Vitamin K as a Diet Supplement with Impact in Human Health: Current Evidence in Age-Related Diseases
Abstract
1. Introduction
2. Clinical Context of Vitamin K
2.1. Vitamin K Deficiency
2.2. Vitamin K Antagonists (VKAs)
3. Vitamin K1 and K2: Similar Function but Different Absorption, Storage, Bioavailability, and Targets
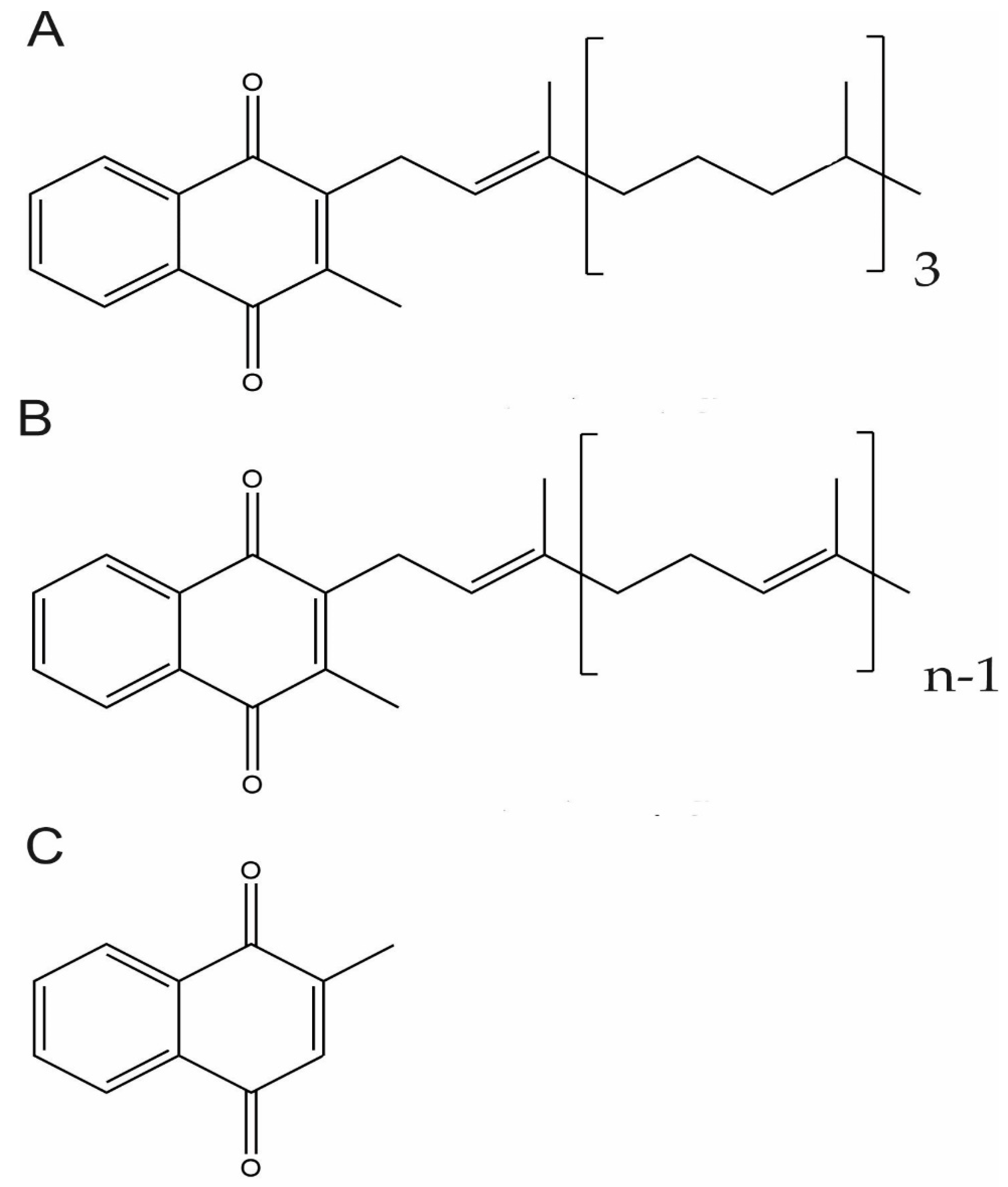
3.1. Vitamin K Chemical Structure
3.2. Vitamin K Functions
3.3. Vitamin K1 and K2 Absorption, Storage, and Bioavailability
4. Dietary Sources of Vitamin K1 and K2
5. Vitamin K1 and K2 Chemical and Biotechnological Production Methods
5.1. Vitamin K1
5.2. Vitamin K2
5.3. Aquatic Organisms as Sources of Vitamin K with a Potential Biotechnological Application
6. Vitamin K Formulations and Impact on Absorption and Bioavailability
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CHD | Coronary heart disease |
| CKD | Chronic kidney disease |
| CRP | C-reactive protein |
| CVD | Cardiovascular disease |
| DOACs | Direct oral anticoagulants |
| GGCX | γ-glutamyl carboxylase |
| Gas6 | Growth arrest-specific protein 6 |
| Gla | γ-carboxyglutamic acid |
| Glu | Glutamic acid |
| GRAS | Generally recognized as safe |
| GRP | Gla-rich protein |
| HD | Haemodialysis |
| IKB | IkappaB kinase |
| INR | International normalized ratio |
| KH2 | Vitamin K hydroquinone |
| KO | Vitamin K 2,3-epoxide |
| LDL | Low-density lipoproteins |
| 12-LOX | 12-lipoxygenase |
| LPS | Liposaccharide |
| LSF | Liquid-state fermentation process |
| MGP | Matrix-Gla protein |
| MKs | Menaquinones |
| MM | Mixed micelles |
| NF-κB | Nuclear factor kappa B |
| OA | Osteoarthritis |
| OC | Osteocalcin |
| PRGP1 | Proline-rich Gla proteins |
| RA | Rheumatoid arthritis |
| ROS | Reactive oxygen species |
| SSF | Solid-state fermentation process |
| TLR | Triacylglycerol-rich lipoproteins |
| TMG | Transmembrane Gla proteins |
| VC | Vascular calcification |
| VKAs | Vitamin K antagonists |
| VKDB | Vitamin K deficiency bleeding |
| VKDPs | Vitamin K-dependent proteins |
| VKORC1 | Vitamin K epoxide reductase complex subunit 1 |
| VKORC1L1 | Vitamin K epoxide reductase complex subunit 1-like 1 |
| WHO | World Health Organization |
References
- Dam, B.Y.H.; Schnheyder, F. The occurence and chemical nature of vitamin k. Biochem. J. 1936, 30, 897–901. [Google Scholar] [PubMed]
- Nelsestuen, G.L.; Suttie, W. Mode of Action of Vitamin K. Calcium Binding Properties of Bovine Prothrombint. Biochem. J. 1972, 11, 4961–4964. [Google Scholar] [CrossRef] [PubMed]
- Willems, B.A.G.; Vermeer, C.; Chris, P.; Reutelingsperger, M.; Schurgers, L.J. The realm of vitamin K dependent proteins: Shifting from coagulation toward calcification. Mol. Nutr. Food Res. 2014, 58, 1620–1635. [Google Scholar] [CrossRef] [PubMed]
- Cranenburg, E.C.M.; Schurgers, L.J.; Uiterwijk, H.H.; Beulens, J.W.J.; Dalmeijer, G.W.; Westerhuis, R.; Magdeleyns, E.J.; Herfs, M.; Vermeer, C.; Laverman, G.D. Vitamin K intake and status are low in hemodialysis patients. Kidney Int. 2012, 82, 605–610. [Google Scholar] [CrossRef]
- Misra, D.; Booth, S.L.; Tolstykh, I.; Felson, D.T.; Nevitt, M.C.; Lewis, C.E.; Torner, J.; Neogi, T. Vitamin K Deficiency Is Associated with Incident Knee Osteoarthritis. Am. J. Med. 2013, 126, 243–248. [Google Scholar] [CrossRef]
- Simes, D.C.; Viegas, C.S.B.; Araujo, N.; Marreiros, C. Vitamin K as a Powerful Micronutrient in Aging and Age-Related Diseases: Pros and Cons from Clinical Studies. IJMS 2019, 20, 4150. [Google Scholar] [CrossRef]
- Lippi, G.; Favaloro, E.J. Laboratory hemostasis: From biology to the bench. Clin. Chem. Lab. Med. 2018, 56, 1035–1045. [Google Scholar] [CrossRef]
- Dahlbäck, B. Blood coagulation and its regulation by anticoagulant pathways: Genetic pathogenesis of bleeding and thrombotic diseases. J. Intern. Med. 2005, 257, 209–223. [Google Scholar] [CrossRef]
- Van De Wouwer, M.; Collen, D.; Conway, E.M. Thrombomodulin-protein C-EPCR system integrated to regulate coagulation and inflammation. Arter. Thromb. Vasc. Biol. Biol. 2004, 24, 1374–1383. [Google Scholar] [CrossRef]
- Riewald, M.; Petrovan, R.J.; Donner, A.; Mueller, B.M.; Ruf, W. Activation of endothelial cell protease activated receptor 1 by the protein C pathway. Science 2002, 296, 1880–1882. [Google Scholar] [CrossRef]
- Maillard, C.; Berruyer, M.; Serre, C.; Dechavanne, M.; Delmas, P. Protein-S, a vitamin K-dependent protein, is a bone matrix component synthesized and secreted by osteoblasts. Endocrinology 1992, 130, 1599–1604. [Google Scholar]
- Rezende, S.M.; Simmonds, R.E.; Lane, D.A. Coagulation, inflammation, and apoptosis: Different roles for protein S and the protein S-C4b binding protein complex. Blood 2004, 103, 1192–1201. [Google Scholar] [CrossRef]
- Vasse, M. Protein Z, a protein seeking a pathology. Thromb. Haemost. 2008, 100, 548–556. [Google Scholar] [CrossRef]
- Almawi, W.Y.; Al-Shaikh, F.S.; Melemedjian, O.K.; Almawi, A.W. Protein Z, an anticoagulant protein with expanding role in reproductive biology. Reproduction 2013, 146, R73–R80. [Google Scholar] [CrossRef] [PubMed]
- Neve, A.; Corrado, A.; Cantatore, F.P. Osteocalcin: Skeletal and extra-skeletal effects. J. Cell. Physiol. 2013, 228, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Zoch, M.L.; Clemens, T.L.; Riddle, R.C. New Insights into the Biology of Osteocalcin. Bone 2016, 27, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Bjorklund, G.; Svanberg, E.; Dadar, M.; David, J.C.; Salvatore, C.; Dominic, J.H.; Jan, A. The role of matrix Gla protein (MGP) in vascular calcification. Curr. Med. Chem. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gheorghe, S.R.; Craciun, A.M. Matrix Gla protein in tumoral pathology. Clujul Med. 2016, 89, 319–321. [Google Scholar] [CrossRef]
- Boström, K.; Zebbondj, A.F.; Yao, Y.; Lin, T.S.; Torres, A. Matrix GLA protein stimulates VEGF expression through increased transforming growth factor-β1 activity in endothelial cells. J. Biol. Chem. 2004, 279, 52904–52913. [Google Scholar] [CrossRef]
- Fernández-Fernández, L.; Bellido-Martín, L.; De Frutos, P.G. Growth arrest-specific gene 6 (GAS6): An outline of its role in haemostasis and inflammation. Thromb. Haemost. 2008, 100, 604–610. [Google Scholar] [CrossRef]
- Cosemans, J.M.E.M.; Van Kruchten, R.; Olieslagers, S.; Schurgers, L.J.; Verheyen, F.K.; Munnix, I.C.A.; Waltenberger, J.; Angelillo-Scherrer, A.; Hoylaerts, M.F.; Carmeliet, P.; et al. Potentiating role of Gas6 and Tyro3, Axl and Mer (TAM) receptors in human and murine platelet activation and thrombus stabilization. J. Thromb. Haemost. 2010, 8, 1797–1808. [Google Scholar] [CrossRef] [PubMed]
- Hasanbasic, I.; Rajotte, I.; Blostein, M. The role of γ-carboxylation in the anti-apoptotic function of gas6. J. Thromb. Haemost. 2005, 3, 2790–2797. [Google Scholar] [CrossRef] [PubMed]
- Viegas, C.S.B.; Costa, R.M.; Santos, L.; Videira, P.A.; Silva, Z.; Araújo, N.; Macedo, A.L.; Matos, A.P.; Vermeer, C.; Simes, D.C. Gla-rich protein function as an anti-inflammatory agent in monocytes/macrophages: Implications for calcification-related chronic inflammatory diseases. PLoS ONE 2017, 12, e0177829. [Google Scholar] [CrossRef] [PubMed]
- Viegas, C.S.B.; Rafael, M.S.; Enriquez, J.L.; Teixeira, A.; Vitorino, R.; Luís, I.M.; Costa, R.M.; Santos, S.; Cavaco, S.; Neves, J.; et al. Gla-Rich Protein acts as a Calcification Inhibitor in the Human Cardiovascular System. Arter. Thromb. Vasc. Biol. 2015, 35, 399–408. [Google Scholar] [CrossRef]
- Cavaco, S.; Viegas, C.S.B.; Rafael, M.S.; Ramos, A.; Magalhães, J.; Blanco, F.J.; Vermeer, C.; Simes, D.C. Gla-rich protein is involved in the cross-talk between calcification and inflammation in osteoarthritis. Cell. Mol. Life Sci. 2016, 73, 1051–1065. [Google Scholar] [CrossRef]
- Viegas, C.S.B.; Santos, L.; Macedo, A.L.; Matos, A.A.; Silva, A.P.; Neves, P.L. Chronic Kidney Disease Circulating Calciprotein Particles and Extracellular Vesicles Promote Vascular Calcification: A Role for GRP (Gla-Rich Protein). Arter. Thromb. Vasc. Biol. 2018, 38, 575–587. [Google Scholar] [CrossRef]
- Hamilton, D.W. Functional role of periostin in development and wound repair: Implications for connective tissue disease. J. Cell Commun. Signal. 2008, 2, 9–17. [Google Scholar] [CrossRef]
- Kim, B.R.; Kwon, Y.W.; Park, G.T.; Choi, E.J.; Seo, J.K.; Jang, I.H.; Kim, S.C.; Ko, H.C.; Lee, S.C.; Kim, J.H. Identification of a novel angiogenic peptide from periostin. PLoS ONE 2017, 12, e0187464. [Google Scholar] [CrossRef]
- Kühn, B.; Del Monte, F.; Hajjar, R.J.; Chang, Y.S.; Lebeche, D.; Arab, S.; Keating, M.T. Periostin induces proliferation of differentiated cardiomyocytes and promotes cardiac repair. Nat. Med. 2007, 13, 962–969. [Google Scholar] [CrossRef]
- Kulman, J.D.; Harris, J.E.; Xie, L.; Davie, E.W. Proline-rich Gla protein 2 is a cell-surface vitamin K-dependent protein that binds to the transcriptional coactivator Yes-associated protein. Proc. Natl. Acad. Sci. USA 2007, 104, 8767–8772. [Google Scholar] [CrossRef]
- Kulman, J.D.; Harris, J.E.; Haldeman, B.A.; Davie, E.W. Primary structure and tissue distribution of two nove proline-rich γ-carboxyglutamic acid proteins. Proc. Natl. Acad. Sci. USA 1997, 94, 9058–9062. [Google Scholar] [CrossRef] [PubMed]
- Khazi, F.R.; Chu, K.C.; High, K.A. Transmembrane Gla Protein 4 as a Novel Modulator of ERK2. Blood 2006, 108, 544. [Google Scholar] [CrossRef]
- Presnell, S.R.; Stafford, D.W. The Vitamin K-dependent Carboxylase. Thromb. Haemost. 2002, 87, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Shikdar, S.; Bhattacharya, P.T. International Normalized Ratio (INR) [Updated 2019 Mar 25]. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2019. Available online: https://www.ncbi.nlm.nih.gov/books/NBK507707/ (accessed on 12 December 2019).
- Conly, J.; Stein, K.; Worobetz, L.; Rutledge-Harding, S. The contribution of vitamin K2 (menaquinones) produced by the intestinal microflora to human nutritional requirements for vitamin K. Am. J. Gastroenterol. 1994, 89, 915–923. [Google Scholar]
- Aziz, F.; Patil, P. Role of Prophylactic Vitamin K in Preventing Antibiotic Induced Hypoprothrombinemia. Indian J. Pediatr. 2015, 82, 363–367. [Google Scholar] [CrossRef]
- Shevcbuk, Y.M.; Conly, J.M. Antibiotic-Associated Hypoprothrombinemia: A Review of Prospective Studies, 1966–1988. Rev. Infect. Dis. 1990, 12, 1109–1126. [Google Scholar] [CrossRef]
- Cinaz, S.Y.; Tavi, B.; Sarı, S.; Cinaz, P. Vitamin k deficiency because of ceftriaxone usage and prolonged diarrhoea. JPCH 2011, 47, 314–315. [Google Scholar]
- Chen, L.; Hsiao, F.; Shen, L.; Wu, F.L.; Tsay, W. Use of Hypoprothrombinemia-Inducing Cephalosporins and the Risk of Hemorrhagic Events: A Nationwide Nested Case-Control Study. PLoS ONE 2016, 27, e0158407. [Google Scholar] [CrossRef]
- Vroonhof, K.; Van Rijn, H.J.M.; Van Hattum, J. Vitamin K deficiency and bleeding after long-term use of cholestyramine. Neth. J. Med. 2003, 61, 19–21. [Google Scholar]
- MacWalter, R.; Fraser, H.; Armstrong, K. Orlistat Enhances Warfarin Effect. Ann. Pharmacother. 2003, 37, 510–512. [Google Scholar] [CrossRef]
- Sutor, A.; von Kries, R.; Cornelissen, E.; McNinch, A.; Andrew, M. Vitamin K deficiency bleeding (VKDB) in infancy. ISTH Pediatric/Perinatal Subcommittee. International Society on Thrombosis and Haemostasis. Thromb. Haemost. 1999, 81, 456–461. [Google Scholar] [PubMed]
- American Academy of Pediatrics Committee on Fetus and Newborn. Controversies concerning vitamin K and the newborn. Pediatrics 2003, 112, 191–192. [Google Scholar]
- Pichler, E.; Pichler, L. The neonatal coagulation system and the vitamin K deficiency bleeding—A mini review. Wien. Med. Wochenschr. 2008, 158, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Schulte, R.; Jordan, L.C.; Morad, A.; Naftel, R.P.; Iii, J.C.W.; Sidonio, R. Rise in Late Onset Vitamin K Defi ciency Bleeding in Young Infants Because of Omission or Refusal of Prophylaxis at Birth. Pediatr. Neurol. 2014, 50, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Chen, J.; Duan, L.; Li, S. Vitamin K-dependent proteins involved in bone and cardiovascular health. Mol. Med. Rep. 2018, 18, 3–15. [Google Scholar] [CrossRef]
- Alisi, L.; Cao, R.; De Angelis, C.; Cafolla, A.; Caramia, F.; Cartocci, G.; Librando, A.; Fiorelli, M. The Relationships Between Vitamin K and Cognition: A Review of Current Evidence. Front. Neurol. 2019, 10, 239. [Google Scholar] [CrossRef]
- Shea, M.K.; Kritchevsky, S.B.; Hsu, F.; Nevitt, M.; Booth, S.L.; Kwoh, C.K.; Mcalindon, T.E.; Drummen, N.; Harris, T.B.; Womack, C.; et al. The association between vitamin K status and knee osteoarthritis features in older adults: The Health, Aging and Body Composition Study. Osteoarthr. Cartil. 2016, 23, 370–378. [Google Scholar] [CrossRef]
- Shea, M.K.; Booth, S.L.; Weiner, D.E.; Brinkley, T.E.; Kanaya, A.M.; Murphy, R.A.; Simonsick, E.M.; Wassel, C.L.; Vermeer, C.; Kritchevsky, S.B. Circulating Vitamin K Is Inversely Associated with Incident Cardiovascular Disease Risk among Those Treated for Hypertension in the Health, Aging, and Body Composition Study (Health ABC) 1–3. J. Nutr. 2017, 147, 888–895. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, L.; Bu, C. Vitamin K status and cardiovascular events or mortality: A meta-analysis. Eur. J. Prev. Cardiol. 2019, 26, 549–553. [Google Scholar] [CrossRef]
- Ferland, G. Vitamin K, an emerging nutrient in brain function. Biofactors 2012, 38, 151–157. [Google Scholar] [CrossRef]
- Chatrou, M.L.; Winckers, K.; Hackeng, T.M.; Reutelingsperger, C.P.; Schurgers, L.J. Vascular calcification: The price to pay for anticoagulation therapy with vitamin K-antagonists. Blood Rev. 2012, 26, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Dinicolantonio, J.J.; Bhutani, J.; Keefe, J.H.O. The health benefits of vitamin K. Open Heart 2015, 2, e000300. [Google Scholar] [CrossRef] [PubMed]
- Namba, S.; Yamaoka-tojo, M.; Hashikata, T.; Ikeda, Y.; Kitasato, L.; Hashimoto, T.; Shimohama, T.; Tojo, T.; Takahira, N.; Masuda, T.; et al. Long-term warfarin therapy and biomarkers for osteoporosis and atherosclerosis. BBA Clin. 2015, 12, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Van Gorp, R.H.; Schurgers, L.J. New Insights into the Pros and Cons of the Clinical Use of Vitamin K Antagonists (VKAs) Versus Direct Oral Anticoagulants (DOACs). Nutrients 2015, 7, 9538–9557. [Google Scholar] [CrossRef] [PubMed]
- Schurgers, L.J.; Uitto, J.; Reutelingsperger, C.P. Vitamin K-dependent carboxylation of matrix Gla-protein: A crucial switch to control ectopic mineralization. Trends Mol. Med. 2013, 19, 217–226. [Google Scholar] [CrossRef]
- Schurgers, L.J.; Spronk, H.M.H.; Soute, B.A.M.; Schiffers, P.M.; Demey, J.G.R.; Vermeer, C. Regression of warfarin-induced medial elastocalcinosis by high intake of vitamin K in rats. Blood 2007, 109, 2823–2831. [Google Scholar] [CrossRef]
- Lerner, R.G.; Aronow, W.S.; Sekhri, A.; Palaniswamy, C.; Ahn, C.; Singh, T.; Sandhu, R. Warfarin use and the risk of valvular calcification. J. Thromb. Haemost. 2009, 7, 2023–2027. [Google Scholar] [CrossRef]
- Rennenberg, R.J.M.W.; Van Varik, B.J.; Schurgers, L.J.; Hamulyak, K.; Cate, H.; Leiner, T.; Vermeer, C.; De Leeuw, P.W.; Kroon, A.A. Chronic coumarin treatment is associated with increased extracoronary arterial calcification in humans. Blood 2010, 115, 5121–5123. [Google Scholar] [CrossRef]
- Han, K.H.; O’Neill, W.C. Increased Peripheral Arterial Calcification in Patients Receiving Warfarin. J. Am. Heart Assoc. 2016, 5, e002665. [Google Scholar] [CrossRef]
- Schurgers, L.J.; Joosen, I.A.; Laufer, E.M.; Chatrou, M.L.L.; Herfs, M.; Winkens, M.H.M.; Westenfeld, R.; Veulemans, V.; Krueger, T.; Shanahan, C.M.; et al. Vitamin K-Antagonists Accelerate Atherosclerotic Calcification and Induce a Vulnerable Plaque Phenotype. PLoS ONE 2012, 7, e43229. [Google Scholar] [CrossRef]
- Koos, R.; Mahnken, A.H.; Mühlenbruch, G.; Brandenburg, V.; Pflueger, B.; Wildberger, J.E.; Kühl, H.P. Relation of Oral Anticoagulation to Cardiac Valvular and Coronary Calcium Assessed by Multislice Spiral Computed Tomography. Am. J. Cardiol. 2005, 96, 747–749. [Google Scholar] [CrossRef] [PubMed]
- Weijs, B.; Blaauw, Y.; Rennenberg, R.; Schurgers, L.; Timmermans, C.; Pison, L.; Nieuwlaat, R.; Hofstra, L.; Kroon, A.; Wildberger, J.; et al. Patients using vitamin K antagonists show increased levels of coronary calcification: An observational study in low-risk atrial fibrillation patients. Eur. Heart J. 2011, 32, 2555–2562. [Google Scholar] [CrossRef] [PubMed]
- Villines, T.C.; Malley, P.G.O.; Feuerstein, I.M.; Thomas, S.; Taylor, A.J. Does Prolonged Warfarin Exposure Potentiate Coronary Calcification in Humans? Results of the Warfarin and Coronary Calcification Study. Calcif. Tissue Int. 2009, 85, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Mac-Way, F.; Poulin, A.; Utescu, M.S.; De Serres, S.A.; Marquis, K.; Douville, P.; Desmeules, S.; Larivière, R.; Lebel, M.; Agharazii, M. The impact of warfarin on the rate of progression of aortic stiffness in hemodialysis patients: A longitudinal study. Nephrol. Dial. Transpl. 2014, 29, 2113–2120. [Google Scholar] [CrossRef]
- Verdalles Guzmán, Ú.; De La Cueva, P.; Verde, E.; De Vinuesa, S.G.; Goicoechea, M.; Mosse, A.; López Gómez, J.M.; Luño, J. Calciphylaxis: Fatal complication of cardiometabolic syndrome in patients with end stage kidney disease. Nefrologia 2008, 28, 32–36. [Google Scholar]
- Eggebrecht, L.; Prochaska, H.; Schulz, A.; Arnold, N.; Junger, C.; Gobel, S.; Laubert-reh, D.; Binder, H.; Beutel, M.E.; Pfeiffer, N.; et al. Intake of Vitamin K Antagonists and Worsening of Cardiac and Vascular Disease: Results From the Population-Based Gutenberg Study. J. Am. Heart Assoc. 2018, 7, e008650. [Google Scholar] [CrossRef]
- Andrews, J.; Psaltis, P.J.; Bayturan, O.; Shao, M.; Stegman, B.; Elshazly, M.; Kapadia, S.R.; Tuzcu, E.M.; Nissen, S.E.; Nicholls, S.J.; et al. Warfarin Use Is Associated With Progressive Coronary Arterial Calcification: Insights From Serial Intravascular Ultrasound. JACC Cardiovasc. Imaging 2018, 11, 1315–1323. [Google Scholar] [CrossRef]
- Peeters, F.; Dudink, E.; Kimenai, D.; Weijs, B.; Altintas, S.; Heckman, L.; Mihl, C.; Schurgers, L.; Wildberger, J.; Meex, S.; et al. Vitamin K Antagonists, Non-Vitamin K Antagonist Oral Anticoagulants, and Vascular Calcification in Patients with Atrial Fibrillation. TH Open 2018, 2, e391–e398. [Google Scholar] [CrossRef]
- Schwarb, H.; Tsakiris, D.A. New Direct Oral Anticoagulants (DOAC) and Their Use Today. Dent. J. 2016, 4, 5. [Google Scholar] [CrossRef]
- Shearer, M.; Newman, P. Metabolism and cell biology of vitamin K. Thromb. Haemost. 2008, 100, 530–547. [Google Scholar]
- Shearer, M.J.; Newman, P. Recent trends in the metabolism and cell biology of vitamin K with special reference to vitamin K cycling and MK-4 biosynthesis. J. Lipid Res. 2014, 55, 345–362. [Google Scholar] [CrossRef] [PubMed]
- Booth, S.L. Vitamin K: Food composition and dietary intakes. Food Nutr. Res. 2012, 1, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, K.; Hirota, Y.; Sawada, N.; Yuge, N.; Watanabe, M.; Uchino, Y.; Okuda, N.; Shimomura, Y.; Suhara, Y.; Okano, T. Identification of UBIAD1 as a novel human menaquinone-4 biosynthetic enzyme. Nature 2010, 468, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Tie, J.; Stafford, D. Structural and functional insights into enzymes of the vitamin K cycle. J. Thromb. Haemost. 2016, 14, 236–247. [Google Scholar] [CrossRef] [PubMed]
- Stafford, D.W. The vitamin K cycle. J. Thromb. Haemost. 2005, 3, 1873–1878. [Google Scholar] [CrossRef]
- Rishavy, M.A.; Berkner, K.L. Vitamin K Oxygenation, Glutamate Carboxylation, and Processivity: Defining the Three Critical Facets of Catalysis by the Vitamin K-Dependent Carboxylase. Adv. Nutr. 2012, 3, 135–148. [Google Scholar] [CrossRef]
- Shearer, M.J.; Fu, X.; Booth, S.L. Vitamin K Nutrition, Metabolism, and Requirements: UBIAD and Future Research. Adv. Nutr. 2012, 3, 182–195. [Google Scholar] [CrossRef]
- Buitenhuis, H.; Soute, B.; Vermeer, C. Comparison of the vitamins K1, K2 and K3 as cofactors for the hepatic vitamin K-dependent carboxylase. Biochim. Biophys. Acta 1990, 1034, 170–175. [Google Scholar] [CrossRef]
- Ohsaki, Y.; Shirakawa, H.; Hiwatashi, K.; Furukawa, Y.; Mizutani, T.; Komai, M. Vitamin K Suppresses Lipopolysaccharide-Induced Inflammation in the Rat. Biosci. Biotechnol. Biochem. 2006, 70, 926–932. [Google Scholar] [CrossRef]
- Ohsaki, Y.; Shirakawa, H.; Miura, A.; Giriwono, P.E.; Sato, S.; Ohashi, A.; Iribe, M.; Goto, T.; Komai, M. Vitamin K suppresses the lipopolysaccharide-induced expression of inflammatory cytokines in cultured macrophage-like cells via the inhibition of the activation of nuclear factor κB through the repression of IKKα/β phosphorylation. J. Nutr. Biochem. 2010, 21, 1120–1126. [Google Scholar] [CrossRef]
- Fujii, S.; Shimizu, A.; Takeda, N.; Oguchi, K.; Katsurai, T. Systematic synthesis and anti-inflammatory activity of x-carboxylated menaquinone derivatives. Investigations on identified and putative vitamin K2 metabolites. Bioorg. Med. Chem. 2015, 23, 2344–2352. [Google Scholar] [CrossRef] [PubMed]
- Mukai, K.; Shingo, I.; Morimoto, H. Stopped-flow Kinetic Study of Vitamin E Regeneration Reaction with Biological Hydroquinones (Reduced Forms of Ubiquinone, Vitamin K and Tocopherolquinone) in Solution. Biol. Chem. 1992, 267, 22277–22281. [Google Scholar]
- Vervoort, L.M.T.; Ronden, J.E.; Thijssen, H.H.W. The Potent Antioxidant Activity of the Vitamin K Cycle in Microsomal Lipid Peroxidation. Biochem. Pharmacol. 1997, 54, 871–876. [Google Scholar] [CrossRef]
- Westhofen, P.; Watzka, M.; Marinova, M.; Hass, M.; Kirfel, G.; Mu, J.; Bevans, C.G.; Mu, C.R.; Oldenburg, J. Human Vitamin K 2,3-Epoxide Reductase Complex Subunit 1-like 1 (VKORC1L1) Mediates Vitamin K-dependent Intracellular Antioxidant Function. Biol. Chem. 2011, 286, 15085–15094. [Google Scholar] [CrossRef]
- Li, J.; Lin, J.C.; Wang, H.; Peterson, J.W.; Furie, B.C.; Furie, B.; Booth, S.L.; Volpe, J.J.; Rosenberg, P.A. Novel Role of Vitamin K in Preventing Oxidative Injury to Developing Oligodendrocytes and Neurons. J. Neurosci. 2003, 23, 5816–5826. [Google Scholar] [CrossRef]
- Li, J.; Wang, H.; Rosenberg, P.A. Vitamin K Prevents Oxidative Cell Death by Inhibiting Activation of 12-Lipoxygenase in Developing Oligodendrocytes. J. Neurosci. Res. 2009, 87, 1997–2005. [Google Scholar] [CrossRef]
- Ambrożewicz, E.; Muszyńska, M.; Tokajuk, G.; Grynkiewicz, G.; Žarković, N.; Skrzydlewska, E. Beneficial Effects of Vitamins K and D3 on Redox Balance of Human Osteoblasts Cultured with Hydroxypatite-Based Biomaterials. Cells 2019, 8, 325. [Google Scholar] [CrossRef]
- Cutler, R.G.; Kelly, J.; Storie, K.; Pedersen, W.A.; Tammara, A.; Hatanpaa, K.; Troncoso, J.C.; Mattson, M.P. Involvement of oxidative stress-induced abnormalities in ceramide and cholesterol metabolism in brain aging and Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2004, 101, 2070–2075. [Google Scholar] [CrossRef]
- Jana, A.; Hogan, E.L.; Pahan, K. Ceramide and neurodegeneration: Susceptibility of neurons and oligodendrocytes to cell damage and death. J. Neurol. Sci. 2009, 278, 5–15. [Google Scholar] [CrossRef]
- De Chaves, E.P.; Sipione, S. Sphingolipids and gangliosides of the nervous system in membrane function and dysfunction. FEBS Lett. 2010, 584, 1748–1759. [Google Scholar] [CrossRef]
- Lev, M.; Milford, A. The 3-Ketodihydrosphingosine melaninogenicus: Synthetase of Bacteroides melaninogenicus: Induction by Vitamin K. Arch. Biochem. Biophys. 1973, 157, 500–508. [Google Scholar] [CrossRef]
- Sundaram, K.S.; Lev, M. Regulation of sulfotransferase activity by vitamin k in mouse brain. Arch. Biochem. Biophys. 1990, 277, 109–113. [Google Scholar] [CrossRef]
- Sundaram, K.S.; Lev, M. Warfarin administration reduces synthesis of sulfatides and other sphingolipids in mouse brain. J. Lipid Res. 1988, 29, 1475–1479. [Google Scholar] [PubMed]
- Thijssen, H.; Drittij-Reijnders, M. Vitamin K status in human tissues: Tissue-specific accumulation of phylloquinone and menaquinone-4. Br. J. Nutr. 1996, 75, 121–127. [Google Scholar] [CrossRef]
- Carrie, I.; Portoukalian, J.; Vicaretti, R.; Rochford, J.; Potvin, S.; Ferland, G. Menaquinone-4 Concentration Is Correlated with Sphingolipid Concentrations in Rat Brain. J. Nutr. 2004, 134, 167–172. [Google Scholar] [CrossRef]
- Sundaram, K.S.; Engelke, J.A.; Foley, A.L.; Suttie, J.; Lev, M. Vitamin K Status Influences Brain Sulfatide Metabolism in Young Mice and Rats. J. Nutr. 1996, 126, 2746–2751. [Google Scholar]
- Okano, T.; Shimomura, Y.; Yamane, M.; Suhara, Y.; Kamao, M.; Sugiura, M.; Nakagawa, K. Conversion of Phylloquinone (Vitamin K1) into Menaquinone-4 (Vitamin K2) in Mice. Two Possible Routes for Menaquinone-4 Accumulation in Cerebra of Mice. J. Biol. Chem. 2008, 25, 11270–11279. [Google Scholar] [CrossRef]
- Presse, N.; Shatenstein, B.; Kergoat, M.J.; Ferland, G. Low Vitamin K Intakes in Community-Dwelling Elders at an Early Stage of Alzheimer’s Disease. J. Am. Diet. Assoc. 2008, 108, 2095–2099. [Google Scholar] [CrossRef]
- Presse, N.; Belleville, S.; Gaudreau, P.; Greenwood, C.E.; Kergoat, M.; Morais, J.A.; Payette, H.; Shatenstein, B.; Ferland, G. Vitamin K status and cognitive function in healthy older adults. Neurobiol. Aging 2013, 34, 2777–2783. [Google Scholar] [CrossRef]
- Xv, F.; Chen, J.; Duan, L.; Li, S. Research progress on the anticancer effects of vitamin K2. Oncol. Lett. 2018, 15, 8926–8934. [Google Scholar] [CrossRef]
- Hitomi, M.; Yokoyama, F.; Nonomura, T.; Masaki, T.; Yosiji, H.; Inoeu, H.; Kinekawa, F.; Kurokohchi, K.; Uchida, N.; Watanabe, S.; et al. Antitumor effects of vitamins K1, K2 and K3 on hepatocellular carcinoma in vitro and in vivo. Int. J. Oncol. 2005, 26, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Dasari, S.; Ali, S.M.; Zheng, G.; Chen, A.; Dontaraju, V.S.; Bosland, M.C.; Kajdacsy-balla, A.; Gnanasekar, M. Vitamin K and its analogs: Potential avenues for prostate cancer management. Oncotarget 2017, 8, 57782–57799. [Google Scholar] [CrossRef] [PubMed]
- Schurgers, L.J.; Vermeer, C. Determination of Phylloquinone and Menaquinones in Food. Effect of Food Matrix on Circulating Vitamin K Concentrations. Haemostasis 2000, 30, 298–307. [Google Scholar] [PubMed]
- Schurgers, L.J.; Teunissen, K.J.F.; Hamulya, K.; Knapen, M.H.J.; Vik, H.; Vermeer, C. Vitamin K- containing dietary supplements: Comparison of synthetic vitamin K1 and natto-derived menaquinone-7. Blood 2007, 109, 3279–3283. [Google Scholar] [CrossRef] [PubMed]
- Schurgers, L.J.; Vermeer, C. Differential lipoprotein transport pathways of K-vitamins in healthy subjects. Biochim. Biophys. Acta 2002, 1570, 27–32. [Google Scholar] [CrossRef]
- Hirota, Y.; Tsugawa, N.; Nakagawa, K.; Suhara, Y.; Tanaka, K.; Uchino, Y.; Takeuchi, A.; Sawada, N.; Kamao, M.; Wada, A.; et al. Menadione (Vitamin K3) Is a Catabolic Product of Oral Phylloquinone (Vitamin K1) in the Intestine and a Circulating Precursor of Tissue Menaquinone-4 (Vitamin K2) in Rats. J. Biol. Chem. 2013, 288, 33071–33080. [Google Scholar] [CrossRef]
- Tsugawa, N.; Shiraki, M.; Suhara, Y.; Kamao, M.; Tanaka, K.; Okano, T. Vitamin K status of healthy Japanese women: Age-related vitamin K requirement for gamma-carboxylation of osteocalcin. Am. J. Clin. Nutr. 2006, 83, 380–386. [Google Scholar] [CrossRef]
- Geleijnse, J.M.; Vermeer, C.; Grobbee, D.E.; Schurgers, L.J.; Knapen, M.H.J.; Van Der Meer, I.M.; Hofman, A.; Witteman, J.C.M. Dietary Intake of Menaquinone Is Associated with a Reduced Risk of Coronary Heart Disease: The Rotterdam Study. J. Nutr. 2004, 134, 3100–3105. [Google Scholar] [CrossRef]
- Gast, G.C.; de Roos, N.M.; Sluijs, I.; Bots, M.L.; Beulens, J.W.; Geleijnse, J.M.; Witteman, J.C.; Grobbee, D.E.; Peeters, P.H.; van der Schouw, Y.T. A high menaquinone intake reduces the incidence of coronary heart disease. Nutr. Metab. Cardiovasc. Dis. 2009, 19, 504–510. [Google Scholar] [CrossRef]
- Shea, M.K.; Booth, S.L.; Miller, M.E.; Burke, G.; Chen, H.; Cushman, M.; Tracy, R.P.; Kritchevsky, S.B. Associations between circulating vitamin K1 and coronary calcium progression in community-dwelling adults: the Multi-Etcnic Study of Atherosclerosis. Am. J. Clin. Nutr. 2013, 98, 197–208. [Google Scholar] [CrossRef]
- Usui, Y.; Tanimura, H.; Nishimura, N.; Kobayashi, N.; Okanou, T.; Zawa, K. Vitamin K concentrations in the plasma and liver of surgical patients. Am. J. Clin. Nutr. 1990, 51, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Karl, J.P.; Booth, S.L.; Boyaval, P. Menaquinones, Bacteria, and the Food Supply: The Relevance of Dairy and Fermented Food Products to Vitamin K Requirements. Adv. Nutr. 2013, 4, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Shearer, M.J.; Bolton-Smith, C. The UK food data-base for vitamin K and why we need it. Food Chem. 2000, 68, 213–218. [Google Scholar] [CrossRef]
- Bolton-smith, C.; Price, R.J.G.; Fenton, S.T.; Harrington, D.J.; Shearer, M.J. Compilation of a provisional UK database for the phylloquinone (vitamin K1) content of foods. Br. J. Nutr. 2000, 83, 389–399. [Google Scholar] [PubMed]
- Ben-shem, A.; Frolow, F.; Nelson, N. Crystal structure of plant photosystem I. Nature 2003, 426, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Gross, J.; Cho, W.K.; Lezhneva, L.; Falk, J.; Krupinska, K.; Shinozaki, K.; Seki, M.; Herrmann, R.G.; Meurer, J. A Plant Locus Essential for Phylloquinone (Vitamin K1) Biosynthesis Originated from a Fusion of Four Eubacterial Genes. J. Biol. Chem. 2006, 281, 17189–17196. [Google Scholar] [CrossRef]
- Piironen, V.; Koivu, T.; Tammisalo, O.; Mattila, P. Determination of phylloquinone in oils, margarines and butter by high-performance liquid chromatography with electrochemical detection. Food Chem. 1997, 59, 473–480. [Google Scholar] [CrossRef]
- Peterson, J.W.; Muzzey, K.L.; Haytowitz, D.; Exler, J.; Lemar, L.; Booth, S.L. Phylloquinone (vitamin K1) and Dihydrophylloquinone Content of Fats and Oils. JAOCS 2002, 79, 641–646. [Google Scholar] [CrossRef]
- Elder, S.J.; Haytowitz, D.B.; Howe, J.; Peterson, J.W.; Booth, S.L. Vitamin K Contents of Meat, Dairy, and Fast Food in the U.S. Diet. J. Agric. Food Chem. 2006, 54, 463–467. [Google Scholar] [CrossRef]
- Kamao, M.; Yoshitomo, S.; Tsuwaga, N.; Uwano, M.; Yamaguchi, N.; Uenishi, K.; Ishida, H.; Sasaki, S.; Okano, T. Vitamin K Content of Foods and Dietary Vitamin K Intake in Japanese Young Women. J. Nutr. Sci. Vitaminol. 2007, 53, 464–470. [Google Scholar] [CrossRef]
- Bito, T.; Teng, F.; Watanabe, F. Bioactive Compounds of Edible Purple Laver Porphyra sp. (Nori). J. Agric. Food Chem. Agric. Food Chem. 2017, 65, 10685–10692. [Google Scholar] [CrossRef] [PubMed]
- Suttie, J. The importance of menaquinones in human nutrition. Ann. Rev. Nutr. 1995, 15, 399–417. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.D.; Jones, D. Distribution of isoprenoid quinone structural types in bacteria and their taxonomic implications. Microbiol. Rev. 1981, 45, 316–354. [Google Scholar] [PubMed]
- Fernandez, F.; Collins, M.D. Vitamin K composition of anaerobic gut bacteria. FEMS Microbiol. Lett. 1987, 41, 175–180. [Google Scholar] [CrossRef]
- Conly, J.; Stein, K. Quantitative and qualitative measurements of K vitamins in human. Am. J. Gastroenterol. 1992, 87, 311–316. [Google Scholar]
- Conly, J.; Stein, K. The production of menaquinones (vitamin K2) by intestinal bacteria and their role in maintaining coagulation homeostasis. Prog. Food Nutr. Sci. 1992, 16, 307–343. [Google Scholar]
- Bourdichon, F.; Casaregola, S.; Farrokh, C.; Frisvad, J.C.; Gerds, M.L.; Hammes, W.P.; Harnett, J.; Huys, G.; Laulund, S.; Ouwehand, A.; et al. Food fermentations: Microorganisms with technological beneficial use. Int. J. Food Microbiol. 2012, 154, 87–97. [Google Scholar] [CrossRef]
- Morishita, T.; Tamura, N.; Makino, T.; Kudo, S. Production of Menaquinones by Lactic Acid Bacteria. J. Dairy Sci. 1999, 82, 1897–1903. [Google Scholar] [CrossRef]
- Manoury, E.; Jourdon, K.; Boyaval, P.; Fourcassié, P. Quantitative measurement of vitamin K2 (menaquinones) in various fermented dairy products using a reliable high-performance liquid chromatography method. J. Dairy Sci. 2013, 96, 1335–1346. [Google Scholar] [CrossRef]
- Vermeer, C.; Raes, J.; Van Hoofd, C.; Knapen, M.H.J.; Xanthoulea, S. Menaquinone Content of Cheese. Nutrients 2018, 10, 446. [Google Scholar] [CrossRef]
- Hojo, K.; Watanabe, R.; Mori, T.; Taketomo, N. Quantitative measurement of tetrahydromenaquinone-9 in cheese fermented by propionibacteria. J. Dairy Sci. 2007, 90, 4078–4083. [Google Scholar] [CrossRef] [PubMed]
- Kaneki, M.; Hedges, S.J.; Hosoi, T.; Fujiwara, S.; Lyons, A.; Crean, S.J.; Ishida, N.; Nakagawa, M.; Takechi, M.; Sano, Y.; et al. Japanese Fermented Soybean Food as the Major Determinant of the Large Geographic Difference in Circulating Levels of Vitamin K2: Possible Implications for Hip-Fracture Risk. Nutrition 2001, 4, 315–321. [Google Scholar] [CrossRef]
- Villa, J.K.; Diaz, M.A.; Pizziolo, V.R.; Martino, H.S. Effect of vitamin K in bone metabolism and vascular calcification: A review of mechanisms of action and evidences. Crit. Rev. Food Sci. Nutr. 2017, 57, 3959–3970. [Google Scholar] [CrossRef] [PubMed]
- Sakane, R.; Kimura, K.; Hirota, Y.; Ishizawa, M.; Takagi, Y.; Akimori, W.; Shigefumi, K.; Makoto, M.; Suhara, Y. Synthesis of novel vitamin K derivatives with alkylated phenyl groups introduced at the ω-terminal side chain and evaluation of their neural differentiation activities. Bioorg. Med. Chem. 2017, 27, 4881–4884. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Hirota, Y.; Kuwahara, S.; Takeuchi, A.; Tode, C.; Wada, A.; Osakabe, N.; Suhara, Y. Synthesis of Novel Synthetic Vitamin K Analogues Prepared by Introduction of a Heteroatom and a Phenyl Group That Induce Highly Selective Neuronal Differentiation of Neuronal Progenitor Cells. J. Med. Chem. 2017, 60, 2591–2596. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.; Kagechika, H. Medicinal Chemistry of Vitamin K Derivatives and Metabolites. Vital Health Wellbeing 2017, 239–255. [Google Scholar] [CrossRef]
- Isler, O.; Doebel, K.; Hoffman-La Roche Inc. Synthesis of Vitamin K1 Using Boron Trifluoride Catalysts. U.S. Patent 2,683,176, 6 July 1954. [Google Scholar]
- Fieser, L.F. Synthesis of Vitamin K1. J. Am. Chem. Soc. 1939, 61, 3467–3475. [Google Scholar] [CrossRef]
- Afanasjeva, J. Administration of Injectable Vitamin K Orally. Hosp. Pharm. 2017, 52, 645–649. [Google Scholar] [CrossRef]
- Daines, A.M.; Payne, R.J.; Humphries, M.E.; Abell, A.D. The Synthesis of Naturally Occurring Vitamin K and Vitamin K Analogues. Org. Chem. 2003, 7, 1–15. [Google Scholar] [CrossRef]
- Coman, S.M.; Parvulescu, V.I.; Wuttke, S.; Kemnitz, E. Synthesis of Vitamin K1 and K1-Chromanol by Friedel–Crafts Alkylation in Heterogeneous Catalysis. ChemCatChem 2010, 2, 92–97. [Google Scholar] [CrossRef]
- European Commission. Opinion on Vitamin K1 (Phytonadione); SCCP/1105/07; Scientific Committee on Consumer Safety: Brussels, Belgium, 2007; pp. 1–31. [Google Scholar]
- Tien, J.-H.; Pang, C.-Y.; Hsu, N.-H.; Sunny Pharmatec Inc. Method of Making Vitamin K1. U.S. Patent Application WO2016060670A1, 21 April 2016. [Google Scholar]
- EFSA FEEDAP Panel (EFSA Panel on Additives and Products or Substances used in Animal Feed). Scientific Opinion on the safety and efficacy of vitamin K3 (menadione sodium bisulphite and menadione nicotinamide bisulphite) as a feed additive for all animal species. EFSA J. 2014, 12, 3532. [Google Scholar]
- Tarento, T.D.C.; Mcclure, D.D.; Talbot, A.M.; Regtop, H.L.; Biffin, J.R.; Valtchev, P.; Dehghani, F.; Kavanagh, J.M.; Tarento, T.D.C.; Mcclure, D.D.; et al. A potential biotechnological process for the sustainable production of vitamin K1. Crit. Rev. Biotechnol. 2019, 39, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Snyder, C.D.; Rapoport, H. Synthesis of Menaquinones. J. Am. Chem. Soc. 1974, 96, 8046–8054. [Google Scholar] [CrossRef]
- Baj, A.; Wa, P.; Kutner, A.; Morzycki, J.W.; Witkowski, S. Convergent synthesis of menaquinone-7 (MK-7). Org. Process Res. Dev. 2016, 20, 1026–1033. [Google Scholar] [CrossRef]
- Suhara, Y.; Watanabe, M.; Motoyoshi, S.; Nakagawa, K.; Wada, A.; Takeda, K.; Takahashi, K.; Tokiwa, H.; Okano, T. Synthesis of new vitamin K analogues as steroid and xenobiotic receptor (SXR) agonists: Insights into the biological role of the side chain part of vitamin K. J. Med. Chem. 2011, 54, 4918–4922. [Google Scholar] [CrossRef] [PubMed]
- Suhara, Y.; Hanada, N.; Okitsu, T.; Sakai, M.; Watanabe, M.; Nakagawa, K.; Wada, A.; Takeda, K.; Takahashi, K.; Tokiwa, H.; et al. Structure-activity relationship of novel menaquinone-4 analogues: Modification of the side chain affects their biological activities. J. Med. Chem. 2012, 55, 1553–1558. [Google Scholar] [CrossRef]
- Vermeer, C.; van’t Hoofd, C.; Knapen, M.H.J.; Xanthoulea, S. Synthesis of 2-methyl-1,4-naphthoquinones with higher gamma-glutamyl carboxylase activity than MK-4 both in vitro and in vivo. Bioorg. Med. Chem. 2017, 27, 208–211. [Google Scholar] [CrossRef]
- Mahanama, R.; Berenjian, A.; Valtchev, P.; Talbot, A.; Biffin, R.; Regtop, H.; Dehghani, F.; Kavanagh, J.M. Enhanced production of menaquinone 7 via solid substrate fermentation from Bacillus subtilis. Int. J. Food Eng. 2011, 7. [Google Scholar] [CrossRef]
- Berenjian, A.; Mahanama, R.; Talbot, A.; Regtop, H.; Kavanagh, J.; Dehghani, F. Advances in menaquinone-7 production by bacillus subtilis natto: Fed-batch glycerol addition. Am. J. Biochem. Biotechnol. 2012, 8, 105–110. [Google Scholar]
- Singh, R.; Puri, A.; Panda, B.P. Development of menaquinone-7 enriched nutraceutical: Inside into medium engineering and process modeling. J. Food Sci. Technol. 2015, 52, 5212–5219. [Google Scholar] [CrossRef]
- Berenjian, A.; Mahanama, R.; Kavanagh, J.; Dehghani, F.; Berenjian, A.; Mahanama, R.; Kavanagh, J.; Dehghani, F. Critical Reviews in Biotechnology Vitamin K series: Current status and future prospects. Crit. Rev. Biotechnol. 2015, 35, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Schallmey, M.; Singh, A.; Ward, O.P. Developments in the use of Bacillus species for industrial production. Can. J. Microbiol. 2004, 50, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Yamada, Y.; Ohtani, Y.; Mitsui, N.; Murasawa, H.; Araki, S. Production of menaquinone (vitamin K2)-7 by Bacillus subtilis. J. Biosci. Bioeng. 2001, 91, 16–20. [Google Scholar] [CrossRef]
- Song, J.; Liu, H.; Wang, L.; Dai, J.; Liu, Y.; Liu, H.; Zhao, G.; Wang, P.; Zheng, Z. Enhanced Production of Vitamin K2 from Bacillus subtilis (natto) by Mutation and Optimization of the Fermentation Medium. Braz. Arch. Biol. Technol. 2014, 57, 606–612. [Google Scholar]
- Berenjian, A.; Chan, N.L.C.; Mahanama, R.; Talbot, A.; Regtop, H.; Kavanagh, J.; Dehghani, F. Effect of biofilm formation by Bacillus subtilis natto on menaquinone-7 biosynthesis. Mol. Biotechnol. 2013, 54, 371–378. [Google Scholar] [CrossRef]
- Sato, T.; Yamada, Y.; Ohtani, Y.; Mitsui, N.; Murasawa, H.; Araki, S. Efficient production of menaquinone (vitamin K2) by a menadione-resistant mutant of Bacillus subtilis. J. Ind. Microbiol. Biotechnol. 2001, 26, 115–120. [Google Scholar] [CrossRef]
- Tsukamoto, Y.; Kasai, M.; Kakuda, H. Construction of a Bacillus subtilis (natto) with High Productivity of Vitamin K2 (Menaquinone-7) by Analog Resistance. Biosci. Biotechnol. BioChem. 2001, 65, 2007–2015. [Google Scholar] [CrossRef]
- Nishito, Y.; Osana, Y.; Hachiya, T.; Popendorf, K.; Toyoda, A.; Fujiyama, A.; Itaya, M.; Sakakibara, Y. Whole genome assembly of a natto production strain Bacillus subtilis natto from very short read data. BMC Genom. 2010, 11, 243. [Google Scholar] [CrossRef]
- Ma, Y.; McClure, D.D.; Somerville, M.V.; Proschogo, N.W.; Dehghani, F.; Kavanagh, J.M.; Coleman, N.V. Metabolic Engineering of the MEP Pathway in Bacillus subtilis for Increased Biosynthesis of Menaquinone-7. ACS Synth. Biol. 2019, 8, 1620–1630. [Google Scholar] [CrossRef]
- Yang, S.; Cao, Y.; Sun, L.; Li, C.; Lin, X.; Cai, Z.; Zhang, G.; Song, H. Modular Pathway Engineering of Bacillus subtilis to Promote de Novo Biosynthesis of Menaquinone-7. ACS Synth. Biol. 2019, 8, 70–81. [Google Scholar] [CrossRef]
- Sanghvi, A.; Lo, Y. Present and potential industrial applications of macro- and microalgae. Recent Pat. Food Nutr. Agric. 2010, 2, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Collins, K.G.; Fitzgerald, G.F.; Stanton, C.; Ross, R.P. Looking beyond the terrestrial: The potential of seaweed derived bioactives to treat non-communicable diseases. Mar. Drugs 2016, 14, 60. [Google Scholar] [CrossRef] [PubMed]
- Romano, G.; Costantini, M.; Sansone, C.; Lauritano, C.; Ruocco, N.; Ianora, A. Marine microorganisms as a promising and sustainable source of bioactive molecules. Mar. Environ. Res. 2017, 128, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Stengel, D.B.; Connan, S. Marine Algae: A Source of Biomass for Biotechnological Applications. Methods Mol. Biol. 2015, 1308, 1–37. [Google Scholar] [PubMed]
- De Roeck-holtzhauer, Y.; Quere, I.; Claire, C. Vitamin analysis of five planktonic microalgae and one macroalga. J. Appl. Phycol. 1991, 3, 259–264. [Google Scholar] [CrossRef]
- Tarento, T.D.C.; Mcclure, D.D.; Vasiljevski, E.; Schindeler, A.; Dehghani, F.; Kavanagh, J.M. Microalgae as a source of vitamin K1. Algal Res. 2018, 36, 77–87. [Google Scholar] [CrossRef]
- Johnson, T.W.; Shen, G.; Zybailov, B.; Kolling, D.; Reategui, R.; Beauparlant, S.; Vassiliev, I.R.; Bryant, D.A.; Jones, A.D.; Golbeck, J.H.; et al. Recruitment of a foreign quinone into the A1 site of photosystem I. I. Genetic and physiological characterization of phylloquinone biosynthetic pathway mutants in Synechocystis sp. PCC 6803. J. Biol. Chem. 2000, 275, 8523–8530. [Google Scholar] [CrossRef]
- Mimuro, M.; Tsuchiya, T.; Inoue, H.; Sakuragi, Y.; Itoh, Y.; Gotoh, T.; Miyashita, H.; Bryant, D.A.; Kobayashi, M. The secondary electron acceptor of photosystem I in Gloeobacter violaceus PCC 7421 is menaquinone-4 that is synthesized by a unique but unknown pathway. FEBS Lett. 2005, 579, 3493–3496. [Google Scholar] [CrossRef]
- Sakuragi, Y.; Zybailov, B.; Shen, G.; Bryant, D.A.; Golbeck, J.H.; Diner, B.A.; Karygina, I.; Pushkar, Y.; Stehlik, D. Recruitment of a foreign quinone into the A1 site of photosystem I: Characterization of a menB rubA double deletion mutant in Synechococcus sp. PCC 7002 devoid of FX, FA, and FB and containing plastoquinone or exchanged 9,10-anthraquinone. J. Biol. Chem. 2005, 280, 12371–12381. [Google Scholar] [CrossRef]
- Ikeda, Y.; Komura, M.; Watanabe, M.; Minami, C.; Koike, H.; Itoh, S.; Kashino, Y.; Satoh, K. Photosystem I complexes associated with fucoxanthin-chlorophyll-binding proteins from a marine centric diatom, Chaetoceros gracilis. Biochim. Biophys. Acta 2008, 1777, 351–361. [Google Scholar] [CrossRef]
- Yoshida, E.; Nakamura, A.; Watanabe, T. Reversed-phase HPLC determination of chlorophyll a′ and naphthoquinones in photosystem I of red algae: Existence of two menaquinone-4 molecules in photosystem I of Cyanidium caldarium. Anal. Sci. 2003, 19, 1001–1005. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Koivu, T.J.; Piironen, V.I.; Henttonen, S.K.; Mattila, P.H. Determination of Phylloquinone in Vegetables, Fruits, and Berries by High-Performance Liquid Chromatography with Electrochemical Detection. J. Agric. Food Chem. Food Chem. 1997, 45, 4644–4649. [Google Scholar] [CrossRef]
- Booth, S.L.; Sadowski, J.A.; Penningtont, J.A.T. Phylloquinone (Vitamin K1) Content of Foods in the US. Food and Drug Administration’s Total Diet Study. J. Agric. Food Chem. Food Chem. 1995, 43, 1574–1579. [Google Scholar] [CrossRef]
- Salvaterra, T.; Green, D.S.; Crowe, T.P.; O’Gorman, E.J. Impacts of the invasive alga Sargassum muticum on ecosystem functioning and food web structure. Biol. Invasions 2013, 15, 2563–2576. [Google Scholar] [CrossRef]
- Epstein, G.; Smale, D.A. Undaria pinnatifida: A case study to highlight challenges in marine invasion ecology and management. Ecol. Evol. 2017, 7, 8624–8642. [Google Scholar] [CrossRef]
- von Kries, R.; Hachmeister, A.; Gobel, U. Oral mixed micellar vitamin K for prevention of late vitamin K deficiency bleeding. Arch. Dis Child. Fetal Neonatal Ed. 2003, 88, 109–112. [Google Scholar] [CrossRef]
- Hasselt, P.M.; Van Janssens, G.E.P.J.; Slot, T.K.; Van Der Ham, M.; Minderhoud, T.C.; Talelli, M.; Akkermans, L.M.; Rijcken, C.J.F.; Van Nostrum, C.F. The influence of bile acids on the oral bioavailability of vitamin K encapsulated in polymeric micelles. J. Control. Release 2009, 133, 161–168. [Google Scholar] [CrossRef]
- Leiro, V.; Moreno, P.; Sarmento, B.; Durão, J.; Gales, L.; Pêgo, A.; Barrias, C. 1-Design and preparation of biomimetic and bioinspired materials. In Bioinspired Materials for Medical Applications; Rodrigues, L., Mota, M., Eds.; Woodhead Publishing: Cambridge, UK, 2017; pp. 1–44. [Google Scholar]
- Sun, F.; Ye, C.; Thanki, K.; Leng, D.; Van Hasselt, P.M.; Hennink, W.E.; van Nostrum, C.F. Mixed micellar system stabilized with saponins for oral delivery of vitamin K. Colloids Surf. B Biointerfaces 2018, 170, 521–528. [Google Scholar] [CrossRef]
- Hamrick, H.J.; Gable, K.; Freeman, H.; Dunn, L.; Zimmerman, P.; Rusin, M.M.; Linthavong, R.; Wright, E.; Moss, A.; Skinner, C. Reasons for Refusal of Newborn Vitamin K Prophylaxis: Implications for Management and Education. Hosp. Pediatr. 2019, 6, 15–21. [Google Scholar] [CrossRef]
- Gomaa, Y.A.; Garland, M.J.; Mcinnes, F.; El-khordagui, L.K.; Wilson, C.; Donnelly, R.F. Laser-engineered dissolving microneedles for active transdermal delivery of nadroparin calcium. Eur. J. Pharm. Biopharm. 2012, 82, 299–307. [Google Scholar] [CrossRef]
- Quinn, H.L.; Bonham, L.; Hughes, C.M.; Donnelly, R.F. Design of a Dissolving Microneedle Platform for Transdermal Delivery of a Fixed-Dose Combination of Cardiovascular Drugs. J. Pharm. Sci. 2015, 104, 3490–3500. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S.P.; Koutsonanos, D.G.; Martin, P.; Lee, J.; Zarnitsyn, V.; Murthy, N.; Compans, R.W.; Skountzou, I.; Prausnitz, R. Dissolving Polymer Microneedle Patches for Influenza Vaccination. Nat. Med. 2010, 16, 915–920. [Google Scholar] [CrossRef] [PubMed]
- González-vázquez, P.; Larrañeta, E.; Mccrudden, M.T.C.; Jarrahian, C.; Rein-weston, A.; Quintanar-solares, M.; Zehrung, D.; Mccarthy, H.; Courtenay, A.J.; Donnelly, R.F. Transdermal delivery of gentamicin using dissolving microneedle arrays for potential treatment of neonatal sepsis. J. Control. Release 2017, 265, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Hutton, A.R.J.; Quinn, H.L.; Mccague, P.J.; Jarrahian, C.; Rein-weston, A.; Co, P.S.; Gerth-guyette, E.; Zehrung, D.; Larrañeta, E.; Donnelly, R.F. Transdermal delivery of vitamin K using dissolving microneedles for the prevention of vitamin K deficiency bleeding. Int. J. Pharm. 2018, 541, 56–63. [Google Scholar] [CrossRef]
- Kidd, P.M. Vitamins D and K as pleiotropic nutrients: Clinical importance to the skeletal and cardiovascular systems and preliminary evidence for synergy. Altern. Med. Rev. 2010, 15, 199–222. [Google Scholar]
- Van Ballegooijen, A.J.; Pilz, S.; Tomaschitz, A.; Grübler, M.R.; Verheyen, N. The Synergistic Interplay between Vitamins D and K for Bone and Cardiovascular Health: A Narrative Review. Int. J. Endocrinol. 2017, 2017, 7454376. [Google Scholar] [CrossRef]
- Viegas, C.; Araújo, N.; Marreiros, C.; Simes, D. The interplay between mineral metabolism, vascular calcification and inflammation in Chronic Kidney Disease (CKD): Challenging old concepts with new facts. Aging 2019, 11, 4274–4299. [Google Scholar] [CrossRef]
- Theuwissen, E.; Cranenburg, E.C.; Knapen, M.H.; Magdeleyns, E.J.; Teunissen, K.J.; Schurgers, L.J.; Smit, E.; Vermeer, C. Low-dose menaquinone-7 supplementation improved extra-hepatic vitamin K status, but had no effect on thrombin generation in healthy subjects. Br. J. Nutr. 2012, 108, 1652–1657. [Google Scholar] [CrossRef]
- Schulman, S.; Furie, B. How I treat poisoning with vitamin K antagonists. Blood 2015, 125, 438–442. [Google Scholar] [CrossRef]
- Spahr, J.E.; Maul, J.S.; Rodgers, G.M. Superwarfarin Poisoning: A Report of Two Cases and Review of the Literature. Am. J. Hematol. 2007, 82, 656–660. [Google Scholar] [CrossRef]
| Designation | Function | |
|---|---|---|
| Hepatic | Factor II (Prothrombin) | Pro-coagulant [7] |
| Factor VII | Pro-coagulant [7] | |
| Factor IX | Pro-coagulant [7] | |
| Factor X (Stuart Factor) | Pro-coagulant [7] | |
| Protein C | Anti-coagulant, anti-inflammatory, anti-apoptotic [7,8,9,10] | |
| Protein S | Co-factor for activated protein C, anti-coagulant, bone turnover, anti-inflammatory [7,8,11,12] | |
| Protein Z | Regulation of coagulation, anti-thrombotic [13,14] | |
| Extra Hepatic | OC | Negative regulator of bone formation, regulator of mineral maturation rate, mechanical stabilizer of bone matrix, regulator of glucose metabolism [15,16] |
| MGP | Inhibitor of soft tissue calcification, modulator of angiogenesis and tumorigenesis [17,18,19] | |
| Gas6 | Signal transduction, regulator of proliferation, migration, differentiation, adhesion, and apoptosis, anti-inflammatory, platelet activation, thrombus stabilization [20,21,22] | |
| GRP | Inhibitor of soft tissue calcification, inhibitor of mineral crystal maturation and growth in blood, anti-inflammatory [23,24,25,26] | |
| Periostin (isoforms 1–4) | Regulator of cell-matrix interactions, adhesion, proliferation, and differentiation processes, tissue remodelling and wound repair, angiogenesis [27,28,29] | |
| PRGP1/PRGP2 | Signal transduction [30,31] | |
| TGM3/TGM4 | Signal transduction [32] | |
| Endoplasmic Reticulum/Golgi Apparatus | GGCX | γ-carboxylation of VKDPs [33] |
| Phylum | Species Designation | Content (µg/g) | |
|---|---|---|---|
| Macroalgae | Ochrophyta | Undaria pinnatifida *1 | 12.9 [121] |
| Sargassum fusiforme *2 | 1.75 [121] | ||
| Sargassum muticum | 750 [169] | ||
| Rhodophyta | Porphyra sp. *3 | 26 [122,169] | |
| Microalgae | Bacillariophyta | Skeletonema costatum | 5.5 [169] |
| Chlorophyta | Tetraselmis suecica | 28 [169] | |
| Dunaliella salina | 0.1 [170] | ||
| Desmodesmus asymmetricus | 0.46 [170] | ||
| Chlorella vulgaris | 0.73 [170] | ||
| Cyanobacteria | Anabaena cylindrica | 200.25 [170] | |
| Spirulina sp. | 12.70 [170] | ||
| Haptophyta | Isochrysis galbana | 8 [169] | |
| Pavlova lutheri | 6.5 [169] | ||
| Ochrophyta | Nannochloropsis oculata | 0.17 [171] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simes, D.C.; Viegas, C.S.B.; Araújo, N.; Marreiros, C. Vitamin K as a Diet Supplement with Impact in Human Health: Current Evidence in Age-Related Diseases. Nutrients 2020, 12, 138. https://doi.org/10.3390/nu12010138
Simes DC, Viegas CSB, Araújo N, Marreiros C. Vitamin K as a Diet Supplement with Impact in Human Health: Current Evidence in Age-Related Diseases. Nutrients. 2020; 12(1):138. https://doi.org/10.3390/nu12010138
Chicago/Turabian StyleSimes, Dina C., Carla S. B. Viegas, Nuna Araújo, and Catarina Marreiros. 2020. "Vitamin K as a Diet Supplement with Impact in Human Health: Current Evidence in Age-Related Diseases" Nutrients 12, no. 1: 138. https://doi.org/10.3390/nu12010138
APA StyleSimes, D. C., Viegas, C. S. B., Araújo, N., & Marreiros, C. (2020). Vitamin K as a Diet Supplement with Impact in Human Health: Current Evidence in Age-Related Diseases. Nutrients, 12(1), 138. https://doi.org/10.3390/nu12010138






