Use of Heating Configuration to Control Marangoni Circulation during Droplet Evaporation
Abstract
:1. Introduction
Synthesis and Objectives of the Work
2. Mathematical Model
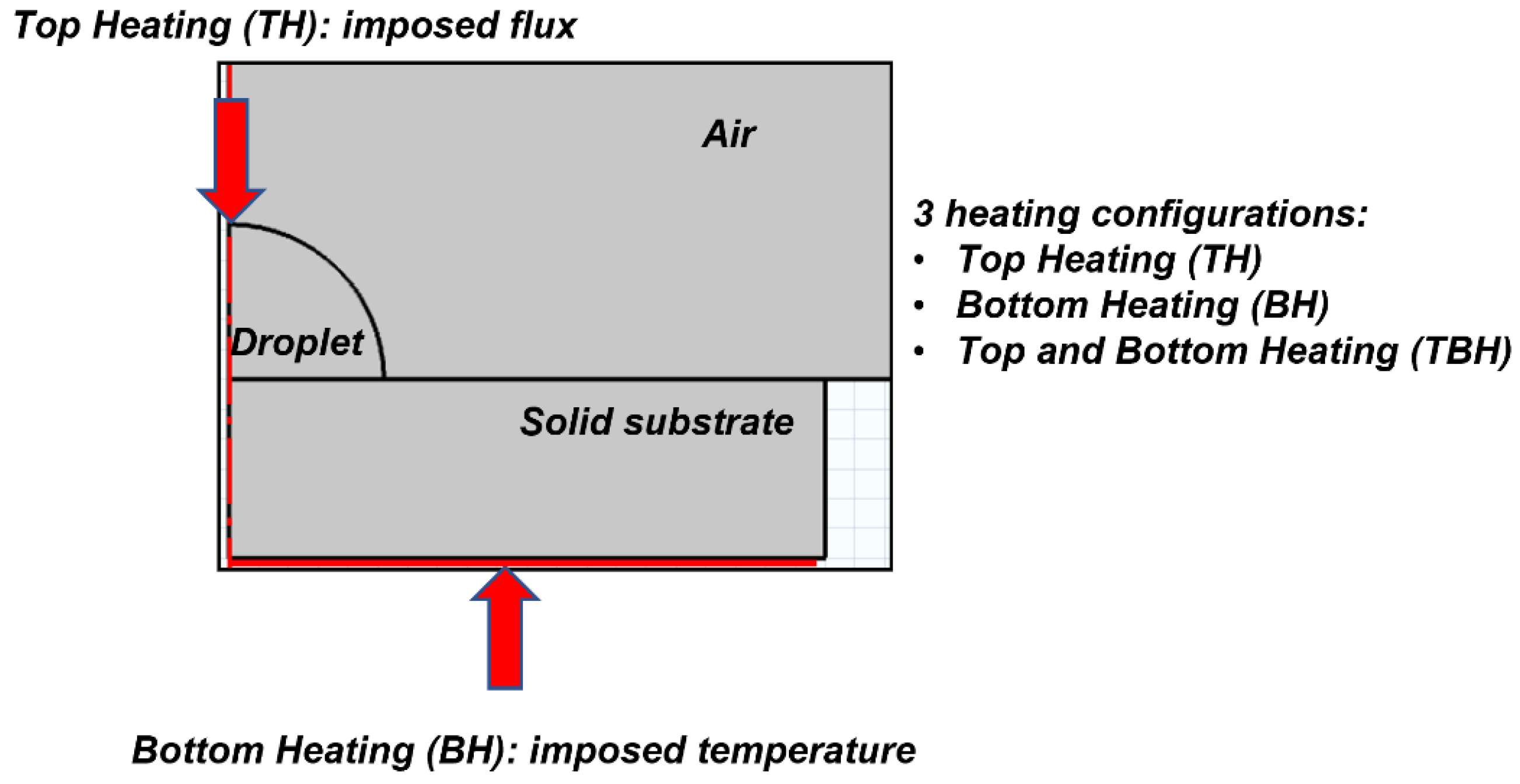
2.1. Physical Domain
2.2. ALE Formulation
- move with the materials (at the interface liquid–gas) to accurately reproduce the moving boundaries and interfaces of multi-domain systems;
- be fixed in space inside the material domain;
- be fixed in one direction and move with the material in other directions.
2.3. Governing Equations System
- Conduction equation in the solid substrate:
- Continuity, Navier–Stokes, and energy equations in the liquid droplet and gas domain:
- Advection-diffusion equation in the gas domain, with air surrounding the droplet:
- At the liquid–gas interface.
2.4. Initial and Boundary Conditions
2.4.1. Boundary Conditions
- At r = 0,
- At r = 0, z = h,
- At z = −e,
- At z = 0, 0 < r < R,
- At z = 0, R < r < L,
- At z = ∞,
2.4.2. Heating Configurations
3. Numerical Simulation
3.1. Mesh Velocity and Balanced Stresses
3.2. Computer Code and Used Grid
4. Results and Discussion
4.1. Model Validation
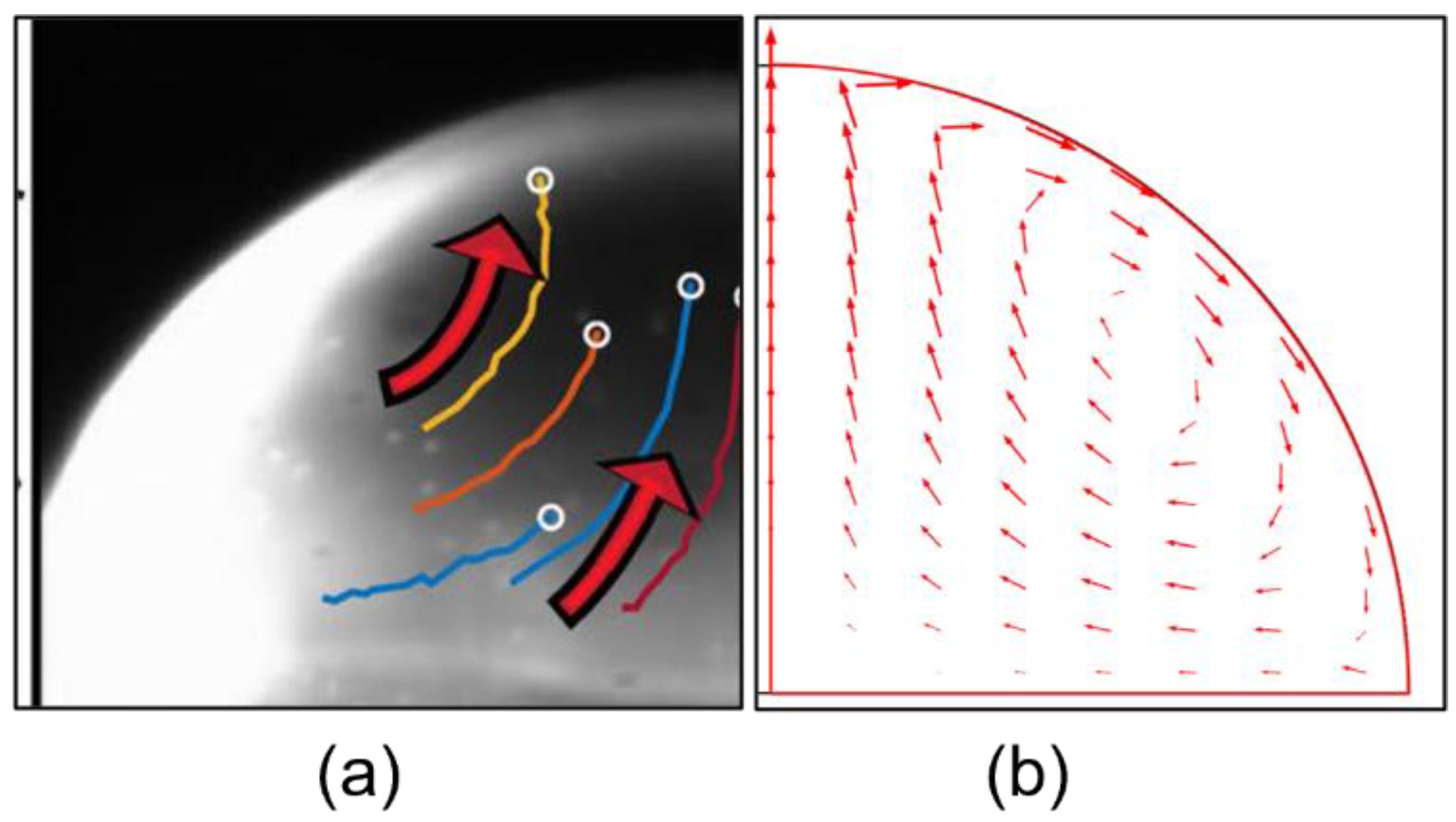
4.2. Marangoni Circulation in the Case of One Single Heat Source (TH and BH Configurations)
4.3. TBH Configuration and Nature Substrate Effect
4.3.1. Marangoni Circulation
4.3.2. Substrate Type Effect
4.3.3. Effect of the Substrate Thickness
5. Conclusions
- Using one single source of heat, the direction of Marangoni circulation is monotonic and can be chosen; upward flow occurs when the substrate is heated (BH) and a downward flow occurs when heat is supplied at the top of the droplet (TH);
- The combination of the two types of heating (TBH) triggered a Marangoni flow with two vortices separated by a stagnation point;
- The balance between the magnitude of the two heat sources was changed by the nature and the thickness of the substrate. The results show that the respective importance of the two vortices and the position of the stagnation point can be controlled.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| Symbols | Abbreviation |
| ALE | Arbitrary Lagrangian–Eulerian (-) |
| BH | Bottom Heating (-) |
| Bo | Bond number (-) |
| C | concentration (mol·L−1 or g·L−1) |
| Cp | heat capacity (J·kg−1·K−1) |
| D | diffusion coefficient (m2·s−1) |
| e | substrate thickness (m) |
| fst | force per unit area (N·m−2) |
| g | gravity acceleration (m·s−2) |
| h | droplet height (m) |
| H | humidity (%) |
| L | substrate length (m) |
| Lc | capillary length (m) |
| Lv | latent heat (J·kg−1) |
| mev | local evaporation rate (kg·m−2·s−1) |
| Mw | molar Mass (kg·mol−1) |
| n | normal direction (-) |
| p | pressure (Pa) |
| rc | curvature radius (m) |
| R | contact radius (m) |
| Rm | universal gas constant (J·mol−1·K−1) |
| (r,z) | cylindrical coordinates (m) |
| SZ | Stagnation zone (-) |
| t | time (s) |
| tangential direction (-) | |
| T | temperature (K or °C) |
| TH | Top Heating (-) |
| TBH | Top and Bottom Heating (-) |
| (u,w) | velocity components (m·s−1) |
| U | norm of velocity (m·s−1) |
| V | droplet volume (mm3 or µL) |
| x | space coordinates (m) |
| X | domain coordinates (m) |
| Xm | mesh coordinates (m) |
| Greek symbols | |
| ε | relative difference (%) |
| θ | contact angle (°) |
| λ | thermal conductivity (W·m−1·K−1) |
| μ | dynamic viscosity (Pa·s) |
| ρ | density (kg·m−3) |
| σ | surface tension (N·m−1) |
| τ | total stress tensor (-) |
| Φ | heat flux (W) |
| Subscripts | |
| c | convective |
| g | gas (air) |
| h | hot |
| l | liquid (water) |
| ∝ | infinite |
| 0 | ambient, reference |
| s | solid (substrate) |
| sat | saturation |
References
- Leenaars, A.F.M.; Huethorst, J.A.M.; Van Oekel, J.J. Marangoni drying: A new extremely clean drying process. Langmuir 1990, 6, 1701–1703. [Google Scholar] [CrossRef]
- Kim, J. Spray cooling heat transfer: The state of the art. Int. J. Heat Fluid Flow 2007, 28, 753–767. [Google Scholar] [CrossRef]
- Park, J.; Moon, J. Control of Colloidal Particle Deposit Patterns within Picoliter Droplets Ejected by Ink-Jet Printing. Langmuir 2006, 22, 3506–3513. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Dong, Z.; Kuang, M.; Li, Y.; Li, F.; Jiang, L.; Song, Y. Printing Patterned Fine 3D Structures by Manipulating the Three Phase Contact Line. Adv. Funct. Mater. 2015, 25, 2237–2242. [Google Scholar] [CrossRef]
- MacBeath, G.; Schreiber, S.L. Printing Proteins as Microarrays for High-Throughput Function Determination. Science 2000, 289, 1760–1763. [Google Scholar] [CrossRef]
- Dugas, V.; Broutin, A.J.; Souteyrand, E. Droplet Evaporation Study Applied to DNA Chip Manufacturing. Langmuir 2005, 21, 9130–9136. [Google Scholar] [CrossRef]
- Sempels, W.; De Dier, R.; Mizuno, H.; Hofkens, J.; Vermant, J. Auto-production of biosurfactants reverses the coffee ring effect in a bacterial system. Nat. Commun. 2013, 4, 1757. [Google Scholar] [CrossRef] [Green Version]
- Liu, H. Science and Engineering of Droplets—Fundamentals and Applications; Noyes Publications: Norwich, CT, USA, 2000. [Google Scholar]
- de Gennes, P.-G.; Brochard-Wyart, F.; Quere, D. Capillarity and Wetting Phenomena: Drops, Bubbles, Pearls, Waves; 2004th ed.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Sirignano, W.A. Fluid Dynamics and Transport of Droplets and Sprays, 2nd ed; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Brutin, D. (Ed.) Droplet Wetting and Evaporation: From Pure to Complex Fluids; Academic Press: San Diego, CA, USA, 2015. [Google Scholar]
- Murisic, N.; Kondic, L. On evaporation of sessile drops with moving contact lines. J. Fluid Mech. 2011, 679, 219–246. [Google Scholar] [CrossRef]
- Picknett, R.; Bexon, R. The evaporation of sessile or pendant drops in still air. J. Colloid Interface Sci. 1977, 61, 336–350. [Google Scholar] [CrossRef]
- Rowan, S.M.; Newton, M.I.; McHale, G. Evaporation of Microdroplets and the Wetting of Solid Surfaces. J. Phys. Chem. 1995, 99, 13268–13271. [Google Scholar] [CrossRef]
- Deegan, R.; Bakajin, O.; Dupont, T.F.; Huber, G.; Nagel, S.R.; Witten, T.A. Contact line deposits in an evaporating drop. Phys. Rev. E 2000, 62, 756–765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, H.; Larson, R.G. Evaporation of a Sessile Droplet on a Substrate. J. Phys. Chem. B 2002, 106, 1334–1344. [Google Scholar] [CrossRef]
- Erbil, H.Y. Evaporation of pure liquid sessile and spherical suspended drops: A review. Adv. Colloid Interface Sci. 2012, 170, 67–86. [Google Scholar] [CrossRef]
- Gelderblom, H.; Bloemen, O.; Snoeijer, J.H. Stokes flow near the contact line of an evaporating drop. J. Fluid Mech. 2012, 709, 69–84. [Google Scholar] [CrossRef] [Green Version]
- David, S.; Sefiane, K.; Tadrist, L. Experimental investigation of the effect of thermal properties of the substrate in the wetting and evaporation of sessile drops. Colloids Surf. A Physicochem. Eng. Asp. 2007, 298, 108–114. [Google Scholar] [CrossRef]
- Lu, G.; Duan, Y.-Y.; Wang, X.-D.; Lee, D.-J. Internal flow in evaporating droplet on heated solid surface. Int. J. Heat Mass Transf. 2011, 54, 4437–4447. [Google Scholar] [CrossRef]
- Lopes, M.C.; Bonaccurso, E.; Gambaryan-Roisman, T.; Stephan, P. Influence of the substrate thermal properties on sessile droplet evaporation: Effect of transient heat transport. Colloids Surfaces A Physicochem. Eng. Asp. 2013, 432, 64–70. [Google Scholar] [CrossRef]
- Maatar, A.; Chikh, S.; Saada, M.A.; Tadrist, L. Transient effects on sessile droplet evaporation of volatile liquids. Int. J. Heat Mass Transf. 2015, 86, 212–220. [Google Scholar] [CrossRef]
- Khilifi, D.; Foudhil, W.; Harmand, S.; Ben Jabrallah, S. Evaporation of a sessile oil drop in the Wenzel-like regime. Int. J. Therm. Sci. 2020, 151, 106236. [Google Scholar] [CrossRef]
- Saada, M.A.; Chikh, S.; Tadrist, L. Numerical investigation of heat and mass transfer of an evaporating sessile drop on a horizontal surface. Phys. Fluids 2010, 22, 112115. [Google Scholar] [CrossRef]
- Yang, K.; Hong, F.; Cheng, P. A fully coupled numerical simulation of sessile droplet evaporation using Arbitrary Lagrangian–Eulerian formulation. Int. J. Heat Mass Transf. 2013, 70, 409–420. [Google Scholar] [CrossRef]
- Askounis, A.; Sefiane, K.; Koutsos, V.; Shanahan, M.E. Effect of particle geometry on triple line motion of nano-fluid drops and deposit nano-structuring. Adv. Colloid Interface Sci. 2015, 222, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Girard, F.; Antoni, M.; Faure, S.; Steinchen, A. Influence of heating temperature and relative humidity in the evaporation of pinned droplets. Colloids Surfaces A Physicochem. Eng. Asp. 2008, 323, 36–49. [Google Scholar] [CrossRef]
- Diddens, C. Detailed finite element method modeling of evaporating multi-component droplets. J. Comput. Phys. 2017, 340, 670–687. [Google Scholar] [CrossRef]
- Diddens, C.; Tan, H.; Lv, P.; Versluis, M.; Kuerten, J.G.M.; Zhang, X.; Lohse, D. Evaporating pure, binary and ternary droplets: Thermal effects and axial symmetry breaking. J. Fluid Mech. 2017, 823, 470–497. [Google Scholar] [CrossRef] [Green Version]
- Diddens, C.; Kuerten, J.; van der Geld, C.; Wijshoff, H. Modeling the evaporation of sessile multi-component droplets. J. Colloid Interface Sci. 2017, 487, 426–436. [Google Scholar] [CrossRef]
- Foudhil, W.; Chen, P.; Fahem, K.; Harmand, S.; Ben Jabrallah, S. Study of the evaporation kinetics of pure and binary droplets: Volatility effect. Heat Mass Transf. 2021, 57, 1773–1790. [Google Scholar] [CrossRef]
- Hu, H.; Larson, R.G. Analysis of the Microfluid Flow in an Evaporating Sessile Droplet. Langmuir 2005, 21, 3963–3971. [Google Scholar] [CrossRef]
- Ristenpart, W.D.; Kim, P.G.; Domingues, C.; Wan, J.; Stone, H.A. Influence of Substrate Conductivity on Circulation Reversal in Evaporating Drops. Phys. Rev. Lett. 2007, 99, 234502. [Google Scholar] [CrossRef] [Green Version]
- Xu, X.; Luo, J.; Guo, D. Criterion for Reversal of Thermal Marangoni Flow in Drying Drops. Langmuir 2009, 26, 1918–1922. [Google Scholar] [CrossRef]
- Shi, W.-Y.; Tang, K.-Y.; Ma, J.-N.; Jia, Y.-W.; Li, H.-M.; Feng, L. Marangoni convection instability in a sessile droplet with low volatility on heated substrate. Int. J. Therm. Sci. 2017, 117, 274–286. [Google Scholar] [CrossRef]
- Tan, H.; Diddens, C.; Lv, P.; Kuerten, J.G.M.; Zhang, X.; Lohse, D. Evaporation-triggered microdroplet nucleation and the four life phases of an evaporating Ouzo drop. Proc. Natl. Acad. Sci. USA 2016, 113, 8642–8647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karapetsas, G.; Sahu, K.C.; Matar, O.K. Evaporation of Sessile Droplets Laden with Particles and Insoluble Surfactants. Langmuir 2016, 32, 6871–6881. [Google Scholar] [CrossRef] [PubMed]
- Deegan, R.D.; Bakajin, O.; Dupont, T.F.; Huber, G.; Nagel, S.R.; Witten, T.A. Capillary flow as the cause of ring stains from dried liquid drops. Nature 1997, 389, 827–829. [Google Scholar] [CrossRef]
- Mampallil, D.; Eral, H.B. A review on suppression and utilization of the coffee-ring effect. Adv. Colloid Interface Sci. 2018, 252, 38–54. [Google Scholar] [CrossRef] [PubMed]
- Hu, A.H.; Larson, R.G. Marangoni Effect Reverses Coffee-Ring Depositions. J. Phys. Chem. B 2006, 110, 7090–7094. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, X.; Xin, Z.; Deng, M.; Wen, Y.; Song, Y. Controlled Inkjetting of a Conductive Pattern of Silver Nanoparticles Based on the Coffee-Ring Effect. Adv. Mater. 2013, 25, 6714–6718. [Google Scholar] [CrossRef]
- Lee, K.-H.; Kim, S.-M.; Jeong, H.; Jung, G.-Y. Spontaneous nanoscale polymer solution patterning using solvent evaporation driven double-dewetting edge lithography. Soft Matter 2011, 8, 465–471. [Google Scholar] [CrossRef]
- Cui, L.; Zhang, J.; Zhang, X.; Li, Y.; Wang, Z.; Gao, H.; Wang, T.; Zhu, S.; Yu, H.; Yang, B. Avoiding coffee ring structure based on hydrophobic silicon pillar arrays during single-drop evaporation. Soft Matter 2012, 8, 10448–10456. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, J.; Su, B.; Shi, L.; Wang, J.; Chen, S.; Wang, L.; Zi, J.; Song, Y.; Jiang, L. Colloidal Photonic Crystals with Narrow Stopbands Assembled from Low-Adhesive Superhydrophobic Substrates. J. Am. Chem. Soc. 2012, 134, 17053–17058. [Google Scholar] [CrossRef]
- Crivoi, A.; Duan, F. Three-dimensional Monte Carlo model of the coffee-ring effect in evaporating colloidal droplets. Sci. Rep. 2014, 4, 4310. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yen, T.M.; Fu, X.; Wei, T.; Nayak, R.U.; Shi, Y.; Lo, Y.-H. Reversing Coffee-Ring Effect by Laser-Induced Differential Evaporation. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Gaalen, R.; Diddens, C.; Wijshoff, H.; Kuerten, J. Marangoni circulation in evaporating droplets in the presence of soluble surfactants. J. Colloid Interface Sci. 2020, 584, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Legros, J.; Limbourg-Fontaine, M.; Petre, G. Influence of a surface tension minimum as a function of temperature on the marangoni convection. Acta Astronaut. 1984, 11, 143–147. [Google Scholar] [CrossRef]
- Limbourg-Fontaine, M.C.; Pétré, G.; Legros, J.C. Thermocapillary movements under microgravity at a minimum of surface tension. Sci. Nat. 1986, 73, 360–362. [Google Scholar] [CrossRef]
- Savino, R.; Cecere, A.; Di Paola, R. Surface tension-driven flow in wickless heat pipes with self-rewetting fluids. Int. J. Heat Fluid Flow 2009, 30, 380–388. [Google Scholar] [CrossRef]
- Ouenzerfi, S.; Harmand, S. Experimental Droplet Study of Inverted Marangoni Effect of a Binary Liquid Mixture on a Nonuniform Heated Substrate. Langmuir 2016, 32, 2378–2388. [Google Scholar] [CrossRef]
- Ruiz, O.E.; Black, W.Z. Evaporation of Water Droplets Placed on a Heated Horizontal Surface. J. Heat Transf. 2002, 124, 854–863. [Google Scholar] [CrossRef]
- Mollaret, R.; Sefiane, K.; Christy, J.R.; Veyret, D. Experimental and Numerical Investigation of the Evaporation into Air of a Drop on a Heated Surface. Chem. Eng. Res. Des. 2004, 82, 471–480. [Google Scholar] [CrossRef]
- Ganesan, S.; Tobiska, L.U.T.Z. Finite element simulation of a droplet impinging a horizontal surface. Proc. Algoritm. 2005, 2005, 1–11. [Google Scholar]
- Bozorgmehr, B.; Murray, B.T. Numerical Simulation of Evaporation of Ethanol–Water Mixture Droplets on Isothermal and Heated Substrates. ACS Omega 2021, 6, 12577–12590. [Google Scholar] [CrossRef] [PubMed]
- Comsol Multiphysics. Micro-Fluidics Module, User’s Guide, Theory for the Two Phase Flow, Moving Mesh User Interface; Comsol Multiphysics: Kingston, Australia, 2012; pp. 113–121. [Google Scholar]
- Song, H.; Lee, Y.; Jin, S.; Kim, H.-Y.; Yoo, J.Y. Prediction of sessile drop evaporation considering surface wettability. Microelectron. Eng. 2011, 88, 3249–3255. [Google Scholar] [CrossRef]
















| Grid 1 29,261 Elements | Grid 2 35,557 Elements | Grid 3 41,034 Elements | |
|---|---|---|---|
| TH (Φ = 40 mW) | |||
| V (mm3) | 0.7239 | 0.7219 | 0.7235 |
| T (°C) | 33.061 | 33.161 | 33.080 |
| mev (g·m−2·s−1) | 3.9119 | 3.9045 | 3.8955 |
| BH (Th = 50 °C) | |||
| V (mm3) | 0.3211 | 0.3282 | 0.3271 |
| T (°C) | 45.532 | 45.738 | 45.732 |
| mev (g·m−2·s−1) | 2.3767 | 2.2721 | 2.2824 |
| TBH (Th = 50 °C, Φ = 3 mW) | |||
| V (mm3) | 0.3193 | 0.3151 | 0.3184 |
| T (°C) | 47.683 | 47.708 | 47.692 |
| mev (g·m−2·s−1) | 3.2700 | 3.2499 | 3.2213 |
| λ (W·m−1·K−1) | ρ (kg·m−3) | Cp (J·kg−1·K−1) | |
|---|---|---|---|
| Glass | 1.38 | 2203 | 703 |
| PTFE | 0.25 | 2200 | 1010 |
| e (mm) | 0.4 | 0.6 | 0.8 | 1 |
| XSZ (mm) | 0.837 | 0.762 | 0.576 | 0.240 |
| (Xi+1 − Xi)/Xi+1 | - | 8.96% | 24.41% | 58.33% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Foudhil, W.; Aricò, C.; Perré, P.; Ben Jabrallah, S. Use of Heating Configuration to Control Marangoni Circulation during Droplet Evaporation. Water 2022, 14, 1653. https://doi.org/10.3390/w14101653
Foudhil W, Aricò C, Perré P, Ben Jabrallah S. Use of Heating Configuration to Control Marangoni Circulation during Droplet Evaporation. Water. 2022; 14(10):1653. https://doi.org/10.3390/w14101653
Chicago/Turabian StyleFoudhil, Walid, Costanza Aricò, Patrick Perré, and Sadok Ben Jabrallah. 2022. "Use of Heating Configuration to Control Marangoni Circulation during Droplet Evaporation" Water 14, no. 10: 1653. https://doi.org/10.3390/w14101653
APA StyleFoudhil, W., Aricò, C., Perré, P., & Ben Jabrallah, S. (2022). Use of Heating Configuration to Control Marangoni Circulation during Droplet Evaporation. Water, 14(10), 1653. https://doi.org/10.3390/w14101653









