Ferroelectric Properties and Ambipolar Carrier Transport of 9-Fluorenone-Based Liquid Crystals
Abstract
1. Introduction
2. Materials and Methods
2.1. General Procedures and Materials
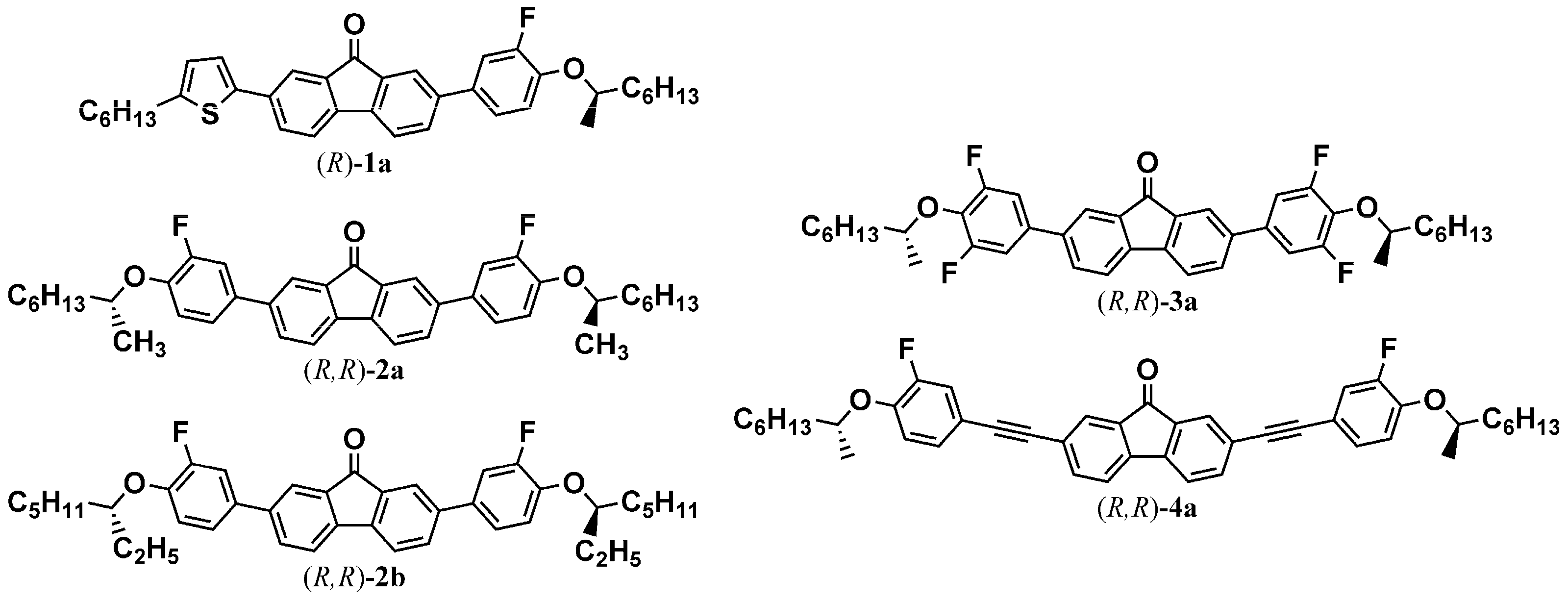
2.2. Synthesis and Characterization of Target Compounds
2.2.1. Characterization of (R)-1a
2.2.2. Characterization of (R,R)-2a
2.2.3. Characterization of (R,R)-2b
2.2.4. Characterization of (R,R)-3a
2.2.5. Characterization of (R,R)-4a
2.3. Characterization of Light-Absorption Properties
2.4. Characterization of LC Properties
2.5. Evaluation of Ferroelectric Properties
2.6. Evaluation of Carrier Transport Characteristics
2.7. Evaluation of Current–Voltage Characteristics
3. Results and Discussion
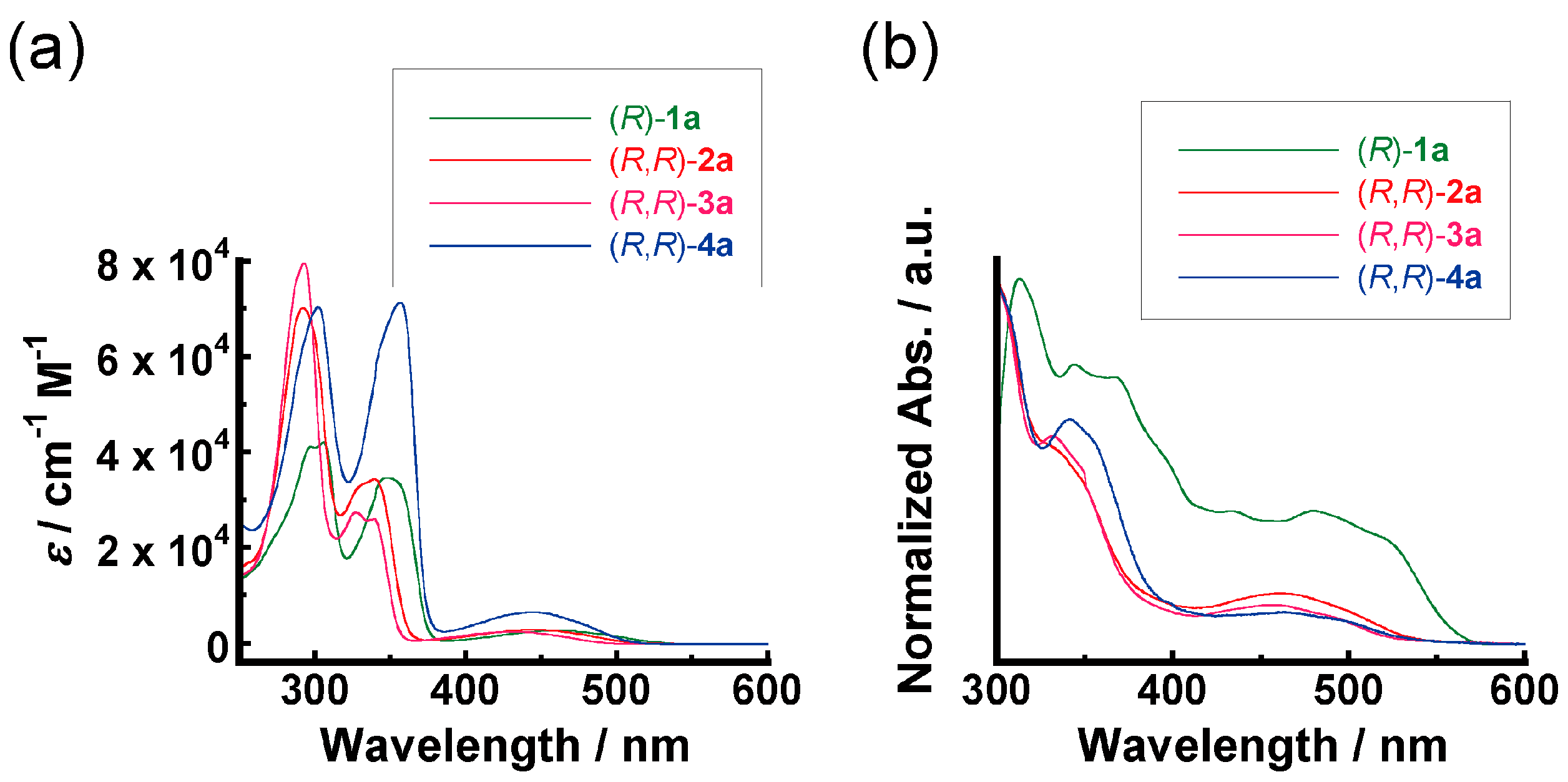
3.1. Light-Absorption Properties
3.2. Liquid-Crystalline Properties
3.2.1. Polarizing Optical Microscopy
3.2.2. Differential Scanning Calorimetry
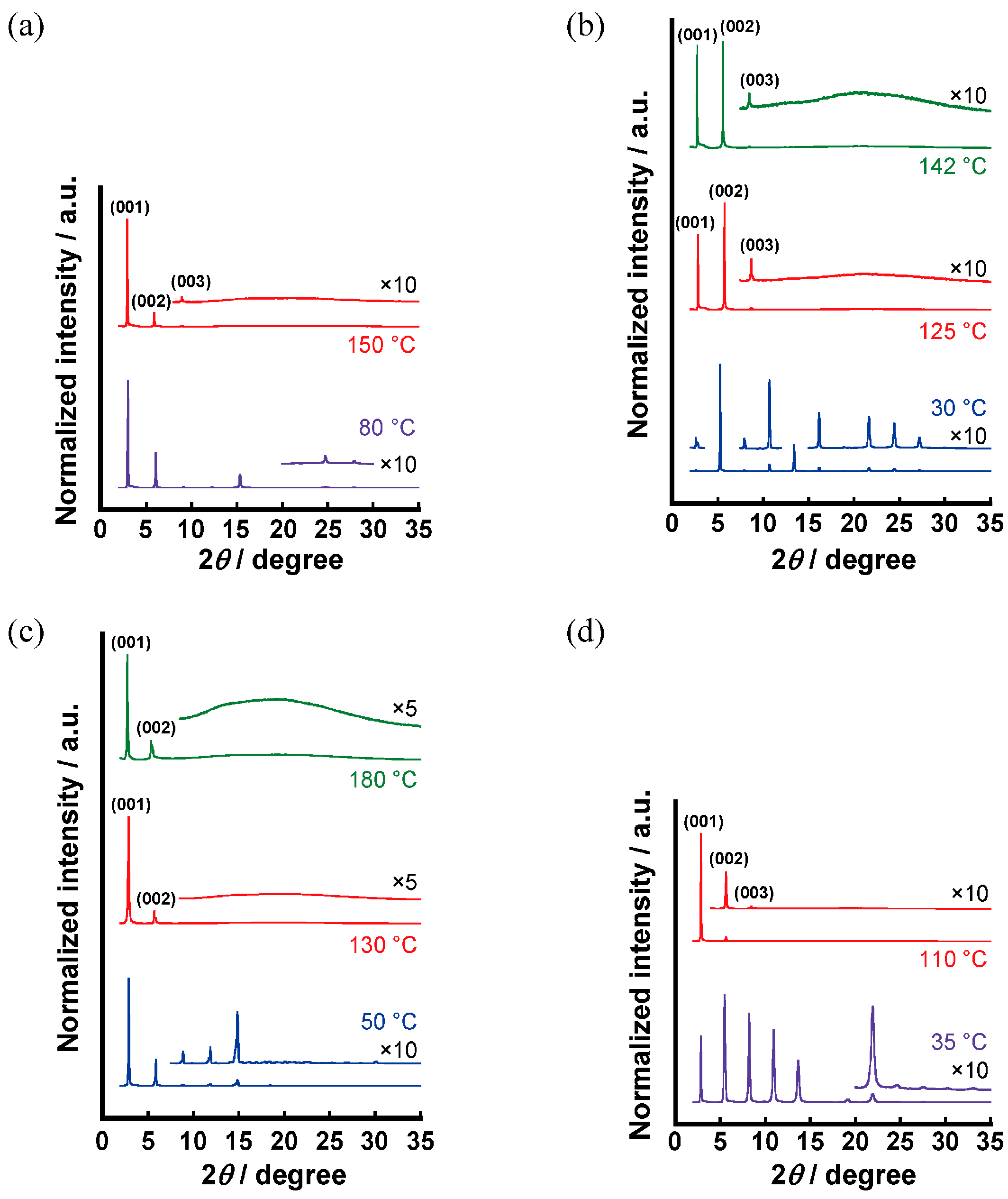
3.2.3. X-Ray Diffraction
3.3. Ferroelectric Properties
3.4. Carrier Transport Characteristics
3.5. Current–Voltage Characteristics
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goodby, J.W. Symmetry and Chirality in Liquid Crystals. In Handbook of Liquid Crystals, 1st ed.; Demus, D., Goodby, J.W., Gray, G.W., Spiess, H.-W., Vill, V., Eds.; Wiley-VCH: Weinheim, Germany, 1998; Volume 1, pp. 115–132. [Google Scholar]
- Kitzerow, H.-S.; Bahr, C. (Eds.) Chirality in Liquid Crystals, 1st ed.; Springer: New York, NY, USA, 2001. [Google Scholar]
- Dierking, I. Chiral Liquid Crystals: Structures, Phases, Effects. Symmetry 2014, 6, 444–472. [Google Scholar] [CrossRef]
- Zhang, L.; Qin, L.; Wang, X.; Cao, H.; Liu, M. Supramolecular Chirality in Self-Assembled Soft Materials: Regulation of Chiral Nanostructures and Chiral Functions. Adv. Mater. 2014, 26, 6959–6964. [Google Scholar] [CrossRef]
- Yashima, E.; Ousaka, N.; Taura, D.; Shimomura, K.; Ikai, T.; Maeda, K. Supramolecular Helical Systems: Helical Assemblies of Small Molecules, Foldamers, and Polymers with Chiral Amplification and Their Functions. Chem. Rev. 2016, 116, 13752–13990. [Google Scholar] [CrossRef] [PubMed]
- Evers, F.; Aharony, A.; Bar-Gill, N.; Entin-Wohlman, O.; Hedegård, P.; Hod, O.; Jelinek, P.; Kamieniarz, G.; Lemeshko, M.; Michaeli, K.; et al. Theory of Chirality Induced Spin Selectivity: Progress and Challenges. Adv. Mater. 2022, 34, 2106629. [Google Scholar] [CrossRef]
- He, S.; Jiang, Z.; Dou, X.; Gao, L.; Feng, C. Chiral Supramolecular Assemblies: Controllable Construction and Biological Activity. ChemPlusChem 2023, 88, e202300226. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, L.; Wei, Z. Chiral Conjugated Molecular Assemblies Interact with Substances and Light. Acc. Mater. Res. 2024, 5, 329–346. [Google Scholar] [CrossRef]
- Fernando, P.S.; Chen, Y.-C.; Baek, J.M.; Diao, Y. Chiral Assemblies of π-Conjugated Molecules: Fundamentals, Processing Strategies, and Applications in (Opto)Electronics. Annu. Rev. Chem. Biomol. Eng. 2025, 16, 59–91. [Google Scholar] [CrossRef] [PubMed]
- Meyer, R.B.; Libert, L.; Strzelecki, L.; Keller, P. Ferroelectric liquid crystals. J. Phys. 1975, 36, L69–L71. [Google Scholar] [CrossRef]
- Lagerwall, S.T.; Dahl, I. Ferroelectric Liquid Crystals. Mol. Cryst. Liq. Cryst. 1984, 114, 151–187. [Google Scholar] [CrossRef]
- Takanishi, Y.; Takezoe, H.; Suzuki, Y.; Kobayashi, I.; Yajima, T.; Terada, M.; Mikami, K. Spontaneous Enantiomeric Resolution in a Fluid Smectic Phase of a Racemate. Angew. Chem. Int. Ed. 1999, 38, 2353–2356. [Google Scholar] [CrossRef]
- Young, C.Y.; Pindak, R.; Clark, N.A.; Meyer, R.B. Light-Scattering Study of Two-Dimensional Molecular-Orientation Fluctuations in a Freely Suspended Ferroelectric Liquid-Crystal Film. Phys. Rev. Lett. 1978, 40, 773–776. [Google Scholar] [CrossRef]
- Clark, N.A.; Lagerwall, S.T. Submicrosecond bistable electro-optic switching in liquid crystals. Appl. Phys. Lett. 1980, 36, 899–901. [Google Scholar] [CrossRef]
- Lago-Silva, M.; Fernández-Míguez, M.; Rodríguez, R.; Quiñoá, E.; Freire, F. Stimuli-responsive synthetic helical polymers. Chem. Soc. Rev. 2024, 53, 793–852. [Google Scholar] [CrossRef]
- Zhang, M.; Kim, M.; Choi, W.; Choi, J.; Kim, D.H.; Liu, Y.; Lin, Z. Chiral macromolecules and supramolecular assemblies: Synthesis, properties and applications. Prog. Polym. 2024, 151, 101800. [Google Scholar] [CrossRef]
- García, F.; Gómez, R.; Sánchez, L. Chiral supramolecular polymers. Chem. Soc. Rev. 2023, 52, 7524–7548. [Google Scholar] [CrossRef] [PubMed]
- Seki, A.; Funahashi, M. Photovoltaic Effects in Ferroelectric Liquid Crystals based on Phenylterthiophene Derivatives. Chem. Lett. 2016, 45, 616–618. [Google Scholar] [CrossRef]
- Seki, A.; Funatsu, Y.; Funahashi, M. Anomalous photovoltaic effect based on molecular chirality: Influence of enantiomeric purity on the photocurrent response in π-conjugated ferroelectric liquid crystals. Phys. Chem. Chem. Phys. 2017, 19, 16446–16455. [Google Scholar] [CrossRef]
- Seki, A.; Funahashi, M. Chiral photovoltaic effect in an ordered smectic phase of a phenylterthiophene derivative. Org. Electron. 2018, 62, 311–319. [Google Scholar] [CrossRef]
- Seki, A.; Yoshio, M.; Mori, Y.; Funahashi, M. Ferroelectric Liquid-Crystalline Binary Mixtures Based on Achiral and Chiral Trifluoromethylphenylterthiophenes. ACS Appl. Mater. Interfaces 2020, 12, 53029–53038. [Google Scholar] [CrossRef]
- Seki, A.; Shimizu, K.; Aoki, K. Chiral π-Conjugated Liquid Crystals: Impacts of Ethynyl Linker and Bilateral Symmetry on the Molecular Packing and Functions. Crystals 2022, 12, 1278. [Google Scholar] [CrossRef]
- Seki, A.; Funahashi, M.; Aoki, K. Ferroelectric Photovoltaic Effect in the Ordered Smectic Phases of Chiral π-Conjugated Liquid Crystals: Improved Current-Voltage Characteristics by Efficient Fixation of Polar Structure. Bull. Chem. Soc. Jpn. 2023, 96, 1224–1233. [Google Scholar] [CrossRef]
- Mulder, D.J.; Schenning, A.P.H.J.; Bastiaansen, C.W.M. Chiral-nematic liquid crystals as one-dimensional photonic materials in optical sensors. J. Mater. Chem. C 2014, 2, 6695–6705. [Google Scholar] [CrossRef]
- Hartmann, W.J.A.M. Ferroelectric Liquid Crystal Displays for Television Application. Ferroelectrics 1991, 122, 1–26. [Google Scholar] [CrossRef]
- Mondal, R.; Tönshoff, C.; Khon, D.; Neckers, D.C.; Bettinger, H.F. Synthesis, Stability, and Photochemistry of Pentacene, Hexacene, and Heptacene: A Matrix Isolation Study. J. Am. Chem. Soc. 2009, 131, 14281–14289. [Google Scholar] [CrossRef]
- Mushrush, M.; Facchetti, A.; Lefenfeld, M.; Katz, H.E.; Marks, T.J. Easily Processable Phenylene–Thiophene-Based Organic Field-Effect Transistors and Solution-Fabricated Nonvolatile Transistor Memory Elements. J. Am. Chem. Soc. 2003, 125, 9414–9423. [Google Scholar] [CrossRef]
- Wu, J. Polycyclic Aromatic Compounds for Organic Field-effect Transistors: Molecular Design and Syntheses. Curr. Org. Chem. 2007, 11, 1220–1240. [Google Scholar] [CrossRef]
- Fichou, D. Structural order in conjugated oligothiophenes and its implications on opto-electronic devices. J. Mater. Chem. 2000, 10, 571–588. [Google Scholar] [CrossRef]
- Zhang, L.; Colella, N.S.; Cherniawski, B.P.; Mannsfeld, S.C.B.; Briseno, A.L. Oligothiophene semiconductors: Synthesis, characterization, and applications for organic devices. ACS Appl. Mater. Interfaces 2014, 6, 5327–5343. [Google Scholar] [CrossRef] [PubMed]
- Warman, J.M.; de Haas, M.P.; Dicker, G.; Grozema, F.C.; Piris, J.; Debije, M.G. Charge Mobilities in Organic Semiconducting Materials Determined by Pulse-Radiolysis Time-Resolved Microwave Conductivity: π-Bond-Conjugated Polymers versus π–π-Stacked Discotics. Chem. Mater. 2004, 16, 4600–4609. [Google Scholar] [CrossRef]
- Pisula, W.; Zorn, M.; Chang, J.Y.; Müllen, K.; Zentel, R. Liquid Crystalline Ordering and Charge Transport in Semiconducting Materials. Macromol. Rapid Commun. 2009, 30, 1179–1202. [Google Scholar] [CrossRef]
- O’Neill, M.; Kelly, S.M. Ordered Materials for Organic Electronics and Photonics. Adv. Mater. 2011, 23, 566–584. [Google Scholar] [CrossRef]
- Funahashi, M. Nanostructured liquid-crystalline semiconductors—A new approach to soft matter electronics. J. Mater. Chem. C 2014, 2, 7451–7459. [Google Scholar] [CrossRef]
- Funahashi, M.; Kato, T. Design of liquid crystals: From a nematogen to thiophene-based π-conjugated mesogens. Liq. Cryst. 2015, 42, 909–917. [Google Scholar] [CrossRef]
- Seki, A.; Funahashi, M. Nanostructure Formation Based on the Functionalized Side Chains in Liquid-Crystalline Heteroaromatic Compounds. Heterocycles 2016, 92, 3–30. [Google Scholar]
- Kato, T.; Yoshio, M.; Ichikawa, T.; Soberats, B.; Ohno, H.; Funahashi, M. Transport of ions and electrons in nanostructured liquid crystals. Nat. Rev. Mater. 2017, 2, 17001. [Google Scholar] [CrossRef]
- Funahashi, M.; Hanna, J. Photoconductive Behavior in Smectic A Phase of 2-(4′-Heptyloxyphenyl)-6-Dodecylthiobenzothiazole. Jpn. J. Appl. Phys. 1996, 35, L703. [Google Scholar] [CrossRef]
- Funahashi, M.; Hanna, J.-I. High Carrier Mobility up to 0.1cm2V−1s−1 at Ambient Temperatures in Thiophene-Based Smectic Liquid Crystals. Adv. Mater. 2005, 17, 594–598. [Google Scholar] [CrossRef]
- Funahashi, M.; Sonoda, A. High electron mobility in a columnar phase of liquid-crystalline perylene tetracarboxylic bisimide bearing oligosiloxane chains. J. Mater. Chem. 2012, 22, 25190–25197. [Google Scholar] [CrossRef]
- Adam, D.; Closs, F.; Frey, T.; Funhoff, D.; Haarer, D.; Ringsdorf, H.; Schuhmacher, P.; Siemensmeyer, K. Transient photoconductivity in a discotic liquid crystal. Phys. Rev. Lett. 1993, 70, 457–460. [Google Scholar] [CrossRef]
- Boden, N.; Bushby, R.J.; Clements, J.; Movaghar, B.; Donovan, K.J.; Kreouzis, T. Mechanism of charge transport in discotic liquid crystals. Phys. Rev. B Condens. Matter Mater. Phys. 1995, 52, 13274–13280. [Google Scholar] [CrossRef]
- Simmerer, J.; Glüsen, B.; Paulus, W.; Kettner, A.; Schuhmacher, P.; Adam, D.; Etzbach, K.-H.; Siemensmeyer, K.; Wendorff, J.H.; Ringsdorf, H.; et al. Transient photoconductivity in a discotic hexagonal plastic crystal. Adv. Mater. 1996, 8, 815–819. [Google Scholar] [CrossRef]
- Ban, K.; Nishikawa, K.; Ohta, K.; van de Craats, A.M.; Warman, J.M.; Yamamoto, I.; Shirai, H. Discotic liquid crystals of transition metal complexes 29: Mesomorphism and charge transport properties of alkylthiosubstituted phthalocyanine rare-earth metal sandwich complexes. J. Mater. Chem. 2001, 11, 321–331. [Google Scholar] [CrossRef]
- Demenev, A.; Eichhorn, S.H.; Taerum, T.; Perepichka, D.F.; Patwardhan, S.; Grozema, F.C.; Siebbeles, L.D.A.; Klenkler, R. Quasi temperature independent electron mobility in hexagonal columnar mesophases of an H-bonded benzotristhiophene derivative. Chem. Mater. 2010, 22, 1420–1428. [Google Scholar] [CrossRef]
- Pisula, W.; Menon, A.; Stepputat, M.; Lieberwirth, I.; Kolb, U.; Tracz, A.; Sirringhaus, H.; Pakula, T.; Mullen, K. A Zone Cast-ing Technique for Device Fabrication of Field-Effect Transistors Based on Discotic Hexa-peri-Hexabenzocoronene. Adv. Mater. 2005, 17, 684–689. [Google Scholar] [CrossRef]
- van Breemen, A.J.J.M.; Herwig, P.T.; Chlon, C.H.T.; Sweelssen, J.; Schoo, H.F.M.; Setayesh, S.; Hardeman, W.M.; Martin, C.A.; de Leeuw, D.M.; Valeton, J.J.P.; et al. Large Area Liquid Crystal Monodomain Field-Effect Transistors. J. Am. Chem. Soc. 2006, 128, 2336–2345. [Google Scholar] [CrossRef] [PubMed]
- Funahashi, M.; Zhang, F.; Tamaoki, N. High Ambipolar Mobility in a Highly Ordered Smectic Phase of a Dialkylphenylterthiophene Derivative That Can Be Applied to Solution-Processed Organic Field-Effect Transistors. Adv. Mater. 2007, 19, 353–358. [Google Scholar] [CrossRef]
- Mori, T.; Komiyama, H.; Ichikawa, T.; Yasuda, T. A liquid-crystalline semiconducting polymer based on thienylene–vinylene–thienylene: Enhanced hole mobilities by mesomorphic molecular ordering and thermoplastic shape-deformable characteristics. Polym. J. 2020, 52, 313–321. [Google Scholar] [CrossRef]
- Hassheider, T.; Benning, S.A.; Kitzerow, H.-S.; Achard, M.-F.; Bock, H. Color-Tuned Electroluminescence from Columnar Liquid Crystalline Alkyl Arenecarboxylates. Angew. Chem. Int. Ed. 2001, 40, 2060–2063. [Google Scholar] [CrossRef]
- Benning, S.A.; Oesterhaus, R.; Kitzerow, H.-S. Polarized electroluminescence of a discotic mesogenic compound. Liq. Cryst. 2004, 31, 201–205. [Google Scholar] [CrossRef]
- Aldred, M.P.; Contoret, A.E.A.; Farrar, S.R.; Kelly, S.M.; Mathieson, D.; O’Neill, M.; Tsoi, W.C.; Vlachos, P. A Full-Color Electroluminescent Device and Patterned Photoalignment Using Light-Emitting Liquid Crystals. Adv. Mater. 2005, 17, 1368–1372. [Google Scholar] [CrossRef]
- Hori, T.; Miyake, Y.; Yamasaki, N.; Yoshida, H.; Fujii, A.; Shimizu, Y.; Ozaki, M. Solution Processable Organic Solar Cell Based on Bulk Heterojunction Utilizing Phthalocyanine Derivative. Appl. Phys. Express 2010, 3, 101602. [Google Scholar] [CrossRef]
- Shin, W.; Yasuda, T.; Watanabe, G.; Yang, Y.S.; Adachi, C. Self-Organizing Mesomorphic Diketopyrrolo-pyrrole Derivatives for Efficient Solution-Processed Organic Solar Cells. Chem. Mater. 2013, 25, 2549–2556. [Google Scholar] [CrossRef]
- Kogo, K.; Maeda, H.; Kato, H.; Funahashi, M.; Hanna, J. Photoelectrical Properties of a Ferroelectric Liquid Crystalline Photoconductor. Appl. Phys. Lett. 1999, 75, 3348–3350. [Google Scholar] [CrossRef]
- Funahashi, M.; Hanna, J. High Ambipolar Carrier Mobility in Self-Organizing Terthiophene Derivative. Appl. Phys. Lett. 2000, 76, 2574–2576. [Google Scholar] [CrossRef]
- Funatsu, Y.; Sonoda, A.; Funahashi, M. Ferroelectric Liquid-crystalline Semiconductors Based on a Phenylterthiophene Skeleton: Effect of the Introduction of Oligosiloxane Moieties and Photovoltaic Effect. J. Mater. Chem. C 2015, 3, 1982–1993. [Google Scholar] [CrossRef]
- Yang, M.-C.; Hanna, J.; Iino, H. Novel Calamitic Liquid Crystalline Organic Semiconductors Based on Electron-Deficient Dibenzo[c,h][2,6]naphthyridine: Synthesis, Mesophase, and Charge Transport Properties by the Time-of-Flight Technique. J. Mater. Chem. C 2019, 7, 13192–13202. [Google Scholar] [CrossRef]
- Porzio, W.; Destri, S.; Giovanella, U.; Pasini, M.; Motta, T.; Natali, D.; Sampietro, M.; Campione, M. Fluorenone–thiophene derivative for organic field effect transistors: A combined structural, morphological and electrical study. Thin Solid Films 2005, 492, 212–220. [Google Scholar] [CrossRef]
- Lincker, F.; Attias, A.-J.; Mathevet, F.; Heinrich, B.; Donnio, B.; Fave, J.-L.; Rannou, P.; Demadrille, R. Influence of polymorphism on charge transport properties in isomers of fluorenone-based liquid crystalline semiconductors. Chem. Commun. 2012, 48, 3209–3211. [Google Scholar] [CrossRef]
- Lim, C.J.; Lei, Y.; Wu, B.; Li, L.; Liu, X.; Lu, Y.; Zhu, F.; Ong, B.S.; Hu, X.; Ng, S.-C. Synthesis and characterization of two fluorenone-based conjugated polymers and their application in solar cells and thin film transistors. Tetrahedron Lett. 2016, 57, 1430–1434. [Google Scholar] [CrossRef]
- Suzuki, M.; Seki, A.; Yamada, S.; Aoki, K. Multi-stimuli-responsive behaviours of fluorenone-based donor–acceptor–donor triads in solution and supramolecular gel states. Mater. Adv. 2024, 5, 7401–7412. [Google Scholar] [CrossRef]
- Suzuki, M.; Yamada, S.; Iwai, K.; Aoki, K.; Seki, A. Acid vapor–responsive supramolecular gels of hydrogen-bonding D–A type fluorenones. Chem. Lett. 2025, 54, upaf004. [Google Scholar] [CrossRef]
- Kukhta, N.A.; da Silva Filho, D.A.; Volyniuk, D.; Grazulevicius, J.V.; Sini, G. Can Fluorenone-Based Compounds Emit in the Blue Region? Impact of the Conjugation Length and the Ground-State Aggregation. Chem. Mater. 2017, 29, 1695–1707. [Google Scholar] [CrossRef]
- Rao, P.B.; Rao, N.V.S.; Pisipati, V.G.K.M. The Smectic F Phase in nO.m Compounds. Mol. Cryst. Liq. Cryst. 1991, 206, 9–15. [Google Scholar] [CrossRef]
- Ouchi, Y.; Uemura, T.; Takezoe, H.; Fukuda, A. Molecular Reorientation Process in Chiral Smectic I Liquid Crystal. Jpn. J. Appl. Phys. 1985, 24, 893–895. [Google Scholar] [CrossRef]
- Goodby, J.W. Optical Activity and Ferroelectricity in Liquid Crystals. Science 1986, 231, 350–355. [Google Scholar] [CrossRef]
- Kelly, S.M.; O’Neill, M. Liquid Crystals for Electro-Optical Applications. In Handbook of Advanced Electronic and Photonic Materials and Devices. In Liquid Crystals, Display and Laser Materials; Nalwa, H.S., Ed.; Academic Press: New York, NY, USA, 2001; Volume 7, pp. 1–66. [Google Scholar]




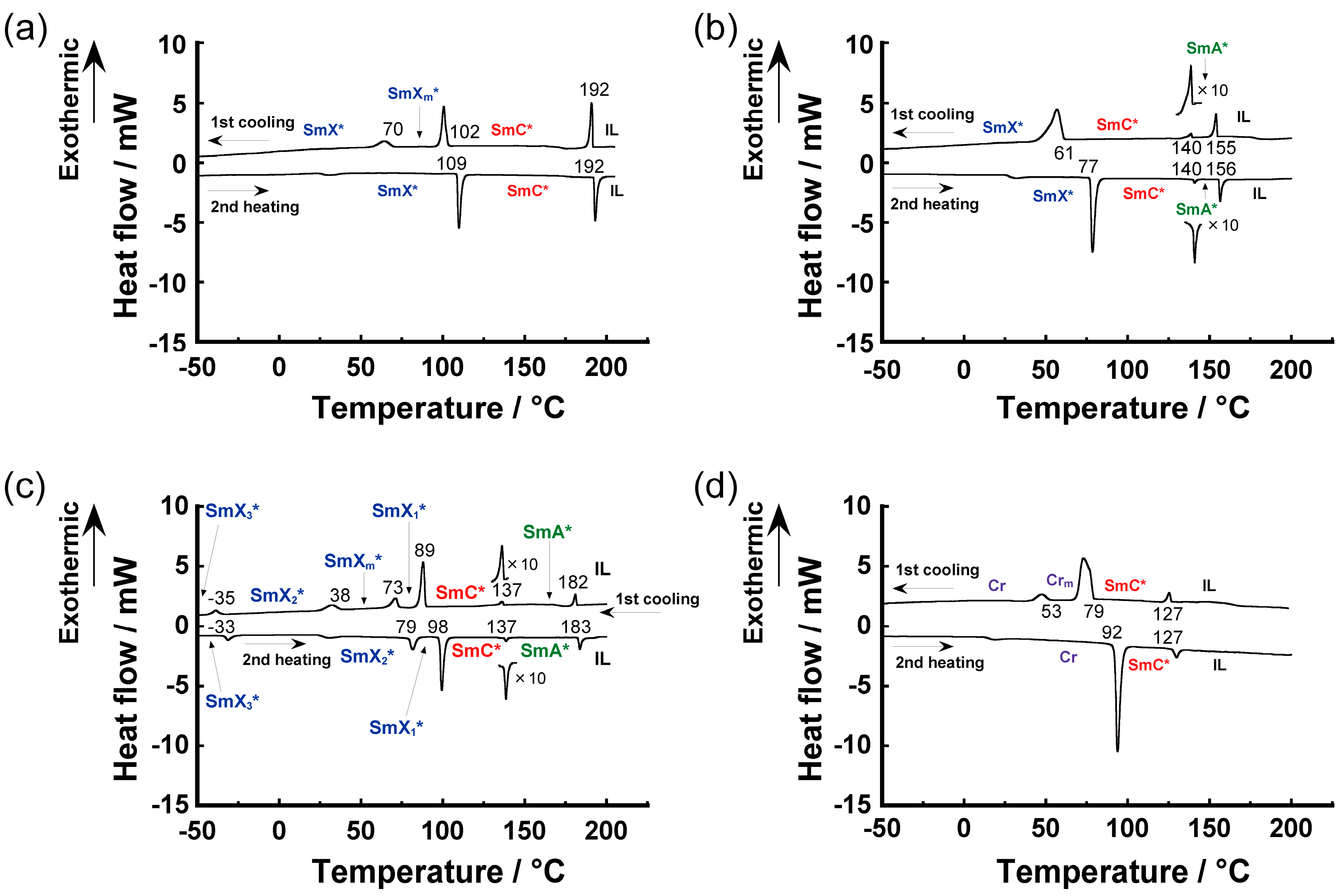
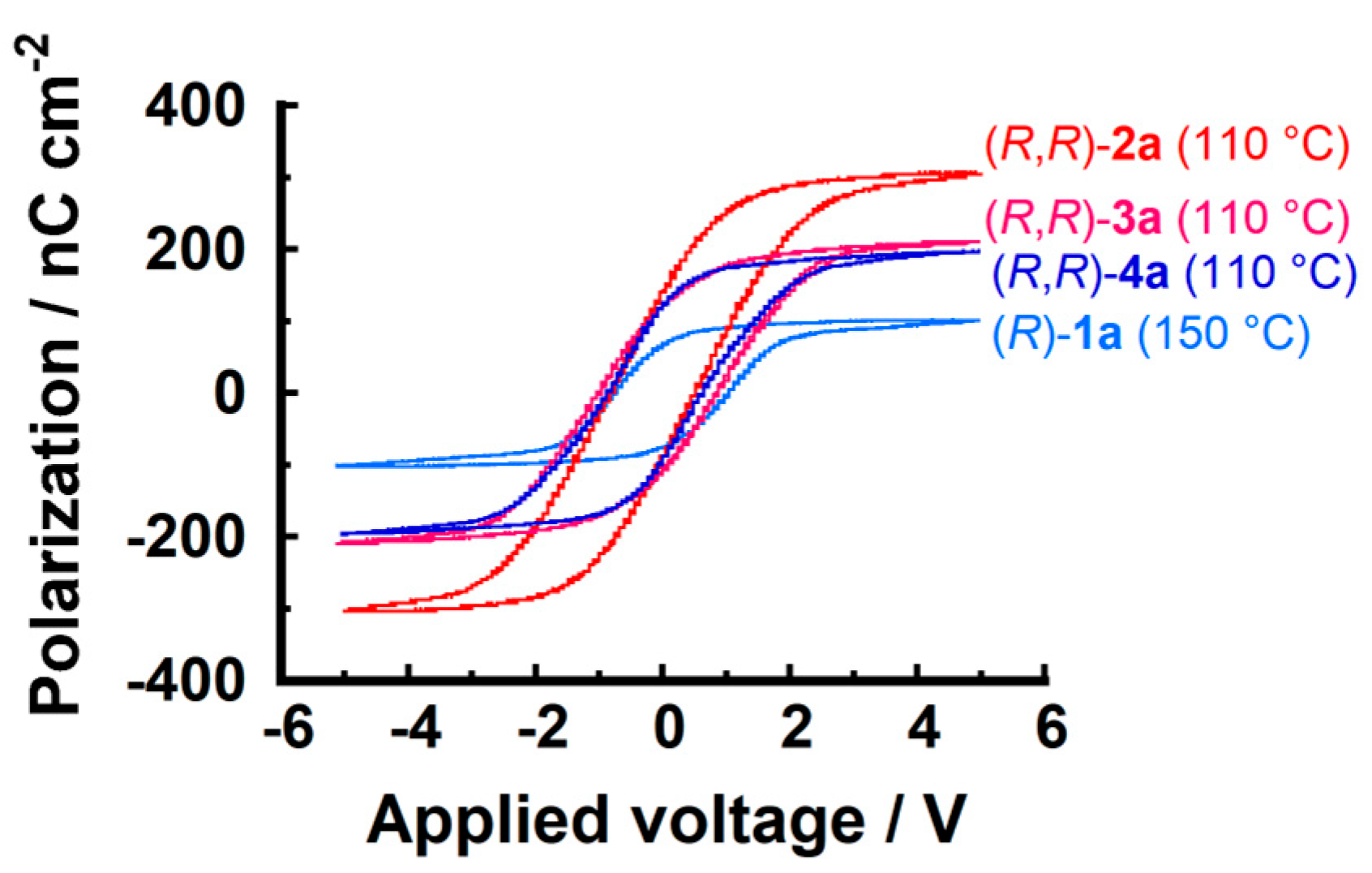
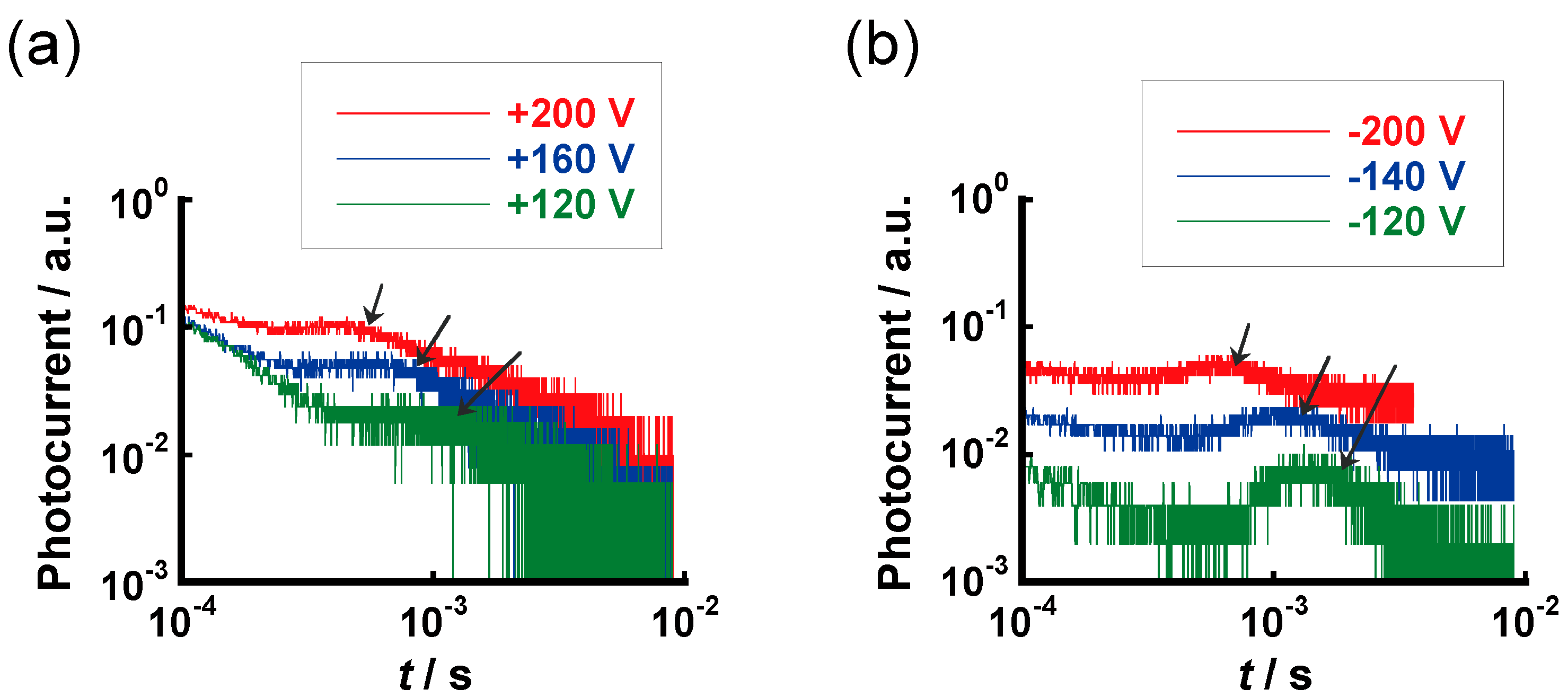
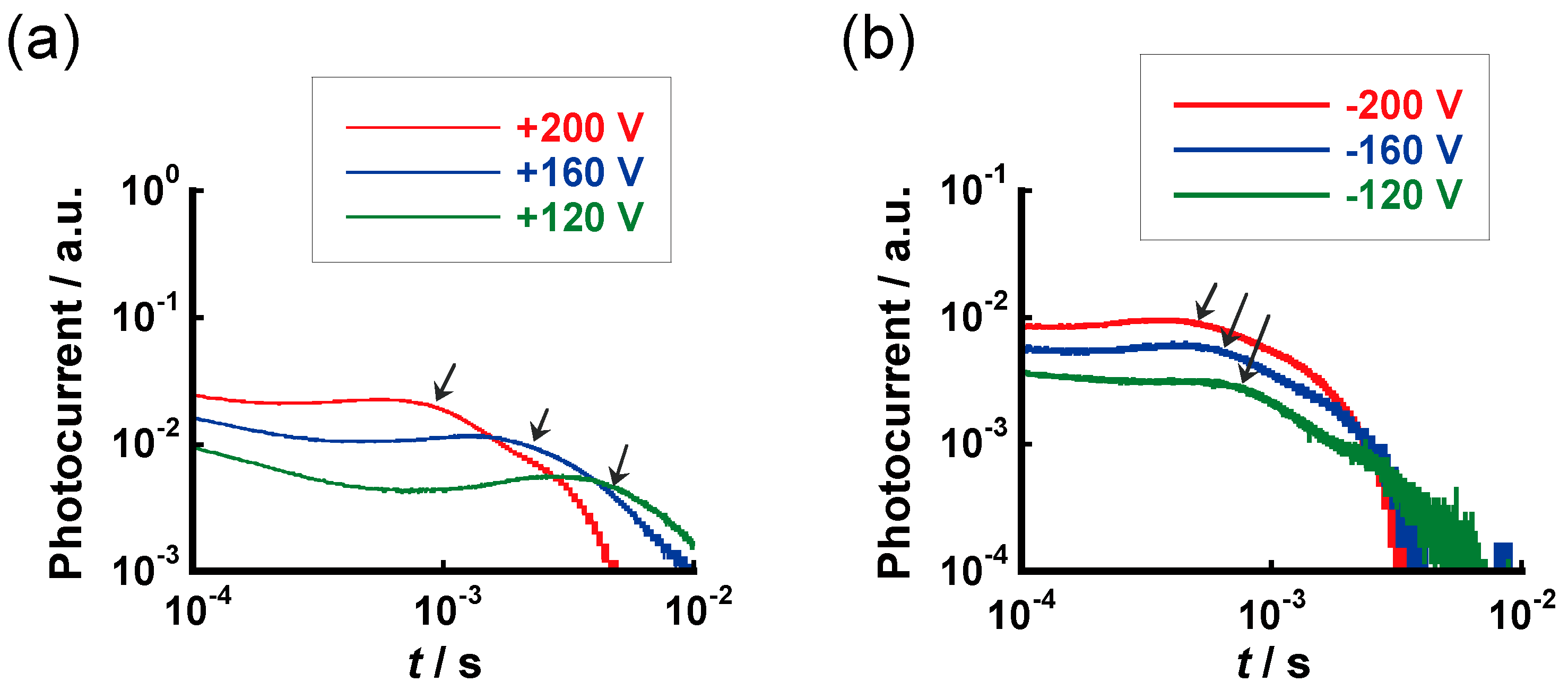
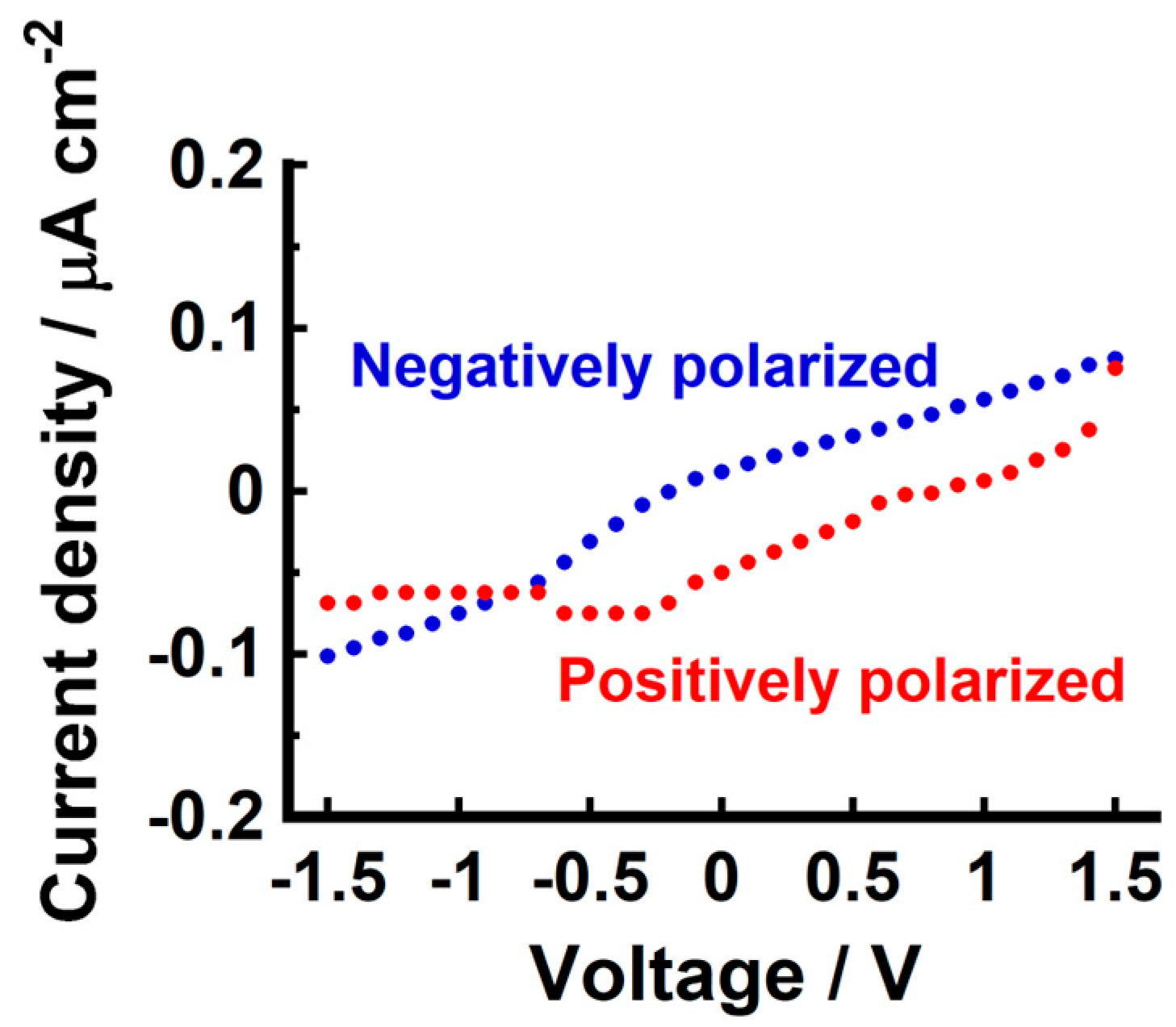
| Compound | Phase Transition Temperature/°C (Enthalpy/kJ mol−1) 1 |
|---|---|
| (R)-1a | SmX* 109 (11) SmC* 192 (8) IL |
| (R,R)-2a | SmX1* 77 (16) SmC* 140 (1) SmA* 156 (3) IL |
| (R,R)-2b | G 44 IL |
| (R,R)-3a | SmX3* −33 (2) SmX2* 79 (4) SmX1* 98 (11) SmC* 137 (1) SmA* 183 (2) IL |
| (R,R)-4a | Cr 92 (23) SmC* 127 (2) IL |
| Compound | T/°C | V/V | μ/cm2 V−1 s−1 (Positive Carrier) | V/V | μ/cm2 V−1 s−1 (Negative Carrier) |
|---|---|---|---|---|---|
| (R)-1a | 160 | +200 | 6 × 10−5 | −200 | 4 × 10−5 |
| (R,R)-2a | 120 | +200 | 3 × 10−5 | −200 | 6 × 10−5 |
| (R,R)-3a | 130 | +200 | 1 × 10−5 | −200 | 1 × 10−5 |
| (R,R)-4a | 120 | +200 | 1 × 10−5 | −200 | 1 × 10−5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doi, S.-u.; Yamada, S.; Aoki, K.; Seki, A. Ferroelectric Properties and Ambipolar Carrier Transport of 9-Fluorenone-Based Liquid Crystals. Crystals 2025, 15, 1021. https://doi.org/10.3390/cryst15121021
Doi S-u, Yamada S, Aoki K, Seki A. Ferroelectric Properties and Ambipolar Carrier Transport of 9-Fluorenone-Based Liquid Crystals. Crystals. 2025; 15(12):1021. https://doi.org/10.3390/cryst15121021
Chicago/Turabian StyleDoi, Sou-un, Syota Yamada, Ken’ichi Aoki, and Atsushi Seki. 2025. "Ferroelectric Properties and Ambipolar Carrier Transport of 9-Fluorenone-Based Liquid Crystals" Crystals 15, no. 12: 1021. https://doi.org/10.3390/cryst15121021
APA StyleDoi, S.-u., Yamada, S., Aoki, K., & Seki, A. (2025). Ferroelectric Properties and Ambipolar Carrier Transport of 9-Fluorenone-Based Liquid Crystals. Crystals, 15(12), 1021. https://doi.org/10.3390/cryst15121021






