Experimental Assessment of the Influence of Fish Passage Geometry Parameters on Downstream Migrating Atlantic Salmon (Salmo salar) Smolts Behavior
Abstract
:1. Introduction
2. Experimental Setup and Methods
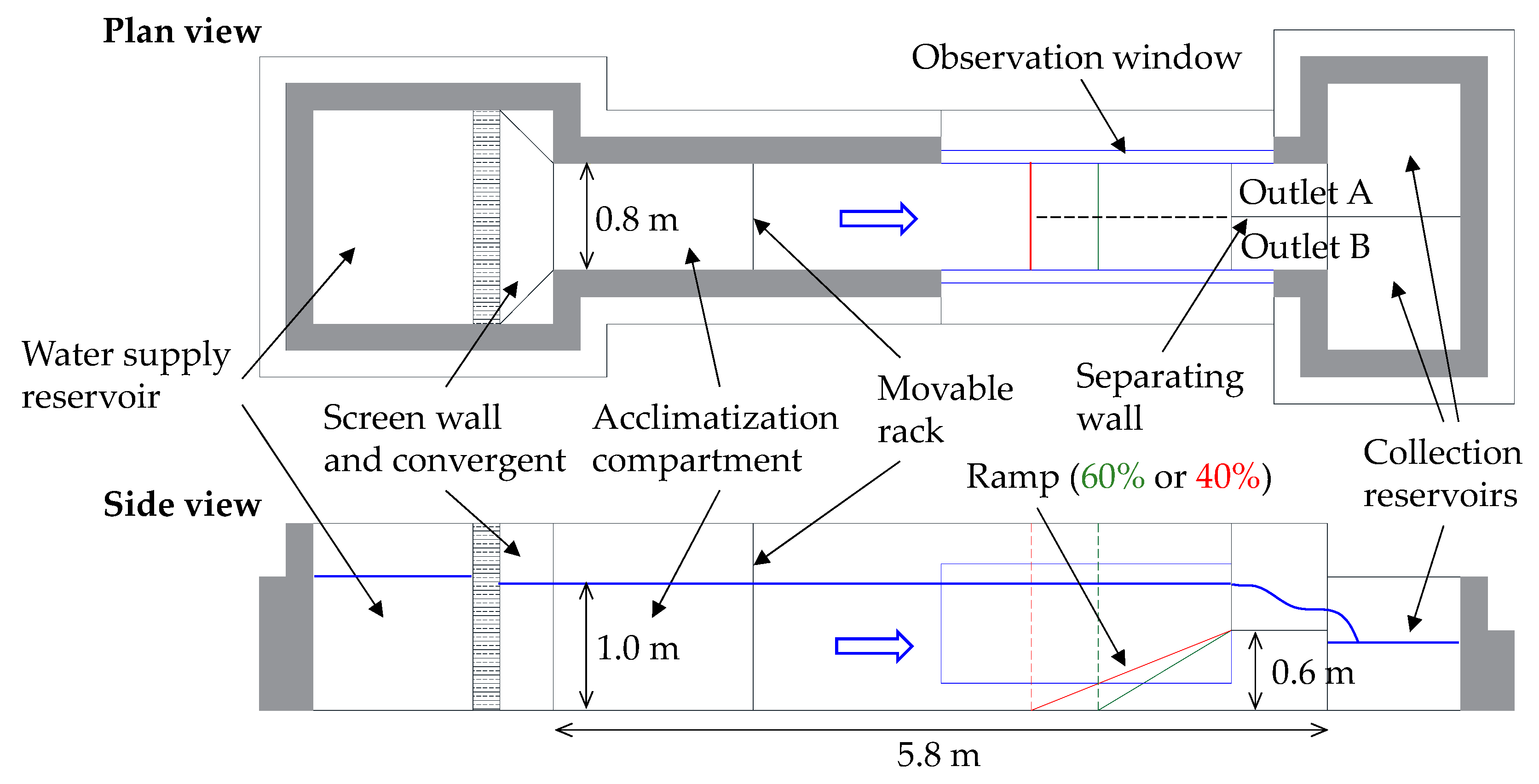
2.1. Experimental Facility and Instrumentation
2.2. Test Configurations
2.3. Smolt Availability
2.4. Experimental Protocol and Tests Methodology
- Placement of a batch of smolts in the acclimatization compartment of the flume for 10 min.
- Opening of the movable rack to allow the smolts to swim downstream and establishment of a steady water discharge of 0.265 m3/s for 30 min.
- Lowering of the water discharge to 0.116 m3/s, counting and removal of the fish that swam through one of the outlets and ended up in the downstream reservoirs. This operation lasted around 5 min, depending on the number of smolts to catch in the reservoirs.
- Set the water discharge back to 0.265 m3/s for another 30 min.
- Repetition of steps 3 and 4 for three more times.
- In case smolts swam through one of the outlets during the fifth 30 min period with 0.265 m3/s discharge, steps 3 and 4 were repeated for two more times and after that the test ended. In case no smolts swam through one of the outlets during the fifth 30 min period with 0.265 m3/s discharge, this period was extended by 30 more minutes and subsequently the test was over.
- Removal of the fish from the downstream reservoir and counting. The fish that were still in the flume were forced, using a brush, to swim downstream and choose one of the two outlets. Removal of these fish from the downstream reservoirs and counting.
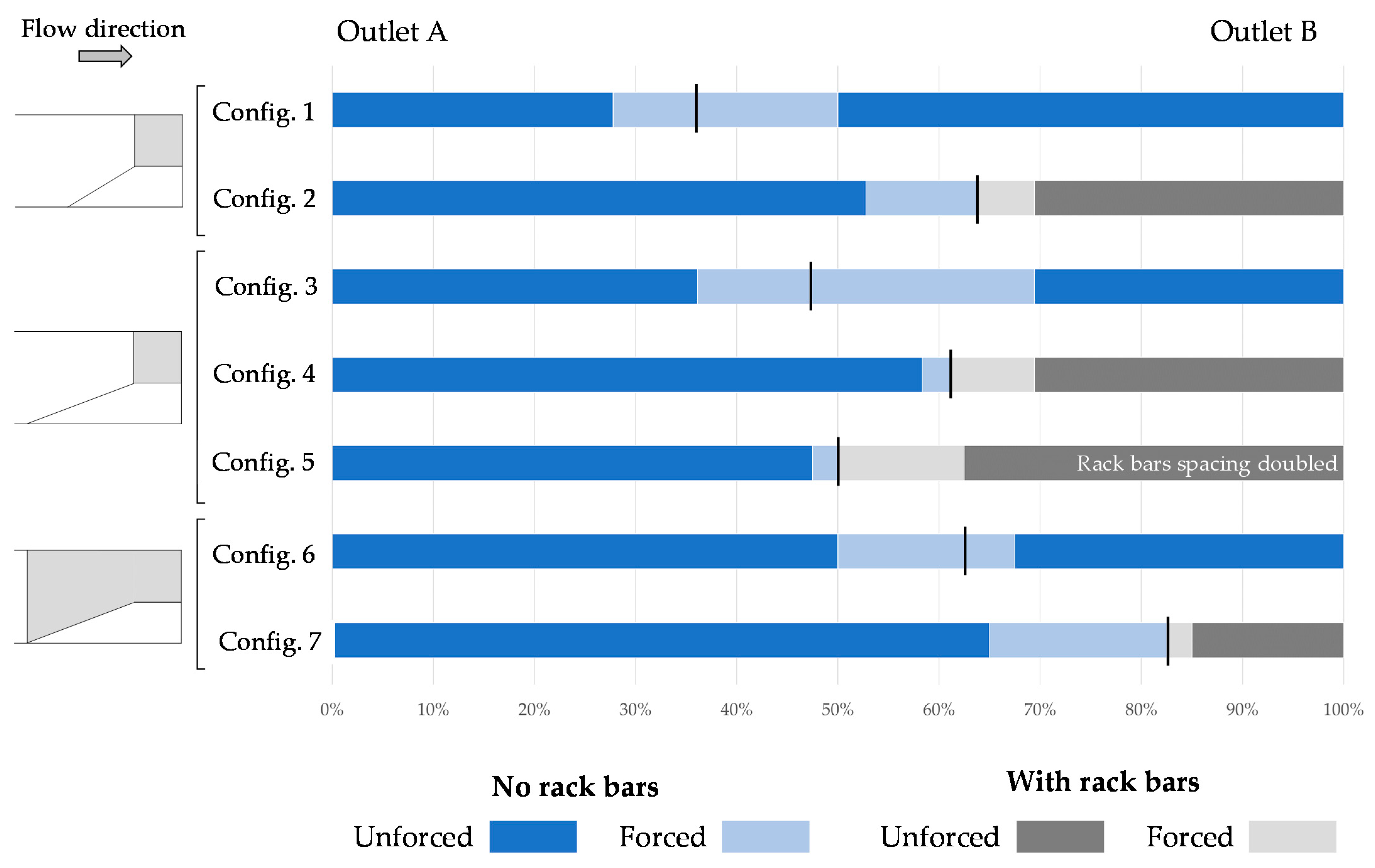
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kitsikoudis, V.; Becker, B.P.J.; Huismans, Y.; Archambeau, P.; Erpicum, S.; Pirotton, M.; Dewals, B. Discrepancies in Flood Modelling Approaches in Transboundary River Systems: Legacy of the Past or Well-grounded Choices? Water Resour. Manag. 2020, 34, 3465–3478. [Google Scholar] [CrossRef]
- Poff, N.L.; Olden, J.; Merritt, D.M.; Pepin, D.M. Homogenization of regional river dynamics by dams and global biodiversity implications. Proc. Natl. Acad. Sci. USA 2007, 104, 5732–5737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liermann, C.R.; Nilsson, C.; Robertson, J.; Ng, R.Y. Implications of Dam Obstruction for Global Freshwater Fish Diversity. Biosci. 2012, 62, 539–548. [Google Scholar] [CrossRef]
- Vörösmarty, C.J.; McIntyre, P.B.; Gessner, M.O.; Dudgeon, D.; Prusevich, A.; Green, P.; Glidden, S.; Bunn, S.E.; Sullivan, C.A.; Liermann, C.R.; et al. Global threats to human water security and river biodiversity. Nature 2010, 467, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Katopodis, C.; Williams, J.G. The development of fish passage research in a historical context. Ecol. Eng. 2012, 48, 8–18. [Google Scholar] [CrossRef]
- Noonan, M.J.; A Grant, J.W.; Jackson, C.D. A quantitative assessment of fish passage efficiency. Fish Fish. 2012, 13, 450–464. [Google Scholar] [CrossRef]
- Silva, A.T.; Lucas, M.C.; Castro-Santos, T.; Katopodis, C.; Baumgartner, L.; Thiem, J.; Aarestrup, K.; Pompeu, P.S.; O’Brien, G.; Braun, D.C.; et al. The future of fish passage science, engineering, and practice. Fish Fish. 2018, 19, 340–362. [Google Scholar] [CrossRef] [Green Version]
- Thorstad, E.B.; Whoriskey, F.; Uglem, I.; Moore, A.; Rikardsen, A.H.; Finstad, B. A critical life stage of the Atlantic salmon Salmo salar: Behaviour and survival during the smolt and initial post-smolt migration. J. Fish Biol. 2012, 81, 500–542. [Google Scholar] [CrossRef]
- Renardy, S.; Takriet, A.; Benitez, J.-P.; Dierckx, A.; Baeyens, R.; Coeck, J.; Pauwels, I.S.; Mouton, A.; Archambeau, P.; Dewals, B.; et al. Trying to choose the less bad route: Individual migratory behaviour of Atlantic salmon smolts (Salmo salar L.) approaching a bifurcation between a hydropower station and a navigation canal. Ecol. Eng. 2021, 169, 106304. [Google Scholar] [CrossRef]
- Ovidio, M.; Renardy, S.; Dierckx, A.; Matondo, B.N.; Benitez, J.-P. Improving bypass performance and passage success of Atlantic salmon smolts at an old fish-hostile hydroelectric power station: A challenging task. Ecol. Eng. 2021, 160, 106148. [Google Scholar] [CrossRef]
- Larinier, M.; Travade, F. Downstream migration: Problems and facilities. Bull. Français Pêche Piscic. 2002, 364, 181–207. [Google Scholar] [CrossRef] [Green Version]
- Nyqvist, D.; McCormick, S.D.; Greenberg, L.; Ardren, W.R.; Bergman, E.; Calles, O.; Castro-Santos, T. Downstream Migration and Multiple Dam Passage by Atlantic Salmon Smolts. N. Am. J. Fish. Manag. 2017, 37, 816–828. [Google Scholar] [CrossRef]
- Szabo-Meszaros, M.; Silva, A.; Bærum, K.; Baktoft, H.; Alfredsen, K.; Hedger, R.; Økland, F.; Gjelland, K.; Fjeldstad, H.-P.; Calles, O.; et al. Validation of a Swimming Direction Model for the Downstream Migration of Atlantic Salmon Smolts. Water 2021, 13, 1230. [Google Scholar] [CrossRef]
- Ovidio, M.; Dierckx, A.; Bunel, S.; Grandry, L.; Spronck, C.; Benitez, J. Poor Performance of a Retrofitted Downstream Bypass Revealed by the Analysis of Approaching Behaviour in Combination with a Trapping System. River Res. Appl. 2017, 33, 27–36. [Google Scholar] [CrossRef]
- Calles, O.; Karlsson, S.; Hebrand, M.; Comoglio, C. Evaluating technical improvements for downstream migrating diadromous fish at a hydroelectric plant. Ecol. Eng. 2012, 48, 30–37. [Google Scholar] [CrossRef]
- Williams, J.G. Mitigating the effects of high-head dams on the Columbia River, USA: Experience from the trenches. Hydrobiologia 2008, 609, 241–251. [Google Scholar] [CrossRef]
- Harbicht, A.B.; Watz, J.; Nyqvist, D.; Virmaja, T.; Carlsson, N.; Aldvén, D.; Nilsson, P.A.; Calles, O. Guiding migrating salmonid smolts: Experimentally assessing the performance of angled and inclined screens with varying gap widths. Ecol. Eng. 2022, 174, 106438. [Google Scholar] [CrossRef]
- Greenberg, L.; Calles, O.; Andersson, J.; Engqvist, T. Effect of trash diverters and overhead cover on downstream migrating brown trout smolts. Ecol. Eng. 2012, 48, 25–29. [Google Scholar] [CrossRef]
- Albayrak, I.; Boes, R.M.; Kriewitz-Byun, C.R.; Peter, A.; Tullis, B.P. Fish guidance structures: Hydraulic performance and fish guidance efficiencies. J. Ecohydraulics 2020, 5, 113–131. [Google Scholar] [CrossRef]
- Beck, C.; Albayrak, I.; Meister, J.; Peter, A.; Selz, O.M.; Leuch, C.; Vetsch, D.F.; Boes, R.M. Swimming Behavior of Downstream Moving Fish at Innovative Curved-Bar Rack Bypass Systems for Fish Protection at Water Intakes. Water 2020, 12, 3244. [Google Scholar] [CrossRef]
- Leander, J.; Klaminder, J.; Hellström, G.; Jonsson, M. Bubble barriers to guide downstream migrating Atlantic salmon (Salmo salar): An evaluation using acoustic telemetry. Ecol. Eng. 2021, 160, 106141. [Google Scholar] [CrossRef]
- Vowles, A.S.; Kemp, P.S. Effects of light on the behaviour of brown trout (Salmo trutta) encountering accelerating flow: Application to downstream fish passage. Ecol. Eng. 2012, 47, 247–253. [Google Scholar] [CrossRef]
- Williams, J.G.; Armstrong, G.; Katopodis, C.; Larinier, M.; Travade, F. Thinking like a fish: A key ingredient for development of effective fish passage facilities at river obstructions. River Res. Appl. 2012, 28, 407–417. [Google Scholar] [CrossRef] [Green Version]
- Kemp, P.S.; Gessel, M.H.; Sandford, B.P.; Williams, J.G. The behaviour of Pacific salmonid smolts during passage over two experimental weirs under light and dark conditions. River Res. Appl. 2006, 22, 429–440. [Google Scholar] [CrossRef]
- Enders, E.C.; Gessel, M.H.; Williams, J.G. Development of successful fish pasage structures for downstream migrants requires knowledge of their behavioural response to accelerating flow. Can. J. Fish. Aquat. Sci. 2009, 66, 2109–2117. [Google Scholar] [CrossRef]
- Kemp, P.S.; Gessel, M.H.; Williams, J.G. Fine-Scale Behavioral Responses of Pacific Salmonid Smolts as They Encounter Divergence and Acceleration of Flow. Trans. Am. Fish. Soc. 2005, 134, 390–398. [Google Scholar] [CrossRef]
- Courret, D.; Larinier, M. Guide Pour la Conception de Prises d’eau “Ichtyocompatibles” Pour les Petites Centrales Hydroélectriques; Rapport GHAAPPE RA.08.04. November, 2008; ONEMA: Vincennes, France, 2008. [Google Scholar]
- Riley, W.D.; Ibbotson, A.T.; Maxwell, D.L.; Davison, P.I.; Beaumont, W.R.C.; Ives, M.J. Development of schooling behaviour during the downstream migration of Atlantic salmon Salmo salar smolts in a chalk stream. J. Fish Biol. 2014, 85, 1042–1059. [Google Scholar] [CrossRef]
- Vowles, A.S.; Anderson, J.J.; Gessel, M.H.; Williams, J.G.; Kemp, P.S. Effects of avoidance behaviour on downstream fish passage through areas of accelerating flow when light and dark. Anim. Behav. 2014, 92, 101–109. [Google Scholar] [CrossRef] [Green Version]


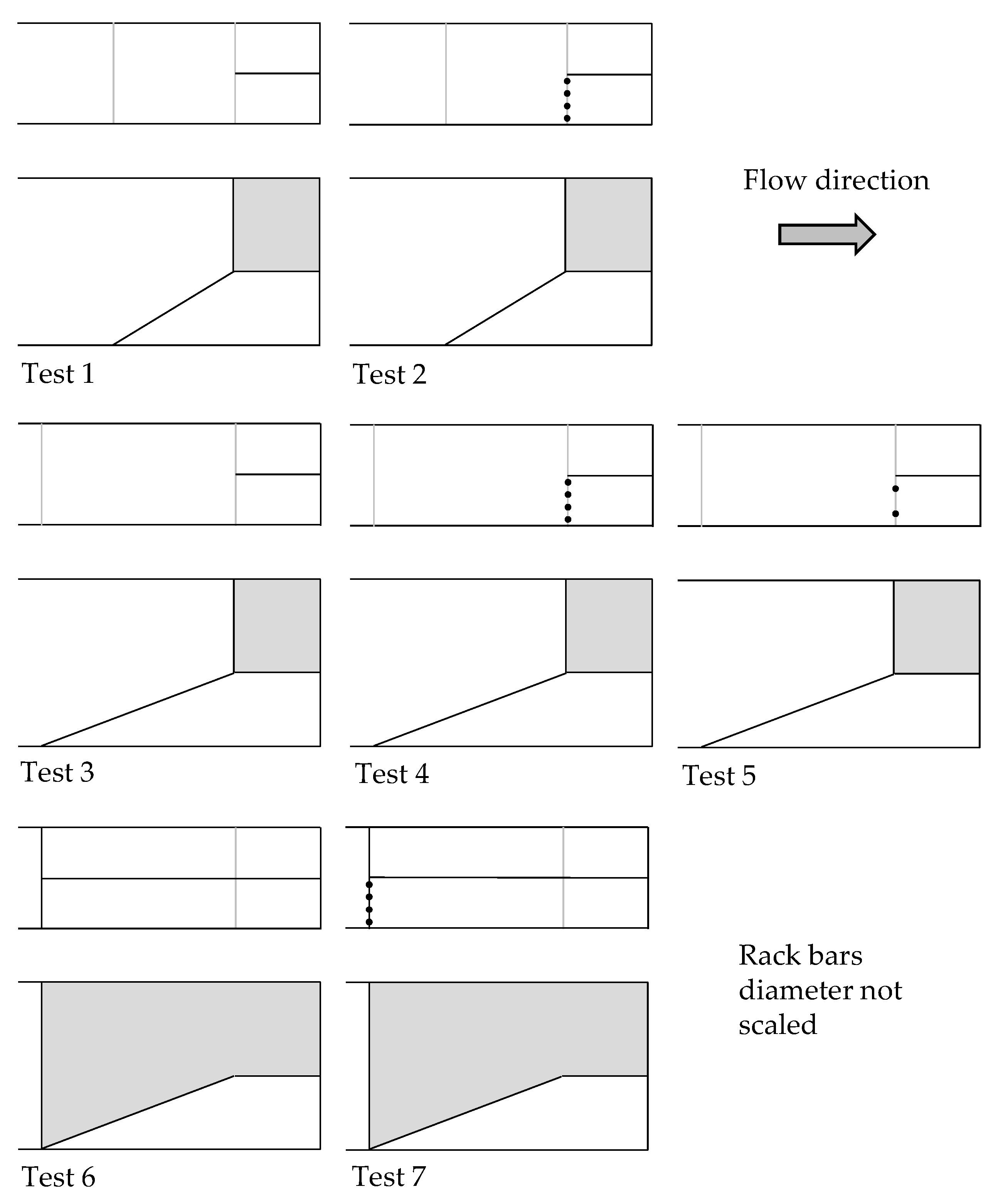
| Config. Number | Groups Number | Batches Number | Smolts per Batch | Slope of Ramp | Spacing of Rack Bars | Location of Rack |
|---|---|---|---|---|---|---|
| 1 | 2 | 2.1–2.2 | 18 | 60% | No rack 1 | |
| 2 | 2 | 2.3–2.4 | 18 | 60% | 10 cm | Weir crest 1 |
| 3 | 3 | 3.1–3.2 | 18 | 40% | No rack 1 | |
| 4 | 3 | 3.3–3.4 | 18 | 40% | 10 cm | Weir crest 1 |
| 5 | 4 | 4.1–4.2 | 20 | 40% | 20 cm | Weir crest 1 |
| 6 | 4 | 4.3–4.4 | 20 | 40% | No rack 2 | |
| 7 | 4 | 4.5–4.6 | 20 | 40% | 10 cm | Ramp toe 2 |
| Batch Number | Number of Smolts | m < 30 g | 30 g ≤ m ≤ 40 g | m > 40 g |
|---|---|---|---|---|
| 1.1 | 18 | 6 | 9 | 3 |
| 1.2 | 18 | 6 | 10 | 2 |
| 1.3 | 18 | 6 | 10 | 2 |
| 1.4 | 18 | 6 | 10 | 2 |
| Config. Number | Batch Number | Number of Smolts in Batch | Smolts in Outlet A within the Test Duration | Smolts in Outlet B within the Test Duration | Smolts in Outlet A when Forced | Smolts in Outlet B when Forced |
|---|---|---|---|---|---|---|
| 1 | 2.1 | 18 | 7 | 8 | 1 | 2 |
| 2.2 | 18 | 3 | 10 | 2 | 3 | |
| 2 | 2.3 | 18 | 12 | 5 | 1 | 0 |
| 2.4 | 18 | 7 | 6 | 3 | 2 | |
| 3 | 3.1 | 18 | 8 | 3 | 2 | 5 |
| 3.2 | 18 | 5 | 8 | 2 | 3 | |
| 4 | 3.3 | 18 | 13 | 2 | 1 | 2 |
| 3.4 | 18 | 8 | 9 | 0 | 1 | |
| 5 | 4.1 | 20 | 9 | 10 | 0 | 1 |
| 4.2 | 20 | 10 | 5 | 1 | 4 | |
| 6 | 4.3 | 20 | 10 | 8 | 2 | 0 |
| 4.4 | 20 | 10 | 5 | 3 | 2 | |
| 7 | 4.5 | 20 | 17 | 1 | 2 | 0 |
| 4.6 | 20 | 9 | 5 | 5 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erpicum, S.; Kitsikoudis, V.; Archambeau, P.; Dewals, B.; Pirotton, M. Experimental Assessment of the Influence of Fish Passage Geometry Parameters on Downstream Migrating Atlantic Salmon (Salmo salar) Smolts Behavior. Water 2022, 14, 616. https://doi.org/10.3390/w14040616
Erpicum S, Kitsikoudis V, Archambeau P, Dewals B, Pirotton M. Experimental Assessment of the Influence of Fish Passage Geometry Parameters on Downstream Migrating Atlantic Salmon (Salmo salar) Smolts Behavior. Water. 2022; 14(4):616. https://doi.org/10.3390/w14040616
Chicago/Turabian StyleErpicum, Sebastien, Vasileios Kitsikoudis, Pierre Archambeau, Benjamin Dewals, and Michel Pirotton. 2022. "Experimental Assessment of the Influence of Fish Passage Geometry Parameters on Downstream Migrating Atlantic Salmon (Salmo salar) Smolts Behavior" Water 14, no. 4: 616. https://doi.org/10.3390/w14040616
APA StyleErpicum, S., Kitsikoudis, V., Archambeau, P., Dewals, B., & Pirotton, M. (2022). Experimental Assessment of the Influence of Fish Passage Geometry Parameters on Downstream Migrating Atlantic Salmon (Salmo salar) Smolts Behavior. Water, 14(4), 616. https://doi.org/10.3390/w14040616







