Abstract
COVID-19 affects brain function, as deduced by the “brain fog” that is often encountered in COVID-19 patients and some cognitive impairment that is observed in many a patient in the post-COVID-19 period. Approximately one-third of patients, even when they have recovered from the acute somatic disease, continue to show posttraumatic stress disorder (PTSD) symptoms. We hypothesized that the persistent changes induced by COVID-19 on brain structure would overlap with those associated with PTSD. We performed a thorough PubMed search on 25 April 2023 using the following strategy: ((posttraumatic OR PTSD) AND COVID-19 AND (neuroimaging OR voxel OR VBM OR freesurfer OR structural OR ROI OR whole-brain OR hippocamp* OR amygd* OR “deep gray matter” OR “cortical thickness” OR caudate OR striatum OR accumbens OR putamen OR “regions of interest” OR subcortical)) OR (COVID-19 AND brain AND (voxel[ti] OR VBM[ti] OR magnetic[ti] OR resonance[ti] OR imaging[ti] OR neuroimaging[ti] OR neuroimage[ti] OR positron[ti] OR photon*[ti] OR PET[ti] OR SPET[ti] OR SPECT[ti] OR spectroscop*[ti] OR MRS[ti])), which produced 486 records and two additional records from other sources, of which 36 were found to be eligible. Alterations were identified and described and plotted against the ordinary PTSD imaging findings. Common elements were hypometabolism in the insula and caudate nucleus, reduced hippocampal volumes, and subarachnoid hemorrhages, while white matter hyperintensities were widespread in both PTSD and post-COVID-19 brain infection. The comparison partly supported our initial hypothesis. These data may contribute to further investigation of the effects of long COVID on brain structure and function.
1. Introduction
The Coronavirus Disease-2019 (COVID-19) pandemic, first declared on 11 March 2020 [1], has been associated with 687,021,745 infections worldwide up to now and 6,863,517 deaths (approximately 1% mortality), with deaths peaking and plateauing between April 2020 and April 2022 and new cases peaking between January and April 2022 [2]. A vast majority of patients infected with the Coronavirus recovered, but some of them—in the range of 6.2% [3] to 45% [4]—go on to develop long COVID. Of them, approximately 30% develop frank posttraumatic stress disorder (PTSD) or PTSD symptoms [5,6], but figures of approximately 50% have also been reported [7]. We suspected that the coronavirus somehow induces brain morphological and functional abnormalities that could be similar to those that patients with PTSD exhibit upon neuroimaging. These could either precede (creating vulnerability towards the development of PTSD) or follow the establishment of the disorder. To date, some specific brain alterations have been shown to accompany PTSD. Brain structure and function alterations have been hypothesized to occur after exposure to traumatic exposure and condition subsequent brain maturational processes, thus pointing to a neurodevelopmental origin of trauma-related pathology [8]. PTSD youths were shown to significantly increase hippocampal activation in response to threatening images compared to typically developing youth; also, patients with pediatric PTSD with a remitting condition show increasing functional connectivity (FC) between the hippocampus and visual cortex while viewing threat stimuli. The increased hippocampal activation in response to threat and the decreased FC in the hippocampal–visual cortex 4 network could be one of the reasons why PTSD persists in a pediatric population [9]. Persistence in youth could be also attributable to atypical insular neurodevelopment since the insula is expected to increase across development and consequent brain maturation; the failure to do so, as shown with longitudinally employed magnetic resonance imaging (MRI), renders the affected people resistant to treatment [10].
Multiple factors have concurred with the determination of the traumatic impact of contracting COVID-19 [11], and social factors like the presentation of the pandemic by social and mass media cannot be ignored [12]. Although much work has focused on demonstrating the traumatic meaning of contracting COVID-19 and long COVID [13,14], to date, no study has focused on the neuroimaging cross-section of trauma and COVID-19. Establishing the existence of a common cross-section between the neuroimaging underpinnings of trauma and COVID-19 would pave the way to treatments sharing common elements in dealing with both groups of patients.
1.1. Studies of the Effects of PTSD on Brain Structure and Function
Other nuclei that have been involved with the development of PTSD are the amygdala, hippocampus, rostral anterior cingulate cortex (ACC), and ventromedial prefrontal cortex (PFC). Results generally indicate that smaller hippocampal volumes are associated with PTSD [15], but it is not known whether this is a cause or effect (it may well be both); the evidence for rostral ACC, ventromedial PFC, and amygdala is less clear or strong [16]. Youths with PTSD showed reduced gray matter (GM) volumes in the right ventromedial PFC and bilateral ventrolateral PFC, whereas they showed increases in the dorsolateral PFC; this is different from typically developing youths, who show maturation-related decreases in dorsolateral PFC GM volumes with decreased PFC–amygdalar FC and PFC–hippocampal FC [17]. Such findings may relate to alterations that are not specific to PTSD but are shared by other comorbid psychiatric disorders [18].
We especially focused on the neuroimaging findings of patients with neurological symptoms of any origin. Neurological symptoms may not be the result of a viral invasion of the brain or viral replication in the brain [19], but they are compatible with immunological reactivity [20], possibly due to an “original antigenic sin” [21] leading to “immunological imprinting”, i.e., to the tendency of the immune system to respond to antigenic challenges the same way it responded previously (this holds true for any biological system and constitutes the core element of learning and adaptive responses); this could lead to increased antibodies against other viruses and lower responses to COVID-19 [22], something that has been observed in some individuals with COVID-19 [23,24]. It has been recently supported that the original antigenic sin could be responsible for shaping the humoral immune response to COVID-19 and may be related to the development of neurological symptoms in patients with long COVID [25]. Immune dysregulation in COVID-19 could lead to the commonly observed microbleeding and endotheliitis that involve various organs, including the brain [26]; the finding of immunologically-induced endothelial alterations is universal in the brains of patients who died during their COVID-19 infection [27].
1.2. Aim
Having hypothesized that the brain alterations that are associated with PTSD would overlap with COVID-19–induced brain alterations, we were prompted to seek literature reporting on the neuroimaging findings of patients who had recently suffered from COVID-19. We here systematically review this evidence.
2. Materials and Methods
To review systematically the neuroimaging findings of the brain alterations in patients who either currently or previously had COVID-19, we carried out a PubMed search using the following search strategy: ((posttraumatic OR PTSD) AND COVID-19 AND (neuroimaging OR voxel OR VBM OR freesurfer OR structural OR ROI OR whole-brain OR hippocamp* OR amygd* OR “deep gray matter” OR “cortical thickness” OR caudate OR striatum OR accumbens OR putamen OR “regions of interest” OR subcortical)) OR (COVID-19 AND brain AND (voxel[ti] OR VBM[ti] OR magnetic[ti] OR resonance[ti] OR imaging[ti] OR neuroimaging[ti] OR neuroimage[ti] OR positron[ti] OR photon*[ti] OR PET[ti] OR SPET[ti] OR SPECT[ti] OR spectroscop*[ti] OR MRS[ti])). The choice of the above search strategy was based on the need to be omnicomprehensive; all authors added their expertise to refine the search. Inclusion criteria were having performed structural neuroimaging and reporting data. MRI (either voxel-based morphometry (VBM) or regions-of-interest (ROI) approaches), positron emission tomography (PET), single-photon emission computerized tomography (SPECT), and computerized tomography (CT) were considered appropriate. Functional MRI (fMRI) or FC studies were considered when the aforementioned techniques were involved, but they were excluded if they included only examination. Excluded were also case reports or case series; opinion papers, such as editorials or letters to the editor; comments on other articles; duplicates; article corrections referring to an article that was already present in the search; articles unrelated to what we were searching for; unfocused or inadequate designs (inadequate for our purposes); animal or in vitro studies; papers not reporting on COVID-19 patients; articles of side effects of COVID-19 vaccination (labeled as unfocused); protocols, which usually do not report data but pave the way for future studies (however, if reporting preliminary data, they were taken into consideration); exclusively post-mortem studies (but if they reported on brain pathological findings of patients dying during their COVID-19 infection, they were considered and included); studies containing no neuroimaging data; and reviews, but the latter were downloaded and hand-searched for possible additional eligible references that could have eluded our search.
After completing the search, we labeled all resulting records according to whether they were to be included or excluded (Supplementary Table S1). The principal reason for exclusion was provided for each article (Supplement). In our review, we followed the 2020 PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) Statement indications [28]. In Figure 1, we display the PRISMA flow diagram. We provide the 2020 PRISMA Checklist in the Online Supplement (Supplementary Table S2). Thus, the labels for exclusion were the following: Unrelated, Review, Unfocused, Case reports/series, No COVID-19, No neuroimaging, Unsuitable data presentation, Opinion, Protocol, Animal, Functional connectivity only, fMRI only, Duplicate, In vitro, and Post-mortem.
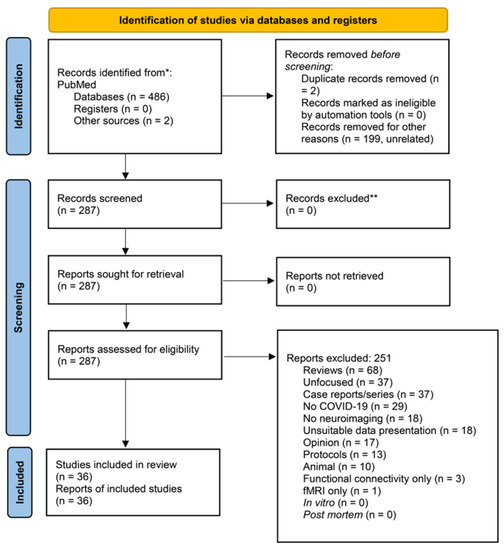
Figure 1.
PRISMA-flow diagram of our search with exclusion criteria specified. PRISMA 2020 flow diagram for new systematic reviews which included searches of databases and registers only ((posttraumatic OR PTSD) AND COVID-19 AND (neuroimaging OR voxel OR VBM OR freesurfer OR structural OR ROI OR whole-brain OR hippocamp* OR amygd* OR “deep gray matter” OR “cortical thickness” OR caudate OR striatum OR accumbens OR putamen OR “regions of interest” OR subcortical)) OR (COVID-19 AND brain AND (voxel[ti] OR VBM[ti] OR magnetic[ti] OR resonance[ti] OR imaging[ti] OR neuroimaging[ti] OR neuroimage[ti] OR positron[ti] OR photon*[ti] OR PET[ti] OR SPET[ti] OR SPECT[ti] OR spectroscop*[ti] OR MRS[ti])) PubMed, 29 April 2023, 486 articles + 2 from other sources * Consider, if feasible to do so, reporting the number of records identified from each database or register searched (rather than the total number across all databases/registers). ** If automation tools were used, indicate how many records were excluded by a human and how many were excluded by automation tools. From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi:10.1136/bmj.n71. For more information, visit: http://www.prisma-statement.org/.
Three authors independently conducted the agreed search and compared their results. Eligibility was based on being an original study on patients who had contracted COVID-19 or who had a current COVID-19 infection, including humans, that provided data on patients’ neuroimaging. To establish eligibility for each study, all authors ran Delphi rounds until a full consensus was reached. All studies were downloaded with the exception of manifestly unrelated studies. Thereafter, all titles were introduced into our database where their characteristics were defined.
To assess the quality of eligible studies, we used the appraisal tool for cross-sectional studies (AXIS) [29] on each cross-sectional included study. The results of the assessment are shown in Supplementary Table S3.
There were no ethical concerns to address or ethical committee approval to obtain. The authors adhered to the principles of the WMA Helsinki Declaration of Human Rights and its subsequent amendments in their handling of information related to the patients involved in the eligible studies.
3. Results
On 29 April 2023, our above PubMed search yielded 486 articles; 2 more were obtained from the references of the obtained literature. Eligible were 36 studies (Figure 1; Table 1) [30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65]. These studies found a multitude of brain alterations with and after COVID-19. Most frequent were hypometabolism in the frontal cortex, ACC, insula, and caudate nucleus performed with 18F-fluorodeoxyglucose-PET/computerized tomography (8 studies), hemorrhages (nontraumatic subdural hemorrhages, nonaneurysmal subarachnoid hemorrhages, microhemorrhages, large parenchymal hemorrhage) (7 studies), hypoxic changes (7 studies), and supratentorial, middle cerebellar peduncular subcortical, periventricular, and deep white matter lesions (7 studies). Details are shown in Table 1.

Table 1.
Summary of studies (in chronological order) on the neuroimaging of patients with COVID-19.
Of the 36 eligible studies, the majority (N = 23) were single-site, while 11 were multicenter (5 2-center, 2 3-center, one each 4-, 5- and 6-center, and 1 11-center); one used BioBank data across the entire United Kingdom (UK) and another was an international call on behalf of the American Society of Pediatric Neuroradiology (ASPN), which recruited from 10 countries. As is currently fashionable, no multicenter study reported on intersite differences. The majority of studies were conducted in the United States (USA) (N = 9, while a tenth study participated in a 5-center/4-country study), 7 were conducted in France, 2 were conducted in Italy (plus 1 in the aforementioned 5-center/4-country study), 2 were conducted in Sweden, 2 were conducted in Brazil (plus 1 in the 5-center/4-country study), 2 were conducted in the UK, 2 were conducted in Turkey, 2 were conducted in China, 1 was conducted in Spain (plus 1 in the 5-center/4-country study), 1 was conducted in the Netherlands, 1 was conducted in Switzerland, and the international call of the ASPN resulted in recruitment from France, the UK, the USA, Brazil, Argentina, India, Peru, and Saudi Arabia. Location did not affect study quality (AXIS assessment, Supplementary Table S3). Details about locations and correspondence with eligible studies are shown in Supplementary Table S4.
Neuroimaging in these studies was generally arranged according to patient needs and correctly started first with a computerized tomography (CT) scan, identifying patients with putative abnormalities, and progressing thereafter to more sophisticated and in-depth investigation methods like MRI and PET. MRI used more commonly 3Tesla instrumentation (6 studies) and less frequently 1.5Tesla apparatuses, indicating a transition in instrumentation use towards upgraded tools. We did not exclude articles including 1.5Tesla apparatuses as the studies using them still provide valid results. Seven studies used PET with 2-deoxy-2-[fluorine-18]fluoro-D-glucose, four studies combined it with MRI, and three used it alone. The studies included here [30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65] identified brain alterations in a subset of patients—those with identifiable alterations—and classified them roughly, without focusing on GM- or WM-specific alterations and brain nuclei/area volumes. In contrast, studies on brain consequences or correlates of PTSD [8,9,10,15] were more detailed as concerns structural and functional alterations in patients with PTSD. The findings of the studies included in this review only partially overlapped with those already reported in the literature for PTSD. Therefore, we started from the reported alterations in the brains of patients with COVID-19, attempting to identify such alterations in the PTSD literature. The evidence will be presented in the discussion that follows.
4. Discussion
In this review, we included studies reporting brain alterations in patients with recent or current COVID-19 infections; these included patients with long COVID, whom we expected to manifest PTSD symptoms. However, just one study reported PTSD symptoms [50], probably due to the lack of application of instruments that could reliably identify PTSD symptoms in most studies. The most frequently reported alterations were hypometabolism in the frontal cortex, ACC, insula, and caudate nucleus; hemorrhages, which all too frequently were microbleeds; hypoxia; and supratentorial, middle cerebellar, peduncular subcortical, periventricular, and deep white matter lesions. Surprisingly, there was not much reference to the hippocampus (only 4 of the 36 eligible studies), but studies reporting on the hippocampus identified hippocampal abnormalities. Results were not meta-analyzable due to extreme heterogeneity due to the variety of objectives and aims of each research team. Focusing on only methodologically consistent studies would have resulted in including a very reduced number of articles that would have been unreviewable.
By performing specific PubMed searches, we identified one study identifying frontal cortex hypometabolism in torture victims with PTSD [66] and two which found hypometabolism in the insula [66,67], the latter of which included one patient with PTSD following domestic violence and one finding moderate hypometabolism in the caudate nucleus [66]. The latter study also identified hippocampal volume reductions.
Subarachnoid hemorrhages are related to PTSD. People with a subarachnoid hemorrhage are more likely to develop PTSD in a Chinese population [68] and the subarachnoid hemorrhage population shows more PTSD than other populations, but the time course is different among individual patients [69]. Post-ictus patients have a >33% chance to develop PTSD [70,71]. However, there were no studies investigating radiologically present hemorrhages, macro-, or microbleeds in patients with PTSD. At any rate, the above studies do show a link between brain hemorrhages and PTSD, but they do not indicate the direction of this link. While it is probable that one-third of patients suffering these hemorrhages will go on to develop PTSD, it is not known whether patients with PTSD will develop intracranial hemorrhages.
Hypoxia is a factor in PTSD allowing memory loss of a traumatic event in PTSD patients [72]. Post-avalanche survivors develop PTSD in approximately 11% of cases and it is presumable, but not demonstrated, that suffering hypoxia is a factor in this development [73]. Most studies investigating hypoxia in PTSD were animal studies; the directionality of hypoxia in PTSD has not been demonstrated and no study has investigated radiological signs of hypoxia in PTSD patients.
Many studies investigated white matter integrity and identified diffuse hyperintensities in patients with PTSD. These studies employed more sophisticated techniques, like diffusion tensor imaging, while, among our eligible studies, few of them used it [50,60]. One study observed increased fractional anisotropy in multiple white matter tracts in patients with PTSD compared with controls subjected to trauma that did not develop PTSD [74]. Another study found reduced fractional anisotropy in patients with PTSD undergoing trauma-focused cognitive behavior therapy to correlate with dysphoric symptom reduction [75]. Still another study identified white matter abnormalities in patients with PTSD, i.e., reduced fractional anisotropy and increased radial diffusivity in white matter tracts like the corpus callosum, the external and internal capsules, cingulum, and inferior and superior longitudinal fasciculi [76], all findings shared also by the COVID-19 patients of our review. A role of trauma in white matter disruption may be suspected, inasmuch as both patients with PTSD and non-PTSD trauma-exposed individuals showed increased fractional anisotropy in white matter tracts like the anterior limb of the internal capsule, the forceps of the corpus callosum, and the corona radiata compared to a healthy control group [77]. Decreased baseline fractional anisotropy was confirmed by a Chinese study in the right cingulate gyrus, uncinate fasciculus, superior longitudinal fasciculus, corticospinal tract, inferior fronto-occipital fasciculus, inferior longitudinal fasciculus, and forceps major for parents having lost their only child, but it did not persist at follow-up, with PTSD progressively resolving [78]. Significant negative correlations between PTSD symptom severity and fractional anisotropy values were found in the left corticospinal tract and left inferior cerebellar peduncle [79], matching some of the results of our review. The most recent study on this subject reported reduced fractional anisotropy and increased radial diffusivity in patients with PTSD or mild traumatic brain injury who displayed psychological symptoms [80]. Making sense of all these findings, white matter alterations in PTSD match white matter alterations that accompany COVID-19.
Limitations
This review has not been registered to PROSPERO. Furthermore, due to design heterogeneity, we could not perform meta-analyses or assess the risk of bias of each included study. However, we used the AXIS instrument to assess the quality of the cross-sectional studies we included [29] (although there were studies with longitudinal aspects, none was fully prospective longitudinal) and found the mean quality to be medium-high. Due to the restricted time of the COVID-19 pandemic and the recent time period involved in these studies, this was to be expected (i.e., recent studies are expected to be more methodologically sound). It should also be stressed that some of the alterations that we found to be shared among PTSD patients and survivors of COVID-19 infection (PFC, ACC, insula, and hippocampus) may also be found in patients with emotional disturbances, like anxiety and depressive disorders [81,82,83,84,85], and even in severe psychotic disorders, like schizophrenia [86]. With these disturbances and disorders all having a shared background with childhood trauma and adversity [87,88,89], it is almost impossible to disentangle what is specific to PTSD and COVID-19 and what could be nonspecific.
The lack of prospective longitudinal studies does not allow strong conclusions to be drawn about predictors and risk factors, but, in COVID-19 times, cross-sectional studies on the particular issue we examined were all that we could obtain. Despite these limitations, the included studies allowed us to draw some conclusions that could constitute the basis of future studies.
Summarizing, of the brain alterations we identified in COVID-19 patients, some match the alterations encountered in PTSD and for some others, which are the most frequently observed changes in this review, the evidence is weaker.
5. Conclusions
Taking together all the evidence of brain alterations in COVID-19 and in PTSD, they overlap only partially, thus partially backing our hypothesis. One-third of patients with COVID-19 who developed PTSD could likely find a neurobiological basis of COVID-19-induced brain alterations. Since neuroinflammatory and immune reactivity mechanisms were advocated for the neurological symptoms of COVID-19, studies focusing on neuroinflammation and immunity are warranted. Such studies, despite appearing in the late nineties, are still scarce. It is important to continue to study this issue since it could open-up new ways of treating both PTSD cases and people who have suffered COVID-19 by enforcing treatment programs shared by both populations.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jpm13071140/s1, Table S1: Studies included vs. excluded with reasons for exclusion for each study resulting in our search strategy; Table S2: PRISMA Checklist; Table S3: Quality assessment through AXIS. [Bracketed numbers] refer to papers’ References; N/A, not applicable; Table S4: Locations of the eligible studies.
Author Contributions
G.D.K. and A.S. Conceptualization; O.M.F., S.M., V.I., A.R., E.B., A.F. and A.C. methodology; L.M., D.P.R.C., A.S. and G.D.K. software and validation; G.D.K., A.S., O.M.F., S.M., V.I., A.R. and E.B. formal analysis, investigation, and writing—original draft preparation; G.D.K., O.M.F., S.M., A.S. and G.S. supervised the writing of the review; G.D.K., O.M.F., S.M., A.S. and G.S. resources, data curation, writing—review and editing, and supervision; A.S. and G.S. visualization, supervision, project administration, Nobody data acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in the article and Supplementary Materials.
Acknowledgments
The authors are grateful to the Scientific Administration of the Bibliographic and Bibliometric Support Service, Fondazione Policlinico A. Gemelli IRCCS, in particular, Maria Pattuglia, Mimma Ariano, Ales Casciaro, Teresa Prioreschi, and Susanna Rospo, and the librarians of the Sant’Andrea Hospital, Faculty of Medicine and Psychology, Sapienza University of Rome, for rendering precious bibliographical material accessible. They all have consented to the acknowledgment.
Conflicts of Interest
The authors declare no conflict of interest.
References
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—11 March 2020. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 29 April 2023).
- Worldometer. COVID-19 Coronavirus Pandemic. Available online: https://www.worldometers.info/coronavirus/ (accessed on 29 April 2023).
- Global Burden of Disease Long COVID Collaborators; Wulf Hanson, S.; Abbafati, C.; Aerts, J.G.; Al-Aly, Z.; Ashbaugh, C.; Ballouz, T.; Blyuss, O.; Bobkova, P.; Bonsel, G.; et al. Estimated global proportions of individuals with persistent fatigue, cognitive, and respiratory symptom clusters following symptomatic COVID-19 in 2020 and 2021. JAMA 2022, 328, 1604–1615. [Google Scholar] [CrossRef] [PubMed]
- O’Mahoney, L.L.; Routen, A.; Gillies, C.; Ekezie, W.; Welford, A.; Zhang, A.; Karamchandani, U.; Simms-Williams, N.; Cassambai, S.; Ardavani, A.; et al. The prevalence and long-term health effects of Long COVID among hospitalised and non-hospitalised populations: A systematic review and meta-analysis. EClinicalMedicine 2022, 55, 101762, Erratum in EClinicalMedicine 2023, 59, 101959. [Google Scholar] [CrossRef] [PubMed]
- Janiri, D.; Carfì, A.; Kotzalidis, G.D.; Bernabei, R.; Landi, F.; Sani, G.; Gemelli against COVID-19 Post-Acute Care Study Group. Posttraumatic stress disorder in patients after severe COVID-19 infection. JAMA Psychiatry 2021, 78, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Grover, S.; Sahoo, S.; Mishra, E.; Gill, K.S.; Mehra, A.; Nehra, R.; Suman, A.; Bhalla, A.; Puri, G.D. Fatigue, perceived stigma, self-reported cognitive deficits and psychological morbidity in patients recovered from COVID-19 infection. Asian J. Psychiatr. 2021, 64, 102815. [Google Scholar] [CrossRef]
- Jakhar, K.; Lone, K.Y.; Gupta, R.; Srivastava, A.; Solanki, H.K. A cross-sectional study on the psychological impact of stress, anxiety and depressive symptoms in COVID-19 patients. Cureus 2022, 14, e27724. [Google Scholar] [CrossRef]
- Weems, C.F.; Russell, J.D.; Neill, E.L.; McCurdy, B.H. Annual Research Review: Pediatric posttraumatic stress disorder from a neurodevelopmental network perspective. J. Child. Psychol. Psychiatry 2019, 60, 395–408. [Google Scholar] [CrossRef]
- George, G.C.; Keding, T.J.; Heyn, S.A.; Herringa, R.J. Longitudinal hippocampal circuit change differentiates persistence and remission of pediatric posttraumatic stress disorder. Depress. Anxiety 2022, 39, 8–18. [Google Scholar] [CrossRef]
- Zantvoord, J.B.; Zhutovsky, P.; Ensink, J.B.M.; Op den Kelder, R.; van Wingen, G.A.; Lindauer, R.J.L. Trauma-focused psychotherapy response in youth with posttraumatic stress disorder is associated with changes in insula volume. J. Psychiatr. Res. 2021, 132, 207–214. [Google Scholar] [CrossRef]
- Kordi, A.; Sarabi-Jamab, A.; Shariat, S.V.; Rezaee, N.; Shariati, B.; Faiz, S.H.R.; Mirfazeli, F.S. Higher anxiety and perceived trauma among COVID-19 patients: A prospective comparative study. BMC Psychiatry 2023, 23, 100. [Google Scholar] [CrossRef]
- Liu, C.; Liu, Y. Media Exposure and anxiety during COVID-19: The mediation effect of media vicarious traumatization. Int. J. Environ. Res. Public. Health 2020, 17, 4720. [Google Scholar] [CrossRef]
- Somma, A.; Krueger, R.F.; Markon, K.E.; Gialdi, G.; Colanino, M.; Ferlito, D.; Liotta, C.; Fossati, A. A nine-month study on the course of COVID-19 related perceived post-traumatic stress disorder among Italian community-dwelling adults. J. Psychiatr. Res. 2021, 141, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Jeftić, A.; Ikizer, G.; Tuominen, J.; Chrona, S.; Kumaga, R. Connection between the COVID-19 pandemic, war trauma reminders, perceived stress, loneliness, and PTSD in Bosnia and Herzegovina. Curr. Psychol. 2023, 42, 8582–8594. [Google Scholar] [CrossRef] [PubMed]
- Logue, M.W.; van Rooij, S.J.H.; Dennis, E.L.; Davis, S.L.; Hayes, J.P.; Stevens, J.S.; Densmore, M.; Haswell, C.C.; Ipser, J.; Koch, S.B.J.; et al. Smaller hippocampal volume in posttraumatic stress disorder: A multisite ENIGMA-PGC study: Subcortical volumetry results from Posttraumatic Stress Disorder Consortia. Biol. Psychiatry 2018, 83, 244–253. [Google Scholar] [CrossRef]
- Henigsberg, N.; Kalember, P.; Petrović, Z.K.; Šečić, A. Neuroimaging research in posttraumatic stress disorder—Focus on amygdala, hippocampus and prefrontal cortex. Prog. Neuropsychopharmacol. Biol. Psychiatry 2019, 90, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Heyn, S.A.; Keding, T.J.; Ross, M.C.; Cisler, J.M.; Mumford, J.A.; Herringa, R.J. Abnormal prefrontal development in pediatric posttraumatic stress disorder: A longitudinal structural and functional magnetic resonance imaging study. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2019, 4, 171–179. [Google Scholar] [CrossRef]
- Kroes, M.C.; Rugg, M.D.; Whalley, M.G.; Brewin, C.R. Structural brain abnormalities common to posttraumatic stress disorder and depression. J. Psychiatry Neurosci. 2011, 36, 256–265. [Google Scholar] [CrossRef]
- Aschman, T.; Mothes, R.; Heppner, F.L.; Radbruch, H. What SARS-CoV-2 does to our brains. Immunity 2022, 55, 1159–1172. [Google Scholar] [CrossRef]
- Matschke, J.; Lütgehetmann, M.; Hagel, C.; Sperhake, J.P.; Schröder, A.S.; Edler, C.; Mushumba, H.; Fitzek, A.; Allweiss, L.; Dandri, M.; et al. Neuropathology of patients with COVID-19 in Germany: A post-mortem case series. Lancet Neurol. 2020, 19, 919–929. [Google Scholar] [CrossRef]
- Francis, T.J. On the doctrine of original antigenic sin. Proc. Am. Philos. Soc. 1960, 104, 572–578. [Google Scholar]
- Koutsakos, M.; Ellebedy, A.H. Immunological imprinting: Understanding COVID-19. Immunity 2023, 56, 909–913. [Google Scholar] [CrossRef]
- Reincke, S.M.; Prüss, H.; Wilson, I.A.; Kreye, J. Antigenic imprinting in SARS-CoV-2. Clin. Transl. Med. 2022, 12, e923. [Google Scholar] [CrossRef]
- King, S.M.; Bryan, S.P.; Hilchey, S.P.; Wang, J.; Zand, M.S. First impressions matter: Immune imprinting and antibody cross-reactivity in influenza and SARS-CoV-2. Pathogens 2023, 12, 169. [Google Scholar] [CrossRef]
- Spatola, M.; Dinoto, A.; Bozzetti, S.; Chiodega, V.; Ferrari, S.; Mariotto, S.; Alter, G. Presentation S21: Serum and cerebrospinal fluid antibody signatures track with outcome of neurologic post-acute sequelae of SARS-Cov-2 Infection (NeuroPASC) (Congress Abstract) Acute and Post-Acute Sequelae of COVID-19. In Proceedings of the American Academy of Neurology 75th Annual Meeting, Boston Convention and Exhibition Center (BCEC), Boston, MA, USA, 22–27 April 2023. [Google Scholar]
- Wenzel, J.; Lampe, J.; Müller-Fielitz, H.; Schuster, R.; Zille, M.; Müller, K.; Krohn, M.; Körbelin, J.; Zhang, L.; Özorhan, Ü.; et al. The SARS-CoV-2 main protease Mpro causes microvascular brain pathology by cleaving NEMO in brain endothelial cells. Nat. Neurosci. 2021, 24, 1522–1533. [Google Scholar] [CrossRef]
- Lee, M.H.; Perl, D.P.; Steiner, J.; Pasternack, N.; Li, W.; Maric, D.; Safavi, F.; Horkayne-Szakaly, I.; Jones, R.; Stram, M.N.; et al. Neurovascular injury with complement activation and inflammation in COVID-19. Brain 2022, 145, 2555–2568. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Downes, M.J.; Brennan, M.L.; Williams, H.C.; Dean, R.S. Development of a critical appraisal tool to assess the quality of cross-sectional studies (AXIS). BMJ Open 2016, 6, e011458. [Google Scholar] [CrossRef]
- Kremer, S.; Lersy, F.; Anheim, M.; Merdji, H.; Schenck, M.; Oesterlé, H.; Bolognini, F.; Messie, J.; Khalil, A.; Gaudemer, A.; et al. Neurologic and neuroimaging findings in patients with COVID-19: A retrospective multicenter study. Neurology 2020, 95, e1868–e1882. [Google Scholar] [CrossRef] [PubMed]
- Klironomos, S.; Tzortzakakis, A.; Kits, A.; Öhberg, C.; Kollia, E.; Ahoromazdae, A.; Almqvist, H.; Aspelin, Å.; Martin, H.; Ouellette, R.; et al. Nervous system involvement in Coronavirus Disease 2019: Results from a retrospective consecutive neuroimaging cohort. Radiology 2020, 297, E324–E334. [Google Scholar] [CrossRef]
- O’Shea, A.; Parakh, A.; Hedgire, S.; Lee, S.I. Multisystem assessment of the imaging manifestations of coagulopathy in hospitalized patients with coronavirus disease (COVID-19). AJR Am. J. Roentgenol. 2021, 216, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Lin, E.; Lantos, J.E.; Strauss, S.B.; Phillips, C.D.; Campion, T.R., Jr.; Navi, B.B.; Parikh, N.S.; Merkler, A.E.; Mir, S.; Zhang, C.; et al. Brain imaging of patients with COVID-19: Findings at an academic institution during the height of the outbreak in New York City. AJNR Am. J. Neuroradiol. 2020, 41, 2001–2008. [Google Scholar] [CrossRef] [PubMed]
- Sawlani, V.; Scotton, S.; Nader, K.; Jen, J.P.; Patel, M.; Gokani, K.; Denno, P.; Thaller, M.; Englezou, C.; Janjua, U.; et al. COVID-19-related intracranial imaging findings: A large single-centre experience. Clin. Radiol. 2021, 76, 108–116. [Google Scholar] [CrossRef]
- Lindan, C.E.; Mankad, K.; Ram, D.; Kociolek, L.K.; Silvera, V.M.; Boddaert, N.; Stivaros, S.M.; Palasis, S.; ASPNR PECOBIG Collaborator Group. Neuroimaging manifestations in children with SARS-CoV-2 infection: A multinational, multicentre collaborative study. Lancet Child. Adolesc. Health 2021, 5, 167–177. [Google Scholar] [CrossRef]
- Orman, G.; Desai, N.K.; Kralik, S.F.; Meoded, A.; Seghers, V.J.; Annapragada, A.V.; Huisman, T.A.G.M. Neuroimaging offers low yield in children positive for SARS-CoV-2. AJNR Am. J. Neuroradiol. 2021, 42, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Rapalino, O.; Pourvaziri, A.; Maher, M.; Jaramillo-Cardoso, A.; Edlow, B.L.; Conklin, J.; Huang, S.; Westover, B.; Romero, J.M.; Halpern, E.; et al. Clinical, imaging, and lab correlates of severe COVID-19 leukoencephalopathy. AJNR Am. J. Neuroradiol. 2021, 42, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Kas, A.; Soret, M.; Pyatigoskaya, N.; Habert, M.O.; Hesters, A.; Le Guennec, L.; Paccoud, O.; Bombois, S.; Delorme, C.; on behalf of the CoCo-Neurosciences Study Group and COVID SMIT PSL Study Group. The cerebral network of COVID-19-related encephalopathy: A longitudinal voxel-based 18F-FDG-PET study. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2543–2557. [Google Scholar] [CrossRef] [PubMed]
- Conklin, J.; Frosch, M.P.; Mukerji, S.S.; Rapalino, O.; Maher, M.D.; Schaefer, P.W.; Lev, M.H.; Gonzalez, R.G.; Das, S.; Champion, S.N.; et al. Susceptibility-weighted imaging reveals cerebral microvascular injury in severe COVID-19. J. Neurol. Sci. 2021, 421, 117308. [Google Scholar] [CrossRef] [PubMed]
- Guedj, E.; Campion, J.Y.; Dudouet, P.; Kaphan, E.; Bregeon, F.; Tissot-Dupont, H.; Guis, S.; Barthelemy, F.; Habert, P.; Ceccaldi, M.; et al. 18F-FDG brain PET hypometabolism in patients with long COVID. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2823–2833. [Google Scholar] [CrossRef]
- Büttner, L.; Bauknecht, H.C.; Fleckenstein, F.N.; Kahn, J.; Tietze, A.; Bohner, G.; Siebert, E. Neuroimaging findings in conjunction with severe COVID-19. Rofo 2021, 193, 822–829. [Google Scholar] [CrossRef]
- Thurnher, M.M.; Boban, J.; Röggla, M.; Staudinger, T. Distinct pattern of microsusceptibility changes on brain magnetic resonance imaging (MRI) in critically ill patients on mechanical ventilation/oxygenation. Neuroradiology 2021, 63, 1651–1658. [Google Scholar] [CrossRef]
- Agarwal, S.; Melmed, K.; Dogra, S.; Jain, R.; Conway, J.; Galetta, S.; Lewis, A. Increase in ventricle size and the evolution of white matter changes on serial imaging in critically ill patients with COVID-19. Neurocrit. Care 2021, 35, 491–500. [Google Scholar] [CrossRef]
- Mahammedi, A.; Ramos, A.; Bargalló, N.; Gaskill, M.; Kapur, S.; Saba, L.; Carrete, H., Jr.; Sengupta, S.; Salvador, E.; Hilario, A.; et al. Brain and lung imaging correlation in patients with COVID-19: Could the severity of lung disease reflect the prevalence of acute abnormalities on neuroimaging? A global multicenter observational study. AJNR Am. J. Neuroradiol. 2021, 42, 1008–1016. [Google Scholar] [CrossRef]
- Lambrecq, V.; Hanin, A.; Munoz-Musat, E.; Chougar, L.; Gassama, S.; Delorme, C.; Cousyn, L.; Borden, A.; Damiano, M.; Frazzini, V.; et al. Association of clinical, biological, and brain magnetic resonance imaging findings with electroencephalographic findings for patients with COVID-19. JAMA Netw. Open 2021, 4, e211489. [Google Scholar] [CrossRef]
- Yan, K.; Xiao, F.-F.; Jiang, Y.-W.; Xiao, T.-T.; Zhang, D.-J.; Yuan, W.-H.; Shao, J.-B.; Cheng, G.-Q.; Zeng, L.-K. Effects of SARS-CoV-2 infection on neuroimaging and neurobehavior in neonates. World J. Pediatr. 2021, 17, 171–179. [Google Scholar] [CrossRef]
- Uginet, M.; Breville, G.; Hofmeister, J.; Machi, P.; Lalive, P.H.; Rosi, A.; Fitsiori, A.; Vargas, M.I.; Assal, F.; Allali, G.; et al. Cerebrovascular complications and vessel wall imaging in COVID-19 encephalopathy-A pilot study. Clin. Neuroradiol. 2022, 32, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Jegatheeswaran, V.; Chan, M.W.K.; Chakrabarti, S.; Fawcett, A.; Chen, Y.A. Neuroimaging findings of hospitalized COVID-19 patients: A Canadian retrospective observational study. Can. Assoc. Radiol. J. 2022, 73, 179–186. [Google Scholar] [CrossRef]
- Tu, Y.; Zhang, Y.; Li, Y.; Zhao, Q.; Bi, Y.; Lu, X.; Kong, Y.; Wang, L.; Lu, Z.; Hu, L. Post-traumatic stress symptoms in COVID-19 survivors: A self-report and brain imaging follow-up study. Mol. Psychiatry 2021, 26, 7475–7480. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, F.; Palladini, M.; Paolini, M.; Melloni, E.; Vai, B.; De Lorenzo, R.; Furlan, R.; Rovere-Querini, P.; Falini, A.; Mazza, M.G. Brain correlates of depression, post-traumatic distress, and inflammatory biomarkers in COVID-19 survivors: A multimodal magnetic resonance imaging study. Brain Behav. Immun. Health 2021, 18, 100387. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.C.; Fujiki, R.H.M.; Glória, L.F.P.; Fragoso, D.C.; Pacheco, F.T.; Calvi, C.; Rosa-Júnior, M.; Junior, A.C.M.M.; Rocha, A.J.D. Neurological imaging findings in hospitalized COVID-19 patients: A retrospective observational study in two Brazilian reference centers. Arq. Neuropsiquiatr. 2022, 80, 490–496. [Google Scholar] [CrossRef]
- Nelson, D.W.; Granberg, T.; Andersen, P.; Jokhadar, E.; Kåhlin, J.; Granström, A.; Hallinder, H.; Schening, A.; Thunborg, C.; Walles, H.; et al. The Karolinska NeuroCOVID study protocol: Neurocognitive impairment, biomarkers and advanced imaging in critical care survivors. Acta Anaesthesiol. Scand. 2022, 66, 759–766. [Google Scholar] [CrossRef]
- Andriuta, D.; Si-Ahmed, C.; Roussel, M.; Constans, J.M.; Makki, M.; Aarabi, A.; Basille, D.; Andrejak, C.; Godefroy, O. Clinical and imaging determinants of neurocognitive disorders in post-acute COVID-19 patients with cognitive complaints. J. Alzheimers Dis. 2022, 87, 1239–1250. [Google Scholar] [CrossRef]
- Widemon, R.S.; Huang, S.; Capaccione, K.M.; Mitchell, R.P.; Salvatore, M.M.; Lignelli, A.; Nguyen, P. The impact of the COVID-19 pandemic on neuroimaging volume in New York City. Neuroradiol. J. 2022, 35, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Lersy, F.; Bund, C.; Anheim, M.; Mondino, M.; Noblet, V.; Lazzara, S.; Phillipps, C.; Collange, O.; Oulehri, W.; Mertes, P.M.; et al. Evolution of neuroimaging findings in severe COVID-19 patients with initial neurological impairment: An observational study. Viruses 2022, 14, 949. [Google Scholar] [CrossRef] [PubMed]
- de Paula, J.J.; Paiva, R.E.R.P.; Souza-Silva, N.G.; Rosa, D.V.; Duran, F.L.S.; Coimbra, R.S.; Costa, D.S.; Dutenhefner, P.R.; Oliveira, H.S.D.; Camargos, S.T.; et al. Selective visuoconstructional impairment following mild COVID-19 with inflammatory and neuroimaging correlation findings. Mol. Psychiatry 2023, 28, 553–563. [Google Scholar] [CrossRef]
- Callen, A.L.; Tanabe, J.; Thaker, A.A.; Pollard, R.; Sauer, B.; Jones, W.; Pattee, J.; Steach, B.; Timpone, V.M. Evaluation of cerebrovascular reactivity and vessel wall imaging in patients with prior COVID-19: A prospective case-control MRI study. AJR Am. J. Roentgenol. 2023, 220, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Díez-Cirarda, M.; Yus, M.; Gómez-Ruiz, N.; Polidura, C.; Gil-Martínez, L.; Delgado-Alonso, C.; Jorquera, M.; Gómez-Pinedo, U.; Matias-Guiu, J.; Arrazola, J.; et al. Multimodal neuroimaging in post-COVID syndrome and correlation with cognition. Brain 2022, 146, 2142–2152. [Google Scholar] [CrossRef] [PubMed]
- Goehringer, F.; Bruyere, A.; Doyen, M.; Bevilacqua, S.; Charmillon, A.; Heyer, S.; Verger, A. Brain 18F-FDG PET imaging in outpatients with post-COVID-19 conditions: Findings and associations with clinical characteristics. Eur. J. Nucl. Med. Mol. Imaging 2023, 50, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Paolini, M.; Palladini, M.; Mazza, M.G.; Colombo, F.; Vai, B.; Rovere-Querini, P.; Falini, A.; Poletti, S.; Benedetti, F. Brain correlates of subjective cognitive complaints in COVID-19 survivors: A multimodal magnetic resonance imaging study. Eur. Neuropsychopharmacol. 2023, 68, 1–10. [Google Scholar] [CrossRef]
- Kamasak, B.; Ulcay, T.; Nisari, M.; Gorgulu, O.; Akca, V.; Alpaslan, M.; Yetis, A.; Hizmali, L.; Karahocagil, M.K.; Aycan, K. Effects of COVID-19 on brain and cerebellum: A voxel based morphometrical analysis. Bratisl. Lek. Listy. 2023, 124, 442–448. [Google Scholar] [CrossRef]
- Klinkhammer, S.; Horn, J.; Duits, A.A.; Visser-Meily, J.M.A.; Verwijk, E.; Slooter, A.J.C.; Postma, A.A.; van Heugten, C.M.; NeNeSCo Study Group. Neurological and (neuro)psychological sequelae in intensive care and general ward COVID-19 survivors. Eur. J. Neurol. 2023, 30, 1880–1890. [Google Scholar] [CrossRef]
- Douaud, G.; Lee, S.; Alfaro-Almagro, F.; Arthofer, C.; Wang, C.; McCarthy, P.; Lange, F.; Andersson, J.L.R.; Griffanti, L.; Duff, E.; et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature 2022, 604, 697–707. [Google Scholar] [CrossRef]
- Burulday, V.; Muluk, N.B.; Akgül, M.H.; Sayar, M.S. Diffusion-weighted imaging measurements of central smell regions in COVID-19 patients: Insular gyrus, corpus amygdala, and thalamus. Eur. Rev. Med. Pharmacol. Sci. 2023, 27, 3201–3207. [Google Scholar] [CrossRef]
- Debs, P.; Khalili, N.; Solnes, L.; Al-Zaghal, A.; Sair, H.I.; Yedavalli, V.; Luna, L.P. Post-COVID-19 brain [18F] FDG-PET findings: A retrospective single-center study in the United States. AJNR Am. J. Neuroradiol. 2023, 44, 517–522. [Google Scholar] [CrossRef]
- Zandieh, S.; Bernt, R.; Knoll, P.; Wenzel, T.; Hittmair, K.; Haller, J.; Hergan, K.; Mirzaei, S. Analysis of the metabolic and structural brain changes in patients with Torture-Related Post-Traumatic Stress Disorder (TR-PTSD) using ¹⁸F-FDG PET and MRI. Medicine 2016, 95, e3387. [Google Scholar] [CrossRef] [PubMed]
- Stevanovic, D.; Brajkovic, L.; Srivastava, M.K.; Krgovic, I.; Jancic, J. Widespread cortical PET abnormalities in an adolescent related to a PNES dissociative state, PTSD, ADHD, and domestic violence exposure. Scand. J. Child. Adolesc. Psychiatr. Psychol. 2018, 6, 98–106. [Google Scholar] [CrossRef]
- Jiang, C. Posttraumatic stress disorder after a first-ever intracerebral hemorrhage in the Chinese population: A pilot study. Appl. Neuropsychol. Adult 2020, 27, 1–8. [Google Scholar] [CrossRef]
- Wajer, I.M.C.H.; Smits, A.R.; Rinkel, G.J.E.; van Zandvoort, M.J.E.; Wijngaards-de Meij, L.; Visser-Meily, J.M.A. Exploratory study of the course of posttraumatic stress disorder after aneurysmal subarachnoid hemorrhage. Gen. Hosp. Psychiatry 2018, 53, 114–118. [Google Scholar] [CrossRef]
- Noble, A.J.; Baisch, S.; Covey, J.; Mukerji, N.; Nath, F.; Schenk, T. Subarachnoid hemorrhage patients’ fears of recurrence are related to the presence of posttraumatic stress disorder. Neurosurgery 2011, 69, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Noble, A.J.; Baisch, S.; Mendelow, A.D.; Allen, L.; Kane, P.; Schenk, T. Posttraumatic stress disorder explains reduced quality of life in subarachnoid hemorrhage patients in both the short and long term. Neurosurgery 2008, 63, 1095–1104. [Google Scholar] [CrossRef]
- Layton, B.S.; Krikorian, R.; Dori, G.; Martin, G.A.; Wardi, K. Posttraumatic stress disorder with amnesia following asphyxiation. Ann. N. Y. Acad. Sci. 2006, 1071, 488–490. [Google Scholar] [CrossRef]
- Léonard, C.; Charriau-Perret, A.; Debaty, G.; Belle, L.; Ricard, C.; Sanchez, C.; Dupré, P.M.; Panoff, G.; Bougerol, T.; Viglino, D.; et al. Survivors of avalanche accidents: Posttraumatic stress disorder symptoms and quality of life: A multicentre study. Scand. J. Trauma. Resusc. Emerg. Med. 2021, 29, 96. [Google Scholar] [CrossRef] [PubMed]
- Siehl, S.; Wicking, M.; Pohlack, S.; Winkelmann, T.; Zidda, F.; Steiger-White, F.; King, J.; Burgess, N.; Flor, H.; Nees, F. Structural white and gray matter differences in a large sample of patients with Posttraumatic Stress Disorder and a healthy and trauma-exposed control group: Diffusion tensor imaging and region-based morphometry. Neuroimage Clin. 2020, 28, 102424. [Google Scholar] [CrossRef] [PubMed]
- Korgaonkar, M.S.; Felmingham, K.L.; Klimova, A.; Erlinger, M.; Williams, L.M.; Bryant, R.A. White matter anisotropy and response to cognitive behavior therapy for posttraumatic stress disorder. Transl. Psychiatry 2021, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.Z.; Cumming, P.; Nasrallah, F.A.; Department of Defense Alzheimer’s Disease Neuroimaging Initiative. White matter alterations are associated with cognitive dysfunction decades after moderate-to-severe traumatic brain injury and/or posttraumatic stress disorder. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2021, 6, 1100–1109. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.J.; Qi, R.; Ke, J.; Qiu, J.; Xu, Q.; Zhong, Y.; Lu, G.M.; Chen, F. White matter abnormalities in patients with typhoon-related posttraumatic stress disorder. Front. Hum. Neurosci. 2021, 15, 665070. [Google Scholar] [CrossRef]
- Lan, Q.Y.; Cao, Z.H.; Qi, R.F.; Luo, Y.F.; Zhang, J.Y.; Ge, H.H.; Dai, P.; Liu, F.; Chen, L.J.; Li, G.M.; et al. A study on longitudinal changes in white matter microstructure of parents who have lost their only child based on diffusion tensor imaging and its relationship with symptoms of posttraumatic stress disorder. Zhonghua Yi Xue Za Zhi 2022, 102, 1760–1765. (In Chinese) [Google Scholar] [CrossRef]
- Romaniuk, M.; Xia, Y.; Fisher, G.; Pannek, K.; Fripp, J.; Evans, J.; Rose, S. The relationship between chronic PTSD, cortical volumetry and white matter microstructure among Australian combat veterans. Mil. Med. Res. 2022, 9, 50. [Google Scholar] [CrossRef]
- de Souza, N.L.; Esopenko, C.; Jia, Y.; Parrott, J.S.; Merkley, T.L.; Dennis, E.L.; Hillary, F.G.; Velez, C.; Cooper, D.B.; Kennedy, J.E.; et al. Discriminating mild traumatic brain injury and posttraumatic stress disorder using latent neuroimaging and neuropsychological profiles in active-duty military service members. J. Head Trauma Rehabil. 2022, 38, E254–E266. [Google Scholar] [CrossRef]
- Guo, Z.P.; Chen, L.; Tang, L.R.; Gao, Y.; Chand, T.; Sen, Z.D.; Li, M.; Walter, M.; Wang, L.; Liu, C.H. Association between decreased interhemispheric functional connectivity of the insula and duration of illness in recurrent depression. J. Affect. Disord. 2023, 329, 88–95. [Google Scholar] [CrossRef]
- Hein, T.P.; Gong, Z.; Ivanova, M.; Fedele, T.; Nikulin, V.; Herrojo Ruiz, M. Anterior cingulate and medial prefrontal cortex oscillations underlie learning alterations in trait anxiety in humans. Commun. Biol. 2023, 6, 271. [Google Scholar] [CrossRef]
- Steinhäuser, J.L.; Teed, A.R.; Al-Zoubi, O.; Hurlemann, R.; Chen, G.; Khalsa, S.S. Reduced vmPFC-insula functional connectivity in generalized anxiety disorder: A Bayesian confirmation study. Sci. Rep. 2023, 13, 9626. [Google Scholar] [CrossRef]
- Janiri, D.; Simonetti, A.; Piras, F.; Ciullo, V.; Spalletta, G.; Sani, G. Predominant polarity and hippocampal subfield volumes in Bipolar disorders. Bipolar Disord. 2020, 22, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Kotzalidis, G.D.; Rapinesi, C.; Savoja, V.; Cuomo, I.; Simonetti, A.; Ambrosi, E.; Panaccione, I.; Gubbini, S.; De Rossi, P.; De Chiara, L.; et al. Neurobiological Evidence for the Primacy of Mania Hypothesis. Curr. Neuropharmacol. 2017, 15, 339–352. [Google Scholar] [CrossRef] [PubMed]
- Farmer, C.B.; Roach, E.L.; Bice, L.R.; Falgout, M.E.; Mata, K.G.; Roche, J.K.; Roberts, R.C. Excitatory and inhibitory imbalances in the trisynaptic pathway in the hippocampus in schizophrenia: A postmortem ultrastructural study. J. Neural Transm. 2023, 130, 949–965. [Google Scholar] [CrossRef] [PubMed]
- Janiri, D.; De Rossi, P.; Kotzalidis, G.D.; Girardi, P.; Koukopoulos, A.E.; Reginaldi, D.; Dotto, F.; Manfredi, G.; Jollant, F.; Gorwood, P.; et al. Psychopathological characteristics and adverse childhood events are differentially associated with suicidal ideation and suicidal acts in mood disorders. Eur. Psychiatry 2018, 53, 31–36. [Google Scholar] [CrossRef]
- Janiri, D.; Kotzalidis, G.D.; De Chiara, L.; Koukopoulos, A.E.; Aas, M.; Sani, G. The ring of fire: Childhood trauma, emotional reactivity, and mixed states in mood disorders. Psychiatr. Clin. N. Am. 2020, 43, 69–82. [Google Scholar] [CrossRef]
- Trovini, G.; Amici, E.; Bauco, P.; Matrone, M.; Lombardozzi, G.; Giovannetti, V.; Kotzalidis, G.D.; De Filippis, S. A comprehensive evaluation of adverse childhood experiences, socialemotional impairments, and neurodevelopmental disorders in cannabis use disorder: Implications for clinical practice. Eur. Psychiatry 2023, in press. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).