Simple Summary
Many spider species show large differences in size between males and females, but biologists still do not fully understand how or why this evolves. In this study, we focused on the systematics and evolution of a little-known African spider, Megaraneus gabonensis, in which females are about four times larger than males. We provided an overdue taxonomic description of the species and examined where the genus Megaraneus fits in the spider tree of life. Our findings show that this spider is a part of a clade that has previously been thought to live only in East Asia and Australia, but we now know it also occurs in Africa. We also found that in Megaraneus, the large size difference between the sexes resulted primarily because females became larger, not because males would become smaller. This research helps us better understand how spider body size evolves differently in males and females and highlights the importance of studying lesser-known species to fill in knowledge gaps about the natural world.
Abstract
Among terrestrial animals, spiders exhibit the most striking examples of sexual size dimorphism (SSD) but better understanding of its evolution requires improved taxonomy and phylogeny. Many sexually dimorphic spiders lack adequate description, phylogenetic placement, and natural history observations. In South Africa, we documented the natural history of a poorly known spider, Megaraneus gabonensis (Lucas, 1858), with extreme, female-biased SSD (eSSD, female:male approximately 4:1). Here, we redescribe M. gabonensis, place Megaraneus Lawrence, 1968 phylogenetically for the first time, assess whether the observed eSSD represents an independent evolutionary origin, and test whether the macroevolutionary pattern is better explained by male dwarfism or female gigantism. The recovered phylogenetic placement of Megaraneus in the araneid ‘backobourkiines’, a clade previously considered as restricted to East Asia and Australasia, extends the range of this clade to the Afrotropics. We find that eSSD was present in the common ancestor of the ‘backobourkiines’, with further increases in female body length occurring independently in Megaraneus, Backobourkia Framenau, Dupérré, Blackledge & Vink, 2010, and the currently misplaced Parawixia dehaani (Doleschall, 1859). We conclude that the evolution of eSSD reflects a complex pattern of sex-specific size changes across spider phylogeny, but that in Megaraneus it results from female gigantism.
1. Introduction
Spiders exhibit some of the most striking examples of female-biased sexual size dimorphism (SSD) in the animal kingdom [1,2,3,4,5,6]. In some orb-weaving species, females are more than twice the linear size of males, a pattern termed extreme sexual size dimorphism (eSSD) [1]. Phylogenetic studies have inferred between four [4] and nine [7] independent evolutionary origins of eSSD in araneoid spiders. Early phylogenetic research on male and female evolutionary size changes focused on whether eSSD in particular clades is better attributable to patterns of “male dwarfism” versus “female gigantism” [4,8]. Recent studies instead suggest that sex-specific size evolution is often more complex and lacks a universal pattern across clades [9].
Despite recent advances, the selective forces driving SSD in spiders remain incompletely understood. Several hypotheses have been proposed, emphasizing different interactions of natural and sexual selection, ecological pressures, and sexual conflict (reviewed in [1]). Female gigantism is associated with fecundity selection whereby larger body size allows for greater egg production and increased reproductive success [10,11,12]. In contrast, small male size may result from selection for increased agility, more efficient mate searching, and improved predator avoidance [2,13,14,15]. To explain the evolution of SSD in spiders, Kuntner & Coddington [1] built on previous work [2,3,5] to propose a differential equilibrium model [16], where reinforcing and opposing selection pressures act together and where the direction and magnitude of SSD vary across lineages.
A major obstacle to understanding SSD evolution across spiders is incomplete taxonomic sampling and poor understanding of intra- and interspecific size variation. Many species are known only from one sex and are described based on minimal morphological data, often without intraspecific variation or ecological context [17]. For example, some of the best-known examples of eSSD in spiders are found in the family Araneidae [1], whose significant portion of species diversity remains underexplored, particularly in the tropics. Continued field research in poorly studied regions is essential for discovering new species, documenting SSD, and refining phylogenetic frameworks that underpin evolutionary inference.
In South Africa, we documented the natural history of Megaraneus gabonensis (Lucas, 1858), a conspicuous (Figure 1) but poorly known araneid. Its eSSD is particularly striking, with males only about a fourth the body length of females (Figure 1A). This eSSD case has remained elusive as over 100 years passed since the description of the species to the discovery of its male. When Lawrence [18] described the male, he noted the dimorphism in “[t]he genus, with regard to the discrepancy of form and size in the sexes, agrees more with Argyope, Cyrtophora, Caerostris and Gasteracantha than with Araneus…” (p. 110) and “[t]here is a much greater discrepancy of size in the male and female than in the case of Caerostris…” (p. 114). Further males have never been reported, and the genus and species lack recent taxonomic treatment. In part due to the convoluted taxonomic history of M. gabonensis—originally described as Epeira, and transferred to Cyrtophora Simon, 1864 and Caerostris Thorell, 1868—the genus has never been placed phylogenetically.

Figure 1.
Megaraneus gabonensis (Lucas, 1858) from South Africa. (A) Male (above) and female (below) resting in a web retreat. (B) Color variation in female abdomens. (C) Female web. Photos by Matjaž Kuntner.
We here provide these missing links for a more comprehensive understanding of araneid SSD evolution by redescribing M. gabonensis based on both sexes, reconstructing its phylogenetic relations to other araneids, and investigating the evolutionary origins of its eSSD. Specifically, we ask: (i) Where does Megaraneus fit within the broader araneid phylogeny? (ii) Does eSSD in Megaraneus represent an independent origin, or is it part of a shared evolutionary trajectory with other eSSD araneid lineages? (iii) Can we attribute the observed eSSD in Megaraneus to female gigantism, male dwarfism, or a more complex interplay of sex-specific selection pressures?
2. Materials and Methods
2.1. Fieldwork
Two adult male and twelve adult female specimens of Megaraneus gabonensis were collected in 2019 at multiple sites within iSimangaliso Wetland Park, KwaZulu-Natal Province, South Africa (see Appendix A for details). Specimens were hand-collected during diurnal and nocturnal surveys and preserved in 70% ethanol for morphological examination. One leg of each specimen was removed and preserved in 96% ethanol for molecular analyses.
2.2. Microscopy and Imaging
Micrographs were taken at the Harry Butler Institute, Murdoch University (Australia), using a Leica DMC4500 digital camera (Leica, Wetzlar, Germany) mounted on a Leica M205C stereomicroscope. Images were captured in multiple focal planes and focus-stacked using Leica Application Suite X v.3.6.0.20104. Final image editing and figure assembly were carried out in Adobe Photoshop CC 2023. Color patterns of described specimens were based on specimens preserved in 96% ethanol. The protocol to expand the male pedipalp and to clarify the female genitalia was carried out by alternately submerging these structures for around 5 to 10 min in 10% KOH and distilled water until the pedipalp was fully expanded and the female genitalia sufficiently clarified with internal ducts and spermathecae visible.
2.3. Molecular Procedures
We extracted DNA from all 14 specimens using the DNeasy Blood & Tissue Kit (Qiagen, Hilden, Germany), following the manufacturer’s instructions. PCR protocol, including primers used, followed the procedure outlined in Yu et al. [19]. From one male and one female, we targeted five standard molecular markers: mitochondrial cytochrome c oxidase subunit I (COI) and 16S rRNA, as well as the nuclear ribosomal 18S, 28S rRNA, and Histone H3. From all remaining specimens, we only amplified COI. PCR products were purified and sequenced by Macrogen Europe B. V. (Amsterdam, The Netherlands). Sequences were assembled and edited using Geneious Pro v.5.6.7. and deposited in GenBank (for accession numbers see Appendix A).
2.4. Phylogenetic Reconstruction
To infer the phylogenetic placement of Megaraneus, we aligned the new sequences to the dataset of Scharff et al. [20], which includes 133 araneid taxa. To further increase taxon sampling we added newly available sequences of Cyphalonotus [19], belonging to another lineage containing a case of eSSD. The concatenated matrix is available as Supplementary File S1. Alignment was performed in MAFFT v.7 [21] using default settings and concatenated in Mesquite v.3.8.1 [22]. We reconstructed phylogenetic relationships using MrBayes v.3.2.7 [23], run via the CIPRES Science Gateway [24]. We used the generalized time-reversible model with gamma distribution and invariant sites (GTR + G + I) for each partition, following Scharff et al. [20]. We ran two independent runs, each with four MCMC chains, for 30 million generations, with a sampling frequency of 1000 and relative burn-in set to 25%. The starting tree was random [20].
To further corroborate the phylogenetic placement of Megaraneus as suggested by the result of the Bayesian analysis, we ran a second, Maximum Likelihood phylogenetic analysis on a representative subset of 63 taxa. We used IQ-TREE 2 v.3.0.1 [25] with a partitioning scheme from Scharff et al. [20]. Substitution model for each partition was determined by IQ-TREE 2. Bootstrap support was assessed with 1000 ultrafast bootstrap replicates [26,27] and SH-like approximate likelihood ratio test [28].
In order to evaluate evolutionary trends of sex-specific size evolution trajectories within the focal M. gabonensis group in downstream analyses, we reconstructed a separate phylogeny only including taxa with available body size measurements (see Section 2.6 Trait evolution). We expanded the focal group by adding Backobourkia brounii (GenBank accession sode: FJ873121) and Backobourkia collina (GenBank accession code: FJ873123), and eliminated representatives of Acroaspis and Carepalxis, for which such data is unavailable. Phylogenetic reconstruction used the same settings as above.
2.5. Morphological Examination
Measurements were taken under a Keyence VHX-7000 digital microscope and are reported in millimeters. Body and appendage measurements were recorded to the nearest 0.1 mm, while finer structures such as the eyes and labium were measured to the nearest 0.01 mm for greater precision. Terminology follows standard conventions in recent ‘backobourkiines’ systematics [29,30,31,32,33]. The terminology of the views of the male pedipalp considers its position as a limb, i.e., the full view of the bulb with the cymbium in the background is retrolateral (not ventral) because in Araneidae, the pedipalp is twisted so that the cymbium is situated mesally [32,33]. The epigyne has two main parts, the base (encapsulating the internal genitalia) and the scape. We refer to the central part of the base in ventral view as atrium, which becomes the central division in posterior view [29,30,31].
2.6. Trait Evolution
For comparative analyses, we compiled body length data for taxa, reconstructed as members of the focal Megaraneus clade, from the literature (Supplementary Table S1). SSD was calculated as the ratio of female to male body length, following Kuntner & Coddington [1]. We projected continuous trait data (body length and SSD) onto our focal clade phylogeny in Mesquite v.3.8.1 [22] using squared-change parsimony, requiring no assumptions about the underlying statistical distribution of trait evolution. This allowed a descriptive assessment of general trends in trait evolution within the clade, including ancestral state estimation and size trajectory comparisons for: (1) male body length vs. SSD, (2) female body length vs. SSD, and (3) male body length vs. female body length.
3. Results
The freshly collected material allows for thorough taxonomic treatment of M. gabonensis and, by current monotypy, of the genus Megaraneus (see Section 5, Taxonomy). Analyses of body length data suggest that M. gabonensis females are between 3.4 and 5.1 times larger than males, with an average SSD value of 4.1.
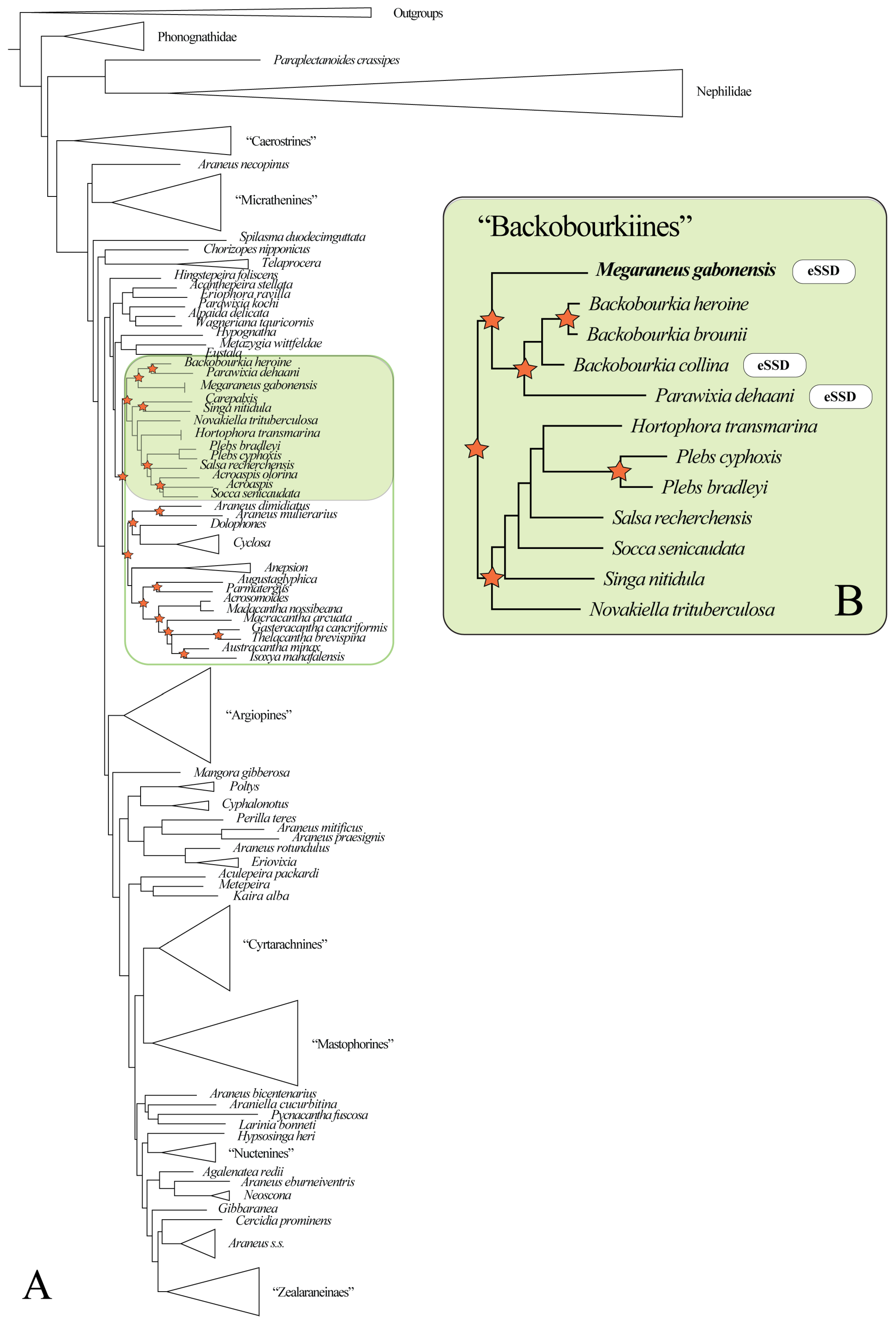
3.1. Phylogenetic Placement
The reconstructed Bayesian araneid phylogeny generally exhibits low support for deeper nodes, and the recovered topology deviates from that of Scharff et al. [20]. Nevertheless, it places M. gabonensis within a well-supported clade informally named ‘backobourkiines’ sensu Scharff et al. [20] (Figure 2A; Supplementary File S2). Within ‘backobourkiines’ (Figure 2B; Supplementary File S3), Megaraneus is sister to a clade containing Backobourkia and Parawixia dehaani (but see Discussion for a comment on probable generic misattribution), with the latter and Backobourkia collina exhibiting eSSD. The remaining two Backobourkia species exhibit modest SSD, reflecting intrageneric variation. Other genera within the broader ‘backobourkiines’ assemblage, i.e., Novakiella, Socca, Salsa, Plebs, Hortophora, and Singa (but see Results section Size evolution), are all sexually size monomorphic.
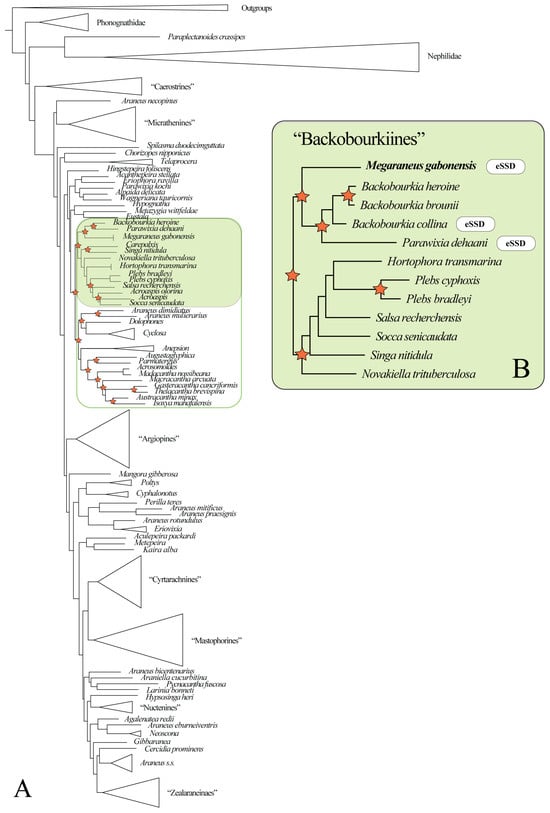
Figure 2.
(A) Bayesian phylogeny of Araneidae (for full results, see Supplementary File S2). Megaraneus gabonensis is placed within the ‘backobourkiines’ (highlighted in green) sensu Scharff et al. [20]. Stars indicate nodes with posterior probability > 0.90 within ‘backobourkiines’ and its sister clade (green outline). (B) Bayesian reconstruction of a subset of ‘backobourkiines’ (for full results, see Supplementary File S3). Stars indicate nodes with posterior probability > 0.90.
The Maximum Likelihood phylogeny corroborates the placement of Megaraneus within ‘backobourkiines’ (Supplementary File S4) and as sister to Parawixia dehaani + Backobourkia with high support (98/99).
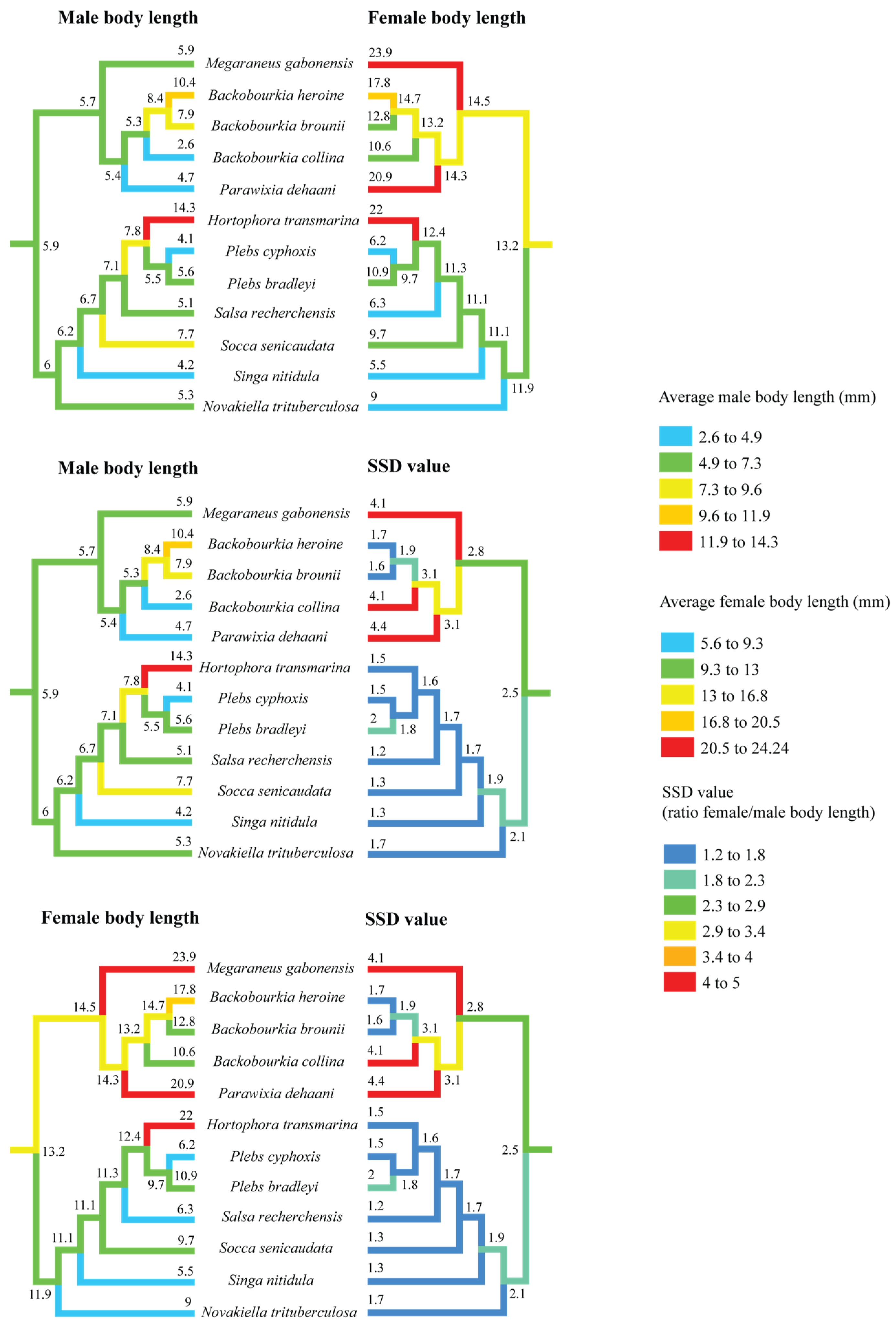
3.2. Size Evolution
Ancestral state reconstruction of body length and SSD across the ‘backobourkiines’ clade rejects an independent origin of eSSD in M. gabonensis. The common ancestor of Megaraneus, Backobourkia, Parawixia dehaani, Hortophora, Plebs, Salsa, Socca, Singa, and Novakiella is reconstructed to already exhibit SSD values above the 2.0 threshold, typically used to define eSSD (Figure 3). Although not directly relevant for the present study, note that the placement of Singa nitidula within the ‘backobourkiines’ in Scharff et al. [20] and other recent, yet unpublished phylogenies [34], is inconsistent and thus debatable.
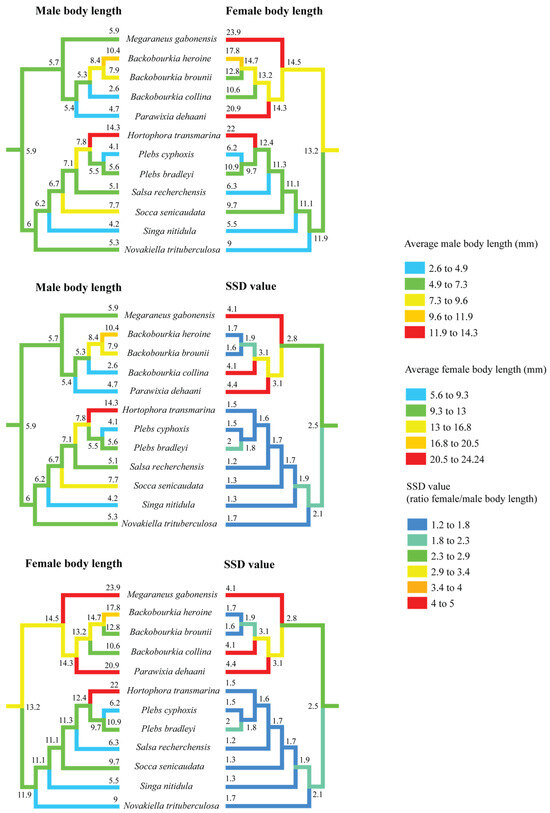
Figure 3.
Ancestral state reconstruction of body length and sexual size dimorphism (SSD) in ‘backobourkiines’ using squared-change parsimony. The results demonstrate the complexity of evolutionary change in male and female size that are often decoupled and combined produce SSD.
Within this clade, SSD trajectories show divergent trends. Except for Plebs bradleyi, the lineage containing Hortophora, Plebs, Salsa, Socca, Singa, and Novakiella has seen a decline to SSD values below 2.0. In contrast, the sister clade comprising Megaraneus, Backobourkia, and Parawixia dehaani exhibits increases in SSD values beyond the ancestral state. The highest reconstructed values occur in Parawixia dehaani (SSD = 4.4), followed by M. gabonensis and Backobourkia collina (both SSD = 4.1), while the other two species of Backobourkia show a decrease in SSD (see Supplementary Table S1).
Separate reconstructions of male and female body length (Figure 3) indicate that female body length has increased towards Megaraneus, whereas male body length has remained relatively unchanged from the ancestral values. This directional asymmetry in trait evolution underlies the observed increases in SSD toward the terminals.
4. Discussion
By integrating morphological variation, taxonomy, phylogenetics, and trait reconstruction, we shed new light on the evolutionary history of Megaraneus gabonensis, the sole representative of its genus, and its placement within Araneidae. Prior descriptions of the genus and species are, by today’s standards, outdated. We therefore provide a redescription (see Section 5, Taxonomy) that adheres to modern standards in araneology [17]. The major taxonomic contribution of our present description is the inclusion of microscopic imaging that shows detailed morphological features used in genus and species diagnoses.
Our original phylogenetic placement of Megaraneus recovers it nested within ‘backobourkiines’ sensu Scharff et al. [20], a clade previously thought geographically restricted to East Asia and Australia. This unexpected phylogenetic affinity significantly expands the known biogeographic range of the clade to include the Afrotropics and raises the question of whether the current distribution reflects an isolated case of long-distance dispersal or undersampled diversity in Africa.
Although the ‘backobourkiines’ portion of the tree is well supported, many deeper nodes of the araneid tree of life are not. Scharff et al. [20] reconstructed the araneid phylogeny using five Sanger markers, unsurprisingly not fully resolving relationships within an ancient family of over 100 million years [35,36]. The limits of Sanger data and the lack of sufficient detail on phylogenetic methodology used by Scharff et al. [20] are mirrored in our inability to recover their deeper topology. These topological uncertainties limit broader inferences about the evolutionary origins of SSD across the family. We therefore limit our discussion to the part of the topology relevant for Megaraneus and the size evolution within its immediate phylogenetic proximity.
SSD within the ‘backobourkiines’ is variable, ranging from minimal to eSSD. Our ancestral trait reconstructions indicate that eSSD was likely present in the common ancestor of Megaraneus, Backobourkia, and Parawixia dehaani. It should be noted, however, that the latter species is a probable misattribution to the genus Parawixia and in fact constitutes a separate genus [20].
These reconstructions suggest a broad pattern of fluctuating SSD across the clade, with independent trajectories of male and female size change and thus SSD magnitude. These trajectories include secondary reductions in Hortophora and Novakiella and further increases in Megaraneus and Parawixia. In Megaraneus, the observed pattern of sex-specific size changes suggests that its eSSD is a case of female gigantism [8], given that male body length has remained conserved, but the female body length has steadily increased. Note, however, that the observed trait evolution patterns are based only on ‘backobourkiines’ with available size data, omitting genera such as Acroaspis and Carepalxis. Additional sampling is thus necessary to further test our interpretations.
In conclusion, the pronounced eSSD in Megaraneus (Figure 1A) is not an isolated phenomenon, but rather part of a more complex and dynamic pattern of size evolution within a phylogenetically coherent group. Within Backobourkia alone, SSD varies from extreme to only slight; moreover, the eSSD species B. collina is significantly smaller overall and occupies a different habitat (grasslands vs. trees) compared to its congeners, making it an interesting object of future exploration. Results across the clade are consistent with the view that both female gigantism and male dwarfism have contributed to spider SSD across evolutionary timescales [1,37], and that the intensity and direction of size divergence are lineage-, or even species-specific, possibly shaped by ecological and reproductive factors [9].
5. Taxonomy
- Order Araneae Clerck, 1757Family Araneidae Clerck, 1757Genus Megaraneus Lawrence, 1968Type species. Megaraneus gabonensis (Lucas, 1858); by monotypy.Diagnosis. Somatically, specimens of Megaraneus resemble those of the phylogenetically close Backobourkia Framenau, Dupérré, Blackledge & Vink, 2010 and Parawixia dehaani (Doleschall, 1859) due to the similar abdomen shape and coloration of the three genera (enlarged, oblong with olive brown and light brown markings dorsally on paler area) (Figure 1A and Figure 4A vs. e.g., [38], figures 5A–F; [39], figure 25D). However, the genital morphology of males and females of Megaraneus can easily identify this genus from P. dehaani and especially from Backobourkia. Females of Megaraneus and P. dehaani differ from Backobourkia by having an epigyne with a bulky scape, triangular and not wrinkled, almost completely covering the atrium (Figure 4C; [33], figures 6D,H, 7A, 10D, 12D; [39], figure 3J), while males have pedipalps lacking a bubble-shaped terminal apophysis and the typical flange on the median apophysis of Backobourkia (Figure 5C and Figure 6C; [33], figures 6A, 10A, 12A; [40], figure 290g). In females, Megaraneus is distinguished from P. dehaani by the scape of the epigyne uniformly tapering to its tip, which ventrally overreaches the epigyne border by around half of the scape’s length (vs. scape very wide anteriorly, abruptly tapering to a thin tip that overreaches the epigyne border by less than half of the scape’s length) (Figure 4C,D; [39], figure 3J; [40], figure 290d,e). Finally, males of Megaraneus are distinguished from those of P. dehaani by the pedipalp with a larger conductor and conductor lobe, and by the presence of a strong triangular protrusion on the base of median apophysis. In contrast, P. dehaani males exhibit a smaller conductor and conductor lobe, and median apophysis without protrusions (Figure 5C and Figure 6C; [40], figure 290g).
 Figure 4. Megaraneus gabonensis, female (WAM T170877). (A) dorsal habitus; (B) ventral habitus; (C) epigyne, ventral view; (D) epigyne lateral view; (E) epigyne posterior view. Scale bars: (A,B) 2 mm; (C–E) 0.2 mm.
Figure 4. Megaraneus gabonensis, female (WAM T170877). (A) dorsal habitus; (B) ventral habitus; (C) epigyne, ventral view; (D) epigyne lateral view; (E) epigyne posterior view. Scale bars: (A,B) 2 mm; (C–E) 0.2 mm. Figure 5. Megaraneus gabonensis, male (WAM T170875). (A) dorsal habitus; (B) ventral habitus; (C) left pedipalp, ventral view; (D) left pedipalp, dorsal view. Scale bars: (A,B), 2 mm; (C,D) 0.2 mm.
Figure 5. Megaraneus gabonensis, male (WAM T170875). (A) dorsal habitus; (B) ventral habitus; (C) left pedipalp, ventral view; (D) left pedipalp, dorsal view. Scale bars: (A,B), 2 mm; (C,D) 0.2 mm.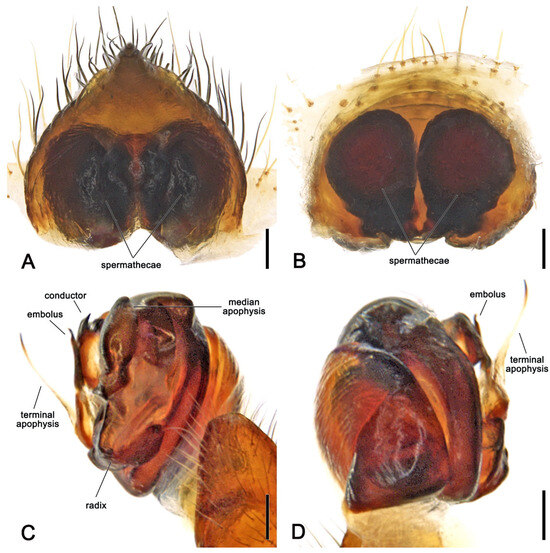 Figure 6. Megaraneus gabonensis, female internal genitalia cleared (WAM T170877). (A) posterior view; (B) anterior view. Megaraneus gabonensis, male left pedipalp expanded (WAM T170875). (C) baso-ventral view; (D) prolateral-dorsal view. Scale bars: (A–D) 0.2 mm.Description. Large orb-weaving spiders, with females (ca. TL 20.2–29.1) between 3.4 and 5.1 times larger than males (ca. TL 5.7–5.9). Preserved specimens with carapace longer than wide, pear-shaped; reddish brown with dusky sides and numerous setae with cup-shaped bases (Figure 4A and Figure 5A). Living specimens sometimes with a black carapace (Figure 1A). Fovea transverse (Figure 4A and Figure 5A). Row of posterior eyes slightly recurved, lateral eyes almost touching on a small tubercle; anterior median eyes slightly protruding from the carapace in both sexes (Figure 4A and Figure 5A). Sternum longer than wide, orange to reddish brown with dark contour and mottled beige (Figure 4B and Figure 5B). Labium wider than long, dark brown with anterior pale edge (Figure 4B and Figure 5B). Maxillae dark brown with pale antero-mesal section (Figure 4B and Figure 5B). Chelicerae dark brown. Leg formula IV > I > II > III for females and I > IV > II > III for males. Abdomen oblong, elongate cylindrical, posteriorly much narrower, with six strong humps (Figure 4A and Figure 5A); female with projected posterior end, rounded in males (Figure 4A and Figure 5A); at least two pairs of conspicuous black sigillae, the central ones larger (Figure 4A and Figure 5A); abdomen otherwise without specialized setae, condyles or other specific structures; females dorsally with brown background exposed anteriorly and followed by a large golden brown area, while males dorsally with olive gray background, medially lighter (Figure 4A and Figure 5A). Venter of females golden brown with two parallel black marks and other sparse black dots; venter of males olive grey to black (Figure 4B and Figure 5B). Female epigyne subquadrate, atrium centrally concave with excavated sclerotized sides (Figure 4C); scape broad, elongated, covering most of the atrium, and reaching over the epigyne base (Figure 4C,D); posterior plate with strong sclerotized sides, slightly tapering posteriorly with central division narrow (Figure 4E); internal genitalia with spherical enlarged spermathecae occupying most of the epigyne area (Figure 6A,B). Male pedipalp patella with a single short macroseta (Figure 5C); tibia with two strong macrosetae, only socket visible dorsally (Figure 5C,D); paracymbium very short with rounded tip (Figure 5D); median apophysis relatively short, medially enlarged with rounded tip, bearing an extremely enlarged and triangular protrusion on its base (Figure 5C and Figure 6C); radix wide and curved (Figure 5C and Figure 6C); stipes inconspicuous (Figure 5C and Figure 6C); terminal apophysis elongated, filiform, straight and transparent (Figure 5C,D and Figure 6C,D); conductor lobe enlarged and triangular, with a sclerotized base bearing scale-like structures (Figure 5C); conductor prominent, spoon-shaped, centrally fleshy and slightly concave, projected retrolaterally as two pointed and sclerotized tips (Figure 5C and Figure 6C); embolus basally broad, tapering apically to an elongated and filiform uncapped tip (Figure 5C and Figure 6C).Distribution. According to the World Spider Catalogue, specimens of Megaraneus gabonensis have so far been recorded in Sierra Leone, Cameroon, Gabon, Republic of Congo, Angola, Mozambique, and South Africa [41]. For an up-to-date species occurrence map, see GBIF [42]. Our specimens were collected in iSimangaliso Wetland Park, South Africa.Megaraneus gabonensis (Lucas, 1858)Epeira gabonensis Lucas [43]: 420–422, pl. 12, figure 6.Epeira angolensis Brito Capello [44]: 79–80, pl. 2, figure 4.Epeira chinchoxensis Karsch [45]: 333–334.Aranea basilissa Thorell [46]: 44.Cyrtophora angolensis Pocock [47]: 853–854.Cyrtophora gabonensis Simon [48]: 286.Caerostris basilissa Roewer [49]: 888.Megaraneus campbelli Lawrence [18]: 110–115, figures 1a–e and 2a–e,g (first male description).Megaraneus gabonensis Grasshoff [50]: 763–764.Megaraneus gabonensis Dippenaar-Schoeman et al. [51]: 47–48, p. 47: 1 figure, p. 48: 6 figures.Type-material. Holotype female of Epeira gabonensis Lucas, 1858 from Gabon, western Africa coast (MNHN?), not examined.Female syntype of Epeira angolensis Brito Capello, 1866 from Rio Cuilo (Kwilo or Quilo), Angola/Democratic Republic of Congo, José de Anchieta coll. (ACL?), not examined.4 syntype females of Epeira chinchoxensis Karsch, 1879 from Chinchoxo (Tschinschotscho), Cabinda, Angola, Falkenstein J. coll. (ZMB 2950), not examined.Female syntype of Aranea basilissa Thorell, 1899 from Cameroon, Sjöstedt coll. (NHRS?), not examined.Holotype female of Megaraneus campbelli Lawrence, 1968, from eastern shore of Lake Sibayi, I. D. J. Jones coll., July 1967 (RU), not examined; male allotype from same locality of holotype, except R. F. Lawrence coll., January1968 (RU), not examined; 2 females and 1 male paratypes, same data as allotype (RU), not examined.Material examined. See Appendix A.Diagnosis. As for genus, Megaraneus is monotypic.Description. Female (based on WAM T170877): Total length 20.6. Carapace 10.0 long, 8.9 wide; entirely reddish brown with darker sides and a central dark spot at cephalic area (Figure 4A). Eyes: AME 0.56, ALE 0.48, PME 0.53, PLE 0.45. Chelicerae black (Figure 4B). Legs black with femora lighter ventrally near coxae; all legs with sparse yellowish setae (Figure 4A,B). Pedipalp length of segments (femur + patella + tibia + tarsus = total length): 3.4 + 1.7+ 3.0 + 4.5 = 12.6. Leg formula IV > I > II > III; length of segments (femur + patella + tibia + metatarsus + tarsus = total length): I—11.1 + 5.0 + 9.8 + 8.6 + 3.4 = 37.9, II—10.4 + 4.6 + 8.9 + 8.5 + 3.6 = 36.0, III—8.1 + 4.1 + 5.6 + 4.9 + 2.5 = 25.2, IV—12.5 + 5.8 + 9.6 + 8.8 + 3.5 = 40.2. Labium 1.53 long, 2.36 wide, dark brown, almost black, and maxillae dark brown, both anteriorly light brown and pale (Figure 4B). Sternum 5.7 long, 4.7 wide, orange brown with lighter streaks (Figure 4B). Abdomen 10.6 long, 18.6 wide, bearing six humps, three on each side, and a projected and pointed posterior end; dorsally with olive brown background exposed anteriorly, medially and posteriorly strongly covered in golden brown; four pairs of small to median sized brown sigillae (Figure 4A), venter golden brown with two parallel transverse black streaks and sparse black dots, laterally olive brown (Figure 4B). Epigyne and spermathecae descriptions as for genus (Figure 4C–E and Figure 6A,B).Male (based on WAM T170875): Total length 5.9. Carapace 2.8 long, 2.3 wide, chelicerae, sternum 1.2 long, 1.1 wide, labium 0.57 long, 0.42 wide, and maxillae as in females (Figure 5A,B). Eyes: AME 0.23, ALE 0.16, PME 0.20, PLE 0.14. Legs orange brown mottled dark (Figure 5A,B). Leg formula I > II > IV > III; length of segments (femur + patella + tibia + metatarsus + tarsus = total length): I—2.0 + 0.9 + 1.8 + 1.6 + 0.8 = 7.1, II—2.0 + 0.8 + 1.6 + 1.5 + 0.8 = 6.7, III—1.4 + 0.6 + 0.9 + 0.8 + 0.6 = 4.3, IV—1.7 + 0.7 + 1.4 + 1.3 + 0.7 = 5.8. Abdomen 3.1 long, 2.7 wide, with similar humps as females, with a rounded posterior end; dorsally olive gray background, medially lighter, and bearing two pairs of small sigillae (Figure 5A); venter with yellowish brown background covered by a large black patch (Figure 5B). Pedipalp length of segments (femur + patella + tibia + cymbium = total length): 0.6 + 0.3 + 0.2 + 0.8 = 1.9; description as for genus (Figure 5C,D and Figure 6C,D).Variation. Total length females 20.2–29.1 (n = 12). Total length males 5.7–5.9 (n = 2). Live female specimens can vary from entirely olive brown to entirely black; or black with white abdomen (Figure 1B). The white area of the abdomen varies between specimens.Natural history. The orb web of the female (Figure 1C) is suspended among shrubs, on short anchor threads close to vegetation. During the day, the spiders hide in an off-web leaf retreat enforced by silk threads. The web is large, outermost spiral area between 43 and 58 cm wide and between 60 and 75 cm in height, with the upper orb portion of 16 to 24 cm (n = 4 female webs). With the capture area of 0.20 to 0.34 m2, hub displacement index of 0.65 to 0.73, and ladder index of 1.29 to 1.40, the webs are typical of large araneid spiders [52,53]. The webs are sparse, typically with 25–29 radii and 25–28 capture spirals along one web axis, always with an open hub. For additional natural history observations, see [18].Taxonomic notes. The taxonomic history of the genus Megaraneus and its species M. gabonensis is complex [41]. Megaraneus was proposed by Lawrence [18] to initially accommodate the species M. campbelli Lawrence, 1968, while Megaraneus gabonensis was first described by Lucas [43] in the genus Epeira Walckenaer, 1805 (today a junior synonym of Araneus Clerck, 1757). After its original description, two other species in the genus Epeira were described, E. angolensis (Brito Capello, 1866) and E. chinchoxensis Karsch, 1879, both later transferred to Cyrtophora Simon, 1864 and synonymized with C. citricola (Forsskål, 1775) [54]. Simon [48] later rejected the synonymy with C. citricola and synonymized both species with E. gabonensis in Cyrtophora, possibly due to the abdominal humps, forming the combination Cyrtophora gabonensis (Lucas, 1858).
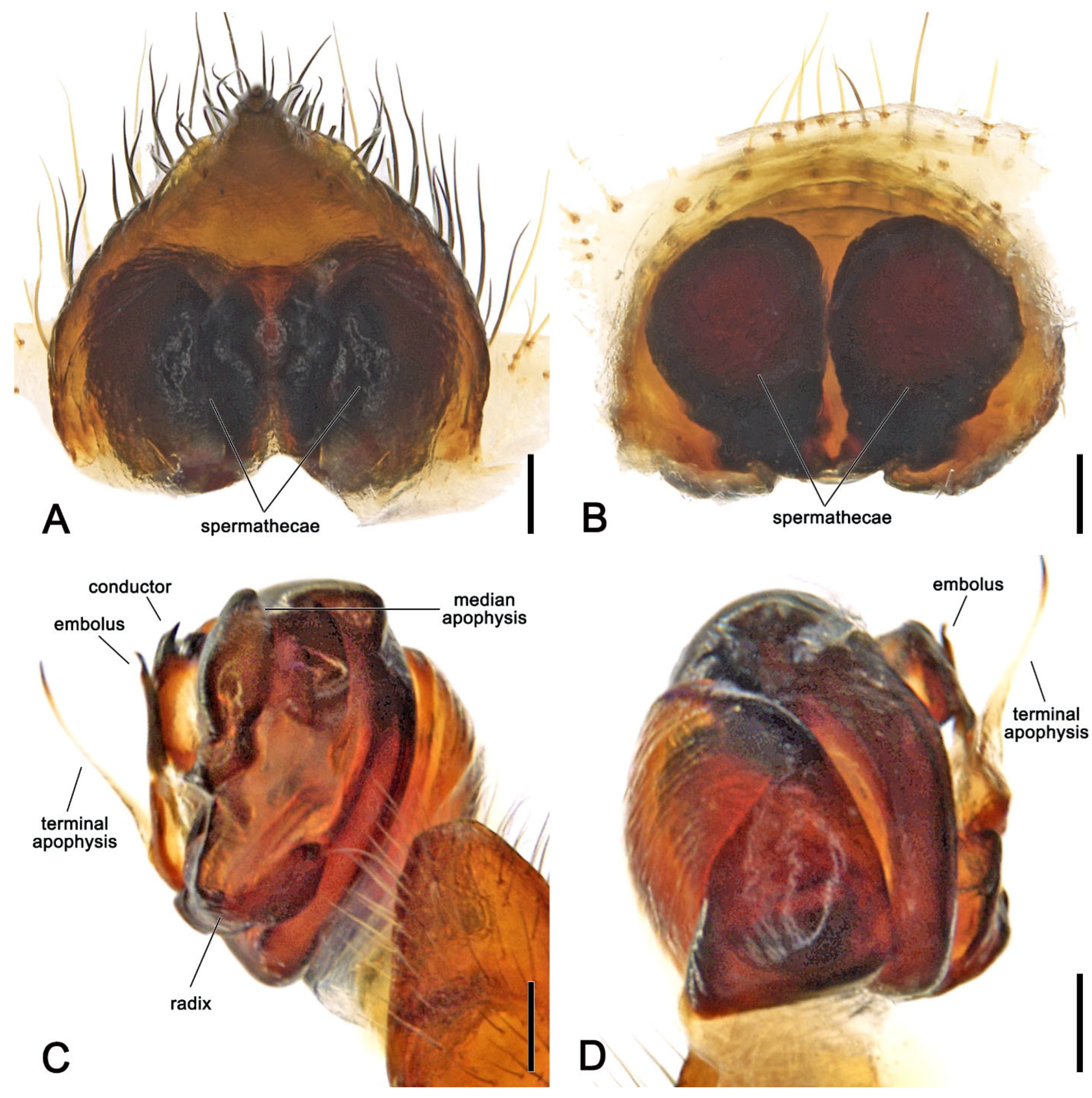
Figure 6. Megaraneus gabonensis, female internal genitalia cleared (WAM T170877). (A) posterior view; (B) anterior view. Megaraneus gabonensis, male left pedipalp expanded (WAM T170875). (C) baso-ventral view; (D) prolateral-dorsal view. Scale bars: (A–D) 0.2 mm.Description. Large orb-weaving spiders, with females (ca. TL 20.2–29.1) between 3.4 and 5.1 times larger than males (ca. TL 5.7–5.9). Preserved specimens with carapace longer than wide, pear-shaped; reddish brown with dusky sides and numerous setae with cup-shaped bases (Figure 4A and Figure 5A). Living specimens sometimes with a black carapace (Figure 1A). Fovea transverse (Figure 4A and Figure 5A). Row of posterior eyes slightly recurved, lateral eyes almost touching on a small tubercle; anterior median eyes slightly protruding from the carapace in both sexes (Figure 4A and Figure 5A). Sternum longer than wide, orange to reddish brown with dark contour and mottled beige (Figure 4B and Figure 5B). Labium wider than long, dark brown with anterior pale edge (Figure 4B and Figure 5B). Maxillae dark brown with pale antero-mesal section (Figure 4B and Figure 5B). Chelicerae dark brown. Leg formula IV > I > II > III for females and I > IV > II > III for males. Abdomen oblong, elongate cylindrical, posteriorly much narrower, with six strong humps (Figure 4A and Figure 5A); female with projected posterior end, rounded in males (Figure 4A and Figure 5A); at least two pairs of conspicuous black sigillae, the central ones larger (Figure 4A and Figure 5A); abdomen otherwise without specialized setae, condyles or other specific structures; females dorsally with brown background exposed anteriorly and followed by a large golden brown area, while males dorsally with olive gray background, medially lighter (Figure 4A and Figure 5A). Venter of females golden brown with two parallel black marks and other sparse black dots; venter of males olive grey to black (Figure 4B and Figure 5B). Female epigyne subquadrate, atrium centrally concave with excavated sclerotized sides (Figure 4C); scape broad, elongated, covering most of the atrium, and reaching over the epigyne base (Figure 4C,D); posterior plate with strong sclerotized sides, slightly tapering posteriorly with central division narrow (Figure 4E); internal genitalia with spherical enlarged spermathecae occupying most of the epigyne area (Figure 6A,B). Male pedipalp patella with a single short macroseta (Figure 5C); tibia with two strong macrosetae, only socket visible dorsally (Figure 5C,D); paracymbium very short with rounded tip (Figure 5D); median apophysis relatively short, medially enlarged with rounded tip, bearing an extremely enlarged and triangular protrusion on its base (Figure 5C and Figure 6C); radix wide and curved (Figure 5C and Figure 6C); stipes inconspicuous (Figure 5C and Figure 6C); terminal apophysis elongated, filiform, straight and transparent (Figure 5C,D and Figure 6C,D); conductor lobe enlarged and triangular, with a sclerotized base bearing scale-like structures (Figure 5C); conductor prominent, spoon-shaped, centrally fleshy and slightly concave, projected retrolaterally as two pointed and sclerotized tips (Figure 5C and Figure 6C); embolus basally broad, tapering apically to an elongated and filiform uncapped tip (Figure 5C and Figure 6C).Distribution. According to the World Spider Catalogue, specimens of Megaraneus gabonensis have so far been recorded in Sierra Leone, Cameroon, Gabon, Republic of Congo, Angola, Mozambique, and South Africa [41]. For an up-to-date species occurrence map, see GBIF [42]. Our specimens were collected in iSimangaliso Wetland Park, South Africa.Megaraneus gabonensis (Lucas, 1858)Epeira gabonensis Lucas [43]: 420–422, pl. 12, figure 6.Epeira angolensis Brito Capello [44]: 79–80, pl. 2, figure 4.Epeira chinchoxensis Karsch [45]: 333–334.Aranea basilissa Thorell [46]: 44.Cyrtophora angolensis Pocock [47]: 853–854.Cyrtophora gabonensis Simon [48]: 286.Caerostris basilissa Roewer [49]: 888.Megaraneus campbelli Lawrence [18]: 110–115, figures 1a–e and 2a–e,g (first male description).Megaraneus gabonensis Grasshoff [50]: 763–764.Megaraneus gabonensis Dippenaar-Schoeman et al. [51]: 47–48, p. 47: 1 figure, p. 48: 6 figures.Type-material. Holotype female of Epeira gabonensis Lucas, 1858 from Gabon, western Africa coast (MNHN?), not examined.Female syntype of Epeira angolensis Brito Capello, 1866 from Rio Cuilo (Kwilo or Quilo), Angola/Democratic Republic of Congo, José de Anchieta coll. (ACL?), not examined.4 syntype females of Epeira chinchoxensis Karsch, 1879 from Chinchoxo (Tschinschotscho), Cabinda, Angola, Falkenstein J. coll. (ZMB 2950), not examined.Female syntype of Aranea basilissa Thorell, 1899 from Cameroon, Sjöstedt coll. (NHRS?), not examined.Holotype female of Megaraneus campbelli Lawrence, 1968, from eastern shore of Lake Sibayi, I. D. J. Jones coll., July 1967 (RU), not examined; male allotype from same locality of holotype, except R. F. Lawrence coll., January1968 (RU), not examined; 2 females and 1 male paratypes, same data as allotype (RU), not examined.Material examined. See Appendix A.Diagnosis. As for genus, Megaraneus is monotypic.Description. Female (based on WAM T170877): Total length 20.6. Carapace 10.0 long, 8.9 wide; entirely reddish brown with darker sides and a central dark spot at cephalic area (Figure 4A). Eyes: AME 0.56, ALE 0.48, PME 0.53, PLE 0.45. Chelicerae black (Figure 4B). Legs black with femora lighter ventrally near coxae; all legs with sparse yellowish setae (Figure 4A,B). Pedipalp length of segments (femur + patella + tibia + tarsus = total length): 3.4 + 1.7+ 3.0 + 4.5 = 12.6. Leg formula IV > I > II > III; length of segments (femur + patella + tibia + metatarsus + tarsus = total length): I—11.1 + 5.0 + 9.8 + 8.6 + 3.4 = 37.9, II—10.4 + 4.6 + 8.9 + 8.5 + 3.6 = 36.0, III—8.1 + 4.1 + 5.6 + 4.9 + 2.5 = 25.2, IV—12.5 + 5.8 + 9.6 + 8.8 + 3.5 = 40.2. Labium 1.53 long, 2.36 wide, dark brown, almost black, and maxillae dark brown, both anteriorly light brown and pale (Figure 4B). Sternum 5.7 long, 4.7 wide, orange brown with lighter streaks (Figure 4B). Abdomen 10.6 long, 18.6 wide, bearing six humps, three on each side, and a projected and pointed posterior end; dorsally with olive brown background exposed anteriorly, medially and posteriorly strongly covered in golden brown; four pairs of small to median sized brown sigillae (Figure 4A), venter golden brown with two parallel transverse black streaks and sparse black dots, laterally olive brown (Figure 4B). Epigyne and spermathecae descriptions as for genus (Figure 4C–E and Figure 6A,B).Male (based on WAM T170875): Total length 5.9. Carapace 2.8 long, 2.3 wide, chelicerae, sternum 1.2 long, 1.1 wide, labium 0.57 long, 0.42 wide, and maxillae as in females (Figure 5A,B). Eyes: AME 0.23, ALE 0.16, PME 0.20, PLE 0.14. Legs orange brown mottled dark (Figure 5A,B). Leg formula I > II > IV > III; length of segments (femur + patella + tibia + metatarsus + tarsus = total length): I—2.0 + 0.9 + 1.8 + 1.6 + 0.8 = 7.1, II—2.0 + 0.8 + 1.6 + 1.5 + 0.8 = 6.7, III—1.4 + 0.6 + 0.9 + 0.8 + 0.6 = 4.3, IV—1.7 + 0.7 + 1.4 + 1.3 + 0.7 = 5.8. Abdomen 3.1 long, 2.7 wide, with similar humps as females, with a rounded posterior end; dorsally olive gray background, medially lighter, and bearing two pairs of small sigillae (Figure 5A); venter with yellowish brown background covered by a large black patch (Figure 5B). Pedipalp length of segments (femur + patella + tibia + cymbium = total length): 0.6 + 0.3 + 0.2 + 0.8 = 1.9; description as for genus (Figure 5C,D and Figure 6C,D).Variation. Total length females 20.2–29.1 (n = 12). Total length males 5.7–5.9 (n = 2). Live female specimens can vary from entirely olive brown to entirely black; or black with white abdomen (Figure 1B). The white area of the abdomen varies between specimens.Natural history. The orb web of the female (Figure 1C) is suspended among shrubs, on short anchor threads close to vegetation. During the day, the spiders hide in an off-web leaf retreat enforced by silk threads. The web is large, outermost spiral area between 43 and 58 cm wide and between 60 and 75 cm in height, with the upper orb portion of 16 to 24 cm (n = 4 female webs). With the capture area of 0.20 to 0.34 m2, hub displacement index of 0.65 to 0.73, and ladder index of 1.29 to 1.40, the webs are typical of large araneid spiders [52,53]. The webs are sparse, typically with 25–29 radii and 25–28 capture spirals along one web axis, always with an open hub. For additional natural history observations, see [18].Taxonomic notes. The taxonomic history of the genus Megaraneus and its species M. gabonensis is complex [41]. Megaraneus was proposed by Lawrence [18] to initially accommodate the species M. campbelli Lawrence, 1968, while Megaraneus gabonensis was first described by Lucas [43] in the genus Epeira Walckenaer, 1805 (today a junior synonym of Araneus Clerck, 1757). After its original description, two other species in the genus Epeira were described, E. angolensis (Brito Capello, 1866) and E. chinchoxensis Karsch, 1879, both later transferred to Cyrtophora Simon, 1864 and synonymized with C. citricola (Forsskål, 1775) [54]. Simon [48] later rejected the synonymy with C. citricola and synonymized both species with E. gabonensis in Cyrtophora, possibly due to the abdominal humps, forming the combination Cyrtophora gabonensis (Lucas, 1858).
Finally, in 1984 Grasshoff transferred Cyrtophora gabonensis to Megaraneus and proposed M. campbelli, as well as Caerostris basilissa (Thorell, 1899) (initially described as Aranea basilissa Thorell, 1899) as junior synonyms of M. gabonensis, rendering Megaraneus monotypic [50]. The morphology of M. gabonensis is more comparable with other ‘backobourkiines’ that display eSSD, such as the genus Backobourkia and P. dehaani (see diagnosis above), rather than Cyrtophora and Caerostris Thorell, 1868 by females with a long and wide scape covering the atrium (Figure 4C) and males with a strong median apophysis with an arch over the radix and a very thin and elongated terminal apophysis (Figure 5C,D).
- Distribution. As for genus, Megaraneus is monotypic.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/insects16100992/s1, Table S1: compiled body length data; File S1: aligned phylogenetic matrix; File S2: raw Bayesian phylogenetic tree (note that taxonomic names are from Scharff et al. [20]); File S3: raw Bayesian phylogeny of the focal group containing Megaraneus; File S4: Maximum Likelihood phylogeny on an araneid subset. References [18,29,30,31,32,38,40,43,46,55,56,57,58,59,60,61,62,63,64] are cited in the Supplementary Materials.
Author Contributions
Conceptualization, M.K. and K.Č.; formal analysis, K.-P.Y., K.Č. and P.d.S.C.; investigation, M.K., M.G., I.A. and K.-P.Y.; resources, M.K. and V.W.F.; data curation, E.T. and K.-P.Y.; writing—original draft preparation, E.T., K.Č. and M.K.; writing—review and editing, all authors; visualization, P.d.S.C., K.-P.Y., K.Č. and E.T.; supervision, M.K.; project administration, M.K.; funding acquisition, M.K., M.G. and I.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Slovenian Research and Innovation Agency, grants numbers P1-0255, P1-0236, Z1-8143, J1-9163, J1-50015, J1-60005; National Science Foundation (USA), grant number IOS-1656460. Additional funding was provided by the Australian Biological Resources Study (ABRS) grant number 4-EHPVRMK [2021,2022,2023,2024] to VWF, PSC, N. Scharff, D. Dimitrov, A. Chopra and R. Baptista.
Data Availability Statement
Original sequence data presented in the study is openly available on GenBank. For accession numbers, refer to Appendix A.
Acknowledgments
We thank R.-C. Cheng and T.-K. Lin for providing additional measurements of Parawixia dehaani. Research in South Africa was performed under a research agreement with the iSimangaliso Wetland Park Authority and the sampling permission by the Ezemvelo KZN Wildlife. We thank J. Bond, C. Haddad and R. Booysen for logistical help.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ALE | Anterior lateral eyes |
| AME | Anterior median eyes |
| PLE | Posterior lateral eyes |
| PME | Posterior median eyes |
| ACL | Academia das Ciências de Lisboa (Lisbon Science Academy), Lisbon, Portugal |
| NHRS | Naturhistoriska Riksmuseet (Swedish Museum of Natural History), Stockholm, Sweden |
| NIB | National Institute of Biology, Ljubljana, Slovenia |
| RU | Rhodes University, Grahamstown, South Africa |
| WAM | Western Australian Museum, Perth, Australia |
| ZMB | Museum für Naturkunde, Berlin, Germany |
Appendix A
The Megaraneus gabonensis material examined and sequenced in this study. We provide specimen information as: species name; database code; sex and number; locality details; locality coordinates (longitude, latitude); collection date (day, month, year); and DNA barcode identifiers (GenBank accession numbers for COI, 16S, 18S, 28S, and H3). Unless otherwise indicated, specimen vouchers are deposited at the National Institute of Biology, Ljubljana, Slovenia. Specimens were collected under a general permit (No. OP1869) issued by Ezemvelo KZN Wildlife and research agreement with iSimangaliso Wetland Park Authority.
- Megaraneus gabonensis; ARA2067; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 11. 1. 2019; COI: PV625041, 16S: PV624644, 18S: PV624643; 28S: PV624640, H3: PV631755.
- Megaraneus gabonensis; ARA5105; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625040.
- Megaraneus gabonensis; ARA5106; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Kosi Bay; lat −26.959247, lon 32.824499; 14. 1. 2019; COI: PV625050.
- Megaraneus gabonensis; ARA5107; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625045.
- Megaraneus gabonensis; ARA5108; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625039.
- Megaraneus gabonensis; ARA5109; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 11. 1. 2019; COI: PV625038.
- Megaraneus gabonensis; ARA5110; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625037.
- Megaraneus gabonensis; ARA5111; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625048.
- Megaraneus gabonensis; ARA5135; 1 male; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 11. 1. 2019; COI: PV625044.
- Megaraneus gabonensis; ARA5136; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 11. 1. 2019; COI: PV625049.
- Megaraneus gabonensis; ARA5137; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625043.
- Megaraneus gabonensis; WAM T170875; 1 male; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625047, 16S: PV624645, 18S: PV624642, 28S: PV624641, H3: PV631756. Deposited at Western Australian Museum, Perth, Australia.
- Megaraneus gabonensis; WAM T170876; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Eastern Shores, uMziki near Mission Rocks; lat −28.264910, lon 32.481620; 7. 1. 2019; COI: PV625046. Deposited at Western Australian Museum, Perth, Australia.
- Megaraneus gabonensis; WAM T170877; 1 female; South Africa, KwaZulu-Natal, iSimangaliso Wetland Park, Cape Vidal; lat −28.125640, lon 32.558201; 17. 1. 2019; COI: PV625042. Deposited at Western Australian Museum, Perth, Australia.
References
- Kuntner, M.; Coddington, J.A. Sexual Size Dimorphism: Evolution and Perils of Extreme Phenotypes in Spiders. Annu. Rev. Entomol. 2020, 65, 57–80. [Google Scholar] [CrossRef]
- Vollrath, F.; Parker, G.A. Sexual Dimorphism and Distorted Sex Ratios in Spiders. Nature 1992, 360, 156–159. [Google Scholar] [CrossRef]
- Kuntner, M.; Elgar, M.A. Evolution and Maintenance of Sexual Size Dimorphism: Aligning Phylogenetic and Experimental Evidence. Front. Ecol. Evol. 2014, 2, 26. [Google Scholar] [CrossRef]
- Hormiga, G.; Scharff, N.; Coddington, J.A. The Phylogenetic Basis of Sexual Size Dimorphism in Orb-Weaving Spiders (Araneae, Orbiculariae). Syst. Biol. 2000, 49, 435–462. [Google Scholar] [CrossRef] [PubMed]
- Foellmer, M.W.; Moya-Laraño, J. Sexual Size Dimorphism in Spiders: Patterns and Processes. In Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism Sex, Size and Gender Roles: Evolutionary Studies of Sexual Size Dimorphism; Fairbairn, D.J., Blanckenhorn, W.U., Székely, T., Eds.; Oxford University Press: Oxford, UK, 2007; pp. 71–81. [Google Scholar]
- Framenau, V.W.; Castanheira, P.d.S. A New Genus of Australian Orb-Weaving Spider with Extreme Sexual Size Dimorphism (Araneae, Araneidae). Zoosyst. Evol. 2022, 98, 137–149. [Google Scholar] [CrossRef]
- Kuntner, M.; Agnarsson, I.; Li, D. The Eunuch Phenomenon: Adaptive Evolution of Genital Emasculation in Sexually Dimorphic Spiders. Biol. Rev. 2015, 90, 279–296. [Google Scholar] [CrossRef]
- Coddington, J.A.; Hormiga, G.; Scharff, N. Giant Female or Dwarf Male Spiders? Nature 1997, 385, 687–688. [Google Scholar] [CrossRef]
- Kuntner, M.; Hamilton, C.A.; Cheng, R.-C.; Gregorič, M.; Lupše, N.; Lokovšek, T.; Lemmon, E.M.; Lemmon, A.R.; Agnarsson, I.; Coddington, J.A.; et al. Golden Orbweavers Ignore Biological Rules: Phylogenomic and Comparative Analyses Unravel a Complex Evolution of Sexual Size Dimorphism. Syst. Biol. 2019, 68, 555–572. [Google Scholar] [CrossRef]
- Head, G. Selection on Fecundity and Variation in the Degree of Sexual Size Dimorphism among Spider Species (Class Araneae). Evolution 1995, 49, 776–781. [Google Scholar] [CrossRef]
- Prenter, J.; Elwood, R.W.; Montgomery, W.I. Sexual Size Dimorphism and Reproductive Investment by Female Spiders: A Comparative Analysis. Evolution 1999, 53, 1987–1994. [Google Scholar] [CrossRef]
- Higgins, L.E. Developmental Plasticity and Fecundity in the Orb-Weaving Spider Nephila clavipes. J. Arachnol. 1992, 20, 94–106. [Google Scholar]
- Corcobado, G.; Rodríguez-Gironés, M.A.; De Mas, E.; Moya-Laraño, J. Introducing the Refined Gravity Hypothesis of Extreme Sexual Size Dimorphism. BMC Evol. Biol. 2010, 10, 236. [Google Scholar] [CrossRef] [PubMed]
- Moya-Laraño, J.; Vinković, D.; Allard, C.M.; Foellmer, M.W. Optimal Climbing Speed Explains the Evolution of Extreme Sexual Size Dimorphism in Spiders. J. Evol. Biol. 2009, 22, 954–963. [Google Scholar] [CrossRef] [PubMed]
- Moya-Laraño, J.; Halaj, J.; Wise, D.H. Climbing to Reach Females: Romeo Should Be Small. Evolution 2002, 56, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Blanckenhorn, W.U. Behavioral Causes and Consequences of Sexual Size Dimorphism. Ethology 2005, 111, 977–1016. [Google Scholar] [CrossRef]
- Bond, J.E.; Godwin, R.L.; Colby, J.D.; Newton, L.G.; Zahnle, X.J.; Agnarsson, I.; Hamilton, C.A.; Kuntner, M. Improving Taxonomic Practices and Enhancing Its Extensibility—An Example from Araneology. Diversity 2021, 14, 5. [Google Scholar] [CrossRef]
- Lawrence, R.F. Four New Spiders from Southern Africa (Araneae). Ann. Natal Mus. 1968, 20, 109–121. [Google Scholar]
- Yu, K.-P.; Kuntner, M.; Cheng, R.-C. Phylogenetic Evidence for an Independent Origin of Extreme Sexual Size Dimorphism in a Genus of Araneid Spiders (Araneae: Araneidae). Invertebr. Syst. 2022, 36, 48–62. [Google Scholar] [CrossRef]
- Scharff, N.; Coddington, J.A.; Blackledge, T.A.; Agnarsson, I.; Framenau, V.W.; Szűts, T.; Hayashi, C.Y.; Dimitrov, D. Phylogeny of the Orb-Weaving Spider Family Araneidae (Araneae: Araneoidea). Cladistics 2020, 36, 1–21. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef]
- Maddison, W.P.; Maddison, D.R. Mesquite: A Modular System for Evolutionary Analysis, Version 3.8.1; 2025. Available online: http://www.mesquiteproject.org (accessed on 14 July 2025).
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian Phylogenetic Inference and Model Choice across a Large Model Space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Miller, M.A.; Pfeiffer, W.; Schwartz, T. Creating the CIPRES Science Gateway for Inference of Large Phylogenetic Trees. In Proceedings of the Gateway Computing Environments Workshop (GCE), New Orleans, LA, USA, 14 November 2010; pp. 1–8. [Google Scholar]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Minh, B.Q.; Nguyen, M.A.T.; von Haeseler, A. Ultrafast Approximation for Phylogenetic Bootstrap. Mol. Biol. Evol. 2013, 30, 1188–1195. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Framenau, V.W.; Vink, C.J.; Scharff, N.; Baptista, R.L.C.; Castanheira, P.d.S. Review of the Australian and New Zealand Orb-Weaving Spider Genus Novakiella (Araneae, Araneidae). Zoosyst. Evol. 2021, 97, 393–405. [Google Scholar] [CrossRef]
- Framenau, V.W.; Castanheira, P.d.S.; Vink, C.J. Taxonomy and Systematics of the New Australo-Pacific Orb-Weaving Spider Genus Socca (Araneae: Araneidae). N. Z. J. Zool. 2022, 49, 263–334. [Google Scholar] [CrossRef]
- Framenau, V.W.; Baptista, R.L.C.; Oliveira, F.S.M.; Castanheira, P.d.S. Taxonomic Revision of the New Spider Genus Hortophora, the Australasian Garden Orb-Weavers (Araneae, Araneidae). Evol. Syst. 2021, 5, 275–334. [Google Scholar] [CrossRef]
- Framenau, V.W.; Castanheira, P.d.S. Revision of the New Australasian Orb-Weaving Spider Genus Salsa (Araneae, Araneidae). Zookeys 2022, 1102, 107–148. [Google Scholar] [CrossRef]
- Castanheira, P.d.S.; Baptista, R.L.C.; Framenau, V.W. The Male of the Orb-Weaving Spider Socca australis (Araneae, Araneidae) and New Distribution Records in the Genus. Aust. J. Taxon. 2025, 89, 1–5. [Google Scholar] [CrossRef]
- Castanheira, P.d.S.; Dimitrov, D.; Baptista, R.L.C.; Scharff, N.; Framenau, V.W. Taxonomy and systematics of the Australasian gum nut orb-weaving spider genus Carepalxis (Araneae, Araneidae). Invertebr. Syst. 2025, 39, IS25009. [Google Scholar] [CrossRef]
- Kuntner, M.; Čandek, K.; Gregorič, M.; Turk, E.; Hamilton, C.A.; Chamberland, L.; Starrett, J.; Cheng, R.-C.; Coddington, J.A.; Agnarsson, I.; et al. Increasing Information Content and Diagnosability in Family-Level Classifications. Syst. Biol. 2023, 72, 964–971. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, I.L.F.; Azevedo, G.H.F.; Michalik, P.; Ramírez, M.J. The Fossil Record of Spiders Revisited: Implications for Calibrating Trees and Evidence for a Major Faunal Turnover since the Mesozoic. Biol. Rev. 2020, 95, 184–217. [Google Scholar] [CrossRef]
- Cheng, R.-C.; Kuntner, M. Phylogeny Suggests Nondirectional and Isometric Evolution of Sexual Size Dimorphism in Argiopine Spiders. Evolution 2014, 68, 2861–2872. [Google Scholar] [CrossRef]
- Framenau, V.W.; Dupérré, N.; Blackledge, T.A.; Vink, C.J. Systematics of the New Australasian Orb-Weaving Spider Genus Backobourkia (Araneae: Araneidae: Araneinae). Arthropod Syst. Phylogeny 2010, 68, 79–111. [Google Scholar] [CrossRef]
- Caleb, J.T.D. Spider (Arachnida: Araneae) Fauna of the Scrub Jungle in the Madras Christian College Campus, Chennai, India. J. Threat. Taxa 2020, 12, 15711–15766. [Google Scholar] [CrossRef]
- Yin, C.M.; Peng, X.J.; Yan, H.M.; Bao, Y.H.; Xu, X.; Tang, G.; Zhou, Q.S.; Liu, P. Fauna Hunan: Araneae in Hunan, China; Hunan Science and Technology Press: Changsha, China, 2012. [Google Scholar]
- World Spider Catalog; Version 26; Natural History Museum Bern: Bern, Switzerland, 2025; Available online: http://wsc.nmbe.ch (accessed on 14 July 2025).
- GBIF Occurrence Download. Available online: https://doi.org/10.15468/39omei (accessed on 14 July 2025).
- Lucas, H. Aptères. In Voyage au Gabon; Thomson, J., Ed.; Archives Entomologiques de M. J. Thomson; Au Bureau du Trésorier de la Société Entomologique de France Place of publishing: Paris, France, 1858; Volume 2, pp. 377–445. [Google Scholar]
- de Brito Capello, F. Especies novas ou pouco conhecidas d’arachnidios d’Africa occidental. J. Sci. Math. Phys. Naturaes 1866, 1, 79–88. [Google Scholar]
- Karsch, F. West-afrikanische Arachniden, gesammelt von Herrn Stabsarzt Dr. Falkenstein. Z. Gesammten Naturwissenschaften 1879, 52, 329–373. [Google Scholar]
- Thorell, T. Araneae Camerunenses (Africae occidentalis) quas anno 1891 collegerunt Cel. Dr Y. Sjöstedt aliique. Bih. Till Kongliga Sven. Vetensk.-Akad. Handl. 1899, 25, 1–105. [Google Scholar]
- Pocock, R.I. On the scorpions, pedipalps and spiders from tropical West-Africa, represented in the collection of the British Museum. Proc. Zool. Soc. Lond. 1900, 67, 833–885. [Google Scholar]
- Simon, E. Arachnides recueillis par L. Fea sur la côte occidentale d’Afrique. 1re partie. Ann. Mus. Civ. Stor. Nat. Genova 1907, 43, 218–323. [Google Scholar]
- Roewer, C.F. Katalog der Araneae von 1758 bis 1940. 1. Band (Mesothelae, Orthognatha, Labidognatha: Dysderaeformia, Scytodiformia, Pholciformia, Zodariiformia, Hersiliaeformia, Argyopiformia); Natura, Buchhandlung für Naturkunde und Exakte Wissenschaften Paul Budy Place of Publishing: Bremen, Germany, 1942; p. 1040. [Google Scholar]
- Grasshoff, M. Die Radnetzspinnen-Gattung Caerostris (Arachnida: Araneae). Rev. Zool. Afr. 1984, 98, 725–765. [Google Scholar]
- Dippenaar-Schoeman, A.S.; Haddad, C.R.; Foord, S.H.; Lotz, L.N.; Webb, P. The Araneidae of South Africa. Version 2: Part 2 (E-Ne); South African National Survey of Arachnida Photo Identification Guide, Irene; 2022; p. 64. Available online: https://zenodo.org/records/6619195 (accessed on 14 July 2025).
- Kuntner, M.; Haddad, C.R.; Aljančič, G.; Blejec, A. Ecology and Web Allometry of Clitaetra irenae, an Arboricolous African Orb-Weaving Spider. J. Arachnol. 2008, 36, 583–594. [Google Scholar] [CrossRef]
- Gregorič, M.; Agnarsson, I.; Blackledge, T.A.; Kuntner, M. Darwin’s Bark Spider: Giant Prey in Giant Orb Webs (Caerostris darwini, Araneae: Araneidae)? J. Arachnol. 2011, 39, 287–295. [Google Scholar] [CrossRef]
- Simon, E. Histoire Naturelle des Araignées. Deuxième Édition, Tome Premier; Roret: Paris, France, 1895; pp. 761–1084. [Google Scholar]
- Almquist, S. Swedish Araneae, part 1—families Atypidae to Hahniidae (Linyphiidae excluded). Insect Syst. Evol. Suppl. 2005, 62, 1–284. [Google Scholar]
- Barrion, A.T.; Litsinger, J.A. Riceland Spiders of South and Southeast Asia; University Pres: Cambridge, UK, 1995. [Google Scholar]
- Biswas, V.; Raychaudhuri, D. A new record of the orb-weaving spider genus Parawixia FO Pickard Cambridge, 1904 from Bangladesh (Araneae: Araneidae). Serket 2017, 15, 113–118. [Google Scholar]
- Joseph, M.M.; Framenau, V.W. Systematic review of a new orb-weaving spider genus (Araneae: Araneidae), with special reference to the Australasian-Pacific and South-East Asian fauna. Zool. J. Linn. Soc. 2012, 166, 279–341. [Google Scholar] [CrossRef]
- Mcheidze, T.S. Spiders of Georgia: Systematics, Ecology, Zoogeographic Review; Tbilisi University: Tbilisi, Georgia, 1997; 390p. [Google Scholar]
- Roberts, M.J. Collins Field Guide: Spiders of Britain & Northern Europe; HarperCollins: London, UK, 1995; 383p. [Google Scholar]
- Roy, T.K.; Saha, S.; Raychaudhuri, D. On the araneid fauna (Araneae: Araneidae) of the tea estates of Dooars, West Bengal, India. World Sci. News 2017, 67, 1–67. [Google Scholar]
- Sen, S.; Dhali, D.C.; Saha, S.; Raychaudhuri, D. Spiders (Araneae: Arachnida) of reserve forests of Dooars: Gorumara National Park, Chapramari Wildlife Sanctuary and Mahananda Wildlife Sanctuary. World Sci. News 2015, 20, 1–339. [Google Scholar]
- Thorell, T. Studi sui ragni Malesi e Papuani. IV, 1. Ann. Mus. Civ. Stor. Nat. Genova 1890, 28, 5–421. [Google Scholar]
- Tikader, B.K. Part 1. Family Araneidae (=Argiopidae). Typical orb-weavers. In The Fauna of India. Spiders: Araneae. Vol. II; Zoological Survey of India: Calcutta, India, 1982; pp. 1–293. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).