Influence of Intravenous Magnesium Sulfate Infusion on the Subjective Postoperative Quality of Recovery: A Meta-Analysis of Randomized Controlled Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source and Protocol Registration
2.2. Selection Criteria
2.3. Data Extraction
2.4. Outcomes and Definition
2.5. Quality Assessment
2.6. Certainty of Evidence Assessment
2.7. Statistical Analysis
3. Results
3.1. Selection and Characteristics of Studies
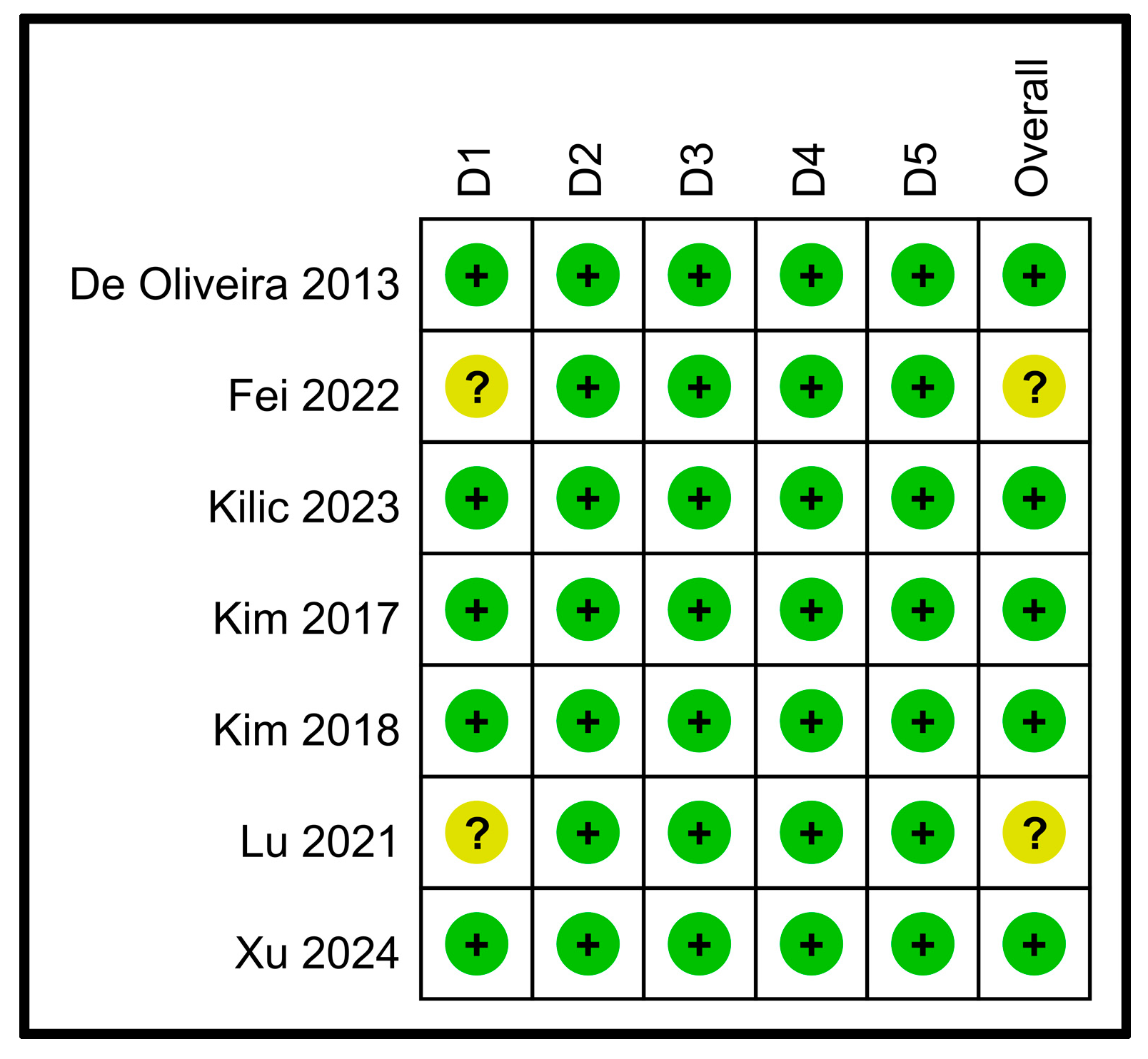
3.2. Quality of Studies
3.3. Outcomes
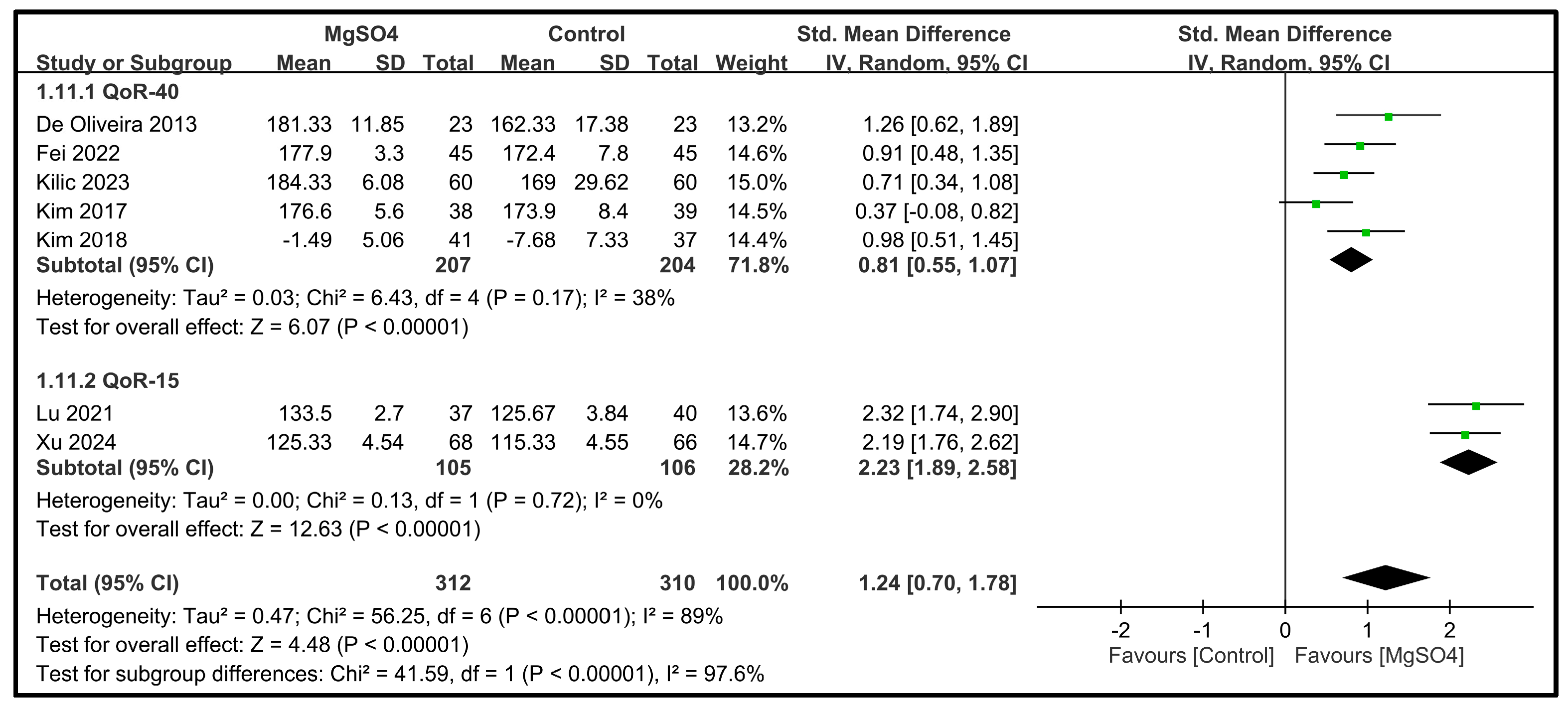
3.3.1. Primary Outcomes—Global QoR Scores on POD 1
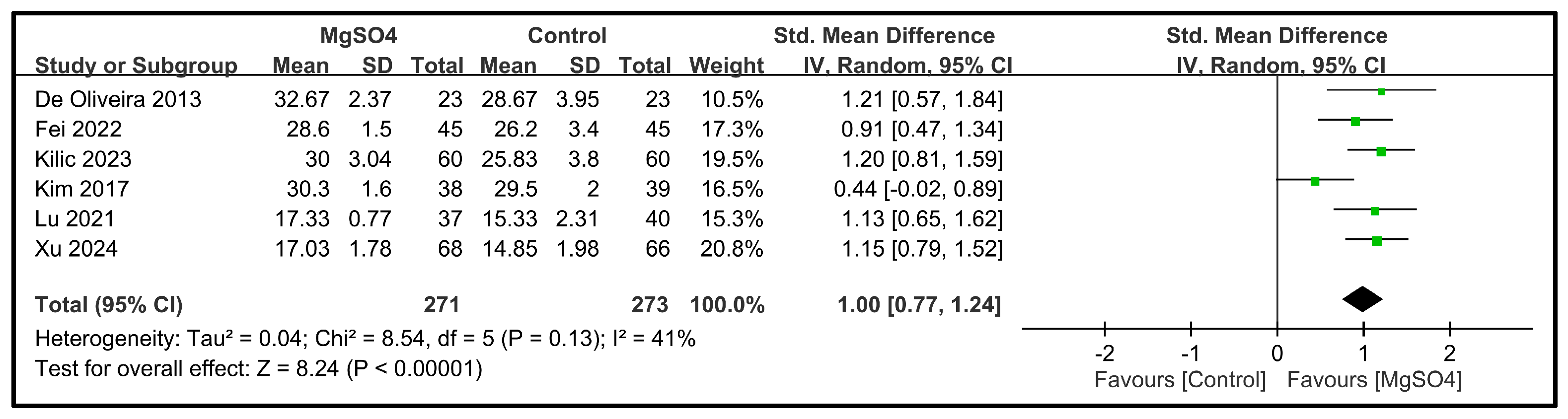
3.3.2. Secondary Outcomes—QoR Dimensions
3.3.3. Secondary Outcome—Intraoperative Opioid Use and Other Recovery Characteristics
3.3.4. Sensitivity Analysis
3.3.5. Certainty of Evidence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Royse, C.F.; Chung, F.; Newman, S.; Stygall, J.; Wilkinson, D.J. Predictors of patient satisfaction with anaesthesia and surgery care: A cohort study using the Postoperative Quality of Recovery Scale. Eur. J. Anaesthesiol. 2013, 30, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Myles, P.S.; Reeves, M.D.; Anderson, H.; Weeks, A.M. Measurement of quality of recovery in 5672 patients after anaesthesia and surgery. Anaesth. Intensive Care 2000, 28, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Bowyer, A.; Royse, C. A matter of perspective—Objective versus subjective outcomes in the assessment of quality of recovery. Best Pract. Res. Clin. Anaesthesiol. 2018, 32, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Qiu, Y.; Fu, Y.; Liu, J. Evaluation of postoperative recovery: Past, present and future. Postgrad. Med. J. 2023, 99, 808–814. [Google Scholar] [CrossRef]
- Choi, J.Y.; Lee, H.S.; Kim, J.Y.; Han, D.W.; Yang, J.Y.; Kim, M.J.; Song, Y. Comparison of remimazolam-based and propofol-based total intravenous anesthesia on postoperative quality of recovery: A randomized non-inferiority trial. J. Clin. Anesth. 2022, 82, 110955. [Google Scholar] [CrossRef] [PubMed]
- Kukreja, P.; Uppal, V.; Kofskey, A.M.; Feinstein, J.; Northern, T.; Davis, C.; Morgan, C.J.; Kalagara, H. Quality of recovery after pericapsular nerve group (PENG) block for primary total hip arthroplasty under spinal anaesthesia: A randomised controlled observer-blinded trial. Br. J. Anaesth. 2023, 130, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Campfort, M.; Cayla, C.; Lasocki, S.; Rineau, E.; Léger, M. Early quality of recovery according to QoR-15 score is associated with one-month postoperative complications after elective surgery. J. Clin. Anesth. 2022, 78, 110638. [Google Scholar] [CrossRef] [PubMed]
- Le Bescond, V.; Petit-Phan, J.; Campfort, M.; Nicolleau, C.; Conté, M.; Bouhours, G.; Rony, L.; Lasocki, S.; Léger, M. Validation of the postoperative Quality of Recovery-15 questionnaire after emergency surgery and association with quality of life at three months. Can. J. Anaesth. 2024, 71, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Kinugasa, Y.; Ida, M.; Nakatani, S.; Uyama, K.; Kawaguchi, M. Quality of recovery in hospital and disability-free survival at three months after major abdominal surgery. Korean J. Anesthesiol. 2023, 76, 567–574. [Google Scholar] [CrossRef]
- Myles, P.S.; Shulman, M.A.; Reilly, J.; Kasza, J.; Romero, L. Measurement of quality of recovery after surgery using the 15-item quality of recovery scale: A systematic review and meta-analysis. Br. J. Anaesth. 2022, 128, 1029–1039. [Google Scholar] [CrossRef]
- Gornall, B.F.; Myles, P.S.; Smith, C.L.; Burke, J.A.; Leslie, K.; Pereira, M.J.; Bost, J.E.; Kluivers, K.B.; Nilsson, U.G.; Tanaka, Y.; et al. Measurement of quality of recovery using the QoR-40: A quantitative systematic review. Br. J. Anaesth. 2013, 111, 161–169. [Google Scholar] [CrossRef]
- Tang, L.; Sun, Y.; Hao, X.; Sun, X.; Xie, C.; Wang, T.; Hu, C.; Lu, Y.; Liu, X. Effect of general anaesthesia with remimazolam versus propofol on postoperative quality of recovery in patients undergoing ambulatory arthroscopic meniscus repair: A randomised clinical trial. BJA Open 2023, 8, 100237. [Google Scholar] [CrossRef] [PubMed]
- Carella, M.; Beck, F.; Piette, N.; Lecoq, J.P.; Bonhomme, V.L. Effect of preoperative warming on intraoperative hypothermia and postoperative functional recovery in total hip arthroplasty: A randomized clinical trial. Minerva Anestesiol. 2024, 90, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Zhou, Y.; Zheng, X.; Zhou, W.; Gu, Y.; Jiang, Z.; Yao, Y.; Wei, W. Effects of S-ketamine added to patient-controlled analgesia on early postoperative pain and recovery in patients undergoing thoracoscopic lung surgery: A randomized double-blinded controlled trial. J. Clin. Anesth. 2024, 92, 111299. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Lin, C.; He, Q.; Gao, H.; Jin, L.; Zheng, X. Ultrasound-guided bilateral superficial cervical plexus blocks enhance the quality of recovery in patients undergoing thyroid cancer surgery: A randomized controlled trial. J. Clin. Anesth. 2020, 61, 109651. [Google Scholar] [CrossRef] [PubMed]
- Mayer, M.L.; Westbrook, G.L.; Guthrie, P.B. Voltage-dependent block by Mg2+ of NMDA responses in spinal cord neurones. Nature 1984, 309, 261–263. [Google Scholar] [CrossRef] [PubMed]
- Begon, S.; Pickering, G.; Eschalier, A.; Mazur, A.; Rayssiguier, Y.; Dubray, C. Role of spinal NMDA receptors, protein kinase C and nitric oxide synthase in the hyperalgesia induced by magnesium deficiency in rats. Br. J. Pharmacol. 2001, 134, 1227–1236. [Google Scholar] [CrossRef] [PubMed]
- Mantyh, P.W.; Allen, C.J.; Rogers, S.; DeMaster, E.; Ghilardi, J.R.; Mosconi, T.; Kruger, L.; Mannon, P.J.; Taylor, I.L.; Vigna, S.R. Some sensory neurons express neuropeptide Y receptors: Potential paracrine inhibition of primary afferent nociceptors following peripheral nerve injury. J. Neurosci. 1994, 14, 3958–3968. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.; de Melo Nunes, R.; de Freitas Carvalho, W.V.; Girão, V.C.C.; Rocha, F.A.C. Systemic and local antiinflammatory effect of magnesium chloride in experimental arthritis. Adv. Rheumatol. 2024, 64, 6. [Google Scholar] [CrossRef]
- Albrecht, E.; Kirkham, K.; Liu, S.; Brull, R. Peri-operative intravenous administration of magnesium sulphate and postoperative pain: A meta-analysis. Anaesthesia 2013, 68, 79–90. [Google Scholar] [CrossRef]
- Choi, G.J.; Kim, Y.I.; Koo, Y.H.; Oh, H.-C.; Kang, H. Perioperative magnesium for postoperative analgesia: An umbrella review of systematic reviews and updated meta-analysis of randomized controlled trials. J. Pers. Med. 2021, 11, 1273. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.-H.; Kang, M.-H.; Park, K.-S.; Do, S.-H. Effects of magnesium sulphate on intraoperative anaesthetic requirements and postoperative analgesia in gynaecology patients receiving total intravenous anaesthesia. Br. J. Anaesth. 2008, 100, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Zhang, Y.; Li, Q. Magnesium sulfate reduces postoperative pain in women with cesarean section: A meta-analysis of randomized controlled trials. Pain Pract. 2022, 22, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, H.; Nakajima, D.; Mihara, T.; Sato, H.; Goto, T. Effectiveness of Magnesium in Preventing Shivering in Surgical Patients: A Systematic Review and Meta-analysis. Anesth. Analg. 2019, 129, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Fairley, J.L.; Zhang, L.; Glassford, N.J.; Bellomo, R. Magnesium status and magnesium therapy in cardiac surgery: A systematic review and meta-analysis focusing on arrhythmia prevention. J. Crit. Care 2017, 42, 69–77. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, G.S.; Bialek, J.; Fitzgerald, P.; Kim, J.Y.; McCarthy, R.J. Systemic magnesium to improve quality of post-surgical recovery in outpatient segmental mastectomy: A randomized, double-blind, placebo-controlled trial. Magnes. Res. 2013, 26, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Kim, M.S.; Lee, J.H.; Kim, S.T.; Lee, J.R. Intravenously Administered Lidocaine and Magnesium During Thyroid Surgery in Female Patients for Better Quality of Recovery After Anesthesia. Anesth. Analg. 2018, 127, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Lee, K.Y.; Park, S.; Kim, S.I.; Park, H.S.; Yoo, Y.C. Effects of systemic lidocaine versus magnesium administration on postoperative functional recovery and chronic pain in patients undergoing breast cancer surgery: A prospective, randomized, double-blind, comparative clinical trial. PLoS ONE 2017, 12, e0173026. [Google Scholar] [CrossRef] [PubMed]
- Hung, K.C.; Kao, C.L.; Ho, C.N.; Hsing, C.H.; Chang, Y.J.; Wang, L.K.; Liao, S.W.; Chen, I.W. The impact of perioperative ketamine or esketamine on the subjective quality of recovery after surgery: A meta-analysis of randomised controlled trials. Br. J. Anaesth. 2024, 132, 1293–1303. [Google Scholar] [CrossRef]
- Sterne, J.A.; Sutton, A.J.; Ioannidis, J.P.; Terrin, N.; Jones, D.R.; Lau, J.; Carpenter, J.; Rücker, G.; Harbord, R.M.; Schmid, C.H.; et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011, 343, d4002. [Google Scholar] [CrossRef]
- Fei, F.; Xiongxiong, P. Effect of intravenous infusion of magnesium sulfate or lidocaine during anesthesia on postoperative recovery quality in early stage in patients undergoing upper airway surgery. J. Clin. Med. Pract. 2022, 26, 119–124. [Google Scholar]
- Kilic, K.; Sakat, M.S.; Sahin, A.; Ahiskalioglu, E.O.; Altunok, H. Efficacy of intravenous magnesium sulfate infusion on postoperative pain and quality of recovery for septorhinoplasty: A randomized controlled study. Acta Otolaryngol. 2023, 143, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Wang, J.F.; Guo, C.L.; Yin, Q.; Cheng, W.; Qian, B. Intravenously injected lidocaine or magnesium improves the quality of early recovery after laparoscopic cholecystectomy: A randomised controlled trial. Eur. J. Anaesthesiol. 2021, 38, S1–S8. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Hao, C.; Wang, X.; Du, J.; Zhang, T.; Zhang, X. Effect of Intraoperative infusion Magnesium Sulfate Infusion on Postoperative Quality of Recovery in Patients Undergoing Total Knee Arthroplasty: A Prospective, Double-Blind, Randomized Controlled Trial. Drug Des. Dev. Ther. 2024, 18, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Hung, K.C.; Chu, C.C.; Hsing, C.H.; Chang, Y.P.; Li, Y.Y.; Liu, W.C.; Chen, I.W.; Chen, J.Y.; Sun, C.K. Association between perioperative intravenous lidocaine and subjective quality of recovery: A meta-analysis of randomized controlled trials. J. Clin. Anesth. 2021, 75, 110521. [Google Scholar] [CrossRef] [PubMed]
- Miao, M.; Xu, Y.; Li, B.; Chang, E.; Zhang, L.; Zhang, J. Intravenous administration of dexmedetomidine and quality of recovery after elective surgery in adult patients: A meta-analysis of randomized controlled trials. J. Clin. Anesth. 2020, 65, 109849. [Google Scholar] [CrossRef] [PubMed]
- Hung, K.C.; Ko, C.C.; Hsu, C.W.; Pang, Y.L.; Chen, J.Y.; Sun, C.K. Association of peripheral nerve blocks with patient-reported quality of recovery in female patients receiving breast cancer surgery: A systematic review and meta-analysis of randomized controlled studies. Can. J. Anaesth. 2022, 69, 1288–1299. [Google Scholar] [CrossRef]
- Xu, M.; Zhang, G.; Tang, Y.; Wang, R.; Yang, J. Impact of Regional Anesthesia on Subjective Quality of Recovery in Patients Undergoing Thoracic Surgery: A Systematic Review and Meta-Analysis. J. Cardiothorac. Vasc. Anesth. 2023, 37, 1744–1750. [Google Scholar] [CrossRef]
- Zhang, M.; Zhang, H.; Li, P.; Li, J. Effect of transcutaneous electrical acupoint stimulation on the quality of postoperative recovery: A meta-analysis. BMC Anesthesiol. 2024, 24, 104. [Google Scholar] [CrossRef]
- Seyfi, S.; Amiri, H.A.; Banihashem, N.; Ezoji, K.; Ziaei, S.G. An Investigation Into Quality of Recovery After General Anesthesia With Isoflurane and Propofol in Patients Undergoing Abdominal Hysterectomy. Asian J. Anesthesiol. 2022, 60, 109–116. [Google Scholar] [CrossRef]
- Shui, M.; Xue, Z.; Miao, X.; Wei, C.; Wu, A. Intravenous versus inhalational maintenance of anesthesia for quality of recovery in adult patients undergoing non-cardiac surgery: A systematic review with meta-analysis and trial sequential analysis. PLoS ONE 2021, 16, e0254271. [Google Scholar] [CrossRef] [PubMed]
- Ng, K.T.; Yap, J.L.L.; Izham, I.N.; Teoh, W.Y.; Kwok, P.E.; Koh, W.J. The effect of intravenous magnesium on postoperative morphine consumption in noncardiac surgery: A systematic review and meta-analysis with trial sequential analysis. Eur. J. Anaesthesiol. EJA 2020, 37, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Avci, Y.; Rajarathinam, M.; Kalsekar, N.; Tawfic, Q.; Krause, S.; Nguyen, D.; Liu, E.; Nagappa, M.; Subramani, Y. Unravelling the analgesic effects of perioperative magnesium in general abdominal surgery: A systematic review and meta-analysis of randomized controlled trials. Braz. J. Anesthesiol. 2024, 74, 844524. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.C.; Pan, P.T.; Chiu, H.Y.; Huang, C.J. Neuraxial magnesium sulfate improves postoperative analgesia in Cesarean section delivery women: A meta-analysis of randomized controlled trials. Asian J. Anesthesiol. 2017, 55, 56–67. [Google Scholar] [CrossRef]
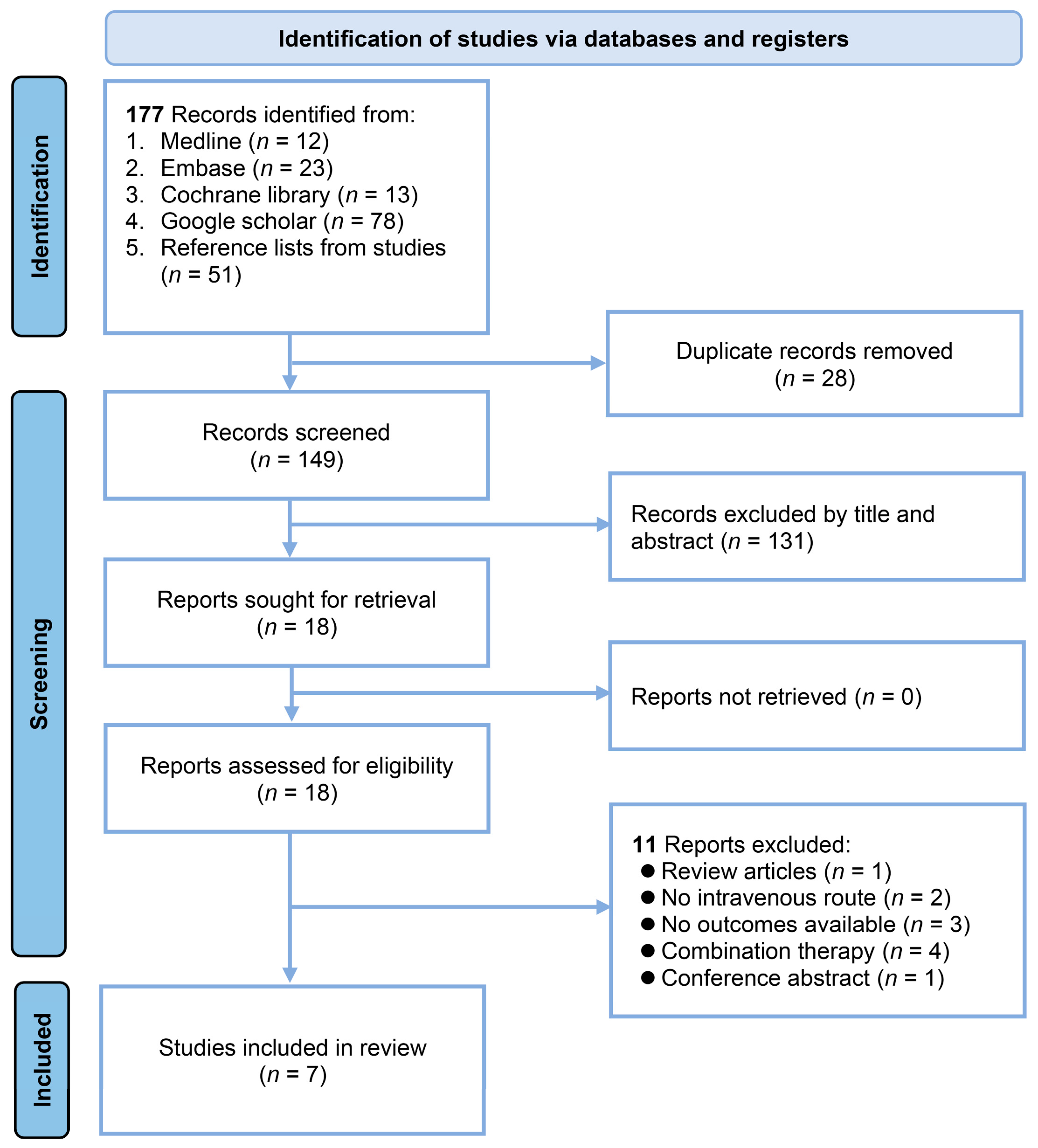



| 1 | (“(operative or surgical) adj4 (procedure* or technique*)” or “surger*” or “operation*”).mp. |
| 2 | exp “Surgical Procedures, Operative”/ |
| 3 | (“Magnesium Hydroxide” or “Magnesium Chloride” or “Magnesium Sulfate” or “Magnesium” or “Magnesium Oxide” or “Magnesium Silicates” or “MgSO4”).mp. |
| 4 | exp “Magnesium Sulfate”/ |
| 5 | (“quality of recovery score” or “QoR-40” or “Quality of Recovery-40” or “Quality of Recovery-15” or “QoR-15” or “Quality of Recovery scale” or “quality of recovery”).mp. |
| 6 | (1 or 2) and (3 or 4) and 5 |
| 7 | 6 and (((randomized controlled trial or controlled clinical trial).pt. or randomi*ed.ab. or placebo.ab. or drug therapy.fs. or randomly.ab. or trial.ab. or groups.ab.) not (exp animals/ not humans.sh.)) |
| Age (Years) | Male (%) | BMI (kg/m2) | ASA | N | IV Magnesium Sulfate (Bolus, Infusion) | Control | Procedures | Surgical Duration (Minutes) | QoR | Country | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| De Oliveira 2013 [26] | 53/52 | 0 | 24/25 | I–II | 46 | 50 mg/kg; 15 mg/kg/h | Saline | Mastectomy | 413/442 | 40 | USA |
| Fei 2022 [31] | 44/47 | 53/47 | 25/24 | I–II | 90 | 20 mg/kg; 20 mg/kg/h | Saline | Airway surgery | 38/35 | 40 | China |
| Kilic 2023 [32] | 18–45 † | 33/28 | na | na | 120 | 30 mg/kg; 9 mg/kg/h | Saline | Septorhinoplasty surgery | na | 40 | Turkey |
| Kim 2018 [27] | 44/45 | 0 | 58/56 | I–II | 78 | 20 mg/kg; 20 mg/kg/h | Saline | Thyroidectomy | 94/91 | 40 | Korea |
| Kim 2017 [28] | 48/49 | 0 | 56/55 | I–II | 77 | 20 mg/kg; 20 mg/kg/h | Saline | Breast cancer surgery | 105/98 | 40 | Korea |
| Lu 2021 [33] | 47/45 | 22/28 | 24/24 | I–II | 77 | 20 mg/kg; 20 mg/kg/h | Saline | Cholecystectomy | 58/59 | 15 | China |
| Xu 2024 [34] | 67/66 | 21/24 | 25/26 | II–III | 134 | 40 mg/kg; 15 mg/kg/h | Saline | Total knee replacement | 69/71 | 15 | China |
| Outcomes | N † | Participants | Certainty Assessment (Domains) | Effect Size [95% CI] | I2 | Sensitivity Analysis | Certainty | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | |||||||
| Global QoR scores | 7 | 622 |  |  |  |  | - | SMD 1.24 [0.7, 1.78] p < 0.00001 | 89% | Consistent | ⨁⨁⨁◯ Moderate |
| Pain domain | 6 | 544 |  |  |  |  | - | SMD 1.0 [0.77, 1.24] p < 0.00001 | 41% | Consistent | ⨁⨁⨁⨁ High |
| Physical comfort | 6 | 544 |  |  |  |  | - | SMD 0.85 [0.47, 1.24] p < 0.0001 | 78% | Consistent | ⨁⨁⨁◯ Moderate |
| Emotional domain | 6 | 544 |  |  |  |  | - | SMD 0.65 [0.24, 1.06] p = 0.002 | 81% | Consistent | ⨁⨁⨁◯ Moderate |
| Physical independence | 6 | 544 |  |  |  |  | - | SMD 0.43 [0.24, 0.61] p < 0.00001 | 15% | Consistent | ⨁⨁⨁◯ Moderate |
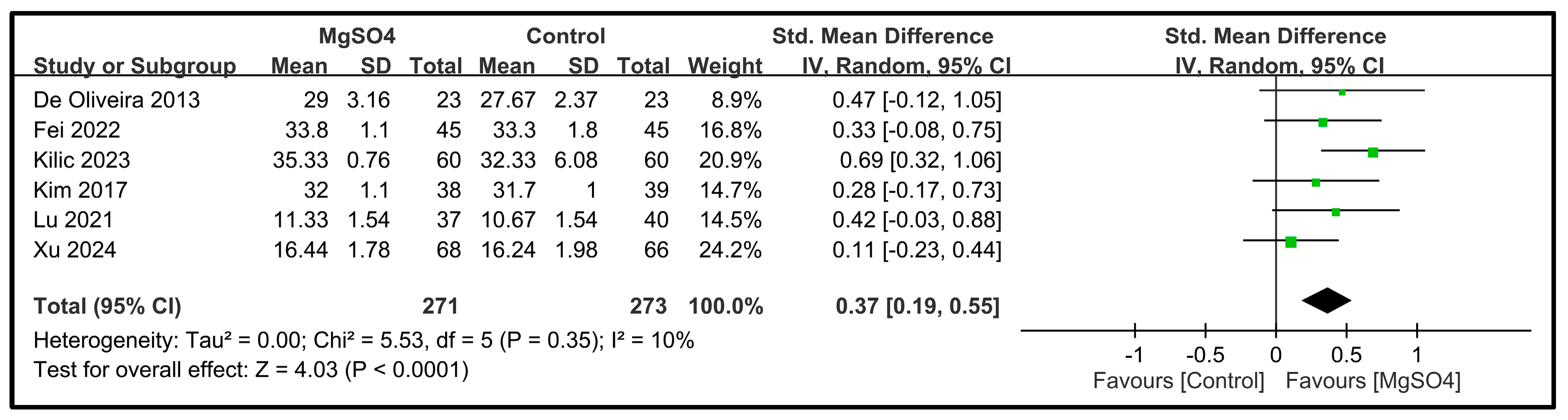
| Psychological support | 6 | 544 |  |  |  |  | - | SMD 0.37 [0.19, 0.55] p < 0.0001 | 10% | Consistent | ⨁⨁⨁◯ Moderate |
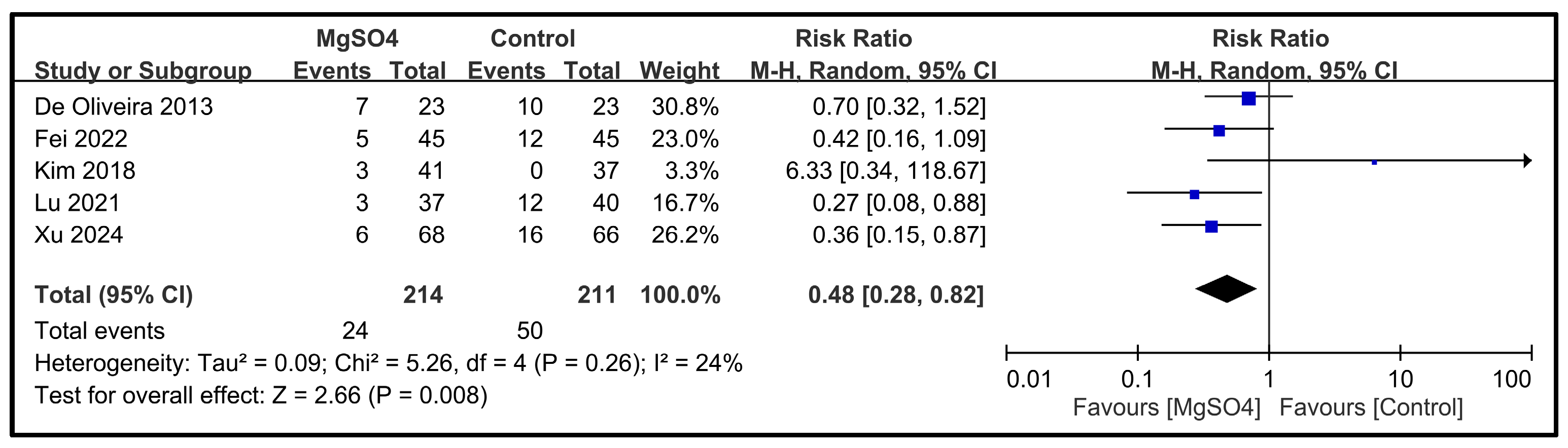
| PONV | 5 | 425 |  |  |  |  | - | RR 0.48 [0.28, 0.82] p = 0.008 | 24% | Inconsistent | ⨁⨁⨁⨁ High |
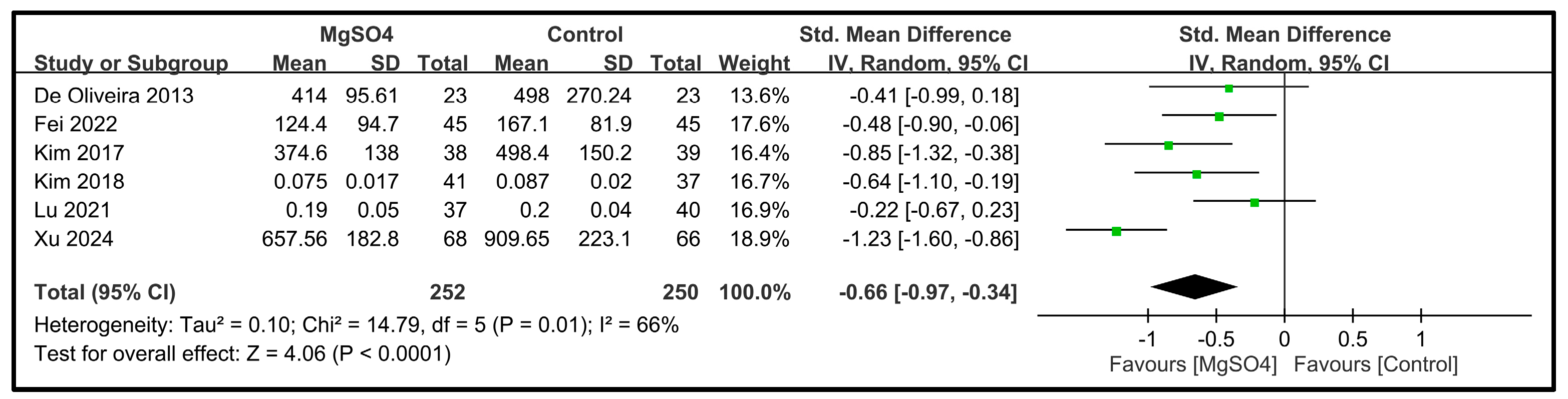
| Remifentanil | 6 | 502 |  |  |  |  | - | SMD −0.66 [−0.97, −0.34] p < 0.0001 | 66% | Consistent | ⨁⨁⨁◯ Moderate |
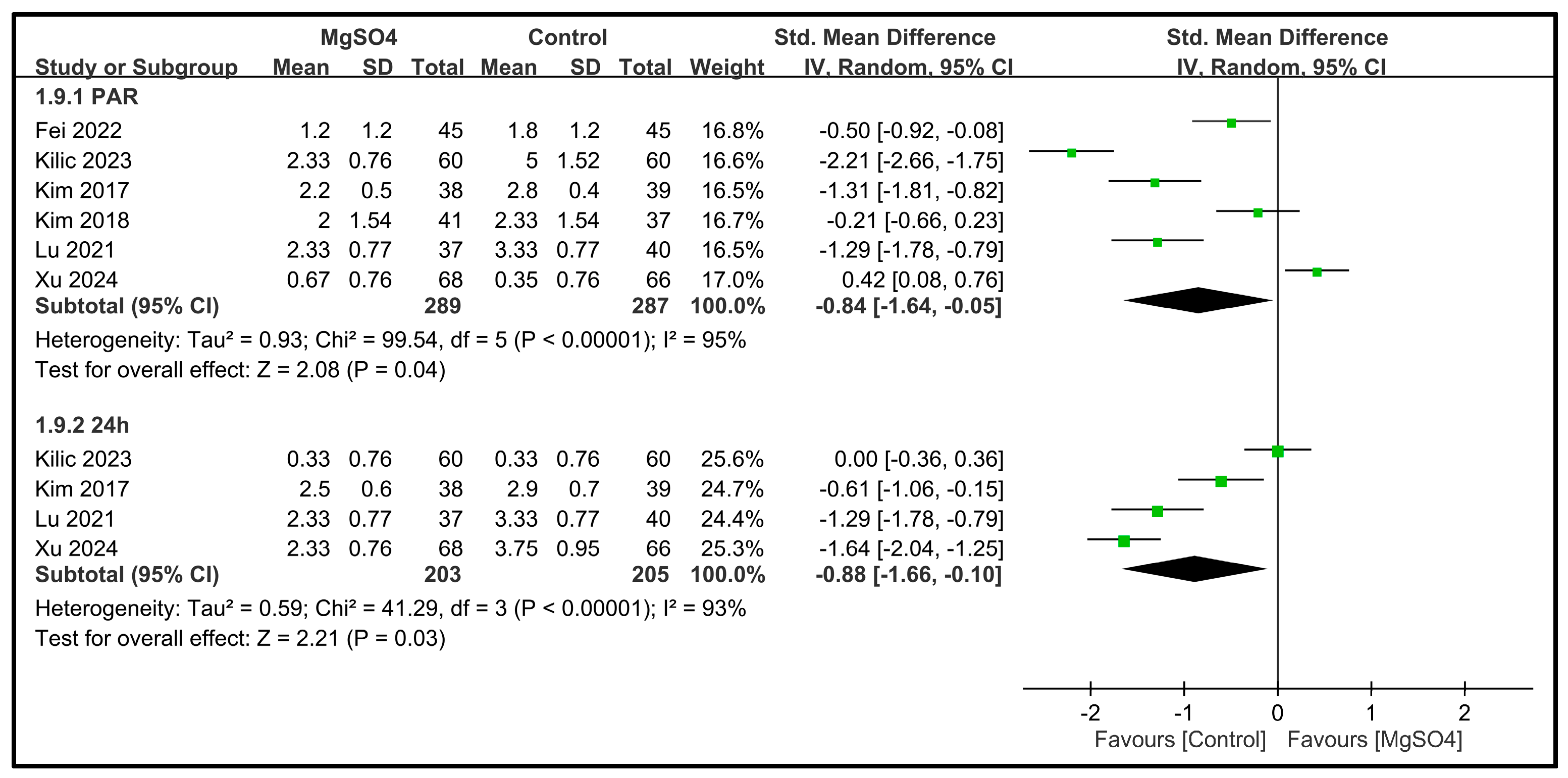
| Pain score at PACU | 6 | 576 |  |  |  |  | - | SMD −0.84 [−1.64, −0.05] p = 0.04 | 95% | Inconsistent | ⨁⨁◯◯ Low |
| Pain score at 24 h | 4 | 408 |  |  |  |  | - | SMD −0.88 [−1.66, −0.10] p = 0.03 | 93% | Inconsistent | ⨁⨁◯◯ Low |
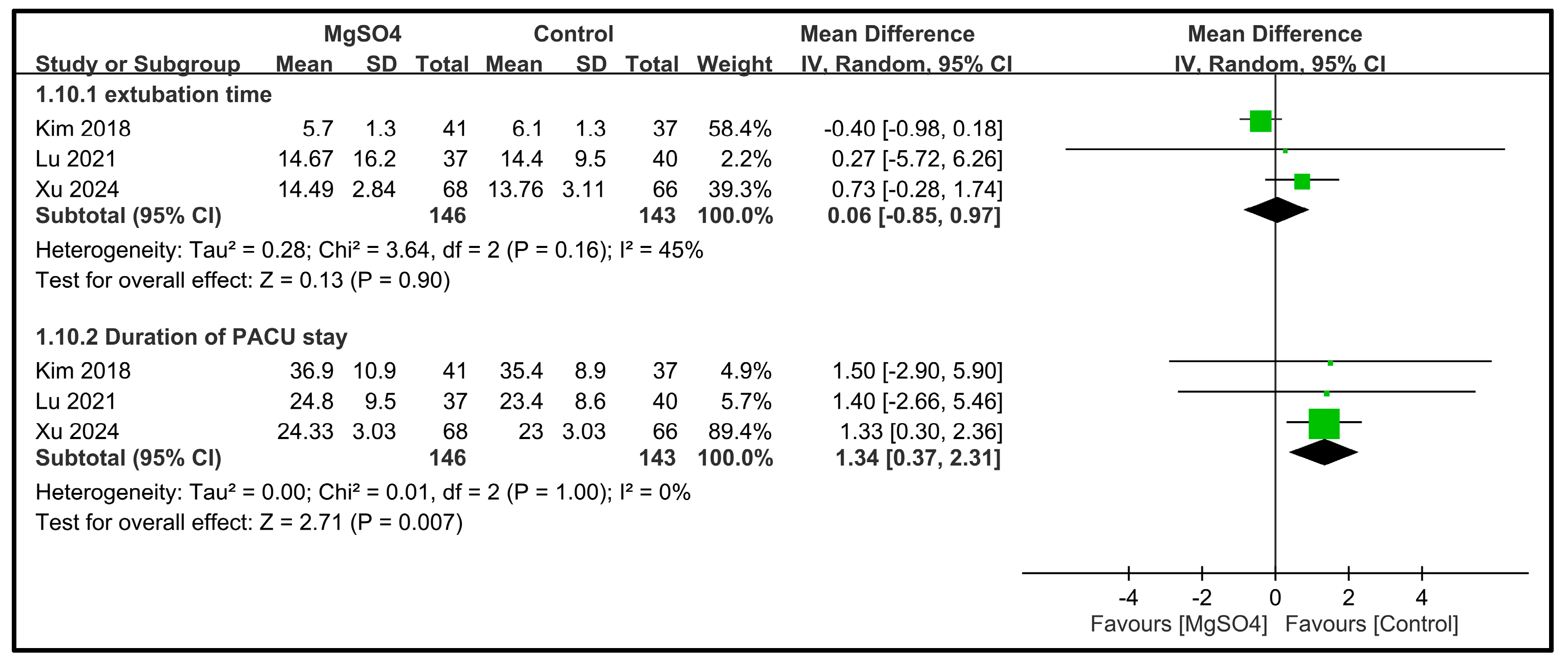
| Extubation time | 3 | 289 |  |  |  |  | - | MD 0.06 [−0.85, 0.97] p = 0.9 | 45% | Consistent | ⨁⨁⨁⨁ High |
| Duration of PACU stay | 3 | 289 |  |  |  |  | - | MD 1.34 [0.37, 2.31] p = 0.007 | 0% | Inconsistent | ⨁⨁⨁◯ Moderate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, K.-C.; Chang, L.-C.; Ho, C.-N.; Hsu, C.-W.; Wu, J.-Y.; Lin, Y.-T.; Chen, I.-W. Influence of Intravenous Magnesium Sulfate Infusion on the Subjective Postoperative Quality of Recovery: A Meta-Analysis of Randomized Controlled Trials. Nutrients 2024, 16, 2375. https://doi.org/10.3390/nu16142375
Hung K-C, Chang L-C, Ho C-N, Hsu C-W, Wu J-Y, Lin Y-T, Chen I-W. Influence of Intravenous Magnesium Sulfate Infusion on the Subjective Postoperative Quality of Recovery: A Meta-Analysis of Randomized Controlled Trials. Nutrients. 2024; 16(14):2375. https://doi.org/10.3390/nu16142375
Chicago/Turabian StyleHung, Kuo-Chuan, Li-Chen Chang, Chun-Ning Ho, Chih-Wei Hsu, Jheng-Yan Wu, Yao-Tsung Lin, and I-Wen Chen. 2024. "Influence of Intravenous Magnesium Sulfate Infusion on the Subjective Postoperative Quality of Recovery: A Meta-Analysis of Randomized Controlled Trials" Nutrients 16, no. 14: 2375. https://doi.org/10.3390/nu16142375
APA StyleHung, K.-C., Chang, L.-C., Ho, C.-N., Hsu, C.-W., Wu, J.-Y., Lin, Y.-T., & Chen, I.-W. (2024). Influence of Intravenous Magnesium Sulfate Infusion on the Subjective Postoperative Quality of Recovery: A Meta-Analysis of Randomized Controlled Trials. Nutrients, 16(14), 2375. https://doi.org/10.3390/nu16142375








