Effect of Nuts on Gastrointestinal Health
Abstract
1. Introduction
2. Food Matrix and Digestion
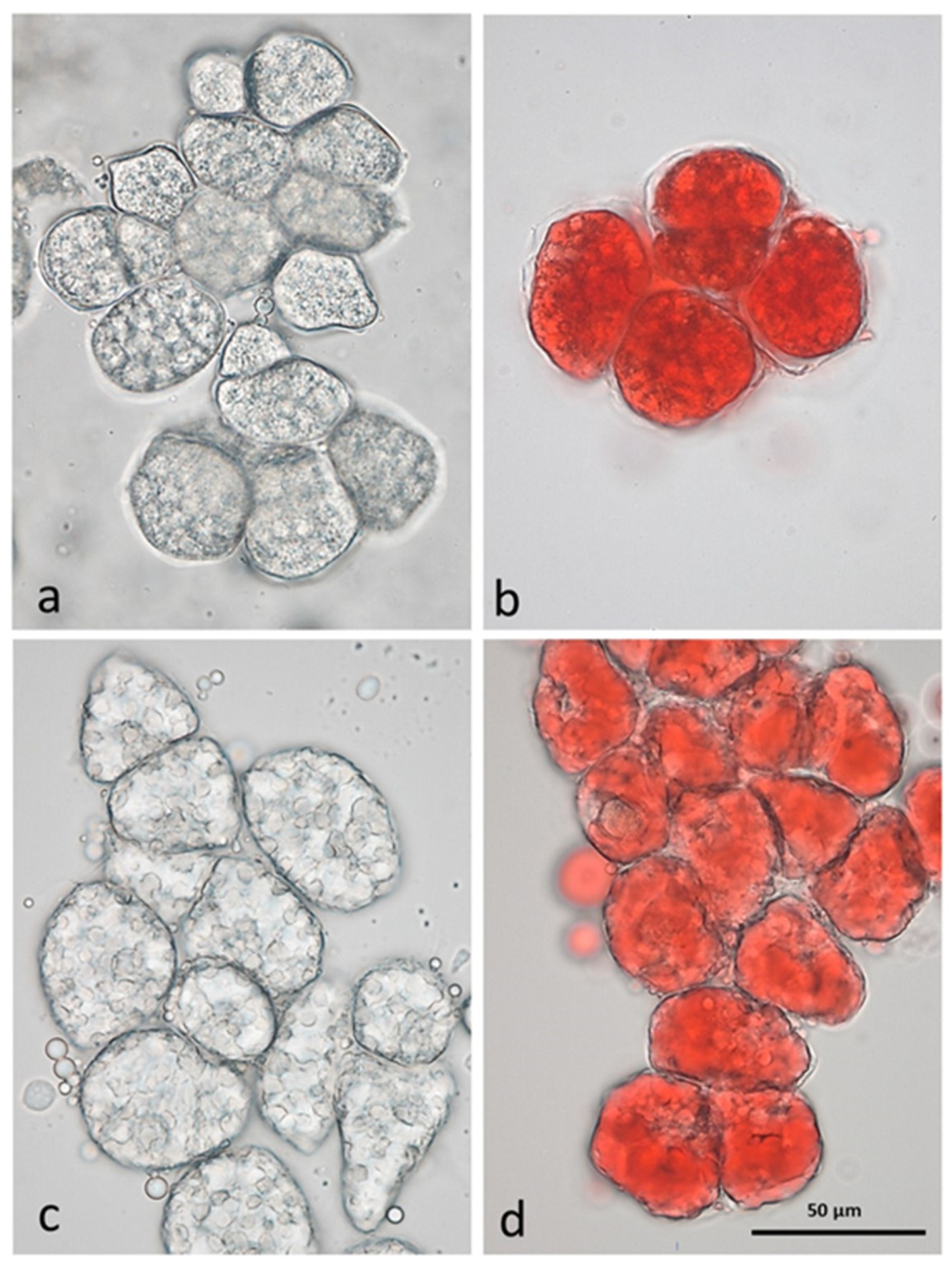
2.1. Microstructure and Cell Properties
2.2. Bioaccessibility of Nutrients and Phytochemicals in the Upper Gastrointestinal Tract
2.2.1. Nutrient Bioaccessibility
2.2.2. Phytochemicals Bioaccessibility
2.2.3. Effect of Processing and Food Matrix on Digestion
3. Effect of Nuts on Gastrointestinal Health
3.1. Microbiota Composition and Diversity
3.1.1. Changes in Relative Proportion at the Phyla Level
3.1.2. Changes in Relative Proportion at the Genus Level
3.2. Effect of Nuts on Microbial End Products
3.2.1. Short Chain Fatty Acids
3.2.2. Bile Acids
3.3. Walnut Consumption and Gastrointestinal Disease
4. Conclusions
5. Research Gaps and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alasalvar, C.; Bolling, B.W. Review of nut phytochemicals, fat-soluble bioactives, antioxidant components and health effects. Br. J. Nutr. 2015, 113, S68–S78. [Google Scholar] [CrossRef]
- Alasalvar, C.; Salas-Salvado, J.; Ros, E. Bioactives and health benefits of nuts and dried fruits. Food Chem. 2020, 314, 126192. [Google Scholar] [CrossRef] [PubMed]
- Dagbasi, A.; Lett, A.M.; Murphy, K.; Frost, G. Understanding the interplay between food structure, intestinal bacterial fermentation and appetite control. Proc. Nutr. Soc. 2020, 79, 514–530. [Google Scholar] [CrossRef] [PubMed]
- ILSI Europe. Dietary probiotics, prebiotics and the gut microflora in human health. ILSI Eur. Concise Monogr. Ser. 2022, 1–50. [Google Scholar]
- McArthur, B.M.; Higgins, K.A.; Hunter, S.R.; Mattes, R.D. Energetics of Nut Consumption. In Health Benefits of Nuts and Dried Fruits; Alasalvar, C., Salas-Salvado, J., Ros, E., Sabate, S., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 126–156. [Google Scholar]
- Lamuel-Raventos, R.M.; St. Onge, M.P. Prebiotic nut compounds and human microbiota. Crit. Rev. Food Sci. Nutr. 2017, 57, 3154–3163. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, E.; Lambert, K.; Stanford, J.; Neale, E.P. The effect of nut consumption (treenuts and peanuts) on the gut microbiota of humans: A systematic review. Br. J. Nutr. 2021, 125, 508–520. [Google Scholar] [CrossRef]
- Creedon, A.C.; Hung, E.S.; Berry, S.E.; Whelan, K. Nuts and their Effect on Gut Microbiota, Gut Function and Symptoms in Adults: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Nutrients 2020, 12, 2347–2368. [Google Scholar] [CrossRef]
- Mead, L.C.; Hill, A.M.; Carter, S.; Coates, A.M. The Effect of Nut Consumption on Diet Quality, Cardiometabolic and Gastrointestinal Health in Children: A Systematic Review of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2021, 18, 454–469. [Google Scholar] [CrossRef]
- Neale, E.P.; Tapsell, L.C. Nuts in Healthy Dietary Patterns and Dietary Guidelines. In Health Benefits of Nuts and Dried Fruits; Alasalvar, C., Salas-Salvado, J., Ros, E., Sabate, S., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 290–314. [Google Scholar]
- Galie, S.; Garcia-Gavilan, J.; Camacho-Barcia, L.; Atzenia, A.; Muralidharan, J.; Papandreou, C.; Arcelin, P.; Palau-Galindo, A.; Garcia, D.; Basora, J.; et al. Effects of the Mediterranean Diet or Nut Consumption on Gut Microbiota Composition and Fecal Metabolites and their Relationship with Cardiometabolic Risk Factors. Mol. Nutr. Food Res. 2021, 65, 2000982–2000991. [Google Scholar] [CrossRef]
- Rinott, E.; Yaskolka, M.; Tsaban, G.; Zelicha, H.; Kaplan, A.; Knights, D.; Tuohy, K.; Scholz, M.U.; Koren, O.; Stampfer, M.J.; et al. The effects of the Green-Mediterranean diet on cardiometabolic health are linked to gut microbiome modifications: A randomized controlled trial. Genome Med. 2022, 14, 29–44. [Google Scholar] [CrossRef]
- Li, C.H.; Shelp, G.; Wright, A.J. Influence of Nut Structure and Processing on Lipid Bioaccessibility and Absorption. Curr. Opin. Food Sci. 2022, 49, 100966. [Google Scholar] [CrossRef]
- Mandalari, G.; Parker, M.L.; Grundy, M.M.; Grassby, T.; Smeriglio, A.; Bisignano, C.; Raciti, R.; Trombetta, D.; Baer, D.J.; Wilde, P.J. Understanding the Effect of Particle Size and Processing on Almond Lipid Bioaccessibility through Microstructural Analysis: From Mastication to Faecal Collection. Nutrients 2018, 10, 213. [Google Scholar] [CrossRef] [PubMed]
- Holland, C.; Ryden, P.; Edwards, C.H.; Grundy, M.M. Plant Cell Walls: Impact on Nutrient Bioaccessibility and Digestibility. Foods 2020, 9, 201. [Google Scholar] [CrossRef] [PubMed]
- Grassby, T.; Picout, D.R.; Mandalari, G.; Faulks, R.M.; Kendall, C.W.; Rich, G.T.; Wickham, M.S.J.; Lapsley, K.; Ellis, P.R. Modelling of nutrient bioaccessi-bility in almond seeds based on the fracture properties of their cell walls. Food Funct. 2014, 5, 3096–3106. [Google Scholar] [CrossRef] [PubMed]
- Grassby, T.; Mandalari, G.; Grundy MM, L.; Edwards, C.H.; Bisignano, C.; Trombetta, D.; Smeriglio, A.; Chessa, S.; Ray, S.; Sanderson, J.; et al. In vitro and in vivo modeling of lipid bioaccessibility and digestion from almond muffins: The importance of the cell-wall barrier mechanism. J. Funct. Foods 2017, 37, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Creedon, A.C.; Dimidi, E.; Hung, E.S.; Rossi, M.; Probert, C.; Grassby, T.; Miguens-Blanco, J.; Marchesi, J.R.; Scott, S.M.; Berry, S.E.; et al. The impact of almonds and almond processing on gastrointestinal physiology, luminal microbiology and gastrointestinal symptoms: A randomized controlled trial and mastication study. Am. J. Clin. Nutr. 2022, 116, 1790–1804. [Google Scholar] [CrossRef] [PubMed]
- Ellis, P.R.; Kendall, C.W.; Ren, Y.; Parker, C.; Pacy, J.F.; Waldron, K.W.; Jenkins, D.J. Role of cell walls in the bioaccessibility of lipids in almond seeds. Am. J. Clin. Nutr. 2004, 80, 604–613. [Google Scholar] [CrossRef]
- Kumari, S.; Gray, A.R.; Webster, K.; Bailey, K.; Reid, M.; Kelvin, K.A.H.; Tey, S.L.; Chisholm, A.; Brown, R.C. Does ‘activating’ nuts affect nutrient bioavailability? Food Chem. 2020, 319, 126529. [Google Scholar] [CrossRef]
- McArthur, B.M.; Mattes, R.D. Energy extraction from nuts: Walnuts, almonds and pistachios. Br. J. Nutr. 2020, 123, 361–371. [Google Scholar] [CrossRef]
- Grundy, M.M.; Grassby, T.; Mandalari, G.; Waldron, K.W.; Butterworth, P.J.; Berry, S.E.; Ellis, P.R. Effect of mastication on lipid bioaccessibility of almonds in a randomized human study and its implications for digestion kinetics, metabolizable energy, and postprandial lipemia. Am. J. Clin. Nutr. 2015, 101, 25–33. [Google Scholar] [CrossRef]
- Mandalari, G.; Faulks, R.M.; Rich, G.T.; Lo Turco, V.; Picout, D.R.; Lo Curto, R.B.; Bisignano, G.; Dugo, P.; Dugo, G.; Waldron, K.W.; et al. Release of protein, lipid, and vitamin E from almond seeds during digestion. J. Agric. Food Chem. 2008, 56, 3409–3416. [Google Scholar] [CrossRef] [PubMed]
- Grundy, M.M.L.; Carrière, F.; Mackie, A.R.; Gray, D.A.; Butterworth, P.J.; Ellis, P.R. The role of plant cell wall encapsulation and porosity in regulating lipolysis during the digestion of almond seeds. Food Funct. 2016, 7, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Maier, C.S. The chemistry of gut microbial metabolism of polyphenols. Phytochem. Rev. 2016, 15, 425–444. [Google Scholar] [CrossRef]
- Williamson, G.; Clifford, M.N. Role of the small intestine, colon and microbiota in determining the metabolic fate of polyphenols. Biochem. Pharmacol. 2017, 139, 24–39. [Google Scholar] [CrossRef] [PubMed]
- Harborne, J.B. (Ed.) The Flavonoids—Advances in Research since 1986; Chapman & Hall: London, UK, 1993. [Google Scholar]
- Stevens-Barrón, J.C.; de la Rosa, L.A.; Wall-Medrano, A.; Álvarez-Parrilla, E.; Rodríguez-Ramirez, R.; Robles-Zepeda, R.E.; Astiazaran-García, H. Chemical composition and in vitro bioaccessibility of antioxidant phytochemicals from selected edible nuts. Nutrients 2019, 11, 2303. [Google Scholar] [CrossRef]
- Mandalari, G.; Bisignano, C.; Filocamo, A.; Chessa, S.; Sarò, M.; Torre, G.; Faulks, R.M.; Dugo, P. Bioaccessibility of pistachio polyphenols, xanthophylls, and tocopherols during simulated human digestion. Nutrition 2013, 29, 338–344. [Google Scholar] [CrossRef]
- Urpi-Sarda, M.; Garrido, I.; Monagas, M.; Gómez-Cordovés, C.; Medina-Remón, A.; Andres-Lacueva, C.; Bartolomé, B. Profile of plasma and urine metabolites after the intake of almond [Prunus dulcis (Mill.) D.A. Webb] polyphenols in humans. J. Agric. Food Chem. 2009, 57, 10134–10142. [Google Scholar] [CrossRef]
- Bartolomé, B.; Monagas, M.; Garrido, I.; Gómez-Cordovés, C.; Martín-Alvarez, P.J.; Lebrón-Aguilar, R.; Urpí-Sardà, M.; Llorach, R.; Andrés-Lacueva, C. Almond (Prunus dulcis (Mill.) D.A. Webb) polyphenols: From chemical characterization to targeted analysis of phenolic metabolites in humans. Arch. Biochem. Biophys. 2010, 501, 124–133. [Google Scholar] [CrossRef]
- Garrido, M.; Urpi-Sarda, M.; Monagas, C.; Gomez-Cordoves, P.J.; Martin-Alvarez, R.; Llorach, B.; Bartolome, C.; Andres-Lacueva, C. Targeted analysis of conjugated and microbial-derived phenolic metabolites in human urine after consumption of an almond skin phenolic extract. J. Nutr. 2010, 140, 1799–1807. [Google Scholar] [CrossRef]
- Verghese, M.; Willis, S.; Boateng, J.; Gomaa, A.; Kaur, R. Effect of food processing on antioxidant potential, availability and bioavailability. Ann. Rev. Food Sci. Technol. 2012, 12, 307–329. [Google Scholar] [CrossRef]
- Grundy, M.M.-L.; Wilde, P.J.; Butterworth, P.J.; Gray, R.; Ellis, P.R. Impact of cell wall encapsulation of almonds on in vitro duodenal lipolysis. Food Chem. 2015, 185, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Tu, X.; Wu, B.; Xie, Y.; Xu, S.-L.; Wu, Z.-Y.; Lv, X.; Wei, F.; Du, L.-Q.; Chen, H. A comprehensive study of raw and roasted macadamia nuts: Lipid profile, physicochemical, nutritional, and sensory properties. Food Sci. Nutr. 2021, 9, 1688–1697. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, I.; Meyer, A.S.; Afonso, S.; Sequeira, A.; Vilela, A.; Goufo, P.; Trindade, H.; Gonçalves, B. Effects of Different Processing Treatments on Almond (Prunus dulcis) Bioactive Compounds, Antioxidant Activities, Fatty Acids, and Sensorial Characteristics. Plants 2020, 9, 1627. [Google Scholar] [CrossRef] [PubMed]
- Mandalari, G.; Vardakou, M.; Faulks, R.; Bisignano, C.; Martorana, M.; Smeriglio, A.; Trombetta, D. Food Matrix Effects of Polyphenol Bioaccessibility from Almond Skin during Simulated Human Digestion. Nutrients 2016, 8, 568. [Google Scholar] [CrossRef]
- Provatas, A.A.; Ayers, S.A.; Callas, A.A.; Birk, J.W.; Lacson, T.A.; Rosenberg, D.W. Quantitative determination of selected urolithin metabolites in human urine by simple sample preparation and UPLC-MS/MS analysis. Curr. Top. Anal. Chem. 2021, 13, 69–80. [Google Scholar]
- Pfundstein, B.; Haubner, R.; Würtele, G.; Gehres, N.; Ulrich, C.M.; Owen, R.W. Pilot walnut intervention study of urolithin bioavailability in human volunteers. J. Agric. Food Chem. 2014, 62, 10264–10273. [Google Scholar] [CrossRef]
- Bhosale, P.B.; Ha, S.E.; Vetrivel, P.; Kim, H.H.; Kim, S.M.; Kim, G.S. Functions of polyphenols and its anticancer properties in biomedical research: A narrative review. Transl. Cancer Res. 2020, 9, 7619–7631. [Google Scholar] [CrossRef]
- Vinson, J.A.; Cai, Y. Nuts, especially walnuts, have both antioxidant quantity and efficacy and exhibit significant potential health benefits. Food Funct. 2012, 3, 134–140. [Google Scholar] [CrossRef]
- Sanchez-Gonzalez, C.; Ciudad, C.J.; Noe, V.; Izquierdo-Pulido, M. Health benefits of walnut polyphenols: An exploration beyond their lipid profile. Crit. Rev. Food Sci. Nutr. 2017, 57, 3373–3383. [Google Scholar] [CrossRef]
- Wu, S.; Shen, D.; Wang, R.; Li, Q.; Mo, R.; Zheng, Y.; Zhou, Y.; Liu, Y. Phenolic profiles and antioxidant activities of free, esterified and bound phenolic compounds in walnut kernel. Food Chem. 2021, 350, 129217. [Google Scholar] [CrossRef]
- Lozupone, C.A.; Knight, R. Species divergence and the measurement of microbial diversity. FEMS Microbiol. Rev. 2008, 32, 557–578. [Google Scholar] [CrossRef] [PubMed]
- Manor, O.; Dai, C.L.; Kornilov, S.A.; Smith, B.; Price, N.D.; Lovejoy, J.C.; Gibbons, S.M.; Magis, A.T. Health and disease markers correlate with gut microbiome composition across thousands of people. Nat. Commun. 2020, 11, 5206. [Google Scholar] [CrossRef] [PubMed]
- Human Microbiome Project, Consortium. Structure, function and diversity of the healthy human microbiome. Nature 2012, 486, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Holscher, H.D.; Guetterman, H.M.; Swanson, K.S.; An, R.; Matthan, N.R.; Lichtenstein, A.H.; Novotny, J.A.; Baer, D.J. Walnut Consumption Alters the Gastrointestinal Microbiota, Microbially Derived Secondary Bile Acids, and Health Markers in Healthy Adults: A Randomized Controlled Trial. J. Nutr. 2018, 148, 861–867. [Google Scholar] [CrossRef]
- Tindall, A.M.; McLimans, C.J.; Petersen, K.S.; Kris-Etherton, P.M.; Lamendella, R. Walnuts and Vegetable Oils Containing Oleic Acid Differentially Affect the Gut Microbiota and Associations with Cardiovascular Risk Factors: Follow-up of a Randomized, Controlled, Feeding Trial in Adults at Risk for Cardiovascular Disease. J. Nutr. 2020, 150, 806–817. [Google Scholar] [CrossRef]
- Holscher, H.D.; Taylor, A.M.; Swanson, K.S.; Novotny, J.A.; Baer, D.J. Almond Consumption and Processing Affects the Composition of the Gastrointestinal Microbiota of Healthy Adult Men and Women: A Randomized Controlled Trial. Nutrients 2018, 10, 126. [Google Scholar] [CrossRef]
- Burns, A.M.; Zitt, M.A.; Rowe, C.C.; Langkamp-Henken, B.; Mai, V.; Nieves, C., Jr.; Ukhanova, M.; Christman, M.C.; Dahl, W.J. Diet quality improves for parents and children when almonds are incorporated into their daily diet: A randomized, crossover study. Nutr. Res. 2016, 36, 80–89. [Google Scholar] [CrossRef]
- Ukhanova, M.; Wang, X.; Baer, D.J.; Novotny, J.A.; Fredborg, M.; Mai, V. Effects of almond and pistachio consumption on gut microbiota composition in a randomised cross-over human feeding study. Br. J. Nutr. 2014, 111, 2146–2152. [Google Scholar] [CrossRef]
- Dhillon, J.; Newman, J.W.; Fiehn, O.; Ortiz, R.M. Almond Consumption for 8 Weeks Altered Host and Microbial Metabolism in Comparison to a Control Snack in Young Adults. J. Am. Nutr. Assoc. 2022, 42, 242–254. [Google Scholar] [CrossRef]
- Bamberger, C.; Rossmeier, A.; Lechner, K.; Wu, L.; Waldmann, E.; Fischer, S.; Stark, R.G.; Altenhofer, J.; Henze, K.; Parhofer, K.G. A Walnut-Enriched Diet Affects Gut Microbiome in Healthy Caucasian Subjects: A Randomized, Controlled Trial. Nutrients 2018, 10, 244. [Google Scholar] [CrossRef]
- Dhillon, J.; Li, Z.; Ortiz, R.M. Almond Snacking for 8 wk Increases Alpha-Diversity of the Gastrointestinal Microbiome and Decreases Bacteroides fragilis Abundance Compared with an Isocaloric Snack in College Freshmen. Curr. Dev. Nutr. 2019, 3, nzz079. [Google Scholar] [CrossRef] [PubMed]
- Sapp, P.A.; Kris-Etherton, P.M.; Arnesen, E.A.; Chen See, J.R.; Lamendella, R.; Petersen, K.S. Peanuts as a nighttime snack enrich butyrate-producing bacteria compared to an isocaloric lower-fat higher-carbohydrate snack in adults with elevated fasting glucose: A randomized crossover trial. Clin. Nutr. 2022, 41, 2169–2177. [Google Scholar] [CrossRef] [PubMed]
- Parilli-Moser, I.; Dominguez-Lopez, I.; Trius-Soler, M.; Castellvi, M.; Bosch, B.; Castro-Barquero, S.; Estruch, R.; Hurtado-Barroso, S.; Lamuela-Raventos, R.M. Consumption of peanut products improves memory and stress response in healthy adults from the ARISTOTLE study: A 6-month randomized controlled trial. Clin. Nutr. 2021, 40, 5556–5567. [Google Scholar] [CrossRef] [PubMed]
- Zouiouich, S.; Loftfield, E.; Huybrechts, I.; Viallon, V.; Louca, P.; Vogtmann, E.; Wells, P.M.; Steves, C.J.; Herzig, K.H.; Menni, C.; et al. Markers of metabolic health and gut microbiome diversity: Findings from two population-based cohort studies. Diabetologia 2021, 64, 1749–1759. [Google Scholar] [CrossRef] [PubMed]
- Gebauer, S.K.; Novotny, J.A.; Bornhorst, G.M.; Baer, D.J. Food processing and structure impact the metabolizable energy of almonds. Food Funct. 2016, 7, 4231–4238. [Google Scholar] [CrossRef] [PubMed]
- Baer, D.J.; Gebauer, S.K.; Novotny, J.A. Walnuts consumed by healthy adults provide less available energy than predicted by the Atwater factors. J. Nutr. 2016, 146, 9–13. [Google Scholar] [CrossRef]
- Baer, D.J.; Gebauer, S.K.; Novotny, J.A. Measured energy value of pistachios in the human diet. Br. J. Nutr. 2012, 107, 120–125. [Google Scholar] [CrossRef]
- Novotny, J.A.; Gebauer, S.K.; Baer, D.J. Discrepancy between the Atwater factor predicted and empirically measured energy values of almonds in human diets. Am. J. Nutr. 2012, 96, 296–301. [Google Scholar] [CrossRef]
- Baer, D.J.; Novotny, J.A. Metabolizable energy from cashew nuts is less than that predicted by Atwater factors. Nutrients 2018, 11, 33. [Google Scholar] [CrossRef]
- Husted, A.S.; Trauelsen, M.; Rudenko, O.; Hjorth, S.A.; Schwartz, T.W. GPCR-Mediated Signaling of Metabolites. Cell. Metab. 2017, 25, 777–796. [Google Scholar] [CrossRef]
- Liu, P.; Wang, Y.; Yang, G.; Zhang, Q.; Meng, L.; Xin, Y.; Jiang, X. The role of short-chain fatty acids in intestinal barrier function, inflammation, oxidative stress, and colonic carcinogenesis. Pharmacol. Res. 2021, 165, 105420. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Formation of propionate and butyrate by the human colonic microbiota. Environ. Microbiol. 2017, 19, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Choo, J.M.; Tran, C.D.; Luscombe-Marsh, N.D.; Stonehouse, W.; Bowen, J.; Johnson, N.; Thompson, C.H.; Watson, E.J.; Brinkworth, G.D.; Rogers, G.B. Almond consumption affects fecal microbiota composition, stool pH, and stool moisture in overweight and obese adults with elevated fasting blood glucose: A randomized controlled trial. Nutr. Res. 2021, 85, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Reifen, R.; Karlinsky, A.; Stark, A.H.; Berkovich, Z.; Nyska, A. alpha-Linolenic acid (ALA) is an anti-inflammatory agent in inflammatory bowel disease. J. Nutr. Biochem. 2015, 26, 1632–1640. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mantrana, I.; Selma-Royo, M.; Gonzalez, S.; Parra-Llorca, A.; Martinez-Costa, C.; Collado, M.C. Distinct maternal microbiota clusters are associated with diet during pregnancy: Impact on neonatal microbiota and infant growth during the first 18 months of life. Gut Microbes 2020, 11, 962–978. [Google Scholar] [CrossRef] [PubMed]
- Martin-Gallausiaux, C.; Marinelli, L.; Blottiere, H.M.; Larraufie, P.; Lapaque, N. SCFA: Mechanisms and functional importance in the gut. Proc. Nutr. Soc. 2021, 80, 37–49. [Google Scholar] [CrossRef]
- Thomas, J.P.; Modos, D.; Rushbrook, S.M.; Powell, N.; Korcsmaros, T. The Emerging Role of Bile Acids in the Pathogenesis of Inflammatory Bowel Disease. Front. Immunol. 2022, 13, 829525. [Google Scholar] [CrossRef]
- Martinot, E.; Sedes, L.; Baptissart, M.; Lobaccaro, J.M.; Caira, F.; Beaudoin, C.; Volle, D.H. Bile acids and their receptors. Mol. Asp. Med. 2017, 56, 2–9. [Google Scholar] [CrossRef]
- Rodriguez-Morato, J.; Matthan, N.R. Nutrition and Gastrointestinal Microbiota, Microbial-Derived Secondary Bile Acids, and Cardiovascular Disease. Curr. Atheroscler. Rep. 2020, 22, 47. [Google Scholar] [CrossRef]
- Singh, R.; Chandrashekharappa, S.; Bodduluri, S.R.; Baby, B.V.; Hegde, B.; Kotla, N.G.; Hiwale, A.A.; Saiyed, T.; Patel, P.; Vijay-Kumar, M.; et al. Enhancement of the gut barrier integrity by a microbial metabolite through the Nrf2 pathway. Nat. Commun. 2019, 10, 89. [Google Scholar] [CrossRef]
- Nakanishi, M.; Matz, A.; Klemashevich, C.; Rosenberg, D.W. Dietary Walnut Supplementation Alters Mucosal Metabolite Profiles During DSS-Induced Colonic Ulceration. Nutrients 2019, 11, 1118. [Google Scholar] [CrossRef] [PubMed]
- Marin, M.; Maria Giner, R.; Rios, J.L.; Recio, M.C. Intestinal anti-inflammatory activity of ellagic acid in the acute and chronic dextrane sulfate sodium models of mice colitis. J. Ethnopharmacol. 2013, 150, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Al-Maghout, T.; Cao, H.; Pelzl, L.; Salker, M.S.; Veldhoen, M.; Cheng, A.; Lang, F.; Singh, Y. Gut Bacterial Metabolite Urolithin A (UA) Mitigates Ca(2+) Entry in T Cells by Regulating miR-10a-5p. Front. Immunol. 2019, 10, 1737. [Google Scholar] [CrossRef] [PubMed]
- Koh, S.J.; Choi, Y.I.; Kim, Y.; Kim, Y.S.; Choi, S.W.; Kim, J.W.; Kim, B.G.; Lee, K.L. Walnut phenolic extract inhibits nuclear factor kappaB signaling in intestinal epithelial cells, and ameliorates experimental colitis and colitis-associated colon cancer in mice. Eur. J. Nutr. 2019, 58, 1603–1613. [Google Scholar] [CrossRef]
- Bartoszek, A.; Makaro, A.; Bartoszek, A.; Kordek, R.; Fichna, J.; Salaga, M. Walnut Oil Alleviates Intestinal Inflammation and Restores Intestinal Barrier Function in Mice. Nutrients 2020, 12, 1302. [Google Scholar] [CrossRef]
- Arab, H.H.; Salama, S.A.; Omar, H.A.; Arafa el, S.A.; Maghrabi, I.A. Diosmin protects against ethanol-induced gastric injury in rats: Novel anti-ulcer actions. PLoS ONE 2015, 10, e0122417. [Google Scholar] [CrossRef]
- Liu, R.; Hao, Y.T.; Zhu, N.; Liu, X.R.; Kang, J.W.; Mao, R.X.; Hou, C.; Li, Y. The Gastroprotective Effect of Small Molecule Oligopeptides Isolated from Walnut (Juglans regia L.) against Ethanol-Induced Gastric Mucosal Injury in Rats. Nutrients 2020, 12, 1138. [Google Scholar] [CrossRef]
- Park, J.M.; Han, Y.M.; Park, Y.J.; Hahm, K.B. Dietary intake of walnut prevented Helicobacter pylori-associated gastric cancer through rejuvenation of chronic atrophic gastritis. J. Clin. Biochem. Nutr. 2021, 68, 37–50. [Google Scholar] [CrossRef]
| Study Number (REF) | Intervention Nut | Study Design | Sample Size | Dose | Study Duration | Diversity Changes | Microbial Composition Change |
|---|---|---|---|---|---|---|---|
| [47] (Holscher et al., 2018) | Walnut | Crossover, controlled diet | 18 | 42 g/d | 3 wk | No effect on α diversity; β diversity, weighted principal coordinates analysis of UniFrac distances between samples based on their 97% OTU composition and abundances showed that bacterial communities were affected by walnut consumption. | Compared with after the control period, walnut consumption resulted in higher relative abundance of Faecalibacterium, Clostridium, Dialister, and Roseburia and lower relative abundances of Ruminococcus, Dorea, Oscillospira, and Bifidobacterium. |
| [48] (Tindall et al., 2020) | Walnut | Crossover, controlled diet | 42 | 18% of energy | 6 wk | No effect on α diversity; β diversity, weighted principal coordinates analysis of UniFrac distances between samples based on their 97% OTU composition and abundances showed that bacterial communities were affected by walnut consumption. | Compared with after the control period, walnut consumption resulted in higher relative abundance of Faecalibacterium, Clostridium, Dialister, and Roseburia and lower relative abundances of Ruminococcus, Dorea, Oscillospira, and Bifidobacterium. |
| [49] (Holscher et al., 2018) | Almond (whole, whole roasted, chopped roasted, butter) | Crossover, controlled diet | 18 | 42 g/d | 3 wk | No effect on α and β diversity. | Almond consumption increased the relative abundances of Lachnospira, Roseburia, and Dialister. Comparisons between control and the four almond treatments revealed that chopped almonds increased Lachnospira, Roseburia, and Oscillospira compared with the control; whole almonds increased Dialister compared with the control. There were no differences between almond butter and the control. |
| [50] (Burns et al., 2016) | Almond | Crossover, free-living | 50 | 40 g/g | 6 wk | No differences in overall microbiota diversity measures (Shannon diversity index and inverse Simpson diversity index). | Targeted qPCR analysis did not show almond intake- associated changes in the quantities of Bifidobacteria spp or lactic acid bacteria. When individual OTUs from 16S rRNA were combined at the phylum level, there were no significant differences in abundances correlating with almond intake. Some changes in the prevalence of various bacterial signatures at the genus and species levels were observed with the almond intervention at final vs. baseline. |
| [51] (Ukhanova et al., 2014) | Almond | Crossover, controlled diet | 18 | 42 g/d and 84 g/d | 18 d | α-diversity was not affected by the intake of almonds. | Numbers of bifidobacteria were not affected by the consumption of almonds. |
| [51] (Ukhanova et al., 2014) | Pistachio | Crossover, controlled diet | 16 | 42 g/d and 84 g/d | 18 d | α-diversity was not affected by the intake of pistachios. | Numbers of bifidobacteria were not affected by the consumption of pistachio. Pistachio consumption appeared to decrease the number of lactic acid bacteria. |
| [52] (Dhillon et al., 2022) | Almond | Parallel arm, free-living | 73 | 57 g/d | 8 wk | Microbial amino acid biosynthesis, and amino sugar and nucleotide sugar metabolism pathways were differentially enriched at the end of the intervention. | |
| [53] (Bamberger et al., 2018) | Walnut | Crossover, free-living | 142 | 43 g/d | 4 wk | Supplementing walnuts in the diet did not significantly affect bacterial diversity measured by Shannons effective, and Simpsons effective counts. There was no significant difference in evenness as well as in richness for the walnut diet compared with the control diet. Beta-diversity increased with walnut consumption. | The abundance of Ruminococcaceae and Bifidobacteria increased significantly while Clostridium sp. cluster XIVa species (Blautia; Anaerostipes) decreased significantly during walnut consumption. |
| [54] (Dhillon et al., 2019) | Almond | Parallel arm, free-living | 73 | 57 g/d | 8 wk | Almond snacking resulted in 3% greater quantitative alpha-diversity (Shannon index) and 8% greater qualitative alpha-diversity (Chao1 index) than the cracker group. | Almond snacking decreased overall Bacteroides fragilis relative abundance by 48%. |
| [55] (Sapp et al., 2022) | Peanut | Crossover, controlled diet | 50 | 28 g/d | 6 wk | No between-condition differences in alpha- or beta- diversity were observed. | Following peanut intake, Ruminococcaceae were significantly more abundant compared with a lower-fat higher-carbohydrate snack. Metatranscriptomics showed increased expression of the K03518 (aerobic carbon-monoxide dehydrogenase small subunit) gene following peanut intake, and Roseburia intestinalis L1-82 was identified as a contributor to the increased expression. |
| [56] (Choo et al., 2021) | Almond | Parallel arm, free-living | 69 | 56 g/d | 8 wk | In the almond intervention group, there were significant increases in bacterial community richness, evenness and diversity. | Increases in both the relative and absolute abundance of operational taxonomic units in the Ruminococcaceae family, including Ruminiclostridium, Ruminococcaceae NK4A214, and Ruminococcaceae UCG-003 were the principal drivers of microbiota-level changes. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mandalari, G.; Gervasi, T.; Rosenberg, D.W.; Lapsley, K.G.; Baer, D.J. Effect of Nuts on Gastrointestinal Health. Nutrients 2023, 15, 1733. https://doi.org/10.3390/nu15071733
Mandalari G, Gervasi T, Rosenberg DW, Lapsley KG, Baer DJ. Effect of Nuts on Gastrointestinal Health. Nutrients. 2023; 15(7):1733. https://doi.org/10.3390/nu15071733
Chicago/Turabian StyleMandalari, Giuseppina, Teresa Gervasi, Daniel W. Rosenberg, Karen G. Lapsley, and David J. Baer. 2023. "Effect of Nuts on Gastrointestinal Health" Nutrients 15, no. 7: 1733. https://doi.org/10.3390/nu15071733
APA StyleMandalari, G., Gervasi, T., Rosenberg, D. W., Lapsley, K. G., & Baer, D. J. (2023). Effect of Nuts on Gastrointestinal Health. Nutrients, 15(7), 1733. https://doi.org/10.3390/nu15071733











