Gut Symptoms during FODMAP Restriction and Symptom Response to Food Challenges during FODMAP Reintroduction: A Real-World Evaluation in 21,462 Participants Using a Mobile Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Ethical Considerations
2.3. Low FODMAP Diet App
- (a)
- food category, checking the suitability of generic foods rather than specific brands for a low FODMAP diet (e.g., suitable types of cheese under dairy category);
- (b)
- free text, to enable all branded food products matching free-text descriptions;
- (c)
- pre-determined food category groups within the app, which generated branded food products available within that food category (e.g., suitable cheese brands under dairy category);
- (d)
- barcode scanner, which used the camera function to scan the barcode of food products to identify it and display the ingredients related to that product, including whether it was suitable or not to be consumed during the low FODMAP diet.
2.4. Outcome Measures and Data Extraction
2.4.1. Gastrointestinal Symptoms
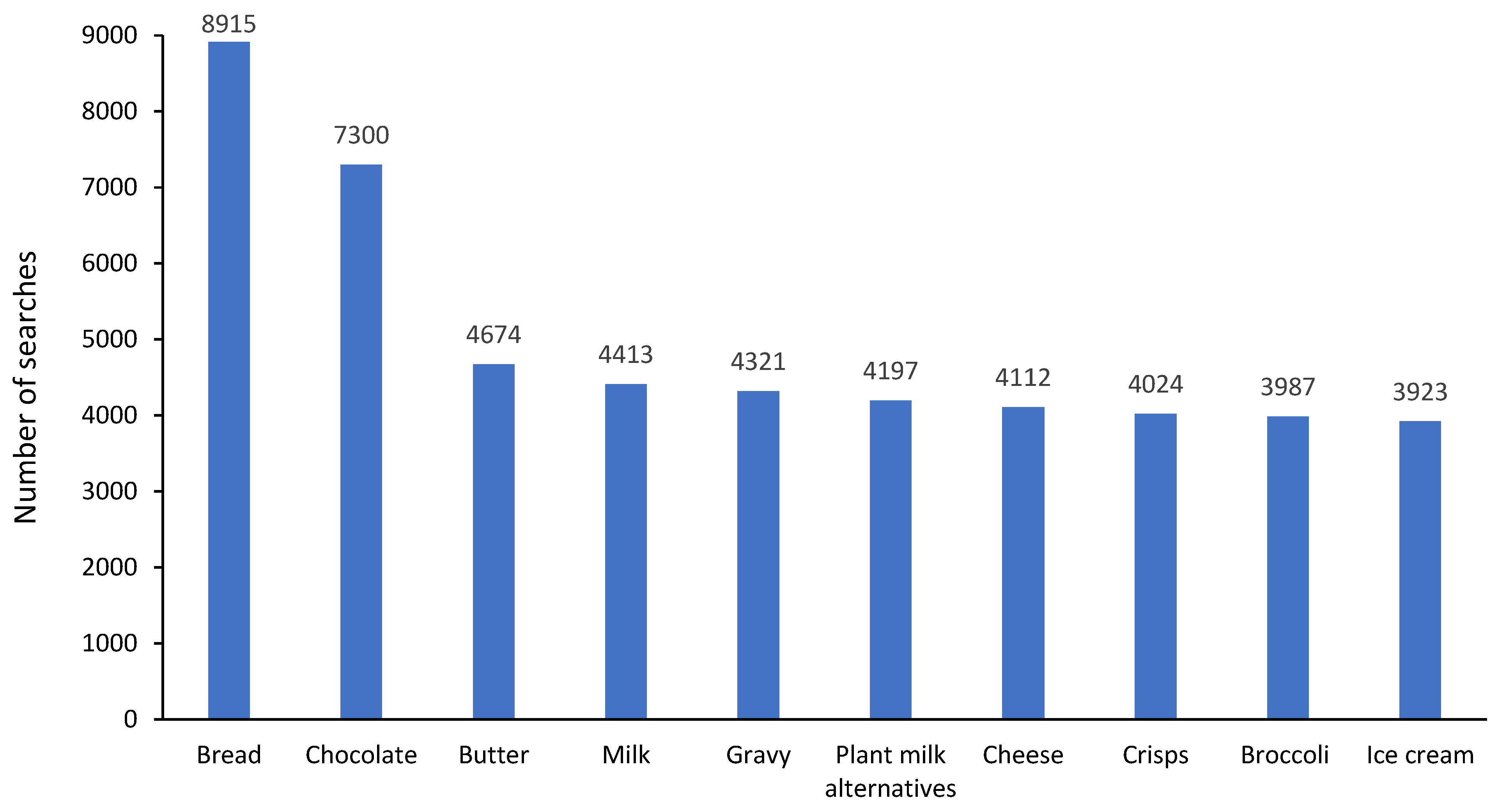
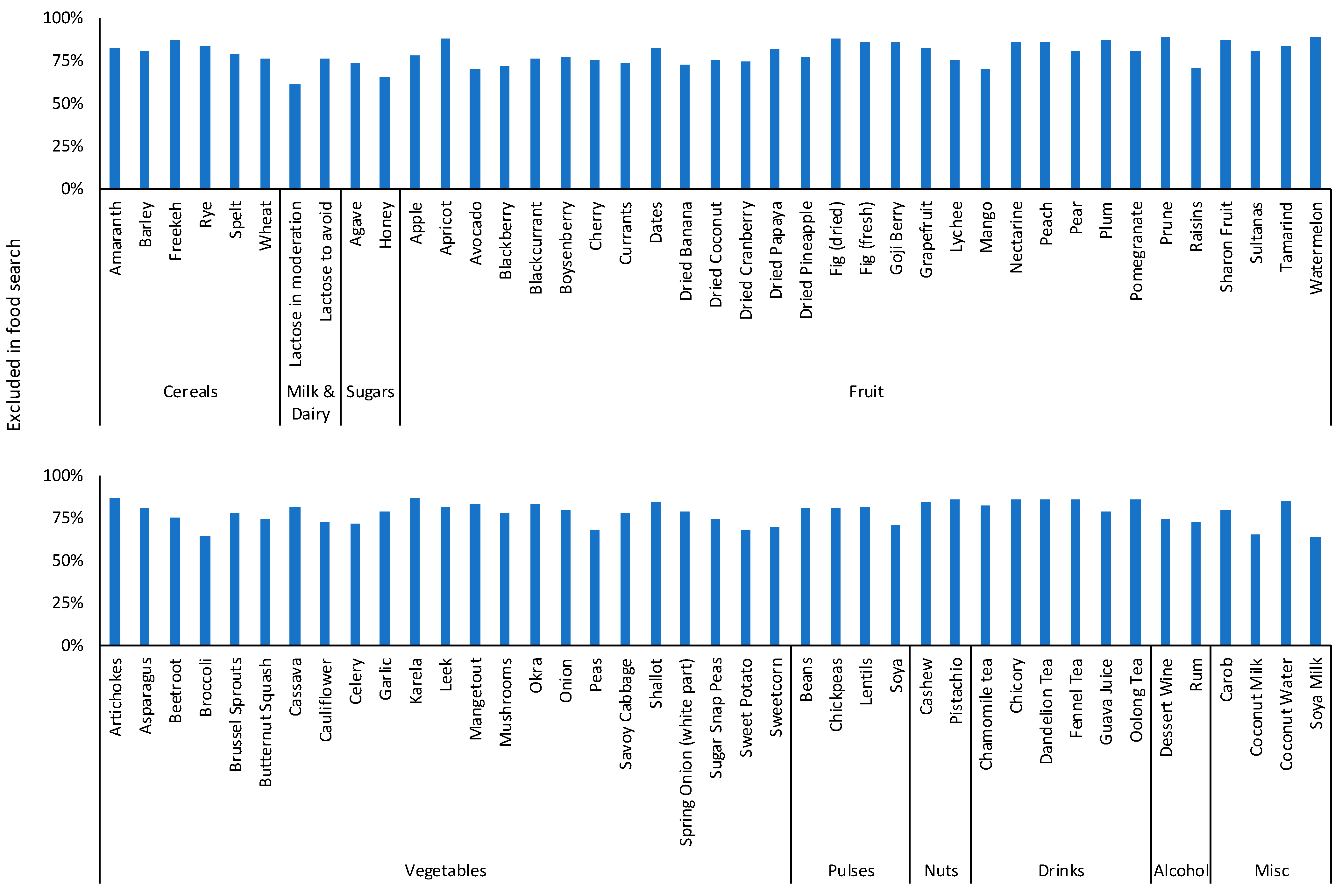
2.4.2. FODMAP Restriction: Symptoms at Baseline and End, and the Most Common Food Searches
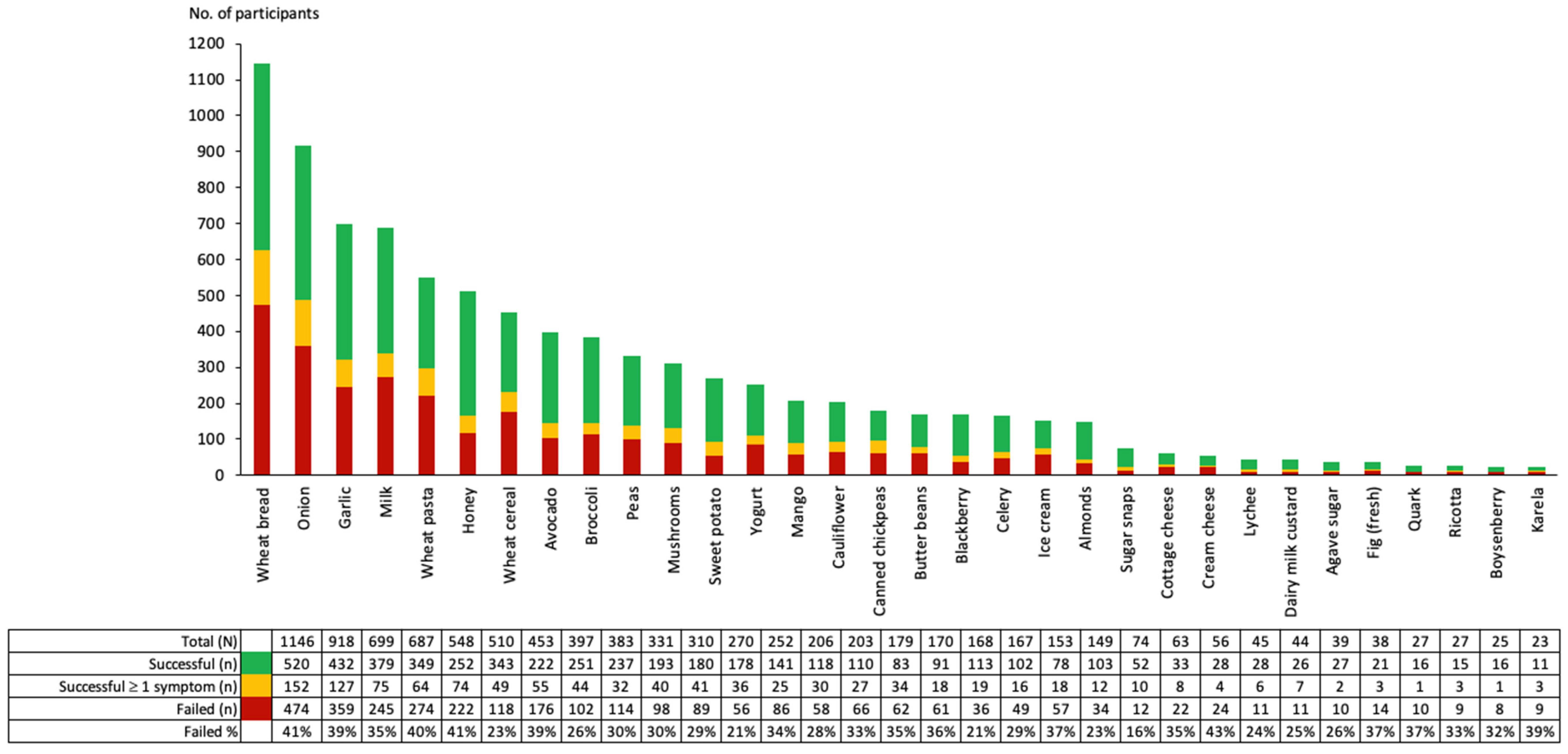
2.4.3. FODMAP Reintroduction: Most Common Food Challenges and Whether Failed or Successful
2.4.4. FODMAP Personalisation: Symptom Data and FODMAP Filters
2.5. Statistical Analyses
3. Results
3.1. FODMAP Restriction
3.2. FODMAP Reintroduction
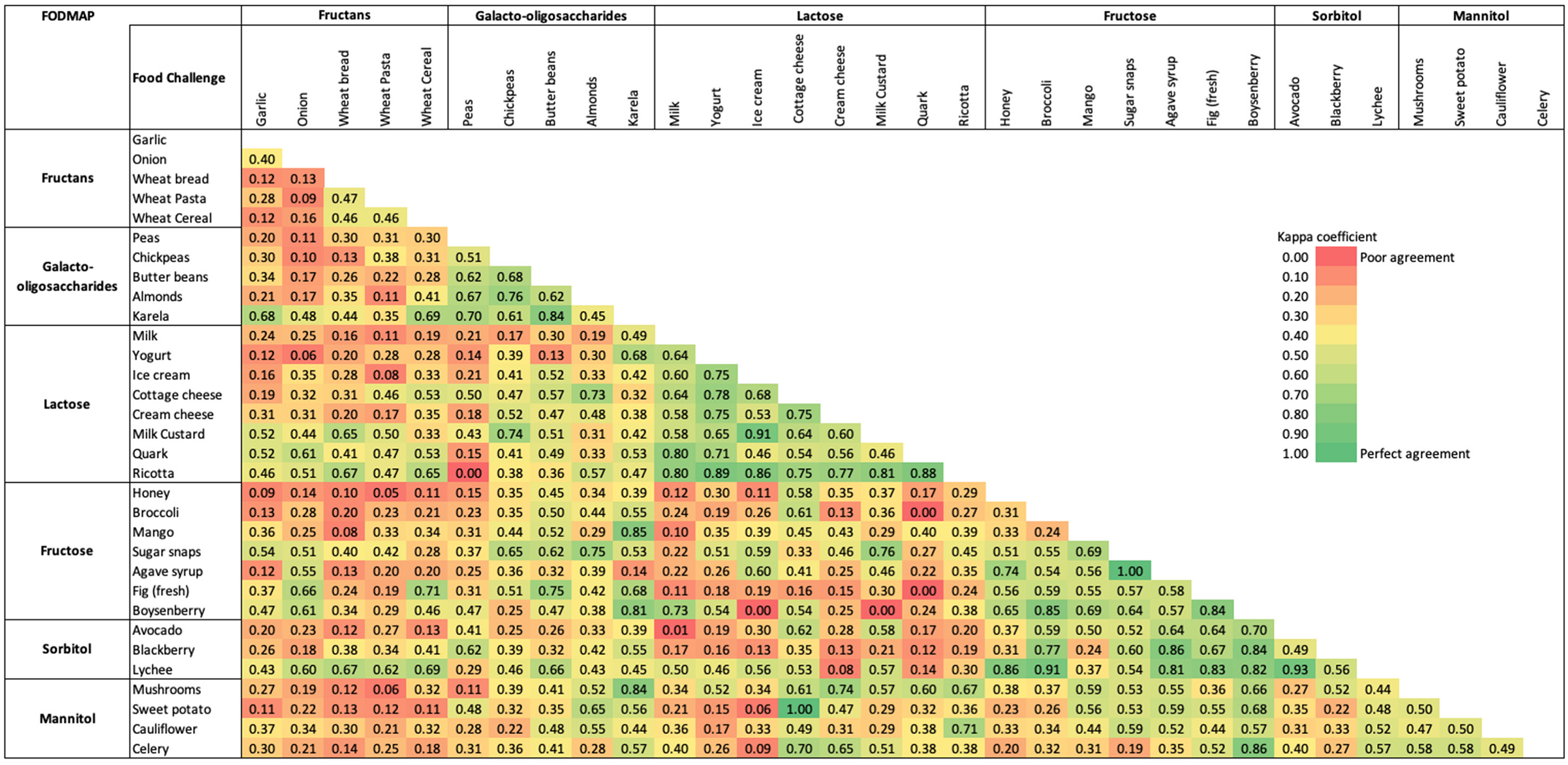
3.3. FODMAP Personalisation
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mearin, F.; Lacy, B.E.; Chang, L.; Chey, W.D.; Lembo, A.J.; Simren, M.; Spiller, R. Bowel Disorders. Gastroenterology 2016, 150, 1393–1407. [Google Scholar]
- Enck, P.; Aziz, Q.; Barbara, G.; Farmer, A.D.; Fukudo, S.; Mayer, E.A.; Niesler, B.; Quigley, E.M.; Rajilic-Stojanovic, M.; Schemann, M.; et al. Irritable bowel syndrome. Nat. Rev. Dis. Primers 2016, 2, 16014. [Google Scholar] [CrossRef] [PubMed]
- Oka, P.; Parr, H.; Barberio, B.; Black, C.J.; Savarino, E.V.; Ford, A.C. Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 908–917. [Google Scholar] [CrossRef] [PubMed]
- Lovell, R.M.; Ford, A.C. Global prevalence of and risk factors for irritable bowel syndrome: A meta-analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721.e4. [Google Scholar] [CrossRef] [PubMed]
- Ballou, S.; McMahon, C.; Lee, H.N.; Katon, J.; Shin, A.; Rangan, V.; Singh, P.; Nee, J.; Camilleri, M.; Lembo, A.; et al. Effects of Irritable Bowel Syndrome on Daily Activities Vary Among Subtypes Based on Results from the IBS in America Survey. Clin. Gastroenterol. Hepatol. 2019, 17, 2471–2478.e3. [Google Scholar] [CrossRef]
- Hahn, B.A.; Yan, S.; Strassels, S. Impact of irritable bowel syndrome on quality of life and resource use in the United States and United Kingdom. Digestion 1999, 60, 77–81. [Google Scholar] [CrossRef]
- Hu, Z.; Li, M.; Yao, L.; Wang, Y.; Wang, E.; Yuan, J.; Wang, F.; Yang, K.; Bian, Z.; Zhong, L.L. The level and prevalence of depression and anxiety among patients with different subtypes of irritable bowel syndrome: A network meta-analysis. BMC Gastroenterol. 2021, 21, 23. [Google Scholar] [CrossRef]
- Chang, L.; Sultan, S.; Lembo, A.; Verne, G.N.; Smalley, W.; Heidelbaugh, J.J. AGA Clinical Practice Guideline on the Pharmacological Management of Irritable Bowel Syndrome with Constipation. Gastroenterology 2022, 163, 118–136. [Google Scholar] [CrossRef]
- Lembo, A.; Sultan, S.; Chang, L.; Heidelbaugh, J.J.; Smalley, W.; Verne, G.N. AGA Clinical Practice Guideline on the Pharmacological Management of Irritable Bowel Syndrome with Diarrhea. Gastroenterology 2022, 163, 137–151. [Google Scholar] [CrossRef]
- Vasant, D.H.; Paine, P.A.; Black, C.J.; Houghton, L.A.; Everitt, H.A.; Corsetti, M.; Agrawal, A.; Aziz, I.; Farmer, A.D.; Eugenicos, M.P.; et al. British Society of Gastroenterology guidelines on the management of irritable bowel syndrome. Gut 2021, 70, 1214–1240. [Google Scholar] [CrossRef]
- McKenzie, Y.A.; Thompson, J.; Gulia, P.; Lomer, M.C. British Dietetic Association systematic review of systematic reviews and evidence-based practice guidelines for the use of probiotics in the management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 2016, 29, 576–592. [Google Scholar] [CrossRef]
- Whelan, K.; Martin, L.D.; Staudacher, H.M.; Lomer, M.C.E. The low FODMAP diet in the management of irritable bowel syndrome: An evidence-based review of FODMAP restriction, reintroduction and personalisation in clinical practice. J. Hum. Nutr. Diet. 2018, 31, 239–255. [Google Scholar] [CrossRef]
- van Lanen, A.S.; de Bree, A.; Greyling, A. Efficacy of a low-FODMAP diet in adult irritable bowel syndrome: A systematic review and meta-analysis. Eur. J. Nutr. 2021, 60, 3505–3522. [Google Scholar] [CrossRef]
- Wang, J.; Yang, P.; Zhang, L.; Hou, X. A Low-FODMAP Diet Improves the Global Symptoms and Bowel Habits of Adult IBS Patients: A Systematic Review and Meta-Analysis. Front. Nutr. 2021, 8, 683191. [Google Scholar] [CrossRef]
- Black, C.J.; Staudacher, H.M.; Ford, A.C. Efficacy of a low FODMAP diet in irritable bowel syndrome: Systematic review and network meta-analysis. Gut 2022, 71, 1117–1126. [Google Scholar] [CrossRef]
- McKenzie, Y.A.; Bowyer, R.K.; Leach, H.; Gulia, P.; Horobin, J.; O’Sullivan, N.A.; Pettitt, C.; Reeves, L.B.; Seamark, L.; Williams, M.; et al. British Dietetic Association systematic review and evidence-based practice guidelines for the dietary management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 2016, 29, 549–575. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Irritable Bowel Syndrome in Adults: Diagnosis and Management Clinical Guideline [CG61] 2017. Available online: https://www.nice.org.uk/guidance/cg61 (accessed on 12 March 2023).
- So, D.; Loughman, A.; Staudacher, H.M. Effects of a low FODMAP diet on the colonic microbiome in irritable bowel syndrome: A systematic review with meta-analysis. Am. J. Clin. Nutr. 2022, 116, 943–952. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Ralph, F.S.E.; Irving, P.M.; Whelan, K.; Lomer, M.C.E. Nutrient Intake, Diet Quality, and Diet Diversity in Irritable Bowel Syndrome and the Impact of the Low FODMAP Diet. J. Acad. Nutr. Diet. 2020, 120, 535–547. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Rossi, M.; Kaminski, T.; Dimidi, E.; Ralph, F.S.; Wilson, B.; Martin, L.D.; Louis, P.; Lomer, M.C.; Irving, P.M.; et al. Long-term personalized low FODMAP diet improves symptoms and maintains luminal Bifidobacteria abundance in irritable bowel syndrome. Neurogastroenterol. Motil. 2022, 34, e14241. [Google Scholar] [CrossRef]
- O’Keeffe, M.; Lomer, M.C. Who should deliver the low FODMAP diet and what educational methods are optimal: A review. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. 1), 23–26. [Google Scholar] [CrossRef]
- Chen, J.; Lieffers, J.; Bauman, A.; Hanning, R.; Allman-Farinelli, M. The use of smartphone health apps and other mobile health (mHealth) technologies in dietetic practice: A three country study. J. Hum. Nutr. Diet. 2017, 30, 439–452. [Google Scholar] [CrossRef] [PubMed]
- Whelan, K. Group education by dietitians in patients with gastrointestinal disorders: Potentially clinically effective and time for randomised trials. J. Hum. Nutr. Diet. 2022, 35, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Whigham, L.; Joyce, T.; Harper, G.; Irving, P.M.; Staudacher, H.M.; Whelan, K.; Lomer, M.C.E. Clinical effectiveness and economic costs of group versus one-to-one education for short-chain fermentable carbohydrate restriction (low FODMAP diet) in the management of irritable bowel syndrome. J. Hum. Nutr. Diet. 2015, 28, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Trott, N.; Aziz, I.; Rej, A.; Surendran Sanders, D. How Patients with IBS Use Low FODMAP Dietary Information Provided by General Practitioners and Gastroenterologists: A Qualitative Study. Nutrients 2019, 11, 1313. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.; Cox, S.R.; Whelan, K. Challenges of the low FODMAP diet for managing irritable bowel syndrome and approaches to their minimisation and mitigation. Proc. Nutr. Soc. 2021, 80, 19–28. [Google Scholar] [CrossRef]
- Ankersen, D.V.; Weimers, P.; Burisch, J. Whats ‘App-ening’: The help of new technologies in nutrition in digestive diseases. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 426–431. [Google Scholar] [CrossRef]
- Varney, J.; Barrett, J.; Scarlata, K.; Catsos, P.; Gibson, P.R.; Muir, J.G. FODMAPs: Food composition, defining cutoff values and international application. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. 1), 53–61. [Google Scholar] [CrossRef]
- Tuck, C.; Barrett, J. Re-challenging FODMAPs: The low FODMAP diet phase two. J. Gastroenterol. Hepatol. 2017, 32 (Suppl. 1), 11–15. [Google Scholar] [CrossRef]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Lomer, M.C.; Anderson, J.L.; Barrett, J.S.; Muir, J.G.; Irving, P.M.; Whelan, K. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J. Nutr. 2012, 142, 1510–1518. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Lomer, M.C.E.; Farquharson, F.M.; Louis, P.; Fava, F.; Franciosi, E.; Scholz, M.; Tuohy, K.M.; Lindsay, J.O.; Irving, P.M.; et al. A Diet Low in FODMAPs Reduces Symptoms in Patients with Irritable Bowel Syndrome and A Probiotic Restores Bifidobacterium Species: A Randomized Controlled Trial. Gastroenterology 2017, 153, 936–947. [Google Scholar] [CrossRef]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef]
- Wilson, B.; Rossi, M.; Kanno, T.; Parkes, G.C.; Anderson, S.; Mason, A.J.; Irving, P.M.; Lomer, M.C.; Whelan, K. beta-Galactooligosaccharide in Conjunction With Low FODMAP Diet Improves Irritable Bowel Syndrome Symptoms but Reduces Fecal Bifidobacteria. Am. J. Gastroenterol. 2020, 115, 906–915. [Google Scholar] [CrossRef]
- Halmos, E.P.; Power, V.A.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014, 146, 67–75.e5. [Google Scholar] [CrossRef]
- Rej, A.; Sanders, D.S.; Shaw, C.C.; Buckle, R.; Trott, N.; Agrawal, A.; Aziz, I. Efficacy and Acceptability of Dietary Therapies in Non-Constipated Irritable Bowel Syndrome: A Randomized Trial of Traditional Dietary Advice, the Low FODMAP Diet, and the Gluten-Free Diet. Clin. Gastroenterol. Hepatol. 2022, 20, 2876–2887.e15. [Google Scholar] [CrossRef]
- Dimidi, E.; Whelan, K.; Lomer, M.C.E. Investigating optimal education regarding the low FODMAP diet in functional bowel disorders: A feasibility randomised controlled trial of leaflet vs mobile application vs dietetic consultation. Proc. Nutr. Soc. 2020, 79, E7. [Google Scholar] [CrossRef]
- Gravina, A.G.; Dallio, M.; Romeo, M.; Di Somma, A.; Cotticelli, G.; Loguercio, C.; Federico, A. Adherence and Effects Derived from FODMAP Diet on Irritable Bowel Syndrome: A Real Life Evaluation of a Large Follow-Up Observation. Nutrients 2020, 12, 928. [Google Scholar] [CrossRef]
- Rafferty, A.J.; Hall, R.; Johnston, C.S. A Novel Mobile App (Heali) for Disease Treatment in Participants with Irritable Bowel Syndrome: Randomized Controlled Pilot Trial. J. Med. Internet Res. 2021, 23, e24134. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Whelan, K. The low FODMAP diet: Recent advances in understanding its mechanisms and efficacy in IBS. Gut 2017, 66, 1517–1527. [Google Scholar] [CrossRef]
- Katsirma, Z.; Dimidi, E.; Rodriguez-Mateos, A.; Whelan, K. Fruits and their impact on the gut microbiota, gut motility and constipation. Food Funct. 2021, 12, 8850–8866. [Google Scholar] [CrossRef]
- Chen, J.; Lieffers, J.; Bauman, A.; Hanning, R.; Allman-Farinelli, M. Designing Health Apps to Support Dietetic Professional Practice and Their Patients: Qualitative Results from an International Survey. JMIR Mhealth Uhealth 2017, 5, e40. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.J.; Reed, D.E.; Muir, J.G.; Vanner, S.J. Implementation of the low FODMAP diet in functional gastrointestinal symptoms: A real-world experience. Neurogastroenterol. Motil. 2020, 32, e13730. [Google Scholar] [CrossRef] [PubMed]
- Seamark, L.; Barclay, Y.; Marchant, C.; Williams, M.; Hickson, M. Long-term symptom severity in people with irritable bowel syndrome following dietetic treatment in primary care: A service evaluation. J. Hum. Nutr. Diet. 2021, 34, 890–900. [Google Scholar] [CrossRef] [PubMed]
- Lackner, J.M.; Jaccard, J.; Keefer, L.; Firth, R.; Carosella, A.M.; Sitrin, M.; Brenner, D.; Representing the IBSOS Research Group. The accuracy of patient-reported measures for GI symptoms: A comparison of real time and retrospective reports. Neurogastroenterol. Motil. 2014, 26, 1802–1811. [Google Scholar] [CrossRef]
- Beckers, A.B.; Snijkers, J.T.W.; Weerts, Z.Z.R.M.; Vork, L.; Klaassen, T.; Smeets, F.G.M.; Masclee, A.A.M.; Keszthelyi, D. Digital Instruments for Reporting of Gastrointestinal Symptoms in Clinical Trials: Comparison of End-of-Day Diaries Versus the Experience Sampling Method. JMIR Form. Res. 2021, 5, e31678. [Google Scholar] [CrossRef]
- Habuš, M.; Mykolenko, S.; Iveković, S.; Pastor, K.; Kojić, J.; Drakula, S.; Ćurić, D.; Novotni, D. Bioprocessing of Wheat and Amaranth Bran for the Reduction of Fructan Levels and Application in 3D-Printed Snacks. Foods 2022, 11, 1649. [Google Scholar] [CrossRef]
- Pejcz, E.; Spychaj, R.; Gil, Z. Technological Methods for Reducing the Content of Fructan in Wheat Bread. Foods 2019, 8, 663. [Google Scholar] [CrossRef]
- Mollakhalili-Meybodi, N.; Ehrampoush, M.H.; Hajimohammadi, B.; Mosaddegh, M.H. Formulation optimization of functional wheat bread with low glycemic index from technological and nutritional perspective. Food Sci. Nutr. 2023, 11, 284–294. [Google Scholar] [CrossRef]
- Whelan, K.; Abrahmsohn, O.; David, G.J.P.; Staudacher, H.; Irving, P.; Lomer, M.C.E.; Ellis, P.R. Fructan content of commonly consumed wheat, rye and gluten-free breads. Int. J. Food Sci. Nutr. 2011, 62, 498–503. [Google Scholar] [CrossRef]
- Thomas, B.; Rabinowitch, H.D. (Eds.) Future prospects for allium crops. In Edible Alliums: Botany, Production and Uses; CABI: Wallingford, UK, 2022; pp. 296–300. [Google Scholar]
- Rej, A.; Trott, N.; Aziz, I.; Sanders, D.S. A Gluten-Free Diet: The Express Route to Fructan Reduction. Am. J. Gastroenterol. 2019, 114, 1553. [Google Scholar] [CrossRef]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierød, M.B.; Henriksen, C.; Lundin, K.E. Fructan, Rather Than Gluten, Induces Symptoms in Patients with Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterology 2018, 154, 529–539.e2. [Google Scholar] [CrossRef]
- Mohseni, F.; Agah, S.; Ebrahimi-Daryani, N.; Taher, M.; Nattagh-Eshtivani, E.; Karimi, S.; Rastgoo, S.; Bourbour, F.; Hekmatdoost, A. The effect of low FODMAP diet with and without gluten on irritable bowel syndrome: A double blind, placebo controlled randomized clinical trial. Clin. Nutr. ESPEN 2022, 47, 45–50. [Google Scholar] [CrossRef]
- Lomer, M.C.; Parkes, G.C.; Sanderson, J.D. Review article: Lactose intolerance in clinical practice--myths and realities. Aliment. Pharmacol. Ther. 2008, 27, 93–103. [Google Scholar] [CrossRef]
- O’Keeffe, M.; Jansen, C.; Martin, L.; Williams, M.; Seamark, L.; Staudacher, H.M.; Irving, P.M.; Whelan, K.; Lomer, M.C. Long-term impact of the low-FODMAP diet on gastrointestinal symptoms, dietary intake, patient acceptability, and healthcare utilization in irritable bowel syndrome. Neurogastroenterol. Motil. 2018, 30, e13154. [Google Scholar] [CrossRef]
- Bohn, L.; Storsrud, S.; Tornblom, H.; Bengtsson, U.; Simren, M. Self-reported food-related gastrointestinal symptoms in IBS are common and associated with more severe symptoms and reduced quality of life. Am. J. Gastroenterol. 2013, 108, 634–641. [Google Scholar] [CrossRef]
- Hayes, P.; Corish, C.; O’Mahony, E.; Quigley, E.M. A dietary survey of patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2014, 27 (Suppl. 2), 36–47. [Google Scholar] [CrossRef]
- Bellini, M.; Tonarelli, S.; Barracca, F.; Morganti, R.; Pancetti, A.; Bertani, L.; De Bortoli, N.; Costa, F.; Mosca, M.; Marchi, S.; et al. A Low-FODMAP Diet for Irritable Bowel Syndrome: Some Answers to the Doubts from a Long-Term Follow-Up. Nutrients 2020, 12, 2360. [Google Scholar] [CrossRef]
- Lomer, M.C. Review article: The aetiology, diagnosis, mechanisms and clinical evidence for food intolerance. Aliment. Pharmacol. Ther. 2015, 41, 262–275. [Google Scholar] [CrossRef]
- Johansson, C. Studies of gastrointestinal interactions. VII. Characteristics of the absorption pattern of sugar, fat and protein from composite meals in man. A quantitative study. Scand. J. Gastroenterol. 1975, 10, 33–42. [Google Scholar] [CrossRef]
- Lentle, R.G.; Sequeira, I.R.; Hardacre, A.K.; Reynolds, G. A method for assessing real time rates of dissolution and absorption of carbohydrate and other food matrices in human subjects. Food Funct. 2016, 7, 2820–2832. [Google Scholar] [CrossRef]
- Cox, S.R.; Prince, A.C.; Myers, C.E.; Irving, P.M.; Lindsay, J.O.; Lomer, M.C.; Whelan, K. Fermentable Carbohydrates [FODMAPs] Exacerbate Functional Gastrointestinal Symptoms in Patients with Inflammatory Bowel Disease: A Randomised, Double-blind, Placebo-controlled, Cross-over, Re-challenge Trial. J. Crohn’s Colitis 2017, 11, 1420–1429. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, S.J.; Parker, F.C.; Muir, J.G.; Gibson, P.R. Dietary triggers of abdominal symptoms in patients with irritable bowel syndrome: Randomized placebo-controlled evidence. Clin. Gastroenterol. Hepatol. 2008, 6, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Major, G.; Pritchard, S.; Murray, K.; Alappadan, J.P.; Hoad, C.L.; Marciani, L.; Gowland, P.; Spiller, R. Colon Hypersensitivity to Distension, Rather Than Excessive Gas Production, Produces Carbohydrate-Related Symptoms in Individuals with Irritable Bowel Syndrome. Gastroenterology 2017, 152, 124–133.e2. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Wilkinson-Smith, V.; Hoad, C.; Costigan, C.; Cox, E.; Lam, C.; Marciani, L.; Gowland, P.; Spiller, R.C. Differential effects of FODMAPs (fermentable, o.l.i.g.o.-; di-; mono-saccharides polyols) on small large intestinal contents in healthy subjects shown by, M.R.I. Am. J. Gastroenterol. 2014, 109, 110–119. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Irving, P.M.; Lomer, M.C.; Whelan, K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 256–266. [Google Scholar] [CrossRef]
- Peters, S.L.; Gibson, P.R.; Halmos, E.P. Smartphone app-delivered gut-directed hypnotherapy improves symptoms of self-reported irritable bowel syndrome: A retrospective evaluation. Neurogastroenterol. Motil. 2023, 35, e14533. [Google Scholar] [CrossRef]
- Thompson, W.G.; Heaton, K.W.; Smyth, G.T.; Smyth, C. Irritable bowel syndrome: The view from general practice. Eur. J. Gastroenterol. Hepatol. 1997, 9, 689–692. [Google Scholar] [CrossRef]
- Lenhart, A.; Ferch, C.; Shaw, M.; Chey, W.D. Use of Dietary Management in Irritable Bowel Syndrome: Results of a Survey of Over 1500 United States Gastroenterologists. J. Neurogastroenterol. Motil. 2018, 24, 437–451. [Google Scholar] [CrossRef]

| Symptom | Baseline n (%) | End of FODMAP Restriction n (%) | p Value |
|---|---|---|---|
| Overall symptoms | 11,689 (100%) | 6116 (52%) | <0.001 |
| Abdominal pain | 8196 (100%) | 3966 (48%) | <0.001 |
| Bloating | 11,265 (100%) | 6170 (55%) | <0.001 |
| Flatulence | 10,318 (100%) | 5289 (51%) | <0.001 |
| Diarrhoea | 6284 (100%) | 2808 (45%) | <0.001 |
| Constipation | 5448 (100%) | 3013 (55%) | <0.001 |
| Covariates * | Hazard Ratio | 95% Confidence Intervals | p Value |
|---|---|---|---|
| Fructans (n failed = 939, N total= 2901) | |||
| Abdominal pain | 2.36 | 1.99–2.80 | <0.001 |
| Bloating | 3.12 | 2.60–3.76 | <0.001 |
| Flatulence | 1.20 | 1.02–1.41 | 0.028 |
| Diarrhoea | 1.56 | 1.34–1.81 | <0.001 |
| Constipation | 1.20 | 1.02–1.41 | 0.028 |
| GOS (n failed = 157, N total = 664) | |||
| Abdominal pain | 3.35 | 2.10–5.35 | <0.001 |
| Bloating | 3.05 | 1.82–5.13 | <0.001 |
| Flatulence | 2.43 | 1.52–3.89 | <0.001 |
| Diarrhoea | 1.31 | 0.90–1.93 | 0.163 |
| Constipation | 1.28 | 0.84–1.93 | 0.252 |
| Lactose (n failed = 325, N total = 1044) | |||
| Abdominal pain | 3.31 | 2.38–4.61 | <0.001 |
| Bloating | 2.38 | 1.69–3.36 | <0.001 |
| Flatulence | 1.37 | 1.05–1.78 | 0.021 |
| Diarrhoea | 1.81 | 1.40–2.35 | <0.001 |
| Constipation | 1.48 | 1.12–1.94 | 0.005 |
| Fructose (n failed = 188, N total = 1029) | |||
| Abdominal pain | 3.07 | 2.02–4.67 | <0.001 |
| Bloating | 3.99 | 2.61–6.10 | <0.001 |
| Flatulence | 2.32 | 1.56–3.45 | <0.001 |
| Diarrhoea | 1.99 | 1.36–2.92 | <0.001 |
| Constipation | 1.01 | 0.67–1.53 | 0.951 |
| Sorbitol (n failed = 149, N total = 610) | |||
| Abdominal pain | 2.72 | 1.75–4.25 | <0.001 |
| Bloating | 3.75 | 2.35–6.00 | <0.001 |
| Flatulence | 1.60 | 1.01–2.51 | 0.044 |
| Diarrhoea | 1.39 | 0.90–2.15 | 0.141 |
| Constipation | 0.76 | 0.49–1.19 | 0.229 |
| Mannitol (n failed = 154, N total = 761) | |||
| Abdominal pain | 0.76 | 0.49–1.17 | 0.205 |
| Bloating | 3.26 | 2.01–5.30 | <0.001 |
| Flatulence | 2.40 | 1.52–3.78 | <0.001 |
| Diarrhoea | 1.81 | 1.24–2.64 | 0.002 |
| Constipation | 1.14 | 0.74–1.75 | 0.553 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dimidi, E.; Belogianni, K.; Whelan, K.; Lomer, M.C.E. Gut Symptoms during FODMAP Restriction and Symptom Response to Food Challenges during FODMAP Reintroduction: A Real-World Evaluation in 21,462 Participants Using a Mobile Application. Nutrients 2023, 15, 2683. https://doi.org/10.3390/nu15122683
Dimidi E, Belogianni K, Whelan K, Lomer MCE. Gut Symptoms during FODMAP Restriction and Symptom Response to Food Challenges during FODMAP Reintroduction: A Real-World Evaluation in 21,462 Participants Using a Mobile Application. Nutrients. 2023; 15(12):2683. https://doi.org/10.3390/nu15122683
Chicago/Turabian StyleDimidi, Eirini, Katerina Belogianni, Kevin Whelan, and Miranda C. E. Lomer. 2023. "Gut Symptoms during FODMAP Restriction and Symptom Response to Food Challenges during FODMAP Reintroduction: A Real-World Evaluation in 21,462 Participants Using a Mobile Application" Nutrients 15, no. 12: 2683. https://doi.org/10.3390/nu15122683
APA StyleDimidi, E., Belogianni, K., Whelan, K., & Lomer, M. C. E. (2023). Gut Symptoms during FODMAP Restriction and Symptom Response to Food Challenges during FODMAP Reintroduction: A Real-World Evaluation in 21,462 Participants Using a Mobile Application. Nutrients, 15(12), 2683. https://doi.org/10.3390/nu15122683





