Abstract
This study quantitatively evaluates the effect of solid-phase pre-reduction of chromite concentrate on the energy efficiency and techno-economic performance of high-carbon ferrochrome (HC FeCr) smelting. Laboratory pre-reduction experiments were conducted at 1200–1400 °C using Shubarkol coal, metallurgical coke, and special coke as carbonaceous reducing agents. Structural and phase transformations were characterized by X-ray diffraction (XRD) and scanning electron microscopy with energy-dispersive spectroscopy (SEM-EDS). At 1200 °C, the degree of metallization remained low (<5%), whereas at 1400 °C it increased to 41.3% under laboratory conditions and up to 65% in pilot-scale tests due to the decomposition of the spinel matrix and the formation of metallic and carbide phases. The application of pre-reduced feedstock in a submerged arc furnace reduced specific electricity consumption by up to 33.5% compared with conventional smelting and increased chromium recovery to 89.71%. Industrial-scale extrapolation indicates the potential to decrease power consumption to approximately 3190 kWh/t of alloy. Techno-economic analysis demonstrates that the use of pre-reduced feedstock reduces the production cost by approximately 10–23%, depending on the type of carbonaceous reducing agent (Shubarkol coal, metallurgical coke, or special coke). Special coke provided the highest energy efficiency, whereas Shubarkol coal ensured the greatest direct economic benefit. The integrated microstructural, energetic, and economic assessment confirms the industrial applicability of the proposed pre-reduction approach.
1. Introduction
With the continuous development of modern technologies and engineering industries, the demand for stainless steel grades is steadily increasing, which in turn drives the demand for ferrochrome as the principal alloying element. More than 80% of global ferrochrome production is consumed in stainless steel manufacturing, making ferrochrome a strategically important ferroalloy for the metallurgical industry as a whole. In 2019, global stainless steel production reached approximately 52.2 million tons, of which 29.4 million tons were produced in China. All stainless steel grades contain at least 10.5% chromium [1,2].
Global chromite reserves are unevenly distributed. The three largest resource holders are South Africa (72%), Zimbabwe (12%), and Kazakhstan (5%). Ore quality is primarily determined by the Cr/Fe ratio; metallurgical-grade ores typically exhibit a Cr/Fe ratio exceeding 2.8. In 2024, global chromite production exceeded 39 million tons, while total world resources are estimated at approximately 12 billion tons. According to official ICDA data, global production in 2024 amounted to 17 million tons of high-carbon ferrochrome (HC FeCr), 23 thousand tons of medium-carbon ferrochrome, and 0.9 million tons of low-carbon ferrochrome. Of this total, 9.4 million tons were produced in China, 3.9 million tons in South Africa and Zimbabwe, and 1.9 million tons in the CIS countries [3,4].
The conventional production of high-carbon ferrochrome in submerged arc furnaces (SAF) is among the most energy-intensive pyrometallurgical processes. Depending on furnace type, charge composition, and smelting regime, specific electricity consumption typically ranges from 2400 to 4700 kWh per ton of alloy [4,5,6,7]. For open AC furnaces, values of 4000–4300 kWh/t are commonly reported, whereas the use of closed furnaces with pelletized and preheated feed allows a reduction to approximately 3100–3400 kWh/t. The lowest electrical energy demand is achieved in process routes combining pre-reduction with smelting in closed SAF units, where electricity consumption may decrease to around 2400 kWh/t (excluding fuel consumption during the pre-reduction stage). Under conditions of rising energy prices and increasing decarbonization requirements in metallurgy, reducing specific energy consumption remains a key industry challenge [8].
Modern approaches to improving ferrochrome production efficiency are largely based on preliminary preparation and partial solid-state reduction of chromite raw materials. The most widespread industrial scheme involves pelletizing the concentrate with a carbonaceous reductant, followed by pre-reduction in rotary kilns and subsequent smelting in a closed submerged arc furnace [9,10]. Shifting a portion of the endothermic reduction reactions from the electric arc zone to a fuel-fired unit reduces electrical load, increases chromium metallization, and stabilizes furnace operation. However, the effectiveness of such schemes strongly depends on the type of carbonaceous reductant, the mineralogical and chemical composition of the feedstock, and the thermal treatment regime.
The energy efficiency and environmental sustainability of ferrochrome production are also influenced by gaseous emissions and solid wastes generated during processing. Carbothermic reduction is accompanied by the formation of CO and CO2, dust emissions, and solid residues containing chromium compounds. Particular attention is given to controlling the formation of hexavalent chromium (Cr (VI)) in off-gas dust and slags, which necessitates effective capture and stabilization systems [11,12,13]. Therefore, technological solutions aimed at enhancing chromium reduction and minimizing Cr2O3 losses to slag simultaneously contribute to improved energy and environmental performance [14,15,16,17,18].
Despite the availability of industrial pre-reduction technologies, the influence of various carbonaceous reductants available in regional markets on the degree of chromite pre-reduction and the subsequent techno-energy performance of high-carbon ferrochrome smelting remains insufficiently investigated. Particular interest lies in evaluating such processes under pilot-scale conditions and in small-capacity submerged arc furnaces, where specific thermal and electrical losses are generally higher than in large industrial units.
This study presents the results of pilot-scale investigations of chromite concentrate pre-reduction conducted at 1400 °C with a holding time of 20 h using different carbonaceous reductants in a chamotte-lined chamber furnace with internal dimensions of 0.92 × 0.80 × 1.10 m (depth × width × height), corresponding to a working volume of approximately 0.81 m3. Four crucibles containing approximately 10 kg of ore–carbon mixture each were placed in the furnace, resulting in a total charge mass exceeding 40 kg per experiment. The obtained pre-reduced materials were subsequently smelted in a closed submerged arc furnace, enabling quantitative assessment of the influence of reductant type and degree of pre-reduction on specific electricity consumption and techno-economic performance.
The scientific novelty of this work lies in the comprehensive experimental validation of chromite concentrate pre-reduction using various carbonaceous reductants at pilot scale under conditions of 1400 °C and a holding time of 20 h, combined with microstructural (SEM-EDS), phase (XRD), and techno-economic analysis of the subsequent high-carbon ferrochrome smelting process.
2. Materials and Methods
Chrome concentrate from the Khromtau deposit (Aktobe region, Kazakhstan) was used as the feedstock. The following carbonaceous reducing agents were used: special coke, metallurgical coke (Karaganda region, Kazakhstan), and coal from the Shubarkol deposit (Ulytau region, Kazakhstan). The chemical composition of the chrome concentrate, as well as the technical and chemical composition of the carbonaceous reducing agents, are presented below in Table 1 and Table 2.

Table 1.
Chemical composition of chrome concentrate.

Table 2.
Technical and chemical composition of carbon reducing agents.
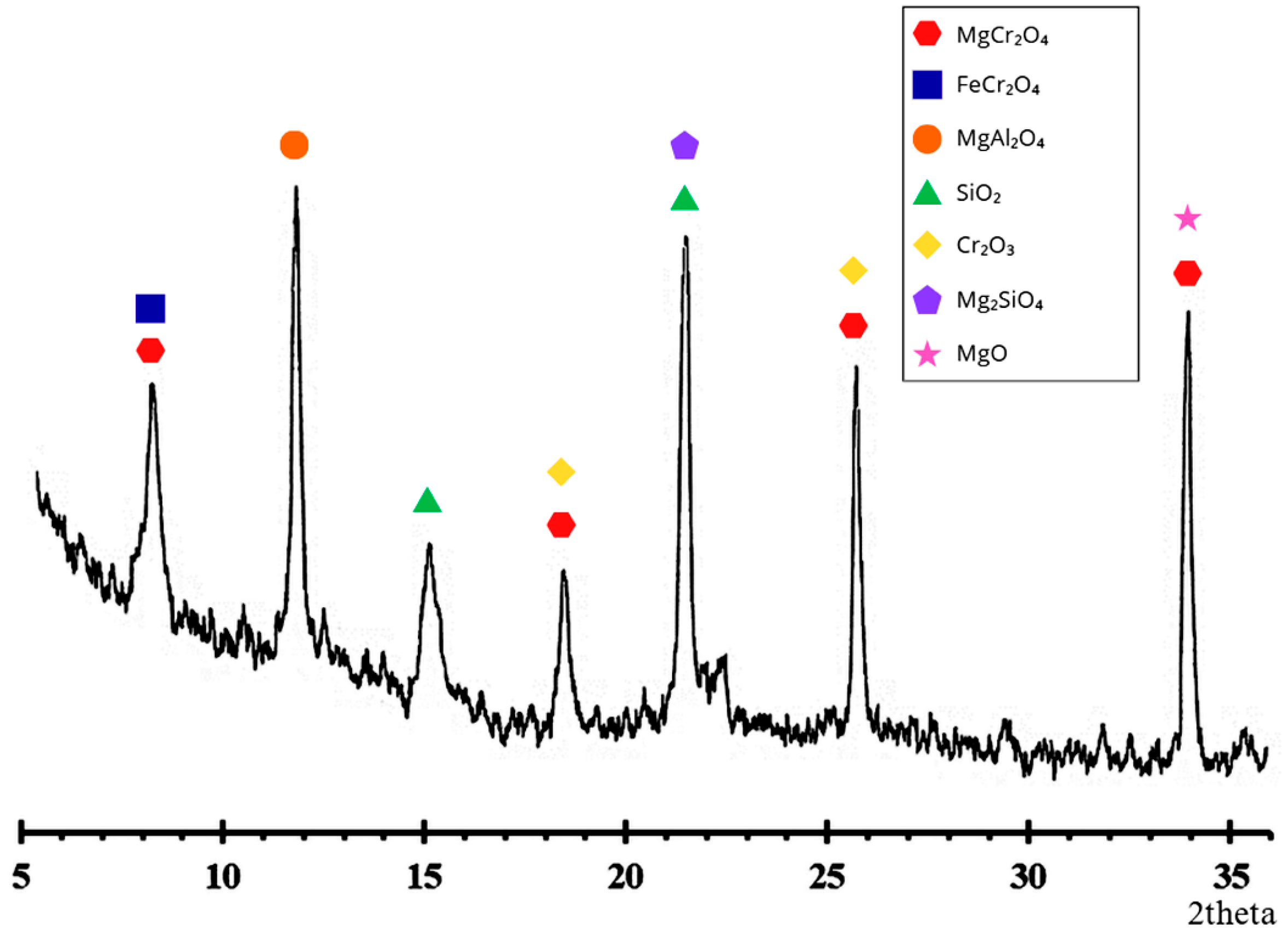
X-ray diffraction (XRD) analysis was performed on an Empyrean Malvern Panalytical diffractometer (Malvern Panalytical, Almelo, The Netherlands) (CuKα, 2θ range 0–80°) with a scanning step of 0.01° to determine the phase compounds of the chrome concentrate. The XRD results are shown in Figure 1.
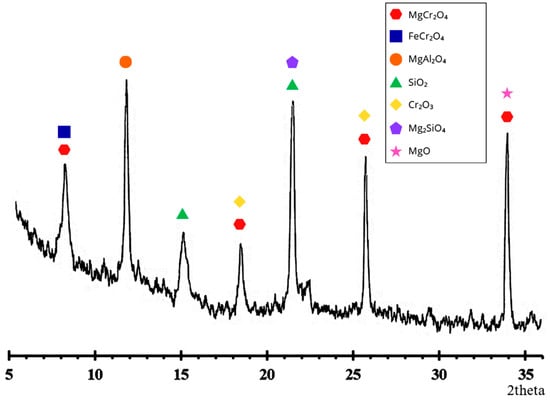
Figure 1.
Diffraction pattern of chrome concentrate.
X-ray phase analysis results (Figure 1) indicate that the original chromium concentrate consists predominantly of spinel phases of chromite. The main identified compounds are MgCr2O4 and FeCr2O4, which is typical of natural chromite concentrates.
Additionally, the sample contained MgAl2O4, indicating isomorphic substitution of Cr3+ and Fe2+ by aluminum in the spinel structure. SiO2 and Mg2SiO4 phases were also detected, indicating the presence of silicate impurities and associated minerals in the concentrate. The presence of individual Cr2O3 peaks may be due to partial spinel decomposition or the presence of free chromium oxide in the feedstock. MgO peaks are weak and are likely due to residual mineral formation products or ore processing [19,20].
Thus, the phase composition of the concentrate is represented predominantly by the spinel system MgO-FeO-Cr2O3 with an admixture of aluminum- and silicon-containing compounds, which corresponds to the typical mineralogical composition of chromite raw materials and determines its reducing behavior during heat treatment.
Scanning electron microscopy (SEM) with energy-dispersive spectroscopy (EDS) was performed on a ZEM20 microscope (ZEPTOOLS, Tongling City, China) equipped with an Oxford energy-dispersive spectroscopy (EDS) detector (Oxford Instruments, Abingdon, UK). Spectra were processed automatically using the AzTec One software package (AzTec One version 6.0 SP1) and the TrueQ method.
According to SEM data, the chrome concentrate is represented by angular particles of varying sizes with a developed surface and microcracks, which is typical of crushed raw materials and promotes the intensification of solid-phase reduction reactions due to the increased reaction surface (Figure 2a). EDS analysis (Table 3) revealed pronounced microchemical heterogeneity: individual areas are enriched in Cr and correspond to chromium-containing spinel phases (spectrum 1), others are characterized by elevated Si and Mg contents and belong to a silicate-oxide matrix (spectrum 2), while zones with elevated Fe and moderate Cr (spectrum 3) may be associated with iron-containing spinel or oxide phases. A slight presence of sulfur is recorded locally. The presence of carbon in all spectra is due to the peculiarities of sample placement on carbon tape and surface carbon contamination during electron irradiation in the microscope chamber. The element distribution maps (Figure 2b) confirm the uneven distribution of Cr, Fe, and Si, and the co-presence of Mg and Al corresponds to the typical spinel mineralogy of chromite concentrate.

Figure 2.
Energy dispersive spectroscopy (EDS) image of chrome concentrate: (a) spectral analysis of chrome concentrate; (b) distribution map of elements in chrome concentrate.

Table 3.
Results of energy dispersive spectroscopy (EDS) of chrome concentrate.
Pre-reduction annealing was performed in a laboratory muffle furnace SNOL (SnolTherm, UAB, Narkūnai, Lithuania) with a maximum operating temperature of 1600 °C. The charge ratio was calculated based on the stoichiometry of the basic reaction of chromium oxide and carbon:
and the ratio of solid carbon to chromium oxide was taken to be 0.4 (2).
This value was selected based on stoichiometric calculations for the carbothermic reduction reaction Cr2O3 + 3C = 2Cr + 3CO, for which the theoretical mass ratio C/Cr2O3 is approximately 0.24. In practical systems, an excess of carbon is required due to carbon consumption in side reactions such as the Boudouard reaction, reduction of iron-bearing oxides, and carbon losses associated with CO gas evolution. Therefore, the selected ratio ensures sufficient reducing agent availability for effective reduction of chromium-bearing phases during the pre-reduction process.
The percentage ratio of the charge mixture is given in Table 4.

Table 4.
Percentage ratio of the charge mixture.
The resulting pre-reduced chrome concentrates were sent for chemical composition analysis to study the degree of metallization. The degree of metallization was determined from the ratio of metallic chromium to the total chromium content in the pre-reduced feedstock using Formula (3):
the obtained materials were also studied using a scanning electron microscope (SEM) and a diffractometer (XRD) [21,22].
The total chromium content in the samples was determined by atomic absorption spectroscopy after complete dissolution of the sample. Metallic chromium was determined by selective dissolution of the metallic phase followed by analysis of the obtained solution. The chromium concentration in the solution was measured using an atomic absorption spectrometer. The degree of metallization was calculated as the ratio of metallic chromium content to the total chromium content in the sample.
Based on laboratory studies, pilot tests of pre-reduction of chrome concentrate with various carbonaceous reducing agents were conducted in a chamber furnace. A total of three experiments were conducted for each type of carbonaceous reducing agent, with a firing temperature of 1400 °C and a holding time of 20 h. The range of parameter variations was determined based on the laboratory results. Four crucibles with different charge compositions were used in each experiment.
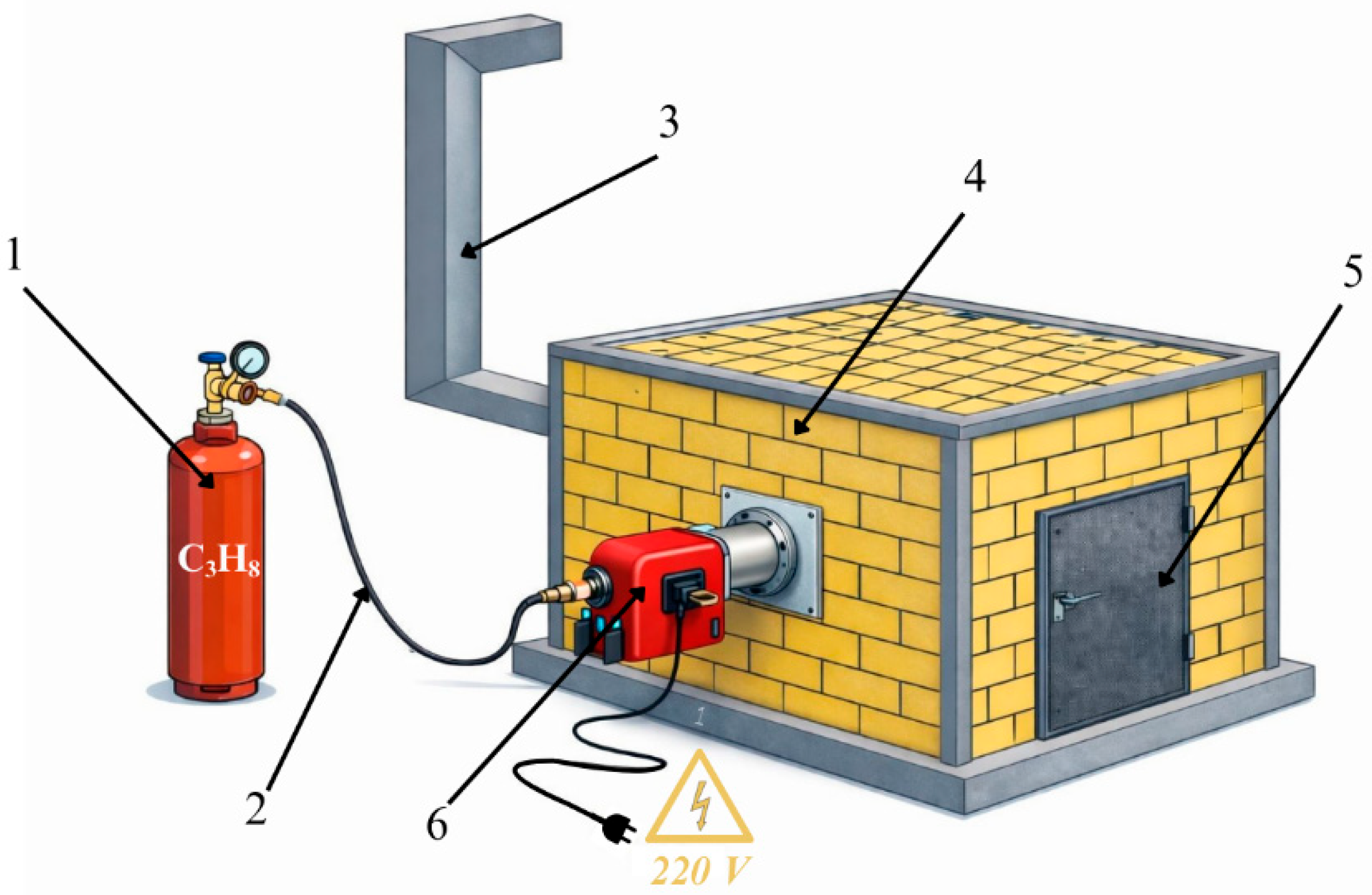
A chamber furnace was used for the experiments. The chamber furnace was rectangular, with a lining made of fireclay bricks coated with refractory sand. Propane (C3H8) was used as fuel because it is more readily available for laboratory testing, is supplied and filled in cylinders, and the flame and gas-air mixing are easier to regulate. The furnace was equipped with two gas burners, each rated at 70 kW, one burner on each side of the chamber furnace. The gas burners had a short combustion head with a diameter of 90 mm. They were equipped with turbochargers for mixing air and propane under pressure. The gas–air mixing was controlled on the gas burner itself. Exhaust gases were exhausted through the rear wall, where a gas cleaning system was installed. A schematic diagram of the chamber furnace is shown in Figure 3.
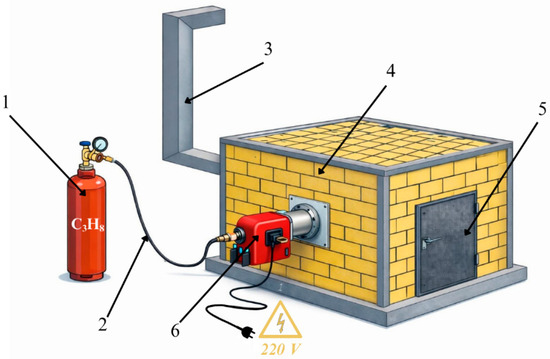
Figure 3.
Chamber furnace diagram: 1—gas cylinder (propane); 2—fuel supply system; 3—gas cleaning system; 4—chamber furnace body; 5—chamber furnace door; 6—gas burner.
The temperature in the gas-fired chamber furnace was controlled manually by adjusting the propane–air ratio supplied to the burners. Two temperature controllers equipped with thermocouples were used for temperature monitoring. The first thermocouple was installed in the furnace chamber and was used to control the overall furnace temperature. The second thermocouple was placed in one of the crucibles to monitor the temperature directly in the reaction zone. During the experiments, the temperature was maintained at approximately 1400 ± 40–50 °C for a holding time of 20 h. Such temperature fluctuations are typical for laboratory-scale gas-fired furnaces with manual burner regulation. The chamotte lining of the furnace ensured relatively uniform heat distribution within the working chamber. The endothermic effects associated with carbothermic reduction reactions were compensated by the burner system and did not significantly affect the overall thermal balance of the furnace.
Four crucibles were installed in the furnace for each experiment, each loaded with an average of 10 kg of ore–coal mixture without compaction. When the crucible was completely filled, the ore–coal mixture column was 500 mm high and 200 mm in diameter. The filled crucible was covered with a silicon carbide lid. The total weight of the ore–coal mixture for one experiment was over 40 kg.
After loading the charge materials into the furnace and closing the furnace chamber, the burners were ignited. First, the furnace chamber interior was dried at low heat at 100–150 °C, operating at this temperature for 2 h. The temperature was raised to 800 °C at a rate of 100 °C/h, and above 800 °C, up to an operating temperature of 1400 °C, at a rate of 200 °C/h.
The loaded crucibles were held at operating temperature for 20 h, after which they were cooled with the furnace closed to 800 °C at a rate of 100 °C/h. Furnace cooling was accomplished by reducing gas flow and then stopping the gas supply to the burners. Further cooling of the furnace working space was accomplished by blowing air through the burners. Once the furnace temperature reached 200–300 °C, the crucibles were removed and cooled in air to room temperature.
When unloading pre-reduced material from crucibles, partial sintering and adhesion to the crucible walls were observed in some cases, complicating unloading and homogenization. In some crucibles, particularly those using −0.2 mm Shubarkol coal as the reducing agent, the reducing agent was observed to migrate to the top of the crucible.
To assess the degree of chromium recovery, after unloading the crucibles, samples were taken from the top, middle, and bottom layers, along the height of each quarter of the crucible, and from the center and periphery of the horizontal cross-section of each level. To determine the weighted average degree of chromium metallization for each crucible, two parallel samples were taken from the averaged material of the same batch. The samples were analyzed for total chromium and metallic chromium content.
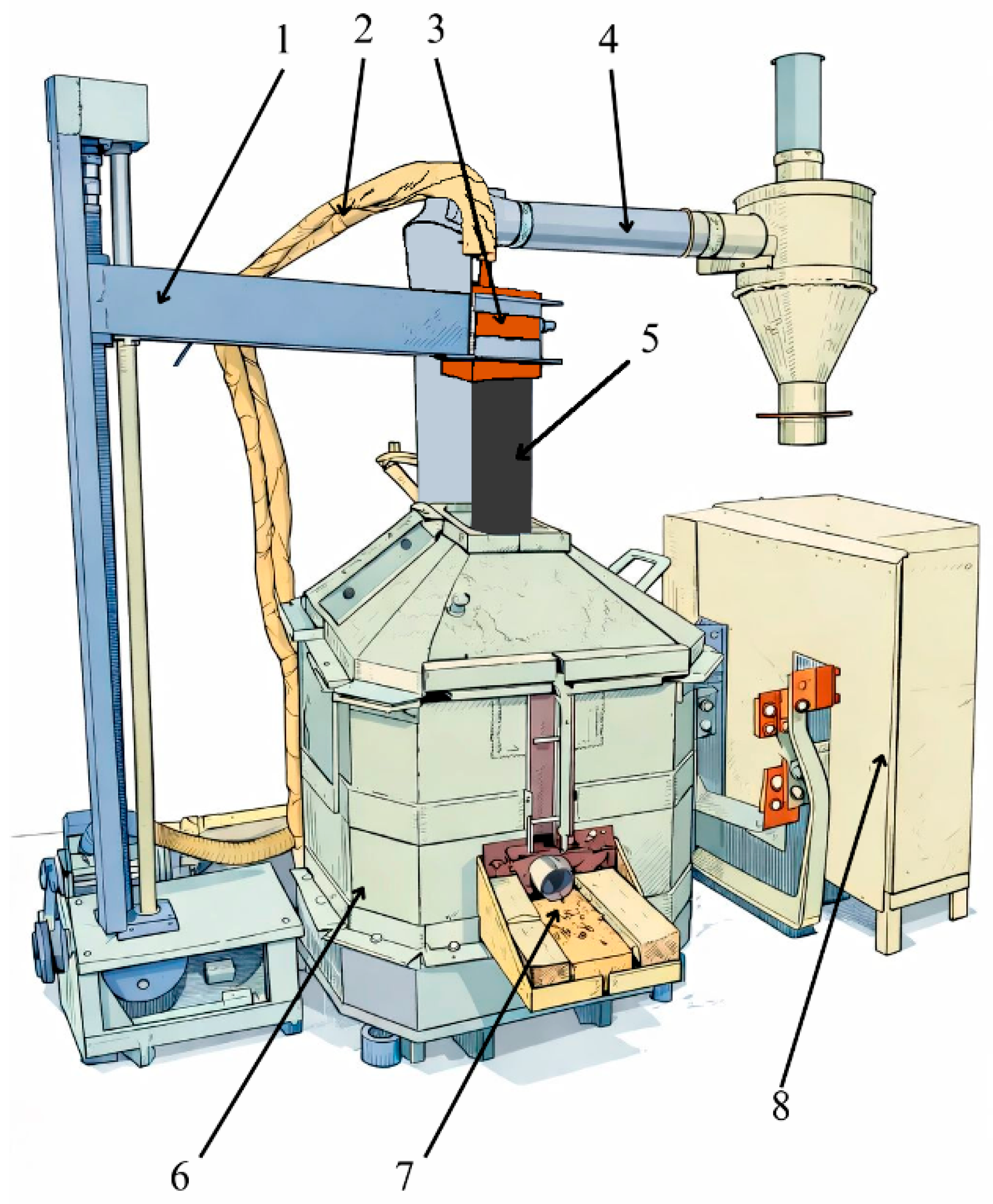
Technological studies of the process of obtaining HC FeCr using a pre-reduced charge were carried out on an ore-thermal single-phase arc furnace with a transformer capacity of 0.1 MVA, equipped with a single graphite electrode. A copper busbar, which is coked with an electrode mass, is connected to the furnace bottom from above and below. The electrode diameter is 100 mm. The raising and lowering of the upper electrode are carried out by a motor connected to the control panel. The furnace has a rectangular shape without a bath slope. The furnace throat is closed and belongs to the closed type of ore-thermal furnaces. The furnace lining is made of magnesite brick. The furnace has a single taphole for tapping metal and slag. Metal and slag are tapped together. The diagram of the ore-thermal furnace is shown in Figure 4 [23,24].
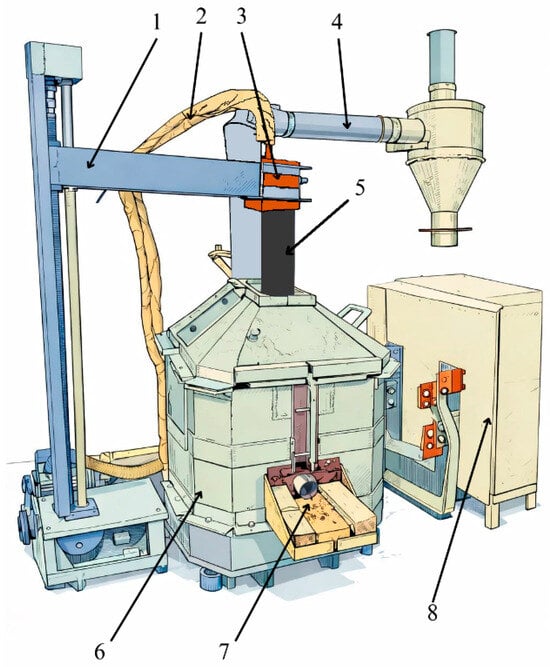
Figure 4.
Schematic diagram of an ore-thermal furnace with a transformer capacity of 0.1 MVA: 1—Electrode holder; 2—cooling system and cable connecting to the copper cheek; 3—copper cheek; 4—gas cleaning system; 5—graphite electrode; 6—furnace body; 7—metal and slag taphole; 8—transformer.
The electric furnace was heated for 4 h on a coke bed, which acts as a conductor of electric current. Upon completion of the heating period, the furnace was completely cleared of any remaining coke bed. The electrical mode during the heating period was maintained at a secondary voltage of 24.6 V and a high-side current of 50–70 A.
3. Results
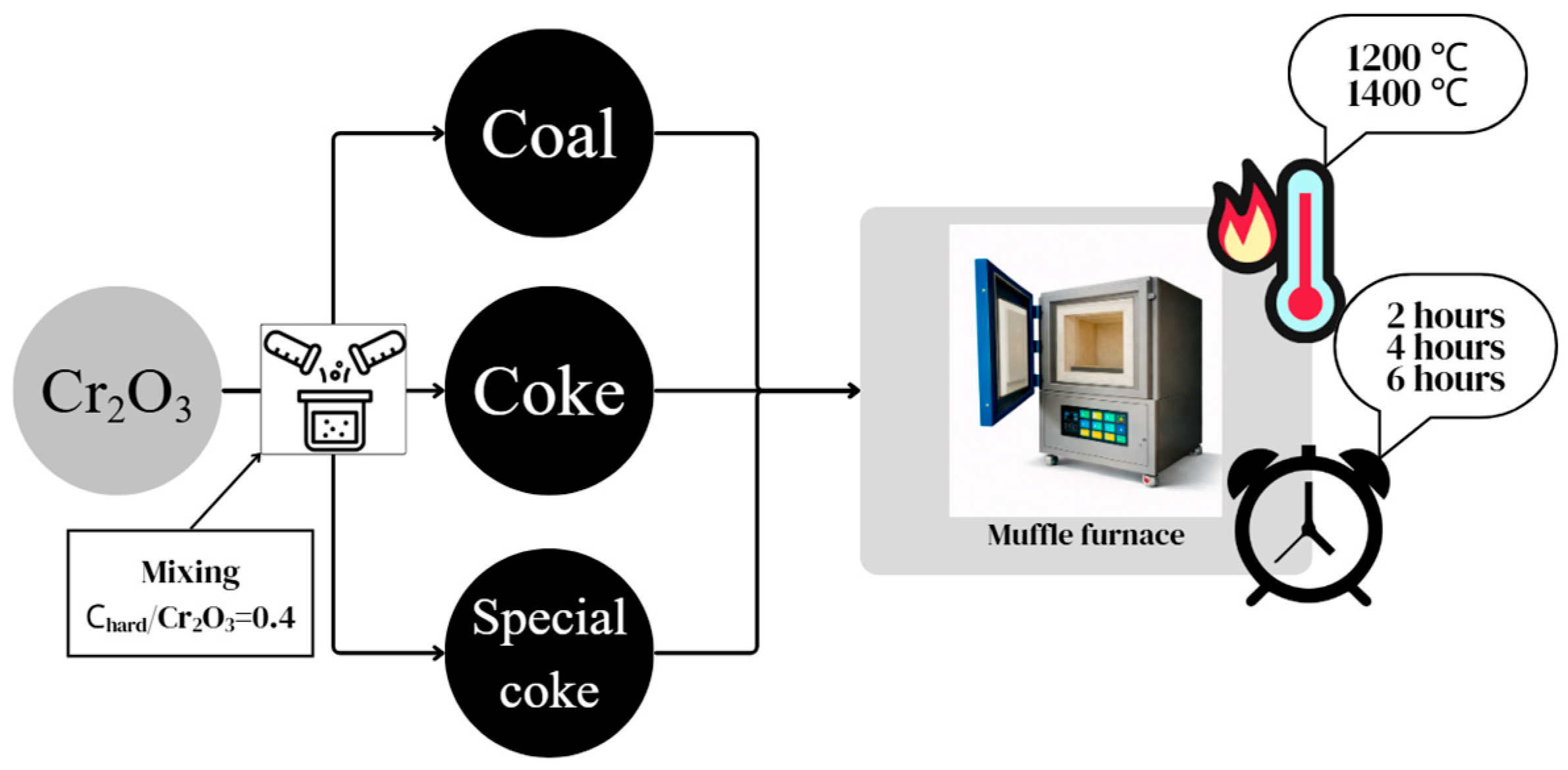
Laboratory studies on the pre-reduction of chrome concentrate with various carbonaceous reducing agents, including coal from the Shubarkol deposit, coke, and special coke, were conducted at the Zh. Abishev Chemical and Metallurgical Institute using a SNOL muffle furnace. Pre-reduction roasting involved mixing the chrome concentrate separately with the carbonaceous reducing agents and roasting at temperatures of 1200 and 1400 °C with holding times of 2, 4, and 6 h. The laboratory study flow chart for pre-reduction of chrome concentrate is shown in Figure 5.
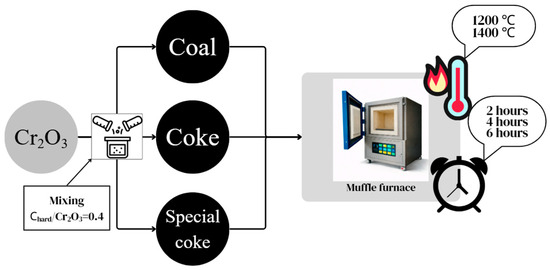
Figure 5.
Scheme of laboratory studies on pre-reduction of chrome concentrate with various carbonaceous reducing agents.
Experimental studies on the pre-reduction of chromium concentrate with various carbonaceous reducing agents have shown that the temperature and duration of roasting have a significant effect on the process of pre-reduction of chromium.
The degree of metallization was calculated based on the results of chemical analysis of the pre-reduction roasting of chrome concentrate using carbonaceous reducing agents. The results of determining the degree of metallization of chrome concentrate are presented in Table 5.

Table 5.
Degree of metallization of pre-reduced chrome raw materials at different temperatures and holding times.
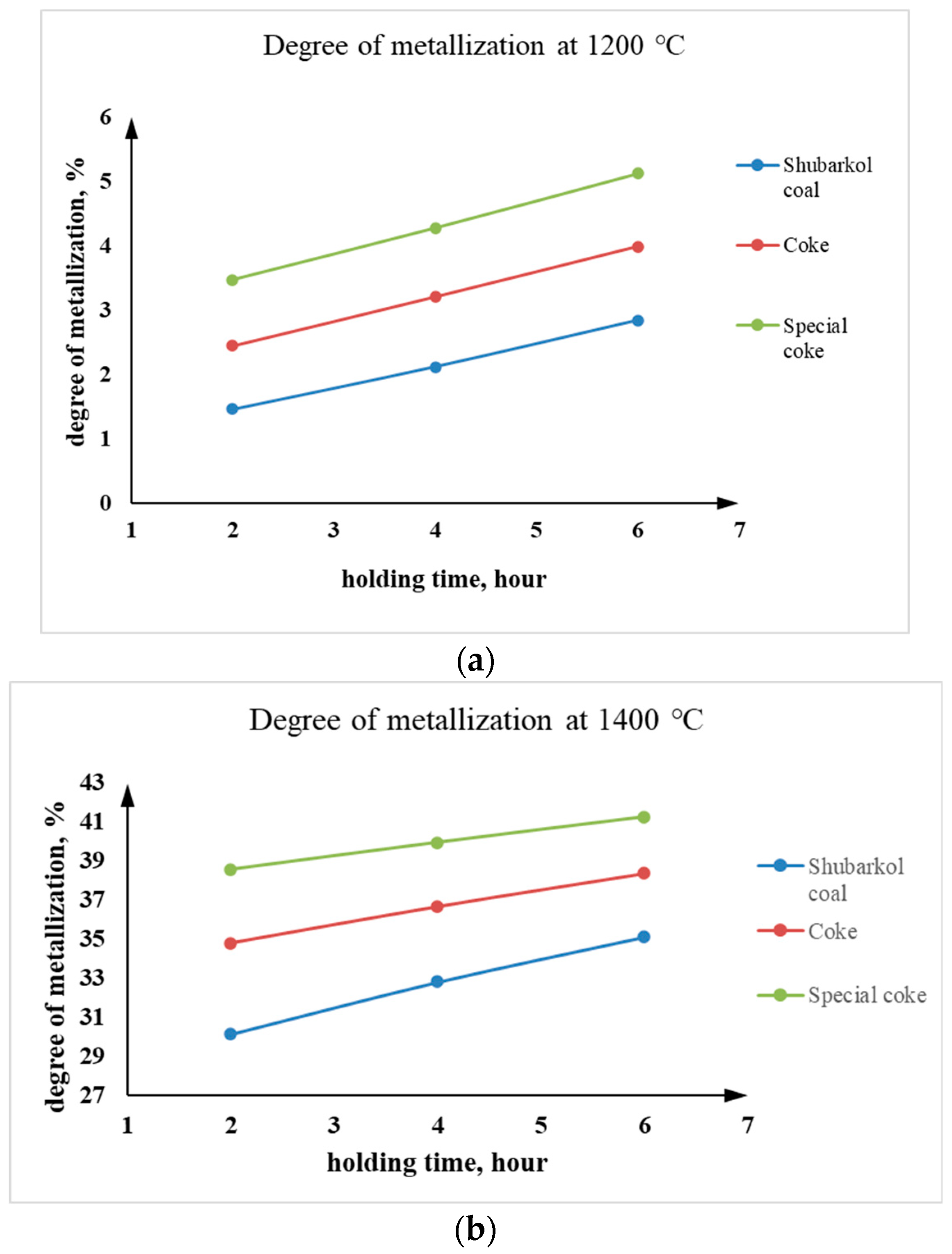
As can be seen from Table 5, at 1200 °C, slight reduction of Cr2O3 was observed. Increasing the holding time from 2 to 6 h contributed to a gradual increase in the degree of metallization of the chromite concentrate. For example, at 1200 °C the metallization degree increased from 1.46–3.47% to 2.84–5.13%, while at 1400 °C it increased from 30.15–38.58% to 35.13–41.28%, depending on the type of carbonaceous reducing agent.
At 1400 °C, the process was more intense, as confirmed by calculations of the degree of metallization. The use of special coke demonstrated the greatest efficiency, ensuring the formation of metallic chromium and chromium-containing phases to a greater extent than other reducing agents. Coke and Shubarkol coal demonstrated lower activity, which is explained by their different chemical and structural compositions. At a temperature of 1400 °C, with a holding time of 6 h, the highest metallization rate of 41.28% was achieved using special coke.
Based on the chemical analysis results, it was decided to conduct scanning electron microscopy (SEM) to study the morphology and element distribution within the pre-reduced chrome concentrate. The particle morphology of the pre-reduced chrome concentrate was studied at 2000× magnification in backscattered electron detection mode. For comparison, samples were collected from the pre-reduced material using special coke as the reducing agent at calcination temperatures of 1200 and 1400 °C and a holding time of 6 h. Images of the chrome concentrate before pre-reduction calcination are also provided for comparison (Figure 6).

Figure 6.
SEM images of pre-reduced chrome raw materials using special coke as a reducing agent: (a) original version of chrome concentrate; (b) at 1200 °C with a holding time of 6 h; (c) at 1400 °C with a holding time of 6 h.
Based on the obtained SEM images (Figure 6), an EDS analysis of the light and dark phases was performed. The results are presented in Table 6.

Table 6.
EDS analysis of pre-reduced chrome concentrates at a temperature of 1200 and 1400 °C with a holding time of 6 h using special coke as a reducing agent.
As shown in Figure 6a, the initial chromite concentrate is characterized by a dense granular structure composed of angular particles. The particle surfaces are relatively smooth, indicating low reactivity of the material in its original state. When the temperature is increased to 1200 °C, bright phases with a sponge-like morphology appear on the particle surfaces, which are typical of metallic formations. This observation is confirmed by EDS analysis: 55.55 wt.% metallic Cr and 29.72 wt.% Fe were detected in the bright regions (Spectrum 1), whereas the darker regions (Spectrum 2) contained 11.26 wt.% Cr and 2.35 wt.% Fe. The contents of Mg, Al, and Si in the bright metallic phase (Spectrum 1) did not exceed 0.3 wt.%, while in the darker oxide phases (Spectrum 2) they reached 16.02, 7.64, and 5.47 wt.%, respectively.
With increasing temperature from 1200 to 1400 °C, the reduction of Cr from oxide compounds becomes more pronounced. SEM micrographs (Figure 6b,c) reveal a significant growth of the bright metallic phase, which nearly envelops the chromite particle. Spectral analysis indicates that the metallic Cr content increases from 55 to 88 wt.%, while the Fe content decreases from 29.72 to 8.51 wt.%. This trend may be attributed to chromium enrichment and recalculation by the EDS software—AzTec One v.6.0 SP1. In the dark oxide phase at 1400 °C (Spectrum 2), the Cr content decreases to 1.7 wt.% and Fe to 0.44 wt.%, indicating substantial chromium extraction from the oxide matrix.
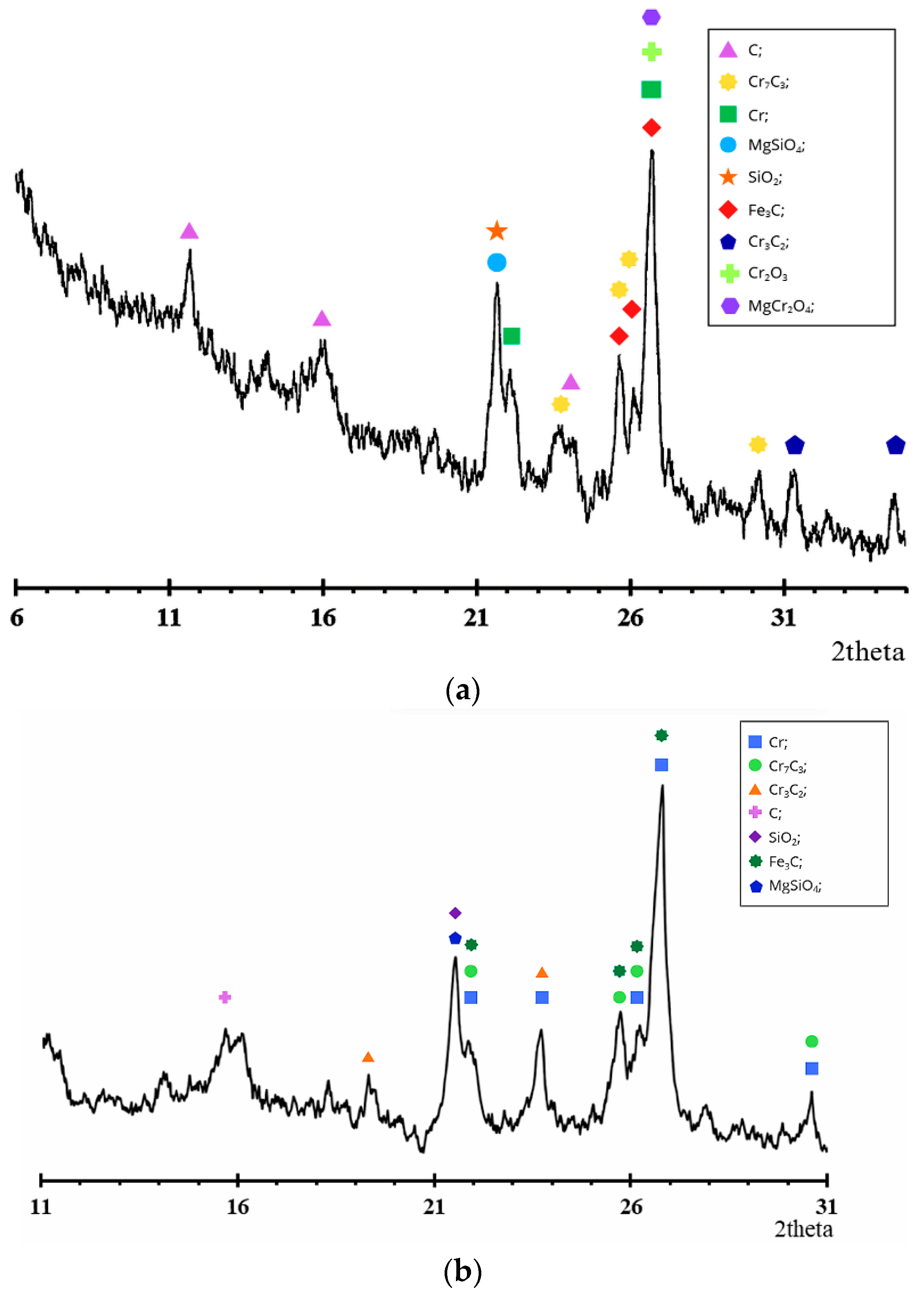
The XRD results (Figure 7) confirm the data of chemical and SEM-EDS studies and reflect the consistent transformation of the chromite structure with increasing pre-reduction roasting temperature.
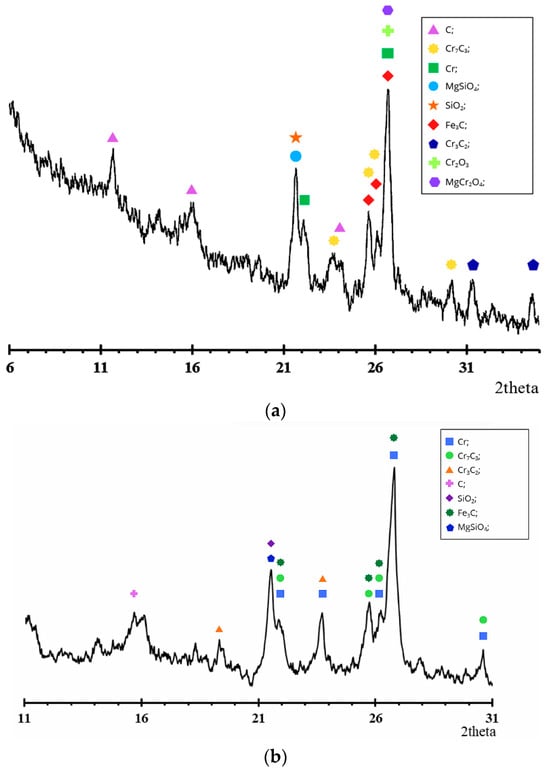
Figure 7.
Derivative diagram of pre-reduced chrome concentrate at a temperature of (a) 1200 °C and (b) 1400 °C with a holding time of 6 h.
The initial chromium concentrate (Figure 1) is characterized by intense reflections of the spinel phase (Mg,Fe)(Cr,Al)2O4, as well as the presence of MgAl2O4 and minor amounts of SiO2. The absence of metallic phases in the diffraction pattern is consistent with the SEM data, which reveal an oxide structure without metallic inclusions.
After firing at 1200 °C for 6 h (Figure 7a), no significant phase changes occur: the main spinel peaks are retained, but their intensity decreases slightly. The diffraction pattern shows weak reflections of metallic chromium and carbide phases, consistent with a low degree of metallization (up to 5.13% for special coke). These results are consistent with SEM-EDS data, where the Cr content in the light phase increases to ~55%, but the oxide matrix remains predominant.
At an increase in temperature to 1400 °C with a holding time of 6 h (Figure 7b), a pronounced change in phase composition is observed. The intensity of spinel reflections decreases significantly, while the peaks of metallic chromium become dominant, indicating extensive reduction of the chromite spinel structure. Chromium carbide phases Cr3C2, Cr7C3, and Fe3C are formed, confirming the active progress of carbothermic reduction followed by carbide formation.
The XRD pattern reveals characteristic reflections of chromium carbides. In particular, reflections assigned to Cr7C3 are observed at 2θ ≈ 21.7°, 25.5–26.2°, and 30.7°, while peaks corresponding to Cr3C2 appear at approximately 2θ ≈ 18.8° and 23.3°. Phase identification was performed by comparison with reference patterns from the ICDD PDF database and structural data from the Materials Project database. According to the Materials Project database, both Cr7C3 (mp-19855) and Cr3C2 (mp-20937) crystallize in an orthorhombic structure with space group Pnma, and their calculated diffraction patterns exhibit reflections in the same angular regions observed in the present study. The agreement between the experimental peaks and the reference diffraction data confirms the formation of chromium carbide phases during high-temperature carbothermic reduction [21,22].
These changes are consistent with the increase in the metallization degree up to 41.28% when special coke is used as the carbonaceous reductant. According to EDS analysis, the chromium content in the bright metallic phase increases up to 88.76%, whereas the oxygen concentration decreases sharply, indicating significant destruction of the spinel structure and the formation of metallic chromium together with chromium carbide phases.
In the dark phases at 1400 °C, magnesium- and aluminum-containing compounds are recorded, corresponding to the residual spinel and silicate components, which is also confirmed by the low content of Cr and Fe in spectrum 2. This indicates the redistribution of Mg and Al into stable oxide phases after the extraction of chromium.
Thus, the results of X-ray diffraction, chemical analysis, and SEM-EDS demonstrate a consistent picture: at 1200 °C, reduction is limited, whereas at 1400 °C, profound destruction of the chromite matrix occurs, forming metallic chromium and carbide phases. The most effective reducing agent is special coke, which ensures the maximum degree of metallization and the highest intensity of metallic phases in the diffraction patterns.
Based on laboratory studies of pre-reduction of chrome concentrate with various carbonaceous reducing agents at temperatures of 1200 and 1400 °C with holding times of 2, 4, and 6 h, it can be concluded that the temperature and holding time are directly proportional to the degree of metallization. That is, the higher the temperature and the longer the holding time of solid-phase roasting, the higher the degree of metallization of the chrome raw material.
As can be seen from Table 5, at a calcination temperature of 1200 °C, there is virtually no noticeable increase in the degree of metallization across all three holding times. This may indicate insufficient temperature and time for solid-phase pre-reduction to allow for significant physicochemical reactions between the ore material and the carbonaceous reducing agent. As the temperature increases from 1200 °C to 1400 °C, the degree of metallization increases sharply. This is expected: at higher temperatures, reduction reactions accelerate, the error in oxygen and carbon diffusion decreases, and the rate of metallic Cr formation increases. At 1400 °C, significant metallization is observed in just 2 h (especially with special coke), and at 6 h, the best degree of metallization in the laboratory experiments is achieved. The dependence of the degree of metallization on holding time at temperatures of 1200 °C and 1400 °C is shown in Figure 8.
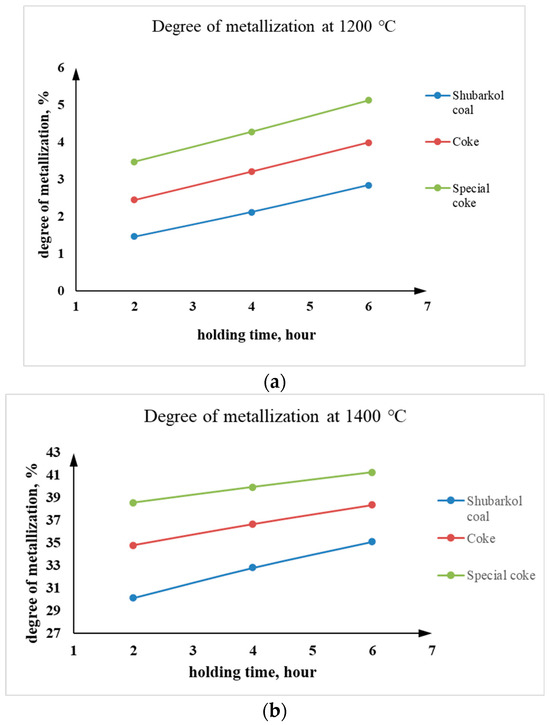
Figure 8.
Dependence of the degree of metallization on the holding time at temperatures (a) 1200 °C and (b) 1400 °C.
Based on the results obtained in laboratory studies, it was decided that the optimal temperature for pre-reduction firing is above 1400 °C and further pilot tests were carried out at this temperature.
Pilot tests of pre-reduction of chrome concentrate using different types of carbonaceous reducing agents. As a result of pilot tests of pre-reduction of chrome concentrates using Shubarkol coal, coke, and special coke as carbonaceous reducing agents, all three types of carbonaceous reducing agents demonstrated relatively identical performance. The tests yielded 40 kg of ore–coal mixture. The appearance of the crucibles before and after pre-reduction roasting of the chrome concentrate is shown in Figure 9.

Figure 9.
Pre-reduction of chrome concentrate (a) before roasting and (b) after roasting.
After pre-reduction of the chrome concentrate, sintering of the charge materials was observed to varying degrees: below average with Shubarkol coal; significant with coke; and moderate with special coke. This affected the extraction of the charge material from the crucibles.
The best results in terms of chromium metallization were achieved using special coke as the reducing agent. However, mixtures containing it are more prone to charge material segregation during heat treatment. At a process temperature of 1400 °C, the effect of reducing agent particle size on the degree of chromium metallization is reduced to the experimental error. To simplify charge material preparation, using special coke with a particle size of −1 or −2 mm is sufficient. Table 7 presents the results of chemical analysis of pre-reduced chromium concentrates with different carbonaceous reducing agents and indicates the degree of metallization for each carbonaceous reducing agent.

Table 7.
Results of chemical analysis of pre-reduced chrome concentrates with different carbonaceous reducing agents.
Pilot tests of high-carbon FeCr smelting (HC FeCr) used pre-reduced chromium concentrate. For the pilot tests of high-carbon FeCr smelting, three experimental charge compositions with pre-reduced material with different carbonaceous reducing agents and different metallization degrees were tested, along with one base charge composition with non-pre-reduced chromium concentrate for comparison. The chemical and technical compositions of the starting materials used in the studies are presented in Table 8 and Table 9.

Table 8.
Chemical compositions of average samples of pre-reduced chrome concentrate with different degrees of metallization, mass %.

Table 9.
Technical composition of coke, mass %.
Furnace preheating began with the base charge. After preheating the furnace for two temperatures, the furnace switched to balance smelting using the base charge. Overall, the tests were divided into four stages, each with the following sequence:
1—Base charge;
2—pre-reduced chromite material with Shubarkol coal (53%);
3—pre-reduced chromite material with coke (64%);
4—pre-reduced chromite material with special coke (65%).
Stage 1. Testing began with the base charge used in the smelting of carbon ferrochrome [21,22]. Since the amount of pre-reduced material was 40 kg for each carbon reducing agent and the volume of the ore-smelting furnace bath, it was decided to calculate the charge material weight per 10 kg of chrome raw material. The charge had the following composition, %: chrome concentrate—77.15; coke—15.55; quartzite—7.3.
Four smelting runs were conducted using this method. The average chemical analysis of the smelting products during this period was as follows: metal, %: 71.16 Cr; 2.26 Si; 6.04 C; 0.018 S; 0.026 P; slag, %: 4.01 Cr2O3; 35.57 SiO2; traces of CaO; 41.91 MgO; 14.39 Al2O3; 1.10 FeO.
Stage 2. After refining the process modes for carbon ferrochrome using the base charge, we switched to pre-reduced chromium material with Shubarkol coal. To optimize the slag composition, quartzite was added, and the charge mixture also included the following components (%): chromium material with Shubarkol coal—90.5; coke—3.26; quartzite—6.24.
Four melts were also conducted using this option. Overall, the furnace operation was characterized by stability, good heating of the hearth, and a fairly complete metal recovery. At the same time, in contrast to the accepted technology, where a refractory crust remained on the furnace throat until the end of the melt, complete melting of the bath occurred. Chromium recovery was 84.27%.
The average chemical analysis of the melt products during this period was: metal, %: 67.58 Cr; 2.49 Si; 7.43 C; 0.014 S; 0.0081–0.042 P; slag, %: 2.68 Cr2O3; 30.85 SiO2; traces of CaO; 37.47 MgO; 17.61 Al2O3; 0.76 FeO
Stage 3. During this stage of the pilot testing, approximately 40 kg of pre-reduced chromite material with coke, obtained during the pilot pre-reduction experiments, were smelted in the ore-thermal furnace at an estimated temperature interval of 1900–2000 °C, which is typical for high-carbon ferrochrome smelting. The material ratios were as follows, in %: chromium material with coke—84.6; coke—1.44; quartzite—13.96.
Four smelts were conducted for this variant. The average chemical analysis of the smelting products during this period was: metal, %: 69.73 Cr; 1.71 Si; 8.33 C; 0.028 S; 0.048 P; slag, %: 3.39 Cr2O3; 28.56 SiO2; traces of CaO; 41.96 MgO; 19.08 Al2O3; 1.08 FeO. Chromium recovery was 83.76%.
Stage 4. During this testing period, pre-reduced chromium material with special coke was used as the charge. The charge mixture consisted of the following materials, in %: chromium material with special coke—83.86; coke—0.63; quartzite—15.51.
Four smelts were conducted using this option. The average chemical analysis of the smelting products during this period was: metal, %: 67.62 Cr; 1.68 Si; 7.71 C; 0.018 S; 0.049 P; slag, %: 2.65 Cr2O3; 30.05 SiO2; traces of CaO; 45.91 MgO; 18.43 Al2O3; 0.73 FeO. Chromium recovery was 89.71%.
Figure 10 shows the appearance of the furnace during HC FeCr smelting.

Figure 10.
General view of the process of smelting high-carbon ferrochrome: (a) metal and slag discharge, (b) furnace throat.
There were no obvious process disturbances, melt tapping was intensive, and metal and slag yields were stable. Chromium recovery was 82.14%.
Based on the test results, the specific energy consumption per unit of high-carbon ferrochrome smelting was estimated. Table 10 presents the specific energy consumption for each test stage.

Table 10.
Specific energy consumption for smelting carbon ferrochrome during testing, calculated per ton of alloy.
In industrial ore-smelting furnaces of various designs, 3200–4800 kW of electric power are consumed to smelt 1 ton of carbon ferrochrome using the traditional method, according to various sources [4,5,6,7]. In the conditions of smelting shop No. 4 of the Aktobe ferroalloy plant, 4800 kW of electric power are consumed to smelt 1 ton of carbon ferrochrome using the traditional method [25,26]. Given the large heat losses and design features of the experimental furnace, it is impossible to achieve this figure. According to the data in Table 11 (Stage 1), in the conditions of an ore-smelting furnace with a transformer capacity of 0.1 MVA, 54.76% more electric power is consumed to produce 1 ton of ferrochrome, or 7121.8 kW. Knowing the differences in specific energy consumption for different furnace units, we can calculate the approximate specific energy consumption for smelting carbon ferrochrome using pre-reduced chromite material in industrial furnaces. The upper limit of specific energy consumption per ton of alloy was used for the calculations. The results are presented in Table 11.

Table 11.
Comparative specific energy consumption for the smelting of carbon ferrochrome.
When comparing the specific energy consumption under the conditions of a 0.1 MVA ore-thermal furnace given in Table 11, it can be noted that the specific energy consumption using pre-reduced chromite material with different types of reducing agents and different degrees of metallization is lower than the specific energy consumption of the base charge by: 18.43, 29.71, and 33.52%, respectively.
Evaluation of the performance of an ore-smelting furnace with a 0.1 MVA transformer for smelting carbon ferrochrome used pre-reduced materials. During testing, it was observed that furnace performance increased with the use of pre-reduced charge. Producing a large volume of charge materials and ensuring long-term stable furnace operation enabled optimal smelting performance. Furthermore, with an increasing metallization percentage of pre-reduced materials, metal yield steadily increased, while the specific consumption of electricity and the chromium-containing material itself decreased.
As a result of the smelting stages, the chromium oxide content in the slag does not exceed standard limits and ranges on average from 1.96 to 5.01%. Overall, 86.13 kg of carbon ferrochrome, compliant with GOST 4757-91 (ISO 5448:1981) [27,28], was obtained during pilot testing across all stages.
Technical and economic indicators for the production of high-carbon ferrochrome using pre-reduced chrome concentrate. The technical and economic assessment was performed based on pilot plant data, with all costs adjusted to the production of 1 ton of commercial high-carbon ferrochrome. The calculations took into account the consumption of chrome concentrate, carbonaceous reducing agents, quartzite, propane (at the pre-reduction stage), and electricity. Cost indicators were adopted based on market data [29,30,31,32,33,34]. The analysis was conducted to determine the impact of pre-reduction on the cost structure, energy intensity, and overall economic efficiency of the process (Table 12).

Table 12.
Technical and economic indicators for the production of high-carbon ferrochrome with pre-reduced chrome concentrate.
Basic technology for high-carbon ferrochrome (HCFeCr) production is characterized by a total production cost of 1110.9 USD/t of alloy. Cost structure analysis indicates that chromite concentrate (612 USD/t, or 55.1%) and electricity (288 USD/t, or 25.9%) represent the dominant cost components. Together, these two items account for more than 81% of the total production cost, demonstrating the high sensitivity of the process to raw material and electricity prices. With a concentrate consumption of 1.8 t/t alloy and a specific electricity demand of 4800 kWh/t, the conventional route exhibits high material and energy intensity, which constrains competitiveness under conditions of rising tariffs and raw material prices.
The introduction of a solid-phase pre-reduction stage fundamentally alters the energy and economic balance of the process by transferring part of the endothermic reduction reactions from the electric arc zone to a fuel-fired unit and by increasing chromium utilization efficiency. The most pronounced economic effect is achieved when Shubarkol coal is used as the reductant. In this case, the total production cost decreases to 855.51 USD/t, representing a reduction of 255.39 USD/t (−23.0%) compared with the baseline. This saving is primarily attributed to a decrease in chromite concentrate consumption from 1.8 to 1.3 t/t alloy (−27.8%) and a reduction in specific electricity consumption to 3913.84 kWh/t (−18.4%). Although additional propane is required during the pre-reduction stage (144 USD/t), its impact is offset by the low cost of coal and the reduced electrical energy demand.
When metallurgical coke is applied at the pre-reduction stage, the total cost amounts to 997.7 USD/t, corresponding to a 10.2% reduction relative to the conventional route. While specific electricity consumption decreases to 3373.6 kWh/t (−29.7%), the high price of coke (450 USD/t) significantly limits the economic benefit. Consequently, this option demonstrates limited economic attractiveness compared with alternative reductants.
The most balanced techno-economic performance is achieved with special coke. The total production cost reaches 914.75 USD/t, which is 17.7% lower than the baseline. Specific electricity consumption decreases to 3191.2 kWh/t, corresponding to a 33.5% reduction in electrical load compared with the conventional process. The direct saving in electricity cost amounts to 96.5 USD/t. Simultaneously, a high degree of metallization and chromium recovery is achieved, allowing a reduction in chromite concentrate consumption to 1.5 t/t alloy. Special coke thus provides an optimal balance between energy efficiency and reductant cost.
Sensitivity analysis with respect to electricity tariffs demonstrates the robustness of the pre-reduction schemes under increasing energy prices. At an electricity tariff of 0.08 USD/kWh, the electricity-related savings for the special coke option exceed 120 USD/t, further strengthening the economic advantage of the technology. Therefore, the economic efficiency of the pre-reduction approach increases proportionally with electricity price escalation, which is particularly relevant in the context of global tariff growth.
An additional advantage is the reduction in chromite concentrate consumption, which remains the most capital-intensive charge component. A decrease of 0.3–0.5 t of concentrate per ton of alloy exerts a decisive influence on total production cost, given its dominant share in the cost structure.
From an environmental perspective, a reduction in electricity consumption of approximately 1.6 MWh/t (special coke option), assuming an average emission factor of 0.8 t CO2/MWh, results in a decrease of indirect emissions by approximately 1.28 t CO2 per ton of alloy. This corresponds to a carbon footprint reduction of more than 25% compared with the conventional route. Pre-reduction therefore functions not only as a cost-optimization tool but also as a decarbonization measure.
Industrial-scale extrapolation highlights the significant economic potential of the proposed approach. At an annual production capacity of 100,000 t of ferrochrome, the expected economic benefit may reach approximately 25.5 million USD when Shubarkol coal is used and 19.6 million USD when special coke is applied. Even considering capital expenditures associated with implementing the pre-reduction stage, the estimated payback period is within 2–3 years, which satisfies typical investment criteria in the metallurgical industry.
Overall, the engineering and economic assessment confirms that solid-phase pre-reduction of chromite concentrate is an effective strategy for reducing energy intensity and production cost in HC FeCr manufacturing. The technology enables up to a 33.5% reduction in specific electricity consumption, up to a 27.8% decrease in concentrate consumption, chromium recovery of up to 89.71%, and a substantial reduction in carbon footprint. Special coke provides the highest energy efficiency, whereas Shubarkol coal ensures the greatest direct economic benefit. Pre-reduction should therefore be considered a strategic energy-optimization stage capable of enhancing the long-term competitiveness of ferrochrome production under conditions of rising energy prices and increasingly stringent environmental requirements.
4. Discussion
The obtained results demonstrate a consistent correlation between pre-reduction temperature, the nature of the carbonaceous reductant, and the degree of metallization of the chromite concentrate, which subsequently determines the techno-energy performance of high-carbon ferrochrome (HC FeCr) smelting. Comprehensive analysis of the XRD and SEM-EDS data (Figure 6 and Figure 7) confirms the progressive transformation of the spinel structure (Mg,Fe)(Cr,Al)2O4 with temperature increasing from 1200 to 1400 °C. At 1200 °C, the reduction process remains limited: the spinel phase predominates, and metallic inclusions are localized and weakly developed, corresponding to low metallization degrees (not exceeding 5.13%) (Table 5). Increasing the temperature to 1400 °C results in intensive destruction of the spinel matrix, formation of metallic chromium and carbide phases (Cr3C2, Cr7C3), and a sharp decrease in oxygen content in the bright phases according to EDS analysis (Figure 6, Table 6). The metallization degree increases to 41.28% under laboratory conditions (Table 5) and up to 65% at pilot scale (Table 7). Thus, the limiting factor in pre-reduction is not only reaction stoichiometry but also diffusion constraints within the chromite spinel structure, which are significantly mitigated at 1400 °C due to enhanced mass transfer and accelerated carbothermic reactions.
From a thermodynamic viewpoint, the reaction should be regarded as a simplified overall representation of the carbothermic reduction process. According to Ellingham-type considerations, the reduction ability of carbon increases with temperature because the formation of CO becomes thermodynamically more favorable at elevated temperatures. In chromite-bearing systems, chromium oxide is significantly more stable than iron oxides, and therefore reduction at 1200 °C remains limited, whereas at 1400 °C the thermodynamic conditions become substantially more favorable for the destruction of the spinel matrix and the formation of metallic and carbide phases. This interpretation is in good agreement with the present experimental results, including the low metallization degree at 1200 °C and the formation of Cr3C2, Cr7C3, and Fe3C phases at 1400 °C [35,36,37,38].
Comparison of carbonaceous reductants indicates that special coke provides the highest efficiency, which is associated with its high fixed carbon content, low ash level, and favorable porous structure that enhances CO gas-phase transport and improves carbon–oxide contact. Metallurgical coke exhibits comparable but slightly lower performance, likely due to its higher mineral content. Shubarkol coal demonstrates a lower metallization degree in laboratory experiments; however, from an economic perspective, it represents the most advantageous option owing to its low cost.
The use of pre-reduced material substantially alters the smelting behavior in the submerged arc furnace. Compared with the base charge, improved bath heating, more stable tapping, reduced Cr2O3 content in slag, and increased chromium recovery up to 89.71% are observed. A particularly significant outcome is the reduction in specific electricity consumption. In a 0.1 MVA furnace, electricity demand decreased by 18.43% when Shubarkol coal was used, by 29.71% with metallurgical coke, and by 33.52% with special coke relative to the baseline technology. Even considering the elevated relative heat losses of the small experimental furnace, extrapolation to industrial scale indicates a reduction in specific electricity consumption to approximately 3191 kWh/t when special coke is applied, corresponding to the lower bound of the industrial range (3200–4800 kWh/t). These results confirm the central hypothesis of this study: transferring part of the endothermic reduction reactions from the electric arc zone to a fuel-based pre-reduction stage reduces electrical load and enhances overall process energy efficiency.
Techno-economic assessment shows that the conventional process yields a production cost of approximately 1110.9 USD/t, whereas the use of pre-reduced feedstock reduces the cost to 855.51 USD/t with Shubarkol coal, 997.7 USD/t with metallurgical coke, and 914.75 USD/t with special coke. Therefore, the most economically favorable option is pre-reduction using Shubarkol coal, while the best energy performance is achieved with special coke. The choice of reductant may depend on the strategic priorities of the enterprise, whether minimizing energy consumption, reducing production cost, or achieving an optimal balance between these factors. An additional opportunity for further cost reduction lies in replacing propane with natural gas during industrial implementation.
Despite the positive outcomes, the study has several limitations. Experiments were conducted in a low-power furnace, which results in overestimated specific energy indicators due to relative heat losses. Industrial extrapolation is based on calculated projections. Moreover, long-term effects of pre-reduced feedstock on furnace lining and electrodes were not evaluated, and a comprehensive carbon balance and CO2 emission assessment were not performed. These aspects require further investigation.
Overall, the results confirm that solid-phase pre-reduction of chromite concentrate at 1400 °C ensures deep phase transformation of spinel with the formation of metallic and carbide phases, leading to reduced specific electricity consumption and improved techno-economic performance of HC FeCr smelting. The technology demonstrates both energetic and economic feasibility and may be considered a promising pathway for modernization of conventional ferrochrome production routes under conditions of rising electricity costs and increasing requirements for energy efficiency in metallurgical processes.
It should be noted that the present study was conducted in a pilot-scale ore-thermal furnace (0.1 MVA). When extrapolating the obtained results to industrial-scale ferrochrome production, several scale-related factors should be considered. These include differences in furnace geometry and dimensions, heat transfer conditions, charge bed permeability affecting the transport of reducing gases, and scale-dependent heat losses associated with larger furnace structures. Despite these differences, pilot-scale experiments provide valuable insight into the thermodynamic and kinetic features of chromite pre-reduction and ferrochrome smelting.
5. Conclusions
- –
- Pre-reduction of chromite concentrate at 1200 °C was found to be limited, with the degree of metallization not exceeding 5.13%, even when the holding time was increased from 2 to 6 h, indicating insufficient reduction under these conditions.
- –
- At 1400 °C, the reduction process intensified significantly: the degree of metallization reached 41.28% under laboratory conditions and up to 65% in pilot-scale trials. These results are supported by chemical analysis, SEM-EDS, and XRF data, which confirm the decomposition of the spinel matrix and the formation of metallic and carbide phases.
- –
- Among the investigated reducing agents, special coke demonstrated the highest efficiency in terms of metallization, whereas Shubarkol coal provided the greatest economic benefit due to its lower cost.
- –
- The application of pre-reduced feedstock in high-carbon ferrochrome (HC FeCr) smelting increased chromium recovery (up to 89.71%) and reduced Cr2O3 losses to slag compared with the conventional charge. Specific electricity consumption decreased relative to the baseline scenario by 18.43% (coal), 29.71% (coke), and 33.52% (special coke). Industrial-scale extrapolation indicates a projected energy consumption of approximately 3191 kWh/t when special coke is used.
- –
- Techno-economic assessment demonstrates a reduction in production costs compared with the traditional process route; further optimization potential lies in replacing propane with natural gas during industrial implementation.
Author Contributions
Conceptualization, I.A. and M.Y.; methodology, S.Z.; software, S.Z.; validation, I.A., M.Y. and S.Z.; formal analysis, I.A. and M.Y.; investigation, S.Z. and M.Y.; resources, I.A., S.S. and M.Y.; data curation, S.Z. and M.Y.; manuscript preparation, S.Z.; review and editing, I.A. and M.Y.; visualization, S.S.; scientific supervision, I.A. and M.Y.; project administration, I.A. and M.Y.; funding acquisition, I.A. and S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Scientific Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan, grant number BR24993020.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of this study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Abbreviations
The following abbreviations are used in this manuscript:
| CIS | Commonwealth of Independent States |
| ICDA | International Chromium Development Association |
| FeCr | Ferrochrome |
| HC | High carbon |
| SEM | Scanning electron microscope |
| EDS | Energy dispersive spectroscopy |
| AktFP | Aktobe Ferroalloy Plant |
References
- Erwee, M.; Beukes, J.P.; Dawson, N.F. Fluxing of South African chromite ore with colemanite. In Proceedings of the Infacon XV: International Ferro-Alloys Congress, Cape Town, South Africa, 25–28 February 2018. [Google Scholar]
- Bilyalov, K.S.; Kaliakparov, A.G.; Panfilov, V.P.; Suindikov, D.B.; Baimagambetov, K.N. Technology Development for Chromium Ore Agglomeration Using Bentonite. Metallurgist 2023, 67, 462–468. [Google Scholar] [CrossRef]
- Moyo, L.B.; Simate, G.S.; Hobane, N.; Dube, C. Characterization, kinetics and thermodynamic evaluation of struvite produced using ferrochrome slag as a magnesium source. S. Afr. J. Chem. Eng. 2024, 47, 83–90. [Google Scholar] [CrossRef]
- McCullough, S.; Hockaday, S.; Johnson, C.; Barcza, N.A. Pre-reduction and smelting characteristics of Kazakhstan ore samples. In Proceedings of the INFACON XII, Helsinki, Finland, 6–9 June 2010; pp. 249–262. (In English) [Google Scholar]
- International Stainless Steel Forum (ISSF). Stainless Steel in Figures 2020; International Stainless Steel Forum: Brussels, Belgium, 2020. [Google Scholar]
- International Chromium Development Association (ICDA). Market Insights (Chromite and Ferrochrome Statistics). Available online: https://www.icdacr.com/market-insights/ (accessed on 20 February 2026).
- Dehghanpour, H.; Doğan, F.; Subaşı, S.; Maraşlı, M. Effects of single-walled carbon nanotubes and steel fiber on recycled ferro-hrome filled electrical conductive mortars. J. Sustain. Constr. Mater. Technol. 2022, 7, 250–265. [Google Scholar] [CrossRef]
- Basson, J.; Daavittila, J. High Carbon Ferrochrome Technology. In Handbook of Ferroalloys: Theory and Technology; Butterworth-Heinemann Elsevier: Oxford, UK, 2013; pp. 317–363. ISBN 9780080977539. [Google Scholar]
- Beukes, J.P.; Dawson, N.F.; van Zyl, P.G. Review of Cr (VI) environmental practices in the chromite mining and ferrochrome production industry. J. Clean. Prod. 2017, 165, 874–889. [Google Scholar] [CrossRef]
- Gasik, M.I. Technology of Chromium and Its Ferroalloys. In Handbook of Ferroalloys: Theory and Technology; Butterworth-Heinemann Elsevier: Oxford, UK, 2013; pp. 299–316. ISBN 9780080977539. [Google Scholar]
- du Preez, S.P.; van Kaam, T.P.M.; Ringdalen, E.; Tangstad, M.; Morita, K.; Bessarabov, D.G.; van Zyl, P.G.; Beukes, J.P. An Overview of Currently Applied Ferrochrome Production Processes and Their Waste Management Practices. Minerals 2023, 13, 809. [Google Scholar] [CrossRef]
- Kapure, G.; Tathavadkar, V.; Rao, C.B.; Rao, S.M.; Raju, K.S. Coal based direct reduction of preoxidized chromite ore at high temperature. In Proceedings of the INFACON XII, Helsinki, Finland, 6–9 June 2010; pp. 293–302. [Google Scholar]
- Motovilov, I.Y.; Luganov, V.A.; Chepushtanova, T.A.; Guseynova, G.D.; Itkulova, S.S. Processing of metallurgical wastes with obtaining iron oxides nanopowders. In WASTES—Solutions, Treatments and Opportunities II—Selected Papers from the 4th Edition of the International Conference Wastes: Solutions, Treatments and Opportunities, Porto, Portugal, 25–26 September 2017; CRC Press: Boca Raton, FL, USA, 2018; pp. 191–196. (In English) [Google Scholar]
- Das, A.K.; Khaoash, S.; Das, S.P.; Mohapatra, B.K.; Dash, N.; Singh, S.K.; Mishra, P.; Mohanty, J. Processing of Low-Grade Chromite Ore for Ferroalloy Production: A Case Study from Ghutrigaon, Odisha, India. Trans. Indian Inst. Met. 2020, 73, 2309–2320. [Google Scholar] [CrossRef]
- Panda, C.R.; Mishra, K.K.; Nayak, B.D.; Rao, D.S.; Nayak, B.B. Release behaviour of chromium from ferrochrome slag. Int. J. Environ. Technol. Manag. 2012, 15, 261–274. [Google Scholar] [CrossRef]
- Kleynhans, E.L.J.; Beukes, J.P.; Dawson, N.F. Techno-economic feasibility of a pre-oxidation process to enhance pre-reduction of chromite. J. S. Afr. Inst. Min. Metall. 2017, 117, 441–450. [Google Scholar]
- Shotanov, A.E.; Roshchin, A.V.; Panfilov, V.P.; Nurgali, N.Z. Prereduction of Chromite Raw Materials by the Höganäs Method. Metallurgist 2022, 66, 871–880. [Google Scholar] [CrossRef]
- Shotanov, A.E.; Nurgali, N.Z.; Roshchin, A.V.; Panfilov, V.P.; Baysanov, S.O.; Almagambetov, M.S. Smelting of High-Carbon Ferrochrome from Prereduced Chromite Raw Materials of the Donskoy Ore Mining and Processing Plant. Metallurgist 2023, 66, 1619–1624. [Google Scholar] [CrossRef]
- Kumar, P.; Sahu, N.; Roshan, A.; Rout, B.N.; Tripathy, S.K. Influence of process parameters on impurity level in ferrochrome production-An industrial-scale analysis. Miner. Process. Extr. Metall. Rev. 2022, 43, 622–632. [Google Scholar] [CrossRef]
- Akhmetov, A.; Zulhan, Z.; Sadyk, Z.; Burumbayev, A.; Zhakan, A.; Kabylkanov, S.; Toleukadyr, R.; Saulebek, Z.; Ayaganova, Z.; Makhambetov, Y. Carbon-Free Smelting of Ferrochrome Using FeAlSiCa Alloy. Processes 2025, 13, 1745. [Google Scholar] [CrossRef]
- Materials Project Database. Cr7C3(Chromium Carbide) Crystal Structure and Properties. Available online: https://next-gen.materialsproject.org/materials/mp-19855 (accessed on 20 February 2026).
- Materials Project Database. Cr3C2 (Chromium Carbide) Crystal Structure and Properties. Available online: https://next-gen.materialsproject.org/materials/mp-20937 (accessed on 20 February 2026).
- Makhambetov, Y.; Kutzhanov, M.; Toleukadyr, R.; Myrzagaliyev, A.; Sadyk, Z.; Saulebek, Z.; Akhmetov, A. Utilization of Chromite Spinel Powder in the Metallothermic Smelting of Low-Carbon Ferrochrome. Processes 2025, 13, 2288. [Google Scholar] [CrossRef]
- Shabanov, E.Z.; Saulebek, Z.K.; Akhmetov, A.S.; Mukhtarkhanova, G.K. Smelting of high-carbon ferrochromium from pre-reduced chromite raw materials. CIS Iron Steel Rev. 2024, 27, 15–19. [Google Scholar] [CrossRef]
- Shabanov, Y.; Makhambetov, Y.; Saulebek, Z.; Toleukadyr, R.; Baisanov, S.; Nurgali, N.; Shotanov, A.; Dossekenov, M.; Zhumagaliyev, Y. Pilot Tests of Pre-Reduction in Chromium Raw Materials and Melting of High-Carbon Ferrochromium. Metals 2024, 14, 202. [Google Scholar] [CrossRef]
- U.S. Geological Survey (USGS). Mineral Commodity Summaries 2025: Chromium; U.S. Department of the Interior: Reston, VA, USA, 2025. Available online: https://pubs.usgs.gov/periodicals/mcs2025/mcs2025-chromium.pdf (accessed on 20 February 2026).
- GOST 4757-91; Ferrochrome—Technical Requirements and Delivery Conditions. Interstate Council for Standardization, Metrology and Certification: Moscow, Russia, 1991.
- ISO 5448:1981; Ferrochrome—Specification and Delivery Conditions. International Organization for Standardization: Geneva, Switzerland, 1981.
- U.S. Energy Information Administration (EIA). Quarterly Coal Report; U.S. Department of Energy: Washington, DC, USA. Available online: https://www.eia.gov/coal/production/quarterly/ (accessed on 20 February 2026).
- Flagma Kazakhstan. Semi-Coke (Polukoks): Commercial Offer. Available online: https://flagma.kz/polukoks-o2216698.html (accessed on 20 February 2026).
- World Bank. Commodity Markets (Pink Sheet Data). Available online: https://www.worldbank.org/en/research/commodity-markets (accessed on 20 February 2026).
- Ministry of Energy of the Republic of Kazakhstan. Order on Maximum Wholesale Prices for Liquefied Petroleum Gas (LPG). Available online: https://adilet.zan.kz/rus/docs/G25JVM00489 (accessed on 20 February 2026).
- K-Zharyk LLP. Industrial Electricity Tariffs in the Republic of Kazakhstan. Available online: https://k-zharyk.kz/ru/node/329 (accessed on 20 February 2026).
- Campbell, K.; van Laar, J.H.; Booysen, W.; Kleingeld, M. Comparison of prescribed emission quantification methods and potential carbon tax liability in the South African FeCr industry. Carbon Manag. 2020, 11, 213–229. [Google Scholar] [CrossRef]
- Ellingham, H.J.T. Reducibility of Oxides and Sulphides in Metallurgical Processes. J. Soc. Chem. Ind. 1944, 63, 125–133. [Google Scholar]
- Atkins, P.; de Paula, J. Thermodynamics and the Temperature Dependence of Gibbs Free Energy. In Physical Chemistry, 10th ed.; Oxford University Press: Oxford, UK, 2014; pp. 171–196. ISBN 9780199697403. [Google Scholar]
- Rosenqvist, T. Thermodynamics of Carbothermic Reduction of Metal Oxides. In Principles of Extractive Metallurgy; McGraw-Hill: New York, NY, USA, 2004; pp. 145–168. ISBN 9780070532205. [Google Scholar]
- He, J.; Zhang, G.; Jiang, T.; Xue, X. Carbothermal Reduction Behavior of Chromite in the Fe–Cr–O System. Int. J. Miner. Metall. Mater. 2013, 20, 987–994. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.