Abstract
Introduction: This study aimed to verify the predictors of the diagnostic accuracy of rapid on-site evaluation (ROSE) in endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) among patients with non-small cell lung cancer (NSCLC). Methods: We retrospectively reviewed consecutive patients with NSCLC who underwent EBUS-TBNA for staging or diagnosis at our hospital from June 2016 to June 2018. The patients were divided into two groups—those with a correct diagnosis and an incorrect diagnosis after ROSE. Kaplan–Meier plots and log-rank tests were used to estimate outcomes. Results: A total of 84 patients underwent EBUS-TBNA for staging and diagnosis. Sixty patients with demonstrated malignant mediastinal lymph nodes were enrolled. In the univariate analysis, lymph nodes < 1.5 cm (HR = 3.667, p = 0.031) and a SUVmax > 5 (HR = 41, p = 0.001) were statistically significant for diagnostic accuracy of ROSE. In the multivariate Cox regression analysis, only a SUVmax > 5 (HR = 20.258, p = 0.016) was statistically significant. Conclusions: A SUVmax > 5 is an independent predictor of higher diagnostic accuracy of ROSE in EBUS-TBNA in patients with NSCLC with malignant mediastinal lymph nodes. Therefore, ROSE in patients with a SUVmax < 5 might not be reliable and requires further prudent assessment (more shots or repeated biopsies at mediastinal LNs) in clinical practice.
1. Introduction
Abnormal intrathoracic lymph nodes (LNs) are usually selected for biopsy on the basis of a short axis of more than 1 cm by contrast-enhanced computed tomography (CT) of the chest or increased 18F-fluorodeoxyglucose (FDG) uptake by position emission tomography (PET)-CT. Endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) has revolutionized intrathoracic disease diagnosis and lung cancer staging and is a minimally invasive and pivotal procedure in invasive thoracic workup [1,2]. Due to substantial improvements in molecular medicine for non-small cell lung cancer (NSCLC), EBUS-TBNA is now an efficient method that provides adequate samples to test for epidermal growth factor receptor (EGFR) mutations and anaplastic lymphoma kinase gene arrangement following routine histopathology and immunohistochemistry (IHC)-based subtyping [3]. Although EBUS sonographic factors, such as gray scale, blood flow Doppler, and elastography, can be used to predict malignant and benign intrathoracic LNs, tissue samples should still be obtained to confirm the diagnosis [1,4]. Rapid on-site evaluation (ROSE) of EBUS-TBNA is a well-established tool that improves the procedure yield if performed by a cytopathologist or an experienced cytotechnologist and reduces additional biopsies while preserving the diagnostic yield [5]. However, ROSE still has some limitations [6], and many reports have revealed less than 100% diagnostic accuracy [7,8,9]. To our knowledge, no studies have investigated the predictors of the diagnostic accuracy of ROSE in EBUS-TBNA. The diagnostic accuracy of ROSE plays a particularly important role in clinical practice, especially in nodal staging for immediately intraopeartive treatment decision making. Therefore, this study aimed to correlate the results of ROSE and to determine the predictors of the diagnostic accuracy of ROSE.
2. Methods
2.1. Patient Selection and Study Design
The database of the Thoracic Surgery Division of Tri-Service General Hospital, National Defense Medical Center, Taipei, Taiwan, was searched for patients who underwent EBUS-TBNA for nodal staging and diagnosis of NSCLC between June 2016 and June 2018. The medical records of the study population were reviewed and evaluated. Patients who underwent EBUS-TBNA were suspected to have intrathoracic lymph node metastases on the basis of enlargement (short axis > 10 mm), as visualized by CT or F-18 fluorodeoxyglucose (FDG) uptake ≥ the standard uptake value of 3.5 by PET. All LNs in the thorax and extrathoracic regions with a SUVmax > 3.5 were considered positive, unless they showed high attenuation (>70 HU) or benign calcification (central nodular, laminated, popcorn, or diffuse), as seen on the soft-tissue window of CT images [10,11,12]. The 60 patients with malignant LNs and the 24 patients with benign LNs were proved by the final pathologies or further follow-up in clinical conditions. This study was approved by the Institutional Review Board of Tri-Service General Hospital, National Defense Medical Center (2-108-05-089). Written informed consent for bronchoscopy was obtained from all patients; additional informed consent for this study was waived due to the retrospective design of the chart review through which clinical history and diagnostic results were obtained.
2.2. Mediastinal Lymph Node Sampling
Patients under general anesthesia underwent EBUS-TBNA in an operating room with 8.0 mm endotracheal intubation. After white-light bronchoscopy was performed with a tracheal tube, the target LNs and peripheral vessels were examined by EBUS using a linear array ultrasonic bronchoscope (BF-UC180F-OL8; Olympus Ltd., Tokyo, Japan). The diameters of the target LNs were measured and recorded in the frozen ultrasound images. A dedicated 22G needle was used for aspiration (NA-201SX-4022; Olympus Ltd., Tokyo, Japan). We recommended that at least three needle aspirations be performed for each target lesion, and the number of passes was approximately 20 to 30 times [13,14]. All procedures were performed by experienced bronchoscopists. Needle aspiration of the largest accessible LNs was guided by ultrasound. An internal stylet was removed after the initial puncture, after which negative pressure was applied with a syringe to obtain histological cores and cytological specimens. The aspirated material was smeared onto glass slides and was then fixed in 95% alcohol. Papanicolaou staining and light microscopy were also performed by an independent cytopathologist. Histological cores were formalin-fixed and stained with hematoxylin and eosin. Immunohistochemistry was also performed when necessary. Biopsies from stations 4R, 7, and 4L were routinely obtained.
2.3. Statistical Analyses
Univariate and multivariate analyses were used to assess the independent risk factors for the diagnostic accuracy of ROSE in EBUS-TBNA. A t-test was used to compare continuous variables, while the chi-squared test or Fisher’s exact test was used to compare categorical variables when appropriate. Significance was indicated at p < 0.05, and all analyses were two-sided. Significant variables in the univariate analysis or those deemed clinically important were then entered in a multivariable logistic regression model. The IBM SPSS Statistics for Windows software package (ver. 20.0; IBM Corp., Armonk, NY, USA) was used for the data analysis.
3. Results
3.1. Demographic Characteristics of the Enrolled Patients
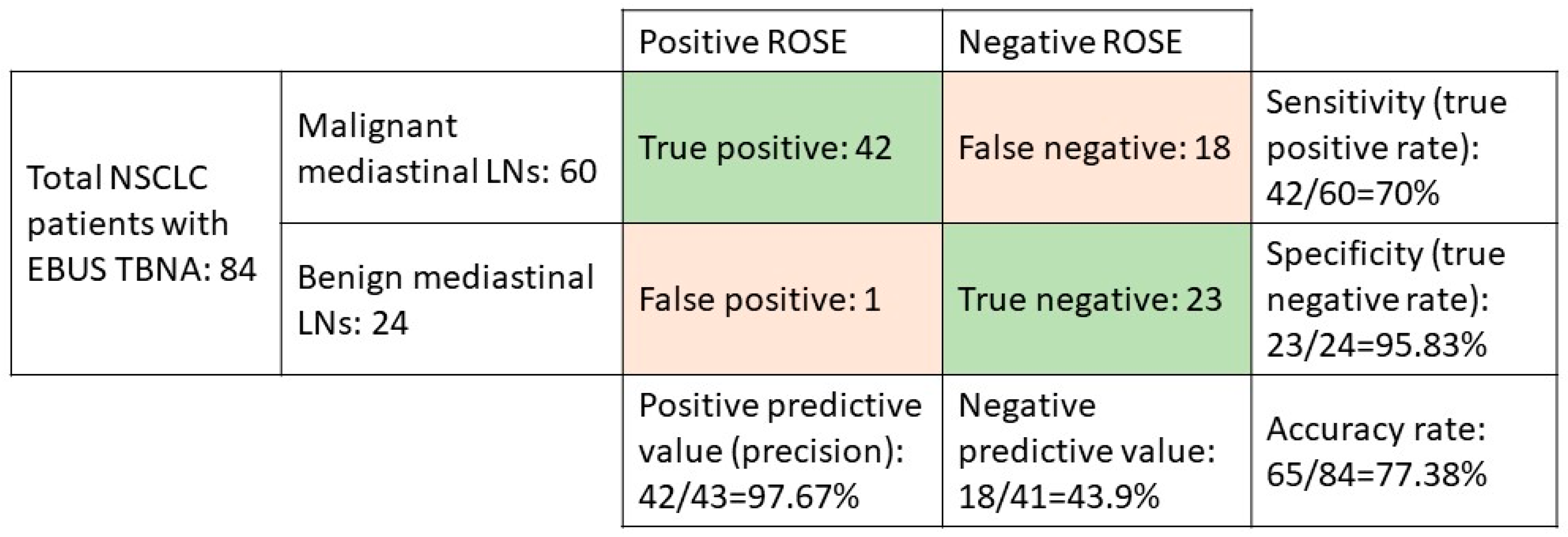
In all, 723 patients with NSCLC were eligible for this retrospective cohort study, and of them, 84 underwent EBUS-TBNA for staging and diagnosis from June 2016 to June 2018. Of those patients, we investigated 60 patients with demonstrated malignant mediastinal LNs to determine the diagnostic accuracy of ROSE. The confusion matrix showed the positive predictive rate was 97.67%, the sensitivity was 70%, the specificity was 95.83%, and the accuracy rate was 77.38% (Figure 1).
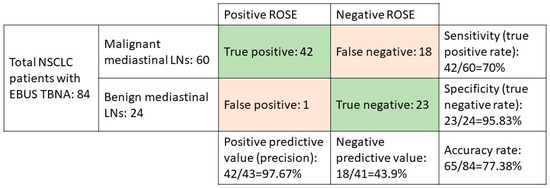
Figure 1.
The confusion matrix of NSCLC patients who underwent EBUS-TBNA.
Table 1 shows the general characteristics of the 60 patients. Forty-two patients (average 65.5 ± 1.69 years of age) with a correct diagnosis by ROSE were compared with 18 patients (average 65.5 ± 2.38 years of age) with an incorrect diagnosis by ROSE. The mean size of the sampled mediastinal LNs in the two groups was 24.55 ± 2.23 vs. 22.86 ± 4.13 mm (p = 0.698). The mean number of mediastinal lymph node stations biopsied per patient was 1.7 (range: 1.0–2.3), and the mean FDG uptake in the two groups was 10.77 ± 0.75 vs. 6.45 ± 0.78 (p = 0.001). In the two groups, a SUVmax of mediastinal LNs > 5 (p < 0.001) and a mediastinal LN size > 1.5 cm (p = 0.027) was statistically significant. No statistical significance was observed for age (p = 1), gender (p = 0.678), smoking history (p = 0.78), histology (p = 0.335), tumor differentiation (p = 0.145), EGFR mutation (p = 0.321), Hounsfield units (HU) (p = 0.852), ROSE slides (p = 0.594), carcinoembryonic antigen (CEA) level (p = 0.695), squamous cell carcinoma antigen level (p = 0.515), or survival (p = 0.309). None of the patients had endobronchial mucosal abnormalities. All EBUS-TBNA procedures were performed under general anesthesia by two experienced pulmonologists. A median of three passes was performed for each node (range: 2–5).

Table 1.
Characteristics of non-small cell lung cancer patients with malignant cells involved in mediastinal lymph node by endobronchial ultrasound with trans-bronchial needle biopsy.
3.2. Univariate and Multivariate Analysis of Predictive Factors of the Diagnostic Accuracy of ROSE in EBUS-TBNA in Non-Small-Cell Lung Cancer Patients with Malignant Mediastinal LNs
Table 2 shows the results of the univariate regression and multivariate regression analyses. In the univariate analysis, mediastinal LNs larger than 1.5 cm (HR = 3.667, p = 0.031) and a SUVmax greater than 5 (HR = 41, p = 0.001) were statistically significant for the diagnostic accuracy of ROSE in EBUS-TBNA. However, according to the multivariate Cox regression analysis, only a SUVmax greater than 5 (HR = 20.258, p = 0.016) was statistically significant.

Table 2.
Univariate and multivariate logistic regression of predictors for diagnostic accuracy of ROSE.
3.3. Survival Analysis
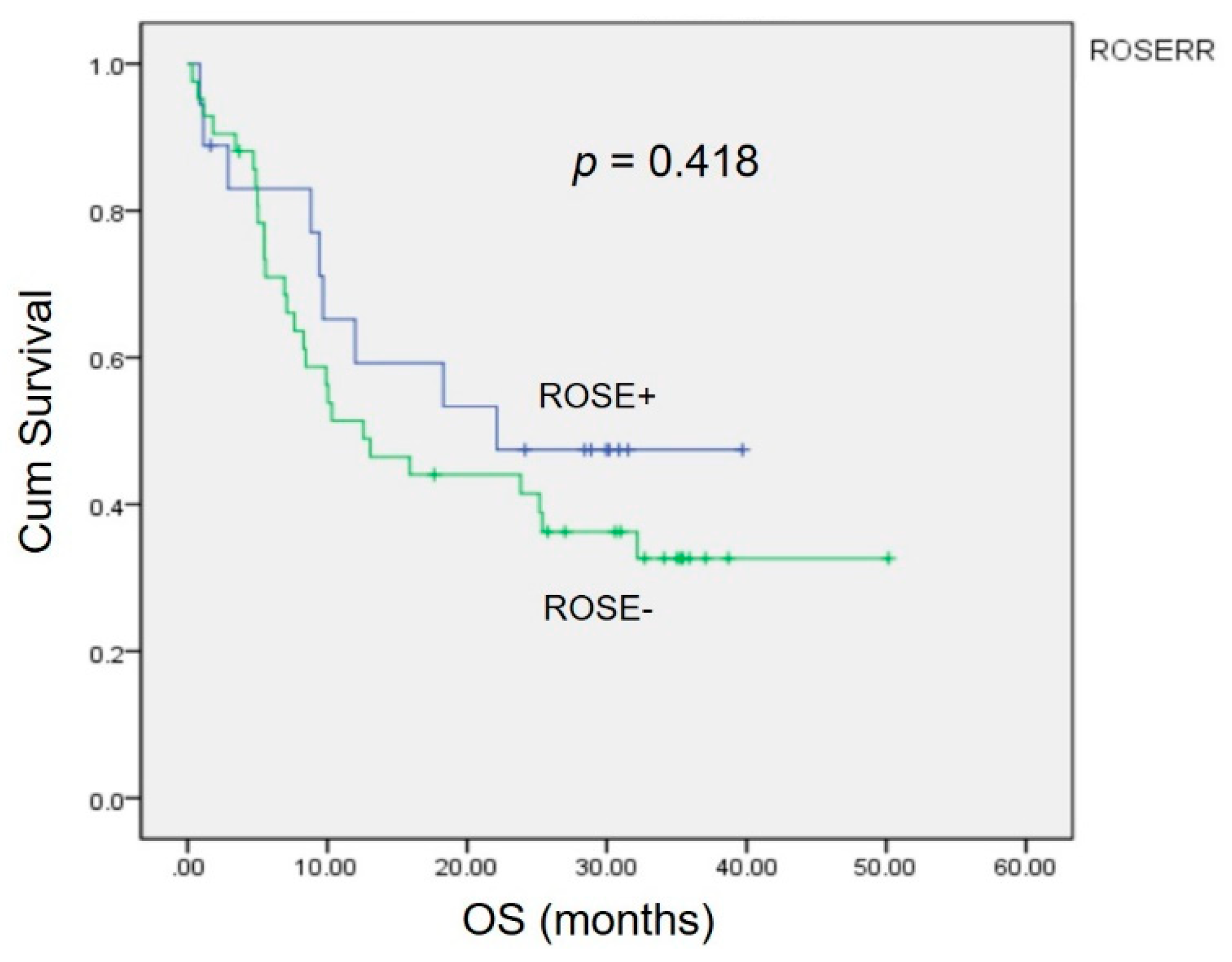
Kaplan–Meier analysis revealed no significant differences (p = 0.418) in the 3 year overall survival (OS) between patients with a correct diagnosis and those with an incorrect diagnosis by ROSE (Figure 2).
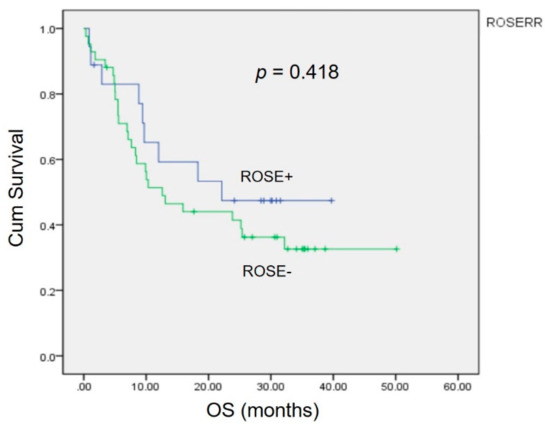
Figure 2.
The impact of the accuracy of ROSE in EBUS on overall survival. The Kaplan–Meier curve showed no statistically significant difference (p = 0.418).
4. Discussion
4.1. The Role of ROSE in Clinical Practice
Precise assessment of lymph node metastasis is important for selecting the optimal treatment for patients with NSCLC. Pathologic diagnosis of LNs is generally considered credible. EBUS-TBNA is a relatively non-invasive procedure compared with mediastinoscopy and video-assisted thoracoscopic surgery with lymph node dissection. The role of ROSE in EBUS-TBNA is to verify sample adequacy and to establish a preliminary diagnosis by performing a rapid stain in the bronchoscopy suite or operating room, followed by evaluation by a cytopathologist or a trained cytotechnologist [7,15]. The utility of ROSE has been critically examined in the literature [4,16]. Therefore, ROSE is believed to be a tool that increases the adequacy rate, diagnostic yield, and accuracy of EBUS-TBNA.
Theoretically, ROSE is not needed because EBUS is a real-time technique in which the needle and target may be observed during the procedure to verify the procedure’s accuracy [6]. Other arguments include the need for an experienced dedicated cytopathologist as a limitation of ROSE because all hospitals and institutes may not be able to accommodate this requirement [6]. Other criticisms of ROSE include the uncertainty of the diagnostic material in a paraffin block, despite the use of ROSE services, the time spent in performing the evaluation of repeated staining and sample analysis, and the cost [17]. The staining method employed during ROSE, however, does not influence the final diagnosis [18]. Moreover, on the basis of its convenience and efficiency in lung cancer patients, we typically perform EBUS-TBNA for nodal staging followed by surgical resection of lung cancer without nodal metastases according to the result of ROSE. Therefore, the diagnostic accuracy of ROSE plays a particularly important role in clinical practice, especially in nodal staging used for the selection of subsequent treatment, including surgical resection or systemic therapy.
In our study, the correlation rate between correct diagnosis by ROSE and correct diagnosis by final pathology was 71.43% (p = 0.003). To the best of our knowledge, no associated studies have investigated predictors of the diagnostic accuracy of ROSE in EBUS-TBNA. We hypothesized that the result of ROSE in selected patients without predictors should be carefully considered, at which point treatment decisions for patient management may be made.
4.2. Predictive Factors of the Diagnostic Accuracy of ROSE in EBUS-TBNA
To the best of our knowledge, the predictive factors of preoperative lymph node metastasis are central tumor localization [19], larger tumor size [19,20,21], age ≤ 67 years [20], high CEA level [20,21], micropapillary-predominant adenocarcinoma [21,22,23], and consolidation/tumor ratio ≥ 89% [20]. Moreover, although EGFR mutations are strongly associated with clinical outcomes in patients with lung adenocarcinoma, the effect of EGFR mutation status on prognosis is still unclear [24,25,26,27,28,29]. In our study, age, gender, smoking history, histology, tumor differentiation, EGFR mutation status, CEA level, and OS were not statistically significant factors related to the diagnostic accuracy of ROSE.
In the univariate regression analysis, the SUVmax and size of mediastinal LNs were significant predictors of the diagnostic accuracy of ROSE. A SUVmax > 5 (area = 0.894) and mediastinal LN size > 1.5 cm (area = 0.643) as cutoffs were determined by the area under the receiver operating characteristic curve. After the multivariate analysis, a SUVmax > 5 was the only independent predictor of the diagnostic accuracy of ROSE in EBUS-TBNA. High FDG uptake, as seen by PET, that is suspicious for nodal metastases requires histopathologic confirmation and represents higher metabolic activity [30]. FDG-PET/CT adds greater value to CT in lymph node staging of NSCLCs [31]. Therefore, the density and behavior of malignant cells in mediastinal LNs could be correlated with the FDG uptake value. In clinical practice, we perform EBUS-TBNA and possible surgical resection of lung cancer at the same time when patients with suspected mediastinal nodal metastases are under general anesthesia. The diagnostic accuracy of ROSE is valuable and helps direct immediate decisions regarding subsequent disease management. Therefore, in clinical practice, we could use predictors of ROSE accuracy to consider the results and make rational decisions. We also noted that the size of the mediastinal LNs corresponds with the volume of the available specimen. Thus, patients with larger LNs benefited from a higher diagnostic accuracy of ROSE and received more needle aspirations per target lesion in our study. A SUVmax less than 5 should be considered a risk factor of lower diagnostic accuracy of ROSE.
One limitation of this study was its retrospective single-center design. Additionally, the small sample size may have led to bias. A prospective, randomized, multicenter study is necessary to confirm the predictive accuracy of ROSE during EBUS-TBNA.
5. Conclusions
A SUVmax greater than 5 is an independent predictor of higher diagnostic accuracy of ROSE in EBUS-TBNA in NSCLC patients with metastatic mediastinal LNs. The patients with larger LNs benefited from a higher diagnostic accuracy of ROSE and received more needle aspirations per target lesion in our study. In clinical practice, the utility of ROSE in patients with certain risk factors that might affect the diagnostic accuracy should be carefully evaluated and requires further prudent assessment (more shots or repeated biopsies at mediastinal LNs).
Author Contributions
Y.-Y.C. and T.-W.H. conceived the project and designed the experiment. Y.-Y.C. and T.-W.H. provided clinical samples and aided in the clinical interpretation. Y.-Y.C., T.-W.H., C.-Y.L., K.-L.C. and H.-Y.H. executed experimental measurements and data analysis. Y.-Y.C. wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Institutional Review Board Statement
Approval for the study was obtained from the Institutional Review Board/Ethics Committee of Tri-Service General hospital (2-108-05-089). All participants provided their written informed consent to participate in this study.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Acknowledgments
The authors declare that there are no conflicts of interest related to the subject matter or materials discussed in this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zhi, X.; Chen, J.; Xie, F.; Sun, J.; Herth, F.J.F. Diagnostic value of endobronchial ultrasound image features: A specialized review. Endosc. Ultrasound 2021, 10, 3–18. [Google Scholar] [PubMed]
- Kinsey, C.M.; Arenberg, D.A. Endobronchial ultrasound-guided transbronchial needle aspiration for non-small cell lung cancer staging. Am. J. Respir. Crit. Care Med. 2014, 189, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xie, F.; Mao, X.; Zheng, X.; Li, Y.; Zhu, L.; Sun, J. Determining factors of endobronchial ultrasound-guided transbronchial needle aspiration specimens for lung cancer subtyping and molecular testing. Endosc. Ultrasound 2019, 8, 404–411. [Google Scholar] [PubMed]
- Wahidi, M.M.; Herth, F.; Yasufuku, K.; Shepherd, R.W.; Yarmus, L.; Chawla, M.; Lamb, C.; Casey, K.R.; Patel, S.; Silvestri, G.A.; et al. Technical Aspects of Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration: CHEST Guideline and Expert Panel Report. Chest 2016, 149, 816–835. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baram, D.; Garcia, R.B.; Richman, P.S. Impact of rapid on-site cytologic evaluation during transbronchial needle aspiration. Chest 2005, 128, 869–875. [Google Scholar] [CrossRef]
- Jain, D.; Allen, T.C.; Aisner, D.L.; Beasley, M.B.; Cagle, P.T.; Capelozzi, V.L.; Hariri, L.P.; Lantuejoul, S.; Miller, R.; Mino-Kenudson, M.; et al. Rapid On-Site Evaluation of Endobronchial Ultrasound-Guided Transbronchial Needle Aspirations for the Diagnosis of Lung Cancer: A Perspective From Members of the Pulmonary Pathology Society. Arch. Pathol. Lab. Med. 2018, 142, 253–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakajima, T.; Yasufuku, K.; Saegusa, F.; Fujiwara, T.; Sakairi, Y.; Hiroshima, K.; Nakatani, Y.; Yoshino, I. Rapid on-site cytologic evaluation during endobronchial ultrasound-guided transbronchial needle aspiration for nodal staging in patients with lung cancer. Ann. Thorac. Surg. 2013, 95, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Yarmus, L.; Akulian, J.; Gilbert, C.; Feller-Kopman, D.; Lee, H.J.; Zarogoulidis, P.; Lechtzin, N.; Ali, S.Z.; Sathiyamoorthy, V. Optimizing endobronchial ultrasound for molecular analysis. How many passes are needed? Ann. Am. Thorac. Soc. 2013, 10, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Dyhdalo, K.S.; Booth, C.N.; Shorie, J.; Underwood, D.L.; Mazzone, P.; Brainard, J.A. Rapid on-site evaluation of endobronchial ultrasound-guided fine-needle aspirates: Correlation of adequacy assessment and final diagnosis in patients with bronchogenic carcinoma. J. Am. Soc. Cytopathol. 2014, 3, 199–205. [Google Scholar] [CrossRef]
- Shim, S.S.; Lee, K.S.; Kim, B.T.; Chung, M.J.; Lee, E.J.; Han, J.; Choi, J.Y.; Kwon, O.J.; Shim, Y.M.; Kim, S. Non-small cell lung cancer: Prospective comparison of integrated FDG PET/CT and CT alone for preoperative staging. Radiology 2005, 236, 1011–1019. [Google Scholar] [CrossRef]
- Kim, B.T.; Lee, K.S.; Shim, S.S.; Choi, J.Y.; Kwon, O.J.; Kim, H.; Shim, Y.M.; Kim, J.; Kim, S. Stage T1 non-small cell lung cancer: Preoperative mediastinal nodal staging with integrated FDG PET/CT--a prospective study. Radiology 2006, 241, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Lee, K.S.; Kim, B.T.; Choi, J.Y.; Kim, H.; Kwon, O.J.; Shim, Y.M.; Yi, C.A.; Kim, H.Y.; Chung, M.J. Mediastinal nodal staging of nonsmall cell lung cancer using integrated 18F-FDG PET/CT in a tuberculosis-endemic country: Diagnostic efficacy in 674 patients. Cancer 2007, 109, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.Y.; Zhao, H.; Zhang, J.; Wang, X.D.; Han, B.H. First 30 endobronchial ultrasound-guided transbronchial needle aspirations: A single institution’s early experience. Chin. Med. J. 2011, 124, 1818–1823. [Google Scholar] [PubMed]
- Casal, R.F.; Staerkel, G.A.; Ost, D.; Almeida, F.A.; Uzbeck, M.H.; Eapen, G.A.; Jimenez, C.A.; Nogueras-Gonzalez, G.M.; Sarkiss, M.; Morice, R.C. Randomized clinical trial of endobronchial ultrasound needle biopsy with and without aspiration. Chest 2012, 142, 568–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, H.; Liu, S.; Guo, J.; Li, B.; Li, W.; Lu, Z.; Sun, J.; Zhang, B.; Yu, J. Rapid on-site evaluation during endobronchial ultrasound-guided transbronchial needle aspiration for the diagnosis of hilar and mediastinal lymphadenopathy in patients with lung cancer. Cancer Lett. 2016, 371, 182–186. [Google Scholar] [CrossRef] [PubMed]
- van der Heijden, E.H.; Casal, R.F.; Trisolini, R.; Steinfort, D.P.; Hwangbo, B.; Nakajima, T.; Guldhammer-Skov, B.; Rossi, G.; Ferretti, M.; Herth, F.F.; et al. Guideline for the acquisition and preparation of conventional and endobronchial ultrasound-guided transbronchial needle aspiration specimens for the diagnosis and molecular testing of patients with known or suspected lung cancer. Respiration 2014, 88, 500–517. [Google Scholar] [CrossRef] [PubMed]
- Layfield, L.J.; Bentz, J.S.; Gopez, E.V. Immediate on-site interpretation of fine-needle aspiration smears: A cost and compensation analysis. Cancer 2001, 93, 319–322. [Google Scholar] [CrossRef]
- Louw, M.; Brundyn, K.; Schubert, P.T.; Wright, C.A.; Bolliger, C.T.; Diacon, A.H. Comparison of the quality of smears in transbronchial fine-needle aspirates using two staining methods for rapid on-site evaluation. Diagn. Cytopathol. 2012, 40, 777–781. [Google Scholar] [CrossRef] [PubMed]
- Moulla, Y.; Gradistanac, T.; Wittekind, C.; Eichfeld, U.; Gockel, I.; Dietrich, A. Predictive risk factors for lymph node metastasis in patients with resected non-small cell lung cancer: A case control study. J. Cardiothorac. Surg. 2019, 14, 11. [Google Scholar] [CrossRef]
- Koike, T.; Koike, T.; Yamato, Y.; Yoshiya, K.; Toyabe, S. Predictive risk factors for mediastinal lymph node metastasis in clinical stage IA non-small-cell lung cancer patients. J. Thorac. Oncol. 2012, 7, 1246–1251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bao, F.; Yuan, P.; Yuan, X.; Lv, X.; Wang, Z.; Hu, J. Predictive risk factors for lymph node metastasis in patients with small size non-small cell lung cancer. J. Thorac. Dis. 2014, 6, 1697–1703. [Google Scholar]
- Roh, M.S.; Lee, J.I.; Choi, P.J.; Hong, Y.S. Relationship between micropapillary component and micrometastasis in the regional lymph nodes of patients with stage I lung adenocarcinoma. Histopathology 2004, 45, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Russell, P.A.; Wainer, Z.; Wright, G.M.; Daniels, M.; Conron, M.; Williams, R.A. Does lung adenocarcinoma subtype predict patient survival?: A clinicopathologic study based on the new International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary lung adenocarcinoma classification. J. Thorac. Oncol. 2011, 6, 1496–1504. [Google Scholar]
- Liu, Y.; Xu, M.L.; Zhong, H.H.; Heng, W.J.; Wu, B.Q. EGFR mutations are more frequent in well-differentiated than in poor-differentiated lung adenocarcinomas. Pathol. Oncol. Res. 2008, 14, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.Y.; Ryu, J.S.; Sim, Y.S.; Kim, D.; Lee, S.Y.; Choi, J.; Park, S.; Ryu, Y.J.; Lee, J.H.; Chang, J.H. Clinical significance of EGFR mutation types in lung adenocarcinoma: A multi-centre Korean study. PLoS ONE 2020, 15, e0228925. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Eom, D.W.; Kim, J.H.; Kim, K.H.; Shin, H.M.; An, J.Y.; Lee, K.M.; Choe, K.H.; Lee, K.H.; Kim, S.T.; et al. EGFR mutation status in primary lung adenocarcinomas and corresponding metastatic lesions: Discordance in pleural metastases. Clin. Lung Cancer 2011, 12, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Tsao, M.S.; Sakurada, A.; Ding, K.; Aviel-Ronen, S.; Ludkovski, O.; Liu, N.; Le Maitre, A.; Gandara, D.; Johnson, D.H.; Rigas, J.R.; et al. Prognostic and predictive value of epidermal growth factor receptor tyrosine kinase domain mutation status and gene copy number for adjuvant chemotherapy in non-small cell lung cancer. J. Thorac. Oncol. 2011, 6, 139–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.W.; Wu, C.T.; Shih, J.Y.; Chang, Y.L.; Yang, P.C. Clinicopathologic characteristics and prognostic significance of EGFR and p53 mutations in surgically resected lung adenocarcinomas ≤ 2 cm in maximal dimension. J. Surg. Oncol. 2014, 110, 99–106. [Google Scholar] [CrossRef]
- Gundogdu, A.G.; Onder, S.; Firat, P.; Dogan, R. EGFR immunoexpression, RAS immunoexpression and their effects on survival in lung adenocarcinoma cases. J. Thorac. Dis. 2014, 6, 778–784. [Google Scholar] [PubMed]
- Kandathil, A.; Kay, F.U.; Butt, Y.M.; Wachsmann, J.W.; Subramaniam, R.M. Role of FDG PET/CT in the Eighth Edition of TNM Staging of Non-Small Cell Lung Cancer. Radiographics 2018, 38, 2134–2149. [Google Scholar] [CrossRef]
- Lee, S.M.; Park, C.M.; Paeng, J.C.; Im, H.J.; Goo, J.M.; Lee, H.J.; Kang, C.H.; Kim, Y.W.; Kim, J.I. Accuracy and predictive features of FDG-PET/CT and CT for diagnosis of lymph node metastasis of T1 non-small-cell lung cancer manifesting as a subsolid nodule. Eur. Radiol. 2012, 22, 1556–1563. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).