Abstract
The present study explores the sustainable potential of volcanic ash sourced from the active Sakurajima volcano (Japan) as an eco-friendly alternative to Portland cement—a binder known for its high carbon emissions—in concrete and mortar production. The abundant pyroclastic material, currently a waste burden for the residents of Sakurajima and the Kagoshima Bay region, presents a unique opportunity for valorization in line with circular economy principles. Rather than treating this ash as a disposal problem, the research investigates its transformation into a valuable supplementary cementitious material (SCM), contributing to more sustainable construction practices. The investigation focused on the material characterization of the ash (including chemical composition, morphology, and PSD) and its pozzolanic activity index, which is a key indicator of its suitability as a cement replacement. Mortars were prepared with 25% of the commercial binder replaced by volcanic ash—both in its raw form and after mechanical activation—and tested for compressive strength after 28 and 90 days of water curing. Additional assessments included workability of the fresh mix (flow table test), apparent density, and flexural strength of the hardened composites. Tests results showed that the applied volcanic ash did not influence the workability of the mix and showed negligible effect on the apparent density (changes of up to 3.3%), although the mechanical strength was deteriorated (decrease by 15–33% after 7 days, and by 25–26% after 28 days). However, further investigation revealed that the simple mechanical grinding significantly enhances the pozzolanic reactivity of Sakurajima ash. The ground ash achieved a 28-day activity index of 81%, surpassing the 75% threshold set by EN 197-1 and EN 450-1 standards for type II mineral additives. These findings underscore the potential for producing low-carbon mortars and concretes using locally sourced volcanic ash, supporting both emissions reduction and sustainable resource management in construction.
1. Introduction and Research Significance
It is estimated that Portland clinker/cement production is responsible for 5–10% of the world’s CO2 emissions [1,2]. This is a huge amount considering that the latest report on worldwide cement production by Madhumitha Jaganmohan (10 March 2025) [3] stated that the global cement production in 2024 is estimated at 4.0 billion tons. Cement production in Asia has a significant share of this amount. According to the abovementioned report, China is the leading cement-producing country, with production reaching nearly 1.9 billion metric tons while India ranked a distant second, producing 450 million metric tons. As for European countries, among the largest cement producers in terms of revenue, the Swiss company Holcim also topped the manufactures list in 2024 [3]. Moreover, cement production is an extremely energy-intensive process. It begins with the extraction and crushing of raw materials, followed by grinding. The most energy-demanding stage involves roasting the mix at temperatures of approximately 1450–1500 °C to produce Portland clinker [4]. Afterward, gypsum is added, and the mix undergoes a final grinding phase to yield the finished cement product. According to estimates, producing one ton of cement requires approximately 1054 kWh of energy [5]. This makes cement production one of the leading causes of greenhouse gas emissions worldwide, and CO2 emissions from cement production have increased almost continuously for several decades, reaching 1.57 billion metric tons in 2023 [3]. Therefore, there is a strong demand to reduce the production and use of Portland clinker/cements on behalf of more environmentally responsible alternatives including the so-called low emission binders [5].
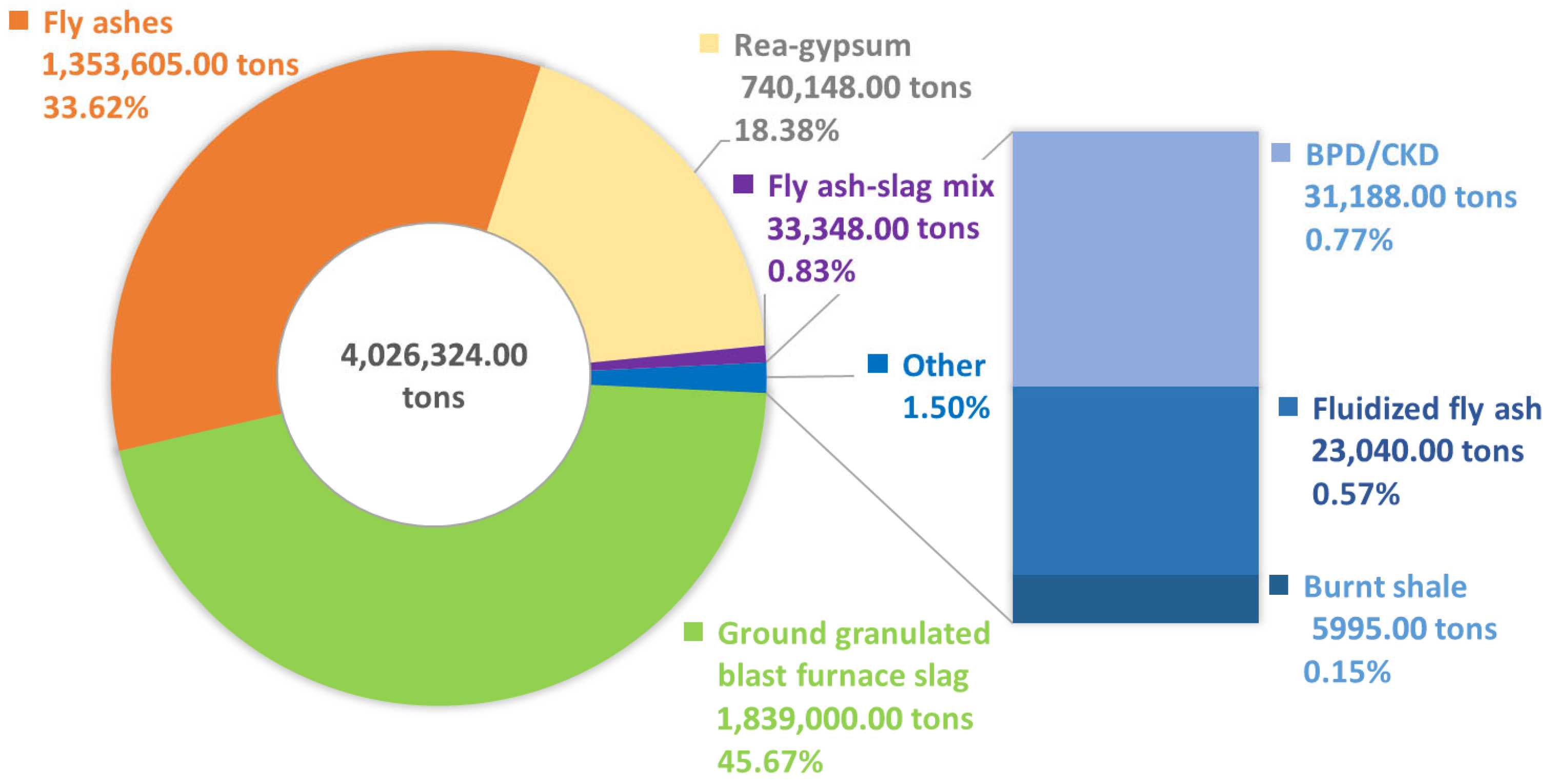
For several years now, there has been a clear decline in the production, and consequently also in the consumption, of Portland cement CEM I in favor of cements containing large shares of constituents other than Portland clinker—e.g., blast furnace cement CEM III or composite cements CEM V and CEM VI. For example, in Poland in 2021, over 4 million tons of secondary materials (including blast furnace slag, siliceous and fluidized fly ashes, by-pass dusts from dedusting cement plants installations, cement kiln dust, burnt shale, or synthetic rea-gypsum—a by-product of flue gas desulfurization in power plants) were used in cement production as the important constituents (for details, see Figure 1) [6].
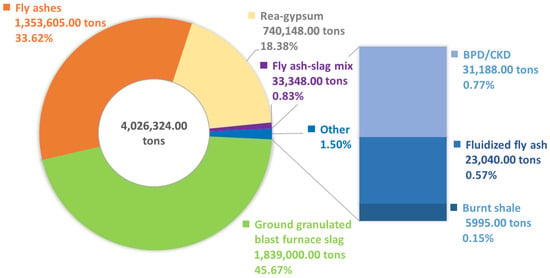
Figure 1.
Consumption of waste materials for cement production in Poland in 2021 (BPD—by-pass dust, CKD—cement kiln dust); chart elaborated using data from [6].
Recently, a widely adopted strategy for reducing emissions and the overall environmental—particularly carbon—footprint [7] of cement-based composites is the partial replacement of Portland cement with supplementary cementitious materials (SCMs) [8]. Ideally, these SCMs are sourced locally to further minimize environmental impact. Among the most commonly used SCMs are ground limestone, siliceous and calcareous fly ashes, and ground granulated blast furnace slag (GGBS) [8,9,10,11,12], all of which contribute to lowering the carbon intensity of concrete production while maintaining or enhancing performance.
While the use of ground limestone powder is environmentally justified in regions with abundant local limestone resources, transporting it over long distances significantly increases the carbon footprint, either of the binder with such a constituent (material carbon footprint) or the overall environmental impact of the final concrete element (product carbon footprint), undermining the sustainability benefits of using this component. In the case of GGBS, the by-product of steel smelting process, its quantities are limited because they result from the amount of steel produced. In some regions—such as Poland—this material is fully consumed in the production of pre-blended blast furnace cement (compare Figure 1), leaving little to no surplus for use as a supplementary cementitious material (SCM) added directly to concrete at the mixing stage [6]. Meanwhile, in the case of fly ash—particularly siliceous fly ash, a by-product of hard coal combustion in power plants or cogeneration facilities in colder regions—its availability is steadily declining. This is primarily due to the global phase-out of coal-fired power plants in favor of cleaner energy sources, such as nuclear power. Generally, due to the shift towards renewable energy, existing SCMs are losing their dominance [13], and it is reasonable to look for other materials that could be used in concrete as a co-binder or binder substitute.
The use of volcanic ash as a partial replacement for Portland cement presents a promising pathway toward the production of sustainable and durable cementitious materials, particularly in seismically active regions. This approach is especially relevant in areas with abundant volcanic activity, such as many Asian countries, the American state of Hawaii (located on the archipelago of 137 volcanic islands), and several countries in Europe [13,14,15], as utilizing locally available volcanic ash not only reduces the carbon footprint of construction materials but also supports regional circular economy initiatives. Some standards (e.g., ASTM C618 Standard [16]) consider the volcanic ash a natural pozzolan that can be used to produce cements and mortars and/or concrete where pozzolanic action is desired. However, it is necessary to determine the pozzolanic activity of the ashes from particular volcanoes, as some of them do not show significant activity. For instance, the pyroclastic materials derived from the active volcano Etna (Italy) shows lower ability to undergo pozzolanic reaction and are considered rather as a filler/aggregate component, rather than a potential SCM for concretes and cementitious mortars [15].
This research paper considers using a particular volcanic ash sourced from Sakurajima, the most active volcano of Japan located in the south of Kyushu Island, as an eco-friendly component in cementitious composites. So far, this material has not received much attention in the context of such an application. In 2013, experimental studies were carried out in Japan to use ashes from Sakurajima and Shinmoe-dake (other active volcano located in the southern Kyushu) as components of so-called water-retention interlocking concrete blocks. Up to 20% of Sakurajima ash and up to 30% of Shinmoe-dake ash was used to obtain materials of flexural strength of c.a. 3.0 MPa and increased water-retention capacity but worsened permeability compared to pure cement blocks [17]. There are also conference mentions from the late 1980s that consideration was given to hardening of the cement–lime–Sakurajima ash system [18], although the author did not have access to the manuscript of that publication. In general, the lack of information on the use of volcanic ash from Sakurajima in concrete technology is surprising in the context of similar studies devoted to pyroclastic materials from other Japanese volcanoes, e.g., volcanic ash from abovementioned active volcano of Shinmoe-dake [19]. There are though few studies on using the natural pozzolan shirasu, the deposit of pyroclastic flows, as a partial fine aggregate replacement [20,21,22]. Also, construction objects such as retaining walls and shelter made using local pyroclastic material as the aggregate are present on the dormant volcano Mount Fuji’s slope (Figure 2). Therefore, the research results obtained by the author seem all the more unique and may constitute valuable materials constituting the foundation for future research in the context of sustainable building materials in Japan.

Figure 2.
Concrete retaining walls and shelter on the slope of volcano made using local pyroclastic material, Mount Fuji, Yamanashi, Japan (author’s own photos).
The paper presents an empirical trial of using volcanic ash from Sakurajima as a partial substitute of Portland cement in cementitious mortars. The study investigates the pozzolanic activity of volcanic ash and the effect of the volcanic ash on the rheological and mechanical properties of composites made with volcanic ash and ordinary Portland cement (OPC). The activity index is usually determined for ashes that are the by-products of coal and, more recently, biomass combustion, for ground granulated blast furnace slag, or for other materials recognized as valuable cement constituents and concrete mineral additives. It is rarely performed for volcanic ashes. However, determination of this parameter seemed crucial to the author, as various volcanoes present very different levels of pozzolanic activity like, for instance, the abovementioned quite active Tajogaite’s ash (index adopted values from the range 59.6–87.7% [13]) or practically inactive Etna’s ash [15].
The tested ash was applied into the mixes in two variants: in its raw form (only pre-fractionated) or after its mechanical activation. The level of substitution of OPC with volcanic ash was 25% (wt.) and resulted from the test procedure used to determine the ash pozzolanic activity coefficient described in the EN 450-1 European Standard [23]. Each time, the consistency of the fresh mix as well as the apparent density, flexural strength, and compressive strength of the hardened composites were tested. The mechanical tests were carried out after 28 and 90 days, which made it possible to determine the pozzolanic activity index of applied volcanic ash. The obtained results indicate that volcanic ash from Sakurajima is a promising low-emission alternative to Portland cement, offering a locally sourced solution for producing sustainable building materials. Its use not only contributes to reducing the carbon footprint of cementitious composites but also provides an environmentally responsible method for managing and repurposing volcanic ash waste.
2. Volcanic Ash in Cementitious Composites Technology
2.1. About Volcanic Ash and Piroclastic Materials
Volcanic ash is a type of pyroclastic material, specifically a type of tephra, i.e., material formed during a volcanic eruption. According to Thorarinsson [24], tephra includes various materials, regardless of composition, size, or emplacement mechanism. Volcanic ash, however, consists of particles not exceeding 2 mm (0.079 inches) [25], and these are fragments of rocks (so-called lithic), crystals, or volcanic glass generated when solid rock shatters and magma separates during explosive volcanic activity [26].
Rose and Durant [25], after Bonadonna et al. [27], also distinguished so-called “fine ash” that includes ash particles with diameters not exceeding 1000 μm (1 mm), which fall in the intermediate flow, and “very fine ash” that includes particles with diameters up to 30 μm, which settle down according to Stokes Law in the laminar flow regime. Volcanic ash particles are generally not spherical, which complicates and further slows their fallout [28] (after [26]). But researchers often assume the spherical shape of ash particles to model their behavior and suspension time in the atmosphere. For simplification, it was established that fine ash particles’ predicted atmospheric residence time is 30 min and in the case of very fine ash particles—c.a. 3 h. In reality, the mass fractions of some volcanic ash with particle diameters smaller than 10 μm presents residence times of even 10 days [25] and can be carried away over long distances and then fading as a dust-like layer across the landscape (so-called “ash fall”).
2.2. Size and Chemical Composition of Selected Volcanic Ashes
The size of volcanic ash particles falling to the ground decreases with distance from the volcano. This is why the grading of volcanic ashes can vary widely [26]. For instance, the volcanic material collected after eruption of Etna (Sicily, Italy) contained over 70% of volcanic ash (i.e., fractions up to 2 mm), of which 57% were particles smaller than 1 mm (fine ash) and 24% were particles up to 0.5 mm [14]. In case of materials collected after the activity of Tajogaite monogenetic volcano (La Palma, Canary Islands), the volcanic ash content was over 90%, of which not less than 65% (depending on the place of collecting) were particles not exceeding 1 mm (i.e., fine ash) [13]. Meanwhile, in case of materials collected after Mount Tavurvu’s stratovolcano eruption (New Britain, Papua New Guinea), the PSD analysis showed that 42% of the ash was “sandy fractions” and the remaining 42% was silty fraction not exceeding 75 μm in size [29]. Therefore, when considering using volcanic ash as a substitute for Portland cement, it may be necessary to fractionate or even grind the material before adding it to the mix. This is also what researchers performed in the abovementioned studies.
The second important issue that should be considered in the context of using volcanic ash as a binder substitute (and not as a filler or aggregate as in [14,30]) is the chemical composition, which partially determines the pozzolanic activity of the ash, i.e., its ability to undergo a pozzolanic reaction during the setting of mineral paste or mortar or concrete. Table 1 contains a summary of the chemical compositions of the selected volcanic ashes collected from not only the already mentioned Mount Etna [24], Mount Tavurvu, and Tajogaite, but also from Mount Bromo [31] and Mount Kelud [32] in Malaysia and from two Pakistani provinces of Cherat and Khyber [33].

Table 1.
Oxide compositions (wt. %) of volcanic ashes collected in Italy, Canary Islands, Papua New Guinea, Malaysia, and Pakistan (on the basis of [13,30,31,32,33]).
Since the volcanic ashes are formed during eruptive processes (including sudden cooling of the magma), they are usually expected to be composed mostly of reactive aluminosilicates [34]. The cited research studies showed that regardless of the volcano’s location, most of the analyzed ashes (excluding Etna’s ash) characterized by a high content of silicon, aluminum, and iron compounds that, when converted into oxides, gave a total value of over 70.0% of the composition, which enables us to conclude that they meet the requirements of the ASTM C618 standard for N-class ash. Therefore, the high content of these compounds was positive in the context of the pozzolanic reaction and the development of strength of the cementitious matrix made with such ashes.
2.3. Pozzolanic Reaction in Cementitious Composites
The concept of using Portland cement as the binder in cementitious composites is generally based on the hydration of cement compounds, mainly hydration of calcium silicates (alite, C3S and belite, C2S) that, first of all, leads to the formation of the calcium silicate hydrates gel (C-S-H), which, after hardening, is the main contributor to the mechanical strength of the cementitious matrix and, secondly, leads to the formation of calcium hydroxide (portlandite, CH) considered a by-product of the reaction [35,36]. If additional pozzolanic materials (e.g., SCMs—fly ash and GGBS, silica fume, metakaolin, or volcanic ash) are added to the mix, they do not react directly with water, but the reactive silicates and aluminosilicates present in them react with calcium hydroxide (product of hydration of calcium silicates from Portland clinker, acting here as an activator), producing more hydrates, such as calcium aluminosilicate hydrates gel (C-A-S-H), densifying the matrix that additionally strengthen the composite. Such reaction is known as the pozzolanic reaction and can be written as follows [34]:
or using abbreviated formulas:
xCa(OH)2 + yAl2O3·zSiO2 + (n − x)H2O → (CaO)x·(Al2O3)y·(SiO2)z·(H2O)n−x,
(CH)x + AySz + H(n−x) → CxAySzH(n−x).
The term “pozzolanic” is generally explained as “consisting of, containing or of the nature of pozzolana” or “having properties similar to those of pozzolana” [37]. It is derived from one of the primary deposits of natural pozzolana, i.e., pyroclastic materials ejected by the Vesuvius used by the ancient Romans in Pozzuoli (Italy). The modern definition of “pozzolana” covers any volcanic origin material (pumice or ash), predominantly composed of fine volcanic glass. The term “pozzolan”, however, applies to all inorganic materials, regardless of their origin, that have the ability to participate in the pozzolanic reaction. The EN 197-1 European Standard [38] distinguishes natural pozzolans (P) and artificial pozzolans (Q), i.e., calcined clay raw materials. However, it should be underlined that not all pyroclastic materials resulting from volcanic eruptions have high pozzolanic activity. For instance, as mentioned before, Mount Etna’s ash presents low or no pozzolanic activity [30].
On the other hand, high content of magnesium compounds (like in case of Tajogaite ash—see Table 1) may lead to production of magnesium silicate hydrates (M-S-H) and decalcification of C-S-H and C-A-S-H gels. As a result, an increase in porosity and permeability of the matrix, and thus lower mechanical strength of the hardened composites, can be observed [35,39].
2.4. Alkalis Content in Volcanic Ashes
It is also worth noting that some of the volcanic ashes contain high amounts of alkalis, i.e., sodium and potassium oxides and hydroxides often recalculated to sodium oxide equivalent (Na2Oeq), which is equal to sum of Na2O and K2O multiplied by 0.658 (coefficient based on the molar masses). High alkali content in cementitious composites is not desirable because they may lead to alkali-aggregate reactions, including alkali–silica reaction (ASR) or alkali–carbonate reaction (ACR) between particular aggregates (e.g., opal, chalcedony, cryptocrystalline quartz [40]) and sodium and potassium hydroxides from cement or mineral additives. As a result, swelling products are formed. In extreme cases, it may lead to the destruction of the composite where changes in the microstructure are reflected by the macroscopic effect [41]. However, Owsiak [40] states that calcium hydroxide plays a significant role in the reaction of alkalis with aggregates because it affects the composition of the sodium–potassium–calcium silicate gel, its viscosity, and the ability to expand. Thus, reducing the Ca(OH)2 content through the pozzolanic reaction should result in a decrease in expansion. Thomas [42] even states that there is a limit in calcium hydroxide content, below which expansion does not occur.
There are no clear recommendations on how little alkalis should be in concrete. According to Owsiak [40], the alkali content in most of the industrial Portland cements is in the range of 0.5–1.3%. Owsiak recalls Stanton’s early work [43], which shows that ASR expansion is unlikely if the Na2Oeq content in cement does not exceed 0.6%, but at the same time, she points out that such an approach does not take into consideration the cement content and type (especially when cement is containing fly ash or blast furnace slag), alkalis soluble in water, or alkalis from the additives and admixtures.
The analyzed volcanic ashes (Table 1) are characterized by Na2Oeq content in the range of 3.9–5.4%. When applying these data to the composition of typical SCMs, it is worth noting that these are higher values than in the case of blast furnace slag (Na2Oeq between 0.3 and 2.6%) but lower than in case of fly ashes (Na2Oeq between 7.0 and 7.8%) [40].
3. Sakurajima Volcano—History and Genesis of Research
The Sakurajima (jap. 桜島) is an active stratovolcano—formerly an island (Figure 3a) and now a peninsula (Figure 3b)—located in the north of Kagoshima Bay, in southern Kyushu, Japan. The lava flows of the 1914 eruption (the largest eruption in twentieth-century Japan) connected the former island with the Ōsumi Peninsula [44]. Sakurajima is the most active volcano in Japan—the summit has three peaks: Kita-dake (northern peak), Naka-dake (central peak) and the currently active Minami-dake (southern peak) [45].
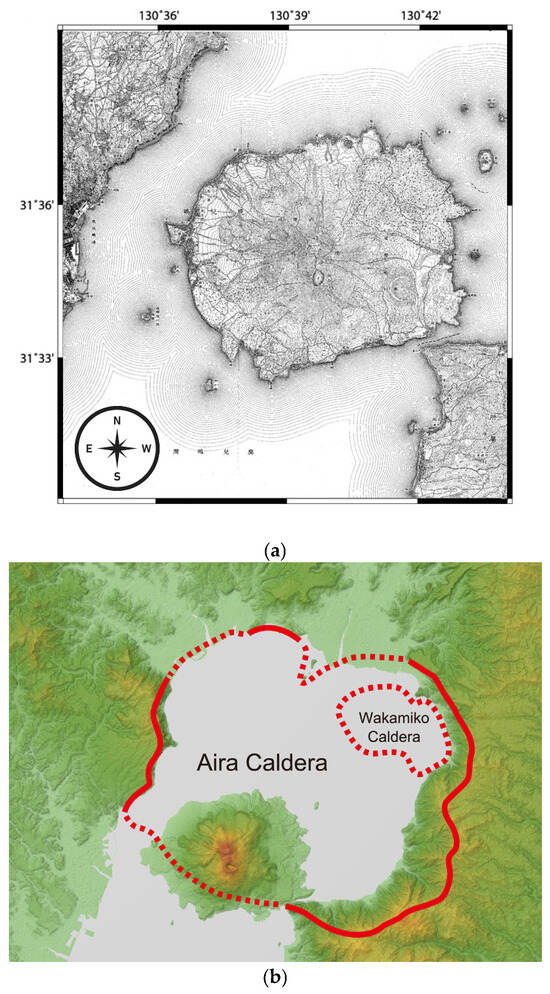
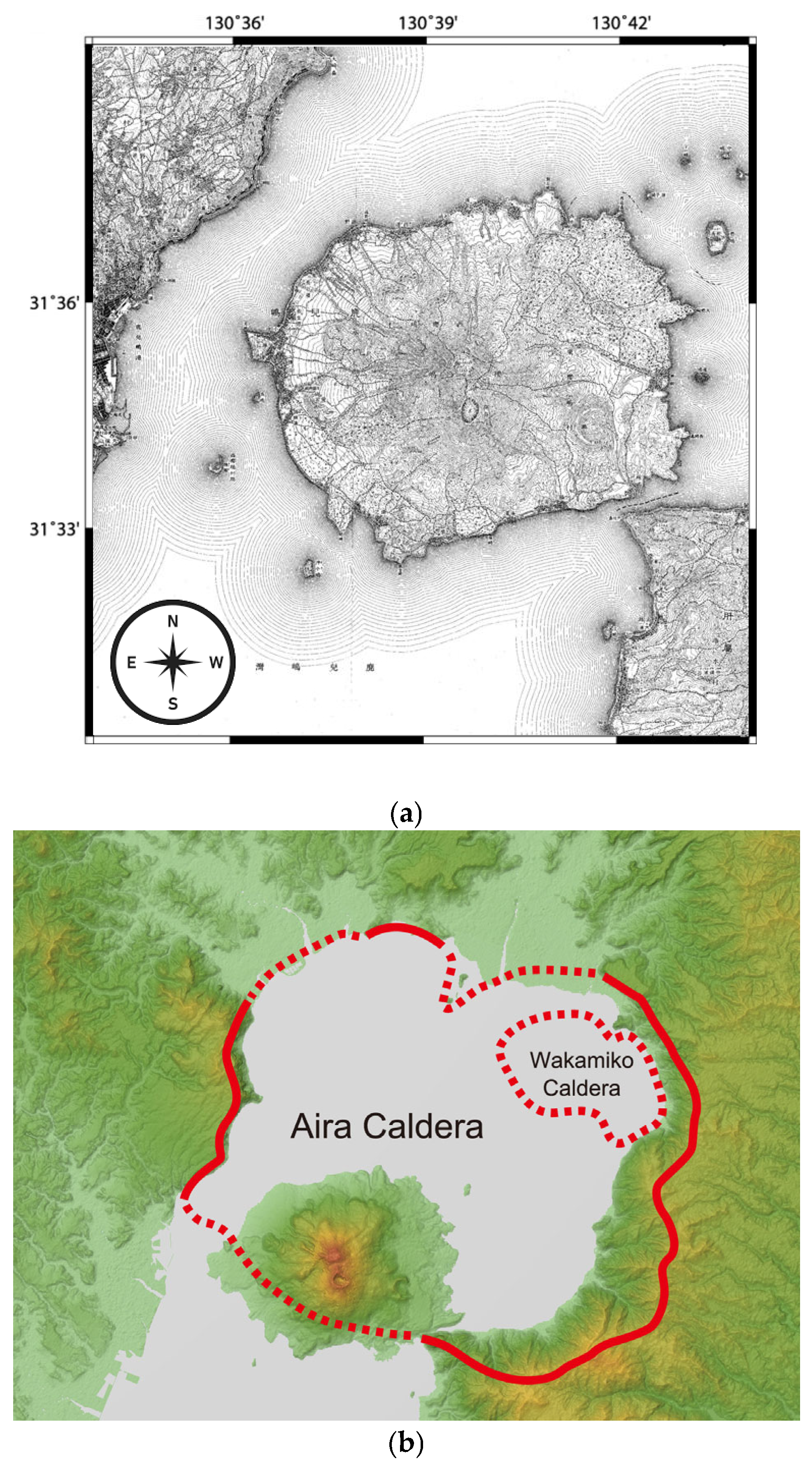
Figure 3.
Situation of the Sakurajima volcano in Kagoshima Bay: (a) A 1909 map of Sakurajima by the Imperial Japanese Land Survey showing Sakurajima as an island before connecting it to the Ōsumi Peninsula after the 1914 eruption [44]; (b) current topographic map with the calderas of the Aira and Wakamiko submerged historical volcanoes marked in red solid and red dotted lines (NASA, public domain [46]).
Sakurajima is located within the other large, at over 23 km across, Aira caldera (Figure 3b) formed from an enormous eruption dating over 22,000 [47] or even over 25,000 years ago (according to tephra 14C dating [48]), which caused tephra to fall as far as 1400 km from the volcano [48] (after [49]). Sakurajima is a post-caldera volcano, formed by later activity within the Aira caldera about 13,000 years ago and is now an active vent of the Aira [47].
The first confirmed eruptions of Sakurajima date back to the end of the first century. Most of the eruptions were of a Strombolian-type (like in Figure 4 and in Video S1 in the Supplementary Materials), but large eruptions of a Plinian-type occurred between 1471 and 1476, 1779 and 1782, and in 1914 [50]. The volcano’s activity surged in 1955, marking the beginning of nearly continuous eruptions. The last Holocene eruptive period began on 25 March 2017, and is still ongoing, and the last three major eruptions included the following [51]:

Figure 4.
The views of the repeatable eruptions and ash ejections from the Sakurajima crater: (a) The view from Kagoshima Port (Kyushu Island) across the Kagoshima Bay; (b) the view of southern Minami-dake crater from Sakurajima Island (author’s own photographs).
On 9 February 2023—Showa crater eruption: plumes rose to an altitude of 1 km, and within a 2 km radius; falling rocks and pyroclastic flows observed.
On 14 February 2024—Minami-dake crater eruption: plumes emitted at over 5 km in height and spewing rocks as far as 1.3 km away.
Between 6 June and 16 June 2025—ongoing eruptive activity at Minami-dake; alert remained at level 3 (on a 5-level scale); the caution area within 2 km of Minima-dake and Showa craters.
Apart from the larger eruptions, thousands of small explosions happen every year as Sakurajima is one of the very few volcanoes that are now showcasing persistent activity. Sakurajima’s ongoing typical activity ranges from a Strombolian-type eruption to large ash explosions every 4–24 h. Each time, the ash is ejected up to a few kilometers above the volcano summit and through the Kagoshima Bay (with a width of c.a. 4.5 km) to the city of Kagoshima and its suburbs, becoming burdensome waste that pollutes the city and impedes its functioning (Figure 5a,b). Residents must regularly collect ash falling on the city and deposit it in designated spots (Figure 5c,d). Aa a consequence, special services collect hundreds of bags marked as ash (jap. kai, 灰, Figure 5c) wastes.

Figure 5.
Sakurajima’s ash in the Kagoshima city located on the other side of Kagoshima Bay (Kagoshima, Japan): (a,b) the view of ash on the pavement; (c,d) the ash waste (“灰”) collected by residents and deposited in the designated place for collection by a dedicated city service of Kagoshima City Office (Kagoshima, Japan) (author’s own photos).
Taking into consideration that Sakurajima’s ash is the burdensome waste that needs to be constantly collected and carefully stored, but at the same time, a natural pozzolanic material, it is worth testing its pozzolanic activity in the context of its potential use in cementitious materials incorporated locally into elements of structures and infrastructure.
4. Materials and Methods
4.1. Qualitative and Qualitative Composition of Tested Composites
The subjects of the research were cementitious mortars unmodified and modified with volcanic ash (from Sakurajima volcano), which was a partial substitute for cement binder. The components included the following:
- CEM I 42.5R, high-early-strength Portland cement (Cementownia Ożarów, Ożarów, Poland) conforming to the requirements of the European standards EN 197-1:2011 [38] and EN 196-1:2016 [52] (for more details, see Table 2);
 Table 2. Selected characteristics of the used cement CEM I 42.5R based on the manufacturer’s data (Cementownia Ożarów, Poland).
Table 2. Selected characteristics of the used cement CEM I 42.5R based on the manufacturer’s data (Cementownia Ożarów, Poland). - Standard or “CEN” sand conforming to the requirements of the European standard EN 196-1:2016 (KWARCMIX, Tomaszów Mazowiecki, Poland);
- Tap water conforming to the requirements of the European standard EN 1008:2002 [53];
- Ash from Sakurajima volcano collected in Kagoshima, Japan (the experimentally determined characteristics of volcanic ash are provided in the next section).
All mortars were prepared on the basis of standard mortar design with the binder-to-cement ratio, b/c = 0.50, and the mass proportion of binder to aggregate 1:3 (according to EN 196-1). The level of Portland cement substitution with volcanic ash was dictated by the methodology for determining the pozzolanic activity index described in the EN 450-1 European Standard [23], which assumes the preparation of mortars in which Portland cement is replaced by the potentially pozzolanic tested material in an amount of 25% by mass. In each case, the testing was performed after 28 days and after 90 days so altogether, 6 sets of specimens (each set including 3 prisms of size 40 mm × 40 mm × 160 mm) were prepared for testing. The quantitative compositions of the tested mortars are listed in Table 3.

Table 3.
Compositions of tested mortars (by wt.); REF—reference standard mortar 1, MNVA—mortar with natural volcanic ash, MAVA—mortar with mechanically activated volcanic ash (25% substitution of cement mass acc. to EN 450-1).
4.2. Characteristics of the Sakurajima’s Volcanic Ash
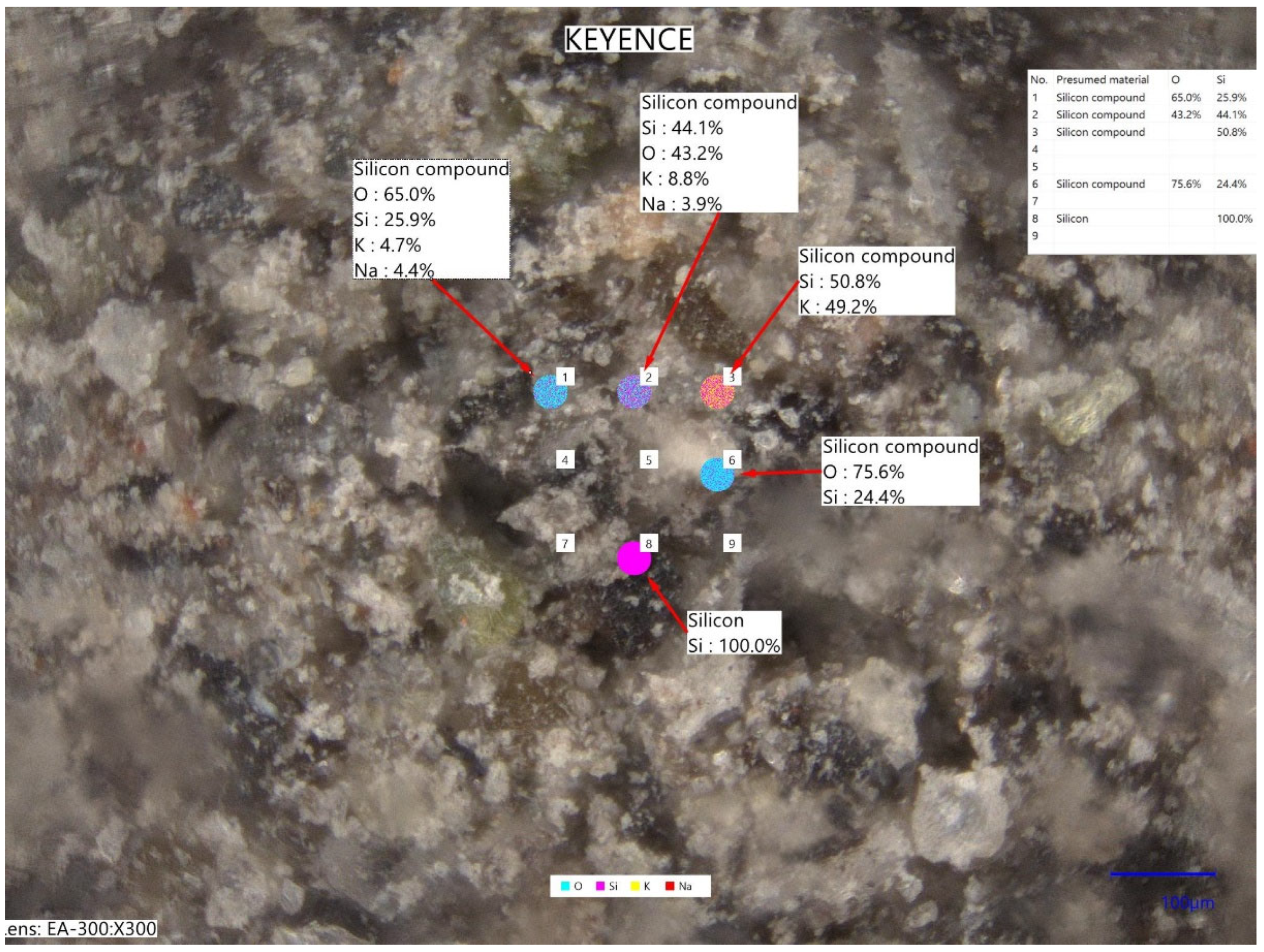
The tested pyroclastic material was collected in the direct vicinity of the Kagoshima Port, which is located on the other side of Kagoshima Bay, so it is a fall ash. The simplified elemental chemical analysis based on laser-induced breakdown spectroscopy (LIBS) performed in Keyence EA-300 VHX analyzer (Keyence Corporation, Osaka, Japan) showed that the tested ash contained sodium and potassium in amounts of 4.1 and 6.5% (on average, by wt.), respectively. Figure 6 shows an exemplary micrograph with the scheme of multi-point measurement of the chemical composition.
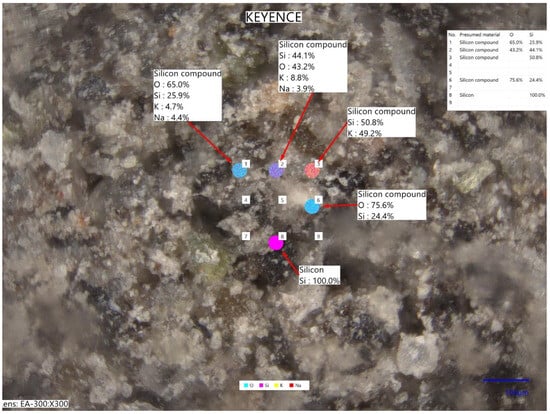
Figure 6.
Micrograph with the scheme of multi-point measurement of the chemical composition (laser-induced breakdown spectroscopy, LIBS) using Keyence EA-300 VHX.
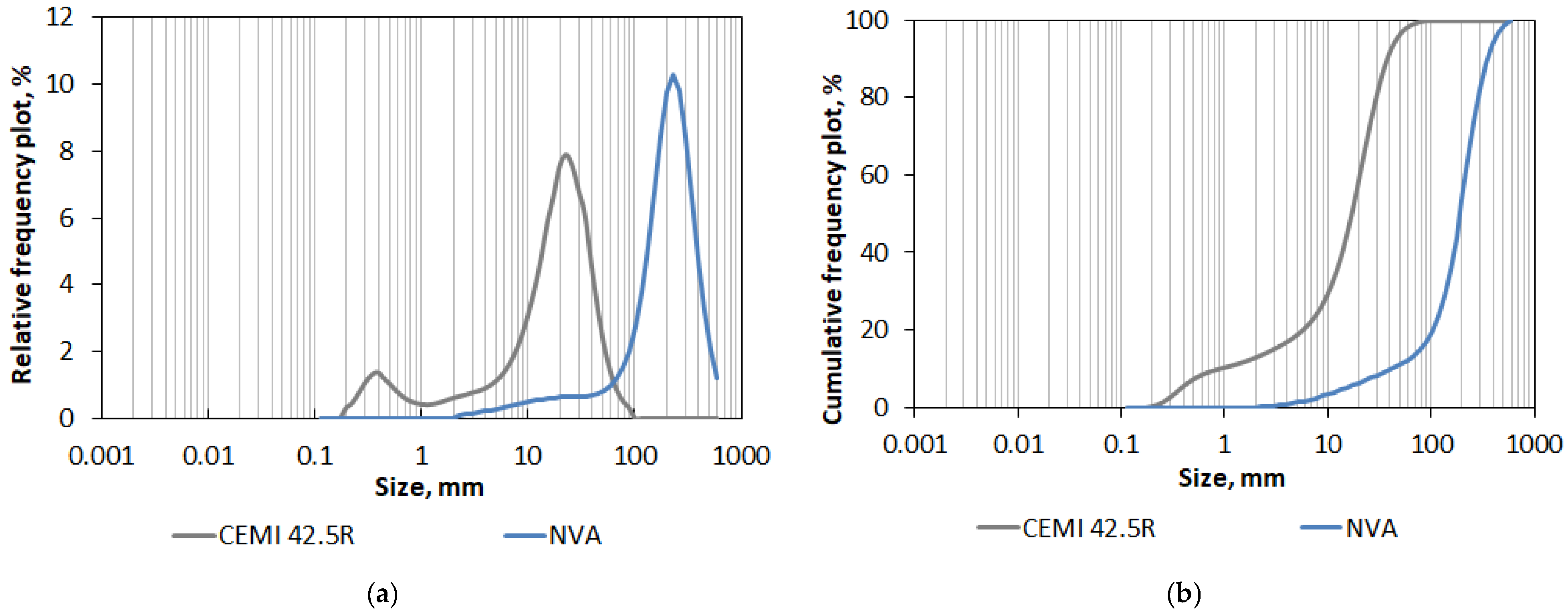
The tested volcanic ash specimen was collected in August, as during the summer months, the Sakurajima volcano is very active. To confirm that the material belongs to the volcanic ash fraction (i.e., grain size no larger than 2 mm), the first process to which the ash was subjected was sifting through standard sieves used for aggregate fractionation. First, a sieve with a mesh size of 2 mm was used. In this way, individual larger particles of pyroclastic material, possible contaminations, and actual volcanic ash were separated. The share of fractions larger than 2 mm was small, as it represented only 1% (by wt.). Then the actual volcanic ash was sifted through sieves with a mesh size of 1 mm and 0.5 mm, so the preliminary macroscale grading was determined. In the next step, the faction 0/0.5 mm (i.e., particles up to 500 μm size) was tested again to determine the particle size distribution in the microscale. The PSD measurement was performed using a laser analyzer, Horiba LA-300 (Horiba Ltd., Kyoto, Japan), and principles of the measurement included laser diffraction phenomenon and Mie LST (light scattering theory) [54,55]. The test involved passing a laser beam through a solution containing dispersed particles of the volcanic ash. A polydisperse solution (methyl alcohol) and ultrasonic were used to better disperse the material particles and break up any particle agglomerates. The method allows measurement in the range of 0.01–600 μm, and larger particles are not registered. Therefore, the volcanic ash tested by this method was sifted through a 500 μm sieve and only this sub-fraction was thoroughly examined. Thanks to this study, it was possible to compare the contents of the finest fractions present in the volcanic ash with the analogical contents of the cement that was replaced by the ash. Statistical parameters describing the particle size distribution and specific surface area obtained during the test in Horiba LA-300 of both materials are listed in Table 4, and PSD in terms of relative and cumulative frequency plots of both materials are shown in Figure 7.

Table 4.
Statistical parameters describing the particle size distribution (PSD) and specific surface area (SPA) of Sakurajima’s ash and Portland cement CEM I 42.5R.
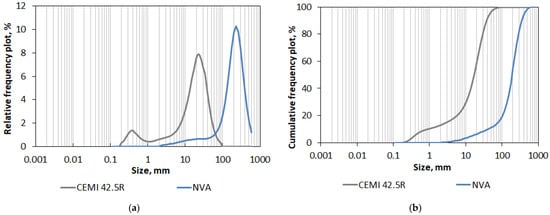
Figure 7.
Particle size distribution (PSD) of Sakurajima’s natural volcanic ash (NVA) and Portland cement CEM I 42.5R: (a) relative frequency plot; (b) cumulative frequency plot.
The obtained results showed that the analyzed volcanic ash is much thicker than the Portland cement CEM I 45.5 R. The mode, i.e., the most common value in the case of ash, is 214.11 µm, which is practically a 10 times higher value than in the case of cement (21.30 µm). The finest detected particles of ash were also 10 times larger than in the case of cement (2.27 µm and 0.20 µm, respectively). In general, the very fine grading and the bimodal size distribution of cement particles (Figure 7a) showing very high content (9.98% by vol.) of particles smaller than 1 µm (Figure 7b) caused that the cement’s specific surface area is much more developed in comparison to the SPA of natural volcanic fall ash that was transported through the air a few kilometers away from the volcano.
The fact that volcanic ash contained larger particles than cement, as well as the results of subsequent research phase (for mechanical test results, see Section 5), prompted the author to grind the ash in order to carry out the mechanical activation process. The fragmentation of the ash particles was supposed to result in developing its specific surface area and therefore increasing its pozzolanic activity as larger content of active compounds, i.e., substrates of the pozzolanic reaction, was exposed (see Section 2.3). Therefore, the volcanic ash was ground in the Fritsch PULVERISETTE 13 disk mill (Fritsch GmbH, Idar-Oberstein, Germany). The morphology of the ash grains before and after grinding (micrographs taken in the digital Keyence Digital Microscope VHX-7000 (Keyence Corporation, Osaka, Japan) with Keyence VHX-E500 high-resolution, high-magnification objective lens, Keyence Corporation, Osaka, Japan) is presented in Figure 8.

Figure 8.
Morphology of Sakurajima’s volcanic ash grains: (a) before mechanical activation; (b) after mechanical activation (micrographs taken in the digital laser microscope Keyence VHX-7000, magnification: 700×).
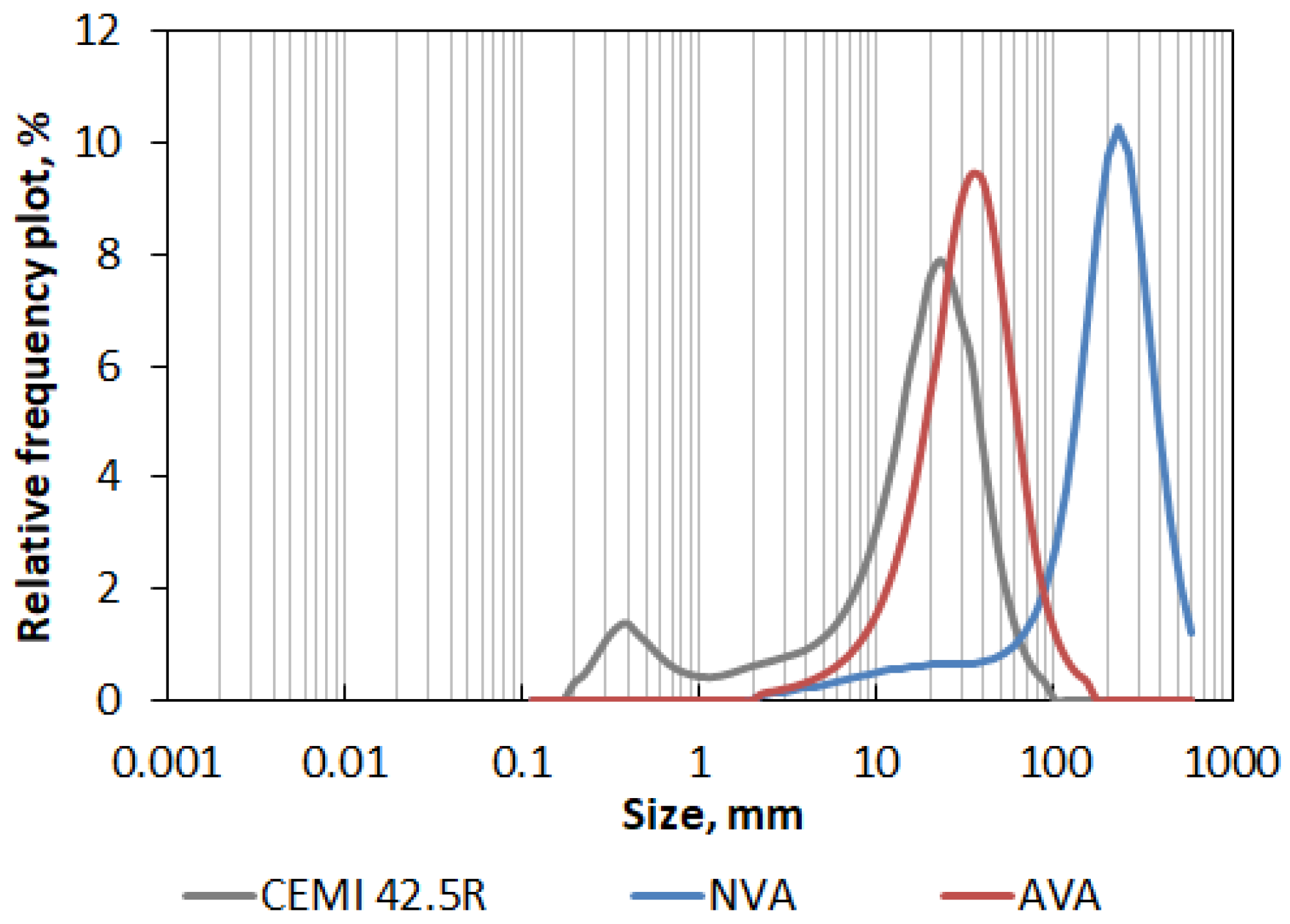
The characteristics of the ash after grinding (including the percentage change compared to the initial values) are listed in Table 5. The obtained data showed that after mechanical activation, the maximal size of the ash was reduced by 82.9%, median by 83.9%, and mode by 85.0%, while the specific surface area was increased by 192.8%, adopting a value of 2722 cm2/cm3. Tested Sakurajima’s ash after mechanical activation has become much more similar to Portland cement CEMI 42.5R in terms of granulation, which is clearly visible in Figure 9, showing particle size distribution plots elaborated for all tested materials.

Table 5.
Statistical parameters describing the particle size distribution and specific surface area (SPA) of Sakurajima’s ash after mechanical activation and percentage change compared to the initial value.
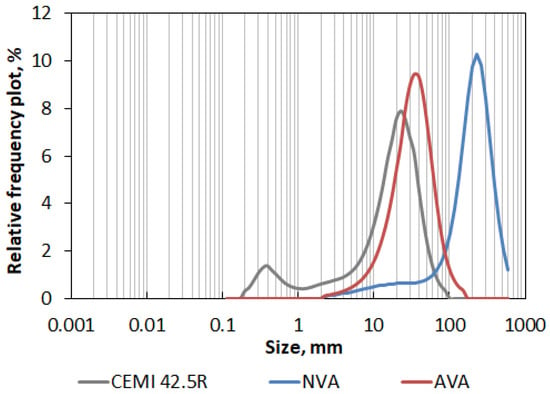
Figure 9.
PSD relative frequency plot of Sakurajima’s volcanic ash in the natural state (NVA) and after mechanical activation (AVA) compared to Portland cement CEM I 42.5R.
4.3. Composites Testing Methods
For all tested mortars, the set of technical properties was determined, including consistency (also known as plasticity), apparent density, flexural strength, and compressive strength.
The consistency of the mortar mixes was determined using a flow table. The flow table test was performed after mixing, corresponding to the initial testing time for rheological measurements, following the method described in EN 1015-3 European Standard [56]. The result of the test was the mean flow diameter (measured in perpendicular directions).
The mechanical properties were determined after 28 and/or 90 days of curing. All specimens were removed from the molds after 24 h and then kept in water under laboratory conditions. Flexural strength was evaluated using a set of three prism-shaped specimens (40 mm × 40 mm × 160 mm) in a three-point flexural test, conducted with an Instron 5567 electromechanical testing machine (Instron, Canton, OH, USA). Compressive strength measurements were performed on the remaining halves of the prisms following the flexural test, using a Controls MC66 hydraulic press (CONTROLS S.p.A., Milan, Italy). Apparent density under the air-dry condition was calculated for the same specimens prior to mechanical testing, based on their mass and measured volume.
The results of the compressive strength tests were a basis for the pozzolanic activity assessment, expressed by the activity index. The method described in the EN 450-1 European standard assumes the preparation of standard mortars, in which Portland cement is replaced by the potentially pozzolanic tested material in an amount of 25% (wt.). The tested specimens are supposed to be sets of three prisms of size 40 mm × 40 mm × 160 mm, first broken in the 3-point flexural test. The compressive strength is determined after 28 days and after 90 days of water curing. The values obtained for composites with pozzolanic compound are compared to analogic data obtained for pure cement reference mortars. According to the EN 450-1 criteria, the level of pozzolanic activity for a bulk mineral material to be considered as a type II active concrete additive is attained when the compressive strength of the modified mortar reaches at least 75% of the reference mortar’s strength after 28 days and at least 85% after 90 days.
5. Results and Discussion
The flow table consistency test (this designation is sometimes called the mortar plasticity test) showed that partial replacement of cement with Sakurajima’s volcanic ash—regardless of its condition (natural or mechanically activated)—did not affect the flow of the mortar mix. The flow diameter (mean values of perpendicular measurements) of the reference standard mortar mix was 206 mm, while the diameters of the mortar mix with natural volcanic ash (MNVA) and mortar mix with mechanically activated volcanic ash (MAVA) were 205 mm and 210 mm, respectively. These are very positive results because they show that the use of Sakurajima volcanic ash in amounts up to 25% (by wt.) does not negatively affect the rheology of the cement-based mortar mix.
The apparent density (mean values) of reference standard mortar tested after 28 and, in the second case, after 90 days was 2214 kg/m3 and 2203 kg/m3, respectively, showing no significant influence of prolonged curing on the mortar density. The apparent density of the mortar with natural volcanic ash (1/4 of binder by wt.) tested after 28 and 90 days was 2141 kg/m3 and 2177 kg/m3, showing slight decrease (by 1.2% and 3.3%, respectively). The density of the mortar with mechanically activated volcanic ash (1/4 of binder by wt.) tested after 90 days was 2243 kg/m3 showing slight increase (by 1.8%). Thus, as expected, the presence of the natural volcanic material showed reducing influence on the apparent density of the mortar—partly because the lower density of the pyroclastic material (Sakurajima volcano has a dense rock equivalent, DRE, density between 2500 and 2700 kg/m3 [57,58]) than in the case of OPC (c.a. 3100 kg/m3), partly because specimens with natural volcanic ash showed the presence of a larger number of macropores present on the smoothed surface of the hardened specimen, which was a sign of greater aeration of the mix with such ash. Meanwhile, grinding to finer fractions made it possible to obtain a material whose particles seemed to disperse better in the mix and then fill the hardened matrix better; thus, the density slightly increased. Also, the fractures of the bended specimens of mortars with mechanically activated volcanic ash (MAVA) showed less macropores and more micropores in comparison to fractures of mortars with natural volcanic ash (MNVA); however, the deeper porosity analysis was not performed in the framework of the presented study.
The results for flexural strength (mean values and basic statistic parameters: standard deviation, SD; coefficient of variation, CV) of reference standard mortar and mortars with volcanic ash (in the original and mechanically activated) tested after 28 and/or 90 days are given in Table 6. And the results for compressive strength (mean values and basic statistic parameters: standard deviation, SD; coefficient of variation, CV) of reference standard mortar and mortars with volcanic ash (in the original and mechanically activated) tested after 28 and 90 days are given in Table 7.

Table 6.
Flexural strength of tested mortars determined after 28 and/or 90 days: REF—reference standard mortar, MNVA—mortar with natural volcanic ash, MAVA—mortar with mechanically activated volcanic ash.

Table 7.
Compressive strength of tested mortars determined after 28 and 90 days: REF—reference standard mortar, MNVA—mortar with natural volcanic ash, MAVA—mortar with mechanically activated volcanic ash.
As expected, mortars with volcanic ash used as the partial binder substitute, in terms of both flexural and compressive strength, obtained lower values in comparison to reference pure cement standard mortar. This trend applies to both 28- and 90-day tests. However, the studies clearly showed that simple grinding of volcanic ash in a disk mill made the properties of mortar with ground ash better than with ash in its natural state. In the case of flexural strength, this improvement is less noticeable (4.2%), but in the case of compressive strength—after 28 days—the strength of MAVA was 29.0 MPa, which is 20.7% more than in the case of MNVA, and after 90 days, it was 43.4 MPa, which is 6.1% more than in the case of MNVA. Moreover, in the case of MAVA, standard deviation (SD) and coefficient of variation (CV) adopted lower values, which proves the greater uniformity of this mortar.
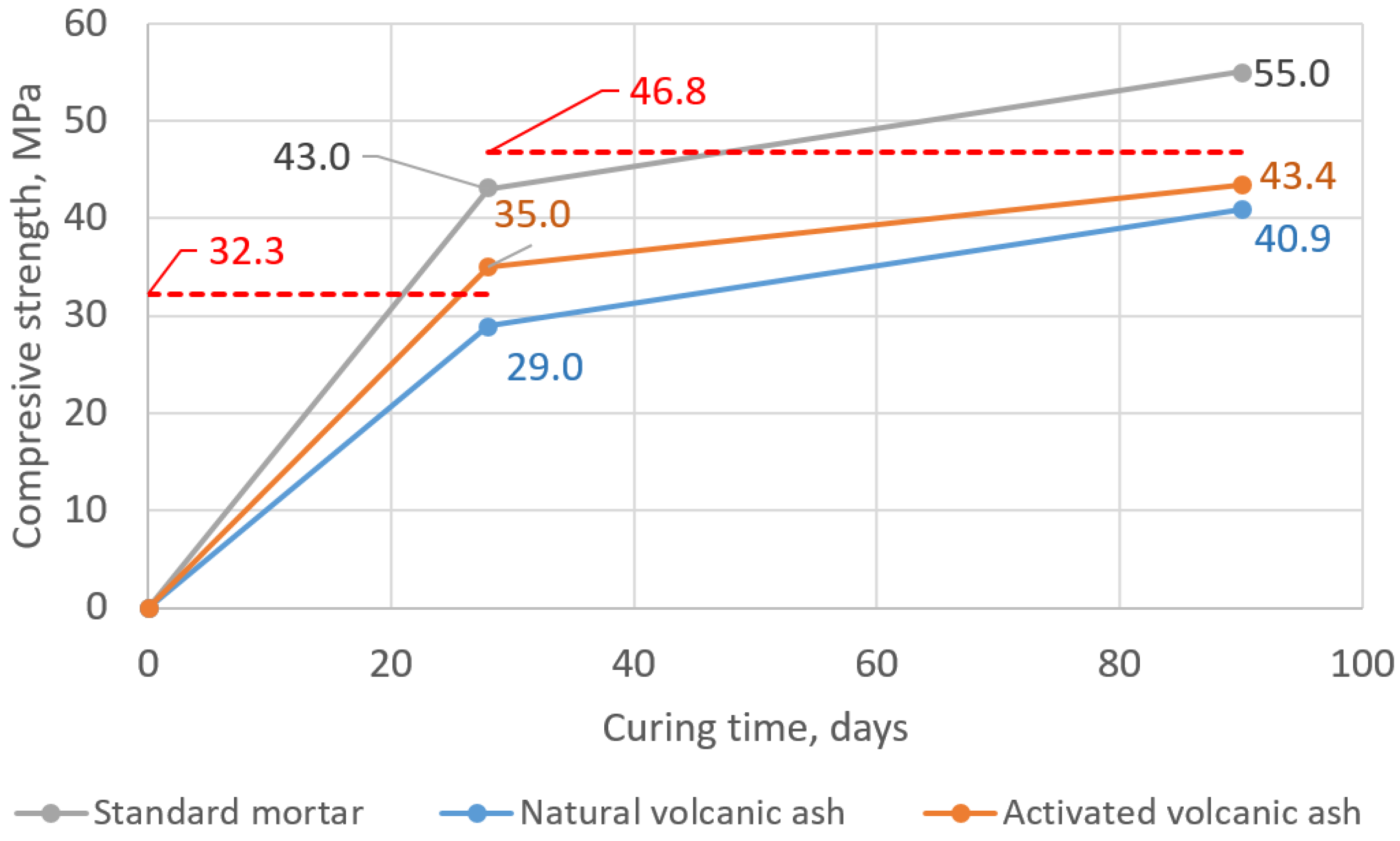
When it comes to the pozzolanic activity assessment, the EN 450-1 European Standard states that the acceptable level of pozzolanic activity is attained when the 28-day compressive strength of the modified mortar constitutes 75% of the value obtained for the 28-day-old reference mortar and 85% after 90 days. In Figure 10, one can see the determined expected levels: 32.3 MPa (i.e., 75% of 43.0 MPa, the mean compressive strength of 28-day-old standard mortar) and 46.8 MPa (i.e., 85% of 55.0 MPa, the mean compressive strength of 90-day-old standard mortar). The mortar with natural volcanic ash did not fulfill both of the criteria. After 4 weeks, it obtained 29.0 MPa, i.e., 67% of 28-day-old reference mortar strength, and after 3 months, it obtained 40.9 MPa, i.e., 74% of 90-day-old reference mortar strength. Meanwhile, in the case of mortars with activated volcanic ash, one of the criteria was fulfilled: After 28 days, the MAVA obtained 35.0 MPa, which corresponds to 81% of the strength of the analogical reference mortar strength, thus more than the required 75%. After 90 days, the MAVA obtained 43.4 MPa, which corresponds to 79% of the strength of the analogical reference mortar strength, i.e., 6% less than the required 85%.
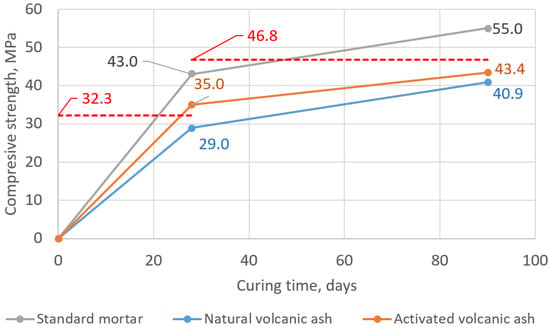
Figure 10.
Development of the compressive strength of tested mortars in time and required level of strength according to pozzolanic activity index criterions—marked with the red dashed lines (acc. to EN 450-1).
6. Conclusions
Based on the results discussed above, the following key conclusions can be drawn from the conducted laboratory investigations:
Grinding the Sakurajima’s volcanic fall ash (i.e., fraction 0/2 mm of pyroclastic fall material from Sakurajima volcano) caused the new material to present the granulation much closer to the granulation of the CEM I 42.5R Portland cement.
The 25% (by wt.) substitution of Portland cement (CEM I 42.5R) with the Sakurajima’s volcanic ash did not influence the consistency of the mortar determined by flow table test. The conclusion covers both the effect of the used volcanic ash in the natural state and in the mechanically activated state.
The 25% (by wt.) substitution of Portland cement (CEM I 42.5R) with the Sakurajima’s volcanic ash influenced the hardened mortar’s apparent density, but the differences were practically negligible. The use of natural volcanic ash caused up to 3.3% decrease in density, and mechanically activated volcanic ash caused a 1.8% increase.
The mechanical strength of mortars with the mechanically activated volcanic ash was higher than that of mortars with natural ash. This trend applies to both flexural and compressive strength after 28-day and 90-day tests. Moreover, in the case of mortar with activated volcanic ash, standard deviation (SD) and coefficient of variation (CV) adopted lower values, which proves the greater uniformity of this mortar.
Mechanical activation by grinding the Sakurajima’s volcanic ash was a successful attempt to increase its pozzolanic activity. The 28-day activity index was increased from 67% to 81%, while the 90-day activity index was increased from 74% to 79%. Also, according to EN 450-1 European Standard, the 28-day activity index determined for the activated volcanic ash fulfilled the requirement of >75% formulated for type II mineral additives for concrete.
In light of the findings presented above, pyroclastic materials sourced from the active Sakurajima volcano demonstrate strong potential as sustainable pozzolanic additives in concrete technology. Utilizing this abundant and locally available volcanic ash—currently considered a problematic waste requiring daily management—offers a dual environmental benefit: reducing the carbon footprint of cementitious composites and providing an eco-friendly waste valorization pathway. Partial substitution of ordinary Portland cement with this natural by-product can lower both material costs and environmental impact, particularly when used in locally manufactured concrete elements and structures, where long-distance transportation of raw materials is minimized. This approach aligns with circular economy principles and supports the transition toward more climate-resilient construction practices.
In future research, the author intends to investigate the chemical composition and granulometric characteristics of ash samples collected from various locations and during different phases of activity of the Sakurajima volcano to develop a comprehensive characterization of this pyroclastic material. Particular emphasis will be placed on enhancing ash activation techniques—such as mechanical refinement using ball milling and/or thermal treatment—and optimizing the formulation of cement–ash composites. Planned analyses include microstructural investigations and chemical characterization of the hardened composites to identify the products of hydration and the pozzolanic reaction of the new cementitious–volcanic ash binder system.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/su17177576/s1; Video S1: The view of the eruption and ash ejections from the southern Minami-dake crater from Sakurajima Island (author’s own video).
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
Selected data was obtained during the completion of the diploma thesis by J. Kurlak and D. Binkowski under the supervision of J.J.S.
Conflicts of Interest
The author declares no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AVA | Activated volcanic ash |
| BPD | By-pass dust |
| CEM | Cement |
| CKD | Cement kiln dust |
| CV | Coefficient of variation |
| Dmin | Minimal diameter |
| D10 | Diameter/size below which 10% of all particles are found |
| D50 | Median diameter |
| Dm | Mean diameter |
| D90 | Diameter/size below which 90% of all particles are found |
| EN | European Norm (Standard) |
| GGBS | Ground granulated blast furnace slag |
| LIBS | Laser-induced breakdown spectroscopy |
| LST | Light scattering theory |
| MAVA | Mortar with mechanically activated volcanic ash |
| MNVA | Mortar with natural volcanic ash |
| NVA | Natural volcanic ash |
| OPC | Ordinary Portland cement |
| PSD | Particle size distribution |
| SCM | Supplementary cementing material |
| SD | Standard deviation |
| SPA | Specific (surface) area |
| VA | Volcanic ash |
References
- Środa, B. Concrete—A Low-Emission Building Material; SPC: Kraków, Poland, 2021. (In Polish) [Google Scholar]
- Poudyal, L.; Adhikari, K. Environmental sustainability in cement industry: An integrated approach for green and economical cement production. Resour. Environ. Sustain. 2021, 4, 100024. [Google Scholar] [CrossRef]
- Statista. Production Volume of Cement Worldwide from 1995 to 2023. Available online: https://www.statista.com/statistics/1087115/global-cement-production-volume/ (accessed on 27 March 2025).
- Zhanikulov, N.; Sapargaliyeva, B.; Agabekova, A.; Alfereva, Y.; Baidibekova, A.; Syrlybekkyzy, S.; Nurshakhanova, L.; Nurbayeva, F.; Sabyrbaeva, G.; Zhatkanbayev, Y. Studies of Utilization of Technogenic Raw Materials in the Synthesis of Cement Clinker from It and Further Production of Portland Cement. J. Compos. Sci. 2023, 7, 226. [Google Scholar] [CrossRef]
- Mokhtar, A.; Nasooti, M. A decision support tool for cement industry to select energy efficiency measures. Energy Strategy Rev. 2020, 28, 100458. [Google Scholar] [CrossRef]
- The Impact of the Cement Industry on the Polish Economy. December 2024. (Wpływ Branży Cementowej na Gospodarkę Polski. Grudzień 2024). Available online: https://www.polskicement.pl/content/uploads/2025/01/Raport-EY-Wplyw-branzy-cementowej-2024-.pdf (accessed on 18 June 2025).
- Sokołowska, J.J.; Chmielewska, B. Carbon Footprint and CO2 Emissions in the Concrete-Polymer Composites Technology. In Concrete-Polymer Composites in Circular Economy, Proceedings of the ICPIC 2023, Warsaw, Poland, 16–19 September 2023; Springer Proceedings in Materials; Czarnecki, L., Garbacz, A., Wang, R., Frigione, M., Aguiar, J.B., Eds.; Springer: Cham, Switzerland, 2025; Volume 61. [Google Scholar] [CrossRef]
- Farahzadi, L.; Nia, S.B.; Shafei, B.; Kioumarsi, M. Sustainability assessment of ultra-high performance concrete made with various supplementary cementitious materials. Clean. Mater. 2025, 15, 100301. [Google Scholar] [CrossRef]
- Žižková, N.; Hodul, J.; Borg, R.P.; Černý, V. Repair mortars containing fly ash and crystalline admixture. Waste Forum 2019, 3, 254–266. [Google Scholar]
- Hodul, J.; Žižková, N.; Borg, R.P. The Influence of Crystalline Admixtures on the Properties and Microstructure of Mortar Containing By-Products. Buildings 2020, 10, 146. [Google Scholar] [CrossRef]
- Černý, V.; Mészárosová, L.; Melichar, J.; Drochytka, R. The Influence of Secondary Raw Materials on Properties of Aerated Autoclaved Concrete. In Modern Building Materials, Structures and Techniques, Proceedings of the MBMST 2023, Vilnius, Lithuania, 5–6 October 2023; Lecture Notes in Civil Engineering; Barros, J.A.O., Kaklauskas, G., Zavadskas, E.K., Eds.; Springer: Cham, Switzerland, 2023; Volume 392. [Google Scholar] [CrossRef]
- Łukowski, P.; Salih, A.; Sokołowska, J.J. Frost resistance of concretes containing ground granulated blast-furnace slag. MATEC Web Conf. 2018, 163, 05001. [Google Scholar] [CrossRef]
- Pérez, F.J.J.; Vega, E.J.; Orihuela, R.R.; Montesdeoca, R.D.; Díaz, C.H.; Díaz, J.R.; Díaz, E.G. Pozzolanic activity of volcanic ashes produced by the eruption of the Tajogaite Volcano in La Palma, Canary Islands. Constr. Build. Mater. 2024, 419, 135498. [Google Scholar] [CrossRef]
- Scollo, S.; Del Carlo, P.; Coltelli, M. Tephra fallout of 2001 Etna flank eruption: Analysis of the deposit and plume dispersion. J. Volcanol. Geotherm. Res. 2007, 160, 147–164. [Google Scholar] [CrossRef]
- Contrafatto, L. Recycled Etna volcanic ash for cement, mortar and concrete manufacturing. Constr. Build. Mater. 2017, 151, 704–713. [Google Scholar] [CrossRef]
- ASTM C618-23e1; Standard Specification for Coal Ash and Raw or Calcined Natural Pozzolan for Use in Concrete. ASTM International: West Conshohocken, PA, USA, 2023.
- Hosokawa, Y.; Senga, T.; Abe, A. Properties of interlocking concrete block mixed with volcanic ashes in southern Kyusyu Island. Cem. Sci. Concr. Tech. 2013, 67, 304–310. (In Japanese) [Google Scholar] [CrossRef][Green Version]
- Kamon, M.; Tomohisa, S. Reuse of Volcanic Ash Erupted from Sakurajima Volcano by the Cement-Lime Hardening (abs.). In Proceedings of the Kagoshima International Conference on Volcanoes, Kagoshima, Japan, 19–23 July 1988; pp. 757–758. Available online: https://darc.gsj.jp/archives/detail?cls=geolis&pkey=198802219 (accessed on 19 June 2025).
- Hosokawa, Y.; Onoue, K. Basic Study on Effective Utilization of Newly Volcanic Ash from Mt. Shinmoedake into Concrete. In Proceedings of the 3rd International Conference on Sustainable Construction Materials & Technologies—SCMT3, Kyoto, Japan, 18–21 August 2013; Paper No: e408. Available online: http://www.claisse.info/2013%20papers/data/e408.pdf (accessed on 19 June 2025).
- Pham, V.T.; Meng, P.; Bui, P.T.; Ogawa, Y.; Kawai, K. Effects of Shirasu natural pozzolan and limestone powder on the strength and aggressive chemical resistance of concrete. Constr. Build. Mater. 2020, 239, 117679. [Google Scholar] [CrossRef]
- Sodeyama, K.; Tomoyose, A.; Noguchi, T.; Higashi, K. Utilizing all of the Shirasu as construction material through dry gravity sorting and crushing. J. Soc. Mater. Sci. 2017, 66, 574–581. (In Japanese) [Google Scholar] [CrossRef]
- Takewaka, K. Characteristics of Shirasu concrete: Based on contents in the manual established by Kagoshima prefectural office. Concr. J. 2007, 45, 16–23. [Google Scholar] [CrossRef] [PubMed][Green Version]
- EN 450-1:2012; Fly Ash for Concrete—Part 1: Definition, Specifications and Conformity Criteria. European Committee for Standardization: Brussels, Belgium, 2012.
- Thorarinsson, S. Tefrokronologiska Studier På Island. Geografiska Annaler 1944, 26, 1–217. [Google Scholar] [CrossRef]
- Rose, W.I.; Durant, A.J. Fine ash content of explosive eruptions. J. Volcanol. Geotherm. Res. 2009, 186, 32–39. [Google Scholar] [CrossRef]
- Sidique, R. Properties of concrete made with volcanic ash. Resour. Conserv. Recycl. 2012, 66, 40–44. [Google Scholar] [CrossRef]
- Bonadonna, C.; Ernst, G.G.J.; Sparks, R.S.J. Thickness variations and volume estimates of tephra fall deposits; the importance of particle Reynolds number. J. Volcanol. Geotherm. Res. 1998, 81, 173–187. [Google Scholar] [CrossRef]
- Riley, C.M.; Rose, W.I.; Bluth, G.J.S. Quantitative shape measurements of distal volcanic ash. J. Geophys. Res. 2003, 108, 2504. [Google Scholar] [CrossRef]
- Hossain, K.M.A. Blended cement using volcanic ash and pumice. Cem. Concr. Res. 2003, 33, 601–1605. [Google Scholar] [CrossRef]
- Cultrone, G. The use of Mount Etna volcanic ash in the production of bricks with good physical-mechanical performance: Converting a problematic waste product into a resource for the construction industry. Ceram. Int. 2022, 48, 5724–5736. [Google Scholar] [CrossRef]
- Ekaputri, J.J.; Triwulan, M.; Aji, P.; Baihaqi, A. Mechanical Properties of Volcanic Ash Based Concrete. In Proceedings of the International Seminar on Applied Technology, Science, and Arts (3rdAPTECS), Surabaya, Indonesia, 6 December 2011. [Google Scholar]
- Risdanareni, R.; Karjanto, A.; Ekaputri, J.J.; Puspitasari, P.; Khakim, K. Mechanical properties of volcanic ash based geopolymer concrete. Mater. Sci. Forum 2016, 857, 377–381. [Google Scholar] [CrossRef]
- Naseer, A.; Jabbar, A.; Khan, A.N.; Ali, Q.; Hussain, Z.; Mirza, J. Performance of Pakistani volcanic ashes in mortars and concrete. Can. J. Civ. Eng. 2008, 35, 12. [Google Scholar] [CrossRef]
- Seymour, L.M.; Maragh, J.; Sabatini, P.; Di Tommaso, M.; Weaver, J.C.; Masic, A. Hot mixing: Mechanistic insights into the durability of ancient Roman concrete. Sci. Adv. 2023, 9, eadd1602. [Google Scholar] [CrossRef] [PubMed]
- Czarnecki, L.; Broniewski, T.; Henning, O. Chemistry in Civil Engineering; Arkady: Warsaw, Poland, 2006; p. 344. (In Polish) [Google Scholar]
- Kurdowski, W. Cement and Concrete Chemistry; Springer: Cham, Switzerland, 2014; p. 700. [Google Scholar]
- Collins Dictionary. Available online: https://www.collinsdictionary.com/dictionary/english/pozzolanic (accessed on 10 April 2024).
- EN 197-1:2011; Cement-Part 1: Composition, Specifications and Conformity Criteria for Common Cements. European Committee for Standardization: Brussels, Belgium, 2011.
- Tran, H.; Scott, A.; Dhakal, R. Mechanical and durability properties of magnesium silicate hydrate binder concrete. Mag. Concr. Res. 2020, 72, 693–702. [Google Scholar] [CrossRef]
- Owsiak, Z. Internal Corrosion of Concrete; Wydawnictwo Politechniki Świętokrzyska: Kielce, Poland, 2015; p. 269. [Google Scholar]
- Zapała-Sławeta, J. Impact of Aggregate Grain Size on ASR-Induced Expansion. Materials 2023, 16, 7506. [Google Scholar] [CrossRef]
- Thomas, M. The role of calcium hydroxide in alkali recycling in concrete. In Materials Science of Concrete, Special Volume: Calcium Hydroxide in Concrete; Skalny, J.P., Gebauer, J., Odler, I., Eds.; Willey-Blackwell: Cambridge, UK, 2001; pp. 225–236. [Google Scholar]
- Stanton, T.E. Expansion of concrete through reaction between cement and aggregate. Proc. Am. Soc. Civ. Eng. 1940, 66, 1781–1811. [Google Scholar] [CrossRef]
- A 1909 Map of Sakurajima by the Imperial Japanese Land Survey—Copyright © National Land Image Information (Color Aerial Photographs), Ministry of Land, Infrastructure, Transport and Tourism, Attribution, via Wikimedia Commons. Available online: https://commons.wikimedia.org/wiki/File:Sakurajima_1902_survey.jpg (accessed on 21 March 2024).
- Japan Metrological Agency: 90. Sakurajima. Available online: https://www.data.jma.go.jp/vois/data/filing/souran_eng/volcanoes/090_sakurajima.pdf (accessed on 20 June 2025).
- Aira Caldera in Kagoshima Prefecture, Kyushu, Japan. NASA, Public Domain. Available online: https://commons.wikimedia.org/wiki/File:Aira_Caldera_Relief_Map,_SRTM,_English.jpg (accessed on 10 April 2024).
- Kagoshima Sakurajima Aira Kirishima Miyakonojo. NASA, Public Domain. Available online: https://commons.wikimedia.org/wiki/File:ISS034-E-027139.jpg (accessed on 10 April 2024).
- Global Volcanism Program. Report on Aira (Japan) (Sennert, S., ed.). Weekly Volcanic Activity Report, 27 December–2 January 2024. Smithsonian Institution and US Geological Survey. 2023. Available online: https://volcano.si.edu/showreport.cfm?wvar=GVP.WVAR20231227-282080 (accessed on 10 April 2024).
- Miyairi, Y.; Yoshida, K.; Miyazaki, Y.; Matsuzaki, H.; Kaneoka, I. Improved 14C dating of a tephra layer (AT tephra, Japan) using AMS on selected organic fractions. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. At. 2004, 223–224, 555–559. [Google Scholar] [CrossRef]
- Machida, H.; Arai, F. Atlas of Tephra in and Around Japan; University of Tokyo Press: Tokyo, Japan, 1992; p. 278. [Google Scholar]
- Global Volcanism Program. Report on Aira (Japan)—Eruptive History. Available online: https://volcano.si.edu/volcano.cfm?vn=282080&vtab=Eruptions (accessed on 10 April 2024).
- EN 196-1:2016; Methods of Testing Cement-Part 1: Determination of Strength. European Committee for Standardization: Brussels, Belgium, 2016.
- EN 1008:2002; Mixing Water for Concrete-Specification for Sampling, Testing and Assessing the Suitability of Water, Including Water Recovered from Processes in the Concrete Industry, as Mixing Water for Concrete. European Committee for Standardization: Brussels, Belgium, 2002.
- Bohren, C.F.; Huffman, D.R. Absorption and Scattering of Light by Small Particles; Wiley-Interscience: New York, NY, USA, 2010. [Google Scholar]
- Ruello, G.; Lattanzi, R. Scattering from Spheres: A New Look into an Old Problem. Electronics 2021, 10, 216. [Google Scholar] [CrossRef]
- EN 1015-3:2007; Methods of Test for Mortar for Masonry: Determination of Consistence of Fresh Mortar (by Flow Table). European Committee for Standardization: Brussels, Belgium, 2007.
- Bagheri, G.; Rossi, E.; Biass, S.; Bonadonna, C. Timing and Nature of Volcanic Particle Clusters Based on Field and Numerical Investigations. J. Volcanol. Geotherm. Res. 2016, 327, 520–530. [Google Scholar] [CrossRef]
- Miwa, T.; Toramaru, A. Conduit process in vulcanian eruptions at Sakurajima volcano, Japan: Inference from comparison of volcanic ash with pressure wave and seismic data. Bull. Volcanol. 2013, 75, 685. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).