Abstract
The brittleness of 18-karat purple gold originates from the AuAl2 intermetallic compound. This study investigates the microstructural modification of the AuAl2 intermetallic compound by adding silicon (Si) and cobalt (Co) and by rapid solidification in copper molds. The samples with alloy additions from a traditional investment casting were compared with copper mold casting for grain boundary characteristics using SEM, EBSD, and TEM. SEM micrographs showed a reduction in grain size of copper mold casting from approximately within 150–200 μm to within 12–20 μm. EBSD showed a narrow grain size distribution in the Si–Co-modified alloy than in the Si-modified alloy, using the copper mold casting technique. TEM observations show that grain boundaries were closely packed, with ~80 nm-sized voids. XRD confirmed that all alloys retained the AuAl2 intermetallic phase, with peak broadening in the modified and fast-cooling samples indicating crystallographic refinement. These results confirm that Si-Co additions with a fast cooling rate effectively refine the microstructure of the AuAl2 intermetallic compound, making the alloy less brittle while preserving the purple gold color.
1. Introduction
Purple gold is an intermetallic compound that is composed of gold and aluminum with a specific stoichiometric structure of AuAl2. Unlike ordinary metallic alloys, intermetallics exhibit directional bonding with substantial covalent and ionic components, which restricts plastic deformation and leads to brittleness at room temperature [1]. It was discovered in the electronics industry, known as the “purple plague,” where AuAl2 formed at the junction between a Au wire and an Al pad during wire bonding [2]. Later, the jewelry industry adapted this compound to produce 18K purple gold for its distinctive hue and chemical stability [3,4].
The first commercial process was patented by Loh, P.C [5]. This involved a sintering technique that improved the ductility of the stoichiometric AuAl2 intermetallic compound. The compositions were Au with Al in the range of 16.5–21.5 wt%, with addition alloys containing 0.5–4.0 wt% Pd, with Ni substantially absent. Other sintering processes have previously been reported for Au-Al purple gold compositions [6,7]. In contrast, hot-working techniques have been applied to more Al-rich Au–Al alloys, such as Au–66 wt% Al and Au–3 wt% Cu–66 wt% Al [3,8]. These compositions deviate substantially from the AuAl2 stoichiometric and, therefore, do not form a single-phase AuAl2 structure. Therefore, Al-rich Au–Al alloys processed by hot working do not form an intense purple color of the stoichiometric AuAl2 intermetallic compound. Casting of Au-15 wt% Al and Au-25 wt% Al has been reported; however, the resulting alloys were very brittle and were therefore cut into facet gemstones [9]. Later on, Au-25 wt% Al alloys with Al-flux to prevent oxidation were processed by centrifugal casting in a vacuum system for irregular-shaped designs, such as animals [10]. It is known that the microstructure of Au-Al intermetallic compound systems includes very large grain sizes. Preliminary studies have explored high-cooling-rate processing of Au-23.5–25 wt% Al alloys with Si, Co, Ni, and Zn additions to obtain the off-stoichiometric AuAl2 intermetallic compound and improve the ductility of purple gold [11]. Later, blue and purple gold intermetallic compounds were tested using the “crash meter” to quantify fracture resistance for jewelry applications [12]. Recently, the AuAl2 intermetallic powder with added Cu exhibited a decrease in the AuAl2 lattice parameter as the Cu content increased, and changes in mechanical and optical properties [13,14]. Purple gold products were widely introduced to the market due to the high production costs and complexity. Many techniques have been introduced to improve the properties of purple gold compounds for industrial operations. Lower costs and simpler techniques, such as casting, are necessary for mass production.
The study design has two stages. First, the AuAl2 alloy systems are systematically screened under traditional jewelry casting as a practical industrial baseline. The Si and Co additions were designed to reduce the brittle intermetallic microstructure of AuAl2 while preserving the characteristic of purple gold alloy. Second, a copper mold is used for fast-cooling casting. Although this technique is not commonly used in jewelry manufacturing due to its higher cost and process complexity, it offers an alternative for refining grain size and enhancing the AuAl2 phase. Rapid solidification has been shown to improve the ductility of ordered intermetallics for turbine engine applications [15,16,17]. A similar effect has been observed in gold-based systems, enhancing ductility and reducing brittleness [18].
To examine the influence of solidification on the AuAl2 intermetallic compound, Electron Backscatter Diffraction (EBSD) quantitatively measures grain size, grain boundary distribution, and misorientation. The technique allows for evaluating how rapid solidification modifies grain refinement and structure [19,20]. Therefore, the microstructure–property engineering relationship under different casting conditions and microstructures can guide the engineering design of more ductile purple gold alloys.
2. Materials and Methods
2.1. Materials
Purple gold (PG) alloys of 18 karat were prepared with different alloying compositions. A commercial Au–Al binary alloy containing 75 wt% Au and 25 wt% Al with an off-stoichiometric AuAl2 composition [21] was set as a standard alloy and designated as 0-PG. Si and Co additions were introduced to modify the brittle intermetallic microstructure of AuAl2 while preserving the characteristic purple color of the alloy. To purposely modify the off-stoichiometric, 2 wt% silicon (Si) and a 1 wt% silicon (Si)–1 wt% cobalt (Co) mixture were selected to add to AuAl2. 2 wt% Silicon is preferable since the system of Au–Si and Al–Si exhibits relatively simple eutectic reactions and lower-melting constituents, which can improve melt fluidity and solidification behavior [22,23]. Co allows only 1 wt% due to the different crystal structures and phase diagrams of Au-Co and Al-Co, which result in more complex intermetallic phase formation [24,25]. Therefore, two compositions were prepared using traditional investment-casting in jewelry: 1-PG-2Si-TC and 2-PG-SiCo-TC. Correspondingly, two alloys with the same compositions were processed by copper mold casting: 3-PG-2Si-CC and 4-PG-SiCo-CC. The chemical compositions of the alloys are summarized in Table 1. All alloys were melted in a vacuum induction furnace under an argon atmosphere using high-purity raw materials (Au: 99.99%, Al: 99.9%, Si: 99.9%, and Co: 99.9%).

Table 1.
The chemical compositions (wt%) of Au–Al-based purple gold alloys.
2.2. Casting Techniques
Two casting techniques were employed to investigate the influence of cooling rate on the solidification behavior and microstructural evolution of the purple gold alloys. In the traditional investment casting (TC) process, the molten alloy was inserted into gypsum-bonded investment molds under an argon atmosphere using the centrifugal technique. The gypsum-bonded molds were set up for casting at 580 °C and casting was performed at a casting temperature of 1150 °C. This route represents a conventional jewelry casting technique characterized by a relatively low cooling rate.
In contrast, the copper mold casting (CC) process involved centrifugal casting of the molten alloy into a copper mold under an argon atmosphere to achieve rapid solidification. The cooling rate was estimated to be on the order of 30–200 K s−1 [18,26,27]. Owing to the geometry of the copper mold, ring-shaped specimens were produced. The rapid solidification technique was applied to suppress dendritic growth and refine the microstructure. The samples were then finished by using conventional jewelry processes, including grinding, polishing, and buffing [28,29].
2.3. Microstructural Characterization
Microstructural characterization was observed the effects of Si and Co additions and solidification rate on the morphology, grain structure, grain boundary characteristics, and crystallographic features of the 18 karat purple gold alloys by Scanning Electron Microscopy (SEM), Electron Backscatter Diffraction (EBSD), and X-ray Diffraction (XRD), which were employed as the principal analytical techniques, while Transmission Electron Microscopy (TEM) was used as a supplementary method for nanoscale observation.
SEM JEOL JSM-IT210 (JEOL Ltd. Headquarters, Tokyo, Japan) coupled with Energy-Dispersive X-ray Spectroscopy (EDS) was operated at an accelerating voltage of 20 kV under high-vacuum conditions, used to observe microstructure, grain morphology, and mapping elemental distributions, which were processed using integrated SMILE VIEW™ Lab software version 3.21. The samples were polished using Silica Carbide abrasive papers (TKX Corporation, Osaka, Japan) of 600, 800, 1000, and 1200 grit, followed by polishing with 1 µm and 0.5 µm diamond suspensions using a METAPOL-2 polishing machine (Laizhou Lyric Testing Equipment Co., Ltd., Laizhou, China) to achieve a mirror-like surface finish suitable for microscopic analysis. SEM is also used to observe fractography, such as cleavage and river patterns, according to standard fractography [30,31].
Crystallographic information was obtained by X-ray diffraction (XRD, Bruker D8 Discover diffractometer, Billerica, MA, USA) using Cu Kα radiation (λ = 1.5406 Å). Diffraction patterns were collected over a 2θ range of 20°–90°, with a step size of 0.02°, a counting time of 0.4 s per step, and a scan rate of 3° min−1. All samples were observed for phase identification and the detection of diffraction peak broadening associated with processing.
The study was subsequently focused on investigating the microstructural characteristics of 3-PG-2Si-CC and 4-PG-SiCo-CC processed via rapid solidification, using Electron Backscatter Diffraction (EBSD). EBSD is used to observe crystal orientation and grain boundary relationships with the Oxford system. The EBSD data were processed using AZtecCrystal™ software version 2.1 to generate grain orientation maps, grain size distributions, and statistics on grain boundary misorientation angles.
Finally, transmission electron microscopy (TEM, JEOL JEM-2100F, JEOL Ltd. Headquarters, Tokyo, Japan) was employed for nanoscale observation. Thin foils were prepared by twin-jet electropolishing using a 5 vol.% perchloric acid–ethanol solution at 15 V and −30 °C. TEM observations are used to characterize local defects along the grain boundaries of sample 3-PG-2Si-CC and sample 4-PG-SiCo-CC at a fast-cooling technique from copper mold casting.
3. Results and Discussion
3.1. Bulk Samples
The purple gold alloy generally exhibits brittleness [2,3,4,5,6,7,8,9,10], which limits mechanical processing after casting. Traditional investment casting (TC) samples could not be polished during the standard jewelry manufacturing process [28,29] due to their brittleness. It cracks and damages due to the intrinsically ordered structure of intermetallic alloys. Figure 1 compares the surface finish of the 1-PG-2Si-TC alloy prepared by TC (Figure 1a) and by rapid solidification via 3-PG-2Si-CC (Figure 1b). It clearly illustrates the differences in surface finish and polishing ability of the samples. It is consistent with Greer et al., who reported that rapid solidification can significantly reduce long-range order and refine microstructure in the commercial NiAl system [32]. This confirms that the CC technique serves its engineering purpose.

Figure 1.
An example of purple gold (a) 1-PG-2Si-TC: traditional investment casting (TC) (b) 3-PG-2Si-CC: rapid solidification by copper mold casting (CC).
3.2. Microstructure
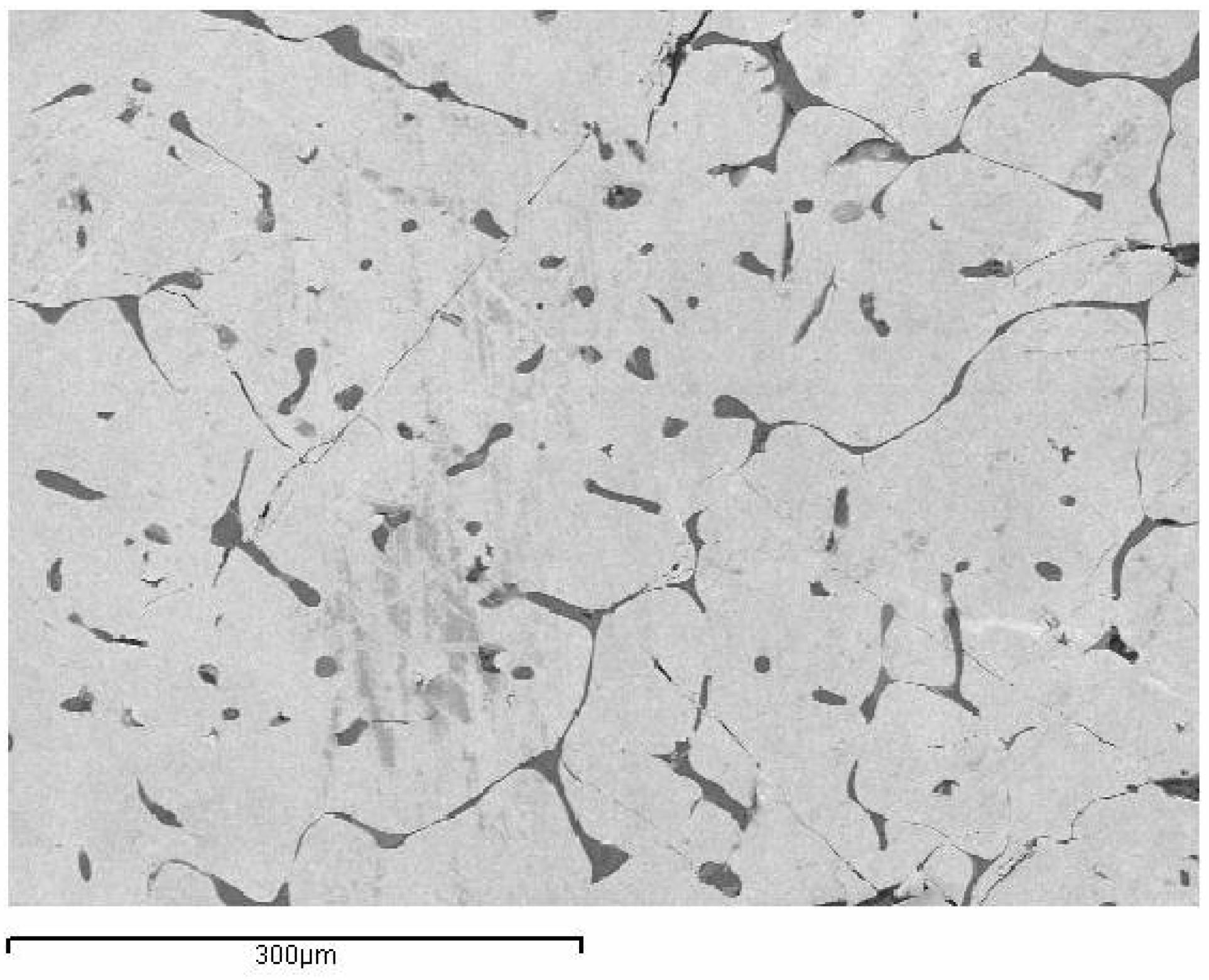
The standard alloy 0-PG, processed by traditional investment casting, exhibited a relatively coarse, non-uniform grain structure under slow-cooling conditions, as shown in Figure 2. With Si- and Si-Co addition alloys, the microstructure was changed. 1-PG-2Si-TC and 2-PG-SiCo-TC exhibit more distinct grains with eutectic-like microstructures along the grain boundaries due to the slow cooling rate, as shown in Figure 3a,b. By contrast, the rapidly solidified samples 3-PG-2Si-CC and 4-PG-SiCo-CC exhibited more pronounced refined grain sizes and homogeneous grain with substantially suppressed eutectic structures, as shown in Figure 3c,d. This is related to the others in that rapid solidification techniques can refine grain size and structure [16,27].
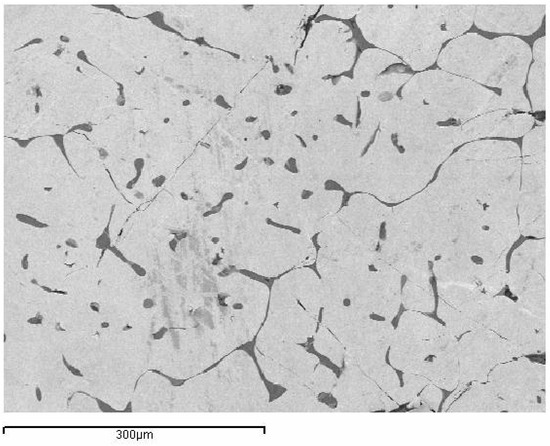
Figure 2.
Secondary electron (SE) micrograph of the base alloy 0-PG (75 wt% Au–25 wt% Al) produced by traditional investment casting, showing coarse and non-uniform grain morphology.

Figure 3.
BSE microstructure comparisons of the grain boundary for different processes and compositions of Traditional Investment casting (TC) and Copper mold casting (CC) (a) 1-PG-2Si-TC, (b) 2-PG-SiCo-TC, (c) 3-PG-2Si-CC, and (d) 4-PG-SiCo-CC.
Si-Co modified addition alloy was selected to investigate the segregation and distribution of the addition alloy of 2-PG-SiCo-TC and 4-PG-SiCo-CC. The two samples have the same composition but different cooling rate casting conditions. At the slow cooling rate, 2-PG-SiCo-TC forms AuAl2 intermetallic compound grains, and Si and Co segregate along grain boundaries, as shown in the SEM mapping in Figure 4a. In contrast, the 4-PG-SiCo-CC, with the fast cooling rate, showed segregation of Al along grain boundaries, while Au remained within the grains. This indicates that the rapid solidification process involves sluggish interdiffusion, as shown in Figure 4b. Both samples show that Si was primarily localized at grain boundaries and exhibited limited solubility in the Au–Al matrix due to the larger atomic size of Si. The segregation behavior of Co followed a similar trend due to a different crystal structure.

Figure 4.
SE image of 2-PG-SiCo-TC (a) and 4-PG-SiCo-CC (b). The mapping shows the composition of Au, Al, Si and Co.
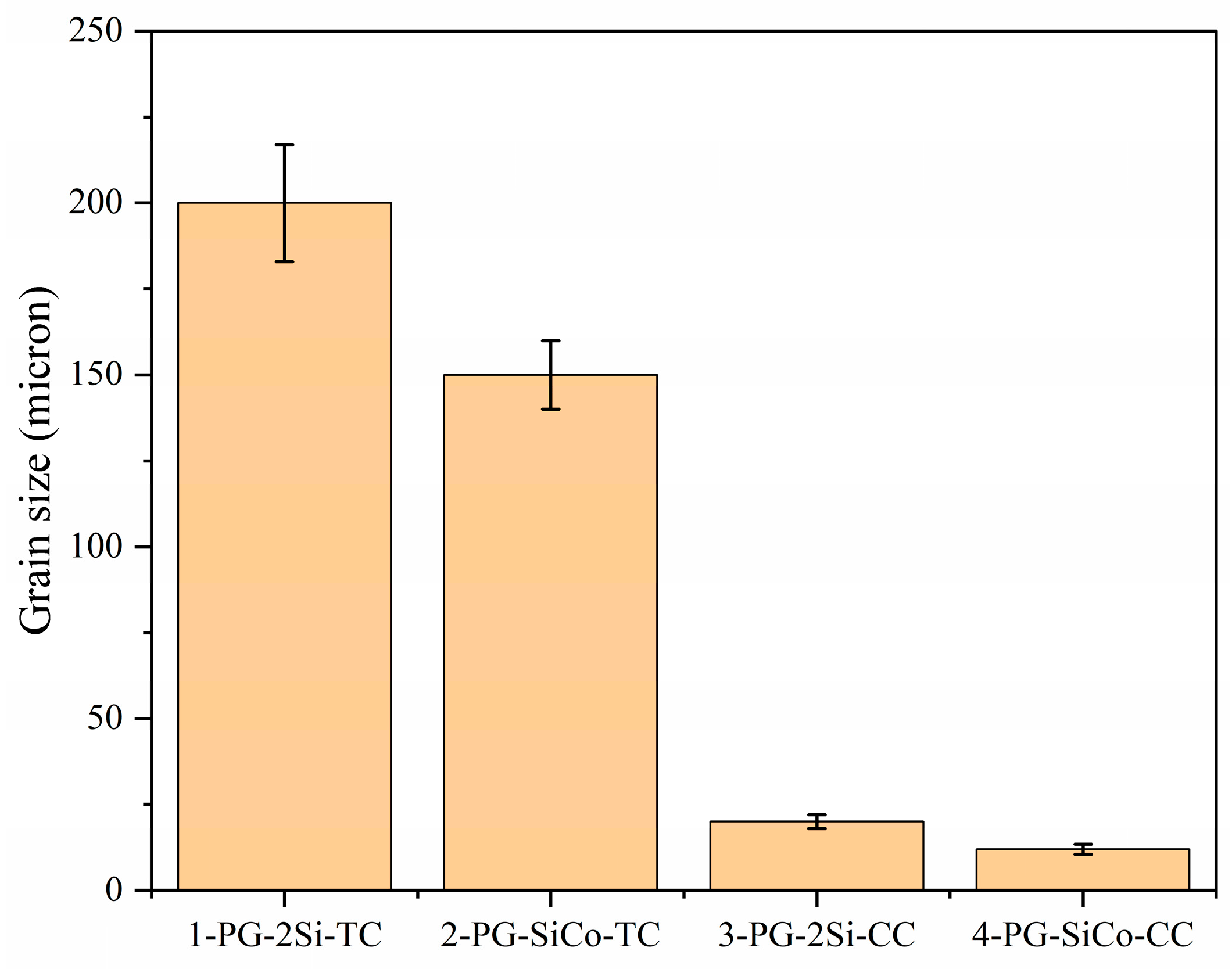
Figure 5 shows the average grain size among alloys produced by traditional investment casting and copper mold casting. The investment-cast samples exhibited relatively coarse-grained structures, with average grain sizes of approximately 200 μm and 150 μm for 1-PG-2Si-TC and 2-PG-SiCo-TC, respectively. By comparison, fast solidification conditions show grain refinement; therefore, the average grain sizes are extremely reduced to approximately 20 μm for 3-PG-2Si-CC and 12 μm for 4-PG-SiCo-CC, respectively. This refinement is primarily attributed to the high cooling rates associated with copper mold casting, which enhance nucleation rates and effectively suppress grain growth during solidification [33].
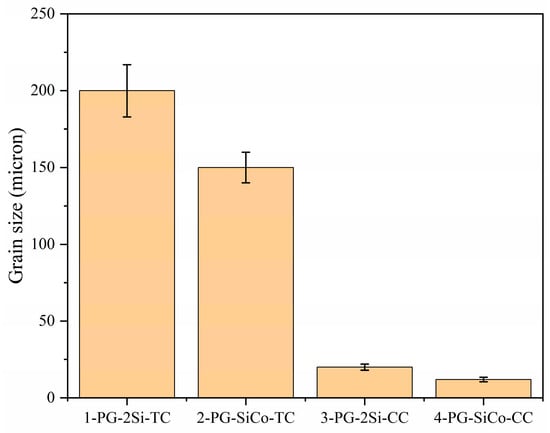
Figure 5.
The grain size of the four samples.
Fractography was performed using SEM micrographs to observe the microstructure. 1-PG-2Si-TC, shown in Figure 6a, exhibited river-like cleavage patterns along the intermetallic of large grains at 200 μm, as shown in Figure 5, indicating brittle-like fracture behavior [34]. 2-PG-SiCo-TC displayed a smaller average grain size of 150 μm, as shown in Figure 6b. By adding 1 wt%Co to the alloys, brittle behavior is observed in intergranular grains, where cracks propagate along grain boundaries. In contrast, the rapidly solidified samples 3-PG-2Si-CC and 4-PG-SiCo-CC represent ductile-like morphologies along grain boundaries, as shown in Figure 6c and Figure 6d, respectively. It is consistent with other reports on the fine-grained size of intermetallics exhibiting ductile dimple rupture [35,36,37].

Figure 6.
Fractography for different processes and compositions of Traditional Investment casting (TC) and Copper mold casting (CC): (a) 1-PG-2Si-TC, (b) 2-PG-SiCo-TC, (c) 3- PG-2Si-CC and (d) 4-PG-SiCo-CC.
3.3. XRD Analysis
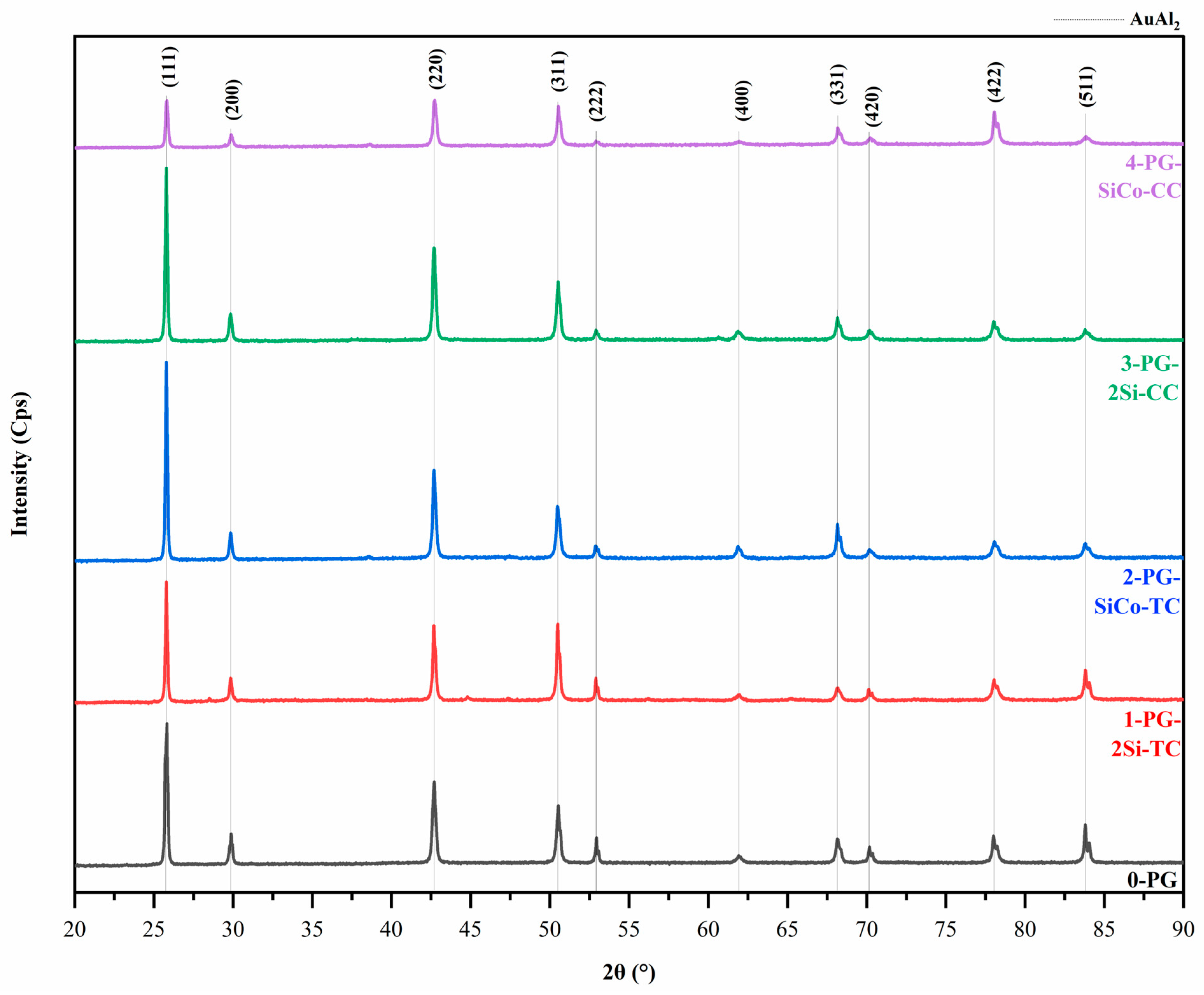
XRD result confirms the majority of AuAl2 intermetallic compound in all samples using JCPDS #17-0877, with the indexing of the CaF2-type cubic structure [38]. No additional phases of Si and Co were detected within the resolution of the measurement due to the composition being too small, as shown in Figure 7. 0-PG exhibits sharp diffraction peaks, indicating an ordered AuAl2 structure under standard conditions. In comparison, the Si- and Si–Co-modified alloys produced by traditional investment casting (1-PG-2Si-TC and 2-PG-SiCo-TC) show minor peak broadening and intensity variation at second-order reflections. The high intensities of (111) and (200) confirm the formation of the AuAl2 intermetallic phase, except in the 4-PG-SiCo-CC. At fast cooling, 4-PG-SiCo-CC shows a lower orientation of the (200) plane, indicating lattice strain in the sample. The peaks of higher-order reflections, such as (222) and (400), show relatively low intensity and broaden for 0-PG, 1-PG-2Si-TC, 2-PG-SiCo-TC, 3-PG-2Si-CC, and 4-PG-SiCo-CC. It indicates lattice distortions that are more pronounced with faster cooling [39].
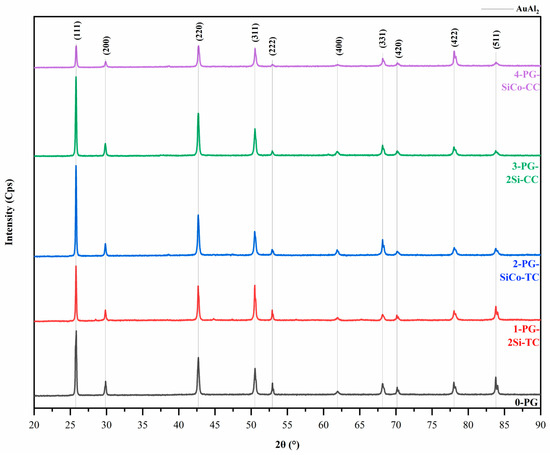
Figure 7.
X-ray diffraction (XRD) patterns of Au–Al-based purple gold alloys with different alloying elements and casting methods, showing AuAl2 intermetallic compound as the dominant phase and minor peak broadening in Si- and Si–Co-modified samples.
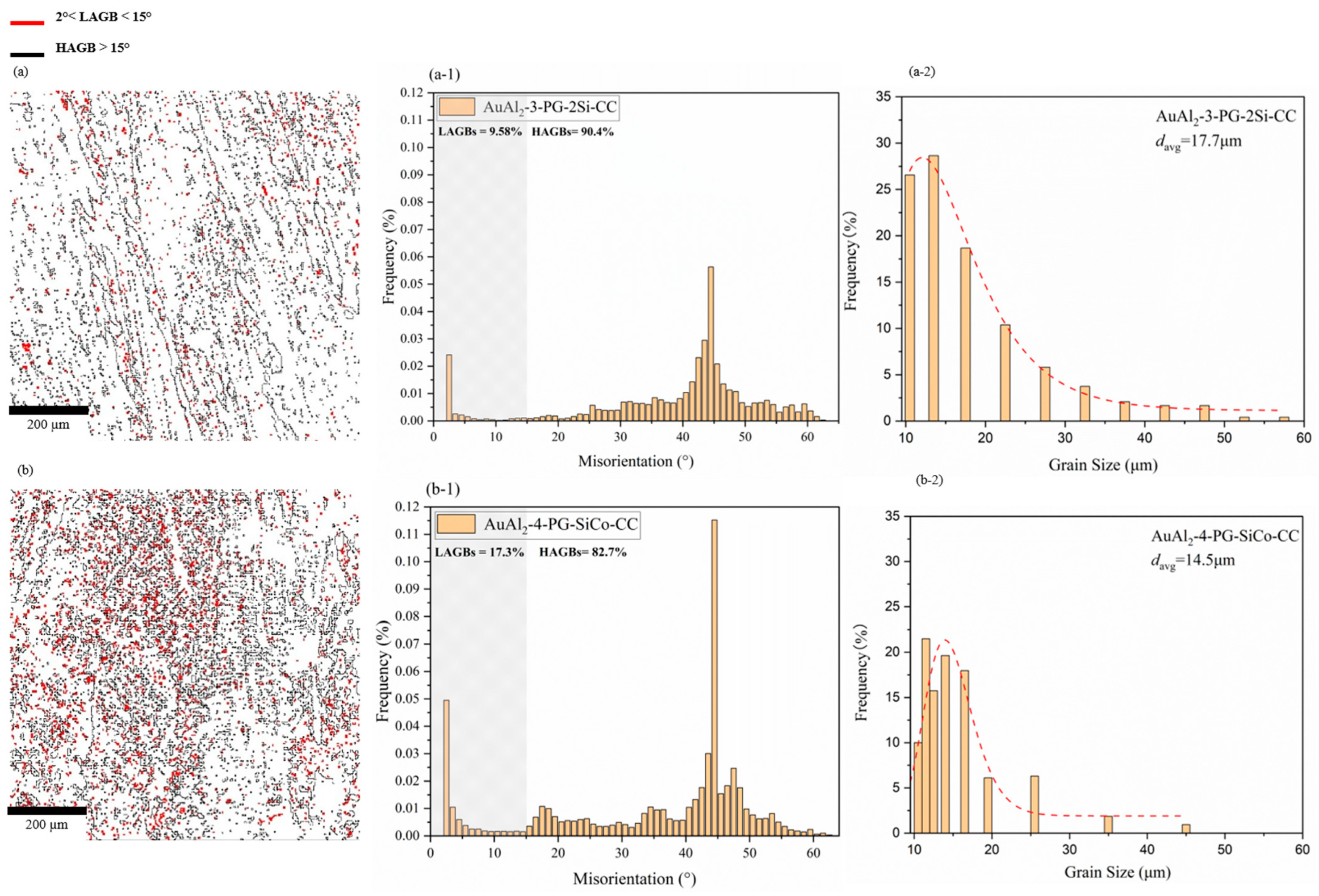
3.4. EBSD Analysis
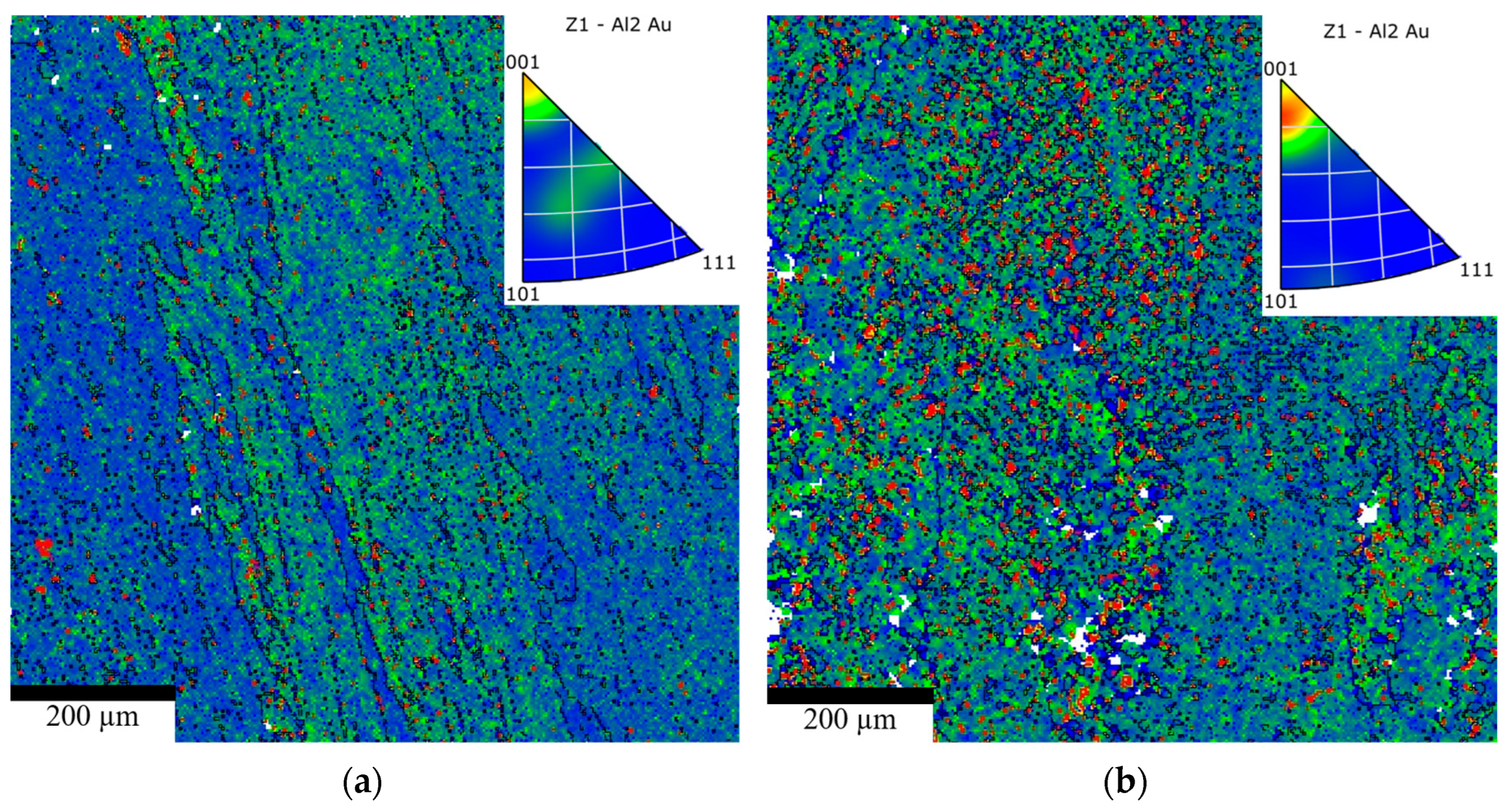
Electron Backscatter Diffraction (EBSD) was employed to characterize the microstructure, including grain boundary distribution, misorientation, and grain size, in AuAl2-based alloys with a fast-cooling rate and different compositions (3-PG-2Si-CC and 4-PG-SiCo-CC). Figure 8a,b shows the inverse pole figure (IPF) maps, which indicate that the grain boundary map of 4-PG-SiCo-CC exhibits a more dispersed and well-developed grain boundary network with a high intensity of low-angle grain boundaries and minimal sub-grain formation, compared with 3-PG-2Si-CC.
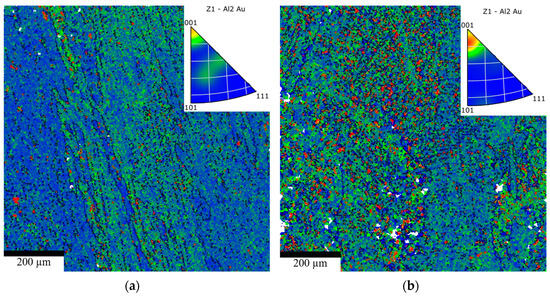
Figure 8.
Inverse pole figure (IPF) maps for (a) 3-PG-2Si-CC and (b) 4-PG-SiCo-CC.
Adding Co introduces local lattice distortion, as atomic-size, crystal-structure, and lattice-parameter mismatches [40,41,42] create local strain fields in the crystal lattice [43,44] during solidification, resulting in a high-density LAGB phenomenon in 4-PG-SiCo-CC. In Figure 9(a-1,b-1), the low-angle grain boundary (LAGB) is measured in the range of angle at 2–15°and high-angle grain boundary (HAGB) in the range more than 15°, indicating in EBSD concept elsewhere [20,45,46]. Si-modified alloy of 3-PG-2Si-CC, as shown in Figure 9(a-1), LAGB is at 9.58%, and HAGB is at 90.4%, whereas Si-Co modified alloy of 4-PG-SiCo-CC, as shown in Figure 9(b-1), LAGB is at 17.3%, and HAGB is at 82.7%. In Figure 9(a-2), 3-PG-2Si-CC indicates an average grain size of approximately 17.7 μm, in agreement with SEM observations and Figure 5. A further improvement in microstructural refinement is observed in the Si–Co alloy of 4-PG-SiCo-CC (Figure 9(b-2)), with a smaller average grain size (~14.5 μm) and a narrower grain size distribution. With a larger percentage of LAGB of Si-Co modified alloy of 4-PG-SiCo-CC, resulting in smaller grain sizes.
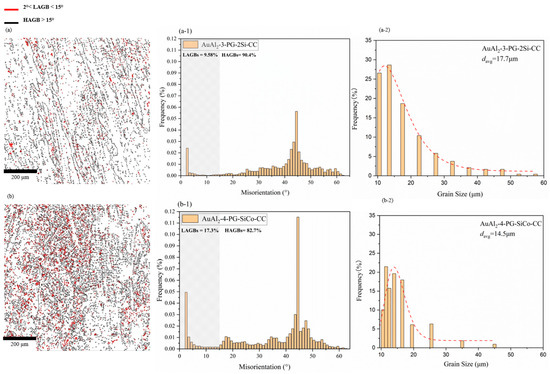
Figure 9.
EBSD orientation maps, misorientation histograms, and grain size distributions of rapidly solidified AuAl2 alloys, (a,b) represent grain boundary maps for 3-PG-2Si-CC and 4-PG-SiCo-CC, (a-1,b-1) show the percentage of LAGBs and HAGBs of misorientation, and (a-2,b-2) display refined grain structures with average grain sizes of 17.7 μm and 14.5 μm, respectively.
Overall, the EBSD observations of grain size and misorientation indicate that the 4-PG-SiCo-CC exhibits a smaller grain size and a higher percentage of LAGB than the 3-PG-2Si-CC under the same fast-cooling-rate conditions. According to Gourgues [20] and Pan [47], LAGB are always less sensitive to cracks than HAGB. This shows that smaller grain sizes and a higher percentage of LAGB are more likely to exhibit ductile behavior, as shown by more ductile fracture-like features along grain boundaries in Figure 6d for 4-PG-SiCo-CC.
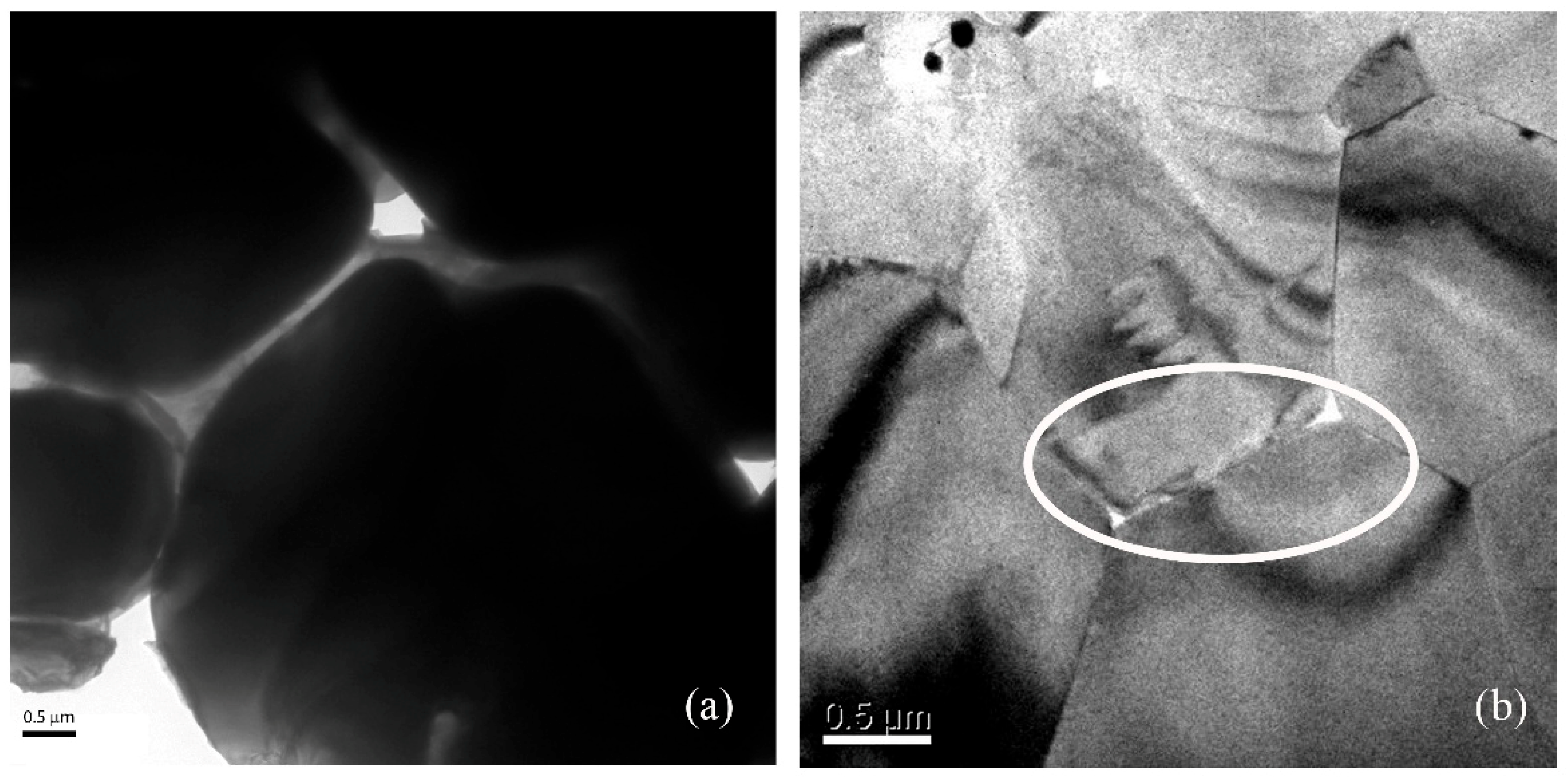
3.5. TEM Analysis
Transmission electron microscopy (TEM) was used to examine the nano-defects at the grain boundary of 3-PG-2Si-CC and 4-PG-SiCo-CC, which have different cooling rates, as shown in Figure 10. The 3-PG-2Si-CC exhibits nanoscale voids with characteristic sizes of approximately ~500 nm along grain boundaries (Figure 10a), indicating incomplete boundary consolidation and localized defect accumulation. In contrast, the 4-PG-SiCo-CC sample shows that grain boundaries were closely packed together with smaller voids of approximately ~80 nm (Figure 10b). The reduction in void size in 4-PG-SiCo-CC is consistent with the finer grain size and lower average local misorientation revealed by SEM and EBSD analyses, respectively. The TEM observations therefore provide nanoscale evidence of increased microstructural refinement, thereby improving grain boundary stability in the Si–Co-modified purple gold alloy. It is also consistent with the results of both EBSD and fractography, as mentioned above.
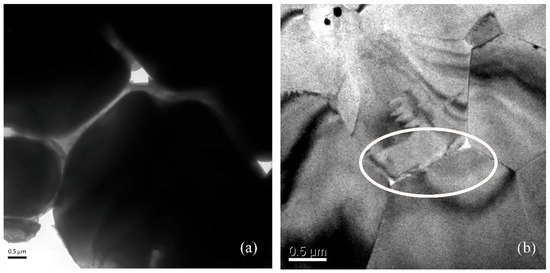
Figure 10.
TEM image of 75Au23Al-1Si1Co shows holes along the grain boundary at different solidification rates: (a) 3-PG-2Si-CC showing voids approximately 500 nm, (b) 4-PG-SiCo-CC showing small voids approximately 80 nm.
4. Conclusions
This study investigated the microstructure of 18-karat purple gold based on the Au–Al intermetallic compound and the effects of Si and Co additions on microstructural evolution and stability, in conjunction with rapid solidification via copper mold casting. The main conclusions are summarized as follows:
- A refinement of the intermetallic microstructure was achieved by engineering design of addition alloys and a rapid cooling technique. The addition of Si and Co, together with a fast cooling rate, effectively reduced the average grain size from approximately within 150–200 μm to within ~12–20 μm, thereby significantly improving microstructural uniformity. The observed diffraction peak broadening in the Si- and Si–Co-modified alloys reflects crystallographic refinement and lattice distortion associated with microstructural modification.
- Grain boundary characteristics were observed by the EBSD technique under rapid solidification conditions. The Si–Co-modified alloy exhibited finer grain sizes and a higher LAGB misorientation (17.3%), indicating a lower risk of cracking.
- At the nanoscale, by TEM observations, Si and Co additions promoted more compact, continuous grain boundary structures with reduced nanoscale intergranular voids, derived from grain refinement, which contribute to the ductility improvement of the AuAl2 intermetallic compound. The reduction in void size with Si and Co additions is consistent with the finer grain size and lower average misorientation revealed by SEM and EBSD analyses, respectively. This microstructure modification provides an engineering design to suppress the intrinsic brittleness of purple gold while preserving its purple color for alternative jewelry applications.
Author Contributions
Conceptualization, A.P., J.Y. (Jingran Yang) and K.W.; methodology, A.P., J.Y. (Jingran Yang), J.Y. (Jie Yu) and J.L., K.W.; investigation, A.P., J.L., J.Y. (Jingran Yang) and K.W.; formal analysis, A.P. and J.Y. (Jingran Yang); data curation, A.P., J.Y. (Jie Yu) and K.W.; validation, A.P., J.Y. (Jingran Yang), and K.W.; resources, J.Y. (Jingran Yang), J.L., J.Y. (Jie Yu) and K.W.; visualization, A.P.; writing—original draft preparation, A.P. and K.W.; writing—review and editing, A.P., J.Y. (Jingran Yang) and K.W.; supervision, J.Y. (Jingran Yang) and K.W.; project administration, K.W.; funding acquisition, J.Y. (Jie Yu) and K.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Commission on Higher Education, Ministry of Education (Industry Section No. 111/2550), and Srinakharinwirot University (Grant No. 234-6/2556). This work was also supported by the Yunnan Province Industrial High-Tech Project (Grant No. 202403AA080015) and the Special Science and Technology Research Project of Yunnan Province for South Asia and Southeast Asia Innovation (Grant No. 202403AP140006).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding authors.
Acknowledgments
The authors acknowledge the technical and administrative support provided by Srinakharinwirot University and Kunming University of Science and Technology. In particular, the authors thank College of Creative Industry, for access to experimental facilities and characterization instruments used in this study. The Faculty of Materials Science and Engineering and the Key Laboratory of Advanced Materials of Precious-Nonferrous Metals, Ministry of Education of China, Kunming University of Science and Technology, Kunming 650093, China, and Precious Metal Unit.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study, in the collection, analysis, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
References
- Russell, A.M.; Lee, K.L. Structure–Property Relations in Nonferrous Metals; Wiley: Hoboken, NJ, USA, 2005; p. 474. [Google Scholar]
- Philofsky, E. Intermetallic formation in gold–aluminum systems. Solid State Electron. 1970, 13, 1391–1399. [Google Scholar] [CrossRef]
- Yukio, M. Purple Gold Alloy Wire and Its Manufacture. Japanese Patent JPH04176829, 24 June 1992. [Google Scholar]
- Cretu, C.; van der Lingen, E. Coloured gold alloys. Gold Bull. 1999, 32, 115–122. [Google Scholar] [CrossRef]
- Loh, P.C. Jewelry Alloy Composition. U.S. Patent 6,929,776, 16 August 2005. [Google Scholar]
- Fukayama, S. Purple Sintered Gold Alloy for Ornamentation. Japanese Patent JP62240729, 21 October 1987. [Google Scholar]
- Yamazaki, S.; Ishii, N.; Sawada, K. Noble Metal Ornament Member and Its Production Method, and Ornament Part and Accessory Part. Japanese Patent JP2003/183710, 3 March 2003. [Google Scholar]
- Steinemann, S. Intermetallic Compound, Method for Producing the Compound, and Use of the Compound. U.S. Patent 4,911,762, 27 March 1990. [Google Scholar]
- Süss, R.; Cretu, C. Proceedings of the Microscopy Society of Southern Africa; McLean, M., Engelbrecht, J.A.A., Neethling, J.H., Dennison, C., Mycock, D., Cross, R.H.M., Eds.; Microscopy Society of Southern Africa: Johannesburg, South Africa, 1998; Volume 28, p. 20. [Google Scholar]
- Wongpreedee, K.; Tansakul, T.; Schuster, H.J.; Chookruvong, K. Purple gold: Past, present and future to ductile intermetallics. In Proceedings of the Gold 2006: International Conference on the Science, Technology, and Industrial Applications, Limerick, Ireland, 3–6 September 2006; p. 163. [Google Scholar]
- Wongpreedee, K.; Ruethaithananon, P. Microstructure of Au–Al systems manipulated by rapid solidification techniques. In Proceedings of the 5th International Conference on Gold Science, Technology and Its Applications, Heidelberg, Germany, 26–29 July 2009. [Google Scholar]
- Bühner, J.F.; Basso, A.; Poliero, M. Metallurgy and processing of coloured gold intermetallics—Part II: Investment casting and related alloy design. Gold Bull. 2010, 43, 11–20. [Google Scholar] [CrossRef]
- Volkova, E.G.; Kozlov, K.A.; Antonov, B.D.; Murzakaev, A.M.; Zavalishin, V.A.; Livinets, A.A.; Volkov, A.Y. Alloying of Al2Au intermetallic compound with Cu by the ball milling technique. J. Alloys Compd. 2022, 900, 163429. [Google Scholar] [CrossRef]
- Volkova, E.G.; Antonov, B.D.; Zavalishin, V.A.; Knyazev, Y.V.; Gavrilova, A.A.; Volkov, A.Y. The structure of cast Al2Au intermetallic compound with added Cu. Phys. Met. Metallogr. 2023, 124, 544–550. [Google Scholar] [CrossRef]
- Liu, C.T.; Stiegler, J.O. Ductile ordered intermetallic alloys. Science 1984, 226, 636–642. [Google Scholar] [CrossRef]
- Jiao, Z.B.; Luan, J.H.; Liu, C.T. Strategies for improving ductility of ordered intermetallics. Prog. Nat. Sci. Mater. Int. 2016, 26, 1–12. [Google Scholar] [CrossRef]
- Koch, C.C. Rapid solidification of intermetallic compounds. Int. Mater. Rev. 1988, 33, 201–219. [Google Scholar] [CrossRef]
- Bai, F.X.; Yao, J.H.; Wang, Y.X.; Pan, J.; Li, Y. Crystallization kinetics of an Au-based metallic glass upon ultrafast heating and cooling. Scr. Mater. 2017, 132, 58–62. [Google Scholar] [CrossRef]
- Randle, V. Microtexture Determination and Its Applications, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2021. [Google Scholar]
- Gourgues, A.-F. Electron backscatter diffraction and cracking. Mater. Sci. Technol. 2002, 18, 119–133. [Google Scholar] [CrossRef]
- Murray, J.L.; Okamoto, H.; Massalski, T.B. The Al–Au (Aluminum–Gold) System. Bull. Alloy Phase Diagr. 1987, 8, 20–30. [Google Scholar] [CrossRef]
- Okamoto, H.; Massalski, T.B. The Au–Si (gold–silicon) system. Bull. Alloy Phase Diagr. 1987, 8, 454–474. [Google Scholar] [CrossRef]
- Asensio-Lozano, J.; Vander Voort, G. The Al–Si phase diagram. Buehler Tech Notes 2007, 5, 1–4. [Google Scholar]
- Yang, K.; Kryutyanskiy, V.; Kolmychek, I.; Murzina, T.V.; Lukaszew, R.A. Experimental correlation between nonlinear optical and magnetotransport properties observed in Au–Co thin films. J. Nanomater. 2016, 2016, 4786545. [Google Scholar] [CrossRef]
- McAlister, A.J. The Al–Co (aluminum–cobalt) system. Bull. Alloy Phase Diagr. 1989, 10, 646–650. [Google Scholar] [CrossRef]
- Kozieł, T. Estimation of cooling rates in suction casting and copper-mould casting processes. Arch. Metall. Mater. 2015, 60, 767–771. [Google Scholar] [CrossRef]
- Lavernia, E.J.; Srivatsan, T.S. The rapid solidification processing of materials: Science, principles, technology, advances, and applications. J. Mater. Sci. 2010, 45, 287–325. [Google Scholar] [CrossRef]
- Faccenda, V.; Ott, D. Handbook of Investment Casting; World Gold Council: London, UK, 2003. [Google Scholar]
- Brepohl, E. The Theory and Practice of Goldsmithing; McCreight, T., Ed.; Lewton-Brain, C., Translator; Brynmorgen Press: Portland, ME, USA, 2001. [Google Scholar]
- ASM International. Fractography, 9th ed.; Metals Handbook; ASM International: Materials Park, OH, USA, 1987; Volume 12. [Google Scholar]
- Lynch, S.P.; Moutsos, S. A brief history of fractography. J. Fail. Anal. Prev. 2006, 6, 54–69. [Google Scholar] [CrossRef]
- Greer, A.L.; Assadi, H. Rapid solidification of intermetallic compounds. Mater. Sci. Eng. A 1997, 226–228, 133–141. [Google Scholar] [CrossRef]
- Eskin, D.; Du, Q.; Ruvalcaba, D.; Katgerman, L. Experimental study of structure formation in binary Al–Cu alloys at different cooling rates. Mater. Sci. Eng. A 2005, 405, 1–10. [Google Scholar] [CrossRef]
- Hertzberg, R.W.; Vinci, R.P.; Hertzberg, J.L. Deformation and Fracture Mechanics of Engineering Materials, 5th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Wongpreedee, K.; Russell, A.M.; Gschneidner, K.A. Mechanical properties of RE–Ag intermetallic compounds. In Proceedings of the International Precious Metals Conference, Phoenix, AZ, USA, 12–15 June 2004. [Google Scholar]
- Sater, J.M.; Jha, S.C.; Sanders, T.H., Jr. Microstructural characterization of rapidly solidified aluminum–transition metal alloys. Mater. Sci. Eng. 1987, 91, 201–216. [Google Scholar] [CrossRef]
- Zhang, Z.; Russell, A.M.; Biner, S.B.; Gschneidner, K.A., Jr.; Lo, C.C.H. Fracture toughness of polycrystalline YCu, DyCu, and YAg. Intermetallics 2005, 13, 559–566. [Google Scholar] [CrossRef]
- Mehl, M.J.; Hicks, D.; Toher, C.; Levy, O.; Hanson, R.M.; Hart, G.; Curtarolo, S. The AFLOW library of crystallographic prototypes: Part 1. Comput. Mater. Sci. 2017, 136, S591–S592. [Google Scholar] [CrossRef]
- Cullity, B.D.; Stock, S.R. Elements of X-Ray Diffraction, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2001. [Google Scholar]
- Lu, J.; Zhan, M.; Yu, J.; Yu, X.; Duan, Y.; Chen, S.; Xu, M.; Lu, W. Insight on the Electronic, Elastic and Thermal Properties of Au–Al Intermetallic Compounds Based on First-Principles Calculations. J. Electron. Mater. 2024, 53, 3809–3821. [Google Scholar] [CrossRef]
- Hubbard, C.R.; Swanson, H.E.; Mauer, F.A. A Silicon Powder Diffraction Standard Reference Material. J. Appl. Crystallogr. 1975, 8, 45–48. [Google Scholar] [CrossRef]
- Jauch, W.; Reehuis, M. Electron density distribution in hexagonal cobalt: A γ-ray diffraction study. Phys. Rev. B 2009, 80, 125126. [Google Scholar] [CrossRef]
- Cheong, K.-S.; Busso, E.P. Effects of lattice misorientations on strain heterogeneities in FCC polycrystals. J. Mech. Phys. Solids 2006, 54, 671–689. [Google Scholar] [CrossRef]
- Li, J.; Chen, Y.; He, Q.; Xu, X.; Wang, H.; Jiang, C.; Liu, B.; Fang, Q.; Liu, Y.; Yang, Y.; et al. Heterogeneous lattice strain strengthening in severely distorted crystalline solids. Proc. Natl. Acad. Sci. USA 2022, 119, e2200607119. [Google Scholar] [CrossRef] [PubMed]
- Dingley, D.J.; Randle, V. Microtexture determination by electron back-scatter diffraction. J. Mater. Sci. 1992, 27, 4545–4566. [Google Scholar] [CrossRef]
- Humphreys, F.J. Review: Grain and subgrain characterisation by electron backscatter diffraction. J. Mater. Sci. 2001, 36, 3833–3854. [Google Scholar] [CrossRef]
- Pan, Y.; Adams, B.L.; Olsen, T.; Panayotou, N. Grain-boundary structure effects on intergranular stress corrosion cracking of Alloy X-750. Acta Mater. 1996, 44, 4685–4695. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.