Safety Assessment and Probiotic Potential of a Novel Species Lactobacillus xujianguonis
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Condition
2.2. Phenotypic Safety Assessment and Probiotic Characteristics
2.2.1. Survivability in Low pH or 0.3% Bile Salt
2.2.2. Auto-Aggregation and Cell Surface Hydrophobicity
2.2.3. Antimicrobial Activity
2.2.4. Antimicrobial Susceptibility
2.2.5. Hemolysin and Gelatinase Production
2.3. In Vivo Toxicology Studies
2.3.1. Animal
2.3.2. Acute Oral Toxicity Study
2.3.3. 28-Day Repeated-Dose Toxicity Study
2.3.4. Hematology and Biochemical Testing
2.3.5. Histopathology Analysis
2.3.6. Quantitative Real-Time Polymerase Chain Reaction
2.4. Statistical Analysis
3. Results
3.1. Probiotic Properties
3.2. Safety Assessment In Vitro
3.3. Acute Toxicity Study
3.4. Twenty-Eight-Day Repeated-Dose Oral Toxicity Study
3.4.1. Clinical Pathology and Immunological Effects
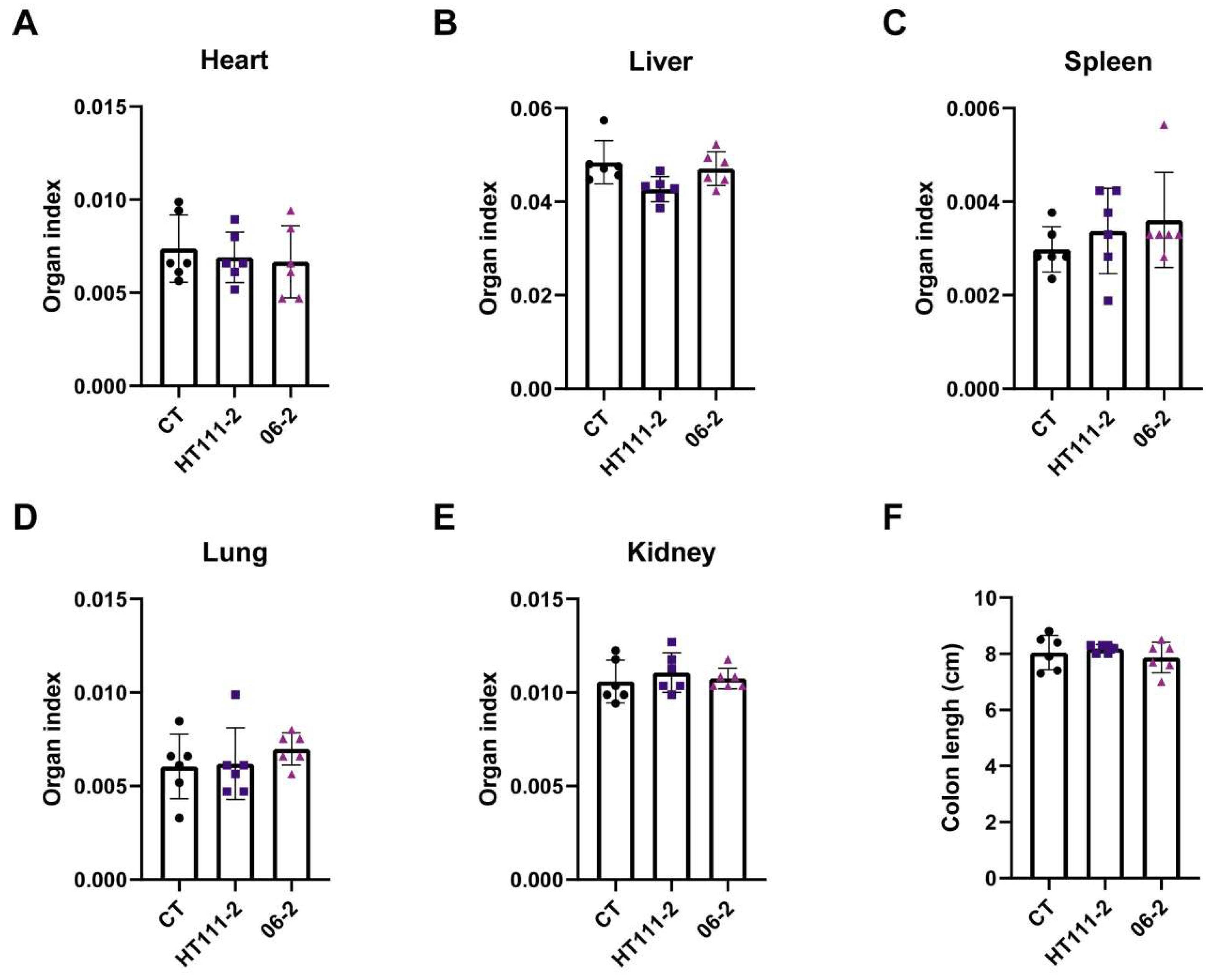
3.4.2. Organ Weight, Macroscopic Observations and Histopathology
3.4.3. Gut Barrier and Translocation Safety
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| LAB | Lactic Acid Bacteria |
| GRAS | Generally Recognized as Safe |
| WHO | World Health Organization |
| FDA | Food and Drug Administration |
| CGMCC | China General Microbiological Culture Collection Center |
| ATCC | American Type Strain Preservation Center |
| MRS | de Man, Rogosa Sharpe |
| BHI | Brain Heart Infusion |
| CLSI | Clinical and Laboratory Standards Institute |
| OECD | Organisation for Economic Co-operation and Development |
| NOAEL | Mo-Observed-Adverse-Effect Level |
| CBC | Complete Blood Count |
| LD50 | Median Lethal Dose |
References
- Araya, M.; Morelli, L.; Reid, G.; Sanders, M.E.; Stanton, C.; Pineiro, M.; Embarek, P.B. Guidelines for the evaluation of probiotics in food. In Joint FAO/WHO Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food; WHO: Geneva, Switzerland, 2002; pp. 1–11. [Google Scholar]
- Shah, A.B.; Baiseitova, A.; Zahoor, M.; Ahmad, I.; Ikram, M.; Bakhsh, A.; Shah, M.A.; Ali, I.; Idress, M.; Ullah, R.; et al. Probiotic significance of Lactobacillus strains: A comprehensive review on health impacts, research gaps, and future prospects. Gut Microbes 2024, 16, 2431643. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Hong, Q.; Cui, J.; Li, W.; Zhang, J.; Sun, Y.; Jiang, J.; Hu, Y. Harnessing lactobacillus: A probiotic revolution in combating metabolic dysfunction-associated steatotic liver disease. Crit. Rev. Food Sci. Nutr. 2025, 1–18, online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Singh, A. Gut microbiome and human health: Exploring how the probiotic genus Lactobacillus modulate immune responses. Front. Pharmacol. 2022, 13, 1042189. [Google Scholar] [CrossRef] [PubMed]
- Donohue, D.C.; Salminen, S. Safety of probiotic bacteria. Asia Pac. J. Clin. Nutr. 1996, 5, 25–28. [Google Scholar]
- Adams, M.R.; Marteau, P. On the safety of lactic acid bacteria from food. Int. J. Food Microbiol. 1995, 27, 263–264. [Google Scholar] [CrossRef]
- Rossi, F.; Amadoro, C.; Gasperi, M.; Colavita, G. Lactobacilli Infection Case Reports in the Last Three Years and Safety Implications. Nutrients 2022, 14, 1178. [Google Scholar] [CrossRef]
- DeMarco, E.; DePetrillo, J.; Qadeer, F. Meropenem resistant Lactobacillus endocarditis in an immunocompetent patient. SAGE Open Med. Case Rep. 2023, 11, 2050313x231152709. [Google Scholar] [CrossRef]
- Kullar, R.; Goldstein, E.J.C.; Johnson, S.; McFarland, L.V. Lactobacillus Bacteremia and Probiotics: A Review. Microorganisms 2023, 11, 896. [Google Scholar] [CrossRef]
- Salminen, M.K.; Rautelin, H.; Tynkkynen, S.; Poussa, T.; Saxelin, M.; Valtonen, V.; Jarvinen, A. Lactobacillus bacteremia, clinical significance, and patient outcome, with special focus on probiotic L. rhamnosus GG. Clin. Infect. Dis. 2004, 38, 62–69. [Google Scholar] [CrossRef]
- Cohen, S.A.; Woodfield, M.; Boyle, N.; Stednick, Z.; Boeckh, M.; Pergam, S. Incidence and outcomes of bloodstream infections among hematopoietic cell transplant recipients from species commonly reported to be in over-the-counter probiotic formulations. Transpl. Infect. Dis. 2016, 18, 699–705. [Google Scholar] [CrossRef]
- Son, H.K.; Chang, H.C.; Lee, J.J. Acute and Subacute Oral Toxicity Evaluation of Crude Antifungal Compounds Produced by Lactobacillus plantarum HD1 in Rats. Prev. Nutr. Food Sci. 2015, 20, 190–197. [Google Scholar] [CrossRef]
- Darbandi, A.; Mirkalantari, S.; Zadeh, R.G.; Esghaei, M.; Talebi, M.; Kakanj, M. Safety evaluation of mutagenicity, genotoxicity, and cytotoxicity of Lactobacillus spp. isolates as probiotic candidates. J. Clin. Lab. Anal. 2022, 36, e24481. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.Y.; Kim, D.-H.; Kim, H.-J.; Jang, Y.-S.; Song, K.-Y.; Bae, D.; Kim, H.; Seo, K.-H. A Combined In Vitro and In Vivo Assessment of the Safety of the Yeast Strains Kluyveromyces marxianus A4 and A5 Isolated from Korean Kefir. Probiotics Antimicrob. Proteins 2023, 15, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Jin, D.; Yang, J.; Lai, X.-H.; Pu, J.; Zhu, W.; Huang, Y.; Liang, H.; Lu, S. Lactobacillus xujianguonis sp. nov., isolated from faeces of Marmota himalayana. Int. J. Syst. Evol. Microbiol. 2020, 70, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Abe Sato, S.T.; Marques, J.M.; da Luz de Freitas, A.; Sanches Progenio, R.C.; Nunes, M.R.T.; Mota de Vasconcelos Massafra, J.; Gomes Moura, F.; Rogez, H. Isolation and Genetic Identification of Endophytic Lactic Acid Bacteria From the Amazonian Açai Fruits: Probiotics Features of Selected Strains and Their Potential to Inhibit Pathogens. Front. Microbiol. 2020, 11, 610524. [Google Scholar] [CrossRef]
- Zulkhairi Amin, F.A.; Sabri, S.; Ismail, M.; Chan, K.W.; Ismail, N.; Esa, N.M.; Lila, M.A.M.; Zawawi, N. Probiotic Properties of Bacillus Strains Isolated from Stingless Bee (Heterotrigona itama) Honey Collected across Malaysia. Int. J. Environ. Res. Public Health 2019, 17, 278. [Google Scholar] [CrossRef]
- Fonseca, H.C.; de Sousa Melo, D.; Ramos, C.L.; Dias, D.R.; Schwan, R.F. Probiotic Properties of Lactobacilli and Their Ability to Inhibit the Adhesion of Enteropathogenic Bacteria to Caco-2 and HT-29 Cells. Probiotics Antimicrob. Proteins 2021, 13, 102–112. [Google Scholar] [CrossRef]
- Li, Y.; Hu, S.; Gong, L.; Pan, L.; Li, D.; Cao, L.; Khan, T.A.; Yang, Y.; Peng, Y.; Ding, X.; et al. Isolating a new Streptomyces amritsarensis N1-32 against fish pathogens and determining its effects on disease resistance of grass carp. Fish Shellfish Immunol. 2020, 98, 632–640. [Google Scholar] [CrossRef]
- Rastogi, S.; Mittal, V.; Singh, A. Selection of Potential Probiotic Bacteria from Exclusively Breastfed Infant Faeces with Antagonistic Activity Against Multidrug-Resistant ESKAPE Pathogens. Probiotics Antimicrob. Proteins 2021, 13, 739–750. [Google Scholar] [CrossRef]
- Ribeiro, S.C.; Coelho, M.; Todorov, S.; Franco, B.; Dapkevicius, M.; Silva, C. Technological properties of bacteriocin-producing lactic acid bacteria isolated from Pico cheese an artisanal cow’s milk cheese. J. Appl. Microbiol. 2014, 116, 573–585. [Google Scholar] [CrossRef]
- GB/T 34240-2017; Laboratory animal—Feed producing. General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China Standardization Administration of China: Beijing, China, 2017.
- Ou, D.; Ling, N.; Wang, X.; Zou, Y.; Dong, J.; Zhang, D.; Shen, Y.; Ye, Y. Safety Assessment of One Lactiplantibacillus plantarum Isolated from the Traditional Chinese Fermented Vegetables-Jiangshui. Foods 2022, 11, 2177. [Google Scholar] [CrossRef]
- Lin, X.; Xu, M.; Lan, R.; Hu, D.; Zhang, S.; Zhang, S.; Lu, Y.; Sun, H.; Yang, J.; Liu, L.; et al. Gut commensal Alistipes shahii improves experimental colitis in mice with reduced intestinal epithelial damage and cytokine secretion. mSystems 2025, 10, e0160724. [Google Scholar] [CrossRef]
- Tan, H.; Wang, C.; Zhang, Q.; Tang, X.; Zhao, J.; Zhang, H.; Zhai, Q.; Chen, W. Preliminary safety assessment of a new Bacteroides fragilis isolate. Food Chem. Toxicol. 2020, 135, 110934. [Google Scholar] [CrossRef]
- Shivangi, S.; Devi, P.B.; Ragul, K.; Shetty, P.H. Probiotic Potential of Bacillus Strains Isolated from an Acidic Fermented Food Idli. Probiotics Antimicrob. Proteins 2020, 12, 1502–1513. [Google Scholar] [CrossRef]
- van Zyl, W.F.; Deane, S.M.; Dicks, L.M.T. Molecular insights into probiotic mechanisms of action employed against intestinal pathogenic bacteria. Gut Microbes 2020, 12, 1831339. [Google Scholar] [CrossRef] [PubMed]
- Breyer, G.M.; Arechavaleta, N.N.; Siqueira, F.M.; de Souza da Motta, A. Characterization of Lactic Acid Bacteria in Raw Buffalo Milk: A Screening for Novel Probiotic Candidates and Their Transcriptional Response to Acid Stress. Probiotics Antimicrob. Proteins 2021, 13, 468–483. [Google Scholar] [CrossRef]
- de Melo Pereira, G.V.; de Oliveira Coelho, B.; Magalhães Júnior, A.I.; Thomaz-Soccol, V.; Soccol, C.R. How to select a probiotic? A review and update of methods and criteria. Biotechnol. Adv. 2018, 36, 2060–2076. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, Z.Y.; Dong, K.; Yuan, J.P.; Guo, X.K. Antibiotic resistance of probiotic strains of lactic acid bacteria isolated from marketed foods and drugs. Biomed. Environ. Sci. 2009, 22, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Brodmann, T.; Endo, A.; Gueimonde, M.; Vinderola, G.; Kneifel, W.; de Vos, W.M.; Salminen, S.; Gómez-Gallego, C. Safety of Novel Microbes for Human Consumption: Practical Examples of Assessment in the European Union. Front. Microbiol. 2017, 8, 1725. [Google Scholar] [CrossRef]
- Metlakunta, A.S.; Soman, R.J. Safety evaluation of Bacillus coagulans SNZ 1969 in Wistar rats. Regul. Toxicol. Pharmacol. 2020, 110, 104538. [Google Scholar] [CrossRef]
- Gómez Del Pulgar, E.M.; Benítez-Páez, A.; Sanz, Y. Safety Assessment of Bacteroides Uniformis CECT 7771, a Symbiont of the Gut Microbiota in Infants. Nutrients 2020, 12, 551. [Google Scholar] [CrossRef]
- Han, J.S.; Back, S.-M.; Cho, J.-W.; Park, H.J.; Kim, W.-J.; Park, S.-H.; Noh, J.-H.; Kim, Y.-B.; Lee, B.-S. Genotoxicity and subchronic general toxicity assessments of Lactobacillus curvatus WiKim 38 using Sprague-Dawley rats. Food Chem. Toxicol. 2021, 152, 112199. [Google Scholar] [CrossRef]
- Liu, B.; Zhong, X.; Liu, Z.; Guan, X.; Wang, Q.; Qi, R.; Zhou, X.; Huang, J. Probiotic Potential and Safety Assessment of Lactiplantibacillus plantarum cqf-43 and Whole-Genome Sequence Analysis. Int. J. Mol. Sci. 2023, 24, 17570. [Google Scholar] [CrossRef]
- Buschmann, J. The OECD guidelines for the testing of chemicals and pesticides. Methods Mol. Biol. 2013, 947, 37–56. [Google Scholar] [CrossRef] [PubMed]
- Petterino, C.; Argentino-Storino, A. Clinical chemistry and haematology historical data in control Sprague-Dawley rats from pre-clinical toxicity studies. Exp. Toxicol. Pathol. 2006, 57, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Volynets, V.; Rings, A.; Bárdos, G.; Ostaff, M.J.; Wehkamp, J.; Bischoff, S.C. Intestinal barrier analysis by assessment of mucins, tight junctions, and α-defensins in healthy C57BL/6J and BALB/cJ mice. Tissue Barriers 2016, 4, e1208468. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, D.; Singh, R.; Tyagi, A.; Rashmi, H.M.; Batish, V.; Grover, S. Assessing safety of Lactobacillus plantarum MTCC 5690 and Lactobacillus fermentum MTCC 5689 using in vitro approaches and an in vivo murine model. Regul. Toxicol. Pharmacol. 2019, 101, 1–11. [Google Scholar] [CrossRef]
- Lazarenko, L.M.; Babenko, L.P.; Gichka, S.G.; Sakhno, L.O.; Demchenko, O.M.; Bubnov, R.V.; Sichel, L.M.; Spivak, M.Y. Assessment of the Safety of Lactobacillus casei IMV B-7280 Probiotic Strain on a Mouse Model. Probiotics Antimicrob. Proteins 2021, 13, 1644–1657. [Google Scholar] [CrossRef]
- Meyer, A.L.; Elmadfa, I.; Herbacek, I.; Micksche, M. Probiotic, as well as conventional yogurt, can enhance the stimulated production of proinflammatory cytokines. J. Hum. Nutr. Diet. 2007, 20, 590–598. [Google Scholar] [CrossRef]
- Samtiya, M.; Bhat, M.I.; Gupta, T.; Kapila, S.; Kapila, R. Safety Assessment of Potential Probiotic Lactobacillus fermentum MTCC-5898 in Murine Model after Repetitive Dose for 28 Days (Sub-Acute Exposure). Probiotics Antimicrob. Proteins 2020, 12, 259–270. [Google Scholar] [CrossRef]
- Muñoz, J.A.; Chenoll, E.; Casinos, B.; Bataller, E.; Ramón, D.; Genovés, S.; Montava, R.; Ribes, J.M.; Buesa, J.; Fàbrega, J.; et al. Novel probiotic Bifidobacterium longum subsp. infantis CECT 7210 strain active against rotavirus infections. Appl. Environ. Microbiol. 2011, 77, 8775–8783. [Google Scholar] [CrossRef]
- Fernández-Murga, M.L.; Sanz, Y. Safety Assessment of Bacteroides uniformis CECT 7771 Isolated from Stools of Healthy Breast-Fed Infants. PLoS ONE 2016, 11, e0145503. [Google Scholar] [CrossRef]
- Pradhan, D.; Singh, R.; Tyagi, A.; Rashmi, H.M.; Batish, V.K.; Grover, S. Assessing the Safety and Efficacy of Lactobacillus plantarum MTCC 5690 and Lactobacillus fermentum MTCC 5689 in Colitis Mouse Model. Probiotics Antimicrob. Proteins 2019, 11, 910–920. [Google Scholar] [CrossRef]
- Bronzwaer, S.; Kass, G.; Robinson, T.; Tarazona, J.; Verhagen, H.; Verloo, D.; Vrbos, D.; Hugas, M. Food Safety Regulatory Research Needs 2030. Efsa J. 2019, 17, e170622. [Google Scholar] [CrossRef]





| Pathogenic Species | Inhibition Zone (mm) | |
|---|---|---|
| HT111-2 | 06-2 | |
| EAEC (CICC 24186) | 3.83 ± 1.34 | 3.90 ± 1.21 |
| EHEC (ATCC 43895) | 3.50 ± 0.40 | 3.17 ± 0.67 |
| S. Typhimurium (ATCC 14028) | 3.03 ± 1.17 | 3.47 ± 0.65 |
| S. aureus (ATCC 26072) | 2.53 ± 0.12 | 3.30 ± 1.77 |
| L. monocytogenes EGD-e (ATCC BAA-679) | 14.47 ± 2.15 | 15.20 ± 1.41 |
| S. flexneri 2a str. 301 | 5.36 ± 0.67 | 3.33 ± 0.95 |
| Day | CT | HT111-2 | 06-2 |
|---|---|---|---|
| D0 | 18.14 ± 1.25 | 18.10 ± 0.49 | 18.06 ± 0.51 |
| D1 | 18.44 ± 1.18 | 18.10 ± 0.45 | 18.05 ± 0.31 |
| D3 | 18.60 ± 1.09 | 18.38 ± 0.70 | 18.26 ± 0.65 |
| D7 | 19.34 ± 1.34 | 19.06 ± 0.92 | 18.85 ± 0.50 |
| D14 | 19.75 ± 1.03 | 19.07 ± 0.91 | 19.20 ± 0.74 |
| Treatment # | WBC | Lymph | Mon | Gran | Lymph% | Mon% | Gran% | RBC | HGB | HCT | MCV | MCH | MCHC | RDW | PLT | MPV | PDW |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 109/L | 109/L | 109/L | 109/L | % | % | % | 1012/L | g/L | % | fL | pg | g/L | % | 109/L | fL | ||
| Female | |||||||||||||||||
| NC | 4.80 ± 0.99 | 3.90 ± 0.78 | 0.12 ± 0.04 | 0.78 ± 0.21 | 81.05 ± 2.35 | 2.65 ± 0.84 | 16.30 ± 1.70 | 10.52 ± 0.36 | 147.00 ± 7.90 | 56.22 ± 2.14 | 53.70 ± 1.20 | 13.68 ± 0.46 | 255.83 ± 14.25 | 15.95 ± 0.49 | 1123.33 ± 333.01 | 6.70 ± 0.54 | 17.13 ± 0.27 |
| H1 | 6.28 ± 0.47 | 5.08 ± 0.28 | 0.13 ± 0.05 | 1.07 ± 0.29 | 80.68 ± 4.20 | 2.55 ± 0.72 | 16.77 ± 3.71 | 10.30 ± 0.38 | 141.33 ± 5.47 | 54.02 ± 2.85 | 52.48 ± 0.94 | 13.70 ± 0.37 | 261.50 ± 9.77 | 15.40 ± 0.36 | 1188.33 ± 291.95 | 6.77 ± 0.30 | 17.32 ± 0.18 |
| H2 | 6.45 ± 2.33 | 5.43 ± 2.09 | 0.17 ± 0.05 | 0.85 ± 0.26 | 83.90 ± 3.13 | 2.68 ± 0.84 | 13.42 ± 2.41 | 10.56 ± 0.34 | 146.67 ± 6.53 | 54.80 ± 2.10 | 52.00 ± 1.15 | 13.85 ± 0.55 | 267.33 ± 13.82 | 16.07 ± 0.74 | 1284.83 ± 416.37 | 6.53 ± 0.69 | 17.02 ± 0.25 |
| M1 | 6.48 ± 2.42 | 5.37 ± 2.17 | 0.20 ± 0.09 | 0.92 ± 0.27 | 81.97 ± 3.59 | 3.18 ± 1.00 | 14.85 ± 3.09 | 9.70 ± 0.32 | 140.50 ± 3.21 | 53.42 ± 1.86 | 55.12 ± 0.80 | 14.43 ± 0.20 | 263.00 ± 6.20 | 15.88 ± 0.75 | 1251.33 ± 279.63 | 6.88 ± 0.47 | 17.32 ± 0.21 |
| M2 | 6.68 ± 2.17 | 5.37 ± 1.80 | 0.13 ± 0.08 | 1.18 ± 0.32 | 79.85 ± 1.93 | 2.53 ± 0.56 | 17.62 ± 1.90 | 9.98 ± 0.46 | 143.00 ± 6.16 | 55.38 ± 2.39 | 55.53 ± 0.39 | 14.28 ± 0.26 | 257.67 ± 3.88 | 15.25 ± 0.29 | 1241.17 ± 253.09 | 6.95 ± 0.46 | 17.30 ± 0.33 |
| L1 | 6.43 ± 2.73 | 5.30 ± 2.36 | 0.15 ± 0.08 | 0.98 ± 0.34 | 81.32 ± 3.21 | 2.63 ± 0.81 | 16.05 ± 2.55 | 10.40 ± 0.76 | 140.17 ± 10.26 | 54.77 ± 4.38 | 52.67 ± 0.56 | 13.42 ± 0.17 | 255.67 ± 3.50 | 16.20 ± 0.41 | 1274.00 ± 178.81 | 6.62 ± 0.55 | 17.00 ± 0.20 |
| L2 | 6.15 ± 2.21 | 5.12 ± 2.09 | 0.13 ± 0.05 | 0.90 ± 0.17 | 81.33 ± 4.52 | 2.87 ± 0.74 | 15.80 ± 3.90 | 9.73 ± 0.17 | 137.67 ± 5.92 | 52.12 ± 1.16 | 53.63 ± 1.73 | 14.27 ± 0.48 | 266.83 ± 6.68 | 16.00 ± 0.68 | 1239.33 ± 347.23 | 6.65 ± 0.32 | 17.32 ± 0.33 |
| Male | |||||||||||||||||
| NC | 7.38 ± 2.68 | 5.87 ± 2.29 | 0.18 ± 0.08 | 1.33 ± 0.36 | 78.80 ± 2.96 | 2.48 ± 0.36 | 18.72 ± 2.70 | 10.38 ± 0.79 | 139.50 ± 12.86 | 54.00 ± 3.34 | 52.17 ± 2.41 | 13.38 ± 0.29 | 257.50 ± 13.43 | 17.32 ± 2.55 | 1327.50 ± 239.63 | 6.57 ± 0.27 | 17.07 ± 0.43 |
| H1 | 9.88 ± 2.47 | 8.13 ± 2.06 | 0.18 ± 0.10 | 1.57 ± 0.45 | 81.87 ± 3.06 | 2.18 ± 0.47 | 15.95 ± 2.77 | 9.86 ± 0.90 | 137.00 ± 13.81 | 52.25 ± 3.95 | 53.15 ± 2.27 | 13.85 ± 0.27 | 261.50 ± 15.42 | 17.27 ± 1.29 | 1421.67 ± 437.78 | 6.63 ± 0.36 | 17.37 ± 0.36 |
| H2 | 9.83 ± 1.93 | 7.97 ± 1.71 | 0.18 ± 0.04 | 1.68 ± 0.26 | 80.50 ± 2.30 | 2.05 ± 0.31 | 17.45 ± 2.03 | 10.22 ± 0.40 | 143.17 ± 4.54 | 51.27 ± 3.07 | 50.17 ± 1.31 | 13.90 ± 0.27 | 279.50 ± 11.91 | 19.82 ± 1.71 | 1299.83 ± 227.69 | 6.37 ± 0.30 | 16.87 ± 0.45 |
| M1 | 10.17 ± 1.52 | 8.74 ± 1.03 | 0.37 ± 0.14 | 1.95 ± 0.46 | 79.85 ± 2.99 | 3.15 ± 0.96 | 17.00 ± 2.21 | 10.07 ± 0.57 | 136.00 ± 7.51 | 52.48 ± 3.91 | 52.18 ± 2.15 | 13.45 ± 0.16 | 259.17 ± 13.45 | 17.03 ± 1.88 | 1546.75 ± 167.92 | 6.52 ± 0.31 | 17.23 ± 0.45 |
| M2 | 9.70 ± 3.68 | 7.75 ± 3.17 | 0.25 ± 0.10 | 1.70 ± 0.52 | 79.23 ± 3.84 | 2.57 ± 0.64 | 18.20 ± 3.35 | 10.26 ± 0.69 | 142.17 ± 11.32 | 51.33 ± 5.09 | 50.05 ± 2.42 | 13.82 ± 0.26 | 277.33 ± 11.38 | 19.65 ± 2.26 | 1485.00 ± 176.13 | 6.67 ± 0.34 | 16.92 ± 0.56 |
| L1 | 8.77 ± 2.72 | 7.17 ± 2.16 | 0.17 ± 0.08 | 1.43 ± 0.52 | 82.12 ± 2.41 | 1.88 ± 0.45 | 16.00 ± 2.28 | 10.33 ± 0.53 | 141.50 ± 5.68 | 50.03 ± 4.34 | 48.43 ± 2.37 | 13.67 ± 0.27 | 275.25 ± 17.25 | 19.53 ± 2.33 | 1328.17 ± 317.71 | 6.38 ± 0.37 | 16.43 ± 0.55 |
| Ref | 0.8–10.6 | 0.6–8.9 | 0.04–1.4 | 0.23–3.6 | 40–92 | 0.9–18 | 6.5–50 | 6.5–11.5 | 110–165 | 35–55 | 41–55 | 13–18 | 250–360 | 12–20 | 400–1600 | 4.0–6.2 | 12.0–17.5 |
| Treatment # | ALT | AST | BUN | CRE | TC | TG | CK | Na | K |
|---|---|---|---|---|---|---|---|---|---|
| Female | |||||||||
| NC | 35.78 ± 3.30 | 160.68 ± 36.35 | 8.74 ± 0.49 | 13.00 ± 0.50 | 2.88 ± 0.08 | 0.44 ± 0.19 | 1235.53 ± 526.45 | 155.67 ± 0.81 | 7.09 ± 0.31 |
| H1 | 34.40 ± 4.60 | 175.03 ± 42.05 | 8.46 ± 0.22 | 13.28 ± 0.86 | 2.75 ± 0.33 | 0.39 ± 0.07 | 1664.27 ± 636.88 | 157.70 ± 2.09 | 7.29 ± 0.80 |
| H2 | 34.37 ± 3.13 | 170.40 ± 30.19 | 8.74 ± 0.35 | 12.80 ± 0.95 | 2.57 ± 0.11 | 0.22 ± 0.08 | 1443.42 ± 515.52 | 157.80 ± 1.11 | 7.08 ± 0.12 |
| M1 | 31.32 ± 3.04 | 173.12 ± 36.41 | 8.16 ± 0.71 | 13.10 ± 0.78 | 2.59 ± 0.35 | 0.30 ± 0.12 | 1707.08 ± 463.81 | 157.75 ± 0.42 | 6.66 ± 0.19 |
| M2 | 30.02 ± 2.26 | 146.05 ± 20.65 | 8.18 ± 0.92 | 12.60 ± 0.81 | 2.57 ± 0.12 | 0.27 ± 0.07 | 1342.58 ± 388.33 | 156.60 ± 1.70 | 6.79 ± 0.40 |
| L1 | 34.95 ± 3.67 | 229.67 ± 88.22 | 7.60 ± 0.68 | 13.22 ± 0.29 | 2.51 ± 0.16 | 0.24 ± 0.08 | 2025.22 ± 480.65 | 155.22 ± 0.84 | 6.54 ± 0.25 |
| L2 | 32.52 ± 4.36 | 192.38 ± 64.60 | 7.60 ± 0.45 | 12.72 ± 1.06 | 2.60 ± 0.15 | 0.26 ± 0.11 | 1754.78 ± 600.88 | 155.16 ± 1.74 | 6.59 ± 0.46 |
| Male | |||||||||
| NC | 40.95 ± 5.83 | 169.57 ± 25.22 | 11.05 ± 0.82 | 11.90 ± 0.94 | 3.00 ± 0.21 | 0.53 ± 0.10 | 1745.62 ± 616.85 | 158.73 ± 1.87 | 7.90 ± 0.48 |
| H1 | 36.40 ± 4.63 | 140.02 ± 30.40 | 10.08 ± 0.59 | 12.80 ± 0.54 | 3.01 ± 0.22 | 0.61 ± 0.08 | 1242.08 ± 469.88 | 156.60 ± 1.17 | 7.73 ± 0.63 |
| H2 | 33.77 ± 4.18 | 165.02 ± 38.64 | 10.94 ± 1.18 | 13.02 ± 1.38 | 2.99 ± 0.13 | 0.51 ± 0.16 | 1698.65 ± 535.90 | 157.87 ± 1.10 | 7.16 ± 0.25 |
| M1 | 37.82 ± 4.60 | 159.08 ± 25.02 | 8.95 ± 1.02 | 12.57 ± 0.56 | 3.03 ± 0.15 | 0.68 ± 0.30 | 1509.68 ± 413.40 | 156.88 ± 1.07 | 7.30 ± 0.81 |
| M2 | 34.67 ± 3.03 | 156.87 ± 18.80 | 9.95 ± 1.12 | 12.48 ± 0.92 | 2.95 ± 0.18 | 0.53 ± 0.18 | 1735.52 ± 487.55 | 156.78 ± 1.25 | 7.19 ± 0.31 |
| L1 | 34.96 ± 4.91 | 133.38 ± 22.16 | 9.95 ± 1.81 | 13.52 ± 4.25 | 3.15 ± 0.13 | 0.47 ± 0.07 | 1666.77 ± 529.52 | 156.45 ± 0.24 | 6.87 ± 0.37 |
| L2 | 35.00 ± 9.28 | 168.70 ± 35.61 | 11.62 ± 2.47 | 15.04 ± 3.99 | 2.77 ± 0.35 | 0.42 ± 0.18 | 2096.00 ± 809.74 | 156.90 ± 1.27 | 7.19 ± 0.73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, X.; Zhou, X.; Lu, Y.; Yuan, Z.; Lan, R.; Du, Y.; Liu, L.; Xu, J. Safety Assessment and Probiotic Potential of a Novel Species Lactobacillus xujianguonis. Nutrients 2025, 17, 3474. https://doi.org/10.3390/nu17213474
Lin X, Zhou X, Lu Y, Yuan Z, Lan R, Du Y, Liu L, Xu J. Safety Assessment and Probiotic Potential of a Novel Species Lactobacillus xujianguonis. Nutrients. 2025; 17(21):3474. https://doi.org/10.3390/nu17213474
Chicago/Turabian StyleLin, Xiaoying, Xiaohui Zhou, Yao Lu, Zheyu Yuan, Ruiting Lan, Ying Du, Liyun Liu, and Jianguo Xu. 2025. "Safety Assessment and Probiotic Potential of a Novel Species Lactobacillus xujianguonis" Nutrients 17, no. 21: 3474. https://doi.org/10.3390/nu17213474
APA StyleLin, X., Zhou, X., Lu, Y., Yuan, Z., Lan, R., Du, Y., Liu, L., & Xu, J. (2025). Safety Assessment and Probiotic Potential of a Novel Species Lactobacillus xujianguonis. Nutrients, 17(21), 3474. https://doi.org/10.3390/nu17213474






