Impact of Vitamin D on Immunopathology of Hashimoto’s Thyroiditis: From Theory to Practice
Abstract
1. Introduction
2. Material and Methods
3. Vitamin D
3.1. Sources, Metabolism, and Function
3.2. The Role of Vitamin D in the Immune System
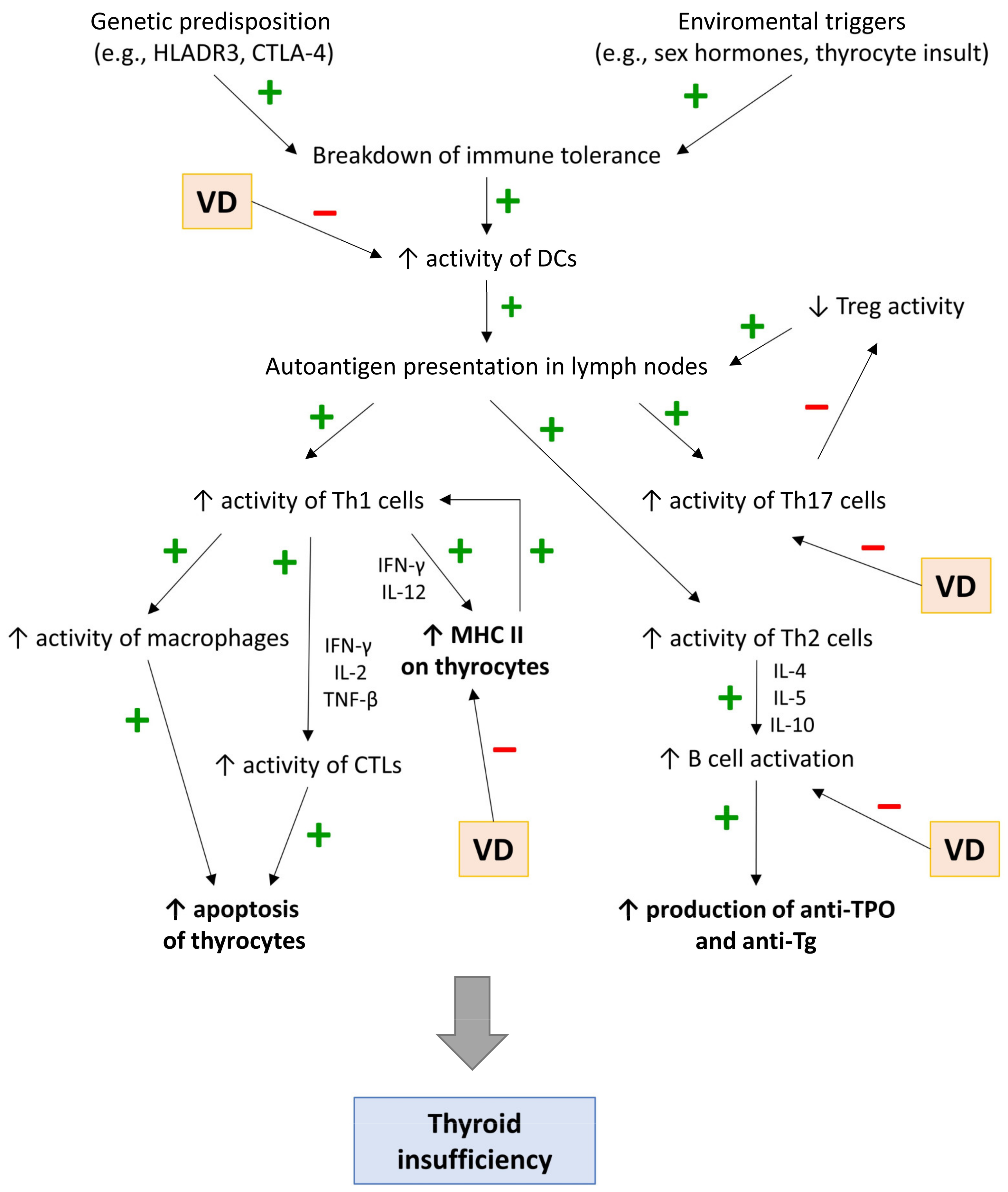
4. Recent Findings in Hashimoto’s Thyroiditis Immunopathology
4.1. Etiological Factors Affecting the Development of HT
4.2. Immunopathological Processes in HT on the Level of Cells and Cytokines
5. Role of Vitamin D in the Immunopathology of Hashimoto’s Thyroiditis
5.1. Immunomodulatory Potential of Vitamin D in HT
5.2. Association between Vitamin D, the Occurrence of HT, and Antibody Levels
5.3. Association between Vitamin D Levels and Immunological Parameters in HT
5.4. Changes in Immunological Parameters and HT Outcomes after Vitamin D Supplementation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weetman, A.P. An Update on the Pathogenesis of Hashimoto’s Thyroiditis. J. Endocrinol. Investig. 2021, 44, 883–890. [Google Scholar] [CrossRef]
- Szklarz, M.; Gontarz-Nowak, K.; Matuszewski, W.; Bandurska-Stankiewicz, E. Iron: Not Just a Passive Bystander in AITD. Nutrients 2022, 14, 4682. [Google Scholar] [CrossRef] [PubMed]
- Ralli, M.; Angeletti, D.; Fiore, M.; D’Aguanno, V.; Lambiase, A.; Artico, M.; de Vincentiis, M.; Greco, A. Hashimoto’s Thyroiditis: An Update on Pathogenic Mechanisms, Diagnostic Protocols, Therapeutic Strategies, and Potential Malignant Transformation. Autoimmun. Rev. 2020, 19, 102649. [Google Scholar] [CrossRef]
- Ihnatowicz, P.; Drywień, M.; Wątor, P.; Wojsiat, J. The Importance of Nutritional Factors and Dietary Management of Hashimoto’s Thyroiditis. Ann. Agric. Environ. Med. 2020, 27, 184–193. [Google Scholar] [CrossRef]
- Ransing, R.S.; Mishra, K.K.; Sarkar, D. Neuropsychiatric Manifestation of Hashimoto’s Encephalopathy in an Adolescent and Treatment. Indian J. Psychol. Med. 2016, 38, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Markomanolaki, Z.S.; Tigani, X.; Siamatras, T.; Bacopoulou, F.; Tsartsalis, A.; Artemiadis, A.; Megalooikonomou, V.; Vlachakis, D.; Chrousos, G.P.; Darviri, C. Stress Management in Women with Hashimoto’s Thyroiditis: A Randomized Controlled Trial. J. Mol. Biochem. 2019, 8, 3–12. [Google Scholar] [PubMed]
- Krysiak, R.; Szkróbka, W.; Okopień, B. The Effect of Vitamin D on Thyroid Autoimmunity in Levothyroxine-Treated Women with Hashimoto’s Thyroiditis and Normal Vitamin D Status. Exp. Clin. Endocrinol. Diabetes 2017, 125, 229–233. [Google Scholar] [CrossRef]
- Krysiak, R.; Szkróbka, W.; Okopień, B. The Effect of Gluten-Free Diet on Thyroid Autoimmunity in Drug-Naïve Women with Hashimoto’s Thyroiditis: A Pilot Study. Exp. Clin. Endocrinol. Diabetes 2019, 127, 417–422. [Google Scholar] [CrossRef]
- Hu, Y.; Feng, W.; Chen, H.; Shi, H.; Jiang, L.; Zheng, X.; Liu, X.; Zhang, W.; Ge, Y.; Liu, Y.; et al. Effect of Selenium on Thyroid Autoimmunity and Regulatory T Cells in Patients with Hashimoto’s Thyroiditis: A Prospective Randomized-Controlled Trial. Clin. Transl. Sci. 2021, 14, 1390–1402. [Google Scholar] [CrossRef]
- Janoušek, J.; Pilařová, V.; Macáková, K.; Nomura, A.; Veiga-Matos, J.; Silva, D.D.d.; Remião, F.; Saso, L.; Malá-Ládová, K.; Malý, J.; et al. Vitamin D: Sources, Physiological Role, Biokinetics, Deficiency, Therapeutic Use, Toxicity, and Overview of Analytical Methods for Detection of Vitamin D and Its Metabolites. Crit. Rev. Clin. Lab. Sci. 2022, 59, 517–554. [Google Scholar] [CrossRef]
- Wierzbicka, A.; Oczkowicz, M. Sex Differences in Vitamin D Metabolism, Serum Levels and Action. Br. J. Nutr. 2022, 128, 2115–2130. [Google Scholar] [CrossRef]
- Matikainen, N.; Pekkarinen, T.; Ryhänen, E.M.; Schalin-Jäntti, C. Physiology of Calcium Homeostasis: An Overview. Endocrinol. Metab. Clin. N. Am. 2021, 50, 575–590. [Google Scholar] [CrossRef]
- Egierska, D.; Pietruszka, P.; Burzyńska, P.; Chruścicka, I.; Buchta, J. Pleiotropic Effects of Vitamin D3. J. Educ. Health Sport 2021, 11, 143–155. [Google Scholar] [CrossRef]
- Huiberts, L.M.; Smolders, K.C.H.J. Effects of Vitamin D on Mood and Sleep in the Healthy Population: Interpretations from the Serotonergic Pathway. Sleep Med. Rev. 2021, 55, 101379. [Google Scholar] [CrossRef]
- Lisowska, K.A.; Bryl, E. The role of vitamin D in the development of autoimmune diseases. Postep. Hig. Med. Dosw. 2017, 71, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Zhang, W.; Ma, C.; Zhao, Y.; Xiong, R.; Wang, H.; Chen, W.; Zheng, S.G. Immunomodulatory Function of Vitamin D and Its Role in Autoimmune Thyroid Disease. Front. Immunol. 2021, 12, 574967. [Google Scholar] [CrossRef] [PubMed]
- Latic, N.; Erben, R.G. Vitamin D and Cardiovascular Disease, with Emphasis on Hypertension, Atherosclerosis, and Heart Failure. Int. J. Mol. Sci. 2020, 21, 6483. [Google Scholar] [CrossRef]
- Mendes, M.M.; Botelho, P.B.; Ribeiro, H. Vitamin D and Musculoskeletal Health: Outstanding Aspects to Be Considered in the Light of Current Evidence. Endocr. Connect. 2022, 11, e210596. [Google Scholar] [CrossRef]
- Chin, K.; Appel, L.J.; Michos, E.D. Vitamin D, Calcium, and Cardiovascular Disease: A “D” Vantageous or “D” Etrimental? An Era of Uncertainty. Curr. Atheroscler. Rep. 2017, 19, 5. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; McGrath, J.J.; Burne, T.H.J.; Eyles, D.W. Vitamin D and Schizophrenia: 20 Years on. Mol. Psychiatry 2021, 26, 2708–2720. [Google Scholar] [CrossRef]
- Littlejohns, T.J.; Henley, W.E.; Lang, I.A.; Annweiler, C.; Beauchet, O.; Chaves, P.H.M.; Fried, L.; Kestenbaum, B.R.; Kuller, L.H.; Langa, K.M.; et al. Vitamin D and the Risk of Dementia and Alzheimer Disease. Neurology 2014, 83, 920–928. [Google Scholar] [CrossRef]
- Szeja, N. Pleiotropic effects of vitamin D—A review of current studies. Pomeranian J. Life Sci. 2017, 63, 128–134. [Google Scholar] [CrossRef]
- Cândido, F.G.; Bressan, J. Vitamin D: Link between Osteoporosis, Obesity, and Diabetes? Int. J. Mol. Sci. 2014, 15, 6569–6591. [Google Scholar] [CrossRef]
- Skowrońska, P.; Pastuszek, E.; Kuczyński, W.; Jaszczoł, M.; Kuć, P.; Jakiel, G.; Wocławek-Potocka, I.; Łukaszuk, K. The Role of Vitamin D in Reproductive Dysfunction in Women—A Systematic Review. Ann. Agric. Environ. Med. 2016, 23, 671–676. [Google Scholar] [CrossRef]
- Kim, C.S.; Kim, S.W. Vitamin D and Chronic Kidney Disease. Korean J. Intern. Med. 2014, 29, 416–427. [Google Scholar] [CrossRef]
- Hii, C.S.; Ferrante, A. The Non-Genomic Actions of Vitamin D. Nutrients 2016, 8, 135. [Google Scholar] [CrossRef]
- Żmijewski, M.A. Nongenomic Activities of Vitamin D. Nutrients 2022, 14, 5104. [Google Scholar] [CrossRef]
- Cui, X.; Gooch, H.; Petty, A.; McGrath, J.J.; Eyles, D. Vitamin D and the Brain: Genomic and Non-Genomic Actions. Mol. Cell. Endocrinol. 2017, 453, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Borel, P.; Caillaud, D.; Cano, N.J. Vitamin D Bioavailability: State of the Art. Crit. Rev. Food Sci. Nutr. 2015, 55, 1193–1205. [Google Scholar] [CrossRef] [PubMed]
- Parva, N.R.; Tadepalli, S.; Singh, P.; Qian, A.; Joshi, R.; Kandala, H.; Nookala, V.K.; Cheriyath, P. Prevalence of Vitamin D Deficiency and Associated Risk Factors in the US Population (2011–2012). Cureus 2018, 10, e2741. [Google Scholar] [CrossRef]
- Bleizgys, A. Vitamin D Dosing: Basic Principles and a Brief Algorithm (2021 Update). Nutrients 2021, 13, 4415. [Google Scholar] [CrossRef]
- Cui, A.; Zhang, T.; Xiao, P.; Fan, Z.; Wang, H.; Zhuang, Y. Global and Regional Prevalence of Vitamin D Deficiency in Population-Based Studies from 2000 to 2022: A Pooled Analysis of 7.9 Million Participants. Front. Nutr. 2023, 10, 1070808. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Köstenberger, M.; Tmava Berisha, A.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D Deficiency 2.0: An Update on the Current Status Worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Gonzalez, L. Is Vitamin D Deficiency a Major Global Public Health Problem? J. Steroid Biochem. Mol. Biol. 2014, 144, 138–145. [Google Scholar] [CrossRef]
- Bizzaro, G.; Antico, A.; Fortunato, A.; Bizzaro, N. Vitamin D and Autoimmune Diseases: Is Vitamin D Receptor (VDR) Polymorphism the Culprit? Isr. Med. Assoc. J. 2017, 19, 438–443. [Google Scholar] [PubMed]
- Ismailova, A.; White, J.H. Vitamin D, Infections and Immunity. Rev. Endocr. Metab. Disord. 2022, 23, 265–277. [Google Scholar] [CrossRef]
- Gilani, S.J.; Bin-Jumah, M.N.; Nadeem, M.S.; Kazmi, I. Vitamin D Attenuates COVID-19 Complications via Modulation of Proinflammatory Cytokines, Antiviral Proteins, and Autophagy. Expert Rev. Anti-Infect. Ther. 2022, 20, 231–241. [Google Scholar] [CrossRef]
- Christakos, S.; Dhawan, P.; Verstuyf, A.; Verlinden, L.; Carmeliet, G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol. Rev. 2016, 96, 365–408. [Google Scholar] [CrossRef]
- Harrison, S.R.; Li, D.; Jeffery, L.E.; Raza, K.; Hewison, M. Vitamin D, Autoimmune Disease and Rheumatoid Arthritis. Calcif. Tissue Int. 2020, 106, 58–75. [Google Scholar] [CrossRef]
- Ao, T.; Kikuta, J.; Ishii, M. The Effects of Vitamin D on Immune System and Inflammatory Diseases. Biomolecules 2021, 11, 1624. [Google Scholar] [CrossRef]
- Bscheider, M.; Butcher, E.C. Vitamin D Immunoregulation through Dendritic Cells. Immunology 2016, 148, 227–236. [Google Scholar] [CrossRef]
- Zhao, H.; Wei, X.; Yang, X. A Novel Update on Vitamin D in Recurrent Pregnancy Loss. Mol. Med. Rep. 2021, 23, 382. [Google Scholar] [CrossRef]
- Cyprian, F.; Lefkou, E.; Varoudi, K.; Girardi, G. Immunomodulatory Effects of Vitamin D in Pregnancy and Beyond. Front. Immunol. 2019, 10, 2739. [Google Scholar] [CrossRef]
- Rolf, L.; Muris, A.-H.; Hupperts, R.; Damoiseaux, J. Illuminating Vitamin D Effects on B Cells—The Multiple Sclerosis Perspective. Immunology 2016, 147, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekara, S.; Patted, A. Role of Vitamin D Supplementation in Improving Disease Activity in Rheumatoid Arthritis: An Exploratory Study. Int. J. Rheum. Dis. 2017, 20, 825–831. [Google Scholar] [CrossRef] [PubMed]
- El Amrousy, D.; El Ashry, H.; Hodeib, H.; Hassan, S. Vitamin D in Children with Inflammatory Bowel Disease: A Randomized Controlled Clinical Trial. J. Clin. Gastroenterol. 2021, 55, 815–820. [Google Scholar] [CrossRef]
- Karagüzel, G.; Sakarya, N.P.; Bahadır, S.; Yaman, S.; Ökten, A. Vitamin D Status and the Effects of Oral Vitamin D Treatment in Children with Vitiligo: A Prospective Study. Clin. Nutr. ESPEN 2016, 15, 28–31. [Google Scholar] [CrossRef]
- Hahn, J.; Cook, N.R.; Alexander, E.K.; Friedman, S.; Walter, J.; Bubes, V.; Kotler, G.; Lee, I.-M.; Manson, J.E.; Costenbader, K.H. Vitamin D and Marine Omega 3 Fatty Acid Supplementation and Incident Autoimmune Disease: VITAL Randomized Controlled Trial. BMJ 2022, 376, e066452. [Google Scholar] [CrossRef]
- Yang, M.; Wu, H.; Zhao, M.; Long, H.; Lu, Q. Vitamin D Status in Patients with Autoimmune Bullous Dermatoses: A Meta-Analysis. J. Dermatol. Treat. 2022, 33, 1356–1367. [Google Scholar] [CrossRef]
- Lee, S.; Kim, B.J.; Lee, C.H.; Lee, W.S. Increased Prevalence of Vitamin D Deficiency in Patients with Alopecia Areata: A Systematic Review and Meta-Analysis. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 1214–1221. [Google Scholar] [CrossRef] [PubMed]
- Upala, S.; Sanguankeo, A. Low 25-Hydroxyvitamin D Levels Are Associated with Vitiligo: A Systematic Review and Meta-Analysis. Photodermatol. Photoimmunol. Photomed. 2016, 32, 181–190. [Google Scholar] [CrossRef]
- Guan, S.-Y.; Cai, H.-Y.; Wang, P.; Lv, T.-T.; Liu, L.-N.; Mao, Y.-M.; Zhao, C.-N.; Wu, Q.; Dan, Y.-L.; Sam, N.B.; et al. Association between Circulating 25-Hydroxyvitamin D and Systemic Lupus Erythematosus: A Systematic Review and Meta-Analysis. Int. J. Rheum. Dis. 2019, 22, 1803–1813. [Google Scholar] [CrossRef]
- Taheriniya, S.; Arab, A.; Hadi, A.; Fadel, A.; Askari, G. Vitamin D and Thyroid Disorders: A Systematic Review and Meta-Analysis of Observational Studies. BMC Endocr. Disord. 2021, 21, 171. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Multiple Nutritional Factors and Thyroid Disease, with Particular Reference to Autoimmune Thyroid Disease. Proc. Nutr. Soc. 2019, 78, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Liu, F.; Zhao, M.; Xu, S.; Li, P.; Cao, J.; Tian, D.; Tan, Y.; Zheng, L.; Cao, X.; et al. Prediction Models Constructed for Hashimoto’s Thyroiditis Risk Based on Clinical and Laboratory Factors. Front. Endocrinol. 2022, 13, 886953. [Google Scholar] [CrossRef]
- Qiu, K.; Li, K.; Zeng, T.; Liao, Y.; Min, J.; Zhang, N.; Peng, M.; Kong, W.; Chen, L. Integrative Analyses of Genes Associated with Hashimoto’s Thyroiditis. J. Immunol. Res. 2021, 2021, 8263829. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.-Y.; Ye, X.-P.; Zhou, Z.; Zhu, C.-F.; Li, R.; Fang, Y.; Zhang, R.-J.; Li, L.; Liu, W.; Wang, Z.; et al. Lymphocyte Infiltration and Thyrocyte Destruction Are Driven by Stromal and Immune Cell Components in Hashimoto’s Thyroiditis. Nat. Commun. 2022, 13, 775. [Google Scholar] [CrossRef]
- Ragusa, F.; Fallahi, P.; Elia, G.; Gonnella, D.; Paparo, S.R.; Giusti, C.; Churilov, L.P.; Ferrari, S.M.; Antonelli, A. Hashimotos’ Thyroiditis: Epidemiology, Pathogenesis, Clinic and Therapy. Best Pract. Res. Clin. Endocrinol. Metab. 2019, 33, 101367. [Google Scholar] [CrossRef]
- Akamizu, T.; Amino, N. Hashimoto’s Thyroiditis. In Endotext; Feingold, K.R., Anawalt, B., Blackman, M.R., Boyce, A., Chrousos, G., Corpas, E., de Herder, W.W., Dhatariya, K., Hofland, J., Dungan, K., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Chandanwale, S.S.; Kulkarni, T.V.; Patel, R.J.; Thakkar, D. A Focal Nodular Hürthle Cell Hyperplasia in Hashimoto’s Thyroiditis: A Diagnostic Dilemma on Fine Needle Aspiration. J. Cytol. 2014, 31, 236–238. [Google Scholar] [CrossRef]
- Luty, J.; Ruckemann-Dziurdzińska, K.; Witkowski, J.M.; Bryl, E. Immunological Aspects of Autoimmune Thyroid Disease—Complex Interplay between Cells and Cytokines. Cytokine 2019, 116, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Stassi, G.; Todaro, M.; Bucchieri, F.; Stoppacciaro, A.; Farina, F.; Zummo, G.; Testi, R.; De Maria, R. Fas/Fas Ligand-Driven T Cell Apoptosis as a Consequence of Ineffective Thyroid Immunoprivilege in Hashimoto’s Thyroiditis. J. Immunol. 1999, 162, 263–267. [Google Scholar] [CrossRef]
- Rocamora-Reverte, L.; Melzer, F.L.; Würzner, R.; Weinberger, B. The Complex Role of Regulatory T Cells in Immunity and Aging. Front. Immunol. 2021, 11, 616949. [Google Scholar] [CrossRef] [PubMed]
- Barragan, M.; Good, M.; Kolls, J.K. Regulation of Dendritic Cell Function by Vitamin D. Nutrients 2015, 7, 8127–8151. [Google Scholar] [CrossRef] [PubMed]
- Konca Degertekin, C.; Aktas Yilmaz, B.; Balos Toruner, F.; Kalkanci, A.; Turhan Iyidir, O.; Fidan, I.; Yesilyurt, E.; Cakır, N.; Kustimur, S.; Arslan, M. Circulating Th17 Cytokine Levels Are Altered in Hashimoto’s Thyroiditis. Cytokine 2016, 80, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Rydzewska, M.; Jaromin, M.; Pasierowska, I.E.; Stożek, K.; Bossowski, A. Role of the T and B Lymphocytes in Pathogenesis of Autoimmune Thyroid Diseases. Thyroid Res. 2018, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, E.A.; Nguyen, J.K.; Liu, J.; Keller, E.; Campbell, N.; Zhang, C.-J.; Smith, H.R.; Li, X.; Jørgensen, T.N. Low Levels of Vitamin D Promote Memory B Cells in Lupus. Nutrients 2020, 12, 291. [Google Scholar] [CrossRef]
- Haas, J.; Schwarz, A.; Korporal-Kuhnke, M.; Faller, S.; Jarius, S.; Wildemann, B. Hypovitaminosis D Upscales B-Cell Immunoreactivity in Multiple Sclerosis. J. Neuroimmunol. 2016, 294, 18–26. [Google Scholar] [CrossRef]
- Chen, S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.; Lipsky, P.E. Modulatory Effects of 1,25-Dihydroxyvitamin D3 on Human B Cell Differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, Y.; Li, H.; Li, H. Effects of Vitamin D on Thyroid Autoimmunity Markers in Hashimoto’s Thyroiditis: Systematic Review and Meta-Analysis. J. Int. Med. Res. 2021, 49, 03000605211060675. [Google Scholar] [CrossRef]
- Wang, J.; Lv, S.; Chen, G.; Gao, C.; He, J.; Zhong, H.; Xu, Y. Meta-Analysis of the Association between Vitamin D and Autoimmune Thyroid Disease. Nutrients 2015, 7, 2485–2498. [Google Scholar] [CrossRef]
- Štefanić, M.; Tokić, S. Serum 25-Hydoxyvitamin D Concentrations in Relation to Hashimoto’s Thyroiditis: A Systematic Review, Meta-Analysis and Meta-Regression of Observational Studies. Eur. J. Nutr. 2020, 59, 859–872. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.R. The Balance of Th17 versus Treg Cells in Autoimmunity. Int. J. Mol. Sci. 2018, 19, 730. [Google Scholar] [CrossRef]
- Li, C.; Yuan, J.; Zhu, Y.-F.; Yang, X.-J.; Wang, Q.; Xu, J.; He, S.-T.; Zhang, J.-A. Imbalance of Th17/Treg in Different Subtypes of Autoimmune Thyroid Diseases. Cell. Physiol. Biochem. 2016, 40, 245–252. [Google Scholar] [CrossRef]
- Shao, S.; Yu, X.; Shen, L. Autoimmune Thyroid Diseases and Th17/Treg Lymphocytes. Life Sci. 2018, 192, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Wang, J.; Li, J.; Li, T.; Chen, Y.; June, R.R.; Zheng, S.G. 1,25-Dihydroxyvitamin D3 Ameliorates Collagen-Induced Arthritis via Suppression of Th17 Cells Through MiR-124 Mediated Inhibition of IL-6 Signaling. Front. Immunol. 2019, 10, 178. [Google Scholar] [CrossRef]
- Dankers, W.; Davelaar, N.; van Hamburg, J.P.; van de Peppel, J.; Colin, E.M.; Lubberts, E. Human Memory Th17 Cell Populations Change into Anti-Inflammatory Cells with Regulatory Capacity upon Exposure to Active Vitamin D. Front. Immunol. 2019, 10, 1504. [Google Scholar] [CrossRef]
- Chang, S.H.; Chung, Y.; Dong, C. Vitamin D Suppresses Th17 Cytokine Production by Inducing C/EBP Homologous Protein (CHOP) Expression. J. Biol. Chem. 2010, 285, 38751–38755. [Google Scholar] [CrossRef] [PubMed]
- Cvek, M.; Kaličanin, D.; Barić, A.; Vuletić, M.; Gunjača, I.; Torlak Lovrić, V.; Škrabić, V.; Punda, A.; Boraska Perica, V. Vitamin D and Hashimoto’s Thyroiditis: Observations from CROHT Biobank. Nutrients 2021, 13, 2793. [Google Scholar] [CrossRef] [PubMed]
- Hanna, H.W.Z.; Rizzo, C.; Abdel Halim, R.M.; El Haddad, H.E.; Salam, R.; El-Sayed Abou-Youssef, H. Vitamin D Status in Hashimoto’s Thyroiditis and Its Association with Vitamin D Receptor Genetic Variants. J. Steroid Biochem. Mol. Biol. 2021, 212, 105922. [Google Scholar] [CrossRef] [PubMed]
- Filipova, L.; Lazurova, Z.; Fulop, P.; Lazurova, I. Vitamin D insufficiency is not associated with thyroid autoimmunity in Slovak women with Hashimoto’s disease. Bratisl. Lek. Listy 2023, 124, 182–186. [Google Scholar] [CrossRef] [PubMed]
- Kim, D. Low Vitamin D Status Is Associated with Hypothyroid Hashimoto’s Thyroiditis. Hormones 2016, 15, 385–393. [Google Scholar] [CrossRef]
- Kim, M.; Song, E.; Oh, H.-S.; Park, S.; Kwon, H.; Jeon, M.J.; Kim, W.G.; Kim, W.B.; Shong, Y.K.; Kim, T.Y. Vitamin D Deficiency Affects Thyroid Autoimmunity and Dysfunction in Iodine-Replete Area: Korea National Health and Nutrition Examination Survey. Endocrine 2017, 58, 332–339. [Google Scholar] [CrossRef]
- Evliyaoğlu, O.; Acar, M.; Özcabı, B.; Erginöz, E.; Bucak, F.; Ercan, O.; Kucur, M. Vitamin D Deficiency and Hashimoto’s Thyroiditis in Children and Adolescents: A Critical Vitamin D Level for This Association? J. Clin. Res. Pediatr. Endocrinol. 2015, 7, 128–133. [Google Scholar] [CrossRef]
- De Pergola, G.; Triggiani, V.; Bartolomeo, N.; Giagulli, V.A.; Anelli, M.; Masiello, M.; Candita, V.; De Bellis, D.; Silvestris, F. Low 25 Hydroxyvitamin D Levels Are Independently Associated with Autoimmune Thyroiditis in a Cohort of Apparently Healthy Overweight and Obese Subjects. Endocr. Metab. Immune Disord. Drug Targets 2018, 18, 646–652. [Google Scholar] [CrossRef]
- Xu, J.; Zhu, X.-Y.; Sun, H.; Xu, X.-Q.; Xu, S.-A.; Suo, Y.; Cao, L.-J.; Zhou, Q.; Yu, H.-J.; Cao, W.-Z. Low Vitamin D Levels Are Associated with Cognitive Impairment in Patients with Hashimoto Thyroiditis. BMC Endocr. Disord. 2018, 18, 87. [Google Scholar] [CrossRef]
- Aktaş, H.Ş. Vitamin B12 and Vitamin D Levels in Patients with Autoimmune Hypothyroidism and Their Correlation with Anti-Thyroid Peroxidase Antibodies. Med. Princ. Pract. 2020, 29, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, N.C.; Karbek, B.; Ucan, B.; Sahin, M.; Cakal, E.; Ozbek, M.; Delibasi, T. The Association between Severity of Vitamin D Deficiency and Hashimoto’s Thyroiditis. Endocr. Pract. 2013, 19, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Sayki Arslan, M.; Topaloglu, O.; Ucan, B.; Karakose, M.; Karbek, B.; Tutal, E.; Caliskan, M.; Ginis, Z.; Cakal, E.; Sahin, M.; et al. Isolated Vitamin D Deficiency Is Not Associated with Nonthyroidal Illness Syndrome, but with Thyroid Autoimmunity. Sci. World J. 2015, 2015, 239815. [Google Scholar] [CrossRef]
- Botelho, I.M.B.; Moura Neto, A.; Silva, C.A.; Tambascia, M.A.; Alegre, S.M.; Zantut-Wittmann, D.E. Vitamin D in Hashimoto’s Thyroiditis and Its Relationship with Thyroid Function and Inflammatory Status. Endocr. J. 2018, 65, 1029–1037. [Google Scholar] [CrossRef]
- Korzeniowska, K.; Jarosz-Chobot, P.; Szypowska, A.; Ramotowska, A.; Fendler, W.; Kalina-Faska, B.; Szadkowska, A.; Mlynarski, W.; Mysliwiec, M. L-Thyroxine Stabilizes Autoimmune Inflammatory Process in Euthyroid Nongoitrous Children with Hashimoto’s Thyroiditis and Type 1 Diabetes Mellitus. J. Clin. Res. Pediatr. Endocrinol. 2013, 5, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Ke, W.; Sun, T.; Zhang, Y.; He, L.; Wu, Q.; Liu, J.; Zham, B. 25-Hydroxyvitamin D serum level in Hashimoto’s thyroiditis, but not Graves’ disease is relatively deficient. Endocr. J. 2017, 64, 581–587. [Google Scholar] [CrossRef]
- Feng, Y.; Qiu, T.; Chen, H.; Wei, Y.; Jiang, X.; Zhang, H.; Chen, D. Association of Serum IL-21 and Vitamin D Concentrations in Chinese Children with Autoimmune Thyroid Disease. Clin. Chim. Acta 2020, 507, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Hisbiyah, Y.; Endaryanto, A.; Setyoboedi, B.; Rochmah, N.; Faizi, M. The Correlation between Vitamin D and Levels of IFN-γ, NF-ΚB, Thyroid Antibodies in down Syndrome: Study in Indonesian Children. Acta Biomed. 2022, 93, e2022342. [Google Scholar] [CrossRef]
- Giuliani, C.; Bucci, I.; Napolitano, G. The Role of the Transcription Factor Nuclear Factor-Kappa B in Thyroid Autoimmunity and Cancer. Front. Endocrinol. 2018, 9, 471. [Google Scholar] [CrossRef] [PubMed]
- Roehlen, N.; Doering, C.; Hansmann, M.-L.; Gruenwald, F.; Vorlaender, C.; Bechstein, W.O.; Holzer, K.; Badenhoop, K.; Penna-Martinez, M. Vitamin D, FOXO3a, and Sirtuin1 in Hashimoto’s Thyroiditis and Differentiated Thyroid Cancer. Front. Endocrinol. 2018, 9, 527. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.S.; Liu, L.; Shu, C.; Yan, L.L.; Zeng, Y. Sex Difference and Interaction of SIRT1 and FOXO3 Candidate Longevity Genes on Life Expectancy: A 10-Year Prospective Longitudinal Cohort Study. J. Gerontol. A Biol. Sci. Med. Sci. 2021, 77, 1557–1563. [Google Scholar] [CrossRef]
- Nho, R.S.; Hergert, P. FoxO3a and Disease Progression. World J. Biol. Chem. 2014, 5, 346–354. [Google Scholar] [CrossRef]
- Chen, C.; Zhou, M.; Ge, Y.; Wang, X. SIRT1 and Aging Related Signaling Pathways. Mech. Ageing Dev. 2020, 187, 111215. [Google Scholar] [CrossRef]
- Tokić, S.; Štefanić, M.; Karner, I.; Glavaš-Obrovac, L. Altered Expression of CTLA-4, CD28, VDR, and CD45 MRNA in T Cells of Patients with Hashimoto’s Thyroiditis—A Pilot Study. Endokrynol. Pol. 2017, 68, 274–828. [Google Scholar] [CrossRef] [PubMed]
- Krysiak, R.; Kowalcze, K.; Okopień, B. The Impact of Exogenous Vitamin D on Thyroid Autoimmunity in Euthyroid Men with Autoimmune Thyroiditis and Early-Onset Androgenic Alopecia. Pharmacol. Rep. 2021, 73, 1439–1447. [Google Scholar] [CrossRef]
- Krysiak, R.; Kowalcze, K.; Okopień, B. The Effect of Vitamin D on Thyroid Autoimmunity in Euthyroid Men with Autoimmune Thyroiditis and Testosterone Deficiency. Pharmacol. Rep. 2019, 71, 798–803. [Google Scholar] [CrossRef] [PubMed]
- Krysiak, R.; Kowalcze, K.; Okopień, B. Hyperprolactinaemia Attenuates the Inhibitory Effect of Vitamin D/Selenomethionine Combination Therapy on Thyroid Autoimmunity in Euthyroid Women with Hashimoto’s Thyroiditis: A Pilot Study. J. Clin. Pharm. Ther. 2020, 45, 1334–1341. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Takacs, I.; Boyanov, M.; Belaya, Z.; Diaconu, C.C.; Mokhort, T.; Zherdova, N.; Rasa, I.; Payer, J.; Pilz, S. Clinical Practice in the Prevention, Diagnosis and Treatment of Vitamin D Deficiency: A Central and Eastern European Expert Consensus Statement. Nutrients 2022, 14, 1483. [Google Scholar] [CrossRef] [PubMed]
- Płudowski, P.; Kos-Kudła, B.; Walczak, M.; Fal, A.; Zozulińska-Ziółkiewicz, D.; Sieroszewski, P.; Peregud-Pogorzelski, J.; Lauterbach, R.; Targowski, T.; Lewiński, A.; et al. Guidelines for Preventing and Treating Vitamin D Deficiency: A 2023 Update in Poland. Nutrients 2023, 15, 695. [Google Scholar] [CrossRef] [PubMed]
- Krysiak, R.; Szkróbka, W.; Okopień, B. Dehydroepiandrosterone Potentiates the Effect of Vitamin D on Thyroid Autoimmunity in Euthyroid Women with Autoimmune Thyroiditis: A Pilot Study. Clin. Exp. Pharmacol. Physiol. 2021, 48, 195–202. [Google Scholar] [CrossRef]
- Krysiak, R.; Szkróbka, W.; Okopień, B. Moderate-Dose Simvastatin Therapy Potentiates the Effect of Vitamin D on Thyroid Autoimmunity in Levothyroxine-Treated Women with Hashimoto’s Thyroiditis and Vitamin D Insufficiency. Pharmacol. Rep. 2018, 70, 93–97. [Google Scholar] [CrossRef]
- Krysiak, R.; Kowalcze, K.; Okopień, B. Selenomethionine Potentiates the Impact of Vitamin D on Thyroid Autoimmunity in Euthyroid Women with Hashimoto’s Thyroiditis and Low Vitamin D Status. Pharmacol. Rep. 2019, 71, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Krysiak, R.; Kowalcze, K.; Okopien, B. Gluten-Free Diet Attenuates the Impact of Exogenous Vitamin D on Thyroid Autoimmunity in Young Women with Autoimmune Thyroiditis: A Pilot Study. Scand. J. Clin. Lab. Investig. 2022, 82, 518–524. [Google Scholar] [CrossRef] [PubMed]
- Malandrini, S.; Trimboli, P.; Guzzaloni, G.; Virili, C.; Lucchini, B. What about TSH and Antithyroid Antibodies in Patients with Autoimmune Thyroiditis and Celiac Disease Using a Gluten-Free Diet? A Systematic Review. Nutrients 2022, 14, 1681. [Google Scholar] [CrossRef] [PubMed]
- Pobłocki, J.; Pańka, T.; Szczuko, M.; Telesiński, A.; Syrenicz, A. Whether a Gluten-Free Diet Should Be Recommended in Chronic Autoimmune Thyroiditis or Not?—A 12-Month Follow-Up. J. Clin. Med. 2021, 10, 3240. [Google Scholar] [CrossRef] [PubMed]
- Mazokopakis, E.E.; Papadomanolaki, M.G.; Tsekouras, K.C.; Evangelopoulos, A.D.; Kotsiris, D.A.; Tzortzinis, A.A. Is Vitamin D Related to Pathogenesis and Treatment of Hashimoto’s Thyroiditis? Hell. J. Nucl. Med. 2015, 18, 222–227. [Google Scholar]
- Chaudhary, S.; Dutta, D.; Kumar, M.; Saha, S.; Mondal, S.A.; Kumar, A.; Mukhopadhyay, S. Vitamin D Supplementation Reduces Thyroid Peroxidase Antibody Levels in Patients with Autoimmune Thyroid Disease: An Open-Labeled Randomized Controlled Trial. Indian J. Endocrinol. Metab. 2016, 20, 391. [Google Scholar] [CrossRef] [PubMed]
- Aghili, A.; Alijanpour Aghamaleki, M.; Pornasrollah, M.; Nooreddini, H.G.; Khafri, S.; Alijanpour, S. Effect of Vitamin D Therapy on Hashimoto’s Thyroiditis in Children with Hypovitaminosis D. Int. J. Pediatr. 2020, 8, 10889–10897. [Google Scholar] [CrossRef]
- Simsek, Y.; Cakir, I.; Yetmis, M.; Dizdar, O.S.; Baspinar, O.; Gokay, F. Effects of Vitamin D Treatment on Thyroid Autoimmunity. J. Res. Med. Sci. 2016, 21, 85. [Google Scholar] [CrossRef] [PubMed]
- Mirhosseini, N.; Brunel, L.; Muscogiuri, G.; Kimball, S. Physiological Serum 25-Hydroxyvitamin D Concentrations Are Associated with Improved Thyroid Function—Observations from a Community-Based Program. Endocrine 2017, 58, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Vondra, K.; Bilek, R.; Matucha, P.; Salatova, M.; Vosatkova, M.; Starka, L.; Hampl, R. Vitamin D Supplementation Changed Relationships, Not Levels of Metabolic-Hormonal Parameters in Autoimmune Thyroiditis. Physiol. Res. 2017, 66, S409–S417. [Google Scholar] [CrossRef] [PubMed]
- Behera, K.K.; Saharia, G.K.; Hota, D.; Sahoo, D.P.; Sethy, M.; Srinivasan, A. Effect of Vitamin D Supplementation on Thyroid Autoimmunity among Subjects of Autoimmune Thyroid Disease in a Coastal Province of India: A Randomized Open-Label Trial. Niger. Med. J. 2020, 61, 237. [Google Scholar] [CrossRef] [PubMed]
- Robat-Jazi, B.; Mobini, S.; Chahardoli, R.; Mansouri, F.; Nodehi, M.; Esfahanian, F.; Saboor Yaraghi, A.A. The Impact of Vitamin D Supplementation on the IFNγ-IP10 Axis in Women with Hashimoto’s Thyroiditis Treated with Levothyroxine: A Double-blind Randomized Placebo-controlled Trial. Iran. J. Allergy Asthma Immunol. 2022, 21, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Karin, N.; Razon, H. Chemokines beyond Chemo-Attraction: CXCL10 and Its Significant Role in Cancer and Autoimmunity. Cytokine 2018, 109, 24–28. [Google Scholar] [CrossRef]
- Antonelli, A.; Ferrari, S.M.; Giuggioli, D.; Ferrannini, E.; Ferri, C.; Fallahi, P. Chemokine (C-X-C Motif) Ligand (CXCL)10 in Autoimmune Diseases. Autoimmun. Rev. 2014, 13, 272–280. [Google Scholar] [CrossRef]
- Nodehi, M.; Ajami, A.; Izad, M.; Omran, H.A.; Chahardoli, R.; Amouzegar, A.; Yekaninejad, S.; Hemmatabadi, M.; Azizi, F.; Esfahanian, F.; et al. Effects of Vitamin D Supplements on Frequency of CD4+ T-Cell Subsets in Women with Hashimoto’s Thyroiditis: A Double-Blind Placebo-Controlled Study. Eur. J. Clin. Nutr. 2019, 73, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
| Disease Group | Vitamin D Functions | Disease Prevention |
|---|---|---|
| Musculoskeletal diseases [18] |
|
|
| Cardiovascular diseases [19] |
|
|
| Brain diseases [20,21] |
|
|
| Cancers [22] |
|
|
| Immune-mediated diseases [15] |
|
|
| Metabolic diseases [23] |
|
|
| Female reproductive system diseases [22,24] |
| PCOS |
| Renal system diseases [25] |
| CKD |
| Author, Year | Type of Study | Number of Participants | Main Findings |
|---|---|---|---|
| Bozkurt et al., 2013 [88] | Case–control study |
|
|
| Evliyaoğlu et al., 2015 [84] | Case–control study |
| The prevalence of 25(OH)D deficiency in HT patients was significantly higher than that in the control group |
| Arslan et al., 2015 [89] | Cross-sectional study | 155 HC |
|
| Wang et al., 2015 [71] | Meta-analysis | (a) The continuous 25(OH)D by AITD * status:
| HT patients had lower 25(OH)D levels and were more likely to have a 25(OH)D deficiency |
| Kim et al., 2016 [82] | Retrospective cross-sectional study | 776 HT patients with measured 25(OH)D | 25(OH)D insufficiency was associated with HT, especially overt hypothyroidism |
| Kim et al., 2017 [83] | Cross-sectional study; a nationwide survey | 4181 participants | Anti-TPO positivity was more prevalent in the 25(OH)D deficient group than in insufficient and sufficient 25(OH)D groups |
| Wencai Ke et al., 2017 [92] | Cross-sectional study |
| 25(OH)D levels were not associated with thyroid function, antithyroid antibodies, and serum cytokines IL-4, IL-17, and TNF-α in patients with AITD * |
| Tokic et al., 2017 [100] | Cross-sectional study |
| Nominally higher expression levels of VDR mRNA were found in T cells of healthy controls when compared to the HT patients |
| De Pergola et al., 2018 [85] | Cross-sectional study | 261 overweight and obese subjects | 25(OH)D deficiency is significantly associated with HT in overweight and obese subjects |
| Botelho et al., 2018 [90] | Cross-sectional study |
| A positive correlation between 25(OH)D and fT4, IL-17, TNF-α and IL-5 in HT group |
| Jun Xu et al., 2018 [86] | Case–control study |
|
|
| Aktaş, 2019 [87] | Retrospective cohort study | 130 HT patients |
|
| Feng et al., 2020 [93] | Cross-sectional study |
|
|
| Štefanić and Tokić, 2020 [55] | Meta-analysis |
| Lower serum 25(OH) in HT compared to healthy controls |
| Cvek et al., 2021 [79] | Case–control study; observations from biobank data |
|
|
| Hanna et al., 2021 [80] | Case–control study |
| 25(OH)D level was statistically indifferent between HT and control groups |
| Taheriniya et al., 2021 [53] | Meta-analysis |
| Significantly lower 25(OH)D level among HT patients compared to healthy controls |
| Hisbiyah et al., 2022 [94] | Cross-sectional study | 80 Down syndrome patients (children) |
|
| Filipova et al., 2023 [81] | Prospective case–control study |
| No significant association between 25(OH)D and thyroid autoantibodies, thyroid hormones, and thyroid volume |
| Author, Year | Dose and Supplementation Duration | Changes in 25(OH)D Levels | Other Changes in Immunological Parameters | Changes in Anti-TPO Titers | Changes in Anti-Tg Titers |
|---|---|---|---|---|---|
| Mazokopakis et al., 2015 [112] | 1200–4000 IU daily, aiming to achieve 25(OH)D concentration of >40 ng/mL, 4 months | ⬆ | ⬇ | ⬇ | |
| Chaudhary et al., 2016 [113] | 60,000 IU weekly, 8 weeks | ⬆ | ⬇ | ||
| Simsek et al., 2016 [115] | 1000 IU daily, 1 month | ⬆ | ⬇ | ⬇ | |
| Mirhosseini et al., 2017 [116] | Doses modified with the aim to achieve 25(OH)D concentration of >40 ng/mL, 1 year | ⬆ | CRP ⬇ | ⬇ | ⬇ |
| Vondra et al., 2017 [117] | 4300 IU daily, 3 months | ⬆ | CRP ⬌ | ⬆ | ⬆ |
| Nodehi et al., 2019 [122] | 50,000 IU weekly, 3 months | ⬆ |
| ||
| Aghili et al., 2020 [114] | Varied depending on initial and rechecked 25(OH)D concentrations | ⬆ | ⬇ | ⬇ | |
| Behera et al., 2020 [118] | 60,000 IU weekly, 2 months, then 60,000 IU monthly, 4 months | ⬆ | ⬆ | ||
| Krysiak et al., 2016–2022 [7,101,102,103,106,107,108] | 2000–4000 IU daily for 6 months | ⬆ | ⬇ | ⬇ | |
| Krysiak et al., 2022 [109] | 4000 IU daily for 6 months | ⬆ | CRP ⬌ | ⬇ | ⬇ |
| Robat-Jazi et al., 2022 [119] | 50,000 IU weekly, 3 months | ⬆ |
| ⬇ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lebiedziński, F.; Lisowska, K.A. Impact of Vitamin D on Immunopathology of Hashimoto’s Thyroiditis: From Theory to Practice. Nutrients 2023, 15, 3174. https://doi.org/10.3390/nu15143174
Lebiedziński F, Lisowska KA. Impact of Vitamin D on Immunopathology of Hashimoto’s Thyroiditis: From Theory to Practice. Nutrients. 2023; 15(14):3174. https://doi.org/10.3390/nu15143174
Chicago/Turabian StyleLebiedziński, Filip, and Katarzyna Aleksandra Lisowska. 2023. "Impact of Vitamin D on Immunopathology of Hashimoto’s Thyroiditis: From Theory to Practice" Nutrients 15, no. 14: 3174. https://doi.org/10.3390/nu15143174
APA StyleLebiedziński, F., & Lisowska, K. A. (2023). Impact of Vitamin D on Immunopathology of Hashimoto’s Thyroiditis: From Theory to Practice. Nutrients, 15(14), 3174. https://doi.org/10.3390/nu15143174







