Abstract
As a continuation of research on 1-vinylazulenes synthesis, on their physico–chemical properties as well as on their use as colorimetric and electrochemical materials for the detection of heavy metals ions, new compounds, 4-(azulen-1-ylmethylene)-2-phenyloxazol-5(4H)-ones, are reported. The exceptional structure of azulene moiety with symmetrical electron distribution only with respect to the x-axis gives some special chemical and physical properties to compounds in which it is inserted. Various azulene-1-carbaldehydes and hippuric acids are condensed with good yields in Erlenmeyer—Plöchl reaction conditions. The obtained 1-vinylzulenes were characterized and their UV-Vis and NMR spectra were briefly discussed.
1. Introduction
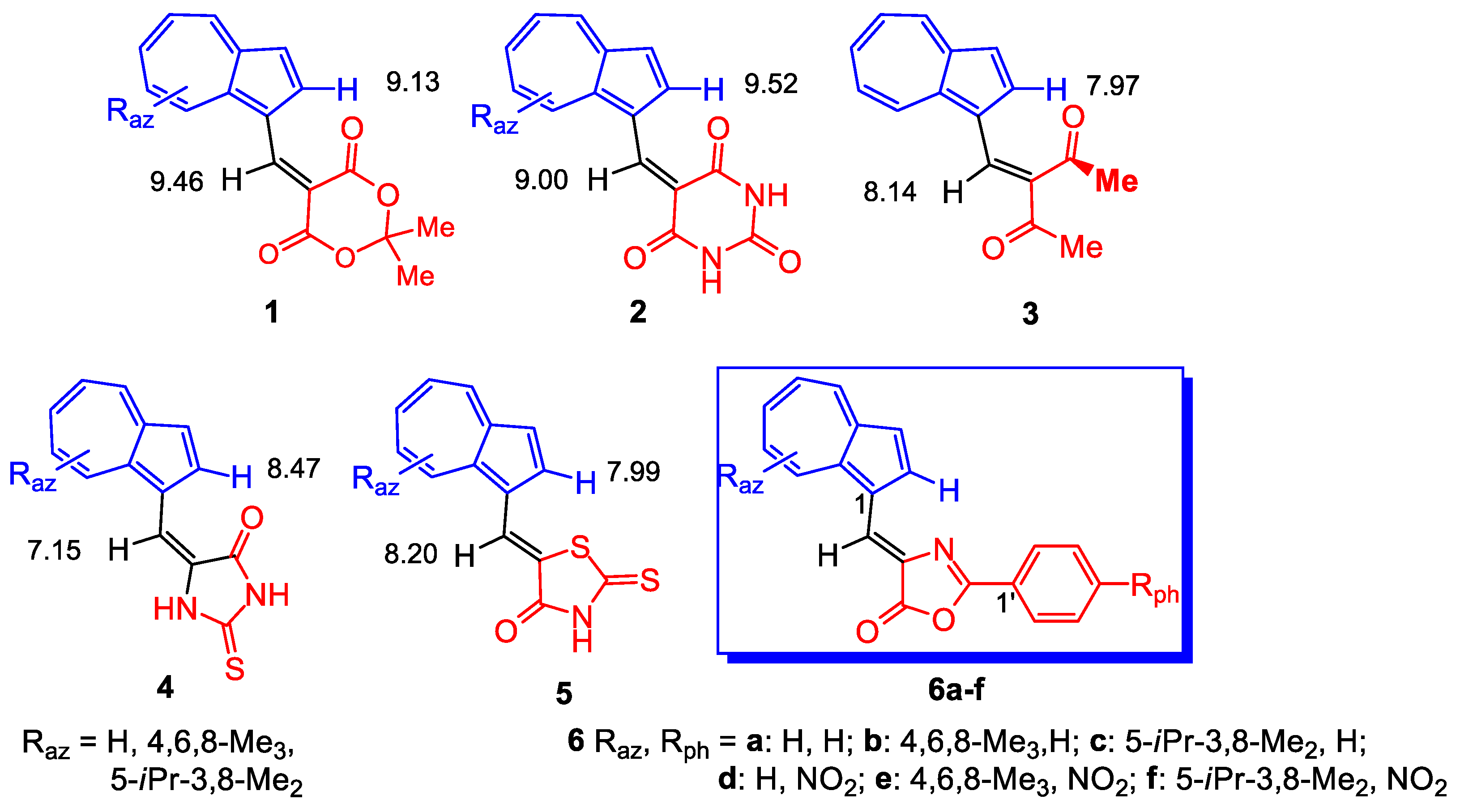
The exceptional structure of azulene with symmetrical electron distribution only with respect to the x-axis gives some special chemical and physical properties to compounds in which it is inserted compared to compounds that contain the well-known naphthalene structure with negative charge supplementary symmetrical with respect to the y-axis [1]. Therefore, the past and also the present interest for the study of these compounds is understandable. Thus, the complexes of some azulene ligands with metals [2,3,4] or some modified electrodes, realized by depositing a polyazulene film, can play a role as heavy metal ion sensors [5,6,7,8,9]. In this purpose several heterocycles substituted with azulen-1-vinyl moietyas such or substituted with alkyl groups, 5-(azulen-1-ylmethylene)-2,2-dimethyl-1,3-dioxane-4,6-dione, 1, 5-(azulen-1-ylmethylene)pyrimidine-2,4,6(1H,3H,5H)-trione, 2, (E)-5-(azulen-1-ylmethylene)-2-thioxoimidazolidin-4-one, 4 and (Z)-5-(azulen-1-ylmethylene)-2-thioxothiazolidin-4-one, 5, shown in Figure 1, were synthesized [4,10]. The compounds 4 and 5 were already investigated for building complexing chemically modified electrodes for metals [11,12,13]. The encouraging obtained results stimulated the expansion of these classes of compounds, and in the present work the synthesis of azulen-1-ylmethylene-2-phenyloxazol-5(4H)-ones, 6a–f was carried out. Determination of electrochemical properties of these compounds and the polymerization attempts on electrodes surface in order to obtain modified electrode as metal sensor are under way.
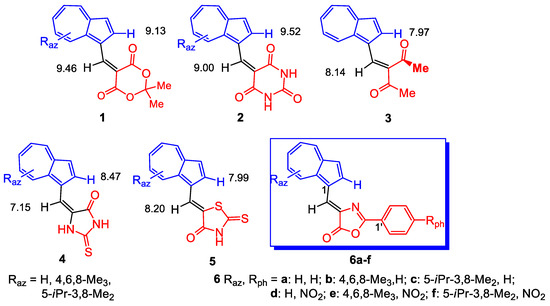
Figure 1.
Azulen-1-yl-methylene substituted heterocycles (1, 2, 4–6) and 3-(azulen-1-yl-methylene)pentane-2,4-dione (3); δ (ppm) for protons in 1H-NMR spectra.
2. Materials and Methods
2.1. Materials and Instrumentation
The starting materials, 2-benzamidoacetic acid (hippuric acid) and 2-(4-nitrobenzamido) acetic acid (4-nitrohippuric acid), were obtained from commercial sources and used without further purification. Starting azulene-1-carbaldehydes (2a–c) were obtained by the Vilsmeier procedure. Melting points (uncorrected) were measured with the Kofler apparatus (Firma Reichert, Vienna, Austria). Elemental analyses were performed using Perkin Elmer CHN 240B (Beakonsfield, England). For UV spectra a Varian Cary 100 spectrophotometer (Varian, Switzerland) was used (λ values in nm and the molar extinction, ε, in M−1 cm−1). 1H- and 13C-NMR: Bruker FOURIER 300 (Bruker Corp., Massachusetts, USA) and Gemini 300 (Bruker Corp., Massachusetts, USA) (1H: 300 MHz, 13C: 75.47 MHz) and Bruker Avance III HD 600 (Bruker Corp., Massachusetts, USA) (1H: 600 MHz, 13C: 150 MHz); δ values in ppm and J values are given in Hz; TMS was used as internal standard in CDCl3; several signals were assigned on the basis of COSY, HETCOR and HMBC experiments. The 1H and 13C spectra are given in the Supplementary Materials. Mass spectra: Varian 1200L Triple Quadrupole LC/MS/MS spectrometer (Varian, Switzerland) by direct injection in ESI. For the column chromatography, silica gel 60 was used. The dichloromethane (DCM) was distilled over CaH2. The compounds’ nomenclature was taken from the Cambridge-Soft package of structure-to-name algorithm included with ChemBioDraw Ultra 11.0.
2.2. Condensation of Azulene-1-Carbaldehydes with Hippuric Acids
A mixture of hippuric acid (8H) (89.5 mg, 0.5 mmol) or 4-nitrohippuric acid (8NO2) (112 mg, 0.5 mmol) and azulen-1-carbaldehydes (7Raz) (0.5 mmol) in 2 mL of acetic anhydride, in the presence of sodium acetate (50 mg, 0.6 mmol) was heated under reflux for 2–4 h in inert atmosphere. When the reaction was complete (determined by TLC), the mixture was cooled to room temperature. Water and a small amount of methanol were added, and the resulting solution was extracted with DCM. The organic layer was washed twice with water to remove acetic anhydride and sodium acetate and was dried on anhydrous Na2SO4, and the solvent was evaporated in vacuum. The crude products were chromatographed on the silica gel column, using as eluent a mixture of petroleum ether and DCM in 1:1 volumetric ratio, with increased gradient in DCM (50–100% DCM). The products (6a–c) eluted as red-brown bands and the products (6d–f) eluted as violet bands. The samples for products’ characterization were realized by the second chromatographic separation in the same conditions, with the specification that in the case of compound 6d, acetone was used as eluent instead of DCM, for a better elution from the column. The reaction yield for all products was reported in Table 1.

Table 1.
Yields of condensation of azulene-1-carbaldehydes (7(Raz)) with hippuric acids (8(Rph)) a.
2.3. Products Characterization
4-(Azulen-1-ylmethylene)-2-phenyloxazol-5(4H)-one, (6a). Brown-reddish (brick) crystals, m.p. 258–261 °C. UV-Vis (MeOH), λ(log ε): 230 (4.53), 282 (4.30), 293 (4.28), 312 (4.17), 317 sh (4.17), 328 (4.17), 410 (4.15), 468 (4.45). 1H-NMR (300 MHz, CDCl3, 20 °C): 7.44 (t, 3J = 9.8 Hz, 1 H, 5-H), 7.49 (t, 3J = 9.8 Hz, 1 H, 7-H), 7.52–7.60 (m, 4 H, 3-H, 3′-H, 5′-H, 4′-H), 7.78 (t, 3J = 9.8 Hz, 1 H, 6-H), 7.94 (s, 1 H, CH=), 8.22 (dd, 3J = 7.6 Hz, 4J = 1.0 Hz, 2 H, 2′-H, 6′-H), 8.41 (d, 3J = 9.5 Hz, 1 H, 4-H), 8.72 (d, 3J = 9.8 Hz, 1 H, 8-H), 9.30 (d, 3J = 4.3 Hz, 1 H, 2-H) ppm. 13C-NMR (75.47 MHz, CDCl3, 20 °C): 122.2, 123.4, 124.3, 126.4, 126.9, 127.8, 128.0, 128.8, 129.1, 132.4, 134.0, 137.9, 139.4, 141.0, 141.9, 145.5, 160.7, 168.6 ppm. IR (neat): 3492 w, 3063 w, 3024 w, 2620 w, 2448 w, 2187 w, 1817 w, 1754 vs, 1626 vs, 1580 s, 1485 s, 1446 s, 1387 s, 1284 s, 1173 s, 1043 m, 1012 m, 969 s, 847 s, 770 s,737 s, 687 s cm−1. MS (+ESI): 300 [M+1]. Calcd. for C20H13NO2: C, 80.25; H, 4.38; N, 4.68. Found: C, 80.21; H, 4.39; N, 4.66.
2-Phenyl-4-((4,6,8-trimethylazulen-1-yl)methylene)oxazol-5(4H)-one, (6b). Dark violet crystals, m.p. 214–216 °C. UV-Vis (MeOH), λ(log ε): 243 (4.43), 295 sh (3.97), 329 (4.14), 404 sh (3.92), 486 (4.40). 1H-NMR (300 MHz, CDCl3, 20 °C): 2.63 (s, 3 H, Me), 2.89 (s, 3 H, Me), 3.19 (s, 3 H, Me), 7.25 (s, 1 H, 5-H), 7.26 (s, 1 H, 7-H), 7.46 (d, 3J = 4.7 Hz, 1 H, 3-H), 8.17 (dd, 3J = 7.9 Hz, 4J = 1.5 Hz, 2 H, 2′-H, 6-H), 7.48–7.55 (m, 3 H, 3′-H, 4′-H, 5′-H), 8.25 (s, 1 H, CH=), 9.16 (d, 3J = 4.7 Hz, 1 H, 2-H) ppm. 13C-NMR (75.47, MHz, CDCl3, 20 °C): 26.0, 28.4, 30.1, 119.8, 125.6, 126.6, 127.7, 128.8, 129.1, 129.5, 132.0, 132.1, 133.8, 138.1, 139.9, 142.6, 146.9, 148.1, 148.2, 160.3, 169.0 ppm. IR (neat): 3103 w, 2918 vs, 2855 s, 2175 w, 1723 vs, 1612 s, 1571 s, 1490 s, 1517 s, 1403 s, 1318 s, 1265 vs, 1152 s, 1020 s, 839 m, 698 m cm−1. MS (+ESI): 342 [M+1]. Calcd. for C23H19NO2: C, 80.92; H, 5.61; N, 4.10. Found: C, 80.90; H, 5.60; N, 4.13.
4-((5-isopropyl-3,8-dimethylazulen-1-yl)methylene)-2-phenyloxazol-5(4H)-one, (6c). Dark green crystals, m.p. 166–168 °C. UV-Vis (MeOH), λ(log ε): 233 sh (4.45), 244 (4.47), 315 (4.07), 363 (3.93), 510 (4.53). 1H-NMR (300 MHz, CDCl3, 20 °C): 1.39 (d, 3J = 6.9 Hz, 6 H, CHMe), 2.67 (s, 3 H, Me3), 3.12 (sept, 3J = 6.9 Hz, 1 H, CHMe2), 3.18 (s, 3 H, Me8), 7.26 (d, 3J = 10.8 Hz, 1 H, 6-H), 7.47–7.55 (m, 4 H, 7-H, 3′-H, 4′-H, 5′-H), 8.17 (d, 4J = 1.7 Hz, 1 H, 4-H), 8.31 (dd, 3J = 7.3 Hz, 4J = 1.7 Hz, 2 H, 2′-H, 6′-H), 8.24 (s, 1 H, CH=), 9.13 (s, 1 H, 2-H) ppm. 13C-NMR (75.47, MHz, CDCl3, 20 °C): 13.3, 24.4, 29.1, 38.0, 123.2, 127.2, 127.6, 127.9, 128.8, 129.7, 133.0, 134.5, 136.4, 140.0, 141.8, 144.1, 147.0, 147.2, 159.9, 169.1 ppm. IR (neat): 3063 w, 2956 m, 2915 m, 2068 w, 1758 vs, 1615 vs, 1582 s, 1525 s, 1422 s, 1342 s, 1324 s, 1151 s, 1094 br, 971 m, 864 m, 684 s cm−1. MS (+ESI): 370 [M+1]. Calcd for C25H23NO2: C, 81.27; H, 6.27; N, 3.79. Found: C, 81.25; H, 6.27; N, 3.80.
4-(Azulen-1-ylmethylene)-2-(4-nitrophenyl)oxazol-5(4H)-one, (6d). Dark-green crystals, m.p. 295–297 °C. UV-Vis (MeOH), λ(log ε): 233 (4.15), 246 sh (4.12), 325 (4.07), 397 (3.99), 507 (3.75). 1H-NMR (600 MHz, CDCl3, 20 °C): 7.50 (t, 3J = 9.8 Hz, 1H, 5-H), 7.56 (t, 3J = 9.8 Hz, 1H, 7-H), 7.60 (d, 3J = 4.3 Hz, 1H, 3-H), 7.83 (t, 3J = 9.8 Hz, 1H, 6-H), 8.03 (s, 1H, CH=), 8.35 (dd, 3J = 7.6 Hz, 4J = 1.0 Hz, 2 H, 2′-H, 6′-H), 8.38 (dd, 3J = 7.6 Hz, 4J = 1.0 Hz, 2 H, 3′-H, 5′-H), 8.45 (d, 3J = 9.5 Hz, 1H, 4-H), 8.75 (d, 3J = 9.8 Hz, 1H, 8-H), 9.26 (d, 3J = 4.3 Hz, 1H, 2-H) ppm. 13C-NMR (150 MHz, CDCl3, 20 °C): 122.9, 124.1, 124.3, 125.7, 127.8, 128.3, 128.4, 128.7, 132.1, 134.2, 138.3, 139.8, 141.2, 142.9, 146.3, 149.7, 158.3, 167.8 ppm. IR (neat): 3083 w, 2918 w, 2610 w, 1767 s, 1723 m, 1632 s, 1585 s, 1401 m, 1317 vs, 1286 s, 1170 m, 1094 m, 848 m, 605 m cm−1. MS (+ESI): 345 [M+1]. Calcd. for C20H12N2O4: C, 69.76; H, 3.51; N, 8.14. Found: C, 69.79; H, 3.50; N, 8.11.
2-(4-Nitrophenyl)-4-((4,6,8-trimethylazulen-1-yl)methylene)oxazol-5(4H)-one, (6e). Dark-green crystals, m.p. 269–270 °C. UV-Vis (MeOH), λ(log ε): 236 (4.06), 257 (4.03), 333 (3.93), 405 (3.70), 521 (3.93). 1H-NMR (300 MHz, CDCl3, 20 °C): 2.67 (s, 3 H, Me6), 2.92 (s, 3 H, Me4), 3.22 (s, 3 H, Me8), 7.34 (s, 2 H, 5-H, 7-H), 7.48 (d, 3J = 4.7 Hz, 1 H, 3-H), 8.37 (s, 1H, CH=), 8.36–8.29 (m, 5 H, 2′-H, 6′-H, 3′-H, 5′-H), 9.16 (d, 3J = 4.5 Hz, 1 H, 2-H) ppm. 13C-NMR (75.47, MHz, CDCl3, 20 °C): 26.1, 28.4, 30.2, 120.5, 124.1, 125.8, 126.8, 128.2, 131.3, 132.2, 133.2, 134.8, 139.1, 140.1, 143.6, 147.5, 148.6, 148.7, 157.8, 168.2 ppm. IR (neat): 3094 w, 2914 w, 1786 w, 1748 s, 1619 m, 1585 s, 1508 s, 1411 w, 1321 vs, 1219 m, 1166 m, 1091 m, 855 s, 713 s cm−1. MS (+ESI): 387 [M+1]. Calcd. for C23H18N2O4: C, 71.49; H, 4.70; N, 7.25. Found: C, 71.47; H, 4.71; N, 7.21.
4-((5-Isopropyl-3,8-dimethylazulen-1-yl)methylene)-2-(4-nitrophenyl)oxazol-5(4H)-one, (6f). Dark-green crystals, m.p. 261–263 °C. UV-Vis (MeOH), λ(log ε): 246 (4.27), 334 (4.13), 423 (3.98), 546 (4.19). 1H-NMR (300 MHz, CDCl3, 20 °C): 1.42 (d, 3J = 6.8 Hz, 6 H, MeCH), 2.69 (s, 3 H, Me3), 3.16 (sept, 3J = 6.8 Hz, 1 H, CHMe2), 3.21 (s, 3 H, Me8), 7.36 (dAB, 3J = 10.3 Hz, 5-H), 7.57 (dAB,d, 3J = 10.3 Hz, 4J = 1.0 Hz, 1 H, 6-H), 8.21 (d, 4J = 1.0 Hz, 1 H, 4-H), 8.30 (d, 3J = 9.0 Hz, 2 H, 2′,6′-Ph), 8.36 (d, 3J = 9.0 Hz, 2 H, 3′,5′-Ph), 8.33 (s, 1 H, CH=), 9.11 (s, 1 H, 2-H). 13C-NMR (75.47 MHz, CDCl3, 20 °C): 13.3, 24.4, 29.2, 38.1, 123.5, 124.3, 126.3, 128.0, 129.9, 130.6, 132.2, 134.1, 134.8, 136.8, 141.2, 141.8, 145.1, 147.6, 148.5, 149.3, 157.2, 168.3. IR (neat): 3482 w, 3072 br, 2918 m, 2856 m, 1738 vs, 1617 s, 1579 s, 1504 vs, 1423 s, 1305 vs, 1147 s, 1086 s, 847 s, 694 m cm−1. MS (+ESI): 415 [M+1]. Calcd. for C25H22N2O4: C, 72.45; H, 5.35; N, 6.76. Found: C, 72.49; H, 5.34; N, 6.73.
3. Results and Discussion
3.1. Synthesis
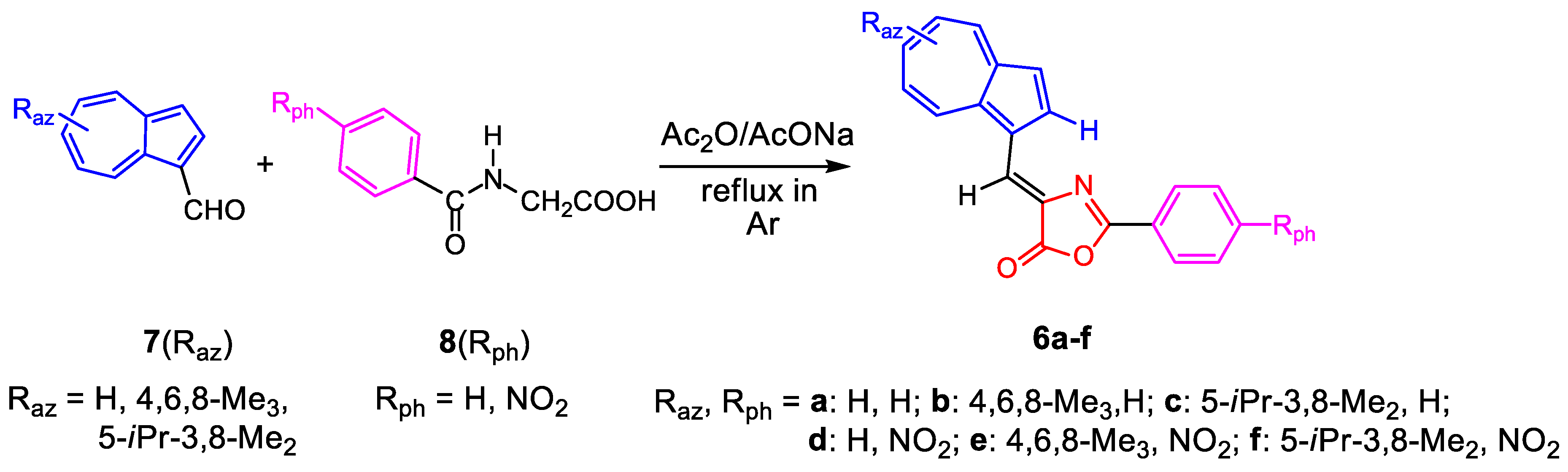
The compounds 6a–f were successfully synthesized following the Erlenmeyer–Plöchl synthesis [14]. Thus, azulene-1-carbaldehydes (7) were condensed with compounds with active methylene unit, hippuric acids (8), at reflux, under inert atmosphere. Acetic anhydride was used as solvent and dehydrating agent in the presence of AcONa as catalyst. The synthesis of compounds, described in Scheme 1, occurred with good yields. Several problems were encountered only in purifying the compound 6d due to its very low solubility.
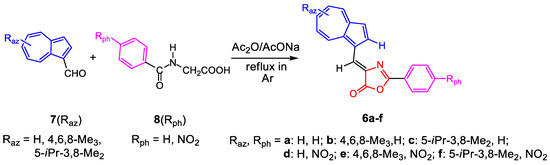
Scheme 1.
Reaction of azulene-1-carbaldehydes 7 with hippuric acids 8.
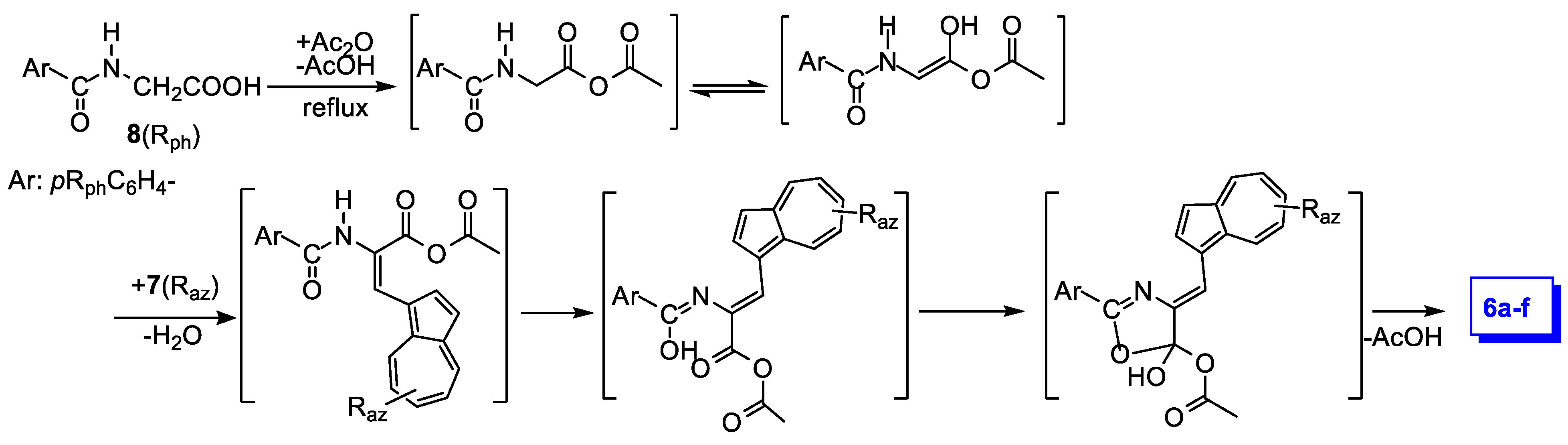
The proposed reaction mechanism [14] applied to the reaction in Scheme 1 is detailed in Scheme 2. The reaction of hippuric acid with acetic anhydride results in a new anhydride, which in turn reacts as enol tautomer with azulene-1-carbaldehyde. In the intermediate, thus formed, a new double bond displacement and subsequent ring closing, with acid removal, generates the desired products, 6a–f.
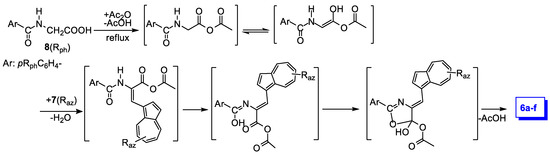
Scheme 2.
Proposed mechanism of azulene-1-carbaldehydes 7 reaction with hippuric acids 8.
3.2. Considerations on UV-Vis and NMR Spectra
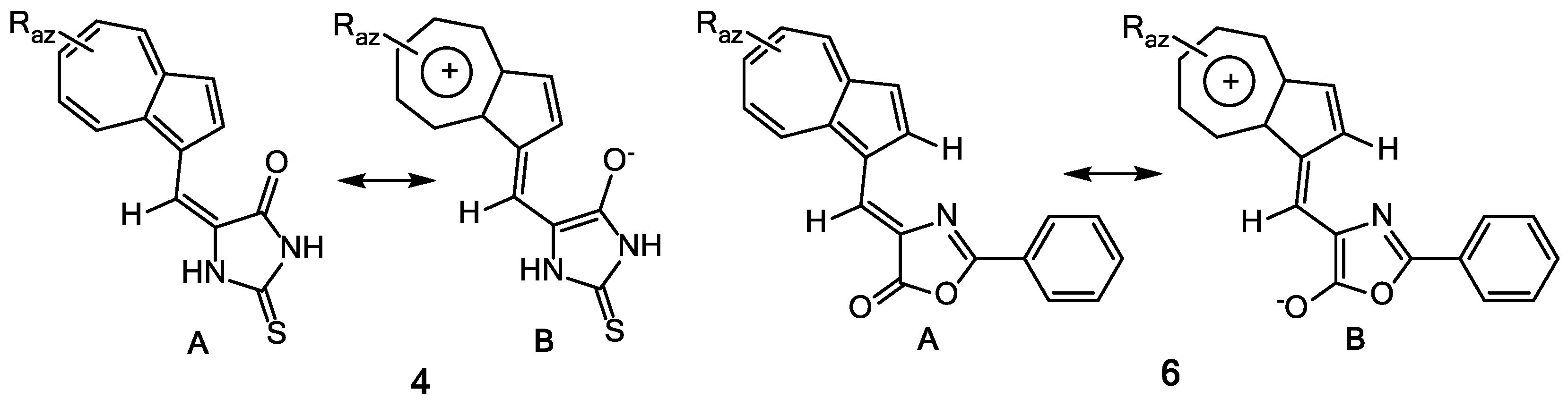
The UV-Vis and NMR spectra of compounds 6a–f (Table 2) are influenced by the nature and position of the grafted substituents on the azulene and phenyl moieties. Figure 2 suggests the stabilization of an entire system of these compounds by generation of tropylium structure (structure 6B) at azulene moiety, simultaneous with the aromatization of 5-membered heterocycle. Nevertheless, the UV-Vis spectra of compounds 6a–c do not differ significantly from those of the similar compounds 4 and 5, possibly due to the low participation of this aromatic structure at the molecules’ properties. Obviously, by the azulene substitution with alkyl groups, the stabilization of polarized system produces a bathochromic shift in visible in order 6c > 6b > 6a (with λ: 510; 486; 468 nm). The presence of nitro group at phenyl results in a bathochromic effect caused by the involvement of this chromophore in the conjugation of electron system.

Table 2.
UV-Vis spectra of compounds 6a–f (λ in nm).
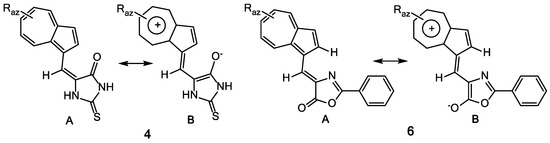
Figure 2.
Resonance structures of compounds 4 and 6.
Interesting information results from the comparison the 1H-NMR spectra of the compounds 6 with those of compounds 1–5. Several significant factors must be taken into account when the chemical shift of these compounds will be examined. The chemical shift of protons belonging to the 7-membered ring does not vary from compound 4 (Raz = H) to 6a. The reduced electron density at 7-membered azulene ring, as a result of the charge displacement towards oxygen, deshields the protons with 0.15–0.42 ppm as compared with the protons at the same positions in the pattern azulene. The higher electronegativity of S [15] present in the heterocycle of compound 5 reduces more efficiently the electron density at 7-membered azulene ring and produces a slight proton deshielding for compound 5 as compared to compounds 6 or 4.
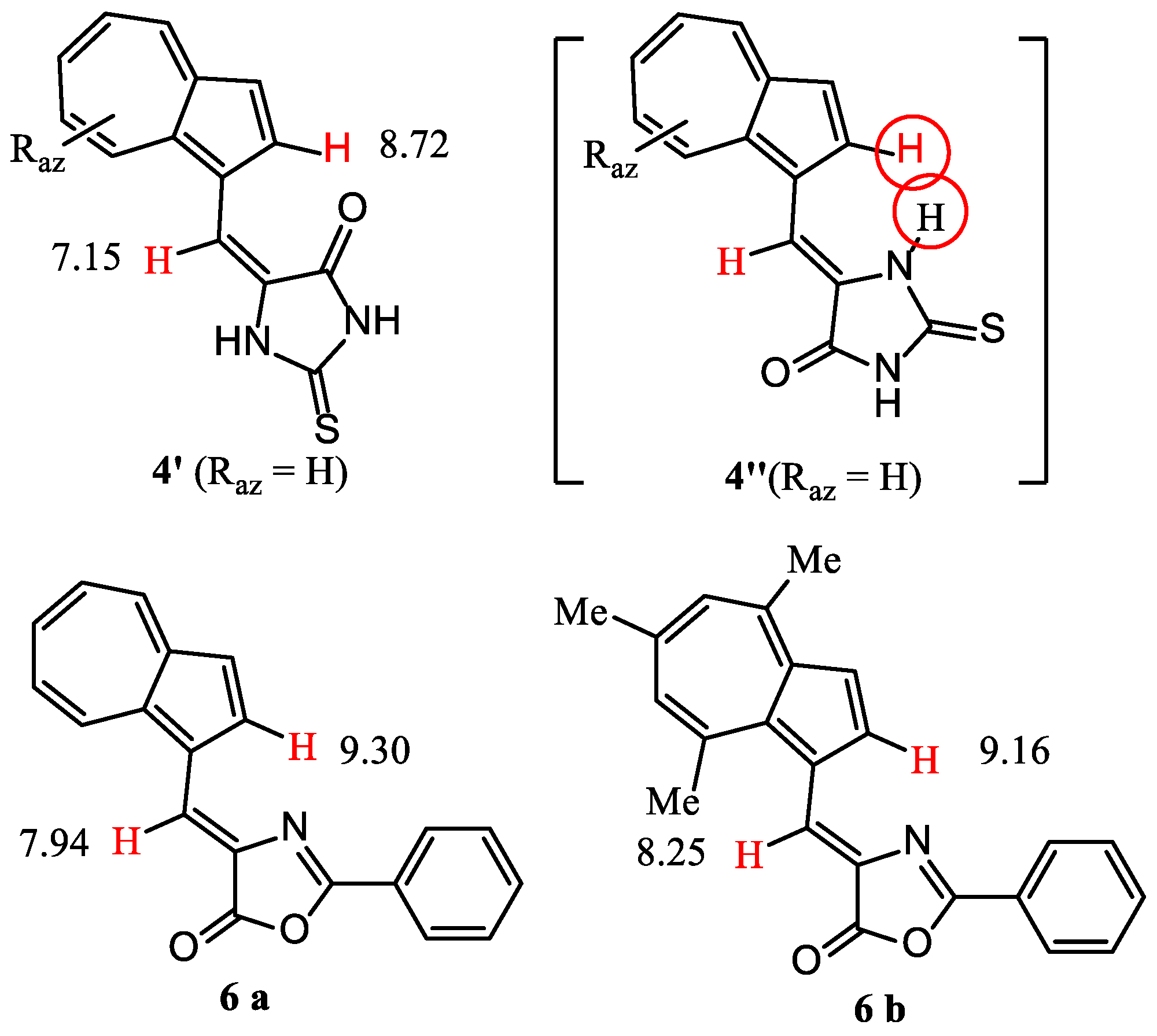
The anisotropy of magnetic fields exerted by different substituents at C1 of azulene on the proton at 2-H is known. As an example, δ = 8.02 ppm was found for 2-H in 1-phenylazulene, whereas for azulene δ value decreases to 7.81 ppm. It has also been found that a double bond develops a remarkable anisotropy of its magnetic field. The effect of this field is evident for the compounds described in Figure 1, where several representative δ are shown. The double bond C=O in compounds 1 and 2 deshields the 2-H until 9.15 and 9.52 ppm, whereas when this double bond is not coplanar with the rest of the structure (compound 3), the value of δ for 2-H is placed next to the value for azulene. The decrease in internal angle for the 5-membered ring in compound 4 compared to that for 6-membered ring for compound 1 or 2, moves the double C=O bond away from 2-H, decreasing the value of δ to 8.47 ppm. A significant difference between the δ values of 2-H for the similar compounds 4 and 5 can be explained by the molecules’ geometry. From Figure 3 it results that isomer 4′ is sterically favored over 4″, whereas the geometry for compound 5, shown in Figure 1, represents the more relaxed structure. Back to compounds synthesized in the present paper, it is found that although the geometry of compounds 5 and 6 are similar, δ for 2-H increases until 9.16 ppm. The magnetic field of C=N double bond belonging to the heterocycle can play a role in the deshielding of this proton.
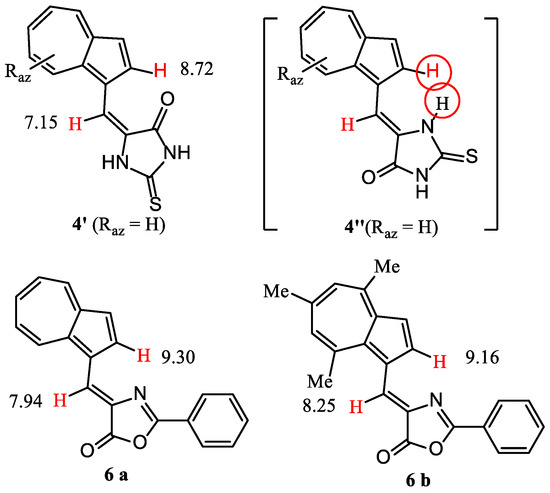
Figure 3.
Geometry of discussed 1-vinylazulenes.
Another aspect to consider further is the δ variation of the proton substituted at the vinyl double bond (=CH). The magnetic field belonging to C=O bond in compounds 1, 2, 5 and 6c deshields this proton as compared with the same proton in compound 4 (in the favorable geometry 4′). It remains, however, difficult to explain the reason for the more intense shielding for compound 6a when compared to 6b. In addition, an important contribution on the downshift of 2-H and vinyl hydrogen can be brought by the hydrogen bond generation between these atoms and carbonylic oxygen of heterocycle in compounds 1, 2, 4 and 6. For the compound 6, the N atom of oxazolone can also form a stable hydrogen bond with 2-H intensifying its downshift.
4. Conclusions
New 1-vinylazulene chromophores, namely 4-(azulen-1-ylmethylene)-2-phenyloxazol-5(4H)-ones, were synthesized. Thus, various azulene-1-carbaldehydes were condensed with good yields with hippuric and 4-nitrohippuric acids in Erlenmeyer–Plöchl reaction conditions, and the obtained products were characterized and considerations about their UV-Vis and NMR spectra were made. A comparison between the resulting compounds and other 1-vinylazulenes obtained in previous works was considered interesting. The influence of the molecules’ geometry was highlighted as well as the activity of the magnetic field anisotropy created by the double bonds on the neighboring protons. The redox behavior, as well as the electrochemically properties and the possible use of products described above as colorimetric and electrochemical materials for detection of heavy metal ions, remains our concern in the future.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/sym13071209/s1, Figure S1. (a) 1H NMR, and (b) 13C NMR spectra of 4-(azulen-1-ylmethylene)-2-phenyloxazol-5(4H)-one 6a; Figure S2. (a) 1H NMR, and (b) 13C NMR spectra of 2-phenyl-4-((4,6,8-trimethylazulen-1-yl)methylene)oxazol-5(4H)-one 6b; Figure S3. (a) 1H NMR, and (b) 13C NMR spectra of 4-((5-isopropyl-3,8-dimethylazulen-1-yl)methylene)-2-phenyloxazol-5(4H)-one 6c; Figure S4. (a) 1H NMR, and (b) 13C NMR spectra of 4-(azulen-1-ylmethylene)-2-(4-nitrophenyl)oxazol-5(4H)-one 6d; Figure S5. (a) 1H NMR, and (b) 13C NMR spectra of 2-(4-nitrophenyl)-4-((4,6,8-trimethylazulen-1-yl)methylene)oxazol-5(4H)-one 6e; Figure S6. (a) 1H NMR, and (b) 13C NMR spectra of 4-((5-isopropyl-3,8-dimethylazulen-1-yl)methylene)-2-(4-nitrophenyl)oxazol-5(4H)-one 6f.
Author Contributions
Conceptualization and methodology, A.C.R., M.C., F.D., and L.B.; writing—original draft preparation A.C.R.; analysis C.E., V.T., A.H., C.D. (Constantin Drăghici), C.D. (Călin Deleanu), A.N., and M.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data available in a publicly accessible repository.
Conflicts of Interest
The authors declare no conflict of interest, financial or otherwise.
References
- Razus, A. Azulene Moiety as Electron Reservoir in Positively Charged Systems; A Short Survey. Symmetry 2021, 13, 526. [Google Scholar] [CrossRef]
- Lash, T.D. Out of the Blue! Azuliporphyrins and Related Carbaporphyrinoid Systems. Acc. Chem. Res. 2016, 49, 471–482. [Google Scholar] [CrossRef] [PubMed]
- Churchill, M.R. Transition Metal Complexes of Azulene and Related Ligands; Wiley: Hoboken, NJ, USA, 2007; Volume 11, pp. 53–98. [Google Scholar]
- Birzan, L.; Cristea, M.; Tecuceanu, V.; Ungureanu, E.-M.; Razus, A.C. Synthesis of ligands based on azulene derivatives used as metal sensors. Rev. Roum. Chim. 2017, 62, 489–497. [Google Scholar]
- Waltman, R.J.; Bargon, J. Electrically conducting polymers: A review of the electropolymerization reaction, of the effects of chemical structure on polymer film properties, and of applications towards technology. Can. J. Chem. 1986, 64, 76–95. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Lai, Y.-H.; Han, M.-Y. Stimuli-Responsive Conjugated Copolymers Having Electro-Active Azulene and Bithiophene Units in the Polymer Skeleton: Effect of Protonation and p-Doping on Conducting Properties. Macromolecules 2004, 37, 3222–3230. [Google Scholar] [CrossRef]
- Buica, G.-O.; Ungureanu, E.-M.; Birzan, L.; Razus, A.C.; Bujduveanu, M.-R. Films of poly(4-azulen-1-yl-2,6-bis(2-thienyl)pyridine) for heavy metal ions complexation. Electrochim. Acta 2011, 56, 5028–5036. [Google Scholar] [CrossRef]
- Buica, G.-O.; Birzan, L.; Tecuceanu, V.; Razus, A.C.; Arnold, G.-L.; Ungureanu, E.-M. Modified Electrodes Based on Poly[(2E)-2-(Azulen-1-ylmethylidene)hydrazinecarbothioamide] for Heavy Metal Ions Complexation. Electroanalysis 2017, 29, 93–102. [Google Scholar] [CrossRef]
- Birzan, L.; Cristea, M.; Draghici, C.C.; Tecuceanu, V.; Maganu, M.; Hanganu, A.; Arnold, G.-L.; Ungureanu, E.-M.; Razus, A.C. 1-vinylazulenes–potential host molecules in ligands for metal ion detectors. Tetrahedron 2016, 72, 2316–2326. [Google Scholar] [CrossRef]
- Razus, A.C.; Birzan, L.; Cristian, L.; Tecuceanu, V. Synthesis and properties of azulene-containing 1,3-dioxanes. Arkivoc 2009, 2009, 31–44. [Google Scholar] [CrossRef] [Green Version]
- Birzan, L.; Cristea, M.; Draghici, C.C.; Tecuceanu, V.; Maganu, M.; Hanganu, A.; Razus, A.C.; Buica, G.-O.; Ungureanu, E.M. Vinylazulenes chromophores: Synthesis and characterization. Dye. Pigment 2016, 131, 246–255. [Google Scholar] [CrossRef]
- Stefaniu, A.; Pop, M.-D.; Arnold, G.-L.; Birzan, L.; Pintilie, L.; Diacu, E.; Ungureanu, E.-M. DFT calculations and electrochemical studies on azulene ligands for heavy metal ions detection using chemically modified electrodes. J. Electrochem. Sci. Eng. 2018, 8, 73–85. [Google Scholar] [CrossRef] [Green Version]
- Ungureanu, E.-M.; Apostoiu, M.P.; Arnold, G.-L.T.; Birzan, L.; Isopescu, R.; Stanciu, G.; Buica, G.-O. Electrochemical Comparison on New (Z)-5-(Azulen-1-Ylmethylene)-2-Thioxo-Thiazolidin-4-Ones. Symmetry 2021, 13, 588. [Google Scholar] [CrossRef]
- Wang, Z. Erlenmeyer-Plöchl Azlactone Synthesis. In Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; Volume 217. [Google Scholar] [CrossRef]
- Detty, M.R.; McKelvey, J.M.; Luss, H.R. Tellurapyrylium dyes. The electron-donating properties of the chalcogen atoms to the chalcogenapyrylium nuclei and their radical dications, neutral radicals, and anions. Organometallics 1988, 7, 1131–1147. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).