Phenology-Informed Strategies for Climate-Resilient Peach Production: Shoot Growth, Leaf Fall, and Flowering of Two Low-Chill Cultivars in Humid Subtropical Central Taiwan
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Plant Materials
2.2. Shoot Growth Measurement
2.3. Shoot Growth and Leaf Fall Curve Fitting
2.4. Leaf Fall and Flowering Observation
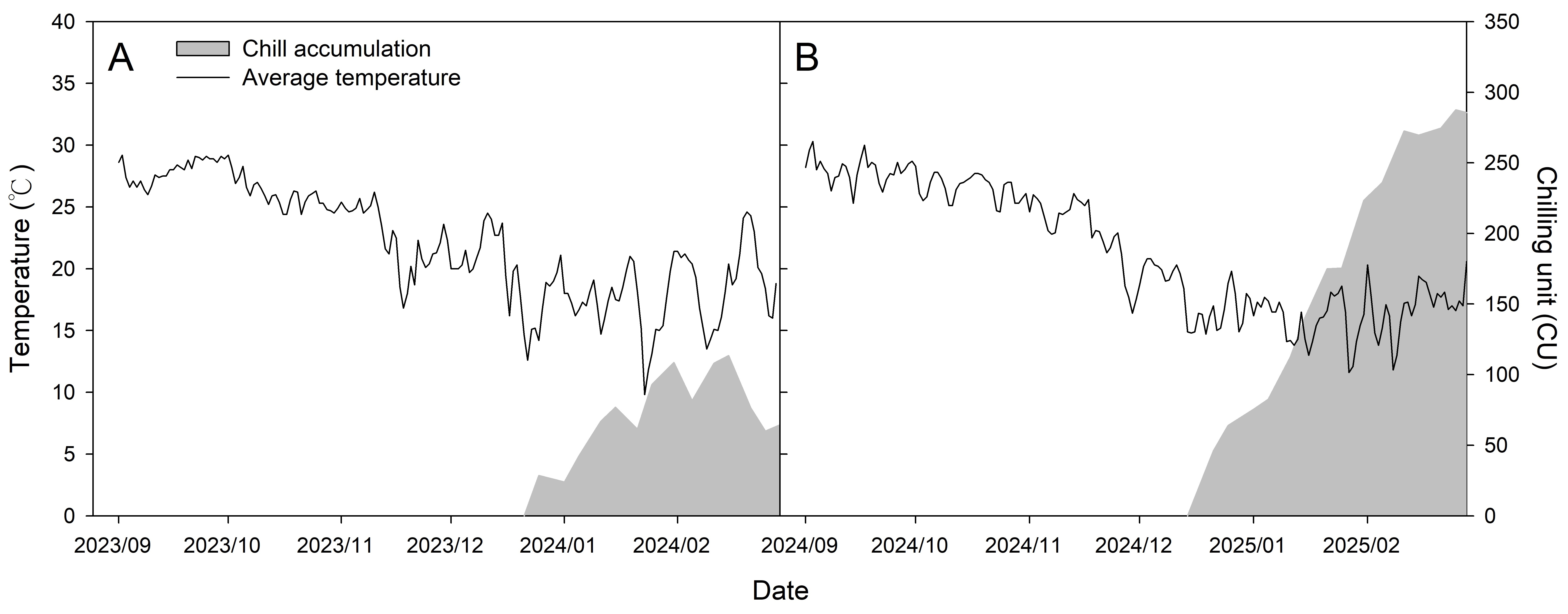
2.5. Calculation of Chilling and Heat Accumulation from the End of Leaf Fall to Budbreak
2.6. Experimental Design and Statistical Analysis
3. Results
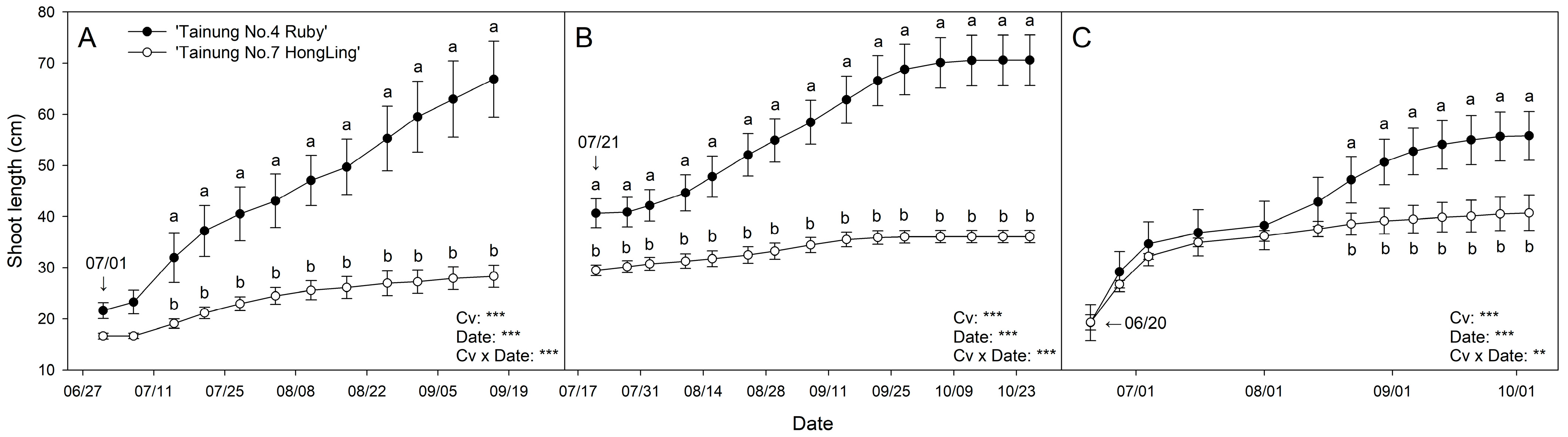
3.1. Shoot Length
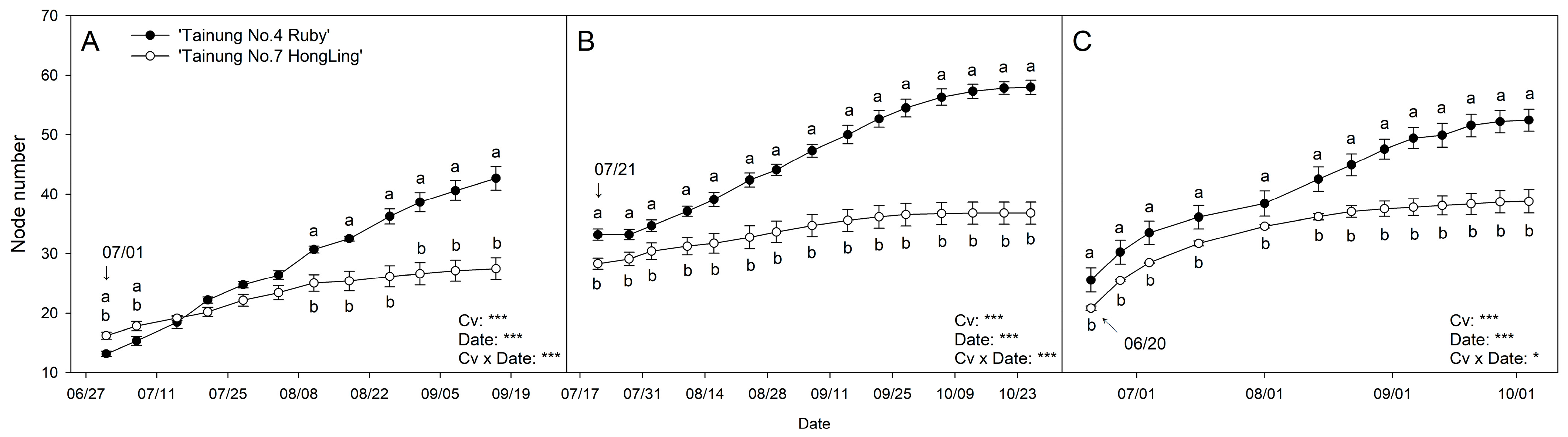
3.2. Node Number
3.3. Nonlinear Model Selection for Shoot Growth
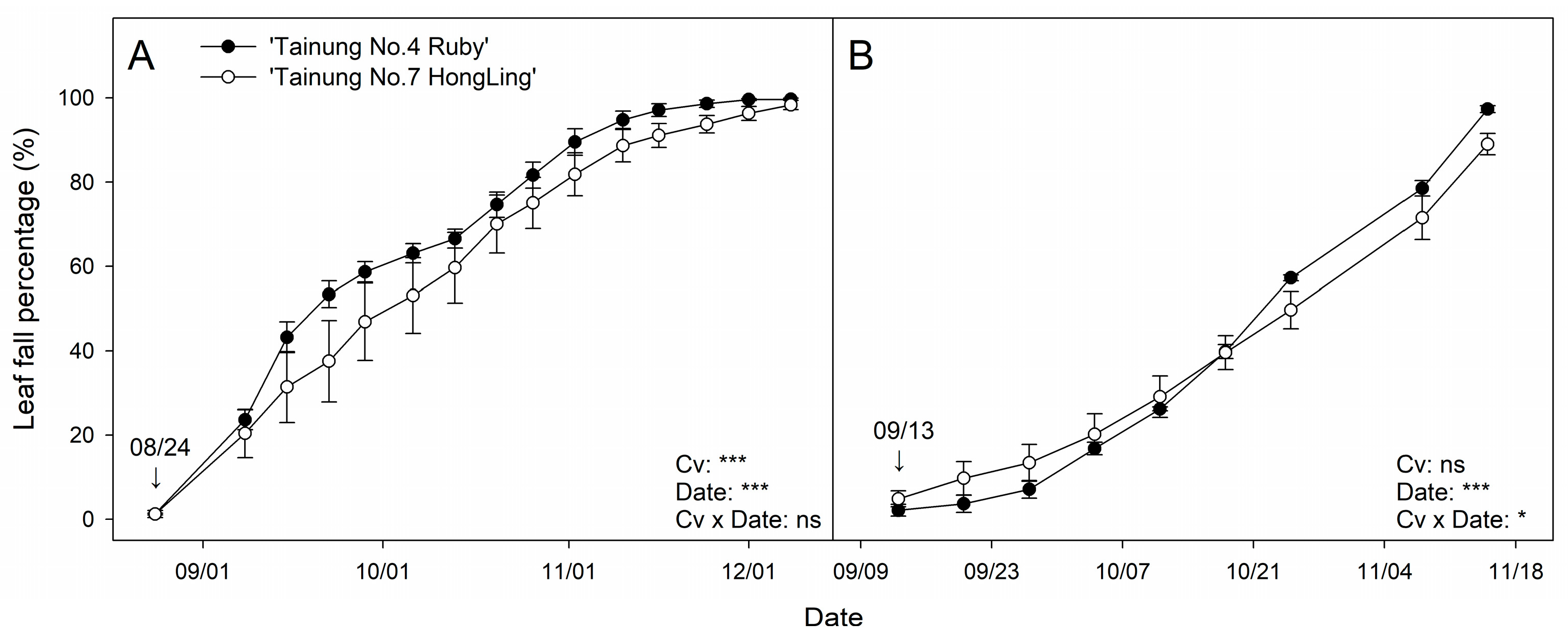
3.4. Leaf Fall Timing and Dynamics
3.5. Flowering Period by Bud Position
3.6. Estimated Chilling Requirements and Heat Accumulation for Apical and Basal Budbreak
4. Discussion
4.1. Low-Chill Cultivars as a Climate-Resilient Strategy
4.2. Impacts of Insufficient Chill on Leaf Fall and Flowering Synchrony
4.3. Adaptation Strategies for Warm-Winter Regions
4.4. Limitations and Future Work
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Campoy, J.A.; Ruiz, D.; Egea, J. Dormancy in temperate fruit trees in a global warming context: A review. Sci. Hortic. 2011, 130, 357–372. [Google Scholar] [CrossRef]
- Salama, A.-M.; Ezzat, A.; El-Ramady, H.; Alam-Eldein, S.M.; Okba, S.K.; Elmenofy, H.M.; Hassan, I.F.; Illés, A.; Holb, I.J. Temperate fruit trees under climate change: Challenges for dormancy and chilling requirements in warm winter regions. Horticulturae 2021, 7, 86. [Google Scholar] [CrossRef]
- Tan, J.C.; Lin, S.-Y. Foliar Urea sprays induce budbreak in peach via rapid non-structural carbohydrate metabolism—A lower-toxicity alternative to hydrogen cyanamide. Sci. Hortic. 2025, 349, 114267. [Google Scholar] [CrossRef]
- Luedeling, E. Climate change impacts on winter chill for temperate fruit and nut production: A review. Sci. Hortic. 2012, 144, 218–229. [Google Scholar] [CrossRef]
- Byrne, D.H. Trends and progress of low chill stone fruit breeding. In Production Technologies for Low-Chill Temperate Fruits, Proceedings of the 2nd International Workshop, Chiang Mai, Thailand, 19–23 April 2004; Australian Centre for International Agricultural Research: Canberra, Australia, 2005; pp. 5–12. [Google Scholar]
- Faust, M.; Erez, A.; Rowland, L.J.; Wang, S.Y.; Norman, H.A. Bud dormancy in perennial fruit trees: Physiological basis for dormancy induction, maintenance, and release. HortScience 1997, 32, 623–629. [Google Scholar] [CrossRef]
- Lang, G.A.; Early, J.D.; Martin, G.C.; Darnell, R.L. Endo-, para-, and ecodormancy: Physiological terminology and classification for dormancy research. HortScience 1987, 22, 371–377. [Google Scholar] [CrossRef]
- Scariotto, S.; Citadin, I.; Raseira, M.D.C.B.; Sachet, M.R.; Penso, G.A. Adaptability and stability of 34 peach genotypes for leafing under Brazilian subtropical conditions. Sci. Hortic. 2013, 155, 111–117. [Google Scholar] [CrossRef][Green Version]
- Clavijo-Herrera, J.; Sarkhosh, A. Subtropical Peach Defoliation and Chill Hours; EDIS; University of Florida: Gainesville, FL, USA, 2025. [Google Scholar] [CrossRef]
- Tang, L.; Chhajed, S.; Vashisth, T.; Olmstead, M.A.; Olmstead, J.W.; Colquhoun, T.A. Transcriptomic study of early responses to the bud dormancy-breaking agent hydrogen cyanamide in ‘TropicBeauty’ peach. J. Am. Soc. Hortic. Sci. 2019, 144, 244–256. [Google Scholar] [CrossRef]
- Wen, I.; Sherman, W.B. Evaluation and breeding of peaches and nectarines for subtropical Taiwan. Acta Hortic. 2002, 592, 191–196. [Google Scholar] [CrossRef]
- Topp, B.L.; Sherman, W.B.; Raseira, M.C.B. Low-chill cultivar development. In The Peach: Botany, Production and Uses; Layne, D.R., Bassi, D., Eds.; CABI: Wallingford, UK, 2008; pp. 106–138. [Google Scholar]
- Sobierajski, G.R.; Harder, I.C.F.; Xavier, D.; Anoni, C.O. Breeding peaches for low-chill in São Paulo State, Brazil. Acta Hortic. 2016, 1127, 35–40. [Google Scholar] [CrossRef]
- Li, Y.; Cao, K.; Zhu, G.; Fang, W.; Chen, C.; Wang, X.; Zhao, P.; Guo, J.; Ding, T.; Guan, L.; et al. Genomic analyses of an extensive collection of wild and cultivated accessions provide new insights into peach breeding history. Genome Biol. 2019, 20, 36. [Google Scholar] [CrossRef]
- Huang, C.-C.; Wen, I.-C.; Lin, S.-Y. Tainung No. 7 HongLing: A Low-chill peach cultivar for early fresh market. HortScience 2024, 59, 304–306. [Google Scholar] [CrossRef]
- Wen, I.-C.; Chang, C.-Y. Breeding of Peach Cultivar “Tainung No. 4 (Ruby)”. J. Taiwan Agric. Res. 2014, 63, 320–323. [Google Scholar]
- Promchot, S.; Boonprakob, U.; Byrne, D.H. Genotype and environment interaction of low-chill peaches and nectarines in subtropical highlands of Thailand. Thai J. Agric. Sci. 2008, 41, 53–61. [Google Scholar]
- Fadón, E.; Herrera, S.; Guerrero, B.; Guerra, M.; Rodrigo, J. Chilling and heat requirements of temperate stone fruit trees (Prunus sp.). Agronomy 2020, 10, 409. [Google Scholar] [CrossRef]
- Agehara, S.; Carrubba, A.; Sarno, M.; Marceddu, R. Phenological assessment of hops (Humulus lupulus L.) grown in semi-arid and subtropical climates through BBCH scale and a thermal-based growth model. Agronomy 2024, 14, 3045. [Google Scholar] [CrossRef]
- Archontoulis, S.V.; Miguez, F.E. Nonlinear regression models and applications in agricultural research. Agron. J. 2015, 107, 786–798. [Google Scholar] [CrossRef]
- Meier, V.U.; Graf, H.; Hack, H.; Heß, M.; Kennel, W.; Klose, R.; Mappes, D.; Seipp, D.; Stauß, R.; Steif, J.; et al. Phänologische entwicklungsstadien des kernobstes (Malus borkh und Pyrus communis L.), des steinobstes (Prunus arten), der johannisbeere (Ribes arten) und der (Fragaria x ananassa Duch.). Nachrichtenbl. Deut. Pflanzenschutzd 1994, 46, 141–153. [Google Scholar]
- Ou, S.-K.; Chen, C.-L. Estimation of the chilling requirement and development of a low-chill model for local peach trees in Taiwan. J. Chin. Soc. Hortic. Sci. 2000, 46, 337–350. [Google Scholar]
- Lu, M.-T.; Song, C.-W.; Ou, S.-K.; Chen, C.-L. A model for estimating chilling requirement of very low-chill peaches in Taiwan. Acta Hortic. 2012, 962, 245–251. [Google Scholar] [CrossRef]
- Marques, L.O.D.; da Silveira Pasa, M.; Sezerino, A.A.; Mello-Farias, P.; Petri, J.L.; Herter, F.G. A Survey of eight chilling models for apple trees in southern Brazil under mild winters. N. Z. J. Crop Hortic. Sci. 2023, 51, 420–431. [Google Scholar] [CrossRef]
- Richardson, E.A.; Seeley, S.D.; Walker, D.R.; Anderson, J.L.; Ashcroft, G.L. Pheno-climatography of spring peach bud development. HortScience 1975, 10, 236–237. [Google Scholar] [CrossRef]
- Parker, L.E.; Abatzoglou, J.T. Warming winters reduce chill accumulation for peach production in the Southeastern United States. Climate 2019, 7, 94. [Google Scholar] [CrossRef]
- Luedeling, E.; Zhang, M.; Girvetz, E.H. Climatic Changes lead to declining winter chill for fruit and nut trees in California during 1950–2099. PLoS ONE 2009, 4, e6166. [Google Scholar] [CrossRef] [PubMed]
- Luedeling, E.; Girvetz, E.H.; Semenov, M.A.; Brown, P.H. Climate change affects winter chill for temperate fruit and nut trees. PLoS ONE 2011, 6, e20155. [Google Scholar] [CrossRef]
- Milech, C.G.; Dini, M.; Franzon, R.C.; Raseira, M.d.C.B. Chilling requirement of four peach cultivars estimated by changes in flower bud weights. Rev. Ceres 2022, 69, 22–30. [Google Scholar] [CrossRef]
- Huang, C.-C.; Wen, I.-C.; Lin, S.-Y. Tainung No.9 HongJin: The first yellow-fleshed, low-chill peach cultivar release from Taiwan. HortScience 2024, 59, 1661–1664. [Google Scholar] [CrossRef]
- Ou, S.-K.; Song, C.-W. ‘Xiami’ peach. HortScience 2006, 41, 1362–1363. [Google Scholar] [CrossRef]
- Ou, S.-K.; Wen, I.-C. ‘SpringHoney’ peach. HortScience 2003, 38, 633–634. [Google Scholar] [CrossRef]
- Malyshev, A.V.; Beil, I.; Zohner, C.M.; Garrigues, R.; Campioli, M. The clockwork of spring: Bud dormancy timing as a driver of spring leaf-out in temperate deciduous trees. Agric. For. Meteorol. 2024, 349, 109957. [Google Scholar] [CrossRef]
- Jacobs, G.; Watermeyer, P.J.; Styrdom, D.K. Aspects of winter rest in apple trees. Crop Prod. 1981, 10, 103–104. [Google Scholar]
- Reig, C.; González-Rossia, D.; Juan, M.; Agustí, M. Effects of fruit load on flower bud initiation and development in peach. J. Hortic. Sci. Biotechnol. 2006, 81, 1079–1085. [Google Scholar] [CrossRef]
- Faust, M.; Liu, D.; Wang, S.Y.; Stutte, G.W. Involvement of apical dominance in winter dormancy of apple buds. Acta Hortic. 1995, 395, 47–56. [Google Scholar] [CrossRef]
- Citadin, I.; Pertille, R.H.; Loss, E.M.S.; Oldoni, T.L.C.; Danner, M.A.; Júnior, A.W.; Lauri, P.-É. Do low chill peach cultivars in mild winter regions undergo endodormancy? Trees 2022, 36, 1273–1284. [Google Scholar] [CrossRef]
- Luedeling, E.; Guo, L.; Dai, J.; Leslie, C.; Blanke, M.M. Differential responses of trees to temperature variation during the chilling and forcing phases. Agric. For. Meteorol. 2013, 181, 33–42. [Google Scholar] [CrossRef]
- Fernandez, E.; Krefting, P.; Kunz, A.; Do, H.; Fadón, E.; Luedeling, E. Boosting statistical delineation of chill and heat periods in temperate fruit trees through multi-environment observations. Agric. For. Meteorol. 2021, 310, 108652. [Google Scholar] [CrossRef]
- Heide, O.M.; Prestrud, A.K. Low temperature, but not photoperiod, controls growth cessation and dormancy induction and release in apple and pear. Tree Physiol. 2005, 25, 109–114. [Google Scholar] [CrossRef]
- Roussos, P.A. Climate change challenges in temperate and sub-tropical fruit tree cultivation. Encyclopedia 2024, 4, 558–582. [Google Scholar] [CrossRef]
- Boutiti, K.; Bellil, I.; Khelifi, D. A Study of chill and heat requirements of resilient apricot (Prunus armeniaca) varieties in response to climate change. Int. J. Hortic. Sci. Technol. 2025, 12, 167–178. [Google Scholar]
- Cifuentes-Carvajal, A.; Chaves-Córdoba, B.; Vinson, E.; Coneva, E.D.; Chavez, D.; Salazar-Gutiérrez, M.R. Modeling the budbreak in peaches: A basic approach using chill and heat accumulation. Agronomy 2023, 13, 2422. [Google Scholar] [CrossRef]
- Ionescu, I.A.; Møller, B.L.; Sánchez-Pérez, R. Chemical control of flowering time. J. Exp. Bot. 2016, 68, erw427. [Google Scholar] [CrossRef]
- Erez, A. Means to compensate for insufficient chilling to improve bloom and leafing. Acta Hortic. 1995, 395, 81–96. [Google Scholar] [CrossRef]
- Ghrab, M.; BenMimoun, M.; Masmoudi, M.M.; BenMechlia, N. Chilling trends in a warm production area and their impact on flowering and fruiting of peach trees. Sci. Hortic. 2014, 178, 87–94. [Google Scholar] [CrossRef]
- Mura, C.; Charrier, G.; Kovaleski, A.P.; Raymond, P.; Deslauriers, A.; Rossi, S. Is it cold enough? Effects of artificial and natural chilling on budbreak and frost hardiness in Acer Saccharum (Marsh.). Physiol. Plant. 2025, 177, e70586. [Google Scholar] [CrossRef]
| Season | Nonlinear Model i | r2 | a | b | c | d |
|---|---|---|---|---|---|---|
| 2022 | Gompertz-3P | 0.992 | 93.22 ± 20.70 ii | 0.02 ± 0.01 | 201.72 ± 12.52 | N/A iii |
| 2023 | Logistic-4P | 0.998 | 71.83 ± 1.02 | 38.48 ± 1.65 | 0.07 ± 0.01 | 242.51 ± 1.95 |
| 2024 | Gompertz-3P | 0.957 | 64.91 ± 14.21 | 0.02 ± 0.01 | 171.75 ± 10.13 | N/A |
| Season | Nonlinear Model i | r2 | a | b | c | d |
|---|---|---|---|---|---|---|
| 2022 | Gompertz-4P | 0.996 | 28.26 ± 0.56 ii | 15.87 ± 1.05 | 0.06 ± 0.01 | 200.81 ± 2.88 |
| 2023 | Logistic-4P | 0.991 | 36.29 ± 0.32 | 29.53 ± 0.67 | 0.08 ± 0.02 | 237.49 ± 3.54 |
| 2024 | Gompertz-3P | 0.974 | 39.50 ± 0.85 | 0.07 ± 0.02 | 166.28 ± 3.10 | N/A iii |
| 2023 | 2024 | |||||
|---|---|---|---|---|---|---|
| Cultivar | Onset of Leaf Fall i | End of Leaf Fall ii | Leaf Fall Period | Onset of Leaf Fall | End of Leaf Fall | Leaf Fall Period |
| ‘Tainung No. 4 Ruby’ | Sep 8 | Nov 10 | 63 | Oct 11 | Nov 15 | 35 |
| ‘Tainung No. 7 HongLing’ | Sep 15 | Nov 16 | 62 | Oct 4 | Nov 15 | 42 |
| Season | Nonlinear Model i | r2 | a | b | c | d |
|---|---|---|---|---|---|---|
| 2023 | Logistic-4P | 0.989 | 1.16 ± 0.27 ii | −5.04 ± 34.66 | 0.02 ± 0.02 | 168.45 ± 376.16 |
| 2024 | Gompertz-3P | 0.996 | 1.54 ± 0.42 | 0.04 ± 0.01 | 300.01 ± 8.48 | N/A iii |
| Season | Nonlinear Model i | r2 | a | b | c | d |
|---|---|---|---|---|---|---|
| 2023 | Logistic-4P | 0.998 | 1.10 ± 0.08 ii | −0.54 ± 0.40 | 0.03 ± 0.01 | 257.35 ± 13.75 |
| 2024 | Gompertz-3P | 0.998 | 2.67 ± 1.26 | 0.02 ± 0.01 | 325.47 ± 22.27 | N/A iii |
| Cultivar | Bud Position | 2023–2024 Season | 2024–2025 Season | ||||
|---|---|---|---|---|---|---|---|
| Onset of Flower Budbreak i | End of Flowering ii | Flowering Period | Onset of Flower Budbreak | End of Flowering | Flowering Period | ||
| ‘Tainung No. 4 Ruby’ | Apical | Feb 16 | Mar 8 | 21 | Feb 14 | Mar 12 | 26 |
| Basal | Feb 23 | Mar 15 | 21 | Feb 21 | Mar 6 | 13 | |
| Apical-basal lag (days) | 7 | 7 | N/A iii | 7 | −6 | N/A | |
| ‘Tainung No. 7 HongLing’ | Apical | Jan 15 | Mar 1 | 46 | Jan 10 | Feb 21 | 42 |
| Basal | Feb 6 | Mar 1 | 24 | Jan 24 | Feb 21 | 28 | |
| Apical-basal lag (days) | 22 | 0 | N/A | 14 | 0 | N/A | |
| Cultivar | Bud Position | 2023–2024 Season | 2024–2025 Season | ||
|---|---|---|---|---|---|
| Chilling Unit i | Growing Degree Hours ii | Chilling Unit | Growing Degree Hours | ||
| ‘Tainung No. 4 Ruby’ | Apical | 113.5 | 7133.0 | 273.5 | 9663.0 |
| Basal | 113.5 | 10,021.3 | 274.6 | 12,046.9 | |
| ‘Tainung No. 7 HongLing’ | Apical | 78.0 | 649.5 | 101.0 | 1166.1 |
| Basal | 81.5 | 7784.2 | 176.5 | 4723.5 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Huang, C.-C.; Lin, S.-Y. Phenology-Informed Strategies for Climate-Resilient Peach Production: Shoot Growth, Leaf Fall, and Flowering of Two Low-Chill Cultivars in Humid Subtropical Central Taiwan. Agronomy 2025, 15, 2748. https://doi.org/10.3390/agronomy15122748
Lee H, Huang C-C, Lin S-Y. Phenology-Informed Strategies for Climate-Resilient Peach Production: Shoot Growth, Leaf Fall, and Flowering of Two Low-Chill Cultivars in Humid Subtropical Central Taiwan. Agronomy. 2025; 15(12):2748. https://doi.org/10.3390/agronomy15122748
Chicago/Turabian StyleLee, Hsuan, Chun-Che Huang, and Syuan-You Lin. 2025. "Phenology-Informed Strategies for Climate-Resilient Peach Production: Shoot Growth, Leaf Fall, and Flowering of Two Low-Chill Cultivars in Humid Subtropical Central Taiwan" Agronomy 15, no. 12: 2748. https://doi.org/10.3390/agronomy15122748
APA StyleLee, H., Huang, C.-C., & Lin, S.-Y. (2025). Phenology-Informed Strategies for Climate-Resilient Peach Production: Shoot Growth, Leaf Fall, and Flowering of Two Low-Chill Cultivars in Humid Subtropical Central Taiwan. Agronomy, 15(12), 2748. https://doi.org/10.3390/agronomy15122748







