Abstract
The global environmental challenges of solid waste accumulation and aquatic eutrophication demand innovative and sustainable strategies. This study introduces a circular “waste-treats-waste” approach by converting dolomite-rich phosphate tailings (PT), a widespread industrial by-product, into a high-value adsorbent for phosphorus (P) removal. Thermal modification at 950 °C for 1 h dramatically enhanced the adsorption capacity by approximately 45 times, from 2.52 mg/g (raw PT) to 112.41 mg/g. This performance is highly competitive with, and often superior to, many engineered adsorbents. The calcination process was pivotal, decomposing carbonates into highly active CaO and MgO while developing a porous structure. Using a multi-technique characterization approach (X-ray diffraction (XRD), Fourier transform infrared spectra (FTIR), TESCAN VEGA3 tungsten filament scanning electron microscope (SEM), the Brunauer–Emmett–Teller method (BET)), the key immobilization mechanism was identified as hydroxyapatite formation, driven by Ca2+/Mg2+-phosphate precipitation and surface complexation. Nonlinear regression analysis revealed that the adsorption kinetics obeyed the pseudo-second-order model, and the equilibrium data were best described by the Freundlich isotherm. This indicates a chemisorption process occurring on a heterogeneous surface, consistent with the complex structure created by thermal modification. Notably, post-adsorption pore structure expansion suggested synergistic pore-filling and surface reorganization. This work not only demonstrates a circular economy paradigm for repurposing industrial solid waste on a global scale but also offers a cost-effective and high-performance pathway for controlling phosphorus pollution in aquatic systems, contributing directly to resource efficiency and sustainable environmental remediation.
1. Introduction
Phosphorus (P) is a critical limiting nutrient in freshwater ecosystems, yet its excessive input has triggered widespread eutrophication, representing a pervasive global environmental issue [1,2,3]. The environmental ramifications extend beyond algal blooms and oxygen depletion. Blooms formed by toxin-producing cyanobacteria (e.g., Microcystis) release potent hepatotoxins and neurotoxins, such as microcystins, into water bodies. These toxins threaten aquatic biodiversity, cause large-scale fish mortality, and accumulate within the food web, ultimately posing severe direct and indirect risks to human health through the consumption of contaminated drinking water and aquatic products [4,5].
The challenge of managing phosphorus pollution is global. In the United States, eutrophication affects numerous water bodies, with annual economic damages estimated in the billions of dollars [6]. Similarly, the European Union’s Water Framework Directive identifies nutrient pollution as a key pressure preventing the achievement of good ecological status in its waters [7]. In China, a leading global producer and consumer of phosphate fertilizers, the situation is particularly pressing. According to the Ministry of Ecology and Environment of China, 12.6% of monitored surface water sections exceeded total phosphorus standards in 2023, with increasing loads in major basins like the Yangtze and Pearl Rivers [8], underscoring the urgent need for effective phosphorus control worldwide. In China, the Environmental Quality Standards for Surface Water [9] mandate, for instance, a Total Phosphorus limit of ≤0.2 mg/L for lakes/reservoirs of Class III (centralized drinking water sources), highlighting the severity of the reported exceedances [8].
Concurrently, the phosphate mining industry, essential for global food security, generates enormous amounts of solid waste—phosphate tailings (PT). Global phosphate rock production results in hundreds of millions of tons of tailings annually, posing a universal waste management challenge [10,11]. These stockpiles are not merely an idle resource but a significant environmental liability [12]. Traditional stockpiling occupies vast land areas and poses a continuous risk of secondary pollution through the leaching of endogenous phosphorus and associated contaminants into groundwater and surface water [13,14], thereby exacerbating the eutrophication problem that phosphorus control aims to mitigate. Current recycling pathways for PT, such as secondary beneficiation or use in construction materials, are hampered by low utilization rates (<10%), high energy consumption, and limited economic viability [10,12,15,16,17,18]. This linear “take-make-dispose” model is unsustainable globally, creating a pressing need for innovative strategies to valorize this waste stream while mitigating aquatic pollution.
Conventional phosphorus removal technologies, including chemical precipitation and biological methods, often suffer from high operational costs, sensitivity to operational conditions, and the generation of chemical sludge, which presents disposal challenges and risks of secondary pollution [19,20,21]. Adsorption has emerged as a promising alternative due to its operational simplicity, potential for resource recovery, and often lower environmental footprint [21,22,23,24]. However, the widespread application of common adsorbents—such as activated carbon [24], zeolites [23], and certain tailings-derived ceramsites [25]—is constrained by their limited adsorption capacity (often below 50 mg/g) and insufficient stability in diverse water matrices [19,26]. Although some studies have explored mineral-based adsorbents, the potential of phosphate tailings themselves remains underexploited, particularly following strategic thermal activation that could profoundly enhance their reactivity and structural properties for P sequestration.
Within this global context, we propose a circular “waste-treats-waste” paradigm, transforming the environmental liability of PT into an effective solution for phosphorus removal from eutrophic waters. The central objective of this study is to investigate the feasibility and mechanism of using thermally modified phosphate tailings for enhanced phosphorus adsorption, directly linking solid waste management to water quality improvement. Specific aims include: (1) optimizing the thermal modification conditions (temperature and time) to maximize P adsorption capacity; (2) characterizing the structural and chemical evolution of PT after calcination; and (3) elucidating the adsorption mechanisms through kinetic, isotherm, and advanced spectroscopic analyses.
This work introduces a novel “high-temperature activation—gradient pore construction” strategy to synergistically enhance the adsorption performance of dolomite-rich PT. We demonstrate that calcination at 950 °C triggers the decomposition of carbonates into highly active CaO/MgO and generates a hierarchical pore structure, facilitating high-capacity phosphorus removal predominantly through chemical precipitation and surface complexation. The outcomes of this study are expected to provide a sustainable and cost-effective solution at the nexus of solid waste management and eutrophication control, contributing to safer aquatic ecosystems and advancing circular economy paradigms in the global phosphorus industry.
2. Materials and Methods
2.1. Materials and Reagents
The natural phosphate tailings (PT) used in this study were obtained from Yunnan Phosphate Group Haikou Phosphate Industry Co., Ltd. (Kunming, China). The major chemical composition, determined by X-ray fluorescence (XRF), was as follows: P2O5 (6.67%), MgO (16.26%), CaO (34.14%), SiO2 (4.78%), Fe2O3 (0.76%), and Al2O3 (0.49%). Mineralogical analysis indicated that the tailings were predominantly composed of dolomite (80%), with minor amounts of collophane (15%) and clay minerals (5%). The raw material was sieved through a 200-mesh sieve, with 95% of particles below 75 μm, and exhibited a pH between 8.3 and 8.6.
A stock phosphorus solution (1000 mg P/L) was prepared using analytical-grade potassium dihydrogen phosphate (KH2PO4). Working solutions of desired concentrations were obtained by dilution. The pH of solutions was adjusted using 0.1 M HCl or NaOH. All chemicals were of analytical grade and used without further purification.
2.2. Thermal Modification of Phosphate Tailings
The raw PT was thermally treated in a programmable muffle furnace (YTH-5-12, Supo Instrument Co., Ltd., Shaoxing, China). Samples were calcined in alumina crucibles under static air conditions, using a heating rate of 10 °C/min. To optimize the modification process, the calcination temperature was varied from 500 to 1050 °C and the holding time from 10 min to 3 h. Based on preliminary adsorption tests, the optimal conditions were determined to be 950 °C for 1 h. The resulting product, denoted as MPT (modified phosphate tailings), was cooled to room temperature in a desiccator and stored in sealed containers for further use.
2.3. Characterization Techniques
The chemical composition of raw and modified tailings was analyzed using an Axios mAX wavelength-dispersive XRF spectrometer (XRF; Malvern Panalytical B.V., Almelo, The Netherlands). Mineral phases were identified by X-ray diffraction (XRD; X’pert3 Powder, Malvern Panalytical B.V., Almelo, The Netherlands). Microstructural morphologies were observed using a TESCAN VEGA3 tungsten filament scanning electron microscope (SEM; TESCAN ORSAY HOLDING, a.s., Brno, Czech Republic). Fourier transform infrared (FTIR) spectra were recorded on Tensor 27 (Bruker Optik GmbH, Karlsruhe, Germany). A spectrophotometer (UV-754N; Shanghai Jinghua Science & Technology Instruments Co., Ltd., Shanghai, China) was used to determine phosphorus concentration. Surface area and pore characteristics were determined by N2 adsorption–desorption at 77 K using a Micromeritics ASAP 2460 analyzer (Micromeritics Instrument Corporation, Norcross, GA, USA). The specific surface area was calculated using the Brunauer–Emmett–Teller (BET) method [27], and pore size distribution was derived from the Barrett–Joyner–Halenda (BJH) model [28].
2.4. Phosphorus Release and Adsorption Experiments
2.4.1. Phosphorus Release Test
To evaluate the endogenous phosphorus release potential, 0.2 g of raw PT was added to 25 mL of deionized water (initial pH = 6) in a 50 mL centrifuge tube. The mixture was shaken at 200 rpm and 37 °C. At predetermined time intervals (1, 12, 24, 48, 72, 120, and 144 h), samples were withdrawn, filtered through a 0.45 μm membrane, and the dissolved phosphorus concentration was measured.
2.4.2. Batch Adsorption Experiments
Adsorption kinetics and isotherms were studied using a batch equilibration technique. Typically, 0.2 g of adsorbent (raw PT or MPT) was added to 25 mL of phosphate solution with varying initial concentrations (1–1000 mg P/L) in capped polyethylene tubes. The pH was adjusted to 6.0 using 0.1 M HCl or NaOH, unless otherwise stated. The tubes were shaken at 200 rpm and 37 °C in a constant-temperature incubator shaker (THZ-100, Shanghai Yiheng Instruments Co., Ltd., Shanghai, China). The solution pH was monitored in selected experiments. For MPT at an initial pH of 6.0, the final equilibrium pH typically ranged from 9.0 to 10.5, indicating the alkaline nature of the material and the dissolution of CaO/MgO.
After adsorption, the suspensions were filtered, and the residual phosphorus concentration was determined using the ammonium molybdate spectrophotometric method (ascorbic acid as reductant) measuring absorbance at 700 nm [29], using a UV-754N spectrophotometer. The adsorption capacity q (mg/g) was calculated as:
where C0 and Ce are the initial and equilibrium phosphorus concentrations (mg/L), respectively, V is the solution volume (L), and m is the mass of adsorbent (g). All experiments were performed in triplicate.
2.5. Adsorption Kinetics and Isotherms
Adsorption kinetics were evaluated by sampling at time intervals from 1 to 24 h. The data were fitted to pseudo-first-order (Equation (2)) [30] and pseudo-second-order (Equation (3)) models [31,32]:
where k1: pseudo-first-order adsorption rate constant (min−1); k2: pseudo-second-order adsorption rate constant (g/mg/min); qt: adsorption capacity at time t (mg/g); qₑ: equilibrium adsorption capacity (mg/g).
qt = qe(1 − e−k1t)
qt = (k2qe2t)/(1 + k2qet)
Adsorption isotherms were obtained at varying initial P concentrations and fitted using Langmuir [33] and Freundlich models [34]. The nonlinearized forms of the models are given as follows:
Langmuir model:
where Qe: equilibrium adsorption capacity (mg/g); qm: maximum adsorption capacity (mg/g); Ce: equilibrium concentration of the adsorbate in solution (mg/L); KL: Langmuir adsorption equilibrium constant (L/mg).
qe = (qmkLce)/(1 + kLce)
Freundlich model:
where Qe: equilibrium adsorption capacity (mg/g); KF: Freundlich adsorption capacity constant (mg/g)·(L/mg)1/n; Ce: equilibrium concentration of adsorbate in solution (mg/L); n: adsorption constant. The value of the adsorption constant n reflects the adsorption capability of the adsorbent. Generally, the value of 1/n falls between 0 and 1.
qe = kFce1/n
2.6. Statistical Analysis
Data processing and graph plotting were conducted using WPS Office 2023 Professional Edition and Origin (Version 2021b), respectively. XRD patterns were analyzed with MDI Jade software (Version 6.5). All experimental values are reported as mean ± standard deviation. During the preparation of this manuscript, we used DeepSeek-V3.2 for translating the text from Chinese to English and for revising the manuscript, particularly in the Discussion section.
3. Results and Discussion
3.1. Analysis of Endogenous Phosphorus Release and Adsorption Performance of Raw Phosphate Tailings
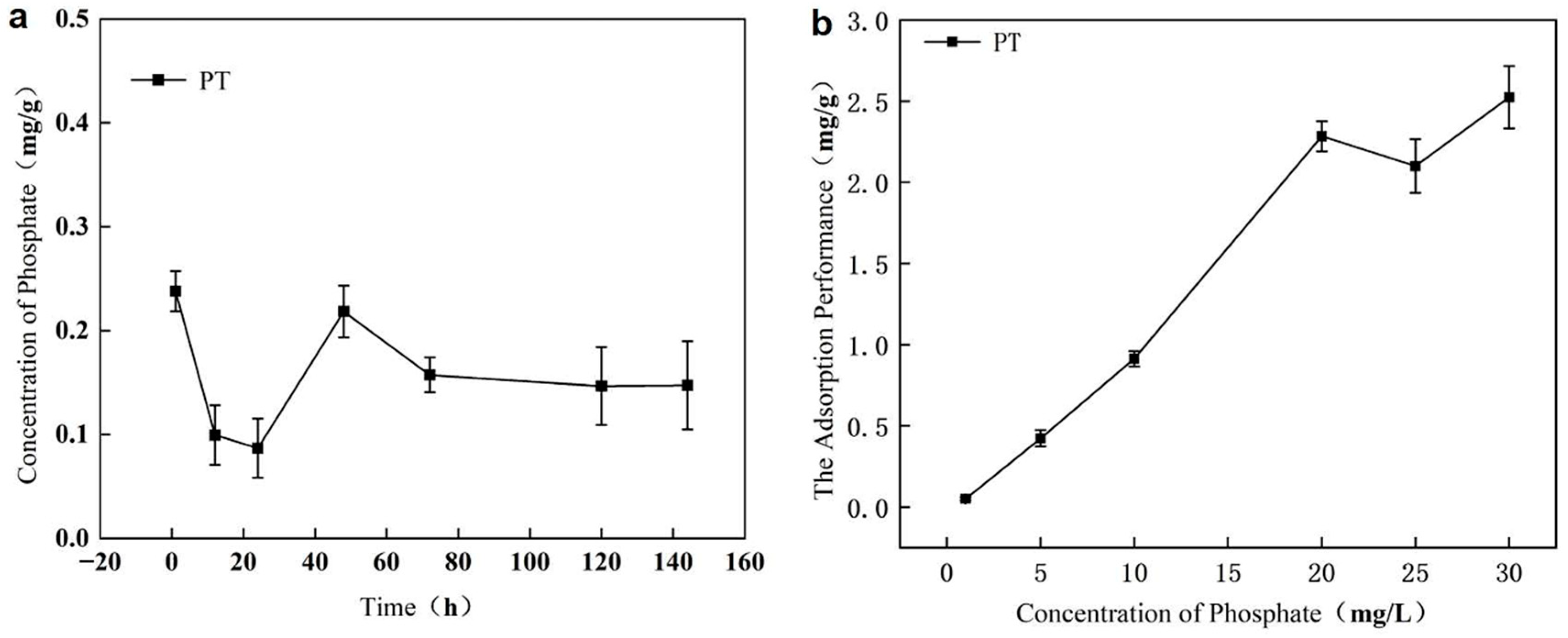
The environmental behavior of raw phosphate tailings (PT) was evaluated through release and adsorption experiments. As shown in Figure 1a, PT continuously released endogenous phosphorus into water, with concentrations ranging between 0.086 and 0.238 mg/g, indicating a persistent risk of secondary phosphorus pollution. In contrast, the adsorption capacity of raw PT was quite limited. Under pH 6 and initial P concentrations of 1–30 mg/L, adsorption equilibrium was reached only after 48 h, with a maximum capacity of 2.52 mg/g (Figure 1b). These results confirm that unmodified PT acts as a source of phosphorus pollution rather than an effective adsorbent, underscoring the necessity of modification for practical application.
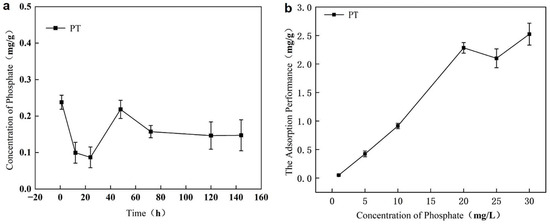
Figure 1.
Assessment of raw phosphate tailings (PT): (a) as a persistent source of secondary phosphorus release; (b) as an inefficient adsorbent with limited capacity and slow kinetics.
3.2. Effect of Calcination Temperature and Time on Adsorption Performance
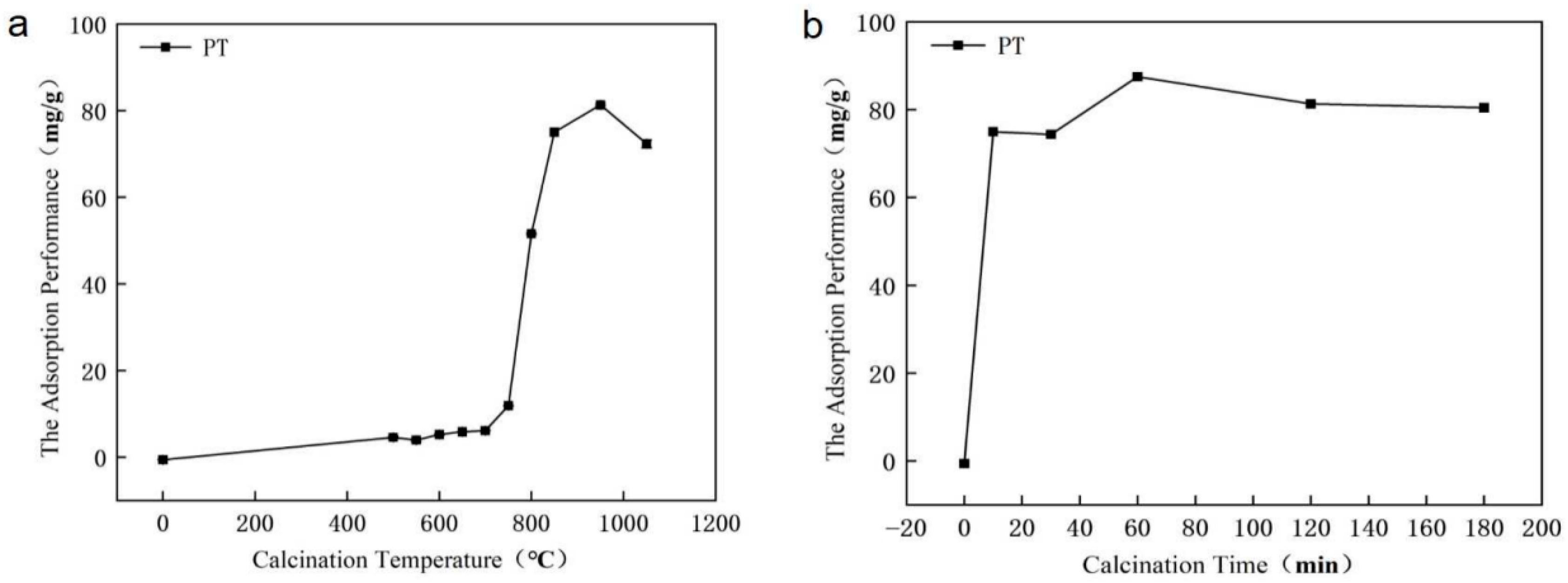
As illustrated in Figure 2a, the phosphorus adsorption capacity of PT exhibited a distinct trend with increasing calcination temperature (2 h holding time, at an initial P concentration of 700 mg/L. In the low-temperature range (0–700 °C), the adsorption capacity increased only gradually, reaching 6.14 mg/g at 700 °C. A remarkable enhancement was observed between 700 °C and 950 °C, where the adsorption capacity rose sharply to a maximum of 81.29 mg/g at 950 °C. This significant improvement is attributed to the thermal decomposition of dolomite [CaMg(CO3)2] into highly active CaO and MgO, which provide abundant sites for phosphorus chemisorption. However, when the temperature exceeded 950 °C, the adsorption capacity declined, likely due to sintering-induced pore collapse and aggregation of active phases, resulting in reduced surface reactivity.
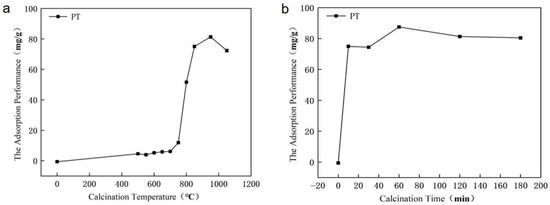
Figure 2.
Variation in phosphorus adsorption capacity of phosphate tailings with (a) different calcination temperatures (2 h holding time) and (b) various calcination times (at 950 °C).
The influence of calcination time was investigated at the optimal temperature of 950 °C. As shown in Figure 2b, the adsorption capacity first increased and then decreased with prolonged duration. A calcination time of 10 min yielded a considerable capacity of 74.94 mg/g. The maximum adsorption capacity of 87.48 mg/g was achieved after 1 h. Further extension to 3 h led to a decrease to 80.40 mg/g, suggesting that prolonged exposure to high temperature may induce structural degradation or phase transformation that diminishes adsorption activity.
These results clearly identify 950 °C for 1 h as the optimal condition for thermal modification, effectively balancing the generation of active sites and the preservation of structural integrity.
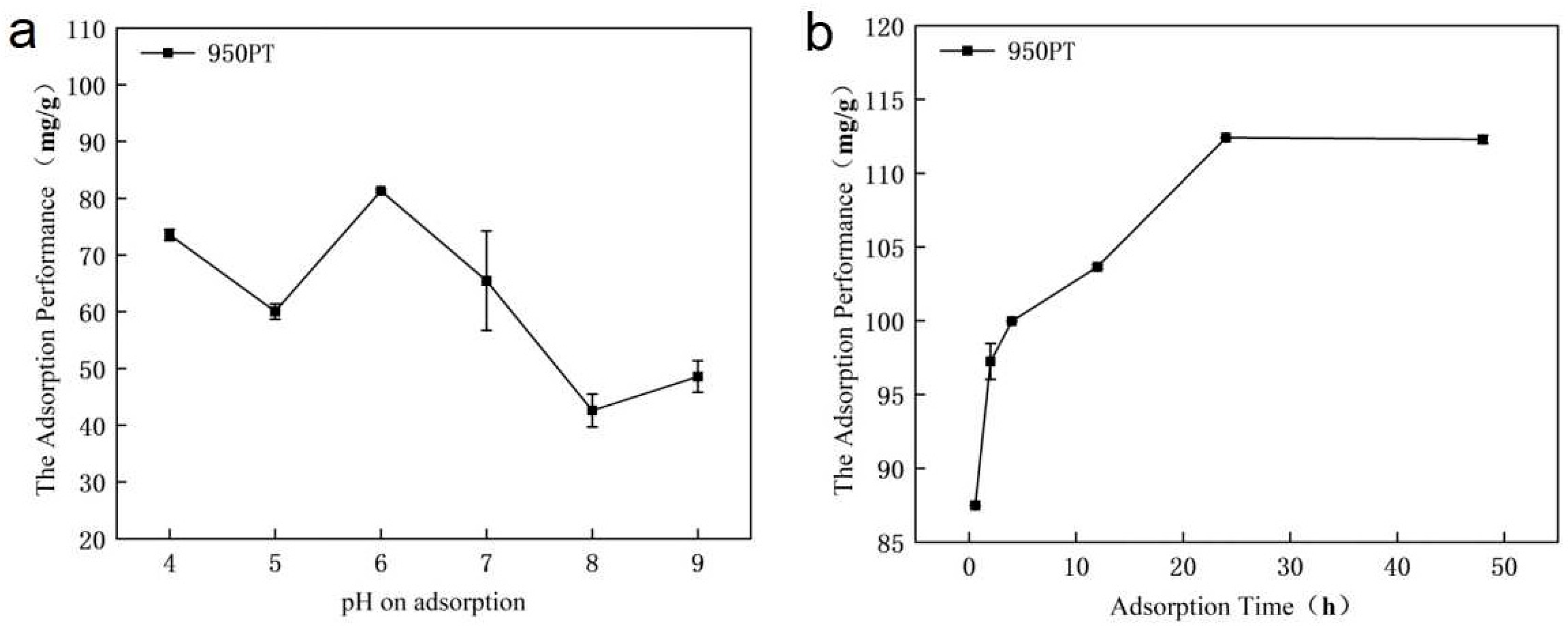
3.3. Effect of Solution pH and Adsorption Time on Adsorption Performance of Modified Phosphate Tailings
The phosphorus adsorption performance of modified phosphate tailings (MPT) was highly dependent on solution pH and contact time (Figure 3). The maximum adsorption capacity (87.48 mg/g) was achieved at pH 6 with 700 mg p/L, where surface protonation enhanced electrostatic attraction and promoted the dissolution of CaO/MgO to release Ca2+/Mg2+ ions for phosphate precipitation. Under alkaline conditions, competition from OH- ions and decreased metal solubility reduced adsorption efficiency. Adsorption kinetics showed rapid initial uptake, reaching 87.49 mg/g within 1 h, followed by gradual attainment of equilibrium at 112.41 mg/g after 24 h. The process involved initial surface complexation followed by intra-particle diffusion and precipitate formation. These results demonstrate that pH 6 and 24 h contact time are optimal for efficient phosphorus removal using MPT.
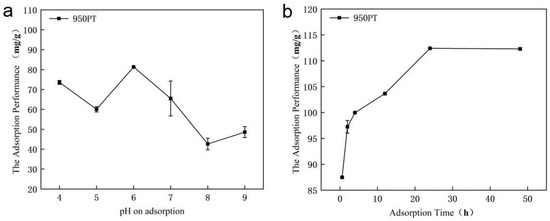
Figure 3.
Effect of (a) pH and (b) adsorption time on phosphorus adsorption by modified phosphate tailings.
3.4. Characterization Analysis of Adsorption Mechanism for Modified Phosphate Tailings
3.4.1. Scanning Electron Microscopy (SEM) Analysis
SEM analysis of the microstructure of phosphate tailings (Figure 4) showed that raw PT exhibited blocky or rod-like agglomerates with dense surface structures and low porosity (Figure 4a). After calcination at 950 °C for 1 h, the material morphology changed significantly, showing increased surface roughness, a fractured and loosened structure, and hierarchical pore networks (Figure 4c). Post-adsorption SEM comparison revealed dense layered deposits on unmodified samples (Figure 4b), while modified samples showed reduced pore volume and surface densification (Figure 4d). These morphological changes confirm that calcination improved the pore structure, suggesting that phosphate adsorption occurs via dual mechanisms: chemical deposition within pores and surface reconstruction.

Figure 4.
SEM images of (a) raw phosphate tailings (×10,000); (b) raw phosphate tailings after adsorption (×10,000); (c) modified phosphate tailings (×10,000); (d) modified phosphate tailings after adsorption (×10,000).
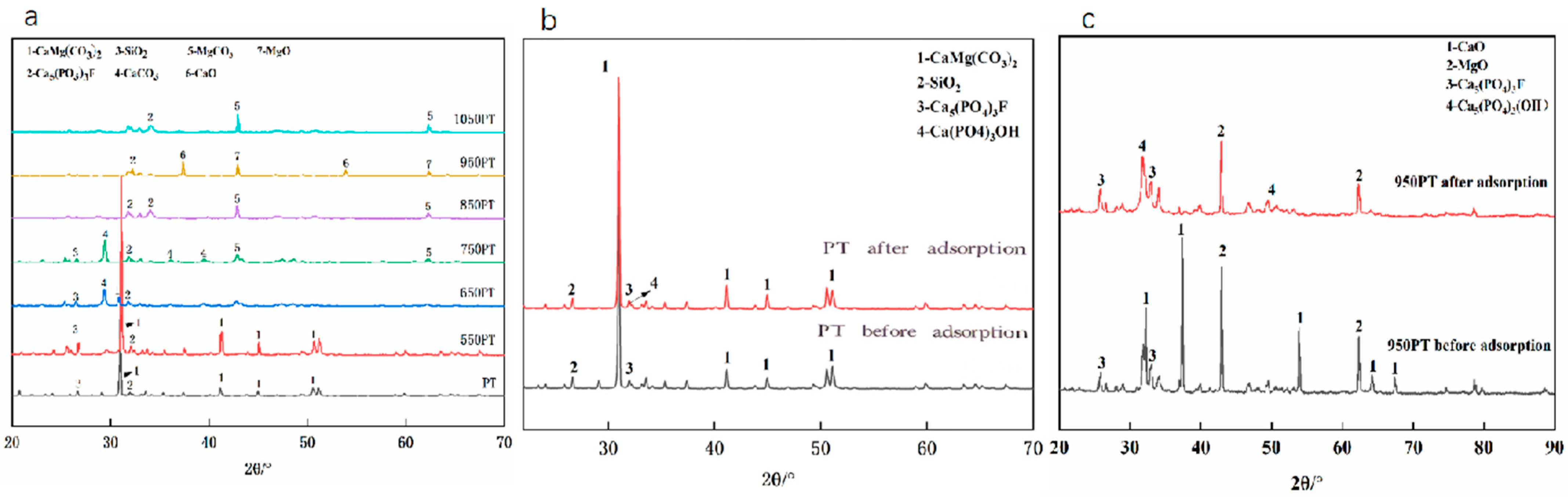
3.4.2. X-Ray Diffraction (XRD) Analysis
The phase evolution of phosphate tailings during calcination and adsorption was systematically analyzed by XRD (Figure 5). The primary phase of the raw PT (Figure 5a) was dolomite (CaMg(CO3)2), with minor mineral phases including fluorapatite (Ca5(PO4)3F) and quartz (SiO2). During pyrolysis, treatment at 550 °C did not induce significant phase changes. At 650 °C, the intensity of the dolomite characteristic peaks decreased, and diffraction peaks of calcite (CaCO3) appeared. Calcination at 750 °C led to the complete decomposition of dolomite, forming a phase composition dominated by calcite along with magnesite (MgCO3). High-temperature treatment at 950 °C completely transformed the carbonate phases into calcium oxide (CaO) and magnesium oxide (MgO). Notably, fluorapatite and quartz remained stable throughout the pyrolysis process. XRD analysis after adsorption experiments (Figure 5b,c) revealed that the characteristic peak intensities of CaO/MgO in the modified samples significantly decreased, while distinct characteristic peaks of hydroxyapatite (Ca5(PO4)3OH) emerged. The peak intensity of hydroxyapatite was much higher than that in the raw PT samples, confirming that phosphate was firmly fixed on the material surface through chemical bonding, forming a stable calcium phosphate mineral phase.
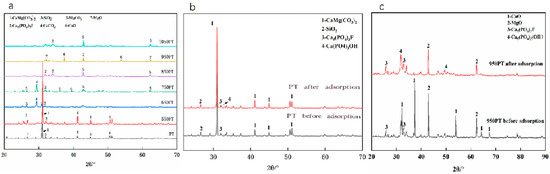
Figure 5.
XRD patterns of (a) phosphate tailings calcined at different temperatures; (b) raw tailings vs. phosphorus removal products; (c) modified tailings vs. phosphorus removal products.
3.4.3. Fourier Transform Infrared Spectroscopy (FTIR) Analysis
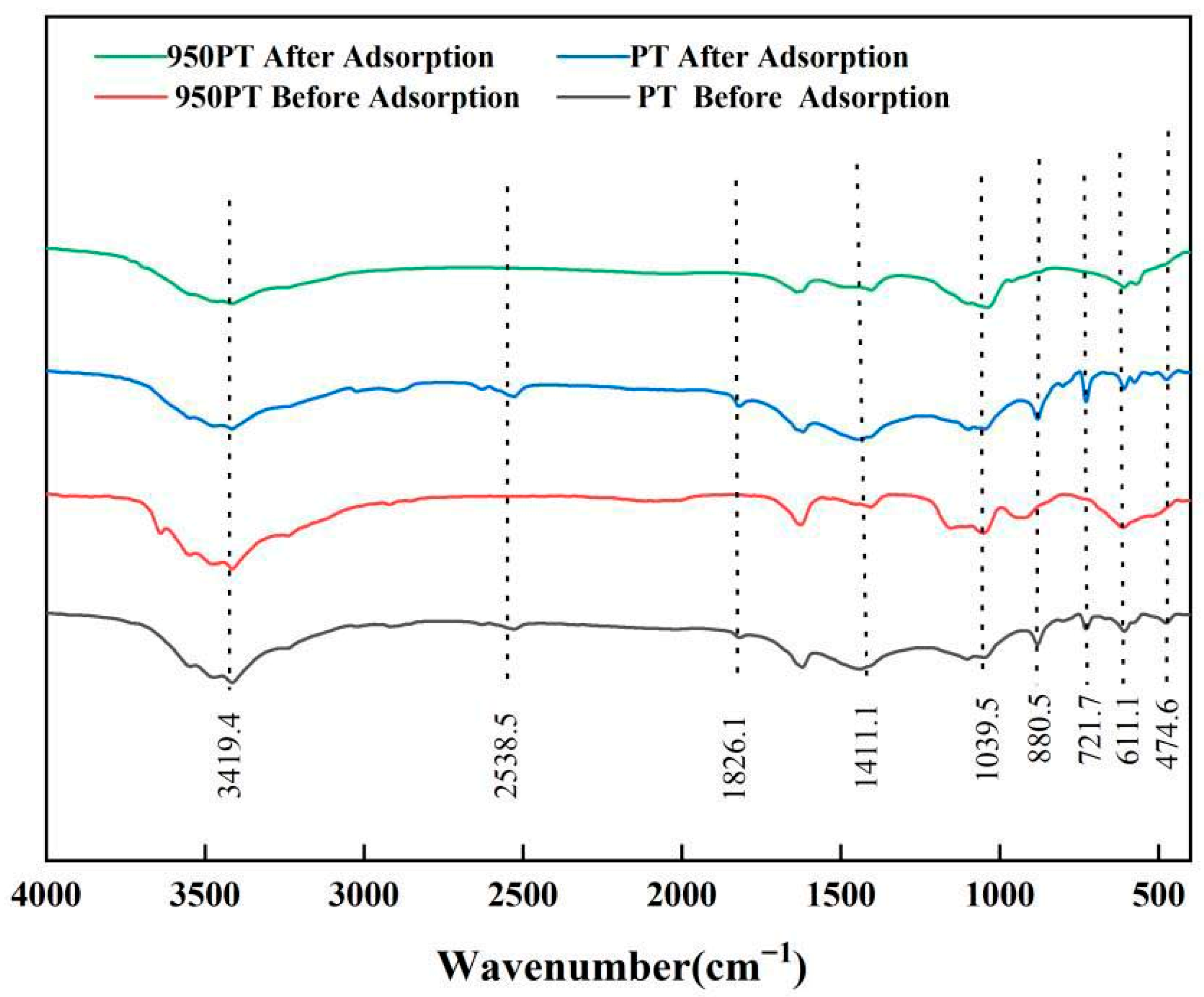
FTIR was employed to characterize changes in chemical bonds of raw and modified PT (950 °C) before and after phosphate adsorption (Figure 6). All samples exhibited characteristic absorption peaks at wavenumbers of 3419.4, 2538.5, 1826.1, 1411.1, 1039.5, 880.5, 721.7, 611.1, and 474.4 cm−1. The peaks at 1039.5, 611.1, and 474.4 cm−1 were attributed to P–O stretching vibrations, confirming the presence of phosphate components. The peaks at 2538.5, 1826.1, 1411.1, 880.5, and 721.7 cm−1 were associated with CO32− vibrational modes [35,36]. The intensity of the 2538.5 cm−1 peak decreased significantly in the calcined sample, indicating that high-temperature treatment promoted carbonate decomposition. The 1826.1 cm−1 peak (characteristic of calcite and dolomite) completely disappeared after 950 °C treatment, confirming the complete conversion of carbonates into CaO and MgO.
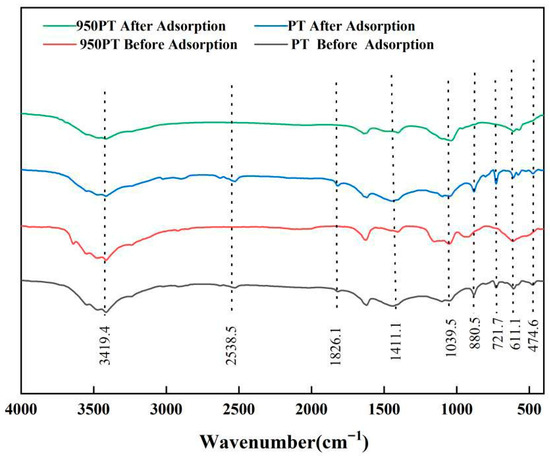
Figure 6.
FTIR spectra of phosphate tailings, modified phosphate tailings, and their phosphorus removal products.
The 1411.1 cm−1 peak (related to the crystal structure of dolomite and calcite) weakened after adsorption, suggesting partial dissolution of carbonates and their participation in phosphate adsorption. The 880.5 cm−1 peak was strong in the unmodified sample but significantly weakened after calcination, further confirming carbonate decomposition. The 721.7 cm−1 peak likely originated from the Si–O–Si bending vibration of quartz (SiO2), showing little change before and after adsorption, indicating the stability of the quartz phase.
The broad peak at 3419.4 cm−1 was assigned to O–H stretching vibration [20], and its broadening suggested the presence of hydrogen bonding or surface-adsorbed water molecules. Notably, this peak intensified significantly in the modified sample after phosphate adsorption, directly correlating with the formation of hydroxyapatite (Ca10(PO4)6(OH)2). This provides direct evidence that phosphate was immobilized through a chemical precipitation mechanism.
3.4.4. Specific Surface Area and Porosity (BET) Analysis
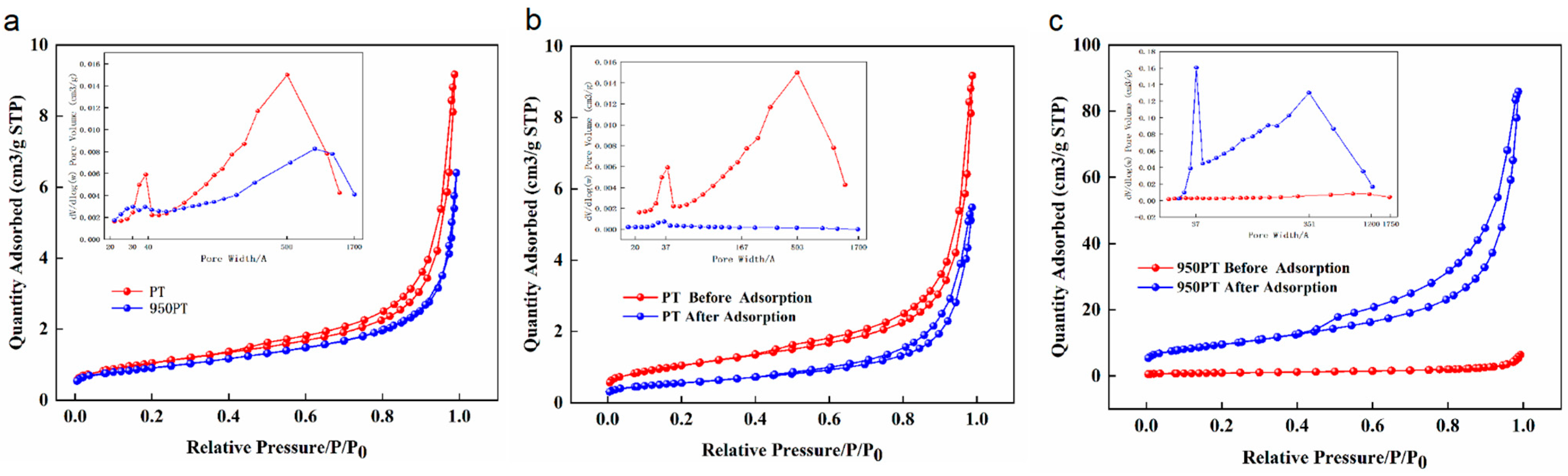
The pore structures of raw PT, modified PT (950 PT), and their phosphorus-adsorbed counterparts were characterized through N2 adsorption–desorption experiments (Figure 7). The specific surface area and pore structure parameters are summarized in Table 1.
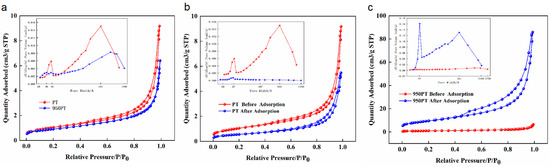
Figure 7.
BET analysis of raw and modified phosphate tailings before and after phosphorus removal. (a): Raw phosphate tailings and modified phosphate tailings; (b): Raw phosphate tailings before and after phosphorus removal; (c): Modified phosphate tailings before and after phosphorus removal.

Table 1.
Specific surface area and pore structure of phosphate tailings and modified phosphate tailings.
The raw PT exhibited a specific surface area of 3.76 m2/g, a pore volume of 0.014 cm3/g, and an average pore diameter of 150.9 Å, consistent with typical dense mineral features and indicating a predominantly mesoporous structure (20–500 Å) with overall low pore development. After phosphorus adsorption, the specific surface area slightly decreased to 3.32 m2/g, while the pore volume increased to 0.020 cm3/g and the average pore diameter expanded to 247.6 Å.
Calcination modification resulted in pore structural changes: the 950 PT sample before adsorption showed a specific surface area of 3.20 m2/g, a pore volume of 0.010 cm3/g, and an average pore diameter of 123.6 Å. Compared to the raw tailings, the pore size distribution shifted toward larger diameters, suggesting that calcination generated a hierarchical meso-macroporous structure through high-temperature decomposition and mineral phase transformation.
Notably, after phosphorus adsorption, the modified tailings demonstrated a substantial increase in specific surface area to 34.05 m2/g, with the pore volume rising to 0.133 cm3/g and the average pore diameter expanding to 156.0 Å. This substantial increase in specific surface area and pore volume post-adsorption is unlikely to stem from the original tailings structure. It may be attributed to the formation of a porous layer of phosphate precipitates (e.g., hydroxyapatite) on the adsorbent surface, which contributes its own micro/mesoporosity to the N2 physisorption measurement. Similar observations of increased surface area due to the porous nature of in situ formed precipitates have been reported in the literature for other adsorbent systems [37]. This suggests that phosphorus immobilization involves significant surface reconstruction and the development of a secondary porous phase.
3.4.5. Adsorption Kinetics
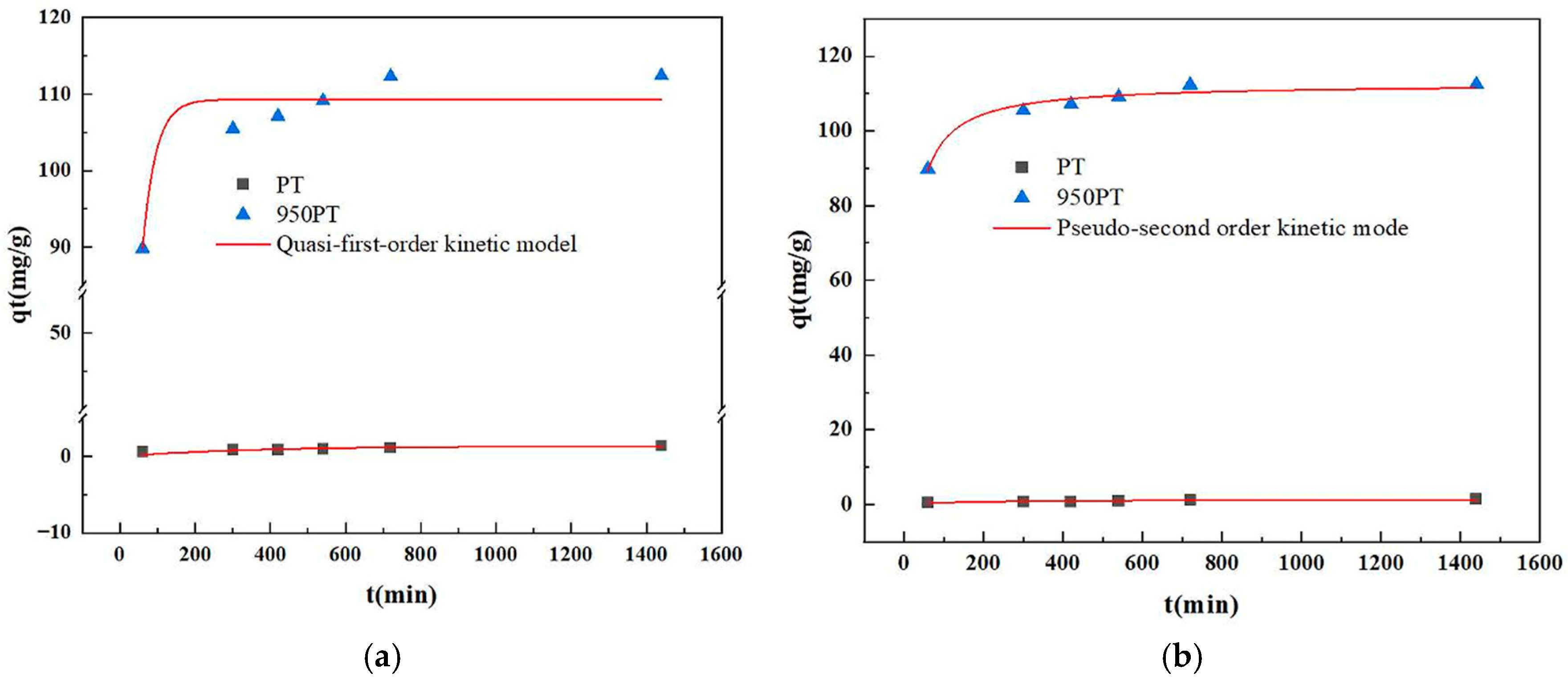
The adsorption kinetics of phosphate onto raw and modified PT were comprehensively evaluated using both pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models (Figure 8). To ensure robustness and model fidelity, the experimental data were analyzed via nonlinear regression analysis, which provides a more statistically rigorous fitting by directly minimizing the sum of squared residuals without the potential distortions introduced by linear transformation.
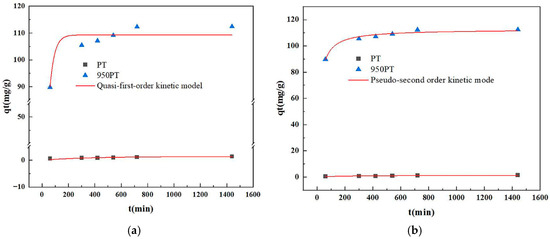
Figure 8.
Nonlinear (a) pseudo-first-order and (b) pseudo-second-order kinetic model fittings for phosphate adsorption on raw and modified phosphate tailings.
The results unequivocally demonstrated that the phosphate adsorption process was best described by the pseudo-second-order (PSO) kinetic model for both raw PT and, most significantly, for the modified PT (MPT). As detailed in Table 2, the nonlinear PSO model achieved near-perfect agreement with the experimental data for MPT, with a correlation coefficient (R2) of 0.9636. The equilibrium adsorption capacity qe,ca predicted by this model was 112.70 mg/g, which is in remarkable agreement with the experimentally measured value qe,exp of 112.43 mg/g. In contrast, the nonlinear PFO model yielded a substantially lower R2 of 0.8654 for MPT, and its predicted qe,cal (109.30 mg/g) showed a greater deviation from the experimental value.

Table 2.
Nonlinear fitting parameters of adsorption kinetics for phosphate tailings and modified phosphate tailings.
For the raw PT, the PSO model (R2 = 0.6256) remained descriptively superior to the PFO model (R2 = 0.4422). The qe,cal from the nonlinear PSO model (1.39 mg/g) was also closer to the measured qe,exp (1.40 mg/g) than the PFO prediction.
The superior fit of the pseudo-second-order model, particularly as confirmed through rigorous nonlinear regression, strongly indicates that chemisorption is the predominant rate-limiting step governing the phosphate adsorption process onto the modified tailings. This mechanism involves valence forces through the sharing or exchange of electrons between the phosphate anions and the active sites (CaO/MgO) on the MPT surface, leading to surface complexation and subsequent precipitation. The transition from a poor kinetic fit for raw PT to an excellent fit for MPT underscores the fundamental change in the adsorption mechanism induced by thermal modification, transforming the material from a passive solid with limited interaction into a highly reactive adsorbent governed by chemical reactions.
3.4.6. Adsorption Isotherms
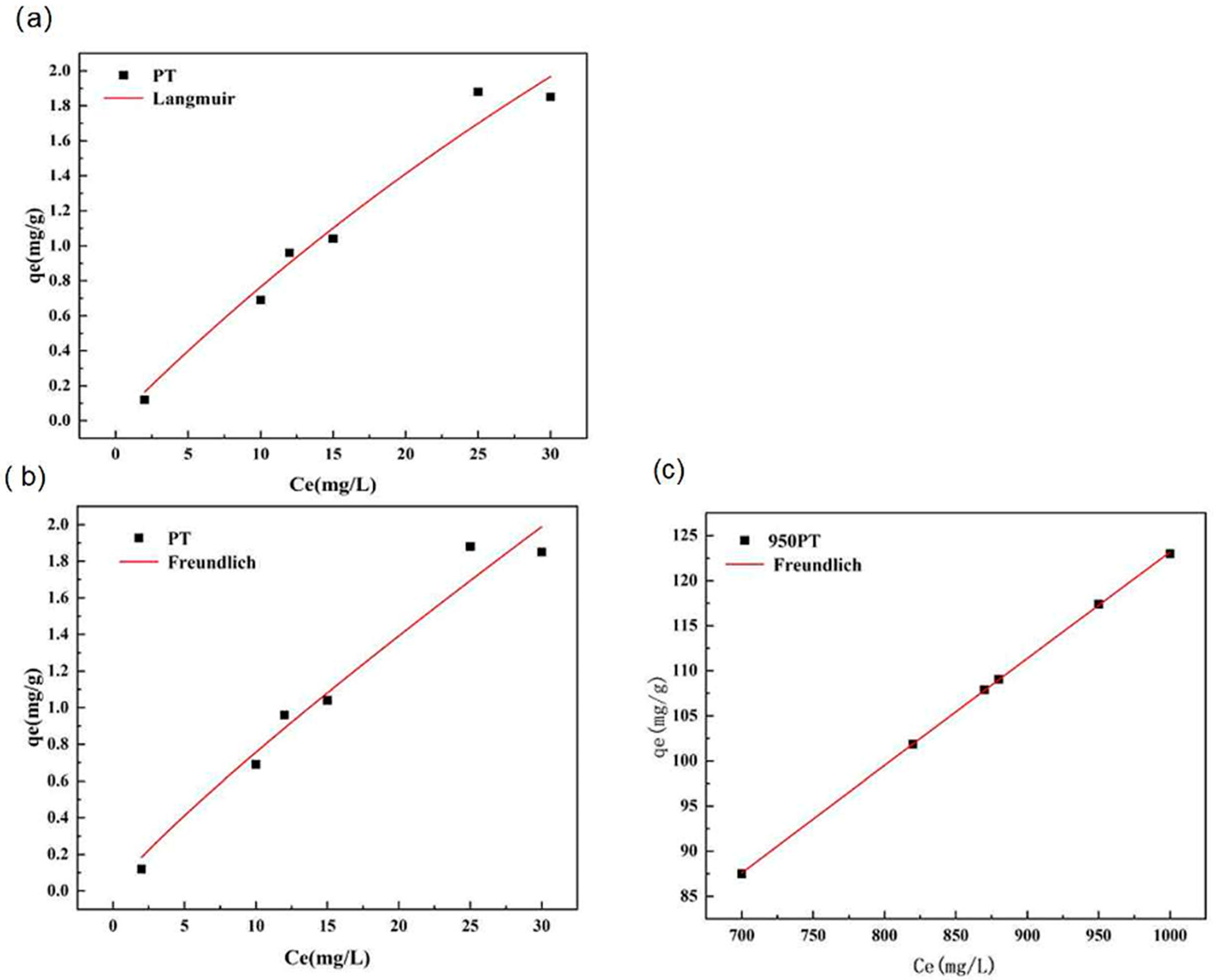
The equilibrium adsorption characteristics of phosphate on raw and modified PT were analyzed using nonlinear regression of the Langmuir and Freundlich isotherm models (Figure 9). Nonlinear fitting was employed as it avoids the inherent biases and statistical limitations associated with the linear transformation of isotherm equations, providing a more accurate estimation of model parameters.
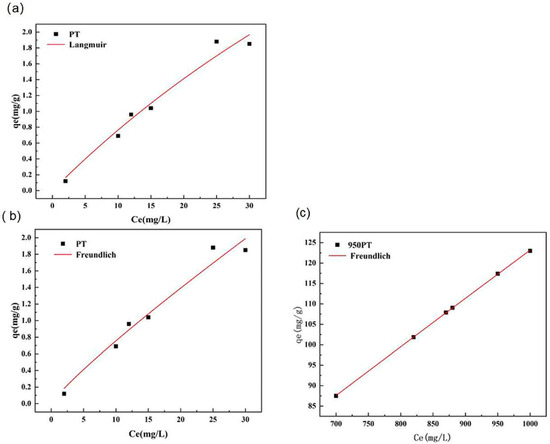
Figure 9.
Nonlinear adsorption isotherm fittings for phosphate tailings and modified phosphate tailings: (a) Langmuir model; (b,c) Freundlich model.
The fitting results revealed a distinct difference between the two adsorbents. For raw phosphate tailings, both the Langmuir and Freundlich models provided a reasonable description of the adsorption data through nonlinear regression (R2 values of 0.9673 and 0.6278, respectively). The Langmuir model predicted a maximum adsorption capacity qm of 9.26 mg/g, which is notably higher than the experimentally observed value of 1.85 mg/g. This discrepancy, along with the moderate Freundlich fit, suggests that the adsorption onto the unmodified material may not strictly follow a simple monolayer mechanism on a homogeneous surface.
In striking contrast, for the thermally modified phosphate tailings (MPTs), the Freundlich model achieved an exceptional fit with a correlation coefficient (R2) of 0.9999 via nonlinear regression. The Freundlich intensity parameter n was 1.046, indicating a highly favorable adsorption process. The model’s description aligns perfectly with the experimental data across the entire concentration range studied. Conversely, attempts to fit the MPT data to the nonlinear Langmuir model failed to converge. This failure indicates that the model’s assumptions—particularly that of a homogeneous surface with identical, non-interacting adsorption sites—are invalid for the modified adsorbent. The system is likely “over-parameterized” for the Langmuir form because the underlying adsorption behavior is more complex.
The superior and robust fit of the Freundlich model to the MPT data is highly significant. It conclusively demonstrates that phosphate adsorption onto the MPT occurs on a heterogeneous surface with sites of different adsorption energies. This heterogeneity is a direct consequence of the thermal modification process, which created a complex assemblage of active phases (CaO, MgO), a hierarchical pore structure, and varied surface chemistries, as evidenced by XRD, SEM, and BET analyses. The adsorption process, therefore, is not a simple monolayer coverage but involves multiple concurrent mechanisms, including chemical precipitation within pores and surface complexation on different active sites, consistent with the kinetic findings. The excellent Freundlich fit quantitatively captures this multifaceted adsorption behavior, which the Langmuir model cannot adequately represent.
4. Discussion
This study demonstrates that high-temperature calcination can transform phosphate tailings (PT), a widespread industrial solid waste, into a high-performance adsorbent for phosphorus (P) removal. Under optimal conditions (950 °C for 1 h, pH = 6, initial concentration of 700 mg p /L), the adsorption capacity of the modified tailings (MPT) reached 112.41 mg/g, representing an approximately 45-fold improvement over the raw material (2.52 mg/g). The adsorption process was well-described by the Langmuir isotherm and pseudo-second-order kinetic models, indicating a chemisorption-dominated, monolayer mechanism, with a theoretical maximum capacity derived from the Langmuir model fitting of 136.4 mg/g. The following sections discuss the structural evolution of the material, the adsorption mechanisms, and contextualize the performance of MPT within the global landscape of P-adsorbents, evaluating its scientific and practical significance.
4.1. Effect of High-Temperature Modification on Material Structure and Adsorption Performance
Under the optimized conditions (pH = 6, initial P concentration of 700 mg/L), the experimentally determined equilibrium adsorption capacity of MPT was 112.41 mg/g. The theoretical maximum adsorption capacity derived from Langmuir model fitting was 136.4 mg/g, while the value of 81.29 mg/g shown in Figure 2a represents a typical measurement obtained under the standard test initial concentration 700 mg/L) and was used for analyzing the influence of process parameters.
Calcination temperature was a critical factor determining the adsorption performance. The capacity increased markedly from 6.14 mg/g at 700 °C to 81.29 mg/g at 950 °C (Figure 2a). This enhancement is closely associated with the material’s phase transformation. XRD analysis (Figure 5a) confirmed that high-temperature treatment promoted the decomposition of dolomite (CaMg(CO3)2) into highly active CaO and MgO. These oxides provide abundant sites for phosphate chemisorption and subsequent precipitation of stable hydroxyapatite (Ca5(PO4)3OH), a mechanism consistently reported for other calcined magnesite slags [38]. Furthermore, SEM observations (Figure 4c) revealed that calcination developed a rougher surface and a hierarchical pore structure, increasing the accessibility of active sites [39]. However, exceeding 950 °C led to a decline in performance (Figure 2a), likely due to sintering-induced pore collapse and a reduction in active surface area, a phenomenon well-documented for other metal oxide adsorbents [40]. BET results indicated a decrease in specific surface area and micropore volume after modification at 950 °C, which aligns with findings from Feng et al. [41] on similarly calcined materials. This suggests that the dramatically enhanced adsorption is primarily attributable to the creation of highly reactive chemical sites (CaO/MgO) rather than an increase in physical surface area.
4.2. Analysis of Adsorption Kinetics and Mechanisms
The adsorption characteristics of MPT were rigorously analyzed using nonlinear regression. Kinetic data were excellently described by the pseudo-second-order model (R2 > 0.96, Figure 8b), with the predicted equilibrium capacity (112.70 mg/g) closely matching the experimental value (112.43 mg/g, Table 2). This confirms chemisorption as the dominant, rate-limiting step. More profoundly, equilibrium isotherm data provided critical mechanistic insight. Nonlinear fitting revealed a near-perfect fit to the Freundlich model (R2 = 0.9999, Table 3), while the Langmuir model failed to converge. This outcome is highly significant: the failure of the Langmuir model invalidates its assumption of a homogeneous surface, whereas the exceptional Freundlich fit unequivocally demonstrates adsorption on an energetically heterogeneous surface. This heterogeneity is a direct manifestation of the complex material architecture created by calcination, as seen in the hierarchical pore structure (Figure 4c) and the multi-phase composition (Figure 5a).

Table 3.
Nonlinear fitting parameters of adsorption isotherm models for phosphate tailings and modified phosphate tailings.
The synergy between kinetic and isotherm findings clarifies the mechanism. The PSO kinetics confirm a chemisorption-dominated process. The Freundlich isotherm specifies that this process occurs on a heterogeneous landscape, aligning with a multi-mechanism pathway. This is corroborated by direct spectroscopic evidence: the post-adservation emergence of distinct hydroxyapatite peaks in XRD (Figure 5c) and intensified P–O bands in FTIR (Figure 6) confirm chemical precipitation. Therefore, phosphate immobilization is driven by a synergistic sequence of surface complexation and chemical precipitation across the heterogeneous reactive surface of MPT, moving beyond simple monolayer coverage to a more complex, multi-faceted adsorption and nucleation process.
While this study indirectly confirmed the crucial role of Ca2+/Mg2+ ions by directly detecting hydroxyapatite formation in the solid phase via XRD and FTIR, future work could provide more direct evidence from the liquid phase for this ‘dissolution-precipitation’ mechanism by monitoring the dynamic changes in Ca2+ and Mg2+ concentrations in solution during adsorption.
4.3. Performance Comparison with Other Adsorbents and Practical Implications
The performance of an adsorbent is comprehensively evaluated based on its efficacy, cost, and environmental footprint. As summarized in Table 4, the theoretical maximum phosphate adsorption capacity of thermally modified phosphate tailings (MPT), derived from Langmuir model fitting, reaches 136.4 mg/g. This capacity is highly competitive, significantly surpassing that of many conventional and low-cost materials, such as activated alumina [42,43,44], thermally treated bentonite [26,45], and unmodified industrial by-products like raw phosphate tailings. Notably, it also compares favorably with or even exceeds several specially engineered adsorbents, including La-modified zeolites (52.2 mg/g) [46].
The key advantage of MPT lies not only in its high adsorption capacity but also in its sustainable origin and straightforward synthesis. Unlike many high-performance adsorbents that rely on expensive precursors (e.g., MOFs) [47] or Layered Double Hydroxide (LDH) [48], MPT is derived entirely from a solid waste stream. This embodies a direct “waste-treats-waste” circular economy strategy, simultaneously addressing the dual environmental challenges of waste stockpile management and aquatic eutrophication control. The modification process, although energy-intensive, is simple and potentially scalable, involving a single-step thermal treatment without the need for complex chemical reagents or synthesis procedures. This positions MPT as a promising, cost-effective, and sustainable alternative, particularly for regions grappling with abundant phosphate tailings and rising concerns over water quality [49,50].

Table 4.
Comparison of the maximum phosphorus adsorption capacity of MPT with other adsorbents reported in the literature.
Table 4.
Comparison of the maximum phosphorus adsorption capacity of MPT with other adsorbents reported in the literature.
| Adsorbents | Maximum P Adsorption | Key Notes/Drawbacks | Reference |
|---|---|---|---|
| Capacity (mg/g) | |||
| Thermally Modified Phosphate | 136.4 | Waste-derived, single-step calcination | This study |
| La-modified Zeolite | 52.2 | Requires rare-earth element (La) | [46] |
| Layered Double Hydroxide (LDH) | 80–245 | High capacity, but synthesis can be complex | [48] |
| Metal–Organic Frameworks (MOFs) | 50–415 | Very high capacity, but high cost and stability issues in water | [47] |
| Thermally Treated Bentonite | 6.9–20.4 | Low cost, moderate capacity | [26,45] |
| Metal-Biochar Composites | 1.2–120 | Wide range, performance depends on metal and pyrolysis conditions | [36,42] |
| Activated Alumina | 0.72–13.7 | Commercial, limited capacity | [42,43,44] |
| Raw Phosphate Tailings | 1.99 | Unmodified, low capacity | This study |
Compared to the conventional chemical precipitation method involving direct addition of calcium or magnesium salts to water bodies, the primary advantage of the MPT technology lies in its transformation of industrial solid waste into a treatment medium, achieving resource closure and avoiding the introduction of new chemicals. Furthermore, the porous structure of MPT provides abundant heterogeneous nucleation sites for phosphate precipitation, potentially enhancing reaction efficiency and product stability. The economic and environmental appeal of this technology is particularly pronounced for regions or enterprises where it can simultaneously reduce waste disposal and water treatment costs.
While the thermal modification at 950 °C entails energy input, the overall cost-effectiveness of the MPT adsorbent should be evaluated within a circular economy framework. This considers the avoided costs of long-term tailings storage and management, the displacement of more expensive commercial adsorbents or precipitation chemicals, and the potential value of the phosphorus-laden spent material. A comprehensive life-cycle assessment is recommended for future scale-up studies to quantify the net economic and environmental benefits.
It is noteworthy to distinguish the proposed MPT technology from conventional chemical precipitation, which relies on direct addition of soluble Ca2+/Mg2+ salts (e.g., CaCl2). While both approaches ultimately remove phosphate via precipitation, MPT functions as a structured, multifunctional adsorbent rather than a mere ion source. Its thermally generated CaO/MgO provides a controlled release of ions, while the hierarchical pore network acts as a confined microenvironment that enhances nucleation and stabilizes the resulting hydroxyapatite. More importantly, MPT embodies a “waste-treats-waste” circular strategy, transforming an industrial solid residue into a treatment medium, thereby avoiding the introduction of external chemicals and simultaneously addressing tailings stockpiling. This integrated design not only improves separation efficiency (the precipitate is anchored within the solid matrix) but also aligns with sustainable resource management, offering a dual environmental benefit that conventional precipitation cannot achieve.
4.4. Influence of Environmental Factors and Future Research Directions
Solution pH critically governed the P adsorption performance, with a maximum capacity achieved at pH 6. This optimum is attributed to the synergistic effect of surface protonation enhancing electrostatic attraction and the promoted dissolution of CaO/MgO to release Ca2+/Mg2+ ions for phosphate precipitation. The effect of contact time showed a rapid initial uptake followed by a slower phase, consistent with the pseudo-second-order kinetic model.
For future practical applications, research should be extended from synthetic to actual wastewater. A key next step is to evaluate the efficacy of MPT in realistic water environments. This includes: (1) conducting batch and dynamic column adsorption experiments in simulated river water or actual surface water containing common coexisting anions (e.g., Cl−, SO42−, HCO3−/CO32−, NO3−) to systematically study competitive adsorption effects; and (2) investigating the potential influence of natural organic matter (NOM) on the adsorption process (e.g., site blockage or complex formation). These studies will provide indispensable parameters for the engineering design of MPT applications. Dynamic column experiments using real eutrophic water are essential to evaluate performance under continuous flow conditions and in the presence of common co-existing ions (e.g., organic matter, competing anions). Furthermore, a detailed techno-economic and life-cycle assessment is needed to quantitatively evaluate the net environmental benefits and economic viability of this adsorbent at scale, factoring in the energy cost of calcination against the avoided costs of tailings management and eutrophication damage.
5. Conclusions
This study successfully demonstrates a sustainable and circular strategy to concurrently address the global challenges of phosphate tailings (PT) accumulation and aquatic eutrophication. By employing a simple thermal modification at 950 °C, we transformed an abundant industrial solid waste into a high-performance adsorbent (MPT) with a phosphorus adsorption capacity of 112.41 mg/g, a 45-fold enhancement over the raw material. Nonlinear regression analysis elucidated the adsorption mechanism: the kinetics followed the pseudo-second-order model, confirming a chemisorption-dominated process, while the equilibrium data were best described by the Freundlich isotherm. This demonstrates that adsorption occurs on a heterogeneous surface with sites of varying energy, a direct consequence of the complex material architecture created by calcination. The primary immobilization mechanism was identified as the chemical precipitation of phosphate with Ca2+/Mg2+ ions released from the generated CaO/MgO to form stable hydroxyapatite. The competitive adsorption capacity of MPT, which stands favorably against many engineered adsorbents, coupled with its origin from a low-cost waste stream, underscores its significant potential as a cost-effective solution.
Therefore, this “waste-treats-waste” technology represents a proactive, source-oriented strategy not only for the phosphate industry but for any region grappling with solid waste management and water quality deterioration. It directly contributes to mitigating a root cause of harmful algal blooms, thereby helping to secure aquatic ecosystem health and public water safety on a global scale. While future work should focus on evaluating its performance in real-water matrices with coexisting ions and conducting a comprehensive life-cycle assessment, the fundamental strategy validated here offers a compelling pathway aligned with circular economy principles. We recommend integrating such waste valorization approaches into broader environmental management and resource recovery policies to accelerate their engineering application and maximize their environmental benefits.
Author Contributions
K.L. and Y.G. were responsible for conceptualization, as well as for reviewing and editing the manuscript. C.G. contributed to the methodology, performed data curation, and prepared the original draft. Y.H. and X.Z. were involved in visualization and formal analysis. J.L. and S.X. provided resources and conducted the investigation. Funding for this research was acquired by K.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research were funded by the Open Fund Program of the China National Engineering Research Center for Phosphate Resources Development and Utilization (Grant No. NECP-2023-12).
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
During the preparation of this manuscript, we used DeepSeek-V3.2 for translating the text from Chinese to English and for revising the manuscript, particularly in the Discussion section. We have reviewed and edited the generated content and take full responsibility for the entire content of this publication.
Conflicts of Interest
Authors Yongjie Guo, Jiangli Li and Shuai Xu are employed by Yunnan Phosphate Chemical Group Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Paerl, H.W.; Gardner, W.S.; Havens, K.E.; Joyner, A.R.; McCarthy, M.J.; Newell, S.E.; Qin, B.Q.; Scott, J.T. Mitigating cyanobacterial harmful algal blooms in aquatic ecosystems impacted by climate change and anthropogenic nutrients. Harmful Algae 2016, 54, 213–222. [Google Scholar] [CrossRef]
- Wang, H.; Molinos, J.G.; Heino, J.; Zhang, H.; Zhang, P.Y.; Xu, J. Eutrophication causes invertebrate biodiversity loss and decreases cross-taxon congruence across anthropogenically-disturbed lakes. Environ. Int. 2021, 153, 106494. [Google Scholar] [CrossRef]
- Li, H.Y.; Zhang, H.C.; Duan, L.Z.; Liu, Q.; Li, D.L.; Huang, J.P.; Fu, J.W.; Zi, L.H.; Xu, T.B. Spatiotemporal variations in water quality parameters and assessment of the current status and challenges of eutrophication in Lake Dian. Ecol. Indic. 2025, 177, 113821. [Google Scholar] [CrossRef]
- Oduor, N.A.; Munga, C.N.; Ong’anda, H.O.; Botwe, P.K.; Moosdorf, N. Nutrients and harmful algal blooms in Kenya’s coastal and marine waters: A review. Ocean Coast. Manag. 2023, 233, 106454. [Google Scholar] [CrossRef]
- Reif, J.S.; Koszalinski, R.; McFarland, M.M.; Parsons, M.L.; Schinbeckler, R.; Kociolek, J.; Rockenstyre, A.; Schaefer, A.M. Evaluating Bias in Self-Reported Symptoms During a Cyanobacterial Algal Bloom. Toxins 2025, 17, 287. [Google Scholar] [CrossRef] [PubMed]
- Dodds, W.K.; Bouska, W.W.; Eitzmann, J.L.; Pilger, T.J.; Pitts, K.L.; Riley, A.J.; Schloesser, J.T.; Thornbrugh, D.J. Eutrophication of US Freshwaters: Analysis of Potential Economic Damages. Environ. Sci. Technol. 2009, 43, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Grizzetti, B.; Vigiak, O.; Udias, A.; Aloe, A.; Zanni, M.; Bouraoui, F.; Pistocchi, A.; Dorati, C.; Friedland, R.; De Roo, A.; et al. How EU policies could reduce nutrient pollution in European inland and coastal waters. Glob. Environ. Change 2021, 69, 102281. [Google Scholar] [CrossRef]
- Ministry of Ecology and Environment of the People’s Republic of China (MEE). China Ecological and Environmental Statistical Annual Report (2023); Ministry of Ecology and Environment of the People’s Republic of China (MEE): Beijing, China, 2024. Available online: https://www.mee.gov.cn/hjzl/sthjzk/sthjtjnb/202412/t20241231_1099687 (accessed on 6 January 2026).
- GB 3838-2002; Environmental Quality Standards for Surface Water. State Environmental Protection Administration & General Administration of Quality Supervision, Inspection and Quarantine: Beijing, China, 2002.
- Wang, C.; Guo, Y.; Liu, L.; Wang, Y.; Luo, A.; Liu, H.; Shi, Y.; Xiao, Y.; Zhou, J. Review and Prospect of Research on Resource Utilization of Phosphorus Tailings. Yunnan Chem. Technol. 2023, 50, 1–6. [Google Scholar]
- Yang, Y.H.; Wei, Z.A.; Chen, Y.L.; Li, Y.J.; Li, X.Y. Utilizing phosphate mine tailings to produce ceramisite. Constr. Build. Mater. 2017, 155, 1081–1090. [Google Scholar] [CrossRef]
- Zhang, S.X.; Xie, X.; Xie, R.Q.; Tong, X.; Wu, Y.Y.; Li, J.W.; Li, Y. Preparation and interface state of phosphate tailing-based geopolymers. J. Cent. South Univ. 2024, 31, 1900–1914. [Google Scholar] [CrossRef]
- Li, H.L.; Yang, J.Q.; Qu, G.F.; Jin, C.Y.; Chen, B.J.; Liu, X.X.; Wu, F.H.; Kuang, L.R.; Liu, Y. Solidification and Stabilization of Fluorine and Phosphorus in Phosphate Mine Tailings and the Mechanism of F and P Fixation During Soilization. Chemistryselect 2024, 9, e202401515. [Google Scholar] [CrossRef]
- Jiao, B.Q.; Peng, Q.K.; Li, D.W.; Guo, S.L. The basic characteristics of the soil in phosphorus tail mining area. Res. J. Chem. Environ. 2013, 17, 89–95. [Google Scholar]
- Segui, P.; Safhi, A.E.; Amrani, M.; Benzaazoua, M. Mining Wastes as Road Construction Material: A Review. Minerals 2023, 13, 90. [Google Scholar] [CrossRef]
- Han, R.B.; Leng, H.X.; Luo, H.; Wu, W.B.; Zhao, Y.R.; Song, B.K.; Liu, M.; He, B.J. Turning waste into treasure: Preparation, physical properties and microstructure of alkali-activated phosphorus tailings-based fully solid waste non-sintered lightweight aggregates. J. Clean. Prod. 2025, 527, 146693. [Google Scholar] [CrossRef]
- Fu, F.H.; Hu, N.Y.; Yang, S.W.; Li, Q.G.; Ye, Y.C.; Tang, R.B.; Chen, G. Effect of green gel system upon the properties of phosphorus tailings foamed ceramics. Ceram. Int. 2024, 50, 38748–38760. [Google Scholar] [CrossRef]
- Gu, K.; Chen, B.; Yan, P.; Wang, J.M. Recycling of phosphate tailings and acid wastewater from phosphorus chemical industrial chain to prepare a high value-added magnesium oxysulfate cement. J. Clean. Prod. 2022, 369, 133343. [Google Scholar] [CrossRef]
- Zheng, Y.L.; Wan, Y.S.; Zhang, Y.; Huang, J.S.; Yang, Y.C.; Tsang, D.C.W.; Wang, H.L.; Chen, H.; Gao, B. Recovery of phosphorus from wastewater: A review based on current phosphorous removal technologies. Crit. Rev. Environ. Sci. Technol. 2022, 53, 1148–1172. [Google Scholar] [CrossRef]
- Akula, V.V.; Philip, L. Enhanced phosphorus reclamation from secondary effluent through Donnan dialysis and vivianite crystallization. Water Reuse 2025, 15, 575–590. [Google Scholar] [CrossRef]
- Gubernat, S.; Maslon, A.; Czarnota, J.; Koszelnik, P. Phosphorus removal from wastewater using marl and travertine and their thermal modifications. Desalination Water Treat. 2022, 275, 35–46. [Google Scholar] [CrossRef]
- He, D.S.; Chen, B.B.; Tang, Y.; Li, Q.Q.; Zhang, K.C.; Li, Z.L.; Xu, C.M. The enhanced adsorption properties of phosphorus from aqueous solutions using lanthanum modified synthetic zeolites. Green Process. Synth. 2023, 12, 20230106. [Google Scholar] [CrossRef]
- Kumari, S.; Chowdhry, J.; Kumar, M.; Chandra Garg, M. Zeolites in wastewater treatment: A comprehensive review on scientometric analysis, adsorption mechanisms, and future prospects. Environ. Res. 2024, 260, 119782. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, N.; Xing, C.; Cui, Q.; Sun, Q. The adsorption, regeneration and engineering applications of biochar for removal organic pollutants: A review. Chemosphere 2019, 223, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.T.; Chen, Y.N.; Xu, J.L.; Liu, J. Removal mechanism of phosphorus in water by calcium hydroxide modified copper tailings. Sci. Rep. 2024, 14, 20326. [Google Scholar] [CrossRef] [PubMed]
- Du, C.M.; Zhang, Y.T.; Yu, W.M. A method for high value-added utilization of BOF slag: Towards slag recycling and phosphorus recovery. Process Saf. Environ. Prot. 2024, 190, 586–597. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecular, layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 1951, 73, 373−380. [Google Scholar] [CrossRef]
- GB/T 11893-1989; Water Quality—Determination of Total Phosphorus—Ammonium Molybdate Spectrophotometric Method. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 1989.
- Lagergren, S. Zur theorie der sogenannten adsorption gelöster stoffe. K. Sven. Vetenskapsakademiens Handlingar 1898, 24, 1–39. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Gosset, Z.T.; Trancart, J.L.; Thevenot, D.R. Batch metal removal by peat kinetics and thermodynamics. Water Res. 1986, 20, 21–26. [Google Scholar] [CrossRef]
- Langmuir, I. The Constitution and fundamental properties of solids and liquids. Part I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017, 3039817. [Google Scholar] [CrossRef]
- Chen, X.; Wu, L.; Liu, F.; Luo, P.; Zhuang, X.L.; Wu, J.S.; Zhu, Z.K.; Xu, S.J.; Xie, G.X. Performance and mechanisms of thermally treated bentonite for enhanced phosphate removal from wastewater. Environ. Sci. Pollut. Res. 2018, 25, 15980–15989. [Google Scholar] [CrossRef] [PubMed]
- Dabbebi, R.; de Aguiar, J.L.B.; Samet, B.; Baklouti, S. Mineralogical and chemical investigation of Tunisian phosphate washing waste during calcination. J. Therm. Anal. Calorim. 2019, 137, 1827–1840. [Google Scholar] [CrossRef]
- Lach, M.; Grela, A.; Komar, N.; Mikula, J.; Hebda, M. Calcined Post-Production Waste as Materials Suitable for the Hydrothermal Synthesis of Zeolites. Materials 2019, 12, 2742. [Google Scholar] [CrossRef]
- Yuan, X.M.; Xie, H.J.; Nie, D.P.; Zhang, Y.; Zhou, L.; Wu, Y.Y.; Wen, Z. Thermogravimetric and spectroscopic analyses along with the kinetic modeling of the pyrolysis of phosphate tailings. RSC Adv. 2023, 13, 16741–16748. [Google Scholar] [CrossRef]
- Liang, H.; Guo, P.L.; Yang, Y.H.; Wang, W.T.; Sun, Z.N. Environmental application of engineering magnesite slag for phosphate adsorption from wastewater. Environ. Sci. Pollut. Res. 2022, 29, 59502–59512. [Google Scholar] [CrossRef]
- Wang, X.Y.; Chai, X.H.; Huang, W.Q.; Li, X.F.; Zhu, B.; Li, X.T.; Zhou, Y.K.; Yang, Z.M.; Sun, X.H.; Fu, L.P. Green synthesis of biomass-derived porous carbon with hierarchical pores and enhanced surface area for superior VOCs adsorption. Mater. Today Commun. 2024, 39, 108906. [Google Scholar] [CrossRef]
- Feng, Y.C.; Jia, J.W.; Wang, N.N.; Ma, S.X. Towards understanding the interaction mechanism of Sintering, hydration and carbonation of calcium oxide during CO2 capture. Chem. Eng. J. 2025, 512, 162598. [Google Scholar] [CrossRef]
- Gao, R.; Lou, L.; Xu, T.; Liu, G.; Fang, J.; Ding, S.J. Phosphorus removal in swine wastewater by lanthanum-modified biochar and its synergistic interaction with Pseudomonas sp. G16. J. Environ. Chem. Eng. 2025, 13, 119362. [Google Scholar] [CrossRef]
- Shaheen, U.; Ye, Z.L.; Abass, O.K.; Zamel, D.; Rehman, A.; Zhao, P.; Huang, F.M. Evaluation of potential adsorbents for simultaneous adsorption of phosphate and ammonium at low concentrations. Microporous Mesoporous Mater. 2024, 379, 113301. [Google Scholar] [CrossRef]
- Genz, A.; Kornmüller, A.; Jekel, M. Advanced phosphorus removal from membrane filtrates by adsorption on activated aluminium oxide and granulated ferric hydroxide. Water Res. 2004, 38, 3523–3530. [Google Scholar] [CrossRef] [PubMed]
- Li, X.D.; Kuang, Y.; Chen, J.B.; Wu, D.Y. Competitive adsorption of phosphate and dissolved organic carbon on lanthanum modified zeolite. J. Colloid Interface Sci. 2020, 574, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Zahed, M.A.; Salehi, S.; Tabari, Y.; Farraji, H.; Ataei-Kachooei, S.; Zinatizadeh, A.A.; Kamali, N.; Mahjouri, M. Phosphorus removal and recovery: State of the science and challenges. Environ. Sci. Pollut. Res. 2022, 29, 58561–58589. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Ji, H.; Li, R.; Tajammal Munir, M.; Wu, X.; Huang, Y.; Li, B. Advancing sustainable phosphorus removal and recovery with Metal-Organic frameworks (MOFs). Chem. Eng. J. 2023, 475, 145949. [Google Scholar] [CrossRef]
- Loganathan, P.; Vigneswaran, S.; Kandasamy, J.; Bolan, N.S. Removal and Recovery of Phosphate From Water Using Sorption. Crit. Rev. Environ. Sci. Technol. 2014, 44, 847–907. [Google Scholar] [CrossRef]
- Li, R.H.; Wang, J.J.; Gaston, L.A.; Zhou, B.Y.; Li, M.L.; Xiao, R.; Wang, Q.; Zhang, Z.Q.; Huang, H.; Liang, W.; et al. An overview of carbothermal synthesis of metal-biochar composites for the removal of oxyanion contaminants from aqueous solution. Carbon 2018, 129, 674–687. [Google Scholar] [CrossRef]
- Gu, K.; Lang, L.; Li, D.S.; Chen, B. Preparation of Magnesium Oxysulfate Cement with Calcined Phosphate Tailings. J. Mater. Civ. Eng. 2022, 34, 04022358. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.