Growth and Magnetocaloric Properties of Co(NH4)2(SO4)2·6H2O Crystal
Abstract
1. Introduction
2. Experiment
2.1. Crystal Growth
2.2. Characterization
3. Results and Discussion
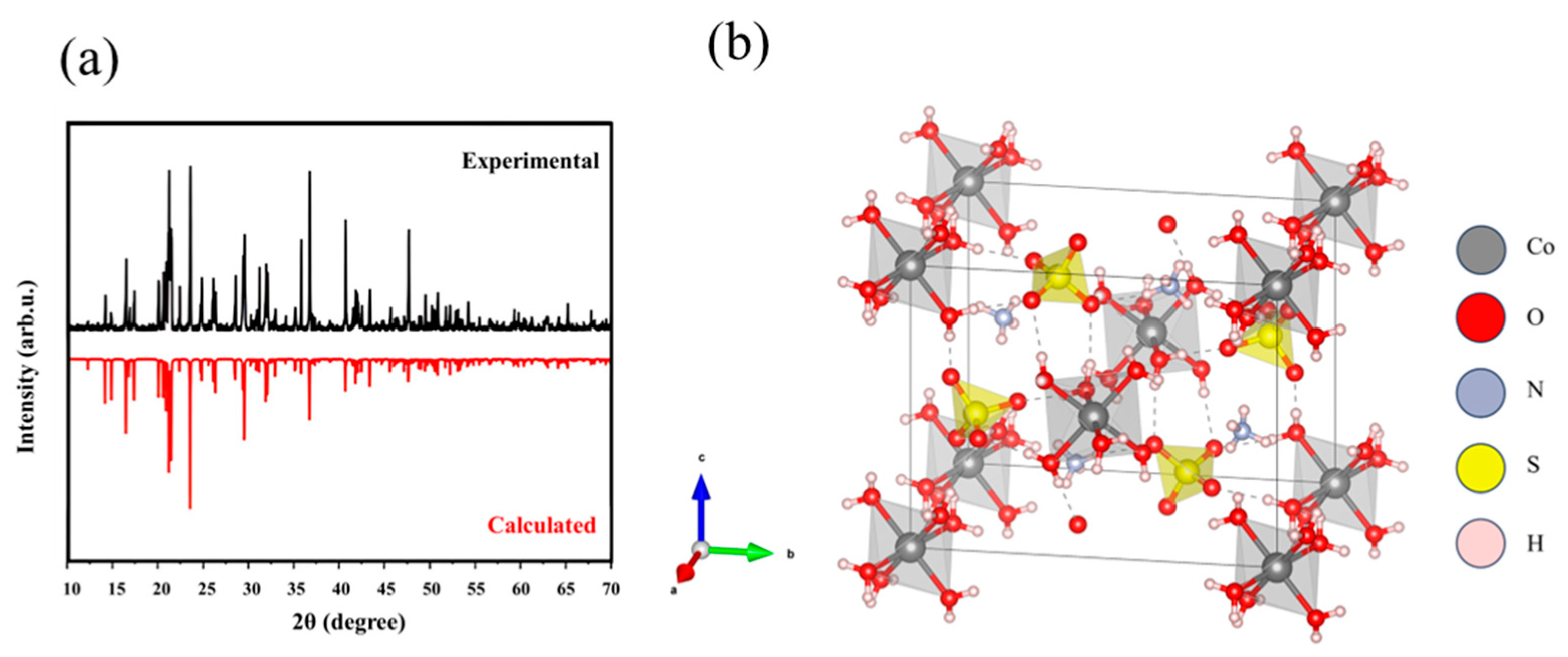
3.1. Crystal Structure
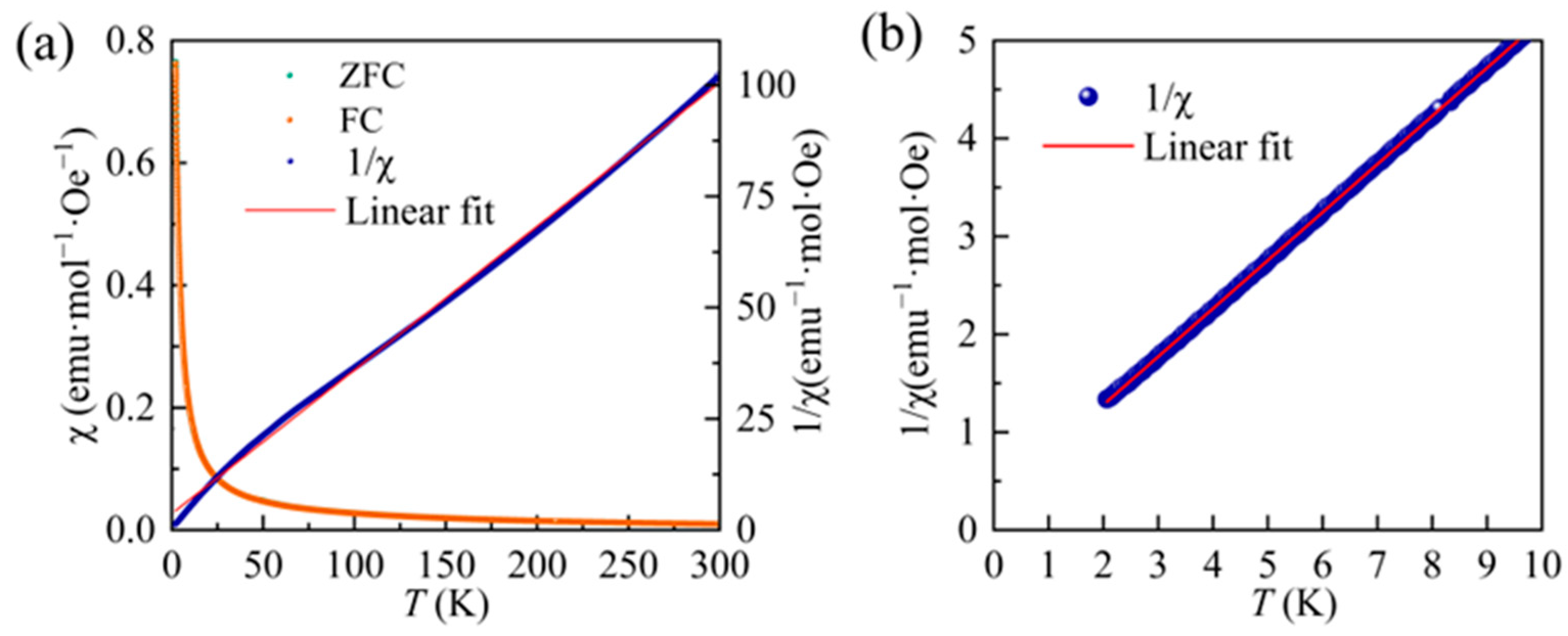
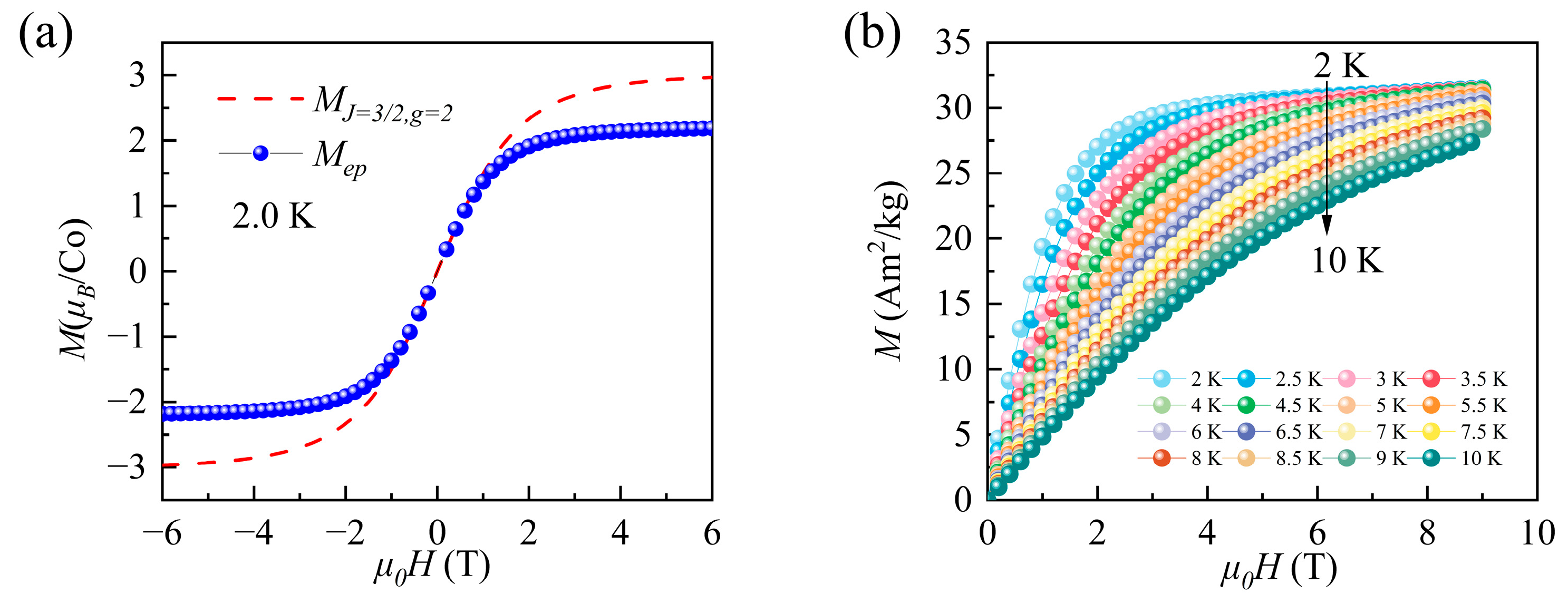
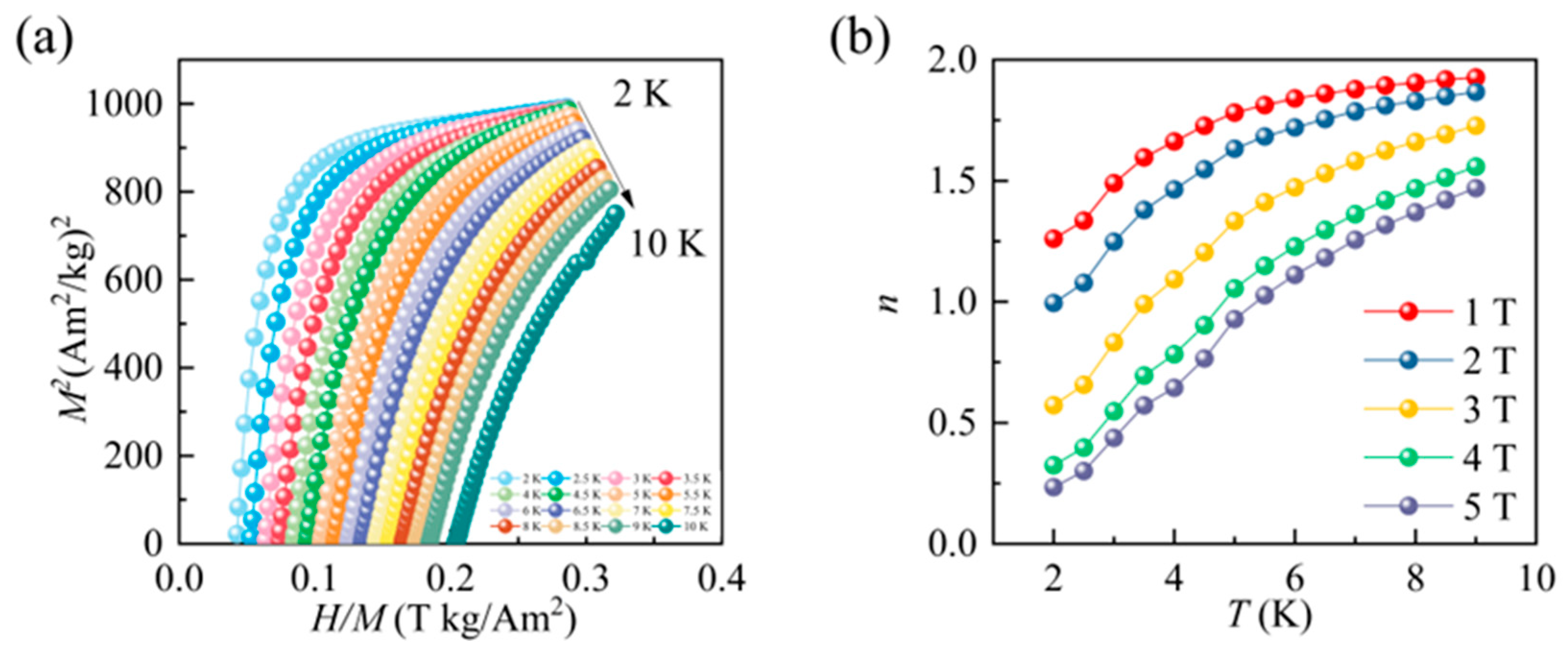
3.2. Magnetic Measurements
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Shen, J.; Mo, Z.; Li, Z.; Gao, X.; Sun, H.; Xie, H.; Liu, R. Development of magnetic refrigeration materials and technology. Sci. Sin. Phys. Mech. Astron. 2021, 51, 067502. [Google Scholar] [CrossRef]
- Zheng, X.; Shen, J.; Hu, F.; Sun, J.; Shen, B. Research progress in magnetocaloric effect materials. Acta Phys. Sin. 2016, 65, 217502. [Google Scholar] [CrossRef]
- Hebb, M.H.; Purcell, E.M. A Theoretical Study of Magnetic Cooling Experiments. J. Chem. Phys. 1937, 5, 338–350. [Google Scholar] [CrossRef]
- Sessoli, R. Chilling with Magnetic Molecules. Angew. Chem. Int. Ed. 2012, 51, 43–45. [Google Scholar] [CrossRef]
- Wang, Q.; Wu, Q.; Cheng, H.; Li, X.; Yu, N.; Pan, M.; Yu, Y.; Fang, J.; Hu, X.; Ge, H.; et al. Review of the research on oxides in low-temperature magnetic refrigeration. J. Eur. Ceram. Soc. 2023, 43, 6665–6680. [Google Scholar] [CrossRef]
- Wang, Y.; Pan, H.; Liu, S. Research Progress of High Efficiency Magnetic Refrigeration Technology and Magnetic Materials. J. Supercond. Nov. Magn. 2025, 38, 86. [Google Scholar] [CrossRef]
- Zhang, H.; Gimaev, R.; Kovalev, B.; Kamilov, K.; Zverev, V.; Tishin, A. Review on the materials and devices for magnetic refrigeration in the temperature range of nitrogen and hydrogen liquefaction. Phys. B Condens. Matter 2019, 558, 65–73. [Google Scholar] [CrossRef]
- Numazawa, T.; Kamiya, K.; Shirron, P.; DiPirro, M.; Matsumoto, K. Magnetocaloric Effect of Polycrystal GdLiF4 for Adiabatic Magnetic Refrigeration. AIP Conf. Proc. 2006, 850, 1579–1580. [Google Scholar]
- Daniels, J.M.; Kurti, N.; Simon, F.E. The thermal and magnetic properties of chromium potassium alum below 0·1 °K. Proc. R. Soc. Lond. A 1954, 221, 243–256. [Google Scholar]
- Vilches, O.E.; Wheatley, J.C. Measurements of the Specific Heats of Three Magnetic Salts at Low Temperatures. Phys. Rev. 1966, 148, 509–516. [Google Scholar] [CrossRef]
- Fritz, J.J.; Clark, J.T.; Cesarano, J. Magnetic and thermodynamic properties of Mn(NH4)2(SO4)2⋅6H2O below 1 °K and at fields up to 24,000 G parallel to the b axis. J. Chem. Phys. 1975, 62, 200–207. [Google Scholar] [CrossRef]
- Tsuru, K.; Uryû, N. Spin Reorientation in Manganese Tutton Salt Mn(NH4)2(SO4)2·6H2O. J. Phys. Soc. Jpn. 1976, 40, 418–426. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, C.; Zhang, Z.; Lu, H.; Wu, L.; Zhang, G.; Tu, H.; Li, Z.; Shen, J.; Wang, D. Large cryogenic magnetocaloric effect in transition metal-based double dinitrates. Appl. Phys. Lett. 2024, 125, 262403. [Google Scholar] [CrossRef]
- Liu, X.H.; Liu, W.; Guo, S.; Gong, W.J.; Lv, X.K.; Zhang, Z.D. Field-induced reversible magnetocaloric effect in CoCl2. J. Alloys Compd. 2010, 507, 26–28. [Google Scholar]
- Giauque, W.F.; Hornung, E.W.; Fisher, R.A.; Brodale, G.E. The Measurement of Magnetic Induction in High Magnetic Fields at Low Temperatures. CoSO4·7H2O. J. Chem. Phys. 1962, 37, 2952–2962. [Google Scholar] [CrossRef]
- de Oliveira Neto, J.G.; Viana, J.R.; Abreu, K.R.; da Silva, L.F.L.; Lage, M.R.; Stoyanov, S.R.; de Sousa, F.F.; Lang, R.; Dos Santos, A.O. Tutton salt (NH4)2Zn(SO4)2(H2O)6: Thermostructural, spectroscopic, Hirshfeld surface, and DFT investigations. J. Mol. Model. 2024, 30, 339. [Google Scholar] [CrossRef]
- Fender, B.E.F.; Figgis, B.N.; Forsyth, J.B.; Mason, R. Spin density and bonding in Mn(H2O)2+6 in ammonium manganese Tutton salt. Proc. R. Soc. Lond. A. 1986, 404, 127–138. [Google Scholar]
- Montgomery, H.; Morosin, B.; Natt, J.J.; Witkowska, A.M.; Lingafelter, E.C. The Crystal Structure of Tutton’s Salts. VI. Vanadium (II), Iron (II) and Cobalt (II) Ammonium Sulfate Hexahydrates. Acta Crystallogr. 1967, 22, 775–780. [Google Scholar] [CrossRef]
- Yilmaz, V.T.; Hacaloglu, J. Thermal Behaviour and Kinetic Analysis of the Thermogravi-metric Data of Double Ammonium Sulphate Hexahydrate Salts of Mn (II), Fe (II), Co (II), Ni (II) and Cu (II). Thermochim. Acta 1994, 236, 179–190. [Google Scholar]
- Manomenova, V.L.; Nikolaeva, N.V.; Shlyapnikova, E.A.; Buchneva, O.N.; Kovaleva, I.Y.; Kruchinin, S.E. The Ammonium Cobalt Sulfate Hexahydrate (ACSH) Crystal Growth from Aqueous Solutions and Some Properties of Solutions and Crystals. J. Cryst. Growth 2020, 533, 125463. [Google Scholar] [CrossRef]
- Oey, Y.M.; Cava, R.J. The Effective Magnetic Moments of Co2+ and Co3+ in SrTiO3 Investigated by Temperature-Dependent Magnetic Susceptibility. Mater. Res. Bull. 2020, 122, 110667. [Google Scholar] [CrossRef]
- Malaker, S.F. Investigation of Nuclear Effects in Paramagnetic Single Crystals at Very Low Temperatures. Phys. Rev. 1951, 84, 133–141. [Google Scholar] [CrossRef]
- Fisher, R.A.; Hornung, E.W.; Brodale, G.E.; Giauque, W.F. Magnetothermodynamics of Ce2Mg3(NO3)12·24H2O. II. The evaluation of absolute temperature and other thermodynamic properties of CMN to 0.6 m °K. J. Chem. Phys. 1973, 58, 5584–5604. [Google Scholar] [CrossRef]
- Bebenin, N.G.; Zainullina, R.I.; Ustinov, V.V.; Mukovskii, Y.M. Magnetic properties of La0.7−xPrxCa0.3MnO3 single crystals: When is Banerjee criterion applicable? J. Magn. Magn. Mater. 2014, 354, 76–80. [Google Scholar] [CrossRef]
- Law, J.Y.; Franco, V.; Moreno-Ramírez, L.M.; Conde, A.; Karpenkov, D.Y.; Radulov, I.; Skokov, K.P.; Gutfleisch, O. A quantitative criterion for determining the order of magnetic phase transitions using the magnetocaloric effect. Nat. Commun. 2018, 9, 2680. [Google Scholar] [CrossRef] [PubMed]
- Gutfleisch, O.; Gottschall, T.; Fries, M.; Benke, D.; Radulov, I.; Skokov, K.P.; Wende, H.; Gruner, M.; Acet, M.; Entel, P.; et al. Mastering hysteresis in magnetocaloric materials. Phil. Trans. R. Soc. A. 2015, 374, 20150308. [Google Scholar] [CrossRef] [PubMed]


| Empirical Formula | CoH20N2O14S2 |
|---|---|
| Formula weight (g mol−1) | 395.21 |
| Temperature (K) | 296.15 |
| Crystal system | Monoclinic |
| Space group | P21/c |
| a (Å) | 6.2437 (49) |
| b (Å) | 12.5259 (44) |
| c (Å) | 9.2560 (95) |
| α (°) | 90 |
| β (°) | 73 |
| γ (°) | 90 |
| Volume (Å3) | 692.1 (73) |
| Z | 2 |
| (g cm−3) | 1.876 |
| Radiation | Cu Kα (λ = 1.5418 Å) |
| 2θ range for data collection (°) | 10–70 |
| Final R indexes (all data) | R1 = 0.0288, wR2 = 0.0369 |
| Materials | Tt (K) | μ0H | Ref. | |
|---|---|---|---|---|
| CoCl2 | 27.0 | 7 | 11.5 | [14] |
| LCoN | 4.0 | 7 | 9.45 | [13] |
| CMN | 1.8 | — | 7.5 | [23] |
| CoSO4·7H2O | 1.2 | 10 | 11.5 | [16] |
| MAS | 0.2 | — | 38.1 | [12] |
| Co(NH4)2(SO4)2·6H2O | 2.0 | 5 | 13.9 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Wan, T.; Chen, Y.; Chen, Z.; Li, Z.; Zhao, Y.; Shen, J.; Zhang, G.; Tu, H. Growth and Magnetocaloric Properties of Co(NH4)2(SO4)2·6H2O Crystal. Crystals 2025, 15, 1022. https://doi.org/10.3390/cryst15121022
Wang Y, Wan T, Chen Y, Chen Z, Li Z, Zhao Y, Shen J, Zhang G, Tu H. Growth and Magnetocaloric Properties of Co(NH4)2(SO4)2·6H2O Crystal. Crystals. 2025; 15(12):1022. https://doi.org/10.3390/cryst15121022
Chicago/Turabian StyleWang, Yunhui, Tingwei Wan, Yuwei Chen, Zuhua Chen, Zhenxing Li, Yanan Zhao, Jun Shen, Guochun Zhang, and Heng Tu. 2025. "Growth and Magnetocaloric Properties of Co(NH4)2(SO4)2·6H2O Crystal" Crystals 15, no. 12: 1022. https://doi.org/10.3390/cryst15121022
APA StyleWang, Y., Wan, T., Chen, Y., Chen, Z., Li, Z., Zhao, Y., Shen, J., Zhang, G., & Tu, H. (2025). Growth and Magnetocaloric Properties of Co(NH4)2(SO4)2·6H2O Crystal. Crystals, 15(12), 1022. https://doi.org/10.3390/cryst15121022





