Dietary Betaine Addition Promotes Hepatic Cholesterol Synthesis, Bile Acid Conversion, and Export in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Design
2.2. Sample Collection
2.3. Analysis of Lipid Metabolites in Serum
2.4. Hepatic Histology and Metabolites Analysis
2.5. Western Blot Analysis
2.6. Treatments of Cells
2.7. Statistical Analysis
3. Results
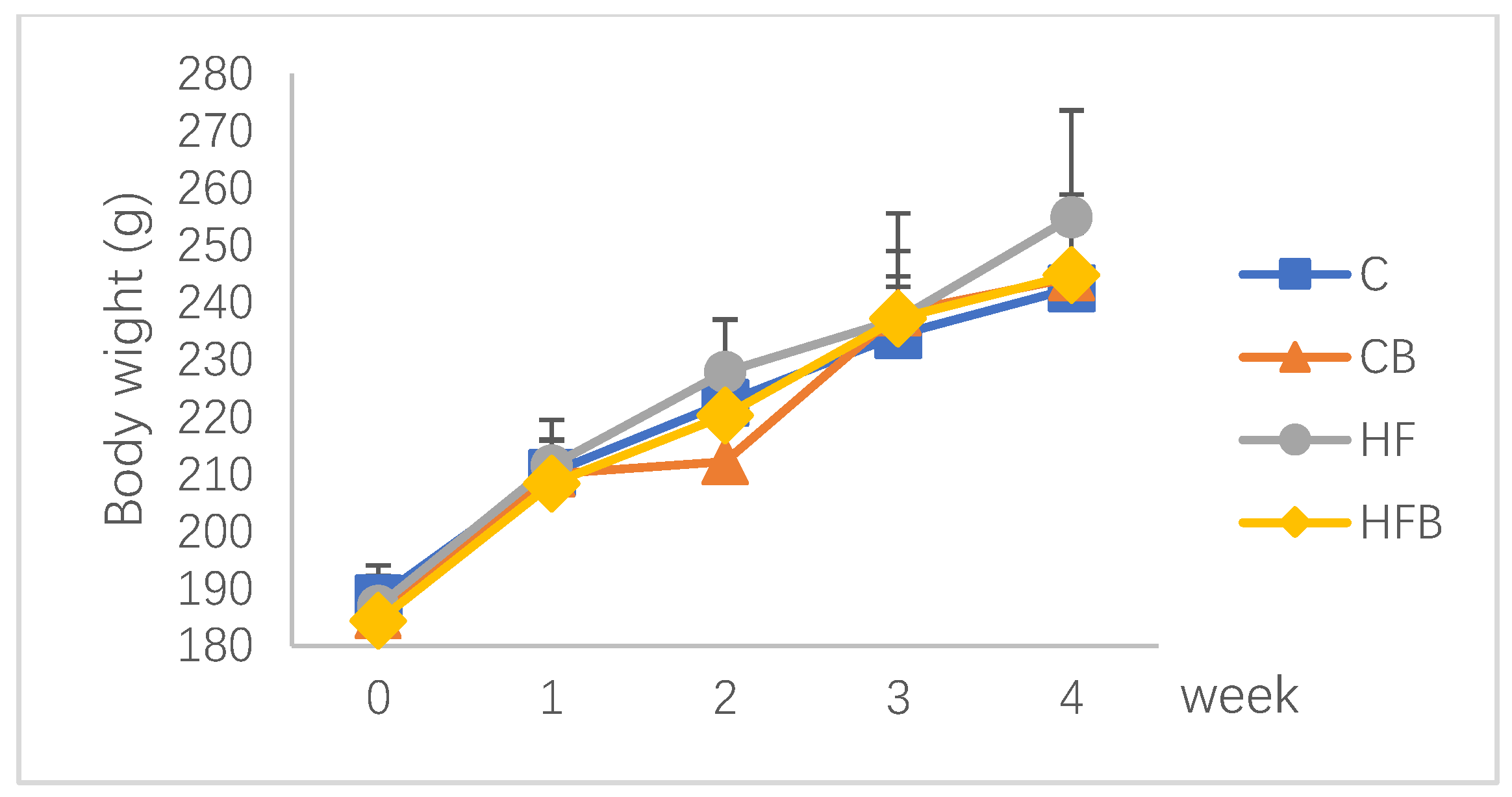
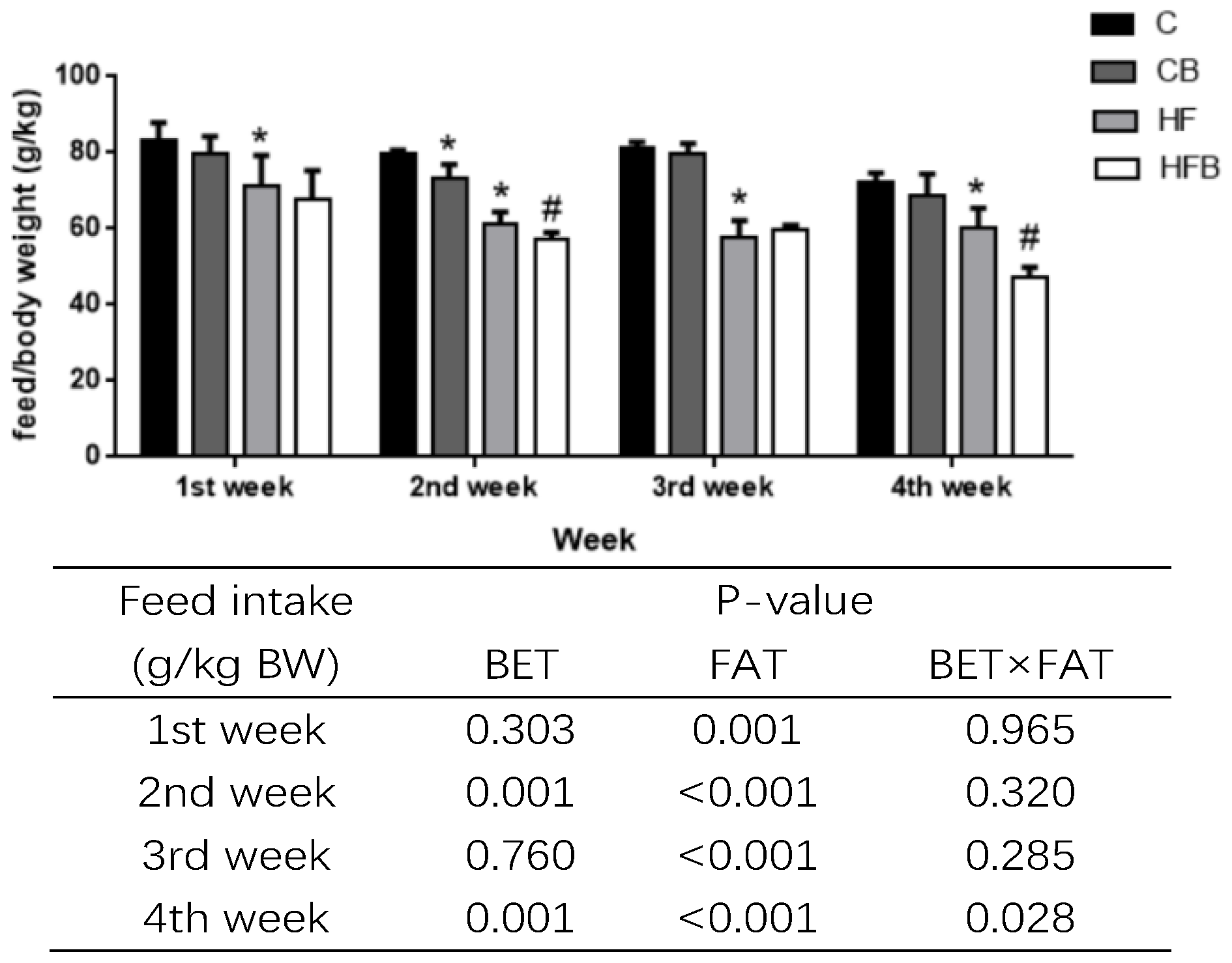
3.1. Growth Performance
3.2. Serum Lipid Metabolites
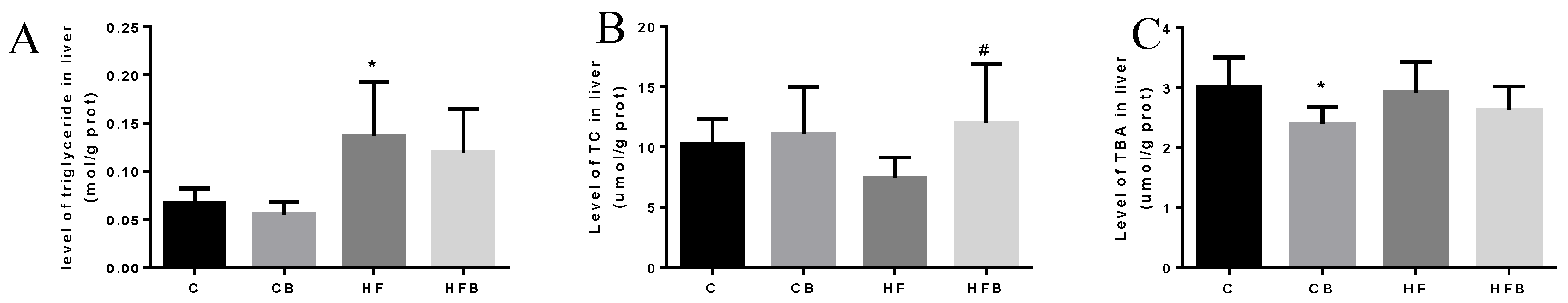
3.3. Hepatic Metabolite
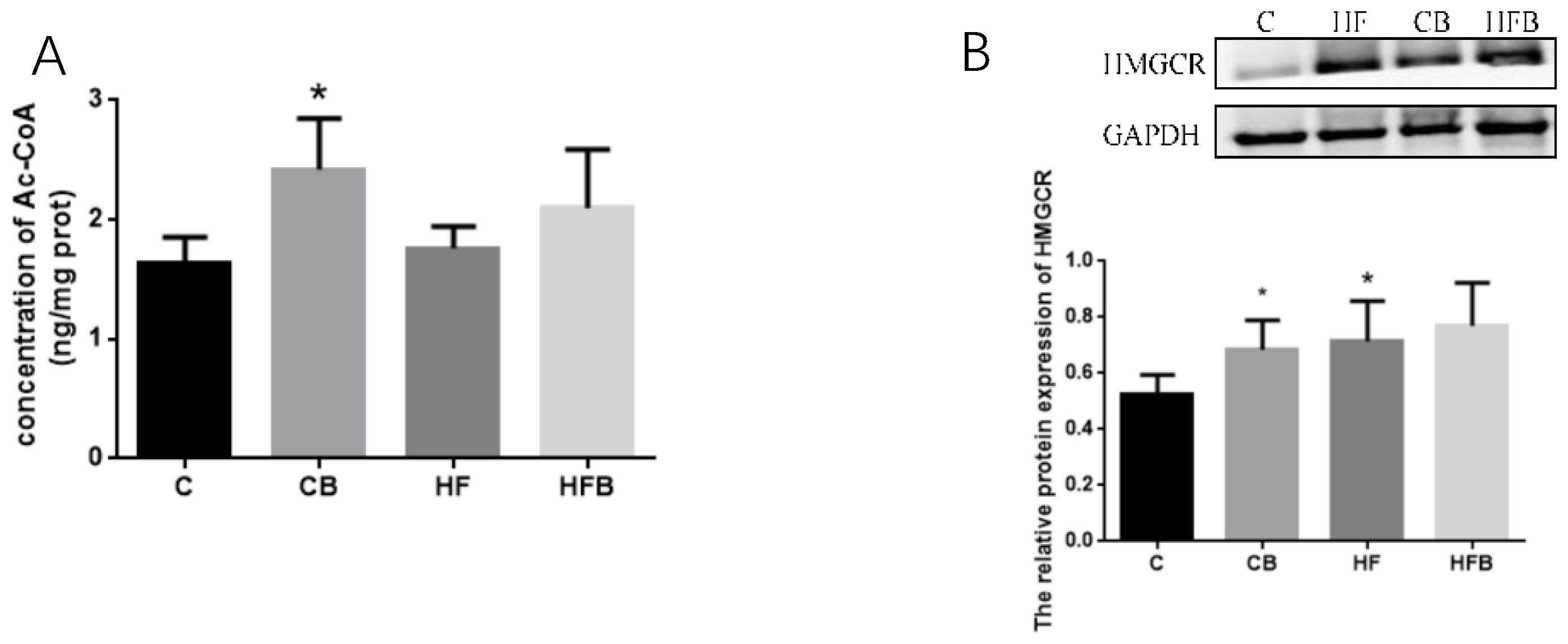
3.4. Key Factors Involved in Cholesterol Metabolism
3.4.1. Cholesterol Synthesis
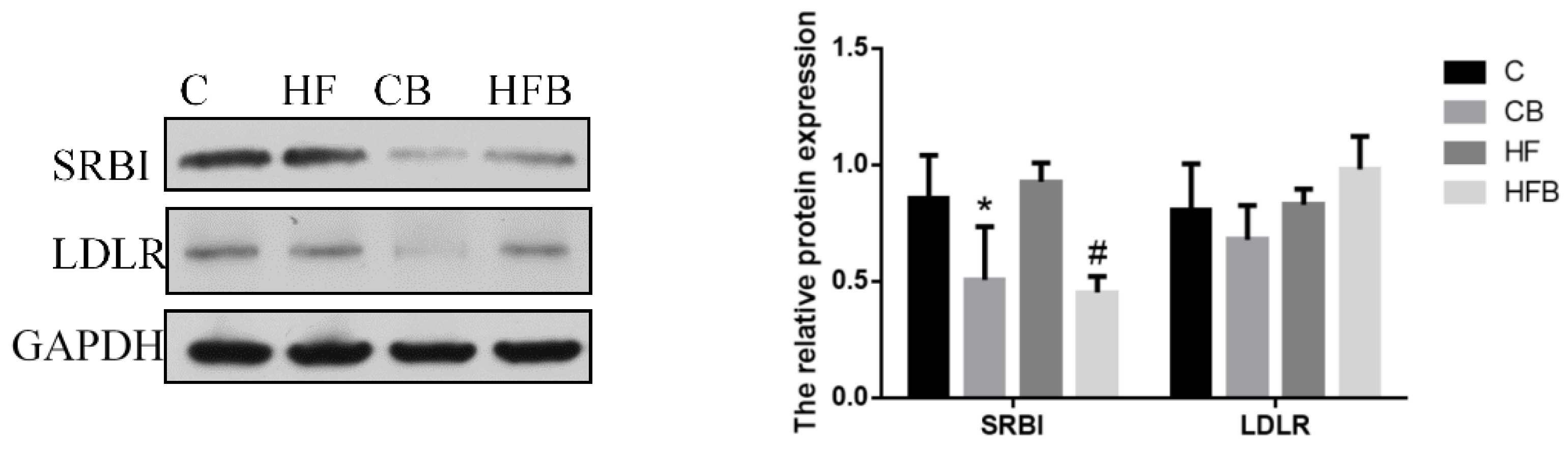
3.4.2. Cholesterol Transport
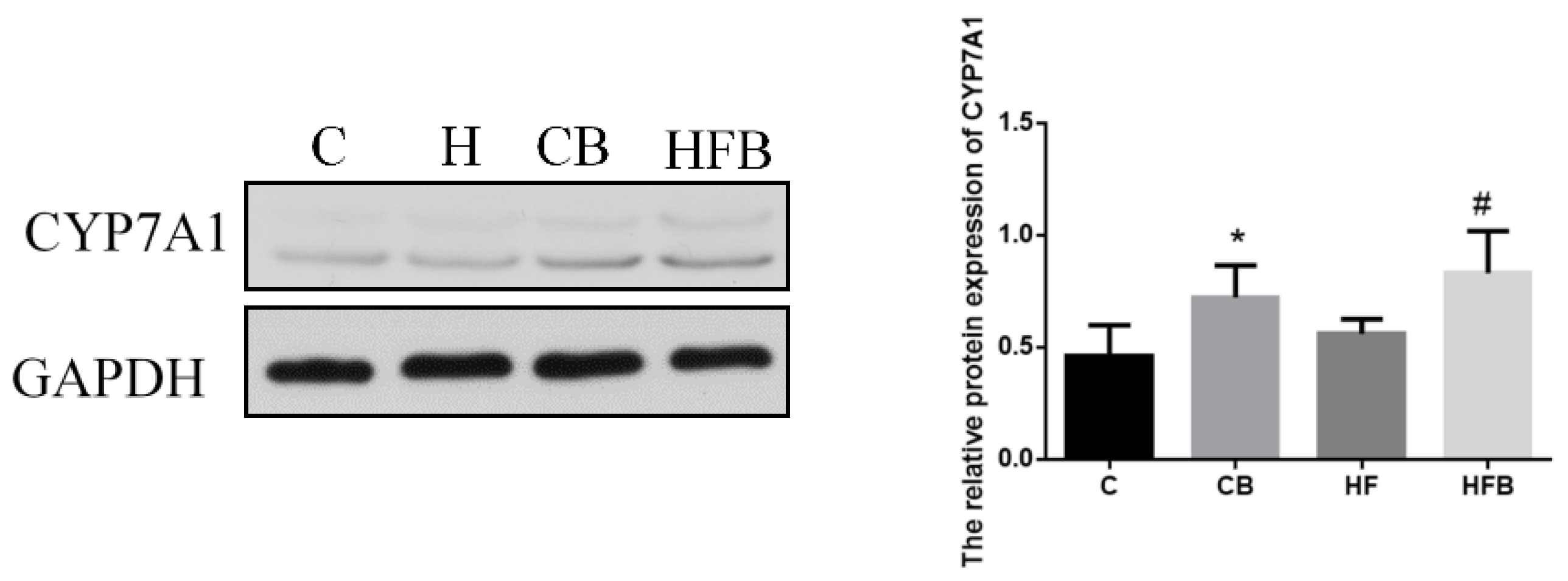
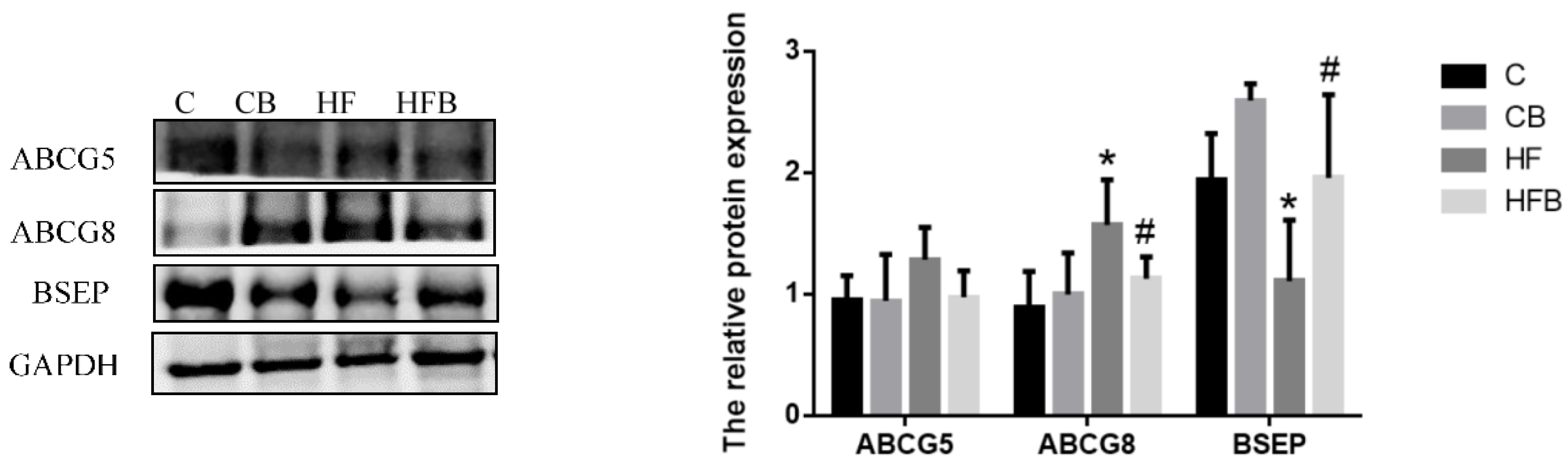
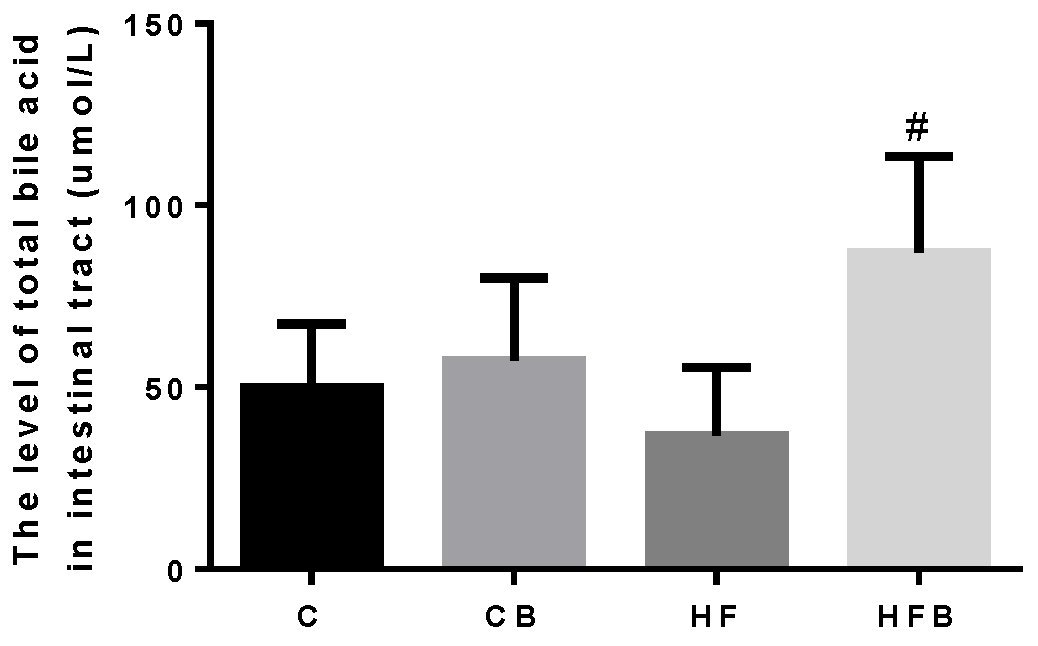
3.4.3. Cholesterol Elimination
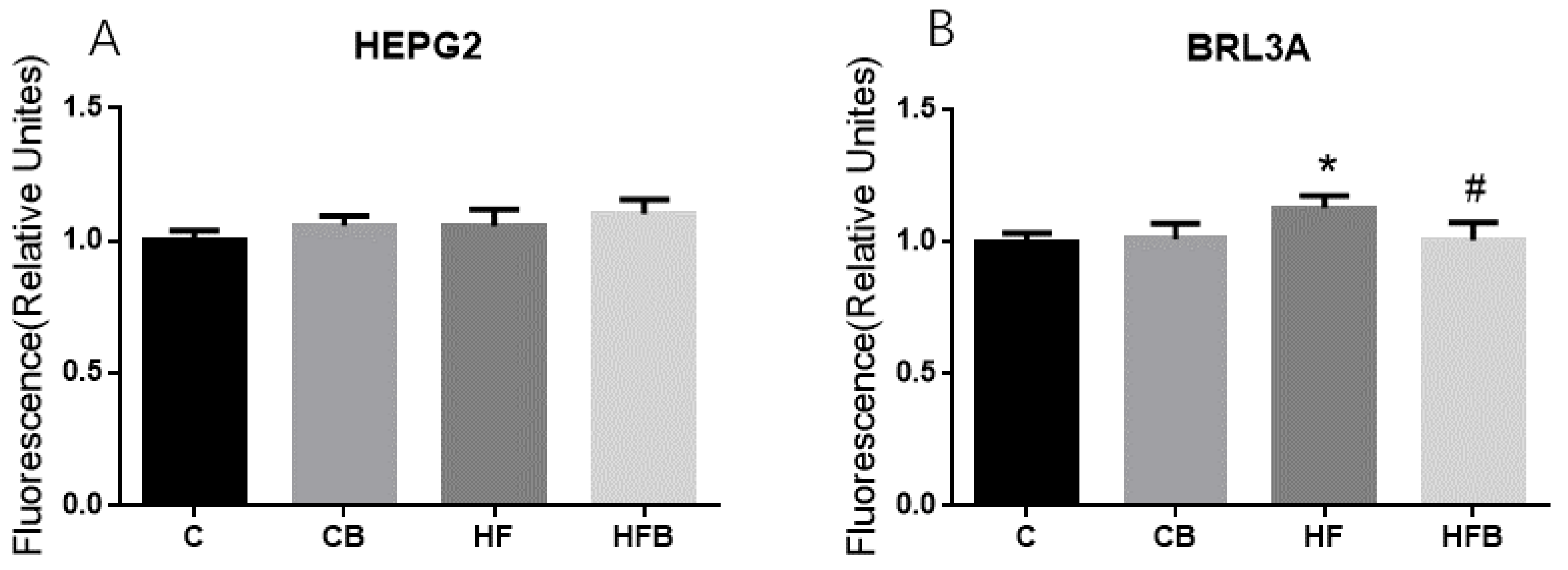
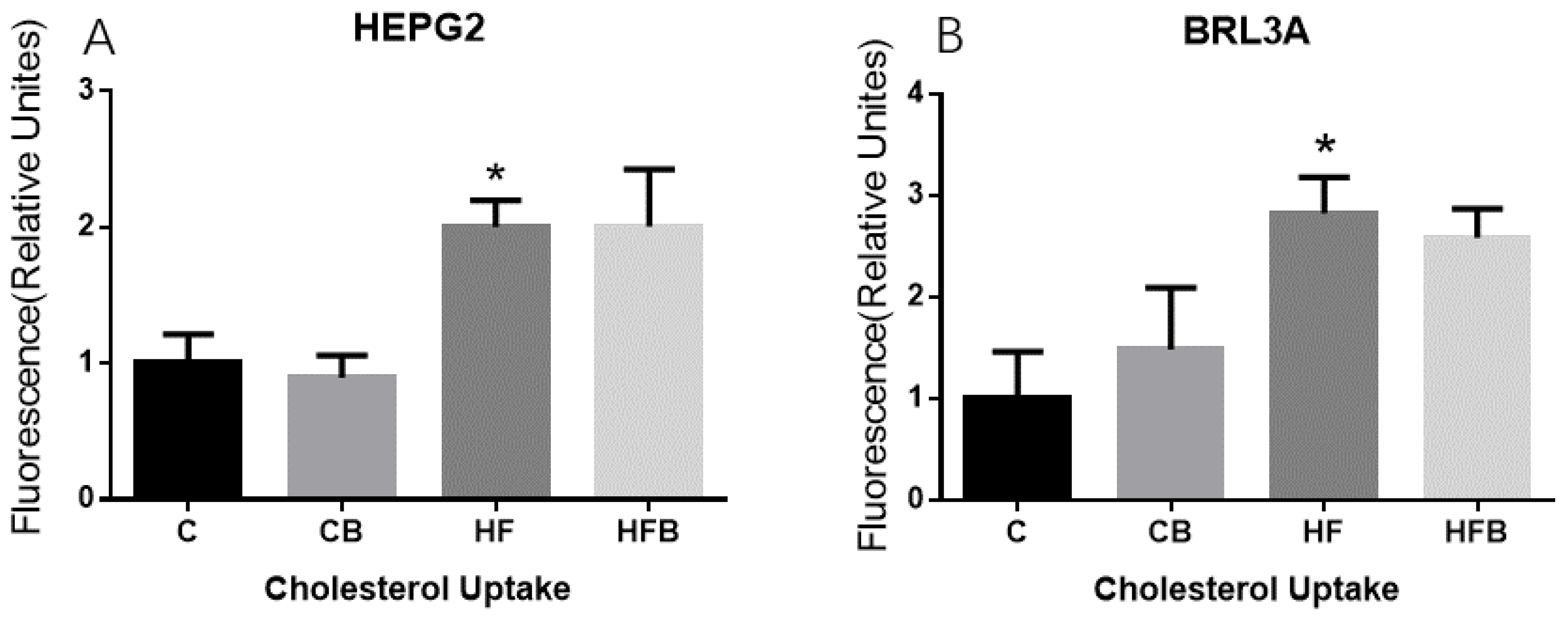
3.5. Cholesterol of Hepatocyte In Vitro
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- De Zwart, F.J.; Slow, S.; Payne, R.J.; Lever, M.; George, P.M.; Gerrard, J.A. Glycine betaine and glycine betaine analogues in common foods. Food Chem. 2003, 83, 197–204. [Google Scholar] [CrossRef]
- Eklund, M.; Bauer, E.; Wamatu, J.; Mosenthin, R. Potential nutritional and physiological functions of betaine in livestock. Nutr. Res. Rev. 2005, 18, 31–48. [Google Scholar] [CrossRef]
- Filipčev, B.; Kojić, J.; Krulj, J.; Marija, B.; Nebojša, I. Betaine in cereal grains and grain-based products. Foods 2018, 7, 49. [Google Scholar] [CrossRef]
- Clow, K.A.; Treberg, J.R.; Brosnan, M.E.; Brosnan, J.T. Elevated tissue betaine contents in developing rats are due to dietary betaine, not to synthesis. J. Nutr. 2008, 138, 1641–1646. [Google Scholar] [CrossRef]
- Lang, F. Mechanisms and significance of cell volume regulation. J. Am. Coll. Nutr. 2007, 26, 613S–623S. [Google Scholar] [CrossRef] [PubMed]
- Natalello, A.; Liu, J.; Ami, D.; Doglia, S.M.; Marco, A.D. The osmolyte betaine promotes protein misfolding and disruption of protein aggregates. Proteins 2009, 75, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, S.; Fang, S.; Yang, X.J.; Feng, J. Betaine Improves Intestinal Functions by Enhancing Digestive Enzymes, Ameliorating Intestinal Morphology, and Enriching Intestinal Microbiota in High-salt stressed Rats. Nutrients 2018, 10, 907. [Google Scholar] [CrossRef] [PubMed]
- Lever, M.; Slow, S. The clinical significance of betaine, an osmolyte with a key role in methyl group metabolism. Clin. Biochem. 2010, 43, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, S.; Ciria, G.; Elisa, M.; Valenzuela, S. Glycine betaine rather than acting only as an osmolyte also plays a role as regulator in cellular metabolism. Biochimie 2018, 147, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Craig, S.A. Betaine in human nutrition. Am. J. Clin. Nutr. 2004, 80, 539–549. [Google Scholar] [CrossRef]
- Pekkinen, J.; Olli, K.; Huotari, A.; Tiihonen, K.; Keski, R.P.; Lehtonen, M.; Auriola, S.; Kolehmainen, M.; Mykknen, H.; Poutanen, K.; et al. Betaine supplementation causes increase in carnitine metabolites in the muscle and liver of mice fed a high-fat diet as studied by nontargeted LCMS metabolomics approach. Mol. Nutr. Food Res. 2013, 57, 1959–1968. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Huang, D.; Hu, Q.; Wu, J.; Wang, Y.; Feng, J. Betaine alleviates hepatic lipid accumulation via enhancing hepatic lipid export and fatty acid oxidation in rats fed with a high-fat diet. Br. J. Nutr. 2015, 113, 1835–1843. [Google Scholar] [CrossRef]
- Martins, J.M.; Neves, J.A.; Freitas, A.; Tirapicos, J.L. Effect of long-term betaine supplementation on chemical and physical characteristics of three muscles from the Alentejano pig. J. Sci. Food Agric. 2012, 92, 2122–2127. [Google Scholar] [CrossRef]
- Huang, Q.C.; Han, X.Y.; Xu, Z.R.; Yang, X.Y.; Chen, T.; Zheng, X.T. Betaine suppresses carnitine palmitoyltransferase I in skeletal muscle but not in liver of finishing pigs. Livest. Sci. 2009, 126, 130–135. [Google Scholar] [CrossRef]
- Abdelmalek, M.F.; Angulo, P.; Jorgensen, R.A.; Sylvestre, P.B.; Lindor, K.D. Betaine, a promising new agent for patients with nonalcoholic steatohepatitis: Results of a pilot study. Am. J. Gastroenterol. 2001, 96, 2711. [Google Scholar] [CrossRef] [PubMed]
- Abdelmalek, M.F.; Sanderson, S.O.; Angulo, P.; Soldevila-Pico, C.; Liu, C.; Peter, J.; Keach, J.; Cave, M.; Chen, T.; McClain, C.J.; et al. Betaine for nonalcoholic fatty liver disease: Results of a randomized placebo-controlled trial. Hepatology 2009, 50, 1818–1826. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zhang, H.; Guo, X.; Li, K.; Li, S.; Li, D. Effect of Betaine on Reducing Body Fat—A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2019, 11, 2480. [Google Scholar] [CrossRef]
- Dietschy, J.M.; Turley, S.D.; Spady, D.K. Role of liver in the maintenance of cholesterol and low density lipoprotein homeostasis in different animal species, including humans. J. Lipid Res. 1993, 34, 1637–1659. [Google Scholar] [CrossRef]
- Millar, J.S.; Cuchel, M. Cholesterol metabolism in humans: A review of methods and comparison of results. Curr. Opin. Lipidol. 2018, 29, 1–9. [Google Scholar] [CrossRef]
- Goldstein, J.L.; Brown, M.S. The LDL receptor. Arterioscl. Throm. Vas. 2009, 29, 431–438. [Google Scholar] [CrossRef]
- Kozarsky, K.F.; Donahee, M.H.; Rigotti, A.; Lqbal, S.N.; Edelman, E.R.; Krieger, M. Overexpression of the HDL receptor SR-BI alters plasma HDL and bile cholesterol levels. Nature 1997, 387, 414. [Google Scholar] [CrossRef] [PubMed]
- DeBose-Boyd, R.A. Feedback regulation of cholesterol synthesis: Sterol-accelerated ubiquitination and degradation of HMG CoA reductase. Cell Res. 2008, 18, 609. [Google Scholar] [CrossRef] [PubMed]
- Stieger, B. The role of the sodium-taurocholate cotransporting polypeptide (NTCP) and of the bile salt export pump (BSEP) in physiology and pathophysiology of bile formation. Drug Transp. 2011, 205–259. [Google Scholar] [CrossRef]
- Calpe-Berdiel, L.; Rotllan, N.; Fievet, C.; Roig, R.; Blanco-Vaca, F.; Escola-Gil, J.C. Liver X receptor-mediated activation of reverse cholesterol transport from macrophages to feces in vivo requires ABCG5/G8. J. Lipid Res. 2008, 49, 1904–1911. [Google Scholar] [CrossRef]
- Cai, D.; Jia, Y.; Lu, J.; Yuan, M.; Sui, S.; Song, H.; Zhao, R. Maternal dietary betaine supplementation modifies hepatic expression of cholesterol metabolic genes via epigenetic mechanisms in newborn piglets. Br. J. Nutr. 2014, 112, 1459–1468. [Google Scholar] [CrossRef]
- Zhao, N.; Yang, S.; Feng, Y.; Sun, B.; Zhao, R. Enhanced hepatic cholesterol accumulation induced by maternal betaine exposure is associated with hypermethylation of CYP7A1 gene promoter. Endocrine 2019, 1–8. [Google Scholar] [CrossRef]
- Hu, Y.; Sun, Q.; Li, X.; Wang, M.; Cai, D.; Li, X.; Zhao, R. In Ovo Injection of Betaine Affects Hepatic Cholesterol Metabolism through Epigenetic Gene Regulation in Newly Hatched Chicks. PLoS ONE 2010, 10, e0122643. [Google Scholar] [CrossRef]
- Albuquerque, A.; Neves, J.A.; Redondeiro, M.; Laranjo, M.; Félix, M.R.; Freitas, A.; Tirapicos, J.L.; Martins, J.M. Long term betaine supplementation regulates genes involved in lipid and cholesterol metabolism of two muscles from an obese pig breed. Meat Sci. 2017, 124, 25–33. [Google Scholar] [CrossRef]
- Martins, J.M.; Neves, J.A.; Freitas, A.; Tirapicos, J.L. Betaine supplementation affects the cholesterol but not the lipid profile of pigs. Eur. J. Lipid Sci. Technol. 2010, 112, 295–303. [Google Scholar] [CrossRef]
- Du, J.; Shen, L.; Tan, Z.; Zhang, P.; Zhao, X.; Xu, Y.; Gan, M.; Yang, Q.; Ma, J.; Jiang, A.; et al. Betaine Supplementation Enhances Lipid Metabolism and Improves Insulin Resistance in Mice Fed a High-Fat Diet. Nutrients 2018, 10, 131. [Google Scholar] [CrossRef]
- Fernández-Fígares, I.; Lachica, M.; Martin, A.; Nieto, R.; González-Valero, L.; Rodríguez-López, J.M.; Aguilera, J.F. Impact of dietary betaine and conjugated linoleic acid on insulin sensitivity, protein and fat metabolism of obese pigs. Animal 2012, 6, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.W.; Choi, Y.J.; Hong, S.H.; Jun, D.S.; Na, J.D.; Choi, Y.J.; Kim, Y.C. Involvement of multiple pathways in the protection of liver against high-fat diet-induced steatosis by betaine. J. Funct. Foods 2015, 17, 66–72. [Google Scholar] [CrossRef]
- Lever, M.; George, P.M.; Atkinson, W.; Molyneux, S.L.; Elmsile, J.L.; Slow, S.; Richards, A.M.; Chambers, S.Y. Plasma lipids and betaine are related in an acute coronary syndrome cohort. PLoS ONE 2011, 6, e21666. [Google Scholar] [CrossRef] [PubMed]
- Rajaie, S.; Esmaillzadeh, A. Dietary choline and betaine intakes and risk of cardiovascular diseases: Review of epidemiological evidence. ARYA Atheroscler. 2011, 7, 78–86. Available online: www.ncbi.nlm.nih.gov/pmc/articles/PMC3347848/ (accessed on 12 July 2011). [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C. AIN-93 purified diets for laboratory rodents—Final report of the American institute of nutrition ad hoc writing committee on the reformulation of the AIN-76a rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef]
- Lee, M.R.; Kwang, P.; Jin, M. Leonurus japonicus Houtt attenuates nonalcoholic fatty liver disease in free fatty acid-induced HepG2 cells and mice fed a high-fat diet. Nutrients 2018, 10, 20. [Google Scholar] [CrossRef]
- Ratriyanto, A.; Mosenthin, R.; Bauer, E.; Eklund, M. Metabolic, osmoregulatory and nutritional functions of betaine in monogastric animals. Asian. Aust. J. Anim. 2009, 22, 1461–1476. [Google Scholar] [CrossRef]
- Sales, J. A meta-analysis of the effects of dietary betaine supplementation on finishing performance and carcass characteristics of pigs. Anim. Feed. Sci. Technol. 2011, 165, 68–78. [Google Scholar] [CrossRef]
- Lipiński, K.; Szramko, E.; Jeroch, H.; Matusevičius, P. Effects of betaine on energy utilization in growing pigs-A review. Ann. Anim. Sci. 2012, 12, 291–300. [Google Scholar] [CrossRef]
- Spady, D.K.; Woollett, L.A.; Dietschy, J.M. Regulation of plasma LDL-cholesterol levels by dietary cholesterol and fatty acids. Annu. Rev. Nutr. 1993, 13, 355–381. [Google Scholar] [CrossRef]
- Cho, A.S.; Jeon, S.M.; Kim, M.J.; Yeo, J.; Seo, K.I.; Choi, M.S.; Lee, M.K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Rang, A.P.; Dale, M.M.; Ritter, J.M. Pharmacology, 3rd ed.; Churchill Livingstone: New York, NY, USA, 1995; pp. 409–410. [Google Scholar]
- Korman, S.H.; Waterham, H.R.; Gutman, A.; Jakobs, C.; Wanders, R.A. Novel metabolic and molecular findings in hepatic carnitine palmitoyltransferase I deficiency. Mol. Genet. Metab. 2005, 86, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Varatharajalu, R.; Garige, M.; Leckey, L.C.; Arellanes-Robledo, J.; Reyes-Gordillo, K.; Shah, R.; Lakshman, R. Adverse signaling of scavenger receptor class B1 and PGC1s in alcoholic hepatosteatosis and steatohepatitis and protection by betaine in rat. Am. J. Pathol. 2014, 184, 2035–2044. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, W.; Huang, L.; Gao, J.; Wen, S.; Tai, Y.; Chen, M.; Huang, Z.; Liu, R.; Tang, C.; Li, J. Betaine attenuates chronic alcohol-induced fatty liver by broadly regulating hepatic lipid metabolism. Mol. Med. Rep. 2017, 16, 5225–5234. [Google Scholar] [CrossRef]
- Nosratola, D.V.; Kai, H.L. Down-regulation of hepatic LDL receptor expression in experimental nephrosis. Kidney Int. 1996, 50, 887–893. [Google Scholar] [CrossRef]
- Idriss, A.A.; Hu, Y.; Hou, Z.; Hu, Y.; Sun, Q.; Omer, N.A.; Abobaker, H.; Ni, Y.; Zhao, R. Dietary betaine supplementation in hens modulates hypothalamic expression of cholesterol metabolic genes in F1 cockerels through modification of DNA methylation. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2018, 217, 14–20. [Google Scholar] [CrossRef]
- Cai, D.; Yuan, M.; Liu, H.; Pan, S.; Ma, W.; Hong, J.; Zhao, R. Maternal Betaine Supplementation throughout Gestation and Lactation Modifies Hepatic Cholesterol Metabolic Genes in Weaning Piglets via AMPK/LXR-Mediated Pathway and Histone Modification. Nutrients 2016, 8, 646. [Google Scholar] [CrossRef]
- Zhu, X.; Parks, J.S. New roles of HDL in inflammation and hematopoiesis. Annu. Rev. Nutr. 2012, 32, 161–182. [Google Scholar] [CrossRef]
- Tall, A.R. An overview of reverse cholesterol transport. Eur. Heart J. 1998, 19, A31–A35. [Google Scholar] [CrossRef]
- Krieger, M. Scavenger receptor class B type I is a multiligand HDL receptor that influences diverse physiologic systems. J. Clin. Investig. 2001, 108, 793–797. [Google Scholar] [CrossRef]
- Li, X.; Yuanwu, L.; Hua, Z.; Liming, R.; Qiuyan, L.; Li, N. Animal models for the atherosclerosis research: A review. Protein Cell 2011, 2, 189–201. [Google Scholar] [CrossRef]
- Li, S.; Wang, H.; Wang, X.; Wang, Y.; Feng, J. Betaine affects muscle lipid metabolism via regulating the fatty acid uptake and oxidation in finishing pig. J. Anim. Sci. Biotechnol. 2017, 8, 72. [Google Scholar] [CrossRef]
- Matthews, J.O.; Southern, L.L.; Higbie, A.D.; Persica, M.A.; Bidner, T.D. Effects of betaine on growth, carcass characteristics, pork quality, and plasma metabolites of finishing pigs. J. Anim. Sci. 2001, 79, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Schwahn, B.C.; Wang, X.L.; Mikael, L.G.; Wu, Q.; Cohn, J.; Jiang, H.; Maclean, K.N.; Rozen, R. Betaine supplementation improves the atherogenic risk factor profile in a transgenic mouse model of hyperhomocysteinemia. Atherosclerosis 2007, 195, e100–e107. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.E.; Basso, F.; Shamburek, R.D.; Amar, M.J.A.; Vaisman, B.; Szakacs, G.; Joyce, C.; Tansey, T.; Freeman, L.; Paigen, B.J.; et al. Hepatic ABCG5 and ABCG8 overexpression increases hepatobiliary sterol transport but does not alter aortic atherosclerosis in transgenic mice. J. Biol. Chem. 2004, 279, 22913–22925. [Google Scholar] [CrossRef] [PubMed]
- Vaz, F.M.; Ferdinandusse, S. Bile acid analysis in human disorders of bile acid biosynthesis. Mol. Asp. Med. 2017, 56, 10–24. [Google Scholar] [CrossRef]
- Chiang, J.Y.L. Bile acids: Regulation of synthesis. J. Lipid Res. 2009, 50, 1955–1966. [Google Scholar] [CrossRef]
- Zapadniuk, V.I.; Panteleĭmonova, T.N. Cholagogic effect of trimethylglycine in normal animals of different ages and in experimental atherosclerosis. Biulleten’ Eksperimental’noi Biol. I Meditsiny 1987, 104, 30–32. [Google Scholar]
- Stieger, B.; Meier, Y.; Meier, P.J. The bile salt export pump. Pflügers Arch. Eur. J. Physiol. 2007, 453, 611–620. [Google Scholar] [CrossRef]
- Westergaard, H.; Dietschy, J.M. The mechanism whereby bile acid micelles increase the rate of fatty acid and cholesterol uptake into the intestinal mucosal cell. J. Clin. Investig. 1976, 58, 97–108. [Google Scholar] [CrossRef]


| Ingredient | Basal Diet (g/kg) | High-Fat Diet (g/kg) |
|---|---|---|
| Maize starch | 504.5 | 293.24 |
| Casein | 230.48 | 227.58 |
| Sucrose | 100.00 | 194.60 |
| Soybean oil | 60.00 | 32.76 |
| Lard oil | 0.0 | 169.00 |
| Fiber | 50.00 | 27.30 |
| Mineral Mix# | 35.00 | 35.00 |
| Vitamin Mix# | 10.00 | 10.00 |
| L-Cys | 3.00 | 3.00 |
| Choline bitartrate | 2.50 | 2.50 |
| Antioxidant | 0.02 | 0.02 |
| Primary Antibody | Order Numbers | Dilution | Size, kDa |
|---|---|---|---|
| Anti-HMGCR | ab174830, Abcam | 1/5000 | 97 |
| Anti-SRBI | ab217318, Abcam | 1/2000 | 60–82 |
| Anti-LDLR | 10785-1-AP, Proteintech | 1/2500 | 150–160 |
| Anti-CYP7A1 | bs-21429R, bioss | 1/1500 | 55 |
| Anti-ABCB11/BSEP | orb259591, Biorbyt | 1/3000 | 146 |
| Anti-ABCG5 | 27722-1-AP, Proteintech | 1/1500 | 68–72 |
| Anti-ABCG8 | orb228808, Biorbyt | 1/1500 | 76 |
| C | CB | HF | HFB | p-Value | |||
|---|---|---|---|---|---|---|---|
| BET | FAT | BET × FAT | |||||
| Initial body weight, g | 188.4 ± 3.9 | 185.3 ± 6.1 | 186.4 ± 7.2 | 184.9 ± 4.7 | 0.252 | 0.569 | 0.700 |
| Final body weight, g | 242.5 ± 10.0 | 244.4 ± 7.9 | 254.9 ± 29.7 | 244.9 ± 14.1 | 0.520 | 0.310 | 0.349 |
| Average daily gain, g/d | 1.93 ± 0.34 | 2.11 ± 0.27 | 2.46 ± 0.43 * | 2.25 ± 0.32 | 0.598 | 0.035 | 0.060 |
| Average daily feed intake, g/kg BW | 79.12 ± 12 | 75.47 ± 6.09 | 62.84 ± 7.46 * | 58.13 ± 8.37# | 0.008 | <0.001 | 0.743 |
| Liver index, % | 3.90 ± 0.22 | 3.84 ± 0.26 | 3.89 ± 0.57 | 3.80 ± 0.17 | 0.542 | 0.863 | 0.894 |
| C | CB | HF | HFB | p-Value | |||
|---|---|---|---|---|---|---|---|
| FAT | BET | FAT × BET | |||||
| TG, mmol/L | 1.47 ± 0.43 | 1.50 ± 0.21 | 1.76 ± 0.60 | 1.77 ± 0.38 | 0.075 | 0.862 | 0.951 |
| NEFA, mmol/L | 0.66 ± 0.15 | 0.52 ± 0.11 * | 0.70 ± 0.18 | 0.57 ± 0.55 | 0.435 | 0.008 | 0.932 |
| VLDL-C, umol/mL | 0.40 ± 0.11 | 0.53 ± 0.20 | 0.33 ± 0.17 | 0.53 ± 0.17 # | 0.639 | 0.021 | 0.596 |
| LPC, ug/mL | 0.71 ± 0.09 | 1.15 ± 0.12 * | 1.11 ± 0.09 | 1.10 ± 0.43 | 0.080 | 0.034 | 0.029 |
| TC, mmol/L | 2.19 ± 0.60 | 2.21 ± 0.51 | 2.38 ± 0.45 | 2.79 ± 0.77 | 0.080 | 0.331 | 0.370 |
| HDLC, mmol/L | 0.83 ± 0.20 | 0.93 ± 0.32 | 0.84 ± 0.15 | 0.92 ± 0.20 | 0.970 | 0.307 | 0.867 |
| LDLC, mmol/L | 0.38 ± 0.07 | 0.36 ± 0.06 | 0.42 ± 0.07 | 0.44 ± 0.12 | 0.045 | 0.845 | 0.538 |
| TBA, umol/L | 43.0 ± 19.5 | 16.0 ± 2.9 * | 14.9 ± 6.7 * | 25.0 ± 10.3 | 0.039 | 0.066 | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Xu, S.; Zhao, Y.; Wang, H.; Feng, J. Dietary Betaine Addition Promotes Hepatic Cholesterol Synthesis, Bile Acid Conversion, and Export in Rats. Nutrients 2020, 12, 1399. https://doi.org/10.3390/nu12051399
Li S, Xu S, Zhao Y, Wang H, Feng J. Dietary Betaine Addition Promotes Hepatic Cholesterol Synthesis, Bile Acid Conversion, and Export in Rats. Nutrients. 2020; 12(5):1399. https://doi.org/10.3390/nu12051399
Chicago/Turabian StyleLi, Sisi, Shuyi Xu, Yang Zhao, Haichao Wang, and Jie Feng. 2020. "Dietary Betaine Addition Promotes Hepatic Cholesterol Synthesis, Bile Acid Conversion, and Export in Rats" Nutrients 12, no. 5: 1399. https://doi.org/10.3390/nu12051399
APA StyleLi, S., Xu, S., Zhao, Y., Wang, H., & Feng, J. (2020). Dietary Betaine Addition Promotes Hepatic Cholesterol Synthesis, Bile Acid Conversion, and Export in Rats. Nutrients, 12(5), 1399. https://doi.org/10.3390/nu12051399




