Simple Summary
Neurologic complications following a hematopoietic stem cell transplantation (HSCT) can be caused by various etiologies and significantly contribute to morbidity and mortality. The aim of our retrospective study was to determine the prognostic indicators for HSCT-associated neurological complications in pediatric HSCT recipients using their clinical characteristics and brain magnetic resonance imaging (MRI) lesions. The demographics, received treatments, treatment-related morbidities, laboratory findings and brain MRI findings were reviewed and compared among 51 patients who had underwent a brain MRI due to newly developed neurological symptoms or infection signs during HSCT and follow-up period. Children with neurologic complications associated with infectious causes, malignant disease or severe brain MRI abnormalities were more likely to have poor outcome.
Abstract
Purpose: To determine the prognostic indicators for hematopoietic stem cell transplantation (HSCT)-associated neurological complications, the clinical characteristics and brain magnetic resonance imaging (MRI) lesions in pediatric HSCT recipients were reviewed. Methods: This retrospective study included 51 patients who had underwent a brain MRI due to newly developed neurological symptoms or infection signs during chemotherapy or HSCT. We reviewed the demographics, received treatments, treatment-related morbidities, laboratory findings and brain MRI findings, which were compared between good and poor neurologic outcome groups. Results: Thirty-seven patients (72.5%) fully recovered from the neurologic deficits and fourteen (27.5%) persisted or aggravated. The children with an underlying malignant disease had significantly poorer neurological outcomes (p = 0.015). The neurologic complications associated with infection were more frequent in the poor outcome group (p = 0.038). In the neuroimaging findings, the extent of the white matter lesions was significantly higher in the poor outcome group, as was that of abnormal enhancement, ventriculomegaly, cortical change, deep gray matter abnormalities and cerebellar abnormalities. Conclusion: Most children with neurologic complications and neuroimaging abnormalities during HSCT had recovered. However, children with neurologic complications associated with infectious causes, malignant disease or severe brain MRI abnormalities should be more carefully monitored during HSCT.
1. Introduction
Advances in hematopoietic stem cell transplantation (HSCT), which is widely used to treat malignant and nonmalignant hematologic disorders, genetic disorders, inborn errors of metabolism and autoimmune disorders, have resulted in increased survival rates [1,2,3,4]. Central nervous system (CNS) complications of HSCT are a significant cause of morbidity and significantly contribute to mortality. Approximately 11–59% of patients who undergo HSCT suffer from neurologic complications [5] and more than 90% of the cases who die after HSCT show neuropathological abnormalities in autopsy studies [6]. Various factors, including the pre-transplant conditioning regimen with high-dose chemotherapy, radiation therapy, immunosuppressive therapy, graft versus host disease (GvHD), infection and disease recurrences, are associated with neurotoxicity [7]. Although the neurotoxic mechanisms underlying each cause remain unknown, there are various theories regarding the mechanisms by which chemotherapy and radiotherapy produce damage to the central nervous system which include vascular injury causing chronic ischemia, progressive demyelination of the white matter (WM) and necrosis, oxidative stress and DNA damage, immune dysregulation and stimulation of neurotoxic cytokines [8,9,10].
In addition to the early diagnosis and accurate management of neurologic complications, a prompt assessment of prognostic indicators is also important to reduce the risk of neurologic complications by developing an appropriate treatment plan that will lead to a good outcome. Neuroimaging is a crucial tool in cases with neurologic complications for the early detection of brain lesions, monitoring of disease progression and assessing treatment responses. Additionally, a recent study has found that patients showing structural abnormalities on brain imaging tended to have a higher mortality rate [11]. This result indicates the potential role of neuroimaging as a prognostic modality in patients with neurologic complications.
In our present study, we hypothesized that the various clinical variables and abnormalities that can be detected by neuroimaging would be useful prognostic indicators of the neurologic outcomes in HSCT candidates with neurologic complications. We therefore retrospectively reviewed the clinical data, neuroimaging findings and associated neurologic symptoms among the pediatric patients who underwent HSCT at our hospital and investigated the most optimal prognostic indicators of the neurologic outcomes in these cases.
2. Materials and Methods
2.1. Patient Population and Data Collection
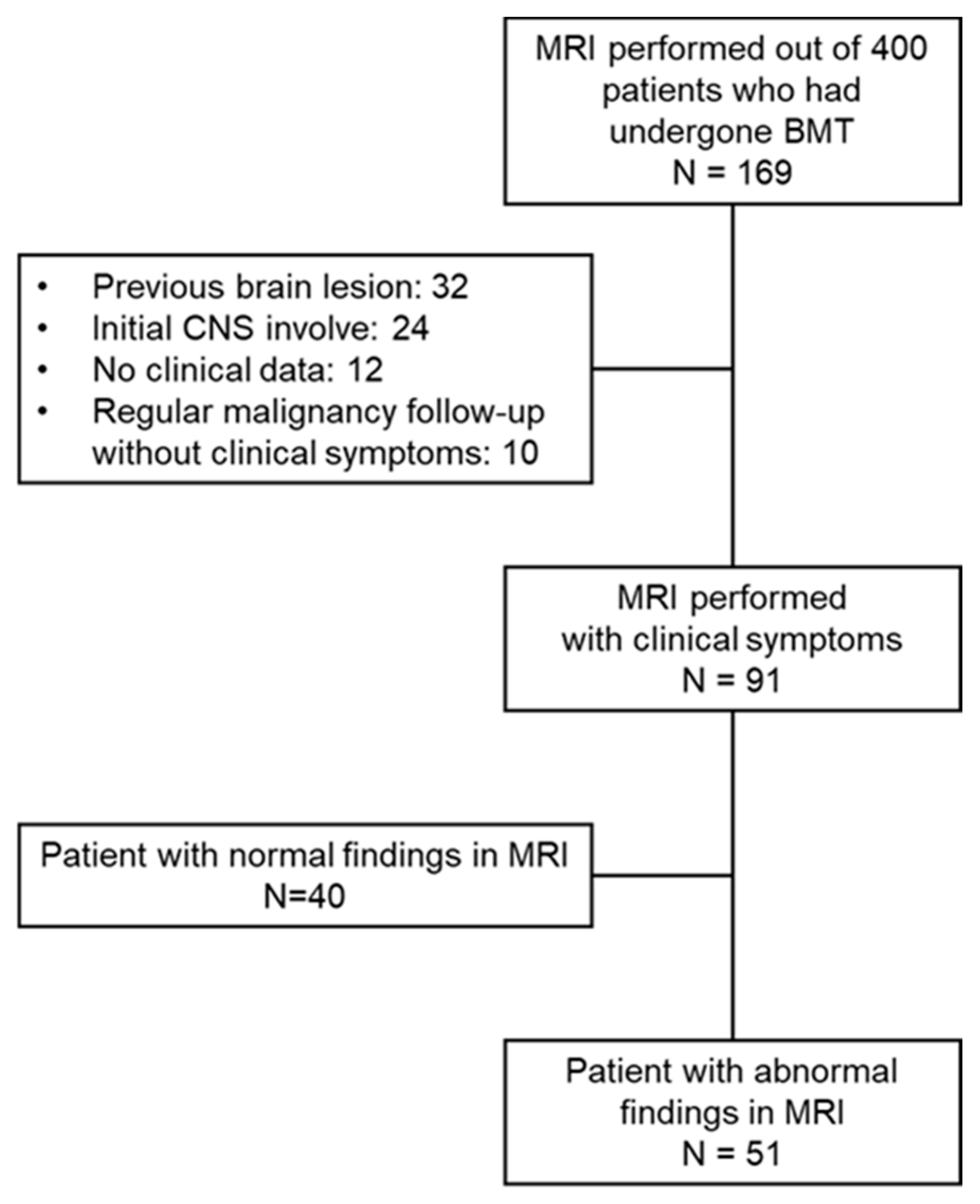
From January 2010 to March 2020, 169 children received HSCT and also underwent a brain MRI at the Department of Pediatrics at Asan Medical Center, Seoul, Korea. Among these cases, except 78 patients with previous brain lesion, tumor involvement of CNS and insufficient medical records, 91 patients that had undergone a brain MRI and had newly developed neurological symptoms after the diagnosis of the causative disease were enrolled in this study (Figure 1). We retrospectively reviewed the clinical and laboratory data for these cases and collected the following information: (1) demographics (sex, age at diagnosis, underlying disease, type of transplantation); (2) pre-transplant treatment regimen (systemic and intrathecal chemotherapy, radiation therapy); (3) transplant-associated treatments or complications (immunosuppressant use, GvHD, infection); and (4) the clinical and laboratory characteristics of any neurologic complications.
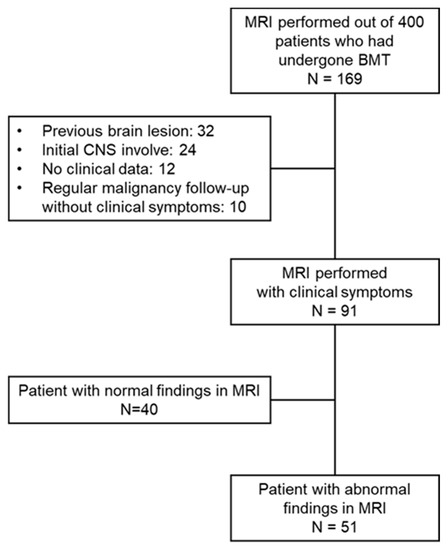
Figure 1.
Study scheme.
The chemotherapy, radiotherapy and immunosuppressant regimens which were administered before the onset of neurologic symptoms were investigated. Patients who received a second HSCT were analyzed based on the transplant procedure closest to the onset of neurological symptoms. Electroencephalogram (EEG) results reported by pediatric neurologists were also reviewed. CSF analysis findings were additionally investigated and any abnormal CSF findings were defined as CSF pleocytosis or positive CSF culture and viral PCR and/or the presence of CSF blasts.
2.2. Neurological Outcome Assessments
To identify effective prognostic indicators of neurologic outcomes in our current series, the patients were classified into good or poor outcome groups. The patients in the good outcome group showed a disappearance of symptoms with or without improvement in their abnormal findings on brain MRI. Patients with a poor outcome was defined as cases with the persistent neurologic sequelae including a bedridden state or death.
2.3. Classification and Scoring of the Neuroimaging Data
The brain MRI findings for our study cohort reported by pediatric neuroradiologists were reviewed. An abnormal lesion was classified as frontal, temporal, corpus callosum, inferomedial, or parieto-occipital and further separated into 14 subdivisions according to the location of the white matter (WM) injury. The number of these WM subdivisions with T2 fluid attenuated inversion recovery (FLAIR) high signal intensities was counted. In addition to the extent of the WM lesions, unidentified scattered spots, abnormal enhancements, ventriculomegaly, cortical changes, deep gray matter abnormalities and cerebellar abnormalities in the T2 FLAIR, or gadolinium enhancement, were regarded as additional findings and investigated. (Table 1) The extent of the WM lesions and existence of other MRI findings were compared between the two neurologic outcome groups.

Table 1.
Division of white matter lesions on brain MRI findings.
2.4. Statistical Analysis
SPSS version 24 (SPSS, Chicago, IL, USA) was used for all statistical analysis of the data. A chi-square test and Fisher’s exact test were used for the univariate analysis of prognostic factors of neurological symptoms, with continuous variables compared using the Student’s t test. Logistic regression analysis was used to evaluate the association between patient outcomes and neuroimaging findings. p- values of less than 0.05 were considered to indicate statistical significance.
3. Results
Among the 91 pediatric patients in our present cohort who underwent neuroimaging testing due to clinical symptoms, 51 patients (51/91, 56.0%) showed abnormal findings. The baseline characteristics, treatments and clinical and laboratory findings of neurological complications for the 51 study patients are listed in Table 2.

Table 2.
Baseline characteristics of the study cohort and received treatments.
3.1. Baseline Demographics and Treatments of the Study Cohort
Twenty-seven of the children in our study series were male (52.9%) and the mean age at diagnosis of the underlying disease was 7.7 years. The underlying diseases in this pediatric cohort included malignancy in 39 patients (76.5%) and non-malignant disease in 12 cases (23.5%). In terms of the transplantation types, 23 children (45.1%) received haplotype HSCT, 20 (39.2%) received allogenic HSCT from an unrelated donor and 8 (15.7%) received allogenic HSCT from a related donor. Abnormal neuroimaging findings were evident in 19 patients (37.3%) prior to HSCT and in the remaining 32 cases (62.7%) after HSCT.
Cyclophosphamide (66.7%), cytarabine (64.7%), etoposide (52.9%) and vincristine (51.0%) were the commonly administered neurotoxic chemotherapeutics among the children with abnormal imaging findings and 36 of these children (70.6%) received intrathecal chemotherapy with cytarabine, methotrexate and hydrocortisone. Cranial irradiation was done in 22 patients (43.1%) prior to symptom onset and almost 50% of the children received immunosuppressants including cyclosporine, tacrolimus (FK506), or mycophenolate mofetil (MMF).
3.2. Clinical and Laboratory Characteristics Associated with Neurologic Complications
The mean age of onset of the neurological or infectious symptoms and signs in our 51 pediatric cases with abnormal findings was 10.2 years. Among the 32 patients with abnormal brain MRI findings after HSCT, the median duration from transplantation to symptom onset was 178.5 days, but this onset occurred within 100 days after HSCT in almost 50% of the patients. Motor symptoms most commonly presented, arising in 20 patients (39.2%), followed by decreased consciousness (12 patients; 23.5%) and headache (10 patients;19.6%). Five patients (9.8%) underwent a brain MRI due to infection signs without neurological symptoms.
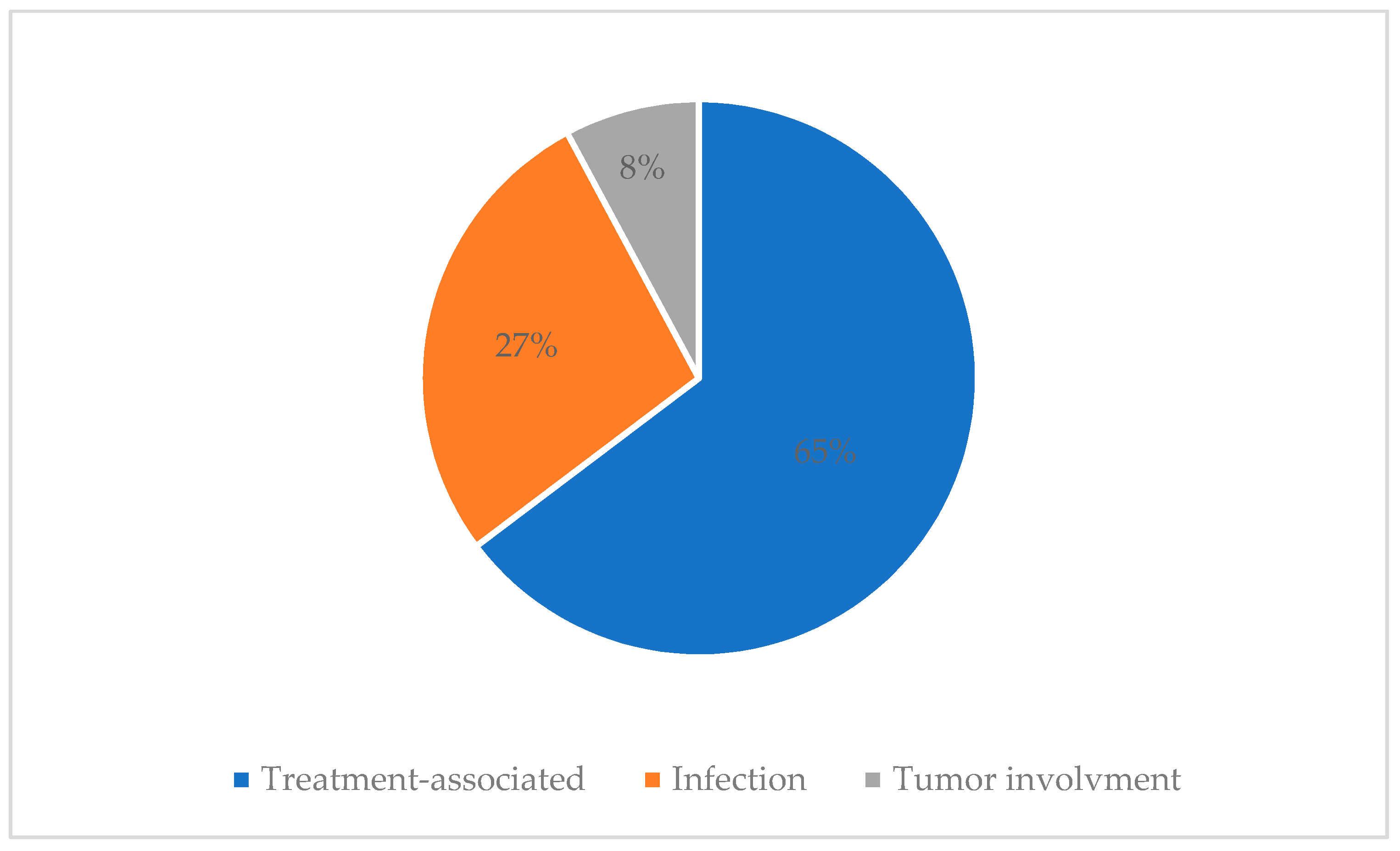
An electroencephalogram (EEG) was performed in 21 children, of which 4 (4/21, 19%) had normal EEG findings, 17 (17/21, 81%) had EEG abnormalities; 6 had focal epileptiform discharge and 11 had cerebral dysfunction. There were 28 patients who underwent a CSF test, of which 14 (14/28, 50%) showed normal CSF findings, 12 (12/28, 42.9%) had CSF abnormalities and 2 (2/28, 7.1%) had newly identified CSF blasts. Details of CSF abnormalities are described in Appendix C. Treatment associated neurological complications were the most common cause of neurologic complications (33 patients; 64.7%), followed by infection (14 patients; 27.5%) and newly involved malignancy (4 patients; 7.8%) (Figure 2).
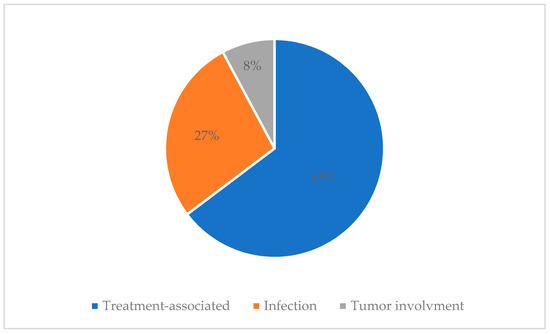
Figure 2.
Causes of neurological complications.
3.3. Neurologic Outcome Assessments
We compared the clinical and laboratory findings between the 37 children (72.5%) with a good outcome and 14 (27.5%) with a poor outcome (Table 3), among the patients showing abnormal findings. Baseline demographics including sex, median age at diagnosis and type of transplantation, were not significantly different between these two groups. On the other hand, a malignancy was significantly associated with a poor outcome (25/37, 67.5% vs. 14/14, 100%; p = 0.015). In terms of the factors associated with the treatments received by the children, there was no statistically significant association found between systemic and intrathecal chemotherapy and outcomes. Cranial radiation was also not significantly associated with a poor outcome (13/37, 35.1% vs. 9/14 64.2%; p = 0.061).

Table 3.
Neurological outcome assessments in patients with MRI abnormalities.
Among the initial presenting neurological symptoms in our cohort, the children with a decreased consciousness were more likely to have a poor outcome, with statistical significance (6/37, 16.2% vs. 6/14, 42.9%; p = 0.045). There was no difference in the EEG and CSF abnormalities between the good and poor outcome groups. Infection was found to be a more frequent cause of neurological symptoms in the poor outcome group (7/37, 18.9% vs. 7/14, 50%; p = 0.038). The causative microorganism was identified in eight of 14 patients whose neurological symptoms and brain MRI abnormalities were due to an infection, i.e., 3 bacterial infections (L. monocytogenes, one case; S. viridians group, two cases), five viral infections (CMV, three cases; adenovirus, one case; BK virus, one case) and an unidentified pathogen in six patients with clinical CNS infections, including three with a brain abscess evident by MRI.
Among the 32 patients that showed neurological symptoms after HSCT, the HSCT-associated factors were compared between the 21 children in this subpopulation with a good neurologic outcome and the 11 remaining cases with a poor neurologic outcome (Table 4). Neither the GvHD nor the type of immunosuppressant affected the neurologic prognosis. More of the children in the poor outcome group had a neurological symptom onset later than 100 days after HSCT.

Table 4.
Neurological outcome assessments in patients with neurological symptom onset after HSCT.
3.4. Comparison of MRI Findings
The extent of the WM lesions was more prevalent in the poor outcome group. Among the other accompanying abnormal brain MRI findings, abnormal enhancements, ventriculomegaly, cortical changes, deep gray matter abnormalities and cerebellar abnormalities were more frequent in the poor outcome group, with statistical significance (Table 5).

Table 5.
MRI findings according to neurologic outcome.
There was no statistically significant difference in outcome between the patients with normal MRI and abnormal MRI findings (Appendix A). However, in patients with normal MRI findings, persistent neurological symptoms such as focal seizure and headache were relatively mild and there was no death due to neurological complications (Appendix B).
In our current study series, six children were diagnosed with PRES, but five of these cases had a good prognosis. All six children diagnosed with PRES had hypertension and neurological symptoms (four with seizures, two with a decreased consciousness) and MRI findings of frontoparietal or parieto-occipital lesions. The extent of the WM lesions according to the classification and scoring in the PRES cases in our current series was 1–2 in the five recovered patients and 5 in the remaining female patient with a poor outcome and that had a tiny petechial hemorrhage in the involved region. This patient had no coagulopathy but mild thrombocytopenia, which was also seen in one of the five recovered patients. She eventually died of multiorgan failure following GI bleeding and pancreatitis caused by uncontrolled GvHD.
4. Discussion
This study retrospectively investigated children that had received HSCT and subsequently underwent a brain MRI due to newly developed neurological symptoms or infection signs during chemotherapy or HSCT. Among the patients in our pediatric cohort, 56% showed abnormal brain MRI findings and we attempted to identify associated factors in accordance with the neurological outcomes of these cases. In our poor outcome group, there were more instances of malignancy as the underlying disease and infection as the cause of the neurological symptoms. Notably, however, there was no differences found in the factors associated with transplantation (immunosuppressants and GvHD) according to the outcome. On the other hand, the severity of the abnormal brain MRI findings was significantly associated with the clinical outcomes in our cohort.
Previous studies [2,12,13,14,15,16] have reported that about 8.3–24% of patients developed neurological symptoms after HSCT. These neurological complications, including encephalopathy, have been associated with a poor prognosis resulting in a high mortality rate in children after HSCT [6,17] and an accurate and timely diagnosis and intervention is crucial to improve the outcomes in these cases. Many of the currently used anticancer drugs and myeloablative drugs administered for HSCT have neurologic side effects [18]. Although it is difficult to find a specific causal relationship between a hazardous dosage or a particular chemotherapeutic agent and CNS toxicity, the mechanisms of neurotoxicity caused by chemotherapy are suggested by several hypotheses [19]. In our present pediatric cohort, an underlying malignancy was associated with poor neurological outcomes, among various clinical factors. Although there were no significant associations identified by our present analyses due to small number of patients examined, our results indicated that a higher dose of chemotherapy, TBI doses and immunosuppressants in patients with malignant disease can be associated with a poor neurologic prognosis, consistent with previous studies [6,7,12,13,14,15,16,20].
Leukoencephalopathy is one of the major side effects of chemotherapy but can be mild and reversible in many cases [19]. Posterior reversible encephalopathy syndrome (PRES), caused by an acute rise in blood pressure associated with calcineurin inhibitors and corticosteroids, is a clinical syndrome typically characterized by vision changes, altered mental status and seizures. Although the prognosis of PRES is known to be favorable in most cases, neurological sequelae can persist in 10–20% of affected patients [21]. Many studies have attempted to identify the etiology and prognostic factors and associated brain MRI findings, for PRES, but there are still no established criteria for this condition. The risk factors identified by each prior study of this disorder have been different, but the following variables have been suggested to be associated with poor outcomes: severe encephalopathy, hypertensive etiology, hyperglycemia, neoplastic etiology, longer time to control the causative factor, presence of multiple comorbidities, elevated CRP, low CSF glucose, coagulopathy and brain MRI findings of corpus callosum involvement, extensive cerebral edema or worsening imaging severity, hemorrhage, subarachnoid hemorrhage and restrictive diffusion [22].
Infection was found in our current analysis to be a more frequent cause of neurologic complications in the poor outcome group. Children who have undergone HSCT are particularly susceptible to viruses, bacteria, or invasive molds and CNS infection has been shown to be indicative of a poor prognosis [2,15,16,20,23]. The incidence of CNS infection after allo-HSCT can be as high as 15% [24]. Aspergillus and toxoplasma are one of the most common causes and other fungi and viruses can also be a source CNS infection. Prior studies have indicated that a high-risk disease status is a risk factor for CNS infection, which in turn significantly reduces the overall survival rate [2,15]. Hence, if any indicators of CNS infection or neurological symptoms are present, a rapid diagnostic test and treatment including antimicrobial agents should be conducted with careful monitoring and concerns. However, there are several limitations to identifying the causative microorganisms in practice because a brain biopsy can be problematic for patients with a poor underlying condition and PCR detection is only possible for a few CSF viruses, such as HSV and CMV, despite the high sensitivity and specificity of this test [25]. Hence, empirical treatments should be initiated depending on the time since HSCT and the extent of the host immune deficiency to improve patient outcomes.
In many previous studies, GvHD has been identified as a risk factor for neurologic sequelae [12,13,25,26,27] and the prognosis of CNS GvHD is alleged to be very poor [28]. In addition to CNS GvHD, CNS infections and thrombotic microangiopathy (TMA)-associated neurological events can be frequent following the increased use of calcineurin inhibitors and corticosteroids in patients with GvHD [29]. In our current study, none of the children were diagnosed with CNS GvHD and the presence of GvHD or the use of immunosuppressant showed no significant association with poor clinical outcomes in our cohort. As CNS GvHD can be diagnosed only when drug toxicity or infection are ruled out, or when the symptoms meet other facultative criteria including abnormal MRI, or cerebrospinal fluid (CSF) abnormalities and pathology revealing GvHD lesions [29], the reported incidence of CNS GvHD can be very rare. Moreover, the differential diagnosis of causality in patients with GvHD and neurological symptoms is very difficult and the risk of immunosuppression and GvHD are always intermixed. This ambiguous causality can result in no significant correlation being found between these factors and clinical outcomes. In addition, it should be noted that our present study series included relatively small number of children with neurological symptoms after HSCT.
Many previous studies have attempted to interpret the changes in brain MRI findings after chemotherapy or HSCT. In a prior systematic review of chemotherapy-induced changes in the brain and cognitive functioning by Mingmei Li et al., neuroimaging data indicated a reduced grey matter density in cancer patients in the frontal, parietal and temporal regions. Data from diffusion-weighted MRI in that report suggested reduced WM integrity involving the superior longitudinal fasciculus, corpus callosum, forceps major and corona radiata and altered structural connectivity across the whole brain network and moderate-to-strong correlations between worsening cognitive function and morphological changes in the frontal brain regions [30]. There have been reports that diffusion tensor imaging (DTI) is a more useful tool than conventional MRI to confirm changes in WM integrity in patients undergoing HSCT [31,32,33,34]. In addition, in previous studies using voxel-based morphometry (VBM), a reduced volume of white and gray matter was observed in patients who underwent chemotherapy [35,36]. However, those investigations did not deal with clinically significant neurological events during and after HSCT, but preclinical quantitative imaging data and cognitive function. To support the clinical diagnosis and treatment of neurological complications of HSCT, we here compared brain MRI findings according to clinical outcomes. In the brain MRI findings, we identified a statistically significant relationship between a poor outcome and a more severe WM injury, abnormal enhancement, ventriculomegaly, cortical change, deep gray matter abnormality, or cerebellar abnormality. On the other hand, only unspecified scattered spots on the MRI were associated with a good outcome in our current pediatric cohort. Hence, along with the various prognostic factors discussed above, a brain MRI is an important prognostic tool in these cases. Moreover, these MRI findings can be combined with other indicators to develop a scoring system for predicting the prognosis.
There were several limitations of our current study of note. There was a selection bias for patients who had undergone a brain MRI. It is difficult to accurately describe the incidence of neurological complications because patients with neurological complications were not first selected. However, in our center, when clinically significant neurological symptoms occur, brain MRI is performed on most patients, so except for children with minor and transient neurological symptoms, most would have been included. The diagnosis of neurological symptoms in these cases was based on clinical and imaging findings. A definitive etiology could therefore not be identified. The cumulative dose of chemotherapeutics, radiation and immunosuppressants was not investigated although the prognosis would have been affected by these parameters. Due to the retrospective design of this study, many patients who were transferred to our center due to refractory diseases were excluded because there were no available data on their previous treatments. In addition, minor neurocognitive dysfunction in patients in our good outcome group, or patients who had no evident abnormalities on a conventional brain MRI scan, could not be evaluated. A further, well-designed, prospective large multicenter study will be needed.
5. Conclusions
Neurological complications and associated brain MRI abnormalities including WM injury are frequent events in children following HSCT and treatments for cancer, but most show a complete recovery. However, children with underlying malignant disease or with neurologic symptoms due to infectious causes should be carefully monitored. In addition, the brain MRI findings for these cases, particularly the extent of WM injury, are associated with their clinical outcomes. A model that predicts the prognosis of children after HSCT in accordance with their brain MRI findings would be very useful. Future large-scale multicenter studies and subgroup analyses of the prognostic indicators of neurological symptoms after HSCT are warranted.
Author Contributions
Conceptualization, M.-J.K. and M.-S.Y.; Methodology, M.-J.K. and M.-S.Y.; software, M.-J.K.; validation, H.S. and M.-J.K.; formal analysis, H.S. and M.-J.K.; investigation, H.S. and M.-J.K.; resources, J.K.S., H.J.I., H.K. and K.-N.K.; data curation, H.S. and M.-J.K.; writing—original draft preparation, H.S. and M.-J.K.; writing—review and editing, M.-S.Y., T.-S.K., J.K.S., H.J.I., H.K. and K.-N.K.; visualization, H.S. and M.-J.K.; supervision, M.-S.Y., T.-S.K.; project administration, M.-S.Y. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board of Asan Medical Center (2021 0626).
Informed Consent Statement
Informed consent was waived because of the retrospective nature of the study and the analysis used anonymous clinical data.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Table A1.
Neurological Outcome Assessments According to MRI Finding.
Table A1.
Neurological Outcome Assessments According to MRI Finding.
| Total, n (%) | Normal MRI, n = 40 | Abnormal MRI, n = 51 | p-Value |
|---|---|---|---|
| Neurologic outcomes, n | 0.321 | ||
| Full recovery | 33 | 37 | |
| Neurologic sequelae | 7 | 14 | |
| Sex, n | 1 | ||
| Male | 22 | 27 | |
| Female | 18 | 24 | |
| Median age at diagnosis, year | 0.265 | ||
| Median [IQR] | 8.00 [4.25, 13.00] | 8.00 [2.00, 14.00] | |
| Underlying disease, n | 0.356 | ||
| Malignant | 27 | 39 | |
| Non-malignant | 13 | 12 | |
| Type of transplantation, n | 0.828 | ||
| HaploPBSCT | 16 | 23 | |
| Unrelated donor alloPBSCT | 16 | 20 | |
| Related donor alloPBSCT | 8 | 8 | |
| Time at symptom appearance, n | 1 | ||
| Before BMT | 15 | 19 | |
| After BMT | 25 | 32 | |
| Systemic chemotherapy, n | |||
| Cyclophosphamide | 27 | 34 | 1 |
| Cytarabine | 15 | 33 | 0.012 |
| Etoposide | 17 | 27 | 0.399 |
| Vincristine | 15 | 26 | 0.212 |
| Methotrexate | 9 | 16 | 0.478 |
| Ifosfamide | 9 | 12 | 1 |
| Blinatumomab | 0 | 3 | 0.253 |
| Cisplatin | 3 | 1 | 0.316 |
| Carboplatin | 3 | 1 | 0.316 |
| Vinblastine | 2 | 1 | 0.58 |
| IT chemotherapy, n | |||
| Not done | 20 | 15 | 0.053 |
| Done | 20 | 36 | |
| Cytarabine | 19 | 33 | 0.135 |
| Methotrexate | 10 | 26 | 0.017 |
| Hydrocortisone | 8 | 15 | 0.341 |
| Cranial radiation, n | 18 | 22 | 1 |
| Immunosuppressant, n (%) | |||
| CS | 15/25 | 19/32 | 1 |
| FK506 | 9/25 | 13/32 | 0.788 |
| MMF | 12/25 | 13/32 | 0.602 |
| none | 4/25 | 7/32 | 0.739 |
| GVHD, n | 12/25 | 19/32 | 0.432 |
| Duration from BMT to symptom onset | 0.788 | ||
| Median [IQR] | 97.00 [58.50, 481.00] | 178.50 [84.75, 487.25] | |
| ≤100day, n | 13/25 | 10/32 | 0.174 |
| >100day, n | 12/25 | 22/32 | |
| Initial presenting symptom, n | |||
| Motor symptom | 14 | 20 | 0.827 |
| Decreased consciousness | 4 | 12 | 0.105 |
| Headache | 19 | 10 | 0.006 |
| Brainstem dysfunction | 1 | 2 | 1 |
| Visual symptom | 0 | 2 | 0.502 |
| Infection sign | 2 | 5 | 0.46 |
Appendix B

Table A2.
Initial Presenting and Remained Neurological Symptom of the Patients.
Table A2.
Initial Presenting and Remained Neurological Symptom of the Patients.
| Patients | Initial Presenting Neurological Symptom | Remained Neurological Symptom |
|---|---|---|
| Normal MRI group | ||
| 1 | Headache | Persistent headache requiring medications |
| 2 | Headache | Progression to confused mentality with brain MRI findings suspicious of CSF metastasis after 2 months |
| 3 | Facial palsy | Persistent facial palsy |
| 4 | Seizure | Continue taking AED due to focal seizure |
| 5 | Seizure | Continue taking AED due to focal seizure |
| 6 | Headache | Intermittent focal hand dystonia probably due to other medication |
| 7 | Seizure | Continue taking AED due to focal seizure |
| Abnormal MRI group | ||
| 1 | Headache | Persistent headache, IICP, Progression to confused mentality |
| Died of graft failure and septic shock after 1 month | ||
| 2 | Decreased consciousness | Persistent headache, vomiting |
| Died of refractory malignancy despite chemotherapy after 1 year | ||
| 3 | Seizure | Persistent seizure |
| Died of multiorgan failure due to GvHD and infection after 2 months | ||
| 4 | Muscle cramping with stiffness | Continue taking gabapentin |
| 5 | Decreased consciousness | Stuporous mental status with rigidity |
| Died of refractory malignancy despite chemotherapy after 4 months | ||
| 6 | Decreased consciousness | Semicomatous mental status |
| Died of multiorgan failure due to GvHD and infection after 1 months | ||
| 7 | Seizure | Semicomatous mental status |
| Died of multiorgan failure due to GvHD and septic shock after 2 months | ||
| 8 | Disorientation | Semicomatous mental status |
| Died of respiratory failure due to lung GvHD and pneumonia after 1 week | ||
| 9 | Progressive muscle weakness, parasthesia | Aggravated weakness |
| Died of respiratory failure due to invasive pulmonary aspergillosis after 2 months | ||
| 10 | Decreased consciousness | Developmental delay |
| 11 | Gait disturbance, dysarthria | Developmental delay, Epilepsy on AEDs |
| 12 | Decreased consciousness | Semicomatous mental status with rigidity, Progressive hydrocephalus |
| Died of refractory malignancy after 3 months | ||
| 13 | Headache | Persistent seizure |
| Died of multiorgan failure due to infection after 2 months | ||
| 14 | Left side weakness, ataxia | Persistent headache |
Appendix C

Table A3.
CSF Analysis and Microbiological Testing Results of Patients with CSF Abnormalities.
Table A3.
CSF Analysis and Microbiological Testing Results of Patients with CSF Abnormalities.
| Patients | Neurologic Outcomes | Cause of Neurologic Symptoms | CSF Analysis | Microbiologic Testing |
|---|---|---|---|---|
| 1 | Full recovery | Tumor involvement | RBC 0/μL, WBC 14/μL, Protein 70.7 mg/dL, Glucose 62 mg/dL | Negative for CSF bacterial culture, Mycoplasma pneumonia DNA PCR, EBV DNA PCR, CMV DNA PCR, Enterovirus RNA PCR, MTB DNA PCR |
| 2 | Full recovery | Infection | RBC 4/μL, WBC 487/μL, Protein 47.8 mg/dL, Glucose 50 mg/dL | Negative for CSF bacterial culture, EBV DNA PCR, CMV DNA PCR, Enterovirus RNA PCR, HSV-1,2 DNA PCR |
| Positive for β-D-Glucan | ||||
| 3 | Full recovery | Infection | RBC 6/μL, WBC 1/μL, Protein 180.4 mg/dL, Glucose 64 mg/dL | Negative for CSF bacterial culture, HSV-1,2 DNA PCR |
| 4 | Full recovery | Infection | RBC 0/μL, WBC 3/μL, Protein 33.2 mg/dL, Glucose 47 mg/dL | Positive for CSF CMV PCR |
| Negative for CSF bacterial culture | ||||
| 5 | Full recovery | Infection | RBC 0/μL, WBC 10/μL, Protein 61.4 mg/dL, Glucose 53 mg/dL | Streptococcus oralis on CSF bacterial culture |
| Negative for CSF Enterovirus RNA PCR, MTB DNA PCR, HSV-1,2 DNA PCR | ||||
| 6 | Full recovery | Infection | RBC 0/μL, WBC 1/μL, Protein 63.9 mg/dL, Glucose 52 mg/dL | Positive for CSF CMV PCR |
| Negative for CSF bacterial culture | ||||
| 7 | Full recovery | Infection | RBC 11800/μL, WBC 87/μL, Protein 1126.5 mg/dL, Glucose 30 mg/dL | Listeria monocytogenes on CSF bacterial culture |
| 8 | Full recovery | Drug associated | RBC 0/μL, WBC 10/μL, Protein 159.4 mg/dL, Glucose 44 mg/dL | Negative for CSF bacterial culture, Mycoplasma pneumonia DNA PCR, EBV DNA PCR, CMV DNA PCR, Enterovirus RNA PCR |
| 9 | Full recovery | Infection | RBC 600/μL, WBC 13/μL, Protein 52.8 mg/dL, Glucose 49 mg/dL | Positive for CSF adenovirus culture |
| Negative for CSF bacterial culture | ||||
| 10 | Neurologic sequelae | Infection | RBC 1/μL, WBC 1/μL, Protein 66.3 mg/dL, Glucose 57 mg/dL | Streptococcus viridans group on CSF bacterial culture |
| 11 | Neurologic sequelae | Infection | RBC 193/μL, WBC 50/μL, Protein 100 mg/dL, Glucose 71 mg/dL | Negative for CSF bacterial culture |
| 12 | Neurologic sequelae | Infection | RBC 0/μL, WBC 1580/μL, Protein 369.6 mg/dL, Glucose 26 mg/dL | Negative for CSF bacterial culture |
| 13 | Neurologic sequelae | Tumor involvement | RBC 0/μL, WBC 16/μL(meningeal involvement of leukemic blasts), Protein 45.9 mg/dL, Glucose 35 mg/dL | Negative for CSF bacterial culture |
| 14 | Neurologic sequelae | Infection | RBC 3/μL, WBC 3/μL(meningeal involvement of leukemic blasts), Protein 29.3 mg/dL, Glucose 61 mg/dL | Positive for CSF CMV PCR |
| Negative for CSF bacterial culture |
References
- Nishiguchi, T.; Mochizuki, K.; Shakudo, M.; Takeshita, T.; Hino, M.; Inoue, Y. CNS Complications of Hematopoietic Stem Cell Transplantation. Am. J. Roentgenol. 2009, 192, 1003–1011. [Google Scholar] [CrossRef]
- Schmidt, K.; Schulz, A.S.; Debatin, K.-M.; Friedrich, W.; Classen, C.F. CNS complications in children receiving chemotherapy or hematopoietic stem cell transplantation: Retrospective analysis and clinical study of survivors. Pediatr. Blood Cancer 2008, 50, 331–336. [Google Scholar] [CrossRef]
- Yoshida, S.; Hayakawa, K.; Yamamoto, A.; Kuroda, H.; Imashuku, S. The central nervous system complications of bone marrow transplantation in children. Eur. Radiol. 2008, 18, 2048–2059. [Google Scholar] [CrossRef] [PubMed]
- Berg, H.V.D.; Gerritsen, E.J.; Noordijk, E.M.; Vossen, J.M. Major complications of the central nervous system after bone marrow transplantation in children with acute lymphoblastic leukemia. Radiother. Oncol. 1990, 18, 94–97. [Google Scholar] [CrossRef]
- Benya, E.C.; Goldman, S. Bone Marrow Transplantation in Children. Pediatr. Clin. North Am. 1997, 44, 741–761. [Google Scholar] [CrossRef]
- Dulamea, A.O.; Lupescu, I.G. Neurological complications of hematopoietic cell transplantation in children and adults. Neural Regen. Res. 2018, 13, 945–954. [Google Scholar] [CrossRef]
- Sostak, P.; Padovan, C.; Yousry, T.; Ledderose, G.; Kolb, H.-J.; Straube, A. Prospective evaluation of neurological complications after allogeneic bone marrow transplantation. Neurology 2003, 60, 842–848. [Google Scholar] [CrossRef]
- Monje, M.L.; Vogel, H.; Masek, M.; Ligon, K.L.; Fisher, P.G.; Palmer, T.D. Impaired human hippocampal neurogenesis after treatment for central nervous system malignancies. Ann. Neurol. 2007, 62, 515–520. [Google Scholar] [CrossRef]
- Seigers, R.; Fardell, J.E. Neurobiological basis of chemotherapy-induced cognitive impairment: A review of rodent research. Neurosci. Biobehav. Rev. 2011, 35, 729–741. [Google Scholar] [CrossRef]
- Monje, M.; Dietrich, J. Cognitive side effects of cancer therapy demonstrate a functional role for adult neurogenesis. Behav. Brain Res. 2012, 227, 376–379. [Google Scholar] [CrossRef]
- Chen, B.T.; Ortiz, A.O.; Dagis, A.; Torricelli, C.; Parker, P.; Openshaw, H. Brain imaging findings in symptomatic patients after allogeneic haematopoietic stem cell transplantation: Correlation with clinical outcome. Eur. Radiol. 2012, 22, 2273–2281. [Google Scholar] [CrossRef] [PubMed]
- Dowling, M.; Li, S.; Dey, B.R.; McAfee, S.L.; Hock, H.R.; Spitzer, T.R.; Chen, Y.-B.; Ballen, K.K. Neurologic complications after allogeneic hematopoietic stem cell transplantation: Risk factors and impact. Bone Marrow Transplant. 2017, 53, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.-M.; Kim, Y.-J.; Kim, J.Y.; Cho, E.J.; Lee, J.H.; Lee, M.H.; Lee, S.-H.; Sung, K.W.; Koo, H.H.; Yoo, K.H. Neurologic Complications after Allogeneic Hematopoietic Stem Cell Transplantation in Children: Analysis of Prognostic Factors. Biol. Blood Marrow Transplant. 2015, 21, 1091–1098. [Google Scholar] [CrossRef][Green Version]
- Azik, F.; Erdem, A.Y.; Tavil, B.; Bayram, C.; Tunc, B.; Uckan, D. Neurological complications after allogeneic hematopoietic stem cell transplantation in children, a single center experience. Pediatr. Transplant. 2014, 18, 405–411. [Google Scholar] [CrossRef]
- Weber, C.; Schaper, J.; Tibussek, D.; Adams, O.; MacKenzie, C.R.; Dilloo, D.; Meisel, R.; Göbel, U.; Laws, H.-J. Diagnostic and therapeutic implications of neurological complications following paediatric haematopoietic stem cell transplantation. Bone Marrow Transplant. 2007, 41, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Faraci, M.; Lanino, E.; Dini, G.; Fondelli, M.; Morreale, G.; Dallorso, S.; Manzitti, C.; Calevo, M.; Gaggero, R.; Castagnola, E.; et al. Severe neurologic complications after hematopoietic stem cell transplantation in children. Neurol. 2002, 59, 1895–1904. [Google Scholar] [CrossRef] [PubMed]
- Woodard, P.; Helton, K.J.; McDaniel, H.M.; Khan, R.B.; Thompson, S.D.; A Hale, G.; Benaim, E.; A Kasow, K.; Leung, W.; Horwitz, E.M.; et al. Encephalopathy in pediatric patients after allogeneic hematopoietic stem cell transplantation is associated with a poor prognosis. Bone Marrow Transplant. 2004, 33, 1151–1157. [Google Scholar] [CrossRef][Green Version]
- Armstrong, C.; Sun, L.R. Neurological complications of pediatric cancer. Cancer Metastasis Rev. 2020, 39, 3–23. [Google Scholar] [CrossRef]
- Ikonomidou, C. Chemotherapy and the pediatric brain. Mol. Cell. Pediatr. 2018, 5, 1–10. [Google Scholar] [CrossRef]
- Maffini, E.; Festuccia, M.; Brunello, L.; Boccadoro, M.; Giaccone, L.; Bruno, B. Neurologic Complications after Allogeneic Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2017, 23, 388–397. [Google Scholar] [CrossRef]
- Fugate, J.; Rabinstein, A. Posterior reversible encephalopathy syndrome: Clinical and radiological manifestations, pathophysiology, and outstanding questions. Lancet Neurol. 2015, 14, 914–925. [Google Scholar] [CrossRef]
- Hinduja, A. Posterior Reversible Encephalopathy Syndrome: Clinical Features and Outcome. Front. Neurol. 2020, 11, 71. [Google Scholar] [CrossRef] [PubMed]
- Hanajiri, R.; Kobayashi, T.; Yoshioka, K.; Watanabe, D.; Watakabe, K.; Murata, Y.; Hagino, T.; Seno, Y.; Najima, Y.; Igarashi, A.; et al. Central nervous system infection following allogeneic hematopoietic stem cell transplantation. Hematol. Stem Cell Ther. 2017, 10, 22–28. [Google Scholar] [CrossRef]
- Schmidt-Hieber, M.; Zweigner, J.; Uharek, L.; Blau, I.W.; Thiel, E. Central nervous system infections in immunocompromised patients –Update on diagnostics and therapy. Leuk. Lymphoma 2009, 50, 24–36. [Google Scholar] [CrossRef]
- Schmidt-Hieber, M.; Silling, G.; Schalk, E.; Heinz, W.; Panse, J.; Penack, O.; Christopeit, M.; Buchheidt, D.; Meyding-Lamadé, U.; Hähnel, S.; et al. CNS infections in patients with hematological disorders (including allogeneic stem-cell transplantation)—Guidelines of the Infectious Diseases Working Party (AGIHO) of the German Society of Hematology and Medical Oncology (DGHO). Ann. Oncol. 2016, 27, 1207–1225. [Google Scholar] [CrossRef]
- Uckan, D.; Çetinkaya, D.U.; Yigitkanli, I.; Tezcan, I.; Tuncer, M.; Karasimav, D.; Oguz, K.K.; Topcu, M. Life-threatening neurological complications after bone marrow transplantation in children. Bone Marrow Transplant. 2004, 35, 71–76. [Google Scholar] [CrossRef][Green Version]
- Koh, K.N.; Park, M.; Kim, B.E.; Im, H.J.; Seo, J.J. Early central nervous system complications after allogeneic hematopoietic stem cell transplantation in children. Korean J. Hematol. 2010, 45, 164–170. [Google Scholar] [CrossRef]
- Ruggiu, M.; Cuccuini, W.; Mokhtari, K.; Meignin, V.; de Latour, R.P.; Robin, M.; de Fontbrune, F.S.; Xhaard, A.; Socié, G.; Michonneau, D. Case report. Medicine 2017, 96, e8303. [Google Scholar] [CrossRef] [PubMed]
- Grauer, O.; Wolff, D.; Bertz, H.; Greinix, H.; Kühl, J.-S.; Lawitschka, A.; Lee, S.J.; Pavletic, S.Z.; Holler, E.; Kleiter, I. Neurological manifestations of chronic graft-versus-host disease after allogeneic haematopoietic stem cell transplantation: Report from the Consensus Conference on Clinical Practice in chronic graft-versus-host disease. Brain 2010, 133, 2852–2865. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Caeyenberghs, K. Longitudinal assessment of chemotherapy-induced changes in brain and cognitive functioning: A systematic review. Neurosci. Biobehav. Rev. 2018, 92, 304–317. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, Y.; Natsume, J.; Kidokoro, H.; Tanaka, M.; Okai, Y.; Ito, Y.; Yamamoto, H.; Ohno, A.; Nakata, T.; Nakane, T.; et al. Change of White Matter Integrity in Children with Hematopoietic Stem Cell Transplantation. Pediatr. Neurol. 2020, 111, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Sabin, N.; Cheung, Y.; Reddick, W.; Bhojwani, D.; Liu, W.; Glass, J.; Brinkman, T.; Hwang, S.; Srivastava, D.; Pui, C.-H.; et al. The Impact of Persistent Leukoencephalopathy on Brain White Matter Microstructure in Long-Term Survivors of Acute Lymphoblastic Leukemia Treated with Chemotherapy Only. Am. J. Neuroradiol. 2018, 39, 1919–1925. [Google Scholar] [CrossRef]
- Correa, D.D.; Wang, Y.; West, J.D.; Peck, K.K.; Root, J.C.; Baser, R.E.; Thaler, H.T.; Shore, T.B.; Jakubowski, A.; Saykin, A.J.; et al. Prospective assessment of white matter integrity in adult stem cell transplant recipients. Brain Imaging Behav. 2015, 10, 486–496. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morioka, S.; Morimoto, M.; Yamada, K.; Hasegawa, T.; Morita, T.; Moroto, M.; Isoda, K.; Chiyonobu, T.; Imamura, T.; Nishimura, A.; et al. Effects of chemotherapy on the brain in childhood: Diffusion tensor imaging of subtle white matter damage. Neuroradiology 2013, 55, 1251–1257. [Google Scholar] [CrossRef]
- Genschaft, M.; Huebner, T.; Plessow, F.; Ikonomidou, V.N.; Abolmaali, N.; Krone, F.; Hoffmann, A.; Holfeld, E.; Vorwerk, P.; Kramm, C.; et al. Impact of Chemotherapy for Childhood Leukemia on Brain Morphology and Function. PLoS ONE 2013, 8, e78599. [Google Scholar] [CrossRef] [PubMed]
- Carey, M.; Haut, M.; Reminger, S.; Hutter, J.; Theilmann, R.; Kaemingk, K. Reduced Frontal White Matter Volume in Long-Term Childhood Leukemia Survivors: A Voxel-Based Morphometry Study. Am. J. Neuroradiol. 2008, 29, 792–797. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).