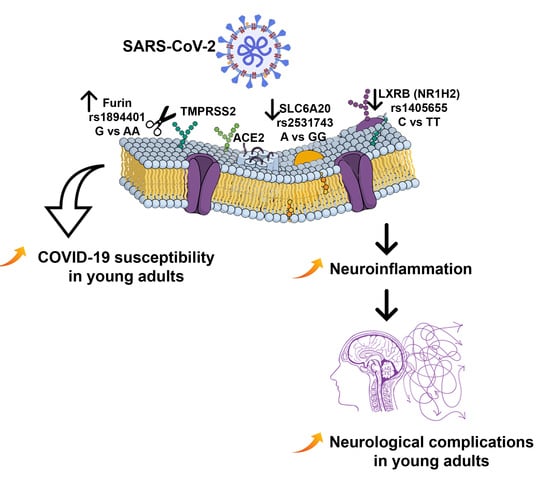
Host Genetic Variants Linked to COVID-19 Neurological Complications and Susceptibility in Young Adults—A Preliminary Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. SNPs Selection and Genotyping
2.3. Statistical Analysis
3. Results
3.1. Statistical Analysis between Clinical and Demographic Data
3.2. Association Analysis between SNPs and COVID-19 Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors Associated with COVID-19-Related Death Using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Brola, W.; Wilski, M. Neurological Consequences of COVID-19. Pharmacol. Rep. 2022, 74, 1208–1222. [Google Scholar] [CrossRef] [PubMed]
- Mustafin, R.; Kazantseva, A.; Kovas, Y.; Khusnutdinova, E. Role of Retroelements in the Development of COVID-19 Neurological Consequences. Russ. Open Med. J. 2022, 11, 313. [Google Scholar] [CrossRef]
- Zguro, K.; Fallerini, C.; Fava, F.; Furini, S.; Renieri, A. Host Genetic Basis of COVID-19: From Methodologies to Genes. Eur. J. Hum. Genet. 2022, 30, 899–907. [Google Scholar] [CrossRef]
- Mapping the Human Genetic Architecture of COVID-19. Nature 2021, 600, 472–477. [CrossRef]
- Ellinghaus, D.; Degenhardt, F.; Bujanda, L.; Buti, M.; Albillos, A.; Invernizzi, P.; Fernández, J.; Prati, D.; Baselli, G.; Asselta, R.; et al. Genomewide Association Study of Severe COVID-19 with Respiratory Failure. N. Engl. J. Med. 2020, 383, 1522–1534. [Google Scholar] [CrossRef] [PubMed]
- Roberts, G.H.L.; Park, D.S.; Coignet, M.V.; McCurdy, S.R.; Knight, S.C.; Partha, R.; Rhead, B.; Zhang, M.; Berkowitz, N.; Haug Baltzell, A.K.; et al. AncestryDNA COVID-19 Host Genetic Study Identifies Three Novel Loci. medRxiv 2020. [Google Scholar] [CrossRef]
- Pairo-Castineira, E.; Clohisey, S.; Klaric, L.; Bretherick, A.D.; Rawlik, K.; Pasko, D.; Walker, S.; Parkinson, N.; Fourman, M.H.; Russell, C.D.; et al. Genetic Mechanisms of Critical Illness in COVID-19. Nature 2021, 591, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Shcherbak, S.G.; Changalidi, A.I.; Barbitoff, Y.A.; Anisenkova, A.Y.; Mosenko, S.V.; Asaulenko, Z.P.; Tsay, V.V.; Polev, D.E.; Kalinin, R.S.; Eismont, Y.A.; et al. Identification of Genetic Risk Factors of Severe COVID-19 Using Extensive Phenotypic Data: A Proof-of-Concept Study in a Cohort of Russian Patients. Genes 2022, 13, 534. [Google Scholar] [CrossRef]
- Torre-Fuentes, L.; Matías-Guiu, J.; Hernández-Lorenzo, L.; Montero-Escribano, P.; Pytel, V.; Porta-Etessam, J.; Gómez-Pinedo, U.; Matías-Guiu, J.A. ACE2, TMPRSS2, and Furin Variants and SARS-CoV-2 Infection in Madrid, Spain. J. Med. Virol. 2021, 93, 863–869. [Google Scholar] [CrossRef]
- Baranova, A.; Cao, H.; Zhang, F. Causal Effect of COVID-19 on Alzheimer’s Disease: A Mendelian Randomization Study. J. Med. Virol. 2022, 95, e28107. [Google Scholar] [CrossRef] [PubMed]
- Kazantseva, A.V.; Enikeeva, R.F.; Davydova, Y.D.; Mustafin, R.N.; Takhirova, Z.R.; Malykh, S.B.; Lobaskova, M.M.; Tikhomirova, T.N.; Khusnutdinova, E.K. The Role of the KIBRA and APOE Genes in Developing Spatial Abilities in Humans. Vavilovskii Zhurnal Genet. Selektsii 2021, 25, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, M.R.; Poland, G.A.; Graeber, A.C.W. Does Apolipoprotein E Genotype Predict COVID-19 Severity? QJM 2020, 113, 529–530. [Google Scholar] [CrossRef] [PubMed]
- Del Ser, T.; Fernández-Blázquez, M.A.; Valentí, M.; Zea-Sevilla, M.A.; Frades, B.; Alfayate, E.; Saiz, L.; Calero, O.; García-López, F.J.; Rábano, A.; et al. Residence, Clinical Features, and Genetic Risk Factors Associated with Symptoms of COVID-19 in a Cohort of Older People in Madrid. Gerontology 2021, 67, 281–289. [Google Scholar] [CrossRef]
- Ji, H.-L.; Zhao, R.; Matalon, S.; Matthay, M.A. Elevated Plasmin(Ogen) as a Common Risk Factor for COVID-19 Susceptibility. Physiol. Rev. 2020, 100, 1065–1075. [Google Scholar] [CrossRef]
- Yuki, K.; Fujiogi, M.; Koutsogiannaki, S. COVID-19 Pathophysiology: A Review. Clin. Immunol. 2020, 215, 108427. [Google Scholar] [CrossRef]
- Zawilska, J.B.; Kuczyńska, K. Psychiatric and Neurological Complications of Long COVID. J. Psychiatr. Res. 2022, 156, 349–360. [Google Scholar] [CrossRef]
- Savino, R.; Polito, A.N.; Arcidiacono, G.; Poliseno, M.; Lo Caputo, S. Neuropsychiatric Disorders in Pediatric Long COVID-19: A Case Series. Brain Sci. 2022, 12, 514. [Google Scholar] [CrossRef]
- Glessner, J.T.; Chang, X.; Mentch, F.; Qu, H.; Abrams, D.J.; Thomas, A.; Sleiman, P.M.A.; Hakonarson, H. COVID-19 in Pediatrics: Genetic Susceptibility. Front. Genet. 2022, 13, 928466. [Google Scholar] [CrossRef]
- Schulert, G.S.; Blum, S.A.; Cron, R.Q. Host Genetics of Pediatric SARS-CoV-2 COVID-19 and Multisystem Inflammatory Syndrome in Children. Curr. Opin. Pediatr. 2021, 33, 549–555. [Google Scholar] [CrossRef]
- Singh, B.; Lant, S.; Cividini, S.; Cattrall, J.W.; Goodwin, L.C.; Benjamin, L.; Michael, B.D.; Khawaja, A.; Matos, A.D.M.B.; Alkeridy, W.; et al. Prognostic Indicators and Outcomes of Hospitalised COVID-19 Patients with Neurological Disease: An Individual Patient Data Meta-Analysis. PLoS ONE 2022, 17, e0263595. [Google Scholar] [CrossRef]
- Mattioli, F.; Stampatori, C.; Righetti, F.; Sala, E.; Tomasi, C.; De Palma, G. Neurological and Cognitive Sequelae of COVID-19: A Four Month Follow-Up. J. Neurol. 2021, 268, 4422–4428. [Google Scholar] [CrossRef]
- Nhung, V.P.; Ton, N.D.; Ngoc, T.T.B.; Thuong, M.T.H.; Hai, N.T.T.; Oanh, K.T.P.; Hien, L.T.T.; Thach, P.N.; Hai, N.V.; Ha, N.H. Host Genetic Risk Factors Associated with COVID-19 Susceptibility and Severity in Vietnamese. Genes 2022, 13, 1884. [Google Scholar] [CrossRef] [PubMed]
- de Rojas, I.; Hernández, I.; Montrreal, L.; Quintela, I.; Calero, M.; Royo, J.L.; Huerto Vilas, R.; González-Pérez, A.; Franco-Macías, E.; Macías, J.; et al. Genomic Characterization of Host Factors Related to SARS-CoV-2 Infection in People with Dementia and Control Populations: The GR@ACE/DEGESCO Study. J. Pers. Med. 2021, 11, 1318. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: www.R-project.org (accessed on 20 October 2022).
- Boscolo-Rizzo, P.; Menegaldo, A.; Fabbris, C.; Spinato, G.; Borsetto, D.; Vaira, L.A.; Calvanese, L.; Pettorelli, A.; Sonego, M.; Frezza, D.; et al. Six-Month Psychophysical Evaluation of Olfactory Dysfunction in Patients with COVID-19. Chem. Senses 2021, 46, bjab006. [Google Scholar] [CrossRef] [PubMed]
- Achom, A.; Das, R.; Pakray, P. An Improved Fuzzy Based GWO Algorithm for Predicting the Potential Host Receptor of COVID-19 Infection. Comput. Biol. Med. 2022, 151, 106050. [Google Scholar] [CrossRef]
- Drak Alsibai, K. Expression of Angiotensin-Converting Enzyme 2 and Proteases in COVID-19 Patients: A Potential Role of Cellular FURIN in the Pathogenesis of SARS-CoV-2. Med. Hypotheses 2020, 143, 109893. [Google Scholar] [CrossRef] [PubMed]
- Coto, E.; Albaiceta, G.M.; Amado-Rodríguez, L.; García-Clemente, M.; Cuesta-Llavona, E.; Vázquez-Coto, D.; Alonso, B.; Iglesias, S.; Melón, S.; Alvarez-Argüelles, M.E.; et al. FURIN Gene Variants (Rs6224/Rs4702) as Potential Markers of Death and Cardiovascular Traits in Severe COVID-19. J. Med. Virol. 2022, 94, 3589–3595. [Google Scholar] [CrossRef]
- Adighibe, O.; Arepalli, S.; Duckworth, J.; Hardy, J.; Wavrant-De Vrièze, F. Genetic Variability at the LXR Gene (NR1H2) May Contribute to the Risk of Alzheimer’s Disease. Neurobiol. Aging 2006, 27, 1431–1434. [Google Scholar] [CrossRef]
- Heath, L.; Earls, J.C.; Magis, A.T.; Kornilov, S.A.; Lovejoy, J.C.; Funk, C.C.; Rappaport, N.; Logsdon, B.A.; Mangravite, L.M.; Kunkle, B.W.; et al. Manifestations of Alzheimer’s Disease Genetic Risk in the Blood Are Evident in a Multiomic Analysis in Healthy Adults Aged 18 to 90. Sci. Rep. 2022, 12, 6117. [Google Scholar] [CrossRef]
- Ferreira, L.C.; Gomes, C.E.M.; Rodrigues-Neto, J.F.; Jeronimo, S.M.B. Genome-Wide Association Studies of COVID-19: Connecting the Dots. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2022, 106, 105379. [Google Scholar] [CrossRef] [PubMed]
- Pastana, L.F.; Silva, T.A.; Gellen, L.P.A.; Vieira, G.M.; de Assunção, L.A.; Leitão, L.P.C.; da Silva, N.M.; Coelho, R.D.C.C.; de Alcântara, A.L.; Vinagre, L.W.M.S.; et al. The Genomic Profile Associated with Risk of Severe Forms of COVID-19 in Amazonian Native American Populations. J. Pers. Med. 2022, 12, 554. [Google Scholar] [CrossRef] [PubMed]
- Semiz, S. SIT1 Transporter as a Potential Novel Target in Treatment of COVID-19. Biomol. Concepts 2021, 12, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Botto, R.; Callai, N.; Cermelli, A.; Causarano, L.; Rainero, I. Anxiety and Depression in Alzheimer’s Disease: A Systematic Review of Pathogenetic Mechanisms and Relation to Cognitive Decline. Neurol. Sci. 2022, 43, 4107–4124. [Google Scholar] [CrossRef]
| Parameters | COVID-19 Patients N = 225 | Neurological Symptoms 1 N = 158 | Long COVID-19 2 N = 81 | Without Complications 3 N = 67 | Controls N = 111 |
|---|---|---|---|---|---|
| Sex, n (%) | |||||
| Women | 173 (76.9) | 119 (75.3) | 64 (79.0) | 46 (68.7) | 93 (83.8) |
| Men | 52 (23.1) | 39 (24.7) | 17 (21.0) | 21 (31.3) | 18 (16.2) |
| COVID-19 severity, n (%) | |||||
| Mild | 185 (82.2) | 126 (79.7) | 81 (75.3) | 60 (89.6) | - |
| Moderate | 40 (17.8) | 32 (20.3) | 20 (24.7) | 7 (10.4) | - |
| Neurological symptoms, n (%) severe headache | - | 23 (14.6) | 0 | - | - |
| memory problems | - | 3 (1.9) | 63 (77.8) | - | - |
| anosmia/dysosmia | - | 147 (93.0) | 11 (13.6) | - | - |
| ageusia/dysgeusia | - | 35 (22.1) | 2 (2.5) | - | - |
| sleep problems | - | 5 (3.2) | 20 (24.7) | - | - |
| depression | - | 5 (3.2) | 7 (8.6) | - | - |
| rsID | Gene Name | EA/NEA | SARS-CoV-2 Infection | Accompanying Neurological Symptoms | Long-Term Neurological Symptoms | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| EAF Cases/Controls | β | p-Value | EAF Cases/Controls | β | p-Value | EAF Cases/Controls | β | p-Value | |||
| rs114067890 | SMRR1 | G/A | 0.01/0 | <0.01 | 0.99 | 0.01/0 | 0.62 | 0.53 | 0.01/0.01 | 0.32 | 0.74 |
| rs10911734 | IVNS1ABP | T/C | 0.30/0.37 | −1.41 | 0.15 | 0.31/0.34 | −0.65 | 0.51 | 0.30/0.33 | −0.57 | 0.56 |
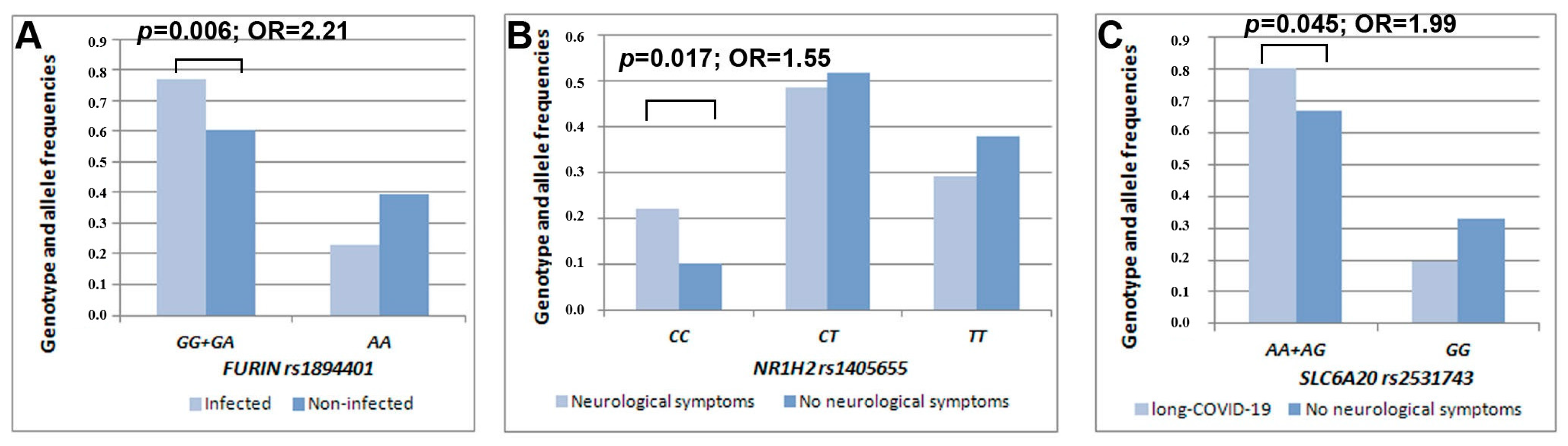
| rs2531743 | SLC6A20 | A/G | 0.50/0.44 | 1.17 | 0.24 | 0.49/0.47 | 0.47 | 0.63 | 0.55/0.46 | 1.99 | 0.045c |
| rs17078348 | LZTFL1 | G/A | 0.09/0.09 | <0.01 | 0.99 | 0.10/0.09 | 0.40 | 0.68 | 0.11/0.09 | 0.55 | 0.57 |
| rs9380142 | HLA-G | G/A | 0.23/0.29 | −1.30 | 0.19 | 0.21/0.28 | −1.70 | 0.09 | 0.26/0.25 | 0.25 | 0.80 |
| rs112640945 | CCHCR1 | T/C | 0.15/0.16 | −0.04 | 0.96 | 0.15/0.16 | −0.48 | 0.63 | 0.14/0.16 | −0.65 | 0.51 |
| rs3131294 | NOTCH4 | A/G | 0.17/0.15 | 0.36 | 0.71 | 0.17/0.15 | 0.67 | 0.49 | 0.14/0.16 | −0.52 | 0.59 |
| rs657152 | ABO | A/C | 0.44/0.46 | - | - | 0.45/0.44 | - | - | 0.47/0.44 | - | - |
| rs505922 | ABO | C/T | 0.39/0.42 | −0.82 | 0.40 | 0.42/0.38 | 0.82 | 0.41 | 0.42/0.39 | 0.70 | 0.48 |
| rs635634 | ABO | T/C | 0.39/0.42 | - | - | 0.38/0.42 | - | - | 0.35/0.41 | - | - |
| rs35705950 | MUC5B | T/G | 0.13/0.16 | −0.60 | 0.54 | 0.13/0.15 | −0.41 | 0.67 | 0.16/0.14 | 0.63 | 0.52 |
| rs4766664 | OAS1 | T/G | 0.28/0.21 | 1.00 | 0.31 | 0.29/0.25 | 0.98 | 0.32 | 0.30/0.26 | 0.88 | 0.37 |
| rs10735079 | OAS3 | G/A | 0.31/0.32 | −0.08 | 0.93 | 0.34/0.29 | 1.19 | 0.23 | 0.26/0.33 | −1.41 | 0.14 |
| rs1894401 | FURIN | G/A | 0.50/0.39 | 2.74 | 0.006a | 0.50/0.44 | 1.52 | 0.12 | 0.49/0.46 | 0.61 | 0.54 |
| rs2109069 | DPP9 | A/G | 0.21/0.20 | 0.25 | 0.79 | 0.21/0.20 | 0.18 | 0.85 | 0.23/0.20 | 0.66 | 0.50 |
| rs2304256 | TYK2 | A/C | 0.13/0.18 | −1.53 | 0.12 | 0.14/0.15 | −0.42 | 0.67 | 0.17/0.14 | 0.79 | 0.42 |
| rs45524632 | KEAP1 | A/C | 0.04/0.04 | −0.08 | 0.93 | 0.02/0.05 | −1.40 | 0.16 | 0.03/0.04 | −0.41 | 0.67 |
| rs769449 | APOE | A/G | 0.11/0.10 | 0.26 | 0.79 | 0.12/0.09 | 0.97 | 0.32 | 0.11/0.11 | 0.03 | 0.97 |
| rs7412 | APOE | T/C | 0.20/0.22 | −0.71 | 0.47 | 0.19/0.22 | −0.74 | 0.45 | 0.18/0.21 | −0.72 | 0.47 |
| rs1405655 | NR1H2 | C/T | 0.43/0.37 | 1.15 | 0.24 | 0.46/0.36 | 2.37 | 0.017b | 0.42/0.41 | 0.34 | 0.73 |
| rs61735789 | TMPRSS2 | A/G | 0.10/0.11 | - | - | 0.11/0.10 | - | - | 0.11/0.11 | - | - |
| rs12329760 | TMPRSS2 | T/C | 0.31/0.31 | <0.01 | 0.99 | 0.30/0.31 | −0.22 | 0.81 | 0.29/0.31 | −0.52 | 0.60 |
| rs4290734 | TMPRSS2 | G/A | 0.31/0.26 | 1.22 | 0.22 | 0.33/0.26 | 1.76 | 0.078 | 0.32/0.29 | 0.64 | 0.51 |
| rs117696554 | TMPRSS2 | A/G | 0.19/0.18 | - | - | 0.18/0.17 | - | - | 0.15/0.18 | - | - |
| rs2838046 | TMPRSS2 | G/A | 0.31/0.31 | - | - | 0.33/0.29 | - | - | 0.30/0.31 | - | - |
| Predictor | Reference Group | Analyzed Group | SARS-CoV-2 Infection | Accompanying Neurological Symptoms | Long-Term Neurological Symptoms | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| β | S.E. | p-Value | β | S.E. | p-Value | β | S.E. | p-Value | |||
| Sex | Men | Women | −0.58 | 0.35 | 0.099 | - | - | - | - | - | - |
| FURIN rs1894401 | AA | GG+GA | 0.85 | 0.29 | 0.0038 | - | - | - | 0.58 | 0.34 | 0.088 |
| SLC6A20 rs2531743 | GG | AA+AG | 0.66 | 0.29 | 0.026 | - | - | - | 0.75 | 0.34 | 0.031 |
| NR1H2 rs1405655 | TT | CC | - | - | - | 1.09 | 0.39 | 0.0059 | - | - | - |
| TMPRSS2 rs4290734 | AA | AG | - | - | - | 0.71 | 0.27 | 0.0099 | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kazantseva, A.; Enikeeva, R.; Takhirova, Z.; Davydova, Y.; Mustafin, R.; Malykh, S.; Karunas, A.; Kanapin, A.; Khusnutdinova, E. Host Genetic Variants Linked to COVID-19 Neurological Complications and Susceptibility in Young Adults—A Preliminary Analysis. J. Pers. Med. 2023, 13, 123. https://doi.org/10.3390/jpm13010123
Kazantseva A, Enikeeva R, Takhirova Z, Davydova Y, Mustafin R, Malykh S, Karunas A, Kanapin A, Khusnutdinova E. Host Genetic Variants Linked to COVID-19 Neurological Complications and Susceptibility in Young Adults—A Preliminary Analysis. Journal of Personalized Medicine. 2023; 13(1):123. https://doi.org/10.3390/jpm13010123
Chicago/Turabian StyleKazantseva, Anastasiya, Renata Enikeeva, Zalina Takhirova, Yuliya Davydova, Rustam Mustafin, Sergey Malykh, Alexandra Karunas, Alexander Kanapin, and Elza Khusnutdinova. 2023. "Host Genetic Variants Linked to COVID-19 Neurological Complications and Susceptibility in Young Adults—A Preliminary Analysis" Journal of Personalized Medicine 13, no. 1: 123. https://doi.org/10.3390/jpm13010123
APA StyleKazantseva, A., Enikeeva, R., Takhirova, Z., Davydova, Y., Mustafin, R., Malykh, S., Karunas, A., Kanapin, A., & Khusnutdinova, E. (2023). Host Genetic Variants Linked to COVID-19 Neurological Complications and Susceptibility in Young Adults—A Preliminary Analysis. Journal of Personalized Medicine, 13(1), 123. https://doi.org/10.3390/jpm13010123