Highlights
What key advances does this study provide for nickel laterite processing?
- Selective comminution–classification generates Ni-rich fines and Co-rich coarse fractions for targeted downstream processing.
- Mechanical activation (stirred-media milling) and thermal calcination pre-treatment enhance Ni–Co leaching via structural disorder and goethite dehydroxylation.
How do ore mineralogy influence extraction performance?
- Ore-specific hydrometallurgical routes (e.g., HPAL/AL for limonite; AL/HL for saprolite) governs acid consumption and value metal extraction.
What emerging processing concepts are demonstrated?
- Resin in Moist Mix (RIMM) technology integrates atmospheric leaching with ion exchange, offering potential for simplified laterite processing with reduced capital costs.
Abstract
The accelerating transition to low carbon energy systems has intensified the demand for nickel and cobalt from low-grade (<1.5 wt.%) refractory lateritic ores. These low-grade laterites are however not amenable to conventional beneficiation due to their complex mineralogy, eclectic physicochemical properties, and fine Ni–Co dissemination. This review examines recent advances made in the extraction of nickel and cobalt from complex low-grade lateritic ores, emphasizing the interplay between ore mineralogy, chemistry, beneficiation, pretreatment, and processing route selection. Developments in selective ore comminution–classification have led to the generation of Ni-rich fine fractions (undersize) and Co-rich coarse fractions (oversize), enabling differentiated extraction strategies that improve resource utilization, frugal energy use, and process efficiency. Mechanical activation via stirred media milling, thermal calcination-induced structural disorder, and dehydroxylate goethite products, are shown to significantly enhance Ni–Co leaching kinetics under both atmospheric and heap leaching conditions. A critical comparison of pyrometallurgical (rotary-kiln electric furnace) and hydrometallurgical (HPAL, EPAL, heap, atmospheric, bioleaching) routes demonstrates that ore-specific optimization is essential to balance recovery, acid consumption, and greenhouse gas emissions. The novel resin in moist mix (RIMM) process, which integrates ambient leaching and in situ ion exchange selective recovery, is shown to offer potential for sustainable values extraction from sub-economic resources. Furthermore, the review highlights the key innovation challenges and concomitant opportunities for enhanced critical battery metal recovery from complex laterite ores.
1. Introduction
The global pursuit of sustainable and clean energy has intensified the demand for critical and strategic battery metals such as cobalt (Co), nickel (Ni), lithium (Li), and rare earth elements (REEs) [1]. These metals are indispensable components of low carbon emission technologies and advanced devices (e.g., electric vehicles, drones, and solar panels) [2]. Their strategic importance has reshaped global resource policies, with resource-rich nations implementing raw mineral export restrictions to promote domestic value addition. Indonesia’s 2019 nickel export ban exemplifies this trend, aiming to stimulate local production of nickel-based materials, particularly battery materials [3]. With the depletion of high-grade sulphide ore deposits and potential future export limitations driven by surging global demand, low-grade lateritic ores have emerged as significant alternative sources of nickel and cobalt.
Co-production of cobalt, primarily as a by-product, further enhances the economic viability of laterite processing [4]. Typically, within a laterite profile, cobalt tends to concentrate in the oxide zones, whereas nickel is predominantly found within magnesium silicate minerals [4,5]. Lateritic ores are, however, mineralogically complex, chemically variable, comprising >98 wt.% of acid consuming gangue clays (e.g., serpentine, kaolinite, smectite, and chlorite) and refractory oxides (e.g., quartz, hematite, magnetite, and goethite) [5,6,7]. These characteristics critically influence the choice and efficiency of extraction routes. The extraction of Ni and Co from such ores can be accomplished through either pyrometallurgical or hydrometallurgical pathways, depending on specific ore characteristics. Prior to extraction, conventional preconcentration (e.g., classification, gravity, dense media, magnetic, flotation separation) and pretreatment methods (e.g., thermal, mechanical, chemical, and biological techniques) are often employed to optimize the feed based on the ore’s physicochemical properties [8,9,10,11].
The success of nickel upgrading depended on the mineralogy of the laterite. However, most attempts were unsuccessful due to the fine dissemination of nickel-bearing minerals, requiring sufficient liberation in the very fine fractions [12,13]. To date, the processing of lateritic ores using conventional techniques remains capital intensive and technically challenging. Numerous studies indicate that use of elevated temperatures and/or pressures can substantially improve Ni and Co extraction efficiency [11,14,15]. Yet, such improvements typically come at the expense of higher capital expenditure (CapEX) and operating expenditure (OpEX). For instance, the rotary kiln–electric furnace (RKEF) technology frequently used in pyrometallurgical treatment of laterite ores is energy demanding and generates considerable greenhouse gas (GHG) emissions [16].
High-pressure acid leaching (HPAL) similarly requires extreme operating conditions including temperatures of 240–270 °C and pressures of 30–50 bar, with costly titanium-lined autoclaves. This results in substantial operating expenses, often comparable to or exceeding those of pyrometallurgical processing [17]. Despite the lower cost of heap leaching process, it is generally marred by long processing time (e.g., up to 500 days), low metal recoveries, and the need for agglomeration pretreatment [18]. Recent research suggests that the application of suitable pretreatment strategies prior to atmospheric leaching could enhance metal recovery while reducing overall processing costs [19,20].
Global Distribution of Nickel and Cobalt
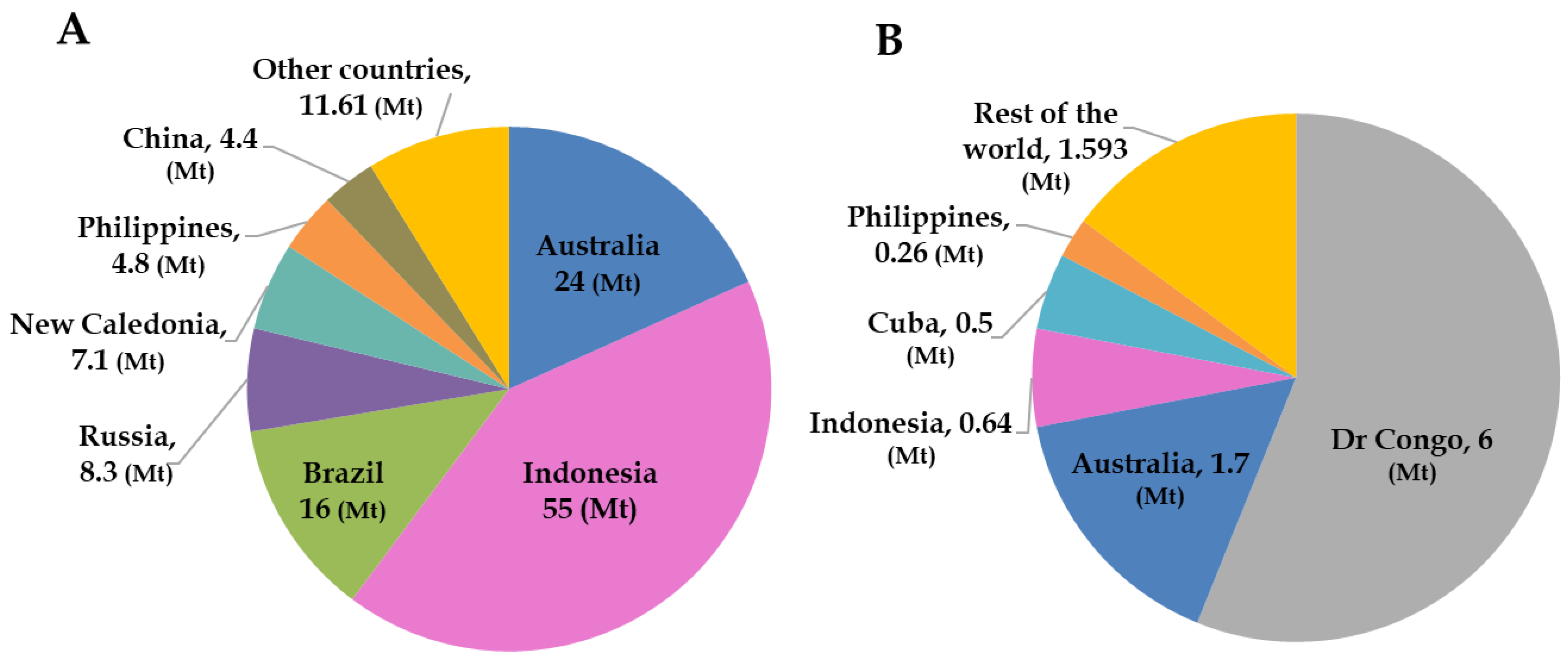
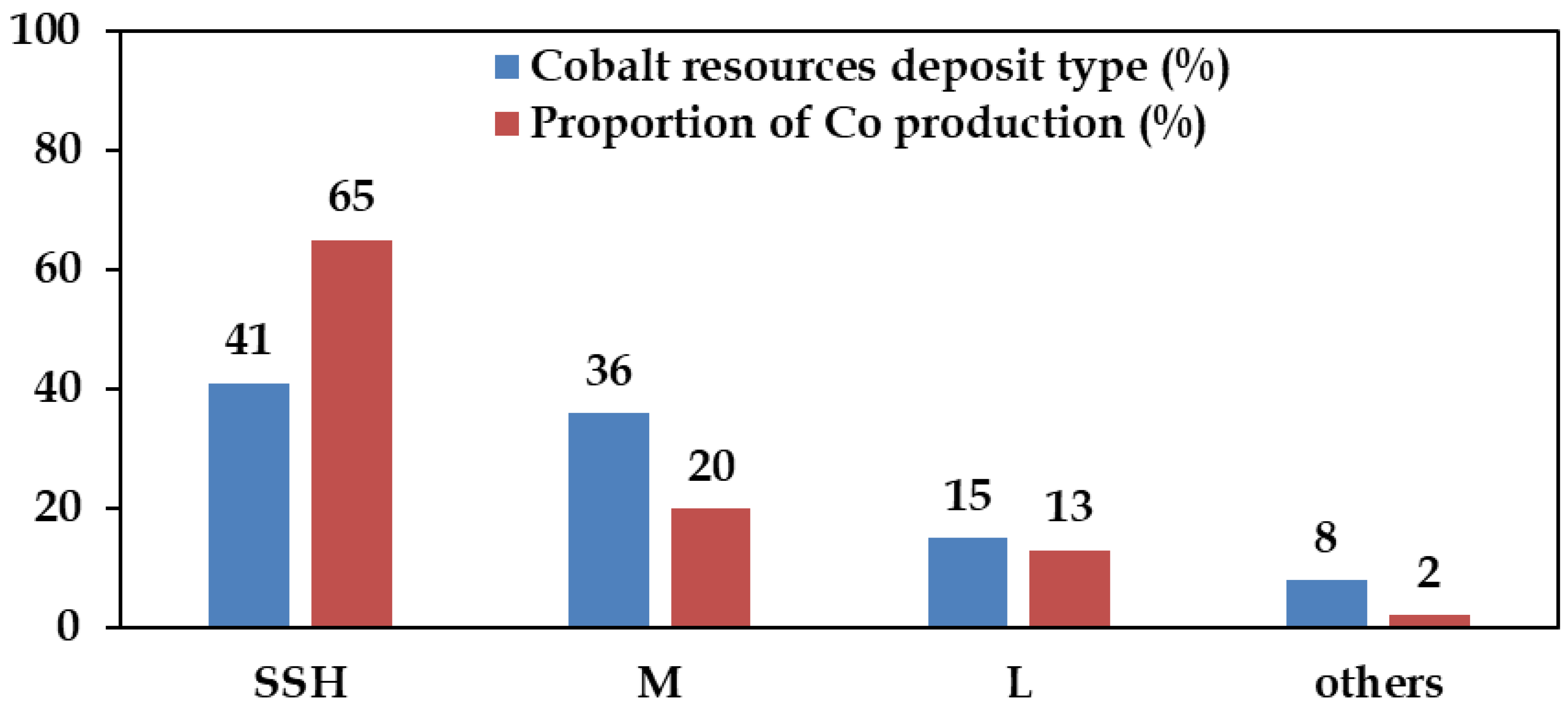
Global reserves of nickel and cobalt are estimated at ~130 Mt and 11 Mt, respectively [21]. The distribution of these reserves among major producing countries is illustrated in Figure 1. Indonesia hosts the world’s largest nickel reserves, estimated at around 55 Mt, followed by Australia, Brazil, and other nations (Figure 1A). In contrast, the Democratic Republic of Congo (DRC) possesses the largest cobalt reserves, estimated at about 6 Mt, with Australia ranking second (Figure 1B) [21]. Globally, nickel resources are predominantly contained in lateritic deposits (~70%), and to a lesser extent in sulphide deposits [22]. Lateritic ores are among the sources from which cobalt is recovered as a by-product of nickel extraction [23]. Overall, cobalt resources are distributed across several deposit types, including Cu-Co oxides (41%), magmatic Ni-Cu sulphide deposits (36%), Ni-Co laterite (15%), and other minor deposits (8%) [24,25,26].
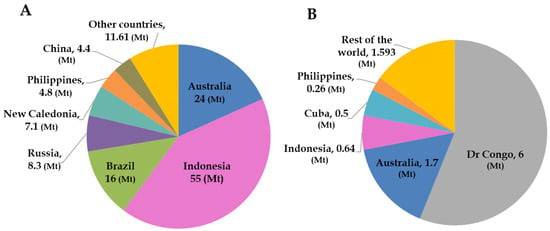
Figure 1.
Distribution of world’s land-based (A) Ni and (B) Co deposits by country (in million metric tons). Modified after U.S.G.S [21].
Figure 1.
Distribution of world’s land-based (A) Ni and (B) Co deposits by country (in million metric tons). Modified after U.S.G.S [21].
Global nickel consumption has shown a steady long term upward trajectory between 2010 and 2024, increasing from ~1620 kt to nearly 3700 kt (Figure 2) [27]. Following moderate growth during the early 2010s, consumption experienced a temporary slowdown between 2014 and 2017 before recovering and accelerating from 2018 onward. This renewed growth aligns with surging demand driven by stainless steel production and the expanding battery industry [27]. According to the Australian Government [28] and Argus [29], the slight contraction observed in 2024 reflects short term market adjustments, particularly softening in stainless steel demand, inventory corrections, and slower electric vehicle (EV) growth amid broader macroeconomic uncertainty. In comparison, global cobalt consumption, though lower in absolute magnitude, demonstrates a sustained and progressive rise from about 107 kt in 2010 to ~300 kt in 2024. Growth remained relatively moderate until around 2019, after which a more pronounced increase occurred, largely attributable to the expanding use of cobalt in lithium-ion battery chemistries. Collectively, these trends underscore the dominant scale of nickel demand and highlight the growing strategic importance of cobalt in supporting global clean energy and electrification initiatives.
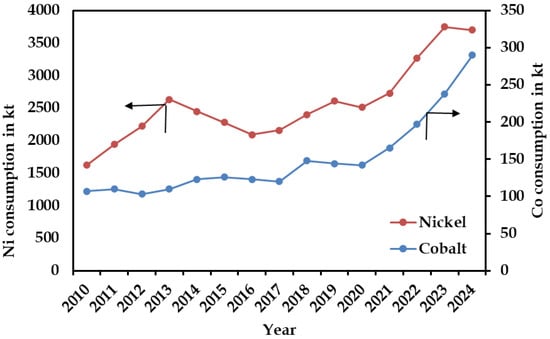
Figure 2.
Global consumption trends of nickel and cobalt from 2010 to 2024, expressed in thousand metric tons. Modified after Statista [27].
Figure 2.
Global consumption trends of nickel and cobalt from 2010 to 2024, expressed in thousand metric tons. Modified after Statista [27].
2. Cobalt Mineral Occurrence
Cobalt is a relatively rare element, constituting ~0.001% of the Earth’s crust. It is a critical metal used in manufacturing high-strength alloys, magnets, and rechargeable batteries [24]. Typically, global cobalt output usually is derived as a by-product of extracting other metals, primarily nickel and copper [26]. Co-bearing ores’ mineralogy is diverse, encompassing both hypogene and supergene phases [30]. In primary deposits, cobalt commonly occurs in stratiform, sediment-hosted copper deposits, while secondary sources are associated with nickel laterites and magmatic Ni–Cu–Co sulphide deposits hosted within mafic and ultramafic rocks. Other deposit types may also contain significant cobalt concentrations [24,31].
2.1. Stratiform Sediment-Hosted (SSH) Cu–Co Deposits
These deposits form within siliciclastic or carbonate sedimentary rocks and represent the world’s largest cobalt source. The most significant examples lie within the Central African Copperbelt in the Democratic Republic of Congo (Kinshasa) and Zambia [30]. Mineral compositions typically include chalcopyrite, pyrite, and carrollite, with minor bornite and chalcocite. Cobalt is predominantly hosted in carrollite. SSH Cu–Co deposits account for ~65% of global cobalt mine production (Figure 3), with cobalt concentrations ranging from 0.001% to 1.08% [32].
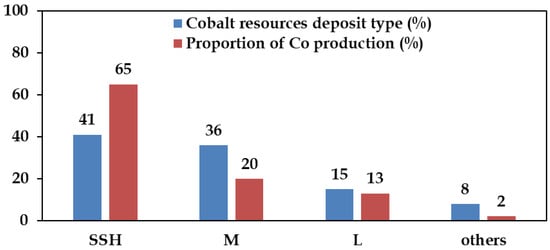
Figure 3.
Various Cobalt resources and proportion of Co-produced. SSH-sediment hosted, M-magmatic, L-Ni-Co laterites. Redacted after Huang et al. [25].
Figure 3.
Various Cobalt resources and proportion of Co-produced. SSH-sediment hosted, M-magmatic, L-Ni-Co laterites. Redacted after Huang et al. [25].
2.2. Magmatic Ni–(Cu–Co) Sulphide Deposits
Magmatic sulphide deposits represent a significant global source of Ni and Co, accounting for ~36% of global reserves and contribute to ~20% of total Co production (Figure 3). They are formed through high temperature magmatic processes containing nickel, copper, minor platinum group metals (PGMs), and cobalt [24,31]. When sulphur saturation is reached, an immiscible sulphide melts separates from the silicate magma, concentrating chalcophile elements such as Ni, Cu, and Co [33]. The ore minerals typically include pyrrhotite, pentlandite, and chalcopyrite, with pentlandite serving as the principal cobalt-bearing phase, containing 0.05%–0.1% Co [30]. Cobalt, along with platinum-group elements (PGMs), are among the by-products extracted from these deposits.
Ni–Co laterite deposits, discussed in the subsequent section, represent a smaller proportion of global cobalt reserves (Figure 3) but offer growing potential as a by-product (pay metal) of nickel extraction. As primary cobalt resources are progressively depleted, increasing attention is being directed toward optimizing recovery from lateritic and other secondary sources.
3. Nickel Mineral Occurrence
Nickel is a transition metal commonly associated with iron and sulphur and is among the abundant metals in the earth’s crust. Its unique properties (e.g., corrosion resistance, ductility, mechanical strength, and magnetism) make it indispensable in modern metallurgy [34]. Approximately 40% of global nickel production is derived from lateritic ores, which account for about 70% of known nickel resources, while the remainder originates from sulphide deposits (Table 1) [35]. The metal’s exceptional blend of properties underpins its widespread use in stainless steel production and the rapidly growing rechargeable battery sector. In the last 10 years, the production of nickel from magmatic sulphide deposits has experienced a gradual decline and may even plateau [36] due to their ruthless exploitation. This calls for the exploration of underutilized laterite deposit for nickel production tied to gradual depletion of sulfidic reserves.

Table 1.
Publicly reported nickel sources and production by mineralization style.
3.1. Nickel–Cobalt Laterite Mineralogy
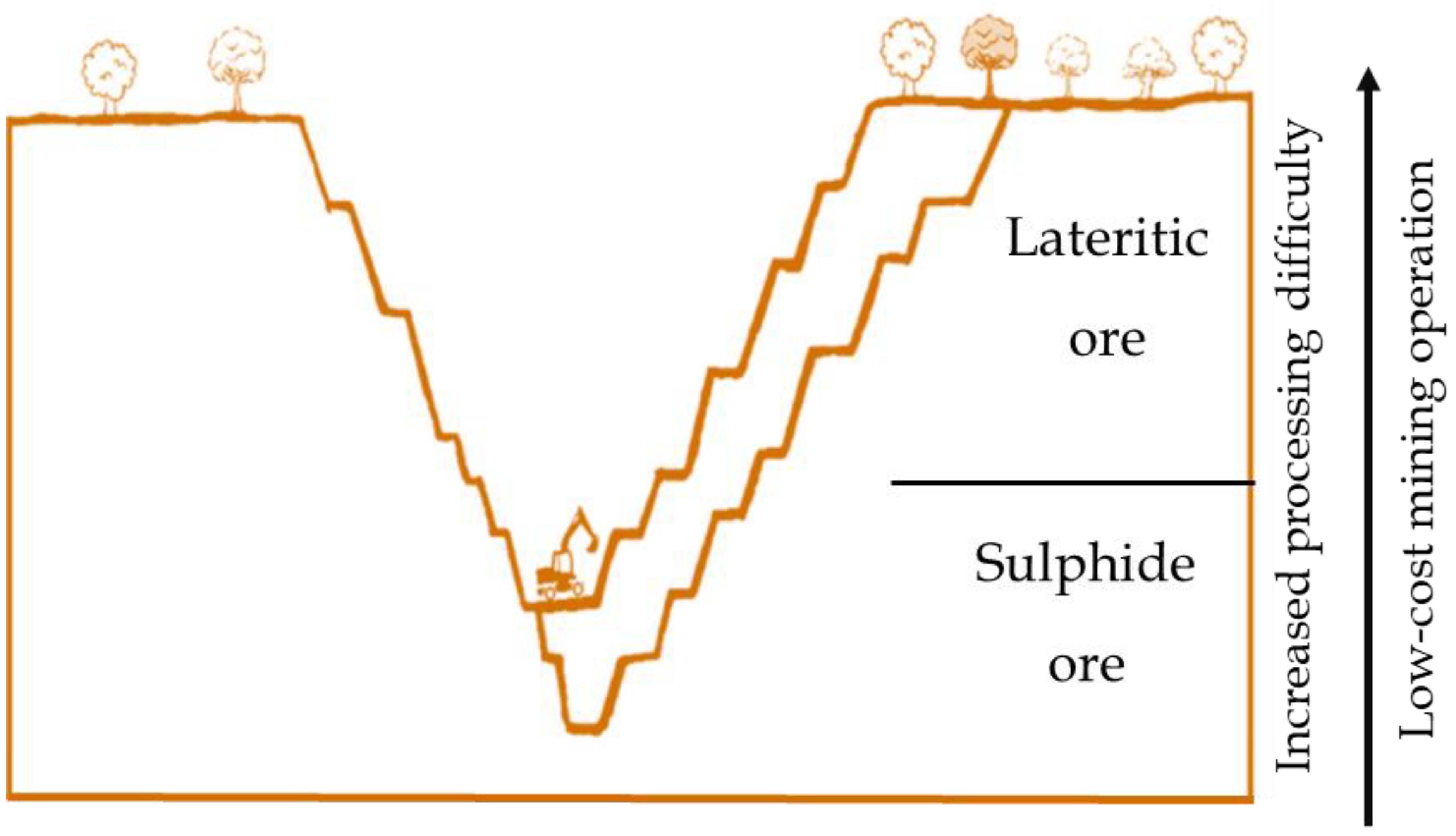
Unlike sulphide ores formed from volcanic or hydrothermal processes, typically containing copper, cobalt, and precious metals (e.g., gold, PGMs, rhodium), laterite deposits are developed through prolong weathering of ultramafic rocks occurring near the earth surface (Figure 4) [37]. They are generally characterized as mineralogically complex and chemically variable, low, often sub-economic, metal grades. Nickel–cobalt laterites are a heterogenous mixture of hydrated iron oxides and hydrous magnesium silicates derived from primary ferromagnesian minerals such as olivine, pyroxene, and amphibole, which initially contained around 0.2%–0.4% Ni [4,6]. Acidified water infiltrates weathered material dissolving iron, nickel, magnesium, and quartz. Iron subsequently oxidizes and precipitates as hydrated iron oxides, primarily goethite (FeOOH) and hematite (Fe2O3), forming near-surface horizons capable of hosting appreciable concentrations of Ni and Co. Idealized nickel sulphide and laterite deposit is shown in (Figure 4), depicting a low-cost mining operation for the lateritic ores, as it is formed near the earth surface.
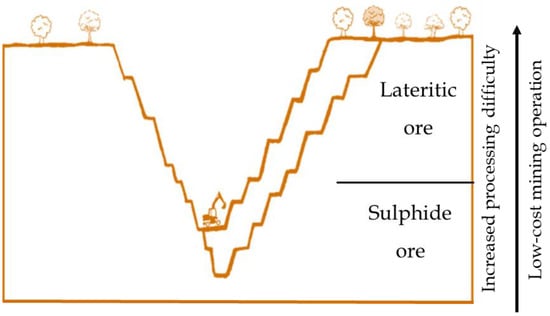
Figure 4.
Idealized cross-section of a nickel deposit showing lateritic ore near surface and sulphide ore at depth.
Closer to the bedrock, there is an increased concentration of Mg, as it is soluble and gets transported with the groundwater. The precipitated minerals undergo continuous attack to fresh acidic groundwater, a consequence of surface erosion and rainwater penetration. This persistent weathering process amplifies the concentration of nickel in secondary minerals, leading to elevated nickel levels in the vicinity of the bedrock [38,39]. Nickel and cobalt are incorporated largely by substitution for Fe and Mg within the iron oxides and silicate minerals, substantiating the absence of a distinct nickel–cobalt phase in lateritic ores. Typically, the weathering profiles a distinct mineral composition from the surface to the bedrock, vertically zoned into ferricrete, limonite, transition, and saprolite (Figure 5), with nickeliferous limonite [(Fe, Ni)(OOH)] and garnierite (Mg[Ni, Co]3Si2O5(OH)4), identified as the principal ore minerals. Identifying these layers is crucial for determining the appropriate extraction method, as nickel laterite deposits even from the same region can be quite variable in nature [14].
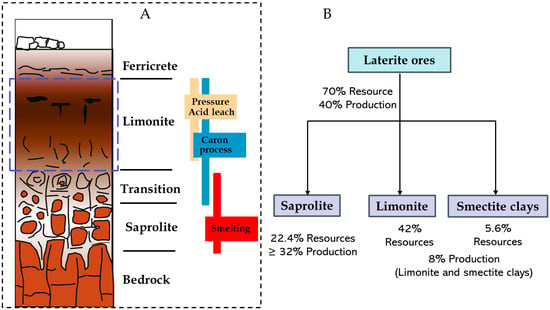
Figure 5.
(A) typical weathering profile for nickel laterite ores depicting their processing route and (B) resource proportion and contribution to current Ni production. Dash blue rectangle highlights the limonite resource fraction. Modified after Mudd and Jowitt [23].
Figure 5.
(A) typical weathering profile for nickel laterite ores depicting their processing route and (B) resource proportion and contribution to current Ni production. Dash blue rectangle highlights the limonite resource fraction. Modified after Mudd and Jowitt [23].
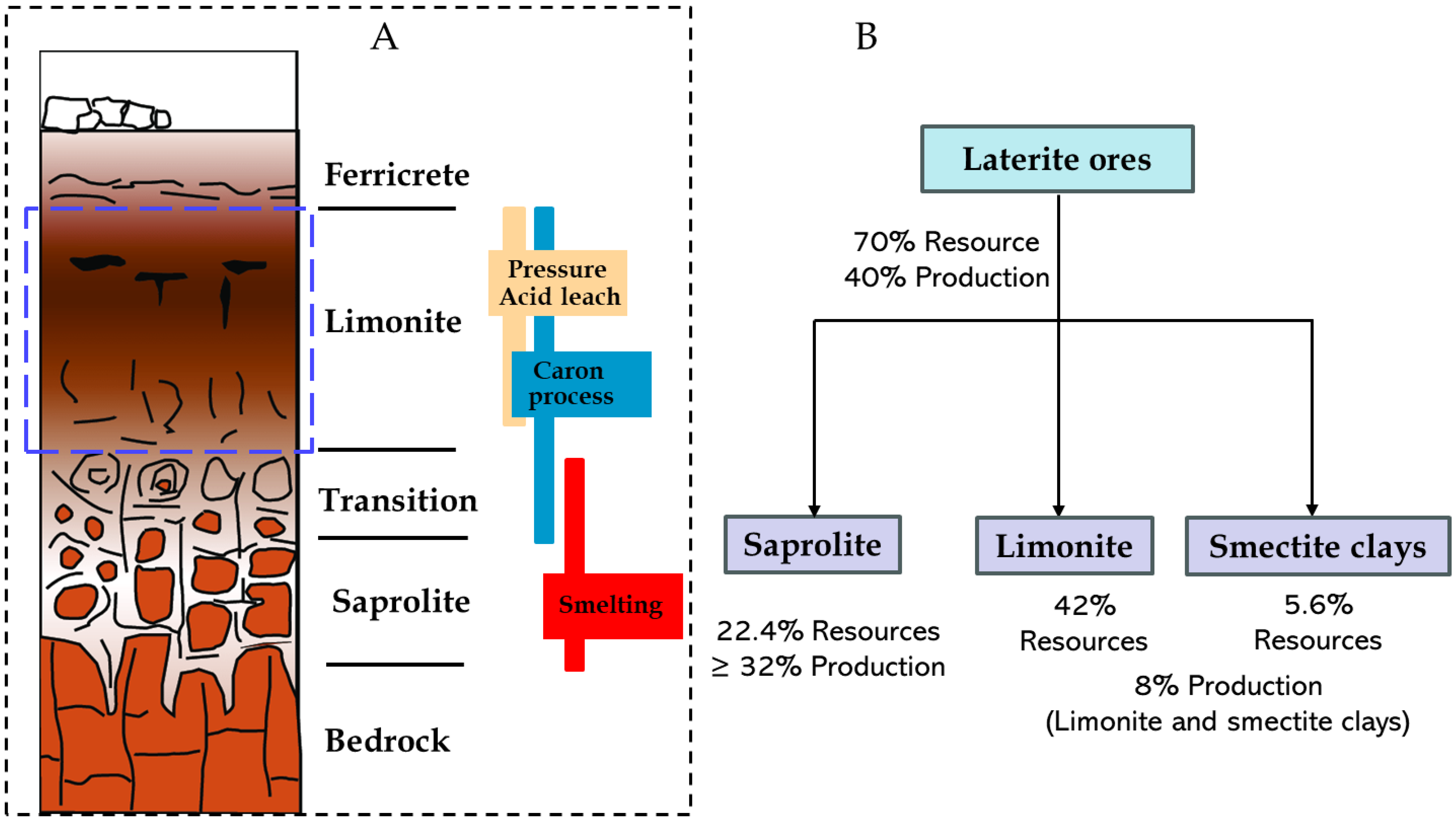
The ferricrete acts as an unmineralized cap that protects underlying rich profile from erosion. The limonite zone, highlighted by the dashed blue box (Figure 5), represents the largest proportion of global laterite deposits, and is characterized by high iron and low MgO content due to downward leaching and removal of Mg through the profile. It is dominated by Fe hydroxides and oxides where nickel is primarily hosted in goethite and occasionally in Co–Ni-rich manganese oxides [40]. The generic formula for goethite, (Fe, Ni)OOH, is used to represent limonitic ores in geochemical modeling, and are sometimes referred to as goethitic ores [7,34]. Nickel and cobalt are considered to be present in goethite in three modes, (i) associated with amorphous or poorly crystalline goethite, (ii) weakly adsorbed to the crystalline goethite surface, and (iii) as a substituent in the goethite structure [41]. Whittington and Muir [15] reported that nickel and cobalt occur predominantly via structural substitution within the goethite lattice rather than surface adsorption. Structure of partial substitution of Ni for Fe is shown in Mweene et al. [42].
The transition zone, rich in Fe, Mg, Ca, Na, and Al are known as layer of smectite clays. It denotes a more weathered, poorly drained areas above the saprolite which contains significant garnierite and clays like nontronite (Na0.3Fe2(Si, Al)4O10(OH)2·nH2O). Nontronite plays a role analogous to goethite where Ni is substituted for Fe in the octahedral sites, so that a substantial proportion of Ni in this zone is clay-hosted [5,43]. Smectite-rich material is sometimes referred to as siliceous goethite when microcrystalline silica is intimately associated with goethite [44]. The saprolite zone, developed closest to the ultramafic bedrock, is less chemically altered than limonite, with lower Fe but higher MgO contents. It is characterized by an absolute enrichment of Ni in the saprolite zone which comprises altered primary minerals such as secondary serpentine, and neoformed goethite, smectite clays, and garnierite.
It is mostly composed of magnesium hydro silicates or serpentines such as chrysotile, Mg3Si2O5(OH)4, with Ni/Co substituting Mg to form garnierite (Mg[Ni, Co]3Si2O5(OH)4) [15,45]. Saprolite ores are generally harder and more serpentinized than the limonite ores, and the serpentine minerals can assume different chemistries through isomorphous substitution. For example, Al may replace Si, and Al for Mg; however, when large species (e.g., Fe2+) replace Mg in the crystal lattice, a structural misfit occurs, hence, that form of substitution hardly happens. Serpentinized structure with Ni substituting Mg is illustrated by Mweene et al. [42]. A summary of laterite deposits and respective metal grades is presented in Table 2.

Table 2.
Various laterite deposits and respective metal (oxide) grades.
Table 2.
Various laterite deposits and respective metal (oxide) grades.
| Type of Ore | Grade % | Deposits | References | ||||
|---|---|---|---|---|---|---|---|
| Ni | Co | Fe | MgO | SiO2 | |||
| Limonite/ oxide | 1.24 | 0.11 | 59.8 | 0.26 | 2.66 | Goro (New Caledonia), Moa Bay (Cuba), Sampla (Indonesia), Cawse (Australia) | [4] |
| Smectite clay | 1.24 | 0.09 | 21.7 | 4.02 | 42.1 | Murrin Murrin (Australia), Camaguey (Cuba) | [15] |
| Saprolite | 1.44 | 0.05 | 9.29 | 13.02 | 21.4 | Koniambo (New Caledonia), Sorowako, Bahodoqi (Indonesia) | [4] |
Mineral Association in Nickel-Cobalt Laterite
Nickel and cobalt have been associated with various minerals in lateritic ores (Table 3) [14]. Notably, encapsulation of nickel and cobalt in the goethite and serpentine structure by a combination of lattice substitution and surface complexation, leads to formation of complex mineralogical structures which influence leaching behavior and extraction efficiency [43]. These structures are exacerbated by fine dissemination of Ni- and Co-bearing minerals which further complicate processing [18,46,47]. Acid consuming saprolitic Mg silicates ores tend to require relatively mild sulphuric acid, whereas refractory limonitic oxide ores commonly demand more aggressive leach conditions and longer residence times [48]. As a result, detailed mineralogical characterization has become central in process selection, modeling leach chemistry, and optimizing hydrometallurgical routes for Ni–Co laterites in both existing and emerging projects [15,40].

Table 3.
Nickel and cobalt mineral associations in nickel laterite ores [14].
3.2. Distribution of Nickel Laterite Deposits
Nickel laterite deposits are predominantly formed under warm, humid, tropical to subtropical climatic regions that promote intense chemical and mechanical weathering of ultramafic rocks. Consequently, the largest laterite-hosted nickel resources occur in Indonesia, New Caledonia, and Australia, which collectively account for roughly 60% of the global inventory [49]. Globally, limonitic ores constitute around 42% of total lateritic nickel resources, while Mg silicate and clay-dominant types make up ~32% and 7% respectively, shown in (Figure 6). The silicate (saprolite) laterites are economically significant, representing the second largest resource but typically the highest in nickel grade among the three deposit types [5,50]. These ores have been the principal feedstocks for laterite-derived nickel production, with major deposits located in New Caledonia, Indonesia, the Philippines, and Colombia [5]. Notably, New Caledonia hosts high grade limonitic and silicate ores ((Ni, Mg)3Si2O5(OH)4), while Indonesian laterites are mineralogically dominated by goethite ((Fe, Ni)OOH), lizardite ((Ni, Fe, Mg)3Si2O5(OH)4), chromium oxide (CrO2), and quartz (SiO2) [51]. Hydrous silicate laterites are characteristic of well-drained, tectonically active tropical regions and are generally absent in Western Australia, where climatic and geological conditions are less conducive to their formation [50]. Australian lateritic nickel deposits, in contrast, are often dominated by smectite clays, less saprolite material, and contain lower Ni grades in their limonitic zones compared with those of New Caledonia or Moa Bay [5,52]. Figure 6 shows the major nickel laterite deposits and their global distribution.
In addition, notable Ni–Co-bearing materials occur in association with chromite deposits in the Sukinda Valley, Odisha State, India. The combined lateritic and chromite overburden reserves in this region are estimated at about 230 Mt. For each ton of chromite mined, approximately 7–10 t of waste overburden containing ~0.5% Ni and 0.03% Co are generated [53,54]. Detailed mineralogical and concentration of Ni laterites from the Sukinda Valley is discussed [18]. Despite multiple attempts, the recovery of Ni and Co from this material has been limited due to high impurity levels and lean metal grades [55]. If the nickel grade could feasibly be increased, this otherwise discarded resource would offer substantial economic potential for India, which currently meets all its nickel demand through imports.
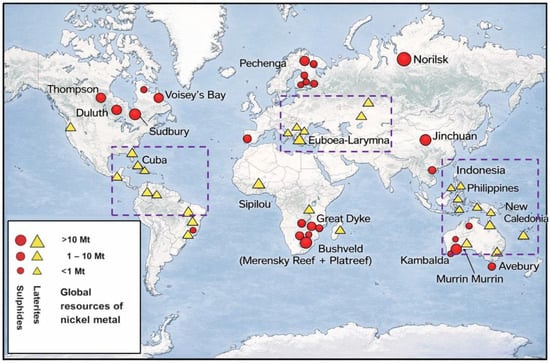
Figure 6.
Global nickel laterite deposits. Dash purple rectangles represent major laterite deposits and their geological setting. Modified after [56].
Figure 6.
Global nickel laterite deposits. Dash purple rectangles represent major laterite deposits and their geological setting. Modified after [56].
3.3. Nickel Resources in Australia
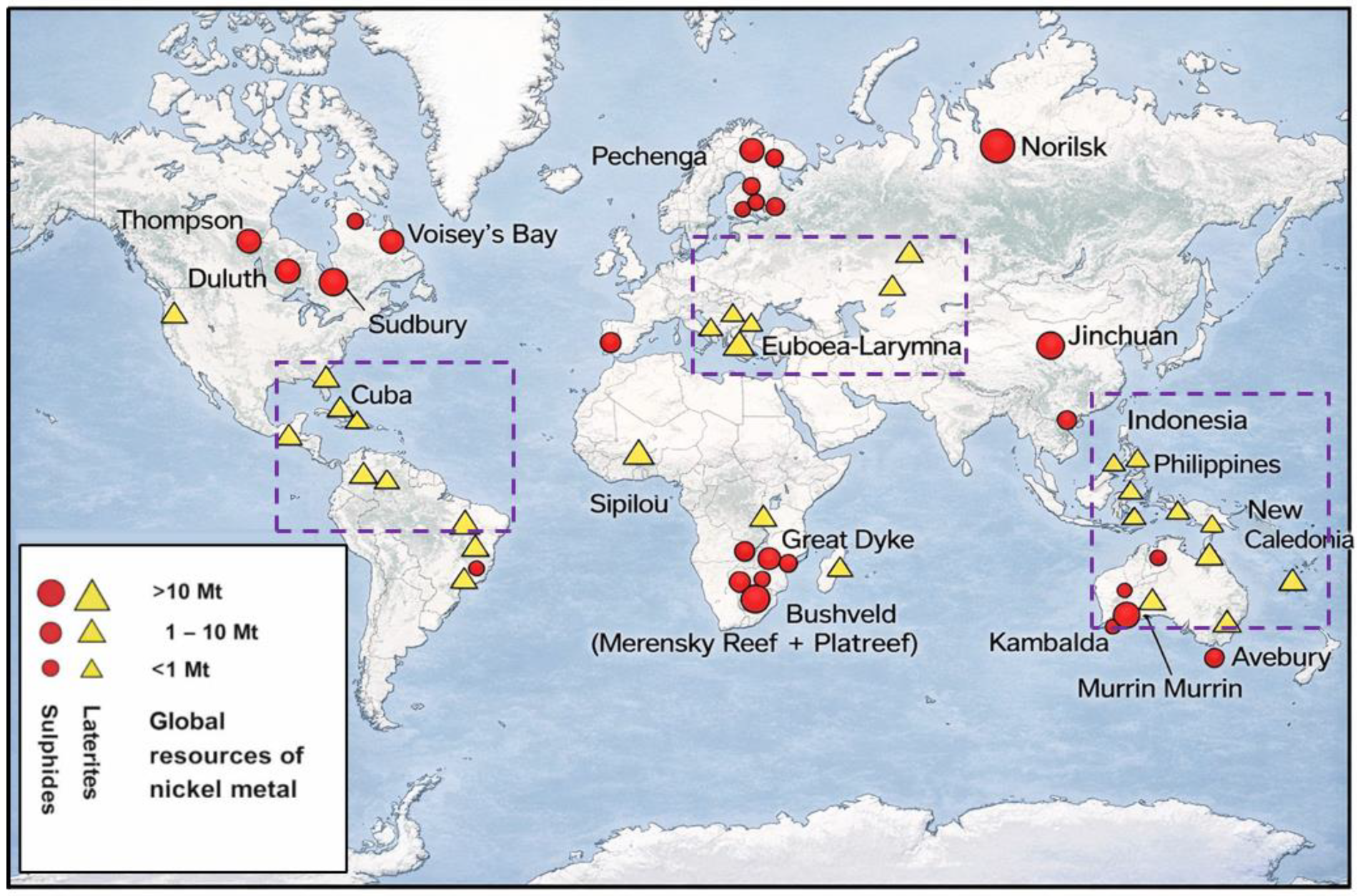
Australia hosts a diverse range of nickel deposit types, including komatiitic volcanic hosted Ni–Cu–PGE sulphide deposits, lateritic nickel deposits, and, to a lesser extent, tholeiitic intrusion-hosted sulphide and hydrothermal deposits [57]. Most of Australia’s nickel resources occur as nickel oxides in lateritic deposits, representing ~69% of total identified reserves. However, 82% of the nation’s annual nickel production is currently derived from sulphide ores hosted in komatiite deposits (Figure 7B) [35,37]. Figure 7A illustrates the distribution of these deposits, while Figure 7B outlines the relative share of production derived from sulphide and laterite sources. Table 4 shows the economic demonstrated Ni and Co resources. As of 2024, Australia contributed around 110,000 t to global nickel production [21], sourced from eight operational mines. Among these operational mines, the Murrin Murrin operation in Western Australia is the only active Australian lateritic nickel producers. Given the projected increase in global nickel consumption, exploration and development of underutilized laterite resources have become increasingly critical as sulphidic reserves approach depletion [58].

Table 4.
Australian economic demonstrated Ni and Co resources. Redacted after Elias [35].
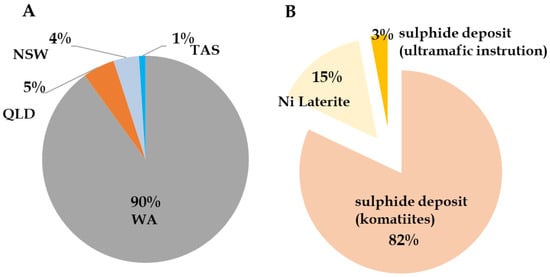
Figure 7.
Distribution of nickel resource (A) and production related to sulphide and laterite deposits (B) in Australia. Modified after [37].
Figure 7.
Distribution of nickel resource (A) and production related to sulphide and laterite deposits (B) in Australia. Modified after [37].
Characteristics of Australian Laterite Ores
The Murrin Murrin Ni laterite deposit lies approximately 60 km E of Leonora within the NE Yilgarn Craton. The deposit is dominated by smectite-type clays, with nontronite as the principal nickel host, containing up to 4% Ni [59]. Other clay minerals, including Mg–Fe-rich and Fe-rich montmorillonite and the Mg-rich variety saponite, are present alongside chlorite, magnesite, and poorly crystalline silica containing trace nickel. The ores developed from serpentinized peridotite, specifically, komatiitic olivine cumulates within the Archean Norseman–Wiluna Greenstone Belt [59]. Comparable smectite-type laterite deposits in the Yilgarn Craton include Bulong and Kalpini [50]. The absence of garnierite at Murrin Murrin is attributed to the fully serpentinized nature of the parent ultramafic rocks [59].
The Ravensthorpe operation, which is currently stopped for economic reasons, has its nickel laterite deposit located 450 km SE of Perth within the Youanmi Terrane of the Yilgarn Craton. The deposits occur in komatiitic volcanics and subordinate high Mg basalts and gabbros. It represents a typical oxide-type lateritic deposit dominated by Fe-oxyhydroxides (goethite), associated with free silica, particularly within the upper saprolite and pedolith zones [60]. The ore bodies are derived from weathering of silica-rich, Al-poor dunites and peridotites, with Ni hosted in Fe and Mn oxides. Cobalt similarly occurs in Mn oxides within the limonite and upper clay zones [59]. Other oxide-type lateritic deposits in Western Australia include Cawse-Siberia, Marshall Pools, and Weld Range [50].
4. Processing Methods
The processing of nickel laterites generally involves two major stages: (i) preconcentration and/or pretreatment, followed by (ii) extraction through either pyrometallurgical or hydrometallurgical routes. Both approaches aim to produce nickel and cobalt concentrations in the form of metal alloys or aqueous solutions. The techno-economic feasibility of each route depends primarily on operational parameters, mineralogical suitability, and project scale [53].
4.1. Preconcentration Methods
The objective of beneficiation or preconcentration is to upgrade ore quality to lower production costs, reduce reagent and energy consumption, and improve throughput while concurrently increasing the production rate [53]. The degree of beneficiation achievable, as well as the final concentrate grade, is strongly influenced by both the characteristics of the ore and the downstream processing route. The complex mineralogy of laterites, particularly the fine dissemination of Ni- and Co-bearing phases and the absence of discrete mineral hosts poses major constraints on beneficiation efficiency [61]. Consequently, coarse fraction with no or low nickel and cobalt rejection from run of mine (ROM) ore, remains the most common preconcentration practice [12]. Other reported separation methods include dense media, gravity, magnetic and electrostatic separation, flotation, and various pretreatment processes such as thermal roasting have been investigated at coarse and fine particle levels under laboratory and pilot plant scale conditions by several researchers [18,46,62,63]. However, none has yet demonstrated sufficient efficiency or selectivity for full scale industrial implementation, highlighting the need for systematic techno-economic comparisons across methods. Most attempts at upgrading have been impeded by the extremely fine grain size of Ni-bearing phases, requiring high degrees of liberation in the very fine fractions that are difficult to achieve by conventional mechanical means [12].
4.1.1. Flotation
Flotation studies have been conducted on various laterite types using a wide range of reagents; however, only minor nickel upgrading has been achieved [12,42,46]. Quast et al. [46] conducted numbers of feed preparation, including desliming, screening, and size classification prior to batch flotation tests on Western Australian SG nickel laterite ore. Collectors such as Flotigam EDA were tested for selective recovery of iron minerals (goethite, hematite) and silicates [64,65]. Under optimal laboratory conditions (400 g/t EDA, 300 g/t starch, pH 10), the Ni content increased from 1.0 to 1.4%, and 43% recovery and 31% mass pull. While the removal of ultrafine slimes slightly improved flotation performance, overall results remained limited due to poor mineral selectivity and fine dissemination of Ni across multiple mineral phases. Additionally, the use of EDA as a frother generated overly stable, voluminous froths in slime-rich samples, indicating poor plant scale applicability due to increased entrainment rather than true particle–bubble attachment.
To address fine particle challenges, selective flocculation flotation was proposed [66]. Application of this technique to SAP, SG, and G laterites resulted in modest performance. Ni grade in SAP was upgraded by ~40% at 70% recovery, slight upgrade for the SG while negligible improvements was observed for the G ore [67]. The inherently fine and complex dissemination of Ni and Co within host gangue minerals necessitates fine grinding for liberation; however, flotation recovery of fine particles <38 µm is intrinsically poor in conventional mechanical flotation cells as illustrated by the elephant curve below (Figure 8) [68]. The tail end of the curve represents poor recoveries of fine particles, highlighted by the solid pink circle. Recent advances in flotation reactor design, such as reactor–separator and reflux flotation cells, offer higher probabilities of particle–bubble contact and improved froth stability for fine and ultrafine particles [69]. These technologies present promising avenues for enhancing laterite flotation efficiency but warrant further pilot scale validation under realistic process conditions.
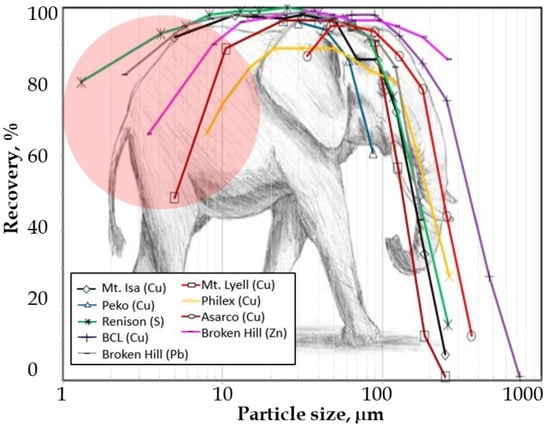
Figure 8.
Typical flotation recoveries at various size fractions in the conventional mechanical flotation cell. The pink circle highlights challenges in fine particle flotation. Modified after [68].
Figure 8.
Typical flotation recoveries at various size fractions in the conventional mechanical flotation cell. The pink circle highlights challenges in fine particle flotation. Modified after [68].
4.1.2. Selective Comminution
Recent studies indicate that selective comminution, combined with classification, can achieve higher nickel upgrades than conventional physical–chemical separation methods such as flotation or magnetic separation [47,70]. Although Ni-and-Co-bearing minerals in laterites are typically fine grained and disseminated among various host phases, variations in Ni and Co deportment across minerals offer opportunities for selective grinding and subsequent particle separation. Differences in mineral hardness and work index lead to different breakage rates during milling, which can be exploited to preferentially liberate and concentrate Ni-rich minerals [71]. By means of selective comminution, two-product distribution is generated, thus coarser, hard fragments, and softer fine material that can be separated effectively using classification techniques. Factors such as grinding time, particle size, grinding media characteristics, and the presence of hard minerals (e.g., quartz) strongly influence breakage and liberation [42,72,73]. For instance, Tong et al. [73] found that Australian SG laterite exhibited non-first order breakage kinetics (Figure 9), with breakage rates decreasing with larger feed sizes and longer grinding durations.
As mentioned earlier, mineralogical composition also plays a key role in selective grinding performance. Differences in hard mineral contents with the softer minerals influence both breakage rates and separation efficiencies. For example, Ou et al. [74] and Yusupov et al. [75] studied the effect of selective grinding of low-grade bauxite and quartz–feldspar ores respectively to optimize the Al/Si ratio in bauxites and quartz–feldspar ratios. It was observed that soft feldspars report in the finer fractions while quartz, which is the hard mineral, reports in the coarser ones. Comparative studies of SAP, SG, and G ores revealed Si/Ni breakage variations of 140%, 87.5%, and 38.5% respectively [47,70,73]. Despite the higher quartz content of SG ore, SAP exhibited a greater Si–Ni ratio, attributed to higher work indexes of its constituent minerals. G ores, being relatively quartz-deficient, displayed the lowest degree of selective breakage.
The size and type of grinding media are additional control factors [71]. Investigation using 12.7 mm, 6 mm, and zero grinding media on −3.35 + 1.70 mm Greek laterite showed that omitting grinding media yielded the best Ni upgrades (<0.075 mm size fraction). Detailed product characteristics after grinding is found in [42,71]. Acquah et al. [72] observed that particle size influenced grinding performance of G ore, with decreasing particle size deteriorating its efficiency. It was reported that selectivity was achieved at short grinding times, typically 0.1 min, for mid-range size fractions (−1180 + 600 µm, and −600 + 300 µm), producing Ni grades of ~1.2% and 1.1%, respectively. Notably, feed sizing was determined as a critical parameter in selective grinding, as the unsized feed significantly reduced upgrading potential and process selectivity. Future approaches should involve mixed-size grinding media or staged milling to accelerate selective breakage of soft minerals and improve Ni enrichment [42].
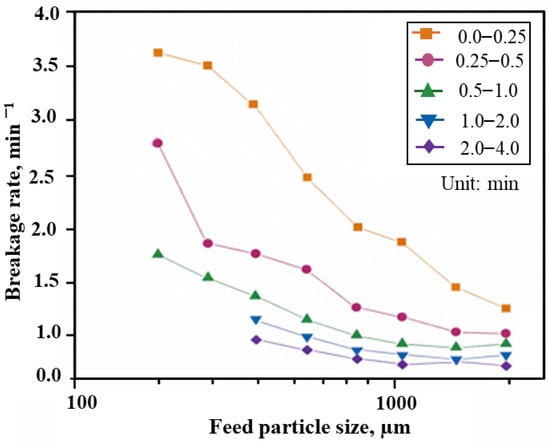
Figure 9.
Effect of feed particle size and grinding time on the breakage rate of siliceous goethite. Modified after Tong et al. [73].
Figure 9.
Effect of feed particle size and grinding time on the breakage rate of siliceous goethite. Modified after Tong et al. [73].
Optimal particle liberation thus depends on careful adjustment of operational parameters and the selection of effective milling equipment [76,77]. Improvements in Ni grade have also been reported for low grade chromite overburden from Sukinda, Odisha through deagglomeration, sieving, and hydrocyclone classification [42]. In many laterites, naturally occurring agglomerates and high isoelectric point (IEP) minerals cause barren particles to coat Ni and Co rich surfaces. Controlled abrasion during mild grinding can liberate these soft, nickel-bearing layers, facilitating effective preconcentration [78]. The addition of dispersants such as sodium hexametaphosphate (SHMP) enhances selective comminution by improving particle dispersion and preventing unwanted particle aggregation [72]. Introduction of SHMP induces strong negative zeta potentials on limonite surfaces, shifting the IEP from ~6.9 to undetectable IEP within the studied pH range (Figure 10) and thereby enabling Ni recovery increase [79]. Notably, determining the optimized concentration is prudent as higher concentrations have minimal additional impact.
While Ni deportment after selective comminution is well understood, Co distribution has received limited attention. Acquah et al. [72] reported that Ni concentrates preferentially in fine fractions, whereas Co is enriched in coarser products, consistent with its association with Mn-oxides (asbolane) [48,80]. The predominance of Co in coarser fractions may result from shorter grinding duration (<0.25 min), which is insufficient to fully liberate Co-rich minerals. Multistage selective comminution could improve Co liberation but risks diluting Ni grade due to over-grinding of barren materials. Instead, application of specific grinding strategies coupled with tailored downstream processing pathways are recommended. On this basis, two integrated extraction strategies were proposed to maximize metal recovery and process efficiency after selective comminution. A proposed conceptual flowsheet for selective comminution-based preconcentration preceding acid leaching is shown in Figure 11.
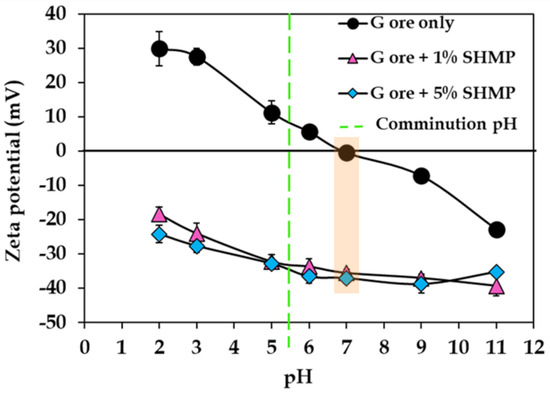
Figure 10.
Ore electrokinetic measurement as a function of pH with and without SHMP. Dashed green line represents the pH at which comminution was performed. G denotes goethitic ore, SHMP means sodium hexametaphosphate, and the orange rectangle represents the pH where IEP is undetectable in the presence of SHMP. Modified after [72].
Figure 10.
Ore electrokinetic measurement as a function of pH with and without SHMP. Dashed green line represents the pH at which comminution was performed. G denotes goethitic ore, SHMP means sodium hexametaphosphate, and the orange rectangle represents the pH where IEP is undetectable in the presence of SHMP. Modified after [72].
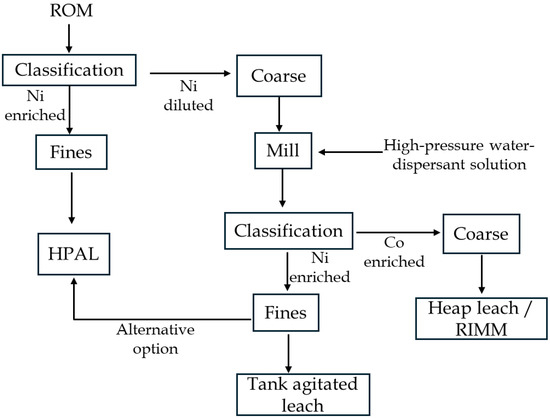
Figure 11.
Proposed flowsheet for laterite preconcentration through selective classification and comminution prior to acid leaching.
Figure 11.
Proposed flowsheet for laterite preconcentration through selective classification and comminution prior to acid leaching.
In the first configuration, (i) subjecting the underscreen Ni-enriched product to atmospheric tank leaching, while allocating Co-rich oversize material to heap leaching/RIMM, with HPAL applied to suitable fractions as required (Ni-rich fine feed). The alternative option is (ii) combining Ni-rich underscreen fraction with the HPAL feed, while maintaining the established processing route for the oversize products. Both strategies aim to enhance overall metal recovery and resource utilization without significant upgrading costs, particularly given that final Ni concentrate grades in laterite processing rarely exceed ~2%. Importantly, incorporating high-pressure water dispersion at an early stage of the flowsheet could potentially enhance feed disaggregation and improve mineral liberation. However, its application to bulk ROM material may increase overall water and dispersant consumption due to the large feed mass involved. A more efficient approach is to classify the ROM feed before milling, followed by controlled dispersant addition during selective grinding. This reduces unnecessary reagent and water consumption while improving selectivity and operational control (Figure 11).
4.1.3. Magnetic Separation
The effectiveness of magnetic separation for nickel laterites is largely constrained by poor mineral liberation arising from the ore’s complex mineralogy [62]. Operational details such as applied magnetic field intensity are not often reported, though a few studies have evaluated their effects [62,63]. The inclusion of dispersants and continuous agitation in magnetic separation operations could help to mitigate particle aggregation and enhance selectivity. Usually, barren covering minerals may suppress Ni–Co particles magnetic susceptibility and hinder effective separation. Farrokhpay et al. [63] demonstrated that magnetic separation improved nickel grade by ~20% for SAP ore, whereas SG and G ores showed negligible responses. The observed Ni upgrade in the SAP ore was attributed mainly to the removal of magnetic siderite and hematite, phases not directly associated with Ni. Complementary QEMSCAN analyses confirmed that silicate minerals (e.g., serpentine and clays) served as the principal Ni hosts. Similar results were obtained by Agatzini-Leonardou et al. [78]), who upgraded Ni from 1.4% to 1.7% in a serpentinite laterite from Greece, emphasizing that beneficiation success is highly dependent on mineralogy, degree of liberation, and the presence of magnetic phases. Other studies involving roasting prior to magnetic separation have been investigated [10,62,81]. Some of these studies have been summarized in Table 5. It was reported that Co and Mn were found preferentially in the magnetic products, while Ni followed weight distribution following roasting [62]. However, Tian et al. [10] reported that an increase of silicate minerals in limonitic laterite ore during selective reduction suppressed iron oxide reduction and enhanced nickel enrichment in the magnetic fraction. Table 5 summarizes selected studies on preconcentration and pretreatment of Ni- and Co-bearing laterites.

Table 5.
Summary of pretreatment/preconcentration of laterite studies prior to extractions.
Table 5.
Summary of pretreatment/preconcentration of laterite studies prior to extractions.
| Ore Type | Metal Grade (%) | Process | Operating Conditions | Recovery (%) | Reference |
|---|---|---|---|---|---|
| W A goethite | Ni: 0.91 Co: 0.06 | Thermal treatment prior to column leaching (CL) | Roasting Cond: 400 °C, 1 h CL Cond: 135 days, 100 g/L H2SO4, 10 L h−1 m−2 flowrate | Ni: 93 Co: 84 | [82] |
| Brazil Laterite (quartz) | Ni: 2.03 Co: 0.11 | Sulphation roasting before AL | Pre-roasting at 265 °C with 50 wt.% H2SO4 for 60 min and roasting at 740 °C for 20 min. AL Cond: 12.5 wt.% solid, 80 °C, 30 min | Ni: 83.3 Co: 90.0 | [83] |
| Colombia nickel laterite | Ni: 1.80 Co: 0.03 | Thermal treatment prior to acid leaching | Calcination Cond: 600 °C, 3% NaCl dosage, 1 h AL Cond: 24 h, ambient, 1M H2SO4, 1:20 S/L ratio | Ni: 64.7 | [9] |
| Philippine Limonite | Ni: 0.69 Co: 0.09 | Roasting ahead of AL | Reductive roasting: 700 °C, 40 min. AL Cond: 20 wt.% solid, 85–90 °C, 6 h, | Ni: 94.64 Co: 90.09 | [84] |
| Philippine Saprolite | Ni: 1.29 | Reduction before Mag. separation | Roasting Cond: 400 °C, 80 min. Wet Magnetic Cond: 0.1 T, 10 min | 6.44 Ni conc recovered at 96.9 | [10] |
| Indonesia limonite | Ni: 0.97 Co: 0.09 | Thermal treatment followed by Mag. separation | Reductive roasting Cond: 1100 °C, 1 h, 5% coal, 40% saprolite dosage WMS Cond. 1800 Gs, 6 min, 74 µm | 5.94 Ni conc. at recovery of 92.3 | [85] |
| China nickel laterite | Ni: 1.92 | Roasting ahead of WMS | RoastingCond:1100–1150 °C. WMS Cond: 50 mT | 7.5 Ni conc. at 96.72 | [86] |
| Türkiye limonite | Ni: 1.20 Co: 0.062 | Mechanical activation prior to AL | Milling time: 2h AL Cond: 85 °C, 2 h, 30% (w/w) H2SO4, ½.5 S/L | Ni: 80.6 Co: 84.5 | [87] |
| Türkiye limonite | Ni: 1.20 Co: 0.06 | Mechanochemical followed by AL | Activation Cond: 20 wt%, 1 h. AL Cond: 2 h, 85 °C, 300 g/L H2SO4, 1:2.5 S/L | Ni: 92.0 Co: 84.8 | [8] |
Attempts to upgrade India’s chromite overburden ores using pilot scale shaking table (for >150 µm fractions at 100 kg/h) followed by wet low intensity magnetic separation (for >45 µm fractions) yielded only a marginal increase in Ni content (from 1.0% to 1.1%) [88]. The limited enrichment was attributed to the complex mineralogy and variable surface potentials of constituent minerals. Earlier studies resulting in slight improvement in grade was linked to complex mineralogy of the ore associated with poor liberation of Ni/Co-containing minerals [12]. Reinforcing that fine grinding is necessary for liberation, however, magnetic separation is limited by the presence of fines. Furthermore, magnetic susceptibility of goethite decreases with decrease in particle size [81,89]. Emerging approaches such as upward feeding configurations in wet high intensity magnetic separators (WHIMS) have improved recovery in hematite-rich systems and may hold promise for Ni laterite applications. Future research could focus on refining separator design and investigating the ferromagnetic behavior of lateritic Ni minerals to optimize field intensities and feed conditions.
4.1.4. Gravity Separation
Specific density of minerals presents in laterite ores provide potential for beneficiation via gravity techniques such as Falcon concentrators, shaking tables, dense media separation, and settling–dispersion methods. Reported Ni upgrades are generally modest, with recoveries often below 50% [42]. Farrokhpay et al. [63] achieved Ni grade improvements of ~36% for SG and ~17% for SAP ores using a Falcon concentrator, while no significant upgrade was observed for G ore. Despite the density difference between iron oxides (4–5 g/cm3) and silicate minerals (~2.5 g/cm3), separation efficiency is limited by the fine dissemination of minerals, poor liberation, and the existence of similar physical characteristics [7,42,63]. The efficiency of gravity separation is particularly low for limonitic laterites, where Fe oxyhydroxides dominate over quartz or silicate phases. Nevertheless, certain improvements may be achieved by tailoring beneficiation routes to individual ore types. Siliceous and saprolitic ores generally show greater potential for upgrading than limonitic ores, though recoveries remain modest (e.g., <50%). For limonitic laterites, preliminary treatments such as partial phase transformation or selective separation of goethite from other constituents could improve subsequent beneficiation and hydrometallurgical processing performance [7]. Overall, while gravity and magnetic methods offer incremental gains, their practical application to lateritic ores remains limited without substantial mineralogical preconditioning or supplementary processing steps.
4.2. Pretreatment Methods
Pretreatment of nickel laterite ores aims to improve subsequent metal extraction efficiency by modifying mineralogical, structural, or surface characteristics. Among the most studied approaches are mechanical activation and thermal treatment, both enhancing mineral reactivity and leaching kinetics while potentially reducing energy and reagent consumption.
4.2.1. Mechanical Activation
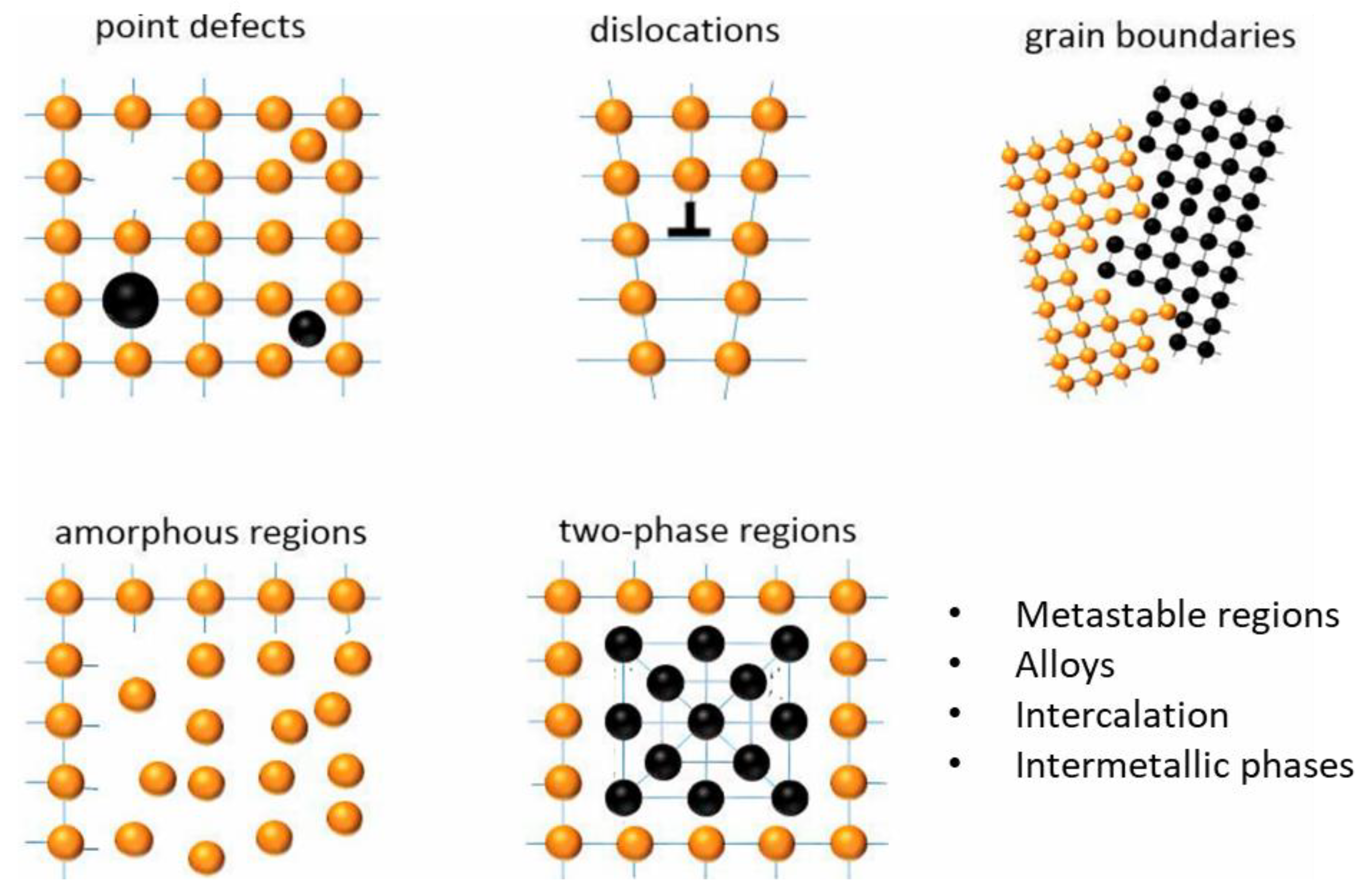
Mechanical activation (MA) involves applying mechanical energy to induce structural and surface modifications in minerals, thereby enhancing their reactivity [90]. The primary effect of MA arises from particle comminution, leading to altered physicochemical properties including increased surface area, lattice disorder, amorphization, and enhanced-defect density [91,92]. Figure 12 shows the type and concentration of defects resulting from mechanical processing of solid substances. The extent to which particles are altered under the influence of mechanical activation processes is dependent on the nature of the particles. Numerous studies [87,93,94,95] have demonstrated that these changes accelerate leaching reactions while lowering thermal and chemical energy requirements. Mechanical activation has proven effective across a variety of mineral systems. For example, improved reactivity was observed in alkali-activated low-grade clays due to structural disorder and increased specific surface area [96]. Gold extraction was improved from refractory flotation concentrates following MA treatment [95]. In sulfide systems such as sphalerite, leaching kinetics improved markedly following MA, supported by reduced crystallinity, altered morphology, and elevated amorphization [97]. Comparable improvements have been reported for other types including ilmenite, chalcopyrite, and REE minerals [98,99,100].
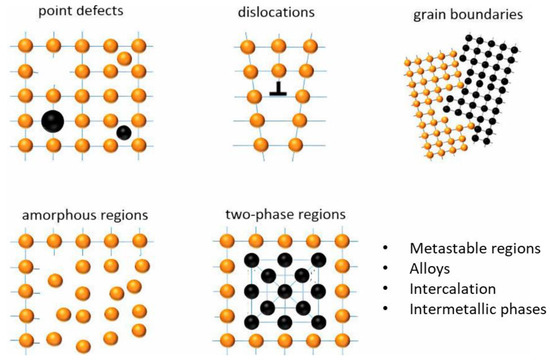
Figure 12.
Schematic illustration of structural changes induced by mechanical processing, including point defects, dislocations, grain boundaries, amorphous regions, and two-phase regions. Orange spheres represent atoms in the crystal lattice, black spheres represent defect or secondary-phase atoms, and blue lines indicate lattice planes. Modified after Asamoah et al. [101].
Figure 12.
Schematic illustration of structural changes induced by mechanical processing, including point defects, dislocations, grain boundaries, amorphous regions, and two-phase regions. Orange spheres represent atoms in the crystal lattice, black spheres represent defect or secondary-phase atoms, and blue lines indicate lattice planes. Modified after Asamoah et al. [101].
In the context of lateritic ores, mechanical activation using various mills has shown promise for enhancing nickel and cobalt extraction. Aaltonen et al. [94] achieved particle size reduction from 40 µm to 1–2 µm in an attritor mill, yielding 98% Ni, 96% Co, and 65% Fe extraction at 2.5% solids after leaching. For Turkish nickeliferous laterites, planetary mono mill activation induced pronounced amorphization and goethite dehydroxylation [20,102]. Similarly, Basturkcu et al. [87] and Basturkcu et al. [8] demonstrated that vibratory milling produced amorphous phases and of hydrosilicate partial dehydroxylation, improving atmospheric leaching amenability (Table 5). High energy mills such as stirred media mills are particularly advantageous for MA because they generate high acceleration forces and strong shear stress on particles [103,104,105].
These mills yield higher mass specific surface areas than planetary or vibratory mills and operate efficiently at lower temperatures, offering scalability for industrial applications [99,106]. Unlike conventional tumbling mills with their low energy densities [105], stirred mills embody sustainability principles by prioritizing attrition-based breakage and controlled stress intensity [104,107]. They utilize 30%–40% lower energy for fine and ultrafine grinding with less carbon footprint [107,108]. These benefits stem from small media (1–8 mm), high power densities, and optimized conditions (e.g., stirrer speed, solid loading) [109]. This lowers kWh/t, minimizes media wear and embodied energy, and leverages MA effects to enhance liberation at coarser sizes and decreases the overall comminution footprint [105].
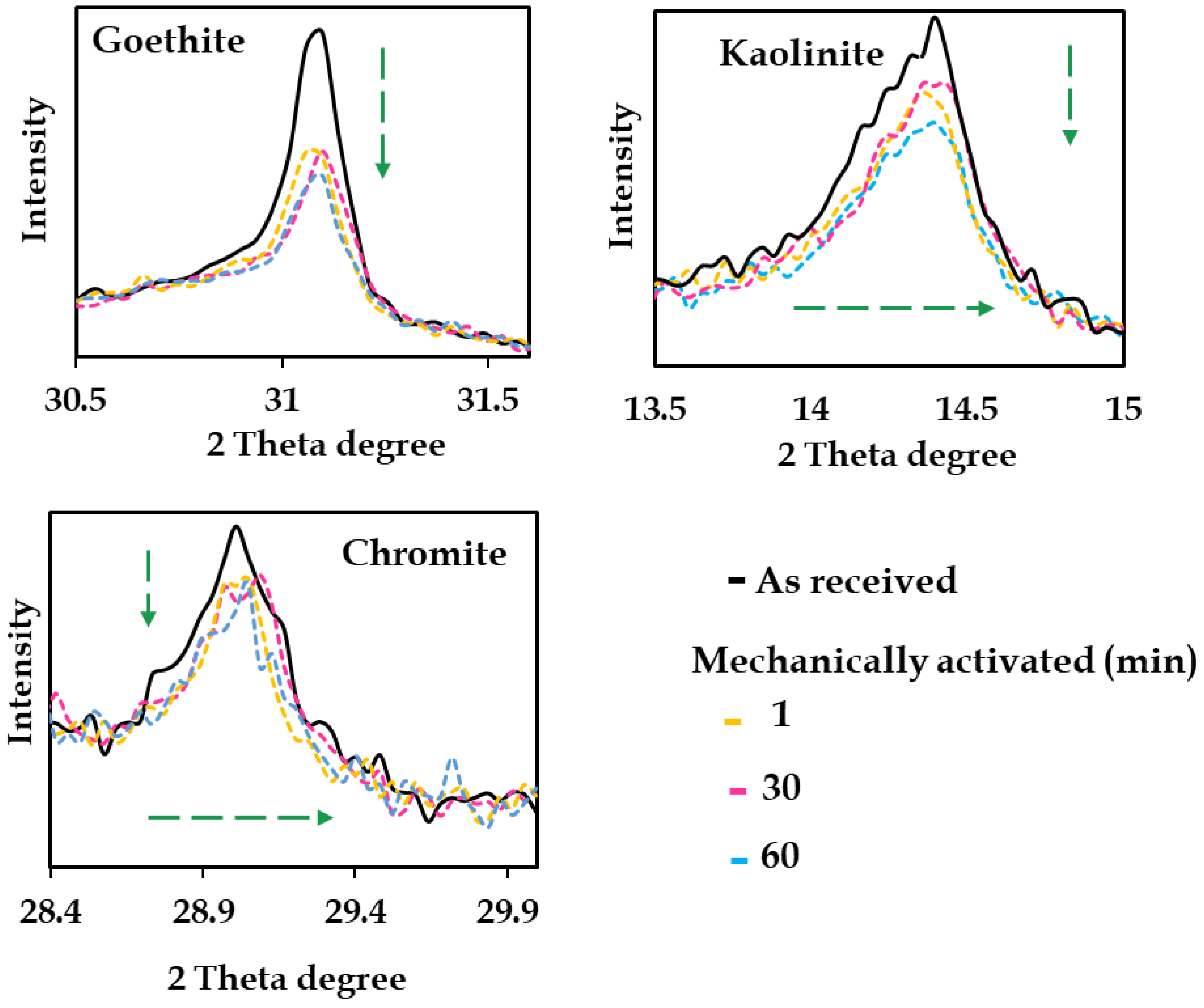
Mechanical activation on Australian nickel laterites has only recently been explored systematically using a stirred mill. Acquah et al. [11] found stirred mill activation of Western Australian limonite ore drove significant physical and structural modifications, including particle size reduction, surface area increase, and amorphization. These transformations exposed encapsulated Ni and Co phases, and improved leach recoveries. However, extended milling (beyond 60 min) yielded no further Ni/Co gains and led to particle aggregation, emphasizing the need for process optimization. While MA had negligible effects on slurry rheology, activation duration was identified as a critical factor controlling extraction kinetics. Typical structural alteration including peak attenuation (e.g., broadening, shifting) is illustrated in Figure 13.
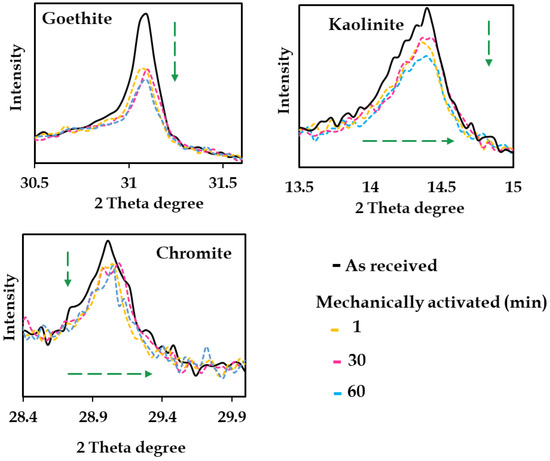
Figure 13.
Phase attenuation of as received and mechanically activated samples for (1, 30, and 60 min). Dashed green arrows show shortening and shifting of phases such as goethite, kaolinite, and chromite phase. Modified after Acquah et al. [11].
Figure 13.
Phase attenuation of as received and mechanically activated samples for (1, 30, and 60 min). Dashed green arrows show shortening and shifting of phases such as goethite, kaolinite, and chromite phase. Modified after Acquah et al. [11].
4.2.2. Thermal Pretreatment
Thermal pretreatment or calcination is another widely studied strategy to enhance metal extraction efficiency without relying on expensive autoclave HPAL technology [9,82,84,86,110,111]. Roasting studies under oxidizing and reducing conditions and additive incorporation have sought to modify mineral phases and magnetic properties ahead of preconcentration or leaching, discussed in detail [7,112]. Calcination induces dehydroxylation of goethite, transforming Fe-oxyhydroxide minerals into poorly ordered and porous minerals such as hematite, maghemite, or magnetite [7,62]. This transformation increases specific surface area and porosity, thereby improving acid accessibility and leaching kinetics [113]. Garces-Granda et al. [114] and Garces-Granda et al. [110] reported preferential Ni leaching post calcination, with congruent Ni–Fe dissolution observed below 800 °C.
Similarly, Landers and Gilkes [113] and Li et al. [82] observed that Ni–Fe dissolution were congruent before roasting. This indicates nickel was likely uniformly incorporated in goethite, but noticed preferential leaching of nickel over iron after roasting at 600 °C to 800 °C. They speculated that some of the nickel migrated toward the surface or voids of the neo-formed hematite during roasting, enhancing accessibility. However, discrete nickel phases were not detected under Transmission Electron Microscope (TEM) [115]. The mechanism of metal redistribution during roasting remains complex and may involve surface adsorption, partial incorporation into hematite lattices, or diffusion into microfractures. Notably, increased reaction rates observed after calcination stem partly from enhanced surface exposure (voids), implying structural rearrangements aids in facilitating ion mobility [82,111]. As shown in Table 5, calcination pretreatment prior to acid leaching enhance enhances Ni and Co extractability.
Calcination conditions, particularly temperature and time, strongly affect the outcome. Prolonged exposure or excessive heat can reduce surface area and promote Ni/Co re-incorporation into iron oxides through recrystallization, ultimately inhibiting leaching [82,116]. Determining the optimum roasting parameters is ore-dependent; notwithstanding, it is crucial to avoid unnecessary operational cost. Reported conditions include 430 °C at 2 h for Colombian limonite [110], 600 °C for Indonesian saprolites [117], and 400 °C at 60 min for Western Australian goethitic ores [82], consistent with subsequent findings by Acquah et al. [118]. Calcination primarily alters internal mineral structure rather than particle size reduction (Figure 14). However, BET specific surface area markedly increased from 26.2 to 41.4 m2/g, indicating a more porous mineral structure, rather than simple size reduction [118]. The enhanced mineral porosity arises from dehydroxylation and associated structural water and OH¯ removal. These structural modifications increase minerals susceptible to acid attack during subsequent leaching.
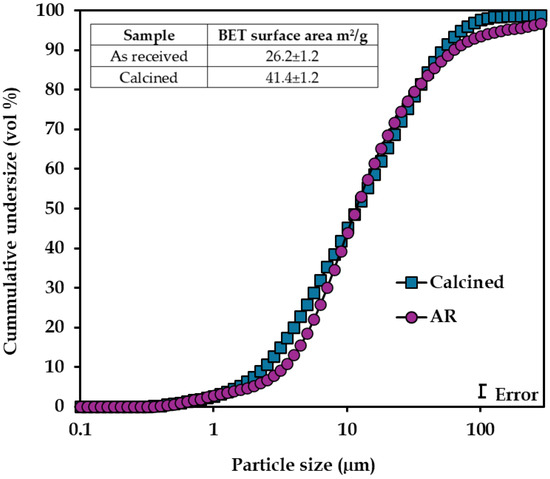
Figure 14.
Cumulative particle size distribution and BET-specific surface area of the as-received (AR) and calcined sample. Modified after Acquah et al. [118].
Figure 14.
Cumulative particle size distribution and BET-specific surface area of the as-received (AR) and calcined sample. Modified after Acquah et al. [118].
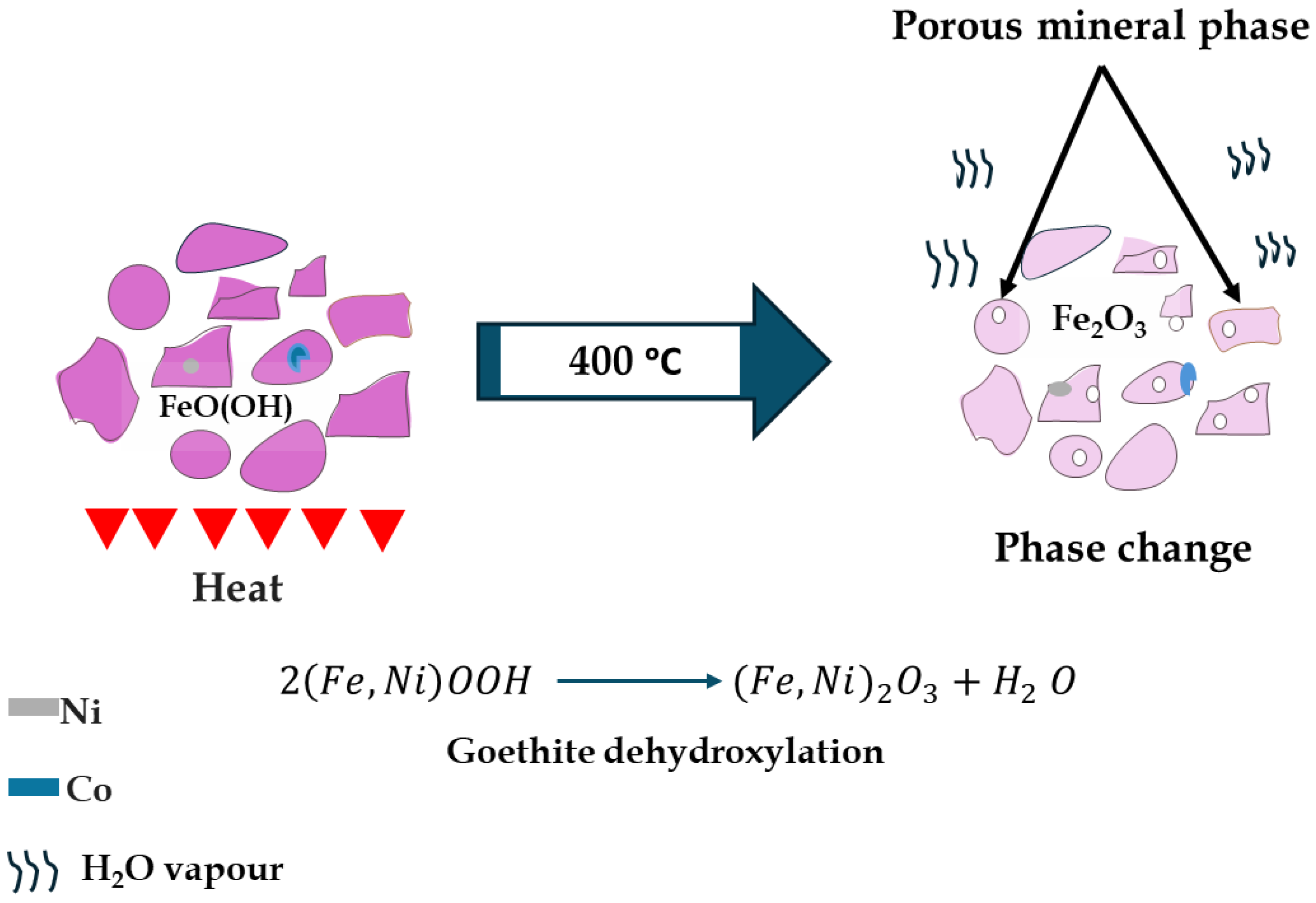
Leaching kinetics modeling revealed a marked reduction in apparent activation energy (Ea) from ~72–75 kJ/mol (pre-calcination) to 27–32 kJ/mol (post-calcination), indicating a shift from chemical control to mixed-control behavior involving both surface reaction and diffusion through a porous product layer. This substantial Ea decrease suggests calcination facilitates more energy-efficient leaching of refractory laterites. A schematic representation of the mineralogical transformations during calcination is illustrated in Figure 15.
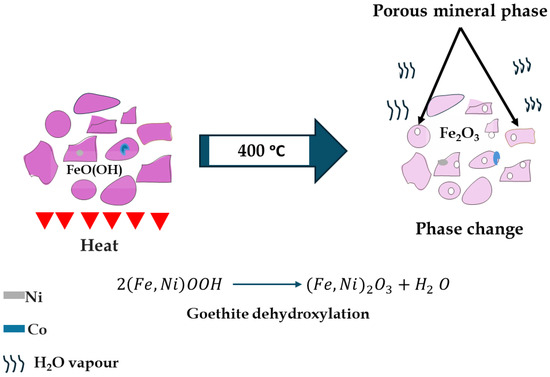
Figure 15.
Schematic representation of phase changes after calcination.
4.3. Pyrometallurgical Methods
Pyrometallurgical processing of nickel laterites is highly energy intensive and generally follows a conventional flowsheet comprising rotary-kiln drying, calcination or reduction at 850–1000 °C, and smelting in an electric furnace at ~1600 °C. The process yields either ferronickel or low iron matte products [36]. Among available technologies, the rotary-kiln electric furnace (RKEF) route remains the most robust and widely adopted, but it is economically feasible for only high-grade lateritic ores containing > 2 wt.% Ni [17]. RKEF processing is best suited to saprolite and sulphide ores that display favorable mineralogical ratios, specifically, Fe/Ni < 12, Ni/Co > 30 and SiO2/MgO < 1.9 for stable smelting and alloy formation. These mineralogical requirements constrain applicability to selective ore types and necessitate careful blending or beneficiation prior to feed preparation. Ferronickel smelting applicability to saprolite laterite ores achieves 90%–95% Ni, and ≤85% Co recoveries. A summary of optimal mineralogical conditions for RKEF operations is provided in Table 6. Process CapEX and OpEX are shown in Table 7.

Table 6.
Conditions for ferronickel smelting process applicability.

Table 7.
Typical costs of leaching technologies employed for laterite nickel extraction. Modified after [17].
4.4. Hydrometallurgical Processes
Hydrometallurgical techniques such as the Caron process, High-pressure acid Leaching (HPAL), Heap leaching (HL), and Atmospheric leaching (AL) are widely applied for low-grade and Fe-oxide-dominated lateritic ores [119,120,121]. These techniques allow efficient treatment of refractory limonite and tailing materials that are unsuitable for pyrometallurgical approaches. The selected extraction route is highly dependent on ore mineralogy, acid-consuming gangue content, and associated trade-offs in reagent demand, recovery rate, and CO2 emissions.
4.4.1. Pyro-Hydro (Caron) Process
The Caron process is a hybrid pyro-hydrometallurgical method developed for Ni–Co extraction from low grade laterites [122]. It is particularly applicable to limonitic ores or mixed limonite–saprolite feeds, as excessive silica and magnesium can impede Ni recovery [123]. The process involves selective reduction by roasting at ~850 °C to convert nickel and cobalt to metallic forms, followed by ammoniacal-ammonium carbonate leaching at 150–200 °C [36]. Fe precipitates as hydroxides during leaching, while Ni and Co dissolve and are later recovered as nickel carbonate and cobalt sulphide. The ammonia used is recycled through calcination of Ni carbonate [22].
Although operationally simpler than HPAL, the Caron process delivers relatively low recoveries (<80%) and exhibits high energy consumption due to the reduction stage. It is also characterized by high greenhouse gas emissions (Table 8) however ammonia regeneration renders it economical in reagent use. The method was largely supplanted by HPAL in commercial practice due to its lower efficiency and higher thermal cost. Recent research seeks to revisit the Caron route by employing green hydrogen as a selective reducing gas to transform Ni–Co phases into more porous, reactive forms, potentially enhancing subsequent leaching at lower temperatures. The key reactions for reduction (at ~850 °C) and ammoniacal leaching (at ~200 °C) are presented in Equations (1) and (2) [22].

Table 8.
Overview of embodied energy demand, GHG emissions and process conditions for various laterite processing routes. Modified after Norgate and Jahanshahi [16] and Stanković et al. [36].
4.4.2. High-Pressure Acid Leaching
The HPAL process is currently the most prevalent and commercially available hydrometallurgical route for processing nickel laterites, with major operations at Murrin Murrin, Ravensthorpe, Ambatovy, Goro, Ramu River, Coral Bay/Tagantio [15]. HPAL achieves high extraction efficiencies, up to ~96% Ni and Co, and is particularly well suited to limonitic ores due to their low acid-consuming minerals concentrations (e.g., Mg, Ca) and their ability to precipitate Fe as hematite under process conditions [22,124]. It is worth noting that, though saprolites are dominated by acid consuming ores, however, the amount of acid (kg) consumed/kg Ni produced is higher for goethitic ores due to their refractoriness [121]. HPAL is conducted in titanium-lined autoclaves under conditions of 30–40 wt.% solids, acid to ore ratio of 0.2–0.4, temperatures of 240–270 °C, and pressures of ~3.5–4.0 MPa, using sulphuric acid as the lixiviant for 60–120 min [15]. Leaching proceeds primarily by proton attack on Fe-oxide and hydroxide matrices (e.g., goethite), dissolving contained Ni and Co. Following leaching, the slurry is flash cooled to ~100 °C, neutralized, and subjected to counter current decantation for liquid–solid separation [125,126]. Ni/Co are subsequently recovered as sulphides, carbonates, or hydroxides. At elevated temperatures, Fe precipitates as hematite, basic ferric sulphate, and jarosite-type compounds, while Al forms alunite (KAl3(SO4)2(OH)6 according to Equations (3–7) [124,126].
Although HPAL achieves high metal recoveries, it involves high CapEX and OpEX (Table 7) due to corrosive operating environments, rapid equipment wear, and scale formation on reactor surfaces. Its leaching conditions, GHG emissions and embodied energy is summarized in Table 8. Acid consumption remains substantial due to limonite’s refractoriness and unwanted dissolution of gangue minerals. Several mitigating strategies have been proposed such as introducing catalytic ions (e.g., Cu2+) or employing staged acid addition to reduce scale formation and control free acidity [2,124,127]. Titanium lining and protective coatings are also required to manage corrosion [22], contributing to high overall cost. Recent laboratory advances by Acquah et al. [128] demonstrated the use of a Milestone SynthWave system (Figure 16) to safely simulate HPAL conditions in polytetrafluoroethylene (PTFE)-lined vessels. This system allows rapid, uniform heating under controlled pressure, eliminates scale formation, and provides a reproducible environment for studying leaching kinetics. Further development and scaling to pilot-plant level could enable cost-effective, low-risk evaluation of HPAL parameters for different laterite feeds.

Figure 16.
Milestone Microwave synthesis system. Modified after [129].
Figure 16.
Milestone Microwave synthesis system. Modified after [129].

4.4.3. Enhanced Pressure Acid Leaching
The Enhanced Pressure Acid Leach (EPAL) process was developed by BHP Billiton for the Ravensthorpe Nickel Operation as an improvement upon the conventional HPAL process [14]. EPAL integrates HPAL with atmospheric leaching (AL) to treat blended limonitic and saprolitic ores [130]; detailed information provided [14]. In this hybrid configuration, limonite is processed under HPAL conditions, generating an acidic pregnant leach solution (PLS). The subsequent atmospheric leaching stage utilizes the saprolite ore, characterized by high magnesium content to neutralize the residual acidity from the HPAL liquor at 80–95 °C under atmospheric pressure. While this two-stage system reduces acid waste and enhances resource utilization, operational results at Ravensthorpe revealed slow and incomplete nickel extraction during the secondary AL step, necessitating extended residence times and careful control of the limonite to saprolite blending ratio. Despite these limitations, EPAL remains a significant advancement in hybrid laterite processing, underscoring the importance of integrated leach strategies in maximizing overall recovery from mixed ore feeds.
4.4.4. Bioleaching
The growing emphasis on sustainable and low-cost processing technologies has renewed interest in bioleaching as a potential route for nickel, copper, gold, and cobalt production [26]. This method employs microorganisms including heterotrophic fungi such as Aspergillus and Penicillium species, and chemolithotrophic bacteria such as Acidithiobacillus species to solubilize metals through biochemical production of organic acids [13,131,132]. Common leaching agents generated by these organisms include oxalic, citric, gluconic, and fumaric acids, which promote leaching of metal bearing phases [34,133,134]. Bioleaching is generally characterized by lower energy requirements and reduced GHG emissions compared to HPAL or RKEF [135]. This is due to its reliance on microbial oxidation and operates at atmospheric pressure and moderate temperatures.
Leaching proceeds by two primary mechanisms including (i) direct bioleaching, where microorganisms adhere to the ore surface and mediate localized dissolution. This has impact on downstream leach liquor purification as a result of presence of the microorganisms in the bioleach solutions, and (ii) indirect bioleaching, in which bio-produced acids are externally applied as lixiviants [136,137]. Comparative studies [138,139] showed that bioleaching of nickel from low-grade laterites was efficient over chemical organic acid leaching due to the synergistic role of microbial metabolism and acid chelation. However, bioleaching has significant limitations that affect its commercial viability. The process is typically slow, often requiring extended leaching times relative to atmospheric sulphuric acid leaching. Recovery rates can be variable (low-moderate) and only suitable for silicates, saprolites, and garnierite [26,43,140].
Its efficiency is heavily dependent on careful control of critical process parameters such as pH, temperature, pulp density, microbial consortium compatibility, substrate concentration, and the presence of inhibitory metals [22,131]. Although bioleaching is environmentally benign, energy efficient, and operationally flexible, and offers potential for more sustainable nickel and cobalt extraction, its slower kinetics, process sensitivity, and lower recoveries constrain its competitiveness [141]. Integration as a pretreatment stage or in combination with conventional methods may be the most viable pathway for industrial application. Recent progress in microbial recovery systems for non-sulphidic ores and recent developments of microbial process are reviewed comprehensively [139,142,143].
4.4.5. Heap Leaching
Heap leaching (HL) is another commercially available method that offers an economically attractive alternative to HPAL, primarily due to its significantly lower CapEX and OpEX (Table 7). In practice, heap leaching involves agglomeration and stacking of ROM or crushed ore on impermeable pads, followed by irrigation with acidic lixiviants, typically sulfuric acid at flow rates of 1–10 L h−1 m−2 for heap heights of 4–10 m [17,121,144]. The resulting pregnant leach solution (PLS) is collected and processed via downstream recovery steps such as solvent extraction and electrowinning. Heap leaching is currently employed industrially at the Glencore Murrin Murrin operation in Australia, Piauí Nickel Project (PNP) in Brazil, and Yuanjiang in China [17,18,123,144,145]. At Murrin Murrin, heap leaching operates alongside the HPAL circuit as part of an integrated laterite processing complex, supporting operational continuity during periods of HPAL plant downtime. While operations in Brazil and China are standalone heap leaching facilities. HL is particularly suited for low-grade (<1.0 wt.% Ni) laterites, especially saprolite and siliceous goethite ores [17,121,146]. Ore mineralogy influences both agglomerate robustness and metal dissolution [147].
For instance, Nosrati et al. [146] reported that goethitic ore’s agglomerates failed after only one day of column leaching (CL), whereas saprolite and siliceous goethite agglomerates remained stable for over 100 days. Subsequent studies by Quaicoe et al. [121] demonstrated that goethitic agglomerates could maintain structural integrity for 100 days of column leaching when the binder dosage was increased from 22.5 to 24 wt.% and higher residual moisture contents were maintained. Additional information relating to Ni laterite ore mineralogy to heap leaching conditions has been reported by Watling et al. [40]. Agglomeration is therefore a critical pretreatment step in heap leaching. Readett and Fox [148] identified effective agglomeration for structural integrity as the most important variable in the successful establishment of the Murrin Murrin heap leach operation. Detailed information on the Murrin Murrin operation is discussed [18].
Despite operating under ambient conditions, heap leaching is characterized by slow kinetics, with residence times commonly exceeding 100–200 days [121,144] (Table 8). Additional challenges include heap instability, reductions in bed porosity/permeability as leaching progresses, and moisture-induced collapse (15%–50% moisture). Problems exacerbated by the fine particles and high clay content typical of many lateritic ores [18,39,149,150]. Nevertheless, when compared with HPAL, heap leaching remains an energy efficient and cost-effective processing route for marginal laterite ores. However, further research is required to improve understanding of agglomeration behavior and leaching mechanisms in lateritic systems as well as the impact of variables (e.g., binder dosage, moisture content) on the performance and optimization of column/heap leaching operations.
4.4.6. Atmospheric Leaching
Atmospheric leaching (AL), also referred to as tank acid leaching, is performed in agitated reactors at atmospheric pressure (1 atm) and moderate temperatures (50–100 °C) [11,14,119]. Increasing global interest in AL stems from its ability to treat both saprolite and limonitic ores, combined with lower capital intensity compared to HPAL. In this process, ground ore is leached in acid media (e.g., H2SO4) under continuous stirring for periods up to 48 h depending on the operation. While technically straightforward, AL is non-selective, dissolving significant quantities of gangue species (e.g., Fe, Al, Mg, Mn, Cr), which leads to high acid consumption and complex liquor purification [11,119]. Post-leach solutions often require neutralization or precipitation using hydroxides and sulphides [151]. Rapid dissolution of Mg from saprolite and nontronite phases yields efficient Ni–Co extraction, but Fe-bearing limonite necessitate aggressive conditions for complete dissolution [119,152]. Integration of pretreatment steps such as mechanical activation or thermal calcination prior to AL has shown successful improvement in Ni–Co recovery from limonitic ores. This provides an energy efficient alternative to autoclave-based HPAL [8,9,20,114]. Process costs (Table 7) and summary of operational conditions, embodied energy and GHG emissions are shown Table 8.
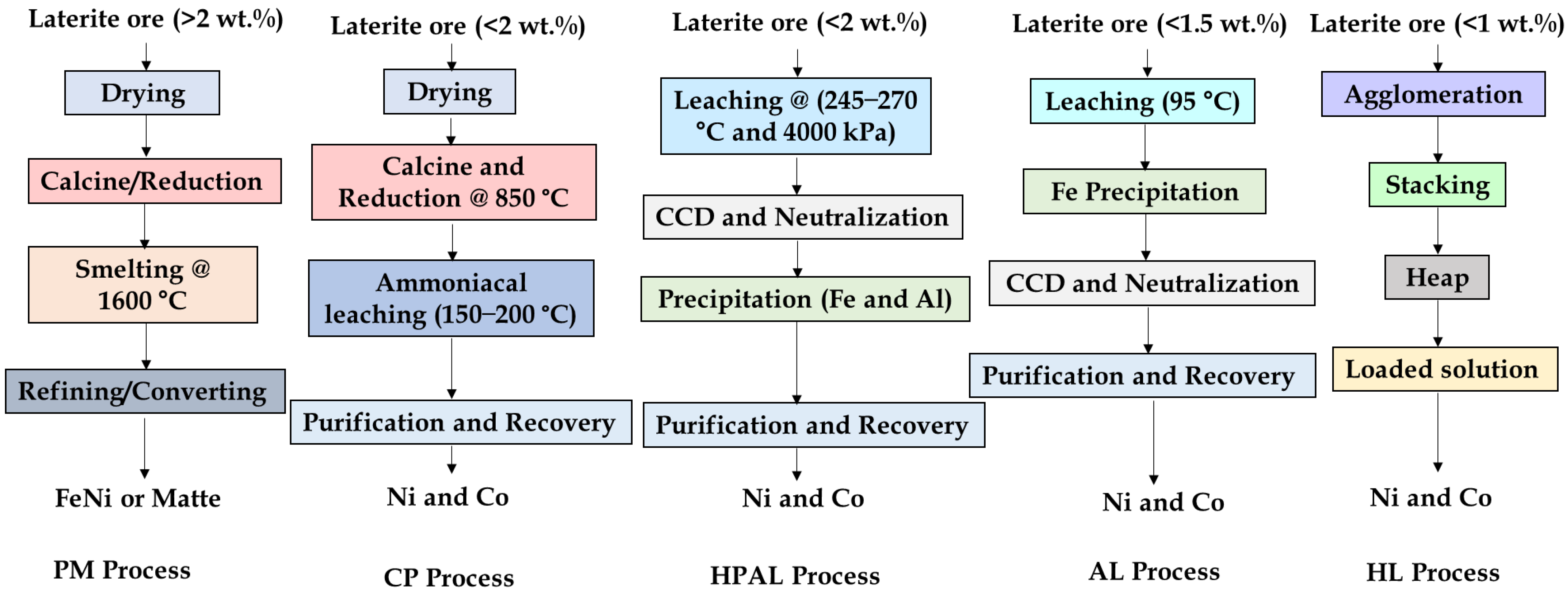
Ongoing research also explores the use of alternative lixiviants, including phosphoric, citric, and nitric acids, as well as synergetic acid systems. For example, dual leaching with sulfuric and citric acids enhances selectivity and reduces reagent consumption [128,153,154], while nitric acid leaching offers rapid dissolution kinetics, and acts as both reagent and oxidant, achieving up to ~95% acid recyclability [53]. Ma et al. [155] reported ~85% Ni/Co recoveries using nitric acid pressure leaching (NAPL), surpassing HPAL performance under similar conditions. Emerging process intensification methods including microwave and ultrasonic-assisted leaching show promising improvements in dissolution kinetics. For instance, 89% Ni and ~62% Co was extracted via microwave-assisted leaching [156]. Nevertheless, the high cost of advanced reagents and specialized equipment remains a barrier to commercialization [53,143]. Figure 17 summarizes the process flowsheet of nickel laterite extraction techniques, while Table 9 presents summary of selected leaching studies.
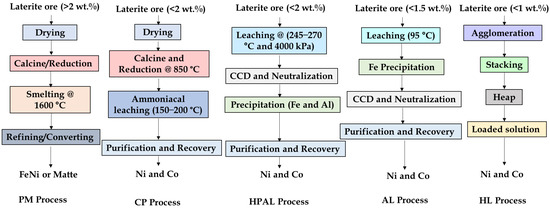
Figure 17.
Typical flowsheet for pyrometallurgical (PM), caron (CP), high-pressure acid leaching (HPAL), atmospheric leaching (AL), and heap leaching (HL) processes.

Table 9.
Summary of various laterite leaching studies.
Table 9.
Summary of various laterite leaching studies.
| Ore Type | Location | Process | Operating Conditions | Recovery (%) | Reference |
|---|---|---|---|---|---|
| Saprolite | Western Australia | AL | 4 h, 95 °C, −2 mm, 40 wt.% solid, 600 rpm, acid consumption: 640 kg/t | Ni: 80 Co: 78 | [119] |
| Goethite | Western Australia | AL | 4 h, 95 °C, −2 mm, 40 wt.% solid, 600 rpm, acid consumption: 571 kg/t | Ni: 54 Co: 35 | [157] |
| Siliceous goethite | Western Australia | AL | 4 h, 95 °C, −2 mm, 40 wt.% solid, 600 rpm, acid consumption: 607 kg/t | Ni: 74 Co: 77 | [152] |
| 50 Ni laterite ores with different mineralogies | Western Australia | CL | 240 g of H2SO4 used as binding medium, 120 days, 200 g/L H2SO4 was irrigated at10 L/h/m2, acid consumption: 110–815 kg/t | Ni: 88–96 Co: 80–93 | [40] |
| Saprolite, and Goethite | Western Australia | CL | 30% w/w H2SO4 used as binding medium, −2 mm, 100 days, 200 g/L H2SO4 irrigated at 8.5 L/h/m2, acid consumptions: 524 (G), and 641 kg/t (SAP) | SAP Ni/Co: 90/73 G Ni/Co: 63/56 | [121] |
| Limonite | Indonesia | AL | 75 °C, 1M H2SO4 | Ni: 79 | [19] |
| Limonite | Çaldağ, Türkiye | HPAL | 325 kg/ton acid load, 250 °C, 1 h, 1 mm | Ni: 94 Co: 94 | [158] |
| Limonite | Philippine | HPAL | −180 μm, 250 °C, 1 h, 250kg/t acid addition, 25 wt.% solid, 300 rpm | Ni: 97 Co: 96 | [159] |
| Limonite | Iran | AL | 2 h, 90 °C, 5 M H2SO4, 370 rpm | Ni: 66 Co: 63 | [160] |
| Chromite overburden | India | AL | 0.15 M oxalic acid, 3 h, 80 °C, 20 wt.% solid, 53 μm | Ni: 64 Co: 44 | [161] |
| Limonite | Papua New Guinea | AL | 600 W, 90 °C, 25 vol.% H2SO4, and 1.5 h | Ni: 91 | [162] |
| Hematite Garnierite | Greece | AL | 0.5 M citric acid, 5 wt.% solid, 30 °C | H: 52 G: 57 | [137] |
| Saprolite | Brazil | Bioleaching | 15% (w/v) glucose, Burkholderia sp. 42 days | Ni: 40 | [131] |
| Limonite | Brazil | Bioleaching and chemical leach | Acidithiobacillus thiooxidans Acidithiobacillus caldus, 7 days | Ni: 17%–56% Co: 68%–88% | [43] |
5. Ion Exchange Application
The application of ion exchange (IX) technology in hydrometallurgy dates back to the 1950s, when it was first employed in the recovery of uranium in the Soviet Union [163]. Since then, IX systems have been adopted across a wide range of metal recovery operations due to their operational simplicity, chemical selectivity, and minimal safety hazards associated with low toxicity media [164]. Among IX-based approaches, the resin in pulp (RIP) process has gained attention as a viable method for dissolved metal adsorption directly from slurries during solid–liquid separation. RIP presents an attractive alternative to conventional liquid–solid clarification steps, which are often costly and prone to product losses. By enabling in situ adsorption of nickel and cobalt from leach pulps, the RIP method eliminates the need for filtration and minimizes valuable metal loss in entrained residues.
Extensive research has been undertaken to evaluate the selectivity and adsorption performance of various resin types for Ni and Co recovery in the presence of impurities (e.g., Fe, Mg, Al) [163,165,166,167,168,169,170]. Chelating resins functionalized with iminodiacetic acid (IDA) or bis-picolylamine (BPA) groups have demonstrated particularly high adsorption capacity for nickel and cobalt in leach slurries, as summarized in Table 10. Despite notable advantages over solvent extraction, membrane, and activated carbon systems, conventional RIP operations are constrained by several drawbacks, including long adsorption times, the need for substantial mechanical agitation to maintain slurry–resin contact, high power demand, and significant water consumption [171].

Table 10.
Summary of various laterites studies using chelating resins as an adsorbent.
Table 10.
Summary of various laterites studies using chelating resins as an adsorbent.
| Resin | Functional Group | Process Options | Recovery (%) | Reference |
|---|---|---|---|---|
| Lewatit TP 207 Lewatit TP 220 | Iminodiacetic acid Bispicolylamine | Conversion of ferric iron to ferrous iron followed by resin in pulp | Ni/Co: 17.45/7.22 Ni/Co: 72.98/74.51 | [172] |
| Purolite S950 | Aminophosphonic acid | Resin in pulp (synthetic bioleach solutions) | Ni: 90 Co: 82–98 | [173,174] |
| Not specified | Iminodiacetic acid | Resin in moist mix (simultaneous leaching and adsorption) | Ni/Co: 50 | [175] |
| Lewatit MonoPlus TP 220 | Bispicolylamine | Resin in pulp scavenging | Ni: 90–95 Co: 75–85 | [165] |
| Amberlite IRC 748 | Iminodiacetic acid | Precipitation of Fe followed by Resin in pulp (HPAL pulp) | Ni: 62.3 Five stages of RIP test Ni/Co: 99.8/9.7 | [166] |
| IRC 748 | Iminodiacetic acid | Resin in pulp (HPAL pulp) | Ni: >90 Co: 80 | [163] |
Recent Novel Atmospheric Resin in Moist Mix Method (RIMM)
An alternative known as the resin in moist mix (RIMM) process has been introduced as a sustainable and simplified approach to address these challenges [171,176]. RIMM integrates atmospheric tumbling leaching with simultaneous IX adsorption, enabling concurrent leaching and resin-mediated recovery of target metals, thereby eliminating the need for separate leaching and adsorption units. The process conceptually resembles heap leach systems but differs by operating with fine grained ore and resin mixtures at high solid loadings (~70 wt.%). In the RIMM process, coarse IX resins are mixed directly with moist ore and lixiviant, allowing selective capture of dissolved metal ions during short tumbling periods under ambient pressure and temperature. Following equilibration, the loaded resins are recovered, eluted, and processed by standard downstream techniques such as electrowinning or crystallization, similar to carbon in leach circuits used in gold extraction [177,178,179]. RIMM has demonstrated successful application at both bench and pilot scale for the recovery of copper from low-grade ores (e.g., 0.6% Cu), achieving recoveries of ~96% [176,180]. A typical RIMM process is shown in Figure 18, and summary of RIMM studies on different copper ores is provided in Table 11.

Table 11.
Summary of resin in moist mix studies.
Table 11.
Summary of resin in moist mix studies.
| Ore Type | Grade (%) | Recovery (%) | Reference |
|---|---|---|---|
| South Australia Cu ore (<300 µm) | Cu/Co: 10.32/0.01 | Cu/Co: 55.1/74.7 | [181] |
| South Australia old Cu tailings (<300 µm) | Cu: 0.19–0.66 | Cu/Co: | [181] |
| Co: 0.1–0.3 | 86.9/91.7 | ||
| South Australia Cu ore Fine fraction (FF) sieved (<425 µm) Coarse fraction (CF) milled (<425 µm) ore milled (425 µm) | Cu: FF: 0.22 CF. 1.1 Ore: 0.81 | FF: 94.5 CF: 98.3 Ore: 96.7 | [171] |
| South Australia Cu ore (<250 µm) | Cu: 1.47 | 98.3 | [176] |
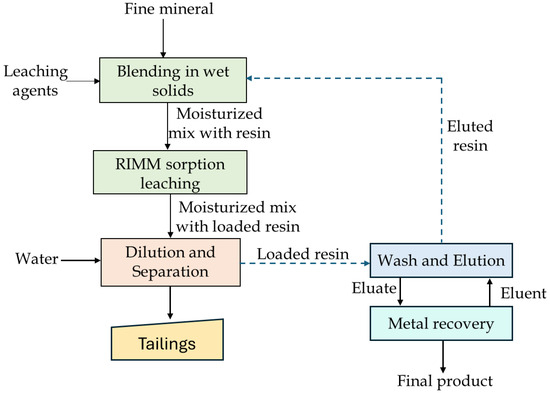
Figure 18.
Basic RIMM flowsheet. Modified after Spiridonov et al. [171].
Figure 18.
Basic RIMM flowsheet. Modified after Spiridonov et al. [171].
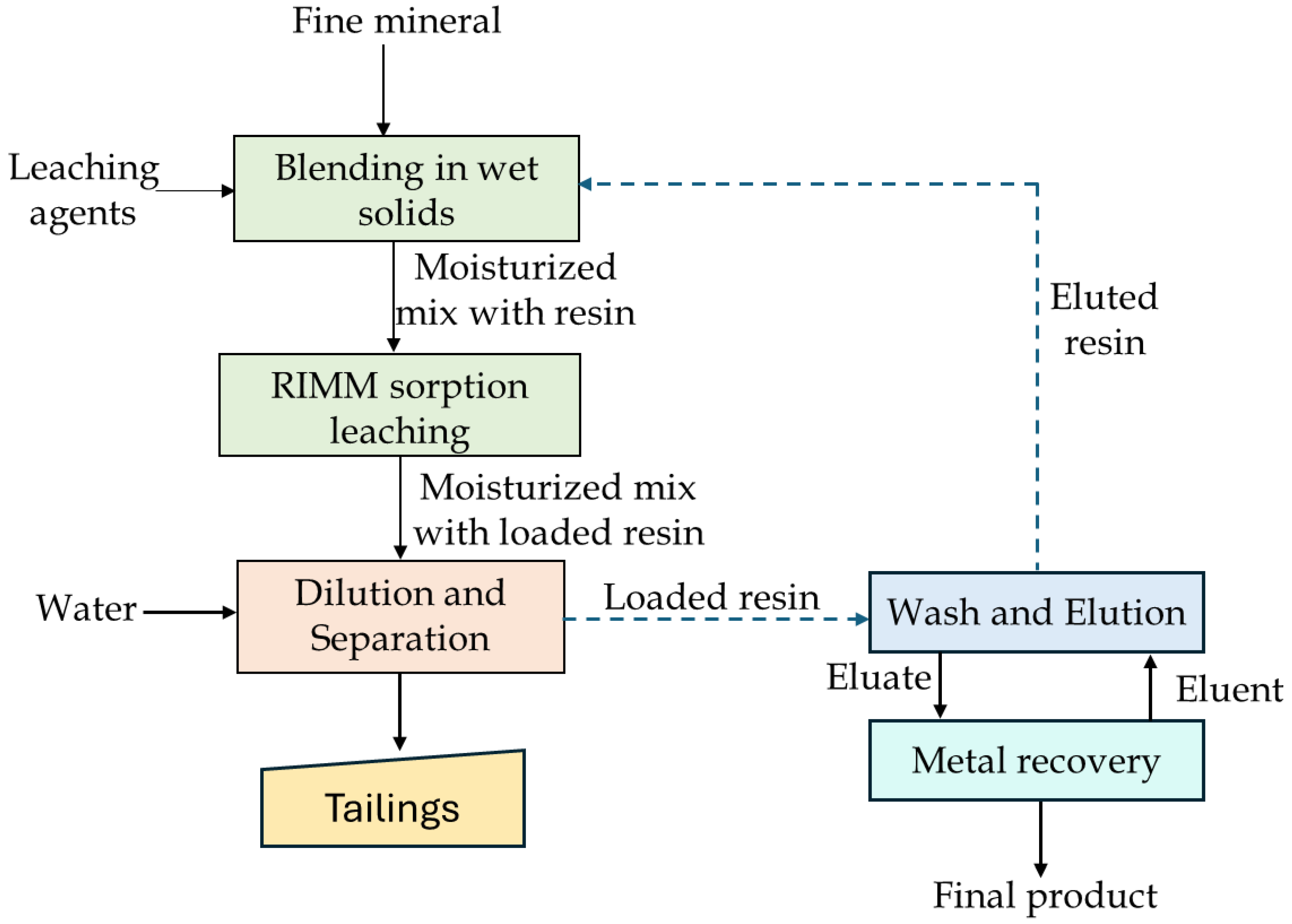
Despite its promising achievements for Cu ores, the application of RIMM to Ni–Co laterite systems remain largely unexplored. Spiridonov et al. [175] conducted preliminary tests on saprolite laterite, attaining ~50% Ni/Co recovery after 72 h at ambient conditions (25 °C, 1 atm). Under similar residence times, comparative heap leach studies reported only ~20% Ni/Co recovery [146,147,182]. However, after 10 days, RIMM extraction rates plateaued, while that of HL showed continued gradual recovery over time. Experimental variables such as resin dosage, acid addition, and water addition (solid loading) were not disclosed, underscoring gaps that require detailed investigation. Future research focusing on elucidating the interactions between ore mineralogy and process parameters, (e.g., particle size, resin-to-solid ratio, lixiviant type and concentration, contact time) is paramount. Such understanding is essential to optimize sorption kinetics and identify the ore mineralogical variation (e.g., saprolite vs. limonite) most amenable to RIMM. Additionally, its integration into an existing flowsheet could offer superior efficiency. With systematic refinement, this method could unlock considerable nickel potential from complex laterite resources, substantially reducing processing cost and environmental footprint relative to multi-stage hydrometallurgical circuits.
6. Conclusions and Future Prospects
This review summarized laboratory and industrial scale developments in the extraction of nickel and cobalt from lateritic ores, highlighting progress in beneficiation, pretreatment, and hydrometallurgical processing. The inherent complexity of laterite mineralogy marked by the fine dissemination of Ni–Co phases within iron and silicate matrices continues to present formidable challenges for efficient extraction. Fine grinding remains essential to liberate Ni-bearing phases. However, conventional milling techniques often fail to achieve sufficient mineral liberation without excessive energy use. The use of stirred media mills has proven particularly effective due to their high energy density and ability to induce structural deformation and surface activation. Nevertheless, fine particle recovery remains problematic in conventional flotation and magnetic circuits. Adoption of modern reactor–separator and reflux flotation cells offers a promising solution to enhance fine particle recovery efficiency and overall beneficiation performance.
By far, both bench and pilot-scale investigations have shown that saprolite and siliceous goethite ores respond more favorably to beneficiation than limonitic types. Selective comminution and classification techniques have achieved encouraging upgrades and should be pursued further. Product fractions enriched in Ni may be efficiently processed via atmospheric leaching, while the Co-rich coarse fractions are better suited to heap leaching or RIMM treatment. The optimization of dispersant concentration is also critical, as nickel laterites are naturally agglomerated, dispersants not only assist in the liberation of fines from coarser particles but can alter the surface charge (isoelectric point) of both ore and gangue minerals, enhancing separation efficiency.
Pretreatment prior to leaching remains a decisive factor in process success. Mechanical activation using stirred media milling introduces substantial structural disorder, lattice defects, and amorphization, which significantly increase leach reactivity and surface accessibility of Ni and Co phases. However, identifying the optimum activation duration is essential to prevent over milling, aggregation, and excess energy consumption. Thermal pretreatment (calcination) dehydroxylates goethite to poorly ordered hematite and has been shown to accelerate acid leaching kinetics and markedly shorten residence times for both heap and atmospheric leaching operations. Albeit calcination temperature and time should be carefully controlled to avoid phase stability.
Extraction efficiency in laterite processing is highly dependent on ore mineralogy and its interaction with the chosen leaching chemistry. In general, Fe-oxide dominated limonitic ores are best suited to strongly acidic routes such as HPAL, and under optimized conditions such as AL, as they contain relatively low acid consuming minerals. In contrast, Mg-rich saprolites and clay-rich feeds tend to consume significant acid under AL or HPAL conditions and can be challenging to treat economically unless blended or partially neutralized. For such silica and Mg-rich ores, hybrid approaches such as EPAL or Caron type pyro-hydro processing or carefully controlled AL with pretreatment may offer more favorable trade-offs between recovery, reagent use, and operating cost. Process route selection therefore strongly influences acid consumption, recovery rates, and overall carbon footprint.
The transition toward energy-efficient, low-emission metallurgical systems will increasingly depend on innovative pretreatment and preconcentration strategies capable of unlocking vast nickel resources presently deemed uneconomical. Among emerging technologies, the Resin in Moist Mix (RIMM) process warrants further investigation, as preliminary studies suggest potential for integrated leaching and selective recovery under ambient conditions through tailored resin chemistry and optimized process parameters. Systematic evaluation across diverse laterite mineralogies and comprehensive techno-economic assessment remain essential to establish its viability.
Overall, the integration of advanced beneficiation, pretreatment, and hybrid leaching approaches presents a path forward for the sustainable extraction of nickel and cobalt from refractory laterites. Through the combined application of mechanical activation, selective comminution, and ambient-pressure extraction technologies, low-grade lateritic deposits may be transformed into commercially viable, sustainable and environmentally responsible sources of critical battery metals.
Author Contributions
Conceptualization, G.A. and R.A.; investigation, G.A.; writing—original draft preparation, G.A.; writing—review and editing, R.A., J.A.-M. and W.S.; visualization, G.A. and R.A.; supervision, R.A., P.S., G.A.-W., J.A.-M. and W.S.; project administration, R.A.; funding acquisition, R.A. and W.S. All authors have read and agreed to the published version of the manuscript.
Funding
The authors acknowledge the funding support from Australia–India Strategic Research Fund (AISRF) for Advanced Recovery of the Battery materials and REE from Ores and Wastes (grant number: AIRXIV000095), and Australian Research Council for the ARC Centre of Excellence for Enabling Eco-Efficient Beneficiation of Minerals (grant number: CE200100009).
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the article.
Acknowledgments
The authors also acknowledge the facilities and technical assistance of staff at the Future Industries Institute, Adelaide University.
Conflicts of Interest
Author Pavel Spiridonov was employed by the company InnovEco Australia Pty Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AL | Atmospheric leaching |
| HL | Heap leaching |
| HPAL | High-pressure acid leaching |
| EPAL | Enhanced pressure acid leaching |
| RIMM | Resin in moist mix |
| SAP | Saprolite |
| SG | Siliceous goethite |
| G | Goethitic |
| RKEF | Rotary kiln electric furnace |
| ROM | Run of mine |
| PTFE | Polytetrafluoroethylene |
| CapEX | Capital expenditure |
| OpEX | Operating expenditure |
| IX | Ion exchange |
References
- IEA. The Role of Critical Minerals in Clean Energy Transitions; IEA: Paris, France, 2021. [Google Scholar]
- Dilshara, P.; Abeysinghe, B.; Premasiri, R.; Dushyantha, N.; Ratnayake, N.; Senarath, S.; Ratnayake, A.S.; Batapola, N. The role of nickel (Ni) as a critical metal in clean energy transition: Applications, global distribution and occurrences, production-demand and phytomining. J. Asian Earth Sci. 2024, 259, 105912. [Google Scholar] [CrossRef]
- Setiani, H. Nickel export ban policy in Indonesia-a path to sustainable economic development? Ecoprofit 2024, 1, 120–130. [Google Scholar] [CrossRef]
- Dalvi, A.D.; Bacon, W.G.; Osborne, R.C. The past and the future of nickel laterites. In PDAC 2004 International Convention, Trade Show & Investors Exchange; The Prospectors and Developers Association of Canada Toronto: Toronto, ON, Canada, 2004; pp. 1–27. [Google Scholar]
- Elias, M. Nickel laterite deposits–geological overview, resources and exploitation. Giant Ore Depos. Charact. Genes. Explor. CODES Spec. Publ. 2002, 4, 205–220. [Google Scholar]
- Golightly, J.P.; Deposits, N.L.; Skinner, B.J. (Eds.) Society of Economic Geologists; Economic Geology Publishing Company: Lancaster, PA, USA, 1981. [Google Scholar] [CrossRef]
- Zappala, L.; McDonald, R.; Pownceby, M.I. Nickel Laterite Beneficiation and Potential for Upgrading Using High Temperature Methods: A Review. Miner. Process. Extr. Metall. Rev. 2024, 45, 767–789. [Google Scholar] [CrossRef]
- Basturkcu, H.; Achimovičová, M.; Kaňuchová, M.; Acarkan, N. Mechanochemical pre-treatment of lateritic nickel ore with sulfur followed by atmospheric leaching. Hydrometallurgy 2018, 181, 43–52. [Google Scholar] [CrossRef]
- Borda, J.; Torres, R. Nickel recovery from low-grade laterites: Study of thermal pre-treatments to improve the efficiency of the hydrometallurgical process. J. Sustain. Min. 2023, 22, 219–226. [Google Scholar] [CrossRef]
- Tian, H.-Y.; Guo, Z.-Q.; Zhan, R.-N.; Pan, J.; Zhu, D.-Q.; Yang, C.-C.; Pan, L.-T.; Huang, X.-Z. Upgrade of nickel and iron from low-grade nickel laterite by improving direct reduction-magnetic separation process. J. Iron Steel Res. Int. 2022, 29, 1164–1175. [Google Scholar] [CrossRef]
- Acquah, G.; Abaka-Wood, G.; Skinner, W.; Addai-Mensah, J.; Asamoah, R. Effect of mechanical activation on nickel and cobalt extraction in refractory nickel laterite ore. Powder Technol. 2026, 468, 121655. [Google Scholar] [CrossRef]
- Quast, K.; Connor, J.N.; Skinner, W.; Robinson, D.J.; Addai-Mensah, J. Preconcentration strategies in the processing of nickel laterite ores Part 1: Literature review. Miner. Eng. 2015, 79, 261–268. [Google Scholar] [CrossRef]
- Senanayake, G.; Childs, J.; Akerstrom, B.D.; Pugaev, D. Reductive acid leaching of laterite and metal oxides—A review with new data for Fe(Ni,Co)OOH and a limonitic ore. Hydrometallurgy 2011, 110, 13–32. [Google Scholar] [CrossRef]
- McDonald, R.; Whittington, B. Atmospheric acid leaching of nickel laterites review: Part I. Sulphuric acid technologies. Hydrometallurgy 2008, 91, 35–55. [Google Scholar] [CrossRef]
- Whittington, B.I.; Muir, D. Pressure Acid Leaching of Nickel Laterites: A Review. Miner. Process. Extr. Metullargy Rev. 2000, 21, 527–599. [Google Scholar] [CrossRef]
- Norgate, T.; Jahanshahi, S. Assessing the energy and greenhouse gas footprints of nickel laterite processing. Miner. Eng. 2011, 24, 698–707. [Google Scholar] [CrossRef]
- Oxley, A.; Smith, M.E.; Caceres, O. Why heap leach nickel laterites? Miner. Eng. 2015, 88, 53–60. [Google Scholar] [CrossRef]
- Quast, K.; Addai-Mensah, J.; Skinner, W. Preconcentration strategies in the processing of nickel laterite ores Part 5: Effect of mineralogy. Miner. Eng. 2017, 110, 31–39. [Google Scholar] [CrossRef]
- Lasut, D.B.E.; Kurniawati, R.; Dahani, W.; Subandrio. The effect of concentration and temperature on nickel extraction from laterite nickel ore using atmospheric pressure acid leaching method. In Proceedings of the Symposium on Advance of Sustainable Engineering 2021 (SIMASE 2021): Post COVID-19 Pandemic: Challenges and Opportunities in Environment, Science, and Engineering Research, 1st ed.; American Institute of Physics: Melville, NY, USA, 2023; Volume 2646. [Google Scholar] [CrossRef]
- Parlak, T.T.; Yildiz, K. Investigation of the effect of mechanical activation on nickel and cobalt extraction from laterite at atmospheric pressure by kinetics and leach residue analysis. Can. Metall. Q. 2025, 64, 680–697. [Google Scholar] [CrossRef]
- U.S. Geological Survey. Mineral Commodity Summaries 2025. In Mineral Commodity Summaries; U.S. Geological Survey: Reston, VA, USA, 2025. Available online: https://pubs.usgs.gov/publication/mcs2025 (accessed on 2 February 2025).
- Caetano, G.C.; Ostroski, I.C.; de Barros, M.A.S.D. Lateritic nickel and cobalt recovery routes: Strategic technologies. Miner. Process. Extr. Metall. Rev. 2025, 46, 400–414. [Google Scholar] [CrossRef]
- Mudd, G.M.; Jowitt, S.M. A detailed assessment of global nickel resource trends and endowments. Econ. Geol. 2014, 109, 1813–1841. [Google Scholar] [CrossRef]
- Dehaine, Q.; Tijsseling, L.T.; Glass, H.J.; Törmänen, T.; Butcher, A.R. Geometallurgy of cobalt ores: A review. Miner. Eng. 2021, 160, 106656. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, Z.; Cao, Y.; Han, G.; Peng, W.; Zhu, X.; Zhang, T.-A.; Dou, Z. Overview of cobalt resources and comprehensive analysis of cobalt recovery from zinc plant purification residue-a review. Hydrometallurgy 2020, 193, 105327. [Google Scholar] [CrossRef]
- Saim, A.K.; Darteh, F.K. A Comprehensive Review on Cobalt Bioleaching from Primary and Tailings Sources. Miner. Process. Extr. Metall. Rev. 2023, 45, 426–452. [Google Scholar] [CrossRef]
- Statista. Global Nickel Mining Industry, 2024. Available online: https://www.statista.com/study/15792/nickel-statista-dossier/ (accessed on 8 January 2026).
- Australia Government, Department of Industry, Science and Resources. Resources and Energy Quarterly. 2024. Available online: https://www.industry.gov.au/sites/default/files/2025-02/Resources-and-energy-quarterly-December-2024.pdf?utm (accessed on 28 January 2026).
- Argus, M. Stainless Steel Prices Stabilise But Downside Risks Remain Amid Weak Demand. 2024. Available online: https://www.argusmedia.com/en/news-and-insights/latest-market-news/2620260-stainless-steel-prices-stabilise-but-downside-remains?utm (accessed on 28 January 2026).
- Slack, J.F.; Kimball, B.E.; Shedd, K.B. Cobalt. In Critical Mineral Resources of the United States—Economic and Environmental Geology and Prospects for Future Supply; Schulz, K.J., DeYoung, J.H., Jr., Seal, R.R., II, Bradley, D.C., Eds.; U.S. Geological Survey Professional Paper 1802; U.S. Geological Survey: Reston, VA, USA, 2017; pp. F1–F4. [Google Scholar] [CrossRef]
- Ziwa, G.; Crane, R.; Hudson-Edwards, K.A. Geochemistry, Mineralogy and Microbiology of Cobalt in Mining-Affected Environments. Minerals 2021, 11, 22. [Google Scholar] [CrossRef]
- Petavratzi, E.; Gunn, G.; Kresse, C. BGS Commodity Review: Cobalt. 2019. Available online: https://www.researchgate.net/publication/337604314_BGS_COMMODITY_REVIEW_Cobalt (accessed on 15 January 2026).
- Eckstrand, O.R.; Hulbert, L.J. Magmatic nickel-copper-platinum group element deposits, Mineral deposits of canada: A synthesis of major deposit types, district metallogeny, the evolution of geological provinces, and exploration methods: Geological association of Canada, mineral deposits division. Spec. Publ. 2007, 5, 205–222. [Google Scholar]
- Behera, S.K.; Mulaba-Bafubiandi, A.F. Advances in microbial leaching processes for nickel extraction from lateritic minerals—A review. Korean J. Chem. Eng. 2015, 32, 1447–1454. [Google Scholar] [CrossRef]
- Elias, M. Overview of the Australian Nickel-Cobalt Industry. AUSIMM Monogr. 2013, 2, 1723–1727. [Google Scholar]
- Stanković, S.; Stopić, S.; Sokić, M.; Marković, B.; Friedrich, B. Review of the past, present, and future of the hydrometallurgical production of nickel and cobalt from lateritic ores. Metall. Mater. Eng. 2020, 26, 199–208. [Google Scholar] [CrossRef]
- Hoatson, D.M.; Jaireth, S.; Jaques, A.L. Nickel sulfide deposits in Australia: Characteristics, resources, and potential. Ore Geol. Rev. 2006, 29, 177–241. [Google Scholar] [CrossRef]
- Butt, C.R.; Cluzel, D. Nickel laterite ore deposits: Weathered serpentinites. Elements 2013, 9, 123–128. [Google Scholar] [CrossRef]
- Kyle, J. Nickel laterite processing technologies–where to next? In Proceedings of the ALTA 2010 Nickel, Cobalt, Copper, Uranium and REE Conference, Perth, Australia, 24–26 May 2010. [Google Scholar]
- Watling, H.; Elliot, A.; Fletcher, H.; Robinson, D.; Sully, D. Ore mineralogy of nickel laterites: Controls on processing characteristics under simulated heap-leach conditions. Aust. J. Earth Sci. 2011, 58, 725–744. [Google Scholar] [CrossRef]
- Swamy, Y.V.; Kar, B.B.; Mohanty, J.K. Physico-chemical characterization and sulphatization roasting of low-grade nickeliferous laterites. Hydrometallurgy 2003, 69, 89–98. [Google Scholar] [CrossRef]
- Mweene, L.; Gomez-Flores, A.; Jeong, H.-E.; Ilyas, S.; Kim, H. Challenges and future in Ni laterite ore enrichment: A critical review. Miner. Process. Extr. Metall. Rev. 2024, 45, 539–563. [Google Scholar] [CrossRef]
- Stanković, S.; Martin, M.; Goldmann, S.; Gäbler, H.-E.; Ufer, K.; Haubrich, F.; Moutinho, V.F.; Giese, E.C.; Neumann, R.; Stropper, J.L.; et al. Effect of mineralogy on Co and Ni extraction from Brazilian limonitic laterites via bioleaching and chemical leaching. Miner. Eng. 2022, 184, 107604. [Google Scholar] [CrossRef]
- Whittle, A.W.G. Mineragraphy. In Applied Geology; Encyclopedia of Earth Sciences Series; Springer: Boston, MA, USA, 1984. [Google Scholar]
- Frank, K.C.; Michael, S.M.; Venkoba, R.; Timothy, G.R.; William, G.D. Extractive Metallurgy of Nickel, Cobalt and Platinum-Group Metals; Elsevier Ltd.: Amsterdam, The Netherlands, 2011; pp. 1–18. [Google Scholar]
- Quast, K.; Otsuki, A.; Fornasiero, D.; Robinson, D.J.; Addai-Mensah, J. Preconcentration strategies in the processing of nickel laterite ores part 3: Flotation testing. Miner. Eng. 2015, 79, 279–286. [Google Scholar] [CrossRef]
- Tong, L.; Klein, B.; Zanin, M.; Quast, K.; Skinner, W.; Addai-Mensah, J.; Robinson, D.J. Stirred milling of nickel laterites for selective comminution. In Proceedings of the SAG Conference, Vancouver, BC, Canada, 24–25 September 2015; pp. 1–20. [Google Scholar]
- Swierczek, Z.; Quast, K.; Addai-Mensah, J.; Connor, J.N.; Robinson, D.J.; Li, J. Challenges in the mineralogical characterisation of low-grade nickel laterites. In Proceedings of the 26th International Mineral Processing Congress, (IMPC), New Delhi, India, 24–28 September 2012; pp. 5352–5364. [Google Scholar]
- Ashcroft, G. Nickel Laterites: The World’s Largest Source of Nickel. 2023. Available online: https://www.geologyforinvestors.com/nickel-laterites/ (accessed on 1 February 2026).
- Elias, M. Lateritic nickel mineralization of the Yilgarn craton. In Nickel Deposits of the Yilgarn Craton: Geology, Geochemistry, and Geophysics Applied to Exploration; Society of Economic Geologists: Littleton, CO, USA, 2006. [Google Scholar]
- Zhang, Y.; Qie, J.; Wang, X.F.; Cui, K.; Fu, T.; Wang, J.; Qi, Y. Mineralogical Characteristics of the Nickel Laterite, Southeast Ophiolite Belt, Sulawesi Island, Indonesia. Min. Metall. Explor. 2020, 37, 79–91. [Google Scholar] [CrossRef]
- Brand, N.; Butt, C.; Hellsten, K. Structural and Lithological Controls in the Formation of the Cawse Nickel Laterite Deposits, Western Australia¿ Implications for Supergene Ore Formation and Exploration in Deeply Weathered Terrains. In Structural and Lithological Controls in the Formation of the Cawse Nickel Laterite Deposits, Western Australia¿ Implications for Supergene Ore Formation and Exploration in Deeply Weathered Terrains; Australasian Institute of Mining and Metallurgy: Carlton, Australia, 1996; pp. 167–171. [Google Scholar]
- Meshram, P.; Abhilash; Pandey, B.D. Advanced Review on Extraction of Nickel from Primary and Secondary Sources. Miner. Process. Extr. Metall. Rev. 2019, 40, 157–193. [Google Scholar] [CrossRef]
- Rao, G.; Sastry, S.; Schneider, F.; Hoberg, H. Amphoteric collectors to enrich nickel from lean lateritic chromite overburden. Miner. Process. Extr. Metall. Rev. 1995, 15, 48. [Google Scholar] [CrossRef]
- Acharya, S.; Jena, M.; Mohanty, J. Aqueous alkaline processing of chromite mine overburden. In Proceedings of the XI International Seminar on Mineral Processing Technology (MPT-2010); NML: Jamshedpur, India, 2010; Volume 2, pp. 874–883. [Google Scholar]
- Stainless Steel. An Overview of World Nickel Resources. 2022. Available online: https://stainless-steel-world.net/an-overview-of-world-nickel-resources/ (accessed on 28 February 2026).
- Teh, M.; Pheeney, J. Australian Resource Reviews Nickel 2020, Geoscience Australia. Available online: https://d28rz98at9flks.cloudfront.net/145883/145883_00_0.pdf (accessed on 15 January 2026).
- Mudd, G.M. Global trends and environmental issues in nickel mining: Sulfides versus laterites. Ore Geol. Rev. 2010, 38, 9–26. [Google Scholar] [CrossRef]
- Marsh, E.E.; Anderson, E.D. Ni-Co Laterite Deposits; U.S. Geological Survey Open-File Report 2011–1259; U.S. Geological Survey: Reston, VA, USA, 2011; 9p.
- Standing, J.G. Structural Geology in Unfamiliar Terrane; Controls on Laterite-Hosted Nickel Mineralisation at Ravensthorpe, WA. In Structural Geology and Resources 2012-Extended Abstracts Volume; Vearncombe, J., Ed.; Australian Institute of Geoscientists Bulletin No. 56; Australian Institute of Geoscientists: Sydney, Australia, 2012; pp. 156–159. [Google Scholar]
- Canterford, J.H. Treatment of Nickeliferous Laterites. Miner Sci Eng. 1975, 7, 3–17. [Google Scholar]
- Quast, K.; Connor, J.N.; Skinner, W.; Robinson, D.J.; Li, J.; Addai-Mensah, J. Preconcentration strategies in the processing of nickel laterite ores part 2: Laboratory experiments. Miner. Eng. 2015, 79, 269–278. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Filippov, L.; Fornasiero, D. Pre-concentration of nickel in laterite ores using physical separation methods. Miner. Eng. 2019, 141, 105892. [Google Scholar] [CrossRef]
- Papini, R.M.; Brandão, P.R.G.; Peres, A.E.C. Cationic flotation of iron ores: Amine characterization and performance. Min. Metall. Explor. 2001, 18, 5–9. [Google Scholar] [CrossRef]
- Vieira, A.M.; Peres, A.E.C. The effect of amine type, pH, and size range in the flotation of quartz. Miner. Eng. 2007, 20, 1008–1013. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Filippov, L. Challenges in processing nickel laterite ores by flotation. Int. J. Miner. Process. 2016, 151, 59–67. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Fornasiero, D.; Filippov, L. Upgrading nickel in laterite ores by flotation. Miner. Eng. 2018, 121, 100–106. [Google Scholar] [CrossRef]
- Lynch, A.J.; Johnson, N.; Manlapig, E.; Thorne, C. Mineral and Coal Flotation Circuits: Their Simulation and Control; Elsevier: Amsterdam, The Netherlands, 1981. [Google Scholar]
- Cole, M.; Dickinson, J.; Galvin, K. Recovery and cleaning of fine hydrophobic particles using the Reflux™ Flotation Cell. Sep. Purif. Technol. 2020, 240, 116641. [Google Scholar] [CrossRef]
- Tong, L.; Klein, B.; Zanin, M.; Quast, K.; Skinner, W.; Addai-Mensah, J.; Robinson, D. Stirred milling kinetics of saprolitic and goethitic nickel laterites for selective comminution. In Proceedings of the Conference of Metallurgists, Toronto, ON, Canada, 23–26 August 2015; pp. 1–14. [Google Scholar]
- Petrakis, E.; Karmali, V.; Komnitsas, K. Factors affecting nickel upgrade during selective grinding of low-grade limonitic laterites. Miner. Process. Extr. Metall. 2021, 130, 192–201. [Google Scholar] [CrossRef]
- Acquah, G.; Abaka-Wood, G.B.; Addai-Mensah, J.; Asamoah, R. Selective comminution for upgrading nickel and cobalt in nickel laterite ores. Miner. Eng. 2025, 233, 109667. [Google Scholar] [CrossRef]
- Tong, L.; Klein, B.; Zanin, M.; Quast, K.; Skinner, W.; Addai-Mensah, J.; Robinson, D. Stirred milling kinetics of siliceous goethitic nickel laterite for selective comminution. Miner. Eng. 2013, 49, 109–115. [Google Scholar] [CrossRef]
- Ou, L.; Feng, Q.; Chen, Y.; Lu, Y.; Zhang, G. Disintegration mode of bauxite and selective separation of Al and Si. Miner. Eng. 2007, 20, 200–203. [Google Scholar] [CrossRef]
- Yusupov, T.; Kirillova, E.; Denisov, G. Dressing of quartz-feldspar ores on the basis of selective grinding and mechanical activation. J. Min. Sci. 2003, 39, 174–177. [Google Scholar] [CrossRef]
- Danha, G.; Hildebrandt, D.; Glasser, D.; Bhondayi, C. Application of basic process modeling in investigating the breakage behavior of UG2 ore in wet milling. Powder Technol. 2015, 279, 42–48. [Google Scholar] [CrossRef]
- Han, Y.; Zhu, Y.; Wang, Z.; Tian, Y. Effects of DA as a grinding aid on selective grinding of low-grade bauxite. In Proceedings of the International Mineral Processing Congress (IMPC), Brisbane, Australia, 6–10 September 2010; pp. 6–10. [Google Scholar]
- Agatzini-Leonardou, S.; Zafiratos, I.G.; Spathis, D. Beneficiation of a Greek serpentinic nickeliferous ore: Part I. Mineral processing. Hydrometallurgy 2004, 74, 259–265. [Google Scholar] [CrossRef]
- Lu, J.; Sun, M.; Yuan, Z.; Qi, S.; Tong, Z.; Li, L.; Meng, Q. Innovative insight for sodium hexametaphosphate interaction with serpentine. Colloids Surf. A Physicochem. Eng. Asp. 2019, 560, 35–41. [Google Scholar] [CrossRef]
- Swierczek, Z.; Quast, K.; Addai-Mensah, J.; Connor, J.; Li, J.; Robinson, D. Mineralogical characterisation of a sample of an Australian nickel laterite. In Proceedings of the CHEMECA 2011, (Engineering A Better World), Sydney, Australia, 1 January 2011. [Google Scholar]
- Kim, J.; Dodbiba, G.; Tanno, H.; Okaya, K.; Matsuo, S.; Fujita, T. Calcination of low-grade laterite for concentration of Ni by magnetic separation. Miner. Eng. 2010, 23, 282–288. [Google Scholar] [CrossRef]
- Li, J.; Bunney, K.; Watling, H.R.; Robinson, D.J. Thermal pre-treatment of refractory limonite ores to enhance the extraction of nickel and cobalt under heap leaching conditions. Miner. Eng. 2013, 41, 71–78. [Google Scholar] [CrossRef]
- Ribeiro, P.P.M.; Santos, I.D.D.; Neumann, R.; Fernandes, A.; Dutra, A.J.B. Roasting and leaching behavior of nickel laterite ore. Metall. Mater. Trans. B 2021, 52, 1739–1754. [Google Scholar] [CrossRef]
- Lucy, E.A.D.; Bello, G.S.P.; Arnejo, C.G.; Naira, C.P.; Diputado, V.J.; Calvez, J.P.S. Extraction of Nickel, Cobalt, and Scandium from Low-Grade Philippine Laterite Ores Using Atmospheric Leaching with Reduction Pre-Treatment; Springer International Publishing: Cham, Switzerland, 2022; pp. 777–786. [Google Scholar]
- Zhu, D.Q.; Cui, Y.; Vining, K.; Hapugoda, S.; Douglas, J.; Pan, J.; Zheng, G.L. Upgrading low nickel content laterite ores using selective reduction followed by magnetic separation. Int. J. Miner. Process. 2012, 106, 1–7. [Google Scholar] [CrossRef]
- Gao, L.; Liu, Z.; Pan, Y.; Ge, Y.; Feng, C.; Chu, M.; Tang, J. Separation and recovery of iron and nickel from low-grade laterite nickel ore using reduction roasting at rotary kiln followed by magnetic separation technique. Min. Metall. Explor. 2019, 36, 375–384. [Google Scholar] [CrossRef]
- Basturkcu, H.; Acarkan, N.; Gock, E. The role of mechanical activation on atmospheric leaching of a lateritic nickel ore. Int. J. Miner. Process. 2017, 163, 1–8. [Google Scholar] [CrossRef]
- Narasimhan, K.; Rao, R.B.; Das, B. Characterisation and concentration of laterites. Miner. Eng. 1989, 2, 425–429. [Google Scholar] [CrossRef]
- Dekkers, M. Magnetic properties of natural goethite-I. Grain-size dependence of some low-and high-field related rockmagnetic parameters measured at room temperature. Geophys. J. Int. 1989, 97, 323–340. [Google Scholar] [CrossRef]
- Baláž, P. Mechanical activation in hydrometallurgy. Int. J. Miner. Process. 2003, 72, 341–354. [Google Scholar] [CrossRef]
- Erdemoğlu, M.; Baláž, P. An Overview of Surface Analysis Techniques for Characterization of Mechanically Activated Minerals. Miner. Process. Extr. Metall. Rev. 2012, 33, 65–88. [Google Scholar] [CrossRef]
- Asamoah, R.K.; Zanin, M.; Skinner, W.; Addai-Mensah, J. Post-BIOX® Physical Surface Activation of Refractory Flotation Concentrates for Improved Gold Recovery. In Asia Pacific Confederation of Chemical Engineering Congress 2015: APCChE 2015, Incorporating CHEMECA 2015; Engineers Australia: Sydney, Australia, 2015. [Google Scholar]
- Kim, S.-B.; Kim, W.-T. Process for Non-Heating Leaching Nickel and Magnesium from Laterite Using Mechano-Chemical Effect. Google Patents C22B23/0415, 2 June 2011. [Google Scholar]
- Aaltonen, A.; Malmström, R.; Karpale, K. Method for Recovering Nickel and Eventually Cobalt by Extraction from Nickel-Containing Laterite Ore. Yugoslavia YU101703A, 6 May 2009. [Google Scholar]
- Asamoah, R.K.; Skinner, W.; Addai-Mensah, J. Leaching behaviour of mechano-chemically activated bio-oxidised refractory flotation gold concentrates. Powder Technol. 2018, 331, 258–269. [Google Scholar] [CrossRef]
- Yamchelou, M.T.; Law, D.W.; Patnaikuni, I.; Li, J. Alkali activation of mechanically activated low-grade clay. J. Sustain. Cem. Based Mater. 2021, 10, 272–288. [Google Scholar] [CrossRef]
- Tian, L.; Zhang, T.A.; Liu, Y.; Lv, G.Z.; Tang, J.J. Oxidative acid leaching of mechanically activated sphalerite. Can. Metall. Q. 2018, 57, 59–69. [Google Scholar] [CrossRef]
- Yang, H.; Zhao, S.; Wang, G.; Zhang, Q.; Jin, Z.; Tong, L.; Chen, G.; Qiu, X. Mechanical activation modes of chalcopyrite concentrate and relationship between microstructure and leaching efficiency. Hydrometallurgy 2022, 207, 105778. [Google Scholar] [CrossRef]
- Wellham, N.; Rowe, J. Mechanical activation of minerals–past, present and future. In Proceedings of the 4th International Conference on Recent Advance in Materials, Mineral Environment and the 2nd Asian Symposium in Materials and Processing (RAMM & ASMP), Penang, Malaysia, 1–3 June 2009; p. 66. [Google Scholar]
- Tan, Q.; Deng, C.; Li, J. Innovative Application of Mechanical Activation for Rare Earth Elements Recovering: Process Optimization and Mechanism Exploration. Sci. Rep. 2016, 6, 19961. [Google Scholar] [CrossRef]
- Asamoah, R.K.; Addai-Mensah, J.; Skinner, W.; Zanin, M.; Research, I.I.W. Improved Understanding of the Fundamentals of Leaching Refractory Gold Ores. Ph.D. Thesis, University of South Australia, Adelaide, Australia, 2017. [Google Scholar]
- Tunç, T.; Apaydin, F.; Yildiz, K. Effects of mechanical activation on the structure of nickeliferous laterite. Acta Phys. Pol. Ser. A 2013, 123, 349–351. [Google Scholar] [CrossRef]
- Alex, T.; Kumar, R.; Roy, S.; Mehrotra, S. Mechanically induced reactivity of gibbsite: Part 2. Attrition milling. Powder Technol. 2014, 264, 229–235. [Google Scholar] [CrossRef]
- Taylor, L.; Skuse, D.; Blackburn, S.; Greenwood, R. Stirred media mills in the mining industry: Material grindability, energy-size relationships, and operating conditions. Powder Technol. 2020, 369, 1–16. [Google Scholar] [CrossRef]
- Mucsi, G. A review on mechanical activation and mechanical alloying in stirred media mill. Chem. Eng. Res. Des. 2019, 148, 460–474. [Google Scholar] [CrossRef]
- Romeis, S.; Schmidt, J.; Peukert, W. Mechanochemical aspects in wet stirred media milling. Int. J. Miner. Process. 2016, 156, 24–31. [Google Scholar] [CrossRef]
- Naeimi, M.; Vakylabad, A.B.; Hosseini, S.M.; Hassanzadeh, A. A Critical Review of Innovations in Stirred Mill Technology for Enhancing Energy Efficiency. Miner. Process. Extr. Metall. Rev. 2025, 1–16. [Google Scholar] [CrossRef]
- Kumar, A.; Sahu, R.; Tripathy, S.K. Energy-Efficient Advanced Ultrafine Grinding of Particles Using Stirred Mills—A Review. Energies 2023, 16, 5277. [Google Scholar] [CrossRef]
- Elbendari, A.M.; Ibrahim, S.S. Optimizing key parameters for grinding energy efficiency and modeling of particle size distribution in a stirred ball mill. Sci. Rep. 2025, 15, 3374. [Google Scholar] [CrossRef]
- Garces-Granda, A.; Lapidus, G.; Restrepo-Baena, O. The effect of calcination as pre treatment to enhance the nickel extraction from low-grade laterites. Miner. Eng. 2018, 120, 127–131. [Google Scholar] [CrossRef]
- Landers, M.; Gilkes, R.J.; Wells, M. Dissolution kinetics of dehydroxylated nickeliferous goethite from limonitic lateritic nickel ore. Appl. Clay Sci. 2009, 42, 615–624. [Google Scholar] [CrossRef]
- Fan, Q.; Yuan, S.; Wen, J.; He, J. Review on comprehensive utilization of nickel laterite ore. Miner. Eng. 2024, 218, 109044. [Google Scholar] [CrossRef]
- Landers, M.; Gilkes, R.J. Dehydroxylation and dissolution of nickeliferous goethite in New Caledonian lateritic Ni ore. Appl. Clay Sci. 2007, 35, 162–172. [Google Scholar] [CrossRef]
- Garces-Granda, A.; Lapidus, G.; Restrepo-Baena, O. Effect of a thermal pretreatment on dissolution kinetics of a limonitic laterite ore in chloride media. Hydrometallurgy 2020, 196, 105428. [Google Scholar] [CrossRef]
- Landers, M.; Gräfe, M.; Gilkes, R.; Saunders, M.; Wells, M. Nickel distribution and speciation in rapidly dehydroxylated goethite in oxide-type lateritic nickel ores: XAS and TEM spectroscopic (EELS and EFTEM) investigation. Aust. J. Earth Sci. 2011, 58, 745–765. [Google Scholar] [CrossRef]
- O’connor, F.; Cheung, W.; Valix, M. Reduction roasting of limonite ores: Effect of dihydroxylation. Int. J. Miner. Process. 2006, 80, 88–99. [Google Scholar] [CrossRef]
- Dong, B.; Tian, Q.; Xu, Z.; Guo, X.; Wang, Q.; Li, D. The effect of pre-roasting on atmospheric sulfuric acid leaching of saprolitic laterites. Hydrometallurgy 2023, 218, 106063. [Google Scholar] [CrossRef]
- Acquah, G.; Spiridonov, P.; Abaka-Wood, G.; Skinner, W.; Addai-Mensah, J.; Asamoah, R. Enhanced extraction of nickel and cobalt from complex refractory laterite ores through thermal pretreatment. 2026; Under review. [Google Scholar]
- MacCarthy, J.; Nosrati, A.; Skinner, W.; Addai-Mensah, J. Atmospheric acid leaching mechanisms and kinetics and rheological studies of a low grade saprolitic nickel laterite ore. Hydrometallurgy 2016, 160, 26–37. [Google Scholar] [CrossRef]
- Wang, F.; Dreisinger, D.; Xiao, Y. Accelerated CO2 mineralization and utilization for selective battery metals recovery from olivine and laterites. J. Clean. Prod. 2023, 393, 136345. [Google Scholar] [CrossRef]
- Quaicoe, I.; Nosrati, A.; Skinner, W.; Addai-Mensah, J. Agglomeration and column leaching behaviour of goethitic and saprolitic nickel laterite ores. Miner. Eng. 2014, 65, 1–8. [Google Scholar] [CrossRef]
- Caron, M. Fundamental and practical factors in ammonia leaching of nickel and cobalt ores. JOM-J. Miner. Met. Mater. Soc. 1950, 2, 67–90. [Google Scholar] [CrossRef]
- Pandey, N.; Tripathy, S.K.; Patra, S.K.; Jha, G. Recent progress in hydrometallurgical processing of nickel lateritic ore. Trans. Indian Inst. Met. 2023, 76, 11–30. [Google Scholar] [CrossRef]
- Çoban, B.; Baş, A.D. HPAL of a lateritic nickel ore: An investigation on the relationship between nickel and cobalt extraction and acid consumption. Miner. Eng. 2024, 218, 109014. [Google Scholar] [CrossRef]
- Muir, D.; Johnson, J. Physical beneficiation and bioleaching of arid region nickel laterites. In Proceedings of the IMPC 2006—Proceedings of 23rd International Mineral Processing Congress, Istanbul, Turkey, 3–8 September 2006. [Google Scholar]
- Gharaei, A.A.; Rezai, B.; Shormasti, H.H. Environmental friendly approach: Atmospheric and high-pressure acid leaching studies, low-grade laterites nickel processing. J. Min. Environ. 2024, 15, 1591–1606. [Google Scholar]
- Kaya, Ş.; Topkaya, Y.A. High pressure acid leaching of a refractory lateritic nickel ore. Miner. Eng. 2011, 24, 1188–1197. [Google Scholar] [CrossRef]
- Acquah, G.; Ritch, S.; Mensah, J.A.; Asamoah, R. Pressure Leaching of Refractory Low-Grade Nickel Laterite Ores: Effect of Additives. In Ni-Co 2025, 6th International Symposium on Nickel and Cobalt; Springer Nature: Cham, Switzerland, 2025; pp. 177–188. [Google Scholar]
- MilestoneSrl. SynthWave Microwave Digestion System: User Manual and Technical Specifications; Milestone Srl: Sorisole, Italy, 2018. [Google Scholar]
- Reid, J.; Barnett, S. Nickel laterite hydrometallurgical processing update. In ALTA 2002 Nickel/Cobalt; ALTA Metallurgical Services: Perth, Australia, 2002; Volume 8. [Google Scholar]
- Carpen, H.L.; Giese, E.C. Enhancement of nickel laterite ore bioleaching by Burkholderia sp. using a factorial design. Appl. Water Sci. 2022, 12, 181. [Google Scholar] [CrossRef]
- Castro, I.; Fietto, J.; Vieira, R.; Trópia, M.; Campos, L.; Paniago, E.; Brandão, R. Bioleaching of zinc and nickel from silicates using Aspergillus niger cultures. Hydrometallurgy 2000, 57, 39–49. [Google Scholar] [CrossRef]
- Nasab, M.H.; Noaparast, M.; Abdollahi, H.; Amoozegar, M.A. Indirect bioleaching of Co and Ni from iron rich laterite ore, using metabolic carboxylic acids generated by P. putida, P. koreensis, P. bilaji and A. niger. Hydrometallurgy 2020, 193, 105309. [Google Scholar] [CrossRef]
- Behera, S.K.; Panda, P.P.; Singh, S.; Pradhan, N.; Sukla, L.B.; Mishra, B.K. Study on reaction mechanism of bioleaching of nickel and cobalt from lateritic chromite overburdens. Int. Biodeterior. Biodegrad. 2011, 65, 1035–1042. [Google Scholar] [CrossRef]
- Dash, J.; Ojha, R.; Pradhan, D. Progress in bioleaching and its mechanism: A short review. Discov. Environ. 2025, 3, 238. [Google Scholar] [CrossRef]
- Duyvesteyn, W.P.; Omofoma, M. Recovery of Nickel from Bioleach Solution. WO1998026100A1, 18 June 1998. [Google Scholar]
- Tzeferis, P.; Agatzini-Leonardou, S. Leaching of nickel and iron from Greek non-sulphide nickeliferous ores by organic acids. Hydrometallurgy 1994, 36, 345–360. [Google Scholar] [CrossRef]
- Valix, M.; Usai, F.; Malik, R. Fungal bio-leaching of low grade laterite ores. Miner. Eng. 2001, 14, 197–203. [Google Scholar] [CrossRef]
- Tang, J.A.; Valix, M. Leaching of low grade limonite and nontronite ores by fungi metabolic acids. Miner. Eng. 2006, 19, 1274–1279. [Google Scholar] [CrossRef]
- Mishra, S.; Panda, S.; Akcil, A.; Dembele, S. Biotechnological avenues in mineral processing: Fundamentals, applications and advances in bioleaching and bio-beneficiation. Miner. Process. Extr. Metall. Rev. 2023, 44, 22–51. [Google Scholar] [CrossRef]
- Msumange, D.A.; Jampaiah, D.; Tardio, J.; Nilsson, M.; Arandiyan, H.; Bhargava, S.K. A Review of Nickel and Cobalt from Laterite Ores: Toward Sustainable Extraction Pathways for the Battery Industry. J. Sustain. Metall. 2026, 1–32. [Google Scholar] [CrossRef]
- Jain, N.; Sharma, D.K. Biohydrometallurgy for Nonsulfidic Minerals—A Review. Geomicrobiol. J. 2004, 21, 135–144. [Google Scholar] [CrossRef]
- McDonald, R.; Whittington, B. Atmospheric acid leaching of nickel laterites review. Part II. Chloride Bio-technologies. Hydrometallurgy 2008, 91, 56–69. [Google Scholar] [CrossRef]
- Readett, D.; Fox, J. Commerciallisation of Ni heap leaching at Murrin Murrin operations. In Proceedings of the XXV International Mineral Processing Congress, Brisbane, Australia, 6–10 September 2010. [Google Scholar]
- Oxley, A.; Gardner, N.; Caceres, O.; Fonseca, C.; Chiron, D. The Piauí Nickel Project, Brejo Seco Deposit, and the Application of Heap Leaching: An Overview of the Deposit. In Ni-Co 2025, 6th International Symposium on Nickel and Cobalt: Proceedings of the Extraction 2025 Meeting & Exhibition, Volume II; Springer Nature: Berlin/Heidelberg, Germany, 2025; Volume 2, p. 309. [Google Scholar]
- Nosrati, A.; Quast, K.; Xu, D.; Skinner, W.; Robinson, D.J.; Addai-Mensah, J. Agglomeration and column leaching behaviour of nickel laterite ores: Effect of ore mineralogy and particle size distribution. Hydrometallurgy 2014, 146, 29–39. [Google Scholar] [CrossRef]
- Xu, D.; Liu, L.X.; Quast, K.; Addai-Mensah, J.; Robinson, D.J. Effect of nickel laterite agglomerate properties on their leaching performance. Adv. Powder Technol. 2013, 24, 750–756. [Google Scholar] [CrossRef]
- Readett, D.; Fox, J. Agglomeration—The key to success for the Murrin Murrin Ni laterite heap. In Proceedings of the Metallurgical Plant Design and Operating Strategies (MetPlant 2011), Perth, Australia, 8–9 August 2011. [Google Scholar]
- Elliot, A.; Fletcher, H.; Li, J.; Watling, H.; Robinson, D. Heap leaching of nickel laterites—A challenge and an opportunity. In Hydrometallurgy of Nickel and Cobalt 2009, Proceeding of the 39th Annual Hydrometallurgy Meeting Held in the Conjunction with the 48th Annual Conference of Metallurgists, Sudbury, ON, Canada, 23–26 August 2009; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2009. [Google Scholar]
- Readett, D.; Fox, J. Development of heap leaching at its integration into the Murrin Murrin operations. In Proceedings of the ALTA Nickel Cobalt Conference, Perth, Australia, 25–27 May 2009. [Google Scholar]
- Kurşunoglu, S. Extraction of nickel from a mixed nickel-cobalt hydroxide precipitate. Bilimsel Madencilik Derg. 2019, 58, 45–52. [Google Scholar] [CrossRef]
- MacCarthy, J.; Nosrati, A.; Skinner, W.; Addai-Mensah, J. Acid leaching and rheological behaviour of a siliceous goethitic nickel laterite ore: Influence of particle size and temperature. Miner. Eng. 2015, 77, 52–63. [Google Scholar] [CrossRef]
- Kursunoglu, S. Synergistic effect of organic acid on the dissolution of mixed nickel-cobalt hydroxide precipitate in sulphuric acid solution. Metall. Res. Technol. 2019, 116, 319. [Google Scholar] [CrossRef]
- Li, G.; Zhou, Q.; Zhu, Z.; Luo, J.; Rao, M.; Peng, Z.; Jiang, T. Selective leaching of nickel and cobalt from limonitic laterite using phosphoric acid: An alternative for value-added processing of laterite. J. Clean. Prod. 2018, 189, 620–626. [Google Scholar] [CrossRef]
- Ma, B.; Wang, C.; Yang, W.; Yang, B.; Zhang, Y. Selective pressure leaching of Fe (II)-rich limonitic laterite ores from Indonesia using nitric acid. Miner. Eng. 2013, 45, 151–158. [Google Scholar] [CrossRef]
- Zhao, Y.; Gao, J.-M.; Yue, Y.; Peng, B.; Que, Z.-Q.; Guo, M.; Zhang, M. Extraction and separation of nickel and cobalt from saprolite laterite ore by microwave-assisted hydrothermal leaching and chemical deposition. Int. J. Miner. Metall. Mater. 2013, 20, 612–619. [Google Scholar] [CrossRef]
- MacCarthy, J.; Nosrati, A.; Skinner, W.; Addai-Mensah, J. Atmospheric acid leaching of nickel laterite: Effect of temperature, particle size and mineralogy. In Chemeca 2014: Processing Excellence; Powering Our Future; Engineers Australia: Barton, ACT, Australia, 2014; pp. 1273–1286. [Google Scholar]
- Önal, M.A.R.; Topkaya, Y.A. Pressure acid leaching of Çaldağ lateritic nickel ore: An alternative to heap leaching. Hydrometallurgy 2014, 142, 98–107. [Google Scholar] [CrossRef]
- Guo, X.-Y.; Shi, W.-T.; Dong, L.; Tian, Q.-H. Leaching behavior of metals from limonitic laterite ore by high pressure acid leaching. Trans. Nonferr. Met. Soc. China 2011, 21, 191–195. [Google Scholar] [CrossRef]
- Nasab, M.H.; Noaparast, M.; Abdollahi, H. Dissolution optimization and kinetics of nickel and cobalt from iron-rich laterite ore, using sulfuric acid at atmospheric pressure. Int. J. Chem. Kinet. 2020, 52, 283–298. [Google Scholar] [CrossRef]
- Biswas, S.; Chakraborty, S.; Chaudhuri, M.G.; Banerjee, P.C.; Mukherjee, S.; Dey, R. Optimization of process parameters and dissolution kinetics of nickel and cobalt from lateritic chromite overburden using organic acids. J. Chem. Technol. Biotechnol. 2014, 89, 1491–1500. [Google Scholar] [CrossRef]
- Che, X.; Su, X.; Chi, R.A.; Yu, J. Microwave assisted atmospheric acid leaching of nickel from laterite ore. Rare Met. 2010, 29, 327–332. [Google Scholar] [CrossRef]
- Nicol, M.J.; Zainol, Z. The development of a resin-in-pulp process for the recovery of nickel and cobalt from laterite leach slurries. Int. J. Miner. Process. 2003, 72, 407–415. [Google Scholar] [CrossRef]
- Sole, K.C.; Mooiman, M.B.; Hardwick, E. Ion exchange in hydrometallurgical processing: An overview and selected applications. Sep. Purif. Rev. 2018, 47, 159–178. [Google Scholar] [CrossRef]
- Littlejohn, P.; Vaughan, J. Recovery of nickel and cobalt from laterite leach tailings through resin-in-pulp scavenging and selective ammoniacal elution. Miner. Eng. 2013, 54, 14–20. [Google Scholar] [CrossRef]
- Mendes, F.D.; Martins, A.H. Recovery of nickel and cobalt from acid leach pulp by ion exchange using chelating resin. Miner. Eng. 2005, 18, 945–954. [Google Scholar] [CrossRef]
- Kuz, V.; Kuz, D. Sorption of nickel and copper from leach pulps of low-grade sulfide ores using Purolite S930 chelating resin. Hydrometallurgy 2014, 141, 76–81. [Google Scholar] [CrossRef]
- McKevitt, B.; Dreisinger, D. Development of an engineering model for nickel loading onto an iminodiacetic resin for resin-in-pulp applications: Part I—Method development and discussion of rate limiting factors. Hydrometallurgy 2012, 121–124, 35–44. [Google Scholar] [CrossRef]
- Liebenberg, C.; Dorfling, C.; Bradshaw, S.; Akdogan, G.; Eksteen, J. The recovery of nickel and cobalt from a sulphate bioleach solution using Dow M4195. In Proceedings of the Southern African Institute of Mining and Metallurgy Base Metals Conference, Mpumalanga, South Africa, 2–4 September 2013; pp. 269–282. [Google Scholar]
- Eksteen, J.; Oraby, E.; Nguyen, V. Leaching and ion exchange based recovery of nickel and cobalt from a low grade, serpentine-rich sulfide ore using an alkaline glycine lixiviant system. Miner. Eng. 2020, 145, 106073. [Google Scholar] [CrossRef]
- Spiridonov, P.; Hein, H.; Hein, R. The RIMM process—An advanced metal extraction method. In Proceedings of the 21st Annual ALTA Conference; Australia ALTA Metallurgical Services: Melbourne, Australia, 2016; p. 301. [Google Scholar]
- Junior, A.B.B.; Espinosa, D.C.R.; Dreisinger, D.; Tenório, J.A.S. Recovery of nickel and cobalt from nickel laterite leach solution using chelating resins and pre-reducing process. Can. J. Chem. Eng. 2019, 97, 1181–1190. [Google Scholar] [CrossRef]
- Deepatana, A.; Tang, J.A.; Valix, M. Comparative study of chelating ion exchange resins for metal recovery from bioleaching of nickel laterite ores. Miner. Eng. 2006, 19, 1280–1289. [Google Scholar] [CrossRef]
- Deepatana, A.; Valix, M. Recovery of nickel and cobalt from organic acid complexes: Adsorption mechanisms of metal-organic complexes onto aminophosphonate chelating resin. J. Hazard. Mater. 2006, 137, 925–933. [Google Scholar] [CrossRef]
- Spiridonov, P.; Skinner, W.; Addai-Mensah, J.; Hein, H.; Hein, R. Investigation of the resin in moist mix process for the nickel and cobalt recovery from laterites. In Proceedings of the ALTA 24th Annual Conference Proceedings Nickel-Cobalt-Copper Conference, Perth, Australia, 20–24 May 2019. [Google Scholar]
- Spiridonov, P.; Abaka-Wood, G.; Skinner, W.; Addai-Mensah, J.; Hein, H.; Hein, R. Further development of the continuous ion exchange process for copper recovery from fines. In Proceedings of the Alta 24th Annual Conference: Nickel-Cobalt-Copper Conference, Perth, Australia, 20–24 May 2019. [Google Scholar]
- Asamoah, R.K.; Zanin, M.; Skinner, W.; Addai-Mensah, J. Influence of activated carbon on leaching behaviour of Eburnean Tectonic refractory bio-oxidised flotation concentrates. In Chemeca 2016: Chemical Engineering-Regeneration, Recovery and Reinvention; Engineers Australia: Melbourne, Australia, 2016; pp. 432–443. [Google Scholar]
- Owusu, C.K.; Konadu, K.T.; Acquah, G.; Mends, E.A.; Amankwah, R.K.; Sasaki, K. Improving gold recovery in carbonaceous gold ores using naphthalene sulphonate as a blanking agent. Miner. Eng. 2023, 202, 108283. [Google Scholar] [CrossRef]
- Owusu, C.; Mends, E.A.; Acquah, G. Enhancing the physical qualities of activated carbon produced from palm kernel shell via response surface methodology—Process variable optimization. In Biomass Conversion and Biorefinery; Springer: Berlin/Heidelberg, Germany, 2022; pp. 1–15. [Google Scholar]
- Acquah, G.; Spiridonov, P.; Addai-Mensah, J.; Asamoah, R. Resin in moist mix behaviour of refractory nickel laterite ore- process variable effect via a factorial design. In Proceedings of the 6th International Symposium, Nickel and Cobalt, Phoenix, AZ, USA, 16–22 November 2025. [Google Scholar]
- Spiridonov, P.; Asamoah, R.; Statsenko, L.; Skinner, W.; Addai-Mensah, J.; Mifsud, J.; Latay, A.; Abaka-Wood, G.; Amankwaa-Kyeremeh, B. Copper and Cobalt Recovery from Old Flotation Tailings. In Proceedings of the Alta 27th Annual Conference: Nickel-Cobalt-Copper Conference, Perth, Australia, 1–5 May 2023. [Google Scholar]
- Quast, K.; Xu, D.; Skinner, W.; Nosrati, A.; Hilder, T.; Robinson, D.J.; Addai-Mensah, J. Column leaching of nickel laterite agglomerates: Effect of feed size. Hydrometallurgy 2013, 134–135, 144–149. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.