz, S.; Müller, A.; Lavrik, I.N.; Buguliskis, J.S.; Schott, B.H.; Schlüter, D.; et al. Chronic Toxoplasma gondii infection enhances beta-amyloid phagocytosis and clearance by recruited monocytes. Acta Neuropathol. Commun. 2016, 4, 25. [Google Scholar] [CrossRef]
- Jung, B.K.; Pyo, K.H.; Shin, K.Y.; Hwang, Y.S.; Lim, H.; Lee, S.J.; Moon, J.H.; Lee, S.H.; Suh, Y.H.; Chai, J.Y.; et al. Toxoplasma gondii infection in the brain inhibits neuronal degeneration and learning and memory impairments in a murine model of Alzheimer’s disease. PLoS ONE 2012, 7, e33312. [Google Scholar] [CrossRef]
- Shin, J.H.; Hwang, Y.S.; Jung, B.K.; Seo, S.H.; Ham, D.W.; Shin, E.H. Reduction of Amyloid Burden by Proliferated Homeostatic Microglia in Toxoplasma gondii-Infected Alzheimer’s Disease Model Mice. Int. J. Mol. Sci. 2021, 22, 2764. [Google Scholar] [CrossRef]
- Ortiz-Guerrero, G.; Gonzalez-Reyes, R.E.; de-la-Torre, A.; Medina-Rincón, G.; Nava-Mesa, M.O. Pathophysiological Mechanisms of Cognitive Impairment and Neurodegeneration by Toxoplasma gondii Infection. Brain Sci. 2020, 10, 369. [Google Scholar] [CrossRef]
- Tong, W.H.; Pavey, C.; O’Handley, R.; Vyas, A. Behavioral biology of Toxoplasma gondii infection. Parasit. Vectors 2021, 14, 77. [Google Scholar] [CrossRef]
- Milne, G.; Webster, J.P.; Walker, M. Toxoplasma gondii: An. Underestimated Threat? Trends Parasitol. 2020, 36, 959–969. [Google Scholar] [CrossRef]
- Boillat, M.; Hammoudi, P.M.; Dogga, S.K.; Pagès, S.; Goubran, M.; Rodriguez, I.; Soldati-Favre, D. Neuroinflammation-Associated Aspecific Manipulation of Mouse Predator Fear by Toxoplasma gondii. Cell Rep. 2020, 30, 320–334. [Google Scholar] [CrossRef] [PubMed]
- Severance, E.G.; Xiao, J.; Jones-Brando, L.; Sabunciyan, S.; Li, Y.; Pletnikov, M.; Prandovszky, E.; Yolken, R. Toxoplasma gondii-A Gastrointestinal Pathogen Associated with Human Brain Diseases. Int. Rev. Neurobiol. 2016, 131, 143–163. [Google Scholar] [PubMed]
- Sutterland, A.L.; Fond, G.; Kuin, A.; Koeter, M.W.J.; Lutter, R.; Gool, T.V.; Yolken, R.; Szoke, A.; Leboyer, M.; de Haan, L. Toxoplasma gondii in schizophrenia, bipolar disorder, and addiction: Systematic review and meta-analysis. Acta. Psychiatr. Scand. 2015, 132, 161–179. [Google Scholar] [CrossRef] [PubMed]
- Gatkowska, J.; Wieczorek, M.; Dziadek, B.; Dzitko, K.; Dlugonska, H. Sex-dependent neurotransmitter level changes in brains of Toxoplasma gondii infected mice. Exp. Parasitol. 2013, 133, 1–7. [Google Scholar] [CrossRef]
- Prandovszky, E.; Gaskell, E.; Martin, H.; Dubey, J.P.; Webster, J.P.; McConkey, G.A. The Neurotropic Parasite Toxoplasma Gondii Increases Dopamine Metabolism. PLoS ONE 2011, 6, e23866. [Google Scholar] [CrossRef] [PubMed]
- Skallova, A.; Kodym, P.; Frynta, D.; Flegr, J. The role of dopamine in Toxoplasma-induced behavioural alterations in mice: An ethological and ethopharmacological study. Parasitology 2006, 133, 525–535. [Google Scholar] [CrossRef]
- Wang, Z.T.; Harmon, S.; O’Malley, K.L.; Sibley, D.L. Reassessment of the role of aromatic amino acid hydroxylases and the effect of infection by Toxoplasma gondii on host dopamine. Infect Immun. 2015, 83, 1039–1047. [Google Scholar] [CrossRef]
- Patten, S.B.; Kennedy, S.H.; Lam, R.W.; O’Donovan, C.; Filteau, M.J.; Parikh, S.V.; Ravindran, A.V. Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults. J. Affect. Disord. 2009, 117, S5–S14. [Google Scholar] [CrossRef]
- Daubener, W.; Spors, B.; Hucke, C.; Adam, R.; Stins, M.; Kim, K.S.; Schroten, H. Restriction of Toxoplasma gondii growth in human brain microvascular endothelial cells by activation of indoleamine 2,3-dioxygenase. Infect. Immun. 2001, 69, 6527–6531. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Hoshi, M.; Hara, A.; Takemura, M.; Arioka, Y.; Yamamoto, Y.; Matsunami, H.; Funato, T.; Seishima, M.; Saito, K. Inhibition of increased indoleamine 2,3-dioxygenase activity attenuates Toxoplasma gondii replication in the lung during acute infection. Cytokine 2012, 59, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, V.B.; Suzuki, Y. Effects of Toxoplasma gondii Infection on the Brain. Schizophr. Bull. 2007, 33, 745–751. [Google Scholar] [CrossRef]
- Dalimi, A.; Abdoli, A. Latent toxoplasmosis and human. Iran. J. Parasitol. 2012, 7, 1–17. [Google Scholar] [PubMed]
- Mahmoud, M.E.; Fereig, R.; Nishikawa, Y. Involvement of Host Defense Mechanisms against Toxoplasma gondii Infection in Anhedonic and Despair-Like Behaviors in Mice. Infect. Immun. 2017, 85, e00007–e00017. [Google Scholar] [CrossRef]
- Webster, J.P.; McConkey, G.A. Toxoplasma gondii-altered host behaviour: Clues as to mechanism of action. Folia. Parasitol. (Praha) 2010, 57, 95–104. [Google Scholar] [CrossRef]
- Bay-Richter, C.; Buttenschøn, H.N.; Mors, O.; Eskelund, A.; Budac, D.; Kærlev, L.; Wegener, G. Latent toxoplasmosis and psychiatric symptom: A role of tryptophan metabolism? J. Psychiatr. Res. 2019, 110, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Notarangelo, F.M.; Wilson, E.H.; Horning, K.J.; Thomas, M.A.R.; Harris, T.H.; Fang, Q.; Hunter, C.A.; Schwarcz, R. Evaluation of kynurenine pathway metabolism in Toxoplasma gondii-infected mice: Implications for schizophrenia. Schizophr Res. 2014, 152, 261–267. [Google Scholar] [CrossRef]
- Erhardt, S.; Schwieler, L.; Imbeault, S.; Engberg, G. The kynurenine pathway in schizophrenia and bipolar disorder. Neuropharmacology 2017, 112, 297–306. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, Y.; Li, H.; Song, X.; Ma, Z.; Lu, H.; Liu, S.; Zhao, Y.; Tan, M.; Wang, S.; et al. Identification of Toxoplasma Gondii Tyrosine Hydroxylase (TH) Activity and Molecular Immunoprotection against Toxoplasmosis. Vaccines 2020, 8, 158. [Google Scholar] [CrossRef] [PubMed]
- Flegr, J.; Horacek, J. Negative Effects of Latent Toxoplasmosis on Mental Health. Front. Psychiatry 2019, 10, 1012. [Google Scholar] [CrossRef] [PubMed]
- Bak, J.; Shim, S.H.; Kwon, Y.J.; Lee, H.Y.; Kim, J.S.; Yoon, H.; Lee, Y.J. The Association between Suicide Attempts and Toxoplasma gondii Infection. Clin. Psychopharmacol. Neurosci. 2018, 16, 95–102. [Google Scholar] [CrossRef]
- Ling, V.J.; Lester, D.; Mortensen, P.B.; Langenberg, P.W.; Postolache, T.T. Toxoplasma gondii seropositivity and suicide rates in women. J. Nerv. Ment. Dis. 2011, 199, 440–444. [Google Scholar]
- Postolache, T.T.; Wadhawan, A.; Rujescu, D.; Hoisington, A.J.; Dagdag, A.; Baca-Garcia, E.; Lowry, C.A.; Okusaga, O.O.; Brenner, L.A. Toxoplasma gondii, Suicidal Behavior, and Intermediate Phenotypes for Suicidal Behavior. Front. Psychiatry 2021, 12, 665682. [Google Scholar] [CrossRef]
- Kar, N.; Misra, B. Toxoplasma seropositivity and depression: A case report. BMC Psychiatry 2004, 4, 1. [Google Scholar] [CrossRef]
- Horacek, J.; Flegr, J.; Tintera, J.; Verebova, K.; Spaniel, F.; Novak, T.; Brunovsky, M.; Bubenikova-Valesova, V.; Holub, D.; Palenicek, T.; et al. Latent toxoplasmosis reduces gray matter density in schizophrenia but not in controls: Voxel-based-morphometry (VBM) study. World J. Biol. Psychiatry 2012, 13, 501–509. [Google Scholar] [CrossRef]
- Torrey, E.F.; Bartko, J.J.; Lun, Z.R.; Yolken, R.H. Antibodies to Toxoplasma gondii in patients with schizophrenia: A meta-analysis. Schizophr Bull. 2007, 33, 729–736. [Google Scholar] [CrossRef]
- Torrey, E.F.; Bartko, J.J.; Yolken, R.H. Toxoplasma gondii and other risk factors for schizophrenia: An update. Schizophr Bull. 2012, 38, 642–647. [Google Scholar] [CrossRef]
- Chen, X.; Chen, B.; Hou, X.; Zheng, C.; Yang, X.; Ke, J.; Hu, X.; Tan, F. Association between Toxoplasma gondii infection and psychiatric disorders in Zhejiang, Southeastern China. Acta Trop. 2019, 192, 82–86. [Google Scholar] [CrossRef]
- Burgdorf, K.S.; Trabjerg, B.B.; Pedersen, M.G.; Nissen, J.; Banasik, K.; Pedersen, O.B.; Sørensen, E.; Nielsen, K.R.; Larsen, M.H.; Erikstrup, C.; et al. Large-scale study of Toxoplasma and Cytomegalovirus shows an association between infection and serious psychiatric disorders. Brain Behav. Immun. 2019, 79, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Avramopoulos, D.; Pearce, B.D.; McGrath, J.; Wolyniec, P.; Wang, R.; Eckart, N.; Hatzimanolis, A.; Goes, F.S.; Nestadt, G.; Mulle, J.; et al. Infection and inflammation in schizophrenia and bipolar disorder: A genome wide study for interactions with genetic variation. PLoS ONE 2015, 10, e0116696. [Google Scholar] [CrossRef] [PubMed]
- Majláth, Z.; Török, N.; Toldi, J.; Vécsei, L. Memantine and Kynurenic Acid: Current Neuropharmacological Aspects. Curr. Neuropharmacol. 2016, 14, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Pocivavsek, A.; Wu, H.Q.; Potter, M.C.; Elmer, G.I.; Pellicciari, R.; Schwarcz, R. Fluctuations in Endogenous Kynurenic Acid Control Hippocampal Glutamate and Memory. Neuropsychopharmacology 2011, 36, 2357–2367. [Google Scholar] [CrossRef] [PubMed]
- Kano, S.I.; Hodgkinson, C.A.; Jones-Brando, L.; Eastwood, S.; Ishizuka, K.; Niwa, M.; Choi, E.Y.; Chang, D.J.; Chen, Y.; Velivela, S.D.; et al. Host-parasite interaction associated with major mental illness. Mol. Psychiatry 2020, 25, 194–205. [Google Scholar] [CrossRef]
- Bhadra, R.; Gigley, J.P.; Weiss, L.M.; Khan, I.A. Control of Toxoplasma reactivation by rescue of dysfunctional CD8+ T-cell response via PD-1-PDL-1 blockade. Proc. Natl. Acad. Sci. USA 2011, 108, 9196–9201. [Google Scholar] [CrossRef]
- Steiner, J.; Jacobs, R.; Panteli, B.; Brauner, M.; Schiltz, K.; Bahn, S.; Herberth, M.; Westphal, S.; Gos, T.; Walter, M.; et al. Acute schizophrenia is accompanied by reduced T cell and increased B cell immunity. Eur. Arch. Psychiatry Clin. Neurosci. 2010, 260, 509–518. [Google Scholar] [CrossRef]
- Torrey, E.F.; Yolken, R.H. Toxoplasma gondii and Schizophrenia. Emerg. Infect. Dis. J. 2003, 9, 1375. [Google Scholar] [CrossRef]
- Hamdani, N.; Daban-Huard, C.; Lajnef, M.; Richard, J.R.; Delavest, M.; Godin, O.; Le Guen, E.; Vederine, F.E.; Lépine, J.P.; Jamain, S.; et al. Relationship between Toxoplasma gondii infection and bipolar disorder in a French sample. J. Affect. Disord. 2013, 148, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Hussein, E.A.M.; Khalifa, H.; Ramadan, G.K.; Hassaan, S.H.; Shaaban, I.; Farrag, H.M.M. Seroprevalence of Toxoplasma gondii among patients with schizophrenia and bipolar disorder in Upper Egypt: A comparative study with a control group. Ann. Parasitol. 2020, 66, 183–192. [Google Scholar] [PubMed]
- Pearce, B.D.; Kruszon-Moran, D.; Jones, J.L. The Relationship Between Toxoplasma gondii Infection and Mood Disorders in the Third National Health and Nutrition Survey. Biol. Psychiatry 2012, 72, 290–295. [Google Scholar] [CrossRef]
- Tedla, Y.; Shibre, T.; Ali, O.; Tadele, G.; Woldeamanuel, Y.; Asrat, D.; Aseffa, A.; Mihret, W.; Abebe, M.; Alem, A.; et al. Serum antibodies to Toxoplasma gondii and Herpesvidae family viruses in individuals with schizophrenia and bipolar disorder: A case-control study. Ethiop. Med. J. 2011, 49, 211–220. [Google Scholar] [PubMed]
- de Barros, J.; Barbosa, I.G.; Salem, H.; Rocha, N.P.; Kummer, A.; Okusaga, O.O.; Soares, J.C.; Teixeira, A.L. Is there any association between Toxoplasma gondii infection and bipolar disorder? A systematic review and meta-analysis. J. Affect. Disord. 2017, 209, 59–65. [Google Scholar] [CrossRef]
- Del Grande, C.; Galli, L.; Schiavi, E.; Dell’Osso, L.; Bruschi, F. Is Toxoplasma gondii a Trigger of Bipolar Disorder? Pathogens 2017, 6, 3. [Google Scholar] [CrossRef]
- Nayeri Chegeni, T.; Sarvi, S.; Amouei, A.; Moosazadeh, M.; Hosseininejad, Z.; Aghayan, S.A.; Daryani, A. Relationship between toxoplasmosis and obsessive compulsive disorder: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2019, 13, e0007306. [Google Scholar] [CrossRef]
- Akaltun, İ.; Kara, S.S.; Kara, T. The relationship between Toxoplasma gondii IgG antibodies and generalized anxiety disorder and obsessive-compulsive disorder in children and adolescents: A new approach. Nord. J. Psychiatry 2018, 72, 57–62. [Google Scholar] [CrossRef]
- Brynska, A.; Tomaszewicz-Libudzic, E.; Wolanczyk, T. Obsessive-compulsive disorder and acquired toxoplasmosis in two children. Eur. Child Adolesc. Psychiatry 2001, 10, 200–204. [Google Scholar] [CrossRef]
- Hodge, J.M.; Coghill, A.E.; Kim, Y.; Bender, N.; Smith-Warner, S.A.; Gapstur, S.; Teras, L.R.; Grimsrud, T.K.; Waterboer, T.; Egan, K.M. Toxoplasma gondii infection and the risk of adult glioma in two prospective studies. Int. J. Cancer 2021. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.; Hurley, S.F.; Johnson, A.M.; Salzberg, M.; Lee, M.W.; North, J.B.; McNeil, J.J.; McMichael, A.J. Tumours of the Brain and Presence of Antibodies to Toxoplasma gondii. Int. J. Epidemiol. 1993, 22, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Thomas, F.; Lafferty, K.D.; Brodeur, J.; Elguero, E.; Gauthier-Clerc, M.; Missé, D. Incidence of adult brain cancers is higher in countries where the protozoan parasite Toxoplasma gondii is common. Biol. Lett. 2012, 8, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Zeiner, G.M.; Norman, K.L.; Thomson, J.M.; Hammond, S.M.; Boothroyd, J.C. Toxoplasma gondii infection specifically increases the levels of key host microRNAs. PLoS ONE 2010, 5, e8742. [Google Scholar] [CrossRef]
- Thirugnanam, S.; Rout, N.; Gnanasekar, M. Possible role of Toxoplasma gondii in brain cancer through modulation of host microRNAs. Infect. Agent Cancer 2013, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Colinot, D.L.; Garbuz, T.; Bosland, M.C.; Wang, L.; Rice, S.E.; Sullivan, W.J.; Arrizabalaga, G.; Jerde, T.J. The common parasite Toxoplasma gondii induces prostatic inflammation and microglandular hyperplasia in a mouse model. Prostate 2017, 77, 1066–1075. [Google Scholar] [CrossRef] [PubMed]
- Cong, W.; Liu, G.H.; Meng, Q.F.; Dong, W.; Qin, S.Y.; Zhang, F.K.; Zhang, X.Y.; Wang, X.Y.; Qian, A.D.; Zhu, X.Q. Toxoplasma gondii infection in cancer patients: Prevalence, risk factors, genotypes and association with clinical diagnosis. Cancer Lett. 2015, 359, 307–313. [Google Scholar] [CrossRef]
- Jung, B.-K.; Song, H.; Kim, M.J.; Cho, J.; Shin, E.H.; Chai, J.Y. High Toxoplasma gondii Seropositivity among Brain Tumor Patients in Korea. Korean J. Parasitol. 2016, 54, 201–204. [Google Scholar] [CrossRef]
- de Faria Junior, G.M.; Murata, F.H.A.; Lorenzi, H.A.; Castro, B.B.P.; Assoni, L.C.P.; Ayo, C.M.; Brandão, C.C.; de Mattos, L.C. The Role of microRNAs in the Infection by T. gondii in Humans. Front. Cell Infect. Microbiol. 2021, 11, 670548. [Google Scholar] [CrossRef]
- Zeiner, G.M.; Boothroyd, J.C. Use of two novel approaches to discriminate between closely related host microRNAs that are manipulated by Toxoplasma gondii during infection. RNA 2010, 16, 1268–1274. [Google Scholar] [CrossRef]
- Carmen, J.C.; Sinai, A.P. Suicide prevention: Disruption of apoptotic pathways by protozoan parasites. Mol. Microbiol. 2007, 64, 904–916. [Google Scholar] [CrossRef] [PubMed]
- Lüder, C.G.; Gross, U. Apoptosis and its modulation during infection with Toxoplasma gondii: Molecular mechanisms and role in pathogenesis. Curr. Top. Microbiol. Immunol. 2005, 289, 219–237. [Google Scholar] [PubMed]
- Qian, Y.; Song, J.; Ouyang, Y.; Han, Q.; Chen, W.; Zhao, X.; Xie, Y.; Chen, Y.; Yuan, W.; Fan, C. Advances in Roles of miR-132 in the Nervous System. Front. Pharmacol. 2017, 8, 770. [Google Scholar] [CrossRef]
- Xiao, J.; Li, Y.; Prandovszky, E.; Karuppagounder, S.S.; Talbot, C.C.; Dawson, V.L.; Dawson, T.M.; Yolken, R.H. MicroRNA-132 dysregulation in Toxoplasma gondii infection has implications for dopamine signaling pathway. Neuroscience 2014, 268, 128–138. [Google Scholar] [CrossRef]
- Pang, J.C.-s.; Kwok, W.K.; Chen, Z.; Ng, H.K. Oncogenic role of microRNAs in brain tumors. Acta Neuropathol. 2009, 117, 599–611. [Google Scholar] [CrossRef]
- Turner, J.D.; Williamson, R.; Almefty, K.K.; Nakaji, P.; Porter, R.; Tse, V.M.; Kalani, Y.S. The many roles of microRNAs in brain tumor biology. Neurosurg. Focus 2010, 28, e3. [Google Scholar]
- Wang, D.; Qiu, C.; Zhang, H.; Wang, J.; Cui, Q.; Yin, Y. Human MicroRNA Oncogenes and Tumor Suppressors Show Significantly Different Biological Patterns: From Functions to Targets. PLoS ONE 2010, 5, e13067. [Google Scholar] [CrossRef]
- Vittecoq, M.; Elguero, E.; Lafferty, K.D.; Roche, B.; Brodeur, J.; Gauthier-Clerc, M.; Missé, D.; Thomas, F. Brain cancer mortality rates increase with Toxoplasma gondii seroprevalence in France. Infect. Genet. Evol. 2012, 12, 496–498. [Google Scholar] [CrossRef]
- Ernst, A.; Campos, B.; Meier, J.; Devens, F.; Liesenberg, F.; Wolter, M.; Reifenberger, G.; Herold-Mende, C.; Lichter, P.; Radlwimmer, B. De-repression of CTGF via the miR-17–92 cluster upon differentiation of human glioblastoma spheroid cultures. Oncogene 2010, 29, 3411–3422. [Google Scholar] [CrossRef] [PubMed]
- Uziel, T.; Karginov, F.V.; Xie, S.; Parker, J.S.; Wang, Y.D.; Gajjar, A.; He, L.; Ellison, D.; Gilbertson, R.J.; Hannon, G.; et al. The miR-17~92 cluster collaborates with the Sonic Hedgehog pathway in medulloblastoma. Proc. Natl. Acad. Sci. USA 2009, 106, 2812. [Google Scholar] [CrossRef] [PubMed]
- Laliberté, J.; Carruthers, V.B. Host cell manipulation by the human pathogen Toxoplasma gondii. Cell. Mol. Life Sci. 2008, 65, 1900–1915. [Google Scholar] [CrossRef]
- Braun, L.; Cannella, D.; Ortet, P.; Barakat, M.; Sautel, C.F.; Kieffer, S.; Garin, J.; Bastien, O.; Voinnet, O.; Hakimi, M.A. A complex small RNA repertoire is generated by a plant/fungal-like machinery and effected by a metazoan-like Argonaute in the single-cell human parasite Toxoplasma gondii. PLoS Pathog. 2010, 6, e1000920. [Google Scholar] [CrossRef]
- Dunay, I.R.; Gajurel, K.; Dhakal, R.; Liesenfeld, O.; Montoya, J.G. Treatment of Toxoplasmosis: Historical Perspective, Animal Models, and Current Clinical Practice. Clin. Microbiol. Rev. 2018, 31, e00057-17. [Google Scholar] [CrossRef]
- Konstantinovic, N.; Guegan, H.; Stäjner, T.; Belaz, S.; Robert-Gangneux, F. Treatment of toxoplasmosis: Current options and future perspectives. Food Waterborne Parasitol. 2019, 15, e00036. [Google Scholar] [CrossRef] [PubMed]
- Blume, M.; Seeber, F. Metabolic interactions between Toxoplasma gondii and its host. F1000Research 2018. [Google Scholar] [CrossRef] [PubMed]
- Lapinskas, P.J.; Ben-Harari, R.R. Perspective on current and emerging drugs in the treatment of acute and chronic toxoplasmosis. Postgrad Med. 2019, 131, 589–596. [Google Scholar] [CrossRef]
- Remington, J.S.; Thulliez, P.; Montoya, J.G. Recent Developments for Diagnosis of Toxoplasmosis. J. Clin. Microbiol. 2004, 42, 941. [Google Scholar] [CrossRef]
- Katlama, C.; Mouthon, B.; Gourdon, D.; Lapierre, D.; Rousseau, F. Atovaquone as long-term suppressive therapy for toxoplasmic encephalitis in patients with AIDS and multiple drug intolerance. Aids 1996, 10, 1107–1112. [Google Scholar]
- Ben-Harari, R.R.; Goodwin, E.; Casoy, J. Adverse Event Profile of Pyrimethamine-Based Therapy in Toxoplasmosis: A Systematic Review. Drugs R D 2017, 17, 523–544. [Google Scholar] [CrossRef] [PubMed]
- Katlama, C.; De Wit, S.; O’Doherty, E.; Van Glabeke, M.; Clumeck, N. Pyrimethamine-clindamycin vs. pyrimethamine-sulfadiazine as acute and long-term therapy for toxoplasmic encephalitis in patients with AIDS. Clin. Infect. 1996, 22, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, M.; Mehrzadi, S.; Sharif, M.; Sarvi, S.; Tanzifi, A.; Aghayan, S.A.; Daryani, A. Drug Resistance in Toxoplasma gondii. Front. Microbiol. 2018, 9, 2587. [Google Scholar] [CrossRef] [PubMed]
- Robert-Gangneux, F.; Binisti, P.; Antonetti, D.; Brezin, A.; Yera, H.; Dupouy-Camet, J. Usefulness of immunoblotting and Goldmann-Witmer coefficient for biological diagnosis of toxoplasmic retinochoroiditis. Eur. J. Clin. Microbiol. Infect. Dis. 2004, 23, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Díaz, J.C.; Martínez-Grueiro, M.M.; Martínez-Fernández, A.R. Comparative activity of several antibiotics against Toxoplasma gondii in a mouse model. Enferm. Infect. Microbiol. Clin. 1993, 11, 543–546. [Google Scholar]
- Schmidt, D.R.; Hogh, B.; Andersen, O.; Hansen, S.H.; Dalhoff, K.; Petersen, E. Treatment of infants with congenital toxoplasmosis: Tolerability and plasma concentrations of sulfadiazine and pyrimethamine. Eur. J. Pediatrics 2006, 165, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Alday, P.H.; Doggett, J.S. Drugs in development for toxoplasmosis: Advances, challenges, and current status. Drug Des. Dev. Ther. 2017, 11, 273–293. [Google Scholar] [CrossRef] [PubMed]
- Montazeri, M.; Sharif, M.; Sarvi, S.; Mehrzadi, S.; Ahmadpour, E.; Daryani, A. A Systematic Review of In vitro and In vivo Activities of Anti-Toxoplasma Drugs and Compounds (2006–2016). Front. Microbiol. 2017, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Değerli, K.; Kilimcioğlu, A.A.; Kurt, O.; Tamay, A.T.; Ozbilgin, A. Efficacy of azithromycin in a murine toxoplasmosis model, employing a Toxoplasma gondii strain from Turkey. Acta Trop. 2003, 88, 45–50. [Google Scholar] [CrossRef]
- Benmerzouga, I.; Checkley, L.A.; Ferdig, M.T.; Arrizabalaga, G.; Wek, R.C.; Sullivan, W.J., Jr. Guanabenz repurposed as an antiparasitic with activity against acute and latent toxoplasmosis. Antimicrob. Agents Chemother. 2015, 59, 6939–6945. [Google Scholar] [CrossRef]
- Hamie, M.; Najm, R.; Deleuze-Masquefa, C.; Bonnet, P.A.; Dubremetz, J.F.; El Sabban, M.; El Hajj, H. Imiquimod Targets Toxoplasmosis Through Modulating Host Toll-Like Receptor-MyD88 Signaling. Front. Immunol. 2021, 12, 629917. [Google Scholar] [CrossRef] [PubMed]
- Mevelec, M.N.; Lakhrif, Z.; Dimier-Poisson, I. Key Limitations and New Insights Into the Toxoplasma gondii Parasite Stage Switching for Future Vaccine Development in Human, Livestock, and Cats. Front. Cell Infect. Microbiol. 2020, 10, 607198. [Google Scholar] [CrossRef]
- Buxton, D.; Innes, E.A. A commercial vaccine for ovine toxoplasmosis. Parasitol 1995, 110, S11–S16. [Google Scholar] [CrossRef]
- Innes, E.A.; Bartley, P.M.; Buxton, D.; Katzer, F. Ovine Toxoplasmosis. Parasitology 2009, 136, 1887–1894. [Google Scholar] [CrossRef]
- Dodangeh, S.; Daryani, A.; Sharif, M.; Aghayan, S.A.; Pagheh, A.S.; Sarvi, S.; Rezaei, F. A systematic review on efficiency of microneme proteins to induce protective immunity against Toxoplasma gondii. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 617–629. [Google Scholar] [CrossRef]
- Faridnia, R.; Daryani, A.; Sarvi, S.; Sharif, M.; Kalani, H. Vaccination against Toxoplasma gondii using rhoptry antigens: A systematic review. Comp. Immunol. Microbiol. Infect. Dis. 2018, 59, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Träskman-Bendz, L.; Janelidze, S.; Langenberg, P.; Saleh, A.; Constantine, N.; Okusaga, O.; Bay-Richter, C.; Brundin, L.; Postolache, T.T. Toxoplasma gondii immunoglobulin G antibodies and nonfatal suicidal self-directed violence. J. Clin. Psychiatry 2012, 73, 1069–1076. [Google Scholar] [CrossRef]
- Siachoque, H.; Guzman, F.; Burgos, J.; Patarroyo, M.E.; Gomez Marin, J.E. Toxoplasma gondii: Immunogenicity and protection by P30 peptides in a murine model. Exp. Parasitol. 2006, 114, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.W.; Wu, C.A.; Morrow, W.J. Cell death induced by vaccine adjuvants containing surfactants. Vaccine 2004, 22, 1524–1536. [Google Scholar] [CrossRef]
- Bonenfant, C.; Dimier-Poisson, I.; Velge-Roussel, F.; Buzoni-Gatel, D.; Del Giudice, G.; Rappuoli, R.; Bout, D. Intranasal immunization with SAG1 and nontoxic mutant heat-labile enterotoxins protects mice against Toxoplasma gondii. Infect. Immun. 2001, 69, 1605–1612. [Google Scholar] [CrossRef]
- Letscher-Bru, V.; Villard, O.; Risse, B.; Zauke, M.; Klein, J.P.; Kien, T.T. Protective effect of vaccination with a combination of recombinant surface antigen 1 and interleukin-12 against toxoplasmosis in mice. Infect. Immun. 1998, 66, 4503–4506. [Google Scholar] [CrossRef] [PubMed]
- Martin, V.; Supanitsky, A.; Echeverria, P.C.; Litwin, S.; Tanos, T.; De Roodt, A.R.; Guarnera, E.A.; Angel, S.O. Recombinant GRA4 or ROP2 Protein Combined with Alum or the GRA4 Gene Provides Partial Protection in Chronic Murine Models of Toxoplasmosis. Clin. Diagn. Lab. Immunol. 2004, 11, 704–710. [Google Scholar] [CrossRef]
- Cérède, O.; Dubremetz, J.F.; Soête, M.; Deslée, D.; Vial, H.; Bout, D.; Lebrun, M. Synergistic role of micronemal proteins in Toxoplasma gondii virulence. J. Exp. Med. 2005, 201, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Jongert, E.; Verhelst, D.; Abady, M.; Petersen, E.; Gargano, N. Protective Th1 immune responses against chronic toxoplasmosis induced by a protein-protein vaccine combination but not by its DNA-protein counterpart. Vaccine 2008, 26, 5289–5295. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, F.; Sarvi, S.; Sharif, M.; Hejazi, S.H.; Pagheh, A.S.; Aghayan, S.A.; Daryani, A. A systematic review of Toxoplasma gondii antigens to find the best vaccine candidates for immunization. Microb. Pathog. 2019, 126, 172–184. [Google Scholar] [CrossRef] [PubMed]
- El Bissati, K.; Chentoufi, A.A.; Krishack, P.A.; Zhou, Y.; Woods, S.; Dubey, J.P.; Vang, L.; Lykins, J.; Broderick, K.E.; Ernest, M.; et al. Adjuvanted multi-epitope vaccines protect HLA-A*11:01 transgenic mice against Toxoplasma gondii. JCI Insight 2020, 1, 15. [Google Scholar] [CrossRef] [PubMed]
- Ahmadpour, E.; Sarvi, S.; Soteh, M.B.H.; Sharif, M.; Rahimi, M.T.; Valadan, R.; Tehrani, M.; Khalilian, A.; Montazeri, M.; Fasihi-Ramandi, M.; et al. Enhancing immune responses to a DNA vaccine encoding Toxoplasma gondii GRA14 by calcium phosphate nanoparticles as an adjuvant. Immunol. Lett. 2017, 185, 40–47. [Google Scholar] [CrossRef]
- Allahyari, M.; Mohabati, R.; Amiri, S.; Rastaghi, A.R.E.; Babaie, J.; Mahdavi, M.; Vatanara, A.; Golkar, M. Synergistic effect of rSAG1 and rGRA2 antigens formulated in PLGA microspheres in eliciting immune protection against Toxoplasama gondii. Exp. Parasitol. 2016, 170, 236–246. [Google Scholar] [CrossRef]
- Lee, D.H.; Kim, A.R.; Lee, S.H.; Quan, F.S. Cross-protection induced by Toxoplasma gondii virus-like particle vaccine upon intraperitoneal route challenge. Acta Trop. 2016, 164, 77–83. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daher, D.; Shaghlil, A.; Sobh, E.; Hamie, M.; Hassan, M.E.; Moumneh, M.B.; Itani, S.; El Hajj, R.; Tawk, L.; El Sabban, M.; et al. Comprehensive Overview of Toxoplasma gondii-Induced and Associated Diseases. Pathogens 2021, 10, 1351. https://doi.org/10.3390/pathogens10111351
Daher D, Shaghlil A, Sobh E, Hamie M, Hassan ME, Moumneh MB, Itani S, El Hajj R, Tawk L, El Sabban M, et al. Comprehensive Overview of Toxoplasma gondii-Induced and Associated Diseases. Pathogens. 2021; 10(11):1351. https://doi.org/10.3390/pathogens10111351
Chicago/Turabian StyleDaher, Darine, Ahmad Shaghlil, Eyad Sobh, Maguy Hamie, Malika Elhage Hassan, Mohamad Bahij Moumneh, Shaymaa Itani, Rana El Hajj, Lina Tawk, Marwan El Sabban, and et al. 2021. "Comprehensive Overview of Toxoplasma gondii-Induced and Associated Diseases" Pathogens 10, no. 11: 1351. https://doi.org/10.3390/pathogens10111351
APA StyleDaher, D., Shaghlil, A., Sobh, E., Hamie, M., Hassan, M. E., Moumneh, M. B., Itani, S., El Hajj, R., Tawk, L., El Sabban, M., & El Hajj, H. (2021). Comprehensive Overview of Toxoplasma gondii-Induced and Associated Diseases. Pathogens, 10(11), 1351. https://doi.org/10.3390/pathogens10111351





