Sodium Butyrate Effectiveness in Children and Adolescents with Newly Diagnosed Inflammatory Bowel Diseases—Randomized Placebo-Controlled Multicenter Trial
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zuo, T.; Ng, S.C. The Gut Microbiota in the Pathogenesis and Therapeutics of Inflammatory Bowel Disease. Front. Microbiol. 2018, 9, 2247. [Google Scholar] [CrossRef] [PubMed]
- Sha, S.; Xu, B.; Wang, X.; Zhang, Y.; Wang, H.; Kong, X.; Zhu, H.; Wu, K. The biodiversity and composition of the dominant fecal microbiota in patients with inflammatory bowel disease. Diagn. Microbiol. Infect. Dis. 2013, 75, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Canani, R.B.; Costanzo, M.D.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519–1528. [Google Scholar] [CrossRef] [PubMed]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.J.; Brummer, R.-J. Review article: The role of butyrate on colonic function. Aliment. Pharmacol. Ther. 2008, 27, 104–119. [Google Scholar] [CrossRef] [PubMed]
- Salvi, P.S.; Cowles, R.A. Butyrate and the Intestinal Epithelium: Modulation of Proliferation and Inflammation in Homeostasis and Disease. Cells 2021, 10, 1775. [Google Scholar] [CrossRef]
- Sitkin, S.; Vakhitov, T.; Pokrotnieks, J. Oral butyrate modulates the gut microbiota in patients with inflammatory bowel disease, most likely by reversing proinflammatory metabolic reprogramming of colonocytes. Neurogastroenterol. Motil. 2021, 33, e14038. [Google Scholar] [CrossRef]
- Dąbek-Drobny, A.; Kaczmarczyk, O.; Woźniakiewicz, M.; Paśko, P.; Dobrowolska-Iwanek, J.; Woźniakiewicz, A.; Piątek-Guziewicz, A.; Zagrodzki, P.; Zwolińska-Wcisło, M. Association between Fecal Short-Chain Fatty Acid Levels, Diet, and Body Mass Index in Patients with Inflammatory Bowel Disease. Biology 2022, 11, 108. [Google Scholar] [CrossRef]
- Kuczyńska, B.; Wasilewska, A.; Biczysko, M.; Banasiewicz, T.; Drews, M. Krótkołańcuchowe kwasy tłuszczowe – mechanizmy działania, potencjalne zastosowania kliniczne oraz zalecenia dietetyczne. [Short-chain fatty acids—Mechanisms of action, potential clinical use and dietary recommendations]. Now. Lek. 2011, 80, 299–304. [Google Scholar]
- Iversen, K.N.; Dicksved, J.; Zoki, C.; Fristedt, R.; Pelve, E.A.; Langton, M.; Landberg, R. The Effects of High Fiber Rye, Compared to Refined Wheat, on Gut Microbiota Composition, Plasma Short Chain Fatty Acids, and Implications for Weight Loss and Metabolic Risk Factors (the RyeWeight Study). Nutrients 2022, 14, 1669. [Google Scholar] [CrossRef]
- Couto, M.R.; Gonçalves, P.; Magro, F.; Martel, F. Microbiota-derived butyrate regulates intestinal inflammation: Focus on inflammatory bowel disease. Pharmacol. Res. 2020, 159, 104947. [Google Scholar] [CrossRef]
- Gonçalves, P.; Araújo, J.R.; Di Santo, J.P. A Cross-Talk Between Microbiota-Derived Short-Chain Fatty Acids and the Host Mucosal Immune System Regulates Intestinal Homeostasis and Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2018, 24, 558–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, G.; Lin, J.; Zhang, C.; Gao, H.; Lu, H.; Gao, X.; Zhu, R.; Li, Z.; Li, M.; Liu, Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes 2021, 13, 1968257. [Google Scholar] [CrossRef] [PubMed]
- Vieira, E.L.; Leonel, A.J.; Sad, A.P.; Beltrão, N.R.; Costa, T.F.; Ferreira, T.M.; Gomes-Santos, A.C.; Faria, A.M.; Peluzio, M.C.; Cara, D.C.; et al. Oral administration of sodium butyrate attenuates inflammation and mucosal lesion in experimental acute ulcerative colitis. J. Nutr. Biochem. 2012, 23, 430–436. [Google Scholar] [CrossRef]
- Turner, D.; Otley, A.R.; Mack, D.; Hyams, J.; de Bruijne, J.; Uusoue, K.; Walters, T.D.; Zachos, M.; Mamula, P.; Beaton, D.E.; et al. Development, Validation, and Evaluation of a Pediatric Ulcerative Colitis Activity Index: A Prospective Multicenter Study. Gastroenterology 2007, 133, 423–432. [Google Scholar] [CrossRef]
- Hyams, J.S.; Ferry, G.D.; Mandel, F.S.; Gryboski, J.D.; Kibort, P.M.; Kirschner, B.S.; Griffiths, A.M.; Katz, A.J.; Grand, R.J.; Boyle, J.T.; et al. Development and validation of a pediatric Crohn’s disease activity index. J. Pediatr. Gastroenterol. Nutr. 1991, 12, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Facchin, S.; Vitulo, N.; Calgaro, M.; Buda, A.; Romualdi, C.; Pohl, D.; Perini, B.; Lorenzon, G.; Marinelli, C.; D’Incà, R.; et al. Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel disease. Neurogastroenterol. Motil. 2020, 32, e13914. [Google Scholar] [CrossRef] [PubMed]
- Vernia, P.; Monteleone, G.; Grandinetti, G.; Villotti, G.; Di Giulio, E.; Frieri, G.; Marcheggiano, A.; Pallone, F.; Caprilli, R.; Torsoli, A. Combined oral sodium butyrate and mesalazine treatment compared to oral mesalazine alone in ulcerative colitis: Randomized, double-blind, placebo-controlled pilot study. Dig. Dis. Sci. 2000, 45, 976–981. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Morera, R.; Ciccocioppo, R.; Cazzola, P.; Gotti, S.; Tinozzi, F.P.; Tinozzi, S.; Corazza, G.R. Oral butyrate for mildly to moderately active Crohn’s disease. Aliment. Pharmacol. Ther. 2005, 22, 789–794. [Google Scholar] [CrossRef]
- Assisi, R.F.; GISDI Study Group. Combined butyric acid/mesalazine treatment in ulcerative colitis with mild-moderate activity. Results of a multicentre pilot study. Minerva Gastroenterol. Dietol. 2008, 54, 231–238. [Google Scholar]
- Jamka, M.; Kokot, M.; Kaczmarek, N.; Bermagambetova, S.; Nowak, J.K.; Walkowiak, J. The effect of sodium butyrate enemas compared with placebo on disease activity, endoscopic scores, and histological and inflammatory parameters in inflammatory bowel diseases: A systematic review of randomised controlled trials. Complement Med. Res. 2021, 28, 344–356. [Google Scholar] [CrossRef]
- Spina, L.; Cavallaro, F.; Fardowza, N.; Lagoussis, P.; Bona, D.; Ciscato, C.; Rigante, A.; Vecchi, M. Butyric acid: Pharmacological aspects and routes of administration. Dig. Liver Dis. Suppl. 2007, 1, 7–11. [Google Scholar] [CrossRef]
- Cole, E.T.; Scott, R.A.; Connor, A.L.; Wilding, I.R.; Petereit, H.-U.; Schminke, C.; Beckert, T.; Cadé, D. Enteric coated HPMC capsules designed to achieve intestinal targeting. Int. J. Pharm. 2002, 231, 83–95. [Google Scholar] [CrossRef]
- Banasiewicz, T.; Borycka-Kiciak, K.; Kiciak, A. Porównanie profilów uwalniania maślanu sodu w jelicie dla produktów dostępnych na polskim rynku (ang. Comparison of release profiles sodium butyrate in the intestine for products available on the Polish market). Farm. Prakt. 2019, 12, 40–42. [Google Scholar]
- Roda, A.; Simoni, P.; Magliulo, M.; Nanni, P.; Baraldini, M.; Roda, G.; Roda, E. A new oral formulation for the release of sodium butyrate in the ileo-cecal region and colon. World J. Gastroenterol. 2007, 21, 1079–1084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lührs, H.; Gerke, T.; Müller, J.G.; Melcher, R.; Schauber, J.; Boxberger, F.; Scheppach, W.; Menzel, T. Butyrate inhibits NF-kappaB activation in lamina propria macrophages of patients with ulcerative colitis. Scand. J. Gastroenterol. 2002, 37, 458–466. [Google Scholar] [CrossRef]
- Vernero, M.; De Blasio, F.; Ribaldone, D.; Bugianesi, E.; Pellicano, R.; Saracco, G.; Astegiano, M.; Caviglia, G. The Usefulness of Microencapsulated Sodium Butyrate Add-On Therapy in Maintaining Remission in Patients with Ulcerative Colitis: A Prospective Observational Study. J. Clin. Med. 2020, 9, 3941. [Google Scholar] [CrossRef]
- Dou, X.; Gao, N.; Yan, D.; Shan, A. Sodium Butyrate Alleviates Mouse Colitis by Regulating Gut Microbiota Dysbiosis. Animals 2020, 10, 1154. [Google Scholar] [CrossRef]
- Chen, G.; Ran, X.; Li, B.; Li, Y.; He, D.; Huang, B.; Fu, S.; Liu, J.; Wang, W. Sodium Butyrate Inhibits Inflammation and Maintains Epithelium Barrier Integrity in a TNBS-induced Inflammatory Bowel Disease Mice Model. EBioMedicine 2018, 30, 317–325. [Google Scholar] [CrossRef] [Green Version]
- Simeoli, R.; Mattace Raso, G.; Pirozzi, C.; Lama, A.; Santoro, A.; Russo, R.; Montero-Melendez, T.; Berni Canani, R.; Calignano, A.; Perretti, M.; et al. An orally administered butyrate-releasing derivative reduces neutrophil recruitment and inflammation in dextran sulphate sodium-induced murine colitis. Br. J. Pharmacol. 2017, 174, 1484–1496. [Google Scholar] [CrossRef] [Green Version]
- Vancamelbeke, M.; Laeremans, T.; Vanhove, W.; Arnauts, K.; Ramalho, A.S.; Farré, R.; Cleynen, I.; Ferrante, M.; Vermeire, S. Butyrate Does Not Protect Against Inflammation-induced Loss of Epithelial Barrier Function and Cytokine Production in Primary Cell Monolayers From Patients With Ulcerative Colitis. J. Crohns. Colitis. 2019, 13, 1351–1361. [Google Scholar] [CrossRef] [Green Version]
- Banasiewicz, T.; Domagalska, D.; Borycka-Kiciak, K.; Rydzewska, G. Determination of butyric acid dosage based on clinical and experimental studies—A literature review. Prz. Gastroenterol. 2020, 15, 119–125. [Google Scholar] [CrossRef] [PubMed]
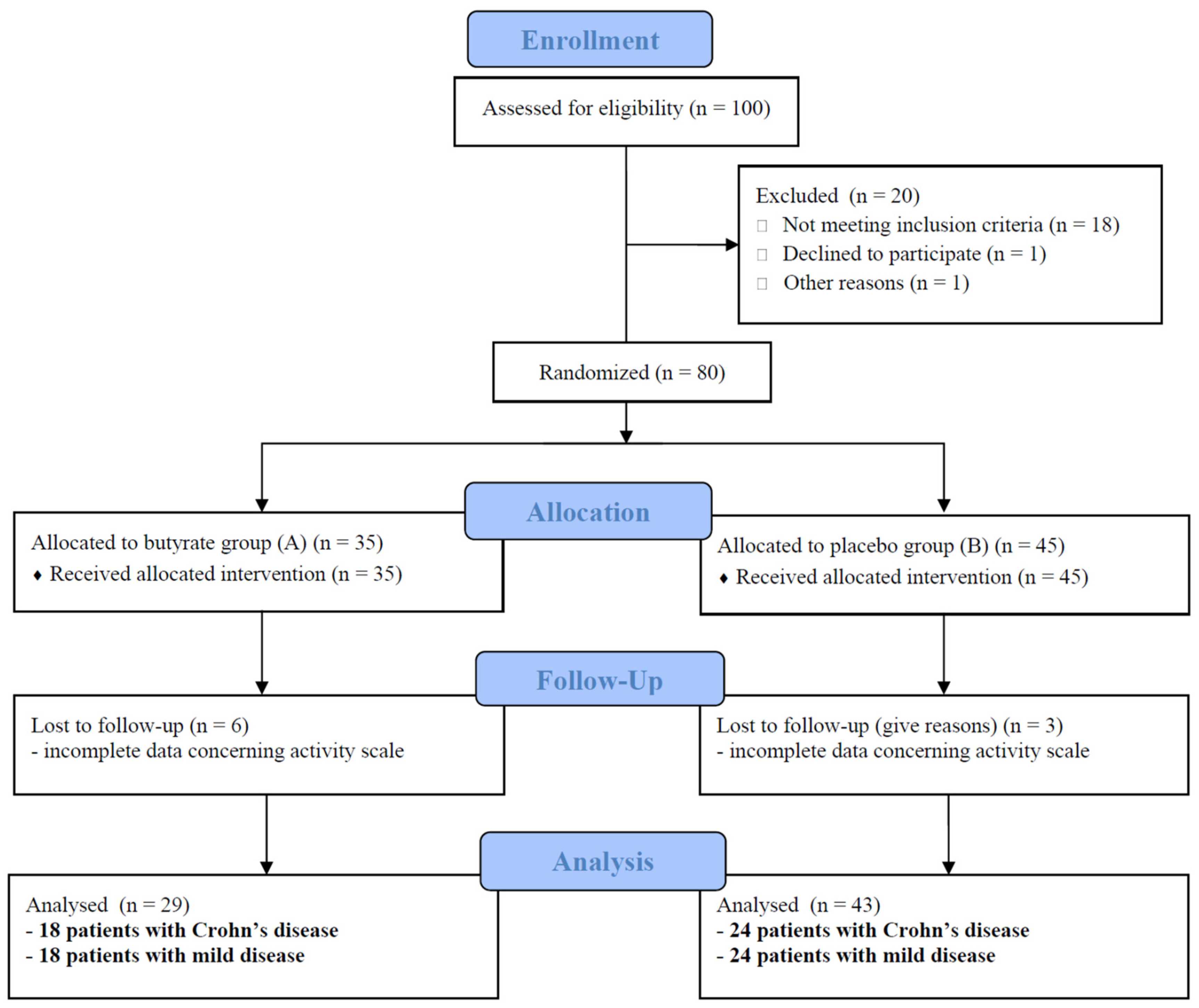
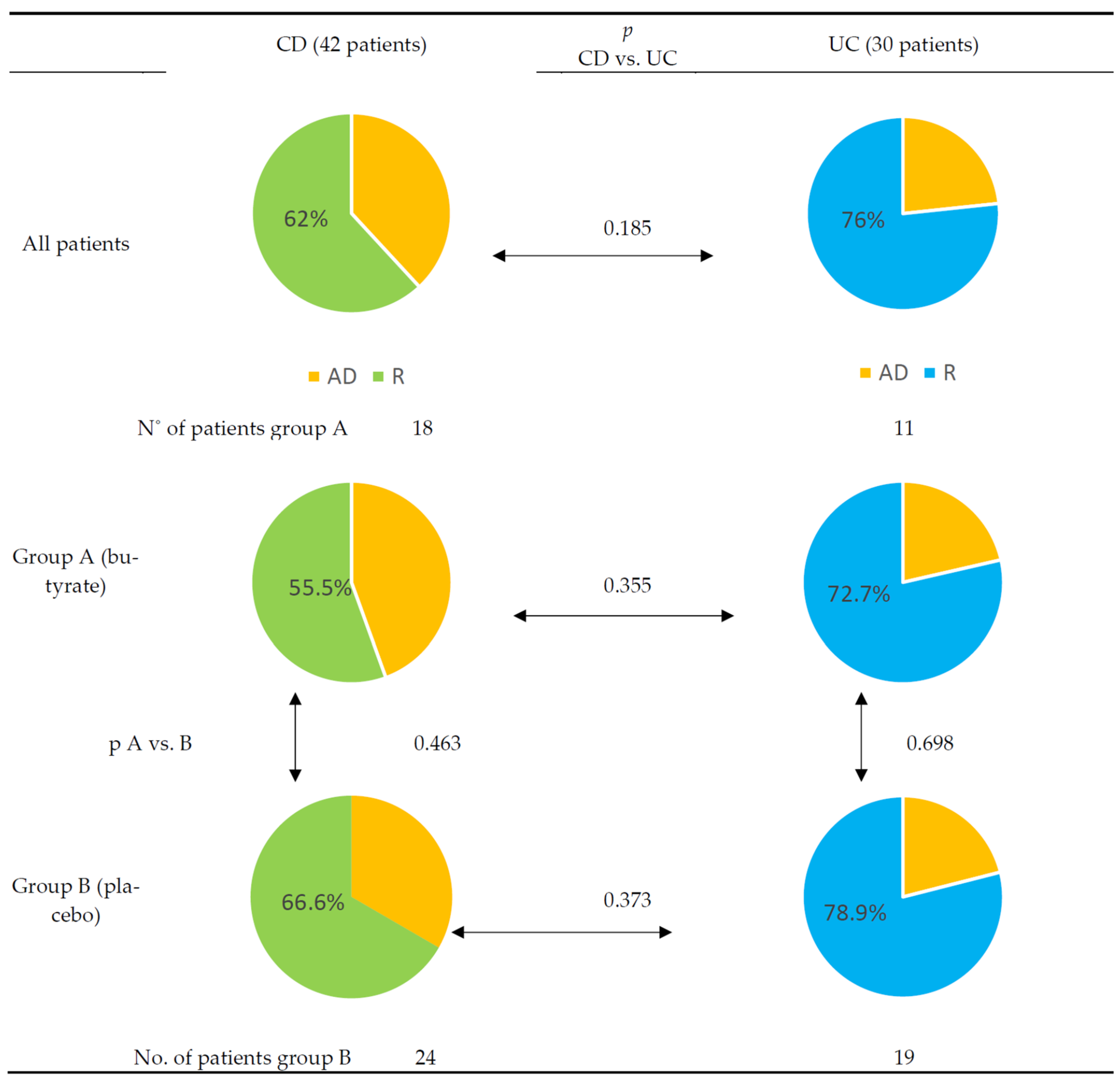
| Number of Patients | Study Group 72 | Group A (Butyrate) 29 | Group B (Placebo) 43 | p | |
|---|---|---|---|---|---|
| Crohn’s disease (No. of patients) | 42 (58.3%) | 18 (62.1%) | 24 (55.8%) | 0.597 | |
| Mean age (years) | 13.3 (6–18) | 12.9 (6–17) | 13.5 (7–18) | 0.6 | |
| Gender (male) | 43 (59.7%) | 14 (48.3%) | 29 (67.1%) | 0.104 | |
| Median weight (kg) | 46 (SD 14.8) | 45 (SD 15.3) | 46.5 (SD 14.2) | 0.329 | |
| Median height (cm) | 158 (SD 19.4) | 158 (SD 20.9) | 159 (SD 17.9) | 0.620 | |
| Baseline activity index score (points) | Mean | 34.5 (12.5–85) | 34.7 (12.5–75) | 34.3 (12.5–85) | 0.554 |
| Median | 31.5 | 30 | 32.5 | - | |
| Calprotectin level (points) | Mean | 1111.1 (50–1800) | 1090 (50–1800) | 1122.2 (50–1800) | 0.599 |
| Median | 1100 | 900 | 1300 | - | |
| Amino-salicylates | 70 (97.2%) | 28 (96.5%) | 42 (97.7%) | 0.776 | |
| Steroids | 8 (11.0%) | 3 (10.3%) | 5 (11.6%) | 0.865 | |
| Thiopurines | 50 (69.5%) | 21 (72.4%) | 29 (67.4%) | 0.653 | |
| Anti-TNF | 10 (13.9%) | 4 (13.8%) | 6 (13.9%) | 0.985 | |
| Antibiotics | 18 (25.0%) | 8 (27.6%) | 10 (23.3%) | 0.677 | |
| Disesae extention (Paris criteria) *—colon involvement (No of patients) | |||||
| UC: E1/E2/E3/E4 | 5/7/3/15 | 3/3/1/8 | 2/4/2/7 | n.s. | |
| CD: L2/L3 | 5/34 | 3/14 | 2/20 | n.s. | |
| Number of Patients | Study Group 72 | Group A 29 | Group B 43 | p | |
|---|---|---|---|---|---|
| Mild disease (≤10 points) | All | 42 | 18 | 24 | - |
| Rem. | 25 (59.5%) | 9 (50%) | 16 (66.7%) | 0.276 | |
| Moderate/severe disease (>10 points) | All | 30 | 11 | 19 | - |
| Rem. | 24 (80%) | 9 (81.8%) | 15 (78.9%) | 0.85 | |
| Excluding patients on steroids | All | 64 | 26 | 38 | - |
| Rem. | 31 48.4%) | 15 (57.7%) | 24 (63.2%) | 0.38 | |
| Excluding patients on antibiotics | All | 54 | 21 | 33 | - |
| Rem. | 40 (70.1%) | 14 (66.7%) | 26 (78.8%) | 0.322 | |
| Excluding patients on steroids, antibiotics and anti-TNF | All | 42 | 15 | 27 | - |
| Rem. | 29 (69.0%) | 9 (60.0%) | 20 (74.1%) | 0.344 | |
| Patients between 6 and 12 years old | All | 27 | 14 | 13 | - |
| Rem. | 19 (70.4%) | 9 (64.3%) | 10 (76.9%) | 0.302 | |
| Patients between 13 and 18 years old | All | 45 | 15 | 30 | - |
| Rem. | 30 (66.7%) | 9 (60.0%0 | 21 (70.0%) | 0.502 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrzak, A.; Banasiuk, M.; Szczepanik, M.; Borys-Iwanicka, A.; Pytrus, T.; Walkowiak, J.; Banaszkiewicz, A. Sodium Butyrate Effectiveness in Children and Adolescents with Newly Diagnosed Inflammatory Bowel Diseases—Randomized Placebo-Controlled Multicenter Trial. Nutrients 2022, 14, 3283. https://doi.org/10.3390/nu14163283
Pietrzak A, Banasiuk M, Szczepanik M, Borys-Iwanicka A, Pytrus T, Walkowiak J, Banaszkiewicz A. Sodium Butyrate Effectiveness in Children and Adolescents with Newly Diagnosed Inflammatory Bowel Diseases—Randomized Placebo-Controlled Multicenter Trial. Nutrients. 2022; 14(16):3283. https://doi.org/10.3390/nu14163283
Chicago/Turabian StylePietrzak, Anna, Marcin Banasiuk, Mariusz Szczepanik, Agnieszka Borys-Iwanicka, Tomasz Pytrus, Jarosław Walkowiak, and Aleksandra Banaszkiewicz. 2022. "Sodium Butyrate Effectiveness in Children and Adolescents with Newly Diagnosed Inflammatory Bowel Diseases—Randomized Placebo-Controlled Multicenter Trial" Nutrients 14, no. 16: 3283. https://doi.org/10.3390/nu14163283
APA StylePietrzak, A., Banasiuk, M., Szczepanik, M., Borys-Iwanicka, A., Pytrus, T., Walkowiak, J., & Banaszkiewicz, A. (2022). Sodium Butyrate Effectiveness in Children and Adolescents with Newly Diagnosed Inflammatory Bowel Diseases—Randomized Placebo-Controlled Multicenter Trial. Nutrients, 14(16), 3283. https://doi.org/10.3390/nu14163283






