Simple Summary
Dendritic cells are important mediators in the activation of the innate and adoptive immune response. The generation of DC/DCleu was comparable under physiological hypoxic and normoxic culture conditions, with no significant differences in frequencies of generated DC/DCleu from leukemic cell lines, peripheral blood mononuclear as well as whole blood cells from patients with acute myeloid leukemia. Moreover, the composition of immune cells and anti-leukemic immune activity after T cell enriched mixed lymphocyte culture with patients’ blood pretreated vs not pretreated with defined response modifiers (Kits) was improved, particularly under hypoxic conditions. These data show that the generation of DC/DCleu as well as the induction of anti-leukemic immune activating functionality is possible under standard normoxic as well as physiological hypoxic culture conditions ex vivo.
Abstract
Hypoxia can modulate the immune system by affecting the function and activity of immune cells, potentially leading to altered immune responses. This study investigated the generation of leukemia-derived dendritic cells (DCleu) from leukemic blasts and their impact on immune cell activation under hypoxic (5–10% O2) compared to normoxic (21% O2) conditions using various immunomodulatory Kits. The results revealed that DC/DCleu-generation was similar under hypoxic and normoxic conditions, with no significant differences observed in frequencies of generated DC/DCleu. Furthermore, the study showed that the activation of immune cells and their anti-leukemic activity improved when T cell-enriched immunoreactive cells were co-cultured with DC/DCleu which were generated with Kit I and M compared to the control after mixed lymphocyte cultures. The anti-leukemic activity was improved under hypoxic compared to normoxic conditions after MLCWB-DC Kit M. These findings suggest that DC/DCleu-cultures of leukemic whole blood with Kits under hypoxic conditions yield comparable frequencies of DC/DCleu and can even increase the anti-leukemic activity compared to normoxic conditions. Overall, this research highlights the potential of utilizing DC/DCleu (potentially induced in vivo with Kits) as a promising approach to enhance immune response in patients with acute myeloid leukemia.
1. Introduction
Acute myeloid leukemia (AML) is a clonal hematological disorder with an overall five-year survival rate in elderly patients of about 28% [1]. Although 60–80% of all patients achieve a complete remission (CR), 70–80% of these patients relapse in the following 2 years [2]. Ongoing clinical trials analyze new immunotherapeutic strategies and immunomodulatory approaches to target leukemic cells, to stabilize remissions and to improve the outcome of patients with AML [3,4].
Dendritic cells (DCs) play a pivotal role as specialized antigen presenting cells (APCs) on the interface of the innate and adoptive immune system [5,6,7]. The generation of DC/DCleu ex vivo from blast-containing peripheral blood mononuclear cells (PBMNCs) or directly from leukemic whole blood (WB) represents an immunotherapy option for patients with AML. With different protocols, myeloid progenitor cells can be converted to DC/DCleu presenting DCs’ together with patients’ blast antigens in a costimulatory manner without the induction of blasts’ proliferation [8,9,10,11,12]. DC/DCleu-generating protocols or Kits contain different response modifiers such as myelopoiesis stimulating factors [e.g., granulocyte-macrophage-colony stimulating factors (GM-CSF)], danger signaling factors [(e.g., bacterial or nucleic stimuli such as Picibanil (OK432)—a lysis product from the streptococcus pyogenes)] and mediators for the maturation of DC/DCleu [(e.g., Prostaglandin (PGE) E1 or E2, Tumor-necrosis-factor alpha (TNF-α) or Interleukin 4 (IL 4)] [6,9,10]. In T cell enriched mixed lymphocyte culture (MLC) generated DC/DCleu are known to increase frequencies of non-naïve T cells, proliferating CD71+ or CD69+ T cells, central memory T cells (Tcm, CD45RO+CCR7+), effector (memory) T cells (Teff-em, CD45RO+CCR7-) and to reduce frequencies of naive T cells and regulatory T cells (Treg, CD25++CD127low) [13,14,15,16]. DC/DCleu moreover contribute by activating cells of the innate immune system and cells on the interface of the innate and the adaptive immune system, such as natural killer cells (NK cells, CD56+CD3-), invariant natural killer cells (iNKT cells, 6B11+CD3+) or cytokine induced killer cells (CIK cells, CD56+CD3+) [11,12,17,18,19].
Hematopoietic stem cells, leukemic cells as well as immune cells reside in the bone marrow (BM) under hypoxic O2 concentrations of about 0.1–1%, in the arterial blood of about 12% or of 4–15% in peripheral blood (PB) [20,21]. That means that hematopoietic cells are exposed to changing O2 concentrations/saturation during their differentiation [22]. It was shown that hypoxic conditions might change the anti-leukemic activity of cells against leukemic blasts [23]. Moreover, reactions of immune cells prepared ex vivo for adoptive transfer under normoxic conditions might not reflect the in vivo situation [22] and might be switched to cell preparations under hypoxic conditions.
The aim of this study was to analyze the impact of hypoxic conditions on the generation of DC/DCleu from leukemic and healthy PBMNCs and WB and furthermore, to analyze the effect of DC/DCleu stimulation on the anti-leukemic immune activation after MLC in comparison to normoxic conditions.
2. Material and Methods
2.1. Sample Collection
After obtaining written informed consent in accordance with the local Ethics Committee (Pettenkoferstraße 8a, 80336 Munich, Ludwig Maximilians University Hospital in Munich; Vote No 339-05), patients’ PB or BM samples were provided by the University Hospital of Oldenburg, Tübingen and Augsburg. Anticoagulation was performed with Lithium heparintubes (7.5 mL, Sarstedt, Nuernberg, Germany) containing standardized concentrations of Heparin. PBMNCs were separated by density gradient centrifugation (density gradient 1.077 g/mL) using the Ficoll Hypaque technique. CD3+T cells were positively selected by using the MACS technology (Milteney Biotech, Bergisch Gladbach, Germany) according to manufacturers’ instructions. The purity of the resulting T cells was on average 90% (range 81–97%). Cells were frozen at −80 °C (using DMSO) and thawed according to standardized protocols.
2.2. Patients’ Characteristics and Diagnostics
DC/DCleu were generated from AML cell lines, PBMNCs- and WB-samples obtained from AML patients (n = 34) in acute phases of the disease and from healthy donors (n = 16). Average age of AML patients was 59 years (range 21–78) and of healthy volunteers 28 years (range 21–56). The female to male ratio of AML patients was 1:1.2 and of healthy volunteers 1:1.3.
The following cell lines were included in analysis: Mono-Mac 6 (AML-M5), THP-1 (AML-M5), KG-1 (AML-M4) and NB-4 (M3). These cell lines were purchased from the DSMZ (German collection of Microorganisms Cell Cultures, Braunschweig, Germany) and were cultured according to the manufacturers’ instructions (Table 1).

Table 1.
Characteristics of AML Patients and AML Cell Lines.
The classification and diagnosis of patients was based on the FAB (French–American–British) classification: AML without maturation (M1: n = 6), AML with granulocytic maturation (M2: n = 7), acute myelomonocytic leukemia (M4: n = 8) and acute monocytic leukemia (M5: n = 8). No FAB classification was available in 5 AML cases (n = 5). Patients presented with primary AML [pAML (n = 24)] or with secondary AML [sAML (n = 8)]. In two cases, no information was available. Patients’ stages were: first diagnosis (n = 29), relapse (n = 4) or relapse after HSCT (n = 1). Patients’ characteristics are given in Table 1.
2.3. Cellular Composition of Uncultured Blood Samples Used for Subsequent Experiments
AML WB samples contained on average 32.0% (e.g., CD34+, CD65+ and/or CD117+) leukemic blasts (range 7.2–92.0%), 13.8% CD3+T cells (range 3.5–18.9%), 2.3% CD19+B cells (range 0.4–8.4%), 5.8% CD56+CD3-NK cells (range 0.8–7.6%) and 3.1% CD14+monocytes (range 0.5–5.1%).
AML PBMNCs samples contained on average 51.3% leukemic blasts (range 21.0–62.0%), 4.2% CD3+T cells (range 0.3–17.3%), 0.9% CD19+B cells (range 0.2–1.9%), 1.4% CD56+CD3-NK-cells (range 0.1–3.0%) and 2.3% CD14+monocytes (range 0.2–4.2%).
Quantification of NK cells and monocytes was not possible in cases with aberrant expression of CD14 or CD 56 on leukemic blasts.
Healthy WB samples contained on average 5.6% CD14+monocytes (range 4.4–8.5%), 19.4% CD3+T cells (range 13.6–26.3%), 4.3% CD56+CD3-NK-cells (range 2.3–6.6%) and 2.3% CD19+B cells (range 0.8–4.8%).
Healthy PBMNCs samples contained on average 6.6% CD14+monocytes (range 2.6–9.4%), 33.3% CD3+T cells (range 27.1–46.7%), 4.8% CD56+CD3-NK-cells (range 0.1–4.3%) and 2.1 CD19+B cells (range 1.3–3.3%).
2.4. Cell Cultures Experiments
Human DC/DCleu-cultures, MLC-cultures as well as the cytotoxicity fluorolysis assay were set up in parallel under standard normoxic laboratory conditions (37 °C, 21% O2 and 5% CO2) as well as under hypoxic conditions (37 °C, 10% O2 and 5% CO2) [22,24]. Cell-culture experiments were set up under 37 °C, 5% O2 and 5% CO2. For the hypoxic experiments, the hypoxic Workstation INVIVO2 400 from Ruskinn Technology (Bridgend, United Kingdom) was used.
2.5. Generation of DC/DCleu from AML Cell Lines
DC/DCleu were generated from AML cell lines with the DC/DCleu-generating protocols Kit I and Kit M [6,25]. Therefore, 3–4 × 106 cells were pipetted in 12-multiwell plates in 2 ml serum-free X-Vivo15-medium (Lonza, Basel, Swiss) and were plated into 12-multiwell plates (Thermo Fisher Scientific, Darmstadt, Germany). Response modifiers and immune modulating factors were added to cultures as described below. A culture without added response modifiers served as a control. All response modifiers used for the DC/DCleu generation are approved for human treatment. Compositions of DC/DCleu-generating protocols are given in Table 2.
2.6. Generation of DC/DCleu from Isolated PBMNC or WB
DC/DCleu were generated from 3–4 × 106 PBMNC (isolated by density gradient centrifugation) from healthy donors and AML patients’ blood using Pici-PGE1 and Pici-PGE2 (Table 2) as described before [6,9,25,26]. PBMNCs were diluted in 2 ml serum-free X-Vivo15-medium (Lonza, Basel, Swiss) and were plated into 12-multiwell plates (Thermo Fisher Scientific, Darmstadt, Germany). Response modifiers were added as described below. Half medium exchange was carried out after 3–4 cell culture days. A culture without added response modifiers served as a control.
DC/DCleu were generated from healthy and leukemic WB (presenting the physiological cellular and soluble composition of the individual samples) with the DC/DCleu-generating protocols Kit I and Kit M, as described before [6,25]. A culture without added response modifiers served as a control. All response modifiers used in Kits for the DC/DCleu generation are approved for human treatment. Compositions of DC/DCleu-generating protocols are given in Table 2.
2.6.1. Picibanil-PGE1 (Pici-PGE1)
DC/DCleu were generated from PBMNCs with the DC/DCleu-generating protocol Pici-PGE1, containing 500 U/mL GM-CSF (Sanofi-Aventis, Frankfurt, Germany) and 250 U/mL IL 4 (PeproTech, Berlin, Germany). After 6–7 days, 10 μg/mL Picibanil (OK 432)—a lysis product from Streptococcus pyogenes (Chugai Pharmaceutical Co., Kajiwara, Japan) and 1 μg/mL PGE1 (PeproTech, Berlin, Germany) were added. After 7–10 days of incubation, cells were harvested and used for subsequent experiments [6,9,27].
2.6.2. Picibanil-PGE2 (Pici-PGE2)
DC/DCleu were generated from PBMNCs with the Pici-PGE2 DC/DCleu-generating protocol, with the same composition as given above for Pici-PGE1; however, PGE1 was substituted by PGE2 (PeproTech, Berlin, Germany) [9,27].
2.6.3. Kit I
DC/DCleu were generated with Kit I from WB and cell lines using 800 U/mL GM-CSF and 10 µg/mL Picibanil [25]. After 2–3 days, the same amounts of response modifiers were added and after in total 7–10 days of incubation, cells were harvested and used for subsequent experiments.
2.6.4. Kit M
DC/DCleu were generated with Kit M from WB and cell lines using 800 U/mL GM-CSF and 1 μg/mL PGE1 [25]. Incubations were performed in analogy to Kit I.

Table 2.
DC/DCleu-generating protocols.
Table 2.
DC/DCleu-generating protocols.
| DC/DCleu-Generating Protocol | Component | Concentration | Sources of DC/DCleu | Mode of Action | Culture Time | References |
|---|---|---|---|---|---|---|
| Pici-PGE1 | GM-CSF | 500 U/mL | PBMNC | GM-CSF: induction of myeloid (DC-) differentiation | 7–10 Days | [6] |
| IL 4 | 250 U/mL | |||||
| OK-432 | 10 μg/mL | |||||
| PGE1 | 1 μg/mL | |||||
| Pici-PGE2 | GM-CSF | 500 U/mL | PBMNC | IL-4: induction of DC-differentiation/maturation | 7–10 Days | [6,28] |
| IL 4 | 250 U/mL | |||||
| OK-432 | 10 μg/mL | |||||
| PGE2 | 1 μg/mL | |||||
| Kit I * | GM-CSF | 800 U/mL | WB/ Cell lines | Picibanil (OK-432): lysis product from streptococcus pyogenes; stimulates DC-differentiation | 7–10 Days | [26] |
| OK-432 | 1 μg/mL | |||||
| Kit M * | GM-CSF | 800 U/mL | WB/ Cell lines | PGE1: increases CCR7-expression and enhances DC/DCleu-maturation | 7–10 Days | [26] |
| PGE1 | 1 μg/mL |
DC dendritic cells; DCleu dendritic cells of leukemic origin; GM-CSF granulocyte macrophage colony stimulating factor; IL 4 interleukin 4; OK-432 Picibanil; PGE2 prostaglandin E2; PGE1 prostaglandin E1; PBMNCs peripheral blood mononuclear cells; WB whole blood; * European Patent 15 801 987.7-1118 and US Patent 15-517627.
2.7. Cell-Characterization by Flow Cytometry
Leukemic blasts, T cell subsets, B cells, monocytes and DC/DCleu subsets in the PBMNC- and WB-fractions were quantified using flow cytometry, as described previously. Panels with several monoclonal antibodies (moAbs) labeled with Fluorescein isothiocyanate (FITC), phycoerythrin (PE), tandem Cy7-PE conjugation (Cy7-PE), or allophycocyanin (APC) were used. Antibodies were provided by Beckman Coulter, Krefeld, Germany (a), Becton Dickinson, Heidelberg, Germany (b), Miltenyi Biotech, Bergisch Gladbach, Germany (c), Thermo Fisher, Darmstadt, Germany (d) and Santa Cruz Biotechnology, Heidelberg, Germany (e). FITC-conjugated moAbs against CD3a, CD15a, CD33a, CD34a, CD45ROa, CD65a, CD71a, CD83a, CD161b and IPO-38e were used. To detect CD3a, CD4a, CD19a, CD33a, CD34a, CD56a, CD80b, CD83a, CD117a, CD206a and 6B11b, PE-conjugated moAbs were used. MoAbs against CD3a, CD4a, CD14b, CD15b, CD19a, CD33a, CD34a, CD56a, CD65c, CD80b, CD117a and CD197b were labeled with Cy7-PE. APC-labeled moAbs against CD3a, CD4b, CD14a, CD15b, CD34a, CD45ROd, CD56a, CD65c, CD69b, CD83b, CD86g, CD117a, CD206b and CD209b were used. 7AADb was used to detect dead cells. To stain intracellular antigens (e.g., IPO-38), the FIX & PERM® Cell Fixation and Cell Permeabilization Kit (Thermo Fisher Scientific, Darmstadt, Germany) were used according to manufacturer’s instructions.
For analysis and quantification of DCs and DCleu in the total- or in the cell-subtype fractions after DC/DCleu-cultures, we used a refined gating strategy, as shown before [8,9,25]. DCleu are characterized by the simultainous expression of DCs’ together with patients’ blast antigens. To quantify generated DCleu, the cells were stained with patients or leukemic cell line specific blast-staining antibodies (e.g., CD34, CD65, and CD117), according to diagnostic reports in combination with DC-staining antibodies (e.g., CD80, CD83, CD86, CD206, and CD209), which were not expressed on blasts before culture. DCleu were quantified in the total cell fraction (DCleu/PBMNC or WB), in the DC-fraction (DCleu/DC+) or in the blast fraction to quantify the amounts of blasts converted to DCleu (DCleu/bla+). Proliferating blasts were characterized by their co-expression of CD71 or IPO-38 without co-expression of DC-markers (Table 3). According to their expression profiles we quantified frequencies of immune-reactive cells in the total cell fraction (e.g., CD3+/cells) or in the subpopulations (e.g., in CD3+cells) as given in Table 3.
2.8. Mixed Lymphocyte Culture (MLC) of T Cell Enriched Immune Reactive Cells with Kit Treated vs Untreated WB from AML-Patients
A total of 1 × 106 (thawn) autologous CD3+T cells from AML patients were co-cultured with IL 2 and a stimulator cell suspension containing approximately 2.5 × 105 generated DC/DCleu which were generated with different DC/DCleu-generating protocols from leukemic WB. MLC of T cell enriched immunoreactive cells with a stimulator cell suspension without pretreatment with different DC/DCleu-generating protocols (MLCWB) served as a control, as shown before [28].
Cells were harvested after 6–7 days; subtypes were quantified by flowcytometry and used for cytotoxicity fluorolysis assay [28].

Table 3.
Monocytes, leukemic blasts, DC/DCleu and T cell substes as evulated by flow cytometry.
Table 3.
Monocytes, leukemic blasts, DC/DCleu and T cell substes as evulated by flow cytometry.
| Name of Subgroup | Referred to | Surface Marker (CD) | Abbreviation | References | |
|---|---|---|---|---|---|
| Monocytes | CD14+ monocytes | PBMNC, WB | CD14+ | [8,9] | |
| Blasts and DC/DCleu | Leukemic blasts | cells (PBMNC, WB) | Bla+ (CD15, CD33, CD34, CD65, CD117) | Bla+/ cells (PBMNC, WB) | [8,9] |
| Dendritic cells | cells (PBMNC, WB) | DC+ (CD80, CD83, CD86, CD206, CD209) | DC+/cells (PBMNC, WB) | [8,9] | |
| Leukemia derived DC | cells (PBMNC, WB | DC+Bla+ | DCleu/cells (PBMNC, WB) | [8,9] | |
| DCleu in DC fraction | DC+ | DC+Bla+ | DCleu/DC+ | [8,9] | |
| DCleu in leukemic blast fraction | Bla+ | DC+Bla+ | DCleu/Bla+ | [8,9] | |
| Mature DC in DC fraction | DC+ | DC+CD197+ | DCmat/DC+ | [8,9] | |
| Proliferating leukemic blasts | WB | Bla+DC- CD71+ | Blaprol-CD71/Bla+ | [29] | |
| Proliferating leukemic blasts | WB | Bla+DC- IPO-38+ | Blaprol-IPO38/ Bla+ | [29] | |
| T cell substes | CD3+ pan-T cells | WB | CD3+ | CD3+/cells | [14] |
| CD4+ T cells | CD3+ | CD3+CD4+ | CD3+CD4+/CD3+ | [14] | |
| CD8+T cells | CD3+ | CD3+CD8+ | CD3+CD8+/CD3+ | [14] | |
| Naive T cells | CD3+ | CD3+CD45RO- | Tnaive/CD3+ | [14] | |
| Non-naive T cells | CD3+ | CD3+CD45RO+ | Tnon-naive/CD3+ | [14] | |
| Central (memory) T cells | CD3+ | CD3+CCR7+CD45RO+ | Tcm/CD3+ | [14] | |
| Effector (memory) T cells | CD3+ | CD3+CCR7-CD45RO+ | Teff-em/CD3+ | [14] | |
| Early proliferating T cells | CD3+ | CD3+CD69+ | Tprol-early/CD3+ | [14] | |
| Late proliferating T cells | CD3+ | CD3+CD71+ | Tprol-late/CD3+ | [14] | |
| CD4+Regulatory T cells | CD3+ | CD3+CD4+CD25++CD127low | CD4+Treg/CD4+ | [14,16]] | |
| CIK cells | Cytokine induced killer cells | MLC | CD3+CD56+ CD3+CD161+ | CD3+CD56+/MLC CD3+CD161+/MLC | [14,16,18,19] |
| NK cells | Natural killer cells | MLC | CD3-CD56+ CD3-CD161+ | CD3-CD56+/MLC CD3-CD161+/MLC | [14,16,18,19] |
| iNKT cells | Invariant natural killer cells | MLC | 6B11+ CD3+6B11+ | 6B11+/MLC CD3+6B11+/MLC | [14,16,18,19] |
PBMNC peripheral blood mononuclear cells; WB whole blood; CD cluster of differentiation; DC dendritic cells; DCleu dendritic cells of leukemic origin; Bla leukemic blasts; MLC mixed lymphocyte culture.
2.9. Cytotoxicity Fluorolysis Assay (CTX)
The Cytotoxicity Fluorolysis Assay was conducted to assess the lytic activity of autologous T cell-enriched immunoreactive cells after stimulation with DC/DCleu containing cell fractions after treatment with Kit M, Kit I or control (without added response modifies) after MLC (‘effector cells’) against autologous leukemic blasts (‘target cells’). Therefore, effector and target cells (with a ratio of 1:1) were co-cultured under hypoxic and normoxic conditions and incubated for 3 and 24 h. Target cells were stained with respective anti-bodies before incubation. After harvest, 7AAD and a defined number of Fluorosphere beads (Beckman Coulter) were added. As a control, effector and target cells were cultured separately and mingled shortly before measurements. Flow cytometric analyses were performed after 3 and 24 h of effector and target cells’ co-incubation using a refined gating strategy [30]. The lytic activity against leukemic target blasts (blast lysis) is defined as the difference in frequencies of viable blasts in the effector target cell cultures as compared to controls. Improved blast lysis is defined as the difference in proportions of blast lysis achieved after MLC with DC/DCleu generated with Kits compared to control.
2.10. Cell Cycle Experiments
The cell cycle profile of cell line samples was determined by staining of DNA with PI-fluorescent dyes. PI intercalates into the groove of double-stranded DNA producing a highly fluorescent signal. As PI can also bind to double-stranded RNA, the cells must be treated with RNAse for DNA resolution. We associated a fixation (paraformaldehyde) with PI staining. The PI staining of DNA allowed the detection of cells in G0/G1, S phase, and G2/M as described earlier [31].
2.11. Quantitative PCR (Real Time PCR)
Total RNA was isolated from 106 cells of each cell line using MagNA Pure LC mRNA HS Kit (Roche, Basel, Switzerland) according to the manufacturer’s instructions. cDNA was synthesized from 1 μg aliquots of total RNA in a 20 μL standard reaction mixture using SuperScript® III First-Strand Synthesis System for RT-PCR (Invitrogen, Camarillo, CA, USA) according to manufacturer’s instructions. Quantitative Real-time polymerase chain reaction (RT-PCR) was performed using the 7900HT Fast Real-Time PCR System (Applie Biosystems, Waltham, MA, USA) with 2 μL of cDNA, Fast SYBR® Green Master Mix (Applied Biosystems, Waltham, MA, USA) [32]. We checked the expression of the fusion genes related to each cell line in Normoxic vs Hypoxic condition. Furthermore, glyceraldehyde-3-phosphate dehydrogenase gene (GAPDH) was used as a reference for the normalization of ΔCT values.
2.12. Statistical Methods
Mean ± standard derivations are given. Statistical comparisons of two groups were performed using the two-tailed t-test (in cases with data normally distributed) and the Mann–Whitney–Wilcoxon Test (in cases with data not normally distributed). Statistical analyses were performed with Microsoft Excel 2010® (Microsoft, Redmond, Washington, USA) and SSPS Statistic 24 software© (IBM, Armonk, NY, USA). Pearson correlation tests were used to evaluate correlations between traits represented in graphs. Differences were defined as ‘not significant’ in cases with p-values > 0.1, as ‘tendentially significant’ (significant *) with p-values between 0.1 and 0.05, as ‘significant’ (significant **) with p-values between 0.05 and 0.005 and as ‘highly significant’ (significant ***) with p-values < 0.005. Figures were created with GraphPad Prism7© (GraphPad Software, San Diego, CA, USA).
3. Results
3.1. Prolog
In a first step, we generated DC/DCleu from four different leukemic cell lines and evaluated mRNA profiles under hypoxic and normoxic conditions. In the next step, we generated DC/DCleu from leukemic and healthy PBMNCs as well as from WB, to simulate physiological conditions with different DC/DCleu-generating protocols. Furthermore, we analyzed the immune stimulating effect of these generated DC/DCleu after MLC under hypoxic and normoxic conditions, evaluated the resulting anti-leukemic activity and correlated these findings with frequencies of DC/DCleu-subtypes.
3.2. AML Cell Lines’ Phenotypic and Genotypic Profiles Do Not Change under Hypoxic Culture
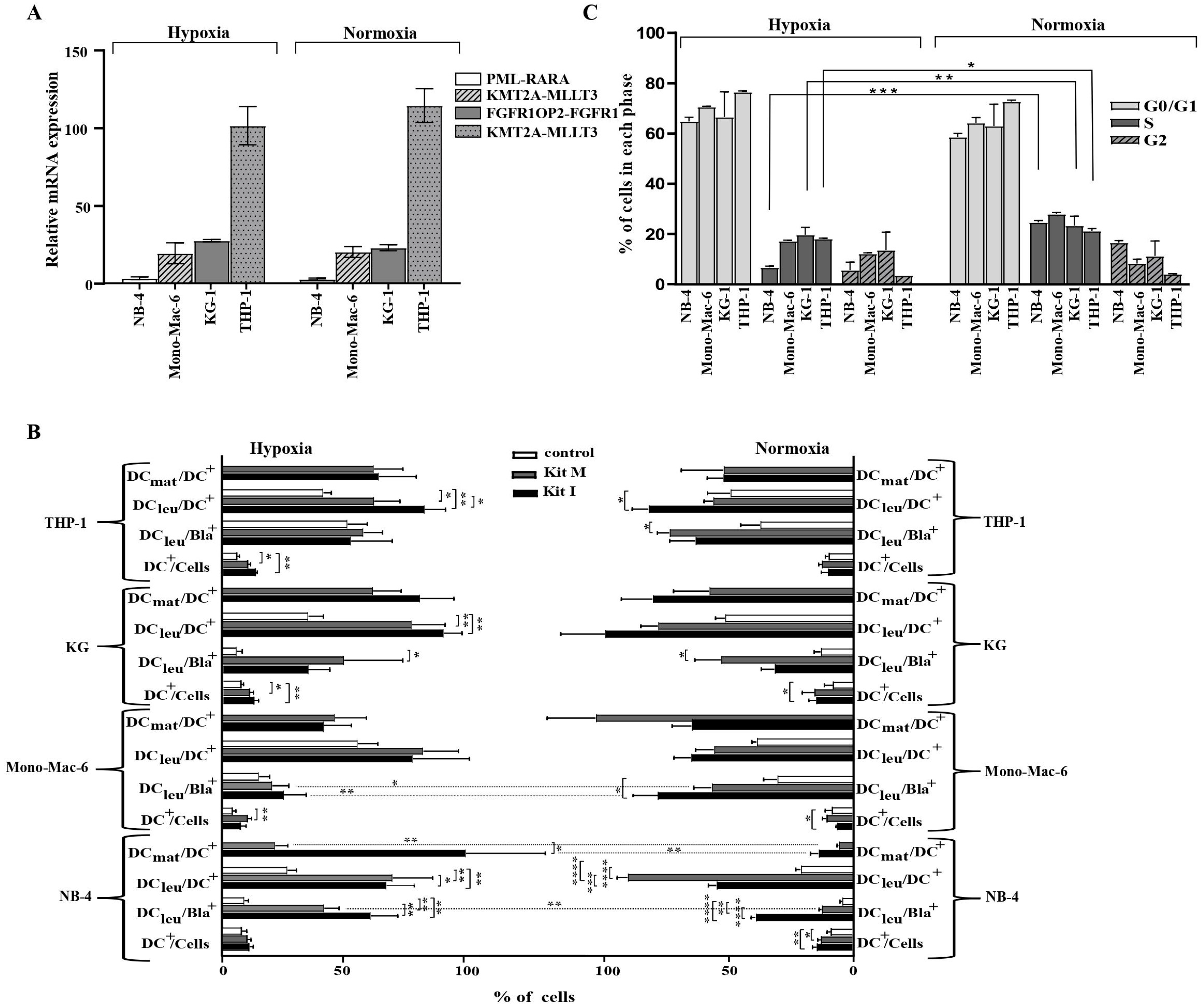
We compared the relative mRNA expression levels of cell line specific fusion genes as mentioned in Table 1 after five passages of AML cell lines under hypoxic and normoxic conditions. Levels of mRNA expression were analyzed by quantitative real-time RT-PCR, using glyceraldehyde-3-phosphate dehydrogenase gene (GAPDH) as the housekeeping control. Our results indicated no significant differences for the expression of these fusion genes under hypoxic vs. normoxic conditions (NB-4 ΔΔCT 3.5 vs 2.9; Mono-Mac-6 ΔΔCT 23.1 vs. 27.6; KG ΔΔCT 19.6 vs. 20.3; THP-1 ΔΔCT 101.2 vs. 114.5) (Figure 1A).
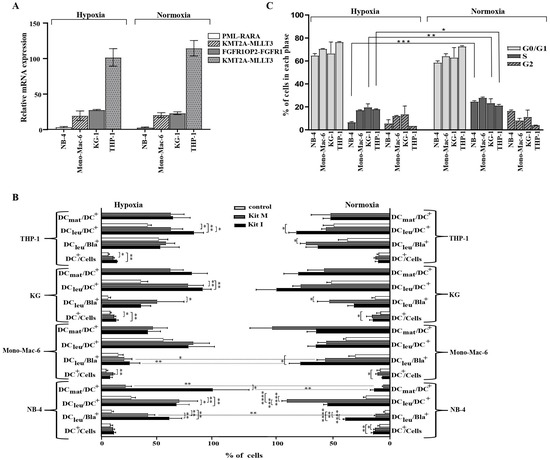
Figure 1.
Characterization of AML cell lines and the generation of DC/DCleu under hypoxic and normoxic conditions. qPCR Real Time Quantitative Polymerase chain reaction; AML acute myeloid leukemia; mRNA messenger RNA; hypoxic conditions (5% O2 saturation); normoxic conditions (21% O2 saturation); CT cycle threshold; GAPDH glyceraldehyde-3-phosphate dehydrogenase gene, DCmat mature DC; DC dendritic cells; DCleu dendritic cells of leukemic origin; Bla leukemic blast, hypoxic conditions (5% O2 saturation); normoxic conditions (21% O2 saturation); S synthesis phase of the cell cycles; G1 gap/growth 1 phase of the cell cycles; G2 gap/growth 2 phase of the cell cycle. * p-values between 0.1 and 0.05, ** p-values between 0.05 and 0.005, *** p-values < 0.005, **** p-values < 0.0005. (A). qPCR analyses of fusion genes Four different AML cell lines (NB-4, Mono-Mac-6, KG-1 and THP-1) were cultured in the RPMI medium under hypoxic as well as normoxic conditions. Results show no differences between the expression of fusion genes (PML-RARA fusion gene in the NB-4 cell line, FGFR1OP2-FGFR1 fusion gene in the KG-1 cell lines, KMT2A-MLLT3 fusion gene in the Mono-Mac-6 cell line and KMT2A-MLLT3 fusion gene in the THP-1 cell line) under hypoxic (left side) or normoxic (right side) conditions. Relative expressions of fusion genes specific for each cell line were tested after five passages. The glyceraldehyde-3-phosphate dehydrogenase gene (GAPDH) was used as the housekeeping control. The y-axis shows the ΔΔCT differences between the genes of interest and the housekeeping gene. Average frequencies ± standard deviation are given. (B). DC/DCleu subsets after DC/DCleu-generation Results of DC/DCleu subsets after DC/DCleu-generation from four different cell lines lines (NB-4, Mono-Mac-6, KG-1, and THP-1) with Kit I, Kit M and control (culture without added response modifiers) are given. Only in the NB-4 cell line could significantly higher frequencies of mature DCs be generated with Kit I and Kit M under hypoxic compared to normoxic conditions. For all other subsets, comparable frequencies could be generated. Average frequencies ± standard deviations are given. (C). Cell cycle analysis Cells were fixed in ethanol, stained with propidium iodide (PI) and analysed for DNA content to determine populations in G1 and S phases of the cell cycle. Significantly lower frequencies of cells are found in the S-phase under hypoxic vs. normoxic conditions for the NB-4, KG-1 and THP-1 cell lines. Average frequencies ± standard deviation are given.
3.3. DC/DCleu Generation from Leukemic Cell Lines with Kits Is Comparable under Hypoxic and Normoxic Conditions without Induction of Blast Proliferation
DC/DCleu-generation with Kit I and Kit M was comparable in all four leukemic cell lines under hypoxic as well as normoxic conditions with significantly higher frequencies of DC/DCleu-subtypes found after DC/DCleu-generation with Kits compared to control. In general, Kit I was superior to Kit M in generating DC/DCleu (including subgroups) from leukemic cell lines (Figure 1B). In the direct comparison of hypoxic and normoxic conditions, significantly more mature DCs (DCmat/DC+) could be generated in the NB-4 cell line with Kit I and Kit M under hypoxic compared to normoxic conditions (Figure 1B). Also, significantly more DCleu/Bla+ could be generated with Kit M under hypoxic compared to normoxic conditions. For the other cell lines, no significant differences could be found in the direct comparison of the amounts of generated DC/DCleu as well as DC/DCleu subgroups.
3.4. Significantly Lower Frequencies of Cells from Leukemic Cell Lines Found in S-Phase of Cell Cycles under Hypoxic Compared to Normoxic Conditions
We found significantly lower frequencies of cells in the S-phase of the cell cycle under hypoxic compared to normoxic conditions for the cell lines NB-4, KG-1 and THP-1. For the cell line Mono-mac-6, no significant differences were found. Frequencies of cells in the remaining cell cycle phases did not differ significantly (Figure 1C).
3.5. Generation of DC/DCleu from Leukemic and Healthy PBMNCs Is Comparable under Hypoxic and Normoxic Conditions
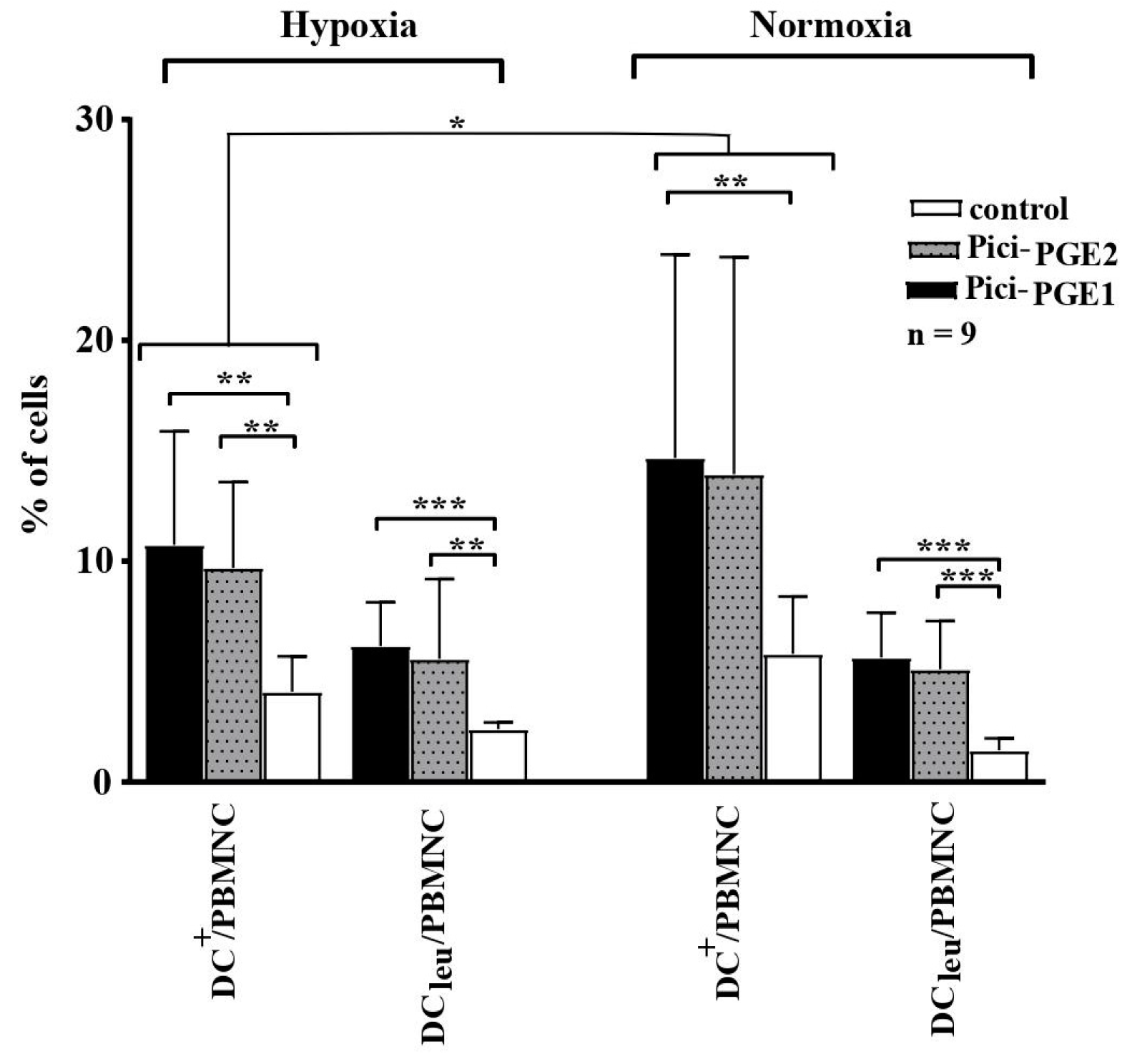
DC/DCleu were generated from healthy and leukemic PBMNCs (n = 9) under hypoxic and normoxic conditions with the two DC/DCleu-generating protocols Pici-PGE1 and Pici-PGE2 compared to control. The following patients’ blood samples were included in this analysis: Pat. 15, 16, 17, 19, 20, 21, 22, 24, 25. Further characteristics of the patients and stages of the disease are given in Table 1. Significantly higher frequencies of DC/DCleu (including subgroups) were found after culture with Pici-PGE1 and Pici-PGE2 under hypoxic as well as normoxic conditions compared to control (Figure 2). Comparable results were found for healthy cell samples.
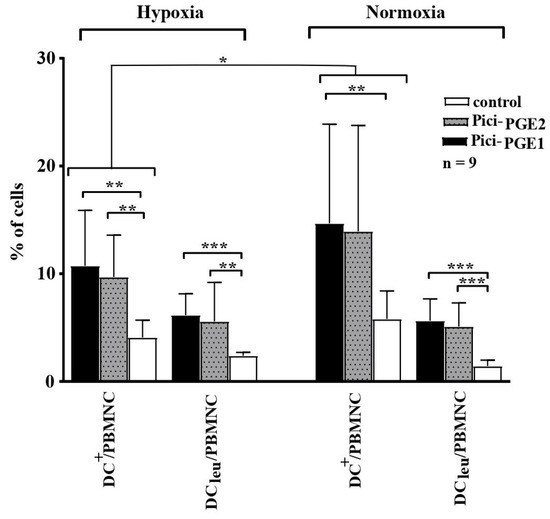
Figure 2.
DC/DCleu generation from leukemic PBMNCs under hypoxic and normoxic conditions. Hypoxic conditions (5% O2 saturation); normoxic conditions (21% O2 saturation); DC dendritic cells; DCleu dendritic cells of leukemic origin; PBMNCs peripheral blood mononuclear cells. * p-values between 0.1 and 0.05, ** p-values between 0.05 and 0.005, *** p-values < 0.005.
Comparable frequencies of DC/DCleu could be generated with Pici-PGE1 and Pici-PGE2 from leukemic PBMNCs under hypoxic and normoxic conditions. Compositions of Pici-PGE1 and Pici-PGE2 protocols are given in Table 2. Average frequencies ± standard deviations of DC/DCleu and their subtypes under hypoxic (left side) and normoxic (right side) conditions are given.
3.6. Generation of DC/DCleu with Kits from Leukemic and Healthy WB Is Comparable under Hypoxic and Normoxic Conditions—without Induction of Blasts’ Proliferation
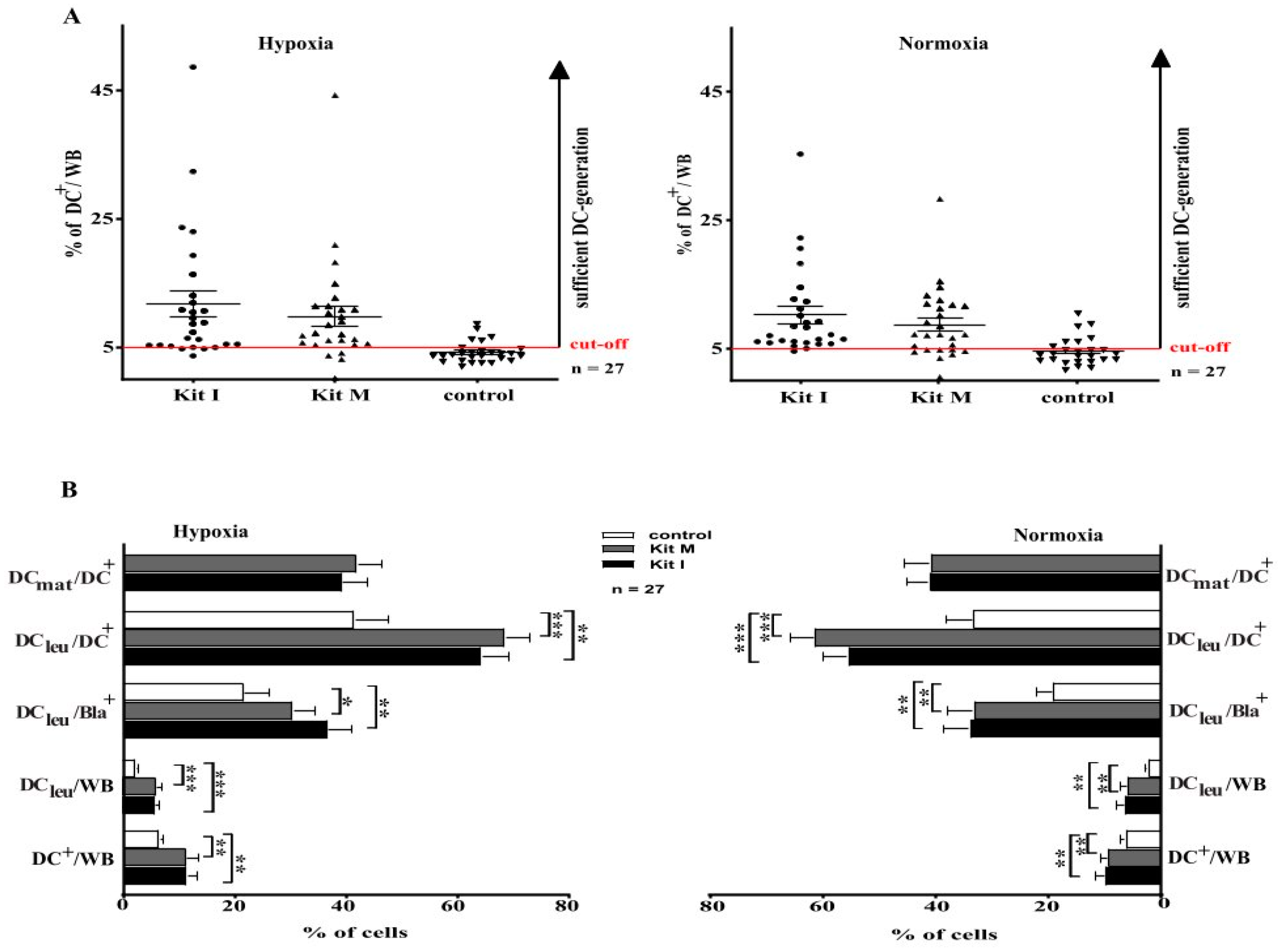
We defined a cut-off value of ≥ 5% DC+/WB as a ‘sufficient DC-generation’ from leukemic WB with Kits. Under hypoxic conditions, a sufficient DC-generation was possible in 88% of cases with Kit I, in 85% of cases with Kit M and in 18% of control cases (Figure 3A). Under normoxic conditions, an adequate and sufficient DC-generation from leukemic WB was possible in 96% of cases cultured with Kit I, in 77% of cases with Kit M and in 25% in the control group (Figure 3A). Comparisons showed no significant differences in sufficient DC-generation under hypoxic vs normoxic conditions.
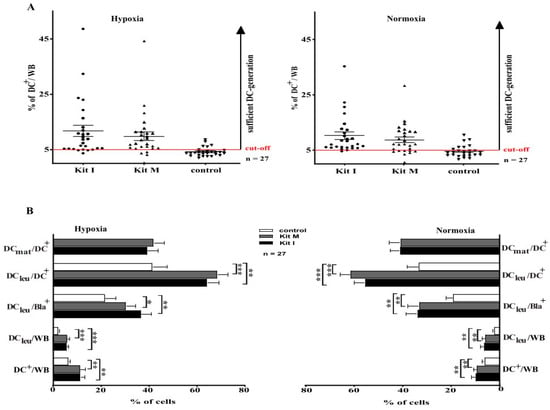
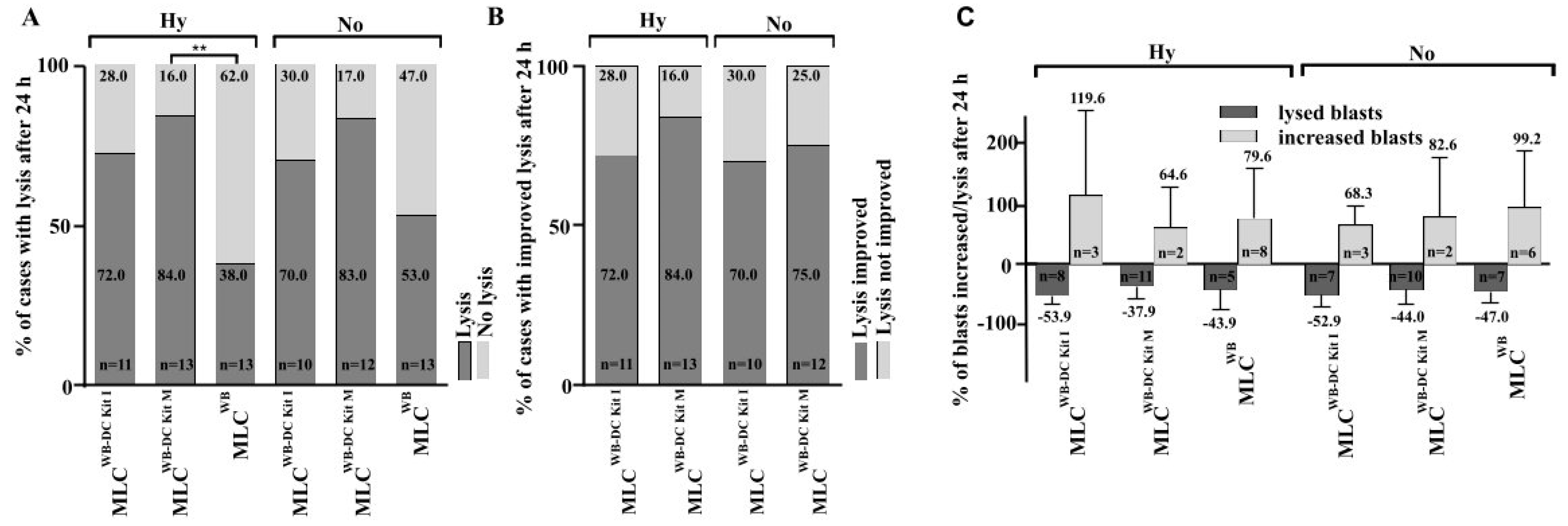
Figure 3.
DC/DCleu generation from leukemic WB under hypoxic and normoxic conditions. WB whole blood; DC dendritic cells; hypoxic conditions (10% O2 saturation); normoxic conditions (21% O2 saturation), DC dendritic cells; DCleu dendritic cells of leukemic origin; DCmat mature dendritic cells. (A). Leukemic WB samples were cultured in parallel under hypoxic and normoxic conditions with Kit I and Kit M compared to controls (without added response modifiers). Proportions of cases with ‘sufficient DC-generation’ (setting a cut-off-value at 5% DC+/WB) from leukemic WB were not different under hypoxic (left side) and normoxic conditions (right sight). Each dot (● ▼) characterizes DC-frequencies generated from each AML-patient in each given case. (B). Leukemic WB samples were cultured with Kit I, Kit M and without added response modifiers (control) under hypoxic (left side) and normoxic (right sight) conditions. Average frequencies ± standard deviation of DCs and their subtypes are given. * p-values between 0.1 and 0.05, ** p-values between 0.05 and 0.005, *** p-values < 0.005.
A detailed analysis revealed that we generated significantly higher frequencies of DC (including subtypes) from leukemic WB with Kits compared to control—under hypoxic as well as under normoxic conditions (Figure 3B). Comparable results were found for healthy samples.
We found comparable frequencies of proliferating blasts (Blaprol-CD71/Bla+ and Blaprol-Ipo-38/Bla+) after culture of leukemic WB with Kits compared to control under hypoxic and normoxic conditions: (hypoxic conditions: %Blaprol-CD71/Bla+: control: 9.2 ± 7.3%; Kit I: 9.3 ± 10.9%, p < 0.2; Kit M: 12.3 ± 11.9%, p < 0.2; normoxic conditions: %Blaprol-CD71/Bla+: control: 9.8 ± 12.6% Kit I: 10.5 ± 11.7%, p < 0.2; Kit M: 10.6 ± 11.4%, p < 0.2).
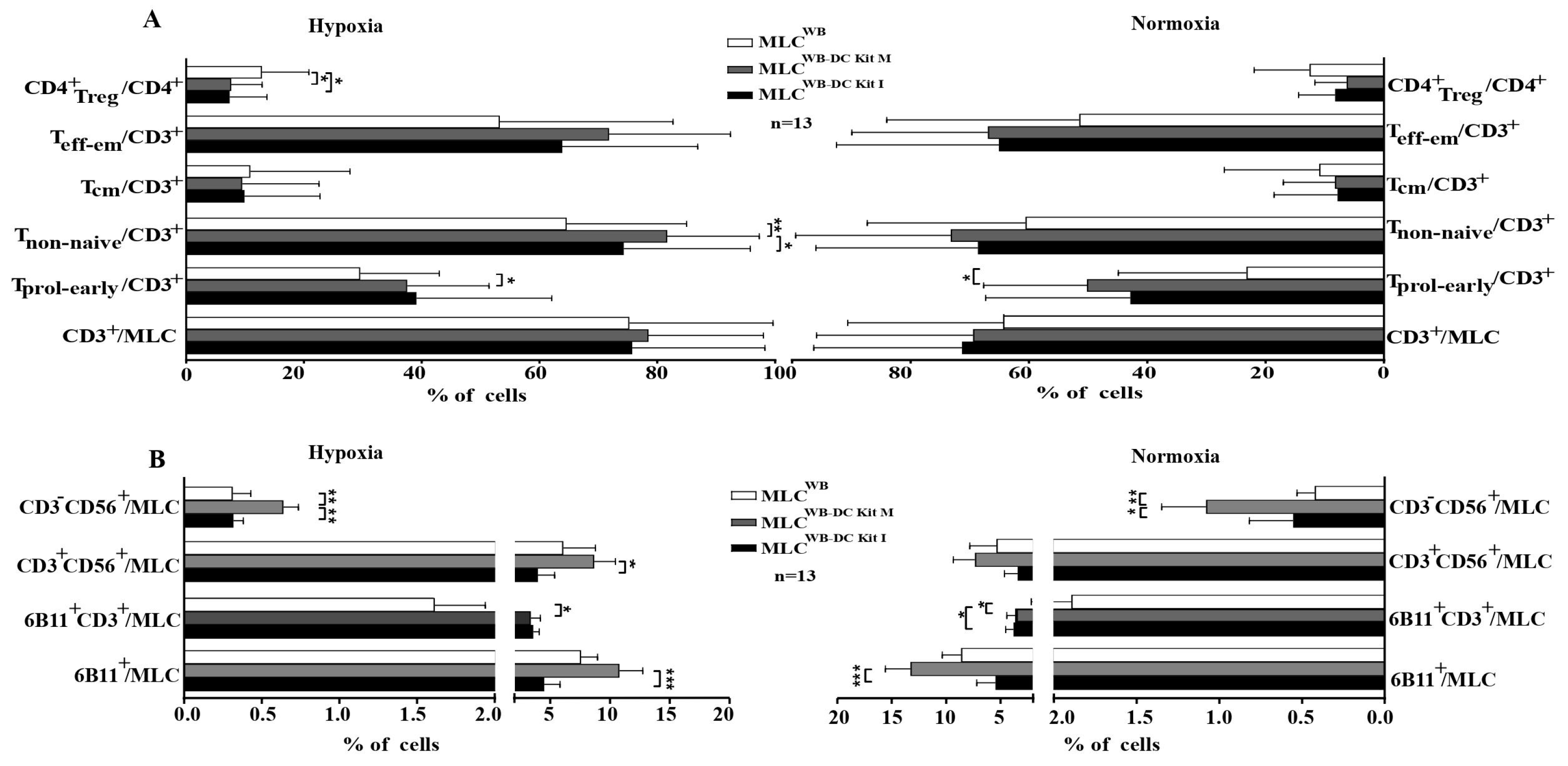
3.7. Significantly Lower Frequencies of Treg Cells Found after MLCWB-DC Kits under Hypoxic Conditions Compared to Control
In general, we found a significantly higher activation status of immunoreactive T cell subtypes after MLCWB-DC Kits compared to MLCWB (e.g., Tprol-eraly, Tnon-naiv) and reduced frequencies of Treg. Frequencies of Tregs significantly decreased after MLCWB-DC Kit M and MLCWB-DC Kit I vs control under hypoxic conditions (Figure 4A).
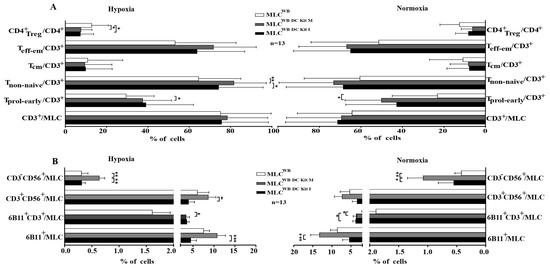
Figure 4.
Composition of immunoreactive cells after T cell enriched MLC under hypoxic and normoxic conditions. % percentage; (A) shows amount of cells of the adaptive immune system, (B) shows amounts of cells of the innate immune system. MLC mixed lymphocyte cultures; MLCWB-DC Kit I and MLCWB-DC Kit M MLC with Kit I or Kit M pretreated WB, MLCWB control; hypoxic conditions (10% O2 saturation); normoxic conditions (21% O2 saturation). * p-values between 0.1 and 0.05, ** p-values between 0.05 and 0.005, *** p-values < 0.005.
Furthermore, we found significantly higher frequencies of NK cells after MLCWB-DC Kit M compared to the control, pointing to a stimulating effect of generated DC/DCleu on cells of the innate immune system under hypoxic and normoxic conditions (Figure 4B).
In the direct comparison of hypoxic and normoxic conditions no significant difference were observed.
T cells enriched immunoreactive cells were stimulated with Kit I or Kit M pretreated (DC/DCleu containing) WB compared to the control without pretreatment. Average frequencies of stimulated cells after MLCWB-DC Kit I, MLCWB-DC Kit M and MLCWB (control) ± standard deviation are given in Figure 4A. Frequencies of iNKT and NK cells under hypoxic and normoxic conditions are given in Figure 4B. Explanations of all analyzed immunoreactive cells subtypes are given in Table 3.
3.8. Kit Pre-Treated Leukemic WB, Leads to Significantly Improved Anti-Leukemic Activation after T Cell Enriched MLC, Especially in Hypoxic Conditions
We analyzed the blast lytic effect of immunoreactive cells after T cell enriched MLCWB-DC Kits and MLCWB under hypoxic as well as normoxic conditions. Blast lysis was evaluated after 3 h and 24 h of incubation of blast targets with effector cells after MLC.
After 24 h of incubation of target cells with effector cells, blast lysis was achieved in 72% of cases after MLCWB-DC Kit I, in 84% after MLCWB-DC Kit M vs 38% of cases in the control group (MLCWB) under hypoxic conditions. Compared to the control group, significantly more cases achieved blast lysis after MLCWB-DC Kit M (Figure 5A). We could see a clear advantage in the improvement of blast lysis after MLCWB-DC Kit M compared to the control (Figure 5B) and in the frequency of improved lysis after 24 h although differences were not significant under hypoxic and normoxic conditions (Figure 5C).
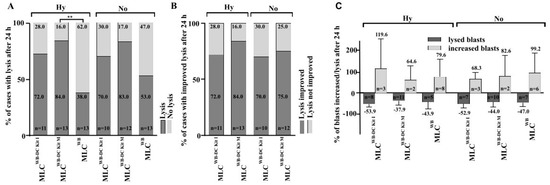
Figure 5.
Lysis of leukemic blasts after T cell enriched MLC under hypoxic and normoxic conditions. % percentage; h hours; MLC T cell enriched mixed lymphocyte cultures; MLCWB-DC Kit I and MLCWB-DC Kit M MLC with Kit I or Kit M pretreated WB; MLCWB control. ** p-values between 0.05 and 0.005.
Achieved blast lysis after MLCWB-DC Kit I, MLCWB-DC Kit M and MLCWB (control) after 24 h of co-cultures with leukemic blats (target cells). Percentage of cases with lysis after 24 h (Figure 5A), percentage of cases with improved lysis after 24 h (Figure 5B) and percentage of blasts increased/lysed after 24 h (Figure 5C) under hypoxic and normoxic conditions are presented.
3.9. Significant Correlation of Anti-Leukemic Reactivity and DC/DCleu Subtypes under Hypoxic as Well as Normoxic Conditions
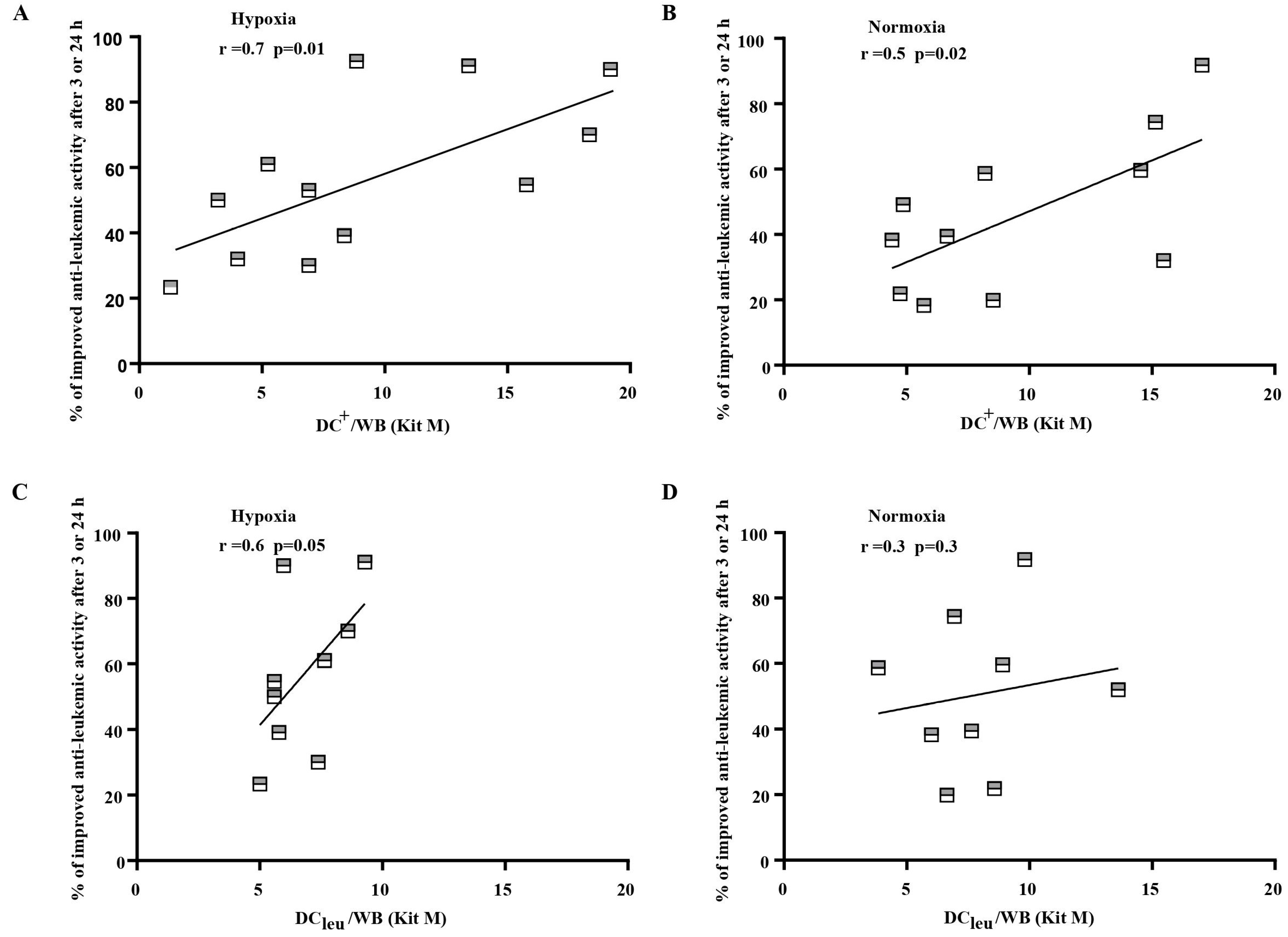
Correlating improved anti-leukemic reactivity of immune reactive cells after MLCWB-DC Kit I and MLCWB-DC Kit M vs MLCWB (control) with frequencies of generated DC and DCleu in cultures with Kits, we found significant positive correlations with generated DC+/WB under hypoxia and normoxia (r = 0.7; p < 0.01; r = 0.5; p < 0.02) and with DCleu/WB under Hypoxia (r < 0.6; p < 0.05), but not under normoxia with Kit M. (Figure 6). These correlations were not found for results obtained with Kit I.
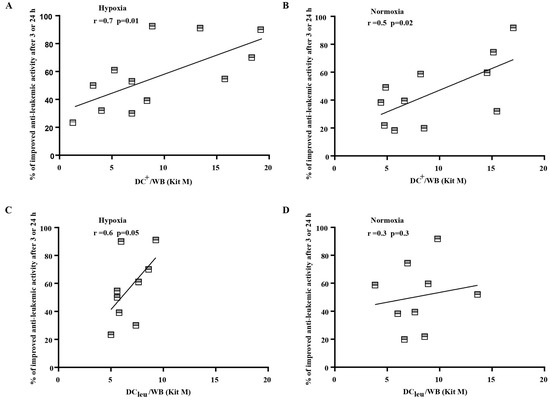
Figure 6.
Correlation analyses of cases with improved anti-leukemic reactivity and DC and DCleu under hypoxic (A,C) and normoxic conditions (B,D). DC dendritic cells; WB whole blood; DCleu dendritic cells of leukemic origin. Improved anti-leukemic activity after MLCWB-DC Kit M in comparison to MLCWB (y axis) correlated positively with frequencies of generated DC+/WB and DCleu/WB with Kit M (x axis) under hypoxic (left) and normoxic (right) conditions. Correlation was evaluated with pearson correlation analyses.
4. Discussion
4.1. DC/DCleu-Based Immunotherapy for AML
AML is a clonal hematopoietic disorder with a high risk of relapse due to blasts’ immune escaping mechanisms, such as impaired or mistaken antigen expressions, resistance to apoptosis or other inhibitory mechanisms [33]. To address this issue, various immunotherapeutic strategies (including DC/DCleu-based strategies) are being explored to reactivate the antitumor immune response [34,35]. On the one hand DCs can be generated ex vivo from monocytes and loaded with different leukemia associated antigens and on the other hand DC/DCleu can be generated directly from leukemic blasts presenting patients’ individual antigen repertoire [9,10,36]. Re-administration of these ex vivo generated and prepared DC/DCleu have shown to increase the frequencies of leukemia-specific (T) cells in vivo and the treatment achieved stable complete remissions in elderly AML patients [37,38,39]. A new and interesting approach could be to induce the production of DC/DCleu from leukemic blasts in vivo after the treatment of patients with DC/DCleu-inducing response modifiers [6].
4.2. Hypoxia, a Condition with Influence on Haematological and Immune Reactions
Hematopoietic and immunoreactive cells are exposed to changing O2 concentrations in the BM of about 0.1–1%, in arterial blood of about 12% and in PB of about 4–15% [20,21]. Several groups have utilized hypoxic conditions at 6% O2 to simulate physiological conditions ex vivo [21,40]. Up to now, no consistent effect of hypoxia on the immune system was shown [41]. Some studies suggest that hypoxia might suppress the anti-leukemic effect of immune cells, weakening the success of current anti-leukemic therapies [42]. Moreover a correlation between the hypoxic tumor microenvironment and poor response to radiation/chemotherapy in patients was seen [42]. Furthermore, physiological hypoxia could stimulate the proliferation and activation of NK cells, contributing to anti-leukemic functions [23]. Different studies have demonstrated that the effects of oxygen on immune cells depend on the tissue and the duration of hypoxia exposure. For example, while 6% O2 is considered as hypoxic, this oxygen concentration represents a normal physiological condition in the BM and stem cell niche [43,44]. Therefore, we used 10% O2 as an average of venous and arterial blood, as described previously [20,21]. Moreover, intense blast proliferation and O2 consumption in leukemic-PB might affect the biology of leukemic blasts [21,45] compared to standard normoxic condition. In hypoxic conditions, the expression of membrane receptors (e.g., CXCR4) and the activation of intracellular signaling cascades of pO2-sensitive tumor suppressor genes (e.g., MAPK, HIF1a) change [40]. Physiological hypoxia was shown to induce cell cycle arrest in the G0/G1-phase of AML blasts (cell lines and primary AML samples) by increasing the expression of the anti-apoptotic XIAP and activation of PI3K/Akt [46].
4.3. Generation of DC/DCleu from Leukemic Cell Lines
We utilized AML cell lines in the initial phase to assess the impact of hypoxia, as previous studies have demonstrated that AML cell lines serve as valuable tools for investigating the effects of hypoxia on leukemic cell proliferation [47]. Our findings indicate that DCs can be successfully generated using DC/DCleu-generating protocols, irrespective of the type and the mutation status of the cell lines, under both hypoxic and normoxic conditions. Interestingly, our results revealed no significant decrease in the frequencies of generated DC/DCleu and no discernible differences in the blast proliferation between the two conditions. The adaptation to low oxygen levels during DC/DCleu-cultures can be attributed to the activity of HIF-1α [45]. Notably, we observed higher frequencies of cells in the S-phase under normoxic conditions compared to hypoxic conditions in our experiments which might result in a lower cell proliferation activity in hypoxic compared to normoxic conditions. In summary, our study confirmed that DC/DCleu-generation from leukemic cell lines is feasible under hypoxic conditions, yielding comparable outcomes to those obtained under normoxic conditions.
4.4. DC/DCleu-Generation from Healthy and AML PBMNCs and WB
The generation of DC/DCleu using DC/DCleu-generating protocols from healthy and AML PBMNCs with Pici-PGE1 and Pici-PGE2 was successful compared to controls. These data confirm previous findings described by others and us [6,9,27,30]. Interestingly, all results obtained were similar under both hypoxic and normoxic conditions. These results demonstrate that hypoxia, as a physiological condition, is not necessary for the ex vivo generation of DC/DCleu for later on adoptive cell transfer.
Whole blood contains all soluble and cellular factors present in the individual AML patient, which may have activating or inhibitory influences on physiological immune reactions [19]. Therefore, we generated DC/DCleu directly from healthy and leukemic WB to simulate most physiological conditions. DC/DCleu generation was feasible under both hypoxic and normoxic conditions and the DCleu subgroups did not significantly differ between the two conditions. Importantly, the used Kits did not induce blasts’ proliferation during cultures, thereby conforming previous findings. In the clinical context, these results support the idea that AML patients could be treated directly with Kits inducing DC/DCleu-generation in vivo [48,49].
4.5. DC/DCleu-Stimulation in T Cell Enriched MLC Results in Activated Cells of the Innate and Adaptive Immune System
T, iNKT, NK and CIK cells and their subsets are important mediators of innate and adaptive immune responses and their anti-tumor and anti-infections functionality are known to be activated by DC/DCleu [15]. This was already confirmed previously under normoxic conditions using DC/DCleu generated with Kits in T cell enriched MLC [9,14,15,30,50]. We can add that this activation was seen under hypoxic as well as normoxic conditions and resulted in comparable frequencies of different immune cell subsets. Memory T cells play a critical role in the maintenance of the complete remission of AML patients if activated in vivo [51,52]. Furthermore, cells of the innate immunity significantly increased after MLC under hypoxic and normoxic conditions.
4.6. DC/DCleu Stimulation after T Cell Enriched MLC Results in Improved Anti-Leukemic Activity
As already shown before treatment of WB with Kits, followed by T cell enriched MLC improved anti-leukemic reactivity under normoxic conditions [25,50]. Here we show that anti-leukemic activity was improved under hypoxic vs normoxic conditions. These results might point to different killing mechanisms under hypoxic vs normoxic conditions or at least a variation of this process (e.g., slow pathway of Fas/FasL- or the fast pathway of perforin-granzyme-mediated killing). These effects might act synergistically or independently [53,54]. Additionally, the superior anti-leukemic activity observed after MLCWB-DC Kit I and MLCWB-DC Kit M appeared to be equally effective under normoxic conditions. However, a significantly higher number of cases with increased lysis after MLCWB-DC Kit M compared to controls (MLCWB) was found under hypoxic versus normoxic conditions. These results suggest that hypoxia, as a physiological condition may enhance the anti-leukemic activity.
4.7. Correlation of Anti-Leukemic Cytotoxicity of Immunoreactive Cells Stimulated by DC/DCleu
We investigated the correlation between the anti-leukemic reactivity of effector cells (T cell-enriched WB treated with or without Kits) and the frequencies of various DC and immune cell subtypes. In normoxic conditions, a significant positive correlation was found between DC+/WB generated with Kit M and the highest anti-leukemic activity after 3 or 24 h of treatment with MLCWB-DC Kit M. Under hypoxic conditions, a significant correlation was observed between DC+/WB and DCleu/WB generated with Kit M and the most effective anti-leukemic reactivity of T effector cells after MLCWB-DC Kit M after 3 or 24 h. Other cell subtypes did not show a direct correlation with achieved cytotoxicity. These findings support previous studies indicating a relationship between DC stimulation and the gained cytotoxicity of stimulated T cells against leukemia [55]. Moreover, the better anti-leukemic reactivity of MLCWB-DC Kit M compared to MLCWB could be a good explanation for the positive correlation between (DCleu/WB) generated with Kit M and the best anti-leukemic activity of T cells (MLCWB-DC Kit M) in hypoxic conditions.
5. Conclusions
DC/DCleu-generation with Kit I and Kit M was shown to increase DC/DCleu frequencies and to activate different immune cells after T cell enriched MLC under hypoxic and normoxic conditions in comparable frequencies using AML cell lines, PBMNCs and WB from leukemic and healthy samples. Induced anti-leukemic reactions were shown to be superior under hypoxic vs normoxic conditions. In summary, we show that immunoreactions induced by DC/DCleu might be underestimated under normoxic conditions—pointing to improved effects of DC/DCleu-immunotherapies in vivo in the hypoxic niches of the body. However, additional studies are needed to evaluate the impact of hypoxic conditions on the immune system and the generation of DC/DCleu in vivo.
Author Contributions
F.D.-G. conducted cell-culture experiments under hypoxic conditions for cell lines, PBMNCs and W.B. experiments, analysed the corresponding data, conducted cell-line-culture experiments also under normoxic conditions. D.C.A. conducted cell-culture experiments for PBMNCs and W.B. under normoxic conditions and rafted this manuscript together with H.M.S., C.A., M.W., C.S. (Christoph Schwepcke), L.K., O.S. supported F.D.-G. with DC/DCleu-culture-, MLC- and CTX-experiments under normoxic and hypoxic conditions. H.H. drafted the figures used in this manuscript. D.K., A.R., C.S. (Christoph Schmid), provided patient samples. H.M.S. designed the study and was responsible for the funding. All authors have read and agreed to the published version of the manuscript.
Funding
The project was supported by intramural funding from the working group of H.M.S. F.D.-G. was funded by grants from the DAAD (ID 21520154 and scholarship 2016–2017 of the Ludwig Maximilian University of Munich). The funders did not influence the study design, data collection or analysis, the publishing decision or the manuscript preparation.
Institutional Review Board Statement
Samples were collected and patients’ informed consent gathered according to the Helsinki guidelines and the vote of the Ethics Committee of the Ludwig Maximilian University of Munich (vote number: 339-05).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data published in this study is openly available in a public repository with a permanent identifier, such as a DOI.
Acknowledgments
The authors thank patients, nurses, and physicians on the wards for their support and the diagnostic laboratories as well as the treating institutions for the patients’ diagnostic reports.
Conflicts of Interest
HM-S. is involved with Modiblast Pharma GmbH (Oberhaching, Germany), which holds the European Patent 15 801 987.7-1118 and US Patent 15-517627, ‘Use of immunomodulatory effective compositions for the immunotherapeutic treatment of patients suffering from myeloid leukemias’. For all other authors, there are no conflicts of interest to declare.
References
- Döhner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef] [PubMed]
- Heuser, M.; Ofran, Y.; Boissel, N.; Brunet Mauri, S.; Craddock, C.; Janssen, J.; Wierzbowska, A.; Buske, C. Acute myeloid leukaemia in adult patients: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 697–712. [Google Scholar] [CrossRef]
- Yuan, C.; Song, G.; Jiang, G. The characterization and role of leukemia cell-derived dendritic cells in immunotherapy for leukemic diseases. Intractable Rare Dis. Res. 2012, 1, 53–65. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Van Acker, H.H.; Versteven, M.; Lichtenegger, F.S.; Roex, G.; Campillo-Davo, D.; Lion, E.; Subklewe, M.; Van Tendeloo, V.F.; Berneman, Z.N.; Anguille, S. Dendritic Cell-Based Immunotherapy of Acute Myeloid Leukemia. J. Clin. Med. 2019, 8, 579. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, S.; Robbiani, D.F.; Du, X.; Rodrigues, E.; Ignatius, R.; Wei, Y.; Ponath, P.; Young, J.W.; Pope, M.; Steinman, R.M.; et al. Expression of a functional eotaxin (CC chemokine ligand 11) receptor CCR3 by human dendritic cells. J. Immunol. 2002, 169, 2925–2936. [Google Scholar] [CrossRef] [PubMed]
- Amberger, D.C.; Doraneh-Gard, F.; Gunsilius, C.; Weinmann, M.; Mobius, S.; Kugler, C.; Rogers, N.; Bock, C.; Kodel, U.; Werner, J.O.; et al. PGE1-Containing Protocols Generate Mature (Leukemia-Derived) Dendritic Cells Directly from Leukemic Whole Blood. Int. J. Mol. Sci. 2019, 20, 4590. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.; Dupasquier, M. Dendritic cells in vivo and in vitro. Cell Mol. Immunol. 2005, 2, 28–35. [Google Scholar] [PubMed]
- Schmetzer, H.M.; Kremser, A.; Loibl, J.; Kroell, T.; Kolb, H.J. Quantification of ex vivo generated dendritic cells (DC) and leukemia-derived DC contributes to estimate the quality of DC, to detect optimal DC-generating methods or to optimize DC-mediated T-cell-activation-procedures ex vivo or in vivo. Leukemia 2007, 21, 1338–1341. [Google Scholar] [CrossRef] [PubMed]
- Kremser, A.; Dressig, J.; Grabrucker, C.; Liepert, A.; Kroell, T.; Scholl, N.; Schmid, C.; Tischer, J.; Kufner, S.; Salih, H.; et al. Dendritic cells (DCs) can be successfully generated from leukemic blasts in individual patients with AML or MDS: An evaluation of different methods. J. Immunother. 2010, 33, 185–199. [Google Scholar] [CrossRef]
- Johnson, L.A.; Jackson, D.G. Control of dendritic cell trafficking in lymphatics by chemokines. Angiogenesis 2014, 17, 335–345. [Google Scholar] [CrossRef]
- Lanzavecchia, A.; Sallusto, F. Regulation of T cell immunity by dendritic cells. Cell 2001, 106, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Grakoui, A.; Bromley, S.K.; Sumen, C.; Davis, M.M.; Shaw, A.S.; Allen, P.M.; Dustin, M.L. Pillars article: The immunological synapse: A molecular machine controlling T cell activation. Science. 1999, 285: 221–227. J. Immunol. 2015, 194, 4066–4072. [Google Scholar] [PubMed]
- Golubovskaya, V.; Wu, L. Different Subsets of T Cells, Memory, Effector Functions, and CAR-T Immunotherapy. Cancers 2016, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Schick, J.; Vogt, V.; Zerwes, M.; Kroell, T.; Kraemer, D.; Kohne, C.H.; Hausmann, A.; Buhmann, R.; Tischer, J.; Schmetzer, H. Antileukemic T-cell responses can be predicted by the composition of specific regulatory T-cell subpopulations. J. Immunother. 2013, 36, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Vogt, V.; Schick, J.; Ansprenger, C.; Braeu, M.; Kroell, T.; Kraemer, D.; Kohne, C.H.; Hausmann, A.; Buhmann, R.; Tischer, J.; et al. Profiles of activation, differentiation-markers, or beta-integrins on T cells contribute to predict T cells’ antileukemic responses after stimulation with leukemia-derived dendritic cells. J. Immunother. 2014, 37, 331–347. [Google Scholar] [CrossRef] [PubMed]
- Pepeldjiyska, E.; Li, L.; Gao, J.; Seidel, C.L.; Blasi, C.; Özkaya, E.; Schmohl, J.; Kraemer, D.; Schmid, C.; Rank, A.; et al. Leukemia derived dendritic cell (DC(leu)) mediated immune response goes along with reduced (leukemia-specific) regulatory T-cells. Immunobiology 2022, 227, 152237. [Google Scholar] [CrossRef] [PubMed]
- Robertson, F.C.; Berzofsky, J.A.; Terabe, M. NKT cell networks in the regulation of tumor immunity. Front. Immunol. 2014, 5, 543. [Google Scholar] [CrossRef] [PubMed]
- Montoya, C.J.; Pollard, D.; Martinson, J.; Kumari, K.; Wasserfall, C.; Mulder, C.B.; Rugeles, M.T.; Atkinson, M.A.; Landay, A.L.; Wilson, S.B. Characterization of human invariant natural killer T subsets in health and disease using a novel invariant natural killer T cell-clonotypic monoclonal antibody, 6B11. Immunology 2007, 122, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Boeck, C.L.; Amberger, D.C.; Doraneh-Gard, F.; Sutanto, W.; Guenther, T.; Schmohl, J.; Schuster, F.; Salih, H.; Babor, F.; Borkhardt, A.; et al. Significance of Frequencies, Compositions, and/or Antileukemic Activity of (DC-stimulated) Invariant NKT, NK and CIK Cells on the Outcome of Patients With AML, ALL and CLL. J. Immunother. 2017, 40, 224–248. [Google Scholar] [CrossRef]
- Marenzana, M.; Arnett, T.R. The Key Role of the Blood Supply to Bone. Bone Res. 2013, 1, 203–215. [Google Scholar] [CrossRef]
- Deynoux, M.; Sunter, N.; Herault, O.; Mazurier, F. Hypoxia and Hypoxia-Inducible Factors in Leukemias. Front. Oncol. 2016, 6, 41. [Google Scholar] [CrossRef] [PubMed]
- Rieger, C.T.; Fiegl, M. Microenvironmental oxygen partial pressure in acute myeloid leukemia: Is there really a role for hypoxia? Exp. Hematol. 2016, 44, 578–582. [Google Scholar] [CrossRef]
- Vasold, J.; Wagner, M.; Drolle, H.; Deniffel, C.; Kutt, A.; Oostendorp, R.; Sironi, S.; Rieger, C.; Fiegl, M. The bone marrow microenvironment is a critical player in the NK cell response against acute myeloid leukaemia in vitro. Leuk. Res. 2015, 39, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Sironi, S.; Wagner, M.; Kuett, A.; Drolle, H.; Polzer, H.; Spiekermann, K.; Rieger, C.; Fiegl, M. Microenvironmental hypoxia regulates FLT3 expression and biology in AML. Sci. Rep. 2015, 5, 17550. [Google Scholar] [CrossRef]
- Schwepcke, C.; Klauer, L.K.; Deen, D.; Amberger, D.C.; Fischer, Z.; Doraneh-Gard, F.; Gunsilius, C.; Hirn-Lopez, A.; Kroell, T.; Tischer, J.; et al. Generation of Leukaemia-Derived Dendritic Cells (DC(leu)) to Improve Anti-Leukaemic Activity in AML: Selection of the Most Efficient Response Modifier Combinations. Int. J. Mol. Sci. 2022, 23, 8333. [Google Scholar] [CrossRef]
- Hirn Lopez, A.; Deen, D.; Fischer, Z.; Rabe, A.; Ansprenger, C.; Stein, K.; Vogt, V.; Schick, J.; Kroell, T.; Kraemer, D.; et al. Role of Interferon (IFN)α in “Cocktails” for the Generation of (Leukemia-derived) Dendritic Cells (DCleu) From Blasts in Blood From Patients (pts) With Acute Myeloid Leukemia (AML) and the Induction of Antileukemic Reactions. J. Immunother. 2019, 42, 143–161. [Google Scholar] [CrossRef]
- Sato, M.; Takayama, T.; Tanaka, H.; Konishi, J.; Suzuki, T.; Kaiga, T.; Tahara, H. Generation of mature dendritic cells fully capable of T helper type 1 polarization using OK-432 combined with prostaglandin E(2). Cancer Sci. 2003, 94, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Klauer, L.K.; Schutti, O.; Ugur, S.; Doraneh-Gard, F.; Amberger, D.C.; Rogers, N.; Krämer, D.; Rank, A.; Schmid, C.; Eiz-Vesper, B.; et al. Interferon Gamma Secretion of Adaptive and Innate Immune Cells as a Parameter to Describe Leukaemia-Derived Dendritic-Cell-Mediated Immune Responses in Acute Myeloid Leukaemia in vitro. Transfus. Med. Hemother. 2022, 49, 44–61. [Google Scholar] [CrossRef]
- Plett, C.; Klauer, L.K.; Amberger, D.C.; Ugur, S.; Rabe, A.; Fischer, Z.; Deen, D.; Hirn-Lopez, A.; Gunsilius, C.; Werner, J.O.; et al. Immunomodulatory kits generating leukaemia derived dendritic cells do not induce blast proliferation ex vivo: IPO-38 as a novel marker to quantify proliferating blasts in acute myeloid leukaemia. Clin. Immunol. 2022, 242, 109083. [Google Scholar] [CrossRef]
- Grabrucker, C.; Liepert, A.; Dreyig, J.; Kremser, A.; Kroell, T.; Freudenreich, M.; Schmid, C.; Schweiger, C.; Tischer, J.; Kolb, H.J.; et al. The quality and quantity of leukemia-derived dendritic cells from patients with acute myeloid leukemia and myelodysplastic syndrome are a predictive factor for the lytic potential of dendritic cells-primed leukemia-specific T cells. J. Immunother. 2010, 33, 523–537. [Google Scholar] [CrossRef]
- Kim, K.H.; Lee, J.M.; Zhou, Y.; Harpavat, S.; Moore, D.D. Glucocorticoids Have Opposing Effects on Liver Fibrosis in Hepatic Stellate and Immune Cells. Mol. Endocrinol. 2016, 30, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, Y.Q.; Jin, G.; Wu, S.; Cui, J.; Wang, R.F. Selection of reference genes for gene expression studies in human bladder cancer using SYBR-Green quantitative polymerase chain reaction. Oncol. Lett. 2017, 14, 6001–6011. [Google Scholar] [CrossRef] [PubMed]
- Cassier, P.A.; Castets, M.; Belhabri, A.; Vey, N. Targeting apoptosis in acute myeloid leukaemia. Br. J. Cancer 2017, 117, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Geiger, T.L.; Rubnitz, J.E. New approaches for the immunotherapy of acute myeloid leukemia. Discov. Med. 2015, 19, 275–284. [Google Scholar] [PubMed]
- Subklewe, M.; von Bergwelt-Baildon, M.; Humpe, A. Chimeric Antigen Receptor T Cells: A Race to Revolutionize Cancer Therapy. Transfus. Med. Hemother. 2019, 46, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Platt, A.M.; Randolph, G.J. Dendritic cell migration through the lymphatic vasculature to lymph nodes. Adv. Immunol. 2013, 120, 51–68. [Google Scholar] [CrossRef] [PubMed]
- Van Tendeloo, V.F.; Van de Velde, A.; Van Driessche, A.; Cools, N.; Anguille, S.; Ladell, K.; Gostick, E.; Vermeulen, K.; Pieters, K.; Nijs, G.; et al. Induction of complete and molecular remissions in acute myeloid leukemia by Wilms’ tumor 1 antigen-targeted dendritic cell vaccination. Proc. Natl. Acad. Sci. USA 2010, 107, 13824–13829. [Google Scholar] [CrossRef] [PubMed]
- Rosenblatt, J.; Stone, R.M.; Uhl, L.; Neuberg, D.; Joyce, R.; Levine, J.D.; Arnason, J.; McMasters, M.; Luptakova, K.; Jain, S.; et al. Individualized vaccination of AML patients in remission is associated with induction of antileukemia immunity and prolonged remissions. Sci. Trans. Med. 2016, 8, 368ra171. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, M.; Rosenblatt, J.; Avigan, D. Dendritic Cell Therapies for Hematologic Malignancies. Mol. Ther. Methods Clin. Dev. 2017, 5, 66–75. [Google Scholar] [CrossRef]
- Fiegl, M.; Samudio, I.; Clise-Dwyer, K.; Burks, J.K.; Mnjoyan, Z.; Andreeff, M. CXCR4 expression and biologic activity in acute myeloid leukemia are dependent on oxygen partial pressure. Blood 2009, 113, 1504–1512. [Google Scholar] [CrossRef]
- Sitkovsky, M.V.; Hatfield, S.; Abbott, R.; Belikoff, B.; Lukashev, D.; Ohta, A. Hostile, hypoxia-A2-adenosinergic tumor biology as the next barrier to overcome for tumor immunologists. Cancer Immunol. Res. 2014, 2, 598–605. [Google Scholar] [CrossRef]
- Lee, C.T.; Mace, T.; Repasky, E.A. Hypoxia-driven immunosuppression: A new reason to use thermal therapy in the treatment of cancer? Int. J. Hyperth. 2010, 26, 232–246. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, H.; Harris, L.J.; Zhang, P.; McIlhenny, S.; Srinivas, V.; Tulenko, T.; DiMuzio, P.J. The role of hypoxia in stem cell differentiation and therapeutics. J. Surg. Res. 2011, 165, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Carreau, A.; El Hafny-Rahbi, B.; Matejuk, A.; Grillon, C.; Kieda, C. Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J. Cell Mol. Med. 2011, 15, 1239–1253. [Google Scholar] [CrossRef] [PubMed]
- Drolle, H.; Wagner, M.; Vasold, J.; Kutt, A.; Deniffel, C.; Sotlar, K.; Sironi, S.; Herold, T.; Rieger, C.; Fiegl, M. Hypoxia regulates proliferation of acute myeloid leukemia and sensitivity against chemotherapy. Leuk. Res. 2015, 39, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Baharaghdam, S.; Yousefi, M.; Movasaghpour, A.; Solali, S.; Talebi, M.; Ahani-Nahayati, M.; Lotfimehr, H.; Shamsasanjan, K. Effects of Hypoxia on Biology of Human Leukemia T-cell Line (MOLT-4 cells) Co-cultured with Bone Marrow Mesenchymal Stem Cells. Avicenna J. Med. Biotechnol. 2018, 10, 62–68. [Google Scholar] [PubMed]
- Goto, M.; Miwa, H.; Suganuma, K.; Tsunekawa-Imai, N.; Shikami, M.; Mizutani, M.; Mizuno, S.; Hanamura, I.; Nitta, M. Adaptation of leukemia cells to hypoxic condition through switching the energy metabolism or avoiding the oxidative stress. BMC Cancer 2014, 14, 76. [Google Scholar] [CrossRef] [PubMed]
- Köhler, T.; Reizis, B.; Johnson, R.S.; Weighardt, H.; Förster, I. Influence of hypoxia-inducible factor 1α on dendritic cell differentiation and migration. Eur. J. Immunol. 2012, 42, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Rein, L.A.; Chao, N.J. WT1 vaccination in acute myeloid leukemia: New methods of implementing adoptive immunotherapy. Expert Opin. Investig. Drugs 2014, 23, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Unterfrauner, M.; Rejeski, H.A.; Hartz, A.; Bohlscheid, S.; Baudrexler, T.; Feng, X.; Rackl, E.; Li, L.; Rank, A.; Filippini Velázquez, G.; et al. Granulocyte-Macrophage-Colony-Stimulating-Factor Combined with Prostaglandin E1 Create Dendritic Cells of Leukemic Origin from AML Patients’ Whole Blood and Whole Bone Marrow That Mediate Antileukemic Processes after Mixed Lymphocyte Culture. Int. J. Mol. Sci. 2023, 24, 7436. [Google Scholar] [CrossRef]
- Sallusto, F.; Lenig, D.; Förster, R.; Lipp, M.; Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999, 401, 708–712. [Google Scholar] [CrossRef] [PubMed]
- Facciabene, A.; Motz, G.T.; Coukos, G. T-regulatory cells: Key players in tumor immune escape and angiogenesis. Cancer Res. 2012, 72, 2162–2171. [Google Scholar] [CrossRef] [PubMed]
- Hassin, D.; Garber, O.G.; Meiraz, A.; Schiffenbauer, Y.S.; Berke, G. Cytotoxic T lymphocyte perforin and Fas ligand working in concert even when Fas ligand lytic action is still not detectable. Immunology 2011, 133, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Khatri, M.; Murgia, M.V.; Saif, Y.M. Fas/FasL and perforin-granzyme pathways mediated T cell cytotoxic responses in infectious bursal disease virus infected chickens. Results Immunol. 2012, 2, 112–119. [Google Scholar] [CrossRef]
- Bakdash, G.; Schreurs, I.; Schreibelt, G.; Tel, J. Crosstalk between dendritic cell subsets and implications for dendritic cell-based anticancer immunotherapy. Expert. Rev. Clin. Immunol. 2014, 10, 915–926. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).