Abstract
Mining tailings are considered a significant environmental challenge due to their large quantities and high residual metal content, particularly iron. Recent developments in biogenic technologies offer a sustainable approach to recovering valuable materials from these waste streams. We consider a biogenic iron oxide nanoparticles production process from mining tailings as an environmentally friendly route to magnetic materials. Microorganisms, including iron-oxidizing and iron-reducing bacteria, microalgae, and fungi, can convert soluble and mineral-bound iron into iron oxide nanoparticles (NPs) phases such as magnetite, maghemite, and hematite. These biogenic iron oxide NPs often exhibit specific physicochemical properties, including controlled particle size, high surface area, and engineered magnetic properties, which make them potentially important for applications in environmental remediation, catalysis, and agriculture. The processes behind microbial iron conversion, the parameters governing mineral phase formation, and the approaches for optimizing the process are presented. This strategy supports the circular economy concept by combining biogenic synthesis with various forms of mining waste, thereby reducing environmental threats associated with tailings confinement and providing an environmentally friendly mechanism for the production of value-added magnetic materials.
1. Introduction
The rapid expansion of the mining sector has led to the accumulation of substantial volumes of mining tailings, which constitute one of the most significant solid waste streams worldwide [1,2]. These tailings often contain elevated amounts of residual metals, particularly iron, and pose significant environmental and health hazards due to issues such as acid mine drainage, metal leaching, and prolonged ecosystem contamination [2]. Conventional methods for remediation and metal recovery predominantly rely on energy-intensive physicochemical treatments that are resource-intensive and economically unfeasible at large scales [3]. As a result, there is an increasing interest in sustainable technologies that can both reduce environmental harm and reclaim valuable resources from mining byproducts.
Iron oxides rank among the most critically important magnetic materials for industrial use, finding applications in environmental remediation, catalysis, data storage, biomedical fields, and agriculture [4,5,6]. Historically, the production processes for iron oxides have relied on high-temperature chemical synthesis or extraction from primary iron ores, methods associated with significant energy consumption and greenhouse gas emissions [6]. Conversely, mining tailings serve as an underexploited secondary resource rich in iron-bearing minerals that could potentially be converted into valuable magnetic materials through environmentally friendly extraction techniques [7].
Biogenic synthesis pathways offer a non-toxic approach for generating Fe3O4 NPs facilitated by the reduction of Fe(III) minerals to Fe(II), thereby assisting in the nucleation and development of magnetite under anaerobic or microaerophilic conditions [8]. Additionally, microalgae and fungi can influence iron speciation through their metabolic functions, extracellular polymeric substances, and localized redox gradients, which lead to the precipitation of magnetic iron oxide NPs [9]. These biologically mediated processes occur under mild conditions, thus negating the necessity for high-temperature synthesis or hazardous chemical agents [9].
A significant benefit of biogenic Fe3O4 NPs production is its adaptability regarding particle size, morphology, and magnetic response, which can be influenced by adjusting biological and environmental variables [9,10]. The resulting biogenic magnetite typically exhibits a high surface area and strong magnetic susceptibility traits, which are particularly beneficial for magnetic recovery and reuse [11]. By facilitating rapid magnetic separation, biogenic Fe3O4 NPs can be seamlessly integrated into downstream applications, such as contaminant adsorption, nutrient delivery systems, or catalytic transformations, thereby contributing to material loops within a circular economy model [9,12].
Despite encouraging results observed at laboratory scales, various obstacles impede large-scale recovery of biogenic Fe3O4 NPs from mining tailings. Challenges include variability in tailings composition, maintaining phase purity, and controlling crystallinity, as well as scalability issues in the processes [9]. Overcoming these challenges requires a deeper understanding of microbe–mineral interactions, pathways for iron transformation, and enhanced efficiency in magnetic separation under realistic operational conditions [9].
This article provides a critical overview of recent progress in the biogenic production of Fe3O4 NPs from mining tailings, focusing on the mechanisms aspects. By connecting microbial iron transformations with the recovery of magnetic materials, this research underscores the potential for biomagnetic strategies to convert mining tailings from an environmental problem into valuable products.
2. Iron Ore as a Resource for Biosynthesis
A diverse array of iron-containing minerals found in iron ores and mining byproducts can serve as foundational components for the biological generation of iron oxides [13]. Common examples include pyrite (FeS2), siderite (FeCO3), hematite (Fe2O3), magnetite (Fe3O4), and various iron oxyhydroxides such as ferrihydrite and goethite [14]. The oxidation state, degree of crystallinity, and thermodynamic stability of these minerals differ considerably, which impacts their availability to microorganisms [3,15]. Iron-oxidizing bacteria have the capability to oxidize reduced minerals like FeS2 and FeCO3, whereas iron-reducing microorganisms can utilize Fe(III)-rich forms (Fe2O3, FeOOH) as electron acceptors [7,16]. In the context of microbial iron cycling, magnetite, which contains both Fe2+ and Fe3+, plays a dual role as both a substrate and a product [16,17].
Although these minerals are prevalent, many iron-bearing forms exhibit low solubility in water under near-neutral pH levels and oxygen-rich environments, thereby significantly restricting the bioavailability of iron [18]. Crystalline varieties, such as hematite and magnetite, dissolve slowly, hindering direct microbial uptake. To navigate these challenges, microorganisms employ tactics such as acidifying their surrounding environment, producing chelating agents and siderophores, and leveraging redox-driven dissolution processes [15]. In mining tailings, the high crystallinity coupled with surface passivation of iron minerals further diminishes bioaccessibility; thus, biological or physicochemical activation methods are essential to improve the release of iron for biosynthetic purposes [15].
Synthetic iron salts such as FeCl2 and FeCl3 offer readily soluble sources of iron and are commonly utilized in laboratory-scale biosynthesis [19]. However, they are prone to rapid hydrolysis, precipitation issues, and instability which vary with pH levels; this often necessitates stringent management of solution chemistry [20]. On the other hand, natural iron ores provide a more gradual source of iron, facilitating controlled biomineralization while minimizing the risks associated with iron toxicity [13]. While synthetic salts can accelerate reaction rates, mineral-based sources encourage slower microbial transformations that yield iron oxides with adjustable particle sizes, shapes, and magnetic characteristics [7]. From a sustainability perspective, using natural iron minerals from ores or mining residues can support resource circularity and reduce dependence on chemically refined iron salts [7]. Nevertheless, such minerals often contain trace metals and metalloids (e.g., arsenic, lead, and cadmium), which could potentially be released or co-concentrated during processing, resulting in final products [13]. This places the products at risk of contamination and of potential human-health problems, particularly when intended for water treatment, agricultural use, or biomedical applications. As such, a comprehensive risk assessment framework, including impurity profiling, leaching behaviour, and ecotoxicological effects, is mandatory whenever mineral-based feedstocks are applied [21,22]. By life-cycle assessment, secondary mineral resources may entail a lower primary extraction burden, but the additional purification, waste management, and monitoring required to control impurities can partially or fully offset the benefits [13]. Therefore, any perceived sustainability benefit of using mineral-derived precursors must be assessed comprehensively, considering not only resource efficiency but also impurity handling, regulatory constraints, and end-of-life impacts.
FeS2, FeCO3, Fe2O3, Fe3O4, and FeOOH are iron-rich minerals present in ores and tailings that serve as sustainable precursors for the generation of biogenic iron oxide [13]. Shewanella spp. and Geobacter spp. facilitate the reduction of Fe(III) compounds (Fe2O3, FeOOH), which promotes the formation of secondary magnetite [8]. In contrast, Acidithiobacillus spp. oxidize reduced iron minerals (FeS2, FeCO3), leading to an increased release of Fe2+ under acidic conditions [23,24]. Additionally, microalgae such as Chlorella and Scenedesmus contribute to iron solubilization by producing extracellular polymeric substances and manipulating local pH levels, thereby enabling controlled biomineralization [9]. Although mineral-derived iron is not as readily soluble as synthetic iron salts (FeCl2, FeCl3), it provides a prolonged release of iron with reduced toxicity and enhanced regulation over particle growth. This mineral-based approach aligns with sustainable practices for utilizing mining waste and fosters environmentally friendly synthesis pathways (Table 1) [14,25].

Table 1.
Comparison of mineral iron salts from mining tailings.
3. Iron Oxide Biosynthesis
Iron oxides, especially magnetite (Fe3O4) NPs, are crucial materials used in various technological applications, including environmental cleanup, magnetic separation, catalysis, and sustainable farming [4]. Traditional methods for synthesizing Fe3O4 NPs typically involve high temperatures, potent reducing agents, and rigorous pH control, resulting in processes that consume significant energy and produce environmentally harmful byproducts (Table 2) [5,9]. In contrast, the biosynthesis of iron oxide NPs present a more sustainable option by utilizing the metabolic functions and surface interactions of microorganisms to facilitate the transformation of iron minerals under mild conditions (Table 3) [9,12,26]. Though chemical synthesis is generally cheaper at scale, biogenic processes offer benefits in biocompatibility, specific surface functionality, and particle uniformity [27,28]. The cost of the biogenic process is mostly influenced by the cost of bioreactors, the need to control microbial cultivation, and labour and maintenance costs [29].

Table 2.
Comparison of biogenic to chemically synthesized Fe3O4 NPs.

Table 3.
Comparison of chemically synthesized iron oxide to biogenic iron oxide.
Biological entities, such as iron-reducing bacteria, magnetotactic bacteria, and microalgae, play a vital role in the redox cycling between Fe3+ and Fe2+, which is essential for the formation of magnetite [9,12,41]. In systems that utilize microalgae, functional groups found on cell walls and extracellular polymeric substances (EPSs) act as sites for nucleation where iron can be adsorbed and subsequently biomineralized (Table 3) [9]. This process yields biogenic iron oxide NPs characterized by distinct surface chemistry and enhanced dispersion stability. Notably, low-grade iron ores and mining tailings are plentiful yet underutilized sources of iron that can be biologically transformed into valuable magnetic materials, aligning with principles of a circular economy [7,10,13]. The biogenic synthesis of Fe3O4 NPs from iron sourced from mining waste not only diminishes mining waste generation and environmental repercussions but also produces magnetically responsive materials that are advantageous for recovery, reuse, and application in biomagnetic separation as well as remediation technologies [1,10,42].
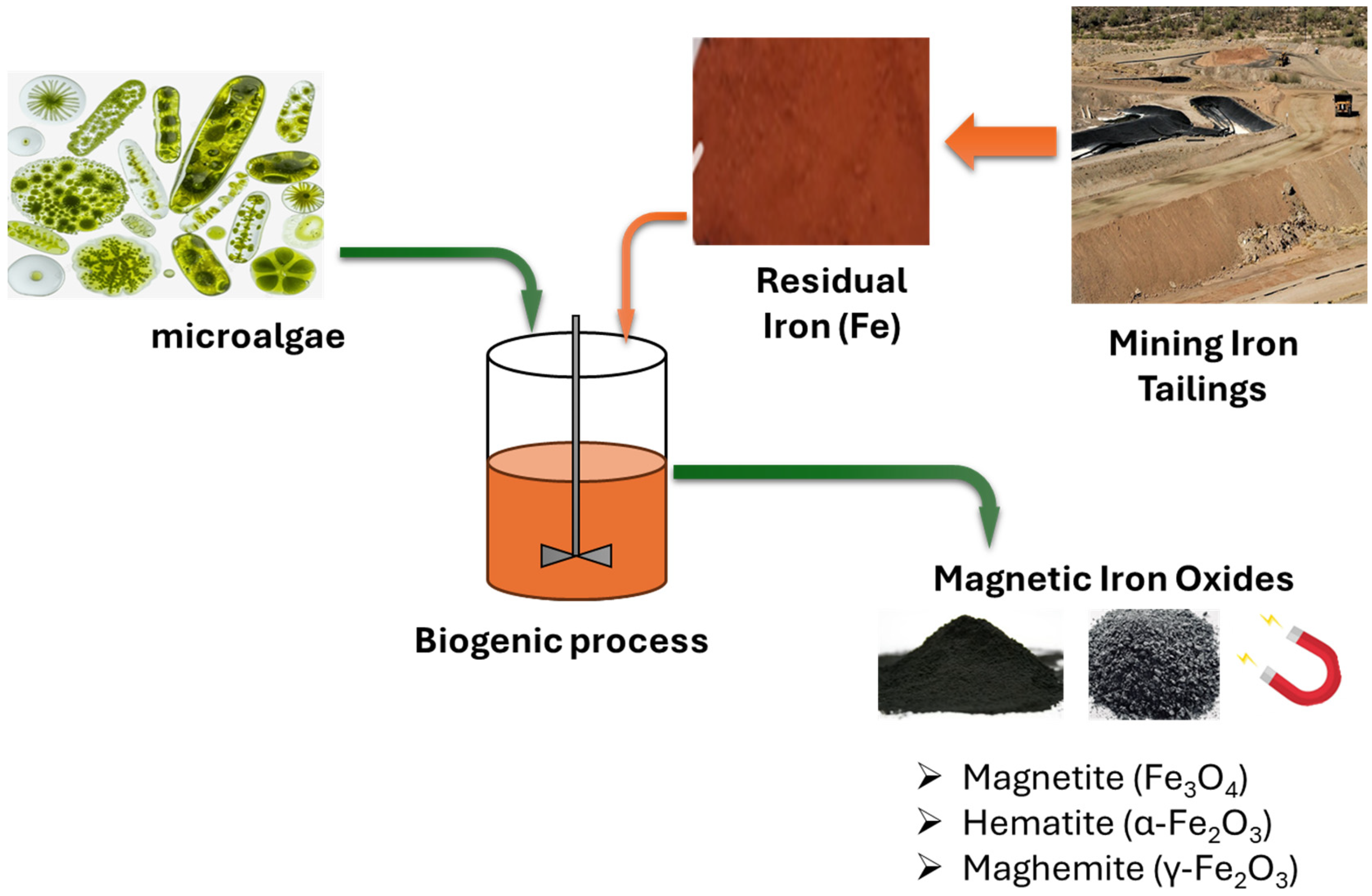
Figure 1 illustrates the formation of biogenic iron oxide (Fe3O4) from mining tailings, facilitated by microalgae. The dissolution of Fe(III)-containing minerals releases Fe3+ ions that adhere to algal cell walls through carboxyl, hydroxyl, and amino functional groups [9]. The partial biological reduction of Fe3+ to Fe2+, combined with EPS templating, encourages the nucleation and development of magnetite nanoparticles both on the cell surface and within the biomatrix [9]. The resulting biogenic Fe3O4 exhibits enhanced magnetic responsiveness, enabling effective recovery and reuse through magnetic means.
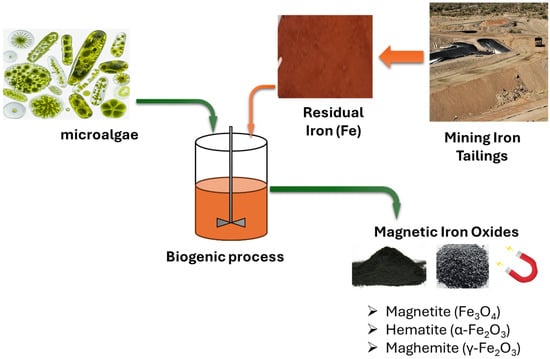
Figure 1.
Schematic diagram of microalgae-assisted biogenic iron oxide produced from mining tailings.
3.1. Mechanisms of Biosynthesis
The formation of iron oxides is influenced by biologically mediated redox reactions, surface interactions, and mineral transformation processes that occur under mild environmental conditions [43].
Biological systems can either directly reduce Fe3+ or indirectly enhance its transformation through metabolic byproducts, creating suitable conditions for the precipitation of mixed-valence iron oxides [44].
One significant mechanism involves biosorption and surface-mediated nucleation, where dissolved iron species from iron-rich minerals or mining tailings adhere to cell walls containing carboxyl, hydroxyl, and phosphate functional groups [8,9]. These functional sites serve as nucleation centres, concentrating iron and reducing the activation energy required for mineral formation. In microalgae, extracellular polymeric substances (EPSs) further stabilize iron complexes and direct the growth of nanoparticles, leading to well-dispersed biogenic iron oxides [8,9,45].
Another important mechanism includes enzymatic and metabolic redox control. Iron-reducing microorganisms, such as Shewanella and Geobacter, facilitate the reduction of Fe3+ to Fe2+, enabling the conversion of poorly crystalline ferrihydrite into magnetite [46]. In microalgal systems, photosynthetically induced redox gradients and organic exudates similarly promote partial reduction of Fe3+, aiding in magnetite nucleation [46,47].
Biomineral transformation and maturation dictate changes in phases, crystallinity, and magnetic characteristics [9]. Initially amorphous or poorly ordered iron hydroxides gradually convert to crystalline Fe3O4 through aging and redox equilibration. These biologically mediated pathways produce iron oxides with inherent surface functionalization and improved magnetic responsiveness, rendering them particularly advantageous for magnetic recovery and environmental applications [9].
3.1.1. Bioleaching of Iron Ore
Bioleaching is a biologically mediated through redox, acid, and complexation processes [48]. Bioleaching is an eco-friendly and sustainable method for mobilising iron in iron-rich ores and mining tailings. It produces bioavailable iron species that aid in biomineralization formation of biogenic magnetite (Fe3O4) [48]. Chemolithotrophic bacteria are responsible for iron ore bioleaching by oxidising ferrous iron (Fe2+) and reducing sulphur compounds. Acidophilic iron-oxidizing bacteria such as Acidithiobacillus ferrooxidans, Leptospirillum ferrooxidans, and Acidithiobacillus ferrivorans drive the oxidation of Fe2+ to Fe3+ in acidic environments, playing a key role in iron redox cycling [46].
The resultant Fe3+ serves as a good oxidant with the secondary dissolving effects on iron-bearing minerals, such as magnetite, hematite, and iron sulphides [49]. At the same time, small sulphur-oxidizing organisms also facilitate proton generation from the oxidation of the elemental sulphur and sulphide phases, and thereby the low pH that is necessary to achieve appropriate solubilization of Fe2+ [49,50]. Bioleaching has both direct and indirect pathways. In a direct manner, microbial cells attach to the mineral surface and enzymatically oxidize the structural iron, breaking down the iron crystal lattice [49]. In the indirect mechanism, the mineral abiotically oxidises microbially regenerated Fe3+, leaving the reduced Fe2+ that is produced re-oxidised by microorganisms to create a continuous Fe2+/Fe3+ redox cycle [49]. Such a cyclic step is particularly important in tailings systems. Iron is often found in mixed-valence oxide phases with almost no solubility at neutral conditions [49].
The bioleaching environment is closely regulated by microorganisms. The pH is controlled as proton production from sulphur oxidation and ferric hydrolysis is the result, while redox potential is maintained through balancing biological redox rates due to iron oxidation (i.e., iron is a reactive iron) with mineral-based iron reduction [48,50,51]. They not only help with iron mobilisation, but they also influence iron speciation, aggregation behaviour, and reprecipitation pathways. Excess ferric iron accumulation or uncontrolled variations in pH may limit microbial activity, underscoring the need for a balanced systemic design of bioleaching-based processes [49].
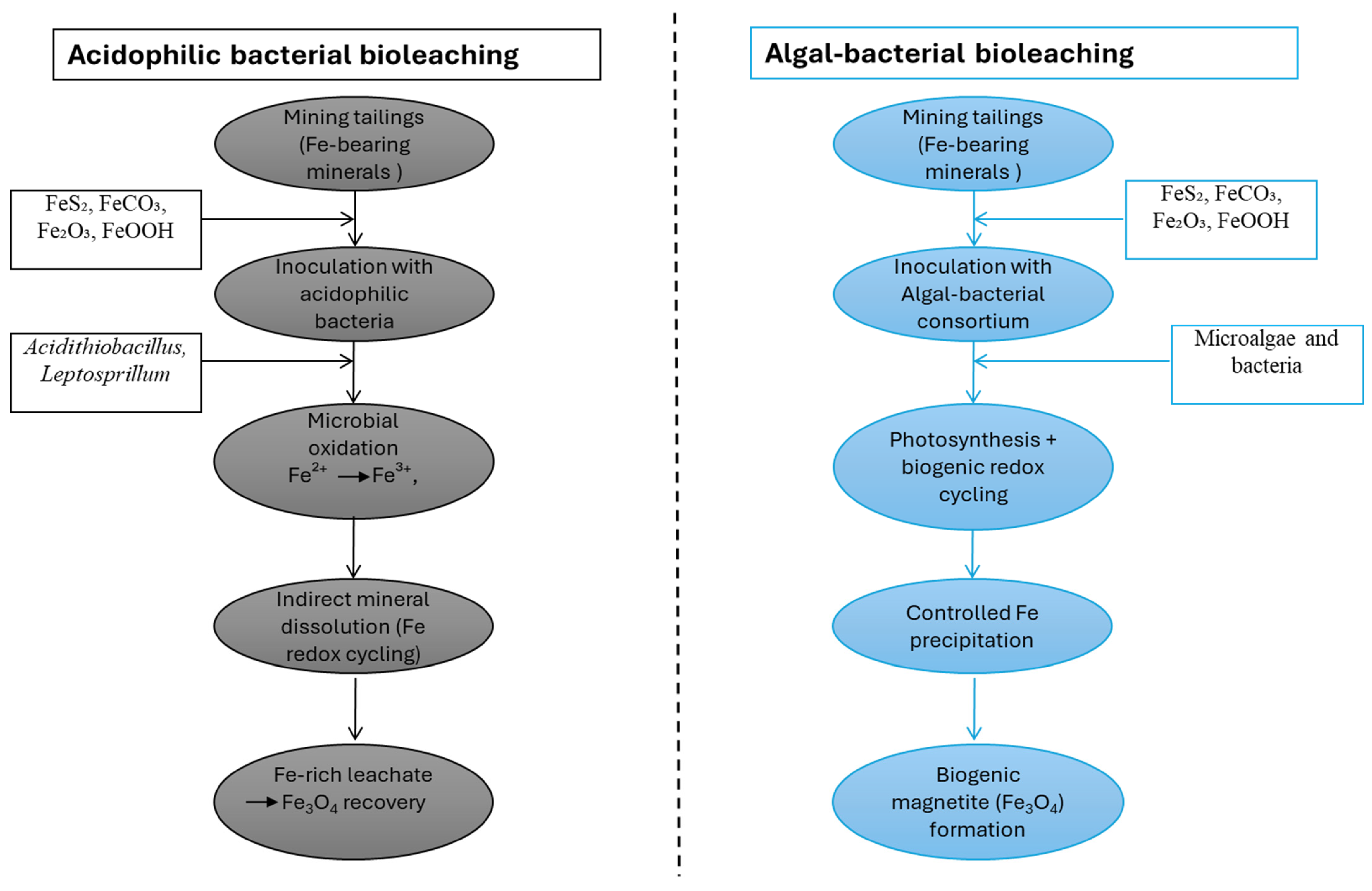
As shown in Figure 2, in algal–bacterial bioleaching systems, algae indirectly enhance iron mobility by providing oxygen, organic acids, and chelating biomolecules, whereas bacteria mediate the core Fe2+/Fe3+ redox transformations responsible for mineral dissolution [52,53]. This metabolic coupling creates localized pH and ORP microenvironments at the ore surface that allow for efficient iron release, even under circumneutral to mildly acidic (pH 5.5–7.5) conditions with moderate and spatially heterogeneous redox potentials (0 to +300 mV) [53]. Algal–bacterial bioleaching processes differ from traditional acid-driven bacterial processes in that they involve such synergistic interactions [52]. In such consortia, microalgae (e.g., Chlorella spp.) provide oxygen through photosynthesis and release organic acids and chelating biomolecules, whereas heterotrophic or iron-cycling bacteria mediate Fe2+/Fe3+ transformations [9,43]. Here, iron mobilization is less governed by extreme acidity and more by localized pH and ORP microenvironments at the mineral surface [54]. Additional operational requirements include light availability, balanced nutrient supply (N, P), and control of oxygen gradients [9,54,55]. These differences in biological agents and physicochemical conditions directly influence dissolution kinetics, impurity mobilization, and downstream iron speciation, which are critical for subsequent material synthesis applications [11,56,57].
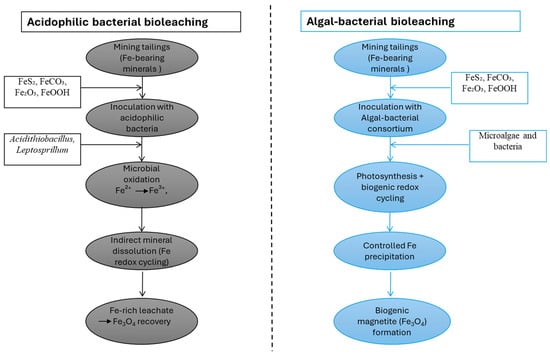
Figure 2.
Comparative schematic of acidophilic bacterial and algal–bacterial bioleaching pathways for iron recovery from mining tailings, highlighting redox transformations and biogenic magnetite (Fe3O4) formation.
In acidophilic bacterial bioleaching systems, iron dissolution is driven by microorganisms such as Acidithiobacillus spp. and Leptospirillum spp., which catalyse the oxidation of Fe2+ to Fe3+ under low-pH conditions (1.5–3.0), with high redox potentials (Eh +400 to +700 mV) and continuous oxygen supply. Ferric iron acts as the primary chemical oxidant of iron-bearing minerals. Continuous microbial regeneration of Fe3+ sustains mineral breakdown, while the acidic environment enhances metal solubility and suppresses abiotic precipitation, resulting in efficient iron mobilization. In terms of materials, bioleaching is not a one-step process solely required for extraction; the specific physicochemical conditions necessary for systematic iron oxide biomineralization must also be established [48]. The release of soluble Fe2+ and Fe3+, accompanied by local redox gradients and the presence of microbial metabolites, is favourable for nucleation of iron oxide nanoparticles [48,49]. In engineered and semi-natural systems, bioleached iron may be selectively converted to magnetite via biologically regulated Fe2+/Fe3+ redox coupling, facilitating efficient magnetic recovery and reuse. In general, the bioleaching of iron ores and tailings is a crucial upstream process that links mineral dissolution to the synthesis of sustainable, functional iron oxides. Its incorporation with redox-controlled biomineralization pathways provides a suitable alternative for transforming low-value mining residues as well as other iron-rich waste from other industries (steel making, alumina refining, etc.) into high-value magnetic materials [58,59].
3.1.2. Redox Transformation of Fe2+/Fe3+
The redox cycling of iron within iron-rich ores and mining tailings is essential for regulating iron solubility and influencing the formation of secondary iron oxides, including magnetite (Fe3O4) [46]. Microorganisms act as key catalysts in maintaining this dynamic equilibrium under environmentally relevant conditions. In acidic and oxic environments, iron-oxidizing microorganisms such as A. ferrooxidans and Leptospirillum spp. [46] facilitate the oxidation of Fe2+ to Fe3+ according to Equation (1). This biologically mediated reaction occurs at rates significantly faster than abiotic oxidation, particularly at low pH levels, where chemical oxidation of Fe2+ is kinetically hindered [49]. In an acidic or microaerophilic environment, the biologically mediated redox cycling of Fe2+/Fe3+ can be much more rapid than solely chemical oxidation, as both iron-oxidizing and iron-reducing microbes can couple electron transfer to the metabolic process and readily operate under low-oxygen conditions, where abiotic rates are slow [60]. On the contrary, under circumneutral pH and oxic conditions, the intrinsic chemical oxidation of Fe2+ by dissolved O2 becomes so fast that the measured bulk oxidation rates of biotic and abiotic systems are practically indistinguishable [60]. Under these conditions, microbial processes exert less of an impact on iron behaviour and more on iron speciation, favourable mineral phases, as well as nucleation growth pathways, such as the direction of the formation of certain Fe(III) (oxyhydroxides), stabilizing nanoscale phases, and mediating the Fe(II)–Fe(III) partitioning in mixed-valence minerals [61]. The resulting Fe3+ serves as a potent oxidant, promoting the dissolution of Fe-bearing minerals and thereby supporting the indirect bioleaching process [49].
Fe2+ + ¼O2 + H+ → Fe3+ + ½H2O
Dissimilatory iron-reducing bacteria, including species from Shewanella and Geobacter, utilize Fe3+ as a terminal electron acceptor, linking the reduction of ferric iron with the oxidation of organic substrates [47,62]. This reduction results in a decreased redox potential, alongside an increase in Fe2+ concentration, which is critical for magnet formation, as Fe3O4 requires mixed-valence Fe2+/Fe3+ within its inverse spinel structure [46,47]. Geologically speaking, the interplay between iron-oxidizing and -reducing processes during bioleaching generates spatial and temporal redox gradients that significantly influence iron speciation and precipitation behaviour [49]. Such gradients can be found extensively within heterogeneous tailings matrices, biofilms, and algal–bacterial consortia where local oxygen diffusion, microbial activity, and organic matter availability vary [46].
These microenvironments foster simultaneous oxidation of Fe2+ and reduction of Fe3+ while preparing conditions for redox-driven biomineralization [63]. The pH level notably affects transformations between Fe2+ and Fe3+; at lower pH values, ferric iron remains soluble and bioavailable, whereas higher pH levels encourage ferric hydrolysis, leading to precipitation as ferric oxyhydroxides [63]. Controlled increases in pH can also occur due to microbial metabolism or intentional system design to facilitate partial ferric reduction, along with co-precipitation with ferrous iron, which favours magnetite nucleation over fully oxidized forms such as hematite or goethite [64]. However, uncontrolled oxidation may result in the formation of non-magnetic iron oxides, which diminishes magnetic recovery efficiency [64].
In sustainable materials recovery efforts, this biologically regulated cycle of Fe2+/Fe3+ facilitates the elimination of passive precipitation, followed by the development of functional magnetite [48]. When integrated with microbial operations that control redox processes, iron extracted from tailings can be selectively transformed into magnetically recoverable Fe3O4 nanoparticles, thereby merging the geological cycling of iron with the creation of value from resources [46].
3.1.3. Nucleation and Growth of Iron Oxide Nanoparticles
An essential consideration in the biodependent formation of iron oxide nanoparticles is the interplay between iron speciation, redox conditions, pH, and microbial interfaces [43]. Following, e.g., bioleaching and cycling processes of Fe2+ and Fe3+, localized supersaturation of dissolved iron species can be observed, leading to the nucleation of secondary iron oxides [44,50]. Under such circumstances, biomineralization occurs under relatively mild conditions to selectively generate nanostructured iron oxides with distinctive magnetic properties.
Nucleation typically occurs when localized changes in pH or redox potential disrupt the solubility of iron complexes [44,48]. For bioleaching-derived solutions, ferric hydrolysis yields short-range ordered clusters of iron oxyhydroxide, which can serve as precursors to more crystalline structures [48]. However, ferrous iron also supports redox coupling reactions favourable for the formation of mixed-valence iron oxides. Notably, magnetite (Fe3O4) nucleates under the coexistence of Fe2+ and Fe3+, which accounts for an inverse spinel structure, having mixed oxidation states [9]. Microbial cells, EPSs, and algal surfaces are essential heterogeneous nucleation sites.
The adhering functional groups (carboxyls, hydroxyls, phosphoryls, amines, etc.) can be observed to attach strongly to those of iron ions, increasing local supersaturation and lowering the energy barrier for nucleation [43]. These biochemically generated interfaces not only facilitate particle generation but also influence crystal orientation, size distribution, and aggregation behaviour [10]. On the other hand, biogenic nucleation can usually produce nanoparticles with limited spacing due to spatial confinement and biological matrices that guide kinetic regulation, unlike in abiotic systems [65].
The growth process of iron oxide nanoparticles may be initiated with ion-by-ion addition, primary nanocrystals aggregated, or metamorphosis from metastable precursor phases [9]. Ferrihydrite is often an initial material, which can be transformed into magnetite in the presence of Fe2+ through either solid-state transformation or dissolution–reprecipitation pathways. These transformations are significantly influenced by redox gradients and the availability of electron donors/acceptors in microbial communities [11,66]. Optimal growth conditions are also more conducive to magnetite formation than hematite or goethite, which are less favourable for magnetic separation [9].
Not only do biological activities contribute to controlling particle size and shape, but they are also crucial in this process. Adsorbed biomolecules can prevent excessive crystal growth by stabilizing nanoparticles within the superparamagnetic or single-domain size range [43]. This type of control over size can be particularly beneficial for magnetic recovery processes, as it enhances magnetic sensitivity while also preventing irreversible aggregation of the particles [9,43].
Biogenic magnetite often exhibits better surface reactivity than chemically synthesized compounds due to its organic coating and high defect density [9,43]. Taken together, the biologically driven mechanism for the nucleation and growth of iron oxide nanoparticles provides fundamental linkages between iron mobilization and material recovery [44,65]. Iron obtained from low-grade ores or tailings can be readily converted into magnetite nanoparticles with variable properties when supported by microbial redox activity, in addition to biological templating conditions [7,44,66]. This technique offers a sustainable approach to transforming mining residues into useful magnetic materials for separation, recycling, and environmental applications.
3.2. Influence of Biomolecules as Natural Capping Agents
Microorganisms and microalgae produce biomolecules that play a major role in controlling both the nanoscale nucleation and growth of biogenic iron oxide nanoparticles, as well as their adhesion properties [67,68,69,70]. In systems developed by bioleaching, extracellular polymeric substances (EPSs) (in addition to proteins, polysaccharides, lipids, and low-molecular-weight organic acids) are incorporated into bioplate interfaces with iron species [45,71,72]. These components act as natural capping agents, influencing their size, morphology, surface chemistry, and aggregation abilities.
Biogenic iron oxides exhibit distinct biomolecular interactions that distinguish them from their chemically synthesized counterparts and are crucial to their performance in magnetic recovery methods [9]. In biomineralization, functional groups such as carboxylate, hydroxyl, phosphate, and amine moieties bind Fe2+ and Fe3+ via electrostatic attraction, complexation, and coordination bonding, among other mechanisms [66]. This binding causes iron to localize at biological interfaces, increasing effective supersaturation while inhibiting uncontrolled crystal growth [9,10].
As a result, iron oxide nanoparticles coated with biomolecules have narrow size distributions and improved colloidal stability [71]. Magnetite (Fe3O4) requires precise control in this domain as its magnetic properties are highly related to particle size [73]. Superparamagnetic or single-domain behaviour is preferred for magnetic separation. In addition, proteins and peptides can selectively adsorb on specific crystallographic planes, thereby affecting crystal habit and phase selectivity as well. Polysaccharides and glycoproteins abundant in microbial EPSs and algal cell walls form hydrated matrices that provide physical support for growing nanoparticles, preventing aggregation, and allowing dispersion [45,72].
Additionally, organic acids—citric acid, oxalic acid, and gluconic acid—commonly generated in microbial metabolism can chelate iron ions, control local pH, and participate in redox buffering that would indirectly affect the Fe2+/Fe3+ ratio during magnetite development [18,63]. The capping effect of biomolecules also changes the surface charge and interfacial characteristics of biogenic iron oxides [71]. The organic coatings lead to steric hindrance and electrostatic repulsion between particles, thereby minimizing irreversible agglomeration during the synthesis and recovery process [72]. This increased stability is beneficial in multiple cycles of magnetic separation, where magnetic responsiveness and active charge can be maintained without issues related to sedimentation or surface passivation [72].
Furthermore, the organic corona provides additional functionalities such as pollutant receptor potential or compatibility with biological systems [74]. At the mechanistic level, biomolecules not only act as passive stabilizers but also play an active role in the redox-induced transformations of minerals [75]. Reducing functional groups in EPSs and cellular metabolites may stimulate partial ferric reduction, establishing an environment of mixed valence compatibility for magnetite formation [45]. On the negative side, oversaturation of organic materials could lead to poor crystallization if the binding strength with iron is too strong; therefore, the need for an interaction between the biomolecule and the ion must be preserved in engineered systems [71]. By exploiting naturally occurring biomolecules used as capping agents, mining tailings valorisation eliminates the need for synthetic surfactants or chemical stabilizers [71,72].
The new technique enhances environmental sustainability in biogenic magnetite production by enabling the synthesis of small, readily controllable nanoparticles with magnetic recovery properties. The combination of bioleaching methods with redox and biomolecular capping procedures can transform low-value iron-rich residues into high-performance magnetic materials appropriate for circular resource applications [46,49].
4. Biomagnetic Recovery
This biomagnetic recovery exploits the natural magnetic activity of biogenic iron oxides, most notably magnetite (Fe3O4), resulting in highly efficient separation and recycling of these products from complex aqueous and solid matrices [76]. Biogenic magnetite can be selectively recovered using external magnetic fields following bioleaching, redox-controlled transformation, and biomolecule-mediated nanoparticle stabilization [7]. This presents a low-energy, reagent-free solid–liquid separation process, suitable for iron-rich mining tailings and process waters [7]. The efficiency of biomagnetic recovery depends heavily on the magnetic phase, particle size, and surface features of the biominerals obtained [77].
As it is mixed-valence inverse spinel-based, magnetite exhibits high saturation magnetisation as compared with other iron oxides, including hematite or goethite. Biogenic magnetite nanoparticles produced at regulated Fe2+/Fe3+ ratios are typically superparamagnetic in character and of single-domain size, which enables a rapid response to magnetic gradients while curbing irreversible agglomerations [9,78]. Such features allow excellent capture even at low particle counts. Biomolecular capping also improves the performance of magnetic recovery [32,78]. Simultaneously, such coatings provide functional interfaces to facilitate attachment to target particles, including mineral fines, organic matter, and contaminants [78].
For tailings systems, this dual mechanism enables flocculation and magnetic harvesting of difficult-to-separate fractions. From a process perspective, biomagnetic recovery can be achieved using low-intensity permanent magnets or high-gradient magnetic separators, depending on the size of the particles and the complexity of the system [77]. By contrast to other flocculants or filtration methods, magnetic separation is fast, produces very limited secondary waste, and enables further cycles [77,79]. Notably, biogenic magnetite is frequently regenerable and reusable, with minimal degradation of magnetic performance, thereby enhancing process economics and sustainability [9,39]. Biomagnetic recovery in tailings valorisation can change the focus from waste management to resource recovery. Iron mobilised in bioleaching is more than just extracted; it is converted into a functional magnetic material, which is also useful for harvesting, recycling, or utilisation in other downstream processes, such as pollutant removal, nutrient delivery, or catalysis [48,50].
Additionally, the selective characteristics of magnetic separation may limit the co-recovery of non-target phases, which may contribute to product purity [80]. This can be seen in ore beneficiation, where low-intensity magnetic separation can recover magnetite while leaving behind non-magnetic minerals such as quartz and silicates [81], or in water treatment, where functionalized magnetite NPs bind to arsenate or phosphate but not to Ca2+, Cl−, or Na+, because these ions are non-magnetic [82]. On a broader scale, biomagnetic recovery is a result of biologically mediated pathways to iron transformation. Innovative microbial iron cycling [83], controlled nanoparticle synthesis, and magnetic separation are among the strategies explored in this study, which offer a sustainable [84] and environmentally friendly option [85] for the value recovery of iron-rich residues through their incorporation into metal-rich, biobased processes [79]. Its use in tailings management focuses on enabling biobased technologies in circular mineral processing and sustainable materials engineering.
4.1. Magnetic Flocculation and Dewatering of Microalgae
Magnetic flocculation is widely recognized as an emerging, potent, and environmentally friendly approach to harvest and dewater microalgae, addressing a significant economic challenge in algal bioprocessing [86,87]. Conventional harvesting and dewatering methods, such as centrifugation, filtration, and chemical flocculation, are energy-intensive, costly, and environmentally detrimental [88,89,90,91,92,93,94]. However, magnetic flocculation utilizes magnetically responsive particles (Fe3O4 nanoparticles) to rapidly aggregate and separate microalgal microorganisms under mild conditions [95,96].
Magnetic flocculation is primarily a functional process driven by electrostatic forces, surface adsorption reactions, and magnetic bridging. Fe3O4 nanoparticles are easily attracted to the negatively charged surfaces of microalgal cells via electrostatic attraction and hydrogen bonding [96]. Experienced by this external magnetic field, these cells and Fe3O4 will accumulate together as dense magnetic flocs, which can easily settle under the influence of a magnetic field [97,98]. Enhancing the surface properties of Fe3O4, particularly through biogenic coatings or extracellular polymeric substances (EPSs), significantly improves flocculation efficiency by increasing particle dispersion and binding affinity, thereby reducing the required nanoparticle dosage [88,96].
There are also advantages to magnetic dewatering through compact floc formation, which reduces water retention and significantly enhances solid–liquid separation. High harvesting efficiency, particularly in short contact times with low doses, can be achieved in systems based on Fe3O4, which, compared to conventional flocculants, yields high harvesting efficiency and minimizes the risk of chemical contamination in biomass [89,95,96]. Following a few cycles of recycling, any remaining magnetite may be recycled for soil remediation or integrated into Fe3O4 nanofertilizers, as part of a circular economy strategy [96]. The Fe3O4 nanoparticles for magnetic flocculation and dewatering serve as a scalable, energy-efficient solution not only for microalgae harvesting but also for promoting environmental sustainability. This approach has considerable potential for integration within closed-loop bioresource recovery frameworks and sustainable agriculture practices [96].
4.2. Biomagnetic Recovery of Metals in Wastewater and Mine Tailings
One such sustainable and rapid method for recovering metals from metal-containing wastewater and mine tailings is biomagnetic recovery. Traditional physicochemical treatment approaches, including precipitation methods such as chemical treatment, ion exchange, and membrane filtration, typically lead to high reagent costs, sludge generation, and low metal selectivity, and are often not very selective either [79]. By contrast, in biomagnetic systems, the synergistic relation between biological materials and magnetically responsive particles, in particular biogenic Fe3O4 particles, including the nanoparticle, is exploited, providing them with the ability to selectively capture, as well as recover, dissolved metals (copper, lead, cadmium, chromium) under mild operation, even in the presence of relatively low operating conditions (pH, temperature, concentration) [17].
The biomagnetic extraction of metals is a novel technique that provides a sustainable and effective means of processing metal-rich wastewater and mining materials, thereby producing clean, productive resources. Conventional methods for physical or chemical treatment, such as chemical precipitation, ion exchange, or membrane filtration processes, can also be limited by the heavy use of reagents, resulting in excessive sludge generation and limited selectivity for individual metals [1,18]. A key difference from conventional systems is that biomagnetic methods rely on the interaction between biological agents (such as microbes) and magnetic reactive materials [1,9,99].
The biogenic Fe3O4 nanoparticles serve as a reference, with the characteristic to harvest and recover soluble metallic materials under mild conditions.
Microorganisms, including microalgae, play a central role in this schema because they are involved in biosorption, bioaccumulation, and biomineralization processes. Characteristics of functional groups on cellular and EPSs, such as carboxyls, hydroxyls, and phosphates, binding sites showing strong affinity for the metal ions Cd2+ (cadmium), Pb2+ (lead), Cu2+ (copper), and Zn2+ (zinc) were found [9,45]. Interest also lies in the magnetic effects of these structures when combined with biogenic Fe3O4 nanoparticles, such as the biotechnology of bioactive surface materials, which generates an attractive magnetic composite that not only improves the uptake efficiency but also allows for quick removal by external magnetic fields [18,98]. It is interesting to note that, in the case of mine tailings, biological redox reactions also contribute, as soluble metallic species can undergo changes into modes that may have diminished mobility or even become mechanically immobilized by magnetic bonds [10,46,77].
Magnetic separation enables the recovery of complexes rich in metal-loaded biomaterials, thereby avoiding secondary waste and providing a means of recycling or immobilizing the recovered materials [77,100]. Importantly, Fe2O3 nanomaterials can go through several cycles before being regarded as depleted [100]. In addition, partially used resources could have new uses (soil remediating projects and positions in the production of nanofertilizer), serving beneficially for circular economic practices [100,101]. Biomagnetic recovery is noted as an energy-saving method with scalability capacity and is environmentally friendly due to its management of pollution problems, including heavy metal contamination from heavy residues left in industrial effluents and remnants from mineral deposits, which have promising prospects for environmental improvement [77].
4.3. Biomedical and Catalytic Uses
Fe3O4 nanoparticles possess unique magnetic, surface, and redox properties that stimulate interest in medicinal and catalytic areas as well as environmental and agricultural applications [102]. Magnetite nanoparticles are extensively investigated in biomedicine for use in MRI contrast enhancement, targeted drug delivery, magnetic hyperthermia, and biosensing [39]. Their superparamagnetic properties enable remote manipulation and targeted localization via external magnetic fields, while surface functionalization with biocompatible polymers or biomolecules enhances stability, prolongs circulation time, and improves cellular compatibility [39]. Biogenic Fe3O4 exhibits advantages such as green production, reduced cytotoxicity, and intrinsic surface biofunctionalization.
Fe3O4 nanoparticles are remarkable heterogeneous catalysts and catalytic supports in redox-driven reactions, including the degradation of organic pollutants in Fenton-like processes [102]. The mixed-valence Fe2+/Fe3+ structure of this species is advantageous for electron transport, as well as improving catalytic activity under mild conditions [102]. Magnetic properties promote catalyst recovery and reuse, boosting process sustainability. The recent application of these biomedical and catalytic applications demonstrates the high adaptability of Fe3O4 nanoparticles, with numerous applications in environmental, industrial, and medicinal fields [33,39].
5. Cost and Environmental Implications
The economic and environmental performance of Fe3O4 nanoparticles is strongly determined by the specific synthesis route employed [103]. Traditional chemical synthesis methods (e.g., co-precipitation, solvothermal, etc.) typically utilize purified iron salts, controlled pH adjustment, and energetically demanding processing, which increases production costs and burdens the environment [104]. The production costs of chemically synthesized Fe3O4 are typically reported to range between USD 10 and USD 50 kg−1, depending on the scale and purity requirements, with associated greenhouse gas emissions of approximately 3–8 kg CO2-eq kg−1 attributed to chemical inputs and energy consumption [31,103,104].
Conversely, biogenic Fe3O4 from microorganisms or microalgae, especially when derived from mining tailings or waste streams, offers both cost and environmental benefits. At the pilot level, lower production costs can be achieved by utilizing low-cost or waste iron sources, operating under ambient conditions, and employing limited chemical additions, resulting in approximately USD 2–10 kg−1 [105]. Life-cycle indicators indicate much less environmental impact, with estimated emissions of <1–3 kg CO2-eq kg−1, with reduced acidification and eutrophication potentials [30]. Biogenic pathways have a longer processing time and are subject to biological variability; however, these challenges are often mitigated by increased sustainability, intrinsic surface functionalization, and alignment with principles of the circular economy [9]. Table 4 highlights some benefits and challenges of laboratory, pilot, and large-scale biogenic processes as reported by [27,28,29]. Biogenic Fe3O4 can be proposed as the likely low-impact source for larger environmental and agricultural applications; this is especially well suited to the use of resources that must be recovered and waste valorised in an environmentally sustainable manner [9,30,104,105].

Table 4.
Comparative benefits and challenges of biogenic and chemical synthesis.
6. Current Limitations and Research Gaps and Future Outlook
Fe3O4-based materials, however, exhibit promising multi-functionality; nevertheless, several issues have been reported that affect their large-scale use. Biological diversity and process conditions have the potential to modify particle size, surface chemistry, and magnetic properties, resulting in a complex series of physicochemical characteristics [40,106]. This is because synthesis routes (e.g., biogenic Fe3O4) often yield differential physicochemical properties [9,106]. This variability complicates the reproducibility, standardization, comparability, and use of similar studies. Furthermore, the long-term environmental fate, transformation, and ecotoxicity of Fe3O4 nanoparticles in soil and aquatic systems, particularly during repeated application scenarios, remain unknown.
Technically, scalability and integration into existing processes remain crucial shortcomings. Although laboratory/pilot-scale studies indicate high efficiency in metal recovery, microalgae harvesting, and soil remediation, the effectiveness of these approaches should also be evaluated in realistic field conditions or even in complex multi-contaminant environments [9,39,96]. Additionally, economic assessments are inadequate, with insufficient life-cycle and techno-economic analyses to compare the cost–benefits of these technologies with traditional ones. Additionally, the regulatory environment surrounding nanomaterials in agriculture and environmental remediation is currently under development, which poses challenges for commercialization and public acceptance [107]. A study by [108] provides a detailed explanation of sustainable biomining of metals using microorganisms from mining tailings, which could be utilized in the biogenic synthesis of iron oxides from mining tailings.
Further research should focus on designing sustainable Fe3O4 materials with a specified size, surface functionality, and degradation profile to mitigate environmental threats. Long-term field studies, in conjunction with consistent ecotoxicity and life-cycle assessments, are crucial for confirming sustainability claims. Innovations in bioprocess design, such as utilizing waste iron sources and implementing process automation, may enhance scalability and reduce costs, respectively. Lastly, the inclusion of Fe3O4-based systems in circular economy models, which connect the treatment of wastewater, harvesting of microalgae, remediation of soil, and recycling of nutrients, is a useful strategy to maximize resource utilization and environmental benefits.
7. Conclusions
In this review, we highlight the opportunity to establish Fe3O4-based materials, particularly biogenic magnetite, as alternative, multi-use platforms for sustainable environmental, agricultural, and resource recovery applications. With improvements such as these for iron oxide biosynthesis, magnetic flocculation, biomagnetic metal recovery, and nanoenabled soil remediation, Fe3O4 is presented as a suitable material to address most pressing problems, including heavy metal contamination, inefficient nutrient management, costly microalgae harvesting, and waste generation resulting from mining and wastewater treatment. Mixed valence redox, strong surface reactivity, and magnetic responsiveness are all characteristic properties of magnetite that significantly favour its versatility and its promising potential in multiple functional domains.
A key point of this review is the unequivocal benefits of the biogenic form of Fe3O4 over the conventional preparation form. Biobased routes have lower manufacturing and emission costs, are more environmentally friendly, are intrinsically functional at the surface level, and are more compatible with biological systems. Biogenically sourced Fe3O4, based on waste streams including mining tailings, enhances the circular economy concept by converting low-value residues into functional elements for nutrient delivery, contaminant immobilization, and magnetic separation. The reuse of magnetite across multiple cycles and the repurposing of spent materials for soil remediation or nanofertilizers are attractive closed-loop solutions.
However, the review also highlights some fundamental shortcomings, including variability in biogenic material properties, limited long-term field data, shortfalls in life-cycle and techno-economic evaluations, and unaddressed regulatory and safety issues. Solving these problems involves well-coordinated and cross-functional initiatives in materials science, bioprocess engineering, agronomy, environmental toxicology, and policy. Further research is needed to establish safe-by-design Fe3O4 systems and standardised evaluation protocols, as well as to conduct large-scale development in a reproducible environment. In short, Fe3O4-driven technologies, particularly those based on biogenic and waste-derived pathways, have real potential to provide sustainable, resilient, and circular solutions across environmental remediation and agro-systems.
Author Contributions
G.A.-D.: Conceptualization, investigation, writing—original draft, writing—review and editing. H.E.: Conceptualization, investigation, supervision, writing—review and editing. E.K.T.: Conceptualization, investigation, supervision, writing—review and editing. P.A.B.: Conceptualization, funding acquisition, supervision, writing—review and editing. N.R.M.: Conceptualization, funding acquisition, supervision, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Murdoch University.
Data Availability Statement
Not applicable.
Acknowledgments
We extend our appreciation to Murdoch University and the Algae Innovation Hub for their assistance. We are grateful to Murdoch University and the Graduate Research Office (GRO) for their financial support.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Giri, S.; Das, N.; Pradhan, G. Synthesis and characterization of magnetite nanoparticles using waste iron ore tailings for adsorptive removal of dyes from aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2011, 389, 43–49. [Google Scholar] [CrossRef]
- Li, Z.; Wang, J.; She, Z.; Gu, J.; Lu, H.; Wang, S.; He, X.; Yue, Z. Tailings particle size effects on pollution and ecological remediation: A case study of an iron tailings reservoir. J. Hazard. Mater. 2024, 476, 135024. [Google Scholar] [CrossRef]
- Moran-Palacios, H.; Ortega-Fernandez, F.; Lopez-Castaño, R.; Alvarez-Cabal, J.V. The potential of iron ore tailings as secondary deposits of rare earths. Appl. Sci. 2019, 9, 2913. [Google Scholar] [CrossRef]
- Coherent Market Insights. Magnetite Nanoparticles Market Analysis and Forecast: 2025–2032. U.S. 2025. Available online: https://www.coherentmarketinsights.com/market-insight/magnetite-nanoparticles-market-5404?utm_source=chatgpt.com (accessed on 22 August 2025).
- Yousif, N.A.; Al-Jawad, S.M.; Taha, A.A.; Stamatis, H. A review of structure, properties, and chemical synthesis of magnetite nanoparticles. J. Appl. Sci. Nanotechnol. 2023, 3, 18–31. [Google Scholar] [CrossRef]
- Niculescu, A.-G.; Chircov, C.; Grumezescu, A.M. Magnetite nanoparticles: Synthesis methods–A comparative review. Methods 2022, 199, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Pan, Q.; Cao, Y.; Gao, P.; Sun, Y.; Han, Y. Preparation of superior magnetite concentrate from waste tailings with clean energy: Semi-industrial utilization, economic profits, and environmental benefits. Process Saf. Environ. Prot. 2024, 188, 1049–1057. [Google Scholar] [CrossRef]
- Jiang, Z.; Shi, M.; Shi, L. Degradation of organic contaminants and steel corrosion by the dissimilatory metal-reducing microorganisms Shewanella and Geobacter spp. Int. Biodeterior. Biodegrad. 2020, 147, 104842. [Google Scholar] [CrossRef]
- Ennaceri, H.; Mkpuma, K.M.; Amo-Duodu, G.; Menkiti, M.; Ishika, T.; Vadiveloo, A.; Moheimani, N.R. Microalgae as sustainable bio-factories for nanoparticle biosynthesis: Progress and challenges. Bioresour. Technol. Rep. 2025, 30, 102089. [Google Scholar] [CrossRef]
- Maass, D.; Valério, A.; Lourenço, L.A.; de Oliveira, D.; Hotza, D. Biosynthesis of iron oxide nanoparticles from mineral coal tailings in a stirred tank reactor. Hydrometallurgy 2019, 184, 199–205. [Google Scholar] [CrossRef]
- Fani, M.; Ghandehari, F.; Rezaee, M. Biosynthesis of iron oxide nanoparticles by cytoplasmic extract of bacteria lactobacillus fermentum. J. Med. Chem. Sci. 2018, 1, 28–30. [Google Scholar]
- Sundaram, P.A.; Augustine, R.; Kannan, M. Extracellular biosynthesis of iron oxide nanoparticles by Bacillus subtilis strains isolated from rhizosphere soil. Biotechnol. Bioprocess Eng. 2012, 17, 835–840. [Google Scholar] [CrossRef]
- Carmignano, O.; Vieira, S.; Teixeira, A.P.; Lameiras, F.; Brandão, P.R.; Lago, R. Iron ore tailings: Characterization and applications. J. Braz. Chem. Soc. 2021, 32, 1895–1911. [Google Scholar] [CrossRef]
- Coelho, P.; Camacho, D. Camacho, The experimental characterization of iron ore tailings from a geotechnical perspective. Appl. Sci. 2024, 14, 5033. [Google Scholar] [CrossRef]
- Chen, M.; Liu, Q.; Gao, S.-S.; Young, A.E.; Jacobsen, S.E.; Tang, Y. Genome mining and biosynthesis of a polyketide from a biofertilizer fungus that can facilitate reductive iron assimilation in plant. Proc. Natl. Acad. Sci. USA 2019, 116, 5499–5504. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Chen, P.; Yu, J.; Liu, J.; Ye, J.; Zhou, S. Recyclable magnetite-enhanced electromethanogenesis for biomethane production from wastewater. Water Res. 2019, 166, 115095. [Google Scholar] [CrossRef] [PubMed]
- Usman, M.; Byrne, J.M.; Chaudhary, A.; Orsetti, S.; Hanna, K.; Ruby, C.; Kappler, A.; Haderlein, S.B. Magnetite and green rust: Synthesis, properties, and environmental applications of mixed-valent iron minerals. Chem. Rev. 2018, 118, 3251–3304. [Google Scholar] [CrossRef]
- Usman, U.A.; Yusoff, I.; Raoov, M.; Hodgkinson, J. The economic potential of the African iron-ore tailings: Synthesis of magnetite for the removal of trace metals in groundwater—A review. Environ. Earth Sci. 2019, 78, 615. [Google Scholar] [CrossRef]
- Bolm, C.; Legros, J.; Le Paih, J.; Zani, L. Iron-catalyzed reactions in organic synthesis. Chem. Rev. 2004, 104, 6217–6254. [Google Scholar] [CrossRef]
- Gammons, C.H.; Allin, N.C. Stability of aqueous Fe (III) chloride complexes and the solubility of hematite between 150 and 300 C. Geochim. Cosmochim. Acta 2022, 330, 148–164. [Google Scholar] [CrossRef]
- Aredes, A.C.; Oliveira, C.D.; Heleno, I.G.; Campanati, M.E.; Sousa, M.R.; Marcelo, N.D.; Almeida, J.R.; Martins, C.D. Iron Ore Tailings: Generation, impacts, applications and challenges. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2025; Volume 1536, p. 012023. [Google Scholar]
- Ferreira, A.D.; Corveloni, H.F.; Boim, A.F.; Queiroz, H.M.; Souza, T.P.; Otero, X.L.; Bernardino, A.F.; Ferreira, T.O. From tailings to tables: Risk assessment of potentially toxic elements in edible crops cultivated in mine tailing impacted soils. Environ. Geochem. Health 2025, 47, 473. [Google Scholar] [CrossRef]
- Malik, L.; Hedrich, S. Ferric iron reduction in extreme acidophiles. Front. Microbiol. 2022, 12, 818414. [Google Scholar] [CrossRef]
- Yi, Q.; Wu, S.; Southam, G.; Robertson, L.; You, F.; Liu, Y.; Wang, S.; Sara, N.; Webb, R.; Wykes, J.; et al. Acidophilic iron-and sulfur-oxidizing bacteria, Acidithiobacillus ferrooxidans, drives alkaline pH neutralization and mineral weathering in Fe ore tailings. Environ. Sci. Technol. 2021, 55, 8020–8034. [Google Scholar] [CrossRef]
- Wang, C.; Jing, J.; Qi, Y.; Zhou, Y.; Zhang, K.; Zheng, Y.; Zhai, Y.; Liu, F. Basic characteristics and environmental impact of iron ore tailings. Front. Earth Sci. 2023, 11, 1181984. [Google Scholar] [CrossRef]
- Mishra, A.; Ahmad, R.; Sardar, M. Biosynthesized iron oxide nanoparticles mimicking peroxidase activity: Application for biocatalysis and biosensing. J. Nanoeng. Nanomanuf. 2015, 5, 37–42. [Google Scholar] [CrossRef]
- Augusto, P.A.; Castelo-Grande, T.; Vargas, D.; Pascual, A.; Hernández, L.; Estevez, A.M.; Barbosa, D. Upscale Design, Process Development, and Economic Analysis of Industrial Plants for Nanomagnetic Particle Production for Environmental and Biomedical Use. Materials 2020, 13, 2477. [Google Scholar] [CrossRef]
- Simeonidis, K.; Morales, M.d.P.; Damartzis, T.; Maniotis, N.; Veintemillas-Verdaguer, S. Toward More Sustainable Magnetic Nanoparticle Synthesis Based on Microwave-Assisted Continuous-Flow Processes. Ind. Eng. Chem. Res. 2024, 63, 20651–20660. [Google Scholar] [CrossRef]
- Fuentes, O.P.; Trujillo, D.M.; Sánchez, M.E.; Abrego-Perez, A.L.; Osma, J.F.; Cruz, J.C. Embracing Sustainability in the Industry: A Study of Environmental, Economic, and Exergetic Performances in Large-Scale Production of Magnetite Nanoparticles. ACS Sustain. Chem. Eng. 2024, 12, 760–772. [Google Scholar] [CrossRef]
- Marimón-Bolívar, W.; González, E.E. Green synthesis with enhanced magnetization and life cycle assessment of Fe3O4 nanoparticles. Environ. Nanotechnol. Monit. Manag. 2018, 9, 58–66. [Google Scholar]
- Harman, A.E.; Nandiyanto, A.B.D. Engineering and Economic Evaluation of Production of Fe3O4 Nanoparticles. J. Tek. Kim. Dan Lingkung. 2020, 4, 68–80. [Google Scholar] [CrossRef]
- Pirsaheb, M.; Gholami, T.; Seifi, H.; Dawi, E.A.; Said, E.A.; Hamoody, A.H.M.; Altimari, U.S.; Salavati-Niasari, M. Green synthesis of nanomaterials by using plant extracts as reducing and capping agents. Environ. Sci. Pollut. Res. 2024, 31, 24768–24787. [Google Scholar] [CrossRef] [PubMed]
- Bharathi, D.; Rajamani, R.; Sibuh, B.Z.; Pandit, S.; Agrawal, S.; Mishra, N.; Sahni, M.; Thakur, V.K.; Gupta, P.K. Biogenic preparation, characterization, and biomedical applications of chitosan functionalized iron oxide nanocomposite. J. Compos. Sci. 2022, 6, 120. [Google Scholar] [CrossRef]
- Runge, E.; Mansor, M.; Chiu, T.H.; Shuster, J.; Fischer, S.; Kappler, A.; Duda, J.P. Hydrothermal sulfidation of biogenic magnetite produces framboid-like pyrite. Commun. Earth Environ. 2024, 5, 252. [Google Scholar] [CrossRef]
- Kahani, S.A.; Yagini, Z. A Comparison between Chemical Synthesis Magnetite Nanoparticles and Biosynthesis Magnetite. Bioinorg. Chem. Appl. 2014, 2014, 384984. (In English) [Google Scholar] [CrossRef]
- Nguyen, M.D.; Tran, H.-V.; Xu, S.; Lee, T.R. Fe3O4 Nanoparticles: Structures, Synthesis, Magnetic Properties, Surface Functionalization, and Emerging Applications. Appl. Sci. 2021, 11, 11301. (In English) [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Li, J.; Ghosen, J.; Zeng, M.; Stebbins, J.; Prasad, P.N.; Swihart, M.T. Standardizing Size- and Shape-Controlled Synthesis of Monodisperse Magnetite (Fe3O4) Nanocrystals by Identifying and Exploiting Effects of Organic Impurities. ACS Nano 2017, 11, 6370–6381. [Google Scholar] [CrossRef]
- Ghazanfari, M.R.; Kashefi, M.; Shams, S.F.; Jaafari, M.R. Perspective of Fe3O4 Nanoparticles Role in Biomedical Applications. Biochem. Res. Int. 2016, 2016, 7840161. (In English) [Google Scholar] [CrossRef]
- Salehirozveh, M.; Dehghani, P.; Mijakovic, I. Synthesis, functionalization, and biomedical applications of iron oxide nanoparticles (IONPs). J. Funct. Biomater. 2024, 15, 340. [Google Scholar] [CrossRef]
- Yadav, S.; Nadar, T.; Lakkakula, J.; Wagh, N.S. Biogenic synthesis of nanomaterials: Bioactive compounds as reducing, and capping agents. In Biogenic Nanomaterials for Environmental Sustainability: Principles, Practices, and Opportunities; Springer: Berlin/Heidelberg, Germany, 2024; pp. 147–188. [Google Scholar]
- Bharde, A.; Rautaray, D.; Bansal, V.; Ahmad, A.; Sarkar, I.; Yusuf, S.M.; Sanyal, M.; Sastry, M. Extracellular biosynthesis of magnetite using fungi. Small 2006, 2, 135–141. [Google Scholar] [CrossRef]
- Dauce, P.D.; de Castro, G.B.; Lima, M.M.F.; Lima, R.M.F. Characterisation and magnetic concentration of an iron ore tailings. J. Mater. Res. Technol. 2019, 8, 1052–1059. [Google Scholar] [CrossRef]
- Palaniyandi, T.; Baskar, G.; V, B.; Viswanathan, S.; Abdul Wahab, M.R.; Govindaraj, M.K.; Sivaji, A.; Rajendran, B.K.; Kaliamoorthy, S. Biosynthesis of iron nanoparticles using brown algae Spatoglossum asperum and its antioxidant and anticancer activities through in vitro and in silico studies. Part. Sci. Technol. 2023, 41, 916–929. [Google Scholar] [CrossRef]
- Brar, K.K.; Magdouli, S.; Othmani, A.; Ghanei, J.; Narisetty, V.; Sindhu, R.; Binod, P.; Pugazhendhi, A.; Awasthi, M.K.; Pandey, A. Green route for recycling of low-cost waste resources for the biosynthesis of nanoparticles (NPs) and nanomaterials (NMs)-A review. Environ. Res. 2022, 207, 112202. [Google Scholar] [CrossRef]
- Zhou, Y.; Cui, X.; Wu, B.; Wang, Z.; Liu, Y.; Ren, T.; Xia, S.; Rittmann, B.E. Microalgal extracellular polymeric substances (EPS) and their roles in cultivation, biomass harvesting, and bioproducts extraction. Bioresour. Technol. 2024, 406, 131054. [Google Scholar] [CrossRef]
- Liu, H.; Liu, T.; Chen, S.; Liu, X.; Li, N.; Huang, T.; Ma, B.; Liu, X.; Pan, S.; Zhang, H. Biogeochemical cycles of iron: Processes, mechanisms, and environmental implications. Sci. Total Environ. 2024, 951, 175722. [Google Scholar] [CrossRef]
- Verma, M.; Singh, V.; Mishra, V. Moving towards the enhancement of extracellular electron transfer in electrogens. World J. Microbiol. Biotechnol. 2023, 39, 130. [Google Scholar] [CrossRef]
- Borkar, P.K.; Sharma, N.; Boafo, K.; Amponsah-Berko, D.; Eisele, T. Reductive Bioleaching of Goethite-Rich and Hematite-Rich Iron Tailings by Anaerobic Organisms. Min. Metall. Explor. 2025, 42, 3379–3389. [Google Scholar] [CrossRef]
- Tonietti, L.; Esposito, M.; Cascone, M.; Barosa, B.; Fiscale, S.; Muscari Tomajoli, M.T.; Sbaffi, T.; Santomartino, R.; Covone, G.; Cordone, A.; et al. Unveiling the bioleaching versatility of Acidithiobacillus ferrooxidans. Microorganisms 2024, 12, 2407. [Google Scholar] [CrossRef] [PubMed]
- Eze, C.; Ugwoju, N.; Nnabuife, O.; Enemuor, S.; Okechukwu, V.; Ezeh, C. Bioleaching and beneficiation of Agbaja iron ore using Providencia vermicola KUBT-1 under varying process conditions. Metallurgy 2025, 125, 711–717. [Google Scholar] [CrossRef]
- Ramirez-Torres, L.A.; Nuñez-Ramirez, D.M.; Medina-Torres, L.; Manero, O. Study of the impurity dissolution kinetics, rheological characterization, and hydrodynamic aspects during the bioleaching of iron ore pulp in a bioreactor. Korea-Aust. Rheol. J. 2024, 37, 29–42. [Google Scholar] [CrossRef]
- Maltsev, Y.; Maltseva, S.; Maltseva, I. Diversity of cyanobacteria and algae during primary succession in iron ore tailing dumps. Microb. Ecol. 2022, 83, 408–423. [Google Scholar] [CrossRef]
- Figueroa-Estrada, J.; Aguilar-López, R.; Rodríguez-Vázquez, R.; Neria-González, M. Bioleaching for the extraction of metals from sulfide ores using a new chemolithoautotrophic bacterium. Hydrometallurgy 2020, 197, 105445. [Google Scholar] [CrossRef]
- Chan, S.S.; Low, S.S.; Chew, K.W.; Ling, Y.C.; Rinklebe, J.; Juan, J.C.; Ng, E.P.; Show, P.L. Prospects and environmental sustainability of phyconanotechnology: A review on algae-mediated metal nanoparticles synthesis and mechanism. Environ. Res. 2022, 212, 113140. [Google Scholar] [CrossRef] [PubMed]
- Brayner, R.; Coradin, T.; Beaunier, P.; Grenèche, J.M.; Djediat, C.; Yéprémian, C.; Couté, A.; Fiévet, F. Intracellular biosynthesis of superparamagnetic 2-lines ferri-hydrite nanoparticles using Euglena gracilis microalgae. Colloids Surf. B Biointerfaces 2012, 93, 20–23. [Google Scholar] [CrossRef]
- Fatemi, M.; Mollania, N.; Momeni-Moghaddam, M.; Sadeghifar, F. Extracellular biosynthesis of magnetic iron oxide nanoparticles by Bacillus cereus strain HMH1: Characterization and in vitro cytotoxicity analysis on MCF-7 and 3T3 cell lines. J. Biotechnol. 2018, 270, 1–11. [Google Scholar] [CrossRef]
- Mahdavi, M.; Namvar, F.; Bin Ahmad, M.; Mohamad, R. Green Biosynthesis and Characterization of Magnetic Iron Oxide (Fe3O4) Nanoparticles Using Seaweed (Sargassum muticum) Aqueous Extract. Molecules 2013, 18, 5954–5964. [Google Scholar] [CrossRef] [PubMed]
- Solmaz, A.; Bölükbaşi, Ö.S.; Sari, Z.A. Green industry work: Production of FeCl3 from iron and steel industry waste (mill scale) and its use in wastewater treatment. Environ. Sci. Pollut. Res. 2024, 31, 19795–19814. [Google Scholar] [CrossRef] [PubMed]
- Baena-Moreno, F.M.; Leventaki, E.; Riddell, A.; Wojtasz-Mucha, J.; Bernin, D. Effluents and residues from industrial sites for carbon dioxide capture: A review. Environ. Chem. Lett. 2023, 21, 319–337. [Google Scholar] [CrossRef]
- Lentini, C.J.; Wankel, S.D.; Hansel, C.M. Enriched Iron(III)-Reducing Bacterial Communities are Shaped by Carbon Substrate and Iron Oxide Mineralogy. Front. Microbiol. 2012, 3, 404. (In English) [Google Scholar] [CrossRef]
- Lv, Z.; Lin, J.-M.; He, Y.-Z.; Chen, Y.-W.; Chen, Y.-T. Insights into Fe(II) speciation influence on kinetics, microbial networks, and metabolism in Fe(II) oxidation coupled with carbon assimilation in paddy soil. Environ. Technol. Innov. 2025, 40, 104601. [Google Scholar] [CrossRef]
- Perez-Gonzalez, T.; Jimenez-Lopez, C.; Neal, A.L.; Rull-Perez, F.; Rodriguez-Navarro, A.; Fernandez-Vivas, A.; Iañez-Pareja, E. Magnetite biomineralization induced by Shewanella oneidensis. Geochim. Cosmochim. Acta 2010, 74, 967–979. [Google Scholar] [CrossRef]
- Chen, G.; Thompson, A.; Gorski, C.A. Disentangling the size-dependent redox reactivity of iron oxides using thermodynamic relationships. Proc. Natl. Acad. Sci. USA 2022, 119, e2204673119. [Google Scholar] [CrossRef]
- Chen, P.; De Meulenaere, E.; Deheyn, D.D.; Bandaru, P.R. Iron redox pathway revealed in ferritin via electron transfer analysis. Sci. Rep. 2020, 10, 4033. [Google Scholar] [CrossRef] [PubMed]
- Gahlawat, G.; Choudhury, A.R. A review on the biosynthesis of metal and metal salt nanoparticles by microbes. RSC Adv. 2019, 9, 12944–12967. [Google Scholar] [CrossRef] [PubMed]
- Hulkoti, N.I.; Taranath, T.C. Biosynthesis of nanoparticles using microbes—A review. Colloids Surf. B Biointerfaces 2014, 121, 474–483. [Google Scholar] [CrossRef]
- Ennaceri, H.; Benyoussef, A.; Ennaoui, A.; Khaldoun, A. Optical Properties of Front and Second Surface Silver-Based and Molybdenum-Based Mirrors. Int. J. Eng. Technol. 2016, 8, 410–413. [Google Scholar] [CrossRef]
- Ennaceri, H.; El Alami, H.; Brik, H.; Mokssit, O.; Khaldoun, A. Lotus effect and super-hydrophobic coatings for concentrated solar power systems (CSP). In Proceedings of the 2014 International Conference on Composite Materials & Renewable Energy Applications (ICCMREA), Sousse, Tunisia, 22–24 January 2014; pp. 1–4. [Google Scholar] [CrossRef]
- El Baraka, A.; Ennaceri, H.; Ennaoui, A.; Ghennioui, A.; Jorio, A.; Khaldoun, A. A novel approach to evaluate soiling adhesion on the surface of CSP reflectors via extended DLVO theory. Appl. Phys. A 2019, 125, 515. [Google Scholar] [CrossRef]
- Boujnah, M.; Ennaceri, H.; El Kenz, A.; Benyoussef, A.; Chavira, E.; Loulidi, M.; Ez-Zahraouy, H. The impact of point defects on the optical and electrical properties of cubic ZrO2. J. Comput. Electron. 2020, 19, 940–946. [Google Scholar] [CrossRef]
- Sidhu, A.K.; Verma, N.; Kaushal, P. Role of biogenic capping agents in the synthesis of metallic nanoparticles and evaluation of their therapeutic potential. Front. Nanotechnol. 2022, 3, 801620. [Google Scholar] [CrossRef]
- Mbuyazi, T.B.; Ajibade, P.A. Influence of different capping agents on the structural, optical, and photocatalytic degradation efficiency of magnetite (Fe3O4) nanoparticles. Nanomaterials 2023, 13, 2067. [Google Scholar] [CrossRef]
- El Ghandoor, H.; Zidan, H.; Khalil, M.M.; Ismail, M. Synthesis and some physical properties of magnetite (Fe3O4) nanoparticles. Int. J. Electrochem. Sci. 2012, 7, 5734–5745. [Google Scholar] [CrossRef]
- Yang, H.; Chen, Z.; Kong, L.; Xing, H.; Yang, Q.; Wu, J. A Review of Eco-Corona Formation on Micro/Nanoplastics and Its Effects on Stability, Bioavailability, and Toxicity. Water 2025, 17, 1124. [Google Scholar] [CrossRef]
- Chen, Z. Iron-Mediated Biogeochemical Cycling of Carbon in (Sub-) Arctic Soils. Ph.D. Thesis, Freie Universität Berlin, Berlin, Germany, 2025. [Google Scholar]
- Tetteh, E.K.; Rathilal, S. Application of biomagnetic nanoparticles for biostimulation of biogas production from wastewater treatment. Mater. Today Proc. 2021, 45, 5214–5220. [Google Scholar] [CrossRef]
- Mohammadi, M.; Reinicke, B.; Wawrousek, K. Biosorption and biomagnetic recovery of La3+ by Magnetospirillum magneticum AMB-1 biomass. Sep. Purif. Technol. 2022, 303, 122140. [Google Scholar] [CrossRef]
- Hu, F.; Meng, L.; Wang, M.; Zhang, Y.; Xu, Z. Roles of naturally occurring biogenic iron-manganese oxides (BFMO) in PMS-based environmental remediation: A complete electron transfer pathway. J. Environ. Sci. 2025, 155, 795–805. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Knowles, W.; Brown, R.; Hondow, N.; Arakaki, A.; Baldwin, S.; Staniland, S.; Matsunaga, T. Biomagnetic recovery and bioaccumulation of selenium granules in magnetotactic bacteria. Appl. Environ. Microbiol. 2016, 82, 3886–3891. [Google Scholar] [CrossRef] [PubMed]
- Borlido, L.; Azevedo, A.; Roque, A.; Aires-Barros, M. Magnetic separations in biotechnology. Biotechnol. Adv. 2013, 31, 1374–1385. [Google Scholar] [CrossRef]
- Cotten, G.B. Magnetic Separations with Magnetite: Theory, Operation and Limitations; University of Idaho: Moscow, ID, USA, 2000. [Google Scholar]
- Caballero-Mejía, B.; Moliner, A.; Escolástico, C.; Hontoria, C.; Mariscal-Sancho, I.; Pérez-Esteban, J. Use of magnetite nanoparticles and magnetic separation for the removal of metal(loid)s from contaminated mine soils. J. Hazard. Mater. 2025, 486, 137081. [Google Scholar] [CrossRef]
- Gagen, E.J.; Filip, J.; Coker, V.S.; Sadhukhan, J.; Safarik, I.; Bagshaw, H.; Lloyd, J.R. Accelerating microbial iron cycling promotes re-cementation of surface crusts in iron ore regions. Microb. Biotechnol. 2020, 13, 1960–1971. (In English) [Google Scholar] [CrossRef]
- Joshi, N.; Filip, J.; Coker, V.S.; Sadhukhan, J.; Safarik, I.; Bagshaw, H.; Lloyd, J.R. Microbial reduction of natural Fe(III) minerals; toward the sustainable production of functional magnetic nanoparticles. Front. Environ. Sci. 2018, 6, 127. [Google Scholar] [CrossRef]
- Sharma, R.; Sharma, S.; Mohammad, I.; Singh, V.; Sharma, G.K.; Nunia, V. Harnessing microbial diversity for nanoparticle synthesis from mineral ores: Toward sustainable agriculture. AMB Express 2025, 15, 170. (In English) [Google Scholar] [CrossRef]
- Cerff, M.; Morweiser, M.; Dillschneider, R.; Michel, A.; Menzel, K.; Posten, C. Harvesting fresh water and marine algae by magnetic separation: Screening of separation parameters and high gradient magnetic filtration. Bioresour. Technol. 2012, 118, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Zhao, G.; Li, J.; Shi, Y.; Zhu, Q.; Peng, X. Enhanced Recovery of Magnetic Materials from Fly Ash Using Spiral Dry Magnetic Separation Technique. Waste Biomass Valorization 2025, 16, 1215–1228. [Google Scholar] [CrossRef]
- Pahariya, R.; Chauhan, A.; Ranjan, A.; Basniwal, R.K.; Upadhyay, S.; Thakur, S.K.; Jindal, T. Critical Review on the Efficacy and Mechanism of Nanoparticle-Based Flocculants for Biodiesel Feedstock Production from Microalgae. BioEnergy Res. 2024, 17, 1065–1079. [Google Scholar] [CrossRef]
- Almomani, F. Algal cells harvesting using cost-effective magnetic nano-particles. Sci. Total Environ. 2020, 720, 137621. [Google Scholar] [CrossRef]
- Mkpuma, V.O.; Moheimani, N.R.; Ennaceri, H. Effect of light intensity on Chlorella sp. biofilm growth on anaerobically digested food effluents (ADFE). J. Environ. Manag. 2024, 371, 123015. [Google Scholar] [CrossRef] [PubMed]
- Mkpuma, V.O.; Moheimani, N.R.; Ennaceri, H. Biofilm and suspension-based cultivation of microalgae to treat anaerobic digestate food effluent (ADFE). Sci. Total Environ. 2024, 924, 171320. [Google Scholar] [CrossRef]
- Mkpuma, V.O.; Moheimani, N.R.; Ennaceri, H. Biofilm cultivation of chlorella species. MUR 269 to treat anaerobic digestate food effluent (ADFE): Total ammonia nitrogen (TAN) concentrations effect. Chemosphere 2024, 354, 141688. [Google Scholar] [CrossRef]
- Mkpuma, V.O.; Moheimani, N.R.; Ennaceri, H. Photosynthetic performance and nutrient removal from anaerobically digested effluents by microalgal biofilms. J. Water Process Eng. 2024, 65, 105811. [Google Scholar] [CrossRef]
- Mkpuma, V.O.; Najafi, M.; Farahbakhsh, J.; Zargar, M.; Moheimani, N.R.; Ennaceri, H. Membrane fouling dynamics and flux optimization in the ultrafiltration of Chlorella sp. MUR 269 and Dunaliella salina CS 744/01. J. Water Process Eng. 2025, 78, 108808. [Google Scholar] [CrossRef]
- Fu, Y.; Hu, F.; Li, H.; Cui, L.; Qian, G.; Zhang, D.; Xu, Y. Application and mechanisms of microalgae harvesting by magnetic nanoparticles (MNPs). Sep. Purif. Technol. 2021, 265, 118519. [Google Scholar] [CrossRef]
- Amo-Duodu, G.; Ennaceri, H.; Bahri, P.A.; Moheimani, N.R. Magnetic flocculation of microalgae using magnetite nanoparticles: Progress and challenges. Algal Res. 2025, 91, 104340. [Google Scholar] [CrossRef]
- Boli, E.; Savvidou, M.; Logothetis, D.; Louli, V.; Pappa, G.; Voutsas, E.; Kolisis, F.; Magoulas, K. Magnetic harvesting of marine algae Nannochloropsis oceanica. Sep. Sci. Technol. 2021, 56, 730–737. [Google Scholar] [CrossRef]
- Abo Markeb, A.; Llimós-Turet, J.; Ferrer, I.; Blánquez, P.; Alonso, A.; Sánchez, A.; Moral-Vico, J.; Font, X. The use of magnetic iron oxide based nanoparticles to improve microalgae harvesting in real wastewater. Water Res. 2019, 159, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zou, W.; Cao, Y. Bioelectromagnetic fields as signaling currents of life. Radiat. Med. Prot. 2025, 6, 112–118. [Google Scholar] [CrossRef]
- Ku, J.; Wang, K.; Wang, Q.; Lei, Z. Application of magnetic separation technology in resource utilization and environmental treatment. Separations 2024, 11, 130. [Google Scholar] [CrossRef]
- Shakoor, N.; Adeel, M.; Rui, Y.; Azeem, I.; Li, Y.; Ahmad, M.A.; Hussain, M.; Xu, M.; Zain, M. Iron-based nanomaterials are emerging nanofertilizers to fulfil iron deficiency. In Engineered Nanoparticles in Agriculture: From Laboratory to Field; Walter de Gruyter GmbH & Co KG: Berlin, Germany, 2023; p. 145. [Google Scholar]
- Jiménez-Vázquez, A.; Jaimes-López, R.; Morales-Bautista, C.M.; Pérez-Rodríguez, S.; Gochi-Ponce, Y.; Estudillo-Wong, L.A. Catalytic Applications of Natural Iron Oxides and Hydroxides: A Review. Catalysts 2025, 15, 236. [Google Scholar] [CrossRef]
- Prabowo, B.; Khairunnisa, T.; Nandiyanto, A.B.D. Economic perspective in the production of magnetite (Fe3O4) nanoparticles by co-precipitation method. World Chem. Eng. J. 2018, 2, 1–4. [Google Scholar]
- Rahman, A.; Kang, S.; McGinnis, S.; Vikesland, P.J. Life cycle impact assessment of iron oxide (Fe3O4/γ-Fe2O3) nanoparticle synthesis routes. ACS Sustain. Chem. Eng. 2022, 10, 3155–3165. [Google Scholar] [CrossRef]
- Iqbal, J.; Abbasi, B.A.; Ahmad, R.; Shahbaz, A.; Zahra, S.A.; Kanwal, S.; Munir, A.; Rabbani, A.; Mahmood, T. Biogenic synthesis of green and cost effective iron nanoparticles and evaluation of their potential biomedical properties. J. Mol. Struct. 2020, 1199, 126979. [Google Scholar] [CrossRef]
- Siddiqi, K.S.; Rahman, A.U.; Tajuddin, N.; Husen, A. Biogenic fabrication of iron/iron oxide nanoparticles and their application. Nanoscale Res. Lett. 2016, 11, 498. [Google Scholar] [CrossRef]
- Li, S.; Li, C.; Gao, X.; Zhu, M.; Li, H.; Wang, X. Biomineralization Mediated by Iron-Oxidizing Microorganisms: Implication for the Immobilization and Transformation of Heavy Metals in AMD. Minerals 2025, 15, 868. [Google Scholar] [CrossRef]
- Thiyagarajulu, N.; Yuvaraj, D.; Gopinathan, P.; Paramasivam, D.; Rajkumar, M.; Subramani, T.; Rangappa, E.S.; Sivagamasundari, U.; Huang, Z. Assessment on Sustainable Biomining: Integrating Environmental Responsibility and Economic Viability. Land Degrad. Dev. 2025, 37, 3–27. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.