Abstract
Various extracts are tested for anti-allergic or anti-inflammatory properties on in vitro models. RBL-2H3 cells are widely used in allergic or immunological studies. FCεRI and its downstream signaling cascades, such as MAPK, NF-κB, and JAK/STAT signaling pathways, are important allergic or inflammatory signaling mechanisms in mast and basophil cells. This systematic review aims to study common signaling pathways of the anti-allergic or anti-inflammatory compounds on RBL-2H3 cells. We selected the relevant research articles published after 2015 from the PubMed, Scopus, Science Direct and Web of Science databases. The risk of bias of the studies was assessed based on the modified CONSORT checklist for in vitro studies. The cell lines, treatments, assay, primary findings, and signaling pathways on RBL-2H3 cells were extracted to synthesize the results. Thirty-eight articles were included, and FCεRI and its downstream pathways, such as Lyn, Sky, PLCγ, and MAPK, were commonly studied. Moreover, the JAK/STAT pathway was a potential signaling mechanism in RBL-2H3 cells. However, the findings based on RBL-2H3 cells needed to be tested along with human mast cells to confirm its relevance to human health. In conclusion, a single plant extract may act as an anti-inflammatory reagent in RBL-2H3 cells via multiple signaling pathways besides the MAPK signaling pathway.
1. Introduction
Allergy is an immunological disorder due to non-toxic environmental factors such as pollen, food, dust, drugs, insect venom, latex, hormones, fungal spores, and vaccines [1,2,3,4,5,6,7]. Allergies are increasing across developed and developing countries [1,2,6,8,9,10], affecting about 20% of the global population [11,12]. The four types of allergies or hypersensitivities are anaphylactic, cytotoxic, immune complex, and delayed, which are called type I, II, III, or IV, respectively [8,13,14]. Type I allergies (e.g., food allergies, asthma, and allergic rhinitis) are the most common [3,4,8,11,13].
How do type I allergies begin? There are two stages: initially, when an allergen is exposed for the first time, which is referred to as the sensitization or induction phase [15], antigen-presenting cells (APCs) or macrophages recognize, engulf, and present it to naïve T cells, which will differentiate into T helper 2 (Th2) cells. These Th2 cells produce pro-inflammatory cytokines such as interleukin-4 (IL-4), IL-5, or IL-13, which convert B cells into IgE-producing cells (i.e., plasma cells). The produced IgE binds with the α-subunit of high-affinity IgE receptors (FCεRIs) of mast and basophil cells [3,16], which are primary effector cells in type I allergy [14]. When the same allergen is present again, it is called the effector phase [15]. The allergen will crosslink two adjacent FCεRI -bound IgE, triggering various downstream signaling cascades such as tyrosine kinase, protein kinase C (PKC), mitogen-activated protein kinase (MAPK) [3,12,17], Janus kinase-signal transducer and activator of transcription (JAK/STAT), and nuclear factor κB (NF-κB) [9], as well as calcium influx [12] and cytoskeleton remodeling, recruiting secondary cells such as neutrophils [15], causing degranulation (i.e., histamine and β-hexosaminidase release), the production of reactive oxygen species (ROS) [18], and various pro-inflammatory cytokines and chemokines production [2,3,4,6,11,13,17,19,20,21,22,23].
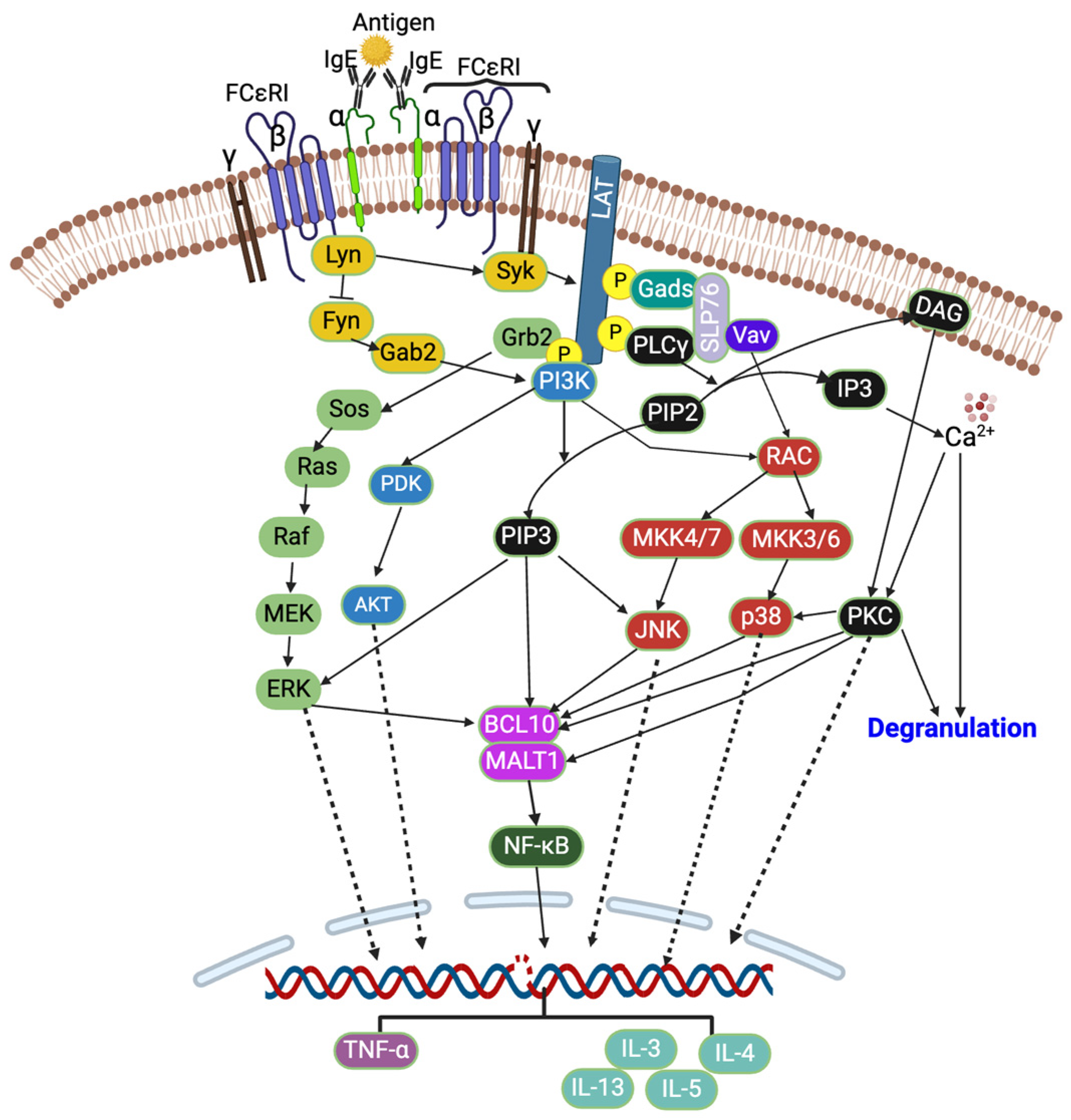
The crosslinking of IgE by an allergen activates the heterotetrameric (one α, one β, and two γ subunits) FCεRI receptors, which further activate two Src family kinases, Lyn and Fyn, which are protein tyrosine kinases (PTKs) (Figure 1). These Lyn and Fyn activate FCεRIβ-immunoreceptor tyrosine-based activation motifs (ITAMs) and recruit spleen tyrosine kinase (Syk) to FCεRIβ-ITAMs [14,24]. The Syk further activates other signaling cascades such as PKC, protein kinase B (Akt), phosphoinositide 3-kinase (PI3K), rat sarcoma (Ras), guanosine triphosphatase (GTPase), and phospholipase Cγ (PLCγ) [13,16,17,20,25,26]. The activated Syk also activates linkers for the activation of T cells (LAT) and src homology 2 (SH2) domain-containing leukocyte-specific phosphoproteins of 76 kd (SLP-76). This is followed by the event where cytosolic adaptor molecules such as growth factor receptor bound protein 2 (Grb2), glutamic acid decarboxylase 2 (Gad2), PLCγ1, and guanine exchange factors (VAV and SOS) bind to LAT, and this further activates PI3K and MAPK signaling pathways [14,24]. The PLCγ converts phosphatidylinositol 4,5-bisphosphate (PIP2) into diacylglycerol (DAG) and inositol triphosphate (IP3), which further reduces the intracellular Ca2+ [27,28]. The Ca2+ and DAG activate PKC [14,28], which then activates p38 MAPK, B cell lymphoma/leukemia 10 (BCL10), and mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MALT1) [28]. The activated PI3K converts PIP2 into phosphatidylinositol 3,4,5-triphosphate (PIP3), and PIP3 further activates JNK (via RAC and MAPK4) and ERK1/2 (Figure 1). The ERK1/2 activates phospholipase A2 (PLA2), increasing leukotriene and prostaglandins [28].
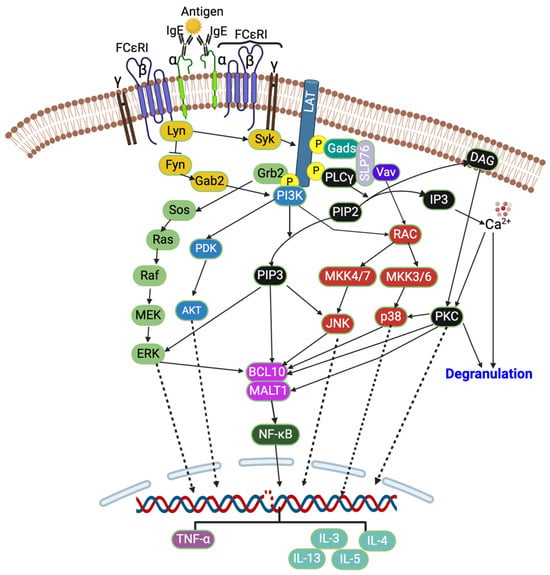
Figure 1.
FCεRI signaling pathway. Created with BioRender.com.
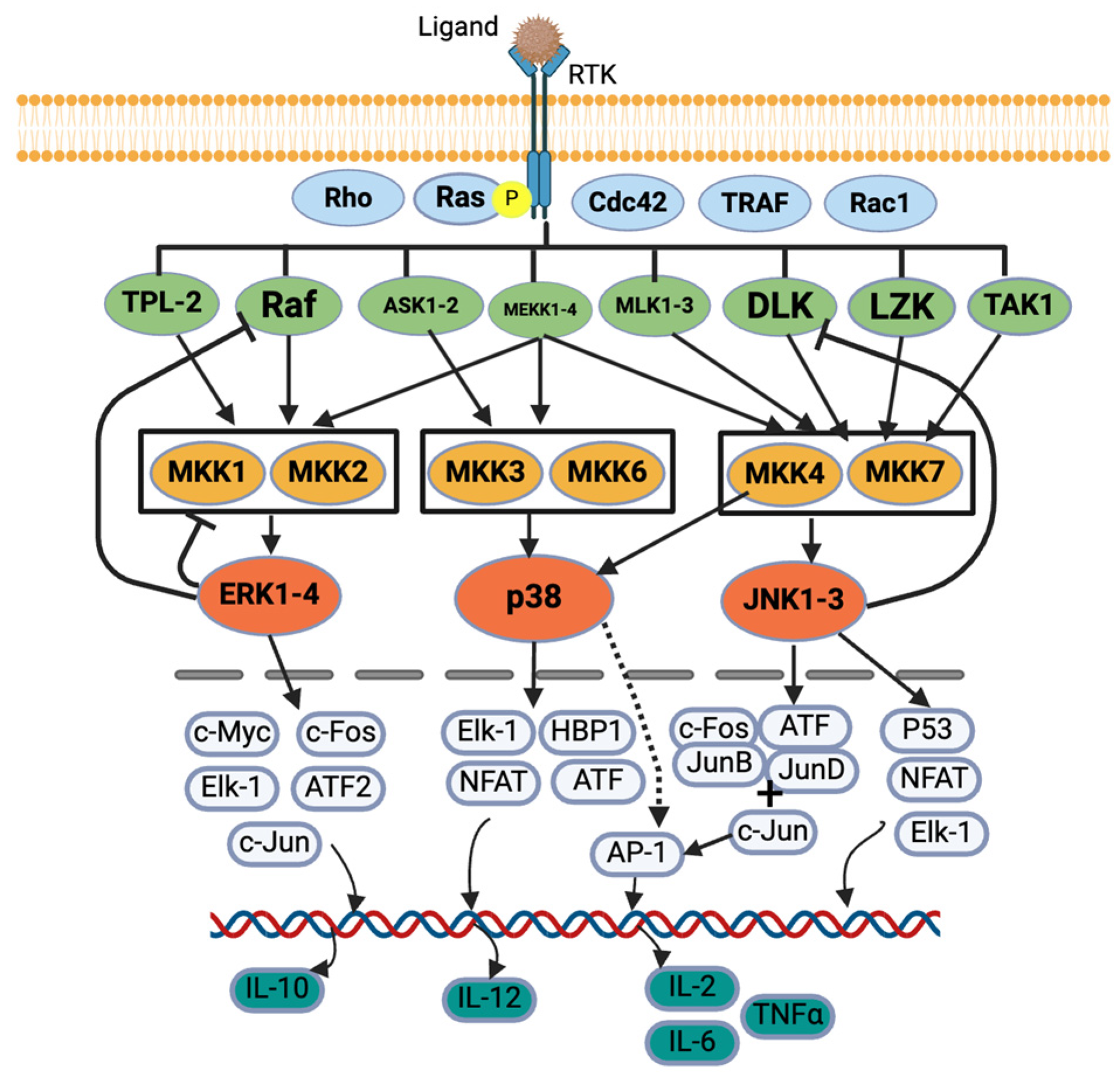
MAPK and JAK/STAT are the most important signaling pathways for allergies [9,25]. Five protein kinases’ sequential activation regulates the MAPK signaling cascade: MAPK kinase kinase kinase (MAP4K), MAPK kinase kinase (MAP3K), MAPK kinase (MAPKK), MAPK, and MAPK-activated protein kinases (MAPKAPK). In general, MAP3K, MAP2K, and MAPK are commonly explained in studies [29,30]. MAPK has four categories based on their structure, activation motif, and function: extracellular signal-regulated kinase 1/2 (ERK1/2), p38 MAPK, c-Jun N terminal kinase 1/2 (JNK1/2) [9,11,12,30], and ERK5 (not commonly explained) [29]. While pro-inflammatory stimuli activate all categories of MAPK, growth factors and hormones activate ERK1/2, and cellular and environmental stresses activate p38 MAPK and JNK 1/2 [30] (Figure 2).
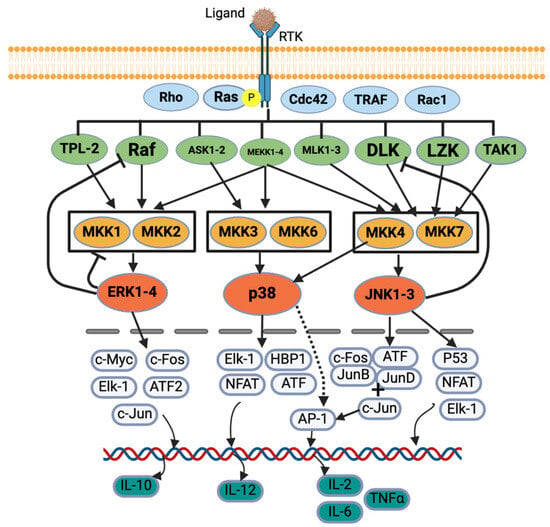
Figure 2.
MAPK signaling pathway. Created with BioRender.com.
In the canonical activation of the ERK1/2 MAPK cascade, a ligand first binds to a receptor tyrosine kinase (RTK), activating a G-protein, Ras. The Ras recruits and activates serine/threonine protein kinase, Raf (a MAP3K), which activates MAPK/ERK kinases (MEK1/2) (a MAP2K also known as MKK1/2), and these MEKs further activate ERK1/2 (a MAPK) [29,30,31,32] (Figure 2). ERK1/2 also provides negative feedback to the proteins, e.g., SOS protein, Raf-1 and MEKs, upstream of the signaling cascades [29]. Once translocated into the nucleus, the ERK regulates various transcription factors such as c-Fos, c-Jun, c-Myc, Elk-1, and ATF2 [29]. The ERK regulates IL-10 production, which helps Th cells to Th2 cells producing IL-4, -5, -9, and -13 [30] (Figure 2). Syk also activates ERK1/2, which further activates the arachidonic signaling pathways and production of TNF-α, IL-2, IL-5, and IL-13 in mast cells.
For p38 MAPK activation, tumor necrosis factor receptor-associated factor 2/3/6 (TRAF) or Rho protein activates MEKK1, SAK1, or TAK1 (a MAPK3K) in response to stress or cytokines. They activate MKK3 or MKK6 (a MAP2K), which finally activates p38 MAPK [30,32,33,34]. The p38 further regulates the transcription factors such as ATF, NFAT, Elk-1, and HBP1, which further regulates the cytokine production [34] (Figure 2). The p38 MAPK regulates IL-12 production, which promotes Th cell differentiation into Th2 cells, which produce IL-2, IFN-γ, and TNF-α/β [30].
Diverse biotic and abiotic stresses, including cytokines (e.g., TNF and IL-1), activate the JNK pathway via various receptors such as TNFR, GPCR, TGFBR, and TLR [35]. Various signals activate Rac1/Cdc42, which activates their downstream proteins such as MLK, ASK, DLK, MEKK, and TAK. They further activate MKK4 or MKK7, which ultimately activates JNK [32,33]. JNK also provides negative feedback to an upstream protein, DLK [35]. The JNK further acts on various transcription factors such as c-Fos, ATF, Jun B, Jun D, and c-Jun, which activates AP-1 that regulates cytokine production [32,33,35]. The JNK also regulates P53, NFAT, and Elk-1 [32] (Figure 2). The p38 MAPK activates IL-4, and the JNK activates IL-2, IL-6, and TNF-α [9,25].
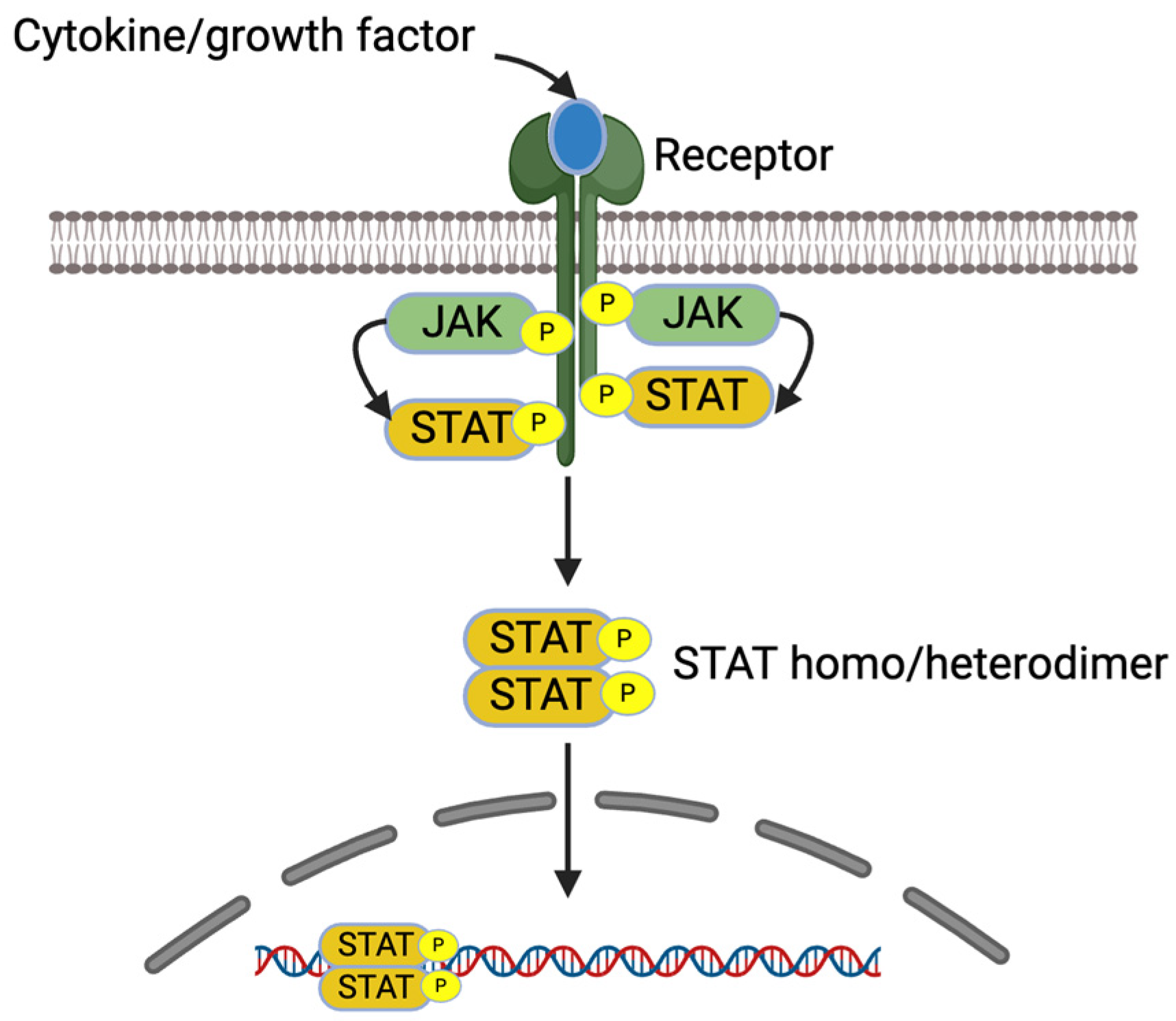
IL-4 and IL-13 increase pro-inflammatory gene expression in allergic diseases via JAK/STAT signaling pathways [7]. These cytokines bind receptors, which lead to receptor dimerization and the recruitment of Janus kinase (JAK). The activated JAKs activate the receptors and recruit STATs to the receptors. The activated STATs dissociate from the receptors as homo or heterodimers and are translocated into the nucleus, bind into DNA, and regulate the gene expression [7,36] (Figure 3).
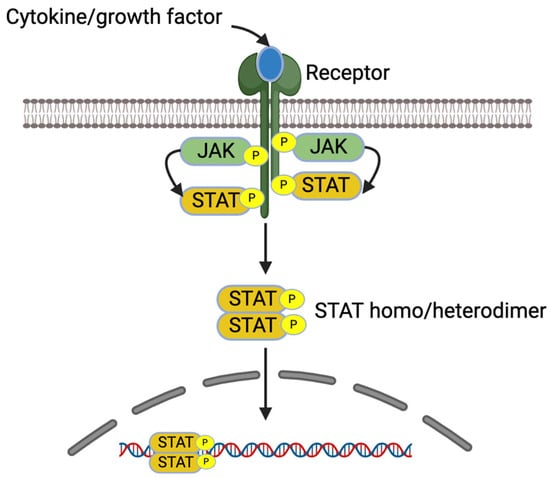
Figure 3.
JAK/STAT signaling pathway. Created with BioRender.com.
MAPK can activate NF-kB [28]. Inactive NF-kB in cytoplasm is found in trimeric form with an NF-kB inhibitor (IkB). Signals from MEKK1 activate the IkB kinase complex (IKK) [37], which further activates IkB (at Ser 32 and Ser 36 residues), releasing NF-kB into the nucleus where it binds to the kB binding site of promotor regions and activates gene expression and mediators—for example, COX-2, TNF-α, IL-1β, -6, and -8 [38,39,40]. The activated NF-kB presents in the heterodimer form of p65 and p50 subunits where the activated IkB is ubiquitinated by the 26S proteasome [39].
Mast cells are vital for allergic reaction, as their activation leads to the release of mast cell mediators such as histamine, leukotriene C4 (LTC4) and prostaglandin D2 (PGD2), causing early reaction. Other mast cell mediators such as IL-3, IL-5, IL-8, and tumor-necrosis factor (TNF) recruit eosinophils, neutrophils, and Th2 cells, and they also interact with other tissue cells to start late-phage allergic reaction [41]. Since mast cells regulate allergic reactions in a multi-faceted way, anti-allergic drugs to control the allergic reaction due to mast cells could be used at various levels such as (a) targeting mast cell mediators and their receptors, (b) mast cell activators, their receptors, and signal transduction, (c) mast cell inhibitory receptors, and (d) reducing mast cell numbers [42]. In other words, mast cells can be targeted with anti-allergic drugs at three levels: the cell membrane (e.g., Omalizumab, which targets free IgE and reduces the IgE attachment to FCεRI), intracellular (e.g., Syk kinase inhibitors, which block IgE-FCεRI-mediated downstream phosphorylation) or extracellular (e.g., H1–4 receptor antagonists which prevent binding and the effect of histamine on target cells) [43,44]. However, since mast-cell-derived mediators are also produced by other cells, targeting those mediators would not provide the answer to the absolute necessity of mast cells for the reaction. On the other hand, mast cell activating receptors are unique to those cells, and blocking them, for example by using anti-IgE antibodies omalizumab and ligelizumab, could provide the definite role of mast cells for the reaction [42]. These monoclonal antibodies block the attachment of IgE to FCεRI on mast cells and indirectly reduce the FCεRI expression in the cell, resulting in downstream signaling reduction [43].
The rat basophilic leukemia cell (RBL-2H3) line is commonly used for various allergic and immunological studies, specially to study the IgE-FCεRI-mediated signaling mechanism [8,10,12,17,23,45], as it has similar characteristics (such as the presence of histamine, β-hexosaminidase, and the expression of FCεRI) [46] and response behavior as those of mast cells and basophil cells [2,10,21,22,25,47]. However, detailed information on RBL-2H3’s potential to test multiple signaling pathways/mechanisms of various anti-allergic or anti-inflammatory compounds or extracts on RBL-2H3 cell lines is scarce. Thus, this systematic review aims to study the common signaling pathways of anti-allergic and anti-inflammatory compounds in RBL-2H3 cells.
2. Materials and Methods
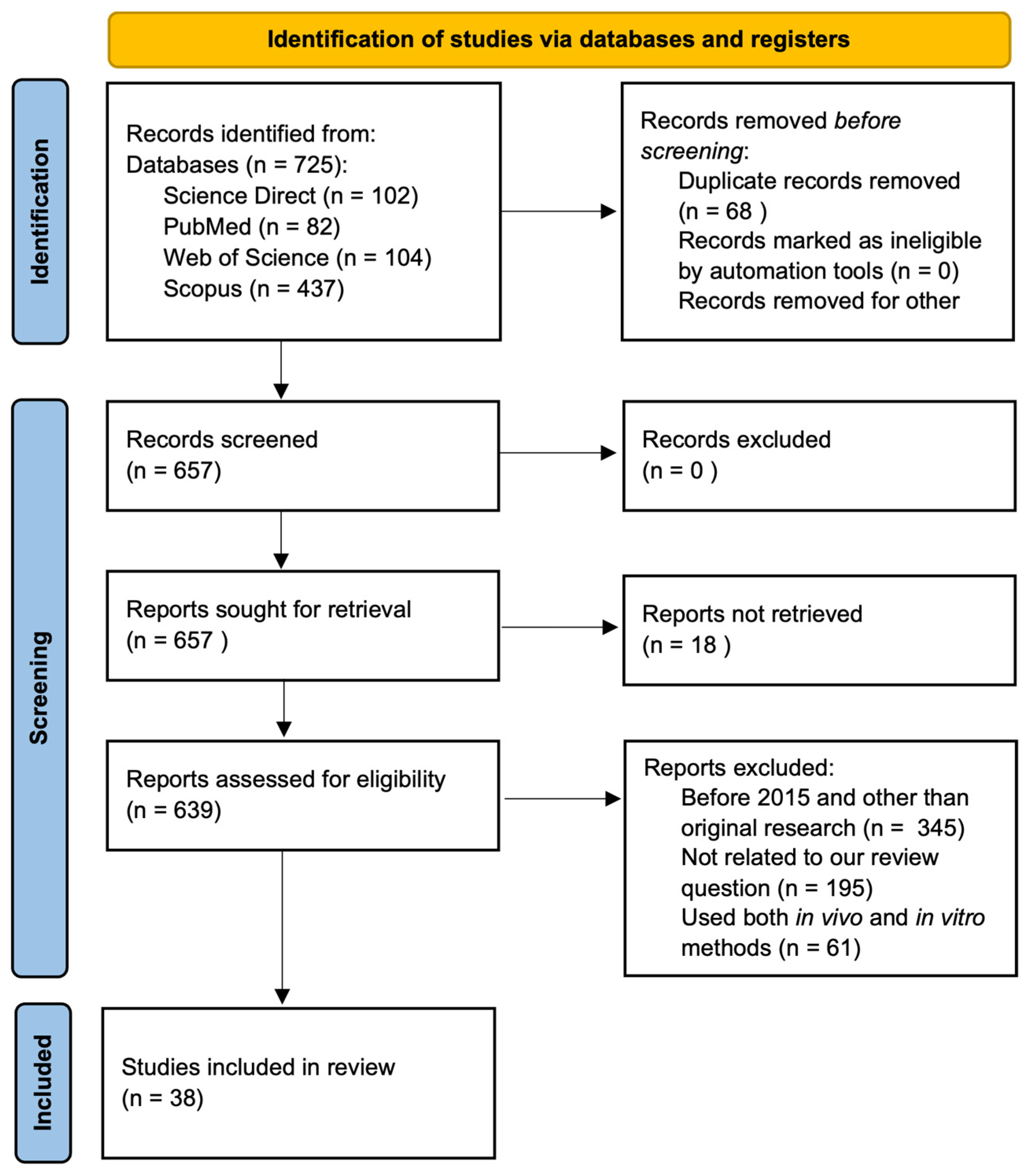
This systematic review was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines [48], and the PRISMA flow chart shows the detail of the articles’ selection process and search results (Figure 4). Out of the 657 total articles, 18 were not fully retrieved, 345 were published before 2015 and were not original research articles, 195 were not closely related to the topics, and 61 included both in vitro and in vivo systems, leading to the final 38 articles for the review.
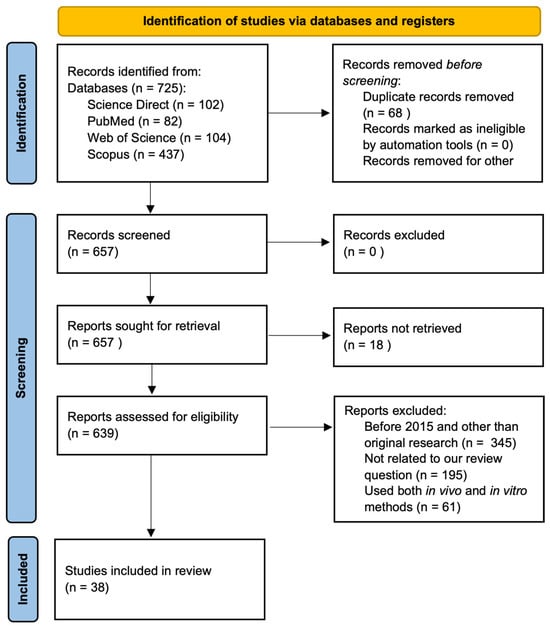
Figure 4.
PRISMA flow diagram showing literature search and selection process.
We searched literature utilizing four databases: PubMed, Scopus, Science Direct, and Web of Science until 5 March 2024. We used the following keywords, in the same order, in Scopus and Science Direct: (“RBL-2H3”) AND (MAPK OR “JAK/STAT” OR “NFkB” OR “FCεRI” OR “signaling pathway”) AND (“allergy” OR “peanut allergy”) AND (polyphenol). In Web of Science, all keywords mentioned above were identical except that “Polyphenol” was omitted. For PubMed, we used following keywords: ((“RBL-2H3”) AND (MAPK OR “JAK/STAT” OR “NFkB” OR “FCεRI” OR “signaling pathway”) AND (“allergy” OR “peanut allergy”)) and the following Medical Subject Headings (MeSH) separately: ((((rbl-2h3) OR (“Mast Cells” [Majr])) AND (“Signal Transduction” [Majr])) AND (“Hypersensitivity” [Majr])). Two authors (T.S.R. and R.B.) screened the articles, and any disagreements were solved through discussion and consultation with a third author (L.L.W.).
The PICO framework was used to determine the evidence in the articles as follows: population: RBL-2H3 cells only (or along with other cell lines); intervention: natural compounds/extracts; comparison: extract/compound-treated cells versus untreated cells; outcome: signaling pathways, cell degranulation, and inflammatory cytokines.
The articles were deduplicated by uploading them in Excel sheets followed by the title and abstract screening for inclusion/exclusion criteria and were further evaluated on full text for their eligibility. Two reviewers (T.S.R. and R.R.B.) independently screened the title and abstract for the inclusion. The original research articles using an in vitro system, published in the English language in peer-reviewed journals from 2015 to 2024 (5 March), were included for the review. Review articles, proceedings, or articles published in languages other than English, articles published before 2015, articles not closely related to the topic (i.e., those that did not include RBL-2H3 cells, pro-inflammatory cytokines, or signaling pathways/molecules), and articles that included both in vitro and in vivo systems were not selected in this review. We excluded studies that used both in vitro and in vivo systems to focus our review on an in vitro system using RBL-2H3 and other cells in order to avoid variability and complexity in our study. Two authors (T.S.R. and R.R.B.) agreed upon the selection criteria and on selected articles.
The following items were manually and independently extracted and agreed by two authors (T.S.R. and R.R.B.) from the selected articles: publication year, objectives, cell lines used, dose and duration of the treatment/control, sequence of sensitization, treatment and stimulation, assay/techniques used, primary findings, and treatment effect on signaling pathways/molecules on RBL-2H3 cells. When information on signaling pathways were not provided in the included articles, cell degranulation signature molecules such as histamine and β-hexosaminidase and cytokines expression data were obtained. Moreover, the compounds’ extraction methods were not included from the selected articles.
The risk of bias of selected articles was independently assessed and agreed by two authors (T.S.R. and R.R.B.) using a modified CONSORT checklist for in vitro study [49,50]. The comprehensiveness and unbiased reporting on the following items of the articles were assessed: abstract, background and rationale, objectives, hypothesis, intervention, outcome, statistical method, outcome estimation, limitations, and funding. As the articles were diverse in terms of treatment used, objectives, and design, we used a narrative synthesis approach to synthesize the articles. The information were tabulated according to cell line used, i.e., either only RBL-2H3 or RBL-2H3 along with other cell lines. The synthesis steps were conducted by two authors (T.S.R. and R.R.B.) and agreed among all three authors (T.S.R., R.R.B., and L.L.W.).
3. Results
3.1. Study Characteristics, Risk of Bias, and Reporting Quality
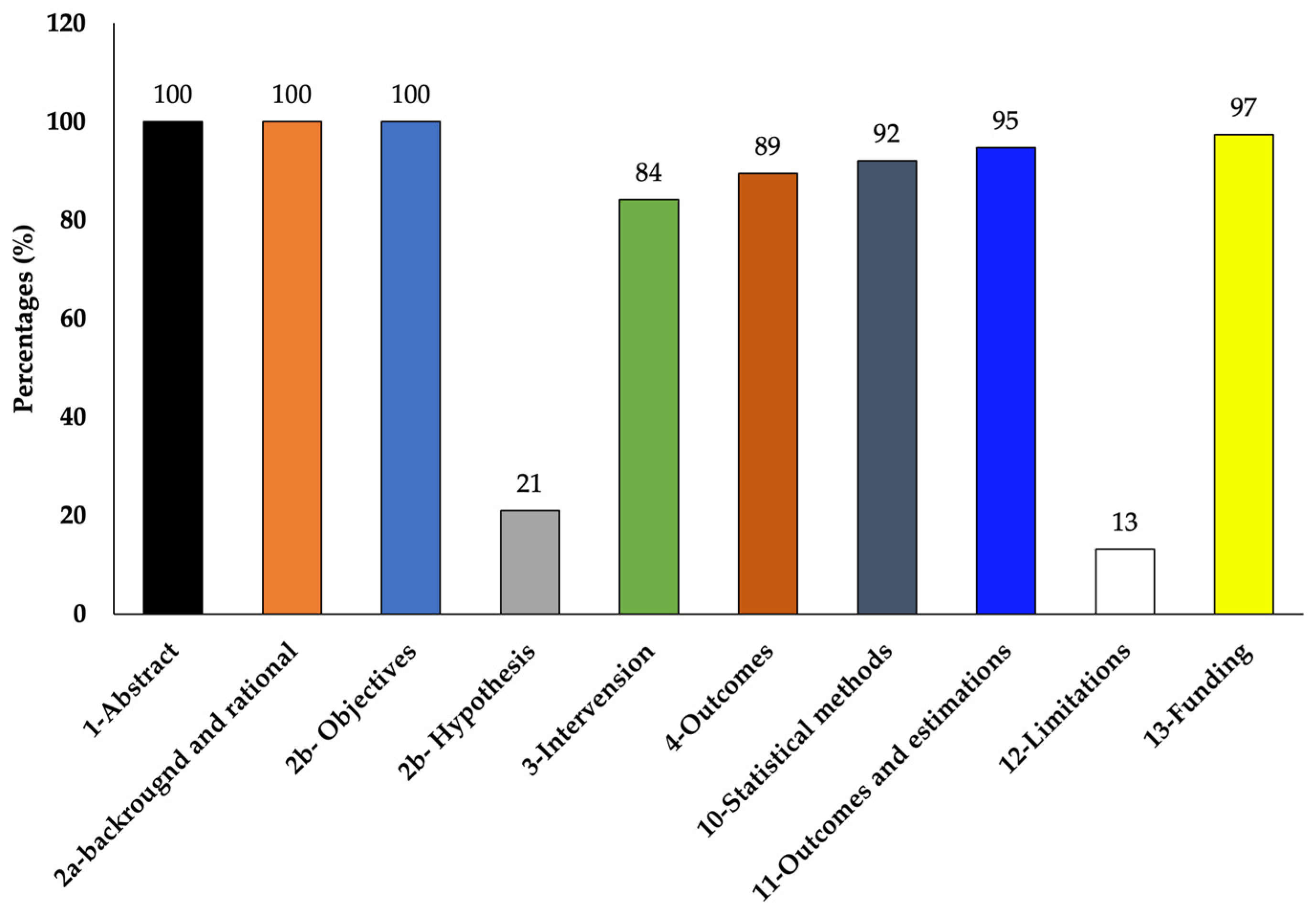
The selected studies varied in their objectives and overall experimentation. Out of the total of 38 chosen studies, 26 studies used only RBL-2H3 cells and 12 studies used RBL-2H3 along with other cells such as RAW264.7 (5 studies), HaCaT (2 studies), HiMC (1 study), BMMC (1 study), Caco2 (1 study), MoLT-4 (1 study), HMC-1 (1 study), KU812 (1 study), human neutrophils (2 studies), human basophils (1 study), and bacteria (Klebestella pneumonia) (2 studies) (Table 1 and Table 2). According to the CONSORT checklist used [49,50], although most of the studies did not mention the hypothesis and limitation of their studies clearly, the majority of them clearly explained the experimental details so that the experiment could be replicated and also reported complete results showing quality results with low risk of bias (Table 3 and Figure 5).

Table 1.
Anti-allergic and anti-inflammatory properties and signaling pathways of various compounds using only RBL-2H3 cells.

Table 2.
Anti-allergic and anti-inflammatory properties and signaling pathways of various compounds using RBL-2H3 cell and other cell lines.

Table 3.
Quality assessment of reviewed studies according to modified CONSORT checklist [49,50].
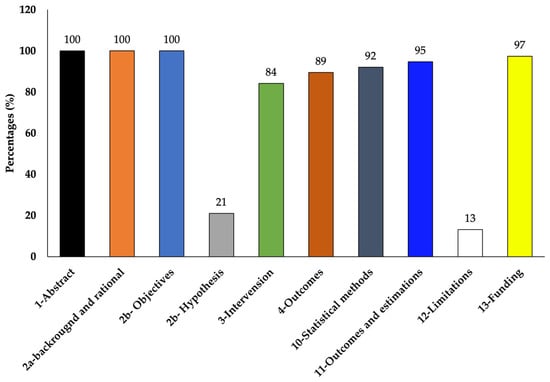
Figure 5.
Percentages of the reviewed articles that met the modified CONSORT checklist.
3.2. Signaling Pathways
Data extracted from the included studies are summarized in Table 1 and Table 2. Studies had diverse compounds tested for their anti-allergic or anti-inflammatory properties and possible signaling mechanisms. For example, studies including only RBL-2H3 had extracts from plants, shrimps, fungi, sponges, and chemicals (sodium sulfite) (Table 1). However, the majority of the studies were related to plant extracts. Similarly, studies those used multiple types of cells also had all plant extracts except one study with enzymatically synthesized glycogen (Table 2). Some studies did not explore the signaling mechanisms of the tested compounds in depth. In contrast, most of the studies had shown that the anti-allergic or anti-inflammatory activities of tested compounds on RBL-2H3 were via the regulation of MAPK, FCεRI, NF-kB signaling pathways, or their upstream or downstream regulators such as Syk, Lyn, Akt, PLCγ, PI3K, PKC, and LAT (Table 1 and Table 2). In addition to FCεRI and its downstream signaling, some studies have also shown that RBL-2H3 cells demonstrate JAK/STAT and NLRP3 signaling pathways when they were tested for anti-allergic mechanisms of fungus extract and sodium sulfite, respectively (Table 1). Moreover, the studies had shown that a single compound can have multiple signaling pathways (Table 1 and Table 2).
4. Discussion
This review has the following limitations. (i) We used peer-reviewed articles published in the English language, which may have excluded important results published in other languages and in preliminary result reports. (ii) We only included in vitro studies and articles published after 2015. (iii) We did not manually search and include the literature from the included articles’ reference lists. (iv) There are no standard methods on quality evaluation of the in vitro studies, so some modified methods recommended for the clinical setting were used. These shortcomings prevent the comparison of in vivo and in vitro results and the relevance of in vitro studies’ findings for further clinical trials. Moreover, exclusion criteria may lead to missing valuable sources regarding the use of RBl-2H3 for novel signaling mechanisms for allergic or immunological research. Furthermore, the lack of a standard protocol for in vitro study quality may also result in variations in the articles’ quality. However, this review provides a trend on the use of RBL-2H3 cells and their potential signaling mechanisms for allergic and inflammatory studies over a decade.
The studies had diverse compounds tested, such as a variety of extracts from plants (on the majority of included articles), fungi [9,21], shrimp [56], sponges [20], agro-industrial waste [6,53,54], and chemical compounds [55]. Thus, there is also a need for multiple studies using the same or similar compounds, which may be met by broadening the literature inclusion criteria. Moreover, the included studies primarily studied MAPK signaling pathways, and other critical pathways such as JAK/STAT pathways were barely reported. This might be related to the fact that RBL-2H3 cells are not exactly the same as mast cells or basophil cells. For example, studies have reported that RBL-2H3 cells lack some properties of mast cells, such as the absence of essential elements such as CD14 and MyD88. However, they have Toll-like receptor 4 (TLR4) on their surface [64], which might have limited the cells from being used in TLR-related signaling pathways such as JAK/STAT pathways. However, some studies suggested that the presence of CD14 does not guarantee that the TLR-acting stimulus, such as LPS, directly activates the cells [65]. However, some of the included studies had reported STAT6 signaling pathways [9] and NLRP3 and caspase pathways [55] in RBL-2H3 cells, indicating their potential use for diverse signaling mechanisms. Moreover, studies related to new applications of tested compounds/extracts, such as using them as a nutraceutical or in industry-standard applications, still need to be included.
From this study, it is also evident that the dose and duration of the tested compound can impact the level of expression or presence and absence of the tested signaling pathways. The literature also reported the variations on the RBL-2H3 phenotype based on various factors such as cultural condition [47,64], age, and biological sex from which the cell line was isolated [46]. Thus, it is important to provide details of the used cell’s properties (e.g., cell passage, catalog number) in the studies for their reproducibility.
Despite the mast cells and basophil cells being similar to RBL-2H3 in many properties, we excluded them in this review because RBL-2H3 cells still differ from those cells in various characteristics such as the amount of histamine in the cells, the absence of CD14, TLR-2, the presence of c-kit, which is a receptor for stem cell factor (SCF) [65], the absence of CD123, which is a signature of primary basophil, and not expressing rat tryptase genes and release tryptase, which are found in primary mast cells [46].
It is also essential to critically consider the relevance or implication of RBL-2H3 studies’ results to human mast cells. There are various limitations on the use of human mast cells or basophil cells for studies. For example, the isolation and purification of human mast cells and basophil cells is expensive, tedious, impractical, and results in issues in the isolated cells’ purity and viability [41,66]. Moreover, various human mast cell lines have been developed but suffered from vital issues over time. For instance, HMC-1 and HMC-α failed to degranulate following immunological activation; LAD-2 has a high cost and slow growth rate, so it is not the first choice for routine experimental studies; ROSA KIT D816V does not have histamine and β-hexosaminidase expression; and ROS KIT WT needs stem cell factor (SCF) to survive, which is very expensive and not affordable for routine studies [41,66]. Due to these limitations for the use of actual human mast cells, the use of RBL-2H3 cells becomes prominent, as these cells are easy to culture, have homogeneity with a rapid growth rate, have functional FCεRI, and have similar degranulation dynamics to human mast cell and basophil cells [41,66]. However, despite some similarities between RBL-2H3 cells and the mast cell (e.g., both have c-kit receptors for SCF), there are differences, too. For example, unlike in mast cells, the TLR4 or its co-receptor CD14 and TLR2 are not expressed in RBL-2H3 [47,65]. Moreover, since RBL-2H3 cells are immortalized cells, their physiology might not be the same as that of primary mast cell physiology, and RBL-2H3 cells also show intra-laboratory reproducibility issues. Thus, the results obtained with these cells should be validated along with the human mast cell lines [41,46,47,66].
In the included studies, other types of cells (not RBL-2H3) were used to study the antioxidant properties, pro-inflammatory cytokines, or gene-level expression of some allergic markers. Moreover, the crude extract versus purified compound may have affected the expression of signaling pathways on RBL-2H3 cells. The variation in the dose and time of the tested compounds and sensitizing/stimulating agents such as anti-DNP-IgE (33.3 ng/mL for 2 h—1 μg/mL for overnight), ionomycin (1 μM for 30 min–250 μM for 30 min), DNP-BSA (50 ng/mL for 1 h—10 μg/mL for 30 min), calcium ionophore A23187 (0.15 ug/mL for 30 min—1 μM for 2.5 h), and DNP-HSA (5 ng/mL for 1 h—100 ng/mL for 18 sec) (Table 1 and Table 2) may also contribute to variation in the signaling mechanisms of the tested compounds on RBL-2H3 cells.
5. Conclusions
Most of the compounds tested on RBL-2H3 cells had shown that FCεRI and MAPK signaling pathways are dominant for anti-allergic and anti-inflammatory mechanisms. Moreover, due to variations in the types of the tested compounds, it is also revealed that a single compound can affect various stages of multiple signaling pathways. The RBL-2H3 cells should have cytokine receptors (required for JAK/STAT signaling) considering their similarities with basophil and mast cells which have cytokine receptors in their cell membranes [41]. Moreover, a fungus extract called viridicatol treatment on RBL-2H3 had reduced the STAT6 expression [9]. Thus, RBL-2H3 may also be able to be tested for JAK/STAT signaling pathway for plant extracts.
Author Contributions
Conceptualization, T.S.R. and R.R.B.; methodology, T.S.R. and R.R.B.; software, T.S.R.; validation, T.S.R., R.R.B. and L.L.W.; formal analysis, T.S.R. and R.R.B.; investigation, T.S.R.; resources, L.L.W.; data curation, T.S.R. and R.R.B.; writing—original draft preparation, T.S.R.; writing—review and editing, R.R.B. and L.L.W.; visualization, T.S.R.; supervision, L.L.W.; project administration, L.L.W.; funding acquisition, L.L.W. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by 1890 Capacity Building Grant Program from the United States Department of Agriculture and National Institute of Food and Agriculture (Project award No. 2023-38821-39979).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Badger-Emeka, L.I.; Emeka, P.M.; Thirugnanasambantham, K.; Ibrahim, H.I.M. Anti-Allergic Potential of Cinnamaldehyde via the Inhibitory Effect of Histidine Decarboxylase (HDC) Producing Klebsiella pneumonia. Molecules 2020, 25, 5580. [Google Scholar] [CrossRef]
- Barbosa, M.; Lopes, G.; Valentão, P.; Ferreres, F.; Gil-Izquierdo, Á; Pereira, D.M.; Andrade, P.B. Edible seaweeds’ phlorotannins in allergy: A natural multi-target approach. Food Chem. 2018, 265, 233. [Google Scholar] [CrossRef]
- Chang, Y.; Hsu, W.; Pan, T. Monascus Secondary Metabolites Monascin and Ankaflavin Inhibit Activation of RBL-2H3 Cells. J. Agric. Food Chem. 2015, 63, 192. [Google Scholar] [CrossRef]
- Kobayashi, S.; Kato, T.; Azuma, T.; Kikuzaki, H.; Abe, K. Anti-allergenic activity of polymethoxyflavones from Kaempferia parviflora. J. Funct. Foods 2015, 13, 100. [Google Scholar] [CrossRef]
- Lorenz, P.; Heinrich, M.; Garcia-Käufer, M.; Grunewald, F.; Messerschmidt, S.; Herrick, A.; Gruber, K.; Beckmann, C.; Knoedler, M.; Huber, R.; et al. Constituents from oak bark (Quercus robur L.) inhibit degranulation and allergic mediator release from basophils and mast cells in vitro. J. Ethnopharmacol. 2016, 194, 642. [Google Scholar] [CrossRef] [PubMed]
- Mwakalukwa, R.; Ashour, A.; Amen, Y.; Niwa, Y.; Tamrakar, S.; Miyamoto, T.; Shimizu, K. Anti-allergic activity of polyphenolic compounds isolated from olive mill wastes. J. Funct. Foods 2019, 58, 207. [Google Scholar] [CrossRef]
- Shankar, A.; Mcalees, J.W.; Lewkowich, I.P. Modulation of IL-4/IL-13 cytokine signaling in the context of allergic disease. J. Allergy Clin. Immunol. 2022, 150, 266. [Google Scholar] [CrossRef]
- Lee, E.; Yu, M.; Garcia, C.V.; Jhee, K.; Yang, S. Inhibitory effect of Zizania latifolia chloroform fraction on allergy-related mediator production in RBL-2H3 cells. Food Sci. Biotechnol. 2017, 26, 481. [Google Scholar] [CrossRef]
- Liu, Y.; Shu, Z.; Li, Y.; Chen, H.; Liu, H.; Yang, X.; Liu, G.; Liu, Q. Deep-sea-derived viridicatol relieves allergic response by suppressing MAPK and JAK-STAT signalling pathways of RBL-2H3 cells. Food Agric. Immunol. 2023, 34, 2207791. [Google Scholar] [CrossRef]
- Ma, J.; Tong, P.; Chen, Y.; Wang, Y.; Ren, H.; Gao, Z.; Yue, T.; Long, F. The inhibition of pectin oligosaccharides on degranulation of RBL-2H3 cells from apple pectin with high hydrostatic pressure assisted enzyme treatment. Food Chem. 2022, 371, 131097. [Google Scholar] [CrossRef]
- Li, X.; Li, H.; Wang, T.; Zhao, Y.; Shao, Y.; Sun, Y.; Zhang, Y.; Liu, Z. Network pharmacology-based analysis of the mechanism of Saposhnikovia divaricata for the treatment of type I allergy. Pharm. Biol. 2022, 60, 1224. [Google Scholar] [CrossRef]
- Yan, Z.; Feng, X.; Li, X.; Gao, Z.; Wang, Z.; Ren, G.; Long, F. Sea Buckthorn Flavonoid Extracted by High Hydrostatic Pressure Inhibited IgE-Stimulated Mast Cell Activation through the Mitogen-Activated Protein Kinase Signaling Pathway. Foods 2024, 13, 560. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, Y.; Inoue, M.; Yoshioka, H.; Kitakaze, T.; Furuyashiki, T.; Abe, N.; Ashida, H. Enzymatically synthesized glycogen inhibited degranulation and inflammatory responses through stimulation of intestine. J. Clin. Biochem. Nutr. 2020, 67, 67–73. [Google Scholar] [CrossRef]
- Li, Y.; Leung, P.S.C.; Gershwin, M.E.; Song, J. New Mechanistic Advances in FcεRI-Mast Cell–Mediated Allergic Signaling. Clin. Rev. Allerg. Immunol. 2022, 63, 431. [Google Scholar] [CrossRef]
- Dera, A.; Rajagopalan, P.; Ahmed, I.; Alfhili, M.; Alsughayyir, J.; Chandramoorthy, H.C. Thymoquinone attenuates IgE-mediated allergic response via pi3k-Akt-NFκB pathway and upregulation of the Nrf2-HO1 axis. J. Food Biochem. 2020, 44, e13216. [Google Scholar] [CrossRef]
- Park, C.; Min, S.; Yu, H.; Kim, K.; Kim, S.; Lee, H.; Kim, J.; Park, Y. Effects of Apigenin on RBL-2H3, RAW264.7, and HaCaT Cells: Anti-Allergic, Anti-Inflammatory, and Skin-Protective Activities. IJMS 2020, 21, 4620. [Google Scholar] [CrossRef]
- Yoo, G.; Lee, K.; Lee, D. Inhibitory effects of 2-oxo-2H-chromen-4-yl 4-methylbenzenesulfonate on allergic inflammatory responses in rat basophilic leukemia cells. Int. Immunopharmacol. 2017, 48, 196. [Google Scholar] [CrossRef]
- Vo, T.S.; Le, T.T.; Kim, S.; Ngo, D. The role of myricetin fromRhodomyrtus tomentosa(Aiton) Hassk fruits on downregulation of FcεRI-mediated mast cell activation. J. Food Biochem. 2020, 44, e13143. [Google Scholar] [CrossRef]
- Hagenlocher, Y.; Kießling, K.; Schäffer, M.; Bischoff, S.C.; Lorentz, A. Cinnamaldehyde is the main mediator of cinnamon extract in mast cell inhibition. Eur. J. Nutr. 2015, 54, 1297. [Google Scholar] [CrossRef]
- Jiao, W.; Cheng, B.; Shi, G.; Chen, G.; Gu, B.; Zhou, Y.; Hong, L.; Yang, F.; Liu, Z.; Qiu, S.; et al. Dysivillosins A–D, Unusual Anti-allergic Meroterpenoids from the Marine Sponge Dysidea villosa. Sci. Rep. 2017, 7, 8947. [Google Scholar] [CrossRef]
- Kawai, J.; Higuchi, Y.; Hirota, M.; Hirasawa, N.; Mori, K. Ergosterol and its derivatives from Grifola frondosa inhibit antigen-induced degranulation of RBL-2H3 cells by suppressing the aggregation of high affinity IgE receptors. Biosci. Biotechnol. Biochem. 2018, 82, 1803. [Google Scholar] [CrossRef]
- Kong, Z.L.; Sudirman, S.; Lin, H.J.; Chen, W.N. In vitro anti-inflammatory effects of curcumin on mast cell-mediated allergic responses via inhibiting FceRI protein expression and protein kinase C delta translocation. Cytotechnology 2020, 72, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Zhu, S.; Tamura, T.; Kadowaki, M.; Wang, Z.; Yoshimatsu, K.; Komatsu, K. Chemical constituents with anti-allergic activity from the root of Edulis Superba, a horticultural cultivar of Paeonia lactiflora. J. Nat. Med. 2016, 70, 234. [Google Scholar] [CrossRef]
- Athari, S.S. Targeting cell signaling in allergic asthma. Sig. Transduct Target Ther. 2019, 4, 45. [Google Scholar] [CrossRef]
- Lee, J.; Park, S.; Jhee, K.; Yang, S. Tricin Isolated from Enzyme-Treated Zizania latifolia Extract Inhibits IgE-Mediated Allergic Reactions in RBL-2H3 Cells by Targeting the Lyn/Syk Pathway. Molecules 2020, 25, 2084. [Google Scholar] [CrossRef]
- Min, S.; Park, C.; Yu, H.; Park, Y. Anti-Inflammatory and Anti-Allergic Effects of Saponarin and Its Impact on Signaling Pathways of RAW 264.7, RBL-2H3, and HaCaT Cells. IJMS 2021, 22, 8431. [Google Scholar] [CrossRef]
- Lim, S.; Oh, S.; Nguyen, Q.T.N.; Kim, M.; Zheng, S.; Fang, M.; Yi, T. Rosa davurica Inhibited Allergic Mediators by Regulating Calcium and Histamine Signaling Pathways. Plants 2023, 12, 1572. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.; Shim, K.; Jeoung, D. The Crosstalk between FcεRI and Sphingosine Signaling in Allergic Inflammation. IJMS 2022, 23, 3892. [Google Scholar] [CrossRef]
- Guo, Y.; Pan, W.; Liu, S.; Shen, Z.; Xu, Y.; Hu, L. ERK/MAPK signalling pathway and tumorigenesis (Review). Exp. Ther. Med. 2020, 19, 1997–2007. [Google Scholar] [CrossRef]
- Soares-Silva, M.; Diniz, F.F.; Gomes, G.N.; Bahia, D. The Mitogen-Activated Protein Kinase (MAPK) Pathway: Role in Immune Evasion by Trypanosomatids. Front. Microbiol. 2016, 7, 183. [Google Scholar] [CrossRef]
- Yuan, J.; Dong, X.; Yap, J.; Hu, J. The MAPK and AMPK signalings: Interplay and implication in targeted cancer therapy. J. Hematol. Oncol. 2020, 13, 113. [Google Scholar] [CrossRef]
- Cui, J.; Zhang, M.; Zhang, Y.; Xu, Z. JNK pathway: Diseases and therapeutic potential. Acta Pharmacol. Sin. 2007, 28, 601. [Google Scholar] [CrossRef]
- Davis, R.J. Signal Transduction by the JNK Group Review of MAP Kinases. Cell 2000, 103, 239–252. [Google Scholar]
- Zarubin, T.; Han, J. Activation and signaling of the p38 MAP kinase pathway. Cell Res. 2005, 15, 11–18. [Google Scholar]
- Zeke, A.; Misheva, M.; Reményi, A.; Bogoyevitch, M.A. JNK Signaling: Regulation and Functions Based on Complex Protein-Protein Partnerships. Microbiol. Mol. Biol. Rev. 2016, 80, 793. [Google Scholar] [CrossRef]
- Hu, X.; Li, J.; Fu, M.; Zhao, X.; Wang, W. The JAK/STAT signaling pathway: From bench to clinic. Sig. Transduct Target Ther. 2021, 6, 402. [Google Scholar] [CrossRef]
- Schulze-Osthoff, K.; Ferrari, D.; Riehemann, K.; Wesselborg, S. Regulation of NF-KB Activation by MAP Kinase Cascades. Immunobiology 1997, 198, 35–49. [Google Scholar]
- Wang, Q.; Gao, H.; Yuan, R.; Han, S.; Li, X.; Tang, M.; Dong, B.; Li, J.; Zhao, L.; Feng, J.; et al. Procyanidin A2, a polyphenolic compound, exerts anti-inflammatory and anti-oxidative activity in lipopolysaccharide-stimulated RAW264.7 cells. PLoS ONE 2020, 15, e0237017. [Google Scholar] [CrossRef]
- Chu, H.; Tang, Q.; Huang, H.; Hao, W.; Wei, X. Grape-seed proanthocyanidins inhibit the lipopolysaccharide-induced inflammatory mediator expression in RAW264.7 macrophages by suppressing MAPK and NF-κb signal pathways. Environ. Toxicol. Pharmacol. 2015, 41, 159. [Google Scholar] [CrossRef]
- Dasiman, R.; Md Nor, N.; Eshak, Z.; Syairah Mohd Mutalip, S.; Suwandi, R.; Bidin, H. A Review of Procyanidin: Updates on Current Bioactivities and Potential Health Benefits. Biointerface Res. Appl. Chem. 2021, 12, 5918. [Google Scholar] [CrossRef]
- Bischoff, S.C. Role of mast cells in allergic and non-allergic immune responses: Comparison of human and murine data. Nat. Rev. Immunol. 2007, 7, 93. [Google Scholar] [CrossRef] [PubMed]
- Kolkhir, P.; Elieh-Ali-Komi, D.; Metz, M.; Siebenhaar, F.; Maurer, M. Understanding human mast cells: Lesson from therapies for allergic and non-allergic diseases. Nat. Rev. Immunol. 2021, 22, 294. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Wilson, T.M.; Metcalfe, D.D. The mast cell and allergic diseases: Role in pathogenesis and implications for therapy. Clin. Exp. Allergy 2007, 38, 4. [Google Scholar] [CrossRef]
- Metz, M.; Kolkhir, P.; Altrichter, S.; Siebenhaar, F.; Levi-schaffer, F.; Youngblood, B.A.; Church, M.K.; Maurer, M. Mast cell silencing: A novel therapeutic approach for urticaria and other mast cell-mediated diseases. Allergy 2023, 79, 37. [Google Scholar] [CrossRef]
- Korinek, M.; Chen, K.; Jiang, Y.; El-Shazly, M.; Stocker, J.; Chou, C.; Hwang, T.; Wu, Y.; Chen, B.; Chang, F. Anti-allergic potential of Typhonium blumei: Inhibition of degranulation via suppression of PI3K/PLCγ2 phosphorylation and calcium influx. Phytomedicine 2016, 23, 1706. [Google Scholar] [CrossRef]
- Wagner, A.; Alam, S.B.; Kulka, M. The effects of age, origin, and biological sex on rodent mast cell (BMMC and MC/9) and basophil (RBL-2H3) phenotype and function. Cell. Immunol. 2023, 391–392, 104751. [Google Scholar] [CrossRef]
- Passante, E.; Frankish, N. The RBL-2H3 cell line: Its provenance and suitability as a model for the mast cell. Inflamm. Res. 2009, 58, 737. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Mckenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021. [Google Scholar] [CrossRef]
- Faggion, C.M. Guidelines for Reporting Pre-clinical In Vitro Studies on Dental Materials. J. Evid. Based Dent. Pract. 2012, 12, 182. [Google Scholar] [CrossRef]
- Lam, T.; Tran, N.N.; Pham, L.D.; Lai, N.V.; Dang, B.N.; Truong, N.N.; Nguyen-Vo, S.; Hoang, T.; Mai, T.T.; Tran, T. Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: A systematic review of in vitro studies. Nat. Prod. Bioprospect. 2024, 14, 4. [Google Scholar] [CrossRef]
- Bansode, R.R.; Plundrich, N.J.; Randolph, P.D.; Lila, M.A.; Williams, L.L. Peanut flour aggregation with polyphenolic extracts derived from peanut skin inhibits IgE binding capacity and attenuates RBL-2H3 cells degranulation via MAPK signaling pathway. Food Chem. 2018, 263, 307. [Google Scholar] [CrossRef]
- Dippenaar, C.; Shimbo, H.; Okon, K.; Miller, N.; Joubert, E.; Yoshida, T.; De Beer, D. Anti-Allergic and Antioxidant Potential of Polyphenol-Enriched Fractions from Cyclopia subternata (Honeybush) Produced by a Scalable Process. Separations 2022, 9, 278. [Google Scholar] [CrossRef]
- Gaihre, Y.R.; Iwamoto, A.; Oogai, S.; Hamajima, H.; Tsuge, K.; Nagata, Y.; Yanagita, T. Perilla pomace obtained from four different varieties have different levels and types of polyphenols and anti-allergic activity. Cytotechnology 2022, 74, 341. [Google Scholar] [CrossRef]
- Hamauzu, Y.; Kawahara, T. Anti-degranulation and bile acid-binding activity of extracts from fruits and agro-industrial by-products. Food Biosci. 2021, 43, 101244. [Google Scholar] [CrossRef]
- Liu, M.; Lu, J.; Chen, Y.; Shi, X.; Li, Y.; Yang, S.; Yu, J.; Guan, S. Sodium Sulfite-Induced Mast Cell Pyroptosis and Degranulation. J. Agric. Food Chem. 2021, 69, 7755. [Google Scholar] [CrossRef]
- Lv, L.; Ahmed, I.; Qu, X.; Ju, G.; Yang, N.; Guo, Y.; Li, Z. Effect of the structure and potential allergenicity of glycated tropomyosin, the shrimp allergen. Int. J. Food Sci. Technol. 2022, 57, 1782. [Google Scholar] [CrossRef]
- Matsui, T.; Ito, C.; Masubuchi, S.; Itoigawa, M. Licarin A is a candidate compound for the treatment of immediate hypersensitivity via inhibition of rat mast cell line RBL-2H3 cells. J. Pharm. Pharmacol. 2015, 67, 1723. [Google Scholar] [CrossRef]
- Matsui, T.; Ito, C.; Itoigawa, M.; Shibata, T. Three phlorotannins from Sargassum carpophyllum are effective against the secretion of allergic mediators from antigen-stimulated rat basophilic leukemia cells. Food Chem. 2022, 377, 131992. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Wei, J.; Li, X.; Jin, Y.; Shi, X. Inhibitory activity of narirutin on RBL-2H3 cells degranulation. Immunopharmacol. Immunotoxicol. 2020, 43, 68. [Google Scholar] [CrossRef]
- Vo, T.S.; Kim, S.; Ryu, B.; Ngo, D.H.; Yoon, N.; Bach, L.G.; Hang, N.; Ngo, D.H. The Suppressive Activity of Fucofuroeckol-A Derived from Brown Algal Ecklonia stolonifera Okamura on UVB-Induced Mast Cell Degranulation. Mar. Drugs 2018, 16, 1. [Google Scholar] [CrossRef]
- Zeng, J.; Hao, J.; Yang, Z.; Ma, C.; Gao, L.; Chen, Y.; Li, G.; Li, J. Anti-Allergic Effect of Dietary Polyphenols Curcumin and Epigallocatechin Gallate via Anti-Degranulation in IgE/Antigen-Stimulated Mast Cell Model: A Lipidomics Perspective. Metabolites 2023, 13, 628. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, H.; Li, X.; Sun, Y.; Shao, Y.; Zhang, Y.; Liu, Z. Network pharmacology-based analysis and experimental in vitro validation on the mechanism of Paeonia lactiflora Pall. in the treatment for type I allergy. BMC Complement Med. Ther. 2022, 22, 199. [Google Scholar] [CrossRef]
- Hanieh, H.; Hairul Islam, V.I.; Saravanan, S.; Chellappandian, M.; Ragul, K.; Durga, A.; Venugopal, K.; Senthilkumar, V.; Senthilkumar, P.; Thirugnanasambantham, K. Pinocembrin, a novel histidine decarboxylase inhibitor with anti-allergic potential in in vitro. Eur. J. Pharmacol. 2017, 814, 178. [Google Scholar] [CrossRef]
- Passante, E.; Frankish, N. Deficiencies in elements involved in TLR4-receptor signalling in RBL-2H3 cells. Inflamm. Res. 2009, 59, 185. [Google Scholar] [CrossRef]
- Falcone, F.H.; Wan, D.; Barwary, N.; Sagi-eisenberg, R. RBL cells as models for in vitro studies of mast cells and basophils. Immunol. Rev. 2018, 282, 47. [Google Scholar] [CrossRef] [PubMed]
- Passante, E. Mast Cells and Basophil Cell Lines: A Compendium. Basophils and Mast Cells: Methods and Protocols, 2nd ed.; Gibbs, B.F., Falcone, F.H., Eds.; Humana Press: New York, NY, USA, 2023; pp. 127–144. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).