Herbal Medicine for Behçet’s Disease: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Study Registration and Protocol Information
2.2. Data Sources
2.3. Study Selection
2.3.1. Types of Studies
2.3.2. Types of Participants
2.3.3. Types of Interventions and Comparison
2.3.4. Types of Outcome Measurements
Primary Outcome
- -
- The total RR: (recovery + marked improvement + improvement)/total number of cases * 100%;
- -
- Recovery rate: clinical cure/total number of cases * 100%;
- -
- Recurrence rate.
Secondary Outcome
- -
- Changes in CRP level and the ESR in laboratory studies;
- -
- Symptom score (oral ulcer, genital ulcer, eye inflammation, skin lesions, arthralgia);
- -
- Adverse events (AEs).
2.4. Data Extraction and Risk of Bias Assessment
2.4.1. Data Extraction
2.4.2. Risk of Bias
2.4.3. Data Analysis
3. Results
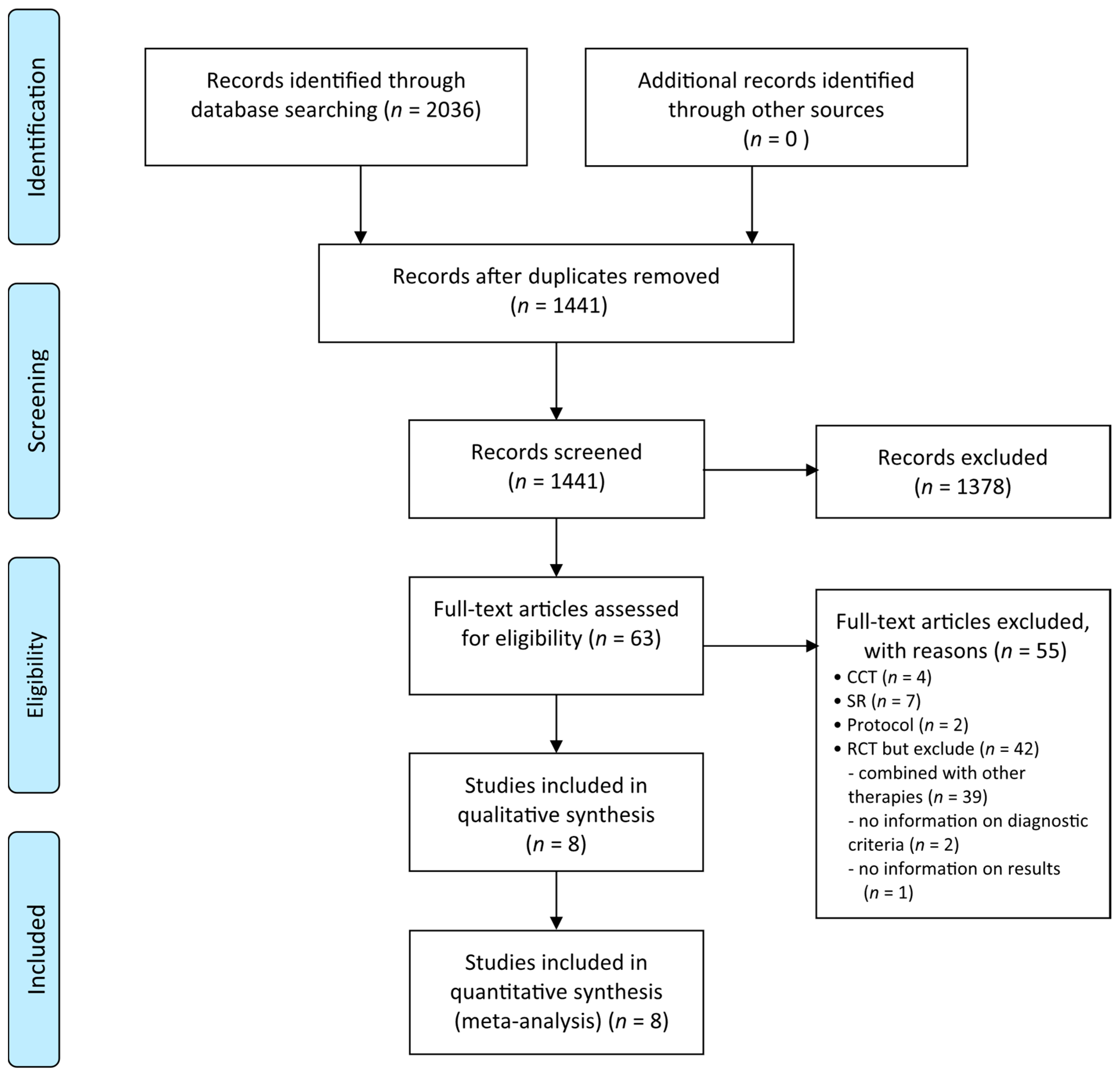
3.1. Description of the Included Trials
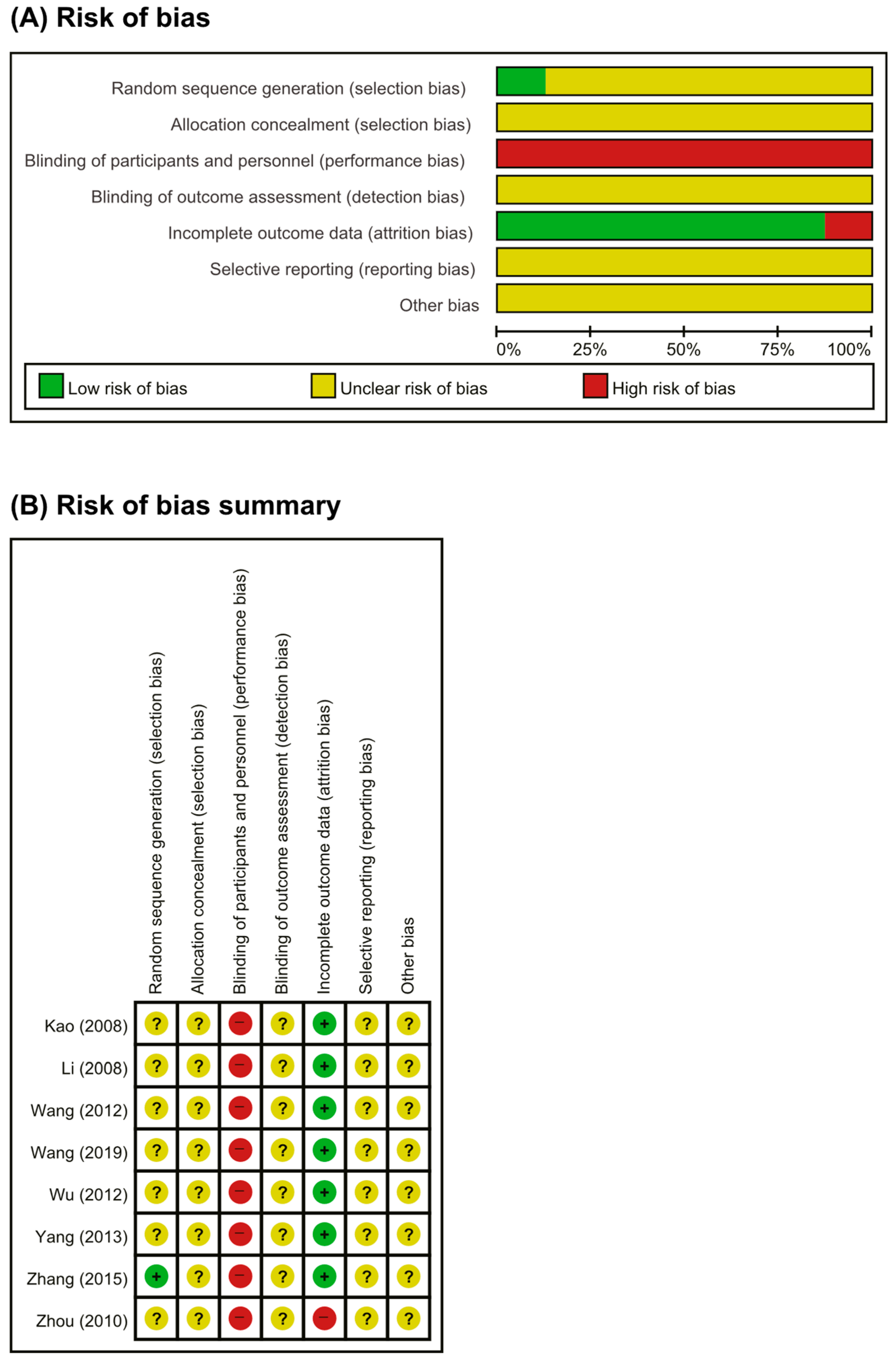
3.2. Risk of Bias
3.3. Certainty of Evidence
3.4. Outcome Measurements
3.4.1. Primary Outcomes
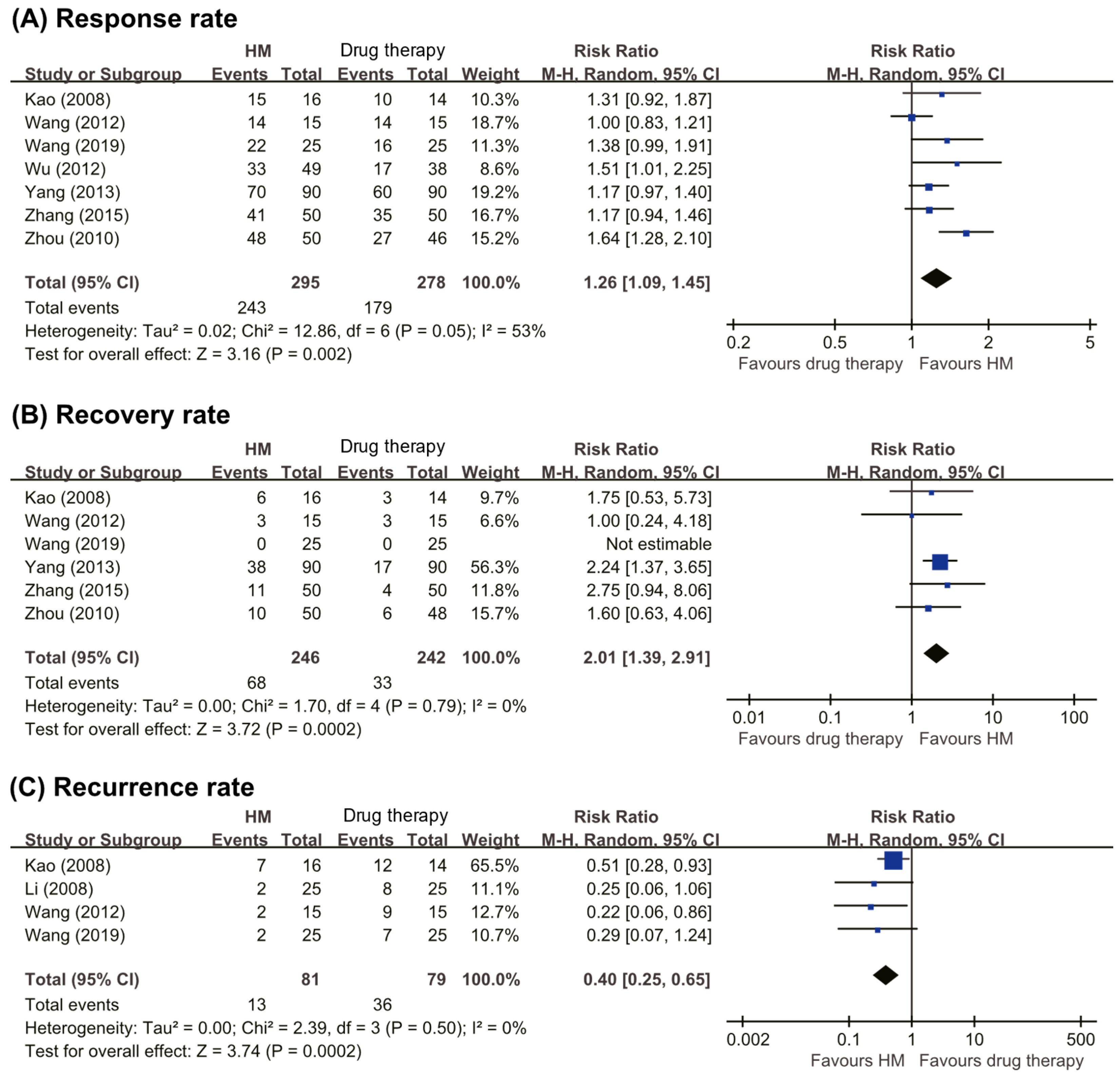
Total Response Rate
Recovery Rate
Recurrence Rate
3.4.2. Secondary Outcomes
ESR and CRP
Symptom Score
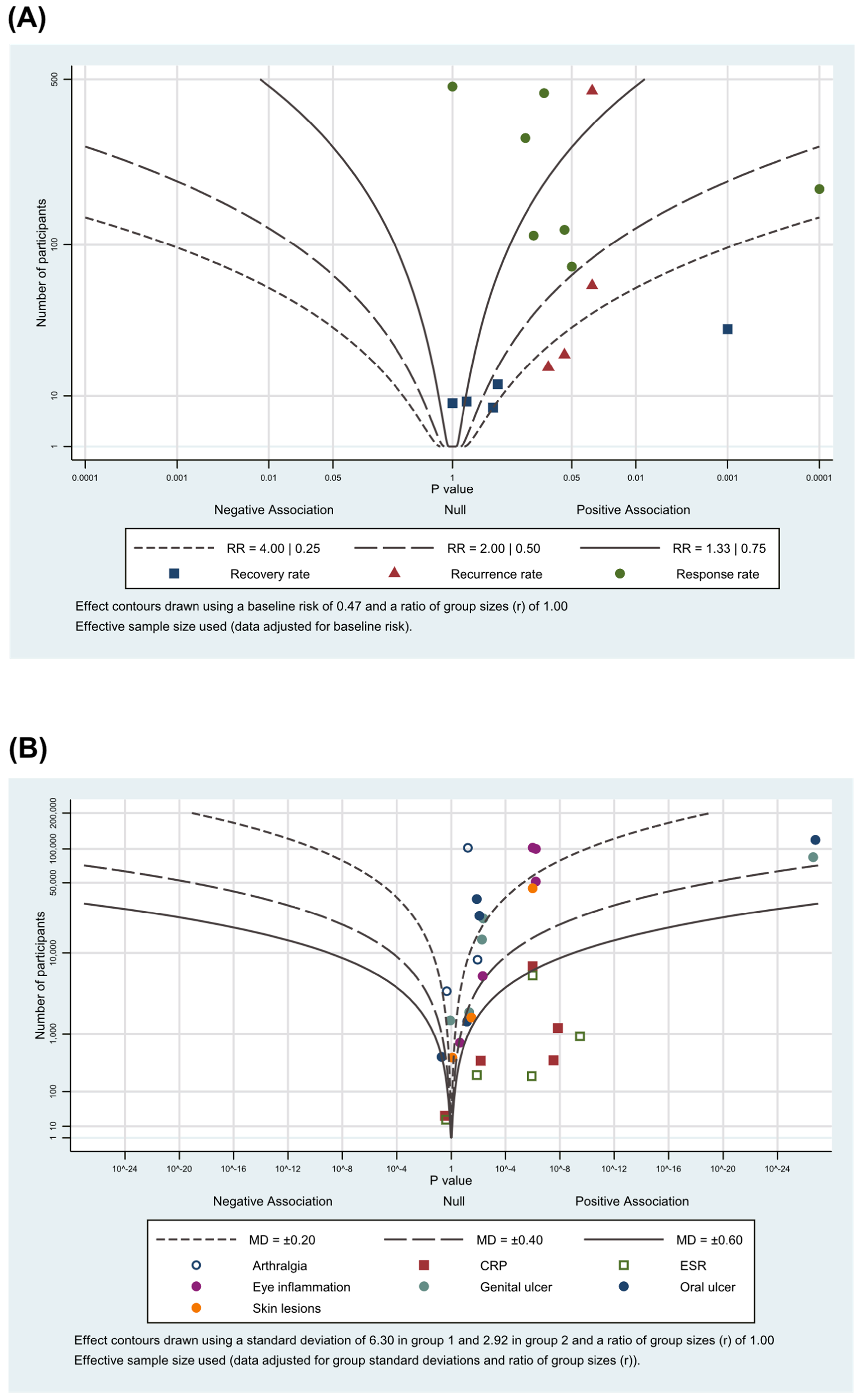
3.5. Albatross Plot
4. Discussion
4.1. Summary of the Main Results
4.2. Overall, Completeness and Applicability of the Evidence
4.3. Quality of the Evidence
4.4. Potential Biases in the Review Process
4.5. Agreements and Disagreements with Other Studies or Review
4.6. Potential Mechanism of Action
4.7. Implications for Nutrients
4.8. Implications for Practice
4.9. Implications for Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jagdish, R.N.; Robert, J.M. Behcet’s disease. Clin. Med. 2017, 17, 71–77. [Google Scholar]
- Yazici, H.; Seyahi, E.; Hatemi, G.; Yazici, Y. Behçet syndrome: A contemporary view. Nat. Rev. Rheumatol. 2018, 14, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Kural, S.E.; Fresko, I.; Seyahi, N.; Ozyazgan, Y.; Mat, C.; Hamuryudan, V.; Yurdakul, S.; Yazici, H. The long-term mortality and morbidity of Behçet syndrome: A 2-decade outcome survey of 387 patients followed at a dedicated center. Medicine 2003, 82, 60–76. [Google Scholar] [CrossRef]
- Mat, C.; Yurdakul, S.; Uysal, S.; Gogus, F.; Ozyazgan, Y.; Uysal, O.; Fresko, I.; Yazici, H. A double-blind trial of depot corticosteroids in behcet’s syndrome. Rheumatology 2005, 45, 348–352. [Google Scholar] [CrossRef] [PubMed]
- Nava, F.; Ghilotti, F.; Maggi, L.; Hatemi, G.; Del Bianco, A.; Merlo, C.; Filippini, G.; Tramacere, I. Biologics, colchicine, corticosteroids, immunosuppressants and interferon-alpha for Neuro-Behçet’s Syndrome. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef]
- Zhang, J.; Onakpoya, I.J.; Posadzki, P.; Eddouks, M. The safety of herbal medicine: From prejudice to evidence. Evid. Based Complementary Altern. Med. 2015, 2015, 316706. [Google Scholar] [CrossRef]
- Wei, L.G.; Jin, L.; Wu, J.L.; Cao, C.H. Progress in traditional chinese medicine treatment of behcet’s disease. Hunan J. TCM 2018, 34, 170–171. [Google Scholar]
- Kim, M.S.; Kim, S.O.; Lim, W.K.; Lee, J.M.; Hwang, C.Y.; Cho, K.H.; Kim, H.M. Novel effects of on-chung-eum, the traditional plant medicine, on cytokine production in human mononuclear cells from behçet’s. Immunopharmacol. Immunotoxicol. 2003, 25, 65–72. [Google Scholar] [CrossRef]
- Borchers, A.T.; Sakai, S.; Henderson, G.L.; Harkey, M.R.; Keen, C.L.; Stern, J.S.; Terasawa, K.; Gershwin, M.E. Shosaiko-to and other Kampo (Japanese herbal) medicines: A review of their immunomodulatory activities. J. Ethnopharmacol. 2000, 73, 1–13. [Google Scholar] [CrossRef]
- Kim, M.S.; Chung, H.S.; Lee, J.G.; Lim, W.K.; Hwang, C.Y.; Lee, E.J.; Cho, K.H.; Wi, D.H.; Kim, H.M. Inhibition of cytokine production by the traditional oriental medicine, ‘Gamcho-Sasim-Tang’ in mitogen-stimulated peripheral blood mononuclear cells from Adamantiades-Behçet’s patients. J. Ethnopharmacol. 2002, 83, 123–128. [Google Scholar] [CrossRef]
- Lee, S.G.; Ahn, K.S. Experimental studies on the kinds of sasim-tang in behcet’s disease symptoms in ICR mice. Korean J. Orient. Physiol. Pathol. 2004, 18, 1061–1070. [Google Scholar]
- Zhang, Z.R.; Xu, J.L.; He, D.Q.; Tian, J.H. Effectiveness of Chinese medicine in the treatment of behcet’s disease: A meta- analysis. J. Basic Chin. Med. 2015, 21, 872–874. [Google Scholar]
- Liu, Y.J.; Du, B.; Huang, S.P. Meta-analysis of clinical efficancy of chinese medicine in the treatment of behcet’s disease. Rheum. Arthritis 2017, 6, 42–45, 62. [Google Scholar]
- Zhang, Z.R.; He, D.Q.; Du, H.L.; Tian, J.W. Traditional medicine and integrative medicine for behcet’s disease: A meta-analysis and systematic review. Sandong Med. 2014, 54, 91–94. [Google Scholar]
- Jun, J.H.; Choi, T.Y.; Zhang, J.; Ko, M.M.; Lee, M.S. Herbal medicine for Behcet’s disease: A protocol for a systematic review and meta-analysis. Medicine 2018, 97, e0165. [Google Scholar] [CrossRef]
- International Study Group for Behcet’s Disease. Criteria for diagnosis of Behcet’s disease. Lancet 1990, 335, 1078–1080. [Google Scholar]
- International Team for the Revision of the International Criteria for Behcet’s Disease. The International Criteria for Behcet’s Disease (ICBD): A collaborative study of 27 countries on the sensitivity and specificity of the new criteria. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 338–347. [Google Scholar] [CrossRef]
- Deeks, J.J.; Higgins, J.P.T.; Altman, D.G. Chapter 10: Analysing data and undertaking meta-analyses. In Cochrane handbook for Systematic Reviews of Interventions, 2nd ed.; Higgins, J.P.T., Thomas, J., Eds.; Wiley Blackwell: Oxford, UK, 2019; pp. 214–284. [Google Scholar]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; DeBeer, H.; et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef]
- Kao, X.L. Clinical observation on 16 cases of behcet’s disease treted by yiqi tuodu decoction. J. TCM 2008, 49, 329. [Google Scholar]
- Li, X.T.; Kong, D.J. Clinical obervation of behcet’s disease treated by traditional chinese medicine. J. Qiqihar Med. Coll. 2008, 29, 1671–1673. [Google Scholar]
- Zhou, Y. Clinical Study of Gan Chi Soup Medicine on Behcet’s Disease. Master’s Thesis, Hebei University, Hebei, China, 2010. [Google Scholar]
- Wang, L.L. Clinical Reserch on Behcet’s Disease TCM Treatment Based on Combination of Disease and Syndrome. Master’s Thesis, Sandong University, Sandong, China, 2012. [Google Scholar]
- Wang, C.T. Clinical Obsevation on the Treatment of Stagnated Qi Transforming into Fire Behcet’s Disease with Jiawei Xiaoyao San. Master’s Thesis, Heilongjiang University, Heilongjiang, China, 2019. [Google Scholar]
- Zhang, Z.R.; He, Z.Q.; Liu, B.Y.; Chen, Y.F. 50 cases of behcet’s syndrome treated with gancao xiexin decoction and sanhuang. TCM Res. 2015, 28, 23–25. [Google Scholar]
- Yang, M. Clinical observation on 90 cases of behcet’s disease treated with bushen huoxue yuyang decoction. J. Sichan TCM 2013, 31, 92–93. [Google Scholar]
- Wu, S.Q.; Gao, F.Y.; Yu, H.M.; Zhang, Y.P.; Hna, Z.Z. Observation on the therapeutic effect of treating behcet’s disease. J. Sichan TCM 2012, 30, 108–109. [Google Scholar]
- Vickers, A.; Goyal, N.; Harland, R.; Rees, R. Do certain countries produce only positive results? a systematic review of controlled trials. Control. Clin. Trials 1998, 19, 159–166. [Google Scholar] [CrossRef]
- Gholijani, N.; Ataollahi, M.R.; Samiei, A.; Aflaki, E.; Shenavandeh, S.; Kamali-Sarvestani, E. An elevated pro-inflammatory cytokines profile in Behcet’s disease: A multiplex analysis. Immunol. Lett. 2017, 186, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.J.; Zhao, L.; Taylor, E.W.; Spelman, K. The influence of traditional herbal formulas on cytokine activity. Toxicology 2010, 278, 140–159. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Z.; Zhang, S.N. Herbal compounds for rheumatoid arthritis: Literatures review and cheminformatics prediction. Phytother. Res. 2020, 34, 51–66. [Google Scholar] [CrossRef]
- Huang, C.-F.; Lin, S.-S.; Liao, P.-H.; Young, S.-C.; Yang, C.-C. The Immunopharmaceutical Effects and Mechanisms of Herb Medicine. Cell. Mol. Immunol. 2008, 5, 23–31. [Google Scholar] [CrossRef]
- Zhang, H.P.; Zhang, D.-D.; Ke, Y.; Bian, K. The vasodilatory effects of anti-inflammatory herb medications: A comparison study of four botanical extracts. Evid. Based Complementary Altern. Med. 2017, 2017, 1021284. [Google Scholar] [CrossRef]
- El-Sayed, S.M.; Youssef, A.M. Potential application of herbs and spices and their effects in functional dairy products. Heliyon 2019, 5, e01989. [Google Scholar] [CrossRef]
- Heinrich, M. Food–Herbal Medicine Interface. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 94–98. [Google Scholar] [CrossRef]
- Kumar, V.; Marković, T.; Emerald, M.; Dey, A. Herbs: Composition and Dietary Importance. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 332–337. [Google Scholar] [CrossRef]
- Li, C.; Wu, F.; Yuan, W.; Ding, Q.; Wang, M.; Zhang, Q.; Zhang, J.; Xing, J.; Wang, S. Systematic Review of Herbal Tea (a Traditional Chinese Treatment Method) in the Therapy of Chronic Simple Pharyngitis and Preliminary Exploration about Its Medication Rules. Evid -Based Complementary Altern. Med. 2019, 2019, 9458676. [Google Scholar] [CrossRef] [PubMed]
- Tapsell, L.C.; Hemphill, I.; Cobiac, L.; Patch, C.S.; Sullivan, D.R.; Fenech, M.; Roodenrys, S.; Keogh, J.B.; Clifton, P.M.; Williams, P.G.; et al. Health benefits of herbs and spices: The past, the present, the future. Med. J. Aust. 2006, 185, 4–24. [Google Scholar] [CrossRef] [PubMed]
- Ruffell, M.J. Nutrition and health claims for food: Regulatory controls, consumer perception, and nutrition labeling. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 93–97. [Google Scholar] [CrossRef]
- Santini, A.; Cammarata, S.M.; Capone, G.; Ianaro, A.; Tenore, G.C.; Pani, L.; Novellino, E. Nutraceuticals: Opening the debate for a regulatory framework. Br. J. Clin. Pharmacol. 2018, 84, 659–672. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Novellino, E. Nutraceuticals—Shedding light on the grey area between pharmaceuticals and food. Expert Rev. Clin. Pharmacol. 2018, 11, 545–547. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. J. Clin. Epidemiol. 2010, 63, 834–840. [Google Scholar] [CrossRef] [PubMed]

| First Author (Year) [Ref] | Sample Size (Randomized/Analyzed) Mean Age (Year) Disease Duration (Year) Diagnosis | Intervention Group (Regimens) | Control Group (Regimens) | Main Outcome | Results | Adverse Effect |
|---|---|---|---|---|---|---|
| Kao (2008) [20] | 30/30 A: 30.2; B: 29.8 A: 7.6; B: 8.1 1989 ISG; 1994 Standards TCM diagnosis criteria | (A) HM (Yiqi Tuodu decoction, 2 times daily for 2 months, n = 16) | (B) Drug therapy (prednisone 10 mg, 2 times daily for 2 months, n = 14) | (1) Response rate (2) Recover rate (3) ESR (4) CRP (5) Symptom score (6) Recurrence rate (n.r.) | (1) RR 1.31 [0.92, 1.87], NS (2) RR 1.75 [0.53, 5.73], NS (3) MD −3.99 [−7.13, −0.85], p = 0.01 (4) MD −3.60 [−6.20, −1.00], p = 0.007 (5) Oral ulcer: MD −1.14 [−2.37, 0.09], NS; genital ulcer: MD −1.06 [−2.10, 0.02], p = 0.05; eye inflammation: MD −0.93 [−1.57, −0.29], p = 0.005; skin lesions: MD −1.19 [−2.28, 0.10], p = 0.03; arthralgia: MD −0.58 [−1.03, −0.13], p = 0.01 (6) RR 0.51 [0.28, 0.93], p = 0.03 | n.r. |
| Li (2008) [21] | 50/50 n.r. 1989 ISG | (A) HM (self-experience prescription, 2 times daily for 1 month, n = 25) | (B) Drug therapy (prednisone 10−30 mg, 1 time daily for 1 month, n = 25) | (1) Symptom score (2) Recurrence rate | (1) Oral ulcer: RR 1.00 [0.85, 1.17], NS; genital ulcer: RR 0.86 [0.54, 1.35], NS; eye inflammation: RR 0.88 [0.58, 1.33], NS; skin lesions: RR 0.83 [0.48, 1.44], NS; arthralgia: 0.96 [0.83, 1.10], NS (2) after 3 months of follow-up: RR 0.25 [0.06, 1.06], NS | Diarrhea (A: 4) |
| Zhou (2010) [22] | 110/96 n.r. 1989 ISG | (A) HM (Ganzhi decoction, 2 times daily for 3 months, n = 50) | (B) Drug therapy (prednisone 10 mg, 1 time daily for 3 months, n = 46) | (1) Response rate (2) Recover rate (3) ESR (4) CRP (5) Symptom score | (1) RR 1.64 [1.28, 2.10], p < 0.0001 (2) RR 1.60 [0.63, 4.06], NS (3) MD −4.47 [−5.87, −3.07], p < 0.00001 (4) MD −3.68 [−4.95, −2.41], p < 0.00001 (5) Oral ulcer: MD −0.34 [−0.59, −0.09], p = 0.008; genital ulcer: MD −0.47 [−0.80, 0.14], p = 0.005; eye inflammation: MD −0.47 [−0.65, −0.29], p < 0.00001 | n.r. |
| Wang (2012) [23] | 30/30 A: 34.2; B: 40.0 A: 6.7; B: 8.5 1989 ISG; 1994 Standards TCM diagnosis criteria | (A) HM (PI prescription, 2 times daily for 2 months, n = 15) | (B) Drug therapy (thalidomide 50 mg, 1 time daily for 2 months, n = 15) | (1) Response rate (2) Recover rate (3) ESR (4) CRP (5) Symptom score (6) Recurrence rate | (1) RR 1.00 [0.83, 1.12], NS (2) RR 1.00 [0.24, 4.18], NS (3) MD 5.13 [−7.25, −17.51], NS (4) MD 5.43 [−6.05, 16.91], NS (5) Oral ulcer: MD 1.60 [−0.84, 4.04], NS; genital ulcer: MD 0.13 [−1.18, 1.44], NS; eye inflammation: MD −0.93 [−2.45, 0.59], NS; skin lesions: MD −0.27 [−2.70, 2.16], NS; arthralgia: MD 0.27 [−0.46, 1.00], NS (6) after 3 of months follow-up: RR 0.29 [0.07, 1.24], p = 0.03 | Diarrhea (A: 2); insomnia (B: 11); dizziness (B: 11); constipation (B: 11) |
| Wang (2019) [24] | 50/50 A: 39.1; B: 39.6 A: 7.5; B: 7.4 1989 ISG | (A) HM (modified Xiaoyao san, 2 times daily for 2 months, n = 25) | (B) Drug therapy (thalidomide 50 mg, 2 times daily for 2 months, n = 25) | (1) Response rate (2) ESR (3) CRP (4) Symptom score (5) Recurrence rate | (1) RR 1.38 [0.99, 1.91], NS (2) MD −11.40 [−11.99, −10.81], p < 0.0001 (3) MD −15.77 [−16.29, −15.25], p < 0.0001 (4) Oral ulcer: MD −0.80 [−0.94, −0.66], p < 0.0001; genital ulcer: MD −0.88 [−1.04, −0.72], p < 0.0001; eye inflammation: MD −0.93 [−2.45, 0.59], NS (5) after 12 months of follow-up: RR 0.29 [0.07, 1.24], NS | n.r. |
| Zhang (2015) [25] | 100/100 A: 40.0; B: 36.2 A: 5.2; B: 3.7 2005 ICBD; 1994 Standards TCM diagnosis criteria | (A) HM (modified Gancao Xiexin decoction, 2 times daily for 3 months, n = 50) | (B) Drug therapy (prednisone 30 mg, 2 times daily; Thalidomide, 50 mg 1 time daily for 3 months, n = 50) | (1) Response rate (2) Recover rate (3) ESR (4) CRP (5) Symptom score | (1) RR 1.17 [0.94, 1.46], NS (2) RR 2.75 [0.94, 8.06], NS (3) MD −5.92 [−8.31, −3.53], p < 0.0001 (4) MD −5.71 [−7.73, −3.69], p < 0.0001 5) Oral ulcer: MD −0.34 [−0.59, −0.09], p = 0.01; genital ulcer: MD −0.43 [−0.72, −0.14], p = 0.004; eye inflammation: MD −0.38 [−0.53, −0.23], p < 0.00001; skin lesions: MD −2.24 [−2.42, −2.06], p < 0.0001; arthralgia: MD −0.12 [−0.24, 0.00], NS | n.r. |
| Yang (2013) [26] | 180/180 32.8 2.5 1989 ISG | (A) HM (Bushen Huoxue Yuyang decoction, 2 times daily for 3 weeks, n = 90) | (B) Drug therapy (loxoprofen sodium 60 mg, 3 times dialy; Thalidomide 50 mg, 1 time daily for 3 weeks, n = 90) | (1) Response rate (2) Recover rate | (1) RR 1.17 [0.97, 1.40], NS (2) RR 2.24 [1.37, 3.65], p = 0.001 | n.r. |
| Wu (2012) [27] | 87/87 n.r. 1989 ISG | (A) HM (PI prescription, 2 times daily for 3 months, n = 49) | (B) Drug therapy (interferon a−2b injection, 3 times daily for 3 months, n = 38) | Response rate | RR 1.51 [1.01, 2.25], p = 0.05 | n.r. detail (A: 1, B: 26) |
| First Author (Year) [Ref] | Prescription | Consists of Herbs |
|---|---|---|
| Kao (2008) [20] | Yiqi Tuodu decoction | Astragali Radix 45 g, Paeoniae Radix Alba 30 g, Isatidis Folium 15 g, Glycyrrhizae Radix et Rhizoma 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 15 g, Tripterygii Cortex 9 g, Angelicae Sinensis Radix 9 g, Angelicae Dauricae Radix 6 g |
| Li (2008) [21] | Modified Wenqin yin + Liuwei Dihuang wan | Atractylodis Rhizoma 30 g, Phragmitis Rhizoma 30 g, Sophorae Tonkinensis Radix et Rhizoma 10 g, Artemisiae Annuae Herba 30 g, Glycyrrhizae Radix et Rhizoma 20 g, Coptidis Rhizoma 10 g, Phellodendri Cortex 15 g, Scutellariae Radix 20 g, Ephedrae Herba 5 g, Paeoniae Radix Rubra 30 g, Coicis Semen 70 g, Alismatis Rhizoma 30 g, Dioscoreae Hypoglaucae Rhizoma 5 g |
| Zhou (2010) [22] | Ganzhi decoction | Scutellariae Radix 9 g, Coptidis Rhizoma 6 g, Pinelliae Rhizoma 15 g, Zingiberis Rhizoma 9 g, Vignae Semen 18 g, Angelicae Sinensis Radix 12 g, Atractylodis Macrocephalae Rhizoma 9 g, Atractylodis Rhizoma 12 g, Coicis Semen 15 g, Rehmanniae Radix 12 g, Gypsum Fibrosum 18 g, Cimicifugae Rhizoma 9 g, Glycyrrhizae Radix et Rhizoma 9 g |
| Wang (2012) [23] | (1) Modified Longdan Xiegan decoction (syndrome of toxic fire ablaze) (2) Modified Liangying Qingqi decoction (syndrome of fire-heat with ablaze, syndrome of toxic blazing of both qi and nutrient) (3) Modified Simiao Longan decoction (syndrome of dampness-heat accumulation) (4) Modified Zhibai Dihuang decoction (syndrome of dampness-heat and yin damage, syndrome deficiency fire with dampness) (5) Modified Xijiao Dihuang decoction (syndrome of toxic heat ablaze, syndrome of frenetic movement of blood due to heat) | (1) Glycyrrhizae Radix et Rhizoma 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 15 g, Astragali Radix 30 g, Isatidis Folium 15 g, Smilacis Glabrae Rhizoma 30 g, Forsythiae Fructus 12 g, Conyzae Herba, Gardeniae Fructus, Scutellariae Radix, Akebiae Caulis, Alismatis Rhizoma, Plantaginis Semen, Bupleuri Radix, Glycyrrhizae Radix et Rhizoma, Angelicae Sinensis Radix, Rehmanniae Radix (2) Glycyrrhizae Radix et Rhizoma 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 15 g, Astragali Radix 30 g, Isatidis Folium 15 g, Smilacis Glabrae Rhizoma 30 g, Forsythiae Fructus 12 g, Rhinocerotis Cornu, Dendrobii Herba, Gardeniae Fructus, Moutan Cortex, Rehmanniae Radix, Menthae Haplocalycis Herba, Coptidis Rhizoma, Paeoniae Radix Rubra, Scrophulariae Radix, Gypsum Fibrosum, Forsythiae Cortex, Lophatheri Herba, Phragmitis Rhizoma Hominis, Excrementum cum Aqua (3) Glycyrrhizae Radix et Rhizoma 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 15 g, Astragali Radix 30 g, Isatidis Folium 15 g, Smilacis Glabrae Rhizoma 30 g, Forsythiae Fructus 12 g, Lonicerae Flos, Scrophulariae Radix, Angelicae Sinensis Radix (4) Glycyrrhizae Radix et Rhizoma 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 15 g, Astragali Radix 30 g, Isatidis Folium 15 g, Smilacis Glabrae Rhizoma 30 g, Forsythiae Fructus 12 g, Dioscoreae Rhizoma, Moutan Cortex, Poria Sclerotium, Corni Fructus, Alismatis Rhizoma, Phellocendri Cortex, Rehmanniae Radix Praeparata, Anemarrhenae Rhizoma (5) Glycyrrhizae Radix et Rhizoma 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 15 g, Astragali Radix 30 g, Isatidis Folium 15 g, Smilacis Glabrae Rhizoma 30 g, Forsythiae Fructus 12 g, Rhinocerotis Cornu, Rehmanniae Radix, Paeoniae Radix Alba, Moutan Cortex |
| Wang (2019) [24] | Modified Xiaoyao san | Bupleuri Radix 15 g, Angelicae Sinensis Radix 15 g, Paeoniae Radix Alba 15 g, Atractylodis Macrocephalae Rhizoma 15 g, Atractylodis Rhizoma 15 g, Moutan Cortex 15 g, Crataegi Fructus 15 g, Cyperi Rhizoma 15 g, Aurantii Fructus 15 g, Magnoliae Officinalis Cortex 15 g, Zingiberis Rhizoma Recens 15 g, Menthae Haplocalycis Herba 10 g, Glycyrrhizae Radix et Rhizoma 10 g Severe case: [Eye inflammation: Sophorae Flos 15 g, Chrysanthemi Flos 15 g, Dendrobii, Herba 20 g], [Genital ulcer: Sophorae Radix 10 g, Plantaginis Semen 20 g, Phellodendri Cortex 10 g], [Skin lesions: Moutan Cortex 15 g, Violae Herba 10 g, Taraxaci Herba 20 g], [Arthralgia: Dioscoreae Hypoglaucae Rhizoma 20 g, Siegesbeckiae Herba 30 g] |
| Zhang (2015) [25] | Modified Gancao Xiexin decoction | Glycyrrhizae Radix et Rhizoma 10 g, Scutellariae Radix 10 g, Codonopsis Pilosulae Radix 30 g, Zingiberis Rhizoma 10 g, Coptidis Rhizoma 6 g, Pinelliae Rhizoma 10 g, Astragali Radix 30 g, Angelicae Sinensis Radix 20 g, Paeoniae Radix Rubra 30 g, Phaseoli Semen 30 g, Cimicifugae Rhizoma 10 g, Jujubae Fructus 10 g |
| Yang (2013) [26] | Bushen Huoxue Yuyang decoction | Rehmanniae Radix Preparata, Lilii Bulbus, Angelicae Gigantis Radix, Anemarrhenae Rhizoma, Phellodendri Cortex, Scrophulariae Radix, Liriopis Tuber, Moutan Cortex, Paeoniae Radix Rubra, Tripterygii Cortex, Coicis Semen, Citri Reticulatae Pericarpium, Glycyrrhizae Radix et Rhizoma |
| Wu (2012) [27] | (1) Xuanhua Jiudu Yin (syndrome of dampness-heat accumulation, syndrome of toxic heat in the collaterals) (2) Erdong Runluo decoction (syndrome of yin deficiency in the collateral) (3) Buyang Tongluo decoction (syndrome of yang deficiency, syndrome of cold congealing in the collateral) (4) Shiwei Rongluo Yin (syndrome of tonify qi and replenish blood) | (1) Scrophulariae Radix 20 g, Lonicerae Flos 30 g, Bubali Cornu 10 g, Forsythiae Fructus 10 g, Angelicae Sinensis Radix 10 g, Taraxaci Herba 15 g, Paeoniae Radix Rubra 10 g, Atractylodis Rhizoma 10 g, Scutellariae Radix 6 g, Pinelliae Rhizoma 10 g, Phellodendri Cortex 10 g, Glycyrrhizae Radix et Rhizoma 10 g (2) Asparagi Radix 15 g, Liriopis Tuber 15 g, Rehmanniae Radix 10 g, Adenophorae Radix 15 g, Lycii Fructus 10 g, Angelicae Sinensis Radix 10 g, Anemarrhenae Rhizoma 10 g, Phellodendri Cortex 6 g, Paeoniae Radix Alba 15 g, Scrophulariae Radix 10 g, Moutan Cortex 10 g, Trionycis Carapax 15 g, Testudinis Carapax et Plastrum 15 g, Glycyrrhizae Radix et Rhizoma Praeparata 10 g (3) Cinnamomi Cortex 9 g, Zingiberis Rhizoma Praeparata 6 g, Rehmanniae Radix Preparata 20 g, Angelicae Sinensis Radix 10 g, Cervi Cornus Colla 10 g, Cinnamomi Ramulus 10 g, Sinapis Semen 6 g, Astragali Radix 15 g, Hirudo 3 g, Eupolyphaga Steleophaga 10 g, Glycyrrhizae Radix et Rhizoma 10 g (4) Asini Gelatinum 10 g, Polygoni Multiflori Radix 15 g, Codonopsis Pilosulae Radix 15 g, Atractylodis Rhizoma 10 g, Atractylodis Macrocephalae Rhizoma 10 g, Rehmanniae Radix Preparata 15 g, Cnidii Rhizoma10 g, Angelicae Sinensis Radix 10 g, Paeoniae Radix Alba 10 g |
| Herbal Medicine for Behçet’s Diseases | |||||
|---|---|---|---|---|---|
| Patient or Population: Behçet’s Diseases Setting: Randomized Controlled Trials Intervention: Herbal Medicine Comparison: Drug Therapy | |||||
| Outcomes | No of Participants (Studies) | Certainty of the Evidence (GRADE) | Relative Effect (95% CI) | Anticipated Absolute Effects * | |
| Risk with Drug Therapy | Risk Difference with Herbal Medicine | ||||
| Response rate | 573 (7 RCTs) | ⨁◯◯◯ VERY LOW a,b,c | RR 1.26 (1.09 to 1.45) | 644 per 1000 | 167 more per 1000 (58 more to 290 more) |
| Recovery rate | 655 (6 RCTs) | ⨁⨁◯◯ LOW a,c | RR 2.01 (1.39 to 2.91) | 136 per 1000 | 138 fewer per 1000 (53 more to 260 more) |
| Recurrence rate | 160 (4 RCTs) | ⨁⨁◯◯ LOW a,c | RR 0.40 (0.25 to 0.65) | 160 per 1000 | 96 fewer per 1000 (387 fewer to 157 fewer) |
| ESR | 306 (5 RCTs) | ⨁◯◯◯ VERY LOW a,c,d | - | MD 5.56 lower (9.99 lower to 1.12 lower) | |
| CRP | 306 (5 RCTs) | ⨁◯◯◯ VERY LOW a,c,d | - | MD 5.44 lower (12.73 lower to 1.86 higher) | |
| Oral ulcers | 306 (5 RCTs) | ⨁◯◯◯ VERY LOW a,b,c | - | MD 0.51 lower (0.85 lower to 0.16 lower) | |
| Genital ulcers | 306 (5 RCTs) | ⨁◯◯◯ VERY LOW a,b,c | - | MD 0.61 lower (0.91 lower to 0.31 lower) | |
| Eye inflammation | 306 (5 RCTs) | ⨁◯◯◯ VERY LOW a,b,c | - | MD 0.63 lower (0.93 lower to 0.34 lower) | |
| Skin lesions | 160 (3 RCTs) | ⨁◯◯◯ VERY LOW a,b,c | - | MD 1.62 lower (2.65 lower to 0.59 lower) | |
| Arthralgia | 160 (3 RCTs) | ⨁◯◯◯ VERY LOW a,b,c | - | MD 0.19 lower (0.56 lower to 0.17 higher) | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jun, J.H.; Choi, T.Y.; Lee, H.W.; Ang, L.; Lee, M.S. Herbal Medicine for Behçet’s Disease: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 46. https://doi.org/10.3390/nu13010046
Jun JH, Choi TY, Lee HW, Ang L, Lee MS. Herbal Medicine for Behçet’s Disease: A Systematic Review and Meta-Analysis. Nutrients. 2021; 13(1):46. https://doi.org/10.3390/nu13010046
Chicago/Turabian StyleJun, Ji Hee, Tae Young Choi, Hye Won Lee, Lin Ang, and Myeong Soo Lee. 2021. "Herbal Medicine for Behçet’s Disease: A Systematic Review and Meta-Analysis" Nutrients 13, no. 1: 46. https://doi.org/10.3390/nu13010046
APA StyleJun, J. H., Choi, T. Y., Lee, H. W., Ang, L., & Lee, M. S. (2021). Herbal Medicine for Behçet’s Disease: A Systematic Review and Meta-Analysis. Nutrients, 13(1), 46. https://doi.org/10.3390/nu13010046







