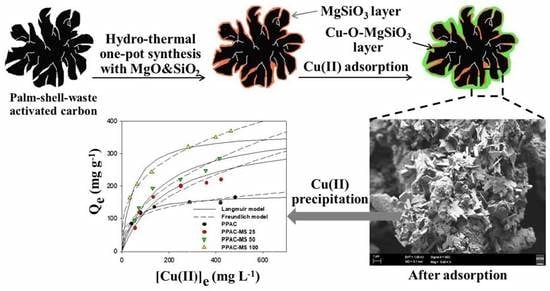
One Step Hydrothermal Synthesis of Magnesium Silicate Impregnated Palm Shell Waste Activated Carbon for Copper Ion Removal
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Preparation of Magnesium Silicate Impregnated on PPAC
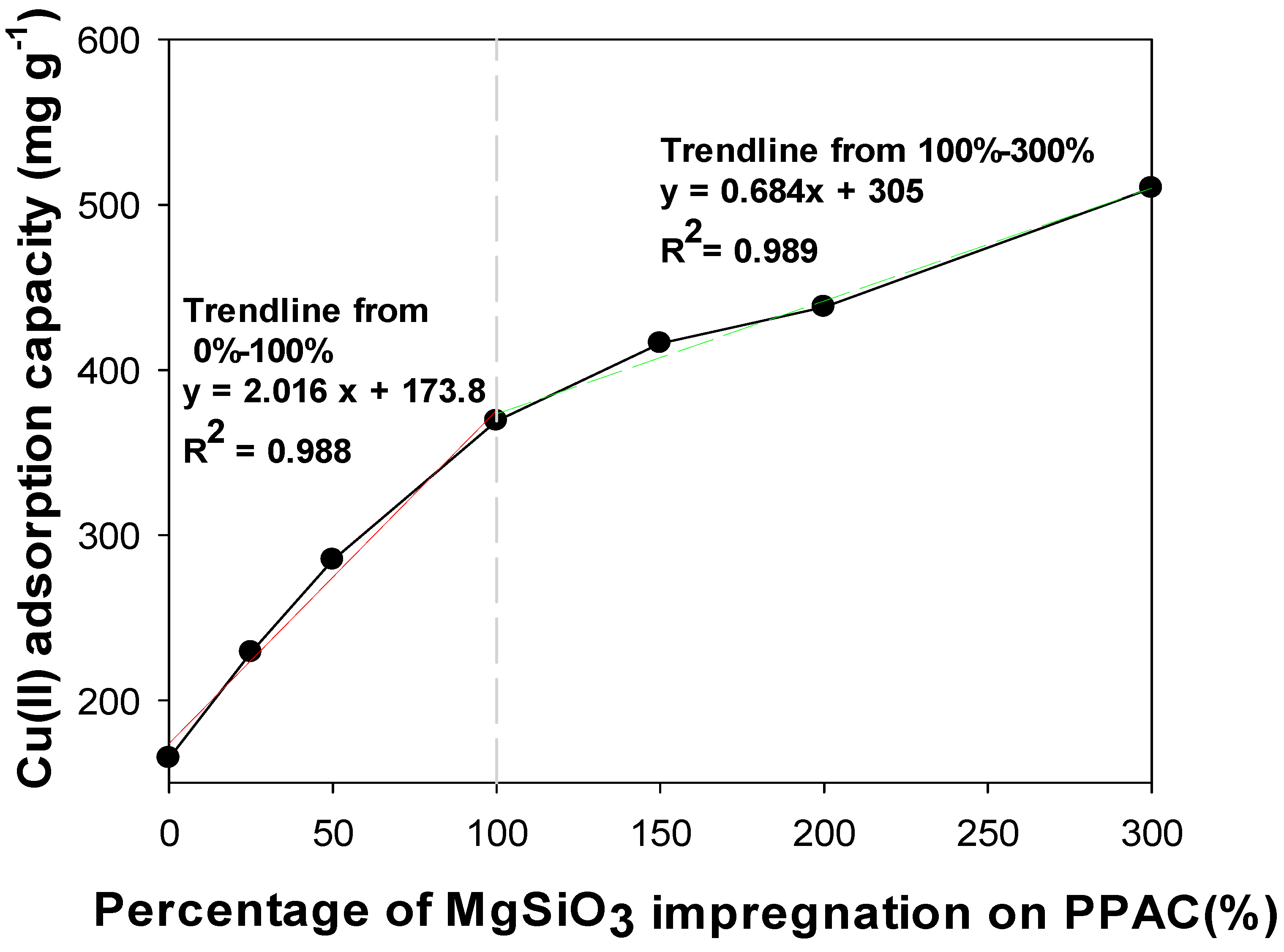
2.1.2. Optimization on Impregnated Ratio of MgSiO3 on PPAC
2.2. Batch Experiments for Cu(II) Removal
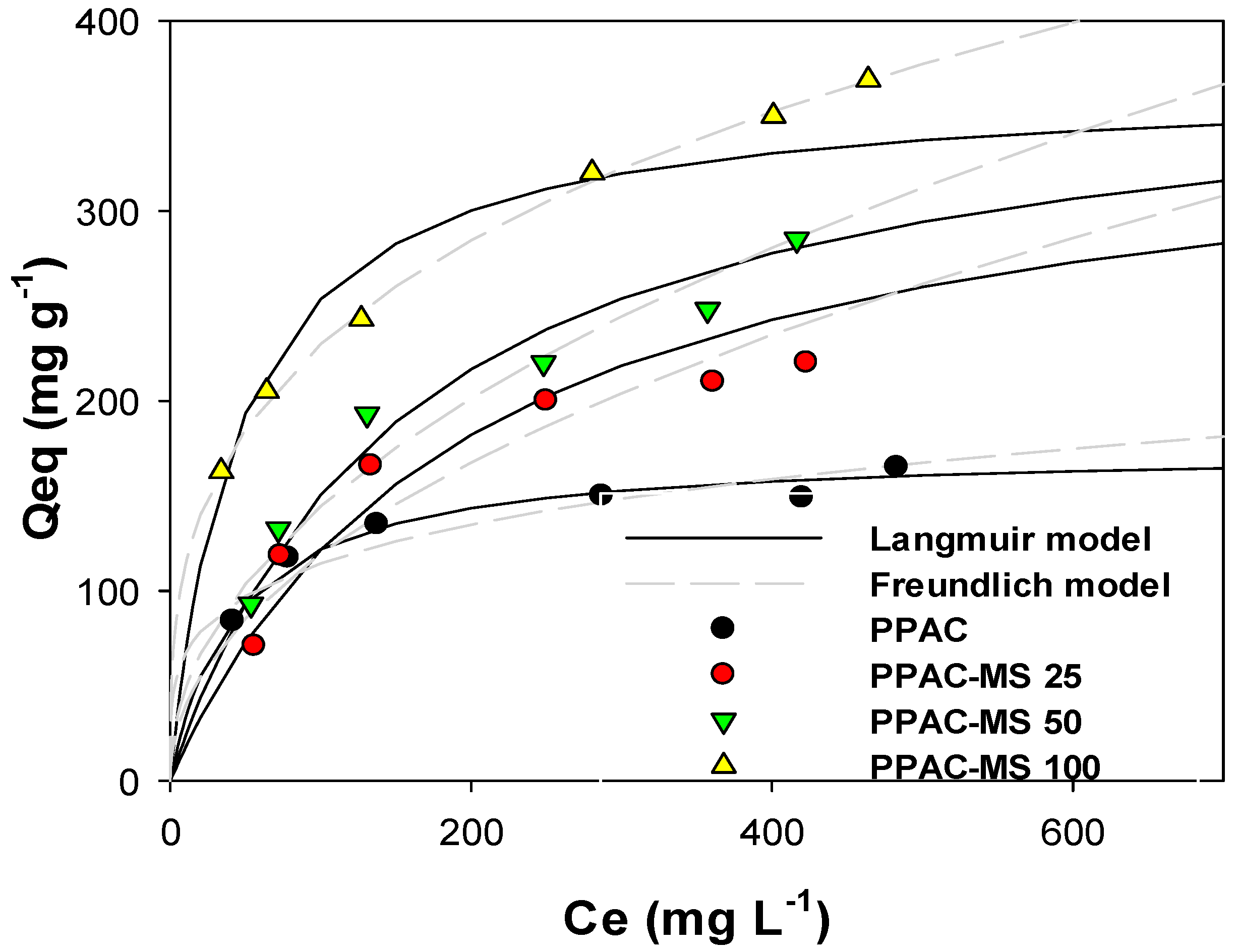
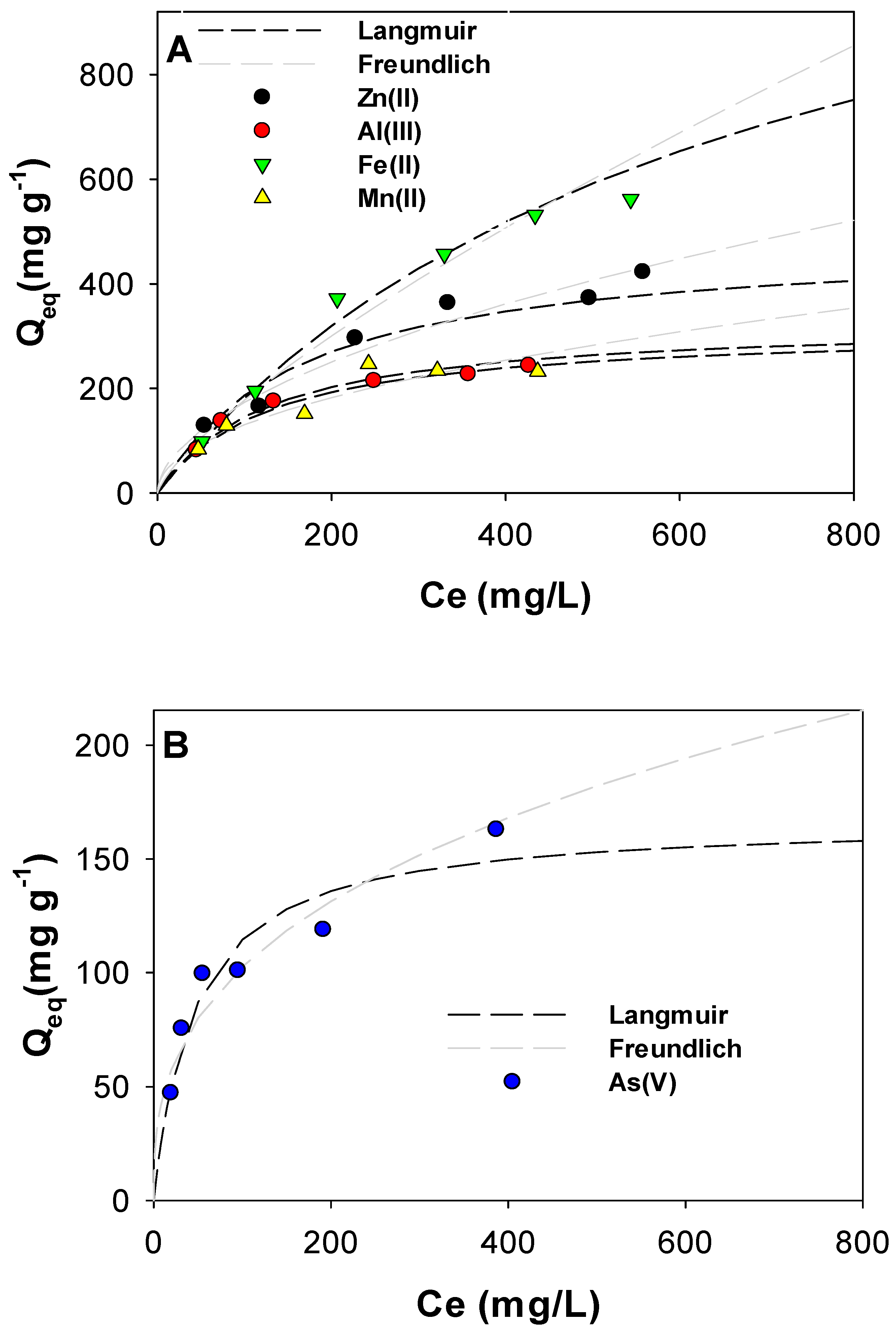
2.2.1. Adsorption Isotherms
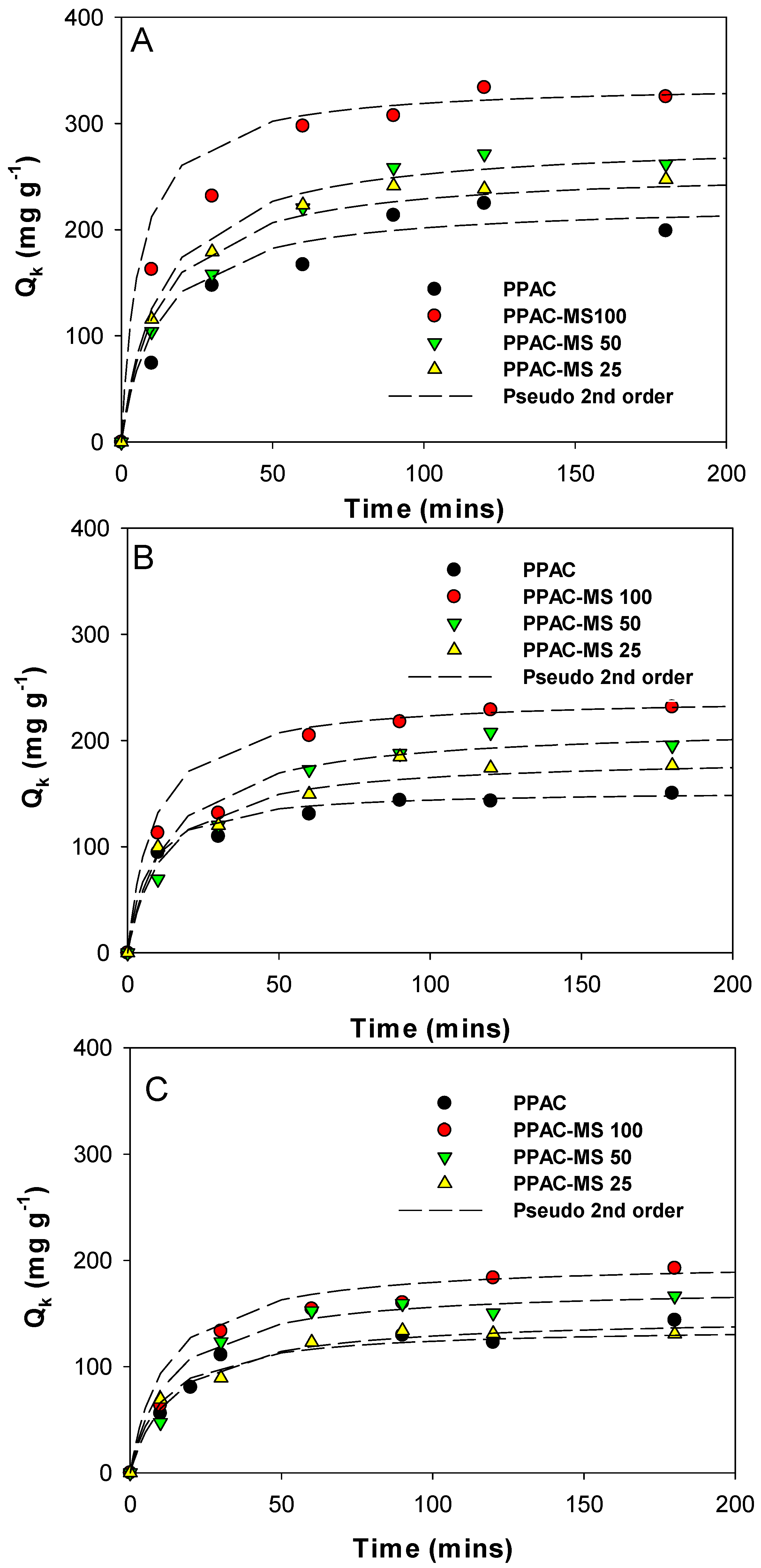
2.2.2. Adsorption Kinetics with Ionic Strength Effect
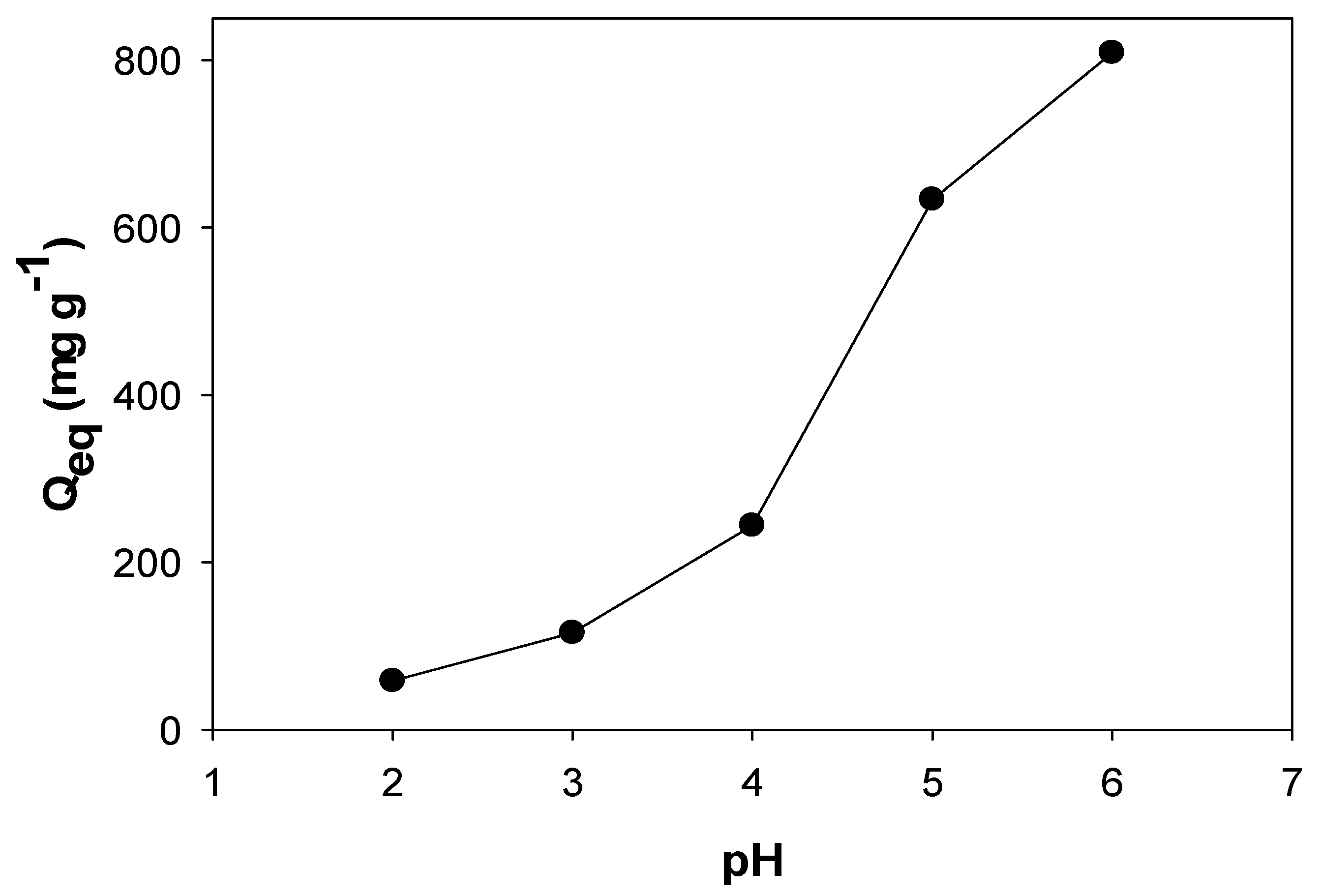
2.2.3. pH Effect on PPAC-MS-100 for Cu(II) Removal
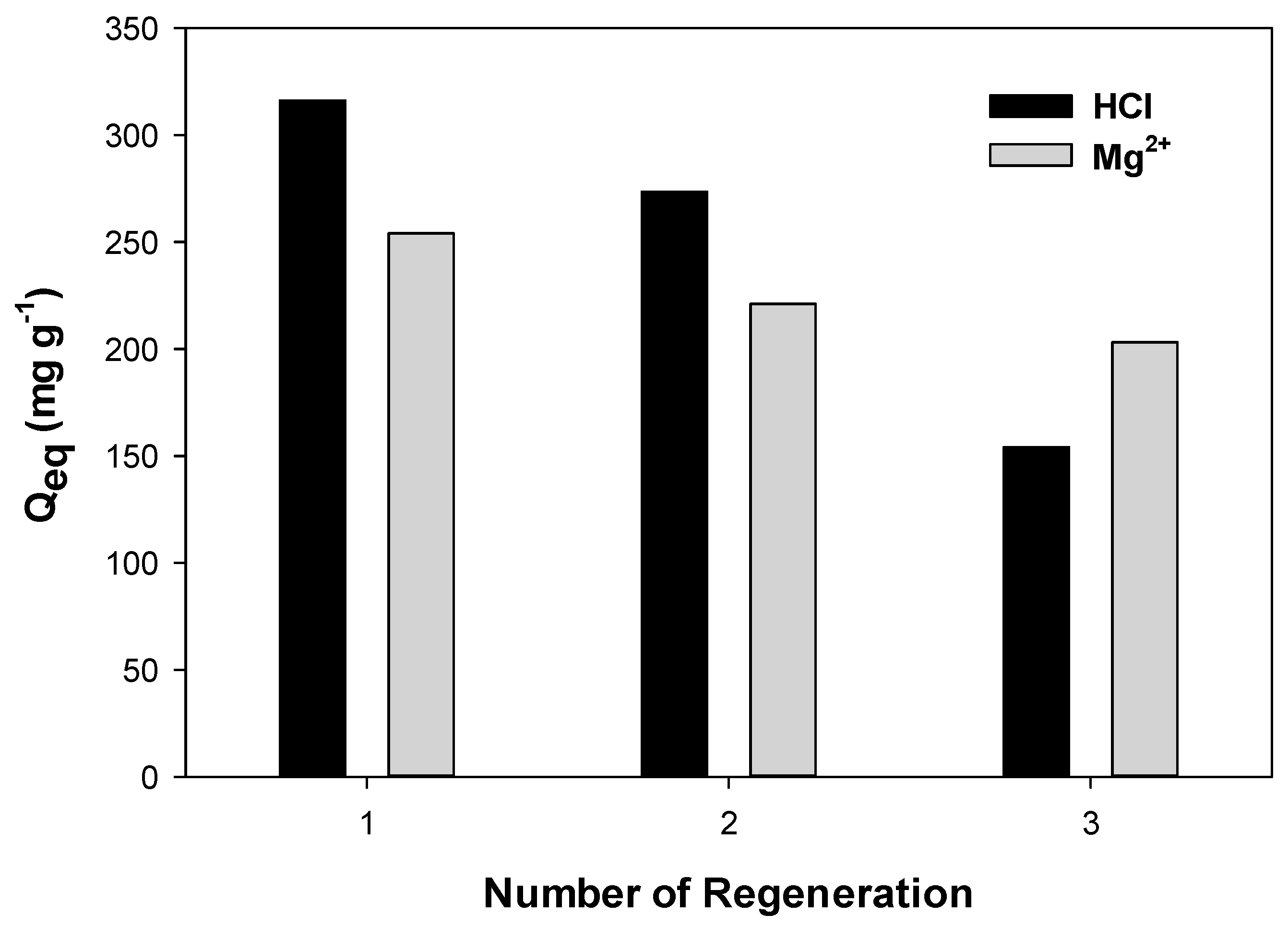
2.2.4. Regeneration of PPAC-MS 100 for Cu(II) Adsorption
2.3. Adsorption Study of Dissociated PTMs by PPAC-MS 100
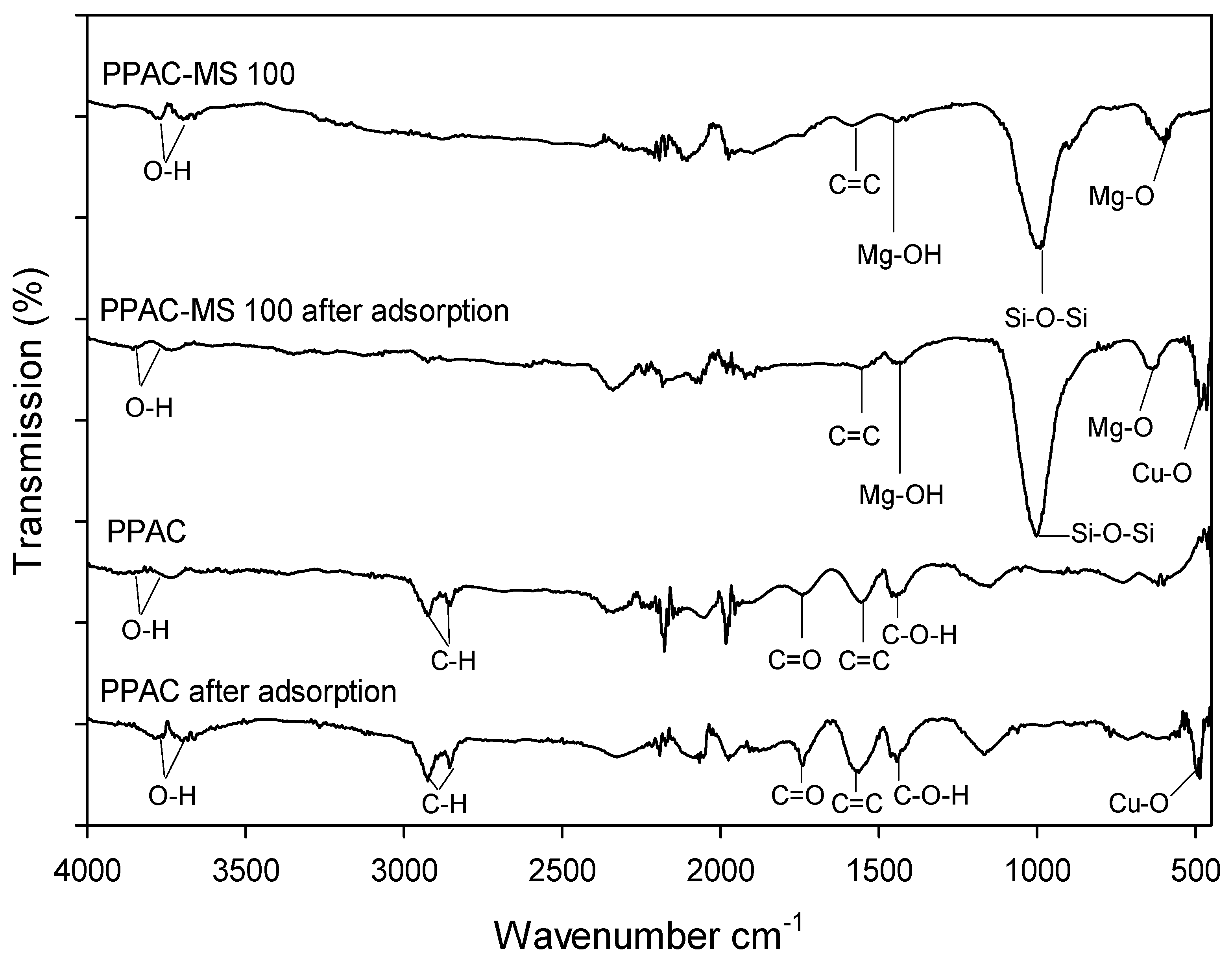
2.4. Material Characterization
3. Results and Discussion
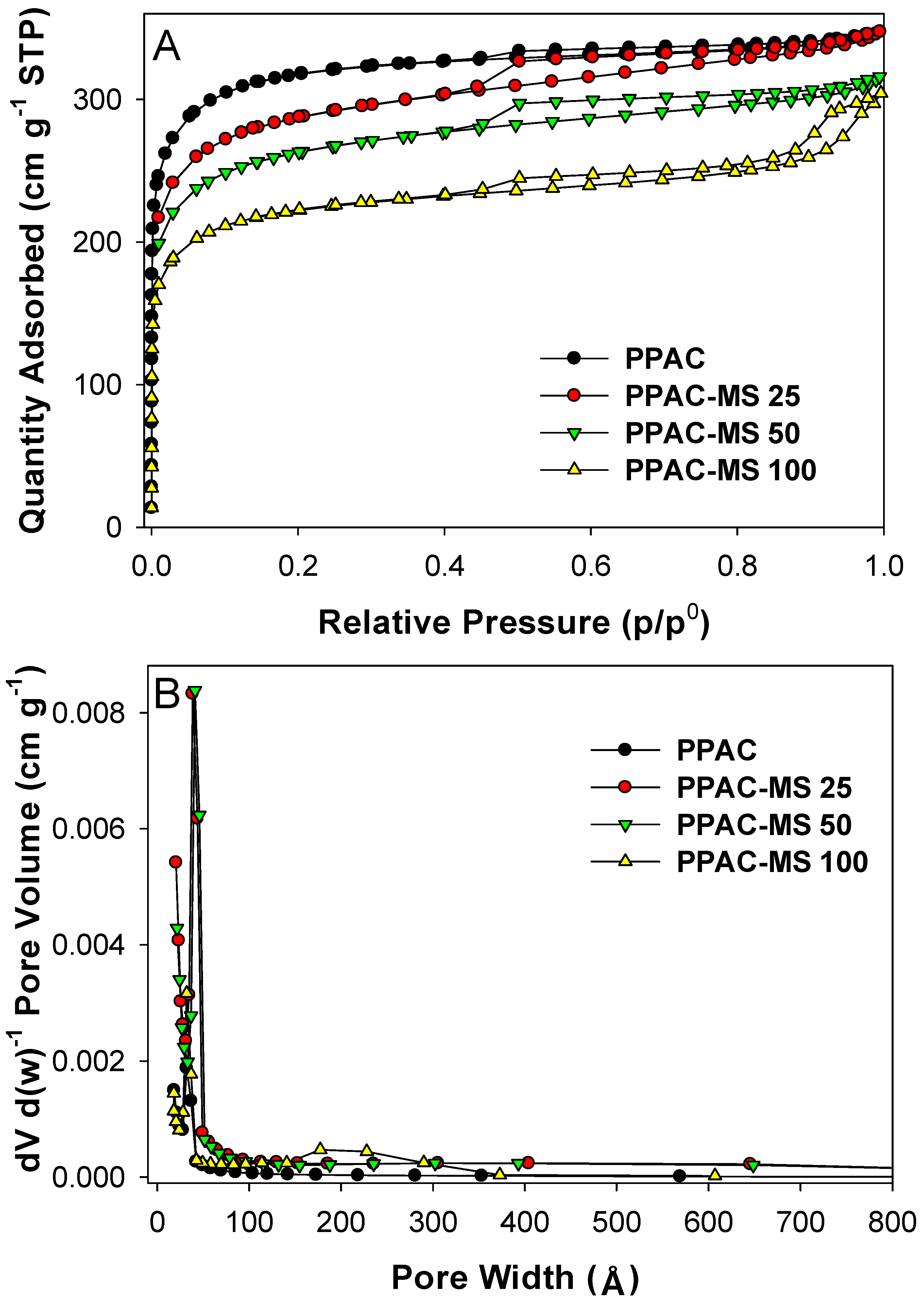
3.1. Optimization on Impregnated Ratio of MgSiO3 on PPAC
3.2. Adsorption Isotherms for Cu(II)
3.3. Adsorption Kinetics with Different Ionic Strength
3.4. Influence of pH on Cu(II) Removal Using PPAC-MS100
3.5. Regeneration PPAC-MS 100
3.6. Adsorption Studies on Zn(II), Al(III), Fe(II), Mn(II) and As(V)
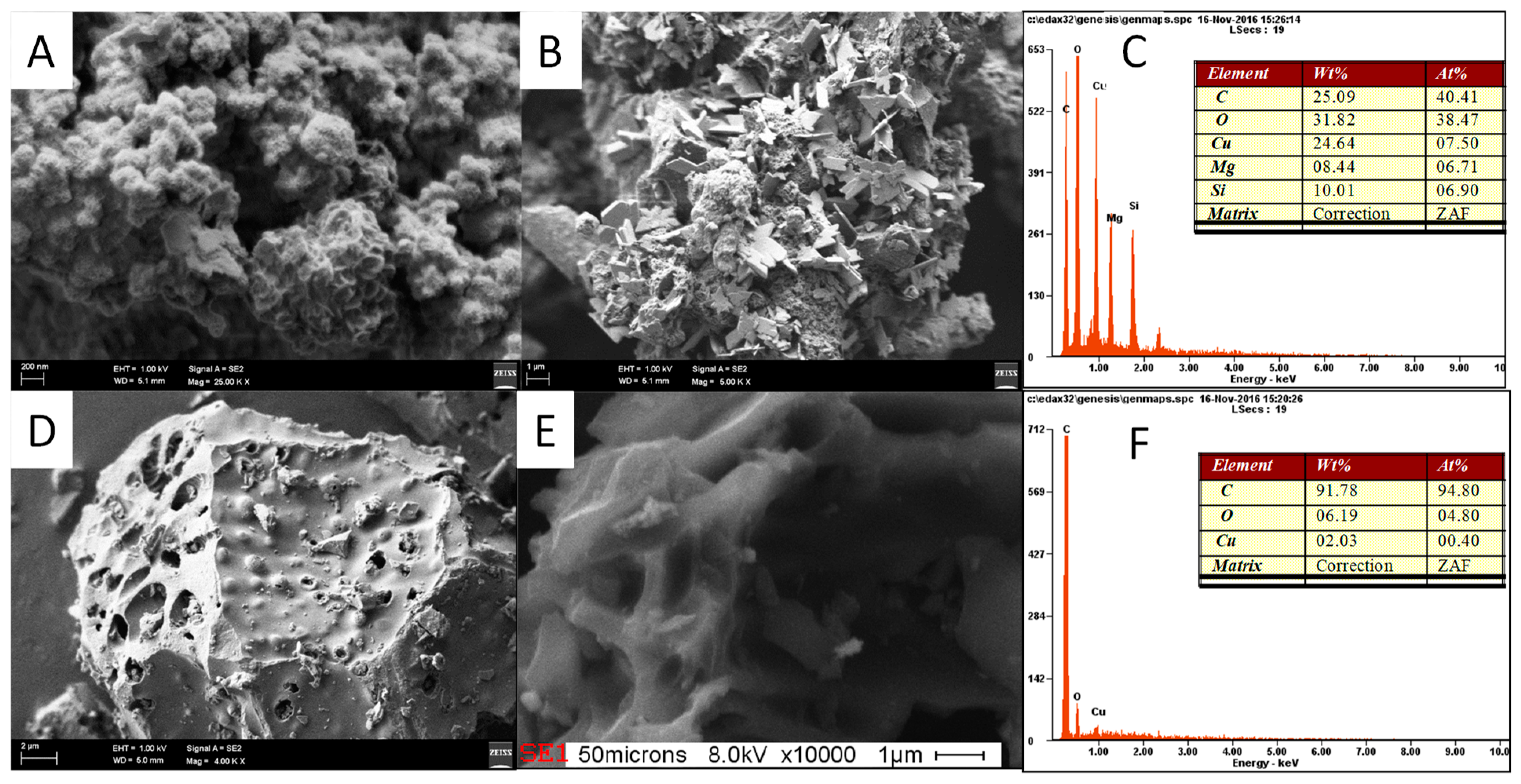
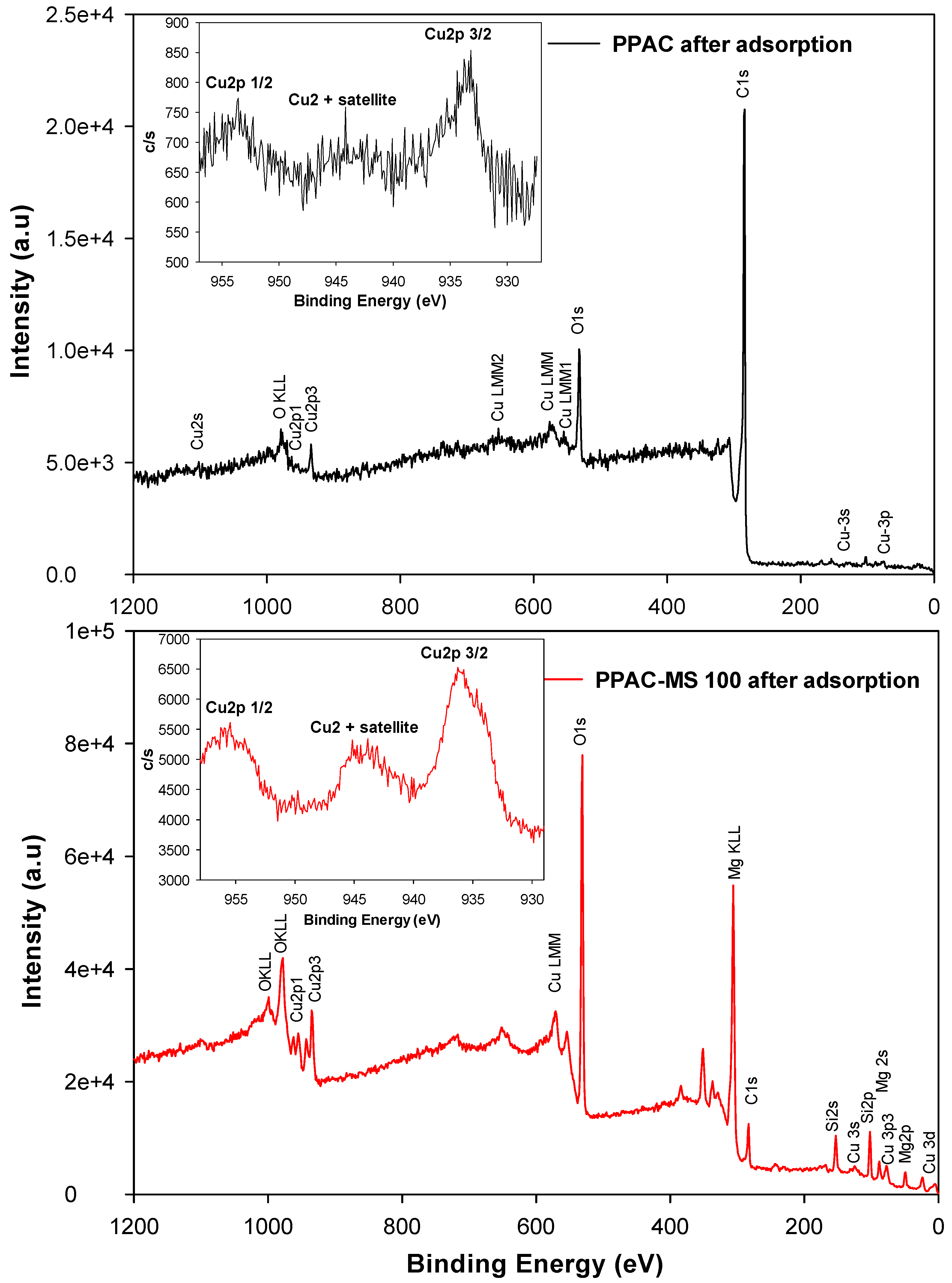
3.7. Copper Removal Mechanisms and Material Characterizations
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Meers, E.; Ruttens, A.; Hopgood, M.J.; Samson, D.; Tack, F.M.G. Comparison of edta and edds as potential soil amendments for enhanced phytoextraction of heavy metals. Chemosphere 2005, 58, 1011–1022. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, R.A.; Williams, H.A.; Rosenoer, V.M.; Sherlock, S. Liver-copper levels in liver disease: Studies using neurtron activation analysis. Lancet 1968, 292, 1310–1313. [Google Scholar] [CrossRef]
- Yang, X.; Li, Q.; Tang, Z.; Zhang, W.; Yu, G.; Shen, Q.; Zhao, F.-J. Heavy metal concentrations and arsenic speciation in animal manure composts in China. Waste Manag. 2017, 64, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Madzin, Z.; Shai-in, M.F.; Kusin, F.M. Comparing heavy metal mobility in active and abandoned mining sites at bestari jaya, selangor. Procedia Environ. Sci. 2015, 30, 232–237. [Google Scholar] [CrossRef]
- Da̧browski, A.; Hubicki, Z.; Podkościelny, P.; Robens, E. Selective removal of the heavy metal ions from waters and industrial wastewaters by ion-exchange method. Chemosphere 2004, 56, 91–106. [Google Scholar] [CrossRef] [PubMed]
- Mobasherpour, I.; Salahi, E.; Ebrahimi, M. Thermodynamics and kinetics of adsorption of Cu(ii) from aqueous solutions onto multi-walled carbon nanotubes. J. Saudi Chem. Soc. 2014, 18, 792–801. [Google Scholar] [CrossRef]
- Barakat, M.A. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Furlan, F.R.; de Melo da Silva, L.G.; Morgado, A.F.; de Souza, A.A.U.; Guelli Ulson de Souza, S.M.A. Removal of reactive dyes from aqueous solutions using combined coagulation/flocculation and adsorption on activated carbon. Resour. Conserv. Recycl. 2010, 54, 283–290. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Deliyanni, E.A.; Bikiaris, D.N.; Mitropoulos, A.C. Graphene composites as dye adsorbents: Review. Chem. Eng. Res. Des. 2018, 129, 75–88. [Google Scholar] [CrossRef]
- Fan, T.-T.; Wang, Y.-J.; Li, C.-B.; He, J.-Z.; Gao, J.; Zhou, D.-M.; Friedman, S.P.; Sparks, D.L. Effect of organic matter on sorption of zn on soil: Elucidation by wien effect measurements and exafs spectroscopy. Environ. Sci. Technol. 2016, 50, 2931–2937. [Google Scholar] [CrossRef] [PubMed]
- Linares, N.; Silvestre-Albero, A.M.; Serrano, E.; Silvestre-Albero, J.; Garcia-Martinez, J. Mesoporous materials for clean energy technologies. Chem. Soc. Rev. 2014, 43, 7681–7717. [Google Scholar] [CrossRef] [PubMed]
- Lua, A.C.; Guo, J. Microporous oil-palm-shell activated carbon prepared by physical activation for gas-phase adsorption. Langmuir 2001, 17, 7112–7117. [Google Scholar] [CrossRef]
- Tsai, W.T.; Chang, C.Y.; Wang, S.Y.; Chang, C.F.; Chien, S.F.; Sun, H.F. Cleaner production of carbon adsorbents by utilizing agricultural waste corn cob. Resour. Conserv. Recycl. 2001, 32, 43–53. [Google Scholar] [CrossRef]
- Yantasee, W.; Lin, Y.; Fryxell, G.E.; Alford, K.L.; Busche, B.J.; Johnson, C.D. Selective removal of copper(ii) from aqueous solutions using fine-grained activated carbon functionalized with amine. Ind. Eng. Chem. Res. 2004, 43, 2759–2764. [Google Scholar] [CrossRef]
- Mahaninia, M.H.; Rahimian, P.; Kaghazchi, T. Modified activated carbons with amino groups and their copper adsorption properties in aqueous solution. Chin. J. Chem. Eng. 2015, 23, 50–56. [Google Scholar] [CrossRef]
- Reza, R.A.; Ahmaruzzaman, M. A novel synthesis of Fe2O3@activated carbon composite and its exploitation for the elimination of carcinogenic textile dye from an aqueous phase. RSC Adv. 2015, 5, 10575–10586. [Google Scholar] [CrossRef]
- Mahdavi, S.; Jalali, M.; Afkhami, A. Removal of heavy metals from aqueous solutions using Fe3O4, ZnO, and CuO nanoparticles. J. Nanopart. Res. 2012, 14, 846. [Google Scholar] [CrossRef]
- Ahn, C.K.; Park, D.; Woo, S.H.; Park, J.M. Removal of cationic heavy metal from aqueous solution by activated carbon impregnated with anionic surfactants. J. Hazard. Mater. 2009, 164, 1130–1136. [Google Scholar] [CrossRef] [PubMed]
- Seymour, M.B.; Su, C.; Gao, Y.; Lu, Y.; Li, Y. Characterization of carbon nano-onions for heavy metal ion remediation. J. Nanopart. Res. 2012, 14, 1087. [Google Scholar] [CrossRef]
- Liao, P.; Yuan, S.; Zhang, W.; Tong, M.; Wang, K. Mechanistic aspects of nitrogen-heterocyclic compound adsorption on bamboo charcoal. J. Colloid Interface Sci. 2012, 382, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Lo, S.-F.; Wang, S.-Y.; Tsai, M.-J.; Lin, L.-D. Adsorption capacity and removal efficiency of heavy metal ions by moso and ma bamboo activated carbons. Chem. Eng. Res. Des. 2012, 90, 1397–1406. [Google Scholar] [CrossRef]
- Wong, S.; Lee, Y.; Ngadi, N.; Inuwa, I.M.; Mohamed, N.B. Synthesis of activated carbon from spent tea leaves for aspirin removal. Chin. J. Chem. Eng. 2018, 26, 1003–1011. [Google Scholar] [CrossRef]
- Shukla, S.R.; Pai, R.S. Adsorption of Cu(ii), Ni(ii) and Zn(ii) on dye loaded groundnut shells and sawdust. Sep. Purif. Technol. 2005, 43, 1–8. [Google Scholar] [CrossRef]
- Lalhmunsiama; Lee, S.M.; Choi, S.S.; Tiwari, D. Simultaneous removal of Hg(ii) and phenol using functionalized activated carbon derived from areca nut waste. Metals 2017, 7, 248. [Google Scholar] [CrossRef]
- Hameed, B.H.; El-Khaiary, M.I. Malachite green adsorption by rattan sawdust: Isotherm, kinetic and mechanism modeling. J. Hazard. Mater. 2008, 159, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Sathishkumar, M.; Binupriya, A.R.; Kavitha, D.; Selvakumar, R.; Sheema, K.K.; Choi, J.G.; Yun, S.E. Organic micro-pollutant removal in liquid-phase using carbonized silk cotton hull. J. Environ. Sci. 2008, 20, 1046–1054. [Google Scholar] [CrossRef]
- Jais, F.M.; Ibrahim, S.; Yoon, Y.; Jang, M. Enhanced arsenate removal by lanthanum and nano–magnetite composite incorporated palm shell waste–based activated carbon. Sep. Purif. Technol. 2016, 169, 93–102. [Google Scholar] [CrossRef]
- Yu, Y.; Hu, Z.; Chen, Z.; Yang, J.; Gao, H.; Chen, Z. Organically-modified magnesium silicate nanocomposites for high-performance heavy metal removal. RSC Adv. 2016, 6, 97523–97531. [Google Scholar] [CrossRef]
- Lu, H.; Wang, J.; Stoller, M.; Wang, T.; Bao, Y.; Hao, H. An overview of nanomaterials for water and wastewater treatment. Adv. Mater. Sci. Eng. 2016, 2016, 4964828. [Google Scholar] [CrossRef]
- Brar, S.K.; Verma, M.; Tyagi, R.D.; Surampalli, R.Y. Engineered nanoparticles in wastewater and wastewater sludge—Evidence and impacts. Waste Manag. 2010, 30, 504–520. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, M.E.; Khalifa, M.A.; El Wakeel, Y.M.; Header, M.S.; Abdel-Fattah, T.M. Engineered nano-magnetic iron oxide-urea-activated carbon nanolayer sorbent for potential removal of uranium (vi) from aqueous solution. J. Nucl. Mater. 2017, 487, 13–22. [Google Scholar] [CrossRef]
- Karnib, M.; Kabbani, A.; Holail, H.; Olama, Z. Heavy metals removal using activated carbon, silica and silica activated carbon composite. Energy Procedia 2014, 50, 113–120. [Google Scholar] [CrossRef]
- Wang, M.C.; Sheng, G.D.; Qiu, Y.P. A novel manganese-oxide/biochar composite for efficient removal of lead(ii) from aqueous solutions. Int. J. Environ. Sci. Technol. 2015, 12, 1719–1726. [Google Scholar] [CrossRef]
- Gaya, U.I.; Otene, E.; Abdullah, A.H. Adsorption of aqueous Cd(ii) and Pb(ii) on activated carbon nanopores prepared by chemical activation of doum palm shell. Springerplus 2015, 4, 458. [Google Scholar] [CrossRef] [PubMed]
- Choong, C.E.; Ibrahim, S.; Yoon, Y.; Jang, M. Removal of lead and bisphenol a using magnesium silicate impregnated palm-shell waste powdered activated carbon: Comparative studies on single and binary pollutant adsorption. Ecotoxicol. Environ. Saf. 2018, 148, 142–151. [Google Scholar] [CrossRef] [PubMed]
- St. Vassileva, P.; Detcheva, A.K. Adsorption of some transition metal ions [Cu(ii), Fe(iii), Cr(iii) and Au(iii)] onto lignite-based activated carbons modified by oxidation. Adsorpt. Sci. Technol. 2010, 28, 229–242. [Google Scholar]
- ASTM. Astm d1976-18. In Standard Test Method for Elements in Water by Inductively-Coupled Plasma Atomic Emission Spectroscopy; ASTM International: West Conshohocken, PA, USA, 2018. [Google Scholar]
- Zhan, W.; Xu, C.; Qian, G.; Huang, G.; Tang, X.; Lin, B. Adsorption of Cu(ii), Zn(ii), and Pb(ii) from aqueous single and binary metal solutions by regenerated cellulose and sodium alginate chemically modified with polyethyleneimine. RSC Adv. 2018, 8, 18723–18733. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Karamesouti, M.; Mitropoulos, A.C.; Kyzas, G.Z. A review for coffee adsorbents. J. Mol. Liq. 2017, 229, 555–565. [Google Scholar] [CrossRef]
- Rafiq, Z.; Nazir, R.; Shah, M.R.; Ali, S. Utilization of magnesium and zinc oxide nano-adsorbents as potential materials for treatment of copper electroplating industry wastewater. J. Environ. Chem. Eng. 2014, 2, 642–651. [Google Scholar] [CrossRef]
- Zhang, K.; Li, H.; Xu, X.; Yu, H. Facile and efficient synthesis of nitrogen-functionalized graphene oxide as a copper adsorbent and its application. Ind. Eng. Chem. Res. 2016, 55, 2328–2335. [Google Scholar] [CrossRef]
- Wang, P.; Ye, Y.; Liang, D.; Sun, H.; Liu, J.; Tian, Z.; Liang, C. Layered mesoporous Mg(OH)2/GO nanosheet composite for efficient removal of water contaminants. RSC Adv. 2016, 6, 26977–26983. [Google Scholar] [CrossRef]
- Kadirvelu, K.; Faur-Brasquet, C.; Cloirec, P.L. Removal of Cu(ii), Pb(ii), and Ni(ii) by adsorption onto activated carbon cloths. Langmuir 2000, 16, 8404–8409. [Google Scholar] [CrossRef]
- Khodaie, M.; Ghasemi, N.; Moradi, B.; Rahimi, M. Removal of methylene blue from wastewater by adsorption onto ZnCl2 activated corn husk carbon equilibrium studies. J. Chem. 2013, 2013, 383985. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–471. [Google Scholar]
- ElShafei, G.M.S.; ElSherbiny, I.M.A.; Darwish, A.S.; Philip, C.A. Silkworms’ feces-based activated carbons as cheap adsorbents for removal of cadmium and methylene blue from aqueous solutions. Chem. Eng. Res. Des. 2014, 92, 461–470. [Google Scholar] [CrossRef]
- Yakout, S.M.; Borai, E.H. Adsorption behavior of cadmium onto natural chabazite: Batch and column investigations. Desalination Water Treat. 2014, 52, 4212–4222. [Google Scholar] [CrossRef]
- Xu, J.; Wang, L.; Zhu, Y. Decontamination of bisphenol a from aqueous solution by graphene adsorption. Langmuir 2012, 28, 8418–8425. [Google Scholar] [CrossRef] [PubMed]
- Nouri, L.; Ghodbane, I.; Hamdaoui, O.; Chiha, M. Batch sorption dynamics and equilibrium for the removal of cadmium ions from aqueous phase using wheat bran. J. Hazard. Mater. 2007, 149, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.D.; Watson, M.A.; Brown, J.; Jefcoat, I.A. Peanut hull pellets as a single use sorbent for the capture of Cu(ii) from wastewater. Waste Manag. 2002, 22, 471–480. [Google Scholar] [CrossRef]
- Ou, Q.; Zhou, L.; Zhao, S.; Geng, H.; Hao, J.; Xu, Y.; Chen, H.; Chen, X. Self-templated synthesis of bifunctional Fe3O4@MgSiO3 magnetic sub-microspheres for toxic metal ions removal. Chem. Eng. J. 2012, 180, 121–127. [Google Scholar] [CrossRef]
- Sharma, Y.C.; Srivastava, V.; Singh, V.K.; Kaul, S.N.; Weng, C.H. Nano-adsorbents for the removal of metallic pollutants from water and wastewater. Environ. Technol. 2009, 30, 583–609. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.X.; Huang, M.; Li, F.; Wen, Z.Q. Controlled synthesis of hierarchical cuo nanostructures for electrochemical capacitor electrodes. Int. J. Electrochem. Sci. 2013, 8, 8645–8661. [Google Scholar]
- Tao, Q.; Zhu, J.; Frost, R.L.; Bostrom, T.E.; Wellard, R.M.; Wei, J.; Yuan, P.; He, H. Silylation of layered double hydroxides via a calcination–rehydration route. Langmuir 2010, 26, 2769–2773. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Zheng, H.; Yin, J.; Lu, Y.; Wu, S.; Wu, X.; Li, B. Mg(OH)2 complex nanostructures with superhydrophobicity and flame retardant effects. J. Phys. Chem. C 2010, 114, 17362–17368. [Google Scholar] [CrossRef]
- Dhaouadi, H.; Chaabane, H.; Touati, F. Mg(OH)2 nanorods synthesized by a facile hydrothermal method in the presence of ctab. Nano-Micro Lett. 2011, 3, 153–159. [Google Scholar] [CrossRef]
- Pei, L.-Z.; Yin, W.-Y.; Wang, J.-F.; Chen, J.; Fan, C.-G.; Zhang, Q.-F. Low temperature synthesis of magnesium oxide and spinel powders by a sol-gel process. Mater. Res. 2010, 13, 339–343. [Google Scholar] [CrossRef]
- Viana, R.B.; da Silva, A.B.F.; Pimentel, A.S. Infrared spectroscopy of anionic, cationic, and zwitterionic surfactants. Adv. Chem. Phys. 2012, 2012, 903272. [Google Scholar] [CrossRef]
- Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta 2007, 1767, 1073–1101. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhao, J.; Li, H.; Zhang, X.; Shi, H. Removal of cadmium(ii) from aqueous solution by granular activated carbon supported magnesium hydroxide. J. Taiwan Inst. Chem. Eng. 2016, 61, 287–291. [Google Scholar] [CrossRef]
- Przepiórski, J.; Czyżewski, A.; Pietrzak, R.; Tryba, B. MgO/CaO-loaded porous carbons for carbon dioxide capture. J. Therm. Anal. Calorim. 2013, 111, 357–364. [Google Scholar] [CrossRef]
- Li, B.; Luo, X.; Zhu, Y.; Wang, X. Immobilization of Cu(ii) in kit-6 supported Co3O4 and catalytic performance for epoxidation of styrene. Appl. Surf. Sci. 2015, 359, 609–620. [Google Scholar] [CrossRef]
- Hu, L.; Shi, L.; Hong, H.; Li, M.; Bao, Q.; Tang, J.; Ge, J.; Lu, J.; Cao, X.; Gu, H. Catalytic epoxidation of stilbene with FePt@Cu nanowires and molecular oxygen. Chem. Commun. 2010, 46, 8591–8593. [Google Scholar] [CrossRef] [PubMed]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choong, C.E.; Lee, G.; Jang, M.; Park, C.M.; Ibrahim, S. One Step Hydrothermal Synthesis of Magnesium Silicate Impregnated Palm Shell Waste Activated Carbon for Copper Ion Removal. Metals 2018, 8, 741. https://doi.org/10.3390/met8100741
Choong CE, Lee G, Jang M, Park CM, Ibrahim S. One Step Hydrothermal Synthesis of Magnesium Silicate Impregnated Palm Shell Waste Activated Carbon for Copper Ion Removal. Metals. 2018; 8(10):741. https://doi.org/10.3390/met8100741
Chicago/Turabian StyleChoong, Choe Earn, Gooyong Lee, Min Jang, Chang Min Park, and Shaliza Ibrahim. 2018. "One Step Hydrothermal Synthesis of Magnesium Silicate Impregnated Palm Shell Waste Activated Carbon for Copper Ion Removal" Metals 8, no. 10: 741. https://doi.org/10.3390/met8100741
APA StyleChoong, C. E., Lee, G., Jang, M., Park, C. M., & Ibrahim, S. (2018). One Step Hydrothermal Synthesis of Magnesium Silicate Impregnated Palm Shell Waste Activated Carbon for Copper Ion Removal. Metals, 8(10), 741. https://doi.org/10.3390/met8100741