Abstract
Micro-segregation of solute elements is inevitable during the casting process of Al–Mg–Si alloys, significantly influencing the precipitation behavior of dispersed phases during subsequent heat treatment, ultimately influencing alloy performance. Mn and Si are typical positive segregation elements and the principal constituents of the dispersed phases in aluminum alloys, and their diffusion behavior directly affects the precipitation of nano-scale α-Al(MnCr)Si phases within grains during subsequent annealing. This study systematically investigates the effects of different annealing conditions (430 °C × 12 h and 530 °C × 12 h) on the precipitation behavior of α-Al(MnCr)Si phases in the Al–Mg–Si–Mn alloy. After annealing at 430 °C, the relatively low diffusion rate promoted the dispersed precipitation of α-Al(MnCr)Si phases as high-density, nano-scale particles within grains. In contrast, annealing at 530 °C substantially enhanced the elements diffusion, accelerating both nucleation and growth of α-Al(MnCr)Si phases and inducing notable Ostwald ripening, resulting in larger α-Al(MnCr)Si phases with a lower number density within grains. This study indicates that the control of annealing parameters can effectively tailor the size, distribution, and number density of nano-scale α-Al(MnCr)Si phases. The findings provide critical theoretical and practical guidance for optimizing annealing processes in Al-Mg-Si-Mn alloys.
1. Introduction
Al–Mg–Si–Mn (6xxx series) alloys are widely used in automotive and aerospace industries due to their excellent formability, corrosion resistance, and moderate strength. The conventional production process for 6xxx series aluminum alloys typically includes melting, casting, annealing, deformation, solution treatment, and aging treatment. Rational design and precise control of each stage in the production process directly influence the final properties of the alloy. Casting and annealing, as the initial steps in alloy manufacturing, play a decisive role in subsequent processing operations through appropriate adjustment of their respective process parameters. However, during the casting cooling process, the inevitable micro-segregation behavior of solute elements influences the transformation and precipitation behavior of second phase, thereby determining the material’s mechanical properties. Post-casting annealing treatment can significantly mitigate the negative effects of element segregation, making it a crucial process in alloy manufacturing. During the casting process of 6xxx aluminum alloys, due to positive segregation of Si and the rapid diffusion of impurity element Fe, brittle intermetallic β-AlFeSi phases form in the interdendritic regions of the as-cast microstructure. These phases possess very high dissolution temperatures and cannot be dissolved back into the matrix during subsequent annealing treatments. Such coarse second phases tend to cause stress concentration and seriously reduce the alloy’s mechanical performance. Therefore, these brittle phases are often modified by adding Mn and Cr during casting. They can replace Fe atoms in the brittle β-AlFeSi phase during the subsequent annealing process, transforming it into fine bead-like α-Al(FeMn)Si phases, thereby reducing the detrimental effects of β-AlFeSi [1,2,3].
Studies have shown that the α-Al(FeMn)Si phase precipitated within grains acts as the primary dispersion phase that retards recrystallization in aluminum alloys and exhibits high thermal stability [4]. Maintaining fine size and stable crystal structure at elevated temperatures, its semi-coherent relationship with the matrix enables it to pin-up dislocation and (sub)grain boundary motion through Zener drag, thereby improving the resistance of recrystallization during deformation and improving the alloy’s thermal stability [5,6,7,8]. Most existing studies aim to regulate the precipitation of Mn-containing dispersoids by adjusting the content of Mn and/or Cr or by adding other slow-diffusing elements, thereby achieving better recrystallization retardation and improved alloy properties. For instance, Liu et al. found that a Mn/Cr ratio of 3.5 promotes the precipitation of a high density of fine dispersoids, enhancing both dispersion strengthening and Zener pinning [9]. Ånes et al. demonstrated that the dragging effect of dispersoids on (sub)grain boundaries exhibits orientation dependence, with Cube-oriented subgrains experiencing the strongest drag after recovery and partial recrystallization [10]. Nagaumi et al. reported that the presence of α-dispersoids in Mn-containing alloys effectively refines the substructure with misorientations in the range of 2–5° and hinders dynamic recovery [11]. Li et al. achieved enhanced pinning force on migrating boundaries by adding Mo, which significantly improved the recrystallization resistance of the alloy [12]. Qian et al. showed that adjusting the Mn content enables dispersoids to effectively pin dislocation motion and subgrain migration, thereby delaying dynamic recovery and suppressing recrystallization [13]. Meanwhile, a high density of Mn-bearing α phases can further enhance the mechanical properties of the alloy via dispersion strengthening [14,15]. Although these methods can improve the precipitation characteristics of dispersoids, such as their size and number density, the increased content of alloying elements and the addition of other elements raise the cost of alloy production. To date, few studies have attempted to tailor the precipitation features of intragranular dispersoids through the design of annealing processes. The annealing treatment after casting directly influences the alleviation of elemental segregation, the modification of the second phase at grain boundaries, and the precipitation of Mn-containing dispersoids, thereby playing a crucial role in the regulation of the alloy’s microstructure and properties.
Based on the above considerations, this study designed a low-cost annealing process and investigated the effects of different annealing temperatures (430 °C and 530 °C) on the precipitation characteristics of intragranular α-Al(FeMnCr)Si phases in a rapidly solidified Al–Mg–Si–Mn alloy. The work elucidated the influence of elemental diffusion behavior on dispersed phases precipitation characteristics (size, number density, and morphology). This research provided a theoretical basis and practical guidance for optimizing the heat treatment process of Al–Mg–Si–Mn alloys and controlling the precipitation of α-Al(MnCr)Si phases.
2. Experimental Procedure
An Al–Mg–Si–Mn alloy with a chemical composition of 1.2 Mg, 1.2 Si, 0.4 Cu, 0.5 Mn, 0.15 Cr, 0.2 Zn, 0.15 Ti, and balance Al (wt.%) was prepared by casting. The elemental composition was measured by spark optical emission spectrometry.
The alloy was melted in a resistance furnace using raw materials, including 99.99% industrial high-purity Al, Mg, and Cu; Al-10Cr alloy; Al-10Mn alloy; Al-20Si alloy; and Al-5Ti-B alloy. The melt was directly poured at 720 °C into a water-cooled copper mold with dimensions of 140 mm × 50 mm × 140 mm, achieving a cooling rate of approximately 60 °C/min. The ingot was equally divided into two parts, designated as Sample 1 and Sample 2, which were subjected to different annealing treatments. Sample 1 was annealed at 530 °C for 12 h, while Sample 2 was annealed at 430 °C for 12 h.
The microstructure of the as-cast and annealed alloys was characterized. An Olympus GX53 optical microscope (OM, Tokyo, Japan) and a ZEISS Sigma 300 scanning electron microscope (SEM, Oberkochen, Germany) were used to observe and characterize the microstructure and second phase. Elemental characterization was performed using an Oxford Energy Dispersive Spectrometer (EDS, Abingdon, UK) attached to the SEM. The JEOL JEM-F200 transmission electron microscope (TEM, Tokyo, Japan) was used to investigate the morphology and distribution of nano-scale α-Al(MnCr)Si phases. The preparation of TEM foils involved mechanically grinding the samples to about 60 μm, and then conducting twinjet electropolishing in a solution consisting of 70 vol% methanol and 30 vol% nitric acid, with the temperature kept within the range of −20 to −30 °C.
3. Results
3.1. Microstructure of As-Cast Alloy
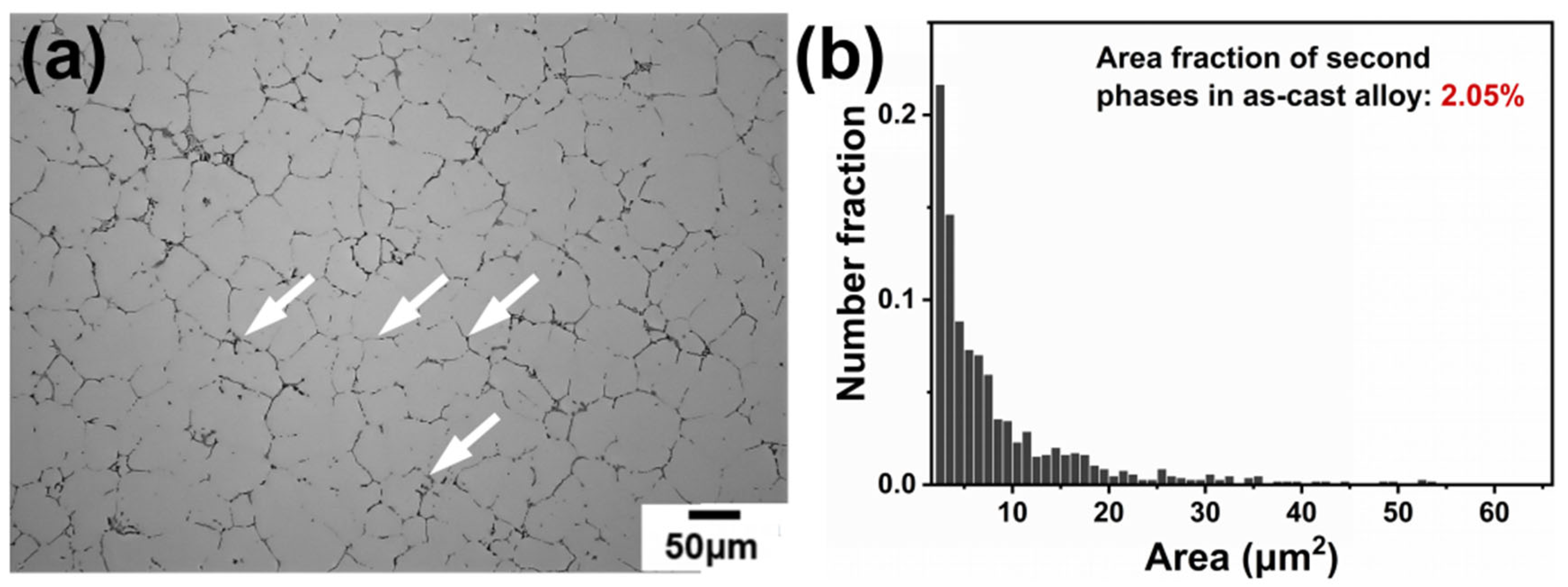
During the casting cooling process, elements exhibit distinct segregation behavior at dendrite arms or interdendritic regions due to differences in their segregation tendencies and diffusion capabilities. Figure 1 shows the optical microstructure of the as-cast alloy. As seen in Figure 1a, many micro-scale second phases are distributed mainly in the interdendritic regions. Based on color and morphology, the second phase can be divided into two types: black elongated phase and gray elongated phase (labeled by white arrows). The area fraction of these phases is 2.05%, as statistically determined using Image J (1.54p) software.
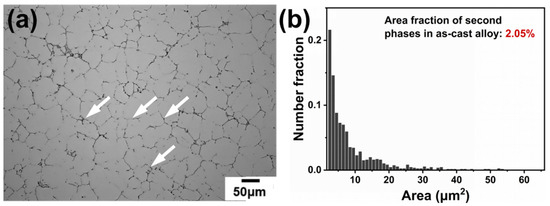
Figure 1.
OM image and area fraction of the second phase of the as-cast alloy: (a) OM image of the as-cast alloy and (b) area fraction of the second phase.
Figure 2 presents the scanning electron microscopy (SEM) microstructure and corresponding energy dispersive spectroscopy (EDS) analysis results of the as-cast alloy. The second phase was characterized using the EDS point analysis, and the results are summarized in Table 1. It is confirmed that the micro-scale second phase in as-cast alloy primarily consists of AlFeMnSi, Mg2Si, and AlCuMgSi phases, as well as eutectic Si phases. The EDS elemental mapping results (Figure 2b) reveal different segregation behavior of the elements. Specifically, Mg, Si, Cu, Mn, and Fe show obvious segregation at grain boundaries. Si, due to its low solid solubility, the high melting temperature relative to Al and relatively high diffusion rate, is less abundant in dendrite arms and predominantly concentrated in interdendritic regions. Cr, owing to its low content and the limited sensitivity of the EDS method used, exhibits slight segregation in the SEM elemental maps compared with other elements. Its segregation location correlates with that of Mn and Si, indicating that Cr is also present in some Fe-containing phases at grain boundaries.

Figure 2.
SEM image and EDS analysis results of the as-cast alloy: (a) SEM image; (b) EDS mapping results; and (c) the EDS line analysis results of Line 1 in (a).

Table 1.
EDS point analysis results of the second phase in the alloy (wt.%).
3.2. Microstructure of Annealed Alloy
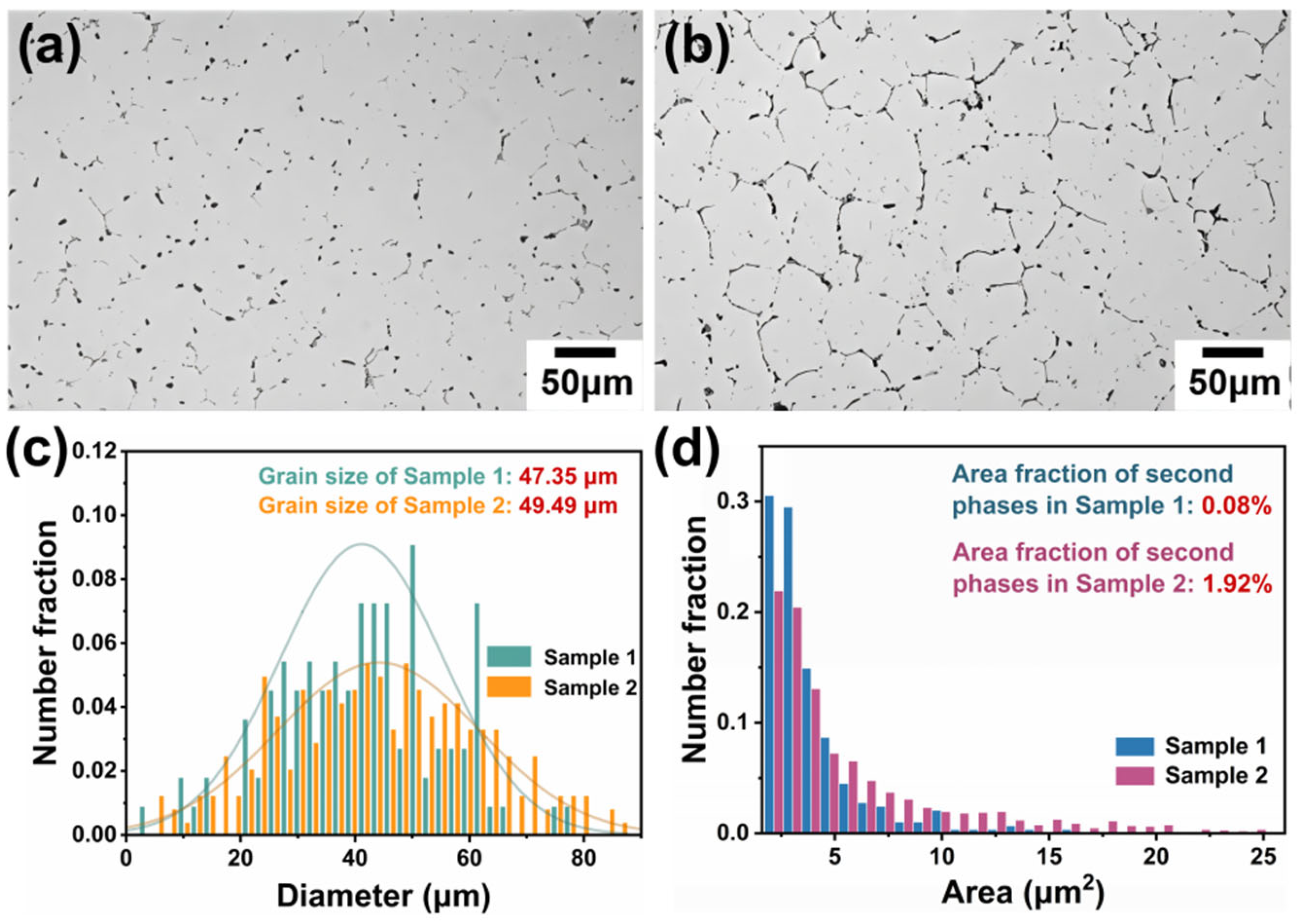
The as-cast alloy was subjected to annealing at different temperatures. The optical microstructure of the annealed Sample 1 (530 °C × 12 h) and Sample 2 (430 °C × 12 h) is shown in Figure 3. As observed in Figure 3a,b, the two alloys exhibited distinct differences in second phase after annealing, but the grain size of both alloys was similar. Statistical analysis using Image J software showed that the equivalent circular diameter of the grain in Sample 1 was 47.35 μm, while that in Sample 2 was 49.49 μm (Figure 3c).
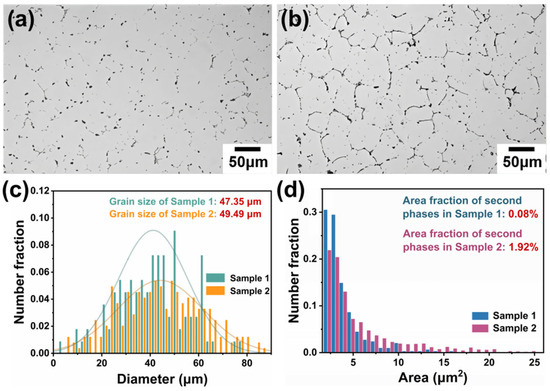
Figure 3.
OM images and corresponding statistical data of the two annealed alloys: (a) Sample 1; (b) Sample 2; (c) the grain size of the two alloys; and (d) the area fraction of the second phase in the two alloys.
Furthermore, the evolution of the second phase at grain boundaries differed significantly. In Sample 1, the second phase was finer, appearing as short rods or nearly spherical particles, which significantly indicated dissolution and transformation during annealing. This phenomenon indicates a significant reduction in element segregation. In contrast, the second phase in Sample 2 only slightly decreased in size, suggesting limited dissolution and transformation under the lower annealing temperature. The area fraction of the second phase in the two alloys, measured by Image J, was 0.08% and 1.92%, respectively (Figure 3d). The significant difference in the area fraction of the second phase also demonstrates the varying degrees of improvement in element segregation. In addition, analysis of the second phase area distribution reveals a greater fraction of small-sized second phase in Sample 1. This implies that annealing at 530 °C promotes more significant dissolution and/or transformation of the second phase, leading to a refined phase size.
The SEM and EDS characterization of the two annealed alloys is presented in Figure 4. Comparing Figure 2a with Figure 4a,c shows that the second phase along grain boundaries evolves differently depending on the annealing temperature. Annealing at 530 °C results in discontinuous, nearly spherical, and coarsened grain-boundary phases, whereas low-temperature annealing leads to only minor changes, with the phases largely preserving their irregular shape. This behavior is attributed to phase dissolution and transformation. The considerably higher equilibrium solid solubility of Mg2Si at elevated temperature promotes substantial dissolution during high-temperature annealing. High temperature supplies a substantial driving force and atomic mobility, which promotes the dissolution of the continuous grain-boundary second phase into the Al matrix. Dissolution starts at weak locations with high curvature (e.g., sharp edges and thin regions), breaking the continuous network into discrete, isolated particles. The change from an irregular to a near-spherical shape reduces the overall interfacial energy. Following this, Ostwald ripening causes smaller particles to dissolve while solute diffuses to and deposits on larger ones, leading to overall coarsening. At low temperature, the limited equilibrium solid solubility of Mg2Si results in only minor dissolution, with no obvious change in phase morphology or size.

Figure 4.
SEM images and EDS mapping results of the two annealed alloys: (a,b) Sample 1 and (c,d) Sample 2.
From the mapping comparison of the two alloys, high-temperature annealing improves the segregation of Mg and Si in Sample 1, weakening their aggregation in interdendritic zones and transforming their grain-boundary distribution from continuous to discrete. This is attributed to the significant dissolution of the Mg2Si phase during annealing. At higher temperatures, most Mg2Si dissolves, and the raised solid solubility of Mg and Si allows them to enter the Al matrix and diffuse inward. The mapping images in Figure 2b and Figure 4b clearly show that Mg and Si are more uniformly distributed within the grains after high-temperature annealing. After low-temperature annealing, however, dissolution of Mg2Si is limited by the lower solid solubility, resulting in fewer Mg and Si atoms entering the matrix and a comparatively lower intragranular concentration.
Cu segregation is substantially reduced after both annealing conditions, owing to the dissolution of AlCuMgSi phases in the as-cast alloy. Given that Cu bearing phases in 6xxx alloys often possess low dissolution temperatures, pronounced dissolution takes place during annealing, followed by Cu diffusion into the grain interiors. The limited diffusion coefficient at low annealing temperature restricts Cu mobility, leading to the lower intragranular Cu content shown in Figure 4d relative to Figure 4b.
Additionally, Fe segregation is alleviated after heat treatment. After 530 °C annealing, Fe accumulation at grain boundaries weakens noticeably, while its concentration within grains increases. This change is attributed to the transformation and/or dissolution of the AlFeMnSi phase, in which Mn and Cr atoms substitute for Fe atoms, resulting in the formation of fine bead-like α-Al(FeMnCr)Si phases and thus reducing Fe segregation. In contrast, after annealing at 430 °C, the segregation of Fe is only slightly improved, with noticeable aggregation still present at grain boundaries and a lower concentration within the grains. This is attributed to the limited transformation and/or dissolution of AlFeMnSi phases at the lower temperature.
High-temperature annealing plays a more pronounced role in reducing Mn segregation. This can be observed by comparing the distribution of Mn in Figure 4b,d, which is also related to the transformation of intermetallic compounds at grain boundaries. Moreover, the increased solid solubility at high temperature allows more Mn to dissolve into the Al matrix. During annealing, Mn gradually diffuses into the grain interiors, resulting in a more uniform intragranular distribution compared to that after low-temperature annealing.
The segregation of Cr is reduced after annealing, as shown in Figure 2b and Figure 4b,d. In the as-cast condition, Cr is bound in intermetallic compounds with Fe, Mn, and Si, which are predominantly distributed along grain boundaries. Annealing promotes partial transformation and refinement of the second phase, mitigating Cr segregation. Nevertheless, some Cr continues to concentrate at grain boundaries after both annealing conditions (Figure 4b,d), reflecting incomplete phase transformation. Additionally, the higher intragranular Cr content in Figure 4b indicates that more intermetallics transform at elevated temperature, enabling Cr to diffuse into the grain interiors.
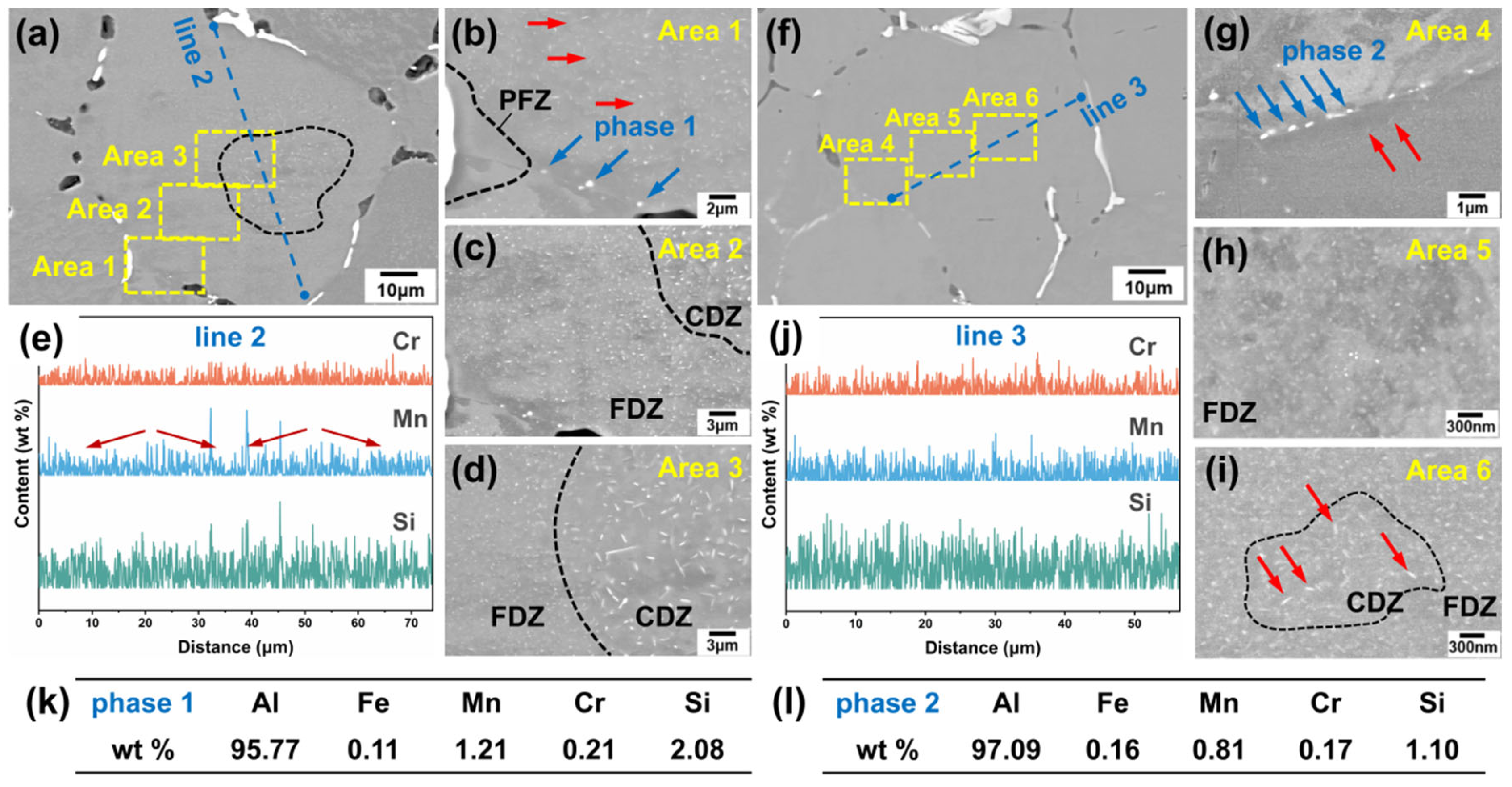
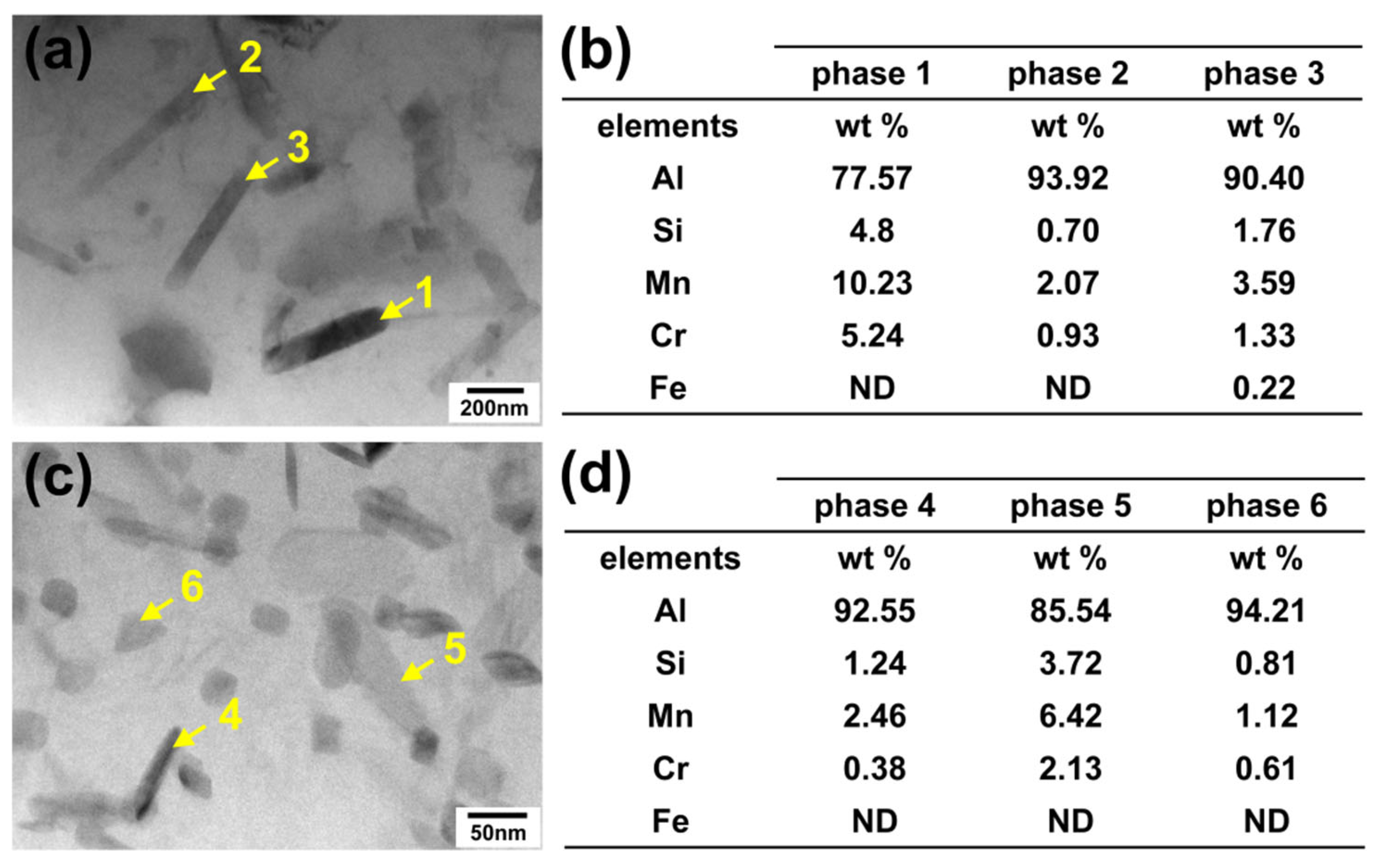
Additionally, different second phases were observed within the grains of both alloys, as marked by yellow and blue arrows in Figure 4a,c. Selected areas within the grain of the two alloys were examined at higher magnification to characterize the precipitated phases (Figure 5a–j). Areas 1, 2, and 3 correspond to enlarged views from different regions within a grain of Sample 1, while Areas 4, 5, and 6 represent similar positions in Sample 2. These locations respectively correspond to the areas near grain boundaries, between grain boundaries and grain centers, and grain centers in both alloys. Observation of Area 1 in Sample 1 (Figure 5b) reveals bead-like bright white precipitates near the grain boundary, as marked by phase 1. The EDS analysis (Figure 5k) confirms that these are α-Al(FeMnCr)Si phases. Additionally, a Precipitation Free Zone (PFZ) is observed near the grain boundary; this is a common microstructural feature in aluminum alloys resulting from the second phase precipitated at the grain boundaries.
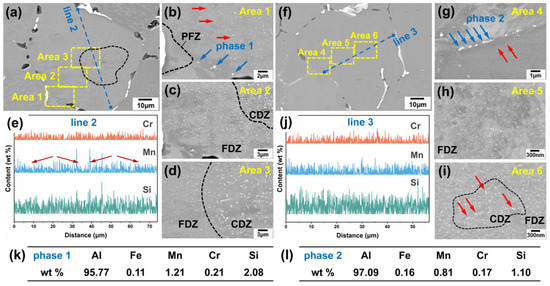
Figure 5.
SEM images, magnified images of different regions and EDS analysis results: (a–d) Sample 1, with (b–d) depicting the region near the grain boundary, between the grain boundary and the grain center, and the grain center, respectively. (e) The result of the EDS line scan at Line 2 in (a). (f–j) Sample 2, with (g–i) depicting the region near the grain boundary, between the grain boundary and the grain center, and the grain center, respectively. (j) The result of the EDS line scan at Line 3 in (b). (k,l) The results of the EDS point analysis of phase 1 in (b) and phase 2 in (g), respectively.
Moreover, a fine white second phase was observed in all three magnified areas in Sample 1. This phase exhibits a high number density and a dispersed distribution, with sizes ranging from several hundred nanometers to a few micrometers, showing a distinct regional distribution characteristic. In the grain center (Area 3), the dispersed phase is the largest (a few micrometers), having the highest aspect ratio, and the lowest number density. This region is designated as the coarse dispersion zone (CDZ), as labeled by the black line in Figure 5d. Far away from the grain center (Area 2), the size of the dispersed phases obviously reduced to several hundred nanometers. The aspect ratio decreases, and the morphology exhibits a blocky appearance. The number density of the dispersed phases in this region has increased significantly. This region is designated as the fine dispersion zone (FDZ), as labeled in Figure 5c,d. It is noteworthy that a small amount of slightly enlarged dispersed phases (labeled by red arrows in Figure 5b) appears near the grain boundaries at the edge of PFZ (Area 1).
Similar regions within the grains of Sample 2 were examined at higher magnification, as shown in Figure 5f–i. A dispersed second phase is also observed in Sample 2. Compared with Sample 1, the phase is obviously finer, on the scale of tens of nanometers, and exhibits no significant size gradient, though it shares similar regional distribution characteristics. In the grain center (Area 6), a zone containing needle-shaped dispersed phases exists, as labeled by red arrows in Figure 5i. However, in Sample 2, this phase is finer and has a lower aspect ratio and a higher number density compared to Sample 1. The CDZ shows a smaller area. Far away from the grain center (Area 5), dispersed phases were reduced to a smaller size and increased the number density. Similarly, near the grain boundary, a slightly larger needle-like phase was observed. A bead-like bright white second phase exists near grain boundaries (labeled by red arrows in Figure 5g, with the EDS analysis results confirming them to be α-Al(FeMnCr)Si phases (Figure 5l). Additionally, relatively large rod-shaped precipitates are observed within the grains of Sample 2, similar to the findings reported by Jin D. et al. [16], which can be identified as Mg2Si phases. As this phase is not the focus of the present work, it will not be discussed further.
The EDS line scans were performed across the grains of both alloys, with the results shown in Figure 5e,j corresponding to Line 2 in Sample 1 and Line 3 in Sample 2, respectively. After annealing, the elemental concentration gradients between the grain boundaries and grain centers were significantly reduced, particularly for Si, whose segregation was markedly improved. In Figure 5e, several strong peaks are observed in the line scan results for Mn and Si. The distribution pattern of Mn content (labelled by red arrows) appears to relate to the size distribution of the dispersed phase. From the grain centre towards the grain boundary, it exhibits an initial increase followed by a decrease. In comparison to Figure 5a, it indicates that these peaks correspond to the areas containing needle-like precipitates in the grain center. This phenomenon was less pronounced in Sample 2, possibly because the α-Al(MnCr)Si particles were too small to be fully captured by the electron beam spot size. The results of EDS line analysis confirmed the regional distribution characteristics of the α-Al(MnCr)Si phases.
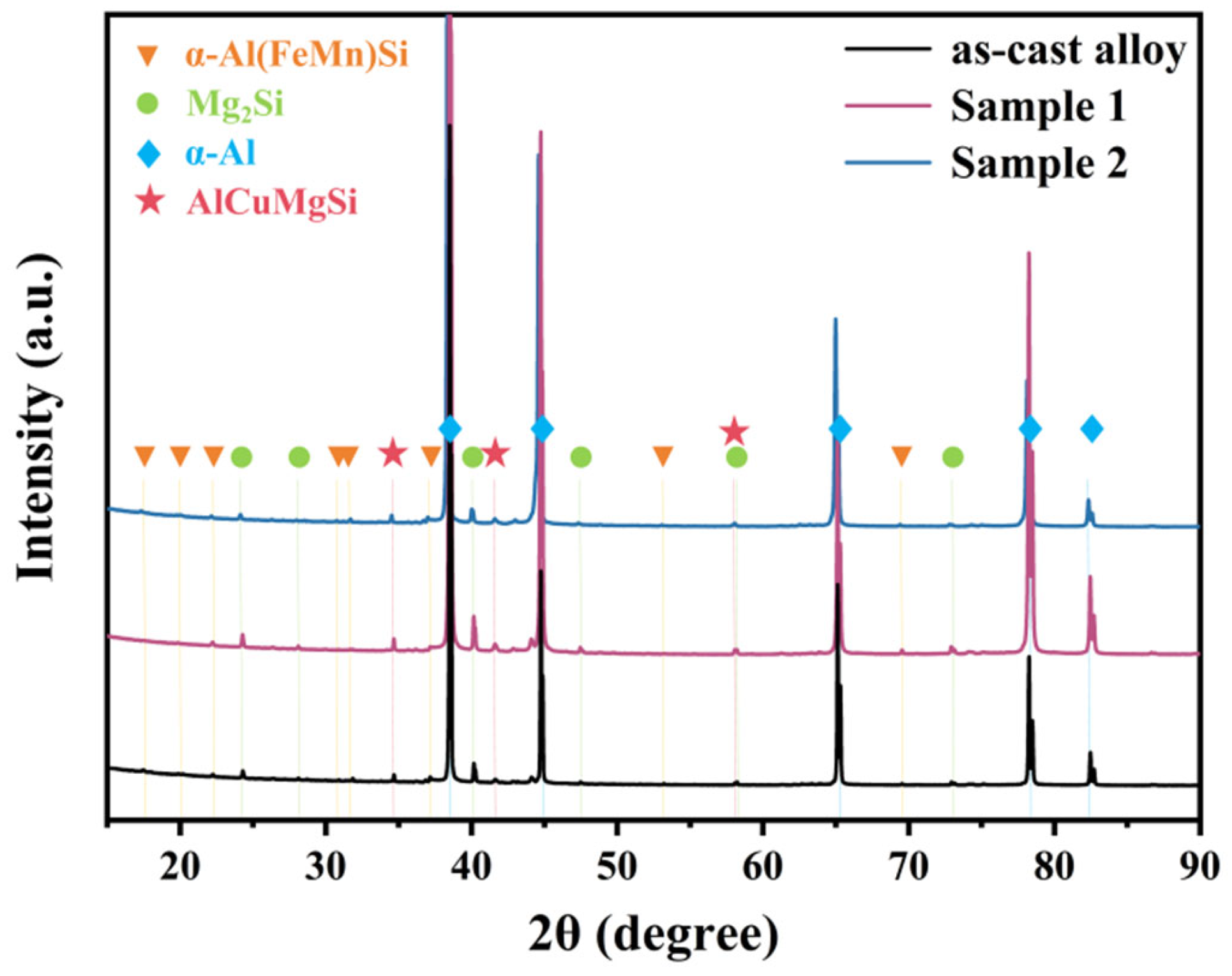
XRD characterization was performed on the alloys before and after annealing, and the results are shown in Figure 6. The results indicate that the as-cast alloy contains α-Al(FeMn)Si, Mg2Si, and AlCuMgSi phases, which are consistent with the EDS point analysis results in Table 1. Since no second phases exist within the grains of the as-cast alloy, the α-Al(FeMn)Si phase detected by XRD should correspond to the Fe-bearing phases located at grain boundaries. After annealing at both temperatures, the types of second phase in the alloy remain unchanged. Due to the low Mn content added in the alloy and the partial consumption of Mn in the formation of grain boundary second phase, the total amount of α-Al(FeMn)Si phase precipitated within the grains during annealing is relatively small, resulting in no significant changes in the XRD peak profiles. After annealing at 530 °C, the peak intensities in the XRD pattern increase, which may be attributed to the significantly improved crystallinity of the α-Al matrix and the equilibration and coarsening of the second phase.
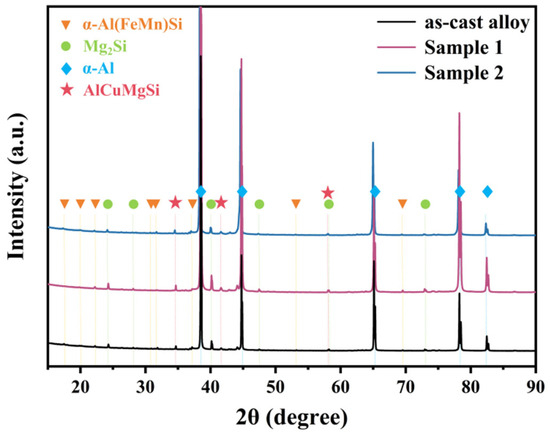
Figure 6.
XRD patterns before and after annealing.
To further identify the composition of the dispersed phases, the TEM characterization combined with an EDS analysis was conducted, as presented in Figure 7. These dispersed phases were confirmed to contain Al, Mn, Si, and Cr. Based on their morphology, distribution characteristics, and previous studies [6], they were identified as α-Al(MnCr)Si and/or α-Al(FeMnCr)Si phases in 6xxx series aluminum alloys. These α-Al(MnCr)Si phases typically precipitate within grains during annealing. Their excellent high-temperature stability is crucial for the alloy’s elevated temperature performance and recrystallization resistance, making them vital precipitates in 6xxx aluminum alloys.
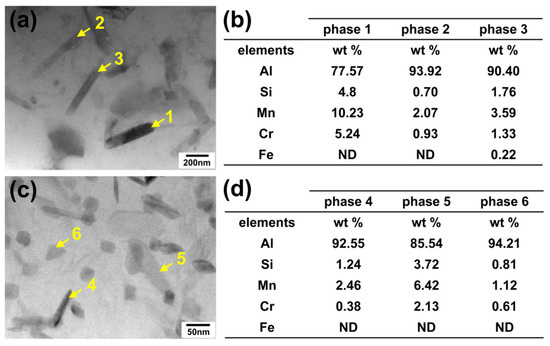
Figure 7.
TEM images and EDS analysis results of intragranular dispersed phases. (a,b) Sample 1; (c,d) Sample 2.
Based on the characterization results presented above, it can be concluded that annealing at different temperatures exerts distinct effects on both the dissolution/transformation of the second phase at grain boundaries and the precipitation of intragranular dispersoids. High-temperature annealing leads to more complete dissolution and transformation of the grain-boundary second phase; however, the resulting dispersoids are larger in size and exhibit a lower number density. Conversely, during low-temperature annealing, the dissolution and transformation of the second phase at grain boundaries are limited, while the intragranular dispersoids are finer and possess a higher number density. Since the elements involved in the formation and growth of dispersoids (e.g., Fe, Mn, Cr, and Si) are also the same elements that constitute and participate in the transformation of the grain-boundary second phase, the dissolution and transformation behavior of the second phase during annealing may alter the elemental concentrations in the alloy matrix. This, in turn, indirectly influences the precipitation characteristics of the intragranular dispersoids.
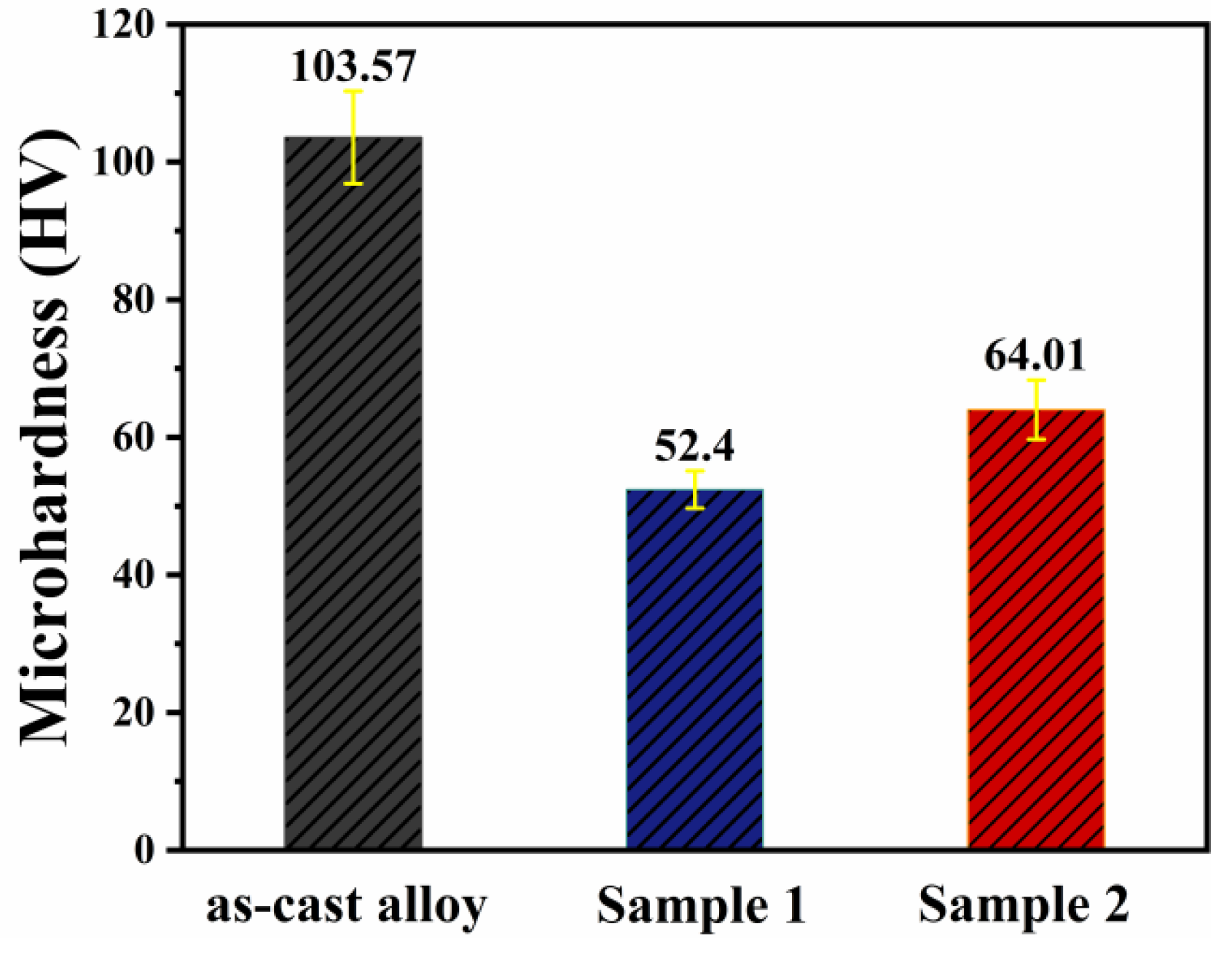
Figure 8 shows the hardness data before and after annealing. The results indicate that the hardness of the as-cast alloy decreases significantly after annealing at both temperatures, which may be attributed to a reduction in dislocation density and the release of internal stresses. After annealing at 430 °C for 12 h, Sample 2 exhibits higher hardness. This is likely due to the combined effect of a larger number of micro-sized phases at grain boundaries and finer dispersoids within the grains. Compared to the aluminum matrix, the micron-sized second phase at grain boundaries generally possesses higher hardness. When the material is subjected to external loading, these hard phases can directly bear part of the load and share the stress in the matrix, thereby enhancing the overall macro-scale resistance to indentation and deformation. Meanwhile, the nanoscale dispersoids inside the grains, with their finer size and higher number density, can effectively hinder dislocation motion during deformation, leading to the higher hardness observed in Sample 2.
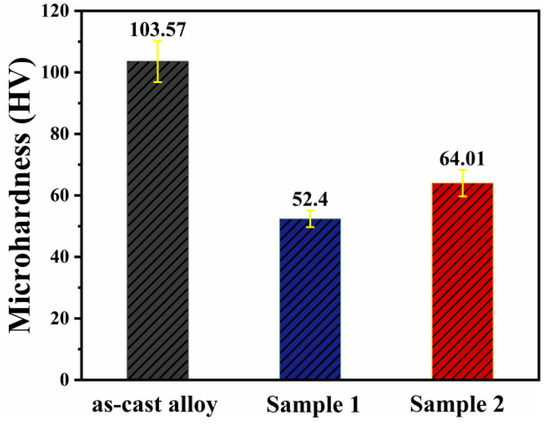
Figure 8.
Microhardness test results before and after annealing.
4. Discussion
4.1. Element Segregation Behavior of As-Cast Alloy
In the as-cast alloy, the elements exhibit distinct segregation behaviors, primarily influenced by cooling conditions and elemental diffusion characteristics. The average partition coefficients(k) of Mg, Si, Cu, Fe, and Mn are all less than one, causing them to be rejected at the solid-liquid interface during solidification and ultimately enriched in the interdendritic regions. Within these regions, atoms combine to form compounds distributed along grain boundaries, resulting in the observation of numerous micro-scale second phases in the as-cast microstructure [16]. Mg and Si atoms combine to form Mg2Si phases. In addition, due to its relatively high diffusion rate, Si shows a pronounced concentration gradient within the grains, increasing gradually from the grain center toward the grain boundaries. Cu, with its low average partition coefficient, combines with elements, such as Mg and Si, at grain boundaries to form AlCuMgSi phases [17]. Fe, an impurity element in 6xxx Al alloys, although present in low content, exhibits marked segregation in interdendritic regions owing to its extremely low solubility and very low average partition coefficient, where it combines with Mn to form AlFeMnSi intermetallic compounds. Due to the slow diffusion rate of Mn [18], aside from the formation of AlFeMnSi phases, no pronounced inhomogeneous distribution within the grain is observed. Cr possesses an average partition coefficient greater than one, causing it to preferentially segregate to the initially solidified dendrite arms during solidification. Under relatively rapid cooling rates and its inherently slow diffusion rate, the low-content Cr atoms do not display significant segregation and mostly remain in solid solution within the Al matrix.
In addition to the second phase mentioned above, the relatively high cooling rate during casting leads to elements not participating in phase formation to dissolve in the AI matrix, forming a supersaturated solid solution. This provides a driving force for phase precipitation during subsequent annealing. The faster cooling rate in the water-cooled copper mold allows a greater proportion of alloying elements to be retained in the supersaturated solid solution at higher concentrations, thereby offering stronger precipitation driving force and more sufficient elemental reserves for the formation of dispersed phases [19]. Thus, the segregation behavior of elements in the as-cast alloy is governed by the casting cooling rate, average partition coefficients, elemental contents, and atomic diffusion rates. Under the combined influence of these factors, the as-cast microstructure of the Al–Mg–Si–Mn alloy is characterized by an interdendritic second phase, consisting mainly of Mg2Si, AlCuMgSi, and AlFeMnSi phases, while within the grains, apart from Si, no significant segregation of other elements is observed.
4.2. Transformation and Precipitation Mechanisms of the Second Phase in Annealed Alloys
In this work, two annealing treatments with significantly different temperatures were employed, resulting in markedly distinct behavior in element diffusion and the nucleation and growth of the second phase. In Sample 1, the area fraction of the second phase at the grain boundary was obviously transformed and dissolved. Concurrently, micro- and submicro-scale α-Al(MnCr)Si phases precipitated within the grains, exhibiting a clear region distribution characteristic. These dispersed phases displayed a distinct size gradient, decreasing first and then increasing from the grain boundaries toward the grain center. In contrast, only a small fraction of the second phase at grain boundaries transformed and dissolved in Sample 2, while α-Al(MnCr)Si phases with similar distribution characteristics precipitated within the grains. These α-Al(MnCr)Si phases were finer (nano-scale), had a higher number density, showed no significant size gradient, and exhibited a lower aspect ratio.
The precipitation behavior and regional distribution characteristics of intragranular α-Al(MnCr)Si phases are also closely related to elemental distribution and diffusion. During annealing, the characteristic diffusion distance x (i.e., the mean diffusion depth) of atoms can be estimated by Fick’s second law [20]:
where D represents the diffusion coefficient, D0 represents the frequency factor (m2/s), t represents the time (s), Q represents the activation energy for diffusion (J/mol), R represents the gas constant (8.314 J/(mol·K)), and T represents the absolute temperature (K). The calculated diffusion distances for each element after 12 h of annealing at 430 °C and 530 °C are listed in Table 2.

Table 2.
Characteristic diffusion distances of elements (μm).
The data in Table 2 indicate that the diffusion distances of these elements increase significantly at higher temperatures, exhibiting a clear difference compared to those at lower temperatures. Combined with the grain size statistics from Section 3.2, it can be observed that at both annealing temperatures, the diffusion distances of Mg and Si are sufficient to fully cover the grain radius (23.68 μm for Sample 1 and 24.75 μm for Sample 2). Therefore, under high-temperature annealing, these two elements can diffuse throughout the entire volume of the grains, achieving a distribution state that approaches equilibrium. Under these conditions, the Mg–Si phase forms and continues to grow until the equilibrium solid solubility of both elements is reached. For the elements Mn, Cr and Si, which participate in the precipitation of α-Al(MnCr)Si phases, their diffusion distance are strongly influenced by temperature. However, the elements Cr and Mn are the slower diffusion elements and therefore become likely the rate-limiting species for α-phase precipitation. Under low-temperature annealing, Cr exhibits a very short diffusion distance (0.71 μm after 12 h) and is therefore not considered to significantly influence dispersoid formation and growth.
The regional distribution characteristics of the α-Al(MnCr)Si phases within grains are likely influenced by Si elemental distribution. Studies have shown that Mg–Si particles are preferred nucleation sites for α-Al(MnCr)Si phases, whose precipitation is accompanied by the dissolution of Mg–Si phases. They precipitate within “Si-rich regions” left by the dissolving Mg–Si particles [2,21,22]. Thus, the distribution of Si determines the nucleation sites of α-Al(MnCr)Si phases, thereby influencing their uniformity. During annealing, Mg-Si phases begin to precipitate around 275 °C and dissolve around 375 °C [22]. Given the heating rate of 5 °C/min used in this study, the time reached for Mg–Si phase precipitation and dissolution was relatively short. This period proved insufficient to achieve complete diffusion of the Si. This demonstrates that during the precipitation of the α-Al(MnCr)Si phases, the distribution of Si atoms within the grains is not uniform. This directly influences nucleation characteristics of the α-Al(MnCr)Si phases, with fewer nucleation sites present in regions of low Si concentration and a greater number of nucleation sites occurring in areas of high Si concentration. Therefore, during high-temperature annealing, the number density of α-Al(MnCr)Si phase precipitates is lower at the grain centers and higher toward the grain boundaries.
During high-temperature annealing, as Si diffusion reaches the grain center, the α-Al(MnCr)Si phases exhibit severe needle-like growth due to the scarcity of nucleation sites and low number density, exacerbated by increased Si concentration. Conversely, the α-Al(MnCr)Si phases at the grain center during low-temperature annealing show no significant coarsening, owing to the abundance of nucleation sites and high number density. Therefore, the initial distribution of Si dictates the uniformity of α-Al(MnCr)Si phases distribution, with regions of higher Si content promoting a higher number density of α-Al(MnCr)Si phases.
The reason for the significant difference in the size of α-Al(MnCr)Si phases under different temperatures is likely related to the diffusion of Mn. Research by C. Li et al. [23] indicates that the influence of heat treatment temperature on dispersed phases can be attributed to its effect on the diffusion rate of Mn. At relatively low temperatures, Mn diffusion is confined to short distances, thereby limiting phase growth. At higher temperatures, however, Mn solute can undergo long-range diffusion, facilitating the growth and coarsening of dispersed phases. According to the element diffusion calculation results, the Si element has achieved complete diffusion and is uniformly distributed throughout the matrix, whereas the Mn element has not diffused sufficiently, resulting in an uneven concentration within the matrix under different annealing temperatures. The reasons for the distinct differences in the size of the α-Al(MnCr)Si phases at different temperatures can be summarized as follows:
Under high-temperature annealing, the diffusion coefficient of Mn increases exponentially, leading to its diffusion over further distances within grains. This results in higher Mn concentrations compared to low-temperature annealing, thereby promoting the growth of α-Al(MnCr)Si phases. Conversely, the lower Mn concentrations within grains at low temperatures contribute to the finer grain size of α-Al(MnCr)Si phases. As the temperature rises, the diffusion rate and distance of atoms increase, thereby promoting the growth of α-Al(MnCr)Si phases. The higher homogenization temperature promotes particle coarse and growth of α-Al(MnCr)Si phases [24]. Ostwald ripening is more likely to occur, wherein smaller particles (with smaller curvature radii) dissolve more readily than larger ones, and atoms diffuse through the matrix to deposit onto the more stable larger particles. This leads to an increased average size and a reduced number density of the dispersed phases at elevated temperatures.
As a typical element with a negative segregation tendency, Cr segregates to the dendrite arms during solidification and cooling. Although it can participate in the precipitation of α-phase and inhibit its growth, the present work shows that even with a significantly longer diffusion distance under high-temperature annealing compared to the low-temperature condition, Cr does not appear to effectively suppress the growth and coarsening of dispersoids at the grain center.
These analysis results indicate that the regional distribution and precipitation characteristics of intragranular α-Al(MnCr)Si phases under different annealing temperatures are collectively determined by the distribution and diffusion behavior of Mn and Si during the casting cooling process and subsequent annealing treatment. Low-temperature annealing precipitates α-Al(MnCr)Si phases with a smaller size and a higher number density within grains.
5. Conclusions
In this study, annealing at different temperatures were applied to the Al–Mg–Si–Mn alloy, achieving different precipitation characteristics of the intragranular α-Al(MnCr)Si phases. The main conclusions are as follows:
- (1)
- At both annealing temperatures, complete diffusion of Mg and Si elements was achieved, whereas the diffusion distance of Mn at the higher temperature was markedly longer than at the lower temperature.
- (2)
- The distribution and coarsening of α-Al(MnCr)Si phases are temperature-dependent. High-temperature annealing promotes non-uniform coarsening, especially at grain centers due to enhanced Si diffusion, whereas low-temperature annealing yields a more uniform phase size. Both nucleation density and number density are controlled by the local Si concentration and annealing temperature.
- (3)
- High-temperature annealing drives pronounced coarsening of α-Al(MnCr)Si phases compared to low-temperature treatment, primarily due to the higher intragranular Mn concentration, accelerated diffusion, and Ostwald ripening.
- (4)
- Low-temperature annealing results in more favorable precipitation characteristics of intragranular α-Al(MnCr)Si phases: a smaller phase size, a higher number density, and a uniform distribution.
Author Contributions
Conceptualization, Y.C. and C.B.; Methodology, Y.C., Q.L. and C.B.; Software, Y.C., H.J., Y.Z. and Y.W.; Validation, Y.C., H.J., H.L., Z.W., X.L., Q.L., Y.Z. and Z.L.; Formal analysis, Y.C., H.J., Z.W., Z.L. and C.B.; Investigation, Y.C., H.J., H.L., Z.W., X.L., Q.L., Y.Z., Z.L. and Y.W.; Resources, Y.C., H.J. and C.B.; Data curation, Y.C.; Writing—original draft, Y.C.; Writing—review & editing, C.B.; Visualization, H.L., Y.W. and C.B.; Supervision, C.B.; Project administration, C.B.; Funding acquisition, C.B. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the National Key Research and Development Program of China (No. 2023YFB3710900).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kumar, S.; Grant, P.S.; O’Reilly, K.A.Q. Evolution of Fe Bearing Intermetallics During DC Casting and Homogenization of an Al-Mg-Si Al Alloy. Metall. Mater. Trans. A 2016, 47, 3000–3014. [Google Scholar] [CrossRef]
- Bayat, N.; Carlberg, T.; Cieslar, M. In-Situ Study of Phase Transformations during Homogenization of 6005 and 6082 Al Alloys. J. Alloys Compd. 2017, 725, 504–509. [Google Scholar] [CrossRef]
- Gao, T.; Wu, Y.; Li, C.; Liu, X. Morphologies and Growth Mechanisms of α-Al(FeMn)Si in Al–Si–Fe–Mn Alloy. Mater. Lett. 2013, 110, 191–194. [Google Scholar] [CrossRef]
- Liu, K.; Chen, X.-G. Development of Al–Mn–Mg 3004 Alloy for Applications at Elevated Temperature via Dispersoid Strengthening. Mater. Des. 2015, 84, 340–350. [Google Scholar] [CrossRef]
- Lu, Q.; Li, K.; Chen, H.; Yang, M.; Lan, X.; Yang, T.; Liu, S.; Song, M.; Cao, L.; Du, Y. Simultaneously Enhanced Strength and Ductility of 6xxx Al Alloys via Manipulating Meso-Scale and Nano-Scale Structures Guided with Phase Equilibrium. J. Mater. Sci. Technol. 2020, 41, 139–148. [Google Scholar] [CrossRef]
- Lodgaard, L.; Ryum, N. Precipitation of Dispersoids Containing Mn and/or Cr in Al–Mg–Si Alloys. Mater. Sci. Eng. A 2000, 283, 144–152. [Google Scholar] [CrossRef]
- Khani Moghanaki, S.; Kazeminezhad, M.; Logé, R. Effect of Concurrent Precipitation on the Texture Evolution during Continuous Heating of Multi Directionally Forged Solution Treated Al-Cu-Mg Alloy. Mater. Charact. 2017, 131, 399–405. [Google Scholar] [CrossRef]
- Li, Y.J.; Muggerud, A.M.F.; Olsen, A.; Furu, T. Precipitation of Partially Coherent α-Al(Mn,Fe)Si Dispersoids and Their Strengthening Effect in AA 3003 Alloy. Acta Mater. 2012, 60, 1004–1014. [Google Scholar] [CrossRef]
- Liu, F.; Zhu, X.; Qin, J.; Zhou, W.; Ling, J.; Dong, Q.; Yu, J.; Nagaumi, H.; Zhang, B. Effect of Mn/Cr Ratio on Precipitation Behaviors of α-Al(FeMnCr)Si Dispersoids and Mechanical Properties of Al–Mg–Si–Cu Alloys. Mater. Sci. Eng. A 2022, 860, 144269. [Google Scholar] [CrossRef]
- Ånes, H.W.; van Helvoort, A.T.J.; Marthinsen, K. Orientation Dependent Pinning of (Sub)Grains by Dispersoids during Recovery and Recrystallization in an Al–Mn Alloy. Acta Mater. 2023, 248, 118761. [Google Scholar] [CrossRef]
- Nagaumi, H.; Qin, J.; Yu, C.; Wang, X.; Wang, L. Quantitative Analysis of Influence of α-Al(MnFeCr)Si Dispersoids on Hot Deformation and Microstructural Evolution of Al-Mg-Si Alloys. Trans. Nonferrous Met. Soc. China 2022, 32, 1805–1821. [Google Scholar] [CrossRef]
- Li, X.; Dong, Q.; Hu, S.; Wang, X.; Yao, Z.; Wei, Z.; Li, Q.; Nagaumi, H. Enhanced Dispersoids Precipitation, Recrystallization Resistance and Mechanical Properties of Al-Mg-Si-Cu-Mn Alloy via Mo Addition. J. Mater. Sci. Technol. 2025, 231, 296–307. [Google Scholar] [CrossRef]
- Qian, X.; Parson, N.; Chen, X.-G. Effects of Mn Addition and Related Mn-Containing Dispersoids on the Hot Deformation Behavior of 6082 Aluminum Alloys. Mater. Sci. Eng. A 2019, 764, 138253. [Google Scholar] [CrossRef]
- Liu, X.; Jia, H.-L.; Wang, C.; Wu, X.; Zha, M.; Wang, H.-Y. Enhancing Mechanical Properties of Twin-Roll Cast Al–Mg–Si–Fe Alloys by Regulating Fe-Bearing Phases and Macro-Segregation. Mater. Sci. Eng. A 2022, 831, 142256. [Google Scholar] [CrossRef]
- Ding, L.; Zhang, X.; Lu, C.; Wang, C.; Weng, Y.; Dong, Q.; Xu, S.; Cao, L.; Jia, Z. Influence of the Combined Additions Mn, Cr or Ni on the Formation of Dispersoids and Mechanical Properties of Al–Mg–Si–Cu Alloys. Mater. Sci. Eng. A 2024, 892, 145962. [Google Scholar] [CrossRef]
- Jin, D.; Li, H.; Zhu, Z.; Yang, C.; Chen, B.; Miao, Y. Effects of Cooling Rate in Homogenization on Microstructure, Hot Deformation Resistance and Subsequent Age-Hardening Behavior of an Al–Mg–Si Alloy. J. Mater. Res. Technol. 2023, 27, 3283–3300. [Google Scholar] [CrossRef]
- Li, Z.; Qin, J.; Zhang, H.; Wang, X.; Zhang, B.; Nagaumi, H. Improved Distribution and Uniformity of α-Al(Mn,Cr)Si Dispersoids in Al-Mg-Si-Cu-Mn (6xxx) Alloys by Two-Step Homogenization. Metall. Mater. Trans. A 2021, 52, 3204–3220. [Google Scholar] [CrossRef]
- Knipling, K.E.; Dunand, D.C.; Seidman, D.N. Criteria for Developing Castable, Creep-Resistant Aluminum-Based Alloys—A Review. Int. J. Mater. Res. 2006, 97, 246–265. [Google Scholar] [CrossRef]
- Li, S.; He, C.; Fu, J.; Xu, J.; Xu, G.; Wang, Z. Evolution of Microstructure and Properties of Novel Aluminum-lithium Alloy with Different Roll Casting Process Parameters during Twin-Roll Casting. Mater. Charact. 2020, 161, 110145. [Google Scholar] [CrossRef]
- Whelan, M.J. On the Kinetics of Precipitate Dissolution. Metal Sci. J. 1969, 3, 95–97. [Google Scholar] [CrossRef]
- Hu, R.; Ogura, T.; Tezuka, H.; Sato, T.; Liu, Q. Dispersoid Formation and Recrystallization Behavior in an Al-Mg-Si-Mn Alloy. J. Mater. Sci. Technol. 2010, 26, 237–243. [Google Scholar] [CrossRef]
- Qian, X.; Wang, Z.; Li, Y.; Wang, Y.; Peng, Y. Formation Mechanism of β”-Mg5Si6 and Its PFZ in an Al-Mg-Si-Mn Alloy: Experiment and First-Principles Calculations. Mater. Charact. 2023, 197, 112617. [Google Scholar] [CrossRef]
- Li, C.; Liu, K.; Chen, X.-G. Improvement of Elevated-Temperature Strength and Recrystallization Resistance via Mn-Containing Dispersoid Strengthening in Al-Mg-Si 6082 Alloys. J. Mater. Sci. Technol. 2020, 39, 135–143. [Google Scholar] [CrossRef]
- Zajac, S.; Hutchinson, B.; Johansson, A.; Gullman, L.-O. Microstructure Control and Extrudability of Al–Mg–Si Alloys Microalloyed with Manganese. Mater. Sci. Technol. 1994, 10, 323–333. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.